
General View of Filters at Hamburg.
[Frontispiece.]
THE FILTRATION
OF
PUBLIC WATER-SUPPLIES.
BY
ALLEN HAZEN,
MEMBER OF THE AMERICAN SOCIETY OF CIVIL ENGINEERS, THE BOSTON SOCIETY OF CIVIL ENGINEERS, THE AMERICAN WATER-WORKS ASSOCIATION, THE NEW ENGLAND WATER-WORKS ASSOCIATION, THE AMERICAN CHEMICAL SOCIETY, THE AMERICAN PUBLIC HEALTH ASSOCIATION, ETC.
THIRD EDITION, REVISED AND ENLARGED.
SECOND THOUSAND.
NEW YORK:
JOHN WILEY & SONS.
London: CHAPMAN & HALL, Limited.
1905.
Copyright, 1900,
BY
ALLEN HAZEN.
ROBERT DRUMMOND, ELECTROTYPER AND PRINTER, NEW YORK.
[Pg iii]
PREFACE TO FIRST EDITION.
The subject of water-filtration is commencing to receive a great deal of attention in the United States. The more densely populated European countries were forced to adopt filtration many years ago, to prevent the evils arising from the unavoidable contaminations of the rivers and lakes which were the only available sources for their public water-supplies; and it has been found to answer its purpose so well that at the present time cities in Europe nearly if not quite equal in population to all the cities of the United States are supplied with filtered water.
Many years ago, when the whole subject of water-supply was still comparatively new in this country, filtration was considered as a means for rendering the waters of our rivers suitable for the purpose of domestic water-supply. St. Louis investigated this subject in 1866, and the engineer of the St. Louis Water Board, the late Mr. J. P. Kirkwood, made an investigation and report upon European methods of filtration which was published in 1869, and was such a model of full and accurate statement combined with clearly-drawn conclusions that, up to the present time, it has remained the only treatise upon the subject in English, notwithstanding the great advances which have been made, particularly in the last ten years, with the aid of knowledge of the bacteria and the germs of certain diseases in water.
Unfortunately the interest in the subject was not maintained in America, but was allowed to lag for many years; it was cheaper to use the water in its raw state than it was to purify it; the people became indifferent to the danger of such use, and [Pg iv] the disastrous epidemics of cholera and typhoid fever, as well as of minor diseases, which so often resulted from the use of polluted water, were attributed to other causes. With increasing study and diffusion of knowledge the relations of water and disease are becoming better known, and the present state of things will not be allowed to continue; indeed at present there is inquiry at every hand as to the methods of improving waters.
The one unfortunate feature is the question of cost. Not that the cost of filtration is excessive or beyond the means of American communities; in point of fact, exactly the reverse is the case; but we have been so long accustomed to obtain drinking-water without expense other than pumping that any cost tending to improved quality seems excessive, thus affording a chance for the installation of inferior filters, which by failing to produce the promised results tend to bring the whole process into disrepute, since few people can distinguish between an adequate filtration and a poor substitute for it. It is undoubtedly true that improvements are made, and will continue to be made, in processes of filtration; so it will often be possible to reduce the expense of the process without decreasing the efficiency, but great care must be exercised in such cases to maintain the conditions really essential to success.
In the present volume I have endeavored to explain briefly the nature of filtration and the conditions which, in half a century of European practice, have been found essential for successful practice, with a view of stimulating interest in the subject, and of preventing the unfortunate and disappointing results which so easily result from the construction of inferior filters. The economies which may possibly result by the use of an inferior filtration are comparatively small, and it is believed that in those American cities where filtration is necessary or desirable it will be found best in every case to furnish filters of the best construction, fully able to do what is required of them with ease and certainty.
[Pg v]
PREFACE TO THIRD EDITION.
There have been several distinct epochs in the development of water purification in the United States. The first may be said to date from Kirkwood’s report on the “Filtration of River Waters,” and the second from the inauguration of the Lawrence Experiment Station by the Massachusetts State Board of Health, and the construction of the Lawrence city filter, with the demonstration of the wonderful biological action of filters upon highly polluted waters.
The third epoch is marked by the experiments at Louisville, Pittsburg and Cincinnati, which have greatly increased our knowledge of the treatment of waters containing enormous quantities of suspended matter, and have reduced to something like order the previously existing confused mass of data regarding coagulation and rapid filtration.
The first edition of this book represented the earlier epochs before the opening of the third. In the five years since it was written, progress in the art of water purification has been rapid and substantial. No apology is needed for the very complete revision required to treat these newly investigated subjects as fully as were other matters in the earlier editions.
In the present edition the first seven chapters remain with but few additions. Experience has strengthened the propositions contained in them. New data might have been added, but in few cases would the conclusions have been altered. The remaining[Pg vi] chapters of the book have been entirely rewritten and enlarged to represent the added information now available, so that the present edition is nearly twice as large as the earlier ones. In the appendices, also, much matter has been added relating to works in operation, particularly to those in America.
New York January, 1900.
[Pg vii]
CONTENTS.
| PAGE | ||
|---|---|---|
| Chapter I. | Introduction. |
1 |
| II. | Continuous Filters and their Construction |
5 |
Sedimentation-basins |
8 | |
Size of Filter-beds |
10 | |
Form of Filter-beds |
11 | |
Covers for Filters |
12 | |
| III. | Filtering-materials |
20 |
Sand |
20 | |
Sands Used in European Filters |
24 | |
Effect of Size of Grain Upon Efficiency of Filtration |
30 | |
Effect of Grain Size Upon Frequency of Scraping |
32 | |
Selection of Sand |
33 | |
Thickness of the Sand Layer |
34 | |
Underdraining |
35 | |
Gravel Layers |
35 | |
Underdrains |
39 | |
Depth of Water on Filters |
45 | |
| IV. | Rate of Filtration and Loss of Head |
47 |
Effect of Rate Upon Cost of Filtration |
48 | |
Effect of Rate Upon Efficiency of Filtration |
50 | |
The Loss of Head |
52 | |
Regulation of the Rate and Loss of Head in the Older Filters |
52 | |
Apparatus For Regulating the Rate and Loss of Head |
55 | |
Apparatus For Regulating the Rate Directly |
57 | |
Apparatus For Regulating the Height of Water Upon Filters |
59 | |
Limit to the Loss of Head |
60 | |
| V. | Cleaning Filters |
68 |
Frequency of Scraping |
72 | |
Quantity of Sand to Be Removed |
74 | |
Wasting the Effluents After Scraping |
74 | |
Sand-washing |
76 | |
| VI. | Theory and Efficiency of Filtration |
83 |
Bacterial Examination of Waters |
93 | |
| VII. | Intermittent Filtration |
97 |
The Lawrence Filter |
100 | |
Chemnitz Water-Works |
107 | |
Application of Intermittent Filtration |
111 | |
| VIII. | Turbidity and Color, and the Effect of Mud upon Sand Filters |
113 |
The Measurement of Color |
114 | |
Amount of Color in American Waters |
115 | |
Removal of Color |
117 | |
Measurement of Turbidity |
117 | |
Relation of Platinum-wire Turbidities to Suspended Matters |
122 | |
Source of Turbidity |
123 | |
The Amounts of Suspended Matters in Water |
129 | |
Preliminary Processes to remove Mud |
133 | |
Effect of Mud upon Sand Filters |
137 | |
Effect of Turbidity Upon the Length of Period |
137 | |
Power of Sand Filters to Produce Clear Effluents from Muddy Water |
139 | |
Effect of Mud Upon Bacterial Efficiency of Filters |
141 | |
Limits to the Use of Subsidence for the Preliminary Treatment of Muddy Waters |
142 | |
| IX. | Coagulation of Waters |
144 |
Substances used for Coagulation |
145 | |
Coagulants Which Have Been Used |
150 | |
Amount of Coagulant required to remove Turbidity |
150 | |
Amount of Coagulant required to remove Color |
153 | |
Successive Applications of Coagulant |
154 | |
The Amount of Coagulant which Various Waters will receive |
155 | |
| [Pg viii] | ||
| X. | Mechanical Filters |
159 |
Providence Experiments |
159 | |
Louisville_Experiments |
161 | |
Lorain Tests |
161 | |
Pittsburg Experiments |
162 | |
Wasting Effluent After Washing Filters |
163 | |
Influence of Amount of Sulphate of Alumina on Bacterial Efficiency of Mechanical Filters |
165 | |
Influence of Degree of Turbidity upon Bacterial Efficiency of Mechanical Filters |
167 | |
Average Results Obtained with Various Quantities of Sulphate of Alumina |
171 | |
Types of Mechanical Filters |
172 | |
Efficiency of Mechanical Filters |
179 | |
Pressure Filters |
180 | |
| XI. | Other Methods of Filtration |
181 |
Worms Tile System |
181 | |
The Use of Asbestos |
181 | |
Filters Using High Rates of Filtration Without Coagulants |
182 | |
Household Filters |
183 | |
| XII. | Removal of Iron from Ground-waters |
186 |
Amount of Iron Required to Render Water Objectionable |
186 | |
Cause of Iron in Ground-waters |
187 | |
Treatment of Iron-containing Waters |
189 | |
Iron-removal Plants in Operation |
192 | |
| XIII. | Treatment of Waters |
197 |
Cost of Filtration |
200 | |
What Waters Require Filtration |
207 | |
| XIV. | Water-supply and Disease—Conclusions |
210 |
| Appendix I. | Rules of the German Government in Regard to the Filtration of Surface-waters Used For Public Water-supplies |
221 |
| II. | Extracts from “Bericht Des Medicinal-inspectorats Des Hamburgischen Staates Für Das Jahr 1892” |
226 |
| III. | Methods of Sand-analysis |
233 |
| IV. | Filter Statistics |
241 |
Statistics of Operation of Sand Filters |
241 | |
Partial List of Cities Using Sand Filters |
244 | |
List of Cities and Towns Using Mechanical Filters |
247 | |
Notes Regarding Sand Filters in the United States |
251 | |
Capacity of Filters |
254 | |
| V. | London’s Water-supply |
255 |
| VI. | The Berlin Water-works |
261 |
| VII. | Altona Water-works |
265 |
| VIII. | Hamburg Water-works |
269 |
| IX. | Notes on Some Other European Water-supplies |
272 |
The Use of Unfiltered Surface-waters. |
275 | |
The Use of Ground-water. |
276 | |
| X. | Literature of Filtration |
277 |
| XI. | The Albany Water-filtration Plant |
288 |
Description of Plant. |
289 | |
Capacity of Plant and Means of Regulation. |
308 | |
Results of Operation. |
314 | |
Cost of Construction. |
314 | |
| Index | 317 | |
[Pg ix]
UNITS EMPLOYED.
The units used in this work are uniformly those in common use in America, with the single exception of data in regard to sand-grain sizes, which are given in millimeters. The American units were not selected because the author prefers them or considers them particularly well suited to filtration, but because he feared that the use of the more convenient metric units in which the very comprehensive records of Continental filter plants are kept would add to the difficulty of a clear comprehension of the subject by those not familiar with those units, and so in a measure defeat the object of the book.
| TABLE OF EQUIVALENTS. | |||
|---|---|---|---|
| Unit. | Metric Equivalent. | Reciprocal. | |
| Foot | 0.3048 | meter | 3.2808 |
| Mile | 1609.34 | meters | 0.0006214 |
| Acre | 4047 | square meters | 0.0002471 |
| Gallon[1] | 3.785 | liters | 0.26417 |
| 1 million gallons | 3785 | cubic meters | 0.00026417 |
| Cubic yard | 0.7645 | cubic meters | 1.308 |
1 million gallons per acre daily |
0.9354 | meter in depth of water daily |
1.070 |
ACKNOWLEDGMENT.
I wish to acknowledge my deep obligation to the large number of European engineers, directors, and superintendents of water-works, and to the health officers, chemists, bacteriologists, and other officials who have kindly aided me in studying the filtration-works in their respective cities, and who have repeatedly furnished me with valuable information, statistics, plans, and reports.
To mention all of them would be impossible, but I wish particularly to mention Major-General Scott, Water-examiner of London; Mr. Mansergh, Member of the Royal Commission on the Water-supply of the Metropolis; Mr. Bryan, Engineer of the East London Water Company; and Mr. Wilson, Manager of the Middlesborough Water-works, who have favored me with much valuable information.
In Holland and Belgium I am under special obligations to Messrs. Van Hasselt and Kemna, Directors of the water companies at Amsterdam and Antwerp respectively; to Director Stang of the Hague Water-works; to Dr. Van’t Hoff, Superintendent of the Rotterdam filters; and to my friend H. P. N. Halbertsma, who, as consulting engineer, has built many of the Dutch water-works.
In Germany I must mention Profs. Frühling, at Dresden, and Flügge, at Breslau; Andreas Meyer, City Engineer of Hamburg; and the Directors of water-works, Beer at Berlin, Dieckmann at Magdeburg, Nau at Chemnitz, and Jockmann at Liegnitz, as well as the Superintendent Engineers Schroeder at Hamburg, Debusmann at Breslau, and Anklamm and Piefke at Berlin, the latter the distinguished head of the Stralau works, the first and most widely known upon the Continent of Europe.
I have to acknowledge my obligation to City Engineer Sechner at Budapest, and to the Assistant Engineer in charge of water-works, Kajlinger; to City Engineer Peters and City Chemist Bertschinger[Pg xii] at Zürich; and to Assistant Engineer Regnard of the Compagnie Générale des Eaux at Paris.
On this side of the Atlantic also I am indebted to Hiram F. Mills, C.E., under whose direction I had the privilege of conducting for nearly five years the Lawrence experiments on filtration; to Profs. Sedgwick and Drown for the numerous suggestions and friendly criticisms, and to the latter for kindly reading the proof of this volume; to Mr. G. W. Fuller for full information in regard to the more recent Lawrence results; to Mr. H. W. Clark for the laborious examination of the large number of samples of sands used in actual filters and mentioned in this volume; and to Mr. Desmond FitzGerald for unpublished information in regard to the results of his valuable experiments on filtration at the Chestnut Hill Reservoir, Boston.
Allen Hazen.
Boston, April, 1895.
[Pg 1]
FILTRATION OF PUBLIC WATER-SUPPLIES.
CHAPTER I.
INTRODUCTION.
The rapid and enormous development and extension of water-works in every civilized country during the past forty years is a matter which deserves our most careful consideration, as there is hardly a subject which more directly affects the health and happiness of almost every single inhabitant of all cities and large towns.
Considering the modern methods of communication, and the free exchange of ideas between nations, it is really marvellous how each country has met its problems of water-supply from its own resources, and often without much regard to the methods which had been found most useful elsewhere. England has secured a whole series of magnificent supplies by impounding the waters of small streams in reservoirs holding enough water to last through dry periods, while on Continental Europe such supplies are hardly known. Germany has spent millions upon millions in purifying turbid and polluted river-waters, while France and Austria have striven for mountain-spring waters and have built hundreds of miles of costly aqueducts to secure them. In the United States an abundant supply of some liquid has too often been the objective point, and the efforts have been most[Pg 2] successful, the American works being entirely unrivalled in the volumes of their supplies. I do not wish to imply that quality has been entirely neglected in our country, for many cities and towns have seriously and successfully studied their problems, with the result that there are hundreds of water-supplies in the United States which will compare favorably upon any basis with supplies in any part of the world; but on the other hand it is equally true that there are hundreds of other cities, including some among the largest in the country, which supply their citizens with turbid and unhealthy waters which cannot be regarded as anything else than a national disgrace and a menace to our prosperity.
One can travel through England, Belgium, Holland, Germany, and large portions of other European countries and drink the water at every city visited without anxiety as to its effect upon his health. It has not always been so. Formerly European capitals drank water no better than that so often dispensed now in America. As recently as 1892 Germany’s great commercial centre, Hamburg, having a water-supply essentially like those of Philadelphia, Pittsburg, Cincinnati, St. Louis, New Orleans, and a hundred other American cities, paid a penalty in one month of eight thousand lives for its carelessness. The lesson was a dear one, but it was not wasted. Hamburg now has a new and wholesome supply, and other German cities the qualities of whose waters were open to question have been forced to take active measures to better their conditions. We also can learn something from their experience.
There are three principal methods of securing a good water-supply for a large city. The first consists of damming a stream from an uninhabited or but sparsely inhabited watershed, thus forming an impounding reservoir. This method is extensively used in England and in the United States. In the latter most of the really good and large supplies are so obtained. It is only applicable to places having suitable watersheds within a reasonable[Pg 3] distance, and there are large regions where, owing to geological and other conditions, it cannot be applied. It is most useful in hilly and poor farming countries, as in parts of England and Wales, in the Atlantic States, and in California. It cannot be used to any considerable extent in level and fertile countries which are sure to be or to become densely populated, as is the case with large parts of France and Germany and in the Middle States.
The second method is to secure ground-water, that is, spring or well water, which by its passage through the ground has become thoroughly purified from any impurities which it may have contained. This was the earliest and is the most widely used method of securing good water. It is specially adapted to small supplies. Under favorable geological conditions very large supplies have been obtained in this manner. In Europe Paris, Vienna, Budapest, Munich, Cologne, Leipzig, Dresden, a part of London, and very many smaller places are so supplied. This method is also extensively used in the United States for small and medium-sized places, and deserves to be most carefully studied, and used whenever possible, but is unfortunately limited by geological conditions and cannot be used except in a fraction of the cases where supplies are required. No ground-water supplies yet developed in the United States are comparable in size to those used in Europe.
The third process of securing a good water-supply is by means of filtration of surface waters which would otherwise be unsuitable for domestic purposes. The methods of filtration, which it is the purpose of this volume to explain, are beyond the experimental stage; they are now applied to the purification of the water-supplies of European cities with an aggregate population of at least 20,000,000 people. In the United States the use of filters is much less common, and most of the filters in use are of comparatively recent installation.
Great interest has been shown in the subject during the last[Pg 4] few years, and the peculiar character of some American waters, which differ widely in their properties from those of many European streams, has received careful and exhaustive consideration. In Europe filtration has been practised with continually improving methods since 1829, and the process has steadily received wider and wider application. It has been most searchingly investigated in its hygienic relations, and has been repeatedly found to be a most valuable aid in reducing mortality. The conditions under which satisfactory results can be obtained are now tolerably well known, so that filters can be built in the United States with the utmost confidence that the result will not be disappointing.
The cost of filtration, although considerable, is not so great as to put it beyond the reach of American cities. It may be roughly estimated that the cost of filtration, with all necessary interest and sinking funds, will add 10 per cent to the average cost of water as at present supplied.
It may be confidently expected that when the facts are better understood and realized by the American public, we shall abandon the present filthy and unhealthy habit of drinking polluted river and lake waters, and shall put the quality as well as the quantity of our supplies upon a level not exceeded by those of any country.
[Pg 5]
CHAPTER II.
CONTINUOUS FILTERS AND THEIR CONSTRUCTION.
Filtration of water consists in passing it through some substance which retains or removes some of its impurities. In its simplest form filtration is a straining process, and the results obtained depend upon the fineness of the strainer, and this in turn is regulated by the character of the water and the uses to which it is to be put. Thus in the manufacture of paper an enormous volume of water is required free from particles which, if they should become imbedded in the paper, would injure its appearance or texture. Obviously for this purpose the removal of the smaller particles separately invisible to the unaided eye, and thus not affecting the appearance of the paper, and the removal of which would require the use of a finer filter at increased expense, would be a simple waste of money. When, however, a water is to be used for a domestic water supply and transparency is an object, the still finer particles which would not show themselves in paper, but which are still able, in bulk, to render a water turbid, should be as far as possible removed, thus necessitating a finer filter; and, when there is reason to think that the water contains the germs of disease, the filter must be fine enough to remove with certainty those organisms so extraordinarily small that millions of them may exist in a glass of water without imparting a visible turbidity.
It is now something over half a century since the first successful attempts were made to filter public water-supplies, and there are now hundreds of cities supplied with clear, healthy, filtered water. (Appendix IV.) While the details of the filters[Pg 6] used in different places present considerable variations, the general form is, in Europe at least, everywhere the same. The most important parts of a filter are shown by the accompanying sketch, in which the dimensions are much exaggerated. The raw water is taken from the river into a settling-basin, where the heaviest mud is allowed to settle. In the case of lake and pond waters the settling-tank is dispensed with, but it is essential for turbid river-water, as otherwise the mud clogs the filter too rapidly. The partially clarified water then passes to the filter, which consists of a horizontal layer of rather fine sand supported by gravel and underdrained, the whole being enclosed in a suitable basin or tank. The water in passing through the sand leaves behind upon the sand grains the extremely small particles which were too fine to settle out in the settling-basin, and is quite clear as it goes from the gravel to the drains and the pumps, which forward it to the reservoir or city.
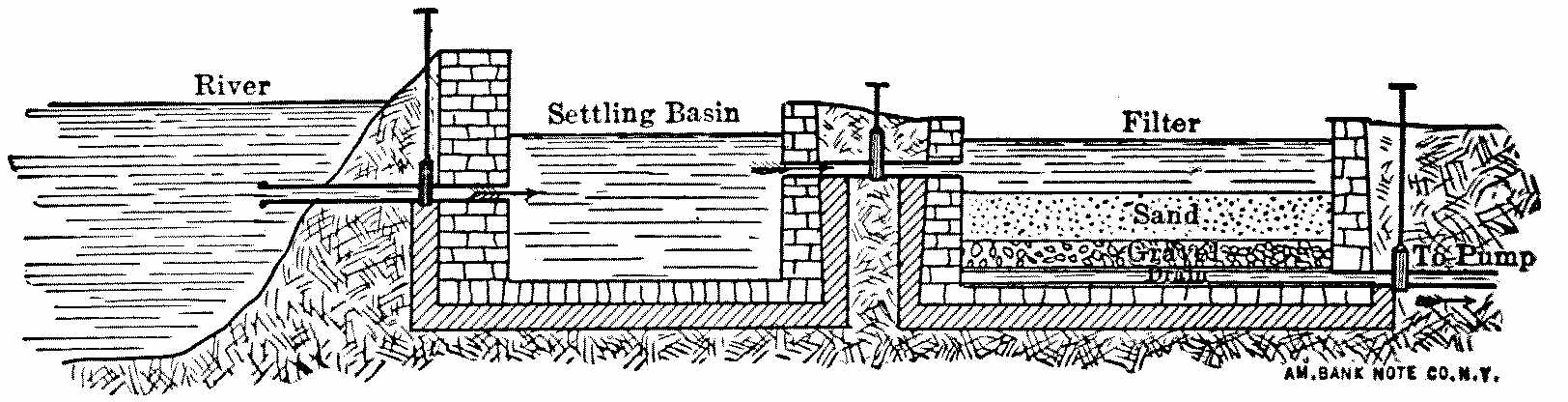
Fig. 1.—Sketch Showing General Arrangement of Filter Plants.
The passages between the grains of sand through which the water must pass are extremely small. If the sand grains were spherical and 1⁄50 of an inch in diameter, the openings would only allow the passage of other spheres 1⁄320 of an inch in diameter, and with actual irregular sands much finer particles are held back. As a result the coarser matters in the water are retained on the surface of the sand, where they quickly form a layer of sediment, which itself becomes a filter much finer than the sand alone, and which is capable of holding back under suitable conditions even the bacteria of the passing water. The water which passes before this takes place may be less perfectly[Pg 7] filtered, but even then, the filter may be so operated that nearly all of the bacteria will be deposited in the sand and not allowed to pass through into the effluent.
As the sediment layer increases in thickness with continued filtration, increased pressure is required to drive the desired volume of water through its pores, which are ever becoming smaller and reduced in number. When the required quantity of water will no longer pass with the maximum pressure allowed, it is necessary to remove, by scraping, the sediment layer, which should not be more than an inch deep. This layer contains most of the sediment, and the remaining sand will then act almost as new sand would do. The sand removed may be washed for use again, and eventually replaced when the sand layer becomes too thin by repeated scrapings. These operations require that the filter shall be temporarily out of use, and as water must in general be supplied without intermission, a number of filters are built together, so that any of them can be shut out without interfering with the action of the others.
The arrangement of filters in relation to the pumps varies with local conditions. With gravity supplies the filters are usually located below the storage reservoir, and, properly placed, involve only a few feet loss of head.
In the case of tidal rivers, as at Antwerp and Rotterdam, the quality of the raw water varies with the tide, and there is a great advantage in having the settling-basins low enough so that a whole day’s supply can be rapidly let in when the water is at its best, without pumping. At Antwerp the filters are higher, and the water is pumped from the settling basins to them, and again from the reservoir receiving the effluents from the filters to the city. In several of the London works (East London, Grand Junction, Southwark and Vauxhall, etc.) the settling-basins are lower than the river, and the filters are still lower, so that a single pumping suffices, that coming between the filter and the city, or elevated distributing reservoir.
[Pg 8]
In many other English filters and in most German works the settling-basins and filters are placed together a little higher than the river, thus avoiding at once trouble from floods and cost for excavation. The water requires to be pumped twice, once before and once after filtration. At Altona the settling-basins and filters are placed upon a hill, to which the raw Elbe water is pumped, and from which it is supplied to the city after filtration by gravity without further pumping. The location of the works in this case is said to have been determined by the location of a bed of sand suitable for filtration on the spot where the filters were built.
When two pumpings are required they are frequently done, especially in the smaller places, in the same pumping-station, with but one set of boilers and engines, the two pumps being connected to the same engine. The cost is said to be only slightly greater than that of a single lift of the same total height. In very large works, as at Berlin and Hamburg and some of the London companies, two separate sets of pumping machinery involve less extra cost relatively than would be the case with smaller works.
SEDIMENTATION-BASINS.
Kirkwood[2] found in 1866 that sedimentation-basins were essential to the successful treatment of turbid river-waters, and subsequent experience has not in any way shaken his conclusion. The German works visited by him, Berlin (Stralau) and Altona, were both built by English engineers, and their settling-basins did not differ materially from those of corresponding works in England. Since that time, however, there has been a well-marked tendency on the part of the German engineers to use smaller, while the English engineers have used much larger sedimentation-basins, so that the practices of the two countries are [Pg 9]now widely separated, the difference no doubt being in part at least due to local causes.
Kirkwood found sedimentation-basins at Altona with a capacity of 21⁄4 times the daily supply. In 1894 the same basins were in use, although the filtering area had been increased from 0.82 acre to 2.20 acres, and still more filters were in course of construction, and the average daily quantity of water had increased from 600,000 to 4,150,000 gallons in 1891-2, or more than three times the capacity of the sedimentation-basins. In 1890 the depth of mud deposited in these basins was reported to be two feet deep in three months. At Stralau in Berlin, also, in the same time the filtering area was nearly doubled without increasing the size of the sedimentation-basins, but the Spree at this point has such a slow current that it forms itself a natural sedimentation-basin. At Magdeburg on the Elbe works were built in 1876 with a filtering area of 1.92 acres, and a sedimentation-basin capacity of 11,300,000 gallons, but in 1894 half of the latter had been built over into filters, which with two other filters gave a total filtering surface of 3.90 acres, with a sedimentation-basin capacity of only 5,650,000 gallons. The daily quantity of water pumped for 1891-2 was 5,000,000 gallons, so that the present sedimentation-basin capacity is about equal to one day’s supply, or relatively less than a third of the original provision. The idea followed is that most of the particles which will settle at all will do so within twenty-four hours, and that a greater storage capacity may allow the growth of algæ, and that the water may deteriorate rather than improve in larger tanks.

Paved Embankment between Two Filters, East London.

Filters and Channels for Raw Water, Antwerp.
[To face page 10.]
At London, on the other hand, the authorities consider a large storage capacity for unfiltered water as one of the most important conditions of successful filtration, the object however, being perhaps as much to secure storage as to allow sedimentation. In 1893 thirty-nine places were reported upon the Thames and the Lea which were giving their sewage systematic treatment before discharging it into the streams from which London’s[Pg 10] water is drawn. These sewage treatments are, with hardly an exception, dry-weather treatments, and as soon as there is a considerable storm crude sewage is discharged into the rivers at every point. The rivers are both short, and are quickly flooded, and afterwards are soon back in their usual condition. At these times of flood, the raw water is both very turbid and more polluted by sewage than at other times, and it is the aim of the authorities to have the water companies provide reservoir capacity enough to carry them through times of flood without drawing any water whatever from the rivers. This obviously involves much more extensive reservoirs than those used in Germany, and the companies actually have large basins and are still adding to them. The storage capacities of the various companies vary from 3 to 18 times the respective average daily supplies, and together equal 9 times the total supply.
In case the raw water is taken from a lake or a river at a point where there is but little current, as in a natural or artificial pond, sedimentation-basins are unnecessary. This is the case at Zürich (lake water), at Berlin when the rivers Havel and Spree spread into lakes, at Tegel and Müggel, and at numerous other works.
SIZE OF FILTER-BEDS.
The total area of filters required in any case is calculated from the quantity of water required, the rate of filtration, and an allowance for filters out of use while being cleaned. To prevent interruptions of the supply at times of cleaning, the filtering area is divided into beds which are operated separately, the number and size of the beds depending upon local conditions. The cost per acre is decreased with large beds on account of there being less wall or embankment required, while, on the other hand, the convenience of operation may suffer, especially in small works. It is also frequently urged that with large filters it is difficult or impossible to get an even rate of filtration over the entire area owing[Pg 11] to the frictional resistance of the underdrains for the more distant parts of the filter. A discussion of this point is given in Chapter III, page 41. At Hamburg, where the size of the single beds, 1.88 acres each, is larger than at any other place, it is shown that there is no serious cause for anxiety; and even if there were, the objectionable resistance could be still farther reduced by a few changes in the under-drains. The sizes of filter-beds used at a large number of places are given in Appendix IV.
At a number of places having severe winters, filters are vaulted over as a protection from cold, and in the most important of these, Berlin, Warsaw, and St. Petersburg, the areas of the single beds are nearly the same, namely, from 0.52 to 0.59 acre. The works with open filters at London (seven companies), Amsterdam, and Breslau have filter-beds from 0.82 to 1.50 acres each. Liverpool and Hamburg alone use filters with somewhat larger areas. Large numbers of works with both covered and open filters have much smaller beds than these sizes, but generally this is to avoid too small a number of divisions in a small total area, although such works have sometimes been extended with the growth of the cities until they now have a considerable number of very small basins.
FORM OF FILTER-BEDS.
The form and construction of the filter-beds depend upon local conditions, the foundations, and building materials available, the principles governing these points being in general the same as for the construction of ordinary reservoirs. The bottoms require to be made water-tight, either by a thin layer of concrete or by a pavement upon a puddle layer. For the sides either masonry walls or embankments are used, the former saving space, but being in general more expensive in construction. Embankments must, of course, be substantially paved near the[Pg 12] water-line to withstand the action of ice, and must not be injured by rapid fluctuations in the water-levels in the filters.
Failure to make the bottoms water-tight has perhaps caused more annoyance than any other single point. With a leaky bottom there is either a loss of water when the water in the filters is higher than the ground-water, or under reverse conditions, the ground-water comes in and mixes with the filtered water, and the latter is rarely improved and may be seriously damaged by the admixture. And with very bad conditions water may pass from one filter to another, with the differences in pressure always existing in neighboring filters, with most unsatisfactory results.
COVERS FOR FILTERS.
The filters in England and Holland are built open, without protection from the weather. In Germany the filters first built were also open, but in the colder climates more or less difficulty was experienced in keeping the filters in operation in cold weather. An addition to the Berlin filters, built in 1874, was covered with masonry vaulting, over which several feet of earth were placed, affording a complete protection against frost. The filters at Magdeburg built two years later were covered in the same way, and since that time covered filters have been built at perhaps a dozen different places.

Interior View of Covered Filter, Ashland, Wis.
When in use the water rises nearly to the springing line of the arches.

Covered Filter in Course of Construction, showing Wooden Centers for Masonry Vaulting, Somersworth, N. H.
[To face page 12.]
It was found at Berlin that, owing to the difficulty of properly cleaning the open filters in winter, it was impossible to keep the usual proportion of the area in effective service, and as a result portions of the filters were greatly overtaxed during prolonged periods of cold weather. This resulted in greatly decreased bacterial efficiency, the bacteria in March, 1889, reaching 3000 to 4000 per cc. (with 100,000 in the raw water), although ordinarily the effluent contained less than 100. An epidemic of typhoid fever followed, and was confined to that part of the city supplied [Pg 13] from the Stralau works, the wards supplied from the covered Tegel filters remaining free from fever. Open filters have since been abandoned in Berlin.
At Altona also, where the water is taken from an excessively polluted source, decreased bacterial efficiency has repeatedly resulted in winter, and the occasional epidemics of typhoid fever in that city, which have invariably come in winter, appear to have been directly due to the effect of cold upon the open filters. The city has just extended the open filters, and hopes with an increased reserve area to avoid the difficulty in future without resource to covered filters. (See Appendices II and VII.)
Brunswick, Lübeck, and Frankfort on Oder with cold winters have open filters, but draw their water-supplies from less polluted sources, and have thus far escaped the fate of Berlin and Altona. The new filters at Hamburg also are open. At Zürich, where open and covered filters were long used side by side, the covered filters were much more satisfactory, and the old open filters have recently been vaulted over.
Königsberg originally built open filters, but was afterward obliged to cover them, on account of the severe winters; and at Breslau, where open filters have long been used, the recent additions are vaulted over.
The fact that inferior efficiency of filtration results with open filters during prolonged and severe winter weather is generally admitted, although there is some doubt as to the exact way in which the disturbance is caused. In some works I am informed that in cutting the ice around the edges of the filter and repeatedly piling the loose pieces upon the floating cake, the latter eventually becomes so thickened at the sides that the projecting lower corners actually touch the sand, with the fluctuating levels which often prevail in these works, and that in this way the sediment layer upon the top of the sand is broken and the water rapidly passes without adequate purification at the points of disturbance.
[Pg 14]
This theory is, however, inadequate to account for many cases where such an accumulation of ice is not allowed. In these cases the poor work is not obtained until after the filters have been scraped. The sand apparently freezes slightly while the water is off, and when water is brought back and filtration resumed, normal results are for some reason not again obtained for a time.
In addition to the poorer work from open filters in cold weather, the cost of removing the ice adds materially to the operating expenses, and in very cold climates would in itself make covers advisable.
I have arranged the European filter plants, in regard to which I have sufficient information, in the table on page 15, in the order of the normal mean January temperatures of the respective places. This may not be an ideal criterion of the necessity of covering filters, but it is at least approximate, and in the absence of more detailed comparisons it will serve to give a good general idea of the case. I have not found a single case where covered filters are used where the January temperature is 32° F. or above. In some of these places some trouble is experienced in unusually cold weather, but I have not heard of any very serious difficulty or of any talk of covering filters at these places except at Rotterdam, where a project for covering was being discussed.
Those places having January temperatures below 30° experience a great deal of difficulty with open filters; so much so, that covered filters may be regarded as necessary for them, although it is possible to keep open filters running with decreased efficiency and increased expense by freely removing the ice, with January temperatures some degrees lower.
Where the mean January temperature is 30° to 32° F. there is room for doubt as to the necessity of covering filters, but, judging from the experience of Berlin and Altona, the covered filters are much safer at this temperature.
[Pg 15]
| TABLE OF PLACES HAVING OPEN AND COVERED FILTERS. | ||
|---|---|---|
| ARRANGED ACCORDING TO THE MEAN JANUARY TEMPERATURES. | ||
| Normal Mean January Temperature. Degrees F. |
Place. | Kind of Filters and Results. |
| 37-40° | All English cities | Open filters only are used, and no great difficulty with ice is experienced. |
| 33-35° | Cities in Holland | All filters are open, and there is little serious trouble with ice; but at Amsterdam and Rotterdam the bacteria in effluents are said to be higher in winter than at other times. |
| 32° | Bremen | Open filters. |
| 31° | Altona | Much difficulty with ice in open filters (see Appendices II and VII). |
| 31° | Brunswick | Open filters. |
| 31° | Hamburg | Open filters. |
| 31° | Lübeck | Open filters. |
| 31° | Berlin | Open filters were formerly used, but owing to decreased efficiency in cold weather they have been abandoned for covered ones. |
| 31° | Magdeburg | Covered filters, but a recent addition is not covered. |
| 30° | Frankfort on Oder | Open filters. |
| 30° | Stuttgart | Part of the filters are covered. |
| 30° | Stettin | Part of the filters are covered. |
| 29° | Zürich | Covered filters were much the most satisfactory, and the open ones were covered in 1894. The raw water has a temperature of 35°. |
| 29° | Liegnitz | Open filters. |
| 29° | Breslau | Open filters have been used, but recent additions are covered. |
| 29° | Budapest | Covered filters only. |
| 29° | Posen | Covered filters only. |
| 26° | Königsberg | The original filters were open, but it was found necessary to cover them. |
| 24° | Warsaw | Covered filters only. |
| 16° | St. Petersburg | Covered filters only. |
In case the raw water was drawn from a lake at a depth where its minimum temperature was above 32°, which is the temperature which must ordinarily be expected in surface-waters in winter, open filters might be successfully used in slightly colder places.
The covers are usually of brick or concrete vaulting supported[Pg 16] by pillars at distances of 11 to 15 feet in each direction, the whole being covered by 2 or 3 feet of earth; and the top can be laid out as a garden if desired. Small holes for the admission of air and light are usually left at intervals. The thickness of the masonry and the sizes of the pillars used in some of the earlier German vaultings are unnecessarily great, and some of the newer works are much lighter. For American use, vaulting like that used for the Newton, Mass., covered reservoir[3] should be amply strong.
Roofs have been used at Königsberg, Posen, and Budapest instead of the masonry vaulting. They are cheaper, but do not afford as good protection against frost, and even with great care some ice will form under them.
Provision must be made for entering the filters freely to introduce and remove sand. This is usually accomplished by raising one section of vaulting and building a permanent incline under it from the sand line to a door above the high-water line in the filter.
The cost of building covered filters is said to average fully one half more than open filters.
Among the incidental advantages of covered filters is that with the comparative darkness there is no tendency to algæ growths on the filters in summer, and the frequency of scraping is therefore somewhat reduced. At Zürich, in 1892, where both covered and open filters were in use side by side, the periods between scrapings averaged a third longer in the covered than in the open filters.
It has been supposed that covered filters kept the water cool in summer and warm in winter, but owing to the large volume of water passing, the change in temperature in any case is very slight; Frühling found that even in extreme cases a change of over 3° F. in either direction is rarely observed.

Removing Ice from a Filter, East London.
This represents the greatest accumulation of ice in the history of the
works.
[To face page 16.]
[Pg 17]
At Berlin, where open and covered filters were used side by side at Stralau for twenty years, it was found that, bacterially, the open filters were, except in severe winter weather, more efficient. It was long supposed that this was caused by the sterilizing action of the sunlight upon the water in the open filters. This result, however, was not confirmed elsewhere, and it was finally discovered, in 1893, that the higher numbers were due to the existence of passages in corners on the columns of the vaulted roof and around the ventilators for the underdrains, through which, practically, unfiltered water found its way into the effluent. This at once removes the evidence in favor of the superior bacterial efficiency of open filters and suggests the necessity of preventing such passages. The construction of a ledge all around the walls and pillars four inches wide and a little above the gravel, as shown in the sketch, might be useful in this way, and the slight lateral movement of the water in the sand above would be of no consequence. The sand would evidently make a closer joint with the horizontal ledge than with the vertical wall.
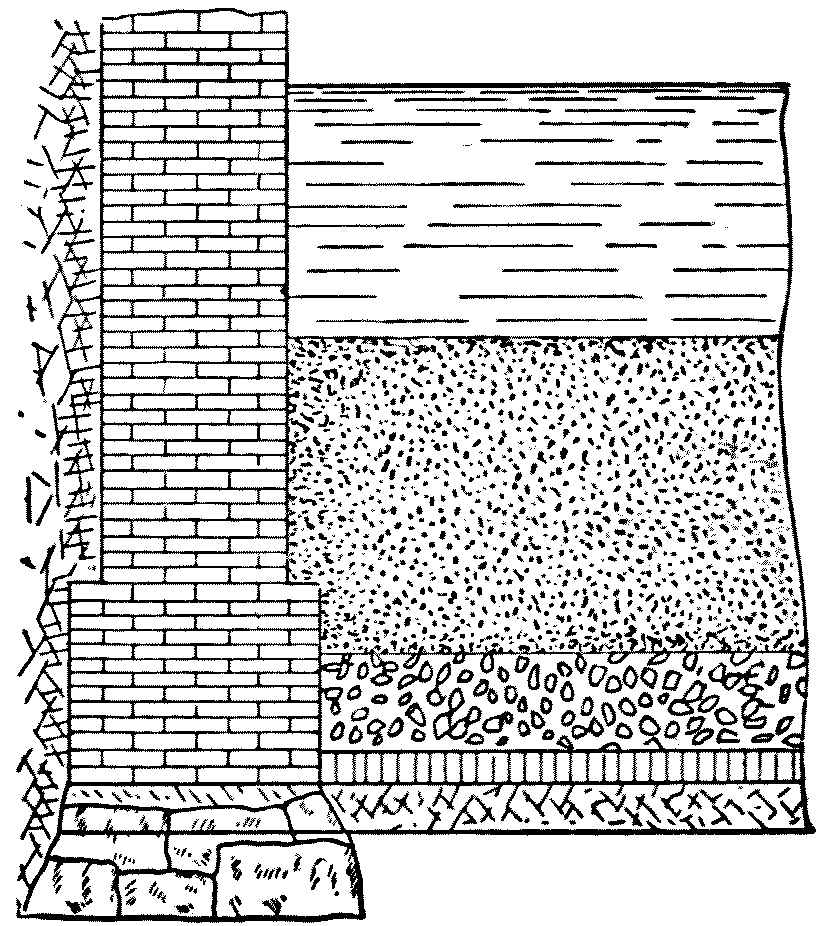
Fig. 2.
In regard to the probable requirement or advisability of covers for filters in the United States, I judge, from the European experience, that places having January temperatures below the freezing-point will have considerable trouble from open filters, and would best have covered filters. Places having higher winter temperatures will be able to get along with the ice which may form on open filters, and the construction of covers would hardly be advisable except under exceptional local conditions, as, for instance, with a water with an unusual tendency to algæ growths.
[Pg 18]
I have drawn a line across a map of the United States on this basis (shown by the accompanying plate) and it would appear that places far north of the line would require covered filters, and that those south of it would not, while for the places in the immediate vicinity of the line (comparable to Hamburg and Altona) there is room for discussion.
In the United States covered filters have been constructed at St. Johnsbury, Vt., Somersworth, N. H., Albany, N. Y., Ashland, Wis., and Grand Forks, N. Dak., all of these places being considerably north of the above-mentioned line.
The filter at Lawrence, Mass., with a mean January temperature of about 25°, is not covered, but serious difficulty and expense have been experienced at times from the ice, so much so that it has been repeatedly recommended to cover it. Open filters have also been in use for many years at Hudson and Poughkeepsie, N. Y., with mean January temperatures about 24°; and although considerable difficulty has been experienced from ice at times, these filters, particularly the ones at Poughkeepsie, have been kept in very serviceable condition at all times, notwithstanding the ice.
At Mount Vernon, N. Y., with a mean January temperature of about 31°, and with a reservoir water, no serious difficulty has been experienced with ice; and at Far Rockaway, L. I., with a slightly higher temperature and well-water, no difficulty whatever has been experienced with open filters. Filters at Ilion, N. Y., with a mean January temperature of about 23°, are not covered, and are fed from a reservoir. No serious difficulty has been experienced with ice, which is probably due to the fact that the water applied to them is taken from near the bottom of the reservoir, and ordinarily has a temperature somewhat above the freezing-point throughout the winter.
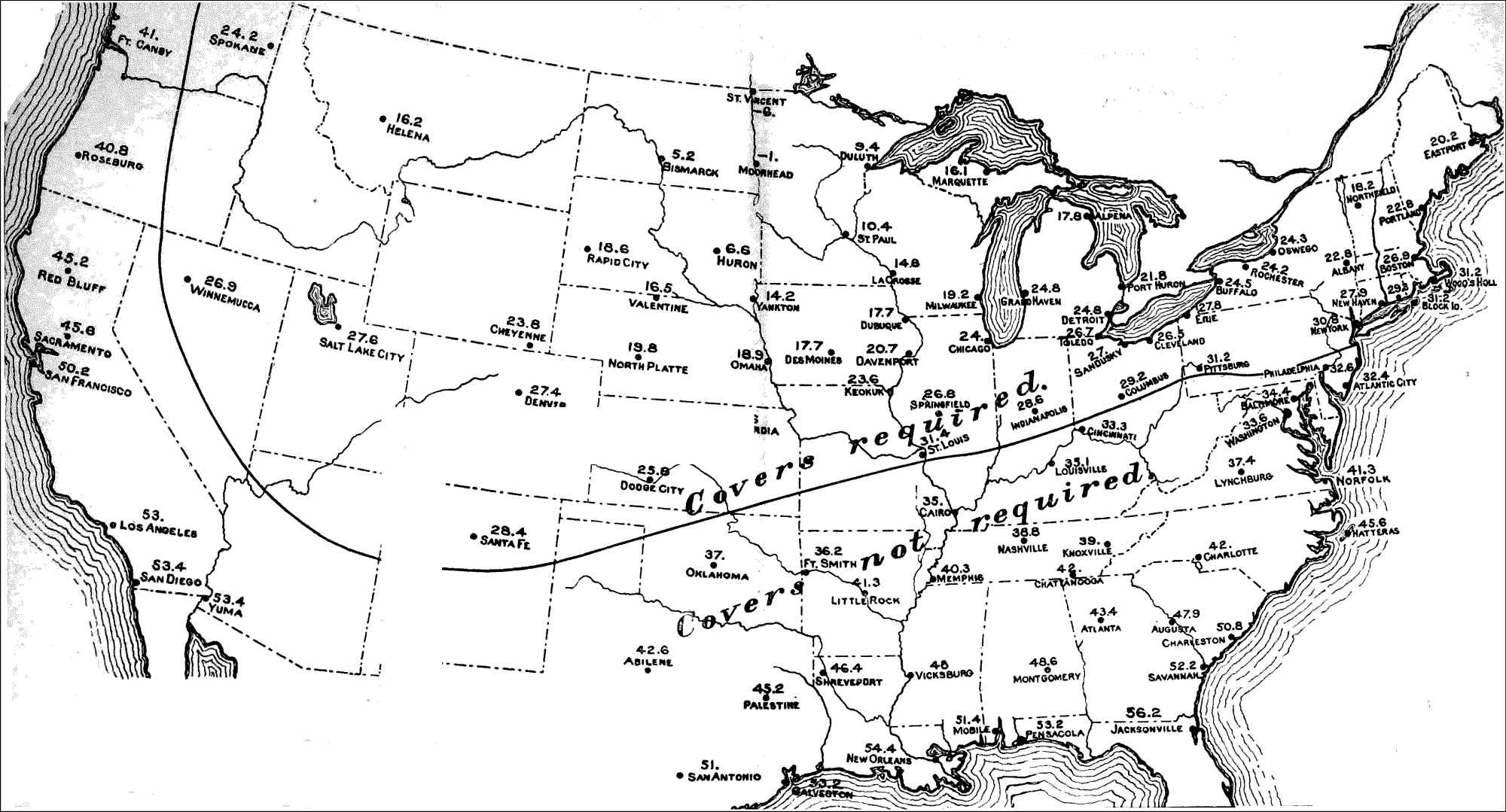
Map showing
Normal Mean January Temperatures
in the United States
and the Area in which Filters should be covered
The cost of removing ice from filters depends, among other things, upon the amount of reserve filter area. When this reserve is small the filters must be kept constantly at work nearly up to their rated capacity; the ice must be removed promptly whenever[Pg 19] the filters require cleaning, and under some conditions the expense of doing this may be considerable. If, on the other hand, there is a considerable reserve area, so that when a filter becomes clogged in severe weather, the work can be turned upon other filters and the clogged filter allowed to remain until more moderate weather, or until a thaw, the expense of ice removal may be kept at a materially lower figure.
In case open filters are built near or north of this line, I would suggest that plenty of space between and around the filters for piling up ice in case of necessity may be found advantageous, and that a greater reserve of filtering area for use in emergencies should be provided than would be considered necessary with vaulted filters or with open filters in a warmer climate.
[Pg 20]
CHAPTER III.
FILTERING MATERIALS.
SAND.
The sand used for filtration may be obtained from the sea-shore, from river-beds or from sand-banks. It consists mainly of sharp quartz grains, but may also contain hard silicates. As it occurs in nature it is frequently mixed with clayey or other fine particles, which must be removed from it by washing before it is used. Some of the New England sands, however, as that used for the Lawrence City filter, are so clean that washing would be superfluous.
The grain size of the sand best adapted to filtration has been variously stated at from 1⁄8 to 1 mm., or from 0.013 to 0.040 inch. The variations in the figures, however, are due more to the way that the same sand appears to different observers than to actual variations in the size of sands used, which are but a small fraction of those indicated by these figures.
As a result of experiments made at the Lawrence Experiment Station[4] we have a standard by which we can definitely compare various sands. The size of a sand-grain is uniformly taken as the diameter of a sphere of equal volume, regardless of its shape. As a result of numerous measurements of grains of Lawrence sands, it is found that when the diameter, as given above, is 1, the three axes of the grain, selecting the longest possible and taking the other two at right angles to it, are, on an average, 1.38, 1.05, and 0.69, respectively and the mean diameter is equal to the cube root of their product.
[Pg 21]
It was also found that in mixed materials containing particles of various sizes the water is forced to go around the larger particles and through the finer portions which occupy the intervening spaces, so that it is the finest portion which mainly determines the character of the sand for filtration. As a provisional basis which best accounts for the known facts, the size of grain such that 10 per cent by weight of the particles are smaller and 90 per cent larger than itself, is considered to be the effective size. The size so calculated is uniformly referred to in speaking of the size of grain in this work.
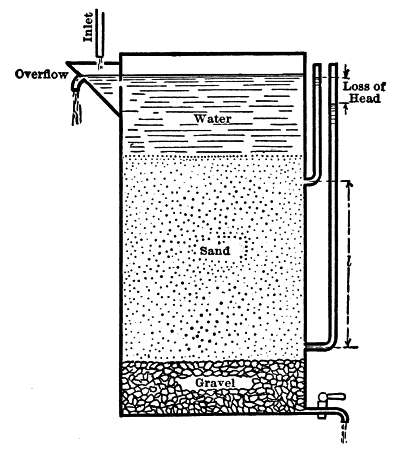
Fig. 3.—Apparatus Used for Measuring the Friction of Water in Sands.
Another important point in regard to a material is its degree of uniformity—whether the particles are mainly of the same size or whether there is a great range in their diameters. This is shown by the uniformity coefficient, a term used to designate the ratio of the size of the grain which has 60 per cent of the sample finer than itself to the size which has 10 per cent finer than itself.
[Pg 22]
The frictional resistance of sand to water when closely packed, with the pores completely filled with water and in the entire absence of clogging, was found to be expressed by the formula
v = cd2(h/l)(t Fah. + 10°)/60,
where v is the velocity of the water in meters daily in a solid column of the same area as that of the sand, or approximately in million gallons per acre daily;
c is an approximately constant factor;
d is the effective size of sand grain in millimeters;
h is the loss of head (Fig. 3);
l is the thickness of sand through which the water passes;
t is the temperature (Fahr.).
| TABLE SHOWING RATE AT WHICH WATER WILL PASS THROUGH EVEN-GRAINED AND CLEAN SANDS OF THE STATED GRAIN SIZES AND WITH VARIOUS HEADS AT A TEMPERATURE OF 50°. | ||||||||
|---|---|---|---|---|---|---|---|---|
| h l |
Effective Size in Millimeters 10 per cent finer than: | |||||||
| 0.10 | 0.20 | 0.30 | 0.35 | 0.40 | 0.50 | 1.00 | 3.00 | |
| Million Gallons per Acre daily. | ||||||||
| .001 | .01 | .04 | .10 | .13 | .17 | .27 | 1.07 | 9.63 |
| .005 | .05 | .21 | .48 | .65 | .85 | 1.34 | 5.35 | 48.15 |
| .010 | .11 | .43 | .96 | 1.31 | 1.71 | 2.67 | 10.70 | 96.30 |
| .050 | .54 | 2.14 | 4.82 | 6.55 | 8.55 | 13.40 | 53.50 | |
| .100 | 1.07 | 4.28 | 9.63 | 13.10 | 17.10 | 26.70 | 107.00 | |
| 1.000 | 10.70 | 42.80 | 96.30 | 131.00 | 171.00 | 267.00 | ||
The above table is computed with the value c taken as 1000, this being approximately the values deduced from the earliest experiments. More recent and extended data have shown that the value of c is not entirely constant, but depends upon the uniformity coefficient, upon the shape of the sand grains, upon their chemical composition, and upon the cleanliness and closeness of packing of the sand. The value may be as high as 1200 for very uniform, and perfectly clean sand, and maybe as low as 400[Pg 23] for very closely packed sands containing a good deal of alumina or iron, and especially if they are not quite clean. The friction is usually less in new sand than in sand which has been in use for some years. In making computations of the frictional resistance of filters, the average value of c may be taken at from 700 to 1000 for new sand, and from 500 to 700 for sand which has been in use for a number of years.
The value of c decreases as the uniformity coefficient increases. With ordinary filter sands with uniformity coefficients of 3 or less the differences are not great. With mixed sands having much higher uniformity coefficients, lower and less constant values of c are obtained, and the arrangement of the particles becomes a controlling factor in the increase in friction.
The friction of the surface layer of a filter is often greater than that of all the sand below the surface. It must be separately computed and added to the resistances computed by the formula, as it depends largely upon other conditions than those controlling the resistance of the sand.
While the value of c is thus not entirely constant, it can be estimated with approximate accuracy for various conditions, from a knowledge of the composition, condition, and cleanliness of the sand, and closeness of packing.
The following table shows the quantity of water passing sands at different temperatures. This table was computed with temperature factors as given above, which were based upon experiments upon the flow of water through sands, checked by the coefficients obtained from experiments with long capillary tubes entirely submerged in water of the required temperature.
| RELATIVE QUANTITIES OF WATER PASSING AT DIFFERENT TEMPERATURES. | |
|---|---|
| 32° | 0.70 |
| 35° | 0.75 |
| 38° | 0.80 |
| 41° | 0.85 |
| 44° | 0.90 |
| 47° | 0.95 |
| 50° | 1.00 |
| 53° | 1.05 |
| 56° | 1.10 |
| 59° | 1.15 |
| 62° | 1.20 |
| 65° | 1.25 |
| 68° | 1.30 |
| 71° | 1.35 |
| 74° | 1.40 |
| 77° | 1.45 |
[Pg 24]
The effect of temperature upon the passage of water through sands and soils has been further discussed by Prof. L. G. Carpenter, Engineering News, Vol. XXXIX, p. 422. This article reviews briefly the literature of the subject, and refers at length to the formula of Poiseuille, published in the Memoires des Savants Etrangers, Vol. XI, p. 433 (1846). This formula, in which the quantity of water passing at 0.0° Cent., is taken as unity, is as follows:
Temperature factor = 1 + 0.033679t + 0.000221t2.
The results obtained by this formula agree very closely with those given in the above table throughout the temperature range for which computations are most frequently required. At the higher and lower temperatures the divergencies are greater, as is shown in a communication in the Engineering News, Vol. XL, p. 26.
The quantity of water passing at a temperature of 50° Fahr. is in many respects more convenient as a standard than the quantity passing at the freezing-point. Near the freezing-point, owing to molecular changes in the water, the changes in its action are rapid, and the results are less certain, and also 50° Fahr. is a much more convenient temperature for precise experiments than is the freezing point.
SANDS USED IN EUROPEAN FILTERS.
To secure definite information in regard to the qualities of the sands actually used in filtration, a large number of European works were visited in 1894, and samples of sand were collected for analysis. These samples were examined at the Lawrence Experiment Station by Mr. H. W. Clark, the author’s method of analysis described in Appendix III being used. In the following table, for the sake of compactness, only the leading points of the analyses, namely, effective size, uniformity coefficient, and albuminoid ammonia, are given. On page 28 full analyses of some samples from a few of the leading works are given.
[Pg 25]
| ANALYSES OF SANDS USED IN WATER FILTRATION. | ||||
|---|---|---|---|---|
| Source. | Effective Size; 10% Finer than (Milli- meters). |
Uni- formity Coeffi-cient. |
Albu- minoid Ammo- ia. Parts in 100,000. |
Remarks. |
| London, E. London Co. | 0.44 | 1.8 | 0.45 | New sand, never used or washed. |
| London, E. London Co. | 0.39 | 2.1 | 26.20 | Dirty sand, very old. |
| London, E. London Co. | 0.37 | 2.0 | 8.60 | Same, washed by hand. |
| London, Grand Junc. | 0.26 | 1.9 | 1.90 | Sand from rough filter. |
| London, Grand Junc. | 0.40 | 3.5 | 10.00 | Old sand in final filter. |
| London, Grand Junc. | 0.41 | 3.7 | 2.70 | Freshly washed old sand. |
| London, Southw’k & V. | 0.38 | 3.5 | 5.00 | Freshly washed old sand. |
| London, Southw’k & V. | 0.30 | 1.8 | 2.80 | Freshly washed new sand. |
| London, Lambeth | 0.36 | 2.3 | 2.60 | Freshly washed old sand. |
| London, Lambeth | 0.36 | 2.4 | 0.35 | New unused sand, washed. |
| London, Lambeth | 0.25 | 1.7 | 0.70 | New extremely fine sand. |
| London, Chelsea | 0.36 | 2.4 | 2.10 | Freshly washed old sand. |
| Middlesborough | 0.42 | 1.6 | 17.60 | Dirty sand, ordinary scraping. |
| Middlesborough | 0.43 | 1.6 | 7.30 | Same, after washing. |
| Birmingham | 0.29 | 1.9 | 33.20 | Dirty sand. |
| Birmingham | 0.29 | 1.9 | 7.20 | Sand below surface of filter. |
| Reading | 0.30 | 2.5 | 4.00 | Dirty sand. |
| Reading | 0.22 | 2.0 | 1.50 | Same, after washing. |
| Antwerp | 0.38 | 1.6 | 7.80 | Dirty sand. |
| Antwerp | 0.39 | 1.6 | 3.40 | Same, after washing. |
| Hamburg | 0.28 | 2.5 | 8.50 | Dirty sand. |
| Hamburg | 0.31 | 2.3 | 0.80 | Same, after washing. |
| Hamburg | 0.34 | 2.2 | 7.90 | Dirty sand, another sample. |
| Hamburg | 0.30 | 2.0 | 0.90 | Same, after washing drums. |
| Hamburg | 0.34 | 2.3 | 1.50 | Same, after washing ejectors. |
| Altona | 0.32 | 2.0 | 9.00 | Dirty sand, old filters. |
| Altona | 0.37 | 2.0 | 1.50 | Same, after washing. |
| Altona | 0.33 | 2.8 | 0.50 | Washed sand for new filters. |
| Berlin, Stralau | 0.33 | 1.9 | 12.20 | Dirty sand-pile. |
| Berlin, Stralau | 0.35 | 1.7 | 4.50 | Filter No. 6, 3″ below surface. |
| Berlin, Stralau | 0.34 | 1.7 | 6.30 | Filter No. 7 3″ below surface. |
| Berlin, Stralau | 0.35 | 1.7 | 4.00 | Filter No. 10 3″ below surface. |
| Berlin, Tegel | 0.38 | 1.6 | 11.00 | Dirty sand, old filters. |
| Berlin, Tegel | 0.38 | 1.5 | 2.80 | Same, after washing, old filters. |
| Berlin, Tegel | 0.35 | 1.6 | 3.20 | Same, after washing, new filters. |
| Berlin, Müggel | 0.35 | 1.8 | 0.80 | Sand from filters below surface. |
| Berlin, Müggel | 0.33 | 2.0 | 6.30 | Dirty sand, ordinary scraping. |
| Berlin, Müggel | 0.34 | 2.0 | 15.30 | Dirty sand, another sample. |
| Charlottenburg | 0.40 | 2.3 | 7.20 | Dirty sand. |
| Chemnitz | 0.35 | 2.6 | 0.20 | New sand not yet used. |
| Magdeburg | 0.39 | 2.0 | 9.50 | Dirty sand. |
| Magdeburg | 0.40 | 2.0 | 2.80 | Same, after washing. |
| Breslau | 0.39 | 1.8 | 1.40 | Normal new sand. |
| Budapest | 0.20 | 2.0 | 0.80 | New washed Danube sand. |
| Zürich | 0.28 | 3.2 | 6.20 | Dirty sand. |
| Zürich | 0.30 | 3.1 | 1.50 | Same, after washing. |
| Hague | 0.19 | 1.6 | 0.70 | Dune-sand used for filtration. [Pg 26] |
| Schiedam | 0.18 | 1.6 | 5.60 | Dune-sand used for filtration; dirty. |
| Schiedam | 0.31 | 1.5 | 13.50 | River-sand; dirty. |
| Amsterdam | 0.17 | 1.6 | 2.40 | Dune-sand. |
| Rotterdam | 0.34 | 1.5 | 2.30 | River-sand; new. |
| Liverpool, Rivington | 0.43 | 2.0 | 0.76 | Sand from bottom of filter. |
| Liverpool, Rivington | 0.32 | 2.5 | 1.00 | New sand unwashed and unscreened. |
| Liverpool, Rivington | 0.43 | 2.7 | 4.10 | Washed sand which has been in use 30 to 40 years. |
| Liverpool, Oswestry | 0.30 | 2.6 | 9.40 | Dirty sand. |
| Liverpool, Oswestry | 0.31 | 4.7 | 2.20 | Same, after washing. |
Note.—It is obvious that in case the sands used at any place are not always of the same character, as is shown to be the case by different samples from some of the works, the examination of such a limited number of samples as the above from each place is entirely inadequate to establish accurately the sizes of sand used at that particular place, or to allow close comparisons between the different works, and for this reason no such comparisons will be made. The object of these investigations was to determine the sizes of the sands commonly used in Europe, and, considering the number and character of the different works represented, it is believed that the results are ample for this purpose.
The English and most of the German sands are washed, even when entirely new, before being used, to remove fine particles. At Breslau, however, sand dredged from the river Oder is used in its natural state, and new sand is used for replacing that removed by scraping. At Budapest, Danube sand is used in the same way, but with a very crude washing, and it is said that only new unwashed sand is used at Warsaw.
In Holland, so far as I learned, no sand is washed, but new sand is always used for refilling. At most of the works visited dune-sand with an effective size of only 0.17 to 0.19 mm. is used, and this is the finest sand which I have ever found used for water filtration on a large scale. It should be said, however, that the waters filtered through these fine sands are fairly clear before[Pg 27] filtration, and are not comparable to the turbid river-waters often filtered elsewhere, and their tendency to choke the filters is consequently much less. At Rotterdam and Schiedam, where the raw water is drawn from the Maas, as the principal stream of the Rhine is called in Holland, river-sand of much larger grain size is employed. It is obtained by dredging in the river and is never washed, new sand always being employed for refilling.
The average results of the complete analyses of sands from ten leading works are shown in the table on page 28. These figures are the average of all the analyses for the respective places, except that one sample from the Lambeth Co., which was not a representative one, was omitted.
The London companies were selected for this comparison both on account of their long and favorable records in filtering the polluted waters of the Thames and Lea, and because they are subject to close inspection; and there is ample evidence that the filtration obtained is good—evidence which is often lacking in the smaller and less closely watched works. For the German works Altona was selected because of its escape from cholera in 1892, due to the efficient action of its filters, and Stralau because of its long and favorable record when filtering the much-polluted Spree water. These two works also have perhaps contributed more to the modern theories of filtration than all the other works in existence. The remaining works are included because they are comparatively new, and have been constructed with the greatest care and attention to details throughout, and the results obtained are most carefully recorded.
Some of the most interesting of these results are shown graphically on page 29. The method of plotting is that described in Appendix III.
[Pg 28]
| TABLE SHOWING THE AVERAGE PER CENT OF THE GRAINS FINER THAN VARIOUS SIZES IN SANDS FROM LEADING WORKS. | ||||||||
|---|---|---|---|---|---|---|---|---|
| Per Cent by Weight Finer than | ||||||||
| 0.106 mm. |
0.186 mm. |
0.316 mm. |
0.46 mm. |
0.93 mm. |
2.04 mm. |
3.89 mm. |
5.89 mm. |
|
| East London | 0.2 | 0.5 | 3.6 | 22.2 | 69.7 | 89.8 | 95.0 | 99.0 |
| Grand Junction | 0 | 0.2 | 3.1 | 17.4 | 47.1 | 68.2 | 84.7 | 93.6 |
| Southwark and Vauxhall | 0.7 | 8.0 | 34.1 | 69.7 | 83.5 | 90.0 | 94.0 | |
| Lambeth | 0 | 0.5 | 5.5 | 26.6 | 63.0 | 79.2 | 88.0 | 94.3 |
| Chelsea | 0 | 0.1 | 5.0 | 28.6 | 63.0 | 76.7 | 86.0 | 93.6 |
| Hamburg | 0.2 | 1.5 | 10.9 | 33.2 | 74.4 | 95.7 | 99.5 | |
| Altona | 0.1 | 1.1 | 7.8 | 28.7 | 72.1 | 92.1 | 95.8 | |
| Stralau | 0.3 | 7.0 | 37.3 | 86.9 | 95.4 | 97.6 | ||
| Tegel | 0.2 | 4.5 | 35.4 | 94.3 | 98.5 | 99.1 | ||
| Müggel | 0.1 | 0.5 | 7.9 | 33.6 | 79.7 | 94.3 | 98.5 | |
Average of all |
0.06 | 0.56 | 6.33 | 29.71 | 71.99 | 87.34 | 93.42 | (97.45) |
| AVERAGE EFFECTIVE SIZE, UNIFORMITY COEFFICIENT, AND ALBUMINOID AMMONIA IN SANDS FROM TEN LEADING WORKS. |
||||
|---|---|---|---|---|
| I. LONDON FILTERS. | ||||
| Effective Size; 10% Finer than (Millimeters). |
Uniformity Coefficient. |
Albuminoid Ammonia. | ||
| Dirty Sand. | Washed Sand. | |||
| East London | 0.40 | 2.0 | 26.00 | 8.60 |
| Grand Junction | 0.40 | 3.6 | 10.00 | 2.70 |
| Southwark and Vauxhall | 0.34 | 2.5 | ..... | 3.90 |
| Lambeth | 0.36 | 2.4 | ..... | 2.60 |
| Chelsea | 0.36 | 2.4 | ..... | 2.10 |
| Average | 0.37 | 2.6 | 18.00 | 3.98 |
| II. GERMAN WORKS. |
||||
| Stralau | 0.34 | 1.7 | 12.20 | 4.00 |
| Tegel | 0.37 | 1.6 | 11.00 | 3.00 |
| Müggel | 0.34 | 2.0 | 10.80 | 0.80 |
| Altona | 0.34 | 2.3 | 9.00 | 1.50 |
| Hamburg | 0.31 | 2.3 | 8.20 | 1.07 |
| Average | 0.34 | 2.0 | 10.25 | 2.07 |

Placing Sand in a Filter, Hamburg.
[To face page 28.
[Pg 29]
The averages show the effective size of the English sands to be slightly greater than that of the German sands—0.37 instead of 0.34 mm.—but the difference is very small. The entire range for the ten works is only from 0.31 to 0.40 mm., and these may be taken as the ordinary limits of effective size of the sands employed in the best European works. The average for the other sixteen works given above, including dune-sands, is 0.31 mm., or, omitting the dune-sands, 0.34 mm.
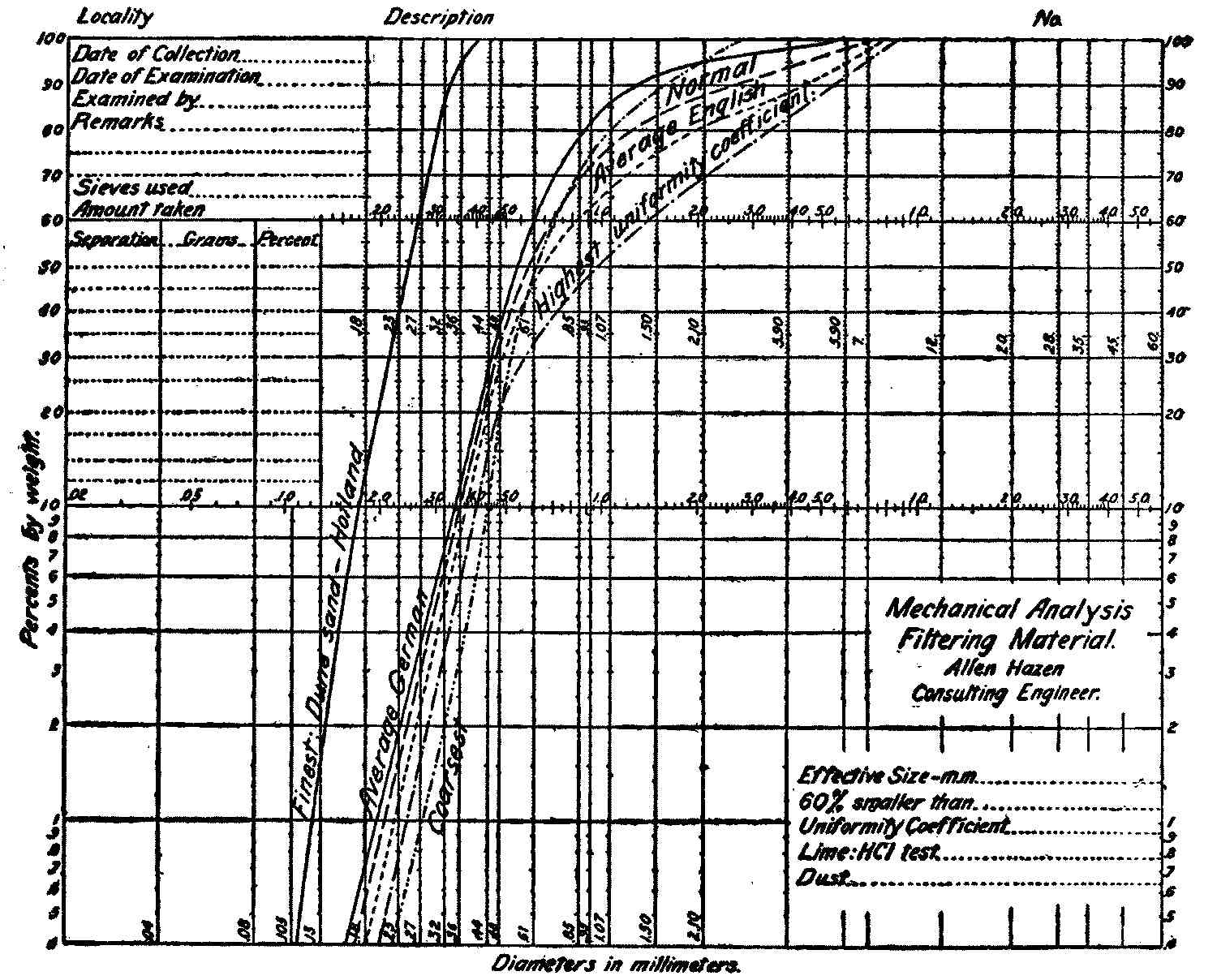
Fig. 3a.—Sand Analysis Sheet, with Analyses of Several European Filter Sands.
It is important that filter sands should be free from lime. When water is filtered through such sands, no increase in hardness results. When, however, water is filtered through sand containing[Pg 30] lime, some of it is usually dissolved and the water is made harder. The amount of lime taken up in this way depends both upon the character of the sand, and upon the solvent power of the water; and it does not necessarily follow that a sand containing lime cannot be used for filtration, but a sand nearly free from lime is to be preferred.
The presence of lime in sand can usually be detected by moistening it with hydrochloric acid. The evolution of gas shows the presence of lime. Some idea of the amount of lime can be obtained from the amount of gas given off, and the appearance of the sample after the treatment, but chemical analysis is necessary to determine correctly the amount.
Experiments with filters at Pittsburg were made with sand containing 1.3 per cent of lime, the result being that the hardness of the water was increased about one part in 100,000; but the amount of lime in the sand was so small that it would be washed out after a time, and then the hardening effect would cease. Larger amounts of lime would continue their action for a number of years and would be more objectionable.
Turning to the circumstances which influence the selection of the sand size, we find that both the quality of the effluent obtained by filtration and the cost of filtration depend upon the size of the sand-grains.
With a fine sand the sediment layer forms more quickly and the removal of bacteria is more complete, but, on the other hand, the filter clogs quicker and the dirty sand is more difficult to wash, so that the expense is increased.
EFFECT OF SIZE OF GRAIN UPON EFFICIENCY OF FILTRATION.
It is frequently stated that it is only the sediment layer which performs the work of filtration, and that the sand which supports it plays hardly a larger part than does the gravel which[Pg 31] carries the sand, and under some circumstances this is undoubtedly the case. Nevertheless sand in itself, without any sediment layer, especially when not too coarse and not in too thin layers, has very great purifying powers, and, in addition, acts as a safeguard by positively preventing excessive rates of filtration on account of its frictional resistance. As an illustration take the case of a filter of sand with an effective size of 0.35 mm. and the minimum thickness of sand allowed by the German Board of Health, namely, one foot, and let us suppose that with clogging the loss of head has reached two feet to produce the desired velocity of 2.57 million gallons per acre daily. Suppose now that by some accident the sediment layer is suddenly broken or removed from a small area, the water will rush through this area, until a new sediment layer is formed, at a rate corresponding to the size, pressure, and depth of the sand, or 260 million gallons per acre daily—a hundred times the standard rate. Under these conditions the passing water will not be purified, but will pollute the entire effluent from the filter. Under corresponding conditions, with a deep filter of fine sand, say with an effective size of 0.20 mm. and 5 feet deep, the resulting rate would be only 17 million gallons per acre daily, or less than seven times the normal, and with the water passing through the full depth of fine sand, the resulting deterioration in the effluent before the sand again became so clogged as to reduce the rate to nearly the normal, would be hardly appreciable.
The results at Lawrence have shown that with very fine sands 0.09 and 0.14 mm., and 4 to 5 feet deep, with the quantity of water which can practically be made to pass through them, it is almost impossible to drive more than an insignificant fraction of the bacteria into the effluent. Even when the sands are entirely new, or have been scraped or disturbed in the most violent way, the first effluent passing, before the sediment layer could have been formed, is of good quality. Still finer materials, 0.04 to 0.06 mm., as far as could be determined, secured the absolute[Pg 32] removal of all bacteria, but the rates of filtration which were possible were so low as to preclude their practical application.
With coarser sands, as long as the filter is kept at a steady rate of filtration, without interruptions of any kind, entirely satisfactory results are often obtained, although never quite so good as with the finer sands. Thus at Lawrence the percentages of bacteria (B. prodigiosus) appearing in the effluents under comparable conditions were as follows:
| 1892 | 1893 | |
|---|---|---|
| With effective grain size 0.38 mm | 0.16 | .... |
| With effective grain size 0.29 mm | 0.16 | .... |
| With effective grain size 0.26 mm | 0.10 | .... |
| With effective grain size 0.20 mm | 0.13 | 0.01 |
| With effective grain size 0.14 mm | 0.04 | 0.03 |
| With effective grain size 0.09 mm | 0.02 | 0.02 |
We may thus conclude that fine sands give normally somewhat better effluents than coarser ones, and that they are much more likely to give at least a tolerably good purification under unusual or improper conditions.
EFFECT OF GRAIN SIZE UPON FREQUENCY OF SCRAPING.
The practical objection to the use of fine sand is that it becomes rapidly clogged, so that filters require to be scraped at shorter intervals, and the sand washing is much more difficult and expensive. The quantities of water filtered between successive scrapings at Lawrence in millions of gallons per acre under comparable conditions have been as follows:
| 1892 | 1893 | |
|---|---|---|
| Effective size of sand grain 0.38 mm | .... | 79 |
| Effective size of sand grain 0.29 mm | .... | 70 |
| Effective size of sand grain 0.26 mm | .... | 57 |
| Effective size of sand grain 0.20 mm | 58 | .... |
| Effective size of sand grain 0.14 mm | 45 | 49 |
| Effective size of sand grain 0.09 mm | 24 | 14 |
[Pg 33]
The increase in the quantities passed between scrapings with increasing grain size is very marked.
With the fine sands, the depth to which the sand becomes dirty is much less than with the coarse sands, but as it is not generally practicable to remove a layer of sand less than about 0.6 inch thick, even when the actual clogged layer is thinner than this, the full quantity of sand has to be removed; and the quantities of sand to be removed and washed are inversely proportional to the quantities of water filtered between scrapings. On the other hand, with very coarse sands the sediment penetrates the sand to a greater depth than the 0.6 inch necessarily removed, so that a thicker layer of sand has to be removed, which may more than offset the longer interval. This happens occasionally in water-works, and a sand coarse enough to allow it occur is always disliked by superintendents, and is replaced with finer sand as soon as possible. It is obvious that the minimum expense for cleaning will be secured with a sand which just does not allow this deep penetration, and I am inclined to think that the sizes of the sands in use have actually been determined more often than otherwise in this way, and that the coarsest samples found, having effective sizes of about 0.40 mm., represent the practical limit to the coarseness of the sand, and that any increase above this size would be followed by increased expense for cleaning as well as by decreased efficiency.
SELECTION OF SAND.
In selecting a sand for filtration, when it is considered that repeated washings will remove some of the finest particles, and so increase slightly the effective size, a new sand coarser than 0.35 mm. would hardly be selected. Perhaps 0.20 might be given as a suitable lower limit. For comparatively clear lake- or reservoir-waters a finer sand could probably be used than would be the case with a turbid river-water. A mixed sand having a[Pg 34] uniformity coefficient above 3.0 would be difficult to wash without separating it into portions of different sizes, and, in general, the lower the coefficient, that is, the more uniform the grain sizes, the better. Great pains should be taken to have the sand of the same quality throughout, especially in the same filter, as any variations in the grain sizes would lead to important variations in the velocity of filtration, the coarser sands passing more than their share of water (in proportion to the square of the effective sizes) and with reduced efficiency.
At Lawrence a sufficient quantity of natural sand was found of the grade required; but where suitable material cannot be so obtained it is necessary to use other methods. A mixed material can be screened from particles which are too large, and can be washed to free it from its finer portions, and in this way a good sand can be prepared, if necessary, from what might seem to be quite unpromising material. The methods of sand-washing will be described in Chapter V.
THICKNESS OF THE SAND LAYER.
The thickness of the sand layer is made so great that when it is repeatedly scraped in cleaning the sand will not become too thin for good filtration for a considerable time. When this occurs the removed sand must be replaced with clean sand. The original thickness of the sand in European filters is usually from 24 to 48 inches, thicknesses between 30 and 40 inches being extremely common, and this is reduced before refilling to from 12 to 24 inches. The Imperial Board of Health of Germany has fixed 12 inches as a limit below which the sand should never be scraped, and a higher limit is recommended wherever possible.
A thick sand layer has the same steadying action as a fine sand, and tends to prevent irregularities in the rate of filtration in proportion to its frictional resistance, and that without increasing the frequency of cleaning; but, on the other hand, it increases[Pg 35] the necessary height of the filter, throughout, and consequently the cost of construction.
In addition to the steadying effect of a deep sand layer, some purification takes place in the lower part of the sand even with a good sediment layer on the surface, and the efficiency of deep filters is greater than that of shallow ones.
Layers of finer materials, as fine sand or loam, in the lower part of a filter, which would otherwise give increased efficiency without increasing the operating expenses, cannot be used. Their presence invariably gives rise sooner or later to sub-surface clogging at the point of junction with the coarser sand, as has been found by repeated tests at Lawrence as well as in some of the Dutch filters where such layers were tried; and as there is no object in putting a coarser sand under a finer, the filter sand is best all of the same size and quality from top to bottom.
UNDERDRAINING.
The underdrains of a filter are simply useful for collecting the filtered water; they play no part in the purification. One of the first requirements of successful filtration is that the rate of filtration shall be practically the same in all parts of the filter. This is most difficult to secure when the filter has just been cleaned and the friction of the sand layer is at a minimum. If the friction of the water in entering and passing through the underdrains is considerable, the more remote parts of the filters will work under less pressure, and will thus do less than their share of the work, while the parts near the outlet will be overtaxed, and filtering at too high rates will yield poor effluents.
To avoid this condition the underdrains must have such a capacity that their frictional resistance will be only a small fraction of the friction in the sand itself just after cleaning.
GRAVEL LAYERS.
The early filters contained an enormous quantity of gravel, but the quantity has been steadily reduced in successive plants.[Pg 36] Thus in 1866 Kirkwood, as a result of his observations, recommended the use of a layer four feet thick, and in addition a foot of coarse sand, while at the present time new filters rarely have more than two feet of gravel. Even this quantity seems quite superfluous, when calculations of its frictional resistance are made. Thus a layer of gravel with an effective size of 20 mm.[5] (which is much finer than that generally employed) only 6 inches thick will carry the effluent from a filter working at a rate of 2.57 million gallons per acre daily for a distance of 8 feet (that is, with underdrains 16 feet apart), with a loss of head of only 0.001 foot, and for longer distances tile drains are cheaper than gravel. To prevent the sand from sinking into the coarse gravel, intermediate sizes of gravel must be placed between, each grade being coarse enough so that there is no possibility of its sinking into the layer below. The necessary thickness of these intermediate layers is very small, the principal point being to have a layer of each grade at every point. Thus on the 6 inches of 20 mm. gravel mentioned above, three layers of two inches each, of 8 and 3 mm. gravel and coarse sand, with a total height of six inches, or other corresponding and convenient depths and sizes, would, if carefully placed, as effectually prevent the sinking of the filter sand into the coarse gravel as the much thicker layers used in the older plants.
The gravel around the drains should receive special attention. Larger stones can be here used with advantage, taking care that adequate spaces are left for the entrance of the water into the drains at a low velocity, and to make everything so solid in this neighborhood that there will be no chance for the stones to settle which might allow the sand to reach the drains.

Reconstructing the Underdrainage System of a Filter after 25 Years of Use, Bremen.

Placing Sand in a Filter, Choisy le Roi (Paris).
[To face page 36.]
At the Lawrence filter, at Königsberg in Prussia, at Amsterdam and other places, the quantity of gravel is reduced by putting the drains in trenches, so that the gravel is reduced from[Pg 37] a maximum thickness at the drain to nothing half way between drains. The economy of the arrangement, however, as far as friction is concerned is not so great as would appear at first sight, and the cost of the bottom may be increased; but on the other hand it gives a greater depth of gravel for covering the drains with a small total amount of gravel.
As even a very small percentage of fine material is capable of getting in the narrow places and reducing the carrying power of the gravel, it is important that all such matters should be carefully removed by washing before putting the gravel in place. In England and Germany gravel is commonly screened for use in revolving cylinders of wire-cloth of the desired sizes, on which water is freely played from numerous jets, thus securing perfectly clean gravel. In getting gravel for the Lawrence filter, an apparatus was used, in which advantage was taken of the natural slope of the gravel bank to do the work, and the use of power was avoided. The respective grades of gravel obtained were even in size, and reasonably free from fine material, but it was deemed best to wash them with a hose before putting them in the filter.
To calculate the frictional resistance of water in passing gravel, we may assume that for the very low velocities which are actually found in filters the quantity of water passing varies directly with the head, which for these velocities is substantially correct, although it would not be true for higher rates, especially with the coarser gravels.[6] In the case of parallel underdrains the friction from the middle point between drains to the drains may be calculated by the formula:
Total head = (1⁄2)[(Rate of filtration × (1⁄2 distance between drains)2)/(Average depth of gravel × discharge coefficient)].
The discharge coefficient for any gravel is 1000 times the quantity [Pg 38] of water which will pass when h⁄l is 1⁄1000 expressed in million gallons per acre daily. The approximate values of this coefficient for different-sized gravels are as follows:
| VALUES OF DISCHARGE COEFFICIENT. | |||
|---|---|---|---|
| For gravel with effective size | 5 mm | c = | 23,000 |
| For gravel with effective size | 10 mm | c = | 65,000 |
| For gravel with effective size | 15 mm | c = | 110,000 |
| For gravel with effective size | 20 mm | c = | 160,000 |
| For gravel with effective size | 25 mm | c = | 230,000 |
| For gravel with effective size | 30 mm | c = | 300,000 |
| For gravel with effective size | 35 mm | c = | 390,000 |
| For gravel with effective size | 40 mm | c = | 480,000 |
Example: What is the loss of head in the gravel at a rate of filtration of 2 million gallons per acre daily, with underdrains 20 feet apart, where the supporting gravel has an effective size of 35 millimeters, and is uniformly 1 ft. deep?
Total head = (1⁄2)[(2 × 102)/(1 × 390,000)] = .000256 ft.
The total friction would be the same with the same average depth of gravel whether it was uniformly 1 foot deep, or decreasing from 1.5 at the drains to 0.5 in the middle, or from 2.0 to 0. The reverse case with the gravel layer thicker in the middle than at the drains does not occur and need not be discussed.
The depth of gravel likely to be adopted as a result of this calculation, when the drains are not too far apart, will be much less than that actually used in most European works, but as the two feet or more there employed are, I believe, simply the result of speculation, there is no reason for following the precedent where calculations show that a smaller quantity is adequate.
The reason for recommending a thin lower layer of coarse gravel, which alone is assumed to provide for the lateral movement[Pg 39] of the water, is that if more than about six inches of gravel is required to give a satisfactory resistance, it will almost always be cheaper to use more drains instead of more gravel; and the reason for recommending thinner upper layers for preventing the sand from settling into the coarse gravel is that no failures of this portion of filters are on record, and in the few instances where really thin layers have been used the results have been entirely satisfactory. In Königsberg filters were built by Frühling,[7] in which the sand was supported by five layers of gravel of increasing sizes, respectively 1.2, 1.2, 1.6, 2.0, 3.2, or, together, 9.2 inches thick, below which there were an average of five inches of coarse gravel. These were examined after eight years of operation and found to be in perfect order.
At the Lawrence Experiment Station filters have been repeatedly constructed with a total depth of supporting gravel layers not exceeding six inches, and among the scores of such filters there has not been a single failure, and so far as they have been dug up there has never been found to have been any movement whatever of the sand into the gravel. The Lawrence city filter, built with corresponding layers, has shown no signs of being inadequately supported. In arranging the Lawrence gravel layers care has always been taken that no material should rest on another material more than three or four times as coarse as itself, and that each layer should be complete at every point, so that by no possibility could two layers of greater difference in size come together. And it is believed that if this is carefully attended to, no trouble need be anticipated, however thin the single layers may be.
UNDERDRAINS.
The most common arrangement, in other than very small filters, is to have a main drain through the middle of the filter,[Pg 40] with lateral drains at regular intervals from it to the sides. The sides of the main drain are of brick, laid with open joints to admit water freely, and the top is usually covered with stone slabs. The lateral drains may be built in the same way, but tile drains are also used and are cheaper. Care must be taken with the latter that ample openings are left for the admission of water at very low velocities. It is considered desirable to have these drains go no higher than the top of the coarsest gravel; and this will often control the depth of gravel used. If they go higher, the top must be made tight to prevent the entrance of the fine gravels or sand. Sometimes they are sunk in part or wholly (especially the main drain) below the floor of the filter. With gravel placed in waves, that is, thicker over the drains than elsewhere, as mentioned above, the drains are covered more easily than with an entirely horizontal arrangement. When this is done, the floor of the filter is trenched to meet the varying thickness of gravel, so that the top of the latter is level, and the sand has a uniform thickness.
Many filters (Lambeth, Brunswick, etc.) are built with a double bottom of brick, the upper layer of which, with open joints, supports the gravel and sand, and is itself supported by numerous small arches or other arrangements of brick, which serve to carry the water to the outlet without other drains. This arrangement allows the use of a minimum quantity of gravel, but is undoubtedly more expensive than the usual form, with only the necessary quantity of gravel; and I am unable to find that it has any corresponding advantages.
The frictional resistance of underdrains requires to be carefully calculated; and in doing this quite different standards must be followed from those usually employed in determining the sizes of water-pipes, as a total frictional resistance of only a few hundredths of a foot, including the velocity head, may cause serious irregularities in the rate of filtration in different parts of the filter.
[Pg 41]
The sizes of the underdrains differ very widely in proportion to the sizes of the filters in European works, some of them being excessively large, while in other cases they are so small as to suggest a doubt as to their allowing uniform rates of filtration, especially just after cleaning.
I would suggest the following rules as reasonably sure to lead to satisfactory results without making an altogether too lavish provision: In the absence of a definite determination to run filters at some other rate, calculate the drains for the German standard rate of a daily column of 2.40 meters, equal to 2.57 million gallons per acre daily. This will insure satisfactory work at all lower rates, and no difficulty on account of the capacity of the underdrains need be then anticipated if the rate is somewhat exceeded. The area for a certain distance from the main drain depending upon the gravel may be calculated as draining directly into it, provided there are suitable openings, and the rest of the area is supposed to drain to the nearest lateral drain.
In case the laterals are round-tile drains I would suggest the following limits to the areas which they should be allowed to drain:
| Diameter of Drain. | To Drain an Area not Exceeding |
Corresponding Velocity of Water in Drain. |
|---|---|---|
| 4 inches | 290 square feet. | 0.30 foot. |
| 6 inches | 750 square feet. | 0.35 foot. |
| 8 inches | 1530 square feet. | 0.40 foot. |
| 10 inches | 2780 square feet. | 0.46 foot. |
| 12 inches | 4400 square feet. | 0.51 foot. |
And for larger drains, including the main drains, their cross-sections at any point should be at least 1⁄6000 of the area drained, giving a velocity of 0.55 foot per second with the rate of filtration mentioned above.
Fig. 4.—Plan of one of the Hamburg Filters, Showing Frictional Resistance of the Underdrains.
The total friction of the underdrains from the most remote points to the outlet will be friction in the gravel, plus friction in[Pg 42] the lateral drains, plus the friction in main drain, plus the velocity head.

Constructing the Underdrainage System of a Filter, Hamburg.
[To face page 42.]
I have calculated in this way the friction of one of the Hamburg filters for the rate of 1,600,000 gallons per acre daily at which it is used. The friction was calculated for each section of the drains separately, so that the friction from intermediate points was also known. Kutter’s formula was used throughout with n = 0.013. On the accompanying plan of the filter I have drawn the lines of equal frictional resistance from the junction of the main drain with the last laterals. My information was incomplete in regard to one or two points, so that the calculation may not be strictly accurate, but it is nearly so and will illustrate the principles involved.
The extreme friction of the underdrains is 11 millimeters = 0.036 foot.
The frictional resistance of the sand 39 inches thick, effective size 0.32 mm. and rate 1.60 million gallons per acre daily, when absolutely free from clogging, is by the formula, page 21, 15mm., or .0490 [Pg 43] foot, when the temperature is 50°. Practically there is some matter deposited upon the surface of the sand before filtration starts, and further, after the first scraping, there is some slight clogging in the sand below the layer removed by scraping. We can thus safely take the minimum frictional resistance of the sand including the surface layer at .07 foot. The average friction of the underdrains for all points is about .023 foot and the friction at starting will be .07 + .023 = .093 foot (including the friction in the last section to the effluent well where the head is measured, .100 foot, but the friction beyond the last lateral does not affect the uniformity of filtration). The actual head on the sand close to the outlet will be .093 and the rate of filtration .093⁄.070 · 1.60 = 2.12. The actual head at the most remote point will be .093 - .036 = .057, and the rate of filtration will there be .057⁄.070 · 160 = 1.30 million gallons per acre daily. The extreme rates of filtration are thus 2.12 and 1.30, instead of the average rate of 1.60. As can be seen from the diagram, only very small areas work at these extreme rates, the great bulk of the area working at rates much nearer the average. Actually the filter is started at a rate below 1.60, and the nearest portion never filters so rapidly as 2.12, for when the rate is increased to the standard, the sand has become so far clogged that the loss of head is more than the .07 foot assumed, and the differences in the rates are correspondingly reduced. Taking this into account, it would not seem that the irregularities in the rate of filtration are sufficient to affect seriously the action of the filter. They could evidently have been largely reduced by moderately increasing the sizes of the lower ends of the underdrains, where most of the friction occurs with the high velocities (up to .97 foot) which there result.
The underdrains of the Warsaw filters were designed by Lindley to have a maximum loss of head of only .0164 foot when[Pg 44] filtering at a rate of 2.57, which gives a variation of only 10 per cent in the rates with the minimum loss of head of .169 foot in the entire filter assumed by him. The underdrains of the Berlin filters, according to my calculations, have .020 to .030 foot friction, of which an unusually large proportion is in the gravel, owing to the excessive distances, in some cases over 80 feet, which the gravel is required to carry the water. In this case, using less or finer gravel would obviously have been fatal, but the friction as well as the expense of construction would be much reduced by using more drains and less gravel.
The underdrains might appropriately be made slightly smaller, with a deep layer of fine sand, than under opposite conditions, as in this case the increased friction in the drains would be no greater in proportion to the increased friction in the sand itself.
The underdrains of a majority of European filters have water-tight pipes connecting with them at intervals, and going up through the sand and above the water, where they are open to the air. These pipes were intended to ventilate the underdrains and allow the escape of air when the filter is filled with water introduced from below. It may be said, however, that in case the drains are surrounded by gravel and there is an opportunity for the air to pass from the top of the drain into the gravel, it will so escape without special provision being made for it, and go up through the sand with the much larger quantity of air in the upper part of the gravel which is incapable of being removed by pipes connecting with the drains.
These ventilator pipes where they are used are a source of much trouble, as unfiltered water is apt to run down through cracks in the sand beside them, and, under bad management, unfiltered water may even go down through the pipes themselves. I am unable to find that they are necessary, except with underdrains so constructed that there is no other chance for the escape of air from the tops of them, or that they serve any useful[Pg 45] purpose, while there are positive objections to their use. In some of the newer filters they have been omitted with satisfactory results.
DEPTH OF WATER ON THE FILTERS.
In the older works with but crude appliances for regulating the rate of filtration and admission of raw water, a considerable depth of water was necessary upon the filter to balance irregularities in the rates of filtration; the filter was made to be, to a certain extent, its own storage reservoir. When, however, appliances of the character to be described in Chapter IV are used for the regulation of the incoming water, and with a steady rate of filtration, this provision becomes quite superfluous.
With open filters a depth of water in excess of the thickness of any ice likely to be formed is required to prevent disturbance or freezing of the sand in winter. It is also frequently urged that with a deep water layer on the filter the water does not become so much heated in summer, but this point is not believed to be well taken, for in any given case the total amount of heat coming from the sun to a given area is constant, and the quantity of water heated in the whole day—that is, the amount filtered—is constant, and variations in the quantity exposed at one time will not affect the average resulting increase in temperature. If the same water remained upon the filter without change it would of course be true that a thin layer would be heated more than a deep one, but this is not the case.
It is also sometimes recommended that the depth of water should be sufficient to form a sediment layer before filtration starts, but this point would seem to be of doubtful value, especially where the filter is not allowed to stand a considerable time with the raw water upon it before starting filtration.
It is also customary to have a depth of water on the filter in excess of the maximum loss of head, so that there can never be a[Pg 46] suction in the sand just below the sediment layer. It may be said in regard to this, however, that a suction below is just as effective in making the water pass the sand as an equal head above. At the Lawrence Experiment Station filters have been repeatedly used with a water depth of only from 6 to 12 inches, with losses of head reaching 6 feet, without the slightest inconvenience. The suction only commences to exist as the increasing head becomes greater than the depth of water, and there is no way in which air from outside can get in to relieve it. In these experimental filters in winter, when the water is completely saturated with air, a small part of the air comes out of the water just as it passes the sediment layer and gets into reduced pressure, and this air prevents the satisfactory operation of the filters. But this is believed to be due more to the warming and consequent supersaturation of the water in the comparatively warm places in which the filters stand than to the lack of pressure, and as not the slightest trouble is experienced at other seasons of the year, it may be questioned whether there would be any disadvantage at any time in a corresponding arrangement on a large scale where warming could not occur.
The depths of water actually used in European filters with the full depth of sand are usually from 36 to 52 inches. In only a very few unimportant cases is less than the above used, and only a few of the older works use a greater depth, which is not followed in any of the modern plants. As the sand becomes reduced in thickness by scraping, the depth of water is correspondingly increased above the figures given until the sand is replaced. The depth of water on the German covered filters is quite as great as upon corresponding open filters. Thus the Berlin covered filters have 51, while the new open filters at Hamburg have only 43 inches.
[Pg 47]
CHAPTER IV.
RATE OF FILTRATION AND LOSS OF HEAD.
The rate of filtration recommended and used has been gradually reduced during the past thirty years. In 1866 Kirkwood found that 12 vertical feet per day, or 3.90 million gallons per acre daily, was recommended by the best engineers, and was commonly followed as an average rate. In 1868 the London filters averaged a yield of 2.18 million gallons[8] per acre daily, including areas temporarily out of use, while in 1885 the quantity had been reduced to 1.61. Since that time the rate has apparently been slightly increased.
The Berlin filters at Stralau constructed in 1874 were built to filter at a rate of 3.21 million gallons per acre daily. The first filters at Tegel were built for a corresponding rate, but have been used only at a rate of 2.57, while the more recent filters were calculated for this rate. The new Hamburg filters, 1892-3, were only intended to filter at a rate of 1.60 million gallons per acre daily. These in each case (except the London figures) are the standard rates for the filter-beds actually in service.
In practice the area of filters is larger than is calculated from these figures, as filters must be built to meet maximum instead of average daily consumptions, and a portion of the filtering area usually estimated at from 5 to 15 per cent, but in extreme cases reaching 50 per cent, is usually being cleaned, and so is for the time out of service. In some works also the rate of filtration on starting a filter is kept lower than the standard rate for a day or two, or the first portion of the effluent, supposed to be of inferior quality, is
[Pg 48]
wasted, the amount so lost reaching in an extreme case 9 to 14 per cent of the total quantity of water filtered.[9] In many of the older works also, there is not storage capacity enough for filtered water to balance the hourly fluctuations in consumption, and the filters must be large enough to meet the maximum hourly as well as the maximum daily requirements. For these reasons the actual quantity of water filtered in a year is only from 50 to 75 per cent of what would be the case if the entire area of the filters worked constantly at the full rate. A statement of the actual yields of a number of filter plants is given in Appendix IV. The figures for the average annual yields can be taken as quite reliable. The figures given for rate, in many cases, have little value, owing to the different ways in which they are calculated at different places. In addition most of the old works have no adequate means of determining what the rate at any particular time and for a single filter really is, and statements of average rates have only limited value. The filters at Hamburg are not allowed to filter faster than 1.60 or those at Berlin faster than 2.57 million gallons per acre daily, and adequate means are provided to secure this condition. Other German works aim to keep within the latter limit. Beyond this, unless detailed information in regard to methods is presented, statements of rate must be taken with some allowance.
EFFECT OF RATE UPON COST OF FILTRATION.
The size of the filters required, and consequently the first cost, depends upon the rate of filtration, but with increasing rates the cost is not reduced in the same proportion as the increase in rate, since the allowance for area out of use is sensibly the same for high and low rates, and in addition the operating expenses depend upon the quantity filtered and not upon the filtering area. Thus, to supply 10 million gallons at a maximum rate of 2 million gallons per acre daily we should require 10 ÷ 2 = 5 acres + 1 acre reserve for cleaning = 6 acres, while with a rate twice[Pg 49] as great, and with the same reserve (since the same amount of cleaning must be done, as will be shown below), we should require 10 ÷ 4 + 1 = 3.5 acres, or 58 per cent of the area required for the lower rate. Thus beyond a certain point increasing the rate does not effect a corresponding reduction in the first cost.
The operating cost for the same quantity of water filtered does not appear to be appreciably affected by the rate. It is obvious that at high rates filters will became clogged more rapidly, and will so require to be scraped oftener than at low rates, and it might naturally be supposed that the clogging would increase more rapidly than the rates, but this does not seem to be the case. At the Lawrence Experiment Station, under strictly parallel conditions and with identically the same water, filters running at various rates became clogged with a rapidity directly proportional to the rates, so that the quantities of water filtered between scrapings under any given conditions are the same whether the rate is high or low.
The statistics bearing upon this point are interesting, if not entirely conclusive. There were eleven places in Germany filtering river waters, from which statistics were available for the year 1891-92. Of these there were four places with high rates, Lübeck, Stettin, Stuttgart, and Magdeburg, yielding 3.70 million gallons per acre daily, which filtered on an average 59 million gallons per acre between scrapings. Three other places, Breslau, Altona, and Frankfurt, yielding 1.85, passed on an average 55 million gallons per acre between scrapings, and four other places, Bremen, Königsberg, Brunswick and Posen, yielding 1.34 million gallons per acre daily, passed only 40 million gallons per acre between scrapings. The works filtering at the highest rates thus filtered more water in proportion to the sand clogged than did those filtering more slowly, but I cannot think that this was the result of the rate. It is more likely that some of the places have clearer waters than others, and that this both allows the higher rate and causes less clogging than the more turbid waters.
[Pg 50]
EFFECT OF RATE UPON EFFICIENCY OF FILTRATION.
The effect of the rate of filtration upon the quality of the effluent has been repeatedly investigated. The efficiency almost uniformly decreases rapidly with increasing rate. Fränkel and Piefke[10] first found that with the high rates the number of bacteria passing some experimental filters was greatly increased. Piefke[11] afterward repeated these experiments, eliminating some of the features of the first series to which objection was made, and confirmed the first results. The results were so marked that Piefke was led to recommend the extremely low limit of 1.28 million gallons per acre daily as the safe maximum rate of filtration, but he has since repeatedly used 2.57 million gallons.
Kümmel,[12] on the other hand, in a somewhat limited series of experiments, was unable to find any marked connection between the rate and the efficiency, a rate of 2.57 giving slightly better results than rates of either 1.28 or 5.14.
The admirably executed experiments made at Zürich in 1886-8 upon this point, which gave throughout negative results, have but little value in this connection, owing to the extremely low number of bacteria in the original water.
At Lawrence in 1892 the following percentages of bacteria (B. prodigiosus) passed at the respective rates:
| No. of Filter. |
Depth. | Effective Size of Sand. |
Rate. Millions gallons per acre daily. | ||||
|---|---|---|---|---|---|---|---|
| 0.5 | 1.0 | 1.5 | 2.0 | 3.0 | |||
| 33A | 60 | 0.14 | 0.002 | ..... | ..... | 0.040 | ..... |
| 34A | 60 | 0.09 | 0.001 | 0.005 | ..... | 0.020 | ..... |
| 36A | 60 | 0.20 | ..... | ..... | 0.050 | ..... | 0.050 |
| 37 | 60 | 0.20 | ..... | ..... | 0.010 | 0.130 | ..... |
| 38 | 24 | 0.20 | 0.018 | ..... | 0.140 | 0.110 | 0.310 |
| 39 | 12 | 0.20 | 0.014 | 0.070 | ..... | 0.080 | 0.520 |
| 40 | 12 | 0.20 | ..... | 0.070 | ..... | 0.090 | ..... |
| 42 | 12 | 0.20 | 0.016 | ..... | ..... | 0.150 | 0.550 |
| Average | 0.010 | 0.048 | 0.067 | 0.088 | 0.356 | ||
[Pg 51]
These results show a very marked decrease in efficiency with increasing rates, the number of bacteria passing increasing in general as rapidly as the square of the rate. The 1893 results also showed decreased efficiency with high rates, but the range in the rates under comparable conditions was less than in 1892, and the bacterial differences were less sharply marked.
While the average results at Lawrence, as well as most of the European experiments, show greatly decreased efficiency with high rates, there are many single cases, particularly with deep layers of not too coarse sand, where, as in Kümmel’s experiments, there seems to be little connection between the rate and efficiency. An explanation of these apparently abnormal results will be given in Chapter VI.
It is commonly stated[13] that every water has its own special rate of filtration, which must be determined by local experiments, and that this rate may vary widely in different cases. Thus it is possible that the rate of 1.60 adopted at Hamburg for the turbid Elbe water, the rate of 2.57 used at Berlin, and about the same at London for much clearer river-waters, and the rate of 7.50 used at Zürich for the almost perfectly clear lake-water are in each case the most suitable for the respective waters. In other cases however, where rates much above 2.57 are used for river-waters, as at Lübeck and Stettin, there is a decided opinion that these rates are excessive, and in these instances steps are now being taken to so increase the filtering areas as to bring the rates within the limit of 2.57 million gallons per acre daily.
From the trend of European practice it would seem that for American river-waters the rate of filtration should not exceed 2.57 in place of the 3.90 million gallons per acre daily recommended by Kirkwood, or even that a somewhat lower rate might be desirable in some cases. Of course, in addition to the area [Pg 52] necessary to give this rate, a reserve for fluctuating rates and for cleaning should be provided, reducing the average yield to 2.00, 1.50, or even less. In the case of water from clear lakes, ponds, or storage reservoirs, especially when they are not subject to excessive sewage pollution or to strong algæ growths, it would seem that rates somewhat and perhaps in some cases very much higher (as at Zürich) could be satisfactorily used.
THE LOSS OF HEAD.
The loss of head is the difference between the heads of the waters above and below the sand layer, and represents the frictional resistance of that layer. When a filter is quite free from clogging this frictional resistance is small, but gradually increases with the deposit of a sediment layer from the water filtered until it becomes so great that the clogging must be removed by scraping before the process can be continued. After scraping the loss of head is reduced to, or nearly to, its original amount. With any given amount of clogging the loss of head is directly proportional to the rate of filtration; that is, if a filter partially clogged, filtering at a rate of 1.0, has a frictional resistance of 0.5 ft., the resistance will be doubled by increasing the rate to 2.00 million gallons per acre daily, provided no disturbance of the sediment layer is allowed. This law for the frictional resistance of water in sand alone also applies to the sediment layer, as I have found by repeated tests, although in so violent a change as that mentioned above, the utmost care is required to make the change gradually and prevent compression or breaking of the sediment layer. From this relation between the rate of filtration and the loss of head it is seen that the regulation of either involves the regulation of the other, and it is a matter of indifference which is directly and which indirectly controlled.
REGULATION OF THE RATE AND LOSS OF HEAD IN THE OLDER FILTERS.
In the older works, and in fact in all but a few of the newest[Pg 53] works, the underdrains of the filters connect directly through a pipe with a single gate with the pure-water reservoir or pump-well, which is so built that the water in it may rise nearly or quite as high as that standing upon the filter.
Fig. 5.—Simplest Form of Regulation: Stralau Filters at Berlin.
A typical arrangement of this sort was used at the Stralau works at Berlin (now discontinued), Fig. 5. With this arrangement the rate of filtration is dependent upon the height of water in the reservoir or pump-well, and so upon the varying consumption. When the water in the receptacle falls with increasing consumption the head is increased, and with it the rate of filtration, while, on the other hand, with decreasing draft and rising water in the reservoir, the rate of filtration decreases and would eventually be stopped if no water were used. This very simple arrangement thus automatically, within limits, adjusts the rate of filtration to the consumption, and at the same time always gives the highest possible level of water in the pump-well, thus also economizing the coal required for pumping.
In plants of this type the loss of head may be measured by floats on little reservoirs built for that purpose, connected with the underdrains; but more often there is no means of determining it, although the maximum loss of head at any time is the difference between the levels of the water on the filter and in the reservoir, or the outlet of the drain-pipe, in case the latter is above[Pg 54] the water-line in the reservoir. The rate of filtration can only be measured with this arrangement by shutting off the incoming water for a definite interval, and observing the distance that the water on the filter sinks. The incoming water is regulated simply by a gate, which a workman opens or closes from time to time to hold the required height of water on the filter.
The only possible regulation of the rate and loss of head is effected by a partial closing of the gate on the outlet-pipe, by which the freshly-cleaned filters with nearly-closed gates are kept from filtering more rapidly than the clogged filters, the gates of which are opened wide. Often, however, this is not done, and then the fresh filters filter many times as rapidly as those which are partially clogged.
A majority of the filters now in use are built more or less upon this plan, including most of those in London and also the Altona works, which had such a favorable record with cholera in 1892.
The invention and application of methods of bacterial examination in the last years have led to different ideas of filtration from those which influenced the construction of the earlier plants. As a result it is now regarded as essential by most German engineers[14] that each filter shall be provided with devices for measuring accurately and at any time both the rate of filtration and the loss of head, and for controlling them, and also for making the rate independent of consumption by reservoirs for filtered water large enough to balance hourly variations (capacity 1⁄4 to 1⁄3 maximum daily quantity) and low enough so that they can never limit the rate of filtration by causing back-water on the filters. These points are now insisted upon by the German Imperial Board of Health,[15] and all new filters are built in accordance with them, while most of the old works are being built over to conform to the requirements.
[Pg 55]
APPARATUS FOR REGULATING THE RATE AND LOSS of HEAD.
Many appliances have been invented for the regulation of the rate and loss of head. In the apparatus designed by Gill and used at both Tegel and Müggel at Berlin the regulation is effected by partially closing a gate through which the effluent passes into a chamber in which the water-level is practically constant (Fig. 6). The rate is measured by the height of water on the weir which serves as the outlet for this second chamber into a third connecting with the main reservoir, while the loss of head is shown by the difference in height of floats upon water in the first chamber, representing the pressure in the underdrains, and upon water in connection with the raw water on the filter. From the respective heights of the three floats the attendant can at any time see the rate of filtration and the loss of head, and when a change is required it is effected by moving the gate.
Fig. 6.—Regulation Apparatus at Berlin (Tegel).
In the apparatus designed in 1866 by Kirkwood for St. Louis and never built (Fig. 7) the loss of head was directly, and the rate indirectly, regulated by a movable weir, which was to have been lowered from time to time by the attendant to secure the required results. This plan is especially remarkable as it meets[Pg 56] the modern requirements of a regular rate independent of rate of consumption and of the water-level in the reservoir, and also allows continual measurements of both rate (height of water on the weir) and head (difference in water-levels on filter and in effluent chamber) to be made, and control of the same by the position of the weir. Mr. Kirkwood found no filters in Europe with such appliances, and it was many years after his report was published before similar devices were used, but they are now regarded as essential.
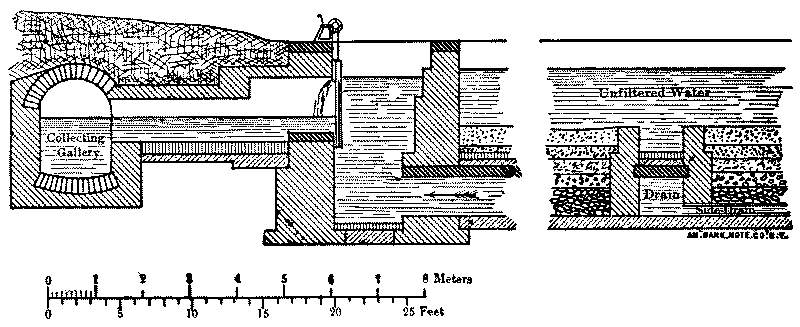
Fig. 7.—Regulation Apparatus and Section of Filter recommended for St. Louis by Kirkwood in 1866.
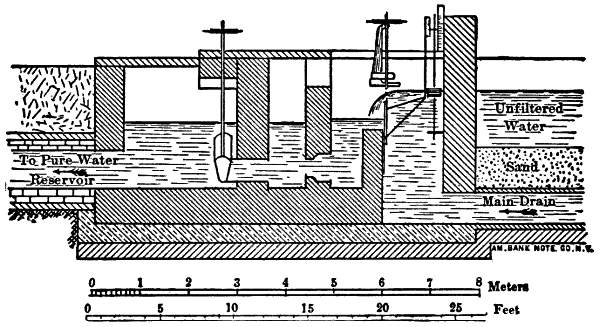
Fig. 8.—Regulation Apparatus used at Hamburg.
The regulators for new filters at Hamburg (Fig. 8) are built upon the principle of Kirkwood’s device, but provision is made for a second measurement of the water if desired by the loss of[Pg 57] head in passing a submerged orifice. Both the rate and loss of head are indicated by a float on the first chamber connecting directly with the underdrain, which at the same time indicates the head on a fixed scale, the zero of which corresponds to the height of the water above the filter, and the rate upon a scale moving with the weir, the zero of which corresponds with the edge of the weir. The water on the filter is held at a perfectly constant level.
The regulators in use at Worms and those recently introduced at Magdeburg act upon the same principle, but the levels of the water on the filters are allowed to fluctuate, and the weirs and in fact, the whole regulating appliances are mounted on big floats in surrounding chambers of water connecting with the unfiltered water on the filters. I am unable to find any advantages in these appliances, and they are much more complicated than the forms shown by the cuts.
APPARATUS FOR REGULATING THE RATE DIRECTLY.
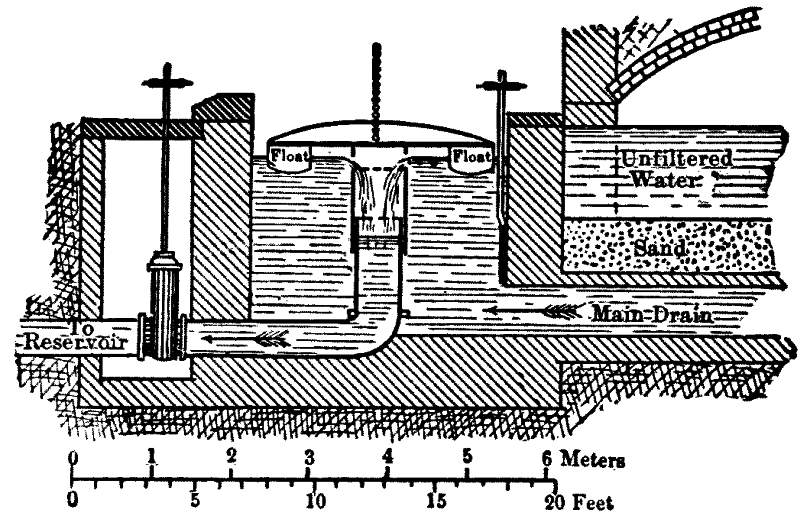
Fig. 9.—Lindley’s Regulation Apparatus at Warsaw, Russia.
The above-mentioned regulators control directly the loss of head, and only indirectly the rate of filtration. The regulators at Warsaw were designed by Lindley to regulate the rate directly and make it independent of the loss of head. The quantity of water flowing away is regulated by a float upon the water[Pg 58] in the effluent chamber, which holds the top of the telescope outlet-pipe a constant distance below the surface and so secures a constant rate. As the friction of the filter increases the float sinks with the water until it reaches bottom, when the filter must be scraped. A counter-weight reduces the weight on the float, and at the same time allows a change in the rate when desired. This apparatus is automatic. All of the other forms described require to be occasionally adjusted by the attendant, but the attention they require is very slight, and watchmen are always on duty at large plants, who can easily watch the regulators. The Warsaw apparatus is reported to work very satisfactorily, no trouble being experienced either by leaking or sticking of the telescope-joint, which is obviously the weakest point of the device, but fortunately a perfectly tight joint is not essential to the success of the apparatus. Regulators acting upon the same principle have recently been installed at Zürich, where they are operating successfully.
Burton[16] has described an ingenious device designed by him for the filters at Tokyo, Japan. It consists of a double acting valve of gun metal (similar to that shown by Fig. 11), through which the effluent must pass. This valve is opened and closed by a rod connecting with a piston in a cylinder, the opposite sides of which connect with the effluent pipe above and below a point where the latter is partially closed, so that the valve is opened and closed according as the loss of head in passing this obstruction is below or above the amount corresponding to the desired rate of filtration.
The use of the Venturi meter in connection with the regulation of filters would make an interesting study, and has, I believe, never been considered.

Regulator-house, showing Rate of Filtration and Loss of Head on the Outside, Bremen.

Inlet for Admission of Raw Water to a Filter, East London.
[To face page 58.]
[Pg 59]
APPARATUS FOR REGULATING THE HEIGHT OF WATER UPON FILTERS.
It will be seen by reference to the diagrams of the Berlin and Hamburg effluent regulators (Figs. 6 and 8) that their perfect operation is dependent upon the maintenance of a constant water-level upon the filters. The old-fashioned adjustment of the inlet-gate by the attendant is hardly accurate enough.
The first apparatus for accurately and automatically regulating the level of the water upon the filters was constructed at Leeuwarden, Holland, by the engineer, Mr. Halbertsma, who has since used a similar device at other places, and improved forms of which are now used at Berlin and at Hamburg.
At Berlin (Müggel) the water-level is regulated by a float upon the water in the filter which opens or shuts a balanced double valve on the inlet-pipe directly beneath, as shown in Fig. 10. It is not at all necessary that this valve should shut water-tight; it is only necessary that it should prevent the continuous inflow from becoming so great as to raise the water-level, and for this reason loose, easily-working joints are employed. The apparatus is placed in a little pit next to the side of the filter, and the overflowing water is prevented from washing the sand by paving the sand around it for a few feet.
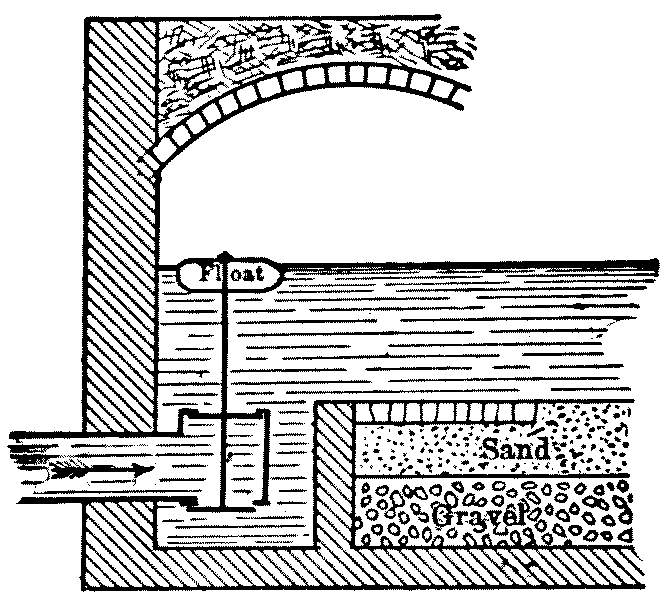
Fig. 10.—Regulation of Inflow used at Müggel, Berlin.
At Hamburg the same result is obtained by putting the valve[Pg 60] in a special chamber outside of the filter and connected with the float by a walking-beam (Fig. 11).
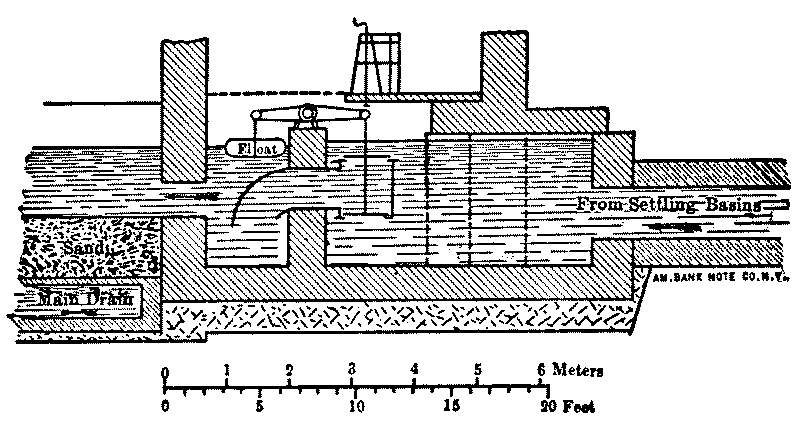
Fig. 11.—Regulation of Inflow used at Hamburg.
The various regulators require to be protected from cold and ice by special houses, except in the case of covered filters, where they can usually be arranged with advantage in the filter itself. In regard to the choice of the form of regulator for both the inlets and outlets of filters, so far as I have been able to ascertain, each of the modern forms described as in use performs its functions satisfactorily, and in special cases any of them could properly be selected which would in the local conditions be the simplest in construction and operation.
LIMIT TO THE LOSS OF HEAD.
The extent to which the loss of head is allowed to go before filters are cleaned differs widely in the different works, some of the newer works limiting it sharply because it is believed that low bacterial efficiency results when the pressure is too great, although the frequency of cleaning and consequently the cost of operation are thereby increased.
At Darlington, England, I believe as a result of the German theories, the loss of head is limited to about 18 inches by a masonry weir built within the last few years. At Berlin, both at[Pg 61] Tegel and Müggel, the limit is 24 inches, while at the new Hamburg works 28 inches are allowed. At Stralau in 1893 an effort was made to not exceed a limit of 40 inches, but previously heads up to 60 inches were used, which corresponds with the 56 inches used at Altona; and, in the other old works, while exact information is not easily obtained because of imperfect records, I am convinced that heads of 60 or even 80 inches are not uncommon. At the Lawrence Experiment Station heads of 70 inches have generally been used, although some filters have been limited to 36 and 24 inches.
In 1866 Kirkwood became convinced that the loss of head should not go much above 30 inches, first, because high heads would, by bringing extra weight upon the sand, make it too compact, and, second, because when the pressure became too great the sediment layer on the surface of the sand, in which most of the loss of head occurs, would no longer be able to support the weight and, becoming broken, would allow the water to pour through the comparatively large resulting openings at greatly increased rates and with reduced efficiency.
In regard to the first point, a straight, even pressure many times that of the water on the filter is incapable of compressing the sand. It is much more the effect of the boots of the workmen when scraping that makes the sand compact. I have found sand in natural banks at Lawrence 70 or 80 feet below the surface, where it had been subjected to corresponding pressure for thousands of years, to be quite as porous as when packed in water in experimental filters in the usual way.
The second reason mentioned, or, as I may call it, the breaking-through theory, is very generally if not universally accepted by German engineers, and this is the reason for the low limit commonly adopted by them.
A careful study of the results at Lawrence fails to show the slightest deterioration of the effluents up to the limit used, 72 inches. Thus in 1892, taking only the results of the continuous[Pg 62] filters of full height (Nos. 33A, 34A, 36A, and 37), we find that for the three days before scraping, when the head was nearly 72 inches, the average number of bacteria in the effluents was 31 per cc., while for the three days after scraping, with very low heads, the number was 47. The corresponding numbers of B. prodigiosus[17] were 1.1 and 2.7. This shows better work with the highest heads, but is open to the objection that the period just after scraping, owing to the disturbance of the surface, is commonly supposed to be a period of low efficiency.
To avoid this criticism in calculating the corresponding results for 1893, the numbers of the bacteria for the intermediate days which could not have been influenced either by scraping or by excessive head are put side by side with the others. Taking these results as before for continuous filters 72 inches high, and excluding those with extremely fine sands and a filter which was only in operation a short time toward the end of the year, we obtain the following results:
| Water Bacteria per cc. |
B. Prodigiosus per cc. |
|
|---|---|---|
| Average 1st day after scraping, low heads | 79 | 6.1 |
| Average 2d day after scraping, low heads | 44 | 4.1 |
| Average 3d day after scraping, low heads | 45 | 3.6 |
| Intermediate days, medium heads | 59 | 4.5 |
| Second from last day, heads of nearly 72 inches | 66 | 2.7 |
| Next to the last day, heads of nearly 72 inches | 56 | 3.2 |
| Last day, heads of nearly 72 inches | 83 | 2.5 |
These figures show a very slight increase of the water bacteria in the effluent as the head approaches the limit, but no such increase as might be expected from a breaking through of the sediment layer, and the B. prodigiosus which is believed to better indicate the removal of the bacteria of the original water,[Pg 63] actually shows a decrease, the last day being the best day of the whole period.
The Lawrence results, then, uniformly and clearly point to a conclusion directly opposite to the commonly accepted view, and I have thus been led to examine somewhat closely the grounds upon which the breaking-through theory rests.
The two works which have perhaps contributed most to the theories of filtration are the Stralau and Altona works. After examining the available records of these works, I am quite convinced that at these places there has been, at times at least, decreased efficiency with high heads. For the Stralau works this is well shown by Piefke’s plates in the Zeitschrift für Hygiene, 1894, after page 188. In both of these works, however, the apparatus (or lack of apparatus) for regulating the rate is that shown by Fig. 5, page 49, and the rate of filtration is thus dependent upon the rate of consumption and the height of water in the reservoir. At the Stralau works, at the time covered by the above-mentioned diagrams, the daily quantity of water filtered was 27 times the capacity of the reservoir, and the rate of filtration must consequently have adapted itself to the hourly consumptions. The data which formed the basis of Kirkwood’s conclusions are not given in detail, but it is quite safe to assume that they were obtained from filters regulated as those at Altona and Stralau are regulated, and what is said in regard to the latter will apply equally to his results.
Piefke[18] shows that among the separate filters at Stralau, all connected with the same pure-water reservoir, those connected through the shorter pipes gave poorer effluents than the more remote filters, and he attributes the difference to the frictional resistance of the connecting pipes, which helped to prevent excessive rates in the filters farthest away when the water in the reservoir became low, and thus the fluctuations in the rates in these filters were less than in those close to the reservoir. He [Pg 64] does not, however, notice, in speaking of the filters in which the decreased efficiencies with high heads were specially marked, that they follow in nearly the same order, and that of the four open filters mentioned three were near the reservoir and only one was separated by a comparatively long pipe, indicating that the deterioration with high heads was only noticeable, or at least was much more conspicuous, in those filters where the rates fluctuated most violently.
It requires no elaborate calculation to show that of two filters connected with the same pure-water reservoir, as shown by Fig. 5, with only simple gates on the connecting pipes, one of them clean and throttled by a nearly closed gate, so that the normal pressure behind the gate is above the highest level of water in the reservoir, and the other clogged so that the normal pressure of the water in the drain is considerably below the highest level of the water in the reservoir, the latter will suffer much the more severe shocks with fluctuating water-levels; and the fact being admitted that fluctuating levels are unfavorable, we must go farther and conclude that the detrimental action will increase with increasing loss of head. I am inclined to think that this theory is adequate to explain the Stralau and Altona results without resource to the breaking-through theory.
While the above does not at all prove the breaking-through theory to be false, it explains the results upon which it rests in another way, and can hardly fail to throw so much doubt upon it as to make us refuse to allow its application to those works where a regular rate of filtration is maintained regardless of variations in the consumption, until proof is furnished that it is applicable to them.
I have been totally unable to find satisfactory European results in regard to this point. The English works can furnish nothing, both on account of the lack of regulating appliances and because the monthly bacterial examinations are inadequate for a discussion of hourly or daily changes. The results from[Pg 65] the older Continental works are also excluded for one or the other, or more often for both, of the above reasons. The Hamburg, Tegel, and Müggel results, so far as they go, show no deterioration with increased heads, but the heads are limited to 24 or 28 inches by the construction of the filters, and the results thus entirely fail to show what would be obtained with heads more than twice as high.
I am thus forced to conclude that there is no adequate evidence of inferior efficiency with high heads in filters where the rates are independent of the water-level in the pure-water reservoir, the only results directly to the point—the Lawrence results mentioned above—indicating that the full efficiency is maintained with heads reaching at least 72 inches.
The principal reason for desiring to allow a considerable loss of head is an economical one; the period will then be lengthened, while the frequency of scraping and the volume of sand to be washed and replaced will be correspondingly reduced. There may be other advantages in long periods, such as less trouble with scraping and better work in cold winter weather, but the cost is the most important consideration.
It is the prevalent idea among the German engineers that the loss of head after reaching 24 to 30 inches would increase very rapidly, so that the quantity of water filtered, in case a much higher head was allowed, would not be materially increased. No careful investigations, however, have been made, and indeed they are hardly possible with existing arrangements, as in the older filters the loss of head fluctuates with varying rates of filtration in such a way that only results of very doubtful value can be obtained, and in the newer works the loss of head is too closely limited, and the curves which can be drawn by extrapolation are evidently no safe indications of what would actually happen if the process was carried farther.
On the other hand, I was told by the attendant at Darlington, England, that since the building of the weir a few years ago,[Pg 66] which now limits the loss of head to about 18 inches instead of the 5 feet or more formerly used, the quantity of sand to be removed has been three times as great as formerly. No records are kept, and this can only be given as the general impression of the man who superintends the work.
At Lawrence the average quantities of water filtered between scrapings with sand of an effective size of 0.20 mm. have been as follows:
| Maximum Loss of Head. |
Million Gallons per Acre filtered between Scrapings. |
||
|---|---|---|---|
| 1892. | 1893. | Average. | |
| 70 inches | 58 | 88 | 73 |
| 34 inches | 32 | 22 | 27 |
| 22 inches | 17 | 16 | 16 |
| With sand of an effective size of 0.29 mm. the results were: | |||
| 1893. | |||
| 70 inches | 70 | ||
| 22 inches | 29 | ||
These results indicate a great increase in the quantity of water filtered between scrapings with increasing heads, the figures being nearly proportional to the maximum heads used in the respective cases. It is, of course, quite possible that the results would differ in different places with the character of the raw water and of the filtering material.
The depth of sand to be removed by scraping at one time is, within limits, practically independent of the quantity of dirt which it has accumulated, and any lengthening of the period means a corresponding reduction in the quantity of sand to be removed, washed and replaced and consequently an important reduction in the operating cost, as well as a reduction in the area of filters out of use while being cleaned, and so, in the capital cost.
Among the minor objections to an increased loss of head are the greater head against which the water must be pumped, and[Pg 67] the possible increased difficulty of filling filters with filtered water from below after scraping, but these would hardly have much weight against the economy indicated by the Lawrence experiments for the higher heads.
High heads will also drive an increased quantity of water through any cracks or passages in the filter. Such leaks have at last been found to be the cause of the inferior work of the covered filters at Stralau, the water going down unfiltered in certain corners, especially at high heads; but with careful construction there should be no cracks, and with the aid of bacteriology to find the possible leaks this ought not to be a valid objection.
In conclusion: the trend of opinion is strongly in favor of limiting the loss of head to about 24 to 30 inches as was suggested by Kirkwood, but I am forced to conclude that there is reason to believe that equally good results can be obtained with lower operating expenses by allowing higher heads to be used, at least in the case of filters with modern regulating appliances, and, I would suggest that filters should be built so as not to exclude the use of moderately high heads, and that the limit to be permanently used should be determined by actual tests of efficiency and length of period with various losses of head after starting the works.
[Pg 68]
CHAPTER V.
CLEANING FILTERS.
When a filter has become so far clogged that it will no longer pass a satisfactory quantity of water with the allowable head it must be cleaned by scraping off and removing the upper layer of dirty sand.
To do this without unnecessary loss of time the unfiltered water standing upon the filter is removed by a drain above the sand provided for that purpose. The water in the sand must then be lowered below the surface of the sand by drawing water from the underdrains until the sand is firm enough to bear the weight of the workmen. By the time that this is accomplished the last water on the surface should have soaked away, and the filter is ready to be scraped. This is done by workmen with wide, sharp shovels, and the sand removed is taken to the sand-washing apparatus to be washed and used again. Special pains are given to securing rapid and cheap transportation of the sand. In some cases it is wheeled out of the filter on an inclined plane to the washer. In other cases a movable crane is provided which lifts the sand in special receptacles and allows it to fall into cars on a tram-line on which the crane also moves. The cars as filled are run to the washer and also serve to bring back the washed sand. When the dirty sand has been removed, the surface of the sand is carefully smoothed and raked. This is especially necessary to remove the effects of the workmen’s boots.
It is customary in the most carefully managed works to fill the sand with filtered water from below, introduced through the underdrains. In case the ordinary level of the water in the[Pg 69] pure-water canal is higher than the surface of the sand in the filters, this is accomplished by simply opening a gate provided for the purpose, which allows the water to pass around the regulating apparatus. Otherwise filters can be filled from a special pipe taking its water from any filter which at that time can deliver its effluent high enough for that purpose. The quantity of water required for filling the sand from below is ordinarily but a fraction of one per cent of the quantity filtered.
Formerly, instead of filling from below, after cleaning, the raw water was brought directly onto the surface of the filter. This was said to only imperfectly fill the sand-pores, which still contained much air. If, however, the water is not brought on too rapidly it will sink into the sand near the point where it is applied, pass laterally through the sand or underlying gravel to other parts of the filter, and then rise, so that even in this case all but a little of the filter will be really filled from below. This is, however, open to the objection that however slowly the water is introduced, the sand which absorbs it around the inlet filters it at a very high rate and presumably imperfectly, so that the water in the underdrains at the start will be poor quality and the sand around the inlet will be unduly clogged. The practice of filling from below is therefore well founded.
As soon as the surface of the sand is covered with the water from below, raw water is introduced from above, filling the filter to the standard height, care being taken at first that no currents are produced which might wash the surface of the sand. It has been recommended by Piefke and others that this water should be allowed to stand for a time up to twenty-four hours before starting the filtration, to allow the formation of a sediment layer, and in some places, especially at Berlin and the works of some of the London companies, this is done; but varying importance is attached to the procedure, and it is invariably omitted, so far as I can learn, when the demand for water is heavy.
The depth of sand removed by scraping must at least equal[Pg 70] the depth of the discolored layer, but there is no sharp dividing line, the impurities gradually decreasing from the surface downward. Fig. 12 shows the relative number of bacteria found in the sand at various depths in one of the Lawrence experimental filters, and is a representative result, although the actual numbers vary at different times. In general it may be said that the bulk of the sediment is retained in the upper quarter inch, but it is desirable to remove also the less dirty sand below and, in fact, it is apparently impossible with the method of scraping in use to remove so thin a layer as one fourth inch. Practically the depth to which sand is removed is stated to be from 0.40 to 1.20 inch. Exact statistics are not easily obtained, but I think that 2 centimeters or 0.79 inch may be safely taken as about the average depth usually removed in European filters, and it is this depth which is indicated on Fig. 12.
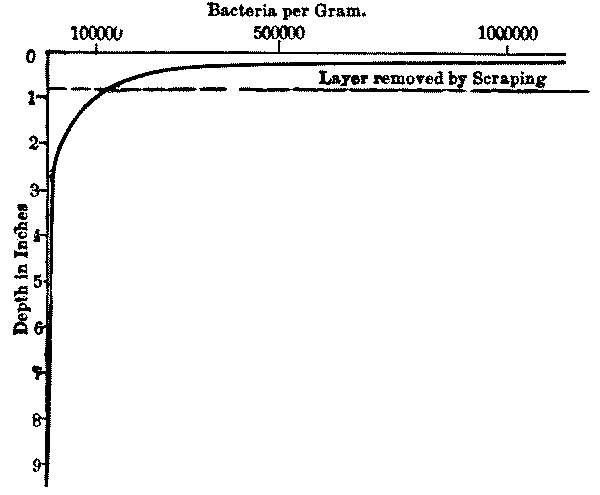
Fig. 12.—Diagram Showing Accumulation of Bacteria near the Surface of the Sand.
At the Lawrence Experiment Station, the depth removed is often much less than this, and depends upon the size of grain of the sand employed, the coarser sands requiring to be more deeply scraped than the finer ones. The method of scraping, however, which allows the removal of very thin sand layers, is[Pg 71] only possible because of the small size of the filters, and as it is incapable of application on a large scale, the depths thus removed are only interesting as showing the results which might be obtained in practice with a more perfect method of scraping.
The replacing of the washed sand is usually delayed until the filter has been scraped quite a number of times—commonly for a year. The last scraping before refilling is much deeper than usual, because the sand below the depth of the ordinary scraping is somewhat dirty, and might cause trouble if left below the clean sand.
In England it is the usual if not the universal practice to replace the washed sand at the bottom between the old sand and the gravel. This is done by digging up the entire filter in sections about six feet wide. The old sand in the first section is removed clear down to the gravel, and the depth of washed sand which is to be replaced is put in its place. The old sand from the next six-foot section is then shovelled upon the first section of clean sand, and its place is in turn filled with fresh sand. With this practice the workmen’s boots are likely to disturb the gravel each year, necessitating a thicker layer of the upper and finest grade than would otherwise be required.
In Germany this is also sometimes done, but more frequently the upper layer of slightly clogged sand below the regular scraping is removed as far as the slightest discoloration can be seen, perhaps 6 inches deep. The sand below is loosened for another 6 inches and allowed to stand dry, if possible, for some days; afterwards the washed sand is brought on and placed above. The washed sand is never replaced without some such treatment, because the slightly clogged sand below the layer removed would act as if finer than the freshly washed sand,[19] and there would be a tendency to sub-surface clogging.
[Pg 72]
FREQUENCY OF SCRAPING.
The frequency of scraping depends upon the character of the raw water, the thoroughness of the preliminary sedimentation, the grain-size of the filter sand, the rate of filtration, and the maximum loss of head allowed. With suitable conditions the period between scrapings should never be less than one week, and will but rarely exceed two months. Under exceptional conditions, however, periods have been recorded as low as one day and as high as one hundred and ten days. Periods of less than a week’s duration are almost conclusive evidence that something is radically wrong, and the periods of one day mentioned were actually accompanied by very inadequate filtration. In 1892 the average periods at the German works varied from 9.5 days at Stettin (with an excessive rate) to 40 days at Brunswick, the average of all being 25 days.[20]
The quantity of water per acre filtered between scrapings forms the most convenient basis for calculation. The effect of rate (page 49), loss of head (page 65), and size of sand grain (page 32) have already been discussed, and it will suffice to say here that the total quantity filtered between scrapings is apparently independent of the rate of filtration, but varies with the maximum loss of head and with the grain-size of the sand, and apparently nearly in proportion to them. Eleven German filter-works in 1892, drawing their waters from rivers, filtered on an average 51 million gallons of water per acre between scrapings, the single results ranging from 28 at Bremen to 71 at Stuttgart, while Zürich, drawing its water from a lake which is but very rarely turbid, filtered 260 million gallons per acre between scrapings. Unfortunately, the quantities at Berlin, where (in 1892 two thirds and now all) the water is drawn from comparatively large ponds on the rivers, are not available for comparison.
At London, in 1884, the average quantities of water filtered [Pg 73] between scrapings varied from 43 to 136 million gallons per acre with the different companies, averaging 85, and in 1892 the quantities ranged from 73 to 157, averaging 90 million gallons per acre. The greater quantity filtered at London may be due to the greater sizes of the sedimentation-basins, which for all the companies together hold a nine days’ supply at London against probably less than one day’s supply for the German works.
There is little information available in regard to the frequency of scraping with water drawn from impounding reservoirs. In some experiments made by Mr. FitzGerald at the Chestnut Hill reservoir, Boston, the results of which are as yet unpublished, a filter with sand of an effective size of only .09 mm. averaged 58 million gallons per acre between scrapings for nine periods, the rate of filtration being 1.50 million gallons per acre daily, while another filter, with sand of an effective size of .18 mm., passed an average of 93 million gallons per acre for ten periods at the same rate. These experiments extended through all seasons of the year, and taking into account the comparative fineness of the sands they show rather high quantities of water filtered between scrapings.
The quantity of water filtered between scrapings is usually greatest in winter, owing to the smaller quantity of sediment in the raw water at this season, and is lowest in times of flood, regardless of season. In summer the quantity is often reduced to a very low figure in waters supporting algæ growths, especially when the filters are not covered. Thus at Stralau in 1893 during the algæ period the quantity was reduced to 14 million gallons per acre for open filters,[21] but this was quite exceptional, the much-polluted, though comparatively clear, Spree water furnishing unusually favorable conditions for the algæ.
[Pg 74]
QUANTITY OF SAND TO BE REMOVED.
In regard to the quantity of sand to be removed and washed, if we take the average result given above for the German works filtering river-waters of 51,000,000 gallons per acre filtered between scrapings, and the depth of sand removed at two centimeters or 0.79 inch, we find that one volume of sand is required for every 2375 volumes of water filtered, or 2.10 cubic yards per million gallons. At Bremen, the highest average result, the quantity would be 3.80 yards, and at Stralau during the algæ season 7.70 yards. At Zürich, on the other hand, the quantity is only 0.41 yard, and at London, with 87,000,000 gallons per acre filtered between scrapings, the quantity of sand washed would be 1.24 yards per million gallons; assuming always that the layer removed is 0.79 inch thick.
These estimates are for the regular scrapings only, and do not include the annual deeper scraping before replacing the sand, which would increase them by about one third.
WASTING THE EFFLUENTS AFTER SCRAPING.
It has already been stated that an important part of the filtration takes place in the sediment layer deposited on top of the sand from the water. When this layer is removed by scraping its influence is temporarily removed, and reduced efficiency of filtration may result. The significance of this reduced efficiency became apparent when the bacteria in the water were studied in their relations to disease, and Piefke suggested[22] that the first effluent after scraping should be rejected for one day after ordinary scrapings and for one week after replacing the sand. In a more recent paper[23] he reduces these estimates to the first million gallons of water per acre filtered after scraping
[Pg 75]
for open and twice as great a quantity for covered filters, and to six days after replacing the sand, which last he estimates will occur only once a year. Taking the quantity of water filtered between scrapings at 13.9 million gallons per acre, the quantity observed at Stralau in the summer of 1893, he finds that it is necessary to waste 9 per cent of the total quantity of effluent from open and 13.8 per cent of that from covered filters.
The eleven German water-works[24] filtering river-waters, however, filtered on an average 51.0 instead of 13.9 million gallons per acre between scrapings, and applying Piefke’s figures to them the quantities of water to be wasted would be only about one fourth of his estimates for Stralau.
The rules of the Imperial Board of Health[25] require that every German filter shall be so constructed “that when an inferior effluent results it can be disconnected from the pure-water pipes and the filtrate allowed to be wasted.” The drain-pipe for removing the rejected water should be connected below the apparatus for regulating the rate and loss of head, so that the filter can be operated exactly as usual, and the effluent can be turned back to the pure-water pipes without stopping or changing the rate. The works at Berlin and at Hamburg conform to this requirement, and most of the older German works have been or are being built over to make them do so.
In regard to the extent of deterioration after scraping, Piefke’s experiments have always shown much larger numbers of bacteria both of the ordinary forms and of special applied forms on the first day after scraping, the numbers frequently being many times as high as at other times.
At the Lawrence Experiment Station it was found in 1892 that on an average the number of water bacteria was increased by 70 per cent (continuous filters only) for the three days following scraping, while B. prodigiosus when applied was increased 140 per [Pg 76] cent, the increase being most marked where the depth of sand was least, and with the highest rate of filtration.
The same tendency was found in 1893, when the increase in the water bacteria on the first day after scraping was only 19 per cent and B. prodigiosus 64 per cent, but for a portion of the year the difference was greater, averaging 132 and 262 per cent, respectively. These differences are much less than those recorded by Piefke, and with the high efficiencies regularly obtained at Lawrence they would hardly justify the expensive practice of wasting the effluent.
The reduction in efficiency following scraping is much less at low rates, and if a filter is started at much less than its normal rate after scraping, and then gradually increased to the standard after the sediment layer is formed, the poor work will be largely avoided. Practically this is done at Berlin and at Hamburg. The filters are started at a fourth or less of the usual rates and are gradually increased, as past experience with bacterial results has shown it can be safely done, and the effluent is then even at first so well purified that it need not be wasted.
Practically in building new filters the provision of a suitable connection for wasting the effluents into the drain which is necessary for emptying them involves no serious expense and should be provided, but it may be questioned how often it should be used for wasting the effluents. If the raw water is so bad that a good effluent cannot be obtained by careful manipulation even just after scraping, the course of the Berlin authorities in closing the Stralau works and seeking a less polluted supply would seem to be the only really safe procedure.
SAND-WASHING.

Cleaning a Filter, East London.

Washing Dirty Sand with Hose, Antwerp.
[To face page 76.]
The sand-washing apparatus is an important part of most European filtering plants. It seldom happens that a natural sand can be found clean enough and sufficiently free from fine particles although such a sand was found and used for the Lawrence filter. Most of the sand in use for filtration in Europe was originally washed. In the operation of the filters also, sand-washing is used for the dirty sand, which can then be used over and over at a much lower cost than would be the case if fresh sand was used for refilling. The methods used for washing sand at the different works present a great variety both in their details and in the underlying principles. Formerly boxes with double perforated bottoms in which the sand was placed and stirred by a man as water from below rose through them, and other similar arrangements were commonly used, but they are at present only retained, so far as I know, in some of the smaller English works. The cleansing obtained is apparently considerably less thorough than with some of the modern devices.
Fig. 13.—Hose-washing for Dirty Sand.
Hose-washing is used in London by the Southwark and Vauxhall, Lambeth and Chelsea companies, and also at Antwerp. For this a platform is constructed about 15 feet long by 8 feet wide, with a pitch lengthwise of 6 to 8 inches (Fig. 13). The[Pg 78] platform is surrounded by a wall rising from one foot at the bottom to three feet high at the top, except the lower end, which is closed by a removable plank weir 5 or 6 inches high. From two to four cubic yards of the sand are placed upon this platform and a stream of water from a hose with a 3⁄4 or 7⁄8-inch nozzle is played upon it, moving it about from place to place. The sand itself is always kept toward the upper end of the platform, while the water with the dirt removed flows down into the pond made by the weir, where the sand settles out and the dirt overflows with the water. When the water comes off clear, which is usually after an hour or a little less, the weir is removed, and, after draining, the sand is removed. These arrangements are built in pairs so that the hose can be used in one while the sand is being changed in the other. They are usually built of brick laid in cement, but plank and iron are also used. The corners are sometimes carried out square as in the figure, but are more often rounded. The washing is apparently fairly well done.
In Germany the so-called “drum” washing-machine, drawings of which have been several times published,[26] has come to be almost universally used. It consists of a large revolving cylinder, on the bottom of the inside of which the sand is slowly pushed up toward the higher end by endless screw-blades attached to the cylinder, while water is freely played upon it all the way. The machine requires a special house for its accommodation and from 2 to 4 horse-power for its operation. It washes from 2.5 to 4 yards of sand per hour most thoroughly, with a consumption of from 11 to 14 times as large a volume of water. The apparatus is not patented or made for sale, but full plans can be easily secured.
A machine made by Samuel Pegg & Sons, Leicester, Eng., pushes the sand up a slight incline down which water flows. It is very heavy and requires power to operate it. The patent has [Pg 79] expired. A machine much like it but lighter and more convenient and moved by water-power derived from the water used for washing instead of steam-power is used at Zürich with good results.
In Greenway’s machine the sand is forced by a screw through a long narrow cylinder in which there is a current of water in the opposite direction. The power required is furnished by a water-motor, as with the machine at Zürich. The apparatus is mounted on wheels and is portable; it has an appliance for piling up the washed sand or loading it onto cars. It is patented and is manufactured by James Gibb & Co., London.
Several of the London water companies are now using ejector washers, and such an apparatus has been placed by the side of the “drum” washers at Hamburg. This apparatus was made by Körting Brothers in Hannover, and combines the ejectors long made by that firm with hoppers from designs by Mr. Bryan, engineer of the East London Water Company. An apparatus differing from this only in the shape of the ejectors and some minor details has been patented in England, and is for sale by Messrs. Hunter, Frazer & Goodman, Bow, London.
Both of these forms consist of a series of conical hoppers, from the bottom of each of which the sand and water are forced into the top of the next by means of ejectors, the excess of dirty water overflowing from the top of each hopper. The apparatus is compact and not likely to get out of order, but is not portable. It can be easily arranged to take the sand at the level of the ground, or even lower if desired, and deliver it washed at some little elevation, thus minimizing hand-labor. The washing is regular and thorough. The objection most frequently raised against its use is the quantity of water required, but at Hamburg I was informed that the volume of water required was only about 15 times that of the sand, while almost as much (13-14 volumes) were required for the “drum” washers, and[Pg 80] the saving in power much more than offset the extra cost for water.
In addition to the above processes of sand-washing, Piefke’s method of cleaning without scraping[27] might be mentioned, although as yet it has hardly passed the experimental stage, and has only been used on extremely small filters. The process consists of stirring the surface sand of the filter with “waltzers” while a thin sheet of water rapidly flows over the surface. This arrangement necessitates a special construction of the filters, providing for rapidly removing the unfiltered water from the surface, and for producing a regular and rapid movement of a thin sheet of water over the surface. In the little filters now in use, one of which I saw in a brewery in Berlin, the cleaning is rapidly, cheaply, and apparently well done.
In washing dirty sand it is obvious that any small sand-grains will be removed with the dirt, and in washing new sand the main object is to remove the grains below a certain size. It is also apparent that the sizes of grains which will and those which will not be removed are dependent upon the mechanical arrangements of the washer, as, for example, with the ejectors, upon the sizes of the hoppers, and the quantity of water passing through them, and care should be taken to make them correspond with the size of grain selected for the filter sand. This can only be done by experiment, as no results are available on this point.
In some places filtered water is used for sand-washing, although this seems quite unnecessary, as ordinary river-water answers very well. It is, however, often cheaper, especially in small works, to use the filtered water from the mains rather than provide a separate supply for the washers.
The quantity of water required for washing may be estimated at 15 times the volume of the sand and the sand as 0.04 per cent of the volume of the water filtered (page 74), so that[Pg 81] 0.6 per cent of the total quantity of water filtered will be required for sand-washing.
The cost of sand-washing in Germany with the “drum” washers is said to be from 14 to 20 cents per cubic yard, including labor, power, and water. In America the water would cost no more, but the labor would be perhaps twice as dear. With an ejector apparatus I should estimate the cost of washing dirty sand as follows: The sand would be brought and dumped near to the washer, and one man could easily feed it in, as no lifting is required. Two men would probably be required to shovel the washed sand into barrows or carts with the present arrangements, but I think with a little ingenuity this handling could be made easier.
| ESTIMATED COST OF OPERATING EJECTOR WASHERS 9 HOURS. | |
|---|---|
Wages of 3 men at $2.00 |
$6.00 |
110,000 gals, water (15 times the volume of sand) at 0.05 a thousand gals. |
5.50 |
Total cost of washing 36 cubic yards |
$11.50 |
The cost of washing new sand might be somewhat less. The other costs of cleaning filters, scraping, transporting, and replacing the sand are much greater than the washing itself. Lindley states that at Warsaw 29 days’ labor of 10 hours for one man are required to scrape an acre of filter surface, and four times as much for the annual deep scraping, digging up, and replacing the sand. The first expense occurs in general monthly, and the second only once a year. At other places where I have secured corresponding data the figures range from 19 to 40 days’ labor to scrape one acre, and average about the same as Lindley estimates.
Under some conditions sand-washing does not pay, and in[Pg 82] still others it is almost impossible. No apparatus has yet been devised which will wash the dirt out of the fine dune-sands used in Holland without washing a large part of the sand itself away, and in these works fresh sand, which is available in unlimited quantities and close to the works, is always used. At Breslau the dirty sand is sold for building purposes for one third of the price paid for new sand dredged from the river, delivered at the works, and no sand is ever washed. Budapest, Warsaw, and Rotterdam also use fresh river-sand without washing, except a very crude washing to remove clay at Budapest.
[Pg 83]
CHAPTER VI.
THEORY AND EFFICIENCY OF CONTINUOUS FILTRATION.
The first filters for a public water-supply were built by James Simpson, engineer of the Chelsea Water Company at London in 1829. They were apparently intended to remove dirt from the water in imitation of natural processes, and without any very clear conception of either the exact extent of purification or the way in which it was to be accomplished. The removal of turbidity was the most obvious result, and a clear effluent was the single test of the efficiency of filtration, as it remains the legal criterion of the work of the London filters even to-day, notwithstanding the discovery and use of other and more delicate tests.
The invention and use of methods for determining the organic matters in water by Wanklyn and Frankland, about 1870, led to the discovery that the proportion of organic matters removed by filtration was disappointingly low, and as, at the time, and for many years afterward, an exaggerated importance was given to the mere quantities of organic matters in water, it was concluded that filtration had only a limited influence upon the healthfulness of the filtered water, and that practically as much care must be given to securing an unpolluted water as would be the case if it were delivered direct without filtration. This theory, although not confirmed by more recent investigation, undoubtedly has had a good influence upon the English works by causing the selection of raw waters free from excessive pollutions, and, in cases like the London supplies, drawn from the Thames and the Lea, in stimulating a most jealous care of the watersheds and the purification of sewage by the towns upon them.
[Pg 84]
It was only after the discovery of the bacteria in water and their relations to health that the hygenic significance of filtration commenced to be really understood. Investigations of the bacteria in the waters before and after filtration were carried out at Berlin by Plagge and Proskauer, at London by Dr. Percy Frankland, and also at Zürich, Altona, and on a smaller scale at other places. These investigations showed that the bacteria were mainly removed by filtration, the numbers in the effluents rarely exceeding two or three per cent of those in the raw water. This gave a new aspect to the problem.
It was further observed, especially at Berlin and Zürich, that the numbers of bacteria in effluents were apparently quite independent of the numbers in the raw water, and the theory was formed that all of the bacteria were stopped by the filters, and that those found in the effluents were the result of contamination from the air and of growths in the underdrains. The logical conclusion from this theory was that filtered water was quite suitable for drinking regardless of the pollution of its source.
It was, however, found that the numbers of bacteria in the effluents were higher immediately after scraping than at other times, and it was concluded that before the formation of the sediment layer some bacteria were able to pass the sand, and it was therefore recommended that the first water filtered after scraping should be rejected.
Piefke at Berlin gave the subject careful study, and came to the conclusion that it was almost entirely the sediment layer which stopped the bacteria, and that the bacteria themselves in the sediment layer formed a slimy mass which completely intercepted those in the passing water. When this layer was removed by scraping, the action was stopped until a new crop of bacteria had accumulated. In support of this idea he stated that he had taken ordinary good filter-sand and killed the bacteria in it by heating it, and that on passing water through, no purification was effected—in fact, the effluent contained more[Pg 85] bacteria than the raw water. After a little, bacteria established themselves in the sand, and then the usual purification was obtained. Piefke concluded that the action of the filter was a biological one; that simple straining was quite inadequate to produce the results obtained; that the action of the filter was mainly confined to the sediment layer, and that the depth of sand beyond the slight depth necessary for the support of this layer had no appreciable influence upon the results. The effect of this theory is still seen in the shallow sand layers used at Berlin and some other German works, although at London the tendency is rather toward thicker sand layers.
Piefke’s deductions, however, are not entirely supported by his data as we understand them in the light of more recent investigation. The experiment with sterilized sand has been repeatedly tried at the Lawrence Experiment Station with results which quite agree with Piefke’s, but it has also been found that the high numbers, often many times as high as in the raw water, do not represent bacteria which pass in the ordinary course of filtration, but instead enormous growths of bacteria throughout the sand supported by the cooked organic matter in it. It has been repeatedly found that ordinary sand quite incapable of supporting bacterial growths, after heating to a temperature capable of killing the bacteria will afterwards furnish the food for most extraordinary numbers. A filter of such sand may stop the bacteria of the passing water quite as effectually as any other filter, but if so, the fact cannot be determined without recourse to special methods, on account of the enormous numbers of bacteria in the sand, a small part of which are carried forward by the passing water, and completely mask the normal action of the filter.
The theory that all or practically all of the bacteria are intercepted by the sediment layer, and that those in the effluent are the result of growths in the sand or underdrains, received two hard blows in 1889 and 1891, when mild epidemics of typhoid fever[Pg 86] followed unusually high numbers of bacteria in the effluents at Altona and at Stralau in Berlin, with good evidence in each case that the fever was directly due to the water. Both of these cases came during, and as the result of, severe winter weather with open filters and under conditions which are now recognized as extremely unfavorable for good filtration.
As a result of the first of these epidemics a series of experiments were made at Stralau by Fränkel and Piefke in 1890. Small filters were constructed, and water passed exactly as in the ordinary filters. Bacteria of special kinds not existing in the raw water or effluents were then applied, and the presence of a very small fraction of them in the effluents demonstrated beyond a doubt that they had passed through the filters under the ordinary conditions of filtration. These experiments were afterwards repeated by Piefke alone under somewhat different conditions with similar results. The numbers of bacteria passing, although large enough to establish the point that some do pass, were nevertheless in general but a small fraction of one per cent of the many thousands applied.
This method of testing the efficiency of filters had already been used quite independently by Prof. Sedgwick at the Lawrence Experiment Station in connection with the purification of sewage, and has since been extensively used there for experiments with water-filtration.
Kümmel also found at Altona that while in the regular samples for bacterial examination, all taken at the same time in the day, there was no apparent connection between the numbers of bacteria in the raw water and effluents, by taking samples at frequent intervals throughout the twenty-four hours, as has been done in a more recent series of experiments, and allowing for the time required for the water to pass the filters, a well-marked connection was found to exist between the numbers of bacteria in the raw water and in the effluents.
Fig. 14.—Showing Bacteria supposed to come through Filters and from the Underdrains.
The subject has more recently been studied in much detail at[Pg 87] the Lawrence Experiment Station, and it now appears that the bacteria in the effluent from a filter are from two sources: directly from the filtered water, and from the lower layers of the filter and underdrains. Thus we may say:
Bacteria in effluent = Bacteria from underdrains + a⁄100 × bacteria in raw water,
where a is the per cent of bacteria actually passing the filter.
Both of these terms depend upon a whole series of complex and but imperfectly understood conditions. In general the bacteria from the underdrains are low in cold winter weather, often almost nil, while at Lawrence with water temperatures of 70 to 75 degrees, and over, in July and August, the numbers from this source may reach 200 or 300, but for the other ten months of the year rarely exceed 50 under normal conditions. In summer especially it seems to be greater at low than at high rates of filtration (although a high rate for a short time only increases it), and so varies in the opposite way from the numbers actually passing the filters. This subject is by no means clearly understood; it is difficult, almost impossible, to separate the numbers of bacteria into the two parts—those which come directly through and may be dangerous, and those which have other origins and are harmless. The sketch, Fig. 14, is drawn to represent my idea of the way they may be divided. It has no statistical basis whatever. The light unshaded section shows the percentage number of bacteria[Pg 88] which I conceive to be coming through a filter under given conditions at various rates of filtration, while the shaded section above represents the bacteria from other sources, and the upper line represents the sum of the two, or the total number of bacteria in the effluent. The relative importance of the two parts would probably vary widely with various conditions. With the conditions indicated by the sketch the number of bacteria in the effluent is almost constant: for a variation of only from 1.4 to 2.5 per cent of the number applied for the whole range is not a wide fluctuation for bacterial results, but the number in the lower and dangerous section is always rapidly increasing with increasing rate.
This theory of filtration accounts for many otherwise perplexing facts. The conclusion reached at Zürich and elsewhere that the efficiency of filtration is independent of rate may be explained in this way. This is especially probable at Zürich, where the number of bacteria in the raw water was only about 200, and an extremely large proportion relatively would have to pass to make a well-marked impression upon the total number in the effluent.
These underdrain bacteria are, so far as we know, entirely harmless; we are only interested in them to determine how far they are capable of decreasing the apparent efficiency of filtration below the actual efficiency, or the per cent of bacteria really removed by the filter.
This efficiency is dependent upon a large number of conditions many of which have already been discussed in connection with grain-size of filter sand, underdrains, rate of filtration, loss of head, etc., and a mere reference to them here will suffice. Perhaps the most important single condition is the rate, the numbers of bacteria passing increase rapidly with it. Next, fine sand and in moderately deep layers tends to give high efficiency. The influence of the loss of head, often mentioned, is not shown to be important by the Lawrence results, nor can I find[Pg 89] satisfactory European results in support of it. Uniformity in the rate of filtration on all parts of the filtering area and a constant rate throughout the twenty-four hours are regarded as essential conditions for the best results. Severe winter weather has indirectly, by disturbing the regular action of open filters, an injurious influence, and has been the cause of most of the cases where filtered waters have been known to injure the health of those who have drunk them. This action is excluded in filters covered with masonry arches and soil, and such construction is apparently necessary for the best results in places subject to cold winters.
The efficiency of filtration under various conditions has been studied by a most elaborate series of experiments at Lawrence with small filters to which water has been applied containing a bacterium (B. prodigiosus) which does not occur naturally in this country and is not capable of growing in the filter, so that the results should represent only the bacteria coming through the filter and not include any additions from the underdrains. These results, which have been published in full in the reports of the Massachusetts State Board of Health, especially for the years 1892 and 1893, show that the number of bacteria passing increases rapidly with increasing rate, and slowly with decreasing sand thickness and increased size of sand-grain.
Assuming that the number of bacteria passing is expressed by the formula
1 [(rate)2 × effective size of sand]
Per cent bacteria passing = — —————————————
2 √thickness of the sand in inches
where the rate is expressed in million gallons per acre daily, and calculating by it the numbers of bacteria for the seventy-three months for which satisfactory data are available from 11 filters in 1892 and 1893, we find that
In 14 cases the numbers observed were 4 to 9 times as great as the calculated numbers;
In 6 cases they were 2 to 3 times as great;
[Pg 90]
In 35 cases they were between 1⁄2 and 2 times the calculated numbers.
In 17 cases they were 1⁄2 to 1⁄3 of them.
In 11 cases they were less than 1⁄3 the calculated numbers.
The agreement is only moderately good, and in fact no such formula could be expected to give more than very rough approximations, because it does not take into consideration the numerous other elements, such as uniformity and regularity of filtration, the influence of scraping, the character of the sediment in the raw water, etc., which are known to affect the results. Perhaps the most marked general difference is the tendency of new or freshly-filled filters to give higher, and of old and well-compacted filters to give lower, results than those indicated by the formula.
Comparing this formula with Piefke’s results given in his “Neue Ermittelungen”[28] the formula gives in the first series (0.34 mm. sand, 0.50 m. thick, and rate 100 mm. per hour), 0.25 per cent passing, while the average number of B. violacious reported, excluding the first day of decreased efficiency after scraping, was 0.26 per cent. In the second series, with half as high a rate the numbers checked exactly the calculated 0.06 per cent.
In other experiments,[29] however, in 1893, when the calculated per cent was also 0.25, only 0.03, 0.04, and 0.07 per cent were observed in the effluents.
Comparing the results from the actual filters, (which numbers also include the bacteria from the underdrains and should therefore be somewhat higher) with the numbers calculated as passing through, I find that for the 46 days, Aug. 20 to Oct. 4, 1893, for which detailed results of the Stralau works are given by Piefke, the average calculated number passing is 0.20 per [Pg 91] cent, while twice as many were observed in the effluents; although three of the filters gave better effluents than the other eight, and the numbers from them approximated closely the calculated numbers. If we calculate the percentages of bacteria passing a number of filters, using the maximum rate of filtration allowed for the German filters where this is accurately determined, and for the English filters taking the maximum rate at one and one-half times the rate obtained by dividing the daily quantity by the area of filters actually in use, we obtain:
| Average Depth of Sand, Inches. |
Effective Size of Sand- grain. |
Maximum Rate of Filtration. |
Per cent Bacteria passing 1 r2d = — ——— 2 √sand |
|
|---|---|---|---|---|
| Hamburg | 32 | 0.31 | 1.60 | 0.07 |
| Altona | 28 | 0.34 | 2.57 | 0.21 |
| Berlin, Stralau | 20 | 0.34 | 2.57 | 0.25 |
| Berlin, Müggel | 20 | 0.34 | 2.57 | 0.25 |
| Berlin, Tegel | 20 | 0.37 | 2.57 | 0.27 |
| London, Southwark & Vauxhall | 36 | 0.34 | 2.81 | 0.22 |
| London, West Middlesex | 39 | 0.37 | 2.81 | 0.23 |
| London, Chelsea | 54 | 0.36 | 3.27 | 0.26 |
| London, Grand Junction | 30 | 0.40 | 3.27 | 0.39 |
| London, Lambeth | 36 | 0.36 | 3.75 | 0.42 |
| Middlesborough | 20 | 0.42 | 5.85 | 1.58 |
| Zürich | 26 | 0.35 | 7.50 | 1.90 |
The numbers actually observed are in every case higher than the calculated per cents passing, as indeed they should be on account of those coming from the underdrains, accidental contamination of the samples, etc.
It may be said that filtration now practised in European works under ordinary conditions never allows over 1 or 2 per cent bacteria of the raw water to pass, and ordinarily not over one fourth to one half of one per cent, although exact data cannot be obtained owing to masking effect of the bacteria which come from below and which bear no relation to those of the raw water. By increasing the size of the filters, fineness and[Pg 92] depth of sand (as at Hamburg), the efficiency can be materially increased above these figures. At the same time it must be borne in mind that the effectiveness of a filter may be greatly impaired by inadequate underdraining, by fluctuating rates of filtration where these are allowed, by freezing in winter in the case of open filters in cold climates, and by other irregularities, all of which can be prevented by careful attention to the respective points.
The action of a continuous filter throughout is mainly that of an exceedingly fine strainer, and like a strainer is mainly confined to the suspended or insoluble matters in the raw water. The turbidity, sediment, and bacteria of the raw water are largely or entirely removed, while hardness, organic matter, and color, so far as they are in solution, are removed to only a slight extent, if at all. Hardness can be removed by the addition of lime in carefully determined quantity before filtration (Clark’s process), by means of which the excess of carbonic acid in the water is absorbed and the lime added, together with that previously in the water, is precipitated.
Ordinary filtration will remove from one fourth to one third of the yellow-brown color of peaty water. A larger proportion can be removed by the addition of alum, which by decomposing forms an insoluble compound of alumina with the coloring matter, while the acid of the alum goes into the effluent either as free acid, or in combination with the lime or other base in the water, according to their respective quantities. Freshly precipitated alumina can be substituted for the alum at increased expense and trouble, and tends to remove the color without adding acid to the water. These will be discussed more in detail in connection with mechanical filters. Alum is but rarely used in slow sand filtration, the most important works where it is used being in Holland with peaty waters.
After all, the most conclusive test of the efficiency of filtration is the healthfulness of the people who drink the filtered water;[Pg 93] and the fact that many European cities take water-supplies from sources which would not be considered fit for use in the United States and, after filtering them, deliver them to populations having death-rates from water-carried diseases which are so low as to be the objects of our admiration, is the best proof of the efficiency of carefully conducted filtration.
It is only necessary to refer to London, drawing its water from the two small and polluted rivers, the Thames and the Lea; to Altona, drawing its water from the Elbe, polluted by the sewage of 6,000,000 people, 700,000 of them within ten miles above the intakes; to Berlin, using the waters of the Havel and the Spree; to Breslau, taking its water from the Oder charged with the sewage of mining districts in Silicia and Galicia, where cholera is so common; to Lawrence, with its greatly decreased death-rate since it has had filtered water, and to the hundred other places which protect themselves from the infectious matters in their raw waters by means of filtration. A few of these cases are described more in detail in Appendices V to IX, and many others in the literature mentioned in Appendix X.
An adequate presentation of even those data which have been already worked up and published would occupy too much space. I think every one who has carefully studied the recent history of water filtration in its relation to disease has been convinced that filtration carefully executed under suitable and normal conditions, even if not an absolute, is at least a very substantial protection against water-carried diseases, and the few apparent failures to remove objectionable qualities have been without exception due to abnormal conditions which are now understood and in future can be prevented.
BACTERIAL EXAMINATION OF WATERS.
Every large filter-plant should have arrangements for the systematic bacterial examination of the water before and after[Pg 94] filtration, especially where the raw water is subject to serious pollution. Such examinations need not be excessively expensive, and they will not only show the efficiency of the plant as a whole, but may be made to show the relative efficiencies of the separate filters, the relative efficiencies at different parts of the periods of operation, the effect of cold weather, etc., and will then be a substantial aid to the superintendent in always securing good effluents at the minimum cost.
In addition a complete record of the bacteria in the water at different times may aid in determining definitely whether the water was connected with outbreaks of disease. Thus if an outbreak of disease of any kind were preceded at a certain interval by a great increase in the number of bacteria,—as has been the case, for example, with the typhoid epidemics at Altona and Berlin (see Appendices II and VII),—a presumption would arise that they might have been connected with each other, and each time it was repeated the presumption would be strengthened, while, on the other hand, outbreaks occurring while the bacteria remained constantly low would tend to discredit such a theory.
Bacterial investigations inaugurated after an epidemic is recognized, as has frequently been done, seldom lead to results of value, both because the local normal bacterial conditions are generally unknown at the commencement of the investigation, and because the most important time, the time of infection, is already long past before the first samples are taken. The fact that such sporadic activities have led to few definite results should throw no discredit upon continued observations, which have repeatedly proved of inestimable value.
Considerable misconception of the use of bacterial examinations exists. The simple bacterial count ordinarily used, and of which I am now speaking, does not and cannot show whether a water contains disease-germs or not. I object to the Chicago water, not so much because a glass of it contains a hundred thousand bacteria more or less, as because I am convinced, by a study[Pg 95] of its source in connection with the city’s death-rate, that it actually carries disease-germs which prove injurious to thousands of those who drink it. Now the fact being admitted that the water is injurious to health, variations in the numbers of bacteria in the water drawn from different intakes and at different times probably correspond roughly with varying proportions of fresh sewage, and indicate roughly the relative dangers from the use of the respective waters. If filters should be introduced, the numbers of bacteria in the effluents under various conditions would be an index of the respective efficiencies of filtration, and would serve to detect poor work, and would probably suggest the measures necessary for better results.
I would suggest the desirability of such investigations where mechanical filters are used, quite as much as in connection with slow filtration; and it would also be most desirable in the case of many water-supplies which are not filtered at all. Such continued observations have been made at Berlin since 1884; at London since 1886; at Boston and Lawrence since 1888; and recently at a large number of places, including Chicago, where observations by the city were commenced in 1894. They are now required by the German Government in the case of all filtered public water-supplies in Germany, without regard to the source of the raw water. The German standard requires that the effluent from each single filter, as well as the mixed effluent and raw water, shall be examined daily, making at some works 10 to 30 samples daily. This amount of work, however, can usually be done by a single man; and when a laboratory is once started, the cost of examining 20 samples a day will not be much greater than if only 20 a week are taken. In England and at some of the Continental works drawing their waters from but slightly polluted sources, much smaller numbers of samples are examined.
The question whether the examinations should be made under the direction of the water-works company or department, or by an independent body—as, for instance, by the Board of Health—will[Pg 96] depend upon local conditions. The former arrangement gives the superintendent of the filters the best chance to study their action, as he can himself control the collection of samples in connection with the operation of the filters, and arrange them to throw light upon the points he wishes to investigate; while examination by a separate authority affords perhaps greater protection against the possible carelessness or dishonesty of water-works officials. An arrangement being adopted in many cases in Germany is to have a bacterial laboratory at the works which is under the control of the superintendent, and in which the very numerous compulsory observations are made, while the Board of Health causes to be examined from time to time by its own representatives, who have no connection with the water-works, samples taken to check the water-works figures, as well as to show the character of the water delivered.
It seems quite desirable to have a man whose principal business is to make these examinations; as in case he also has numerous other duties, the examinations may be found to have been neglected at some time when they are most wanted. Such a man should have had thorough training in the principles of bacterial manipulation, but it is quite unnecessary that he should be an expert bacteriologist, especially if a competent bacteriologist is retained for consultation in cases of doubt or difficulty.
[Pg 97]
CHAPTER VII.
INTERMITTENT FILTRATION.
By intermittent nitration is understood that filtration in which the filtering material is systematically and adequately ventilated, and where the water during the course of filtration is brought in contact with air in the pores of the sand. In continuous filtration, which alone has been previously considered, the air is driven out of the sand as completely as possible before the commencement of filtration, and the sand is kept continuously covered with water until the sand becomes clogged and a draining, with an incidental aeration, is necessary to allow the filter to be scraped and again put in service.
In intermittent filtration, on the other hand, water is taken over the top of the drained sand and settles into it, coming in contact with the air in the pores of the sand, and passes freely through to the bottom when the water-level is kept well down. After a limited time the application of water is stopped, and the filter is allowed to again drain and become thoroughly aerated preparatory to receiving another dose of water.
This system of treating water was suggested by the unequalled purification of sewage effected by a similar treatment. It has been investigated at the Lawrence Experiment Station, and applied to the construction of a filter for the city of Lawrence, both of which are due to the indefatigable energy of Hiram F. Mills, C.E.
In its operation intermittent differs from continuous filtration in that the straining action is less perfect, because the filters yield no water while being aerated, and must therefore filter at a greater velocity when in use to yield the same quantity of water in a given time, and also on account of the mechanical disturbance[Pg 98] which is almost invariably caused by the application of the water; but, on the other hand, the oxidizing powers of the filter, or the tendency to nitrify and destroy the organic matters, are stronger, and in addition, if the rate is not too high, the bacteria die more rapidly in the thoroughly aerated sand than is the case with ordinary filters.
It was found at Lawrence in connection with sewage filters that when nitrification was actively taking place the numbers of bacteria were much lower than under opposite conditions, and it was thought that nitrification in itself might cause the death of the bacteria. Later experiments, however, with pure cultures of bacteria of various kinds applied to intermittent filters with water to which ammonia and salts suitable for nitrification were added, showed that bacteria of all the species tried were able to pass the filter in the presence of nitrification, producing at least one thousand times as much nitrates as could result in any case of water-filtration, as freely as was the case when the ammonia was not added and there was but little nitrification. These results showed conclusively that nitrification in itself is not an important factor in bacterial removal, although nitrification and bacterial purification do to some extent go together; perhaps in part because the nitrification destroys the food of the bacteria and so starves them out, but probably much more because the conditions of aeration, temperature, etc., which favor nitrification also favor equally, and even in its absence, the death of the bacteria.
The rate at which water must pass through an intermittent filter is, on account of the intervals of rest, considerably greater than that required to give a corresponding total yield from a continuous filter, and its straining effect is reduced to an extent comparable to this increase in rate; and if other conditions did not come in, the bacterial efficiency of an intermittent filter would remain below that of a continuous one.
As a matter of fact the bacterial efficiency has usually been[Pg 99] found to be less with intermittent filters at the Lawrence Experiment Station, when they have been run at rates such as are commonly used for continuous filters in Europe, say from one and one half to two million gallons and upwards per acre daily. With lower rates, and especially with rather fine materials, the bacterial efficiency is much greater; but it may be doubted whether it would ever be greater than that of a continuous filter with the same filtering material and the same total yield per acre. The number of bacteria coming from the underdrains is apparently generally less, and with very high summer temperatures much less, than in continuous filters, and this often gives an apparent bacterial superiority to the intermittent filters.
The effluents from intermittent often contain less slightly organic matter than those from continuous filters; but, on the other hand, hardly any water proposed for a public water-supply has organic matter enough to be of any sanitary significance whatever, apart from the living bodies which often accompany it; and if the latter are removed by straining or otherwise, we can safely disregard the organic matters. In addition, the water filtered will in a great majority of cases have enough air dissolved in itself to produce whatever oxidation there is time for in the few hours required for it to pass the filter, and it is only at very low rates of filtration that intermittent filters produce effluents of greater chemical purity than by the ordinary process. The yellow-brown coloring matter present in so many waters appears to be quite incapable of rapid nitrification; and where it is to some extent removed by filtration, the action is dependent upon other and but imperfectly understood causes which seem to act equally in continuous and intermittent filters.
The peculiarities of construction involved by this method of filtration will be best illustrated by a discussion of the Lawrence city filter designed by Hiram F. Mills, C.E., which is the only filter in existence upon this plan.[30]
[Pg 100]
THE LAWRENCE FILTER.
The filter consists of a single bed 21⁄2 acres in area, the bottom of which is 7 feet below low water in the river, and filled with gravel and sand to an average depth of 41⁄2 feet. The filter is all in a single bed instead of being divided into the three or four sections which would probably have been used for a continuous filter of this size. The water-tight bottom also was dispensed with, and the gravel was prevented from sinking into the silt by thin intermediate layers of graded materials. The saving in cost was considerable; but, on the other hand, a considerable quantity of ground-water comes up through the bottom and increases the hardness of the water from 1.5 to 2.6 parts of calcium carbonate in 100,000; and while the water when compared with many other waters is still extremely soft, the addition cannot be regarded as desirable. The ground-water also contains iron, which increases the color of the water above what it would otherwise be.
The underdrains have a frictional resistance ten times as great as would be desirable for a continuous filter, the idea being to check extreme rates of filtration in case of unequal flooding, and also to limit the quantity of water which could be gotten through the filter to that corresponding to a moderate rate of filtration.
The sand, instead of being all of the same-sized grain, is of two grades, with effective sizes respectively 0.25 and 0.30 mm., the coarser sand being placed farthest away from the underdrains, where its greater distance is intended to balance its reduced frictional resistance and make all parts filter at an equal rate.
The surface instead of being level is waved, that is, there are ridges thirty feet apart, sloping evenly to the valleys one foot deep half way between them, to allow water to be brought on[Pg 101] rapidly without disturbing the sand surface. For the same reason, as well as to secure equality of distribution, a system of concrete carriers for the raw water goes to all parts of the filter, reducing the effective filtering area by 4 or 5 per cent. The filter is scraped as necessary in sections, the work being performed when the filter is having its daily rest and aeration. Owing to the difference in frictional resistance before and after scraping, and to the fact that it is impossible to scrape the entire area in one day, considerable variations in the rate of filtration in different parts of the filter must occur. The heavy frictional resistance of the underdrains when more than the proper quantity of water passes them tends to correct this tendency especially for the more remote parts of the filter, but perhaps at the expense of those near to the main drain.
The filter is not covered as the suggestions in Chapter II would require, but this is hardly on account of its being an intermittent filter.
The annual report of the Massachusetts State Board of Health for 1893 states that during the first half of December, 1893, the surface remained covered, that is, it was used continuously, and after December 16th it was so used when the temperature was below 24°, and was drained only when the temperature was 24° or above. The days on which the filter was drained during the remainder of December are not given, but during January and February, 1894, the filter remained covered 29 days and was drained 30 days. Bacterial samples were taken on 44 of these days, 22 days when it was drained and 22 when it was not. The average number of bacteria on the days when it was not drained was 137 and on those days when it was drained 252 per cubic centimeter.
From February 24th to March 12th the number of bacteria were unusually high, averaging 492 per cubic centimeter, or 5.28 per cent of the 9308 applied. During this period the filter was used intermittently; there was ice upon it, and parts of the surface[Pg 102] were scraped under the ice, and high rates of filtration undoubtedly resulted on the scraped areas. After March 12th the ice had disappeared and very much better results were obtained.
While there may be some question as to the direct cause of this decreased efficiency with continued cold weather and ice, the results certainly are not such as to show the advisability of building open filters in the Lawrence climate.
The cost of building the filter in comparison with European filters was extraordinarily low—only $67,000, or $27,000 per acre of filter surface. To have constructed open continuous filters of the same area with water-tight bottoms, divided into sections with separate drains and regulating apparatus, with the necessary piping, would have cost at least half as much more, and with the masonry cover which I regard as most desirable in the Lawrence climate the cost would have been two or three times the expenditure actually required.
It was no easy matter to secure the consent of the city government to the expenditure of even the sum used; there was much skepticism as to the process of filtration in general, and it was said that mechanical filters could be put in for about the same cost. Insisting upon the more complete and expensive form might have resulted either in an indefinite postponement of action, or in the adoption of an inferior and entirely inadequate process. Still I feel strongly that in the end the greater expense would have proved an excellent investment in securing softer water and in the greater facility and security of operating the filter in winter.
In regard to the effect of the Lawrence filter upon the health of the city, I can best quote from Mr. Mills’ paper in the Report of the Massachusetts State Board of Health for 1893, and also published in the Journal of the New England Water-works Association. Mr. Mills says: “In the following diagram [Fig. 15] the average number of deaths from typhoid fever at Lawrence[Pg 103] for each month from October to May, in the preceding five years, are given by the heavy dotted line; and the number during the past eight months are given by the heavy full line.
“The total number for eight months in past years has been forty-three, and in the present year seventeen, making a saving of twenty-six. Of the seventeen who died nine were operatives in the mills, each of whom was known to have drunk unfiltered canal water, which is used in the factories at the sinks for washing.
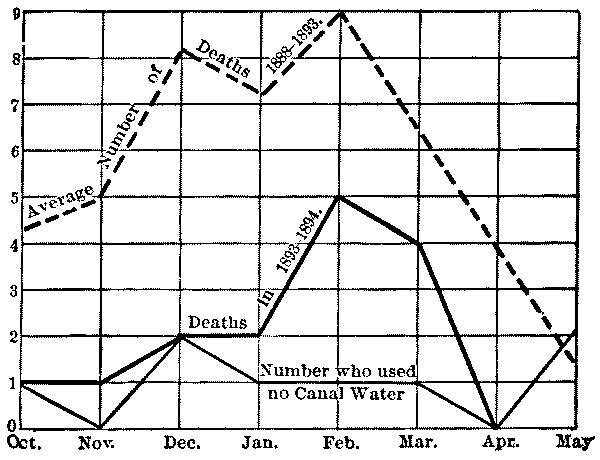
Fig. 15.—Typhoid Fever in Lawrence.
“The finer full line shows the number of those who died month after month who are not known to have used the poisoned canal water. The whole number in the eight months is eight.
“It is evident from the previous diagram [not reproduced] that the numbers above the fine full line, here, follow after those at Lowell in the usual time, and were undoubtedly caused by the sickness at Lowell; but we have satisfactory reason to conclude that the disease was not propagated through the filter but that the germs were conveyed directly into the canals and to those who drank of the unfiltered canal water. Among the operatives[Pg 104] of one of the large corporations not using the canal water there was not a case of typhoid fever during this period. Warnings have been placed in the mills where canal water is used to prevent the operatives from drinking it.
“We find, then, that the mortality from typhoid fever has, during the use of the filter, been reduced to 40 per cent of the former mortality, and that the cases forming nearly one half of this 40 per cent were undoubtedly due to the continued use of unfiltered river water drawn from the canals.”
The records of typhoid fever in Lawrence before and after the introduction of filters are as follows:
| DEATHS FROM TYPHOID FEVER IN LAWRENCE, 1888-98. | ||||
|---|---|---|---|---|
| Years. | Total Number of Deaths. |
Deaths per 10,000 of Population. |
Persons who are known to have been exposed to infection. |
|
| By drinking Canal Water. |
While living out of town just before falling sick in Lawrence. |
|||
| 1888 | 48 | 11.36 | ||
| 1889 | 55 | 12.66 | ||
| 1890 | 60 | 13.44 | ||
| 1891 | 55 | 11.94 | ||
| 1892 | 50 | 10.52 | ||
| 1893 | 39 | 7.96 | ||
| 1894 | 24 | 4.75 | 12 | |
| 1895 | 16 | 3.07 | 9 | 2 |
| 1896 | 10 | 1.86 | 2 | 4 |
| 1897 | 9 | 1.62 | ||
| 1898 | 8 | 1.39 | 1 | |
Filter put in operation September, 1893. |
||||
Average rate before the introduction of filtered water (1888-92) |
11.31 | |||
Average rate afterward (1894-98) |
2.54 | |||
These results show a striking reduction in the deaths from typhoid fever with the introduction of filtered water, which has been most gratifying in every way.
The more recent history of the underdrains of the Lawrence filter is particularly instructive. Owing to the absence of a water-tight bottom to the filter, and its low position, a certain amount of water constantly entered the filter from the ground below.[Pg 105] This water contained iron in solution as ferrous carbonate. When this water came in contact with the filtered water in the gravel and underdrains, the iron was oxidized by the dissolved oxygen carried in the filtered water and precipitated. This was accompanied by a growth of crenothrix in the gravel and underdrains, which gradually reduced their carrying capacity. This reduction in carrying capacity first became apparent in cold weather when the yield from the filter was less free than formerly. There was difficulty in maintaining the supply during the winter of 1896-7 and more difficulty in the following winter.
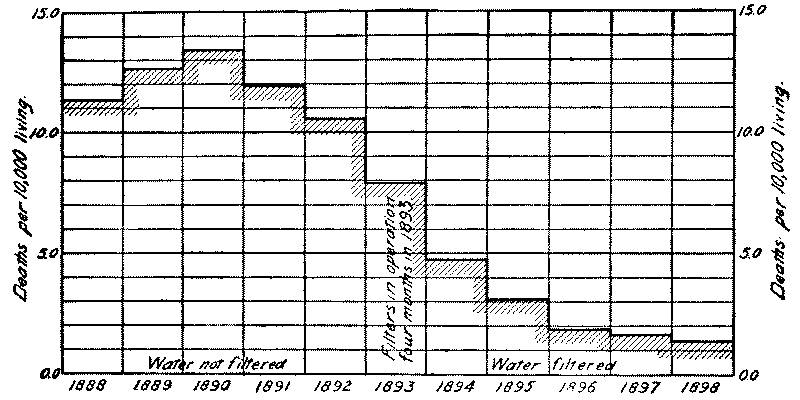
Fig. 16.—Typhoid Fever in Lawrence, 1888 to 1898.
The sand of the filter was as capable of filtering the full supply of water as it ever had been, and the efficiency was as good; but the underdrains were no longer able to collect the filtered water and deliver it. As the filtering area was ample for the supply, it was desired to avoid construction of additional filtering area. The underdrains were dug up and cleaned during the periods when the filter was drained. As the filter is all in one bed, the times when the filter could be allowed to remain drained, and when the work could proceed, were limited. Great care was taken to leave the work in good condition, and free from passages, at the end of each day’s work, but the numbers of bacteria in the[Pg 106] effluent nevertheless increased somewhat. Some weeks afterward the number of cases of typhoid fever in the city increased. The numbers did not become as high as they had been prior to the introduction of filtered water, but they were much higher than they had been since that time, and they pointed strongly to the disturbance of the underdrains as the cause of the increase.
The numbers of bacteria in the applied water and in the effluent from the Lawrence filter by months, from the time the filter was put in operation, compiled from the reports of the State Board of Health, as far as available, are as follows:
| BACTERIA IN WATER APPLIED TO AND EFFLUENT FROM LAWRENCE FILTER. | ||||||
|---|---|---|---|---|---|---|
| RAW WATER. | ||||||
| 1893. | 1894. | 1895. | 1896. | 1897. | 1898. | |
| January | 7,700 | 18,700 | 7,500 | 13,314 | 6,519 | |
| February | 7,600 | 15,040 | 12,600 | 13,113 | 4,653 | |
| March | 6,500 | 20,770 | 5,900 | 12,055 | 3,748 | |
| April | 11,200 | 8,420 | 3,800 | 6,904 | 2,320 | |
| May | 6,000 | 7,000 | 9,600 | 4,625 | 2,050 | |
| June | 8,300 | 9,000 | 6,400 | 4,650 | 6,775 | |
| July | 2,400 | 10,000 | 3,900 | 6,240 | 2,840 | |
| August | 3,100 | 5,000 | 2,700 | 10,700 | 8,575 | |
| September | 57,500 | 6,500 | 5,000 | 12,300 | 27,300 | 6,100 |
| October | 22,200 | 25,300 | 19,000 | 5,300 | 13,200 | 5,120 |
| November | 10,800 | 16,600 | 8,700 | 5,600 | 6,644 | 4,310 |
| December | 8,100 | 23,800 | 6,700 | 9,695 | 5,581 | 5,200 |
| Average | 24,650 | 10,417 | 11,111 | 7,108 | 10,360 | 4,850 |
| EFFLUENT. | ||||||
| January | 129 | 206 | 166 | 91 | 39 | |
| February | 244 | 283 | 315 | 79 | 45 | |
| March | 455 | 405 | 133 | 67 | 34 | |
| April | 281 | 84 | 40 | 47 | 21 | |
| May | 134 | 68 | 56 | 35 | 48 | |
| June | 110 | 68 | 22 | 56 | 50 | |
| July | 25 | 50 | 39 | 106 | 22 | |
| August | 36 | 38 | 146 | 72 | 28 | |
| September | 6,850 | 42 | 40 | 37 | 98 | 67 |
| October | 1,216 | 116 | 60 | 30 | 33 | 28 |
| November | 161 | 175 | 64 | 37 | 27 | 122 |
| December | 111 | 364 | 84 | 67 | 24 | |
| Average | 2,084 | 176 | 121 | 91 | 61 | 46 |
| Average efficiency | 91.55 | 98.31 | 98.91 | 98.72 | 99.41 | 98.95 |
[Pg 107]
CHEMNITZ WATER-WORKS.
The only other place which I have found where anything approaching intermittent filtration of water is systematically employed is Chemnitz, Germany. The method there used bears the same relation to intermittent filtration as does broad irrigation of sewage to the corresponding method of sewage treatment; that is, the principles involved are mainly the same, but a much larger filtering area is used, and the processes take place at a lower rate and under less close control.
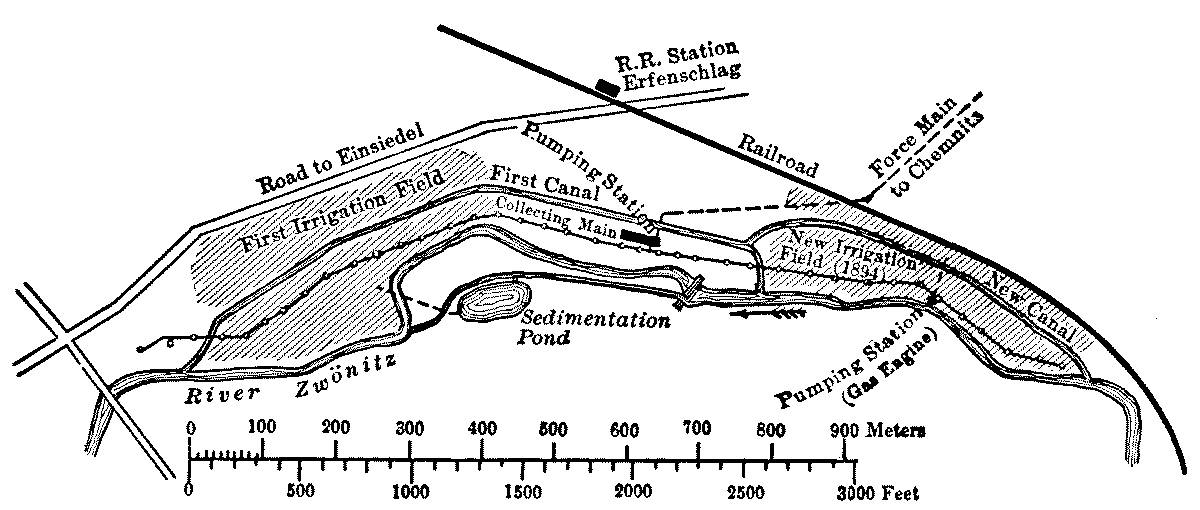
Fig. 17.—Plan of Area used for Intermittent Filtration at Chemnitz.
The water-works were built about twenty years ago by placing thirty-nine wells along the Zwönitz River, connected by siphon pipes, with a pumping-station which forced the water to an elevated reservoir near the city (Fig. 17). The wells are built of masonry, 5 or 6 feet in diameter and 10 or 12 feet deep, and are on the rather low bank of the river. The material, with the exception of the surface soil, and loam about 3 feet deep, is a somewhat mixed gravel with an effective size of probably from 0.25 to 0.50 mm., so that water is able to pass through it freely. The wells are, on an average, about 120 feet apart, and the line is seven eighths of a mile long.
It was found that in dry times the ground-water level in the[Pg 108] entire neighborhood was lowered some feet below the level of the river without either furnishing water enough or stopping the flow of the river below. The channel of the river was so silted that, notwithstanding the porous material, the water could not penetrate it to go toward the wells.
A dam was now built across the river near the pumping-station, and a canal was dug from above the dam, crossing the line of wells and running parallel to it on the back side for about half a mile. Later a similar canal was dug back of the remaining upper wells. Owing to the difference in level in the river above and below, the canals can be emptied and filled at pleasure. They are built with carefully prepared sand bottoms, and the sand sides are protected by an open paving, to allow the percolation of as much water as possible, and the sand is cleaned by scraping, as is usual with ordinary sand filters, once a year or oftener.
The yield from the wells was much increased by these canals, but the water of the river is polluted to an extent which would ordinarily quite prevent even the thought of its being used for water-supply, and it was found that the water going into the ground from the canals, and passing through the always saturated gravel to the wells, without coming in contact with air at any point, after a time contained iron and had an objectionable odor.
To avoid this disagreeable result the meadow below the pumping-station was laid out as an irrigation field (Fig. 16). The water from above the dam was taken by a canal on the opposite side of the river through a sedimentation pond (which, however, is not now believed to be necessary and is not always used), and then under the river by a siphon to a slightly elevated point on the meadow, from which it is distributed by a system of open ditches, exactly as in sewage irrigation. The area irrigated is not exactly defined and varies somewhat from time to time; the rate of filtration may be roughly estimated[Pg 109] at from 100,000 to 150,000 gallons per acre daily, although limited portions may occasionally get five times these quantities for a single day. The water passes through the three feet of soil and loam, and afterward through an average of six feet of drained coarse sand or gravel in which it meets air, and afterward filters laterally through the saturated gravel to the wells. The water so obtained is invariably of good quality in every way, colorless, free from odor and from bacteria. The surface of the irrigated land is covered with grass and has fruit-trees (mostly apple) at intervals over its entire area.
This first system of irrigation is entirely by gravity. On account of natural limits to the land it could not be conveniently extended at this point, and to secure more area, the higher land above the pumping-station was being made into an irrigation field in 1894. This is too high to be flooded by gravity, and will be used only for short periods in extremely dry weather. The water is elevated the few feet necessary by a gas-engine on the river-bank. In times of wet weather enough water is obtained from the wells without irrigation, and the land is only irrigated when the ground-water level is too low.
During December, January, and February irrigation is usually impossible on account of temperature, and the canals are then used, keeping them filled with water so that freezing to the bottom is impossible; but trouble with bad odors in the filtered water drawn from the wells is experienced at these times.
The drainage area of the Zwönitz River is only about 44 square miles, and upon it are a large number of villages and factories, so that the water is excessively polluted. The water in the wells, however, whether coming from natural sources, or from irrigation, or from the canals, has never had as many as 100 bacteria per cubic centimeter, and is regarded as entirely wholesome.
In extremely dry weather the river, even when it is all used for irrigation so that hardly any flows away below, cannot be[Pg 110] made to supply the necessary daily quantity of 2,650,000 gallons, and to supply the deficiency at such times, as well as to avoid the use of the canals in winter, a storage reservoir holding 95,000,000 gallons has recently been built on a feeder of the river. This water, which is from an uninhabited drainage area, is filtered through ordinary continuous filters and flows to the city by gravity. Owing to the small area of the watershed it is incapable of supplying more than a fraction of the water for the city, and will be used to supplement the older works.
This Chemnitz plant is of especial interest as showing the successful utilization of a river-water so grossly polluted as to be incapable of treatment by the ordinary methods. Results obtained at the Lawrence Experiment Station have shown that sewage is incapable of being purified by continuous filtration, the action of air being essential for a satisfactory result. With ordinary waters only moderately polluted this is not so; for they carry enough dissolved air to effect their own purification. In Chemnitz, however, as shown by the results with the canals, the pollution is so great that continuous filtration is inadequate to purify the water, and the intermittent filtration adopted is the only method likely to yield satisfactory results in such cases.
Intermittent filtration is now being adopted for purifying brooks draining certain villages and discharging into the ponds or reservoirs from which Boston draws its water-supply. The water of Pegan Brook below Natick has been so filtered since 1893 with most satisfactory results, and affords almost absolute protection to Boston from any infection which might otherwise enter the water from that town. A similar treatment is soon to be given to a brook draining the city of Marlborough. The sewage from these places is not discharged into the brooks, but is otherwise provided for, but nevertheless they receive many polluting matters from the houses and streets upon their banks.
The filtration used resembles in a measure that at Chemnitz,[Pg 111] and I am informed by the engineer, Mr. Desmond FitzGerald, that it was adopted on account of its convenience for this particular problem, and not because he attaches any special virtue to the intermittent feature.
APPLICATION OF INTERMITTENT FILTRATION.
In regard to the use of waters as grossly polluted as the Zwönitz, the tendency is strongly to avoid their use, no matter how complete the process of purification may be; but in case it should be deemed necessary to use so impure a water for a public supply, intermittent filtration is the only process known which would adequately purify it. And it should be used at comparatively low rates of filtration. I believe that an attempt to filter the Zwönitz at the rate used for the Merrimac water at Lawrence, which is by comparison but slightly polluted, would result disastrously.
The operation in winter must also be considered. Intermittent filtration of sewage on open fields in Massachusetts winters is only possible because of the comparatively high temperature of the sewage (usually 40° to 50°), and would be a dismal failure with sewage at the freezing-point, the temperature to be expected in river-waters in winter.
It is impossible to draw a sharp line between those waters which are so badly polluted as to require intermittent filtration for their treatment and those which are susceptible to the ordinary continuous filtration. Examples of river-waters polluted probably beyond the limits reached in any American waters used for drinking purposes and successfully filtered with continuous filters are furnished by Altona, Breslau, and London.
Intermittent filtration may be considered in those cases where it is proposed to use a water polluted entirely beyond the ordinary limits, and for waters containing large quantities of decomposable organic matters and microscopical organisms; but in those cases where a certain and expeditious removal of mud is[Pg 112] desired, and where waters are only moderately polluted by sewage, but still in their raw state are unhealthy, it is not apparent that intermittent filtration has any advantages commensurate with the disadvantages of increased rate to produce the same total yield and of the increased difficulty of operation, particularly in winter; and in such cases continuous filtration is to be preferred.
In the removal of tastes and odors from pond or reservoir waters which are not muddy, but which are subject to the growths of low forms of plants, which either by their growth or decomposition impart to the water disagreeable tastes and odors, intermittent filtration may have a distinct advantage. In such cases there is often an excess of organic matter to be disposed of by oxidation, and the additional aeration secured by intermittent filtration is of substantial assistance in disposing of these matters.
[Pg 113]
CHAPTER VIII.
TURBIDITY AND COLOR, AND THE EFFECT OF MUD UPON SAND-FILTERS.
The ideal water in appearance is distilled water, which is perfectly clear and limpid, and has a slight blue color. When other waters are compared with it, the divergences in color from the color of distilled water are measured, and not the absolute colors of the waters. Many spring waters and filtered waters are indistinguishable in appearance from distilled water.
Public water-supplies from surface sources contain two substances or classes of substances which injure their appearance, namely, peaty coloring matters, and mud. Waters discolored by peaty matters are most common in New England and in certain parts of the Northwest, while muddy waters are found almost everywhere, but of different degrees of muddiness, according to the physical conditions of the water-sheds from which they are obtained.
Muddy waters are often spoken of as colored waters, and in a sense this is correct where the mud consists of clays or other materials having distinct colors; but it is more convenient to classify impurities of this kind as turbidities only, and to limit the term colored waters to those waters containing in solution vegetable matters which color them.
The removal of either color or turbidity may be called clarification.
Colored waters are usually drawn from water-sheds where the underlying rock is hard and does not rapidly disintegrate, and where the soils are firm and sandy, and especially from swamps. The water here comes in contact with peat or muck, which colors[Pg 114] it, but is so firm as not to be washed by flood flows, and so does not cause turbidity.
Large parts of the United States have for rock foundations shales or other soft materials which readily disintegrate when exposed, and which form clayey soils readily washed by hard rains. Waters from such watersheds are generally turbid and very rarely colored. In fact a water carrying much clay in suspension is usually found colorless when the clay is removed, even if it were originally colored. It thus happens that waters which are colored and turbid at the same time hardly exist in nature.
Color-producing matters and turbidity-producing matters are different in their natures, and the methods which must be used to remove them are different.
THE MEASUREMENT OF COLOR.
The colors of waters are measured and recorded by comparing them with colors of solutions or substances which are permanent, or which can be reproduced at will. One of the earliest methods of measuring colors of waters was to compare them with the colors of the Nessler standards used for the estimation of ammonia in water analysis. The Nessler standards were similar in appearance to yellow waters, and their colors depended upon the amounts of ammonia which had been used in preparing them, and a record was made of the standard which most closely resembled the water under examination.
The method was open to the serious objections that the hues of the standards did not match closely the hues of the waters; that the colors produced with different lots of Nessler reagent differed considerably, and therefore the exact values of results were more or less uncertain; and further, that the numbers obtained for color were not even approximately proportional to the amounts of coloring matter present. Because of this peculiarity, in filtration the percentage of color removal, as determined by the use of these[Pg 115] standards, is not even approximately correct, but is much above the truth.
In the Lovibond tintometer, which has been extensively used in England, the standards of color are based upon the colors of certain glass slips, which are in turn compared with standard originals kept for that purpose. This process answers quite well, but is open to some objections because of possible uncertainties in the standardization of the units.
Another method of measuring colors is to compare them with dilute solutions of platinum and cobalt. The ratio of cobalt to platinum can be varied to make the hue correspond very closely with the hues of natural waters, and the amount of platinum required to match a water affords a measure of its color, one part of metallic platinum in 10,000 parts of water forming the unit of color.
This standard has the advantages that it can be readily prepared with absolute accuracy in any laboratory, and that by varying the ratio of platinum to cobalt the hues of various waters can be most perfectly matched. It is important that the observations should not be made in too great a depth, as the discrepancy in hues increases much more rapidly than the depth of color.
For further information regarding colors the reader is referred to articles in the American Chemical Journal, 1892, vol. xiv, page 300; Journal of the American Chemical Society, vol. ii, page 8; vol. xviii, 1896, pp. 68, 264, and 484; Journal of the Franklin Institute, Dec. 1894, p. 402; Journal of the New England Water Works Association, vol. xiii, 1898, p. 94.
AMOUNT OF COLOR IN AMERICAN WATERS.
New England surface-waters have colors ranging from almost nothing up to 2.00. The colors of the public water-supplies of Massachusetts cities have been recorded in the reports of the State Board of Health for some ten years. The figures given were[Pg 117] recorded first upon the Nessler standard, and afterwards upon a modification of the same, known as the natural water standard. The figures given are approximately equal to those for the platinum color standard, the relations between the two having been frequently determined by various observers and published in the above-mentioned papers. The accompanying diagram shows the colors in several Massachusetts supplies, as plotted from the figures given in the published reports.
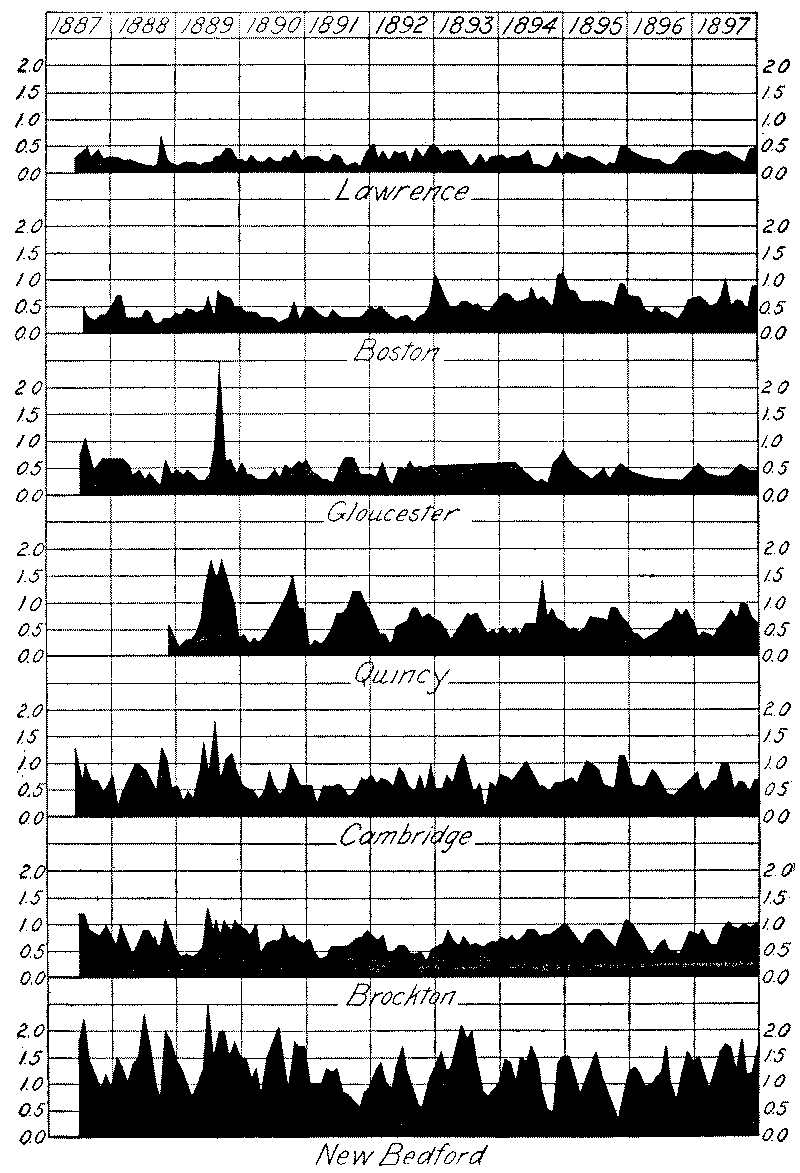
Fig. 18.—Colors of Waters.
(Analyses of the Mass. State Board of Health.)
In Connecticut also the colors of many public water-supplies have been recorded in the reports of the State Board of Health on the platinum color-standard.
The waters of the Middle States, with rare exceptions, are almost free from color. In the Northwest waters are obtained often with very high colors, even considerably higher than the New England waters, and some of the Southern swamps also yield highly colored waters.
REMOVAL OF COLOR.
Peaty coloring-matter is almost perfectly in solution, and only a portion of it is capable of being removed by any form of simple filtration. In order to remove the coloring-matter it is necessary to change it chemically, or to bring it into contact with some substance capable of absorbing it. For this reason sand filtration with ordinary sands, having no absorptive power for color, commonly removes only from one fourth to one third of the color of the raw water.
MEASUREMENT OF TURBIDITY.
The amount of mud or turbidity in a water is often expressed as the weight of the suspended matters in a given weight of the water. Most of the data relating to turbidities of waters are stated in this way, because this was the only method recognized by the earlier investigators.
[Pg 118]
This method of statement has some disadvantages: it fails to take into account the different sizes of particles which are carried in suspension by different waters, and at different times. Thus the Merrimac River in a great flood may carry 100 parts in 100,000 of fine sand in suspension, and still it could hardly be called muddy; while another stream carrying only a fraction of this amount of fine clay would be extremely muddy. Further, an accurate determination of suspended matters is a very troublesome and tedious operation, and cannot be undertaken as frequently as is necessary for an adequate study of the mud question.
Turbidity is principally important as it affects the appearance of water, and it would seem that optical rather than gravimetric methods should be used for its determination. Various optical methods of measuring turbidity have been proposed. The general method employed is to measure the thickness of the layer of water through which some object can be seen under definite conditions of lighting. The most accurate results can probably be obtained in closed receptacles and with artificial light. Such a method has been used by Mr. G. W. Fuller at Louisville and Cincinnati in connection with his experiments, and is described by Parmelee and Ellms in the Technology Quarterly for June, 1899. This apparatus is called by Mr. Fuller a diaphanometer.
At the Lawrence Experiment Station of the Massachusetts State Board of Health as early as 1889 it became necessary to express the turbidities of various waters approximately, and the very simple device of sticking a pin into a stick, and pushing it down into the water under examination as far as it could be seen, was adopted. Afterwards a platinum wire 0.04 of an inch in diameter was substituted for the pin, and the stick was graduated so that the turbidities could be read from it directly. The figures on the stick were inversely proportional to their distances from the wire. When the wire could be seen one inch below the surface, the turbidity was reported as 1.00; when the wire could be seen two inches, the turbidity was 0.50, and when it could be seen ten[Pg 119] inches the turbidity was 0.10, etc. This scale is much more convenient than a scale showing the depth at which the wire can be seen; and within certain limits the figures obtained with it are directly proportional to the amount of the elements which obstruct light in the water. Thus, if a water having a turbidity of 1.00 is mixed with an equal volume of clear water, the mixture will have a turbidity of 0.50. Advantage is taken of this fact for the measurement of turbidities so great that they cannot be accurately determined by direct observation. For turbidities much above 1.00 it is very difficult to read the depth of wire with sufficient accuracy, and such waters are diluted with one, two, or more times their volume of clear water in a pail or other receptacle, the turbidity of the diluted water is taken, and multiplied by the appropriate factor.
For the greatest accuracy it is necessary that the observations should be taken in the open air and not under a roof. They should preferably be made in the middle of the day when the light is strongest, and in case the sun is shining, the wire must be kept in shadow and not in direct sunlight.
The turbidities of effluents are usually so slight that they cannot be taken in this manner; in fact, turbidities of less than 0.02, with the wire visible 50 inches below the surface, cannot be conveniently read in this way. For the estimation of lower turbidities a water is taken having a turbidity of 0.03 or 0.04 and as free as possible from large suspended particles. The turbidity of this water is measured by a platinum wire in the usual way, and the water is then diluted with clear water to make standards for the lower turbidities.
The comparisons between standards and waters are best made in bottles of perfectly clear glass, holding at least a gallon, and the comparison is facilitated by surrounding the bottles with black cloth except at the point of observation, and lighting the water by electric lights so arranged that the light passes through the water but is hidden from the observer. In case the water under[Pg 120] examination is colored, the comparison is rendered difficult, and it is often advisable to add a small amount of methyl orange to the standards to make the colors equal.
Instead of diluting a water of known turbidity for the standards, a standard can be made by precipitating a known amount of silver chloride in the water. For this purpose about one per cent of common salt is dissolved in clear water and small measured amounts of silver nitrate added, until the turbidity produced is equal to that of the water under examination. The relation of the amount of silver nitrate used to the turbidity is entirely arbitrary, and is established by comparisons of standards made in this way with waters having turbidities from 0.02 to 0.04, the turbidities of which are measured with the platinum wire, and which afterwards serve to rate the standards. The silver chloride has a slight color, which is an objection to its use, and perhaps some other substance could be substituted for it with advantage. The standards have to be made freshly each day.
One disadvantage of the platinum-wire method of observing turbidities in the open air, as compared with the diaphanometric method using artificial light, is that observations cannot be made in the night. To get the general character of the water in a stream, daily observations taken about noon will generally be sufficient; but for some purposes it is important to know the turbidity at different hours of the day, and in such cases the platinum-wire method is at a distinct disadvantage. Variations in the amount of light, within reasonable limits, do not affect the results materially, although extreme variations are to be avoided. The size of the wire also influences the results somewhat. The wire commonly used is 0.04 of an inch or one millimeter in diameter. A wire only four tenths of this size in some experiments at Pittsburg gave results 25 per cent higher; with a wire twice as large the results were lower, but the differences were much less. Wire 0.04 of an inch in diameter was adopted as being very well adapted to rather turbid river-waters. For very[Pg 121] clear lake or reservoir waters, usually transparent to a great depth, a much larger object is preferable. Within certain limits the results obtained with an object of any size can be converted into corresponding figures for another object, or another light, by the use of a constant factor. Thus the turbidities obtained with a platinum wire always have approximately the same ratio to the turbidities of the same waters determined by the diaphanometer.
The platinum-wire method has been used in many cases with most satisfactory results. If it lacks something in theoretical accuracy as compared with more elaborate methods, it more than makes up for it by its simplicity; and reliable observations can be taken with it by people who would be entirely incompetent to operate more elaborate apparatus; and it can thus be used in many cases where other methods would be impossible.
Upon this scale the most turbid waters which have come under the observation of the author have turbidities of about 2.50, although waters much more turbid than this undoubtedly exist. A water with a turbidity of 1.00 is extremely muddy, and only one tenth of this turbidity would cause remark and complaint among those who use it for domestic purposes. In an ordinary pressed-glass tumbler a turbidity of 0.02 is just visible to an ordinary observer who looks at the water closely, but it is not conspicuous, nor would it be likely to cause general complaint; and this amount may be taken as approximately the allowable limit of turbidity in a good public water-supply. In a carefully polished, and perfectly transparent glass a turbidity of 0.01 will be visible, and in larger receptacles still lower turbidities may be seen if the water is examined carefully. In gallon bottles of very clear glass, under electric light and surrounded by black cloth, a turbidity of 0.001 can be distinguished, but a turbidity even several times as large as this could hardly be detected except by the use of special appliances, or where water is seen in a depth of several feet.
[Pg 122]
RELATION OF PLATINUM-WIRE TURBIDITIES TO SUSPENDED MATTERS.
The relation of turbidity to the weight of suspended matters is approximately constant for waters from which the coarser matters have been entirely removed by sedimentation. For these waters the suspended matters in parts per 100,000 are about 16 times the turbidity. For river-waters the ratios are always larger. With very sluggish rivers the ratio is only a little larger than for settled waters. For average river-waters the ratio is considerably higher, and increases with the turbidity, and for very rapid rivers and torrents the ratio is much wider, as the suspended matters consist largely of particles which are heavy but do not increase very much the turbidity.
The following table gives the amounts of suspended matters for various classes of waters corresponding to the turbidities stated, which have been deduced from the experience of the author. It is very likely that ratios different from the above would be obtained with waters in which the sediment was of different character.
| Turbidity, Platinum-wire Standard. |
Suspended Matters: Parts in 100,000. | |||
|---|---|---|---|---|
| Settled Waters. |
River Waters, Finest Sediment. |
River Waters, Average Sediment. |
River Waters, Coarsest Sediment. |
|
| 0.01 | 0.16 | |||
| 0.05 | 0.80 | 0.85 | 1.30 | 2.40 |
| 0.10 | 1.60 | 1.75 | 2.60 | 4.90 |
| 0.20 | 3.20 | 3.60 | 5.50 | 10.00 |
| 0.30 | 4.80 | 5.70 | 8.50 | 15.00 |
| 0.40 | 6.40 | 7.80 | 11.60 | 21.00 |
| 0.50 | 8.00 | 10.00 | 15.00 | 26.00 |
| 1.00 | 16.00 | 23.00 | 36.00 | 59.00 |
| 1.50 | 24.00 | 40.00 | 62.00 | 97.00 |
| 2.00 | 32.00 | 61.00 | 94.00 | 140.00 |
| 3.00 | 48.00 | 110.00 | 175.00 | 250.00 |
[Pg 123]
SOURCE OF TURBIDITY.
Much turbidity originates in plowed fields of clayey soil, or in fields upon which crops are growing. If it has not rained for some days, and the surface-soil is comparatively dry, the first rain that falls upon such land is absorbed by the pores of the soil until they are filled. If the rain is not heavy, but little runs off over the surface. If, however, the rain continues rapidly after the surface-soil is saturated, the excess runs off over the surface to the nearest watercourse. The impact of the rain-drops upon the soil loosens the particles, and the water flowing off carries some of them in suspension, and the water is said to be muddy.
The particles carried off in this way are extremely small. Mr. George W. Fuller, in his report upon water purification at Louisville, estimates that many of them are not more than a hundred thousandth of an inch in diameter, and not more than a tenth as large as common water bacteria.
The turbidity of the water flowing from a field of loose soil may be 2.00 or more; that is to say, the wire is hidden by a depth of half an inch of water or less. When the water reaches the nearest watercourse it meets with water from other kinds of land, such as woodlands and grassed fields, and these waters are less turbid. The water in the first little watercourse is thus a mixture and has a turbidity of perhaps 1.00.
The conditions which control the turbidity of any brook are numerous and complicated. The turbidity of a stream receiving various brooks depends upon the turbidities of all the waters coming into it. Generally speaking, the turbidity of a river depends directly upon the turbidities of its feeders, and is not affected materially by erosion of its bed or by sedimentation in it. There are, of course, some streams which in times of great floods cut their banks, and all streams pick up and move about from place to place more or less of the sand and other coarse[Pg 124] materials upon their bottoms. The materials thus moved, however, have but little influence upon the turbidity.
After the rain is over some of the water held by the soil will find its way to the watercourses by underground channels, and will prevent the stream from drying up between rains, but the average volume of the stream-flows between rains will be much less than the volumes during the rains when the water is most turbid.
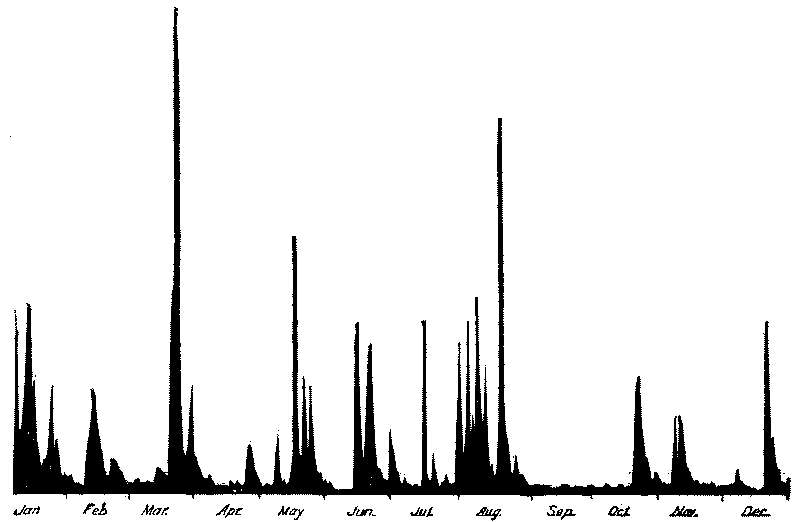
Fig. 19.—Fluctuations in Turbidity of the Water of the Allegheny River at Pittsburg during 1898.
These conditions are well illustrated by a few data upon the turbidity of three Pennsylvania streams, recently collected by the author. One of these streams is a small brook having a drainage area of less than three square miles. The observations extended over a period of 47 days. During this time there were five floods, or an average of one flood in ten days. The duration of floods was less than twenty-four hours in each case. Selecting the days when the turbidity was the highest, to the number of one tenth[Pg 125] of the whole number of days, the sum of the turbidities for these days was 67 per cent of the aggregate turbidities for the whole period. That is to say, 67 per cent of the whole amount of mud was in the water of only a tenth of the days; the water of the other nine tenths of the days contained only 33 per cent of the whole amount of turbidity. The average turbidity of the water for the flood days was eighteen times as great as the average turbidity for the remaining days.
The next stream is a considerable creek having a drainage area of 350 square miles. The observations extended over 117 days, during which time there were seven floods, or an average of one flood in 19 days. The floods lasted in each case one or two days, and the sum of the turbidities for the one tenth of the whole number of days when the water was muddiest was 55 per cent of the aggregate of all the turbidities for the period.
The last case is that of a large river, with a drainage area of over 11,000 square miles. The observations extended over a full year. In this period there were sixteen floods, each lasting from one to six days, and the sum of the turbidities for the one tenth of the whole number of days when the water was muddiest is 45 per cent of the aggregate turbidities for the year. The floods occurred on an average of once in 22 days, and the average duration was two and one half days.
The results are very striking as showing that a very large proportion of the mud is carried by the water in flood flows of comparatively short duration. They also show that in small streams the proportion of mud in the flood-flows is greater, and the average duration of floods is shorter, than in larger streams. In other words, the differences between flood- and low-water flows are greatest in small streams, and gradually become less as the size of the stream increases.
When a stream is used for water-works purposes in the usual way, a certain quantity of water is taken from the stream each day, which quantity is nearly constant, and is not dependent upon[Pg 126] the condition of the stream, or the volume of its flow. The proportions of the total flows taken at high- and low-water stages are very different, and it thus happens that the average quality of the water taken for water-works purposes is different from the average quality of all the water flowing in the stream.
Let us assume, for example, a stream having a watershed of such a size that in times of moderate floods water from the most distant points reaches the water-works intake in twenty-four hours. Let us assume further that rainfalls of sufficient intensity to cause floods and muddy water occur, on an average, once in ten days, and that the turbidity of the water at these times reaches 1.00, and that for the rest of the time the turbidity averages 0.10. Let us assume further that at times of storms the average flow of the stream is 100 units of volume, and for the nine days between storms the average flow is 10 units of volume. We shall then have in a ten days’ period, for one day, 100 volumes of water with a turbidity of 1.00, and nine days with 10 volumes each, or a total of 90 volumes of water with a turbidity of 0.10. The total discharge of the stream will then be 190 volumes, and the average turbidity 0.57. The turbidity of 0.57 represents the average turbidity all the water flowing in the stream, or, in other words, the turbidity which would be found in a lake if all the water for ten days should flow into it and become thoroughly mixed without other change.
Now let us compute the average turbidity of the water taken from the stream for water-works purposes. The water-works require, let us say, one volume each day, and we have for the first day water with a turbidity of 1.00, and then for nine days water with a turbidity of 0.10. The average turbidity of the water taken by the water-works for the period is thus only 0.19 in place of 0.57, the average turbidity of the whole run-off.
The average turbidity of all the water flowing in the stream is thus three times as great as that of the water taken from the stream for water-works purposes.
[Pg 127]
It is often noted that with long streams the water becomes muddier farther down, and it may naturally be thought that it is because of the added erosion of the stream upon its bed in its longer course. This, of course, may be a cause, or the lower tributaries may be muddier than the upper ones, but the fact that the water taken at the lower point is more muddy than farther up is not an indication of this.
Let us take, for example, a watershed of twice the size of that assumed above, that is, so long that 48 hours will be required for the water from the most remote feeders to reach the water-works intake. Let us divide this shed into two parts, which we will assume to be equal, one of which furnishes water reaching the intake within 24 hours, and the other water reaching the intake between 24 and 48 hours. Now suppose a storm upon the watershed producing turbidities equal to those just assumed for the smaller stream. On the first day the water from the lower half of the shed, namely, 100 volumes having a turbidity of 1.00, passes the intake, but this is mixed with 10 volumes of water from the upper half of the watershed, having a turbidity of 0.10, and the total flow is thus 110 volumes of water having a turbidity of 0.92. On the second day the water from the lower half of the watershed has returned to its normal condition, and the flood-flow of the upper half of the watershed, 100 volumes with a turbidity of 1.00, is passing, and mingles with the 10 volumes from the lower half with a turbidity of 0.10, and the total flow is again 110 volumes having a turbidity of 0.92. The following eight days, until the next rain, will have flows of 20 volumes each, with turbidities of 0.10. The average turbidity of all of the water flowing off is 0.57 as before, but the water taken for water-works purposes will consist of 2 volumes of water with turbidities of 0.92, and 8 volumes with turbidities of 0.10 making 10 volumes with an average turbidity of 0.26.
By doubling the length of the watershed we have thus doubled the length of time during which the water is turbid, and have[Pg 128] increased the average turbidity of the water taken for water-works purposes from 0.19 to 0.26, although the average turbidity of all the water running off remains exactly the same.
If now we assume a watershed so long that three days are required for the water from the most remote points to reach the intake, with computations as above, water taken for water-works purposes will have an average turbidity of 0.32; and with still longer watersheds this amount will increase, until with a watershed so long that ten days, or the interval between rains, are required for the water from the upper portions to reach the intake, the average turbidity of the water taken for water-works purposes will reach the average turbidity of the run-off, namely, 0.57.
In the above computations the numbers taken are round ones, and of course do not represent closely actual conditions. They do serve, however, to illustrate clearly the principle that the larger the watershed, other things being equal, the more muddy will be the water obtained from it for water-works purposes, and the longer will be the periods of muddy water, and the shorter the periods of clear water between them.
It cannot be too strongly emphasized that the period of duration of muddy water is, in general, dependent upon the length of time necessary for the muddy water to run out of the stream system after it is once in it, and be replaced by clear water; and that the settling out of the mud in the river has very little to do with it.
Muddy waters result principally from the action of rains upon the surface of ground capable of being washed, and the turbidities of the stream at any point below will occur at the times when the muddy waters reach it in the natural course of flow, and will disappear again when the muddy waters present in the stream system at the end of the rain have run out, and have been replaced with clear water from underground sources, or from clearer surface sources.
[Pg 129]
THE AMOUNTS OF SUSPENDED MATTERS IN WATER.
There is a large class of waters, including most lake and reservoir waters, and surface-waters from certain geological formations, which are almost free from suspended matters and turbidities. That is to say, the average turbidities are less than 0.10, and the average suspended matters are less than 2 parts in 100,000, and are often only small fractions of these figures. This class includes the raw waters of the supplies of many English cities drawn from impounding reservoirs, and also the waters of the rivers Thames and Lea at London, and the raw waters used by both of the Berlin water-works, and in the United States the waters of the great lakes except at special points near the mouths of rivers, nearly all New England waters, and many other waters along the Atlantic coast and elsewhere where the geological formations are favorable.
Data regarding the suspended matters in these waters are extremely meagre. The official examinations of the London waters contain no records of suspended matters, although the clearness of filtered waters is daily reported. Dibden, in his analytical investigations of the London water-supply, mentioned in his book upon “The Purification of Sewage and Water,” reports the average suspended matters in the water of the Thames near the water-works intakes as 0.77 part in 100,000. No figures are available for the raw waters used by the Berlin water-works, but both are taken from lakes, and are generally quite clear. Even in times of floods of the rivers feeding the lakes, the turbidities are not very high, because the gathering grounds for the waters are almost entirely of a sandy nature, yielding waters with low turbidities, and further, the streams flow through successions of lakes before finally reaching the lakes from which the waters are taken. It is safe to assume that the suspended matters and turbidities do not exceed those of the London waters. Even at times when somewhat turbid water is obtained, due to agitation by heavy winds, the suspended matter is mainly of a[Pg 130] sandy nature, readily removed by settling, and it does not seriously interfere with filtration.
The examinations of the Massachusetts State Board of Health, with a very few exceptions, contain no statements of suspended matters. This is due to the fact that the suspended matters, in most of the waters, are so small in amount as to make them hardly capable of determination by the ordinary gravimetric processes, and the determinations if made would have but little value. The Merrimac River at Lawrence, at the time of the greatest flood in fifty years, carried silt to the amount of about 111 parts in 100,000. This was for a very short time, and the suspended matter consisted almost entirely of sand, which deposited in banks, the deposited sand having an effective size of 0.04 or 0.05 millimeter. No clayey matter is ever carried in quantity by the river.
The reports of the Connecticut State Board of Health also contain no records of suspended matters for the same reason. It may be safely said that the average suspended matters of New England waters are almost always less than 1 part in 100,000.
Lake waters are generally almost entirely free from sediment. At Chicago the city water drawn from Lake Michigan has slightly more than 1 part in 100,000 of suspended matters, as determined by Professor Long in 1888-9, and by Professor Palmer in 1896. The suspended matter in this case is probably due to the nearness of the intake to the mouth of the Chicago River, and to mud brought up from the bottom in times of storms. The lake-water further away from the shore would probably give much lower results.
Turning now to waters having considerable turbidities, at Pittsburg the average suspended matters in the Allegheny River water, as shown by the weekly or semi-weekly analyses of the Filtration Commission during 1897-8, were 4 parts in 100,000. During a large part of the time the suspended matters were so small that it was not deemed worth while to determine them, and the results are returned as zero. This is not quite correct, and a recomputation of the amount of suspended matters, based on the[Pg 131] observed amounts, and the amounts calculated from the turbidities when they were very low, leads to an average of a little less than 5 parts in 100,000, which is probably more accurate than the direct average. The average turbidity on the platinum-wire scale was 0.16.
At Cincinnati the suspended matters are about 23 parts in 100,000, and at Louisville about 35 parts, both of these figures being from Mr. Fuller’s reports. In all these cases the enormous and rapid fluctuations in the turbidity of the water is a most striking feature of the results.
Observations on the Mississippi River above the Ohio have been made by Professor Long in 1888-9, and by Professor Palmer in 1896. These results are not as full and systematic as could be desired, but indicate averages of 20 to 30 parts in 100,000 at the different points. Professor William Ripley Nichols, in his work on water-supply, states the amount of suspended matter in the water of the Mississippi, probably referring to the lower river, as 66.66 parts.
Investigations of Professor Long and Professor Palmer for numerous interior Illinois streams extending over considerable periods give average results ranging from 1 to 8 parts in 100,000. The very much lower results for the interior streams as compared with the Mississippi and Ohio rivers may be due to the relative sizes and lengths of the streams, or in part to other causes.
Regarding muddy European rivers there are but few data. The Maas, used for the water-supply of Rotterdam, is reported by Professor Nichols as having from 1.40 to 47.61 and averaging 10 parts of suspended matters in 100,000. More recent information is to the effect that the raw water has at most 30 parts of suspended matters, and that that quantity is very seldom reached.
At Bremen the Weser often becomes quite turbid. The turbidity of the water is noted every day by taking the depth at which a black line on a white surface can be seen. Assuming that this procedure is equivalent to the platinum-wire procedure, the[Pg 132] depths at which the wire can be seen, namely, from 15 to 600 millimeters, correspond to turbidities of from 0.04 to 1.70, a result not very different from the conditions at Pittsburg.
At Hamburg and Altona the water is generally tolerably clear, but at times of flood the Elbe becomes very turbid, and the amount of mud deposited in the sedimentation-basins is considerable. At Dresden, several hundred miles up the river, I have repeatedly seen the river-water extremely turbid with clayey matter, the color of the clay varying from day to day, corresponding to the color of the earth from which it had been washed.
At Budapest, where filters were used temporarily, the Danube water was excessively muddy with clayey material. At first very high rates of filtration were employed and the results were not satisfactory. Afterward the rate of filtration was limited to 1.07 million gallons per acre daily, and good results were secured. There was no preliminary sedimentation. Professor Nichols reports the average suspended matters in the Danube at 32.68 parts in 100,000, but does not state at what place.
Many of the French and German rivers drain prairie country not different in its general aspect from the Mississippi basin, and the soil is probably in many places similar. There is no reason to suppose that the turbidities of these streams in general are materially different from those of corresponding streams in the United States, although it is true that, other things being equal, the average turbidity of water taken for water-works purposes will increase with the size of the stream; and it may be that some American streams, especially the Ohio, Missouri, and Mississippi rivers, are of larger size than European streams, and consequently that the turbidity of the water taken from them for water-works purposes may be greater.
The following are the drainage areas of a number of European and American streams yielding more or less muddy waters at points where they are used for public water-supplies after filtration, with a few other American points for comparison. The[Pg 133] results are obtained in most cases from measurements of the best available maps.
| Place. | River. | Drainage Area, Square Miles. |
|---|---|---|
| New Orleans, La. | Mississippi | 1,261,000 |
| St. Louis, Mo. | Mississippi | 700,000 |
| St. Petersburg | Neva | 108,000 |
| Louisville, Ky. | Ohio | 90,000 |
| Rock Island, Ill. | Mississippi | 88,000 |
| Budapest | Danube | 79,000 |
| Cincinnati, O. | Ohio | 75,700 |
| Dordrecht | Maas | 68,000 |
| Rotterdam | Maas | 68,000 |
| Schiedam | Maas | 68,000 |
| Altona | Elbe | 52,000 |
| Hamburg | Elbe | 52,000 |
| Stettin | Oder | 40,000 |
| Magdeburg | Elbe | 36,000 |
| Warsaw | Weichsel | 34,000 |
| Odessa | Dneister | 26,000 |
| Worms | Rhine | 25,000 |
| Grand Forks, N. Dak. | Red River of the North | 22,000 |
| Frankfort on Oder | Oder | 21,000 |
| Bremen | Weser | 15,000 |
| Suburbs of Paris | Seine | 12,000 |
| Poughkeepsie, N. Y. | Hudson | 11,600 |
| Pittsburg, Penn. | Allegheny | 11,400 |
| Posen | Wartha | 9,400 |
| Hudson, N. Y. | Hudson | 9,200 |
| Albany, N. Y. | Hudson | 8,200 |
| Breslau | Oder | 8,200 |
| Brieg | Oder | 7,500 |
| Lawrence, Mass. | Merrimac | 4,634 |
| Stuttgart | Neckar | 1,660 |
| Brunswick | Ocker | 650 |
| Somersworth, N. H. | Salmon | 171 |
PRELIMINARY PROCESSES TO REMOVE MUD.
With both sand and mechanical filtration the difficulty and expense of treatment of a water increase nearly in direct proportion to the turbidity of the water as applied to the filter; and it is thus highly important to secure a water for filtration with as little turbidity as possible, and thus to develop to their economical limits the preliminary processes for the removal of mud. One of the most important of these processes is the use of reservoirs.
Reservoirs serve two purposes in connection with waters drawn[Pg 134] from streams: they allow sedimentation, and they afford storage. If a water having a turbidity of 1.00 is allowed to remain in a sedimentation-basin for 24 hours, its turbidity may be reduced by as much as 40 per cent, or to 0.60. If it is held a second day the additional reduction is much less.
If samples are taken of the water in the reservoir before and after settling and sent to the chemist for analysis, he will probably report that from 70 to 80 per cent of the suspended matters have been removed by the process. The suspended matters are removed in much larger ratio than the turbidity. This arises from the fact that there is a certain proportion of comparatively coarse material in the water as it is taken from the river. This coarse material increases the weight of the suspended matters without increasing the turbidity in a corresponding degree. In 24 hours the coarser materials are removed completely, and at the end of that time only the clayey or finer particles remain in suspension. It is these clayey particles, however, that constitute the turbidity, which are most objectionable in appearance, and which are most difficult of removal by filtration or otherwise.
Sedimentation thus removes the heavier matters from the water, but it does not remove the finer matters which principally affect the appearance of the water and are otherwise most troublesome. A sedimentation of 24 hours removes practically all of the coarser matters, and the clayey material remaining at the end of that time can hardly be removed by further sedimentation. The economic limit of sedimentation is about 24 hours.
Sedimentation has practically no effect upon the clearer waters between flood periods.
Let us consider the effect of a sedimentation-basin, or reservoir holding a 24-hours’ supply of water, into which water is constantly pumped at one end, and from which an equal quantity is constantly withdrawn from the other, upon the water of a stream of such size that the time of passage of water from the feeders to the intake is less than 24 hours. During the period between storms[Pg 135] the water is comparatively clear and passes through the sedimentation basin without change. When a storm comes the water in the stream promptly becomes muddy, and muddy water is supplied to the reservoir; but owing to the time required for water to pass through it, the outflowing water remains clear for some hours. There is a gradual mixing, however, and long before the expiration of 24 hours somewhat muddy water appears at the outlet. The turbid-water period rarely lasts in streams of this size more than 24 hours, and at the expiration of that time the water in the sedimentation-basin is as muddy or muddier than the water flowing in the stream. After the height of the flood the stream clears itself by the flowing away of the turbid water much more rapidly than the water clears itself by sedimentation in the reservoir. That is to say, if at the time of maximum turbidity we take a certain quantity of water from the stream and put it aside to settle, at no time will the improvement by settling equal the improvement which has taken place in the stream from natural causes. Generally the improvement in the stream is several times as rapid as in the sedimentation-basin, and the water from it will at times have only a fraction of the turbidity of the water in the basin.
Let us now consider what the sedimentation has done to improve the water. During the period of clear water, that is for most of the time, it has done nothing. For the first day of each flood period very much clearer water has been obtained from it than was flowing in the stream. For the first days following floods the water in the sedimentation-basin has been more muddy than the water in the stream. The only time when the sedimentation-basin has been of use is during the first part of floods, that is, when the turbidity of the water in the stream is increasing. During this period it has been of service principally because of its storage capacity, yielding up water received from the stream previously, when it was less muddy. Such sedimentation as has been secured is merely incidental and generally not important in amount.
[Pg 136]
It will be obvious from the above that for these conditions storage is much more important than sedimentation. This brings us back to the old English idea of having storage-reservoirs large enough to carry water-works over flood periods without the use of flood-waters. Reservoirs of this kind were, and still are, considered necessary for the successful utilization of waters of many English rivers, although these waters do not approach in turbidity the waters of some American streams. This idea of storage has been but little used in the United States.
In the above case, if we use our reservoir for storage instead of as a sedimentation-basin, the average quality of the water can be greatly improved. The reservoir should ordinarily be kept full, and pumping to it should be stopped whenever the turbidity exceeds a certain limit, to be determined by experience; and the reservoir is then to be drawn upon for the supply until the turbidity again falls to the normal. In the case assumed above, with a stream in which all of the water reaches the intake in 24 hours, a reservoir holding a 24-hours’ supply, or in practice, to be safe, a somewhat larger one, would yield a water having a very much lower average turbidity than would be obtained with water pumped constantly from the stream without a reservoir.
With a river having a watershed so long that 48 hours are required to bring the water down from the most remote feeders, a reservoir twice as large would be required, and would result in a still greater reduction in the average turbidity.
As the stream becomes larger, and the turbid periods longer, the size of a reservoir necessary to utilize this action rapidly becomes larger, and the times during which it can be filled are shortened, and thus the engineering difficulties of the problem are increased. For moderately short streams, cost for cost, storage is far more effective than sedimentation, and we must come back to the old English practice of stopping our pumps during periods of maximum turbidity.
[Pg 137]
EFFECT OF MUD UPON SAND FILTERS.
There are two aspects of the effect of mud upon the operation of sand filters which require particular consideration. The first relates to the rapidity of clogging, and consequently the frequency of scraping and the cost of operation; while the second relates to the ability of the filters to yield well-clarified effluents.
EFFECT OF TURBIDITY UPON THE LENGTH OF PERIOD.
The amount of water which can be filtered between scrapings is directly dependent upon the turbidity of the raw water. The greater the turbidity, the more frequently will filters require to be scraped. In the experiments of the Pittsburg Filtration Commission, with 4 feet of sand of an effective size of about 0.30 millimeter, and with rates of filtration of about three million gallons per acre daily, and with the loss of head limited to 4 feet, sand filters were operated as follows: For five periods the turbidities of the raw water ranged from 0.035 to 0.062, and averaged 0.051, and the corresponding periods ranged from 102 to 136, and averaged 113 million gallons per acre filtered between scrapings. For ten periods the turbidities of the raw water ranged from 0.079 to 0.128, and averaged 0.102, and the periods averaged 78 million gallons per acre between scrapings. For fifteen other periods the turbidities of the raw water ranged from 0.134 to 0.269, and averaged 0.195, and the periods averaged 52 million gallons per acre between scrapings. In two other periods the turbidities of the raw water averaged 0.67, and the periods between scrapings averaged 16 million gallons. In all cases the turbidity is taken as that of the water applied to the filter. Usually this was the turbidity of the settled water, but in some cases raw water was applied, and in these case the turbidity of the raw water is taken. These results are approximately represented by the formula
Period between scrapings, |
} | = | 12 turbidity + 0.05 |
[Pg 138]
Except for very clear waters the amount of water passed between scrapings is nearly inversely proportional to the turbidity. With twice as great an amount of turbidity, filters will have to be cleaned twice as often, the reserve area for cleaning will require to be twice as great, and the cost of scraping filters and of washing and replacing sand, which is the most important element in the cost of operation, will be doubled.
With waters having turbidities of 0.20 upon this basis, the average period will be about 51 million gallons per acre between scrapings. This is about the average result obtained at the German works filtering river waters, and there is no serious difficulty in operating filters which require to be scraped with this frequency. With more turbid waters the period is decreased. With an average turbidity of 0.50 the average period is only 24 million gallons per acre between scrapings, a condition which means very difficult operation and a very high cost of cleaning. With much more turbid waters the difficulties are increased, and if the duration of turbid water should be long-continued, the operation of sand filters would clearly be impracticable, and the expense, also, would be prohibitive.
In applying these figures to actual cases it must be borne in mind that the turbidity is only one of the several factors which control the length of period; and that the turbidity of a water of a given stream is never constant, but fluctuates within wide limits; and that raw water can be applied to filters for a short time without injurious results, even though it is so turbid that its continued application would be fatal.
It is very likely also that the suspended matters in different streams differ in their natures to such an extent that equal turbidities would give quite different periods, although the Pittsburg results were so regular as to give confidence in their application to other conditions within reasonable limits, and when so applied they afford a most convenient method of computing the approximate cost of operation of filters for waters of known or estimated turbidities.
[Pg 139]
POWER OF SAND FILTERS TO PRODUCE CLEAR EFFLUENTS FROM MUDDY WATER.
When the turbidity of the applied water is not too great it is entirely removed in the course of filtration. With extremely muddy raw waters, however, turbid effluents are often produced with sand filters. The conditions which control the passage of the finest suspended matters through filters have been studied by Mr. Fuller at Cincinnati at considerable length. They are similar in a general way to the conditions which control the removal of bacteria. That is to say, the removal is more complete with fine filter sand than with coarse sand; with a deep sand layer than with a shallow sand layer; and with low rates of filtration than with high rates. The practicable limits to the size of sand grain, depth of sand layer, and rate of filtration are established by other conditions, and the question remains whether within these limits a clear effluent can be produced.
At Pittsburg the turbidity of the effluent from a sand filter operated as mentioned above, which received water which had passed through a sedimentation-basin holding about a 24-hours’ supply, but without taking any advantage of storage to avoid the use of muddy water, was nearly always less than 0.02, which may be taken as the admissible limit of turbidity in a public water-supply. This limit was exceeded on less than 20 days out of 365, these days being during the winter and spring freshets, and on these days the excess was not such as would be likely to be particularly objectionable. For the water of the Allegheny River, then, sand filtration with one day’s sedimentation is capable of yielding a water not absolutely clear, but sufficiently clear to be quite satisfactory for the purpose of municipal water-supply.
At Cincinnati, on the other hand, where the amount of suspended matters was five times as great as at Pittsburg, the effluents which could be obtained by sand filtration without recourse to the use of alum, even under most favorable conditions,[Pg 140] were very much more turbid than those obtained at Pittsburg, and were, in fact, so turbid as to be seriously objectionable for the purpose of public water-supply.
With rivers no more turbid than the Allegheny River at Pittsburg, and rivers having floods of such short duration that the use of flood-flows can be avoided by the use of reservoirs, sand filters are adequate for clarification. For waters which are much muddier than the Allegheny, as, for instance, the Ohio at Cincinnati and at Louisville, sand filtration alone is inadequate. Mr. Fuller,[31] as a result of his Cincinnati experiments, has stated the case as follows:
“For the sake of explicitness it is desired to show, with the data of the fairly normal year of 1898, the proportion of the time when English filters (that is, sand filters) would be inapplicable in the purification of the unsubsided Ohio River water at Cincinnati. This necessitates fixing an average limit of permissible suspended matter in this river water, and is a difficult matter from present evidence.
“In part this is due to variations in the character and in the relative amounts of the suspended silt, clay, and organic matter; and in part it is due to different amounts of clay stored in the sand layer, which affects materially the capacity of the filter to retain the clay of the applied water. During these investigations the unsubsided river-water was not regularly applied to filters; and, with the exception of the results of tests for a few days only, it is necessary to depend upon general information obtained with reference to this point. So far as the information goes, it appears that an average of 125 parts per million is a conservative estimate of the amount of suspended matters in the unsubsided river-water, which could be regularly and satisfactorily handled by English filters. But at times this estimated average would be too low, and at other times too high....
“While English filters are able to remove satisfactorily on an [Pg 141] average about 125 parts of silt and clay of the unsubsided water, actual experience shows that they can regularly handle suspended clay in subsided water in amounts ranging only as high as from 30 to 70 parts (depending upon the amount of the clay stored in the sand layer), and averaging about 50 parts per million. But it is true that for two or three days on short rises in the river, or at the beginning of long freshets, the retentive capacity of the sand layer allows of satisfactory results with the clay in the applied water considerably in excess of 70 parts. If this capacity is greatly overtaxed, however, the advantage is merely temporary, as the stored clay is washed out later, producing markedly turbid effluents.”
Translating Mr. Fuller’s results into terms of turbidity, the 125 parts per million of suspended matters in the raw water represent a turbidity of about 0.40, and the 30 to 70 parts of suspended matters in the settled water represent turbidities from 0.20 to 0.40, the average of 50 parts of suspended matters corresponding to a turbidity of about 0.30.
Upon this basis, then, sand filters are capable of treating raw waters with average turbidities up to 0.40, or settled waters with average turbidities up to 0.30, but waters more turbid than this are incapable of being successfully treated without the use of coagulants or other aids to the process. These results are in general accordance with the results of the experiments at Pittsburg, and demonstrate that while sand filters as generally used in Europe are adequate for the clarification of many, if not most, river waters in the United States, there are other waters carrying mud in such quantities as to make the process inapplicable to them.
EFFECT OF MUD UPON BACTERIAL EFFICIENCY OF FILTERS.
The question is naturally raised as to whether or not the presence of large quantities of mud in the raw water will not seriously[Pg 142] interfere with the bacterial efficiency of filters. Experiments at Cincinnati and Pittsburg have given most conclusive and satisfactory information upon this point. Up to the point where the effluents become quite turbid, the mud in the raw water has no influence upon the bacterial efficiency; and even somewhat beyond this point, with effluents so turbid that they would hardly be suitable for the purpose of a public water-supply, the bacterial efficiency remains substantially equal to that obtained with the clearest waters. Only in the case of excessive quantities of mud, where, for other reasons, sand filters can hardly be considered applicable, is there a moderate reduction in bacterial efficiency. As mentioned above, particles constituting turbidity are often much smaller than the bacteria, and in addition, the bacteria probably have an adhesive power far in excess of that of the clay particles. For these reasons clay particles are able to pass filters under conditions which almost entirely prevent the passage of bacteria.
On the other hand, it does not necessarily follow that the removal of turbidity is accompanied by high bacterial efficiency. Although this is often the case, there are marked exceptions, particularly in connection with the use of coagulants, where very good clarification is obtained, and notwithstanding this, effluents are produced containing comparatively large numbers of bacteria.
LIMITS TO THE USE OF SUBSIDENCE FOR THE PRELIMINARY TREATMENT OF MUDDY WATERS.
When water is too muddy to be applied directly to filters, the most obvious treatment is to remove as much of the sediment as possible by sedimentation. Sedimentation-basins are considered as essential parts of filtration plants for the treatment of muddy waters. The effect of sedimentation, as noted above, is to remove principally the larger particles in the raw water. By doing this the deposit upon the surface of the filters and the cost of operation are greatly reduced.
These larger particles are mainly removed by a comparatively[Pg 143] short period of sedimentation, and the improvement effected after the first 24 hours is comparatively slight. The particles remaining in suspension at the end of this time consist almost entirely of very fine clay, and the rate of their settlement through the water is extremely slow; and currents in the basin, due to temperature changes, winds, etc., almost entirely offset the natural tendency of the sediment to fall to the bottom.
There is thus a practical limit to the effect of sedimentation which is soon reached, and it has not been found feasible to extend the process so as to allow much more turbid waters to be brought within the range which can be economically treated by sand filtration.
[Pg 144]
CHAPTER IX.
THE COAGULATION OF WATERS.
The coagulation of water consists in the addition to it of some substance which forms an inorganic precipitate in the water, the presence of which has a physical action upon the suspended matters, and allows them to be more readily removed by subsidence or filtration.
The most common coagulant is sulphate of alumina. When this substance is added to water it is decomposed into its component parts, sulphuric acid and alumina, the former of which combines with the lime or other base present in the water, or in case enough of this is lacking, it remains partly as free acid and partly undecomposed in its original condition; while the alumina forms a gelatinous precipitate which draws together and surrounds the suspended matters present in the water, including the bacteria, and allows them to be much more easily removed by filtration than would otherwise be the case. In addition, the alumina has a chemical attraction for dissolved organic matters, and the chemical purification may be more complete at very high rates than would be possible with sand filtration without coagulant at any rate, however low.
Coagulants have been employed in connection with filtration from very early times. As early as 1831 D’Arcet published in the “Annales d’hygiène publique,”[32] an account of the purification of Nile water in Egypt by adding alum to the water, and afterwards filtering it through small household filters. More recently alum has been repeatedly used in connection with sand filters, particularly
[Pg 145]
at Leeuwarden, Groningen, and Schiedam in Holland, where the river waters used for public supplies are colored by peaty matter which cannot be removed by simple filtration.
SUBSTANCES USED FOR COAGULATION.
Mr. Fuller[33] has given a very full account of the substances which can be used for the clarification of waters. Without taking up all of the unusual substances which have been suggested, the most important of the coagulants will be briefly described below.
Lime.—Lime has been extensively used in connection with the purification of sewage, and also for softening water. Lime is first slaked and converted into calcium hydrate, which is afterwards dissolved in water, and applied to the water under treatment. The amount of lime to be used is fixed by the amount of carbonic acid in the water. So much lime is always used as will exactly convert the whole of the carbonic acid of the water into normal carbonate of lime. This substance is but slightly soluble in water and it precipitates. The precipitate is crystalline rather than flocculent, and is not as well adapted to aid in the removal of clayey matters as some other substances, although its action in this respect is considerable. The precipitate is quite heavy, and is largely removed by sedimentation, although filtration must be used to complete the process. Water which has been treated with lime is slightly caustic; that is to say, there is a deficiency of carbonic acid in it, and it deposits lime in the pipes, in pumps, etc.; and although the precipitated calcium carbonate is much softer than steel, it rapidly destroys pumps used for lifting it.
Principally for these reasons it is necessary to supply carbonic acid to water which has been treated in this way, and this is done by bringing it in contact with flue-gases, or by the direct addition of carbonic acid.
The use of lime for softening waters is known as Clark’s process. It was patented in England many years ago, and the[Pg 146] patent has now expired. Various ingenious devices have been constructed for facilitating various parts of the operation. The process has hardly been used in the United States, but there is a large field for it in connection with the softening of very hard waters, and where such waters also contain iron or clay, these substances will be incidentally removed by the process.
Larger quantities of lime have an action upon the suspended matters which is entirely different from that secured in Clark’s process, and the action upon bacteria is particularly noteworthy. This action was noted in experiments at Lawrence,[34] where it was found that sewage was almost completely sterilized by the application of considerable quantities of lime. An extremely interesting series of experiments upon the application of large quantities of lime to water was made by Mr. Fuller in 1899.[35] The bacterial results were extremely favorable, although the necessity for removing the excess of lime afterward is a somewhat serious matter, and in these experiments it was not entirely accomplished.
Aluminum Compounds.—Sulphate of alumina is most commonly employed. It can be obtained in a state of considerable purity at a very moderate price, and important improvements in the methods used for its manufacture have been recently introduced. Potash and soda alums have no advantage over sulphate of alumina, and, in fact, are less efficient per pound, while their costs are greater. Chloride of alumina is practically equivalent to the sulphate in purifying power, but is more expensive.
Sodium Aluminate has been examined by Mr. Fuller, who states that experience has shown that its use is impracticable in the case of the Ohio River water.
Compounds of Iron.—Iron forms two classes of compounds, namely, ferrous and ferric salts. When the ferrous salts are applied to water, under certain conditions, ferrous hydrate is precipitated, [Pg 147]but this substance is not entirely insoluble in water containing carbonic acid. Under some conditions the precipitated ferrous hydrate is oxidized by oxygen present in the water to ferric hydrate, and so far as this is the case, good results can be obtained. Ferrous sulphate is not as readily oxidized when applied to water as is the ferric carbonate present in many natural waters, and for this reason ferrous sulphate has not been successfully used in water purification. In the treatment of sewage, where the requirements are somewhat different, it has been one of the most satisfactory coagulants.
Ferric sulphate acts in much the same way as sulphate of alumina, and is entirely suitable for use where sulphate of alumina could be employed, but it has not been used in practice, due probably to its increased cost as compared with its effect, and to the practical difficulties of applying it in the desired quantities due to its physical condition.
Metallic Iron: The Anderson Process.—The use of metallic iron for water purification in connection with a moderately slow filtration through filters of the usual form is known as Anderson’s process (patented), and has been used at Antwerp and elsewhere on a large scale, and has been experimentally examined at a number of other places.
The process consists in agitating the water in contact with metallic iron, a portion of which is taken into solution as ferrous carbonate. Upon subsequent aeration this is supposed to become oxidized and precipitate out as ferric hydrate, with all the good and none of the bad effects which follow the use of alum. The precipitate is partially removed by sedimentation, while filtration completes the process. The process is admirable theoretically, and in an experimental way upon a very small scale often gives most satisfactory results, muddy waters very difficult of filtration, and colored peaty waters yielding promptly clear and colorless effluents.
In applying the process on a larger scale, however, with peaty [Pg 148] waters at least, it seems impossible to get enough iron to go into solution in the time which can be allowed, and the small quantity which is taken up either remains in solution or else slowly and incompletely precipitates out, without the good effects which follow the sudden and complete precipitation of a larger quantity, and in this case the color is seldom reduced, and may even be increased above the color of the raw water by the iron remaining in solution.
The ingenuity of those who have studied the process has not yet found any adequate means of avoiding these important practical objections; and even at Antwerp a great extension of the filtering area, as well as the use of alum at times of unusual pollution, is good evidence that simple filtration, in distinction from the effect of the iron, is relied upon much more than formerly.
At Dordrecht also, where the process has been long in use, the rate of filtration does not exceed the ordinary limits; nor is the result, so far as I could ascertain, in any way superior to that obtained a few miles away at Rotterdam, by ordinary filtration, with substantially the same raw water.
The results obtained at Boulogne-sur-Seine, near Paris, have been closely watched by the public chemist and bacteriologist of Paris, and have been very favorable, and a number of new plants of very considerable capacity have been built, to supply some of the suburbs of Paris, but even in these cases only moderate rates of filtration are employed which would yield excellent effluents without the iron.
Compounds of Manganese.—Manganese forms compounds similar to those of iron, that is to say manganous and manganic salts, but their use in connection with water filtration has not been found possible. In addition, manganese forms a series of compounds, known as manganates and permanganates, quite different in their structure and action from the others. These compounds contain an excess of oxygen which they give up very readily to organic matters capable of absorbing oxygen, and because of this[Pg 149] power, they have been extensively used in the treatment of sewage. Applied to the treatment of waters their action is very slight, and the compounds are so expensive that they have not been employed for this purpose. Theoretically the action is very attractive, as the oxygen liberated by their decomposition oxidizes some of the organic matter of the water, thereby purifying it in part, while the manganese is precipitated as a flocculent precipitate having all of the advantages pertaining to a precipitate of hydrate of alumina, and without the disadvantage of adding acid to the water, as is the case with the compounds of alumina and iron. These chemicals, when used in comparatively concentrated condition, have powerful germicidal actions, but in water purification the amounts which can be used are so small that no action of this kind results. The amount which can be applied to a water is limited to the amount which can be decomposed by the organic matters present in the water, and is not large.
The Use of Metallic Iron and Aluminum, with the Aid of Electricity.—Elaborate experiments were made at Louisville with metallic iron and aluminum oxidized and made available by the aid of electric currents. The use of iron with electric currents was tried in sewage purification some years ago, under the name of the Webster process, but was never put to practical use. The theory is to oxidize the iron or aluminum in contact with the water, with the formation of flocculent hydrates, by the aid of an electric current, thereby securing the advantages of the application of salts of these metals to the water without the disadvantage of the addition of acid.
Other Chemicals Employed.—A solution containing chlorine produced by electrical action has been suggested. Chlorine is a powerful disinfectant, and when used in large quantities kills bacteria. It is not possible to use enough chlorine to kill the bacteria in the water without rendering it unfit for human use. The nature of this treatment has been concisely described by[Pg 150] Dr. Drown,[36] who shows that the electrically prepared fluids do not differ in their action in any way from well-known chemicals, the use of which would be hardly considered.
The use of ozone and peroxide of hydrogen have also been suggested, but I do not know that they have been successfully used on a large scale. The same is true of many other chemicals, the consideration of which is hardly necessary in this connection.
COAGULANTS WHICH HAVE BEEN USED.
In actual work sulphate of alumina is practically the only coagulant which has been employed, excepting the alums, which are practically its equivalent in action, differing only in strength. Nearly all important experiments upon the coagulation of water have been made with sulphate of alumina, and in the further discussion of this subject only this coagulant will be considered.
AMOUNT OF COAGULANT REQUIRED TO REMOVE TURBIDITY.
In the coagulation of turbid waters a certain definite amount of coagulant must be employed. If less than this amount is used either no precipitate will be formed, or it will not be formed in sufficient bulk to effect the desired results. It is necessary that the precipitate should be sufficient, and that it should be formed practically all at one time. The amount of coagulant necessary to accomplish this purpose is dependent upon the turbidity of the raw water. With practically clear waters sulphate of alumina of the ordinary commercial strength, that is to say, with about 17 per cent soluble oxide of aluminum, used in quantities as small as 0.3 or 0.4 of a grain per gallon, will produce coagulation. As the turbidity increases larger amounts must be employed.
A special study was made of this point in connection with the Pittsburg experiments.[37] As an average of these results it was found that two grains per gallon of sulphate of alumina were[Pg 151] required to properly coagulate waters having turbidities of 1.00, so that they could be filtered by the Jewell filter, and 2.75 grains were required for the Warren filter.
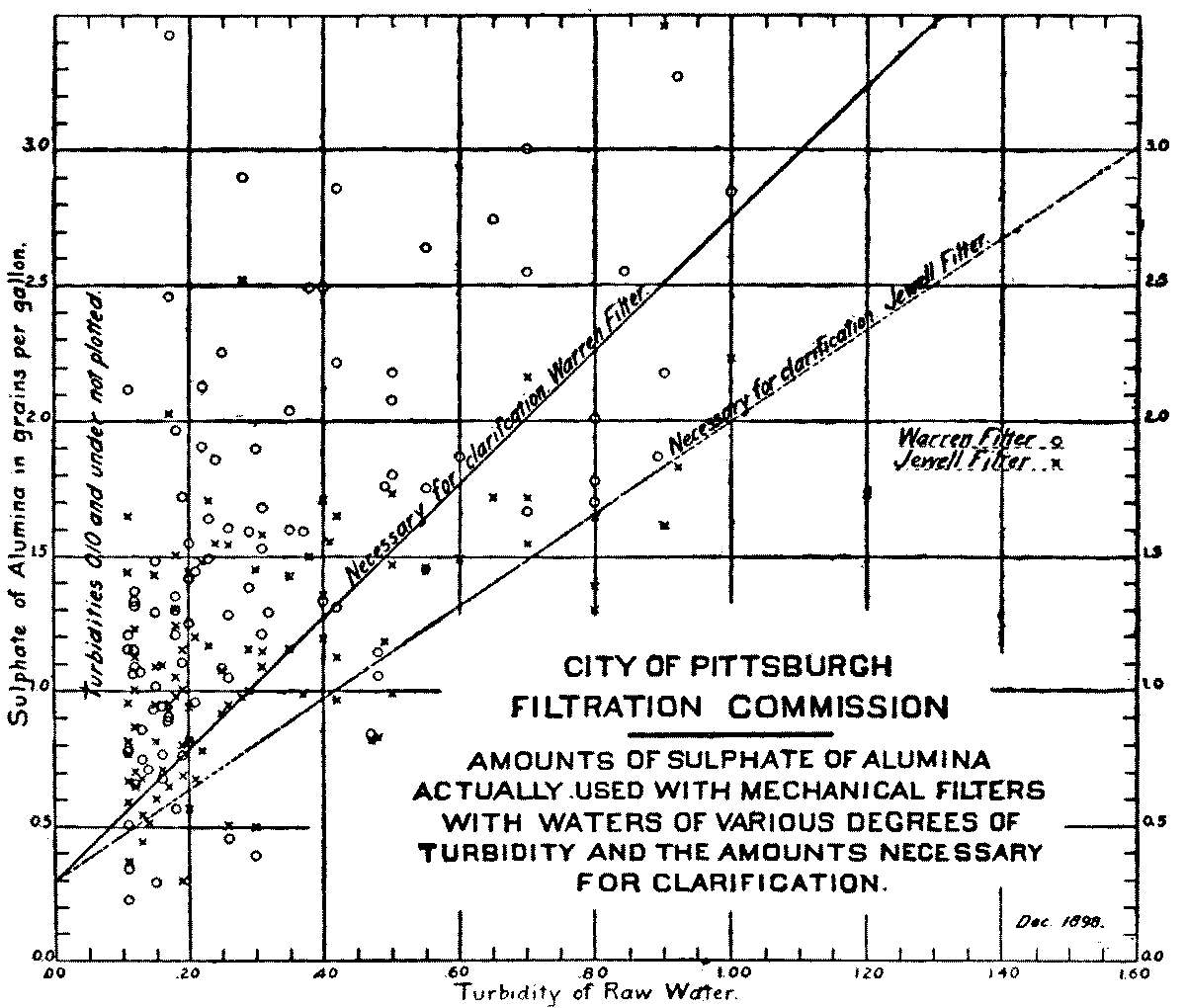
Fig. 20.—Amount of Coagulant Required to Remove Turbidity.
Aside from the amount required to produce a precipitate in the clearest waters, the amount of coagulant required was proportional to the turbidity. As an average for the two filters the required quantity was approximately 0.30 of a grain, and in addition 0.02 of a grain for each 0.01 of turbidity. Thus a water having a turbidity of 0.20 requires 0.70 of a grain per gallon; a water having a turbidity of 0.50 requires 1.30 grains; of 1.00, 2.30 grains; of 2.00, 4.30 grains, etc. These are average minimum results. Occasionally clear effluents were produced with smaller quantities of coagulant, while at other times larger quantities were necessary for satisfactory results.
[Pg 152]
The amount of coagulant required for clarification at Cincinnati has been stated by Mr. Fuller in his report. A number of his results are brought together in the following table, to which has also been added a column showing approximately the corresponding results at Pittsburg.
| ESTIMATED AVERAGE AMOUNTS OF REQUIRED CHEMICAL FOR DIFFERENT GRADES OF WATER. | ||||
|---|---|---|---|---|
| Suspended Matter, Parts in 100,000. |
Chemical Required, Grains per Gallon. | |||
| Raw Water for Sand Filters. Cincinnati Report, Page 290. |
Subsided Water for Cincinnati Report, Page 290. |
Subsided Water for Mechanical Filters. Cincinnati Report, Page 341. |
Minimum for Raw Water for Mechanical Filters. Pittsburg. |
|
| 1.0 | 0 | 0 | 0.75 | 0.40 |
| 2.5 | 0 | 0 | 1.25 | 0.50 |
| 5.0 | 0 | 0 | 1.50 | 0.70 |
| 7.5 | 0 | 1.30 | 1.95 | 0.90 |
| 10.0 | 1.50 | 1.60 | 2.20 | 1.00 |
| 12.5 | 1.60 | 1.80 | 2.45 | 1.15 |
| 15.0 | 1.70 | 2.00 | 2.65 | 1.30 |
| 17.5 | 1.80 | 2.10 | 2.85 | 1.40 |
| 20.0 | 1.95 | 2.20 | 3.00 | 1.60 |
| 30.0 | 2.25 | 2.45 | 3.80 | 2.00 |
| 40.0 | 2.50 | 2.75 | 4.40 | 2.50 |
| 50.0 | 2.80 | |||
| 60.0 | 3.05 | |||
| 75.0 | 3.40 | |||
| 100.0 | 4.00 | |||
| 120.0 | 4.75 | |||
Mr. Fuller’s results seem to show that a greater amount of coagulant is required for the preparation of water for mechanical filters than is necessary in connection with sand filters. The results with sand filters indicate that settled waters and raw waters containing equal amounts of suspended matters are about equally difficult to treat. The results at Pittsburg indicate that the raw waters required much smaller quantities of coagulant for given amounts of suspended matters than was the case with subsided waters at Cincinnati, the results agreeing more closely with the amounts required to prepare raw water for sand filters at Cincinnati.
[Pg 153]
AMOUNT OF COAGULANT REQUIRED TO REMOVE COLOR.
The information upon this point is, unfortunately, very inadequate. In some experiments made by Mr. E. B. Weston at Providence in 1893 with a mechanical filter,[38] with quantities of sulphate of alumina averaging 0.6 or 0.7 of a grain per gallon, the removal of color was usually from 70 to 90 per cent. The standard used for the measurement of color is not stated, and there is no statement of the basis of the scale, consequently no means of determining the absolute color of the raw water upon standards commonly used.
At Westerly, R. I., with a New York filter, the actual quantity of potash alum employed from Oct. 10, 1896, to March 1, 1897, was 1.94 grains per gallon, the amount being regulated to as low a figure as it was possible to use to secure satisfactory decolorization. There is no record of the color of the raw water. A very rough estimate would place it at 0.50 upon the platinum scale. The chemical employed in this case was alum, and two thirds as large a quantity of sulphate of alumina would probably have done corresponding work, had suitable apparatus for applying it been at hand.
At Superior, Wisconsin, the water in the bay coming from the St. Louis River, having a color of 2.40 platinum scale, was treated experimentally with quantities of sulphate of alumina up to 4 grains per gallon, by Mr. R. S. Weston in January, 1899, but even this quantity of coagulant utterly failed to coagulate and decolorize it.
At Greenwich, Conn., during 1898 the average amount of sulphate of alumina employed, as computed from quantities stated in the annual report of the Connecticut State Board of Health for 1898, was about 0.44 of a grain per gallon, and this quantity sufficed to reduce the color of the raw water from 0.40 to 0.30, platinum standard. This reduction is very slight, and it is [Pg 154] obvious that this quantity of coagulant was not enough for decolorization.
Some experiments bearing on color removal were made at East Providence, R. I., by Mr. E. B. Weston, and are described in the Proceedings of the American Society of Civil Engineers for September, 1899. In this case the color is reported to have been reduced from 0.58 to 0.10 platinum standard by the use of one grain of sulphate of alumina, containing 22 per cent of effective alumina, equivalent to about 1.30 grains of the ordinary article per gallon.
The various experiments seem to indicate that a removal from 80 to 90 per cent of the color can be effected by the use of a quantity of sulphate of alumina equal to rather more than two grains per gallon for waters having colors of 1.00, platinum standard, and proportionate quantities for more and less deeply colored waters. With much less sulphate of alumina decolorization is not effected, and even larger quantities do not remove all of the color.
The data are much less complete than could be desired, and it is to be hoped that experiments will be undertaken to throw more light upon this important subject.
SUCCESSIVE APPLICATION OF COAGULANT.
Mr. Fuller, in his experiments at Louisville, has ascertained that when sulphate of alumina is added to extremely muddy water the sediment absorbs some of the chemical before it has time to decompose, and carries it to the bottom, and so far as this is the case, no benefit is derived from that part of the coagulant which is absorbed. In other words, it is necessary to add more coagulant than would otherwise be necessary because of this action. The data showed that different kinds of suspended matters took up very different amounts of coagulant in this way. With only moderately turbid waters the loss of chemical from this source is unimportant. Hardly any trace of it was found at Pittsburg with[Pg 155] the Allegheny River water. At Louisville, however, it was an important factor, as shown by Mr. Fuller’s results.
To avoid this loss of chemical Mr. Fuller has suggested the removal of the greater part of the suspended matters by sedimentation, without chemicals, or with the aid of a small quantity of chemical, followed by the application of the final coagulant prior to filtration. With the worst waters encountered at Louisville the saving in coagulant to be effected in this way is very great.
Mr. Fuller states in “Water Purification at Louisville,” p. 417: “The practical conclusions to be drawn from this experience are that with preliminary coagulation, followed by subsidence for a period of about three hours, the application of coagulants may be divided to advantage, and a considerable portion of the suspended matter kept off the filter, when the total amount of required coagulant ranges from 2 to 2.5 grains or more of ordinary sulphate of alumina per gallon. In the case of a water requiring more than this amount of coagulating treatment, a proper division of the application would increase the saving of coagulants and would diminish the frequency of washing the filter.”
In his final summary and conclusions, page 441, Mr. Fuller estimates the amount of sulphate of alumina required for the clarification of the Ohio River at Louisville at 3.00 grains per gallon of water filtered if all applied at one point, or at 1.75 grains by taking advantage of subsidence to its economical limit prior to the final coagulation. The saving to be effected in this way is sufficient to justify the works necessary to allow it to be carried out. With less turbid waters, or waters highly turbid for only short intervals, the advantages of double coagulation would be less apparent.
THE AMOUNT OF COAGULANT WHICH VARIOUS WATERS WILL RECEIVE.
The amount of coagulant which can be safely used is dependent upon the alkalinity of the raw water. When sulphate of alumina[Pg 156] is added to water it is decomposed, as explained above, with the formation of alumina, which is alone useful in the work of purification, and sulphuric acid, which combines with the calcium carbonate or lime present in the water. There should always be an excess of alkalinity or lime in the raw water. If for any reason there is not, there is nothing to combine with the liberated sulphuric acid, and the decomposition of the coagulant is not complete, and a portion of it goes undecomposed into the effluent. The effluent then has an acid reaction, and is unfit for domestic supply. When distributed through iron pipes, it attacks the iron, rusting the pipes, and giving rise to all the disagreeable consequences of an iron containing water.
The amount of lime in a water available to combine with the sulphuric acid can be determined by a very simple chemical operation, namely, by titration with standard acid with a suitable indicator. The amount of coagulant corresponding to a given quantity of lime can be readily and accurately calculated, but it is not regarded safe to use as much sulphate of alumina as corresponds to the lime. The quantity of coagulant used is not susceptible to exact control, but fluctuates somewhat, and if the exact theoretical quantity should be employed during 24 hours, there would surely be an excess during some portion of that time from which bad results would be experienced. It is therefore considered only prudent to use three quarters as much sulphate of alumina as corresponds to the lime in the water. With sulphate of alumina containing 17 per cent of soluble aluminum oxide and the corresponding amount of sulphuric acid, the amount which can be applied to a water in grains per gallon is slightly less than the alkalinity expressed in terms of parts in 100,000 of calcium carbonate.
Many waters contain sufficient lime to combine with the acid of all the coagulant which is necessary for their coagulation. Others will not, and it thus becomes an important matter to determine whether a given water is capable of decomposing sufficient[Pg 157] coagulant for its treatment. It is usually the flood-flows of rivers which control in this respect. The water at such times requires much larger quantities of coagulant for its clarification, and it also usually contains much less lime than the low-water flows. The reason for this is obviously that the water of the flood-flows is largely rain-water which has come over the surface without coming into very intimate contact with the soil, and consequently without having taken from it much lime, while the low-water flows contain a considerable proportion of water which has percolated through the soil and has thus become charged with lime.
In some parts of the country, as, for instance, in New England, the soil and underlying rock are almost entirely free from lime, and rivers from such watersheds are capable of receiving only very small quantities of coagulant without injurious results.
The deficiency of alkalinity in raw water can be corrected by the addition to it of lime or of soda-ash. Lime has been used for this purpose in many cases. When used only in moderate amounts it hardens the water, and is thus seriously objectionable. The use of so large a quantity as would precipitate out, as in Clark’s process, has not been employed in practice. If it should be attempted, the amount of lime would require to be very accurately controlled, and the effluent would have to be treated with carbonic acid to make it suitable for supply.
Waters so hard as to require the use of the Clark process almost always have sufficient alkalinity, and do not require to be treated with lime in connection with the use of sulphate of alumina.
The use of soda-ash is free from the objections to the use of lime, but is more expensive, and would require to be used with caution. Its use has often been suggested, but I do not know that it has ever been employed in practice. In small works the use of a filtering material containing marble-dust, or other calcareous matter, would seem to have some advantages in case of deficiency of alkalinity, although it would harden the water so treated.
[Pg 158]
The alkalinities of a number of waters computed as parts in 100,000 of calcium carbonate (approximately equal to the safe doses of sulphate to alumina in grains per gallon) are as follows:
| Maximum. | Minimum. | Average. | |
|---|---|---|---|
| Boston water, 1898 | 2.87 | 0.33 | 1.08 |
| Conestoga Creek, Lancaster, Penn. | 12.20 | 3.70 | 6.80 |
| Allegheny River, Pittsburg | 8.00 | 1.02 | 2.90 |
| Mahoning River and tributaries, 1897 | 20.00 | 2.20 | 10.00 |
| Scioto River and tributaries, 1897 | 35.00 | 10.00 | 20.00 |
| Ohio River, Cincinnati, 1898 | 7.00 | 2.00 | 4.50 |
| Ohio River, Louisville | 10.87 | 2.12 | 6.70 |
| Lake Erie, Lorain, Ohio | 9.50 | ||
| Lake Michigan, Chicago | 11.50 |
[Pg 159]
CHAPTER X.
MECHANICAL FILTERS.
The term mechanical filters is used to designate a general class of filters differing in many respects quite radically from the sand filters previously described. They had their origin in the United States, and consisted originally of iron or wooden cylinders filled with sand through which the water was forced at rates of one to two hundred million gallons per acre daily, or from fifty to one hundred times the rates usually employed with sand filters. These filters were first used in paper-mills to remove from the large volumes of water required the comparatively large particles, which would otherwise affect the appearance and texture of the paper; and in their earlier forms they were entirely inadequate to remove the finer particles, such as the bacteria, and the clay particles which constitute the turbidity of river waters. Various improvements in construction have since been made, and, in connection with the use of coagulants, much more satisfactory results can now be obtained with filters of this class; and their use has been extended from manufacturing operations to municipal supplies, in many cases with most satisfactory results.
The information gathered in regard to the conditions essential to the successful design and operation of these filters in the last few years is very great, and may be briefly reviewed.
PROVIDENCE EXPERIMENTS.[39]
The first data of importance were secured from a series of experiments conducted by Mr. Edmund B. Weston of Providence, R. I., in 1893 and 1894, upon the Pawtuxet river water used by[Pg 160] that city. The experimental filter was 30 inches in diameter, and had a layer of sand 2 feet 10 inches deep. The sand was washed by the use of a reverse current, the sand being stirred by a revolving rake at the same time. The amount of coagulant employed was about 0.7 of a grain per gallon. The raw water was practically free from turbidity, and the filter was operated to remove color and bacteria.
The removal of color, as stated in Mr. Weston’s report, amounted to from 70 to 90 per cent. The experiments extended over a period of ten months. The rate of filtration employed was about 128 million gallons per acre daily. The bacterial results of the first six months’ operations were rejected by Mr. Weston on account of defective methods of manipulation.
During the period from November 17, 1893, to January 30, 1894, the average bacterial efficiency of filtration was about 95 per cent, and the manipulation was considered to be in every respect satisfactory. The efficiency was occasionally below 90 per cent, but for four selected weeks was as high as 98.6 per cent. The average amount of sulphate of alumina used, as calculated from Mr. Weston’s tables, was two thirds of a grain per gallon. The highest efficiency followed the application of a solution of caustic soda to the filtering material. The first day following this treatment the bacterial efficiency was above 99 per cent. Afterwards it decreased until January 30, when the experiments were stopped. The high bacterial efficiency following the use of caustic soda was of such short duration as to suggest very grave doubts as to its practical value. It is extremely unfortunate that the experiments stopped only a week after this experiment, and the results were never repeated. I consider that the average bacterial efficiency of about 95 per cent obtained for the period of October 17 to January 30, when the manipulation was considered to be in every way satisfactory, more nearly represents what can be obtained under these conditions than the results for certain periods, particularly after the use of the caustic soda.
[Pg 161]
LOUISVILLE EXPERIMENTS.[40]
These experiments were inaugurated by the Louisville Water Company in connection with the manufacturers of certain patented filters. Mr. Charles Hermany, Chief Engineer of the Company, had general charge of the experiments. Mr. George W. Fuller was Chief Chemist and Bacteriologist and had direct charge of the work and has made a most elaborate report upon the same. In these examinations many devices were investigated; but the two which particularly deserve our attention are the filters known as the Warren Filter and the Jewell Filter.
These filters were operated for two periods, namely, from October 18, 1895, to July 30, 1896, and from April 5 to July 24, 1897. The investigations were directed toward the clarification of the river water from the mud, and to the removal of bacteria. The water was substantially free from color. The character of the water at this point was such that in its best condition at least three fourths of a grain of sulphate of alumina were necessary for its coagulation, and with this and with larger quantities of coagulant fair bacterial purification was nearly always obtained. The problem studied therefore was principally that of clarification from mud. The average efficiencies, as shown by the total averages, (page 248,) were as follows: Warren filter, bacterial efficiency, 96.7 per cent; Jewell filter, 96.0 per cent.
LORAIN TESTS.[41]
These tests were made by the author of a set of Jewell filters at Lorain, Ohio. The filters were six in number, each 17 feet in diameter, having an effective filtering area of 226 square feet each, or 1356 square feet in all. The construction of the filters was in all respects similar to the Jewell filter used at Louisville. The raw water was from Lake Erie, and during the examination was [Pg 162] always comparatively clear, but contained considerable numbers of bacteria. The problem was thus entirely one of bacterial efficiency. The question of clarification hardly presented itself. Although the water became turbid at times it did not approach in muddiness the condition of the Ohio River water, and an amount of coagulant sufficient for a tolerable bacterial efficiency in all cases was more than sufficient for clarification.
A summary of the results obtained is as follows:
| Week Ending 6:00 P.M. |
Average Rate of Filtration, Gallons per Sq. Ft. Min. |
Sulphate of Alumina, Grains per Gallon. |
Bacteria in Lake Water. |
Bacteria in Effluent. |
Bacterial Efficiency per cent. |
|---|---|---|---|---|---|
| June 19 | 1.06 | 2.58 | 1441 | 16 | 98.9 |
| 26 | 1.10 | 2.50 | 385 | 6 | 98.4 |
| July 3 | 1.11 | 2.27 | 367 | 9 | 97.5 |
| 10 | 1.28 | 1.07 | 154 | 14 | 90.9 |
| 17 | 1.14 | 0.94 | 189 | 26 | 86.3 |
| Average | 1.14 | 1.83 | 507 | 14 | 96.4 |
| The average bacterial efficiency was 96.4 per cent with 1.83 grains of sulphate of alumina per gallon. | |||||
PITTSBURG EXPERIMENTS.[42]
The Pittsburg experiments were inaugurated by the Pittsburg Filtration Commission. The operation of the filters extended from January to August, 1898. A Jewell and a Warren filter were used similar in design to those used at Louisville. The raw water contained large numbers of bacteria, and was also often very turbid, although less turbid than at Louisville. At times more coagulant was necessary for clarification than was required for bacterial efficiency; while as a rule more was required for satisfactory bacterial purification than was necessary for clarification. The opportunities were therefore favorable for the study of both of these conditions. The amount of coagulant necessary for clarification has been mentioned in connection with coagulation.
The results secured upon the relation of the quantity of [Pg 163] coagulant to the number of bacteria in the effluent were more complete than any other experiments available, and are therefore here reproduced from the Pittsburg report nearly in full.
It was found that the amount of sulphate of alumina employed was more important than any other factor in determining the bacterial efficiency, and special experiments were made to establish the effect of more and of less coagulant than used in the ordinary work. These experiments were made upon the Warren filter during May, and with the Jewell filter during June. The monthly averages for these months are thus abnormal and are not to be considered. The remaining six months for each filter may be taken as normal and as representing approximately the work of these filters under ordinary careful working conditions.
During the six months when the Warren filter was in normal order the raw water contained 11,531 bacteria and the effluent 201, the average bacterial efficiency being 98.26 per cent. The bacterial efficiency was very constant, ranging only, by months, from 97.48 to 98.96 per cent. During the same period a sand filter receiving the same water yielded an effluent having an average of 105 bacteria per cubic centimeter.
The Jewell filter, for the six months in which it was in normal order, received raw water containing an average of 11,481 bacteria and yielded an effluent containing an average of 293, the bacterial efficiency being 97.45 per cent, and ranging, in different months, from 93.23 to 98.61 per cent.
WASTING EFFLUENT AFTER WASHING FILTERS.
After washing a mechanical filter the effluent for the first few minutes is often inferior in quality to that obtained at other times, and if samples are taken at these times and averaged with other samples taken during the run, an apparent efficiency may be obtained inferior to the true efficiency. To guard against this source of error, whenever samples have been taken at such times, the average work for the day has been taken, not as the numerical[Pg 164] average of the results, but each sample has been given weight in proportion to the amount of time which it could be taken as representing; so that the results represent as nearly as possible the average number of bacteria in the effluent for the whole run. As a matter of fact, however, comparatively few samples were taken during these periods of reduced efficiency, and thus most of the results represent the normal efficiency exclusive of this period. A study has been made, however, of the results of examinations of samples taken directly after washing, somewhat in detail. The following is a tabular statement of the average results obtained from each filter by months, including only the results obtained on those days when samples were taken within twenty minutes after washing, the results of other days being excluded.
| AVERAGE NUMBER OF BACTERIA IN EFFLUENT. | ||||
|---|---|---|---|---|
| Shown by Record Sheets. |
Within Ten Minutes after Washing. |
11 to 20 Minutes after Washing. |
More than Twenty Minutes after Washing. |
|
| WARREN FILTER. | ||||
| February | 115 | 118 | 114 | |
| March | 316 | 50 | 515 | 301 |
| April | 79 | 417 | 207 | 75 |
| May | (Special experiments, omitted.) | |||
| June | 197 | 493 | 272 | 170 |
| July | 300 | 546 | 207 | |
| August | 174 | 356 | 601 | 223 |
| JEWELL FILTER. | ||||
| February | 2453 | 2425 | 2099 | |
| March | 455 | 657 | 958 | 354 |
| April | 99 | 665 | 462 | 165 |
| May | 144 | 998 | 346 | 127 |
| June | (Special experiments, omitted.) | |||
| July | 279 | 1330 | 272 | 274 |
| August | 344 | 612 | 323 | 376 |
The time of inferior work very rarely exceeded twenty minutes. It will be seen from the tables that the results as shown by the record sheets are never very much higher, and are occasionally lower than the results of samples taken on corresponding days more than twenty minutes after washing; and thus while a decrease in bacterial efficiency was noted after washing, no[Pg 165] material increase in the average bacterial efficiency of the mechanical filters would have been obtained if these results had been excluded. The results for the whole time would be affected much less than is indicated by the table, because the table includes only results of those days when samples were taken just after washing, while the much larger number of days when no such samples were taken would show no change whatever.
It has been suggested that these inferior effluents after washing should be wasted. Such a procedure would mean wasting probably on an average two per cent of the water filtered, and a corresponding increase in the cost of filtering. Mr. Fuller[43] in his Louisville report comes to the conclusion that with adequate washing and coagulation it is unnecessary to waste any effluent, and that inferior results after washing usually indicate incomplete washing. While our experiments certainly indicate a reduction in efficiency after washing so regular and persistent as to make it doubtful whether incomplete washing can be the cause of it, it may be questioned whether or not wasting the effluent would be necessary or desirable in actual operation. At any rate the results as given in this report are not materially influenced by this factor.
INFLUENCE OF AMOUNT OF SULPHATE OF ALUMINA ON BACTERIAL EFFICIENCY OF MECHANICAL FILTERS.
The number of bacteria passing a mechanical filter is dependent principally upon the amount of sulphate of alumina used; and by using a larger quantity of sulphate of alumina than was actually used in the experiments the bacterial efficiency could be considerably increased. To investigate this point, the results obtained each day with each of the mechanical filters were arranged in the order of the sulphate of alumina quantities used, and averaged by classes. In this and the following tables a few abnormal results were omitted.[44] A summary of the results is as follows:
[Pg 166]
| SUMMARY OF RESULTS WITH WARREN MECHANICAL FILTER, ARRANGED ACCORDING TO SULPHATE OF ALUMINA QUANTITIES. | ||||||
|---|---|---|---|---|---|---|
| Number of Days Represented. |
Turbidity. | Bacteria. | Per cent remaining. |
Per cent removed. |
Sulphate of Alumina used Grains per Gallon. |
|
| Raw Water. | Effluent. | |||||
| 7 | 0.05 | 4,773 | 1713 | 35.89 | 64.11 | 0.00 |
| 2 | 0.08 | 2,785 | 850 | 30.52 | 69.48 | 0.12 |
| 4 | 0.10 | 5,109 | 726 | 14.21 | 85.79 | 0.26 |
| 2 | 0.20 | 8,713 | 214 | 2.45 | 97.55 | 0.36 |
| 8 | 0.06 | 3,224 | 112 | 3.47 | 96.53 | 0.44 |
| 19 | 0.06 | 3,488 | 123 | 3.53 | 96.47 | 0.55 |
| 11 | 0.06 | 5,673 | 154 | 2.71 | 97.29 | 0.64 |
| 10 | 0.10 | 6,100 | 112 | 1.84 | 98.16 | 0.74 |
| 8 | 0.09 | 8,647 | 148 | 1.71 | 98.29 | 0.85 |
| 5 | 0.16 | 5,645 | 142 | 2.52 | 97.48 | 0.93 |
| 13 | 0.12 | 10,397 | 200 | 1.92 | 98.08 | 1.07 |
| 10 | 0.08 | 12,778 | 121 | 0.95 | 99.05 | 1.13 |
| 13 | 0.14 | 13,397 | 164 | 1.22 | 98.78 | 1.25 |
| 19 | 0.13 | 10,462 | 160 | 1.53 | 98.47 | 1.34 |
| 10 | 0.12 | 12,851 | 107 | 0.83 | 99.17 | 1.46 |
| 4 | 0.27 | 16,015 | 77 | 0.48 | 99.52 | 1.57 |
| 7 | 0.53 | 12,262 | 191 | 1.18 | 98.82 | 1.64 |
| 4 | 0.58 | 26,950 | 347 | 1.29 | 98.71 | 1.74 |
| 5 | 0.29 | 14,570 | 86 | 0.59 | 99.41 | 1.84 |
| 3 | 0.23 | 13,833 | 153 | 1.11 | 98.89 | 1.92 |
| 19 | 0.40 | 18,222 | 92 | 0.50 | 99.50 | 2.48 |
| 5 | 0.45 | 29,300 | 1119 | 3.82 | 96.18 | 3.37 |
| 5 | 1.06 | 33,030 | 535 | 1.62 | 98.38 | 8.06 |
| SUMMARY OF RESULTS WITH JEWELL MECHANICAL FILTER, ARRANGED ACCORDING TO SULPHATE OF ALUMINA QUANTITIES. | ||||||
| Number of Days Represented. |
Turbidity. | Bacteria. | Per cent remaining. |
Per cent removed. |
Sulphate of Alumina used Grains per Gallon. |
|
| Raw Water. | Effluent. | |||||
| 6 | 0.03 | 14,037 | 6217 | 44.29 | 55.71 | 0.00 |
| 5 | 0.07 | 4,267 | 680 | 15.93 | 84.07 | 0.24 |
| 14 | 0.06 | 2,613 | 170 | 6.50 | 93.50 | 0.35 |
| 10 | 0.06 | 2,446 | 113 | 4.62 | 95.38 | 0.44 |
| 9 | 0.11 | 7,303 | 234 | 3.20 | 96.80 | 0.55 |
| 20 | 0.09 | 6,979 | 220 | 3.15 | 96.85 | 0.65 |
| 9 | 0.08 | 5,191 | 130 | 2.50 | 97.50 | 0.75 |
| 16 | 0.12 | 8,504 | 242 | 2.84 | 97.16 | 0.83 |
| 22 | 0.16 | 8,506 | 99 | 1.16 | 98.84 | 0.96 |
| 12 | 0.11 | 11,998 | 246 | 2.05 | 97.95 | 1.05 |
| 14 | 0.18 | 18,982 | 423 | 2.23 | 97.77 | 1.16 |
| 5 | 0.14 | 13,981 | 224 | 1.60 | 98.40 | 1.23 |
| 9 | 0.27 | 19,806 | 325 | 1.64 | 98.36 | 1.34 |
| 14 | 0.27 | 16,549 | 324 | 1.96 | 98.04 | 1.45 |
| 9 | 0.29 | 12,194 | 96 | 0.79 | 99.21 | 1.54 |
| 6 | 0.25 | 13,483 | 51 | 0.38 | 99.62 | 1.65 |
| 7 | 0.53 | 24,243 | 220 | 0.91 | 99.09 | 1.72 |
| 3 | 0.90 | 20,953 | 602 | 2.88 | 97.12 | 1.90 |
| 5 | 0.43 | 25,958 | 307 | 1.19 | 98.81 | 2.19 |
| 4 | 0.84 | 21,017 | 228 | 1.09 | 98.91 | 3.71 |
[Pg 167]
These results are shown graphically by Fig. 21.
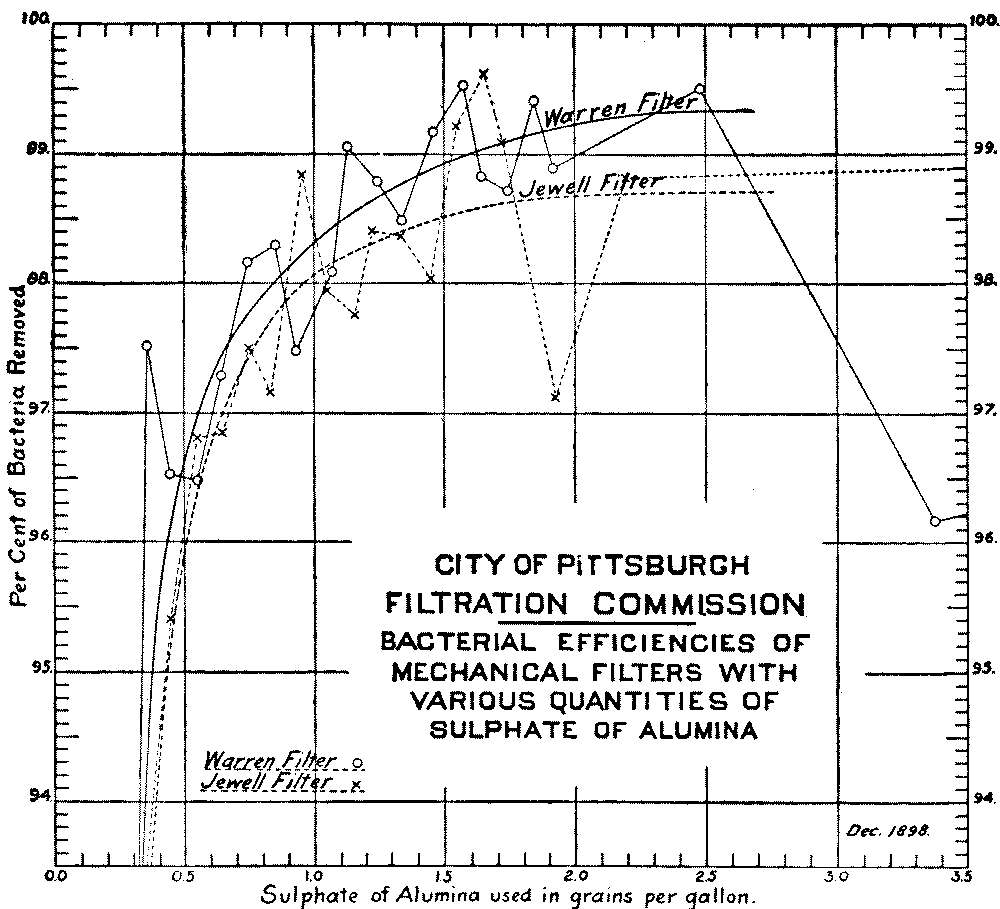
Fig. 21.—Bacterial Efficiencies of Mechanical Filters.
INFLUENCE OF DEGREE OF TURBIDITY UPON BACTERIAL EFFICIENCY OF MECHANICAL FILTERS.
It will be noticed by referring to the tables that as the sulphate of alumina quantities increased the turbidities increased and the numbers of bacteria increased, as well as the bacterial efficiencies. That is to say, with the less turbid waters, small sulphate of alumina quantities have been used, the numbers of bacteria in the raw water have been low, and the bacterial efficiencies have also been low. With turbid waters much larger quantities of sulphate of alumina have been used, the raw water has contained more bacteria, and the bacterial efficiencies have been higher. It may[Pg 168] be then that the increased efficiencies with increased quantities of sulphate of alumina are not due alone to the increased sulphate of alumina, but in part also to other conditions. Thus it may be easier to remove a large percentage of bacteria from a water containing many than from a water containing only a few.
To investigate this matter and eliminate the influence of turbidity and numbers of bacteria in the raw water, the results were first classified with reference to turbidity. The results with waters having turbidities of 0.10 or less, and called for convenience turbid waters, are arranged by alum quantities as before. Afterwards the results obtained with turbidities from 0.11 to 0.50, and called for convenience muddy waters, are grouped; and finally the results with turbid water having turbidities of 0.51 and over, and called for convenience thick waters. The results thus arranged are as follows:
| SUMMARY OF RESULTS WITH WARREN MECHANICAL FILTER, ARRANGED ACCORDING TO TURBIDITIES AND SULPHATE OF ALUMINA QUANTITIES. | ||||||
|---|---|---|---|---|---|---|
| Number of Days Represented. |
Turbidity. | Bacteria. | Per cent remaining. |
Per cent removed. |
Sulphate of Alumina used Grains per Gallon. |
|
| Raw Water. | Effluent. | |||||
| 7 | 0.05 | 4,773 | 1713 | 35.89 | 64.11 | 0.00 |
| 2 | 0.07 | 2,785 | 850 | 30.52 | 69.48 | 0.12 |
| 12 | 0.06 | 3,209 | 224 | 7.00 | 93.00 | 0.42 |
| 31 | 0.06 | 4,238 | 119 | 2.81 | 97.19 | 0.60 |
| 9 | 0.06 | 7,953 | 130 | 1.64 | 98.36 | 0.84 |
| 16 | 0.04 | 11,265 | 137 | 1.22 | 98.78 | 1.11 |
| 29 | 0.06 | 11,500 | 158 | 1.37 | 98.63 | 1.58 |
| 5 | 0.17 | 8,783 | 416 | 4.73 | 95.27 | 0.36 |
| 10 | 0.16 | 6,535 | 165 | 2.54 | 97.46 | 0.85 |
| 13 | 0.19 | 13,253 | 186 | 1.40 | 98.60 | 1.13 |
| 15 | 0.22 | 10,944 | 93 | 0.85 | 99.15 | 1.36 |
| 13 | 0.29 | 14,089 | 112 | 0.80 | 99.20 | 1.73 |
| 10 | 0.35 | 18,088 | 102 | 0.57 | 99.43 | 2.38 |
| 5 | 0.29 | 25,580 | 540 | 2.11 | 97.89 | 4.30 |
| 6 | 0.87 | 25,433 | 369 | 1.45 | 98.55 | 1.74 |
| 6 | 0.73 | 26,566 | 79 | 0.30 | 99.70 | 2.64 |
| 4 | 1.35 | 42,037 | 1388 | 3.30 | 96.70 | 8.16 |
| [Pg 169] SUMMARY OF RESULTS WITH JEWELL MECHANICAL FILTER, ARRANGED ACCORDING TO TURBIDITIES AND SULPHATE OF ALUMINA QUANTITIES. | ||||||
| Number of Days Represented. |
Turbidity. | Bacteria. | Per cent remaining. |
Per cent removed. |
Sulphate of Alumina used Grains per Gallon. |
|
| Raw Water. | Effluent. | |||||
| 6 | 0.03 | 14,037 | 6217 | 44.29 | 55.71 | 0.00 |
| 3 | 0.07 | 5,170 | 991 | 19.15 | 80.85 | 0.21 |
| 25 | 0.05 | 2,403 | 143 | 5.95 | 94.05 | 0.38 |
| 20 | 0.06 | 6,531 | 185 | 2.84 | 97.16 | 0.64 |
| 27 | 0.06 | 5,811 | 122 | 2.10 | 97.90 | 0.88 |
| 14 | 0.06 | 14,978 | 412 | 2.75 | 97.25 | 1.11 |
| 10 | 0.06 | 15,787 | 390 | 2.47 | 97.53 | 1.37 |
| 10 | 0.05 | 10,847 | 47 | 0.43 | 99.57 | 2.17 |
| 14 | 0.16 | 7,525 | 256 | 3.40 | 96.60 | 0.60 |
| 17 | 0.24 | 11,310 | 208 | 1.84 | 98.16 | 0.91 |
| 15 | 0.24 | 15,441 | 262 | 1.70 | 98.30 | 1.13 |
| 10 | 0.28 | 17,842 | 232 | 1.30 | 98.70 | 1.43 |
| 8 | 0.29 | 9,556 | 59 | 0.62 | 99.38 | 1.59 |
| 4 | 0.29 | 20,212 | 135 | 0.67 | 99.33 | 2.00 |
| 5 | 0.66 | 23,680 | 336 | 1.42 | 98.58 | 1.42 |
| 7 | 0.96 | 30,200 | 475 | 1.57 | 98.43 | 1.74 |
| 4 | 1.25 | 37,587 | 496 | 1.32 | 98.68 | 2.81 |
The following table shows the bacterial efficiencies with turbid, muddy, and thick waters, with substantially equal quantities of sulphate of alumina:
| Grains of Sulphate of Alumina. | Corresponding Bacterial Efficiencies. | ||||
|---|---|---|---|---|---|
| Turbid. | Muddy. | Thick. | Turbid. | Muddy. | Thick. |
| WARREN FILTER. | |||||
| 0.42 | 0.36 | 93.00 | 95.27 | ||
| 0.84 | 0.85 | 98.36 | 97.46 | ||
| 1.11 | 1.13 | 98.78 | 98.60 | ||
| 1.58 | 1.73 | 1.74 | 98.63 | 99.20 | 98.55 |
| 2.38 | 2.64 | 99.43 | 99.70 | ||
| 4.30 | 8.16 | 97.89 | 96.70 | ||
| JEWELL FILTER. | |||||
| 0.64 | 0.60 | 97.16 | 96.60 | ||
| 0.88 | 0.91 | 97.90 | 98.16 | ||
| 1.11 | 1.13 | 97.25 | 98.30 | ||
| 1.37 | 1.43 | 1.42 | 97.53 | 98.70 | 98.58 |
| 2.17 | 1.59 | 1.74 | 99.57 | 99.38 | 98.43 |
| 2.00 | 2.81 | 99.33 | 98.68 | ||
[Pg 170]
It appears from this table that waters of various degrees of turbidity give substantially equal bacterial efficiencies with equal quantities of sulphate of alumina, the results varying as often in one direction as the other. Within certain limits it may thus be said that turbidity is without influence upon the bacterial efficiency obtained in mechanical filtration.
It must be borne in mind, however, that the quantities of sulphate of alumina, with very few exceptions, were sufficient to produce full coagulation. Mr. Fuller has shown in his Louisville report that considerable quantities of sulphate of alumina may be added to turbid waters without producing appreciable coagulation; and therefore if a quantity of sulphate of alumina sufficient to produce a certain bacterial efficiency in a clear water should be added to a water so turbid that it was unable to coagulate it, scarcely any effect would be produced. The above statement therefore only applies in those cases where sufficient sulphate of alumina is used to adequately coagulate the water.
As the numbers of bacteria often vary with the turbidity, the variation in the numbers of bacteria in the different classes is much less than in the first tables; but to further investigate the question of whether the numbers of bacteria in the raw water have an important influence upon the bacterial efficiencies, each of the two largest classes in the foregoing tables was divided into two parts, according to the bacterial numbers in the raw water, namely, the results from the Jewell filter with turbid waters and with sulphate of alumina quantities ranging from 0.75 to 1.00 grain per gallon, and the results from the Warren filter with turbid waters and with sulphate of alumina quantities of 1.25 grains per gallon and upward. The results are as follows:
[Pg 171]
| Number of Days Represented. |
Turbidity. | Bacteria. | Per cent remaining. |
Per cent removed. |
Sulphate of Alumina used Grains per Gallon. |
|
|---|---|---|---|---|---|---|
| Raw Water. | Effluent. | |||||
| JEWELL FILTER. | ||||||
| 14 | 0.05 | 3,938 | 81 | 2.06 | 97.94 | 0.88 |
| 13 | 0.07 | 7,827 | 167 | 2.13 | 97.87 | 0.87 |
| WARREN FILTER. | ||||||
| 15 | 0.06 | 3,545 | 59 | 1.66 | 98.34 | 1.67 |
| 14 | 0.06 | 20,022 | 265 | 1.32 | 98.68 | 1.48 |
It will be observed that the bacterial efficiencies are substantially the same, with the lower and with the higher numbers of bacteria in the raw water. That is to say, other things being equal, as the number of bacteria increase in the raw water the number of bacteria in the effluent increase in the same ratio. A further analysis of other groups of results would perhaps show variations in one direction or the other, but on the whole it is believed that the comparison is a fair one, and that there is no well-marked tendency for bacterial efficiencies of mechanical filters to increase or decrease with increasing numbers of bacteria.
AVERAGE RESULTS OBTAINED WITH VARIOUS QUANTITIES OF SULPHATE OF ALUMINA.
As it appears that neither the turbidity nor the number of bacteria in the raw water has a material influence upon the percentage bacterial efficiency obtained, we can take the results given above, which include all the results obtained (except a very few abnormal ones) for computing the various efficiencies obtained with various quantities of sulphate of alumina. These results are graphically shown by Fig. 21, p. 167, on which lines have been drawn indicating the normal efficiencies from various quantities of sulphate of alumina as deduced from our experiments.
In computing the amount of sulphate of alumina which it would be necessary to use in operating a plant at a given place to[Pg 172] give these efficiencies, the quantities of sulphate of alumina shown by the diagram can be taken as those which it would be necessary to use during those days in the year when the raw water was clear, or sufficiently clear, so that the amounts of sulphate of alumina mentioned would suffice to properly coagulate it.
TYPES OF MECHANICAL FILTERS.
Sections of the Warren and Jewell filters used at Pittsburg are presented herewith. The filters here shown are practically identical with those used at Lorain and Louisville, and nearly all the exact information regarding mechanical filters relates to filters of these types. These sections show clearly the constructions used at Pittsburg and Louisville, but there are some points in connection with the designs of these filters which require to be considered more in detail.
The simplest idea of a mechanical filter is a tub, with sand in the bottom and some form of drainage system. Water is run over the sand, passes through it, and is collected by the drainage system. When the sand becomes clogged it is washed by the use of a reverse current of water. This reverse current of water is so rapid as to preclude the use of a drainage system consisting of gravel, tile-drains, etc., such as are used in sand filters operated at lower rates, and instead metallic strainers in some form are used. The sand comes directly against these strainers, which are made as coarse as it is possible to have them, without allowing the sand to pass.
The rate of washing is usually from five to seven gallons per square foot per minute. In the Warren filter the openings in the strainers at the bottom are 6 to 8 per cent of the total area, and during washing the water has an average velocity of 0.20 foot per second upward through them. This velocity is so slow that the friction of the water in passing through the openings in the screen is practically nothing. A result of this is that if there is any[Pg 173] unequal resistance of the sand to the water, the bulk of the water goes up at the points of least resistance in the sand.
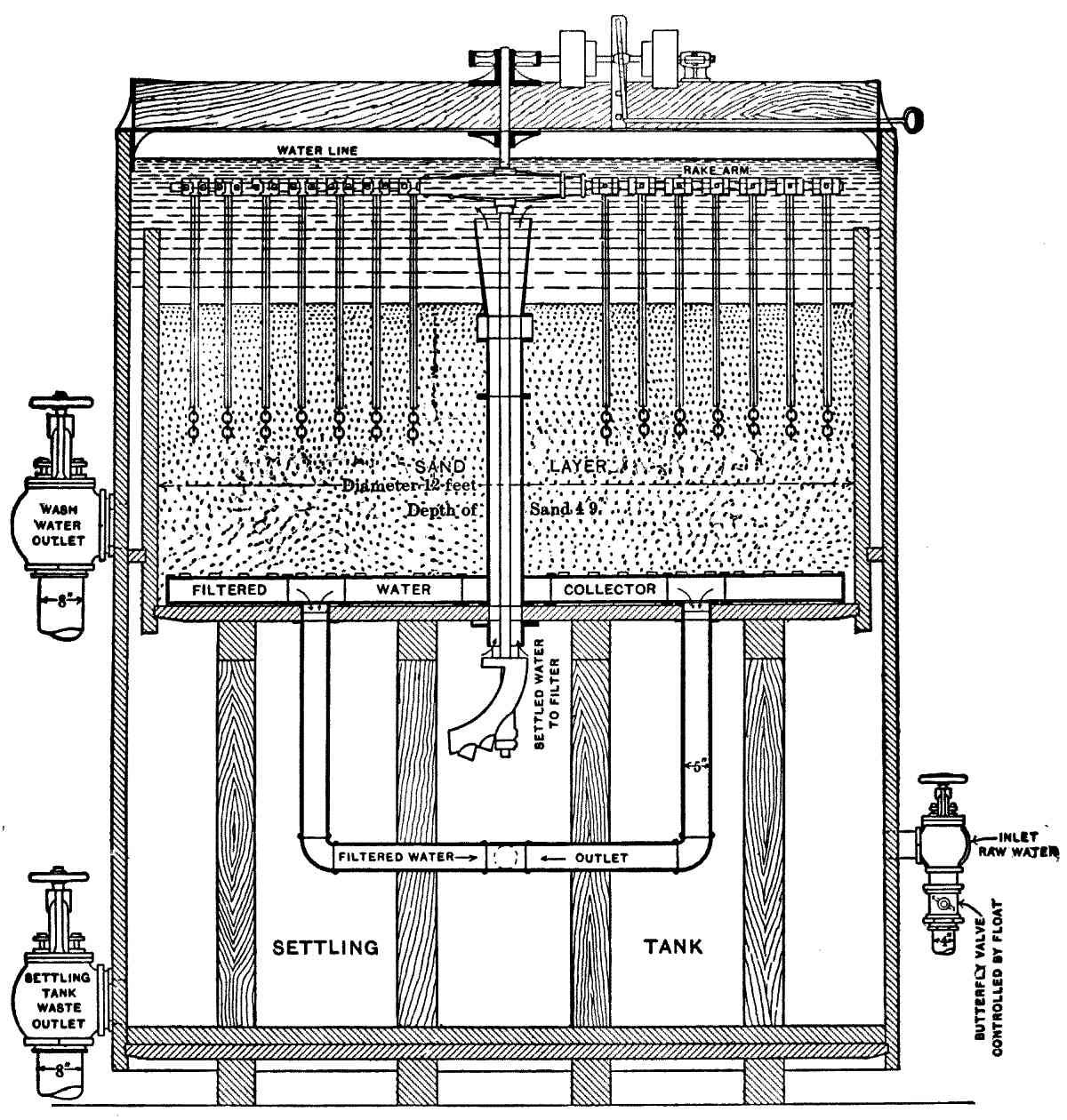
Fig. 22.—Section of Jewell Mechanical Filter used in Pittsburg Experiments.
This tendency would be fatal were it not for the revolving rake which loosens and mixes the sand and largely corrects it. The correction, however, is imperfect, and some parts of the filter are washed more than others.
The rake is also necessary to prevent the separation of sand into coarser and finer particles. It is practically impossible to get[Pg 174] filter sand the grains of which are all of the same size. When a filter is washed the tendency is for the wash water to go up in limited areas. The larger sand grains tend to collect at these points while the finer grains collect in places where there is no upward current, or where it is less rapid. In many filters this tendency is very strong. The revolving rake is necessary to correct it, and to keep the sand thoroughly mixed, otherwise when a filter is put in operation after washing, the frictional resistance through the coarse sand being less, the bulk of the water goes through it, with the result that a part of the area, and the part which is least efficient as a filter, passes nearly all of the water, and with inferior results.
In the Jewell filter provision is made for the distribution of the wash water over the whole area in another way. The strainers have areas at the surface amounting to 1.2 to 1.4 per cent of the whole area, but the water before reaching them passes through throats much smaller in size than the strainer outlets, and amounting in the aggregate to only about 0.07 per cent of the filter area. When washing at a rate of seven gallons per square foot per minute, water passes through these necks at a velocity of 22 feet per second. The friction and velocity head in passing these necks is estimated to be about 30 vertical feet, and is so much greater than the friction of the outlets proper, and of the sand, that the water passes through each strainer with approximately the same velocity, and the wash water is equally distributed over the whole area of the bottom of the filter.
This result is accomplished, however, at a great loss of head in the wash water. When a filter is washed from the pressure-mains without separate pumping, the pressure is usually sufficient and there is no disadvantage in the arrangement. When, however, the water is specially pumped for washing, the required head is much greater than would otherwise be necessary.

Mechanical Filters at Elmira, N. Y. Outlet to Filters with Controller and Pure-water Flume.
[To face page 174.]
It would not be possible to increase the size of the necks, thereby decreasing the friction, without increasing very largely the [Pg 175] size of the pipes in the underdrainage system into which the strainers are fastened. These pipes are so small that during washing the velocity in them is about 13 feet per second, and if the throats of the necks were increased without also enlarging these pipes, the friction would be so reduced that most of the water would go through the necks nearest the supply, thus failing to reach the object to be attained.
A more rational system would be to increase the sizes of all the waterways in the outlet and wash-water system. The Jewell filter is also provided with a rake to keep the sand mixed during washing, as this is necessary even with the complete distribution of wash-water over the area of the filter.
Both the Warren and the Jewell filters are provided with receptacles through which the water passes after receiving the coagulant, and before entering the filter. In the Jewell filter the receptacle, called a sedimentation-basin, is of such size as to hold as much water as is filtered in 15 minutes. In the Warren filter the receptacle is entirely independent and larger, holding about an hour’s supply.
The rates of filtration used in the experiments have ranged from less than 100 to about 130 million gallons per acre daily. To employ a rate much higher than this involves the use of a much coarser sand, or an increase in the height of water upon the filter to an impracticable extent. There would seem to be no material advantage in the use of lower rates within certain limits, while the cost of filters would be greatly increased.
The sand used in the Warren filters has been crushed quartz. In the Jewell filters a silicious sand from Red Wing, Minn., with rounded grains has been used. These sands are somewhat coarser than are commonly used in sand filters, and the uniformity coefficients are very low. It is necessary to use sand with the very lowest uniformity coefficients to avoid the separation of sand particles according to sizes as mentioned above, and for this reason[Pg 178] the sand must be selected with much greater care than is required for sand filters.
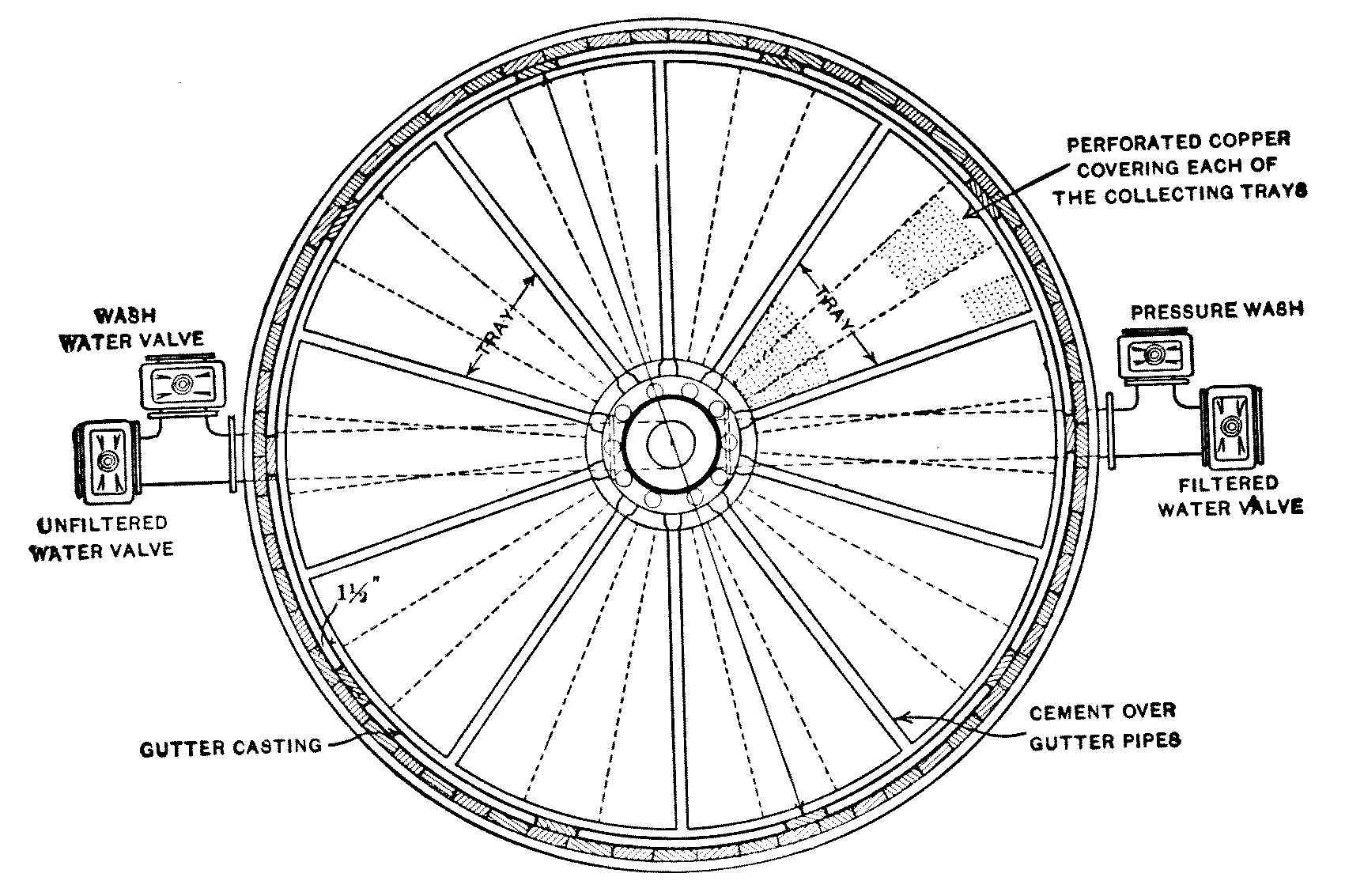
PLAN JUST ABOVE COPPER.
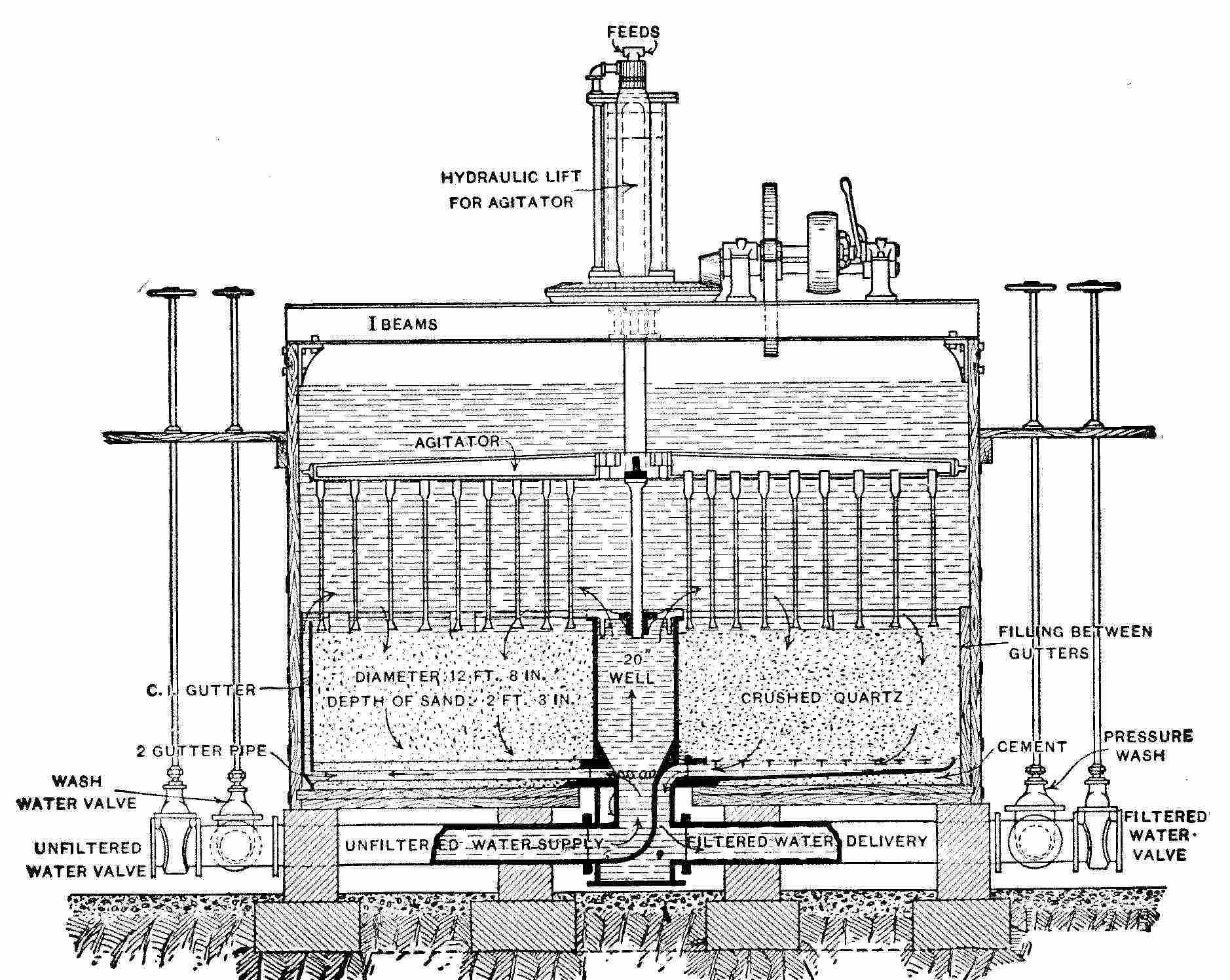
SECTION SHOWING FILTER DURING ORDINARY OPERATION.
Fig. 23.—Warren Filter: Pittsburg Experiments. Section No. 1.
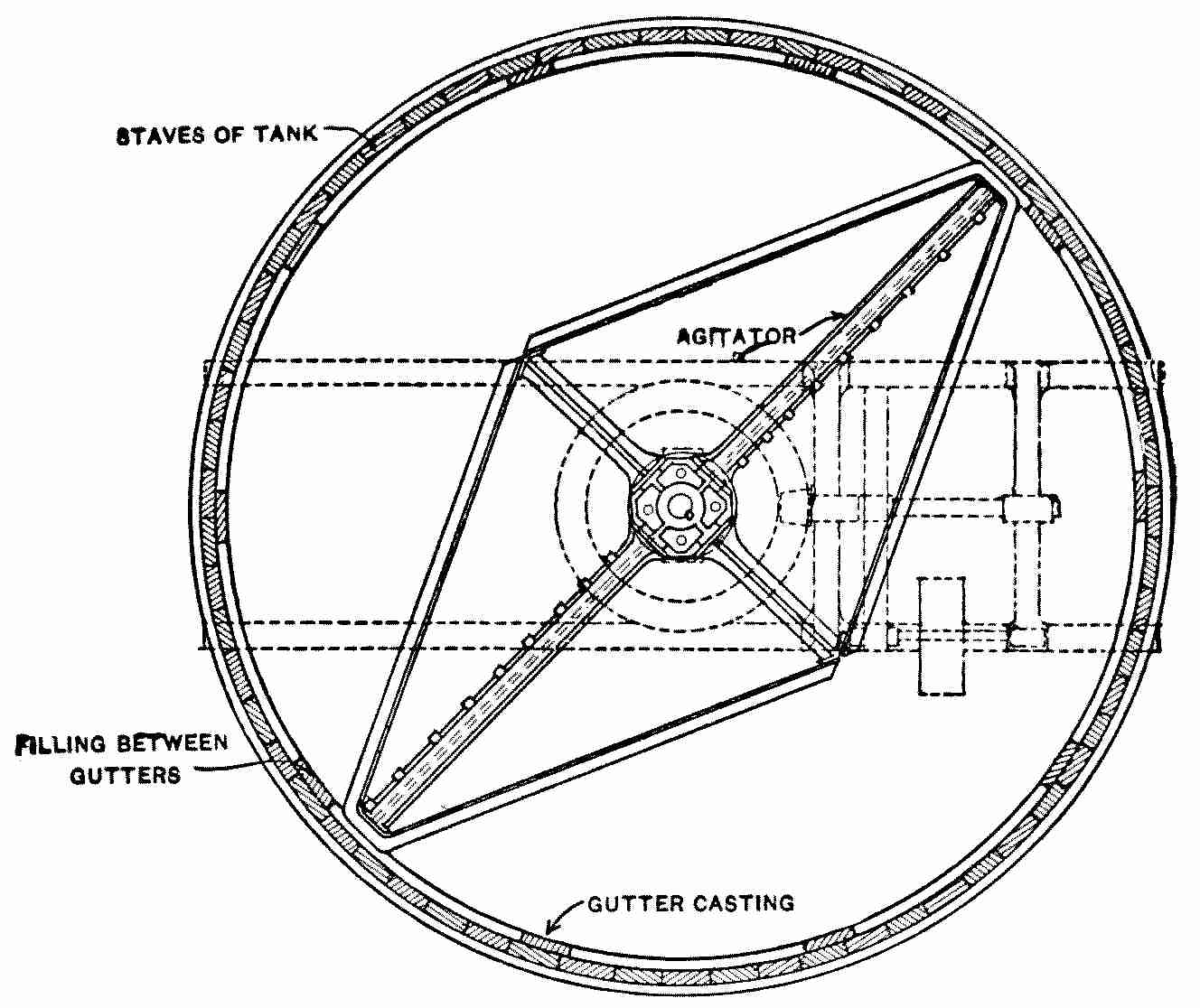
PLAN OF AGITATOR, GUTTER CASTINGS, ETC.
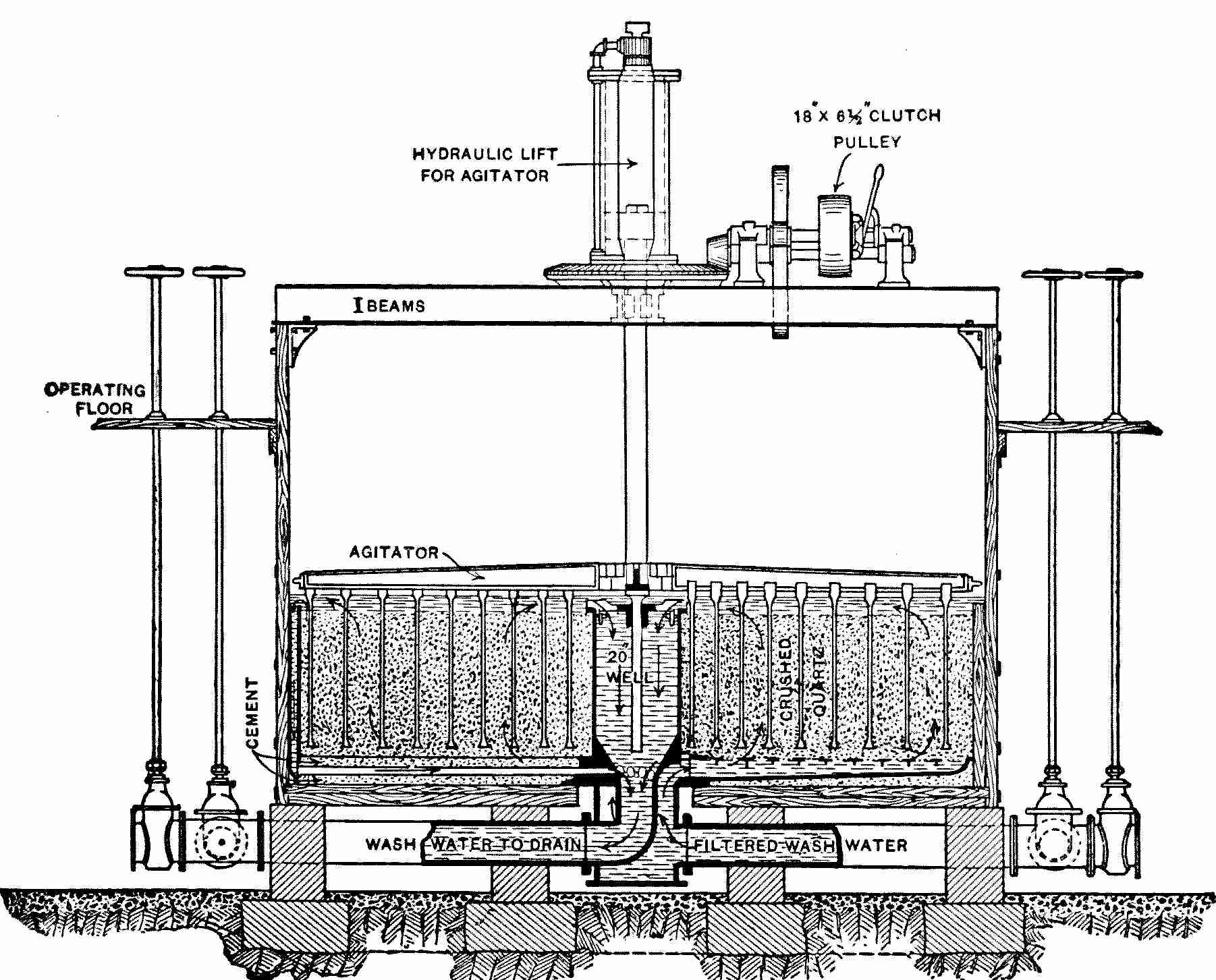
SECTION SHOWING FILTER DURING OPERATION OF WASHING.
Fig. 24.—Warren Filter: Pittsburg Experiments. Section No. 2.
The round-grained sand is more readily and completely washed than the angular crushed quartz. It has been claimed that the crushed quartz is more efficient as a filtering material, but the evidence of this is not very clear.
The amount of water filtered by a filter between washings is, in a general way, about the same as that filtered by a sand filter between scrapings, in relation to its area. The amount of water required for washing is, on an average, about equal to a vertical column 5 or 6 feet high equal in area to the area of the filter, exclusive of water on the top of the filter wasted before the current is reversed. With clear waters, as for instance, the Allegheny at low water, the amount of washing is almost directly proportional to the amount of sulphate of alumina used. With muddy waters the sulphate of alumina required is proportional to the mud, and the frequency of washing and the amount of wash-water are proportional to both. The amount of wash-water required averages about five per cent; with very muddy waters more is required. At Louisville, with the worst waters, the per cents of wash-water rose at times to 30 per cent of the total quantity of water filtered.
The rate of filtration with mechanical filters should be kept as constant as possible, and can be regulated by devices similar to those described in connection with sand filters. Owing to the smaller areas and capacities, the amounts of water to be handled in the units are smaller, and the regulating devices are thus smaller, and have always been made of metal, either cast iron or copper. None of the devices employed in the above-mentioned experiments has been entirely satisfactory in this respect. The devices employed have been too small, and the water has gone through at too high velocities to allow close adjustment.

Mechanical Filters at Elmira, N. Y. Upper Platform and General Arrangement of Filters.
[To face page 178.]
As between the two types of filters, the Jewell filter requires a large loss of head. The water has to be pumped at a sufficient elevation to reach the top of a tank about 18 feet high, while the [Pg 179] effluent must be drawn off at the extreme bottom. The Warren filter is much more economical in head, the plants at Pittsburg and Louisville only requiring about 9 feet from the inlet to the outlet.
The earlier mechanical filters were usually constructed of wrought iron or steel plates. More recently wooden tanks have been commonly employed, although steel is regarded as preferable. Concrete or masonry tanks have been suggested, but they have not as yet been employed.
EFFICIENCY OF MECHANICAL FILTERS.
The efficiency of mechanical filters depends entirely upon the use of coagulants. Without coagulants they can only be used to remove very large particles. The efficiency of the filtration depends much more upon the kind, and amount, and method of application of coagulant than upon the arrangement of the filter. In fact, the arrangements of the filter are more directed to the convenience and economy of operation and washing than towards the efficiency of the results.
The conditions which control the efficiency of mechanical filters have been discussed in connection with coagulation. With sufficient coagulant the removal of turbidity or mud is complete. Color also can be removed with these filters. The bacterial efficiencies secured with them have been discussed at length in connection with the Pittsburg experiments.
With careful coagulation and manipulation it is possible to get 98 per cent bacterial efficiency without difficulty. The results are somewhat irregular, for reasons not as yet fully understood. On some occasions higher bacterial efficiencies are secured with smaller quantities of coagulant, while at other times the efficiencies are less without apparent reason. There seems to be a limit to the bacterial efficiency which can be secured with any amount of sulphate of alumina and rapid filtration, and it is doubtful if a plant could be operated to regularly secure as high a bacterial efficiency as 99 per cent with any amount of sulphate of alumina.
[Pg 180]
PRESSURE FILTERS.
Pressure mechanical filters are constructed in entirely closed receptacles, through which the water is forced under pressure and not by gravity. Many of the earlier mechanical filters were of this type. In small plants this system has the distinct advantage that the water can be pumped from a river or other source of supply through a filter direct to the reservoir or into the mains, while any other system would involve a second pumping. Pressure filters are extensively used for hotel supplies, etc., where, from the conditions, gravity filters are impossible. The practical objections to this system have been found to be so great that it is rarely used under other conditions. Some experiments were made at Louisville with a filter of this type, but they were not long continued, and aside from them there is no precise information as to what can be accomplished with filters of this type.
[Pg 181]
CHAPTER XI.
OTHER METHODS OF FILTRATION.
WORMS TILE SYSTEM.
This system, invented and patented by Director Fischer of the Worms water-works, consists of the filtration of water through artificial hollow sandstone tiles, made by heating a mixture of broken glass and sand, sifted to determined sizes, to a point just below the melting-point of the glass, in suitable moulds or forms. The glass softens and adheres to the sand, forming a strong porous substance through which water can be passed. These tiles are made hollow and are immersed in the water to be treated, the effluent being removed from the centre of each tile. They are connected together in groups corresponding in size to the units of a sand-filtration plant. They are washed by a reverse current of filtered water. These tiles have been used for some years at Worms, Germany, and at a number of smaller places, and were investigated experimentally at Pittsburg. Some difficulty has been experienced in getting tiles with pores small enough to yield an effluent of the desired purity, and at the same time large enough to allow a reasonable quantity of water to pass. In fact, with other than quite clear waters, it has not been found feasible to accomplish both objects at the same time, and it has been necessary to treat the water with coagulants and preliminary sedimentation or filtration before applying it to the tiles. The problem of making the joints between the tiles and the collection-pipes water-tight when surrounded by the raw water also is a matter of some difficulty.
THE USE OF ASBESTOS.
It has been suggested by Mr. P. A. Maignen that the surface of sand filters should be covered with a thin layer of asbestos,[Pg 182] applied in the form of a pulp, with the first water put onto the filter after scraping. The asbestos forms a sort of a paper on the sand which intercepts the sediment of the passing water. The advantage of the process is in the cleaning. When dried to the right consistency this asbestos can be rolled up like a carpet, and taken from the filter without removing any of the sand.
This procedure is almost identical with that which has occurred naturally in iron-removal plants, where algæ grow in the water upon the filters, and form a fibrous substance with the ferric oxide removed from the water, which can be rolled up and removed in the same way as the asbestos. The advantages of the process, from an economical standpoint, are less clear.
FILTERS USING HIGH RATES OF FILTRATION WITHOUT COAGULANTS.
Numerous filters have been suggested, and a few have been constructed for the use of much higher rates of filtration than are usually employed with sand filters, but without the use of coagulants. The results obtained depend upon the requirements and upon the character of the raw water. If a reservoir water contains an algæ growth, it can often be removed by a coarse and rapid filter. The organisms in this case are many times larger than the bacteria, and many times larger than the clay particles which constitute turbidity. The requirements in this case are rather in the nature of straining than of filtration.
The conditions necessary for the removal of bacteria and turbidity are very well understood, and it can be stated with the utmost confidence that no system of filtration through sand at rates many times as high as are used in ordinary sand filtration, and without the use of coagulants, will be satisfactory where either bacterial efficiency or clarification is required. The application of such systems of filtration would therefore seem to be somewhat limited.

Removing Dirty Asbestos Covering from an Experimental Filter. Maignen System.
[To face page 182.]
[Pg 183]
HOUSEHOLD FILTERS.
The subject of household filters is a somewhat broad one, as the variety in these filters is even greater than in the larger filters, and the range in the results to be expected from them is at least as great. I shall only attempt to indicate here some of the leading points in regard to them.
Household filters may be used to remove mud or iron rust from the tap water, or to remove the bacteria in case the latter is sewage-polluted, or to do both at once. Perhaps oftener they are used simply because it is believed to be the proper thing, and without any clear conception either of the desired result or the way in which it can be accomplished. I shall consider them only in their relations to the removal of bacteria, as I credit the people who employ them with being sufficiently good judges of their efficiency in removing visible sediment.
In the first place, as a general rule, which has very few if any exceptions, we may say that all small filters which allow a good stream of water to pass do not remove the bacteria. The reason for this is simply that a material open enough to allow water to pass through it rapidly is not fine enough to stop such small bodies as the bacteria. The filters which are so often sold as “germ-proof,” consisting of sand, animal charcoal, wire-cloth, filter-paper, etc., do not afford protection against any unhealthy qualities which there may be in the raw water. Animal charcoal removes color without retaining the far more objectionable bacteria.
The other household filters have filtering materials of much finer grain, unglazed porcelain and natural sandstone being the most prominent materials, while infusorial earth is also used. The smaller sizes of these filters allow water to pass only drop by drop, and when a fair stream passes them the filters have considerable filtering area (as a series of filter-tubes connected together). On account of their slow action, filters of this class are, as a rule, provided with storage reservoirs so that filtered water to the[Pg 184] capacity of the reservoir can be drawn rapidly (provided the calls do not come too often). Some of these filters are nearly germ-proof, and are comparable in their efficiency to large sand-filters. There is no sharp line between the filters which stop and which do not stop the bacteria; but in general the rule that a filter which works rapidly in proportion to its size does not do so, and vice versa, will be found correct.
In thinking of the efficiency of household filters we must distinguish between the filter carefully prepared for an award at an exhibition and the filter of the same kind doing its average daily work in the kitchen. If we could be sure in the latter case that an unbroken layer of fine sandstone or porcelain was always between ourselves and the raw tap-water we could feel comparatively safe. The manufacturers of the filters claim that leaky joints, cracked tubes, etc., are impossible; but I would urge upon the people using water filtered in this way that they personally assure themselves that this is actually the case with their own filters, for in case any such accident should happen the consequences might be most unpleasant. The increased yield of a filter due to a leaky joint is sure not to decrease it in favor with the cook, who is probably quite out of patience with it because it works so slowly, that is, in case it is good for anything.
The operation of household filters is necessarily, with rare exceptions, left to the kitchen-girl and luck. Scientific supervision is practically impossible. With a large filter, on the other hand, concentrating all the filters for the city at a single point, a competent man can be employed to run them in the best-known way; and if desired, and as is actually done in very many places, an entirely independent bacteriologist can be employed to determine the efficiency of filtration. With the methods of examination now available, and a little care in selecting the times and places of collecting the samples, it is quite impossible for a filter-superintendent to deliver a poor effluent very often or for any considerable length of time without being caught. The safety of properly-conducted central filtration is thus infinitely greater[Pg 185] than that from even the best household filters. Further, it may be doubted whether an infected water can be sent into every house in the city to be used for washing and all the purposes to which water is put except drinking, without causing disease, although less than it would if it were also used for drinking.
The use of household filters must be regarded as a somewhat desperate method of avoiding some of the bad consequences of a polluted water-supply, and they are adopted for the most part by citizens who in some measure realize the dangers from bad water, but who cannot persuade their fellow-citizens to a more thorough and adequate solution of the problem. Such citizens, by the use of the best filters, and by carefully watching their action, or by having their drinking-water boiled, can avoid the principal dangers from bad water, but their vigilance does not protect their more careless neighbors.
[Pg 186]
CHAPTER XII.
REMOVAL OF IRON FROM GROUND-WATERS.
The filtration of ground-waters is a comparatively recent development. Ground-waters are filtered by their passage through soil generally much more perfectly than it is possible to filter other waters, and any further filtration of them is useless. Such waters, however, occasionally contain iron in solution as ferrous carbonate.
Waters containing iron have been used as mineral waters for a very long time. Such waters have an astringent taste, and have been esteemed for some purposes. As ordinary water-supplies, however, they are objectionable. The iron deposits in the pipes when the current is slow, and is flushed out when it is rapid, and makes the water turbid and disagreeable; and still worse, the iron often gets through the pipe-system in solution, and deposits in the wash-tub, coloring the linen a rusty brown and quite spoiling it.
An organism called crenothrix grows in pipes carrying waters containing iron, and after a while this organism dies, and decomposes, and gives rise to very disagreeable tastes and odors. It thus happens that ground-waters containing iron are unsatisfactory as public water-supplies, and are sources of serious complaint.
AMOUNT OF IRON REQUIRED TO RENDER WATER OBJECTIONABLE.
Three hundredths of a part in 100,000 of metallic iron very rarely precipitate or cause any trouble. Five hundredths occasionally precipitate, and this amount may be taken as about the allowable limit of iron in a satisfactory water. One tenth of a part is quite sure to precipitate and give rise to serious complaint. Two or three tenths make the water entirely unsuitable for[Pg 187] laundry purposes, and are otherwise seriously objectionable, and will hardly be tolerated by a community. Under some conditions ground-waters carry as much as 1 part in 100,000 of iron, and such waters are hardly usable. In iron-removal plants an effluent containing less than 0.05 is regarded as satisfactory. One containing less than 0.02, as is the case with many plants, is all that can be desired. The percentage of removal is of no significance, but only the amount left in the effluent.
CAUSE OF IRON IN GROUND-WATERS.
Natural sands, gravels, and rocks almost always contain iron, often in considerable amount. The iron is usually combined with oxygen as ferric oxide, and in this condition it is insoluble in water. Water passing through iron containing materials will not ordinarily take up iron. When, however, the water contains a large amount of organic matter in solution, this organic matter takes part of the oxygen away from the iron, and reduces the ferric oxide to ferrous oxide. The ferrous oxide combines with carbonic acid, always present under these conditions, forming ferrous carbonate, which is soluble and which goes into solution.
Surface-waters nearly always carry free oxygen, and when such waters enter the ground they carry oxygen with them, and the organic matters in the water use up the free oxygen before they commence to take oxygen away from the iron of the ground. It is thus only in the presence of organic matters, and in the absence of free oxygen, that the solution of iron is possible. It sometimes happens that the organic matters which reduce the iron are contained in the soil itself, in which case iron may be taken up even by water originally very pure, as for instance, by rain-water.
Generally speaking, iron is everywhere present in sufficient quantity in the strata from which ground-waters are obtained, and wherever the conditions of the organic matters and oxygen necessary for solution occur, iron-containing waters are secured, and the iron is usually present in the earth in such quantity that the water[Pg 188] can dissolve as much as it will take up for a long series of years, or for centuries, without exhausting the supply. There is thus little prospect of improvement of such waters from exhaustion of the supply of iron.
The circumstances which control the solution of iron are very complicated and difficult to determine. Wells near a river, and drawing their water largely from it by seepage, are apt to yield a water containing iron sooner or later, especially where the river-water carries a large amount of organic matter in solution. Waters drawn from extensive gravel deposits, in which the water is renewed principally by the rainfall upon the surface of the deposits themselves, often remain entirely free from iron indefinitely. The rain-water is almost free from organic matter, and the air is able to take care of decomposing organic matters in the surface soil, and below this there are no accumulations of organic matter sufficient to cause the solution of iron. Under other conditions there are subterranean sources of organic matter which result in the solution of iron under conditions which, on the surface, appear most favorable for securing good water. Wells are often used for many years without developing iron, when suddenly iron will appear. This appearance of iron is often connected with increasing consumption of water. In some cases it may result from drawing water from areas not previously drawn upon.
When iron once makes its appearance in a water, it seldom disappears completely afterward, although it often fluctuates widely at different seasons of the year and under different conditions of pumping. In some cases a decrease in the quantity of iron is noted after a number of years, but in other cases this does not happen.
In a few cases manganese has been found in ground-waters. Manganese in water behaves much like iron, but there are some points of difference, so that the possibility of the presence of this substance should be borne in mind.
Iron-containing waters are generally entirely free from oxygen,[Pg 189] and when first drawn from the ground they are bright and clear and do not differ in appearance from other ground-waters. On exposure to the air they quickly become turbid from the oxidation of the iron, and its precipitation as ferric hydrate. At West Superior, Wisconsin, a water was found containing both iron and dissolved oxygen. It was turbid as pumped from the well. This condition of affairs seemed abnormal, but was repeatedly checked, and the theory was advanced by Mr. R. S. Weston, who made the observations, that it resulted from a mixture in the wells of two entirely different waters, namely, a water resulting from the rainfall on sand deposits back of the wells, containing dissolved oxygen and no iron, and water from the lake which had seeped through the sand, and which contained a considerable amount of iron in solution but no dissolved oxygen. The wells thus drew water from opposite directions, and the two waters were entirely different in character, and the mixture thus had a composition which would not have been possible in a water all of which came from a single source.
TREATMENT OF IRON-CONTAINING WATERS.
The removal of iron from ground-water is ordinarily a very simple procedure. It is simply necessary to aerate the water, by which process the ferrous carbonate is decomposed, and oxidized with the formation of ferric hydrate, which forms a flocculent precipitate and is readily removed by filtration. The aeration required varies in different cases. The quantity of oxygen required to oxidize the iron is only a small fraction of the amount which water will dissolve, and allowing water to simply fall through the air for a few feet in fine streams will usually supply several times as much oxygen as is necessary for this purpose.
Aerating devices of this kind have proved sufficient in a number of cases, as at Far Rockaway, L. I., and at Red Bank, N. J. In some cases, however, a further aeration is necessary, not for the purpose of getting more oxygen into the water,[Pg 190] but to get the excess of carbonic acid out of it. Carbonic acid seems to retard in some way the oxidation of the iron, and it is occasionally present in ground-waters in considerable quantity, and quite seriously interferes with the process. It can be removed sufficiently by aeration, but the necessary amount of exposure to air is much greater than that required to simply introduce oxygen.
Coke-towers have sometimes been used for this purpose. The towers are filled with coarse coke and have open sides, and water is sprinkled over the tops of them and allowed to drip through to the bottoms. In general the simple exposure of water to the air for a sufficient length of time, in any form of apparatus or simply in open channels, will accomplish the desired results.
Mr. H. W. Clark[45] has called attention to the fact that in some cases coke seems to have a direct chemical action upon the water which is entirely independent of its aerating effect. In his experiments there seemed to be some property in the coke which caused the iron to oxidize and flocculate in many cases when it refused to do so with simple aeration and filtration.
When the right conditions are reached the oxidation of the iron is very rapid, and it separates out in flakes of such size that they can be removed by filtration at almost any practicable rate. Mechanical filters have been used for this purpose, with rates of filtration of 100 million gallons per acre daily. In Germany, where plants for the removal of iron are quite common, modified forms of sand filters have usually been employed which have been operated at rates up to 25 million gallons per acre daily.
In experiments made by the Massachusetts State Board of Health rates from 10 to 25 million gallons per acre daily have been employed.
The sand used for filtration may appropriately be somewhat coarser than would be used for treating surface-waters, and the thickness of the sand layer may be reduced. Owing to the higher[Pg 191] rates the underdrainage system must be more ample than is otherwise necessary.
The rate of filtration employed is usually not a matter of vital importance, but by selecting a rate that is not too high it is possible to use a moderate loss of head. It is thus not necessary to clean the filters too often, and the expenses of operation are not as high as with an extreme rate. In some cases it is desired to accomplish other results than the removal of iron by filtration, and this may lead to the selection of a rate lower than would otherwise be used.
Under normal conditions of operation all of the iron separates on the top of the sand. No appreciable amount of it penetrates the sand at all. With open filters at Far Rockaway and at Red Bank there is an algæ growth in the water upon the filters which, with the iron, forms a mat upon the surface of the filter; and when the filter is put out of service and allowed to partially dry, this mat can be rolled up like a carpet and thrown off without removing any sand, and the filters have been in use for several years without renewing any sand and without any important decrease in the thickness of the sand layer.
Some waters contain iron in such a form that it cannot be successfully removed in this manner. Thus at Reading, Mass., it was reported by Dr. Thomas M. Drown that the iron was present in the form of ferrous sulphate instead of ferrous carbonate, and that it was not capable of being separated by simple aeration and filtration. A Warren mechanical filter was installed, and the water is treated by aeration and with the addition of lime and alum. The cost of the process is thereby much increased, and the hardness of the water is increased threefold.
Several other cases have been reported where it was believed that simple aeration and filtration were inadequate; but the advantages of the simple procedure are so great as to make it worth a very careful study to determine if more complete aeration, or the use of coke-towers and perhaps slower filtration, would not serve[Pg 192] in these cases without resorting to the use of chemicals and their attendant disadvantages.
IRON-REMOVAL PLANTS IN OPERATION.
Iron-removal plants are now in use at Amsterdam and The Hague in Holland, at Copenhagen in Denmark, at Kiel, Charlottenburg, Leipzig, Halle, and many other places in Germany; at Reading, Mass.; Far Rockaway, L. I.; Red Bank, Asbury Park, Atlantic Highlands, and Keyport, N. J.
Among the earliest plants for the removal of iron were the filters constructed at Amsterdam and The Hague in Holland. At Amsterdam the water is derived from open canals in the dunes draining a large area. The water has its origin in the rain-water falling upon the sand. The sand is very fine and contains organic matter in sufficient amount so that the ground-water is impregnated with iron. In flowing to a central point in the open canals the water becomes aerated and the iron oxidized. There are also algæ growths in the water which perhaps aid the process. Sand filters of ordinary construction are used, and remove both the iron and the algæ, and the rate of filtration is not higher than is usually used in the treatment of river-waters, although it could probably be largely increased without detriment to the supply.
The works at The Hague are very similar to those at Amsterdam, but covered collectors are used to supplement the open canals. Both of these plants were built before much was known about iron in ground-waters and the means for its removal, but they have performed their work with uniformly satisfactory results. In the more recent German works various aerating devices are employed, and filters similar in general construction to ordinary sand filters, but with larger connections suited to very high rates of filtration, are employed.
The plant at Asbury Park was the first of importance constructed in America. The water is raised from wells from 400 to 1100 feet deep by compressed air by a Pohle lift. It is delivered[Pg 193] into a square masonry receiving-basin holding some hours’ supply. The aeration of the water by this means is very complete. It is afterwards pumped through Continental pressure filters direct into the service-pipes. The reservoir for the aerated water was not a part of the original plant, but was added afterwards to facilitate operation, and to give more complete aeration before filtration.
At Far Rockaway, L. I., the water is lifted from wells by a Worthington Pump, and is discharged over the bell of a vertical 16-inch pipe, from which it falls through the air to the water in a receiving chamber around it. The simple fall through the air aerates the water sufficiently. From the receiving-chamber the water is taken to either or both of two filters, each with an area of 20,000 square feet. These filters are open, with brick walls and concrete bottoms, three feet of sand and one foot of gravel, and the underdrains are of the usual type. The water flows through regulator-chambers to a well 25 feet in diameter and 12 feet deep, from which it is pumped to a stand-pipe in the town. The plant was built to treat easily three million gallons per day, and has occasionally treated a larger quantity. Either filter yields the whole supply while the other is being cleaned. The rate of filtration in this case was made lower than would have otherwise been necessary, as there was an alternate supply, namely, the water from two brooks, which could be used on occasions, and to purify which a lower rate of filtration was regarded necessary, than would have been required for the well-water. The removal of iron is complete.
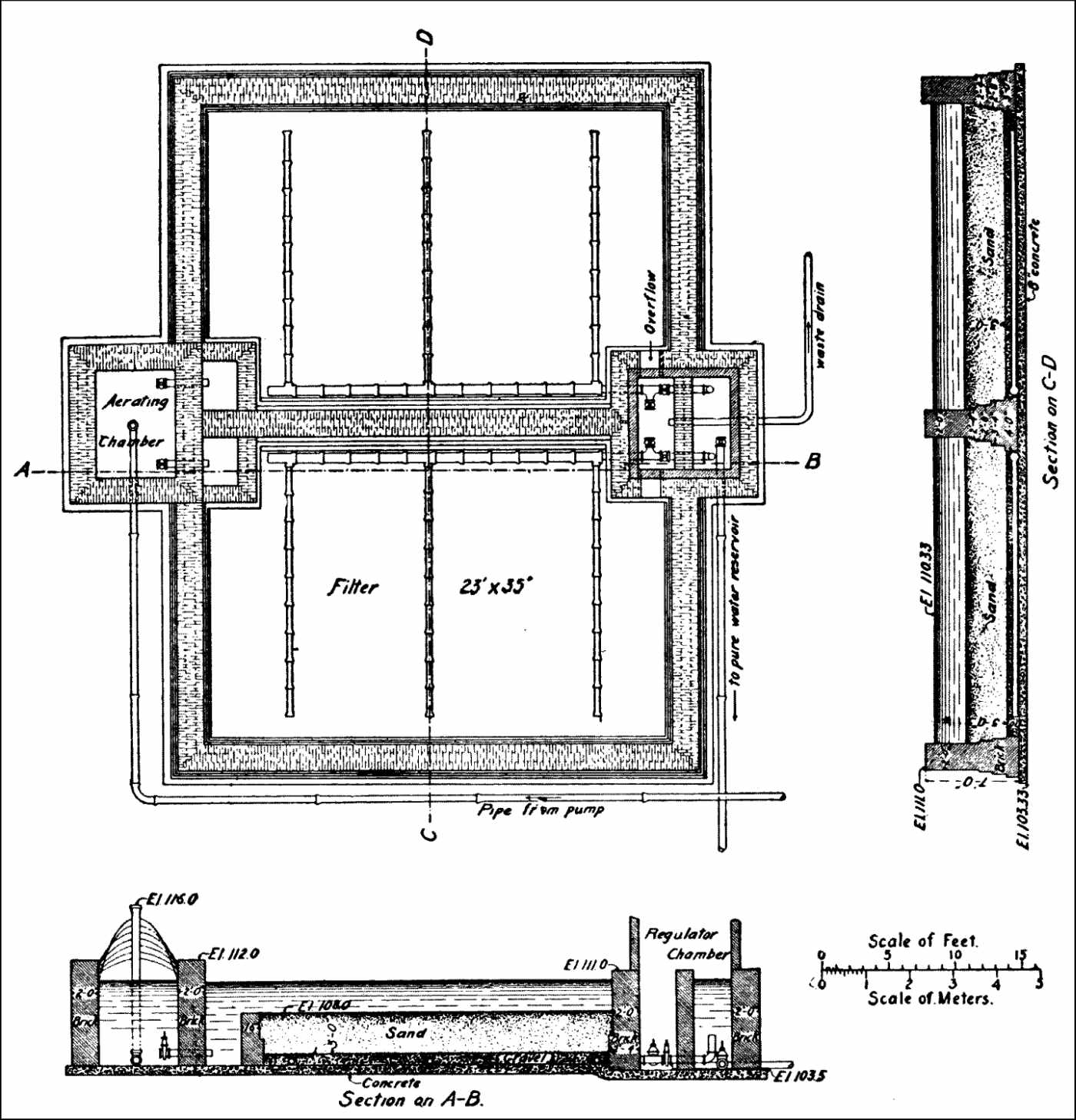
Fig. 25.
The plant of the Rumson Improvement Company at Red Bank, N. J., is quite similar to that at Far Rockaway, but is much smaller. The outlet is a 6-inch pipe perforated with 1⁄4-inch holes which throws the water out in a pine-tree shape to the receiving-tank, thoroughly aerating it. Each of the two filters has 770 square feet of area. The filtering material is three feet of beach sand. From the regulator-chamber the water flows to a circular well 18 feet in diameter, covered by a brick dome and holding[Pg 194] 17,000 gallons, from which it is pumped to the stand-pipe. Either of the filters will treat ten thousand gallons of water per hour, which is equal to the capacity of the pumps; and as the consumption[Pg 195] is considerably less than this figure, they are only in use for a part of each day, the number of hours depending upon the consumption. These filters are shown by the accompanying plan. The cost of the work was as follows:
Filters and pure-water reservoir, with piping and drains complete |
$3,799.47 |
New pump and connections |
492.68 |
Engineering and superintendence |
992.91 |
| Total cost of plant | $5,285.06 |
The engineer who operates the pumps takes care of the filters, and no additional labor has been required. The entire cost of operation is thus represented by the additional coal required for the preliminary lift from the wells to the filters. The effluent is always free from iron.
The plant at Reading,[46] Mass., was installed by the Cumberland Manufacturing Company, and combines aeration, treatment with lime and sulphate of alumina and rapid filtration. The aeration is effected by pumping air through the water, after the water has received the lime. It afterwards receives sulphate of alumina and passes to a settling-tank holding 40,000 gallons, in which the water remains for about an hour. There are six filters of the Warren type, each with an effective filtering area of 54 square feet.
The cost of coagulant is considerable. The chief disadvantage of the process is that it hardens the water, which is naturally soft. From the completion of the plant in July, 1896, to the end of the year the hardness of the water was increased, according to analyses of the State Board of Health, from 4.1 to 11.3 parts in 100,000, and for the year 1897 the increase was from 4.0 to 12.7. The iron, which is present in the raw water to the extent of about 0.26 part in 100,000, is removed sufficiently at all times.
[Pg 196]
Prior to the erection of this plant Mr. Desmond FitzGerald advised aeration followed by sedimentation in two reservoirs holding half a million gallons each, and by rapid filtration. Mr. Bancroft states that in his opinion, if the reservoir recommended by Mr. FitzGerald had been built, the filters could be run with very little or no coagulation, and consequently without increase in hardness, which is the most obvious disadvantage to the procedure. The nominal capacity of the plant is one million gallons, and the average consumption about 200,000 gallons daily.
The plant at Keyport, N. J., is similar, but smaller.
[Pg 197]
CHAPTER XIII.
TREATMENT OF WATERS.
Having now reviewed the most important methods in use for the treatment of waters, we may take a general view of their application to various classes of waters. Different raw waters vary so much, and the requirements of filtration are so different, that it is not possible to outline any general procedure or combination of procedures, but each problem must be taken up by itself. Nevertheless, some general suggestions may be of service.
In the first place, we may consider the case of waters containing very large quantities of oxidizable organic matter. Such waters are obtained from some reservoirs containing very active vegetable and animal growths, or from rivers receiving large amounts of sewage. Waters of both of these classes are, if possible, to be avoided for public water-supplies. When circumstances require their use, they can best be treated by intermittent filtration, this process being best adapted to the destruction by oxygen of excessive quantities of organic matter.
Where the pollution is less, so that the dissolved oxygen contained in the raw water is sufficient for the oxidation of the organic matters, continuous filtration will give substantially as good results as intermittent filtration, and in other respects it has important advantages. The application of intermittent filtration for the treatment of public water-supplies is thus somewhat limited, and, as a matter of fact, it has been used in only a few cases.
For the treatment of very highly polluted waters double filtration has been used in a number of cases, notably by the Grand Junction Company at London, at Schiedam in Holland, and at Bremen and Altona in Germany. At the two first-mentioned[Pg 198] places two separate systems of filters are provided differing somewhat in construction, the first filters being at a higher level than the after filters. The first filters supply water of comparative purity, and very constant composition, to the after filters, which are able to treat it with great efficiency and at very low operating cost.
This procedure is probably the most perfect which has been used for the removal of disease-producing qualities from highly polluted waters; and the cost of the process may not be as much greater than that of simple filtration as would at first appear, because the cost of cleaning the after filters is merely nominal, and the attendance, pumping, etc., are practically common to both sets of filters, and are not materially greater than they would be for a single set.
For very bad waters the first filters might appropriately be intermittent, while the after filters should be continuous. This was the procedure originally intended for Lawrence, but the intermittent filter first constructed yielded such very good results that it has not been considered necessary to complete the plant as originally projected.
At Bremen and at Altona a different procedure has been adopted. The filters are all upon the same level, and of the same construction. When a filter is put in service the effluent from it, instead of being taken to the pure-water reservoir, is taken to another filter which has already been some time in service. After the first filter has been in operation for some time its effluent is taken to the pure-water reservoir, and in turn it is supplied with the effluent from a filter more recently cleaned. The loss of head of water passing a freshly cleaned filter is comparatively slight, and the water of the second filter is allowed to fall a few inches below the high-water mark, at which level it will take the effluent from the other filter. The connections between the filters are made by siphons of large pipe, the summits of which are considerably above the high-water line. These siphons are filled by exhausting the[Pg 199] air, and when opened to the air there is no possibility of a flow of water through them. The process has given extremely good results in practice, yielding effluents of the very greatest purity and at a quite moderate cost of operation.
An objection to the method is the possible filling of a siphon some time when the water standing upon the after-filter is higher than that in the pure-water well of the fore-filter, and while the fore-filter is connected with the pure-water reservoir. Such a connection would send unfiltered water into the pure-water reservoir direct. I do not know that any trouble of this kind has ever been experienced at Bremen or at Altona; and the objection to this system is perhaps not well founded where the management is careful and conscientious. The fact that an unscrupulous attendant can make the connection at any time to help out a deficiency of supply, or simply through carelessness, is certainly objectionable.
For the treatment of river-waters and lake-waters containing only a small quantity of sediment, and where the removal of bacteria or disease-producing qualities is the most important object of filtration, sand filters can be used. Where the rivers are subject to floods and moderate amounts of muddy water, sedimentation-basins or storage reservoirs for raw water will often be found advantageous.
For the treatment of extremely muddy waters, and waters which are continuously muddy for long periods of time, and for the removal of color from very highly colored waters, resource must be had to coagulants. The coagulants which are necessary in each special case and which can be used without injury to the water must be determined by most careful investigation of the raw water.
For the filtration of these waters after coagulation either sand or mechanical filters can be employed. As the principal work in this case is done by the coagulant, the kind of filtration employed is of less consequence than where filtration alone is relied upon,[Pg 200] and the cheapest form of filter will naturally be employed. Under present conditions mechanical filters will usually be cheaper than sand filters for use in this way; but where waters, in addition to the mud, carry bacteria in such large numbers as to make high bacterial efficiency a matter of importance, sand filters may be selected, as the bacterial efficiency obtained with them is not dependent upon the use of coagulant; and is therefore less subject to interruptions from the failure to apply coagulant in the right proportion.
Mechanical filters have also been used for the treatment of comparatively clear waters where bacterial efficiency was the principal object of filtration. For this purpose the efficiencies obtained with them are usually inferior to those obtained with sand filters, while the cost of coagulants is so great as to make their use often more expensive than that of sand filters.
In the case of many streams which are comparatively clear for a part of the year, but occasionally are quite turbid, the use of sand filters has this advantage, that the use of coagulants can be stopped and the cost of operation reduced whenever the water is clear enough to allow of satisfactory treatment by them; and that coagulant can be employed on those days when otherwise insufficient clarification would be obtained.
In this case the high bacterial efficiency is secured at all times, while the cost of coagulant is saved during the greater part of the time. In such cases, also, the preliminary process of sedimentation and storage should be developed as far as possible.
The application of other processes of filtration to special problems are not sufficiently well understood to allow general discussion, and must be taken up separately with reference to the requirements of each special situation.
COST OF FILTRATION.
The cost of filtration of water depends upon the character of the raw water, upon the nature of the plant employed, upon its[Pg 201] size, and upon the skill and economy of manipulation. These conditions affect the cost to such an extent as to make any accurate general estimate quite impossible. Nevertheless a little consideration of the subject, although not leading to exact results, may be helpful as furnishing a rough idea of the probable cost before estimates for local conditions are made.
Open sand filters, with masonry walls, with reasonably favorable conditions of construction, and not too small in area, have averaged to cost in the United States within the last few years perhaps about thirty thousand dollars per acre. The relative cost of small plants is somewhat greater, and with embankments instead of masonry walls, the cost is somewhat reduced. The cost is less where natural deposits of sand can be made use of practically in their original condition, and is increased where the filtering materials have to be transported by rail for long distances, or where the sites are difficult to build upon. Covered filters cost about a half more than open filters. Mechanical filters at current prices cost about $20 per square foot of filtering area, to which must be added the cost of foundations and buildings, which perhaps average to cost half as much more, but are dependent upon local conditions and the character of the buildings.
To these figures must be added the costs of pumps, reservoirs, sedimentation-basins, and pipe-connections, which are often greater than the costs of the filters, but which differ so widely in different cases as to make any general estimate impossible.
Filters must be provided sufficient to meet the maximum and not the average consumption. The excess of maximum over average requirements varies greatly in different cities, and depends largely upon reservoir capacities and arrangements.
As a result of a considerable number of estimates made by the author for average American conditions, the cost of installing filters may be taken very roughly as five dollars per inhabitant, but the amounts differ widely in various cases.
The cost of operation of sand filters in England probably[Pg 202] averages about one dollar per million gallons of water filtered. The following table shows the costs of operation of the filters of the seven London companies for fifteen years, compiled in the office of Mr. W. B. Bryan, Chief Engineer of the East London Water Company. The results have been computed to dollars per million U. S. gallons, and include the cost of all labor, sand, and supplies for the filters, but do not include any pumping or interest costs.
| COST OF FILTRATION, LONDON WATER COMPANIES. | ||||||||
|---|---|---|---|---|---|---|---|---|
| (Computed from data furnished Wm. B. Bryan, C.E., East London Water Works.) | ||||||||
| Dollars per Million U. S. Gallons. | ||||||||
| Chelsea Co. |
East London Co. |
Grand Junction Co. |
Lambeth Co. |
New River Co. |
Southwark & Vauxhall Co. |
West Middlesex Co. |
Average. | |
| 1880-1 | 1.16 | 1.16 | 1.00 | 0.83 | 1.34 | 1.16 | 1.67 | 1.19 |
| 1881-2 | 1.19 | 1.39 | 0.95 | 0.82 | 1.15 | 1.37 | 1.54 | 1.20 |
| 1882-3 | 1.10 | 1.23 | 1.39 | 0.96 | 1.40 | 1.47 | 1.74 | 1.33 |
| 1883-4 | 1.00 | 1.06 | 1.73 | 0.92 | 1.11 | 1.62 | 1.67 | 1.30 |
| 1884-5 | 1.06 | 1.06 | 1.82 | 0.90 | 1.02 | 1.40 | 1.30 | 1.22 |
| 1885-6 | 1.15 | 1.16 | 1.35 | 0.90 | 1.00 | 1.15 | 1.07 | 1.11 |
| 1886-7 | 0.80 | 0.96 | 1.39 | 0.87 | 0.98 | 1.43 | 1.70 | 1.16 |
| 1887-8 | 1.07 | 1.22 | 1.74 | 0.90 | 0.92 | 1.28 | 1.00 | 1.16 |
| 1888-9 | 0.83 | 1.28 | 1.55 | 0.95 | 0.98 | 1.52 | 0.83 | 1.13 |
| 1889-90 | 0.66 | 1.50 | 1.22 | 0.88 | 0.90 | 1.70 | 3.56 | 1.49 |
| 1890-1 | 0.72 | 1.42 | 1.32 | 0.85 | 1.02 | 1.16 | 1.00 | 1.07 |
| 1891-2 | 0.75 | 1.54 | 1.23 | 1.00 | 0.92 | 1.15 | 0.96 | 1.08 |
| 1892-3 | 0.67 | 1.42 | 1.30 | 1.19 | 1.16 | 1.26 | 1.42 | 1.20 |
| 1893-4 | 1.15 | 2.63 | 2.00 | 1.46 | 1.43 | 1.52 | 0.95 | 1.59 |
| 1894-5 | 0.60 | 1.68 | 1.67 | 2.53 | 1.03 | 1.34 | 0.96 | 1.40 |
| Average | 0.93 | 1.38 | 1.44 | 1.06 | 1.09 | 1.37 | 1.43 | 1.24 |
Average of seven companies for 15 years, $1.24 per million gallons. |
||||||||
Variations from year to year are caused by differences in the amounts of ice, and in the quantities of new sand purchased. Wages average about $1.00 per day. At Liverpool for 1896 the cost was $1.08 per million U. S. gallons. |
||||||||
In Germany, with more turbid river-waters, the costs of operation are somewhat higher than the London figures, while at Zürich, where the water is very clear, they are lower.
In the United States the data regarding the cost of operation of sand filters are less complete. At Mt. Vernon, N. Y., with[Pg 203] reservoir-water, the cost has averaged about two dollars per million gallons. At Poughkeepsie, N. Y., with the Hudson River water, which is occasionally moderately turbid, the cost for twenty years has averaged three dollars per million gallons. This cost includes the cost of handling ice, and as the average winter temperature is considerably below that suggested for open filters, the expense of this work has been considerable, and has increased considerably the total cost of operation.
At Far Rockaway, L. I., and Red Bank N. J., for iron-removal plants, the cost of operation has hardly been appreciable. The plants are both close to the pumping-stations, and it has been possible to operate them with the labor necessarily engaged at the pumping-station without additional cost, except a very small amount of labor on the sand at Far Rockaway. No computation has been made in these cases of the additional coal required for pumping.
At Lawrence, Mass., the cost of operation for 1895 was as follows:
Cost of scraping and replacing sand |
$3,467 | |
Cost of care of ice |
2,903 | |
Total cost of operation |
$6,370 | |
Water filtered, millions of gallons |
1,097 | |
Cost per million gallons |
$5.80 |
The cost of care of ice has been excessive at Lawrence, and it has been repeatedly recommended to cover the filter to avoid this expense. The cost of handling sand has been very greatly increased, because the filter is built in one bed, and all work upon it has to be done during the comparatively short intervals when the filter is not in use, an arrangement which is not at all economical in the use of labor. The cost of operation is thus much higher than it would be had the plant been constructed in several units, each of which could be disconnected for the purpose of being cleaned in the ordinary manner. As against this the first cost[Pg 204] of construction was extremely low, and the saving in interest charges should be credited against the increased cost of labor in cleaning.
The cost of operating filters at Ashland, Wis., has been estimated by Mr. William Wheeler at $2.26 per million gallons. This estimate is based upon the performance for the first year that they were in service.
In the operation of mechanical filters one of the largest items of expense is for the coagulant, and the amount of this depends entirely upon the character of the raw water and the thoroughness of the treatment required. The data regarding the other or general costs of operation of mechanical filters are few and unsatisfactory.
I recently made some estimates of cost of clarifying waters of various degrees of turbidity by sand and mechanical filters. These estimates were made for a special set of conditions, and I do not know that they will fit others, but they have at least a suggestive value. The results shown by Fig. 26 include only the cost of operation, and not interest and depreciation charges. These figures, when used for plants in connection with which preliminary treatments are used, should be applied to the turbidity of the water as applied to the filters, and not to the raw water, and the costs of the preliminary processes should be added.
With sand filters the frequency of scraping is nearly proportional to the turbidity; and as scraping represents most of the expenses, the costs of operation are proportional to the turbidity, except the general costs, and the cost of the amount of scraping, which is necessary with even the clearest waters.
With mechanical filters the amount of sulphate of alumina required for clarification increases with the turbidity, and most of the costs of operation increase in the same ratio. The diagram shows the amount of sulphate of alumina in grains per gallon necessary for clarification with different degrees of turbidity.
[Pg 205]
With the clearest waters the costs of operation on the two systems are substantially equal. With muddy waters, the expense of operating sand filters increases more rapidly than the expense of operating mechanical filters.
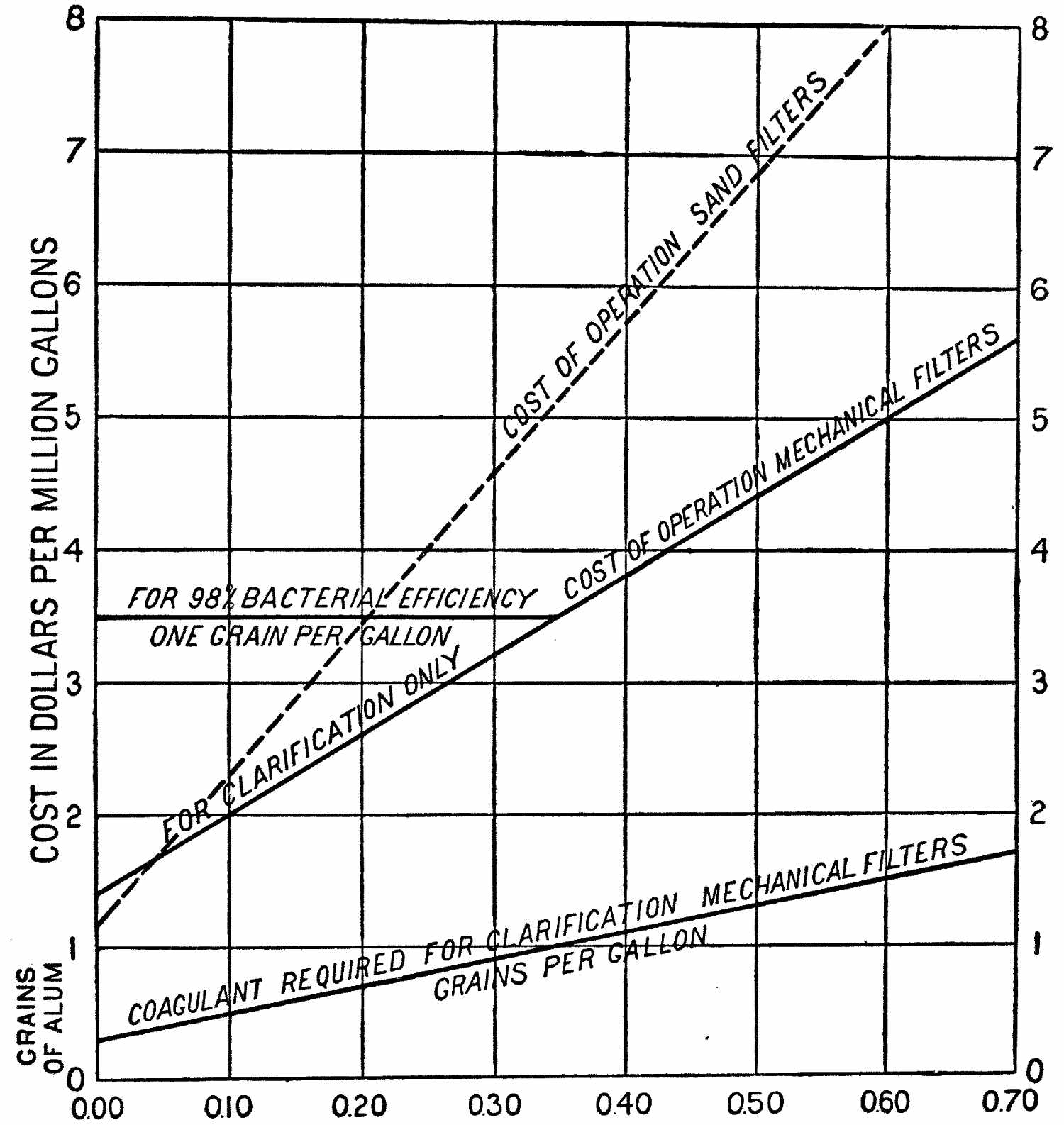
TURBIDITY
Fig. 26.—Cost of Operation of Filters.
There is another element which often comes into the comparison, namely, the question of purification from the effects of sewage-pollution. Nearly all rivers used for public water-supplies[Pg 206] receive more or less sewage, and in filtering such waters it is regarded as necessary to remove as completely as possible the bacteria.
The quantities of sulphate of alumina required for the clarification of the least turbid waters are not sufficient to give even tolerably good bacterial efficiencies. To secure a reasonably complete removal of bacteria with mechanical filters, the use of a considerable quantity of sulphate of alumina is required. Let us assume that 98 per cent bacterial efficiency is required, and that to produce this efficiency it is necessary to use one grain of coagulant to the gallon. With water requiring less than this quantity of coagulant for clarification this quantity must nevertheless be used, and the costs will be controlled by it, and not by the lower quantities which would suffice for clarification, but would not give the required bacterial efficiency.
I have added this line to the diagram, and this, combined with the upper portion of the line showing cost of clarification, represents the cost of treating waters with mechanical filters, where both bacterial efficiency and clarification are required.
This line, considered as a whole, increases much less rapidly with increasing turbidity than does the corresponding line for sand filters, and the two lines cross each other. With the clearest waters sand filters are cheaper than mechanical filters, and for the muddiest waters they are more expensive. It does not appear from the diagram, but it is also true in each case, that the cheaper system is also the more efficient. Sand filters are more efficient in removing bacteria from clear waters than are mechanical filters, and mechanical filters are more efficient in clarifying very muddy waters than are sand filters.
[Pg 207]
WHAT WATERS REQUIRE FILTRATION?
From the nature of the case a satisfactory general answer to this question cannot be given, but a few suggestions may be useful.
In the first place, ground-waters obviously do not require filtration: they have already in most cases been thoroughly filtered in the ground through which they have passed, and in the exceptional cases, as, for instance, an artesian well drawing water through fissures in a ledge from a polluted origin, a new supply will generally be chosen rather than to attempt to improve so doubtful a raw material.
River-waters should be filtered. It cannot be asserted that there are no rivers in mountainous districts in which the water is at once clear and free from pollution, and suitable in its natural state for water-supply; but if so, they are not common, least of all in the regions where water-supplies are usually required. The use of river-waters in their natural state or after sedimentation only, drawn from such rivers as the Merrimac, Hudson, Potomac, Delaware, Schuylkill, Ohio, and Mississippi, is a filthy as well as an unhealthy practice, which ought to be abandoned.
The question is more difficult in the case of supplies drawn from lakes or storage reservoirs. Many such supplies are grossly polluted and should be either abandoned or filtered. Others are subject to algæ growths, or are muddy, and would be much improved by filtration. Still others are drawn either from unpolluted water-sheds, or the pollution is so greatly diluted and reduced by storage that no known disadvantage results from their use.
In measuring the effects of the pollution of water-supplies, the typhoid-fever death-rate is a most important aid. Not that typhoid fever is the sole evil resulting from polluted water, but because it is also a very useful index of other evils for which corresponding statistics cannot be obtained, as, for instance, the causation of diarrhœal diseases or the danger from invasion by cholera.
[Pg 208]
I think we shall not go far wrong at the start to confine our attention to those cities where there are over 25 deaths from typhoid fever per 100,000 of population. This will at once throw out of consideration a large number of relatively good supplies, including those of New York and Brooklyn. It is not my idea that none of these supplies cause disease. Many of them, as for instance that of New York, are known to receive sewage, and it is an interesting question worthy of most careful study whether there are cases of sickness resulting from this pollution. The point that I wish to make now is simply that in those cases the death-rate itself is evidence that, with existing conditions of dilution and storage, the resulting damage of which we have knowledge is not great enough to justify the expense involved by filtration.
In this connection it should not be forgotten that, especially with very small watersheds, there may be a danger as distinct from present damage which requires consideration. Thus a single house or groups of houses draining into a supply may not appreciably affect it for years, until an outbreak of fever on the water-shed results in infecting the water with the germs of disease and in an epidemic in the city below. This danger decreases with increasing size of the water-shed and volume of the water with which any such pollution would be mixed, and also with the population draining into the water, as there is a probability that the amount of infection continually added from a considerable town will not be subject to as violent fluctuation as that from only a few houses.
Thus in Plymouth, Pa., in 1885, there were 1104 cases of typhoid fever and 114 deaths among a population of 8000, as the result of the discharge of the dejecta from a single typhoid patient into the water of a relatively small impounding reservoir. The cost of this epidemic was calculated with unusual care. The care of the sick cost in cash $67,100.17, and the loss of wages for those who recovered amounted to $30,020.08. The 114 persons who died were earning before their sickness at the rate of $18,419.52 annually.
Such an outbreak would hardly be possible with the Croton[Pg 209] water-shed of the New York water-supply, on account of the great dilution and delay in the reservoirs, but it must be guarded against in small supplies.
Of the cities having more than 25 deaths per 100,000 from typhoid fever, some will no doubt be found where milk epidemics or other special circumstances were the cause; but I believe in a majority of them, and in nearly all cases where the rate is year after year considerably above that figure, the cause will be found in the water-supply. Investigation should be made of this point; and if the water is not at fault, the responsibility should be located. If the water is guilty, it should be either purified or a new supply obtained.
[Pg 210]
CHAPTER XIV.
WATER-SUPPLY AND DISEASE—CONCLUSIONS.
One of the most characteristic and uniform results of the direct pollution of public water-supplies is the typhoid fever which results among the users of the water. In the English and German cities with almost uniformly good drinking-water, typhoid fever is already nearly exterminated, and is decreasing from year to year. American cities having unpolluted water-supplies have comparatively few deaths from this cause, although the figures never go so low as in Europe, perhaps on account of the fresh cases which are always coming in from less healthy neighborhoods in ever-moving American communities. In other American cities the death-rates from typhoid fever are many times what they ought to be and what they actually are in other cities, and the rates in various places, and in the same place at different times, bear in general a close relation to the extent of the pollution of the drinking-water. The power of suitable filtration to protect a city from typhoid fever is amply shown by the very low death-rates from this cause in London, Berlin, Breslau, and large numbers of other cities drawing their raw water from sources more contaminated than those of any but the very worst American supplies, and by the marked and great reductions in the typhoid-fever death rates which have followed at once the installation of filters at Zürich, Switzerland; Hamburg, Germany; Lawrence, Mass., and other places.
The following is a list of the cities of 50,000 inhabitants and upward[Pg 211] in the United States, with deaths from typhoid fever and the sources of their water-supplies. The deaths and populations are from the U. S. Census for 1890; the sources of the water-supplies, from the American Water-Works Manual for the same year. Four cities of this size—Grand Rapids, Lincoln, St. Joseph, and Des Moines—are not included in the census returns of mortality. Two cities with less than 50,000 inhabitants with exceptionally high death-rates have been included, and at the foot of the list are given corresponding data for some large European cities for 1893.
| TYPHOID FEVER DEATH-RATES AND WATER-SUPPLIES OF CITIES. | |||||
|---|---|---|---|---|---|
| City. | Population. | Deaths from Typhoid Fever. |
Water-supply. | ||
| Total. | Per 100,000 living. |
||||
| Birmingham | 26,178 | 69 | 264 | Five Mile Creek | |
| 1. | Denver | 106,713 | 232 | 217 | North Platte River and wells |
| 2. | Allegheny | 105,287 | 192 | 182 | Allegheny River |
| 3. | Camden | 58,313 | 77 | 132 | Delaware River |
| 4. | Pittsburg | 238,617 | 304 | 127 | Allegheny and Monongahela rivers |
| Lawrence | 44,654 | 54 | 121 | Merrimac River | |
| 5. | Newark | 181,830 | 181 | 100 | Passaic River |
| 6. | Charleston | 54,955 | 54 | 98 | Artesian wells yielding 1,600,000 gallons daily |
| 7. | Washington | 230,392 | 200 | 87 | Potomac River |
| 8. | Lowell | 77,696 | 64 | 82 | Merrimac River |
| 9. | Jersey City | 163,003 | 134 | 82 | Passaic River |
| 10. | Louisville | 161,129 | 122 | 76 | Ohio River |
| 11. | Philadelphia | 1,046,964 | 770 | 74 | Delaware and Schuylkill rivers |
| 12. | Chicago | 1,099,850 | 794 | 72 | Lake Michigan |
| 13. | Atlanta | 65,533 | 47 | 72 | South River |
| 14. | Albany | 94,923 | 67 | 71 | Hudson River |
| 15. | Wilmington | 61,431 | 43 | 70 | Brandywine Creek |
| 16. | St. Paul | 133,156 | 92 | 69 | Lakes |
| 17. | Troy | 60,956 | 42 | 69 | Hudson River and impounding reservoirs |
| 18. | Los Angeles | 50,395 | 34 | 67 | Los Angeles River and springs |
| 19. | Nashville | 76,168 | 49 | 64 | Cumberland River |
| 20. | Cleveland | 261,353 | 164 | 63 | Lake Erie |
| 21. | Richmond | 81,388 | 50 | 61 | James River |
| 22. | Hartford | 53,230 | 32 | 60 | Connecticut River and impounding reservoir |
| 23. | Fall River | 74,398 | 44 | 59 | Watupa Lake |
| 24. | Minneapolis | 164,738 | 94 | 57 | Mississippi River |
| 25. | San Francisco | 298,997 | 166 | 56 | Lobus Creek, Lake Merced, and mountain streams |
| 26. | Indianapolis | 105,436 | 57 | 54 | White River |
| 27. | Cincinnati | 296,908 | 151 | 51 | Ohio River |
| 28. | Memphis | 64,495 | 33 | 51 | Artesian Wells |
| 29. | Reading | 58,661 | 29 | 49 | Maiden Creek and Springs |
| 30. | Baltimore | 434,439 | 202 | 47 | Impounding reservoir |
| 31. | Omaha | 140,452 | 63 | 45 | Missouri River |
| 32. | Columbus | 88,150 | 38 | 43 | Surface-water and wells |
| 33. | Providence | 132,146 | 53 | 40 | Pawtuxet River |
| 34. | Kansas City | 132,716 | 53 | 40 | Missouri River |
| 35. | Rochester | 133,896 | 53 | 39 | Hemlock and Candice lakes |
| 36. | Evansville | 50,756 | 20 | 39 | Ohio River |
| 37. | Boston | 448,477 | 174 | 39 | Impounding reservoirs |
| 38. | Toledo | 81,434 | 29 | 36 | Maumee River |
| 39. | Cambridge | 70,028 | 24 | 34 | Impounding reservoir |
| 40. | St. Louis | 451,770 | 145 | 32 | Mississippi River |
| 41. | Scranton | 75,215 | 24 | 32 | Impounding reservoir |
| 42. | Buffalo | 255,664 | 80 | 31 | Niagara River |
| 43. | Milwaukee | 204,468 | 61 | 30 | Lake Michigan |
| 44. | New Haven | 81,298 | 22 | 27 | Impounding reservoir |
| 45. | Worcester | 84,655 | 22 | 26 | Impounding reservoir |
| 46. | Paterson | 78,347 | 20 | 26 | Passaic River (higher up) |
| 47. | Dayton | 61,220 | 15 | 25 | Wells |
| 48. | Brooklyn | 806,343 | 194 | 24 | Wells, ponds, and impounding reservoirs |
| 49. | New York | 1,515,301 | 348 | 23 | Impounding reservoir |
| 50. | Syracuse | 88,143 | 18 | 20 | Impounding reservoir and springs |
| 51. | New Orleans | 242,039 | 45 | 19 | Mississippi River |
| 52. | Detroit | 205,876 | 40 | 19 | Detroit River |
| 53. | Lynn | 55,727 | 9 | 16 | Impounding reservoir |
| 54. | Trenton | 57,458 | 9 | 16 | Delaware River |
| London | 4,306,411 | 719 | 17 | Filtered Thames and Lea rivers and 1⁄4 from wells | |
| Glasgow | 667,883 | 138 | 20 | Loch Katrine | |
| Paris | 2,424,705 | 609 | 25 | Spring water | |
| Amsterdam | 437,892 | 69 | 16 | Filtered dune-water | |
| Rotterdam | 222,233 | 12 | 5 | Filtered Maas River | |
| Hague | 169,828 | 3 | 2 | Filtered dune-water | |
| Berlin | 1,714,938 | 161 | 9 | Filtered Havel and Spree rivers | |
| Hamburg | 634,878 | 115 | 18 | Filtered Elbe River | |
| Breslau | 353,551 | 37 | 11 | Filtered Oder River | |
| Dresden | 308,930 | 14 | 5 | Ground-water | |
| Vienna | 1,435,931 | 104 | 7 | Spring-water | |
Any full discussion of these data would require intimate acquaintances with the various local conditions which it is impossible to take up in detail here, but some of the leading facts cannot fail to be instructive.
Each of the places having over 100 deaths per 100,000 from typhoid fever used unfiltered river-water. Lower in the list, but[Pg 213] still very high, Charleston, said to have been supplied only from artesian wells, had an excessive rate; but the reported water-consumption is so low as to suggest that private wells or other means of supply were in common use. Chicago and Cleveland both drew their water from lakes where they were contaminated by their own sewage. St. Paul’s supply came from ponds, of which I do not know the character. With these exceptions all of the 22 cities with over 50,000 inhabitants, at the head of the list, had unfiltered river-water.
The cities supplied from impounding reservoirs as a rule had lower death rates and are at the lower end of the list, together with some cities taking their water supplies from rivers or lakes at points where they were subject to only smaller or more remote infection. Only three of the American cities in the list were reported as being supplied entirely with ground-water.
It is not my purpose to make too close comparisons between the various cities on the list; some of them may have been influenced by unusual local conditions in 1890. Others have in one way or another improved their water-supplies since that date, and there are several cities in which I know the present typhoid-fever death-rates to be materially lower than those of 1890 given in the table. On the other hand, it is equally true that a number of cities, including some of the larger ones, have since had severe epidemics of typhoid fever which have given very much higher rates than those for 1890.
These fluctuations would change the order of cities in the list from year to year; they would not change the general facts, which are as true to-day as they were in 1890. Nearly all of the great cities of the United States are supplied with unfiltered surface-waters, and a great majority of the waters are taken from rivers and lakes at points where they are polluted by sewage. The death-rates from typhoid fever in those cities, whether they are compared with better supplied cities of this country, or with European cities, are enormously high.
[Pg 214]
Such rates were formerly common in European cities, but they have disappeared with better sanitary conditions. The introduction of filters has often worked marvellous changes in Europe, and in Lawrence the improvement in the city’s health with filtered water was prompt and unquestionable. There is every reason to believe that the general introduction of better water in American cities will work corresponding revolutions; and looking at it from a merely money standpoint, the value of the lives and the saving of the expenses of sickness will pay handsomely when compared with the cost of good water.
The reasons for believing that cholera is caused by polluted water are entirely similar to those in the case of typhoid fever. It was no accident that the epidemic of cholera which caused the death of 3400 persons followed the temporary supply of unfiltered water by the East London Water Company in 1866, while the rest of London remained nearly free, or that the only serious outbreak of cholera in Western Europe in 1892 was at Hamburg, which was also the only city in Germany which used raw river-water. This latter caused the sickness of 20,000 and the death of over 8000 people within a month, and an amount of suffering and financial loss, with the panics which resulted, that cannot be estimated, but that exceeded many times the cost of the filters which have since been put in operation. Hamburg had several times before suffered severely from cholera, and the removal of this danger was a leading, although not the sole, motive for the construction of filters.
How little cities supplied with pure water have to dread from cholera is shown by the experience of Altona and other suburbs of Hamburg with good water-supplies, which had but few cases of cholera not directly brought from the latter place, and by the experience of England, which maintained uninterrupted commercial intercourse with the plague-stricken city, absolutely without quarantine, and, notwithstanding a few cases which were directly imported, the disease gained no foothold in England.
[Pg 215]
I do not know of a single modern European instance where a city with a good water-supply not directly infected by sewage has suffered severely from cholera. I shall leave to others more familiar with the facts the discussion of what happened before the introduction of modern sanitary methods, as well as of the present conditions in Asia; although I believe that in these cases also there is plenty of evidence as to the part water plays in the spread of the disease.
A considerable proportion of the water-supplies of the cities of the United States are so polluted that in case cholera should gain a foothold upon our shores we have no ground for hoping for the favorable experience of the English cities rather than the plague of Hamburg in 1892.
The fæces from a man contain on an average perhaps 1,000,000,000 bacteria per gram,[47] most of them being the normal bacilli of the intestines, Bacillus coli communis. Assuming that a man discharges 200 grams or about 7 ounces of fæces daily, this would give 200,000,000,000 bacteria discharged daily per person. The number of bacteria actually found in American sewage is usually higher, often double this number per person; but there are other sources of bacteria in sewage, and in addition growths or the reverse may take place in the sewers, according to circumstances.
This number of bacteria in sewage is so enormously large that the addition of the sewage from a village or city to even a large river is capable of affecting its entire bacterial composition. Thus taking the population of Lowell in 1892 at 85,000, and the average daily flow of the Merrimac at 6000 cubic feet per second, and assuming that 200,000,000,000 bacteria are discharged daily in the sewage from each person, they would increase the number in the river by 1160[Pg 216] per cubic centimeter, or about 300,000 in an ordinary glass of water. The average number found in the water eight miles below, at the intake of the Lawrence water-works, was more than six times as great as this, due in part to the sewage of other cities higher up.
There is every reason to believe that the bulk of these bacteria were harmless to the people of Lawrence, who drank them; but some of them were not. Fæces of people suffering from typhoid fever contain the germs of that disease. What proportion of the total number of bacteria in such fæces are injurious is not known; but assuming that one fourth only of the total number are typhoid germs, and supposing the fæces of one man to be evenly mixed with the whole daily average flow of the river, it would put one typhoid germ into every glass of water at the Lawrence intake, and at low water several times as many proportionately would be added. This gives some conception of the dilution required to make a polluted water safe.
One often hears of the growth of disease-germs in water, but as far as the northern United States and Europe are concerned there is no evidence whatever that this ever takes place. There are harmless forms of bacteria which are capable of growing upon less food than the disease-germs require and they often multiply in badly-polluted waters. Typhoid-fever germs live for a longer or shorter period, and finally die without growth. The few laboratory experiments which have seemed to show an increase of typhoid germs in water have been made under conditions so widely different from those of natural watercourses that they have no value.[48]
[Pg 217]
The proportionate number of cases of typhoid fever among the users of a polluted water varies with the number of typhoid germs in the water. Excessive pollution causes severe epidemics or continued high death-rates according as the infection is continued or intermittent. Slight infection causes relatively few cases of fever. Pittsburg and Allegheny, taking their water-supplies from below the outlets of some of their own sewers, have suffered severely (103.2 and 127.4 deaths from typhoid fever annually per 100,000, respectively, from 1888 to 1892). Wheeling, W. Va., with similar conditions in 1890, was even worse, a death rate of 345 per 100,000 from this cause being reported, while Albany had only comparatively mild epidemics from the less directly and grossly polluted Hudson. Lawrence and Lowell, taking their water from the Merrimac, both had for many years continued excessive rates, increasing gradually with increasing pollution; and the city having the most polluted source had the higher rate.
In Berlin and Altona, in winter, with open filters, epidemics of typhoid fever followed decreased efficiency of filtration, but the epidemics were often so mild that they would have entirely escaped observation under present American conditions. Chicago has for years suffered from typhoid fever, and the rate has fluctuated, as far as reliable information can be obtained, with the fluctuations in the pollution of the lake water. An unusual discharge of the Chicago River results in a higher death-rate. Abandoning the shore inlet near the mouth of the Chicago River in 1892, resulted in the following year in a reduction of 60 per cent in the typhoid fever death-rate.[49] This reduction shows, not that the present intakes are safe, but simply that they are less polluted than the old ones to an extent measured by the reduction in the death-rate.
It is not supposed that in an epidemic of typhoid fever caused by polluted water every single person contracts the disease directly by[Pg 218] drinking the water. On the contrary, typhoid fever is often communicated in other ways. If we have in the first place a thousand cases in a city caused directly by the water, they will be followed by a large number of other cases resulting directly from the presence in the city of the first thousand cases. The conditions favoring this spread may vary in different wards, resulting in considerable local variations in the death-rates. Some persons also will suffer who did not drink any tap-water. These facts, always noted in epidemics, afford no ground for refusing to believe, in the presence of direct evidence, that the water was the cause of the fever. These additional cases are the indirect if not the direct result of the water. The broad fact that cities with polluted water-supplies as a rule have high typhoid-fever death-rates, and cities with good water-supplies do not (except in the occasional cases of milk epidemics, or where they are overrun by cases contracted in neighboring cities with bad water, as is the case with some of Chicago’s suburbs), is at once the best evidence of the damage from bad water and measure of its extent.
The conditions which remove or destroy the sewage bacteria in a water tend to make it safe. The most important of them are: (1) dilution; (2) time, allowing the bacteria to die (sunlight may aid in this process, although effective sunshine cannot reach the lower layer of turbid waters or through ice); (3) sedimentation, allowing them to go to the bottom, where they eventually die; and (4) natural or artificial filtration. In rivers, distance is mainly useful in affording time, and also, under some conditions, in allowing opportunities for sedimentation. Thus a distance of 500 miles requires a week for water travelling three miles an hour to pass, and will allow very important changes to take place. The old theory that water purifies itself in running a certain distance has no adequate foundation as far as bacteria are concerned. Some purification takes place with the time involved in the passage, but its extent has been greatly overestimated.
The time required for the bacteria to die simply from natural[Pg 219] causes is considerable; certainly not less than three or four weeks can be depended upon with any confidence. In storage reservoirs this action is often considerable, and it is for this reason that American water-supplies from large storage reservoirs are, as a rule, much more healthy than those drawn from rivers or polluted lakes, even when the sources of the former are somewhat polluted. The water-supplies of New York and Boston may be cited as examples. In many other water-works operations the entire time from the pollution to the consumption of the water is but a few days or even less, and time does not materially improve water in this period.
Sedimentation removes bacteria only slowly, as might be expected from their exceedingly small size; and in addition their specific gravity probably is but slightly greater than that of water. The Lawrence reservoir, holding from 10 to 14 days’ supply, effected, by the combined effect of time and sedimentation, a reduction of 90 per cent of the bacteria in the raw water. In spite of this the city suffered severely and continuously from fever. It would probably have suffered even more, however, had it not been for this reduction. Nothing is known of the removal of bacteria by sedimentation from flowing rivers, but, considering the slowness with which the process takes place in standing water, it is evident that we cannot hope for very much in streams, and especially rapid streams, where the opportunities for sedimentation are still less favorable.
Filtration as practiced in Europe removes promptly and certainly a very large proportion of the bacteria—probably, under all proper conditions, over 99 per cent, and is thus much more effective in purification than even weeks of storage or long flows in rivers. The places using filtered water have, in general, extremely low death-rates from typhoid fever. The fever which has occurred at a few places drawing their raw water from greatly polluted sources has resulted from improper conditions which can be avoided, and affords no ground for doubt of the efficiency of properly conducted filtration.
[Pg 220]
Corresponding evidence has not yet been produced in connection with the mechanical filters which have been largely used in the United States; but the bacterial efficiencies secured with them, under proper conditions, and with enough coagulant, have been such as to warrant the belief that they also will serve to greatly diminish the danger from such infection, although they have not shown themselves equal in this respect to sand filters.
The main point is that disease-germs shall not be present in our drinking-water. If they can be kept out in the first place at reasonable expense, that is the thing to do. Innocence is better than repentance. If they cannot be kept out, we must take them out afterwards; it does not matter much how this is done, so long as the work is thorough. Sedimentation and storage may accomplish much, but their action is too slow and often uncertain. Filtration properly carried out removes bacteria promptly and thoroughly and at a reasonable expense.
[Pg 221]
APPENDICES.
APPENDIX I.
RULES OF THE GERMAN GOVERNMENT IN REGARD TO THE FILTRATION OF
SURFACE-WATERS USED FOR PUBLIC WATER-SUPPLIES.
Rules somewhat similar to those of which a translation is given below were first issued by the Imperial Board of Health in 1892. These rules were regarded as unnecessarily rigid, and a petition was presented to the government signed by 37 water-works engineers and directors requesting a revision.[50] As a result a conference was organized consisting of 14 members.[51] Köhler presided, and Koch, Gaffsky, Werner, Günther, and Reincke represented the Imperial Board of Health. The bacteriologists were represented by Flügge, Wolffhügel, and Fränkel, while Beer, Fischer, Lindley, Meyer, and Piefke were the engineer members.
This conference prepared the 17 articles given below in the first days of January, 1894. A little later the first 16 articles were issued to all German local authorities, signed by Bosse, minister of the “Geistlichen,” and Haase, minister of the interior, and they are considered as binding upon all water-works using surface-water. The bacterial examinations were commenced April 1, 1894, by most of the cities which had not previously had them.
[Pg 222]
Although the articles do not deal with rate of filtration, or the precautions against snow and ice, they have a very great interest both because they are an official expression, and on account of the personal standing of the men who prepared them.
§ 1. In judging of the quality of a filtered surface-water the following points should be especially observed:
a. The operation of a filter is to be regarded as satisfactory when the filtrate contains the smallest possible number of bacteria, not exceeding the number which practical experience has shown to be attainable with good filtration at the works in question. In those cases where there are no previous records showing the possibilities of the works and the influence of the local conditions, especially the character of the raw water, and until such information is obtained, it is to be taken as the rule that a satisfactory filtration will never yield an effluent with more than about 100 bacteria per cubic centimeter.
b. The filtrate must be as clear as possible, and, in regard to color, taste, temperature, and chemical composition, must be no worse than the raw water.
§ 2. To allow a complete and constant control of the bacterial efficiency of filtration, the filtrate from each single filter must be examined daily. Any sudden increase in the number of bacteria should cause a suspicion of some unusual disturbance in the filter, and should make the superintendent more attentive to the possible causes of it.
§ 3. Filters must be so constructed that samples of the effluent from any one of them can be taken at any desired time for the bacteriological examination mentioned in § 1.
§ 4. In order to secure uniformity of method, the following is recommended as the standard method for bacterial examination:
The nutrient medium consists of 10 per cent meat extract gelatine with peptone, 10 cc. of which is used for each experiment. Two samples of the water under examination are to be taken, one[Pg 223] of 1 cc. and one of 1⁄2 cc. The gelatine is melted at a temperature of 30° to 35° C., and mixed with the water as thoroughly as possible in the test-tube by tipping back and forth, and is then poured upon a sterile glass plate. The plates are put under a bell-jar which stands upon a piece of blotting-paper saturated with water, and in a room in which the temperature is about 20° C.
The resulting colonies are counted after 48 hours, and with the aid of a lens.
If the temperature of the room in which the plates are kept is lower than the above, the development of the colonies is slower, and the counting must be correspondingly postponed.
If the number of colonies in 1 cc. of the water is greater than about 100, the counting must be done with the help of the Wolffhügel’s apparatus.
§ 5. The person entrusted with the carrying-out of the bacterial examinations must present a certificate that he possesses the necessary qualifications, and wherever possible he shall be a regular employé of the water-works.
§ 6. When the effluent from a filter does not correspond to the hygienic requirements it must not be used, unless the cause of the unsatisfactory work has already been removed during the period covered by the bacterial examinations.
In case a filter for more than a very short time yields a poor effluent, it is to be put out of service until the cause of the trouble is found and corrected.
It is, however, recognized from past experience that sometimes unavoidable conditions (high water, etc.) make it impossible, from an engineering standpoint, to secure an effluent of the quality stated in § 1. In such cases it will be necessary to get along with a poorer quality of water; but at the same time, if the conditions demand it (outbreak of an epidemic, etc.), a suitable notice should be issued.
§ 7. Every single filter must be so built that, when an inferior effluent results, which does not conform to the requirements, it can be disconnected from the pure-water pipes and the filtrate allowed[Pg 224] to be wasted, as mentioned in § 6. This wasting should in general take place, so far as the arrangement of the works will permit it:
(1) Immediately after scraping a filter; and
(2) After replacing the sand to the original depth.
The superintendent must himself judge, from previous experience with the continual bacterial examinations, whether it is necessary to waste the water after these operations, and, if so, how long a time will probably elapse before the water reaches the standard purity.
§ 8. The best sand-filtration requires a liberal area of filter-surface, allowing plenty of reserve, to secure, under all local conditions, a moderate rate of filtration adapted to the character of the raw water.
§ 9. Every single filter shall be independently regulated, and the rate of filtration, loss of head, and character of the effluent shall be known. Also each filter shall, by itself, be capable of being completely emptied, and, after scraping, of having filtered water introduced from below until the sand is filled to the surface.
§ 10. The velocity of filtration in each single filter shall be capable of being arranged to give the most favorable results, and shall be as regular as possible, quite free from sudden changes or interruptions. On this account reservoirs must be provided large enough to balance the hourly fluctuation in the consumption of water.
§ 11. The filters shall be so arranged that their working shall not be influenced by the fluctuating level of the water in the filtered-water reservoir or pump-well.
§ 12. The loss of head shall not be allowed to become so great as to cause a breaking through of the upper layer on the surface of the filter. The limit to which the loss of head can be allowed to go without damage is to be determined for each works by bacterial examinations.
§ 13. Filters shall be constructed throughout in such a way as to insure the equal action of every part of their area.
[Pg 225]
§ 14. The sides and bottoms of filters must be made water-tight, and special pains must be taken to avoid the danger of passages or loose places through which the unfiltered water on the filter might find its way to the filtered-water channels. To this end special pains should be taken to make and keep the ventilators for the filtered-water channels absolutely tight.
§ 15. The thickness of the sand-layer shall be so great that under no circumstances shall it be reduced by scraping to less than 30 cm. (= 12 inches), and it is desirable, so far as local conditions allow, to increase this minimum limit.
Special attention must be given to the upper layer of sand, which must be arranged and continually kept in the condition most favorable for filtration. For this reason it is desirable that, after a filter has been reduced in thickness by scraping and is about to be refilled, the sand below the surface, as far as it is discolored, should be removed before bringing on the new sand.
§ 16. Every city in the German empire using sand-filtered water is requested to make a quarterly report of its working results, especially of the bacterial character of the water before and after filtration, to the Imperial Board of Health (Kaiserlichen Gesundheitsamt), which will keep itself in communication with the commission chosen by the water-works engineers in regard to these questions; and it is believed that after such statistical information is obtained for a period of about two years some farther judgments can be reached.
§ 17. The question as to the establishment of a permanent inspection of public water-works, and, if so, under what conditions, can be best answered after the receipt of the information indicated in § 16.
[Pg 226]
APPENDIX II.
EXTRACTS FROM “BERICHT DES MEDICINAL-INSPECTORATS DES HAMBURGISCHEN
STAATES FÜR DAS JAHR 1892.”
The following are translations from Dr. Reincke’s most valuable report upon the vital statistics of Hamburg for 1892. I much regret that I am unable to reproduce in full the very complete and instructive tables and diagrams which accompany the report.
Diarrhœa and Cholera Infantum (page 10). “It is usually assumed that the increase of diarrhœal diseases in summer is to be explained by the high temperature, especially by the action of the heat upon the principal food of infants—milk. Our observations, however, indicate that a deeper cause must be sought.” (Tables and diagrams of deaths from cholera infantum by months for Hamburg and for Altona with the mean temperatures, 1871-1892.)
“From these it appears that the highest monthly mortality of each year in Hamburg occurred 7 times in July, 13 times in August, and 3 times in September, and substantially the same in Altona. If one compares the corresponding temperatures, it is found that in the three years 1886, 1891, and 1892, with high September mortalities, especially the first two of them, had their maximum temperature much earlier, in fact earlier than usual. Throughout, the correspondence between deaths and temperatures is not well marked. Repeated high temperatures in May and June have never been followed by a notable amount of cholera infantum, although such periods have lasted for a considerable time. For example, toward the end of May, 1892, for a long time the temperature was higher than in the following August, when the cholera infantum appeared.
[Pg 227]
“The following observations are still more interesting. As is seen from the diagram, in addition to the annual rise in summer there is also a smaller increase in the winter, which is especially marked in Altona. In 1892 this winter outbreak was greater than the summer one, and nearly as great in 1880 and in 1888. The few years when this winter increase was not marked, 1876-7, 1877-8, 1881-2, 1883-4, were warm winters in which the mean temperature did not go below the freezing-point. It is also to be noted that the time of this winter outbreak is much more variable than that of the summer one. In 1887 the greatest mortality was in November; in 1889 in February; in other years in December or January, and in Altona, in 1886 and 1888, in March, which is sufficient evidence that it was not the result of Christmas festivities.
“Farther, the winter diarrhœa of Hamburg and of Altona are not parallel as is the case in summer. In Hamburg the greatest mortality generally comes before New Year’s; in Altona one to two months later.
“In Bockendahl’s Generalbericht über das öffentliche Gesundheitswesen der Provinz Schleswig-Holstein für das Jahr 1870, page 10, we read: ‘Yet more remarkable was an epidemic of cholera infantum in Altona in February which proved fatal to 43 children. These cases were distributed in every part of the city, and could not be explained by the health officer until he ascertained that the water company had supplied unfiltered water to the city. This occurred for a few days only in January, and was the only time in the whole year that unfiltered Elbe water was delivered. However little reason there may be to believe that there was a connection between these circumstances, future interruptions of the service of filtered water should be most critically watched, as only in this way can reliable conclusions be reached. Without attempting to draw any scientific conclusions from the fact, I cannot do less than record that, prior to the outbreak of cholera on August 20, 1871, unfiltered together with filtered water had been supplied to the city August 11 to 18. The action of the authorities was then justified when[Pg 228] they forbade in future the supply of unfiltered water except in cases of most urgent necessity, as in case of general conflagration; and in such a case, or in case of interruption due to broken pipes, that the public should be suitably warned.’
“The author of this paragraph, Dr. Kraus, became later the health officer of Hamburg, and in an opinion written by him in 1874, and now before me, he most earnestly urged the adoption of sand-filtration in Hamburg, and cites the above observations in support of his position. In the annual report of vital statistics of Hamburg for 1875 he says that it is quite possible that the addition of unfiltered Elbe water to milk is the cause of the high mortality from cholera infantum, as compared with London, and this idea was often afterward expressed by him. Since then so much evidence has accumulated that his view may fairly be considered proved.
“For the information of readers not familiar with local conditions, a mention of the sources of the water-supplies up to the present time used by Hamburg and Altona will be useful. Both cities take their entire water-supplies from the Elbe—Altona from a point about 7 miles below the discharge of the sewage of both cities, Hamburg from about 7 miles above. The raw water at Altona is thus polluted by the sewage from the population of both cities, having now together over 700,000 inhabitants, and contains in general 20,000 to 40,000 or more bacteria per cubic centimeter. The raw water of Hamburg has, however, according to the time of year and tide, from 200 to 5000, but here also occasionally much higher numbers are obtained when the ebb tide carries sewage up to the intake. How often this takes place is not accurately known, but most frequently in summer when the river is low, more rarely in winter and in times of flood. Recent bacterial examinations show that it occurs much more frequently than was formerly assumed from float experiments. This water is pumped directly to the city raw, while that for Altona is carefully filtered.
“Years ago I expressed the opinion that the repeated typhoid[Pg 229] epidemics in Altona stood in direct connection with disturbances of the action of the filters by frost, which result in the supply of insufficiently purified water. Wallichs in Altona has also come to this conclusion as a result of extended observation, and recently Robert Koch has explained the little winter epidemic of cholera in Altona in the same way, thus supporting our theory. When open filters are cleaned in cold, frosty weather the bacteria in the water are not sufficiently held back by the filters. Such disturbances of filtration not only preceded the explosive epidemics of typhoid fever of 1886, 1887, 1888, 1891, and 1892, and the cholera outbreaks of 1871 and 1893, but also the winter outbreaks of cholera infantum which have been so often repeated. It cannot be doubted that these phenomena bear the relation to each other of cause and effect. It is thus explained why in the warm winters no such outbreaks have taken place, and also why the cholera infantum in winter is not parallel in Hamburg and Altona.
“A farther support of this idea is furnished by Berlin, where in the same way frost has repeatedly interfered with filtration. In the following table are shown the deaths from diarrhœa and cholera infantum for a few winter periods having unusual increases in mortality in comparison with the bacteria in the water-supply.” (These tables show that in March, 1886, March, 1888, February-March, 1889, and February, 1891, high numbers of bacteria resulted from frost disturbance at the Stralau works, and in every case they were followed by greatly increased death-rates from diarrhœal diseases.—A. H.)
“No one who sees this exhibition can doubt that here also the supply of inadequately purified water has every time cost the lives of many children.” (100 to 400 or more each time.—A. H.) “Even more conclusive is the evidence, published by the Berlin Health Office, that this increase was confined to those parts of the city supplied from Stralau” (with open filters.—A. H.), “and that the parts supplied from the better Tegel works took no part in the outbreaks,[Pg 230] which was exactly the case with the well-known typhoid epidemic of February and March, 1889.... It was also found that those children nursed by their mothers or by wet-nurses did not suffer, but only those fed on the milk of animals or other substitutes, and which in any case were mixed with more or less water.”
Under Cholera, page 28, he says: “The revised statistics here given differ slightly from preliminary figures previously issued and widely published.” (The full tables, which cannot be here reproduced, show 16,956 cases and 8605 deaths. 8146 of the deaths occurred in the month ending September 21. Of these, 1799 were under 5 years old; 776 were 5 to 15; 744, 15 to 25; 3520, 25 to 50; 1369, 50 to 70; and 397 over 70 or of unknown age. The bulk of the cases were thus among mature people, children, except very young children, suffering the least severely of any age class.)
“The epidemic began on August 16, in the port where earlier outbreaks have also had their origin. The original source of the infection has not been ascertained with certainty, but was probably from one of two sources. Either it came from certain Jews, just arrived from cholera-stricken Russia, who were encamped in large numbers near the American pier, or the infection came from Havre, where cholera had been present from the middle of July. Perhaps the germs came in ships in water-ballast which was discharged at Hamburg, which is so much more probable, as the sewage of Havre is discharged directly into the docks.
“It is remarkable that in Altona, compared to the total number of cases, very few children had cholera, while in the epidemic of 1871 the children suffered severely. This may be explained by supposing that the cholera of 1892 in Altona was not introduced by water, but by other means of infection....
“It is well known that the drinking-water (of Hamburg) is supposed to have been from the first the carrier of the cholera-germs. In support of this view the following points are especially to be noted:
“1. The explosive rapidity of attack. The often-compared epidemic[Pg 231] in Munich in 1854, which could not have come from the water is characteristically different in that its rise was much slower and was followed by a gradual decline. In Hamburg, with six times as large a population, the height of the epidemic was reached August 27, only 12 days after the first cases of sickness, while in Munich 25 days were required. In Hamburg also the bulk of the cases were confined to 12 days, from August 25 to September 5, while in Munich the time was twice as long.
“2. The exact limit of the epidemic to the political boundary between Hamburg and Altona and Wandsbeck, which also agrees with the boundary between the respective water-supplies, while other differences were entirely absent. Hamburg had for 1000 inhabitants 26.31 cases and 13.39 deaths, but Altona only 3.81 cases and 2.13 deaths, and Wandsbeck 3.06 cases and 2.09 deaths.
“3. The old experience of cholera in fresh-water ports, and the analogy of many earlier epidemics. In this connection the above-mentioned epidemic of 1871 in Altona has a special interest, even though some of the conclusions of Bockendahl’s in his report of 1871 are open to objection. First there were 3 deaths August 3, which were not at once followed by others. Then unfiltered Elbe water was supplied August 11 to 18. On the 19th an outbreak of cholera extended to all parts of the city, which reached its height August 25 and 26, and afterwards gradually decreased. In all 105 persons died of cholera and 186 (179 of them children) of diarrhœa. In Hamburg, four times as large, only 141 persons died of cholera at this time, thus proportionately a smaller number. The conditions were then the reverse of those of 1892, an infection of the Altona water and a comparative immunity in Hamburg.
“It is objected that the cholera-germs were not found in the water in 1892. To my knowledge they were first looked for, and then with imperfect methods, in the second half of September. In the after-epidemics at Altona, they were found in the river-water by R. Koch by the use of better methods.
“It is quite evident that the germs were also distributed by other[Pg 232] methods than by the city water, especially by dock-laborers who became infected while at their work and thus set up little secondary epidemics where they went or lived.... These laborers and sailors, especially on the smaller river-boats, had an enormously greater proportionate amount of cholera than others.... These laborers do not live exclusively near the water, but to a measure in all parts of the city.” (And in Altona and Wandsbeck.—A. H.)
“Altona had 5 deaths from cholera December 25 to January 4, and 19 January 23 to February 11, and no more. As noted above, this is attributed to the water-supply, and to defective filtration in presence of frost....
“The cholera could never have reached the proportion which it did, had the improvements in the drinking-water been earlier completed.”
Further accounts of the water-supplies of Altona and of Hamburg and of the new filtration works at the latter city are given in Appendices VII and VIII.
[Pg 233]
APPENDIX III.
METHODS OF SAND ANALYSIS
(From the Annual Report of the Massachusetts State Board of Health for
1892.)
A knowledge of the sizes of the sand-grains forms the basis of many of the computations. This information is obtained by means of mechanical analyses. The sand sample is separated into portions having grains of definite sizes, and from the weight of the several portions the relative quantities of grains of any size can be computed.
Collection of Samples.—In shipping and handling, samples of sand are best kept in their natural moist condition, as there is then no tendency to separation into portions of unequal-sized grains. Under no circumstances should different materials be mixed in the same sample. If the material under examination is not homogeneous, samples of each grade should be taken in separate bottles, with proper notes in regard to location, quantity, etc. Eight-ounce wide-necked bottles are most convenient for sand samples, but with gravels a larger quantity is often required. Duplicate samples for comparison after obtaining the results of analyses are often useful.
Separation into Portions having Grains of Definite Sizes.—Three methods are employed for particles of different sizes—hand-picking for the stones, sieves for the sands, and water elutriation for the extremely fine particles. Ignition, or determination of albuminoid ammonia, might be added for determining the quantity of organic matter, which, as a matter of convenience, is assumed to consist of particles less than 0.01 millimeter in diameter.
[Pg 234]
The method of hand-picking is ordinarily applied only to particles which remain on a sieve two meshes to an inch. The stones of this size are spread out so that all are in sight, and a definite number of the largest are selected and weighed. The diameter is calculated from the average weight by the method to be described, while the percentage is reckoned from the total weight. Another set of the largest remaining stones is then picked out and weighed as before, and so on until the sample is exhausted. With a little practice the eye enables one to pick out the largest stones quite accurately.
With smaller particles this process becomes too laborious, on account of the large number of particles, and sieves are therefore used instead. The sand for sifting must be entirely free from moisture, and is ordinarily dried in an oven at a temperature somewhat above the boiling-point. The quantity taken for analysis should rarely exceed 100-200 grams. The sieves are made from carefully-selected brass-wire gauze, having, as nearly as possible, square and even-sized meshes. The frames are of metal, fitting into each other so that several sieves can be used at once without loss of material. It is a great convenience to have a mechanical shaker, which will take a series of sieves and give them a uniform and sufficient shaking in a short time; but without this good results can be obtained by hand-shaking. A series which has proved very satisfactory has sieves with approximately 2, 4, 6, 10, 20, 40, 70, 100, 140, and 200 meshes to an inch; but the exact numbers are of no consequence, as the actual sizes of the particles are relied upon, and not the number of meshes to an inch.
It can be easily shown by experiment that when a mixed sand is shaken upon a sieve the smaller particles pass first, and as the shaking is continued larger and larger particles pass, until the limit is reached when almost nothing will pass. The last and largest particles passing are collected and measured, and they represent the separation of that sieve. The size of separation of a sieve bears a tolerably definite relation to the size of the mesh, but the relation[Pg 235] is not to be depended upon, owing to the irregularities in the meshes and also to the fact that the finer sieves are woven on a different pattern from the coarser ones, and the particles passing the finer sieves are somewhat larger in proportion to the mesh than is the case with the coarser sieves. For these reasons the sizes of the sand-grains are determined by actual measurements, regardless of the size of the mesh of the sieve.
It has not been found practicable to extend the sieve-separations to particles below 0.10 millimeter in diameter (corresponding to a sieve with about 200 meshes to an inch), and for such particles elutriation is used. The portion passing the finest sieve contains the greater part of the organic matter of the sample, with the exception of roots and other large undecomposed matters, and it is usually best to remove this organic matter by ignition at the lowest possible heat before proceeding to the water-separations. The loss in weight is regarded as organic matter, and calculated as below 0.01 millimeter in diameter. In case the mineral matter is decomposed by the necessary heat, the ignition must be omitted, and an approximate equivalent can be obtained by multiplying the albuminoid ammonia of the sample by 50.[52] In this case it is necessary to deduct an equivalent amount from the other fine portions, as otherwise the analyses when expressed in percentages would add up to more than one hundred.
Five grams of the ignited fine particles are put in a beaker 90 millimeters high and holding about 230 cubic centimeters. The beaker is then nearly filled with distilled water at a temperature of 20° C., and thoroughly mixed by blowing into it air through a glass tube. A larger quantity of sand than 5 grams will not settle uniformly in the quantity of water given, but less can be used if desired. The rapidity of settlement depends upon the temperature of the water, so that it is quite important that no material variation in temperature should occur. The mixed sand and water is allowed[Pg 236] to stand for fifteen seconds, when most of the supernatant liquid, carrying with it the greater part of the particles less than 0.08 millimeter, is rapidly decanted into a suitable vessel, and the remaining sand is again mixed with an equal amount of fresh water, which is again poured off after fifteen seconds, carrying with it most of the remaining fine particles. This process is once more repeated, after which the remaining sand is allowed to drain, and is then dried and weighed, and calculated as above 0.08 millimeter in diameter. The finer decanted sand will have sufficiently settled in a few minutes, and the coarser parts at the bottom are washed back into the beaker and treated with water exactly as before, except that one minute interval is now allowed for settling. The sand remaining is calculated as above 0.04 millimeter, and the portion below 0.04 is estimated by difference, as its direct determination is very tedious, and no more accurate than the estimation by difference when sufficient care is used.
Determination of the Sizes of the Sand-grains.—The sizes of the sand-grains can be determined in either of two ways—from the weight of the particles or from micrometer measurements. For convenience the size of each particle is considered to be the diameter of a sphere of equal volume. When the weight and specific gravity of a particle are known, the diameter can be readily calculated. The volume of a sphere is 1⁄6πd3, and is also equal to the weight divided by the specific gravity. With the Lawrence materials the specific gravity is uniformly 2.65 within very narrow limits, and we have w⁄2.65 = 1⁄6πd3. Solving for d we obtain the formula d = .9∛w, where d is the diameter of a particle in millimeters and w its weight in milligrams. As the average weight of particles, when not too small, can be determinedd with precision, this method is very accurate, and altogether the most satisfactory for particles above 0.10 millimeter; that is, for all sieve separations. For the finer particles the method is inapplicable, on account of the vast number of particles to be counted in the smallest portion[Pg 237] which can be accurately weighed, and in these cases the sizes are determined by micrometer measurements. As the sand-grains are not spherical or even regular in shape, considerable care is required to ascertain the true mean diameter. The most accurate method is to measure the long diameter and the middle diameter at right angles to it, as seen by a microscope. The short diameter is obtained by a micrometer screw, focussing first upon the glass upon which the particle rests and then upon the highest point to be found. The mean diameter is then the cube root of the product of the three observed diameters. The middle diameter is usually about equal to the mean diameter, and can generally be used for it, avoiding the troublesome measurement of the short diameters.
The sizes of the separations of the sieves are always determined from the very last sand which passes through in the course of an analysis, and the results so obtained are quite accurate. With the elutriations average samples are inspected, and estimates made of the range in size of particles in each portion. Some stray particles both above and below the normal sizes are usually present, and even with the greatest care the result is only an approximation to the truth; still, a series of results made in strictly the same way should be thoroughly satisfactory, notwithstanding possible moderate errors in the absolute sizes.
Calculation of Results.—When a material has been separated into portions, each of which is accurately weighed, and the range in the sizes of grains in each portion determined, the weight of the particles finer than each size of separation can be calculated, and with enough properly selected separations the results can be plotted in the form of a diagram, and measurements of the curve taken for intermediate points with a fair degree of accuracy. This curve of results may be drawn upon a uniform scale, using the actual figures of sizes and of per cents by weight, or the logarithms of the figures may be used in one or both directions. The method of plotting is not of vital importance, and the method for any set of materials which gives the most easily and accurately drawn curves[Pg 238] is to be preferred. In the diagram published in the Report of the Mass. State Board of Health for 1891, page 430, the logarithmic scale was used in one direction, but in many instances the logarithmic scale can be used to advantage in both directions. With this method it has been found that the curve is often almost a straight line through the lower and most important section, and very accurate results are obtained even with a smaller number of separations.
Examples of Calculation of Results.—Following are examples of representative analyses, showing the method of calculation used with the different methods of separation employed with various materials.
I. ANALYSIS OF A GRAVEL BY HAND-PICKING, 11,870 GRAMS TAKEN FOR ANALYSIS.
| Number of Stones in Portion. (Largest Selected Stones.) |
Total Weight of Portion. Grams. |
Average Weight of Stones. Milligrams. |
Estimated Weight of Smallest Stones Milligrams. |
Corresponding Size. Millimeters. |
Total Weight of Stones Smaller than this Size. |
Per Cent of Total Weight Smaller than this Size. |
|---|---|---|---|---|---|---|
| .... | .... | .... | .... | 11,870 | 100 | |
| 10 | 3,320 | 332,000 | 250,000 | 56 | 8,550 | 72 |
| 10 | 1,930 | 193,000 | 165,000 | 49 | 6,620 | 56 |
| 10 | 1,380 | 138,000 | 124,000 | 45 | 5,240 | 44 |
| 20 | 2,200 | 110,000 | 93,000 | 41 | 3,040 | 26 |
| 20 | 1,520 | 76,000 | 64,000 | 36 | 1,520 | 13 |
| 20 | 1,000 | 50,000 | 36,000 | 30 | 520 | 4.4 |
| 20 | 460 | 23,000 | 10,000 | 20 | 60 | .5 |
| 10 | 40 | 4,000 | 2,000 | 11 | 20 | .2 |
| Dust | 20 | .... | .... | .... | .... | .... |
The weight of the smallest stones in a portion given in the fourth column is estimated in general as about half-way between the average weight of all the stones in that portion and the average weight of the stones in the next finer portion.
The final results are shown by the figures in full-faced type in the last and third from the last columns. By plotting these figures we find that 10 per cent of the stones are less than 35 millimeters in diameter, and 60 per cent are less than 51 millimeters. The “uniformity coefficient,” as described below, is the ratios of these numbers, or 1.46, while the “effective size” is 35 millimeters.
[Pg 239]
II. ANALYSIS OF A SAND BY MEANS OF SIEVES.
A portion of the sample was dried in a porcelain dish in an air-bath. Weight dry, 110.9 grams. It was put into a series of sieves in a mechanical shaker, and given one hundred turns (equal to about seven hundred single shakes). The sieves were then taken apart, and the portion passing the finest sieve weighed. After noting the weight, the sand remaining on the finest sieve, but passing all the coarser sieves, was added to the first and again weighed, this process being repeated until all the sample was upon the scale, weighing 110.7 grams, showing a loss by handling of only 0.2 gram. The figures were as follows:
| Sieve Marked. |
Size of Separation of this Sieve. Millimeters. |
Quantity of Sand Passing. Grams. |
Per Cent of Total Weight. |
|---|---|---|---|
| 190 | .105 | .5 | .5 |
| 140 | .135 | 1.3 | 1.2 |
| 100 | .182 | 4.1 | 3.7 |
| 60 | .320 | 23.2 | 21.0 |
| 40 | .46 | 56.7 | 51.2 |
| 20 | .93 | 89.1 | 80.5 |
| 10 | 2.04 | 104.6 | 94.3 |
| 6 | 3.90 | 110.7 | 100.0 |
Plotting the figures in heavy-faced type, we find from the curve that 10 and 60 per cent respectively are finer than .25 and .62 millimeter, and we have for effective size, as described above, .25, and for uniformity coefficient 2.5.
III. ANALYSIS OF A FINE MATERIAL WITH ELUTRIATION.
The entire sample, 74 grams, was taken for analysis. The sieves used were not the same as those in the previous analysis, and instead of mixing the various portions on the scale they were separately weighed. The siftings were as follows:
Remaining on sieve marked 10, above 2.2 millimeters 1.5 grams
Remaining on sieve marked 20, above .98 millimeters 7.0 grams
Remaining on sieve marked 40, above .46 millimeters 22.0 grams
Remaining on sieve marked 70, above .24 millimeters 20.2 grams
Remaining on sieve marked 140, above .13 millimeters 9.2 grams
Passing sieve140, below .13 millimeters 14.1 grams
[Pg 240]
The 14.1 grams passing the 140 sieve were thoroughly mixed, and one third, 4.7 grams, taken for analysis. After ignition just below a red heat in a radiator, the weight was diminished by 0.47 gram. The portion above .08 millimeter and between .04 and .08 millimeter, separated as described above, weighed respectively 1.27 and 1.71 grams, and the portion below .04 millimeter was estimated by difference [4.7 - (0.47 + 1.27 + 1.71)] to be 1.25 grams. Multiplying these quantities by 3, we obtain the corresponding quantities for the entire sample, and the calculation of quantities finer than the various sizes can be made as follows:
| Size of Grain. | Weight. Grams. |
Size of Largest Particles. Millimeters. |
Weight of all the Finer Particles. Grams. |
Per Cent by Weight of all Finer Particles. |
|---|---|---|---|---|
| Above 2.20 millimeters | 1.50 | .... | 74.00 | 100 |
| .98-2.20 millimeters | 7.00 | 2.20 | 72.50 | 98 |
| .46- .98 millimeters | 22.00 | .98 | 65.50 | 89 |
| .24- .46 millimeters | 20.20 | .46 | 43.50 | 60 |
| .13- .24 millimeters | 9.20 | .24 | 23.30 | 32 |
| .08- .13 millimeters | 3.81 | .13 | 14.10 | 19 |
| .04- .08 millimeters | 5.13 | .08 | 10.29 | 14 |
| .01- .04 millimeters | 3.75 | .04 | 5.16 | 7 |
Loss on ignition (assumed to be less than .01 millimeter) |
1.41 | .01 | 1.41 | 1.9 |
By plotting the heavy-faced figures we find that 10 and 60 per cent are respectively finer than .055 and .46 millimeter, and we have effective size .055 millimeter and uniformity coefficient 8.
The effective size and uniformity coefficient calculated in this way have proved to be most useful in various calculations, particularly in estimating the friction between the sands and gravels and water. The remainder of the article in the Report of the Mass. State Board of Health is devoted to a discussion of these relations which were mentioned in Chapter III of this volume.
[Pg 241]
APPENDIX IV.
FILTER STATISTICS.
| STATISTICS OF OPERATION OF SAND FILTERS. | ||||||||
|---|---|---|---|---|---|---|---|---|
| Place. | Year Ending. | Total Quantity of Water filtered for One Year. Million Gallons. |
Million Gallons Daily. |
Area of Filters in use, Acres. |
Average Daily Yield, Million Gallons per Acre. |
Area of Filter Surface cleaned in One Year, Acres. |
Period, Million Gallons per Acre filtered between Scrapings. |
|
Altona |
March, | 1895 | 1,620 | 4.44 | 3.08 | 1.45 | 31.0 | 52 |
| March, | 1896 | 1,730 | 4.75 | 3.08 | 1.55 | 48.5 | 36 | |
| March, | 1897 | 1,960 | 5.40 | 3.08 | 1.75 | 44.0 | 45 | |
| March, | 1898 | 1,940 | 5.30 | 3.08 | 1.72 | 36.5 | 53 | |
Amsterdam, River |
Dec., | 1894 | 1,390 | 3.80 | 5.43 | 0.71 | 23 | 62 |
| Dec., | 1896 | 1,490 | 4.08 | 5.43 | 0.75 | 48 | 31 | |
| Dec., | 1897 | 1,600 | 4.40 | 5.43 | 0.81 | 30 | 53 | |
Amsterdam, Dunes |
Dec., | 1894 | 2,330 | 6.40 | 4.94 | 1.29 | 116 | 20 |
| Dec., | 1896 | 2,360 | 6.50 | 4.75 | 1.37 | 90 | 26 | |
| Dec., | 1897 | 2,290 | 6.25 | 4.75 | 1.31 | 109 | 21 | |
Ashland, Wis |
Feb., | 1897 | 398 | 1.09 | 0.50 | 2.18 | 4.83 | 83 |
Berlin, total |
Mar., | 1896 | 13,000 | 35.60 | 25.10 | 1.42 | ||
| Mar., | 1897 | 12,900 | 35.40 | 25.10 | 1.40 | |||
| Mar., | 1898 | 13,200 | 36.20 | 27.00 | 1.34 | |||
Bremen |
Mar., | 1895 | 1,190 | 3.27 | 2.51 | 1.31 | 50 | 24 |
| Mar., | 1896 | 1,220 | 3.34 | 3.21 | 1.04 | 32.5 | 38 | |
| Mar., | 1897 | 1,280 | 3.50 | 3.21 | 1.09 | 25.2 | 50 | |
| Mar., | 1898 | 1,400 | 4.10 | 3.21 | 1.28 | 34.0 | 41 | |
Breslau |
Mar., | 1895 | 2,840 | 7.80 | 5.12 | 1.52 | 45 | 64 |
| Mar., | 1896 | 2,960 | 8.10 | 5.12 | 1.58 | 40.0 | 74 | |
| Mar., | 1897 | 2,990 | 8.20 | 5.12 | 1.60 | 37 | 81 | |
| Mar., | 1898 | 3,060 | 8.40 | 5.12 | 1.64 | 43 | 71 | |
Brunn |
Dec., | 1896 | 1,110 | 3.04 | 1.62 | 1.87 | 8.6 | 128 |
| Dec., | 1897 | 1,190 | 3.25 | 1.62 | 2.00 | 9.1 | 131 | |
Brunswick |
Mar., | 1895 | 815 | 2.23 | 1.48 | 1.51 | 14.8 | 55 |
| Mar., | 1896 | 840 | 2.30 | 1.48 | 1.56 | 13.3 | 63 | |
| Mar., | 1897 | 820 | 2.25 | 1.48 | 1.52 | 13.7 | 60 | |
| Mar., | 1898 | 870 | 2.38 | 1.48 | 1.61 | 11.9 | 73 | |
Budapest |
Dec., | 1892 | 7,360 | 20.20 | 3.00 | 6.70 | 254 | 29 |
| [Pg 242] Copenhagen |
Dec., | 1895 | 2,330 | 6.40 | 2.88 | 2.22 | 45 | 52 |
| Dec., | 1896 | 2,490 | 6.80 | 2.88 | 2.35 | 52 | 48 | |
| Dec., | 1897 | 2,580 | 7.10 | 2.88 | 2.47 | 54 | 48 | |
Dordrecht |
Dec., | 1894 | 365 | 1.00 | 0.56 | 1.79 | ||
Frankfort on Oder |
Dec., | 1895 | 310 | 0.85 | 0.37 | 2.28 | 2.9 | 107 |
| Dec., | 1896 | 325 | 0.89 | 0.37 | 2.40 | 7.4 | 44 | |
| Dec., | 1897 | 356 | 0.98 | 0.37 | 2.65 | 8.8 | 41 | |
Hamburg |
Dec., | 1894 | 11,450 | 31.40 | 34.0 | 0.92 | 350 | 33 |
| Dec., | 1895 | 11,700 | 32.10 | 34.0 | 0.94 | 275 | 43 | |
| Dec., | 1896 | 11,500 | 31.70 | 34.0 | 0.93 | 266 | 43 | |
| Dec., | 1897 | 12,000 | 32.70 | 34.0 | 0.96 | 285 | 42 | |
| Dec., | 1898 | 11,900 | 32.60 | 43.0 | 0.76 | 246 | 48 | |
Hudson, N. Y. |
Dec., | 1892 | 697 | 1.91 | 0.74 | 2.58 | ||
| Dec., | 1893 | 543 | 1.49 | 0.74 | 2.01 | |||
| Dec., | 1895 | 535 | 1.46 | 0.74 | 1.98 | |||
Ilion, N. Y. |
Feb., | 1899 | 182 | 0.50 | 0.14 | 3.57 | 1.40 | 130 |
Königsberg |
Mar., | 1895 | 1,060 | 2.90 | 2.70 | 1.07 | 38.5 | 27 |
| Mar., | 1896 | 1,085 | 2.97 | 2.70 | 1.10 | 35.0 | 31 | |
| Mar., | 1897 | 1,085 | 2.97 | 2.70 | 1.10 | 41.0 | 27 | |
| Mar., | 1898 | 1,140 | 3.12 | 2.70 | 1.16 | 44.0 | 26 | |
Lawrence |
Dec., | 1894 | 1,050 | 2.88 | 2.50 | 1.15 | 10 | 105 |
| Dec., | 1895 | 1,097 | 3.00 | 2.50 | 1.20 | 27 | 41 | |
| Dec., | 1896 | 1,101 | 3.02 | 2.50 | 1.20 | 30 | 37 | |
| Dec., | 1897 | 1,114 | 3.06 | 2.50 | 1.22 | 41 | 27 | |
Liverpool |
Dec., | 1896 | 8,520 | 23.40 | 10.92 | 2.14 | 158 | 54 |
|
[53] London, all filters but not including ground water |
Dec., | 1892 | 65,783 | 180 | 109.75 | 1.64 | 90 | |
| Dec., | 1893 | 195 | 116.00 | 1.68 | ||||
| Dec., | 1894 | 68,700 | 188 | 117.00 | 1.60 | |||
| Dec., | 1895 | 76,900 | 210 | 123.75 | 1.70 | |||
| Dec., | 1896 | 72,482 | 198 | 123.75 | 1.60 | |||
| Dec., | 1897 | 73,340 | 201 | 125.00 | 1.61 | |||
London, Chelsea |
Dec., | 1897 | 5,370 | 14.70 | 8.00 | 1.85 | ||
E. London |
Dec., | 1897 | 18,000 | 49.00 | 31.00 | 1.58 | ||
Grand Junction |
Dec., | 1897 | 8,560 | 23.40 | 21.75 | 1.07 | ||
Lambeth |
Dec., | 1897 | 10,370 | 28.40 | 12.25 | 2.30 | ||
New River |
Dec., | 1897 | 15,750 | 43.00 | 16.50 | 2.60 | ||
Southwark & Vauxhall |
Dec., | 1897 | 14,800 | 40.50 | 20.50 | 1.98 | ||
West Middlesex |
Dec., | 1897 | 8,910 | 24.30 | 15.00 | 1.61 | ||
Lübeck |
Mar., | 1895 | 1,520 | 4.15 | 1.40 | 2.95 | 16.2 | 94 |
| Mar., | 1896 | 1,600 | 4.38 | 1.40 | 3.13 | 24.4 | 66 | |
| Mar., | 1897 | 1,650 | 4.50 | 1.40 | 3.22 | 27.0 | 61 | |
| Mar., | 1898 | 1,750 | 4.80 | 1.40 | 3.42 | 38.5 | 45 | |
Magdeburg [Pg 243] |
Mar., | 1895 | 1880 | 5.15 | 3.76 | 1.37 | 47.5 | 40 |
| Mar., | 1896 | 1950 | 5.35 | 3.76 | 1.42 | 65.0 | 30 | |
| Mar., | 1897 | 1880 | 5.15 | 3.76 | 1.37 | 59.0 | 32 | |
| Mar., | 1898 | 2070 | 5.66 | 3.76 | 1.50 | 63.0 | 33 | |
Mt. Vernon, N. Y |
Dec., | 1895 | 493 | 1.35 | 1.10 | 1.22 | 7.3 | 68 |
| Dec., | 1896 | 608 | 1.66 | 1.10 | 1.51 | 9.2 | 66 | |
| Dec., | 1897 | 808 | 2.21 | 1.10 | 2.00 | 16.6 | 49 | |
| Dec., | 1898 | 933 | 2.56 | 1.10 | 2.34 | 18.4 | 51 | |
Posen |
Mar., | 1895 | 305 | 0.84 | 0.70 | 1.20 | 10.3 | 30 |
| Mar., | 1896 | 346 | 0.94 | 0.70 | 1.35 | 10.4 | 33 | |
| Mar., | 1897 | 325 | 0.89 | 0.70 | 1.27 | 10.1 | 32 | |
| Mar., | 1898 | 360 | 0.99 | 0.70 | 1.42 | 9.6 | 38 | |
Poughkeepsie |
Dec., | 1892 | 696 | 1.91 | 0.68 | 2.81 | 14.0 | 50 |
| Dec., | 1893 | 667 | 1.83 | 0.68 | 2.70 | 12.0 | 56 | |
| Dec., | 1894 | 633 | 1.73 | 0.68 | 2.55 | 14 | 45 | |
| Dec., | 1895 | 686 | 1.88 | 0.68 | 2.77 | 14 | 49 | |
| Dec., | 1896 | 664 | 1.82 | 0.68 | 2.68 | 9 | 73 | |
| Dec., | 1897 | 615 | 1.69 | 1.36 | 1.24 | |||
| Dec., | 1898 | 611 | 1.67 | 1.36 | 1.23 | 10.88 | 57 | |
Rostock |
June, | 1897 | 560 | 1.54 | 1.11 | 1.38 | 9.3 | 60 |
| June, | 1898 | 625 | 1.71 | 1.11 | 1.55 | 9.0 | 70 | |
Rotterdam |
Dec., | 1893 | 4850 | 13.30 | 6.30 | 2.11 | ||
Stettin |
Mar., | 1895 | 1130 | 3.10 | 2.26 | 1.37 | 26.5 | 43 |
| Mar., | 1896 | 1030 | 2.83 | 2.26 | 1.25 | 15.5 | 66 | |
| Mar., | 1897 | 980 | 2.70 | 2.26 | 1.19 | 16.1 | 61 | |
| Mar., | 1898 | 1020 | 2.80 | 2.26 | 1.24 | 20.3 | 50 | |
Stockholm |
Dec., | 1895 | 2375 | 6.50 | 2.78 | 2.33 | 70.0 | 34 |
| Dec., | 1896 | 2500 | 6.85 | 2.78 | 2.45 | 68.0 | 37 | |
| Dec., | 1897 | 2750 | 7.50 | 3.60 | 2.08 | 76.0 | 36 | |
Stralsund |
Mar., | 1897 | 215 | 0.59 | 1.11 | 0.53 | 16.0 | 13 |
| Mar., | 1898 | 210 | 0.58 | 1.11 | 0.51 | 17.3 | 12 | |
Stuttgart |
Mar., | 1895 | 1040 | 2.85 | 1.46 | 1.96 | 13.7 | 76 |
| Mar., | 1896 | 1220 | 3.34 | 1.66 | 2.04 | 17.7 | 69 | |
| Mar., | 1897 | 1270 | 3.48 | 2.32 | 1.50 | 18.7 | 68 | |
| Mar., | 1898 | 1320 | 3.60 | 2.32 | 1.54 | 20.2 | 65 | |
Utrecht |
Dec., | 1896 | 510 | 1.40 | 0.60 | 2.33 | 31 | 16 |
Zürich |
Dec., | 1891 | 2010 | 5.50 | 0.84 | 6.50 | 8 | 250 |
| Dec., | 1892 | 2150 | 5.90 | 0.84 | 7.00 | 10 | 215 | |
| Dec., | 1893 | 2310 | 6.38 | 1.19 | 5.35 | 13 | 177 | |
| Dec., | 1894 | 2250 | 6.15 | 1.19 | 5.18 | 17 | 133 | |
| Dec., | 1895 | 2460 | 6.70 | 1.19 | 5.62 | 27 | 91 | |
| Dec., | 1896 | 2360 | 6.45 | 1.66 | 3.88 | 30 | 79 | |
| Dec., | 1897 | 2500 | 6.84 | 1.66 | 4.13 | 35 | 71 | |
| Dec., | 1898 | 2730 | 7.50 | 1.66 | 4.50 | 47 | 58 | |
[Pg 244]
| PARTIAL LIST OF CITIES USING SAND FILTERS. | |||||
|---|---|---|---|---|---|
| Place. | When Built. |
Population. 1890. |
Area of Filters. |
Number of Filters. |
Average Daily Consumption. |
| UNITED STATES. | |||||
| Poughkeepsie. N. Y. | 1872 | 24,000 | 1.36 | 3 | 1.67 |
| Hudson, N. Y. | 1874 | 9,970 | 0.74 | 2 | 1.50 |
| St. Johnsbury, Vt. | 187(?) | 3,857 | 0.14 | 3 | 0.70 |
| Nantucket, Mass. | 1893 | 3,268 | 0.11 | 1 | 0.09 |
| Lawrence, Mass. | 1893 | 44,654 | 2.50 | 1 | 3.00 |
| Ilion, N. Y. | 1893 | 4,057 | 0.14 | 2 | 0.50 |
| Mount Vernon, N. Y. | 1894 | 10,830 | 1.10 | 3 | 1.66 |
| Grand Forks, N. D. | 1894 | 4,979 | 0.42 | 1 | .... |
| Milford, Mass. | 1895 | 9,956 | 0.25 | 1 | 0.70 |
| Ashland, Wis. | 1895 | 9,956 | 0.50 | 3 | 1.09 |
| Hamilton, N. Y. | 1895 | 1,744 | 0.12 | 1 | 0.03 |
| Lambertville, N. J. | 1896 | 4,142 | 0.28 | 2 | 0.25 |
| Far Rockaway, N. Y. | 1896 | 2,288 | 0.92 | 2 | 0.93 |
| Red Bank, N. J. | 1897 | 500 | 0.03 | 2 | 0.10 |
| Somersworth, N. H. | 1897 | 6,207 | 0.50 | 1 | .... |
| Little Falls, N. Y. | 1898 | 8,783 | 0.76 | 1 | .... |
| Berwyn, Penna. | 1898 | 826 | 0.52 | 3 | .... |
| Harrisburg, Penna. | 1899 | 1,200 | 0.12 | 2 | 0.15 |
| Albany, N. Y. | 1899 | 94,923 | 5.60 | 8 | 11.00[54] |
| Rock Island, Illinois | 1899 | 13,634 | 1.20 | 3 | 3.50 |
| Total | .... | 259,774 | 17.31 | 45 | 26.87 |
| BRITISH COLUMBIA. | |||||
| Victoria | .... | 16,841 | 0.82 | 3 | 1.80 |
| SOUTH AMERICA. | |||||
| Buenos Ayres | .... | 500,000 | 4.15 | 3 | .... |
| Montevidio | .... | .... | Filters | reported | .... |
| HOLLAND. | |||||
| Amsterdam | .... | 555,821 | 10.18 | 12 | 11.20 |
| Rotterdam | .... | 290,000 | 6.30 | 18 | 13.00 |
| The Hague | .... | 191,000 | 2.88 | 6 | 4.20 |
| Schiedam | .... | 25,300 | 1.33 | 5 | 0.68 |
| Utrecht | .... | 140,000 | 0.60 | .... | 1.40 |
| Groningen | .... | 57,900 | 0.59 | 2 | .... |
| Dordrecht | .... | 34,100 | 0.56 | 2 | 1.00 |
| Leeuwarden | .... | 30,700 | 0.31 | 2 | .... |
| Vlaardingen | .... | .... | .... | .... | .... |
| Sliedrecht | .... | .... | .... | .... | .... |
| Gorinchem | .... | 10,000 | .... | .... | .... |
| Zutphen | .... | 18,000 | .... | .... | .... |
| Leyden | .... | 44,200 | .... | .... | .... |
| Enschede | .... | .... | .... | .... | .... |
| Middelburg | .... | 17,000 | .... | .... | .... |
| Total | .... | 1,414,021 | 22.75 | 47 | 31.48 |
| [Pg 246] GREAT BRITAIN. | |||||
| London | .... | 5,030,267 | 125.00 | 120 | 200.00 |
| Liverpool | .... | 790,000 | 10.92 | .... | 26.67 |
| Dublin | .... | 349,000 | 5.00 | 10 | 18.00 |
| Leeds | .... | 420,000 | 6.00 | 8 | 17.99 |
| Bradford | .... | 436,260 | 4.62 | 6 | 13.31 |
| Leicester | .... | 220,005 | 2.50 | .... | 4.75 |
| York | .... | 72,083 | 2.04 | 6 | 3.00 |
| Edinburgh | .... | 292,364 | 2.00 | 4 | 18.00 |
| Darlington | .... | 43,000 | 1.32 | 7 | .... |
| Wakefield | .... | 36,815 | 1.25 | .... | .... |
| Carlisle | .... | 40,000 | 0.90 | .... | .... |
| Dumfries | .... | 17,821 | 0.25 | .... | .... |
| Accrington | .... | 42,000 | .... | .... | .... |
| Birmingham | .... | 680,140 | .... | .... | 19.05 |
| Blackburn | .... | 130,000 | .... | .... | 4.10 |
| Bolton | .... | 250,000 | .... | .... | 6.60 |
| Chester | .... | 40,000 | .... | .... | .... |
| Halifax | .... | 217,000 | .... | .... | 5.18 |
| Hereford | .... | 20,000 | .... | .... | .... |
| Middlesborough | .... | 187,331 | .... | .... | 11.39 |
| Newcastle | .... | 320,000 | .... | .... | 14.00 |
| Oldham | .... | 145,800 | .... | .... | 5.30 |
| Oxford | .... | 53,000 | .... | .... | 1.59 |
| Preston | .... | 113,864 | .... | .... | 4.20 |
| Reading | .... | 71,558 | .... | .... | 3.00 |
| Southampton | .... | 76,430 | .... | .... | 3.45 |
| Wigan | .... | 60,000 | .... | .... | 1.22 |
| Worcester | .... | 45,000 | .... | .... | 1.93 |
| Total | .... | 10,199,738 | 161.80 | 161 | 382.73 |
| GERMANY. | |||||
| Hamburg | .... | 661,200 | 42.00 | 22 | 33.00 |
| Berlin | .... | 1,746,424 | 31.45 | 55 | 36.00 |
| Breslau | .... | 380,000 | 5.12 | 5 | 8.20 |
| Magdeburg | .... | 217,067 | 3.76 | 11 | 5.66 |
| Bremen | .... | 157,500 | 3.21 | 12 | 3.50 |
| Altona | .... | 162,427 | 3.08 | 13 | 5.40 |
| Königsberg | .... | 176,000 | 2.70 | 7 | 3.00 |
| Stuttgart | .... | 162,516 | 2.32 | .... | 4.00 |
| Stettin | .... | 145,000 | 2.26 | 9 | 3.00 |
| Lübeck | .... | 70,000 | 1.40 | 6 | 4.50 |
| Brunswick | .... | 100,883 | 1.48 | 4 | 2.30 |
| Stralsund | .... | 30,105 | 1.11 | 6 | 0.60 |
| Rostock | .... | 49,891 | 1.11 | 3 | 1.54 |
| Lignitz | .... | 46,852 | 0.96 | 6 | 1.40 |
| Posen | .... | 75,000 | 0.70 | 4 | 0.90 |
| Schwerin | .... | 36,000 | 0.65 | 4 | 0.50 |
| Chemnitz | .... | 164,743 | 0.59 | 3 | .... |
| Worms | .... | 30,000 | 0.50 | 3 | 0.64 |
| Ratibor | .... | 20,729 | 0.42 | 3 | .... |
| Frankfort on Oder | .... | 59,161 | 0.37 | 5 | 0.89 |
| Kiel | .... | 69,214 | 0.31 | .... | 1.50 |
| Tilsit | .... | 30,000 | 0.25 | .... | 0.20 |
| Brieg | .... | 20,154 | 0.20 | 4 | .... |
| Gluckstadt | .... | 6,214 | 0.14 | .... | 0.10 |
| Wandsbeck | .... | 22,000 | 0.13 | .... | 0.30 |
| Total | .... | 4,639,080 | 106.22 | 185 | 117.13 |
| OTHER EUROPEAN FILTERS. | |||||
| Warsaw | .... | 500,000 | 6.20 | 12 | 6.00 |
| St. Petersburg | .... | 954,000 | 5.85 | 11 | 39.00 |
| Odessa | .... | 380,000 | 4.75 | 5 | 8.00 |
| Choisy le Roi and | .... | }200,000{ | 3.85 | 25 | 10.00 |
| Neuilly sur Marne | .... | 2.31 | 15 | .... | |
| Copenhagen | .... | 340,000 | 2.88 | 9 | 6.80 |
| Stockholm | .... | 274,000 | 2.78 | .... | 7.00 |
| Antwerp | .... | 240,000 | 2.10 | 8 | 2.00 |
| Zürich | .... | 96,839 | 1.66 | .... | 7.00 |
| Brunn | .... | .... | 1.62 | .... | 3.04 |
| Constantinople, South side | .... | .... | 0.74 | 3 | .... |
| Total | .... | 2,984,839 | 34.74 | 88 | 88.84 |
| ASIA. | |||||
| Blandarwada, India | .... | .... | 1.97 | 6 | .... |
| Agra, India | .... | .... | 1.37 | .... | .... |
| Bombay, India | .... | 821,000 | 1.22 | 4 | .... |
| Shanghai, China | .... | .... | 0.88 | 4 | .... |
| Hong Kong | .... | .... | 0.67 | 6 | .... |
| Yokohama, Japan | .... | 110,000 | 0.58 | 3 | .... |
| Calcutta, India | .... | 466,000 | .... | .... | .... |
| Tokyo, Japan | .... | .... | .... | .... | .... |
| Baroda, India | .... | .... | .... | .... | .... |
| Allahabad, India | .... | .... | .... | .... | .... |
| Total | .... | 1,397,000 | 6.69 | 23 | |
| SUMMARY. | |||||
| United States | .... | 259,774 | 17.31 | 45 | 26.87 |
| British Columbia | .... | 16,841 | 0.82 | 3 | 1.80 |
| South America | .... | 500,000 | 4.15 | 3 | .... |
| Holland | .... | 1,414,021 | 22.75 | 47 | 31.48 |
| Great Britain | .... | 10,199,738 | 161.80 | 161 | 382.73 |
| Germany | .... | 4,639,080 | 106.22 | 185 | 117.13 |
| Other European countries | .... | 2,984,839 | 34.74 | 88 | 88.84 |
| Asia | .... | 1,397,000 | 6.69 | 23 | .... |
| Total | .... | 21,411,293 | 354.48 | 555 | 648.85 |
[Pg 247]
| LIST OF CITIES AND TOWNS USING MECHANICAL FILTERS. ARRANGED BY POPULATIONS. |
||||||
|---|---|---|---|---|---|---|
Abbreviations.--P., Pressure filters; G., Gravity filters; J., Jewell system; N. Y., New York system; W., Warren system; C., Continental system; Am., American system. |
||||||
| Place. | Population, 1890. |
Filters First Installed. |
Nominal Capacity of Filters, 1899. Million Gallons. |
Average Consumption, Million Gallons: Water Works Manual. |
Area of Filters, Sq. Ft., 1899. |
Filter System. |
| Denver, Col. | 108,204 | 2260 | Special. | |||
| Atlanta, Ga. | 65,533 | 1887 | 8 | 4.54 | 2056 | N. Y. P. |
| St. Joseph, Mo. | 52,324 | 1898 | 10.2 | 3842 | J. G. | |
| Oakland, Cal. | 48,682 | 1891 | 5 | 10 | 1960 | N. Y. P. |
| Kansas City, Kan. | 38,316 | 1898 | 6 | 2 | 2260 | J. G. |
| Wilkesbarre, Pa.[55] | 37,718 | 10 | 3166 | J. G. | ||
| Norfolk, Va. | 34,871 | 1899 | 6 | 3.5 | 2112 | J. G. |
| Augusta, Ga. | 33,300 | 1899 | 6 | 3.8 | 2112 | N. Y. G. |
| Quincy. Ill. | 30,494 | 1892 | 4 | 1.2 | 1582 | J. G. |
| Dubuque, Iowa[56] | 30,311 | 1899 | 2 | 880 | J. G. | |
| Terre Haute, Ind. | 30,217 | 1890 | 4 | 3{ | 1076 226 |
N. Y. P. J. G. |
| Elmira, N. Y. | 29,708 | 1897 | 6 | 3 | 2034 | J. G. |
| Chattanooga, Tenn. | 29,100 | 1887 | 9 | 2080 | J. & N. Y. P. | |
| Davenport, Iowa | 26,872 | 1891 | 7.5 | 3 | 2380 | Am. P. |
| Little Rock, Ark. | 25,874 | 1891 | 5.5 | 1544 | Am., J., & N. Y. P. | |
| Winnipeg, Mann. | 25,642 | 1887 | 1.5 | 390 | N. Y. | P. |
| Oshkosh, Wis. | 22,836 | 1891 | 2.4 | 2.1 | 550 | W. G. |
| Macon, Ga. | 22,746 | 1893 | 4 | 1.65 | 1437 | J., W., & N. Y. |
| Burlington, Ia. | 22,565 | 1894 | 3.5 | 1243 | J. G. | |
| Knoxville, Tenn. | 22,535 | 1894 | 5 | 1.93 | 1404 | W. G. |
| Lexington, Ky. | 21,567 | 1895 | 2 | 1.2 | 678 | J. G. |
| Kingston, N. Y. | 21,261 | 1897 | 4 | 1.5 | 1120 | N. Y. P. |
| York, Penna. | 20,793 | 1899 | 4 | 2.37 | 1408 | J. G. |
| Biddeford, Maine | 20,500 | 1896 | 3 | 2 | 780 | W. G. |
| Newport, R. I. | 19,467 | 4 | 2.1 | Special. | ||
| Bangor, Maine | 19,103 | 1897 | 5 | 3 | 1404 | W. G. |
| Cedar Rapids, Ia. | 18,020 | 1896 | 2.5 | 2 | 905 | J. G. |
| Elgin, Ill. | 17,823 | 1888 | 4.3 | 1 | 780 | Am. P. |
| Decatur, Ill. | 16,841 | 1893 | 3 | 2 | 1008 | W. G. |
| Belleville, Ill. | 15,361 | 1 | 0.6 | 339 | J. G. | |
| Columbia, S. C. | 15,353 | 1892 | 3 | 678 | J. G. | |
| Keokuk, Ia. | 14,101 | 1893 | 3 | 980 | N. Y. P. | |
| Ottumwa, Ia. | 14,001 | 1895 | 2 | 1.2 | 678 | J. G. |
| Rock Island, Ill.[55] | 13,634 | 2 | 3.5 | 452 | J. G. | |
| Raleigh, N. C. | 12,678 | 1887 | 1 | 1 | 296 | N. Y. P. |
| Shreveport, La. | 11,979 | 1889 | 1 | 312 | N. Y. P. | |
| New Castle, Penna | 11,600 | 1898 | 4 | 2 | 1408 | N. Y. G. |
| [Pg 248]Charlotte, N. C. | 11,557 | 1896 | 1 | 0.5 | 530 | N. Y. G. |
| Nebraska City, Neb. | 11,494 | 1891 | 0.4 | 0.7 | 116 | N. Y. P. |
| Streator, Ill. | 11,414 | 2 | 1.3 | 100 | Western & Am. P. | |
| Hornelsville, N. Y.[56] | 10,966 | 1899 | 3 | 700 | N. Y. P. | |
| Augusta, Maine | 10,527 | 1887 | 0.6 | 1.6 | 100 | W. |
| St. Thomas, Ont. | 10,370 | 1891 | 2.5 | 0.6 | 700 | N. Y. P. |
| Cairo, Ill. | 10,324 | 1889 | 0.8 | 2.5 | 197 | N. Y. P. |
| Alton, Ill. | 10,294 | 1898 | 3 | 1 | 1056 | N. Y. G. |
| Asheville, N. C. | 10,235 | 1889 | 1 | 0.35 | 312 | N. Y. P. |
| Greenwich, Conn. | 10,131 | 1887 | 2 | 0.4 | 592 | N. Y. P. |
| Huntington, W. Va. | 10,108 | 1899 | 2 | 704 | N. Y. G. | |
| Beaver Falls, Pa. | 9,735 | 2 | 4.5 | N. Y. | ||
| Champaign, Ill.[55] | 9,719 | 0.75 | N. Y. | |||
| Chatham, Ont. | 9,052 | 1895 | 1 | 0.4 | 280 | N. Y. P. |
| Adrian, Mich. | 8,756 | 1899 | 1.75 | 0.45 | 565 | J. G. |
| Athens, Ga. | 8,639 | 1893 | 1 | 0.45 | 420 | W. G. |
| East Providence, R. I. | 8,422 | 1899 | 0.5 | 176 | J. G. | |
| Winston, N. C. | 8,018 | 1895 | 0.5 | 0.3 | 156 | W. G. |
| Danville, Penna. | 7,998 | 1896 | 1 | 1 | 226 | J. G. |
| Clarksville, Tenn.[56] | 7,924 | 1899 | 1.5 | 0.5 | 704 | J. G. |
| Stevens Point, Wis. | 7,896 | 1889 | 0.5 | 0.25 | 156 | N. Y. P. |
| Carlisle, Pa. | 7,620 | 1896 | 1.5 | 339 | J. G. | |
| Calais, Me. | 7,290 | 1893 | 1.5 | 0.85 | 275 | W. G. |
| Long Branch, N. J. | 7,231 | 1888 | 3 | 1.3 | 904 | N. Y. P. |
| Creston, Ia. | 7,200 | 1891 | 0.5 | 0.3 | 150 | J. |
| St. Hyacinthe, Que. | 7,016 | 1898 | 1 | 0.84 | 294 | J. P. |
| Rome, Ga.[56] | 6,957 | 1899 | 1.5 | 1.3 | 528 | J. G. |
| Westerly, R. I. | 6,813 | 1896 | 1.5 | 0.375 | 396 | N. Y. G. |
| Merrill, Wis. | 6,809 | 1897 | 1 | 339 | J. G. | |
| Dennison, Ohio[56] | 6,767 | 1899 | 1.25 | 1 | 528 | J. G. |
| Parsons, Kan. | 6,736 | 1894 | 2 | 0.6 | 452 | J. G. |
| Waterloo, Iowa | 6,674 | 1891 | 1.5 | 0.7 | 565 | J. G. |
| Somerville, N. J. | 6,417 | 1885 | 1.9 | 0.75 | 552 | N. Y. P. |
| Athol, Mass. | 6,319 | 1888 | 1.5 | 0.5 | 350 | N. Y. P. |
| Owego, N. Y. | 6,200 | 1887 | 1 | 0.75 | 234 | N. Y. P. |
| Brunswick, Maine | 6,012 | 1887 | 0.6 | 0.33 | 100 | W. |
| Bucyrus, Ohio | 5,974 | 1887 | 0.5 | 0.55 | 156 | N. Y. P. |
| Warren, Ohio | 5,973 | 1896 | 1.5 | 1.5 | 462 | W. G. |
| Hopkinsville, Ky. | 5,833 | 1895 | 0.5 | 0.15 | 140 | N. Y. P. |
| Brainerd, Minn. | 5,703 | 1897 | 0.5 | 156 | N. Y. P. | |
| New Brighton, Pa. | 5,616 | 1889 | 0.5 | 156 | N. Y. P. | |
| Niagara Falls, N. Y. | 5,502 | 1896 | 4.5 | 2.62 | 1019 | J. G. |
| [Pg 249]Durham, N. C. | 5485 | 1893 | 0.9 | 0.7 | 252 | W. G. |
| Winfield, Kan. | 5184 | 1894 | 1 | 336 | W. G. | |
| Louisiana, Mo. | 5090 | 1888 | 0.8 | 242 | N. Y. P. & G. | |
| Trenton, Mo. | 5039 | 1889 | 0.4 | 128 | N. Y. P. | |
| Lorain, Ohio | 4863 | 1896 | 3 | 1.5 | 1356 | J. G. |
| Sidney, Ohio[55] | 4850 | 0.5 | N. Y. | |||
| Mexico, Mo. | 4789 | 1889 | 0.3 | 0.4 | 66 | N. Y. P. |
| Mt. Clemens, Mich. | 4748 | 1888 | 1 | 0.6 | 251 | N. Y. P. |
| Riverside, Cal. | 4683 | 1892 | 0.09 | 20 | N. Y. P. | |
| Columbus, Miss.[56] | 4559 | 1899 | 0.5 | 0.175 | 176 | J. G. |
| Winchester, Ky. | 4519 | 1894 | 0.75 | 0.107 | 152 | J. P. |
| Salisbury, N. C. | 4418 | 1889 | 0.5 | 0.35 | 156 | N. Y. P. |
| Eufaula, Ala. | 4394 | 1897 | 0.5 | 140 | N. Y. P. | |
| Greenville, Tex. | 4330 | 1888 | 0.8 | 0.175 | 156 | N. Y. P. |
| Exeter, N. H. | 4284 | 1887 | 0.114 | 0.179 | 34 | N. Y. P. |
| Bordentown, N. J. | 4232 | 1890 | 0.5 | 0.5 | 156 | N. Y. P. |
| Lake Forest, Ill. | 4203 | 1892 | 1 | 168 | J. P. | |
| Henderson, N. C. | 4191 | 1899 | 0.25 | 118 | W. G. | |
| Reading, Mass. | 4088 | 1896 | 1 | 0.198 | 336 | W. G. |
| Goldsboro, N. C. | 4017 | 1896 | 0.5 | 0.1 | 156 | W. G. |
| Rich Hill, Mo. | 4008 | 1893 | 0.5 | 0.24 | 140 | N. Y. P. |
| Mt. Pleasant, Ia. | 3997 | 1888 | 0.5 | 156 | N. Y. P. | |
| Murphysboro, Ill. | 3880 | 1890 | 0.2 | 60 | N. Y. P. | |
| Brandon, Manitoba | 3778 | 1893 | 1 | 0.36 | 240 | N. Y. P. |
| Danville, Ky. | 3766 | 1894 | 0.5 | 0.1 | 140 | N. Y. P. |
| Royersford, Pa. | 3612 | 1893 | 1 | 0.08 | 226 | J. G. |
| Warsaw, Ind. | 3514 | 1896 | 0.5 | 0.5 | 156 | N. Y. P. |
| Ashbury Park, N. J. | 3500 | 2 | 0.5 | 670 | C. | |
| Keyport, N. J. | 3411 | 1895 | 0.5 | 0.06 | 156 | W. G. |
| Deseronto, Ont. | 3338 | 1896 | 0.5 | 0.84 | 147 | J. P. |
| Milledgeville, Ga. | 3322 | 1893 | 0.5 | 156 | N. Y. P. | |
| Carlinville, Ill. | 3293 | 0.1 | 38 | Am. or Jackson. | ||
| Gettysburg, Pa. | 3221 | 1894 | 0.3 | 0.075 | 78 | W. G. |
| Independence, Kan. | 3127 | 1891 | 0.75 | 0.25 | 129 | Am. P. |
| LaGrange, Ga. | 3090 | 1893 | 0.25 | 34 | N. Y. P. | |
| Paola, Kan. | 2943 | 1887 | 0.25 | 0.45 | 66 | N. Y. P. |
| Benwood, W. Va.[56] | 2934 | 1899 | 1 | 306 | J. G. | |
| Gadsden, Ala. | 2901 | 1887 | 1.325 | 430 | N. Y. P. & G. | |
| Lamar, Mo. | 2860 | 1891 | 0.25 | 78 | N. Y. P. | |
| Longueuil, Que. | 2757 | 1895 | 0.4 | 0.3 | 100 | N. Y. P. |
| Washington, Mo. | 2725 | 1888 | 0.2 | 0.075 | 50 | N. Y. P. |
| Renfrew, Ont. | 2611 | 1897 | 0.432 | 100 | N. Y. P. | |
| [Pg 250]Oswego, Kan. | 2574 | 1893 | 0.5 | 0.3 | 140 | N. Y. P. |
| Holden, Mo. | 2520 | 1893 | 0.2 | 0.05 | 100 | N. Y. P. |
| Burlington, Kan. | 2239 | 0.5 | 79 | J. | ||
| Council Grove, Kan. | 2211 | 1898 | 0.5 | 0.08 | 78 | N. Y. |
| Wakefield, R. I.[56] | 2170 | 0.15 | 0.25 | N. Y. | ||
| Catonsville, Md. | 2115 | 1890 | 0.25 | 78 | N. Y. P. | |
| Attica, N. Y. | 1994 | 1896 | 0.4 | 100 | N. Y. P. | |
| Hightstown, N. J. | 1875 | 1899 | 0.25 | 0.025 | 78 | N. Y. G. |
| No. Berwick, Me. | 1803 | 1896 | 0.3 | 78 | W. G. | |
| Dunnville, Ont. | 1776 | 1899 | 0.5 | 140 | N. Y. P. | |
| Rogers Park, Ill. | 1708 | 1889 | 0.4 | 0.35 | 100 | N. Y. P. |
| Eatonton, Ga. | 1682 | 1897 | 0.5 | 132 | N. Y. G. | |
| Caldwell, Kan. | 1642 | 1890 | 0.5 | 156 | N. Y. P. | |
| LaGrange, Tex. | 1626 | 1891 | 0.25 | 34 | N. Y. P. | |
| Richfield Springs, N. Y. | 1623 | 1889 | 0.35 | 100 | N. Y. P. | |
| Valatie, N. Y. | 1437 | 1894 | 0.15 | 50 | N. Y. | |
| Tunkhannock, Pa. | 1253 | 0.1 | N. Y. | |||
| Mechanics Falls, Me. | 1030 | 1898 | 0.72 | 176 | W. G. | |
| New Bethlehem, Pa. | 1026 | 1899 | 0.1 | 50 | J. G. | |
| Fairmount, W. Va. | 1023 | 1898 | 1 | 280 | N. Y. P. | |
| Atlantic Highlands, N. J. | 945 | 0.3 | 0.109 | 130 | C. | |
| Rumford Falls, Me. | 898 | 1897 | 0.5 | 113 | W. G. | |
| Lakewood, N. J. | 730 | 1889 | 0.5 | 156 | N. Y. P. | |
| Veazie, Me. | 650 | 1889 | 1 | 0.1 | 176 | W. G. |
| Portersville, Cal. | 606 | 1890 | 0.151 | 0.060 | 34 | N. Y. P. |
| Holmesburg, Pa. | 1896 | 1 | 0.046 | 280 | N. Y. P. | |
| Pickering Creek, Pa. | 1896 | 0.75 | 234 | W. G. | ||
| Overbrook, Penna. | 1895 | 0.25 | 78 | W. G. | ||
| Vandergrift, Pa. | 1897 | 0.5 | 156 | W. G. | ||
| Frazerville, P. Q.[56] | 1899 | 0.2 | 78 | N. Y. G. | ||
| Arnate, Pa. | 1899 | 0.12 | 50 | N. Y. G. | ||
| Chihuahua, Mex.[56] | 1899 | 1 | 612 | J. G. | ||
| West Reading, Pa. | 0.07 | W. G. | ||||
| Totals | 1,565,881 | 252 | 108 | 77,806 | ||
Special filters, neither sand nor mechanical: Wilmington, Del.; Pop., 61,431; area, 10,000 sq. ft.; nominal capacity, 10 million gallons. See Eng. News, Vol. 40, p. 146.
[Pg 251]
NOTES REGARDING SAND FILTERS IN THE UNITED STATES.
Poughkeepsie, N. Y. Designed by James P. Kirkwood, built in 1872, was the earliest of its kind in the United States. It was enlarged by the Superintendent, Charles E. Fowler, in 1896. The walls of the original filters were of rubble, and in course of time developed cracks and leaked badly. The walls of the new filter are of rubble, faced with vitrified brick. The filters treat the water of the Hudson River, which is sewage-polluted and more or less muddy. Description: Jour. N. E. Water Works Assoc., Vol. 12, p. 209.
Hudson, N. Y. Designed by James P. Kirkwood, built in 1874. enlarged in 1888. The filters are open and are used for treating the Hudson River water, which is sewage-polluted and more or less muddy. Description: Eng. News, Vol. 31, p. 487.
St. Johnsbury, Vt. (E. & T. Fairbanks & Co.) These filters were built about 30 years ago, and have been recently enlarged. The filters were originally open, but were afterwards covered with a roof. The single roof proved inadequate to keep them from freezing, and a second roof was added inside and under the main roof. They are used for filtering pond water, which is quite clear and not subject to much pollution. The water supply is one of two, the other is the town supply and is taken from the Passumpsic River. No published description.
Nantucket, Mass. Designed by J. B. Rider, built in 1892. This filter is used to remove organisms from the reservoir water supply. It is only used when the organisms are troublesome, and is satisfactory in preventing the tastes and odors which formerly resulted from their presence. Description: Jour. N. E. Water Works Assoc., Vol. 8, p. 171; Eng. News, Vol. 31, p. 336.
Lawrence, Mass. Designed by Hiram F. Mills, built in 1892-3, and put in operation September, 1893. It is used for treating the water of the Merrimac River, which contains a large amount[Pg 252] of sewage. Description: Report of the Mass. State Board of Health, 1893, p. 543; Jour. N. E. Water Works Assoc., Vol. 9, p. 44; Eng. News, Vol. 30, p. 97.
Ilion, N. Y. Designed by the Stanwix Engineering Company and are used for treating reservoir water, which is generally clear and not subject to pollution. Description: Eng. News, Vol. 31, p. 466.
Mount Vernon, N. Y. (New York Suburban Water Company.) Designed by J. N. Chester, built in 1894. These filters are similar in general construction to the Lawrence filter, although the dimensions both vertical and horizontal are reduced, and the area is divided into three parts. The filters are used for treating reservoir water, which is generally quite clear, but which is polluted by a considerable amount of sewage. Since the use of filters the reduction in the typhoid fever death-rate has been very great. Description: Eng. News, Vol. 32, p. 155.
Milford, Mass. Designed by F. L. Northrop. This filter is very simple in construction, and is used for filtering Charles River water as an auxiliary supply. Description: Jour. N. E. Water Works Assoc., Vol. 10, p. 262.
Grand Forks, N. D. Designed by W. S. Russell. These filters are covered with roofs. They treat the water from the Red River, which is very muddy, and also sewage-polluted, and which formerly caused typhoid fever. Description: Eng. News, Vol. 33, p. 341.
Ashland, Wis. Designed by William Wheeler, built in 1895. The Ashland filters were the first vaulted masonry filters to be constructed in the United States, and are used for treating the bay water, which is polluted with sewage, and is at times muddy from the river water discharging into the bay near the intake. The filters are below the bay level, and receive water from it by gravity. Description: Jour. N. E. Water Works Assoc., Vol. 11, p. 301; Eng. News, Vol. 38, p. 338.
Lambertville, N. J. Designed by Churchill Hungerford, and[Pg 253] built in 1896. These are open filters with earth embankments, for filtration of reservoir water. Description: Eng. News, Vol. 36, p. 4.
Far Rockaway, L. I. (Queens County Water Company.) Designed by Charles R. Bettes, Engineer in Charge; Charles B. Brush & Co., Chief Engineers; and Allen Hazen, Consulting Engineer. Constructed in 1896. These masonry filters were used for the removal of iron from well waters. They are also designed to be suitable for the filtration of certain brook waters which are available as auxiliary supplies, but the brook water has been but rarely used. Description: Eng. Record, Vol. 40, p. 412.
Red Bank, N. J. (Rumson Improvement Company.) Designed by Allen Hazen, built in 1897. They are similar in construction to the Far Rockaway filters, and are used for iron removal only. Description: Eng. Record, Vol. 40, p. 412.
Hamilton, N. Y. Designed by the Stanwix Engineering Company, and were built in 1895 to filter lake water. Description: Eng. News, Vol. 39, p. 254.
Little Falls, N. Y. Designed by Stephen E. Babcock. These filters are open, and were built in 1898, and are used for filtering river water. Description: Eng. Record, Vol. 38, p. 7.
Somersworth, N. H. Designed by William Wheeler. These were the second vaulted filters to be built in the United States. The supply is from the Salmon Falls River and flows to the filters by gravity, the filters being below the river level. Description: Eng. News, Vol 40, p. 358; Eng. Record, Vol. 38, p. 270.
Berwyn, Penna. Designed by J. W. Ledoux. These open filters are used for filtering creek water. Description: Eng. News, Vol. 41, p. 150.
Harrisburg, Penna. (State Lunatic Hospital.) Designed by Allen Hazen; open masonry filters, used for treating the water from a small creek which is often muddy and is subject to pollution. No published description.
[Pg 254]
Albany, N. Y. Designed by Allen Hazen. Constructed 1898-99. This was the third and is the largest vaulted masonry filter plant yet constructed in the United States. It is used for filtering the Hudson River water, which is slightly muddy and much polluted by sewage. Description: Eng. News, Vol. 39, p. 91; Vol. 40, p. 254.
Rock Island, Ill. Designed by Jacob A. Harman. Open filters with embankments, used for filtering the Mississippi River water, which is very muddy and also polluted by sewage. No published description.
CAPACITY OF FILTERS.
Estimating the total additional area of sand filters for which figures are not available at 100 acres, and the maximum capacity of sand filters at three million gallons per acre daily, and of mechanical filters at three million gallons per thousand square feet of filtering area, the total filtering capacity of all the filters in the world used for public water supplies in 1899 is nearly 1600 million gallons daily, of which 15 per cent is represented by mechanical filters and 85 per cent by sand filters. In the United States, including Wilmington, the total filtering capacity is nearly 300 million gallons daily, of which 18 per cent is represented by sand filters, 79 per cent by mechanical filters, and 3 per cent by a special type of filters.
[Pg 255]
APPENDIX V.
LONDON’S WATER-SUPPLY.
London alone among great capitals is supplied with water by private companies. They are, however, under government supervision, and the rates charged for water are regulated by law. There are eight companies, each of which supplies its own separate district, so that there is no competition whatever. One of the companies supplying 460,000 people uses only ground-water drawn from deep wells in the chalk, but the other seven companies depend mainly upon the rivers Thames and Lea for their water. All water so drawn is filtered, and must be satisfactory to the water examiner, who is required to inspect the water supplied by each company at frequent intervals, and the results of the examinations are published each month.
In 1893 the average daily supply was 235,000,000 gallons, of which about 40,000,000 were drawn from the chalk, 125,000,000 from the Thames, and 70,000,000 from the Lea. Formerly some of the water companies drew water from the Thames within the city where it was grossly polluted, and the plagues and cholera which formerly ravaged London were in part due to this fact. These intakes were abandoned many years ago, and all the companies now draw their water from points outside of the city and its immediate suburbs.
The area of the watershed of the Thames above the intakes of the water companies is 3548 square miles, and the population living upon it in 1891 was 1,056,415. The Thames Conservancy Board has control of the main river for its whole length, and of all tributaries within ten miles in a straight line of the main river, but has no[Pg 256] jurisdiction over the more remote feeders. The area drained is essentially agricultural, with but little manufacturing, and there are but few large towns. In the area coming under the conservators there are but six towns with populations above 10,000 and an aggregate population of 170,000, and there are but two or three other large towns on the remaining area more than ten miles from the river. These principal towns are as follows:
| Town. | Population 1891. | Distance above Water Intakes. |
|---|---|---|
| Reading | 60,054 | 49 miles |
| Oxford | 45,791 | 87 miles |
| New Swindon | 27,295 | 116 miles |
| High Wycomb | 13,435 | 33 miles |
| Windsor | 12,327 | 18 miles |
| Maidenhead | 10,607 | 25 miles |
| Guildford | 14,319 | 20 miles |
Guildford is outside of the conservators’ area. All of the above towns treat their sewage by irrigation.
Among the places that are regarded as the most dangerous are Chertsey and Staines, with populations of 9215 and 5060, only 8 and 11 miles above the intakes respectively. These towns are only partially sewered and still depend mainly on cesspools. An attempt is made to treat the little sewage which they produce upon land, but the work has not as yet been systematically carried out. There are also several small towns of 3000 inhabitants or less upon the upper river which do not treat their sewage so far as they have any, but, owing to their great distance, the danger from them is much less than from Chertsey and Staines. Twenty-one of the principal towns upon the watershed have sewage farms, and there are no chemical precipitation plants now in use.
Boats upon the river are not allowed to drain into it, but are compelled to provide receptacles for their sewage, and facilities are provided for removing and disposing of it; and as an additional precaution no boat is allowed to anchor within five miles of the intakes.
[Pg 257]
The conservators of the river Lea have control of its entire drainage area, which is about 460 square miles, measured from the East London water intakes, and has a population of 189,287. On this watershed there is but a single town with more than 10,000 inhabitants, this being Lutton near the headwaters of the river, with a population of 30,005. The sewage from Lutton and from seventeen smaller places is treated upon land. No crude sewage is known to be ordinarily discharged into the river. At Hereford, eleven miles above the East London intakes, there is a chemical precipitation plant. The conservators do not regard this treatment as satisfactory, and have recently conducted an expensive lawsuit against the local authorities to compel them to further treat their effluent. The suit was lost, the court holding that no actual injury to health had been shown. It is especially interesting to note that of the thirty-nine places on the Thames and the Lea giving their sewage systematic treatment there is but a single place using chemical precipitation, and there it is not considered satisfactory. Formerly quite a number of these towns used other processes than land treatment, but in every case but Hereford land treatment has been substituted.
In regard to the efficiency of the sewage farms, it is believed that in ordinary weather the whole of the sewage percolates through the land, and the inspectors of the Conservancy Boards strongly object to its being allowed to pass over the surface into the streams. The land, however, is for the most part impervious, as compared to Massachusetts and German sewage farms, and in times of heavy storms the land often has all the water it can take without receiving even the ordinary flow of sewage, and much less the increased storm-flow. At such times the sewage either does go over the surface, or perhaps more frequently is discharged directly into the rivers without even a pretence of treatment. The conservators apparently regard this as an unavoidable evil and do not vigorously oppose it. It is the theory that, owing to the increased dilution with the storm-flows, the matter is comparatively harmless,[Pg 258] although it would seem that the reduced time required for it to reach the water-works intakes might largely offset the effect of increased dilution.
The water companies have large storage and sedimentation basins with an aggregate capacity equal to nine days’ supply, but the proportion varies widely with the different companies. It is desired that the water held in reserve shall be alone used while the river is in flood, as, owing to its increased pollution, it is regarded as far more dangerous than the water at other times; but as no record is kept of the times when raw sewage is discharged, and no exact information is available in regard to the times when the companies do not take in raw water, it can safely be assumed that a considerable amount of raw sewage does become mixed with the water which is drawn by the companies.
The water drawn from the river is filtered through 113 filters having an area of 116 acres. None of the filters are covered, and with an average January temperature of 39° but little trouble with ice is experienced. A few new filters are provided with appliances for regulating the rate on each filter separately and securing regular and determined rates of filtration, but nearly all of the filters are of the simple type described on page 48, and the rates of filtration are subject to more or less violent fluctuation, the extent of which cannot be determined.
The area of filters is being continually increased to meet increasing consumption; the approximate areas of filters in use having been as follows:
| 1839 | First filters built |
| 1855 | 37 acres |
| 1866 | 47 acres |
| 1876 | 77 acres |
| 1886 | 104 acres |
| 1894 | 116 acres |
There has been a tendency to reduce somewhat the rate of filtration. In 1868, with 51 acres of filters, the average daily quantity of[Pg 259] water filtered was 111,000,000 gallons, or 2,180,000 gallons per acre. In 1884, with 97 acres of filter surface, the daily quantity filtered was 157,000,000 gallons, or 1,620,000 gallons per acre; and in 1893, with 116 acres of filter surface and 195,000,000 gallons daily, the yield per acre was 1,680,000 gallons.
Owing to the area of filter surface out of use while being cleaned, the variations in consumption of water, and the imperfections of the regulating apparatus, the actual rates of filtration are often very much higher and at times may easily be double the figures given.
Evidence regarding the healthfulness of the filtered river-water was collected and examined in a most exhaustive manner in 1893 by a Royal Commission appointed to consider the water-supply of the metropolis in all its aspects with reference to future needs. This commission was unable to obtain any evidence whatever that the water as then supplied was unhealthy or likely to become so, and they report that the rivers can safely be depended upon for many years to come.
The numbers of deaths from all causes and from typhoid fever annually per million of inhabitants for the years 1885-1891 in the populations receiving their waters from different sources in London were as follows:
| Water used. | Deaths from All Causes. | Deaths from Typhoid Fever. |
|---|---|---|
| Filtered Thames water only | 19,501 | 125 |
| Filtered Lea water only | 21,334 | 167 |
| Kent wells only | 18,001 | 123 |
| Thames and Lea jointly | 18,945 | 138 |
| Thames and Kent jointly | 18,577 | 133 |
The population supplied exclusively from the Lea by the East London Company is of a poorer class than that of the rest of London, and this may account for the slightly higher death-rate in this section. Aside from this the rate is remarkably uniform and shows no great difference between the section drinking ground-water only and those drinking filtered river-waters. The death-rate from[Pg 260] typhoid fever is also very uniform and, although higher than that of some Continental cities with excellent water-supplies (Berlin, Vienna, Munich, Dresden), is very low—lower than in any American city of which I have records.
In this connection, it was shown by the Registrar-General that there is only a very small amount of typhoid fever on the watersheds of the Thames and Lea, so that the danger of infection of the water as distinct from pollution is less than would otherwise be the case. Thus for the seven years above mentioned the numbers of deaths from typhoid fever per million of population were only 105 and 120 on the watersheds of the Thames and the Lea respectively, as against 176 for the whole of England and Wales.
| LONDON FILTERS, 1896. | |||||||
|---|---|---|---|---|---|---|---|
| Twenty-sixth Annual Report of the Local Government Board, pages 206-213. | |||||||
| Company. | Amount of Storage Raw Water, Days. |
Average Thickness of Sand, Feet. |
Average Rate of Filtration. |
Bacterial Efficiency. | |||
| Imperial Gallons per Square Foot per Hour. |
Millions U. S. Gallons per Acre Daily. |
Maximum. | Minimum. | Average. | |||
| Chelsea | 12.0 | 4.0 | 1.75 | 2.19 | 99.92 | 99.62 | 99.86 |
| West Middlesex | 5.6 | 2.75 | 1.25 | 1.56 | 99.94 | 91.48 | 99.79 |
| Southwark & Vauxhall | 4.1 | 2.5 | 1.5 | 1.88 | 100.00 | 84.33 | 97.77 |
| Grand Junction | 3.3 | 2.25 | 1.63 | 2.05 | 99.98 | 84.03 | 99.31 |
| Lambeth | 6.0 | 2.8 | 2.08 | 2.60 | 99.97 | 96.45 | 99.81 |
| New River | 2.2 | 4.4 | 1.89 | 2.37 | 100.00 | 77.14 | 99.07 |
| East London | 15.0 | 2.0 | 1.33 | 1.67 | 99.93 | 97.03 | 99.56 |
[Pg 261]
APPENDIX VI.
THE BERLIN WATER-WORKS.
The original works were built by an English company in 1856, and were sold to the city in 1873 for $7,200,000.
The water was taken from the river Spree at the Stralau Gate, which was then above, but is now surrounded by, the growing city. The water was always filtered, and the original filters remained in use until 1893, when they were supplanted by the new works at Lake Müggel. Soon after acquiring the works the city introduced water from wells by Lake Tegel as a supplementary supply, but much trouble was experienced from crenothrix, an organism growing in ground-waters containing iron, and in 1883 this supply was replaced by filtered water from Lake Tegel. With rapidly-increasing pollution of the Spree at Stralau the purity of this source was questioned, and in 1893 it was abandoned (although still held as a reserve in case of urgent necessity), the supply now being taken from the river ten miles higher up, at Müggel.
The watershed of the Spree above Stralau, as I found by map measurement, is about 3800 square miles; the average rainfall is about 25 inches yearly. At extreme low water the river discharges 457 cubic feet per second, or 295 million gallons daily, and when in flood 5700 cubic feet per second may be discharged. The city is allowed by law to take 46 million gallons daily for water-supply, and this quantity can be drawn either at Stralau or at Müggel.
Above Stralau the river is polluted by numerous manufactories and washing establishments, and by the effluent from a considerable part of the city’s extensive sewage farms. The shipping on this part of the river also is heavy, and sewage from the boats is discharged[Pg 262] directly into the river. The average number of bacteria in the Spree at this point is something over ten thousand per cubic centimeter, and 99.6 per cent of them were removed by the filters in 1893.
The watershed of the Spree above the new water-works at Müggel I found by map measurement to be 2800 square miles, and the low water-discharge is said to be 269 million gallons daily. The river at this point flows through Lake Müggel, which forms a natural sedimentation-basin, and the raw water is quite clear except in windy weather.
There were 16 towns on the watershed with populations above 2000 each in 1890, and an aggregate population of 132,000, which does not include the population of the smaller places or country districts. None of these places purify their sewage so far as they have any. Fürstenwalde with a population of 12,935, and 22 miles above Müggel, has surface sewers discharging directly into the river. Above Fürstenwalde the river runs through numerous lakes which probably remove the effect of the pollution from the more distant cities. There is considerable shipping on the river for some miles above Fürstenwalde (which forms a section of the Friedrich Wilhelm Canal), but hardly any between Müggel and Fürstenwalde. The raw water at Müggel contains two or three hundred bacteria per cubic centimeter, and is thus a comparatively pure water before filtration. It is slightly peaty and the filtered water has a light straw color.
Lake Tegel, which supplies the other part of the city’s supply, is an enlargement of the river Havel. The watershed above Tegel I find to be about 1350 square miles, and the annual rainfall is about 22 inches. The low water-discharge is said to be 182 million gallons daily, and the city is allowed by law to take 23 million gallons for water-supply.
There were ten towns upon the watershed with populations above 2000 each in 1890, and with an aggregate population of 44,000. Of these Tegel is directly upon the lake with a population of 3000,[Pg 263] and Oranienburg, 14 miles above, has a population of 6000 and is rapidly increasing. The shipping on the lake and river is heavy. The lake water ordinarily contains two or three hundred bacteria per cubic centimeter. The lake is shallow and becomes turbid in windy weather.
There are 21 filter-beds at Tegel with a combined area of 12.40 acres to furnish a maximum of 23 million gallons of water daily, and 22 filters at Müggel with a combined area of 12.7 acres to deliver the same quantity. Twenty-two more filters will be built at Müggel within a few years to purify the full quantity which can be taken from the river. All of these filters are covered with brick arches supported by pillars about 16 feet apart from centre to centre in each direction, and the whole is covered by nearly 3 feet of earth, making them quite frost-proof. The original filters at Stralau were open, but much difficulty was experienced with them in winter.
The bottom of the filters at Tegel consists of 8 inches of concrete above 20 inches of packed clay and with 2 inches of cement above, and slopes slightly from each side to the centre. The central drain goes the whole length of the filters and has a uniform cross-section of about 1⁄7300 of the area of the whole bed. There are no lateral drains, but the water is brought to the central drain by a twelve-inch layer of stones as large as a man’s fist; above this there is another foot of gravel of graded sizes supporting two feet of fine sand, which is reduced by scraping to half its thickness before the sand is replaced. The average depth of water above the sand is nearly 5 feet. The filters are not allowed to filter at a rate above 2.57 million gallons per acre daily, and at this rate with 70 per cent of the area in service the whole legal quantity of water can be filtered. The filters work at precisely the same rate day and night, and the filtered water is continuously pumped as filtered to ample storage reservoirs at Charlottenburg. The pumps which lift the water from the lake to the filters work against a head of 14 feet. The apparatus for regulating the rate of filtration was described on page 51.
As yet no full description of the Müggel works has been published,[Pg 264] but they resemble closely the Tegel works. Both were designed by or under the direction of the late director of the water-works, Mr. Henry Gill.
The average daily quantity of water supplied for the fiscal year ending March 31, 1893, was 29,000,000 gallons daily, which estimate allows 10 percent for the slip of the pumps. Of this quantity 9,650,000 was furnished by Stralau and 19,350,000 by Tegel. The greatest consumption in a single day was 43,300,000 gallons, or 26.6 gallons per head, while the average quantity for the year was 18.4 gallons per head. All water without exception is sold by meter, the prices ranging from 27.2 cents a thousand gallons for small consumers to 13.6 cents for large consumers and manufacturers. The average receipts for all water pumped, including that used for public purposes and not paid for, were 15.4 cents a thousand gallons, against the cost of production, 9.8 cents, which covers operating expenses, interest on capital, and provision for sinking fund. This leaves a handsome net profit to the city. On account of the comparatively high price of the city water and the ease with which well-water is obtained, the latter is almost exclusively used for running engines, manufacturing purposes, etc., and this in part explains the very low per-capita consumption.
The volume of sewage, however, for the same year, including rain-water, except during heavy showers, was only 29 gallons per head, showing even with the private water-supplies an extraordinarily low consumption.
The friction of the water in the 4.75 miles of 3-foot pipe between Tegel and the reservoir at Charlottenburg presents an interesting point. When well-water with crenothrix was pumped, the friction rose to 34.5 feet, when the velocity was 2.46 feet per second. According to Herr Anklamm, who had charge of the works at the time, the friction was reduced to 19.7 feet when filtered water was used and after the pipe had been flushed, and this has not increased with continued use. He calculated the friction for the velocity according to Darcy 15.0 feet, Lampe 17.8 feet, Weisbach 18.7 feet, and Prony 21.5 feet.
[Pg 265]
APPENDIX VII.
ALTONA WATER-WORKS.
The Altona water-works are specially interesting as an example of a water drawn from a source polluted to a most unusual extent: the sewage from cities with a population of 770,000, including its own, is discharged into the river Elbe within ten miles above the intake and upon the same side.
The area of the watershed of the Elbe above Altona is about 52,000 square miles, and the average rainfall is estimated to be about 28 inches, varying from 24 or less near its mouth to much higher quantities in the mountains far to the south. On this watershed there are 46 cities, which in 1890 had populations of over 20,000 each, and in addition there is a permanent population upon the river-boats estimated at 20,000, making in all 5,894,000 inhabitants, without including either country districts or the numberless cities with less than 20,000 inhabitants each. The sewage from about 1,700,000 of these people is purified before being discharged; and assuming that as many people living in cities smaller than 20,000 are connected with sewers as live in larger places without being so connected, the sewage of over four million people is discharged untreated into the Elbe and its tributaries.
The more important of these sources of pollution are the following:
| City | Population in 1890. |
On what River. |
Approximate Distance, Miles. |
|---|---|---|---|
| Shipping | 20,000 | —— | —— |
| Altona | 143,353 | Elbe | 6 |
| Hamburg | 570,534 | Elbe | 7 |
| Wandsbeck | 20,586 | Elbe | 8 |
| Harburg | 35,101 | Elbe | 11 |
| Magdeburg | 202,325 | Elbe | 185 |
| Dresden | 276,085 | Elbe | 354 |
| Berlin and suburbs | 1,787,859 | Havel | 243 |
| Halle | 101,401 | Saale | 272 |
| Leipzig | 355,485 | Elster | 305 |
| Chemnitz | 138,955 | Mulde | 340 |
| Prague | 310,483 | Moldau | 500 |
The sewage of Berlin and of most of its suburbs is treated before being discharged, and in addition the Havel flows through a series of lakes below the city, allowing better opportunities for natural purification than in the case of any of the other cities. Halle treats less than a tenth of its sewage. Magdeburg will treat its sewage in the course of a few years. Leipzig, Chemnitz, and other places are thinking more or less seriously of purification.
The number of bacteria in the raw water at Altona fluctuates with the tide and is extremely variable; numbers of 50,000 and 100,000 are not infrequent, but 10,000 to 40,000 is perhaps about the usual range.
The works were originally built by an English company in 1860, and have since been greatly extended. They were bought by the city some years ago. The water is pumped directly from the river to a settling-basin upon a hill 280 feet above the river. From this it flows by gravity through the filters to the slightly lower pure-water reservoir and to the city without further pumping. The filters are open, with nearly vertical masonry walls, as described in Kirkwood’s report. The cross-section of the main underdrain is 1⁄2800 of the area of the beds.
Considerable trouble has been experienced from frost. With continued cold weather it is extremely difficult to satisfactorily scrape the filters, and very irregular rates of filtration may result at such times. In the last few years, with systematic bacterial investigation, it has been found that greatly decreased efficiency[Pg 267] frequently follows continued cold weather, and the mild epidemics of typhoid fever from which the city has long suffered have generally occurred after these times. Thus a light epidemic of typhoid in 1886 came in March, following a light epidemic in Hamburg. In 1887 a severe epidemic in February followed a severe epidemic in Hamburg in December and January. In 1888 a severe epidemic in March followed an epidemic in Hamburg lasting from November to January. Hamburg’s epidemic of 1889, coming in warm weather, September and October, was followed by only a very slight increase in Altona. In 1891 Altona suffered again in February from a severe epidemic, although very little typhoid had been in Hamburg. A less severe outbreak also came in February, 1892, and a still slighter one in February, 1893. In the ten years 1882-1892, of five well-marked epidemics, three broke out in February and two in March, while two smaller outbreaks came in December and January. No important outbreak has ever occurred in summer or in the fall months, when typhoid is usually most prevalent, thus showing clearly the bad effect of frost upon open filters (see Appendix II). With steadily increasing consumption the sedimentation-basin capacity of late years has become insufficient as well as the filtering area, and it is not unlikely that with better conditions a much better result could be obtained in winter even with open filters.[63]
The brilliant achievement of the Altona filters was in the summer of 1892, when they protected the city from the cholera which[Pg 268] so ravaged Hamburg, although the raw water at Altona must have contained a vastly greater quantity of infectious matter than that which worked such havoc in Hamburg.
From these records it appears that for about nine months of the year the Altona filters protect the city from the impurities of the Elbe water, but that during cold weather, with continued mean temperatures below the freezing-point, such protection is not completely afforded, and bad effects have occasionally resulted. Notwithstanding the recent construction of open filters in Hamburg it appears to me that there must always be more or less danger from open filters in such a climate. Hamburg’s danger, however, will be much less than Altona’s on account of its better intake above the outlets of the sewers of Hamburg and Altona, which are the most important points of pollution at Altona.
[Pg 269]
APPENDIX VIII.
HAMBURG WATER-WORKS.
The source and quality of the water previously supplied has been sufficiently indicated in Appendix II. It was originally intended to filter the water, but the construction of filters was postponed from time to time until the fall of 1890, when the project was seriously taken up, and work was commenced in the spring of 1891. Three years were allowed for construction. In 1892, however, the epidemic of cholera came, killing 8605 residents and doing incalculable damage to the business interests of the city. The health authorities found that the principal cause of this epidemic was the polluted water-supply. To prevent a possible recurrence of cholera in 1893, the work of construction of the filters was pressed forward much more rapidly than had been intended. Electric lights were provided to allow the work to proceed nights as well as days, and as a result the plant was put in operation May 27, 1893, a full year before the intended time. Owing to the forced construction the cost was materially increased.
The new works take the raw water from a point one and a half miles farther up-stream, where it is believed the tide can never carry the city’s own sewage, as it did frequently to the old intake. The water is pumped from the river to settling-basins against heads varying with tide and the water-level in the basins from 8 to 22 feet. Each of the four settling-basins has an area of about 10 acres, and, with the water 6.56 feet deep, holds 20,500,000 gallons, or 82,000,000 gallons in all. The works are intended to supply a maximum of 48,000,000 gallons daily, but the present average consumption is only about 35,000,000 gallons (1892), or 59 gallons per[Pg 270] head for 600,000 population. This consumption is regarded as excessive, and it is hoped that it will be reduced materially by the more general use of meters. The sedimentation-basins are surrounded by earthen embankments with slopes of 1:3, the inner sides being paved with brick above a clay layer. The water flows by gravity from these basins to the filters, a distance of 11⁄2 miles, through a conduit 81⁄2 feet in diameter. The flow of the water out of the basins and from the lower end of the conduit is regulated by automatic gates connected with floats, shown by Fig. 11, page 60.
The filters are 18 in number, and each has an effective area of 1.89, or 34 acres in all. They are planned to filter at a rate of 1.60 million gallons per acre daily, which with 16 filters in use gives a daily quantity of 48,000,000 gallons as the present limit of the works. The sides of the filters are embankments with 1:2 slopes. Both sides and bottoms have 20 inches of packed clay, above which are 4 inches of puddle, supporting a brick pavement laid in cement. The bricks are laid flat on the bottom, but edge-wise on the sides where they will come in contact with ice.
The main effluent-drain has a cross-section for the whole length of the filter of 4.73 square feet, or 1⁄17000 of the area of the filter; and even at the low rate of filtration proposed, the velocity in the drain will reach 0.97 foot. The drain has brick sides, 1.80 feet high, covered with granite slabs. The lateral drains are all of brick with numerous large openings for admission of water. They are not ventilated, and I am unable to learn that any bad results follow this omission.
The filling of the filters consists of 2 feet of gravel, the top being of course finer than the bottom layers, above which are 40 inches of sand, which are to be reduced to 24 inches by scraping before being refilled. The water over the sand, when the latter is of full depth, is 43 inches deep, and will be increased to 59 inches with the minimum sand-thickness. The apparatus for regulating the rate of filtration was described page 52. The cost of the entire plant, including 34 acres effective filter-surface, 40 acres of sedimentation-basins,[Pg 271] over 2 miles of 81⁄2-foot conduit, pumping-machinery, sand-washing apparatus, laboratory, etc., was about 9,500,000 marks, or $2,280,000. This all reckoned on the effective filter area is $67,000 per acre, or $3.80 per head for a population of 600,000.
The death-rate since the introduction of filtered water has been lower than ever before in the history of the city, but as it is thought that other conditions may help to this result, no conclusions are as yet drawn.
| DEATHS IN HAMBURG FROM ALL CAUSES, AND FROM TYPHOID FEVER, BEFORE AND AFTER THE INTRODUCTION OF FILTERS. | |||
|---|---|---|---|
| Year. | Deaths from all Causes per 1000 Living. |
Deaths from Typhoid Fever per 100,000 Living. |
|
| 1880 | 24.9 | 26 | |
| 1881 | 24.1 | 30 | |
| 1882 | 23.7 | 27 | |
| 1883 | 25.2 | 25 | |
| 1884 | 25.1 | 26 | |
| 1885 | 25.3 | 42 | |
| 1886 | 29.0 | 71 | |
| 1887 | 26.6 | 88 | |
| 1888 | 24.5 | 54 | |
| 1889 | 23.5 | 43 | |
| 1890 | 22.0 | 27 | |
| 1891 | 23.4 | 24 | |
| 1892 | 41.1 | 34 | Cholera year. |
| 1893 | 20.2 | 18 | Filtered water from May 28. |
| 1894 | 17.9 | 7 | |
| 1895 | 19.0 | 11 | |
| 1896 | 17.3 | 6 | |
| 1897 | 17.0 | 7 | |
| 1898 | 17.5 | 5 | |
Average for 5 years, excluding cholera year, before filtration, 1887 to 1891 |
24.0 | 47.2 | |
Average for 5 years with filtration, 1894 to 1898 |
17.7 | 7.2 | |
[Pg 272]
APPENDIX IX.
NOTES ON SOME OTHER EUROPEAN WATER-SUPPLIES.
Amsterdam.—The water is derived from open canals in the dunes. These canals have an aggregate length of about 15 miles, and drain about 6200 acres. The water, as it enters the canals from the fine dune-sand, contains iron, but this is oxidized and deposited in the canals. The water after collection is filtered. It has been suggested that by using covered drains instead of open canals for collecting the water, the filtration would be unnecessary; but, on the other hand, the cost of building and maintaining covered drains in the very fine sand would be much greater than that of the canals, and it is believed, also, that the water so collected would contain iron, the removal of which might prove as expensive as the present filtration. In 1887 filters were built to take water from the river Vecht, but the city has refused to allow the English company which owns the water-works to sell this water for domestic purposes, and it is only used for public and manufacturing purposes, only a fraction of the available supply being required. Leyden, the Hague, and some other Dutch cities have supplies like the dune supply of Amsterdam, and they are invariably filtered.
Antwerp is also supplied by an English company. The raw water is drawn from a small tidal river, which at times is polluted by the sewage of Brussels. It is treated by metallic iron in Anderson revolver purifiers, and is afterward filtered at a rather low average rate. The hygienic results are closely watched by the city authorities, and are said to be satisfactory.
Rotterdam.—The raw water is drawn from the Maas, as the[Pg 273] Dutch call the main stream of the Rhine after it crosses their border. The population upon the river and its tributaries in Switzerland, Germany, Holland, France, and Belgium is very great; but the flow is also great, and the low water flow is exceptionally large in proportion to the average flow, on account of the melting snow in summer in Switzerland, where it has its origin.
The original filters had wooden under-drains, and there was constant trouble with crenothrix until the filters were reconstructed without wood, since which time there has been no farther trouble. The present filters are large and well managed. There is ample preliminary sedimentation.
Schiedam.—The filters at Schiedam are comparatively small, but are of unusual interest on account of the way in which they are operated. The intake is from the Maas just below Rotterdam. The city was unable to raise the money to seek a more distant source of supply, and the engineer, H. P. N. Halbertsma, was unwilling to recommend a supply from so doubtful a source without more thorough treatment than simple sand-filtration was then thought to be. The plan adopted is to filter the supply after preliminary sedimentation through two filters of 0.265 acre each, and the resulting effluent is then passed through three other filters of the same size. River sand is used for the first, and the very fine dune sand for the second filtration. The cost both of construction and operation was satisfactory to the city, and much below that of any other available source; and the hygienic results have been equally satisfactory, notwithstanding the unfavorable position of the intake.
Magdeburg.—The supply is drawn from the Elbe, and is filtered through vaulted filters after preliminary sedimentation. The pollution of the river is considerable, although less than at Altona or even at Hamburg. The city has been troubled at times by enormous discharges of salt solution from salt-works farther up, which at extreme low water have sometimes rendered the whole river brackish and unpleasant to the taste; but arrangements have[Pg 274] now been made which, it is hoped, will prevent the recurrence of this trouble.
Breslau is supplied with filtered water from the river Oder, which has a watershed of 8200 square miles above the intake, and is polluted by the sewage from cities with an aggregate population of about 200,000, some of which are in Galicia, where cholera is often prevalent. In recent years the city has been free from cholera, and from more than a very limited number of typhoid-fever cases; but the pollution is so great as to cause some anxiety, notwithstanding the favorable record of the filters, and there is talk of the desirability of securing another supply. Until 1893 there were four filter-beds, with areas of 1.03 acres each, and not covered. In 1893 a fifth bed was added. This is covered by vaulting and is divided into four sections, which are separately operated, so that it is really four beds of 0.25 acre each. The vaulting is concrete arches, supported by steel I beams in one direction.
Budapest.—A great variety of temporary water-supplies have at different times been used by this rapidly growing city. The filters which for some years have supplied a portion of the supply have not been altogether satisfactory; but perhaps this was due to lack of preliminary sedimentation for the extremely turbid Danube water, and also to inadequate filter-area. The city is rapidly building and extending works for a supply of ground-water, and in 1894 the filters were only used as was necessary to supplement this supply, and it was hoped that enough well-water would be obtained to allow the filters to be abandoned in the near future. The Danube above the intake receives the sewage of Vienna and innumerable smaller cities, but the volume of the river is very great compared to other European streams, so that the relative pollution is not so great as in many other places.
Zürich.—The raw water is drawn by the city from the Lake of Zürich near its outlet, and but a few hundred feet from the heart of the city. Although no public sewers discharge into the[Pg 275] lake, there is some pollution from boats and bathers and other sources, and, judging by the number of bacteria in the raw water, this pollution is increasing. The raw water is extremely free from sediment, and the filters only become clogged very slowly. The rate of filtration is high, habitually reaching 7,000,000 gallons per acre daily; but, with the clear lake water and long periods between scrapings, the results are excellent even at this rate. The filters are all covered with concrete groined arches.
Filtration was commenced in 1886, and was followed by a sharp decline in the amount of typhoid fever, which, up to that time, had been rather increasing; for the six years before the change there were sixty-nine deaths from this cause annually per 100,000 living, and for the six years after only ten, or one seventh as many; and this reduction is attributed by the local authorities to the filtration.[64]
St. Petersburg.—The supply is drawn from the Neva River by an English company, and is filtered through vaulted filters at a very high rate.
Warsaw.—The supply is drawn from the Weichsel River by the city, and is filtered through vaulted filters after preliminary sedimentation at a rate never exceeding 2,570,000 gallons per acre daily.
THE USE OF UNFILTERED SURFACE-WATERS.
The use of surface-water without filtration in Europe is comparatively limited. In Germany this use is now prohibited by the Imperial Board of Health. In Great Britain, Glasgow draws its supply unfiltered from Loch Katrine; and Manchester and some other towns use unfiltered waters from lakes or impounding reservoirs the watersheds of which are entirely free from population. The best English practice, however, as in Germany, requires the filtration of such waters even if they are not known to receive sewage, and the[Pg 276] unpolluted supplies of Liverpool, Bradford, Dublin, and many other cities are filtered before use.
THE USE OF GROUND-WATER.[65]
Ground-waters are extensively used in Europe, and apparently in some localities the geological formations are unusually favorable to this kind of supply. Paris derives all the water it now uses for domestic purposes from springs, but has a supplementary supply from the river for other purposes. Vienna and Munich also obtain their entire supplies from springs, while Budapest, Cologne, Leipzig, Dresden, Frankfurt, many of the great French cities, Brussels, a part of London, and many other English cities derive their supplies from wells or filter-galleries, and among the smaller cities all over Europe ground-water supplies are more numerous than other kinds.
[Pg 277]
APPENDIX X.
LITERATURE OF FILTRATION.
The following is a list of a number of articles on filtration. The list is not complete, but it is believed that it contains the greater part of articles upon slow sand-filtration, and that it will prove serviceable to those who wish to study the subject more in detail.
Anklamm. Glasers Annalen, 1886, p. 48.
A description of the Tegel filters at Berlin, with excellent plans.
Baker. Engineering News.
Water purification in America: a series of descriptions of filters, as follows: Aug. 3, 1893, Lawrence filter and description of apparatus of screening sand and gravel; Apr. 26, 1894, filter at Nantucket, Mass.; June 7, 1894, filters at Ilion, N. Y., plans; June 14, 1894, filters at Hudson, N. Y.; July 12, 1894, filters at Zürich, Switzerland, plans; Aug. 23, 1894, filters at Mt. Vernon, N. Y., plans.
Bertschinger. Journal für Gas- und Wasserversorgung, 1889, p. 1126.
A record of experiments made at Zürich upon the effect of rate of filtration, scraping, and the influence of vaulting. Rate and vaulting were found to be without effect, but poorer results followed scraping. The numbers of bacteria in the lake-water were too low to allow conclusive results.
—— Journal für Gas- und Wasserversorgung, 1891, p. 684.
A farther account of the Zürich results, with full analyses and a criticism of Fränkel and Piefke’s experiments.
Bolton. Pamphlet, 1884.
Descriptions and statistics of London filters.
Böttcher and Ohnesorge. Zeitschrift für Bauwesen, 1876, p. 343.
A description of the Bremen works, with full plans.
Burton. Water-supply of Towns. London, 1894.
Pages 94-115 are upon filtration and mention a novel method of regulating the rate.
Codd. Engineering News, Apr. 26, 1894.
A description of a filter at Nantucket, Mass.
[Pg 278]
Cramer. Centralblatt für Bauwesen, 1886, p. 42.
A description of filters built at Brieg, Germany.
Crook. London Water-supply. London, 1883.
Delbruck. Allgemeine Bauzeitung, 1853, p. 103.
A general article on filtration; particularly valuable for notices of early attempts at filtration and of the use of alum.
Deutsche Verein von Gas- und Wasserfachmänner.
Stenographic reports of the proceedings of this society are printed regularly in the Journal für Gas- und Wasserversorgung, and the discussions of papers are often most interesting.
Drown. Journal Association Eng. Societies, 1890, p. 356.
Filtration of natural waters.
Fischer. Vierteljahresschrift für Gesundheitspflege, 1891, p. 82.
Discussion of papers on water-filtration.
Fränkel. Vierteljahresschrift für Gesundheitspflege, 1891, p. 38.
On filters for city water-works.
Fränkel and Piefke. Zeitschrift für Hygiene, 1891, p. 38, Leistungen der Sandfiltern.
E. Frankland. Report in regard to the London filters for 1893 in the Annual Summary of Births, Deaths, and Causes of Death in London and Other Great Towns, 1893. Published by authority of the Registrar-General.
P. Frankland. Proc. Royal Society, 1885, p. 379.
The removal of micro-organisms from water.
—— Proceedings Inst. Civil Engineers, 1886, lxxxv. p. 197.
Water-purification; its biological and chemical basis.
—— Trans. of Sanitary Institute of Great Britain, 1886.
Filtration of water for town supply.
Frühling. Handbuch der Ingenieurwissenschaften, vol. ii.
Chapter on water-filtration gives general account of filtration, with details of Königsberg filters built by the author and not elsewhere published.
Fuller. Report Mass. State Board of Health, 1892, p. 449.
Report Mass. State Board of Health, 1893, p. 453.
Accounts of the Lawrence experiments upon water-filtration for 1892 and 1893.
—— American Public Health Association, 1893, p. 152.
On the removal of pathogenic bacteria from water by sand filtration.
—— American Public Health Association, 1894, p. 64.
Sand filtration of water with special reference to results obtained at Lawrence, Mass.
[Pg 279]
Gill. Deutsche Bauzeitung, 1881, p. 567.
On American rapid filters. The author shows that they are not to be thought of for Berlin, as they would be more expensive as well as probably less efficient than the usual procedure.
—— Journal für Gas- und Wasserversorgung, 1892, p. 596.
A general account of the extension of the Berlin filters at Müggel. No drawings.
Grahn. Journal für Gas- und Wasserversorgung, 1877, p. 543.
On the filtration of river-waters.
—— Journal für Gas- und Wasserversorgung, 1890, p. 511.
Filters for city water-works.
—— Vierteljahresschrift für Gesundsheitpflege, 1891, p. 76.
Discussion of papers presented on filtration.
—— Journal für Gas- und Wasserversorgung, 1894, p. 185.
A history of the “Rules for Water-filtration” (Appendix I), with some discussion of them.
Grahn and Meyer. Reiseberichte über künstliche central Sandfiltration. Hamburg, 1876.
An account of the observations of the authors in numerous cities, especially in England.
Grenzmer. Centralblatt der Bauverwaltung, 1888, p. 148.
A description of new filters at Amsterdam, with plans.
Gruber. Centralblatt für Bakteriologie, 1893, p. 488.
Salient points in judging of the work of sand-filters.
Halbertsma. Journal für Gas- und Wasserversorgung, 1892, p. 43.
Filter-works in Holland. Gives sand, gravel, and water thickness, with diagrams.
—— Journal für Gas- und Wasserversorgung, 1892, p. 686.
Description of filters built by the author at Leeuwarden, Holland, with plans.
Hart. Proceedings Inst. of Civil Engineers, 1890, c. p. 217.
Description of filters at Shanghai.
Hausen. Journal für Gas- und Wasserversorgung, 1892, p. 332.
An account of experiments made for one year with three 16-inch filters at Helsingfors, Finland, with weekly analyses of effluents.
Hazen. Report of Mass. State Board of Health, 1891, p. 601.
Experiments upon the filtration of water.
—— Report of Mass. State Board of Health, 1892, p. 539.
Physical properties of sands and gravels with reference to their use in filtration. (Appendix III.)
Hunter. Engineering, 1892, vol. 53, p. 621.
Description of author’s sand-washing apparatus.
[Pg 280]
Kirkwood. Filtration of River-waters. New York, 1869.
A report upon European filters for the St. Louis Water Board in 1866. Contains a full account of thirteen filtration-works visited by the author, and of a number of filter-galleries, with a project for filters for St. Louis. This project was never executed, but the report is a wonderful work which appeared a generation before the American public was able to appreciate it. It was translated into German, and the German edition was widely circulated and known.
Koch. Zeitschrift für Hygiene, 1893.
Water-filtration and Cholera: a discussion of the Hamburg epidemic of 1892 in reference to the effect of filtration.
Kröhnke. Journal für Gas- und Wasserversorgung, 1893, p. 513.
An account of experiments made at Hamburg, as a result of which the author recommends the addition of cuprous chloride to the water before filtration to secure greater bacterial efficiency.
Kümmel. Journal für Gas- und Wasserversorgung, 1877, p. 452.
Operation of the Altona filters, with analyses.
—— Vierteljahresschrift für Gesundheitspflege, 1881, p. 92.
The water-works of the city of Altona.
—— Journal für Gas- und Wasserversorgung, 1887, p. 522.
An article opposing the use of rapid filters (David’s process).
—— Journal für Gas- und Wasserversorgung, 1890, p. 531.
A criticism of Fränkel and Piefke’s results, with some statistics of German and English filters. (The English results are taken without credit from Kirkwood.)
—— Vierteljahresschrift für Gesundheitspflege, 1891, p. 87.
Discussion of papers on filtration, with some statistics.
—— Vierteljahresschrift für Gesundheitspflege, 1892, p. 385.
The epidemic of typhoid-fever in Altona in 1891.
—— Journal für Gas- und Wasserversorgung, 1893, p. 161.
Results of experiments upon filtration made at Altona, and bacterial results of the Altona filters in connection with typhoid death-rates.
—— Trans. Am. Society of Civil Engineers, 1893, xxx. p. 330.
Questions of water-filtration.
Leslie. Trans. Inst. Civil Engineers, 1883, lxxiv. p. 110.
A short description of filters at Edinburgh.
Lindley. A report for the commissioners of the Paris Exposition of 1889 upon the purification of river-waters, and published in French or German in a number of journals, among them Journal für Gas- und Wasserversorgung, 1890, p. 501.
[Pg 281]
This is a most satisfactory discussion of the conditions which modern experience has shown to be essential to successful filtration.
Mason. Engineering News, Dec. 7, 1893.
Filters at Stuttgart, Germany, with plans.
Meyer and Samuelson. Deutsche Bauzeitung, 1881, p. 340.
Project for filters for Hamburg, with diagrams. Except in detail, this project is the same as that executed twelve years later.
Meyer. Deutsche Bauzeitung, 1892, p. 519.
Description of the proposed Hamburg filters, with diagrams.
—— The Water-works of Hamburg.
A paper presented to the International Health Congress at Rome, March 1894, and published as a monograph. It contains a full description of the filters as built, with drawings and views in greater detail than the preceding paper.
Mills. Special Report Mass. State Board of Health on the Purification of Sewage and Water, 1890, p. 601.
An account of the Lawrence experiments, 1888-1890.
—— Report Mass. State Board of Health, 1893, p. 543.
The Filter of the Water-supply of the City of Lawrence and its Results.
—— Trans. Am. Society of Civil Engineers, 1893, xxx. p. 350.
Purification of Sewage and Water by Filtration.
Neville. Engineering, 1878, xxvi. p. 324.
A description of the Dublin filters, with plans.
Nichols. Report Mass. State Board of Health, 1878, p. 137.
The filtration of potable water.
Oester. Gesundheits-Ingenieur, 1893, p. 505.
What is the Rate of Filtration? A purely theoretical discussion.
Orange. Trans. Inst. Civil Engineers, 1890, c. p. 268.
Filters at Hong Kong.
Pfeffer. Deutsche Bauzeitung, 1880, p. 399.
A description of filters at Liegnitz, Germany.
Piefke. Results of Natural and Artificial Filtration. Berlin, 1881.
Pamphlet.
—— Journal für Gas- und Wasserversorgung, 1887, p. 595. Die Principien der Reinwassergewinnung vermittelst Filtration.
A sketch of the theory and practical application of filtration.
—— Zeitschrift für Hygiene, 1889, p. 128. Aphorismen über Wasserversorgung.
A discussion of the theory of filtration, with a number of experiments on the thickness of sand-layers, etc.
[Pg 282]
Piefke. Vierteljahresschrift für Gesundheitspflege, 1891, p. 59.
On filters for city water-works.
Fränkel and Piefke. Zeitschrift für Hygiene, 1891, p. 38.
Leistungen der Sandfiltern. An account of the partial obstruction of the Stralau filters by ice, and a typhoid epidemic which followed. Experiments were then made upon the passage of cholera and typhoid germs through small filters.
Piefke. Journal für Gas- und Wasserversorgung, 1891, p. 208. Neue Ermittelungen über Sandfiltration.
The above mentioned experiments being objected to on certain grounds, they were repeated by Piefke alone, confirming the previous observations on the passage of bacteria through filters, but under other conditions.
—— Zeitschrift für Hygiene, 1894, p. 151, Über Betriebsführung von Sandfiltern.
A full account of the operation of the Stralau filters in 1893, with discussion of the efficiency of filtration, etc.
Plagge and Proskauer. Zeitschrift für Hygiene, 11. p. 403.
Examination of water before and after filtration at Berlin, with theory of filtration.
Reincke. Bericht über die Medicinische Statistik des Hamburgischen Staates für 1892.
Contains a most valuable discussion of the relations of filtration to cholera, typhoid fever, and diarrhœa, with numerous tables and charts. (Abstract in Appendix II.)
Reinsch. Centralblatt für Bakteriologie, 1895, p. 881.
An account of the operation of the Altona filters. High numbers of bacteria in the effluents have often resulted from the discharge of sludge from the sedimentation-basins onto the filters, due to the interference of ice on the action of the floating outlet for the basins, and this, rather than the direct effect of cold, is believed to be the direct cause of the low winter efficiency. The author urges the necessity of a deeper sand-layers in no case less than 18 inches thick.
Renk. Gesundheits-Ingenieur, 1886, p. 54.
—— Über die Ziele der künstliche Wasserfiltration.
Ruhlmann. Wochenblatt für Baukunde, 1887, p. 409.
A description of filters at Zürich.
Salbach. Glaser’s Annalen, 1882.
Filters at Groningen, Holland, built in 1880. Alum used.
Samuelson. Translation of Kirkwood’s “Filtration of River-waters” into German, with additional notes especially on the theory of filtration and the sand to be employed. Hamburg, 1876.
[Pg 283]
Samuelson. Filtration and constant water-supply. Pamphlet. Hamburg, 1882.
—— Journal f. Gas- und Wasserversorgung, 1892, p. 660.
A discussion of the best materials and arrangement for sand-filters.
Schmetzen. Deutsche Bauzeitung, 1878, p. 314.
Notice and extended criticism of Samuelson’s translation of Kirkwood.
Sedden. Jour. Asso. Eng. Soc., 1889, p. 477.
In regard to the sedimentation of river-waters.
Sedgwick. New England Water-works Association, 1892, p. 103.
European methods of Filtration with Reference to American Needs.
Sokal. Wochenschrift der östreichen Ingenieur-Verein, 1890, p. 386.
A short description of the filters at St. Petersburg, and a comparison with those at Warsaw.
Sturmhöfel. Zeitschrift f. Bauwesen, 1880, p. 34.
A description of the Magdeburg filters, with plans.
Tomlinson. American Water-works Association, 1888.
A paper on filters at Bombay and elsewhere.
Turner. Proc. Inst. Civil Engineers, 1890, c. p. 285.
Filters at Yokohama.
Van der Tak. Tijdschrift van de Maatschapping van Bouwkunde, 1875(?).
A description (in Dutch) of the Rotterdam water-works, including the wooden drains which caused the trouble with crenothrix, and which have since been removed. Diagrams.
Van Ijsselsteyn. Tijdschrift van het Koninklijk Instituut van Ingenieurs, 1892-5, p. 173.
A description of the new Rotterdam filters, with full drawings.
Veitmeyer. Verhandlungen d. polyt. Gesell. zu Berlin, April, 1880.
Filtration and purification of water.
Wolffhügel. Arbeiten aus dem Kaiserliche Gesundheitsamt, 1886, p. 1.
Examinations of Berlin water for 1884-5, with remarks showing superior bacterial efficiency with open filters.
—— Journal für Gas- u. Wasserversorgung, 1890, p. 516.
On the bacterial efficiency of the Berlin filters, with diagrams.
Zobel. Zeitschrift des Vereins deutsche Ingenieure, 1884, p. 537.
Description of filters at Stuttgart.
[Pg 284]
OTHER LITERATURE.
Many scientific and engineering journals publish from time to time short articles or notices on filtration which are not included in the above list. Among such journals none gives more attention to filtration than the Journal für Gasbeleuchtung und Wasserversorgung, which publishes regularly reports upon the operation of many German filters, and gives short notices of new construction. The first articles upon filtration in this journal were a series of descriptions of German water-works in 1870-73, including descriptions of filters at Altona, Brunswick, Lübeck, etc. Stenographic reports of many scientific meetings have been published, particularly since 1890, and since 1892 there has been much discussion in regard to the “Rules for Filtration” given in Appendix I.
A Report of a Royal Commission to inquire into the water-supply of the metropolis, with minutes of evidence, appendices, and maps (London, 1893-4), contains much valuable material in regard to filtration.
The monthly reports of the water examiner, and other papers published by the Local Government Board, London, are often of interest.
The German “Verein von Gas- u. Wasserfachmänner” prints without publishing a most useful annual summary of German water-works statistics for distribution to members. Many of the statistics given in this volume are from this source.
Description of the filters at Worms was given in the Deutsche Bauzeitung, 1892, p. 508; of the filters at Liverpool in Engineering, 1889, p. 152, and 1892, p. 739. The latter journal also has given a number of descriptions of filters built in various parts of the world by English engineers, but, excepting the articles mentioned in the above list, the descriptions are not given in detail.
[Pg 285]
MORE RECENT ARTICLES.
The following are a few of the more important articles which have appeared since the first edition of this book. In addition many articles of current interest have appeared in the technical journals, particularly in the journals mentioned above.
Clark. Reports of Mass. State Board of Health, 1894 to 1897, inclusive.
Articles on the filtration of water, giving accounts of experiments at the Lawrence Experiment Station, and records of the operation of the Lawrence city filter. These experiments are directed principally to the removal of bacteria from sewage-polluted waters.
—— Jour. New England Water Works Assoc., XI., p. 277.
Removal of Iron from Ground Waters. A description of certain experiments.
Fowler. Jour. New England Water Works Assoc., XII., p. 209.
The Operation of a Slow Sand Filter. A most helpful and thorough description of the operation of sand filters at Poughkeepsie for a long period of years.
Fuller. Water Purification at Louisville. D. Van Nostrand Co., 1898.
A report upon a series of most exhaustive experiments carried out at Louisville, directed principally to the clarification of excessively muddy waters. Contains a full account of methods of coagulation, and of experiments with the electrical treatment of water.
—— Report on Water Filtration at Cincinnati. City document, 1899.
Account of experiments with sand filters, with and without coagulants, and with other processes applied to the Ohio River water at Cincinnati.
Gill. Filters at Muggel. Proc. Institute of Civil Engineers, 1894-5; vol. 119, p. 236.
A description of the new vaulted filter plant designed by the author for Berlin, Germany. Plans and views.
Goetze. Journal für Gasbeleuchtung und Wasserversorgung, 1897, p. 169.
Selbstthätige Wasseraustrittsregler besonders für Filter. A description of the automatic regulating device for filters used at Bremen.
—— Zeitschrift des Vereines deutscher Ingenieure, XXX.
Reinigung des Trinkwassers in Bremen durch mehrmalige Sandfiltration. A description of the method of double filtration used at Bremen, giving results obtained in full. No drawings.
[Pg 286]
Grahn. Journal für Gasbeleuchtung und Wasserversorgung, 1895.
Water purification plant at the city of Magdeburg. A description of the old plant, and the changes which have been made in it to increase its capacity, and make it conform to the requirements of the German official instructions regarding filtration. Many illustrations and plans.
Halbertsma. Journal für Gasbeleuchtung und Wasserversorgung, 1896.
Die Resultate der doppelten Filtration zu Schiedam. A description of double filtration at Schiedam, with the bacterial results for the two years, 1894 and 1895, showing an average bacterial efficiency of 99.76 per cent.
Hazen. Report to Filtration Commission, Pittsburgh. City document, 1899.
A description of experiments upon the treatment of the Allegheny River water by sand and mechanical filters.
—— Ohio State Board of Health Report, 1897, p. 154.
Report on the Mechanical Filtration of the Public Water Supply of Lorain. Gives the results of a five-weeks test of the Jewell mechanical filters at Lorain, treating Lake Erie water.
Kemna. The Biology of Sand Filtration. Read before the annual convention of the British Association of Water Works Engineers. Abstract in Engineering News, XLI., p. 419.
Describing organisms which develop in open sand filters, both animal and vegetable, and their effects upon the process. A quite full account of the author’s extended experience, and the only paper treating this subject.
Magar. Journal für Gasbeleuchtung und Wasserversorgung, 1897, p. 4.
Reinigungsbetrieb der offener Sandfilter des Hamburger Filterwerkes in Frostzeiten. A new method of cleaning open filters in winter without the removal of the ice.
Panwitz. Arbeiten aus dem Kaiserlichen Gesundheitsamte, XIV., p. 153.
Die Filtration von Oberflächenwasser in den deutschen Wasserwerken während der Jahre 1894 bis 1896.
A description of the filtration works in Germany, and the results obtained from them, particularly from the point of view of bacterial efficiency. Results are graphically shown by a series of charts.
Reynard. Le Génie Civil, 1896, XXVIII., p. 321.
Purification of water with the aid of metallic iron. Describing the works of the Compagnie Général des Eaux for supplying the suburbs of Paris with filtered water, the capacity of the works being over 23,000,000 gallons daily.
[Pg 287]
Weston. Rhode Island State Board of Health, 1894.
Report of the Results Obtained with Experimental Filters at the Pattaconset Pumping Station of the Providence Water Works. Relates particularly to the bacterial purification obtained with rapid filtration aided by sulphate of alumina. These were the first systematic experiments made with mechanical filters.
Wheeler. Journal of the New England Water Works Assoc., XI., p. 301. Covered Sand Filter at Ashland, Wis.
A description of the covered filters built by the author at Ashland Wis. for the purification of the bay water. Views and drawings.
[Pg 288]
APPENDIX XI.
THE ALBANY WATER-FILTRATION PLANT.
(Abridged from Proceedings American Society of Civil Engineers, Nov.
1899.)
Albany, N. Y., was originally supplied with water by gravity from certain reservoirs on small streams west and north of the city. In time, with increasing consumption, the supply obtained from these sources became inadequate, and an additional supply from the Hudson River was introduced. The water was obtained from the river through a tunnel under the Erie Basin, and a pumping-station was erected in Quackenbush Street to pump it to reservoirs, one of which served also as the distributing point for one of the gravity supplies. The intake, which was used first in 1873, drew water from the river opposite the heart of the city. In recent years, the amount of water drawn from this source has greatly exceeded that obtained from the gravity sources.
The Hudson River, at the point of intake, has a drainage area of 8240 square miles. Of this, 4541 square miles are tributary to the Hudson above Troy, 3493 are tributary to the Mohawk, and 168 are tributary to the Hudson below the Mohawk.
The minimum flow may be estimated at 1657 cubic feet per second, or 1,060,000,000 gallons per 24 hours, or at least fifty times the maximum consumption.
The cities and larger towns upon the river above the intake, with estimated populations and distances, are as follows:
[Pg 289]
| MOST IMPORTANT CITIES, TOWNS, AND VILLAGES ON THE WATERSHED OF THE HUDSON ABOVE ALBANY. | |||||
|---|---|---|---|---|---|
| Place. | County. | Approximate Distance above Intake, Miles. |
Population in | ||
| 1880. | 1890. | 1900. (Estimated.) |
|||
| Troy | Rensselaer | 4 | 56,747 | 60,956 | 65,470 |
| Watervliet | Albany | 4 | 8,820 | 12,967 | 19,040 |
| Green Island | Rensselaer | 5 | 4,160 | 4,463 | 4,788 |
| Cohoes | Albany | 8 | 19,416 | 22,509 | 26,450 |
| Lansingburg | Rensselaer | 8 | 7,432 | 10,550 | 14,980 |
| Waterford | Saratoga | 9 | (1,822) | 1,822 | (1,822) |
| Schenectady | Schenectady | 28 | 13,655 | 19,002 | 26,450 |
| Hoosic Falls | Rensselaer | 44 | 4,530 | 7,014 | 10,860 |
| Amsterdam | Montgomery | 44 | 9,466 | 17,336 | 31,730 |
| Glens Falls | Warren | 49 | 4,900 | 9,509 | 18,450 |
| Saratoga Springs | Saratoga | 51 | 8,421 | 11,975 | 17,010 |
| Johnstown | Fulton | 56 | 5,013 | 7,768 | 12,040 |
| Gloversville | Fulton | 58 | 7,133 | 13,864 | 26,930 |
| North Adams, Mass. | Berkshire | 68 | 10,191 | 16,074 | 25,340 |
| Adams, Mass. | Berkshire | 75 | 5,591 | 9,213 | 15,181 |
| Little Falls | Herkimer | 82 | 6,910 | 8,783 | 11,160 |
| Utica | Oneida | 107 | 33,914 | 44,007 | 57,090 |
| Rome | Oneida | 127 | 12,194 | 14,991 | 18,430 |
| 32 villages | 52,523 | 61,869 | 76,194 | ||
| Total, not including rural population | 272,838 | 354,672 | 479,415 | ||
| Per square mile | 33 | 43 | 59 | ||
Without entering into a detailed discussion, it may be said that the amount of sewage, with reference to the size of the river and the volume of flow, is a fraction less than that at Lawrence, Mass., where a filter-plant has also been constructed, but the pollution is much greater than that of most American rivers from which municipal water-supplies are taken.
The filtration-plant completed in 1899 takes the water from a point about two miles above the old intake. Pumps lift the water to the sedimentation-basin, from which it flows to the filters and thence through a conduit to the pumping-station previously used.
DESCRIPTION OF PLANT.
Intake.—The intake consists of a simple concrete structure in the form of a box, having an open top covered with rails 6 inches apart, and connected below, through a 36-inch pipe, with[Pg 291] a well in the pumping-station. Before going to the pumps the water passes through a screen with bars 2 inches apart, so arranged as to be raked readily. The rails over the intake and this screen are intended to stop matters which might obstruct the passageways of the pumps, but no attempt is made to stop fish, leaves, or other floating matters which may be in the water. The arrangement, in this respect, is like that of the filter at Lawrence, Mass., where the raw water is not subjected to close screening. There is room, however, to place finer screens in the pump-well, should they be found desirable.
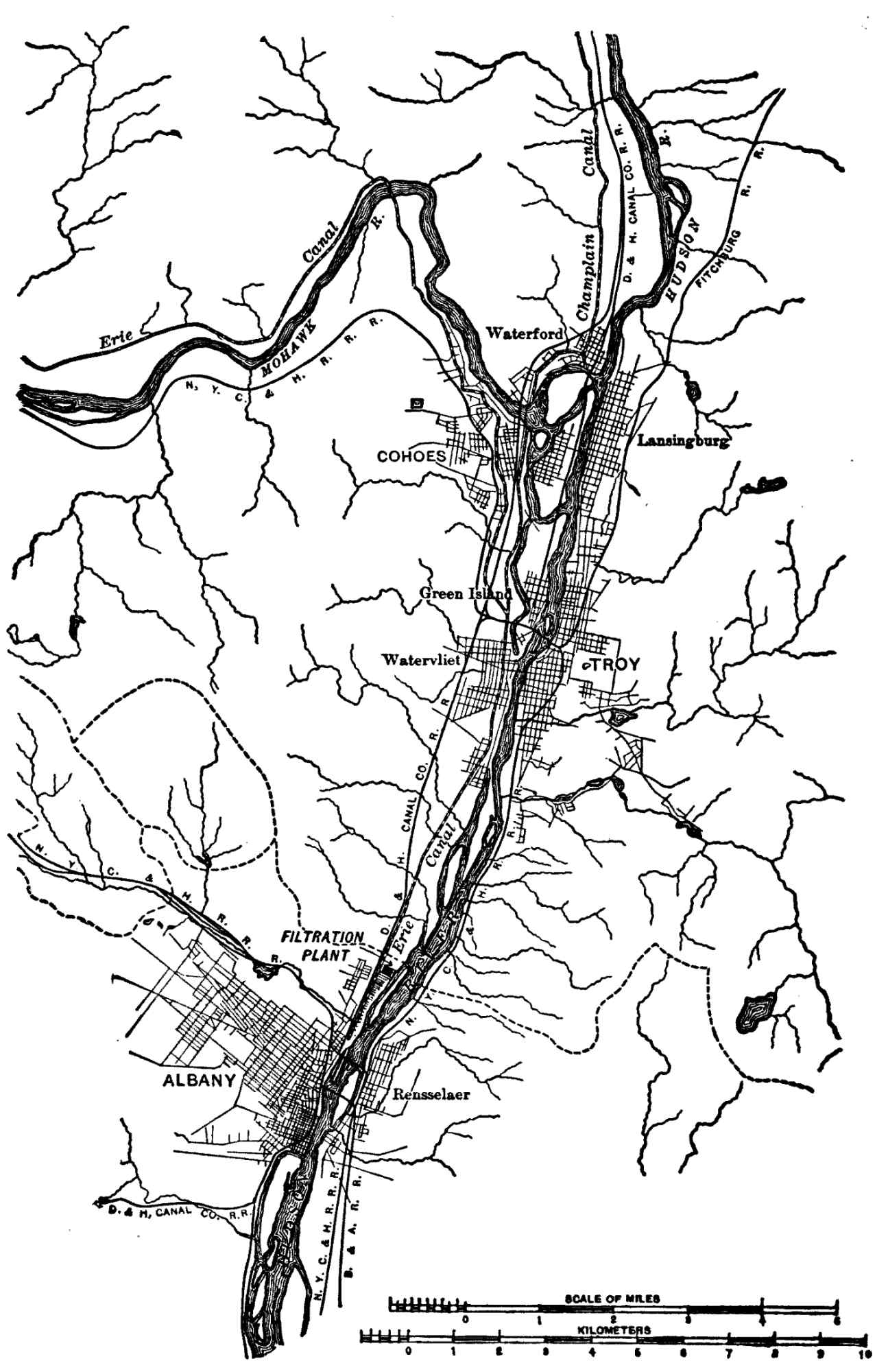
HUDSON RIVER
NEAR INTAKE
Fig. 1.

Sedimentation-basin, Pumping-station, and Outlets.

Sedimentation-basin, an Outlet, and Laboratory.
[To face page 290.]
Pumping-station.—The centrifugal pumps have a guaranteed capacity of 16,000,000 gallons per 24 hours against a lift of 18 feet, or 12,000,000 gallons per 24 hours against a lift of 24 feet. The ordinary pumping at low water is against the higher lift, and under these conditions either pump can supply the ordinary consumption, the other pump being held in reserve.
The pumping-station building, to a point above the highest flood-level, is of massive concrete construction, without openings. Nearly all the machinery is necessarily below this level, and in high water the sluice-gates are closed, and the machinery is thus protected from flooding. The superstructure is of pressed brick, with granite trimmings.
Meter for Raw Water.—Upon leaving the pumping-station the water passes through a 36-inch Venturi meter having a throat diameter of 17 inches, the throat area being two ninths of the area of the pipe. The meter records the quantity of water pumped, and is also arranged to show on gauges in the pumping-station the rate of pumping.
Aeration.—After leaving the meter, the water passes to the sedimentation-basin through eleven outlets. These outlets consist of 12-inch pipes on end, the tops of which are 4 feet above the nominal flow-line of the sedimentation-basin. Each of these outlet-pipes is pierced with 296 3⁄8-inch holes extending from 0.5 to 3.5 feet below the top of the pipe. These holes are computed[Pg 293] so that when 11,000,000 gallons of water per day are pumped all the water will pass through the holes, the water in the pipes standing flush with the tops. The water is thus thrown out in 3256 small streams, and becomes aerated. When more than the above amount is pumped, the excess flows over the tops of the outlet-pipes in thin sheets, which are broken by the jets.
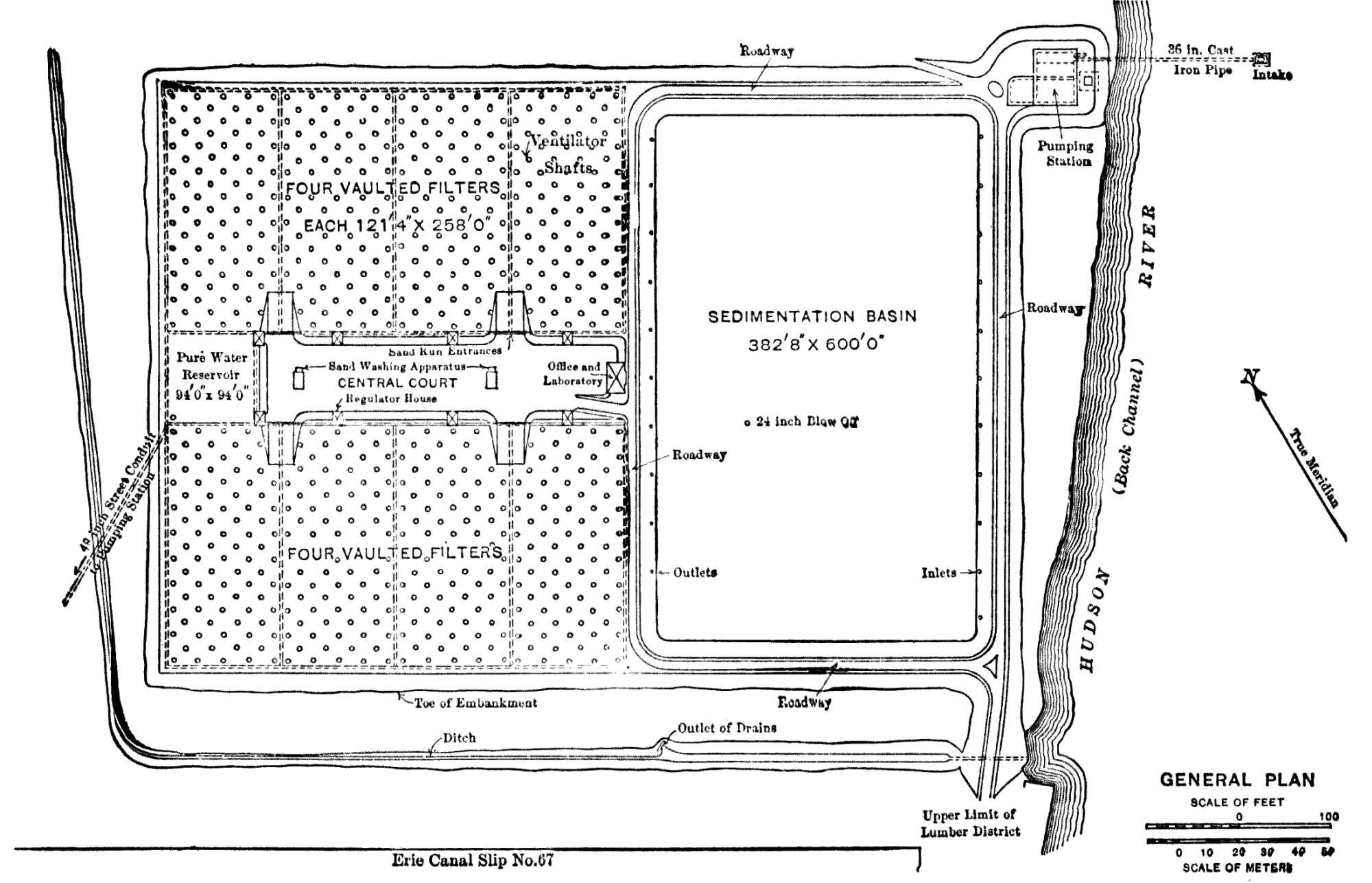
Fig. 2.
Regarding the necessity for aeration, no observations have been taken upon the Hudson River, but, judging from experience with the Merrimac at Lawrence, where the conditions are in many respects similar, the water is at all times more or less aerated, and, for the greater part of the year, it is nearly saturated with oxygen, and aeration is not necessary. During low water in summer, however, there is much less oxygen in the water, and at these times aeration is a distinct advantage. Further, the river-water will often have a slight odor, and aeration will tend to remove it. The outlets are arranged so that they can be removed readily in winter if they are not found necessary at that season.
Sedimentation-basin.—The sedimentation-basin has an area of 5 acres and is 9 feet deep. To the overflow it has a capacity of 14,600,000 gallons, and to the flow-line of the filters 8,900,000 gallons. There is thus a reserve capacity of 5,700,000 gallons between these limits, and this amount can be drawn upon, without inconvenience, for maintaining the filters in service while the pumps are shut down. This allows a freedom in the operation of the pumps which would not exist with the water supplied direct to the filters.
The water enters the sedimentation-basin from eleven inlets along one side, and is withdrawn from eleven outlets directly opposite. The inlets and aerating devices described previously bring the water into the basin without current and evenly distributed along one side. Both inlets and outlets are controlled by gates, so that any irregularities in distribution can be avoided. The concrete floor of the sedimentation-basin is built with even slopes from the toe of each embankment to a sump, the heights of[Pg 295] these slopes being 1 foot, whatever their lengths. The sump is connected with a 24-inch pipe leading to a large manhole in which there is a gate through which water can be drawn to empty the basin. There is an overflow from the basin to this manhole which makes it impossible to fill the basin above the intended level.
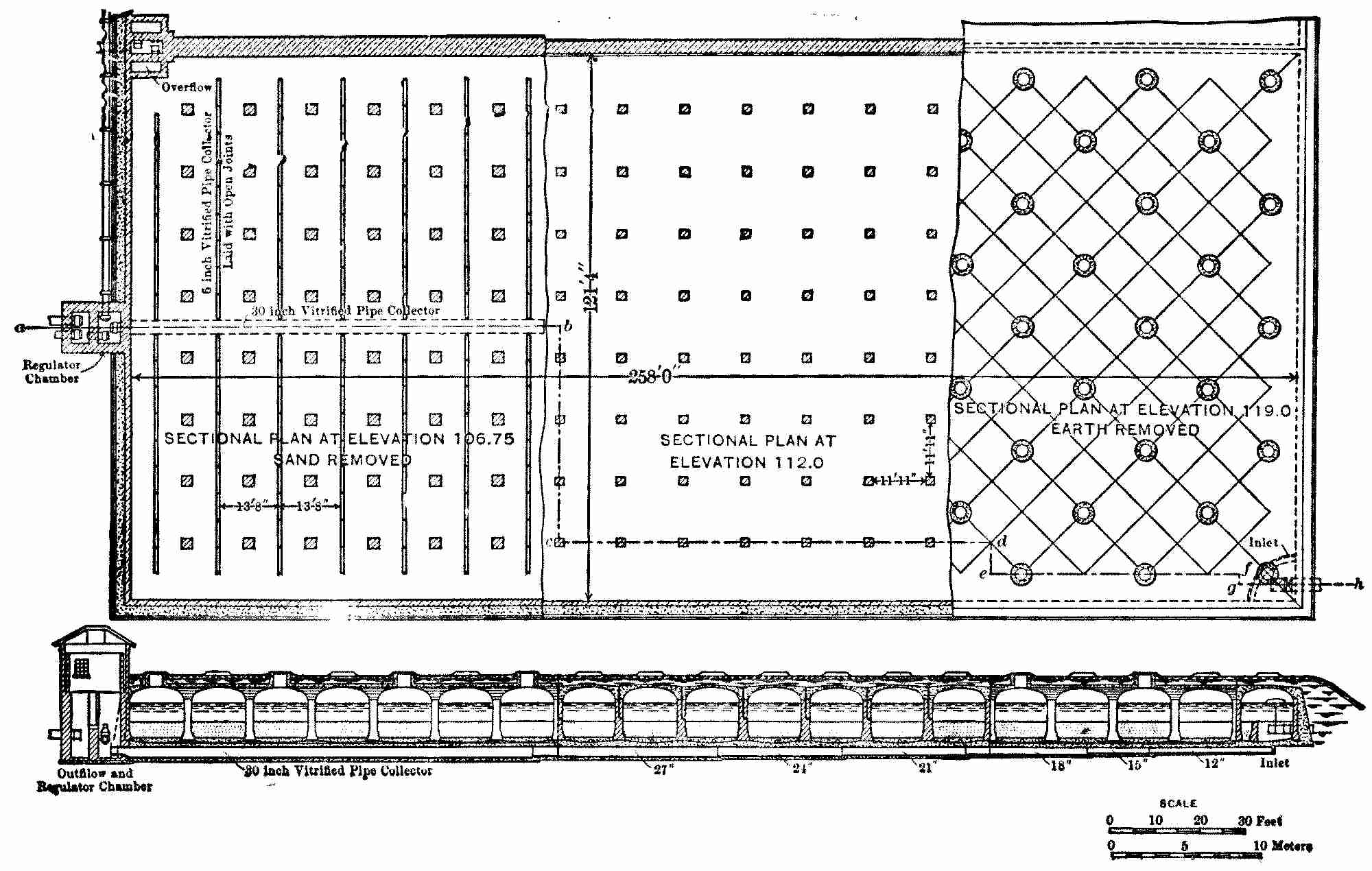
LONGITUDINAL SECTION ON a-b-c-d-e-f-g-h
FILTER BEDS
PLAN AND SECTION OF FILTER NO. 2
Fig. 3.

Outside Wall, ready for Concrete Backing.

Sedimentation-basin: showing Construction of Floor.
[To face page 294.]
Filters.—The filters are of masonry, and are covered to protect them against the winters, which are quite severe in Albany. The piers, cross-walls, and linings of the outside walls, entrances, etc., are of vitrified brick. All other masonry is concrete. The average depth of excavation for the filters was 4 feet, and the material at the bottom was usually blue or yellow clay. In some places shale was encountered. In one place soft clay was found, and there the foundations were made deeper. The floors consisted of inverted, groined, concrete arches, arranged to distribute the weight of the walls and vaulting over the whole area of the bottom.
The groined arch-vaulting is of concrete with a clear span of 11 feet 11 inches, a rise of 21⁄2 feet, and a thickness of 6 inches at the crown. It was put in in squares, the joints being on the crowns of the arches parallel with the lines of the piers, and each pier being the centre of one square. The manholes are in alternate sections, and are of concrete, built in steel forms with castings at the tops, securely jointed to the concrete.
Above the vaulting there are 2 feet of earth and soil, grassed on top. The tops of the manholes are 6 inches above the soil to prevent rain-water from entering them. The drainage of the soil is effected by a depression of the vaulting over each pier, partially filled with gravel and sand, from which water is removed by a 2-inch tile-drain going down the centre of the pier and discharging through its side just above the top of the sand in the filter.
In order to provide ready access to each filter, a part of the vaulting near one side is elevated and made cylindrical in shape, making an inclined runway from the sand-level to a door the threshold of which is 6 inches above the level of the overflow.
[Pg 296]
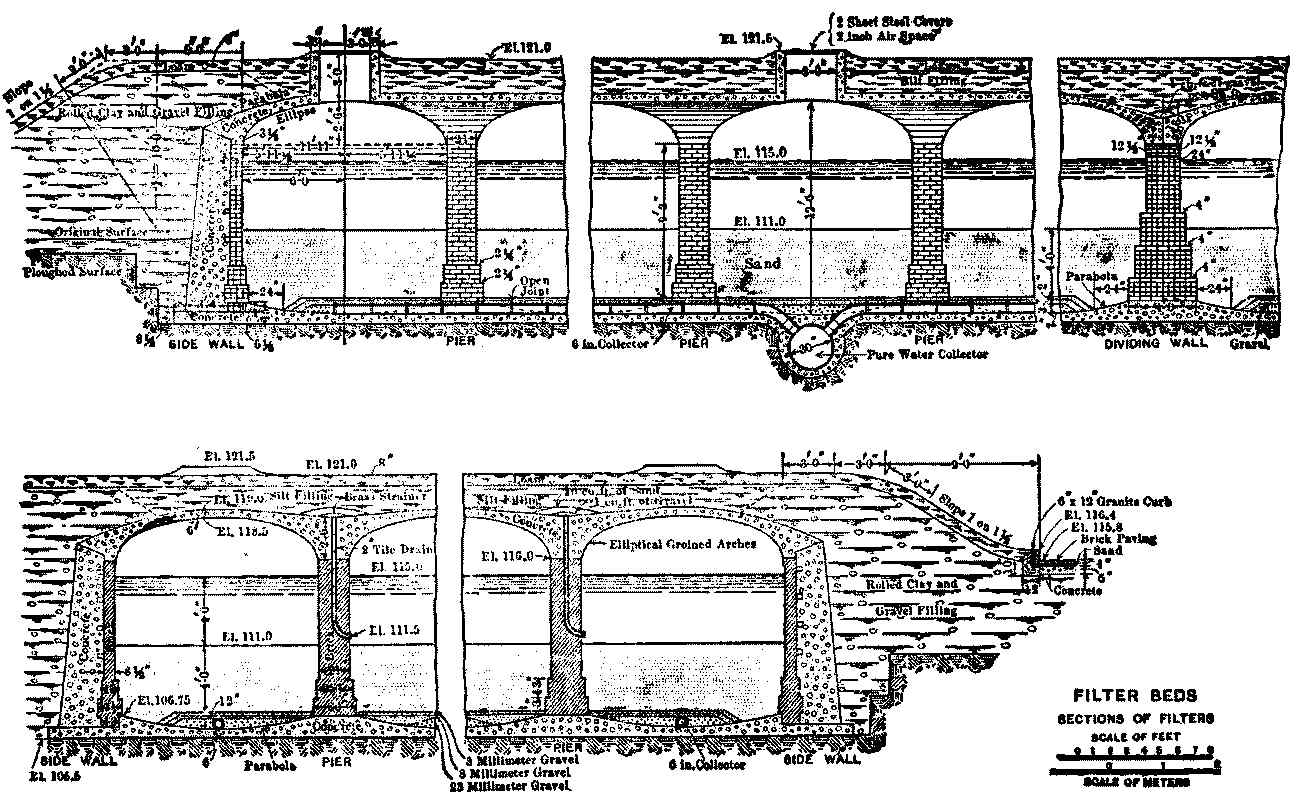
Fig. 4.
[Pg 297]
This sand-run is provided with permanent timber runways and with secure doors.
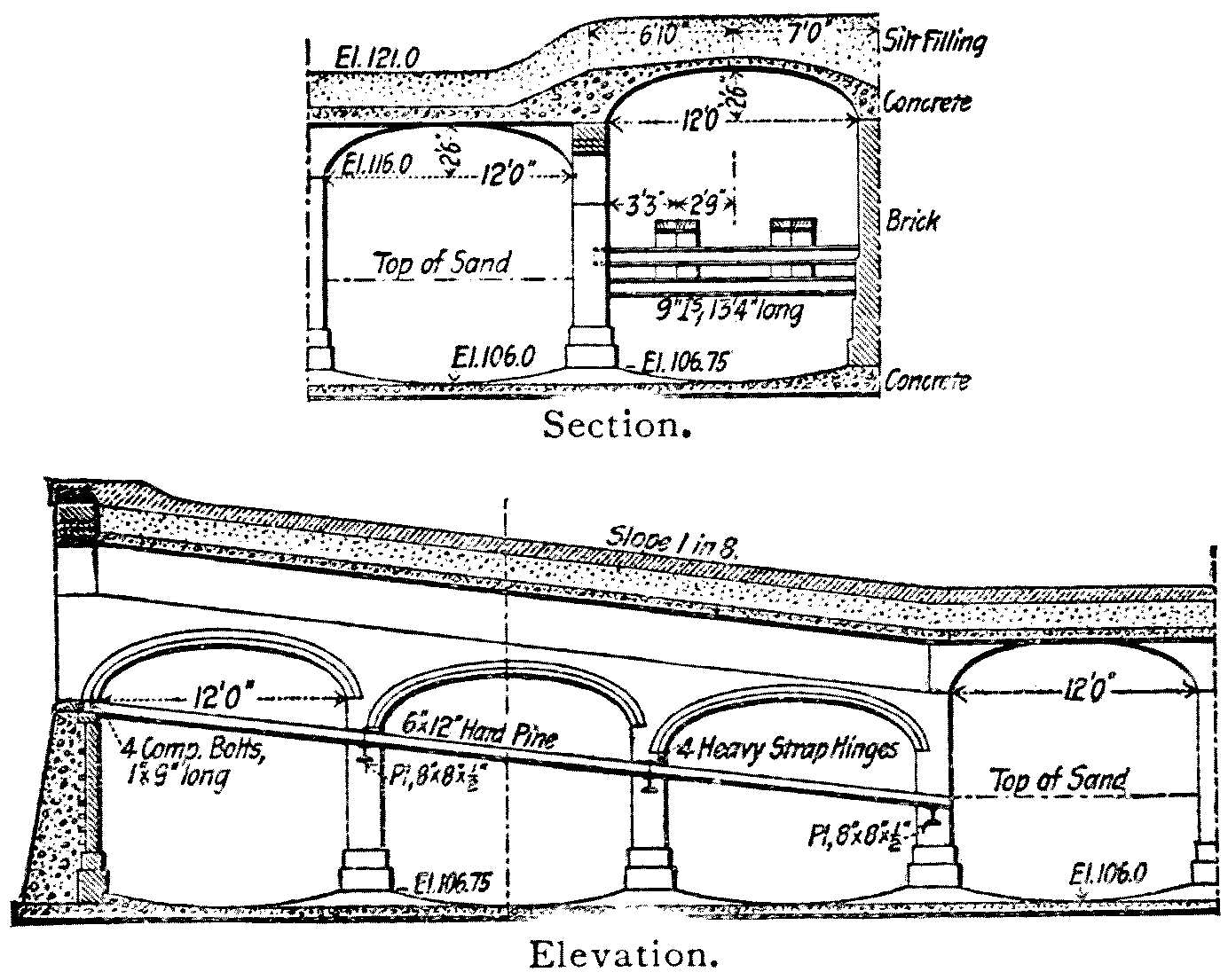
Fig. 5.—Entrance to a Filter.
The manholes of the filters are provided with double covers of steel plates to exclude the cold. The covers also exclude light. When cleaning the filters, light can be admitted by removing the covers. Supports for electric lights are placed in the vaulting, so that the filters can be lighted by electricity and the work of cleaning can be done at night, and in winter under heavy snow, without removing the covers. The electric lights have not yet been installed.
The regulator-houses, the entrances to the sand-runs, and all exposed work are of pressed brick with Milford granite trimmings and slate roofs. The regulator-houses have double walls and double windows and a tight ceiling in the roof, to make them as warm as possible and to avoid the necessity of artificial heat to prevent freezing.
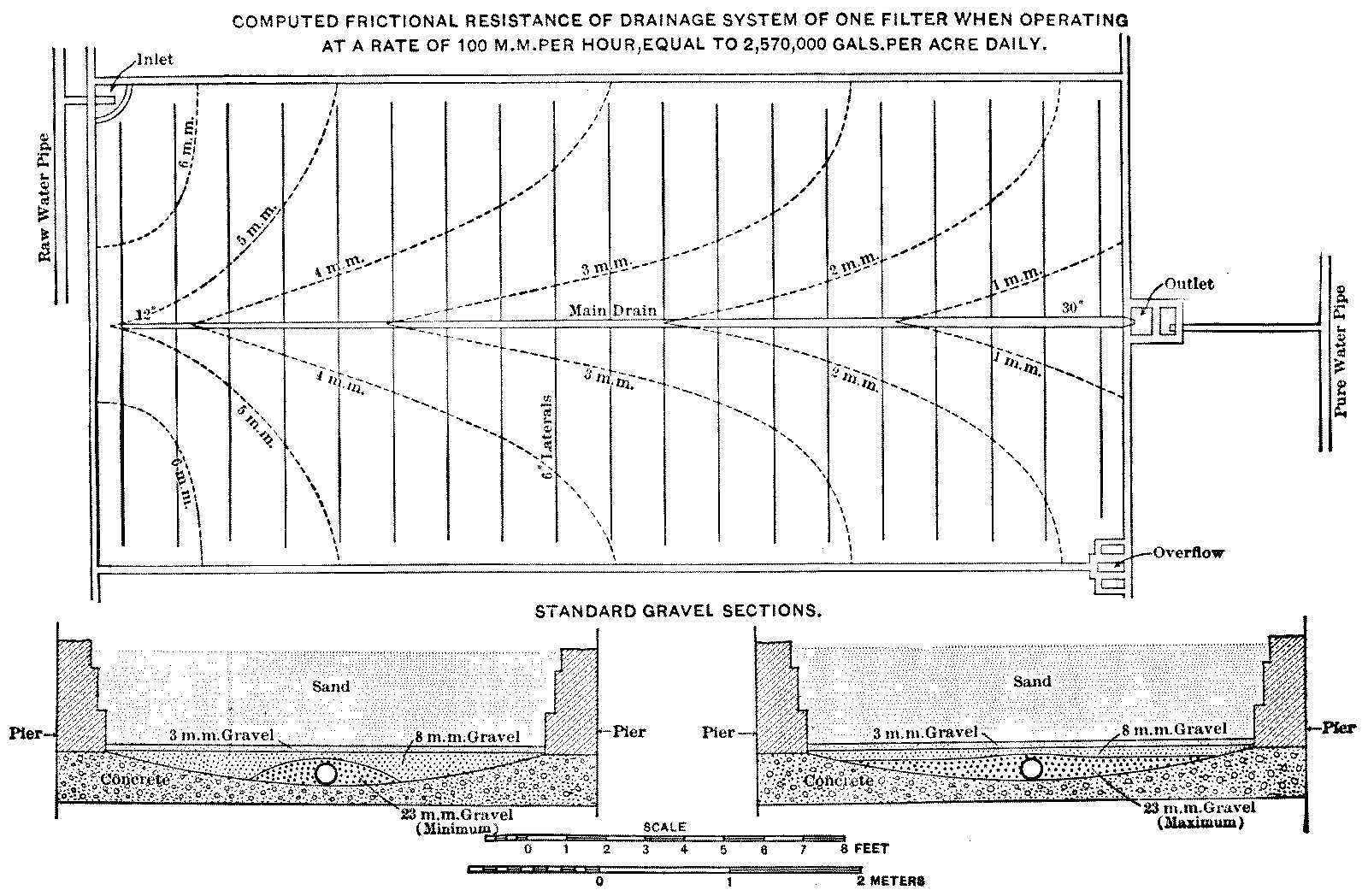
Fig. 6.

Placing the Floor of a Filter.

Building the Brick Piers.
[To face page 298.]
The main underdrains for removing the filtered water are of[Pg 299] vitrified pipe surrounded by concrete and are entirely below the floors of the filters.
Connections with the main drain are made through thirty-eight 6-inch outlets in each filter, passing through the floor and connected with 6-inch lateral drains running through the whole width of the filter. These drains were made with pipes having one side of the bell cut off so that they would lie flat on the floor and make concentric joints, without support and without having to be wedged. They were laid with a space of about 1 inch between the barrels, leaving a large opening for the admission of water from the gravel.
The underdrainage system is so designed that, when starting a filter after cleaning, the friction of the sand is about 50 mm. at a rate of 3,000,000 gallons per acre daily, and the friction of the underdrainage system is estimated at 10 mm. This very low friction, which is necessary, is obtained by the use of ample sizes for the underdrains and low velocities in them. In the outlet and measuring devices moderate losses of head are not objectionable, and the sizes of the pipes and connections are, therefore, smaller than the main underdrains.
The gravel surrounding the underdrains is of three grades. The material was obtained from the river-bed by dredging, and was of the same stock as that used for preparing ballast for the concrete. It was separated and cleaned by a special, cylindrical, revolving screen. The coarsest grade of gravel was that which would not pass round holes 1 inch in diameter, and free from stones more than about 2 inches in diameter. At first it was required to pass a screen with holes 2 inches in diameter, but this screen removed many stones which it was desired to retain, and the screen was afterward changed to have holes 3 inches in diameter. The intermediate grades of gravel passed the 1-inch holes, and were retained by a screen with round holes 33⁄8 inch in diameter. The finest gravel passed the above screens and was retained by a screen with round holes 3⁄16 inch in diameter. The gravel was washed, until free from sand and dirt, by water played upon it during the[Pg 301] process of screening, and it was afterward taken over screens in the chutes, where it was separated from the dirty water, and, when necessary, further quantities of water were played upon it at these points.
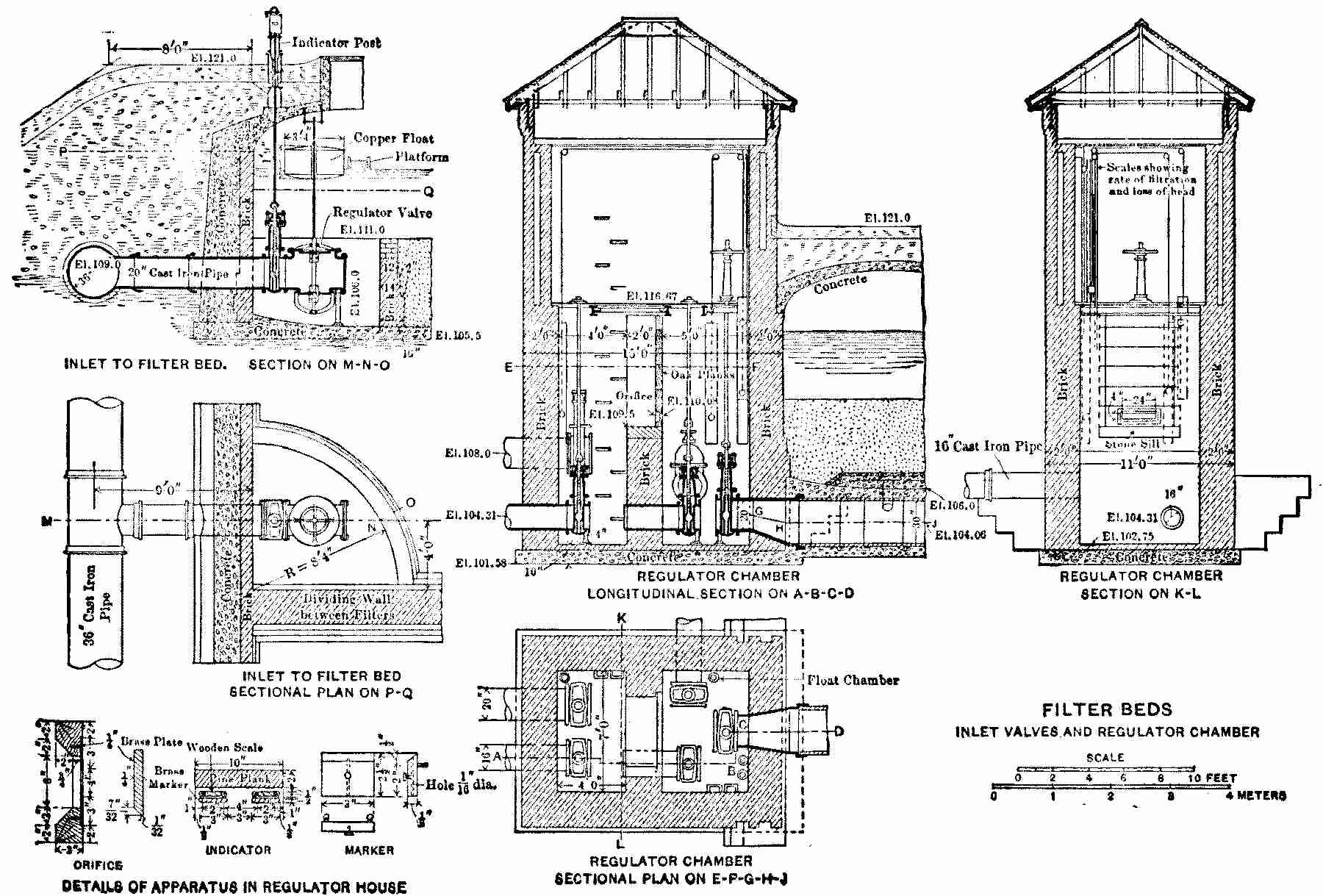
Fig. 7.
The average mechanical analyses of the three grades of gravel are shown by Fig. 8. Their effective sizes were 23, 8, and 3 mm. respectively, and for convenience they are designated by these numbers. The average uniformity coefficient for each grade was about 1.8.
The 23-mm. gravel entirely surrounded the 6-inch pipe-drains, and was carried slightly above their tops. In some cases it was used to cover nearly the whole of the floor, but this was not insisted upon.
The 8-mm. gravel was obtained in larger quantity than the other sizes, and was used to fill all spaces up to a plane 21⁄2 inches below the finished surface of the gravel, this layer being about 2 inches thick over the tops of the drains, and somewhat thicker elsewhere.
The 3-mm. gravel was then applied in a layer 21⁄2 inches deep, and the surface levelled.
The preliminary estimates of cost were based upon the use of filter-sand from a bank near the filter-site. Further examination showed that this sand contained a considerable quantity of lime, and it was found by experiment with a small filter constructed for that purpose that the use of this sand would harden the water by about 2 parts in 100,000, and the amount of lime contained in the sand, namely, about 7 per cent, was sufficient to continue this hardening action for a considerable number of years. This was regarded as a serious objection to its use, and the specifications were drawn limiting the amount of lime in the sand. This excluded all of the local bank sands. The river-sands which were used were nearly free from lime, and in the end the sand as secured was probably not only free from lime, but more satisfactory[Pg 302] in other ways, and also cheaper than the bank-sand would have been.
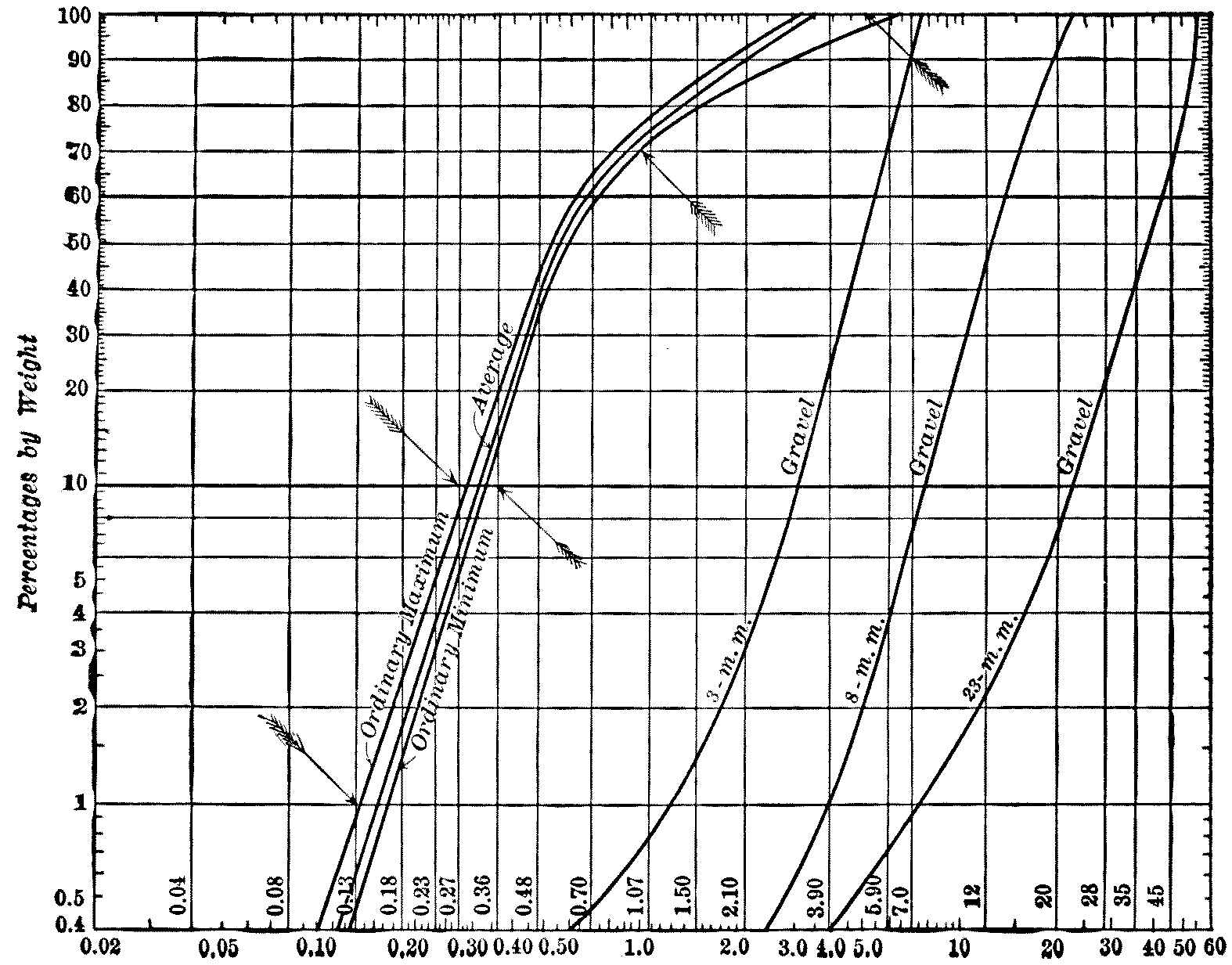
Diameters in Millimeters
MECHANICAL COMPOSITION OF FILTER SAND AND GRAVELS.
(ARROWS SHOW REQUIREMENT OF SPECIFICATION)
Fig. 8.
The specifications of the filter-sand require that “The filter-sand shall be clean river-, beach-, or bank-sand, with either sharp or rounded grains. It shall be entirely free from clay, dust, or organic impurities, and shall, if necessary, be washed to remove such materials from it. The grains shall, all of them, be of hard material which will not disintegrate, and shall be of the following diameters: Not more than 1 per cent, by weight, less than 0.13 mm., nor more than 10 per cent less than 0.27 mm.; at least 10 per cent, by weight, shall be less than 0.36 mm., and at least 70 per cent, by weight, shall be less than 1 mm., and no particles shall be more than 5 mm. in diameter. The diameters of the sand-grains will be computed as the diameters of spheres of[Pg 304] equal volume. The sand shall not contain more than 2 per cent, by weight, of lime and magnesia taken together and calculated as carbonates.”

Placing the Concrete Vaulting.

General View of Vaulting, under Construction.
[To face page 302.
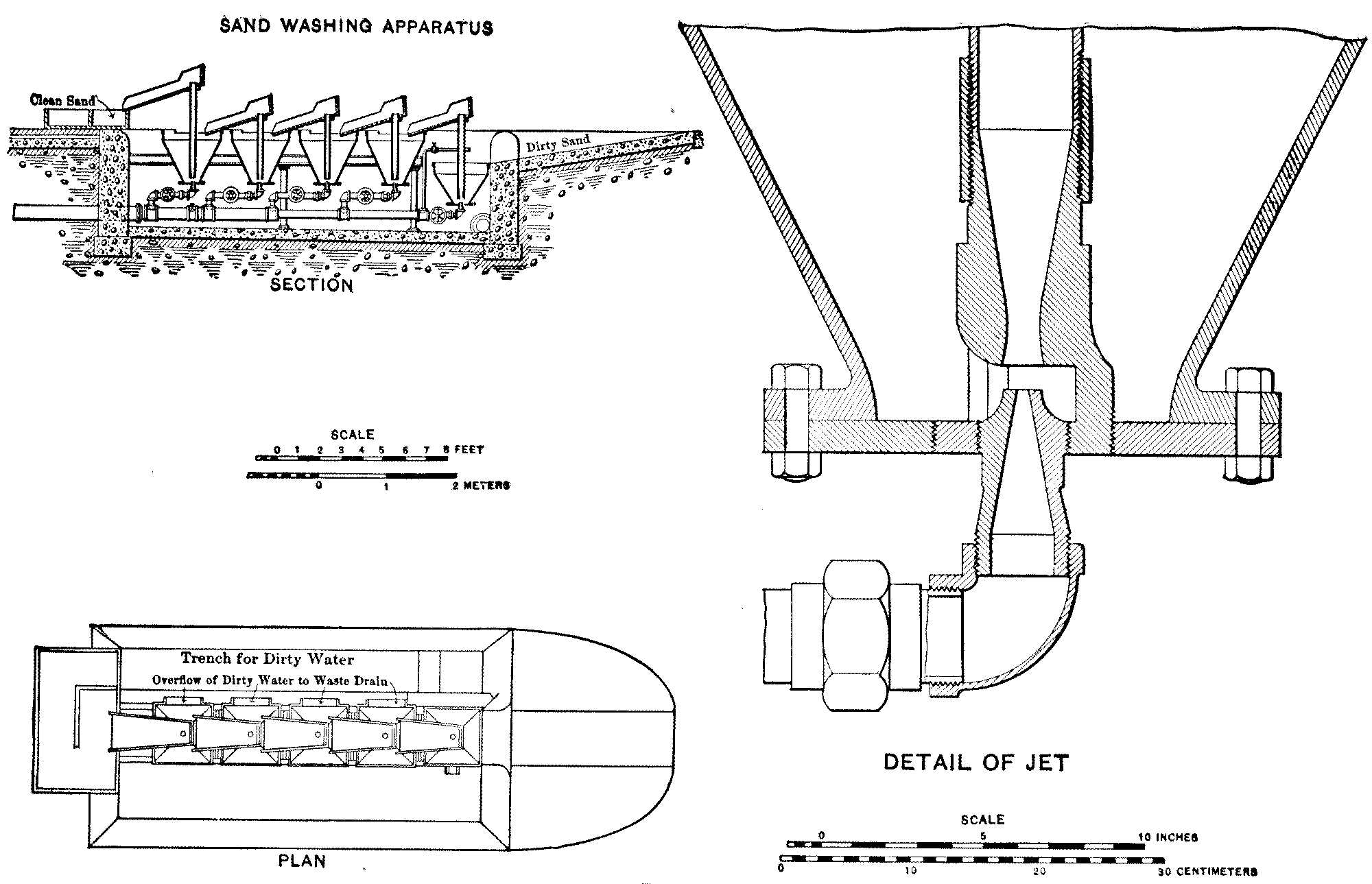
Fig. 9.
The sand was obtained from the river at various places by dredging. It was first taken up by dipper-dredges, and brought in scows to a point in the back channel a little north of the filter-plant. It was there dumped in a specially prepared place in the bottom of the river, from which it was lifted by a hydraulic dredge and pumped through a 15-inch pipe an average distance of 525 feet to points selected, and varied from time to time, on the flats north of the filters. The water containing the sand was then put through screens having meshes which excluded all stones 5 mm. in diameter and over, and was then taken into basins where the sand was deposited and afterward carted to the filters.
Two ejector sand-washing machines, shown in Fig. 9, are provided at convenient places between the filters. In them the dirty sand is mixed with water, and is thrown up by an ejector, after which it runs through a chute into a receptacle, from which it is again lifted by another ejector. It passes in all through five ejectors, part of the dirty water being wasted each time. The sand is finally collected from the last ejector, where it is allowed to deposit from the water.
Water is admitted to each filter through a 20-inch pipe from a pipe system connecting with the sedimentation-basin. Just inside of the filter-wall is placed a standard gate, and beyond that a balanced valve connected with an adjustable float to shut off the water when it reaches the desired height on the filter. These valves and floats were constructed from special designs, and are similar in principle to valves used for the same purpose in the Berlin water-filters.
Each filter is provided with an overflow, so arranged that it cannot be closed, which prevents the water-level from exceeding a fixed limit in case the balanced valve fails to act. An outlet is also provided near the sand-run, so that unfiltered water can be[Pg 305] removed quickly from the surface of the filter, should it be necessary, to facilitate cleaning.
The outlet of each filter is through a 20-inch gate controlled by a standard graduated to show the exact distance the gate is open. The water rises in a chamber and flows through an orifice in a brass plate 4 by 24 inches, the centre of which is 1 foot below the level of the sand-line. At the nominal rate of filtration, 3,000,000 gallons per acre daily, 1 foot of head is required to force the water through the orifice. With other rates the head increases or decreases approximately as the square of the rate and forms a measure of it. With water standing in the lower chamber, so that the orifice is submerged, it is assumed that the same rates will be obtained with a given difference in level between the water on the two sides of the orifice as from an equal head above the centre of the orifice when discharging into air.
Measurement of Effluent.—In order to show the rate of filtration two floats are connected with the water on the two sides of the orifice. These floats are counterbalanced; one carries a graduated scale, and the other a marker which moves in front of the scale and shows the rate of filtration corresponding to the difference in level of the water on the two sides. When the water in the lower chamber falls below the centre of the orifice, the water in the float-chamber is nevertheless maintained at this level. This is accomplished by making the lower part of the tube water-tight, with openings just at the desired level, so that when the water falls below this point in the outer chamber it does not fall in the float-chamber.
To prevent the loss of water in the float-chamber by evaporation or from other causes, a lead pipe is brought from the other chamber and supplies a driblet of water to it constantly; this overflows through the openings, and maintains the water-level at precisely the desired point. The floats thus indicate the difference in water-level on the two sides of the orifice whenever the water in the lower chamber is above the centre of the orifice; otherwise[Pg 306] they indicate the height of water in the upper chamber above the centre of the orifice, regardless of the water-level in the lower chamber. The scale is graduated to show the rates of filtration in millions of gallons per acre of filtering area. In computing this scale the area of the filters is taken as 0.7 acre, and the coefficient of discharge as 0.61.
At the ordinary rates of filtration the errors introduced by the different conditions under which the orifice operates will rarely amount to as much as 100,000 gallons per acre daily, or one thirtieth of the ordinary rate of filtration. Usually they are much less than this. The apparatus thus shows directly, and with substantial accuracy, the rate of filtration under all conditions.
Measurement of Loss of Head.—Two other floats with similar connections show the difference in level between the water standing on the filter and the water in the main drain-pipe back of the gate, or, in other words, the frictional resistance of the filter, including the drains. This is commonly called the loss of head, and increases from 0.2 foot or less, with a perfectly clean filter, to 4 feet with the filter ready for cleaning. When the loss of head exceeds 4 feet the rate of filtration cannot be maintained at 3,000,000 gallons per acre daily with the outlet devices provided, and, in order to maintain the rate, the filter must be cleaned.
Adjustment of Gauges.—The adjustment of the gauges showing the rate of filtration and loss of head is extremely simple. When a filter is put in service the gates from the lower chamber to the pure-water reservoir and to the drain are closed, the outlet of the filter opened, and both chambers allowed to fill to the level of the water on the filter. The length of the wire carrying the gauge is then adjusted so that the gauge will make the desired run without hitting at either end, and then the marker is adjusted. As both the rate of filtration and loss of head are zero under these conditions, it is only necessary to set the markers to read zero on the gauges to adjust them. The gates can then be opened for regular operation, and the readings on the gauges will be correct.

Interior of a Filter: Drain, Gravel and Sand Layers.

Interior of a Filter, Ready for Use.
[To face page 306.]
[Pg 307]
It is necessary to use wires which are light, flexible, and which will not stretch. At first piano-wire, No. 27 B. & S. gauge, was used, and was well adapted to the purpose, except that it rusted rapidly. Because of the rusting it was found necessary to substitute another wire, and cold-drawn copper wire, No. 24 B. & S. gauge, was used with fair results. Stretching is less serious than it would otherwise be, as the correctness of the adjustment can be observed and corrected readily every time a filter is out of service.
From the lower chambers in the regulator-houses the water flows through gates to the pipe system leading to the pure-water reservoir. Drain-pipes are also provided which allow the water to be entirely drawn out of each filter, should that be necessary for any reason, and without interfering with the other filters or with the pure-water reservoir.
The outlets of the filters are connected in pairs, so that filtered water can be used for filling the underdrains and sand of the filters from below prior to starting, thus avoiding the disturbance which results from bringing dirty water upon the sand of a filter not filled with water.
Laboratory Building.—The scientific control of filters is regarded as one of the essentials to the best results, and to provide for this there is a laboratory building at one end of the central court between the filters and close to the sedimentation-basin, supplied with the necessary equipment for full bacterial examinations, and also with facilities for observing the colors and turbidities of raw and filtered waters, and for making such chemical examinations as may be necessary. This building also provides a comfortable office, dark room, and storage room for tools, etc., used in the work.
Pure-water Reservoir.—A small pure-water reservoir, 94 feet square and holding about 600,000 gallons, is provided at the filter-plant. The construction is similar to that of the filters, but the shapes of the piers and vaulting were changed slightly, as there was no necessity for the ledges about the bottoms of the piers and[Pg 308] walls; while provision is made for taking the rain-water, falling upon the vaulting above, to the nearest filters instead of allowing it to enter the reservoir. The floor and roof of the reservoir are at the same levels as those of the filters.
CAPACITY OF PLANT AND MEANS OF REGULATION.
The various filters have effective filtering areas of from 0.702 to 0.704 acre, depending upon slight differences in the thickness of the walls in different places. For the purpose of computation, the area of each filter is taken at 0.7 acre. The nominal rate of filtration is taken as 3,000,000 gallons per acre daily, at which rate each filter will yield 2,100,000 gallons daily, and, with one filter out of use for the purpose of being cleaned, seven filters normally in use will yield 14,700,000 gallons. The entrances and outlets are all made of sufficient size, so that rates 50 per cent greater than the foregoing are possible. The capacities of the intake, pumping-station, and piping are such as to supply any quantity of water which the filters can take, up to an extreme maximum of 25,000,000 gallons in 24 hours. The pure-water conduit from the filters to Quackenbush Street is nominally rated at 25,000,000 gallons per 24 hours, after it has become old and somewhat tuberculated. In its present excellent condition it will carry a larger quantity,
At the pumping-station at Quackenbush Street there are three Allis pumps, each capable of pumping 5,000,000 gallons per 24 hours. In addition to the above there are the old reserve pumps with a nominal capacity of 10,000,000 gallons per 24 hours, which can be used if necessary, but which require so much coal that they are seldom used. For practical purposes the 15,000,000 gallons represents the pumping capacity of this station and also the capacity of the filters, but the arrangements are such that in case of emergency the supply can be increased to 20,000,000 or even 25,000,000 gallons for a short time.
[Pg 309]
The water is pumped through rising mains to reservoirs holding 37,000,000 gallons, not including the Tivoli low-service reservoir, which is usually supplied from gravity sources. The reservoir capacity is such that the pumping can be suspended at Quackenbush Street for considerable periods if necessary, and in practice it has been suspended at certain times, especially on Sundays. The amount of water required is also somewhat irregular. The drainage areas supplying the gravity reservoirs are much larger, relatively, than the reservoirs, and at flood periods the volume of the gravity supply is much greater than that which can be drawn in dry weather. Thus it happens that, at certain seasons of the year, the amount of water to be pumped is but a fraction of the nominal capacity of the pumps, and at these times it is possible to shut the pumps down for greater lengths of time.
Capacity of Pure-water Reservoir.—The storage capacity provided between the filters and the Quackenbush Street pumps is comparatively small, namely, 600,000 gallons, or one hour’s supply at the full nominal rate. A larger basin, holding as much as one third or one half of a day’s supply, would be in many respects desirable in this position, but the conditions were such as to make it practically impossible. The bottom of the reservoir could not be put lower without deepening and increasing greatly the expense of the conduit-line. On the other hand, the flow-line of the reservoir could not be raised without raising the level of the filters, which was hardly possible upon the site selected. The available depth of the reservoir was thus limited between very narrow bounds, and to secure a large capacity would have necessitated a very large area, and consequently a great expense. Under these circumstances, and especially in view of the abundant storage capacity for filtered water in the distributing reservoirs, it was not deemed necessary to provide a large storage, and only so much was provided as would allow the pumps to be started at the convenience of the engineer, and give a reasonable length of time for the filters to be brought into operation. For this the pure-water[Pg 310] reservoir is ample, but it is not enough to balance any continued fluctuations in the rate of pumping.
Method of Regulating and Changing the Rate of Filtration.—With all the Allis pumps running at their nominal capacity, the quantity of water required will just about equal the nominal capacity of the filters. When only one or two pumps are running, the rate of filtration can be reduced. With the plant operating up to its full capacity, the water-level in the pure-water reservoir will be below the level of the standard orifices in the filter outlets. When the rate of pumping is reduced, if no change is made in the gates controlling the filter outlets, the water will gradually rise in the pure-water reservoir and in the various regulator chambers, and will submerge the orifices and gradually reduce the head on the filters, and consequently the rates of filtration, until those rates equal the quantity pumped. In case the pumping is stopped altogether, the filters will keep on delivering at gradually reduced rates until the water-level in the pure-water reservoir reaches that of the water on the filters.
When the pumps are started up, after such stoppage or reduced rate of pumping, the water-levels in the pure-water reservoir and in the gate-chambers will be lowered gradually, and the filters will start to operate it first with extremely low rates, which will increase gradually until the water is depressed below the orifices, when they will again reach the rates at which they were last set. The regulators during all this time will show the rate of filtration on each filter, and, if any inequalities occur which demand correction, the gates on the various outlets can be adjusted accordingly.

Central Court, showing Sand-washer, Dirty Sand, etc.

Sedimentation Basin, Filters, etc.
[To face page 310.
The arrangement, in this respect, combines some of the features of the English and German plants. In the English plants the filters are usually connected directly with the clear-water basin, and that in turn with the pumps, and the speed of filtration is required to respond to the speed of the pumps, increasing and decreasing with it, being regulated at all times by the height of water in the pure-water reservoir. This arrangement has been subject to severe criticism, because the rate of filtration fluctuates with the consumption, and especially because the rates of filtration obtained simultaneously in different filters may be different. There was no way to determine at what rate any individual filter was working, and there was always a tendency for a freshly scraped filter to operate much more rapidly than those which had not been scraped for some time.
This led to the procedure, first formulated by the Commission of German Water-works Engineers in 1894, and provided for in most of the German works built or remodelled since that time, of providing pure-water storage sufficient in amount to make the rate of filtration entirely independent of the operation of the pumps. Each filter was to be controlled by itself, be independent of the others, and deliver its water into a pure-water reservoir lower than itself, so that it could never be affected by back-water, and so large that there would never be a demand for sudden changes in the rate of filtration.
This procedure has given excellent results in the German works; but it leads oftentimes to expensive construction. It involves, in the first place, a much greater loss of head in passing through the works, because the pure-water reservoir must be lower than the filters, and the cost of the pure-water reservoir is increased greatly because of its large size. The regulation of the filters is put upon the attendants entirely, or upon automatic devices, and regulation by what is known as “responding to the pumps” is eliminated.
More recently, the German authorities have shown less disposition to insist rigidly upon the principles advanced in 1894. In a compilation of the results of several years’ experience with German water-filters, Dr. Pannwiz[66] makes a statement of particular interest, of which a free translation is as follows:
“Most of the German works have sufficient pure-water reservoir capacity to balance the normal fluctuations in consumption,[Pg 312] so that the rate of filtration is at least independent of the hourly fluctuations in consumption. Of especial importance is the superficial area of the pure-water reservoir. If it is sufficiently large, there is no objection to allowing the water-level in it to rise to that of the water upon the filters. With very low rates of consumption during the night the filters may work slowly and even stop, without damage to the sediment layers when the stopping and starting take place slowly and regularly, because of the ample reservoir area.”
“The very considerable fluctuations from day to day, especially those arising from unusual and unforeseen occurrences, are not provided for entirely by even very large and well-arranged reservoirs. To provide for these without causing shock, the rate of filtration must be changed carefully and gradually, and the first essential to success is a good regulation apparatus.”
“Responding to the pumps” has a great deal to recommend it. It allows the pure-water reservoir to be put at the highest possible level, it reduces to a minimum the loss of head in the plant, and yet provides automatically, and without the slightest trouble on the part of the attendants, for the delivery of the required quantity of water by the filters at all times. If the filters are connected directly to the pumps there is a tendency for the pulsations of the pumps to disturb their operation, which is highly objectionable, even if the pumps are far removed; and this exists where filters are connected directly to the pumps, and a pure-water reservoir is attached to them indirectly. By taking all the water through the pure-water reservoir and having no connection except through it, this condition is absolutely avoided, and the pull on the filters is at all times perfectly steady.
Much has been said as to the effect of variation in the rate of filtration upon the efficiency of filters. Experiments have been made at Lawrence and elsewhere which have shown that, as long as the maximum rate does not exceed a proper one, and under reasonable regulations, and with the filter in all respects in good[Pg 313] order, no marked decrease in efficiency results from moderate fluctuations in rate. There is probably a greater decrease in efficiency by stopping the filter altogether, especially if it is done suddenly, than by simply reducing the rate. The former sometimes results in loosening air-bubbles in the sand, which rise to the surface and cause disturbances, but this is not often caused by simple change in rate.
On the whole, there is little evidence to show that, within reasonable limits, fluctuations in rate are objectionable, or should be excluded entirely, especially in such cases as at Albany, where arrangements to prevent them would have resulted in very greatly increased first cost. The inferior results sometimes obtained with the system of “responding to the pumps” as it existed in earlier works, and still exists in many important places, undoubtedly arises from the fact that there is no means of knowing and controlling the simultaneous rate of filtration in different filters, and that one filter may be filtering two or three times as fast as another, with nothing to indicate it.
This contingency is fully provided for in the Albany plant. The orifices are of such size that even with a filter just scraped and put in service, with the minimum loss of head, with the outlet-gate wide open, and with the water-level in the pure-water reservoir clear down—that is, with the most unfavorable conditions which could possibly exist—the rate of filtration cannot exceed 5,000,000 or 6,000,000 gallons per acre daily, or double the nominal rate. This rate, while much too high for a filter which has just been cleaned, is not nearly as high as was possible, and in fact actually occurred in the old Stralau filters at Berlin, and in many English works; and, further, such a condition could only occur through the gross negligence of the attendants, because the rate of filtration is indicated clearly at all times by the gauges. These regulating-devices have been specially designed to show the rate with unmistakable clearness, so that no attendant, however stupid, can make an error by an incorrect computation from the[Pg 314] gauge heights. It is believed that the advantage of clearness by this procedure is much more important than any increased accuracy which might be secured by refinements in the method of computation, which should take into account variations in the value of the coefficient of discharge, but which would render direct readings impossible.
In designing the Albany plant the object has been to combine the best features of German regulation with the economical and convenient features of the older English system, and filters are allowed to respond to the pumps within certain limits, while guarding against the dangers ordinarily incident thereto.
RESULTS OF OPERATION.
The filters were designed to remove from the water the bacteria which cause disease. They have already reached a bacterial efficiency of over 99 per cent, and it is expected that their use will result in a great reduction in the death-rate from water-borne diseases in the city. They also remove a part of the color and all of the suspended matters and turbidity, so that the water is satisfactory in its physical properties.
The filters have reached with perfect ease their rated capacity, and on several occasions have been operated to deliver one third more than this amount; that is to say, at a rate of 4,000,000 gallons per acre, daily.
COST OF CONSTRUCTION.
The approximate cost of the filtration-plant complete was as follows:
| Land | $8,290 |
Pumping-station and intake |
49,745 |
Filters and sedimentation-basin, with piping |
323,960 |
Pure-water conduit and connection with Quackenbush Street pumping-station |
86,638 |
Engineering and minor expenses |
28,000 |
| Total | $496,633 |
[Pg 315]
The filters, sedimentation-basin, and pure-water reservoir are connected in such a way as to make an exact separation of their costs impossible; but, approximately, the sedimentation-basin cost $60,000, the pure-water reservoir $9,000, and the filters $255,000. The sedimentation-basin thus cost $4,100 per million gallons capacity; and the filters complete cost $45,600 per acre of net filtering area, including all piping, office and laboratory building, but exclusive of land and engineering.
ACKNOWLEDGMENT.
The general plan and location of the plant were first conceived by the Superintendent of Water-works, George I. Bailey, M. Am. Soc. C. E., and the successful execution is largely due to his efforts. The members of the Water Board, and especially the Construction Committee, have followed the work in detail closely and personally, and their interest and support have been essential factors in the results accomplished. In the designs and specifications for the pure-water conduit the author is greatly indebted to Emil Kuichling, M. Am. Soc. C. E., and also for most valuable suggestions relative to the performance of this part of the work. To William Wheeler, M. Am. Soc. C. E., of Boston, the author is indebted for advice upon the vaulting and cross-sections of the walls, and these matters were submitted to him before the plans were put in final shape. All the architectural designs have been supplied by Mr. A. W. Fuller, of Albany. W. B. Fuller, M. Am. Soc. C. E., as Resident Engineer, has been in direct charge of the work, and its success is largely due to his interest in it and the close attention which he and the assistant engineers have given it.
FOOTNOTES:
[1] The American gallon is 231 cubic inches or 0.8333 of the imperial gallon. In this work American gallons are always used, and English quantities are stated in American, not imperial, gallons.
[2] Filtration of River Waters. Van Nostrand & Co., 1869.
[3] Annual Report of Albert F. Noyes, City Engineer for 1891.
[4] Rept. Mass. State Board of Health, 1892, p. 541. See Appendix III.
[5] The method of calculating the size is given in Appendix III.
[6] A full table of frictions with various velocities and gravels was given in the Rept. of Mass. State Board of Health, 1892, p. 555.
[7] Frühling, Handbuch der Ingenieurwissenschaften, II. Band, VI. Kapitel.
[8] The American gallon is used throughout this book; the English gallon is one fifth larger.
[9] Piefke, Zeitschrift für Hygiene, 1894, p. 177.
[10] Zeitschrift für Hygiene, 1891, page 38.
[11] Journal für Gas- u. Wasserversorgung, 1891, 208 and 228.
[12] Journal für Gas- u. Wasserversorgung, 1893, 161.
[13] Samuelson’s translation of Kirkwood’s “Filtration of River-waters;” Lindley, Die Nutzbarmachung des Flusswassers, Journal für Gas- u. Wasserversorgung, 1890, 501; Kaiserlichen Gesundheitsamt, Grundsätze für die Reinigung von Oberflächenwasser durch Sandfiltration; Journal für Gas- u. Wasserversorgung, 1894, Appendix I.
[14] Lindley, Journal für Gas- u. Wasserversorgung, 1890, 501; Grahn, Journal für Gas- u. Wasserversorgung, 1890, 511; Halbertsma, Journal für Gas- u. Wasserversorgung, 1892, 686; Piefke, Zeitschrift für Hygiene, 1894, 151; and others.
[15] Appendix I.
[16] The Water Supply of Towns. London, 1894.
[17] A special species of bacteria artificially added to secure more precise information in regard to the passage of germs through the filter.
[18] Zeitschrift für Hygiene, 1894, p. 173.
[19] Report Mass. State Board of Health for 1891, p. 438; 1892, page 409.
[20] Appendix IV.
[21] Piefke, Zeitschrift für Hygiene, 1894, p, 177.
[22] Journal für Gas- und Wasserversorgung, 1887, p. 595.
[23] Zeitschrift für Hygiene, 1894, p. 172.
[24] Appendix IV.
[25] Appendix I.
[26] Glaser’s Annalen, 1886, p. 48; Zeit. f. Hygiene, 1889, p. 128.
[27] Vierteljahresschrift für öffentliche Gesundheitspflege, 1891, p. 59.
[28] Journal für Gas- und Wasserversorgung, 1891, 108.
[29] Zeitschrift für Hygiene, 1894, 182.
[30] I am informed that several other filters upon the same principle have been more recently built.
[31] Report on Water Purification at Cincinnati, page 378.
[32] Translation in German in Dingler’s Polytechnical Journal, 1832, 386.
[33] Water Purification at Louisville, page 378.
[34] Special Report Mass. State Board of Health 1890, Purification of Sewage and Water, page 747.
[35] Water Purification at Cincinnati, p. 485.
[36] Jour. of the New England Water Works Assoc., Vol. VIII, page 183.
[37] Report of the Pittsburg Filtration Commission, 1899, page 55.
[38] Rhode Island State Board of Health Report for 1894.
[39] Report of the Rhode Island State Board of Health for 1894.
[40] Report on the Investigations into the Purification of the Ohio River Water at Louisville, Kentucky. D. Van Nostrand & Co., 1898.
[41] Ohio State Board of Health Report, 1897, page 154.
[42] Report of the Pittsburg Filtration Commission, City Document, 1899.
[43] Fuller, Water Purification at Louisville, page 425.
[44] Warren, Feb. 9; June 1; July 6. Jewell, July 1; Feb. 9, 16, 17.
[45] “Removal of Iron from Ground Waters,” Journal of the New England Water Works Association, Vol. xi, 1897, page 277.
[46] Journal of the New England Water Works Association, Vol. ii, page 294. Description of plant by Supt. Lewis M. Bancroft.
[47] This number was the result of numerous counts made from fæces from persons suffering with typhoid fever in the Lawrence City Hospital in 1891 and 1892. Mr. G. W. Fuller afterward made at the Lawrence Experiment Station some further investigation of fæces from healthy people in which the numbers were considerably lower, usually less than 200,000,000, per gram and sometimes as low as 10,000,000 per gram.
[48] These experiments, so far as they have come to the notice of the author, have been made with water sterilized by heating, usually in small tubes stoppered with cotton-wool or other organic matter. In this case the water, no matter how carefully purified in the first place, becomes an infusion of organic matters capable of supporting bacterial growths, and not at all to be compared to natural waters.
In experiments often repeated under my direction, carefully distilled water in bottles, most scrupulously clean, with glass stoppers, and protected from dust, but not sterilized, has uniformly refused to support bacterial growths even when cautiously seeded at the start, and the same is usually true of pure natural waters. Some further experiments showed hardly any bacterial growth even of the most hardy water bacteria in a solution 1 part of peptone in 1,000,000,000 parts of distilled water, and solutions ten times as strong only gave moderate growths.
[49] The Water-supply of Chicago: Its Source and Sanitary Aspects. By Arthur R. Reynolds, M.D., Commissioner of Health of Chicago, and Allen Hazen. American Public Health Association, 1893. Page 146.
[50] Journal für Gas- u. Wasserversorgung, 1893, 694.
[51] Journal für Gas- u. Wasserversorgung, 1894, 185.
[52] The method of making this determination was given in the American Chemical Journal, vol. 12, p. 427.
[53] Some of the companies secure some ground water which they mix with the filtered water, and this is included in the quantities for the separate companies, but is excluded from the totals for all the companies by years.
[54] Exclusive of gravity supplies.
[55] Not in use.
[56] Under construction.
[57] Not in use.
[58] Under construction.
[59] Not in use.
[60] Under construction.
[61] Not in use.
[62] Under construction.
[63] In the Centralblatt für Bakteriologie, 1895, page 881, Reinsch discusses at length the cause of the inferior results at Altona in winter, and has apparently discovered a new factor in producing them. Owing to defective construction of the outlets for the sedimentation-basins they have failed to act properly in presence of excessive quantities of ice, and the sediment from the basins has been discharged in large quantity upon the filters, and a small fraction of the many millions of bacteria in it have passed through the filters. He has experimented with this sediment applied to small filters, and has become convinced that to secure good work under all conditions a much deeper layer of sand than that generally considered necessary must be used, and his work emphasizes the importance of the action of the sand in distinction from the action of the sediment layer, which has often been thought to be the sole, or at least the principal, requirement of good filtration.
[64] Licht- u. Wasserwerke, Zürich, 1892, page 32.
[65] Descriptions of some of the leading European ground-water supplies were given by the author in the Jour. Asso. Eng. Soc., Feb. 1895, p. 113.
[66] “Arbeiten aus dem Kaiserlichen Gesundheitsamte,” vol. xiv. p. 260.
[Pg 317]
INDEX.
- Albany, N. Y., filters at, 254, 288.
- Alkalinity, 155.
- Altona, double filtration at, 198.
- filters at, 265.
- Alum, use of, in filtration, 92, 144.
- American cities, water-supplies of, and typhoid fever in, 211.
- Amsterdam, filters at, 272.
- iron removal at, 192.
- Anderson process, 147.
- Antwerp, filters at, 272.
- Asbestos as filtering material, 181.
- Asbury Park, iron removal at, 192.
- Ashland, Wis., filters at, 252.
- Area of filters to be provided, 47.
- Bacteria, apparent and actual removal of, by filters, 87.
- from underdrains, 87.
- in Elbe at Altona, 228.
- in fæces, 215.
- in water, 84.
- number to be allowed in filtered water, 222.
- of cholera in river water, 231.
- of typhoid fever, life of, in water, 216.
- of special kinds to test efficiency of filtration, 86.
- to be determined daily, 222.
- Bacterial examination of water, 93.
- Berlin, regulation of depth of water, 59.
- cholera infantum from water, 229.
- friction in underdrains, 44.
- regulation of rate, 53, 55.
- water works, 261.
- Berwyn, Penn., filters at, 253.
- Boston, protection of purity of water-supply, 110.
- experimental filters at, 73.
- Bremen, double filtration at, 198.
- Breslau, filters at, 274.
- Brussels, ground-water, supply of, 276.
- Budapest, filters at, 274.
- Burton, regulation of rate at Tokyo, Japan, 58.
- Carpenter, Prof. L. G., 24.
- Chemnitz, intermittent filtration at, 107.
- Chicago, reduced death-rate with new intake, 217.
- Cholera infantum from impure water, 226.
- Cholera, in Hamburg from water, 230.
- caused by water, 214.
- Clarification, definition of, 113.
- Clark, H. W., 24, 190.
- Clark’s process for softening water, 92, 145.
- Clay particles, size of, 123.
- Cleaning filters, 68.
- Coagulant, absorption of, by suspended matters, 154.
- successive applications of, 154.
- Coagulants used in practice, 150.
- Coagulation of waters, 144.
- Cologne, water-supply of, from wells, 276.
- Color, 113.
- amount of coagulant required to remove, 153.
- amount of, in various waters, 115.
- [Pg 318]measurement of, 114.
- Color, removal of, 117.
- Continuous filters, 5.
- filtration, nature of, 83, 92.
- Cost of filters and filtration, 4, 48, 102, 200, 314.
- Covered filters, efficiency of, 17.
- Covers for filters, 12, 15.
- at Albany, 295.
- in the United States, 17.
- omitted at Lawrence, 101.
- Crenothrix, 105, 186.
- Diarrhœa from impure water, 226.
- Dibden, W. J., 129.
- Disease from water, 210.
- Double filtration at Schiedam, 273.
- Drainage areas of a number of rivers, 133.
- Dresden, water-supply of, from filter-gallery, 276.
- Drown, Dr. Thomas M., 150, 191.
- Effective size of sand, 21, 238.
- European sands, 25.
- Efficiency of filtration, 83, 88, 91.
- effect of rate upon, 50.
- effect of size of sand-grain upon, 30.
- effect of thickness of sand layer upon, 34.
- at Lawrence, 106.
- European filters, 91, 260.
- Effluents, wasting after scraping, 74.
- Fæces, number of bacteria in, 215.
- Far Rockaway, L. I., filters at, 193, 253.
- Filling sand with water from below, 68, 307.
- Filter beds, bottoms of, must be water-tight, 12.
- covers for, 12.
- form of, 11.
- size of, 10.
- Filters, aggregate capacity of, 254.
- depths of waters on, 45.
- list of cities using, 244.
- reserve area required, 47.
- first constructed at London, 83.
- for household use, 183.
- general arrangement of, 6.
- Filters, statistics of, at various cities, 241.
- Filtration, cost of, 200.
- degree of purification required, 5.
- general nature of, 92.
- Fischer tile system, 181.
- FitzGerald, Desmond, 73, 111, 196.
- Flood flows not taken for supply, 10.
- Fränkel and Piefke, experiments on removal of disease germs, 86.
- Frankfort on Main, water supply of, from springs, 276.
- Frankland, Dr. Percy, 84.
- Friction of filtered water in pipes, 264.
- water in gravel, 37.
- water in sand, 22.
- water in underdrains, 40.
- Frost, effect of, upon filters, 12, 229, 266.
- Frühling, on the heating of water by sunshine, 16.
- underdraining at Königsberg, 39.
- Fuller, G. W., 118, 123, 131, 139, 140, 145, 152, 154, 161, 165.
- German Imperial Board of Health, 34, 51, 54, 75, 95.
- regulations in regard to filtration, 221.
- Gill, apparatus for regulation, 55.
- Glasgow, water-supply of, from Loch Katrine, 275.
- Gravel at Albany, 299.
- layers, 35.
- friction of water in, 37.
- screening of, for filters, 37.
- Grand Forks, N. D., filters at, 252.
- Ground-water supplies, 3.
- the use of, in Europe, 276.
- Halbertsma, H. P. N., 54, 59.
- Hamburg, apparatus for regulating depth of water, 59.
- health of, 226, 271.
- regulation of rate of filtration, 56.
- underdrains of filters at, 42.
- water-supply of, 269.
- Hamilton, N. Y., filters at, 253.
- Hardness, removal of, 92, 145.
- Harrisburg, Penn., filters at, 253.
- Hermany, Charles, 161.
- [Pg 319] High rates of filtration without coagulant, 182.
- Household filters, 183.
- Hudson, N. Y., filters at, 251.
- Ice on filters, 13.
- Inlet regulators, 59.
- Impounding reservoirs, 2.
- Intermittent filtration, 97.
- application of, 111, 197.
- at Chemnitz, 107.
- at Lawrence, 100.
- of Pegan Brook, 110.
- Iron, compounds of, as coagulants, 146.
- in ground-waters, 186.
- in ground-water at Lawrence, 105.
- metallic, the Anderson process, 147.
- present as ferrous sulphate, 191.
- removal plants in operation, 192.
- Iron waters, treatment of, 189.
- Jewel filter, 151, 161, 162, 172, 173.
- Kirkwood, James P., 8, 36, 47, 51, 55, 61, 63, 67.
- Kümmel, 50, 51, 86.
- Lambertsville, N. J., filters at, 252.
- Lawrence City filter, description of, 100.
- Lawrence Experiment Station, 97.
- air in water filtered in winter at, 46.
- depth of sand removed at, 70.
- depth of water on filters, 46.
- effect of loss of head upon efficiency, 61.
- effect of size of sand-grain upon efficiency, 32.
- effect of size of sand-grain upon frequency of scraping, 32.
- efficiency of filters at various rates, 50.
- efficiency of filtration at, 86, 89.
- experiments with continuous filtration, 110.
- filters of fine sand, 31.
- filters of various sand-grain sizes, 32.
- gravel for filters at, 39.
- growth of bacteria in sterilized sand at, 85.
- intermittent filtration investigated, 97.
- Lawrence Experiment Station, method of sand analysis at, 20.
- quantities of water filtered at various losses of head, 66.
- wasting effluents not necessary, 75.
- Lawrence, typhoid fever at, 102.
- Leipzig, water-supply of, from wells, 276.
- Lime in sand, 29.
- sterilizing effect of, 146.
- as a coagulant, 145.
- application of, to water, 157.
- Lindley, 43, 51, 54, 57, 81.
- Literature on filtration, 277, 285.
- Little Falls, N. Y., filters at, 253.
- Loam in filters, 35.
- London, cost of operating filters at, 202.
- water-supply of, 255.
- Long, Prof., 131.
- Lorain, tests of mechanical filters, 161.
- Loss of head, 52.
- limit to, 60, 67.
- reasons for allowing high, 65.
- Louisville, mechanical filters at, 161.
- Magdeburg, filters at, 273.
- Maignen system, 181.
- Manchester, water-supply of, 275.
- Manganese, compounds of, as coagulants, 148.
- in ground-waters, 188.
- Massachusetts State Board of Health, see Lawrence Experiment
- Station.
- Mechanical filters, 159.
- application of, 199.
- efficiency of, 179.
- list of, 247.
- pressure filters, 180.
- rates of filtration used, 175.
- types of, 172.
- wasting effluent after washing, 163.
- Millford, Mass., filters at, 252.
- Mills, H. F., 97, 99, 102.
- Mount Vernon, N. Y., filters at, 252.
- Mud, see turbidity.
- Muddy waters, 113.
- Munich, water-supply of, from springs, 275.
- [Pg 320]Nichols, Prof., suspended matters in European streams, 131.
- Nitrification, effect of, upon bacteria, 98.
- Odors, removal of, by filtration, 112.
- Organic matters in water, 83.
- removed by intermittent filters, 98.
- Paper manufacturing, filtration of water for, 5.
- Paris, ground-water supply of, 276.
- Palmer, Prof., 131.
- Passages through the sand in filters, 67.
- Pegan Brook, purification of, 110.
- Period, how computed and length of, 72.
- length of, dependent upon turbidity, 137.
- Piefke, 48, 50, 54, 63, 69, 73, 74, 75, 80, 84, 85, 90.
- Pittsburgh, experiments with mechanical filters, 162.
- Plägge and Proskauer, 84.
- Plymouth, Penn., typhoid fever at, 208.
- Pollution of European water-supplies, 93.
- Polluted waters, utilization of excessively, 111.
- Porcelain filters for household use, 183.
- Poughkeepsie, N. Y., filters at, 251.
- Pressure filters, 180.
- Providence, mechanical filters at, 159.
- Rate of filtration, 47, 224.
- at various places, 241.
- effect of, upon cost, 48.
- effect of, upon efficiency, 50.
- lower after scraping, 76.
- regulation of, 52.
- Red Bank, N. J., filters at, 193, 253.
- Regulation of filters, 52.
- old forms of regulators, 52.
- modern forms of regulators, 54.
- at Albany, 305, 308, 310.
- of mechanical filters, 178.
- Reincke, Dr., report on health of Hamburg for 1892, 226.
- Reinsch on the cause of poor filtration at Altona, 267.
- Reserve area required in case of ice, 18.
- Reservoirs, purposes served by, 133.
- Rock Island, Ill., filters at, 254.
- Roofs for filters, 16.
- Rotterdam, filters at, 272.
- St. Johnsbury, Vt., filters at, 251.
- St. Louis, regulators for proposed filters, 55.
- St. Petersburg, filters at, 275.
- Samuelson, 51.
- Sand, 20.
- at Albany, 301.
- analysis of European, 25.
- analysis of, from leading works, 28.
- appliances for moving, 68.
- compactness of, in natural banks, 61.
- depth of, in filters, 34.
- depth to be removed from filters, 69.
- dune, 26.
- dune, washing of, impossible, 82.
- effect of grain-size upon frequency of scraping, 32.
- effect of grain-size upon the efficiency, 30.
- effective size of, 21, 238.
- extra scraping before replacing fresh, 71.
- for filtration, 20, 33.
- for mechanical filters, 175.
- friction of water in, 22.
- grain-size of, 20, 233.
- in European filters, 24.
- in Lawrence filters, two sizes of, 100.
- lime in, 29.
- method of analysis of, 233.
- quantity to be removed by scraping, 74.
- replacing, 71.
- selection of, 33.
- size of passages between grains of, 6.
- sterilized, experiments with, 85.
- thickness of layer, 34.
- uniformity coefficient, 21, 238.
- Sand washing, 26, 76, 304.
- cost of, 81.
- water for, 80.
- Sandstone filters for household use, 183.
- Schiedam, double filtration at, 273.
- Scraping filters, 7, 68.
- [Pg 321] Scraping filters, amount of labor required for, 81.
- depth of sand removed, 33, 66, 69.
- frequency of, 49, 72, 241.
- Sedgwick, Prof. W. T., 86.
- Sediment, removal of, 92, 133.
- Sediment layer, 6, 31.
- influence of, upon bacterial purification, 84.
- thickness of, 33, 66, 69.
- Sedimentation basins, 8, 133, 293.
- effect of, 134.
- Sewage, number of bacteria in, 215.
- Simpson, James, 83.
- Soda-ash, application of, 157.
- Somersworth, N. H., filters at, 253.
- Storage for raw water, 136.
- Subsidence, limits to the use of, 142.
- Sulphate of alumina, action of, upon waters, 144.
- Surface-waters, use of, unfiltered, 275.
- Suspended matters, 113, 117.
- in relation to turbidities, 122.
- in various waters, 129.
- The Hague, iron removal at, 192.
- Tokyo, regulation of rate at, 58.
- Trenched bottoms for filters, 36, 40, 100.
- Turbidity, 92, 113.
- amount which is noticeable, 121.
- amount in several streams, 124.
- duration of, 128.
- in relation to suspended matters, 122.
- measurement of, 117.
- power of sand filters to remove, 139.
- preliminary processes to remove, 133.
- source of, 123.
- Typhoid fever in Berlin and Altona, 12, 85, 267.
- in American cities, 211.
- Typhoid fever in Hamburg, 271.
- in Lawrence, 102.
- in London, 259.
- in Zürich, 275.
- Typhoid-fever germs, life of, in water, 216.
- Underdrains, 35, 39.
- bacteria from, 87.
- friction of, at Albany, 299.
- size of, 41.
- ventilators for, 44.
- Uniformity coefficient of sand, 21, 238.
- Ventilators for underdrains, 44.
- Vienna, water-supply of, from springs, 276.
- Warren filter, 151, 161, 162, 172, 176, 177.
- Warsaw, filters at, 275.
- friction in underdrains, 43.
- regulation of rate at, 57.
- Wasting effluents, 74.
- Water, depth of, on filters, 45, 59.
- heating of, in filters, 45.
- organic matters in, 83.
- Water-supplies of American cities, 211.
- Water-supply and disease, 210.
- Waters, what require filtration, 207.
- Weston, E. B., 153, 154, 159.
- Weston, R. S., 153, 189.
- West Superior, iron in ground-water at, 189.
- Winter, effect of, upon filtration, 12.
- temperatures of places having open and covered filters, 15.
- Worms tile system, 181.
- Zürich, filters at, 274.
[Pg 1]
SHORT-TITLE CATALOGUE
OF THE
PUBLICATIONS
OF
JOHN WILEY & SONS,
New York.
London: CHAPMAN & HALL, Limited.
ARRANGED UNDER SUBJECTS.
Descriptive circulars sent on application. Books marked with an asterisk (*) are sold at net prices only, a double asterisk (**) books sold under the rules of the American Publishers’ Association at net prices subject to an extra charge for postage. All books are bound in cloth unless otherwise stated.
| AGRICULTURE. | ||||
|---|---|---|---|---|
Armsby’s Manual of Cattle-feeding |
12mo, | $1 75 | ||
Principles of Animal Nutrition |
8vo, | 4 00 | ||
Budd and Hansen’s American Horticultural Manual: |
||||
Part I. Propagation, Culture, and Improvement |
12mo, | 1 50 | ||
Part II. Systematic Pomology |
12mo, | 1 50 | ||
Downing’s Fruits and Fruit-trees of America |
8vo, | 5 00 | ||
Elliott’s Engineering for Land Drainage |
12mo, | 1 50 | ||
Practical Farm Drainage |
12mo, | 1 00 | ||
Green’s Principles of American Forestry |
12mo, | 1 50 | ||
Grotenfelt’s Principles of Modern Dairy Practice. (Woll.) |
12mo, | 2 00 | ||
Kemp’s Landscape Gardening |
12mo, | 2 50 | ||
Maynard’s Landscape Gardening as Applied to Home Decoration |
12mo, | 1 50 | ||
* McKay and Larsen’s Principles and Practice of Butter-making |
8vo, | 1 50 | ||
Sanderson’s Insects Injurious to Staple Crops |
12mo, | 1 50 | ||
Insects Injurious to Garden Crops. (In preparation.) |
||||
Insects Injuring Fruits. (In preparation.) |
||||
Stockbridge’s Rocks and Soils |
8vo, | 2 50 | ||
Winton’s Microscopy of Vegetable Foods |
8vo, | 7 50 | ||
Woll’s Handbook for Farmers and Dairymen |
16mo, | 1 50 | ||
| ARCHITECTURE. | ||||
Baldwin’s Steam Heating for Buildings |
12mo, | 2 50 | ||
Bashore’s Sanitation of a Country House |
12mo, | 1 00 | ||
Berg’s Buildings and Structures of American Railroads |
4to, | 5 00 | ||
Birkmire’s Planning and Construction of American Theatres |
8vo, | 3 00 | ||
Architectural Iron and Steel |
8vo, | 3 50 | ||
Compound Riveted Girders as Applied in Buildings |
8vo, | 2 00 | ||
Planning and Construction of High Office Buildings |
8vo, | 3 50 | ||
Skeleton Construction in Buildings |
8vo, | 3 00 | ||
Brigg’s Modern American School Buildings |
8vo, | 4 00 | ||
Carpenter’s Heating and Ventilating of Buildings |
8vo, | 4 00 | ||
Freitag’s Architectural Engineering |
8vo, | 3 50 | ||
Fireproofing of Steel Buildings |
8vo, | 2 50 | ||
French and Ives’s Stereotomy |
8vo, | 2 50 | ||
| [Pg 2]
Gerhard’s Guide to Sanitary House-inspection |
16mo, | 1 00 | ||
Theatre Fires and Panics |
12mo, | 1 50 | ||
* Greene’s Structural Mechanics |
8vo, | 2 50 | ||
Holly’s Carpenters’ and Joiners’ Handbook |
18mo, | 75 | ||
Johnson’s Statics by Algebraic and Graphic Methods |
8vo, | 2 00 | ||
Kidder’s Architects’ and Builders’ Pocket-book. Rewritten Edition |
16mo, mor., | 5 00 | ||
Merrill’s Stones for Building and Decoration |
8vo, | 5 00 | ||
Non-metallic Minerals: Their Occurrence and Uses |
8vo, | 4 00 | ||
Monckton’s Stair-building |
4to, | 4 00 | ||
Patton’s Practical Treatise on Foundations |
8vo, | 5 00 | ||
Peabody’s Naval Architecture |
8vo, | 7 50 | ||
Richey’s Handbook for Superintendents of Construction |
16mo, mor., | 4 00 | ||
Sabin’s Industrial and Artistic Technology of Paints and Varnish |
8vo, | 3 00 | ||
Siebert and Biggin’s Modern Stone-cutting and Masonry |
8vo, | 1 50 | ||
Snow’s Principal Species of Wood |
8vo, | 3 50 | ||
Sondericker’s Graphic Statics With Applications To Trusses, Beams, and Arches |
8vo, | 2 00 | ||
Towne’s Locks and Builders’ Hardware |
18mo, morocco, | 3 00 | ||
Wait’s Engineering and Architectural Jurisprudence |
8vo, | 6 00 | ||
| Sheep, | 6 50 | |||
Law of Operations Preliminary To Construction in Engineering and Architecture |
8vo, | 5 00 | ||
| Sheep, | 5 50 | |||
Law of Contracts |
8vo, | 3 00 | ||
Wood’s Rustless Coatings: Corrosion and Electrolysis of Iron and Steel |
8vo, | 4 00 | ||
Worcester and Atkinson’s Small Hospitals, Establishment and Maintenance, Suggestions for Hospital Architecture, with Plans for a Small Hospital |
12mo, | 1 25 | ||
The World’s Columbian Exposition of 1893 |
Large 4to, | 1 00 | ||
| ARMY AND NAVY. | ||||
Bernadou’s Smokeless Powder, Nitro-cellulose, and the Theory of the Cellulose Molecule |
12mo, | 2 50 | ||
* Bruff’s Text-book Ordnance and Gunnery |
8vo, | 6 00 | ||
Chase’s Screw Propellers and Marine Propulsion |
8vo, | 3 00 | ||
Cloke’s Gunner’s Examiner |
8vo, | 1 50 | ||
Craig’s Azimuth |
4to, | 3 50 | ||
Crehore and Squier’s Polarizing Photo-chronograph |
8vo, | 3 00 | ||
* Davis’s Elements of Law |
8vo, | 2 50 | ||
| * | Treatise on the Military Law of United States |
8vo, | 7 00 | |
| Sheep, | 7 50 | |||
De Brack’s Cavalry Outposts Duties. (Carr.) |
24mo, morocco, | 2 00 | ||
Dietz’s Soldier’s First Aid Handbook |
16mo, morocco, | 1 25 | ||
* Dredge’s Modern French Artillery |
4to, half morocco, | 15 00 | ||
Durand’s Resistance and Propulsion of Ships |
8vo, | 5 00 | ||
* Dyer’s Handbook of Light Artillery |
12mo, | 3 00 | ||
Eissler’s Modern High Explosives |
8vo, | 4 00 | ||
* Fiebeger’s Text-book on Field Fortification |
Small 8vo, | 2 00 | ||
Hamilton’s The Gunner’s Catechism |
18mo, | 1 00 | ||
* Hoff’s Elementary Naval Tactics |
8vo, | 1 50 | ||
Ingalls’s Handbook of Problems in Direct Fire |
8vo, | 4 00 | ||
| * | Ballistic Tables |
8vo, | 1 50 | |
* Lyons’s Treatise on Electromagnetic Phenomena. |
Vols. I. and II. 8vo, each, | 6 00 | ||
* Mahan’s Permanent Fortifications. (Mercur.) |
8vo, half morocco, | 7 50 | ||
Manual for Courts-martial |
16mo, morocco, | 1 50 | ||
* Mercur’s Attack of Fortified Places |
12mo, | 2 00 | ||
| * | Elements of the Art of War |
8vo, | 4 00 | |
| [Pg 3]
Metcalf’s Cost of Manufactures—And the Administration of Workshops |
8vo, | 5 00 | ||
| * | Ordnance and Gunnery. 2 vols |
12mo, | 5 00 | |
Murray’s Infantry Drill Regulations |
18mo, paper, | 10 | ||
Nixon’s Adjutants’ Manual |
24mo, | 1 00 | ||
Peabody’s Naval Architecture |
8vo, | 7 50 | ||
* Phelps’s Practical Marine Surveying |
8vo, | 2 50 | ||
Powell’s Army Officer’s Examiner |
12mo, | 4 00 | ||
Sharpe’s Art of Subsisting Armies in War |
18mo, morocco, | 1 50 | ||
* Walke’s Lectures on Explosives |
8vo, | 4 00 | ||
* Wheeler’s Siege Operations and Military Mining |
8vo, | 2 00 | ||
Winthrop’s Abridgment of Military Law |
12mo, | 2 50 | ||
Woodhull’s Notes on Military Hygiene |
16mo, | 1 50 | ||
Young’s Simple Elements of Navigation |
16mo, morocco, | 2 00 | ||
| ASSAYING. | ||||
Fletcher’s Practical Instructions in Quantitative Assaying with the Blowpipe |
12mo, morocco, | 1 50 | ||
Furman’s Manual of Practical Assaying |
8vo, | 3 00 | ||
Lodge’s Notes on Assaying and Metallurgical Laboratory Experiments |
8vo, | 3 00 | ||
Low’s Technical Methods of Ore Analysis |
8vo, | 3 00 | ||
Miller’s Manual of Assaying |
12mo, | 1 00 | ||
Minet’s Production of Aluminum and its Industrial Use. (Waldo.) |
12mo, | 2 50 | ||
O’Driscoll’s Notes on the Treatment of Gold Ores |
8vo, | 2 00 | ||
Ricketts and Miller’s Notes on Assaying |
8vo, | 3 00 | ||
Robine and Lenglen’s Cyanide Industry. (Le Clerc.) |
8vo, | |||
Ulke’s Modern Electrolytic Copper Refining |
8vo, | 3 00 | ||
Wilson’s Cyanide Processes |
12mo, | 1 50 | ||
Chlorination Process |
12mo, | 1 50 | ||
| ASTRONOMY. | ||||
Comstock’s Field Astronomy for Engineers |
8vo, | 2 50 | ||
Craig’s Azimuth |
4to, | 3 50 | ||
Doolittle’s Treatise on Practical Astronomy |
8vo, | 4 00 | ||
Gore’s Elements of Geodesy |
8vo, | 2 50 | ||
Hayford’s Text-book of Geodetic Astronomy |
8vo, | 3 00 | ||
Merriman’s Elements of Precise Surveying and Geodesy |
8vo, | 2 50 | ||
* Michie and Harlow’s Practical Astronomy |
8vo, | 3 00 | ||
* White’s Elements of Theoretical and Descriptive Astronomy |
12mo, | 2 00 | ||
| BOTANY. | ||||
Davenport’s Statistical Methods, with Special Reference to Biological Variation |
16mo, morocco, | 1 25 | ||
Thomé and Bennett’s Structural and Physiological Botany |
16mo, | 2 25 | ||
Westermaier’s Compendium of General Botany. (Schneider.) |
8vo, | 2 00 | ||
| CHEMISTRY. | ||||
Adriance’s Laboratory Calculations and Specific Gravity Tables |
12mo, | 1 25 | ||
Allen’s Tables for Iron Analysis |
8vo, | 3 00 | ||
Arnold’s Compendium of Chemistry. (Mandel |
Small 8vo, | 3 50 | ||
Austen’s Notes for Chemical Students |
12mo, | 1 50 | ||
Bernadou’s Smokeless Powder.—Nitro-cellulose, and Theory of the Cellulose Molecule |
12mo, | 2 50 | ||
* Browning’s Introduction to the Rarer Elements |
8vo, | 1 50 | ||
| [Pg 4]
Brush and Penfield’s Manual of Determinative Mineralogy |
8vo, | 4 00 | ||
Classen’s Quantitative Chemical Analysis by Electrolysis. (Boltwood.) |
8vo, | 3 00 | ||
Cohn’s Indicators and Test-papers |
12mo, | 2 00 | ||
Tests and Reagents |
8vo, | 3 00 | ||
Crafts’s Short Course in Qualitative Chemical Analysis. (Schaeffer.) |
12mo, | 1 50 | ||
Dolezalek’s Theory of the Lead Accumulator (Storage Battery). (Von Ende.) |
12mo, | 2 50 | ||
Drechsel’s Chemical Reactions. (Merrill.) |
12mo, | 1 25 | ||
Duhem’s Thermodynamics and Chemistry. (Burgess.) |
8vo, | 4 00 | ||
Eissler’s Modern High Explosives |
8vo, | 4 00 | ||
Effront’s Enzymes and their Applications. (Prescott.) |
8vo, | 3 00 | ||
Erdmann’s Introduction to Chemical Preparations. (Dunlap.) |
12mo, | 1 25 | ||
Fletcher’s Practical Instructions in Quantitative Assaying with the Blowpipe. |
12mo, morocco, | 1 50 | ||
Fowler’s Sewage Works Analyses |
12mo, | 2 00 | ||
Fresenius’s Manual of Qualitative Chemical Analysis. (Wells.) |
8vo, | 5 00 | ||
Manual of Qualitative Chemical Analysis. Part I. Descriptive. (Wells.) |
8vo, | 3 00 | ||
System of Instruction in Quantitative Chemical Analysis. (Cohn.) 2 vols |
8vo, | 12 50 | ||
Fuertes’s Water and Public Health |
12mo, | 1 50 | ||
Furman’s Manual of Practical Assaying |
8vo, | 3 00 | ||
* Getman’s Exercises in Physical Chemistry |
12mo, | 2 00 | ||
Gill’s Gas and Fuel Analysis for Engineers |
12mo, | 1 25 | ||
Grotenfelt’s Principles of Modern Dairy Practice. (Woll.) |
12mo, | 2 00 | ||
Hammarsten’s Text-book of Physiological Chemistry. (Mandel.) |
8vo, | 4 00 | ||
Helm’s Principles of Mathematical Chemistry. (Morgan.) |
12mo, | 1 50 | ||
Hering’s Ready Reference Tables (Conversion Factors) |
16mo, morocco, | 2 50 | ||
Hind’s Inorganic Chemistry |
8vo, | 3 00 | ||
| * | Laboratory Manual for Students |
12mo, | 1 00 | |
Holleman’s Text-book of Inorganic Chemistry. (Cooper.) |
8vo, | 2 50 | ||
Text-book of Organic Chemistry. (Walker and Mott.) |
8vo, | 2 50 | ||
| * | Laboratory Manual of Organic Chemistry. (Walker.) |
12mo, | 1 00 | |
Hopkins’s Oil-chemists’ Handbook |
8vo, | 3 00 | ||
Jackson’s Directions for Laboratory Work in Physiological Chemistry |
8vo, | 1 25 | ||
Keep’s Cast Iron |
8vo, | 2 50 | ||
Ladd’s Manual of Quantitative Chemical Analysis |
12mo, | 1 00 | ||
Landauer’s Spectrum Analysis. (Tingle.) |
8vo, | 3 00 | ||
* Langworthy and Austen. The Occurrence of Aluminium in Vegetable Products, Animal Products, and Natural Waters |
8vo, | 2 00 | ||
Lassar-Cohn’s Practical Urinary Analysis. (Lorenz.) |
12mo, | 1 00 | ||
Application of Some General Reactions to Investigations in Organic Chemistry. (Tingle.) |
12mo, | 1 00 | ||
Leach’s The Inspection and Analysis of Food with Special Reference to State Control |
8vo, | 7 50 | ||
Löb’s Electrochemistry of Organic Compounds. (Lorenz.) |
8vo, | 3 00 | ||
Lodge’s Notes on Assaying and Metallurgical Laboratory Experiments |
8vo, | 3 00 | ||
Low’s Technical Method of Ore Analysis |
8vo, | 3 00 | ||
Lunge’s Techno-chemical Analysis. (Cohn.) |
12mo, | 1 00 | ||
Mandel’s Handbook for Bio-chemical Laboratory |
12mo, | 1 50 | ||
* Martin’s Laboratory Guide to Qualitative Analysis with the Blowpipe |
12mo, | 60 | ||
Mason’s Water-supply. (Considered Principally from a Sanitary Standpoint.) 3d Edition, Rewritten |
8vo, | 4 00 | ||
Examination of Water. (Chemical and Bacteriological.) |
12mo, | 1 25 | ||
Matthew’s The Textile Fibres |
8vo, | 3 50 | ||
Meyer’s Determination of Radicles in Carbon Compounds. (Tingle.) |
12mo, | 1 00 | ||
Miller’s Manual of Assaying |
12mo, | 1 00 | ||
Minet’s Production of Aluminum and its Industrial Use. (Waldo.) |
12mo, | 2 50 | ||
Mixter’s Elementary Text-book of Chemistry |
12mo, | 1 50 | ||
Morgan’s Elements of Physical Chemistry |
12mo, | 3 00 | ||
* Physical Chemistry for Electrical Engineers |
12mo, | 1 50 | ||
| [Pg 5] Morse’s Calculations used in Cane-sugar Factories |
16mo, morocco, | 1 50 | ||
Mulliken’s General Method for the Identification of Pure Organic Compounds. Vol. I. |
Large 8vo, | 5 00 | ||
O’Brine’s Laboratory Guide in Chemical Analysis |
8vo, | 2 00 | ||
O’Driscoll’s Notes on the Treatment of Gold Ores |
8vo, | 2 00 | ||
Ostwald’s Conversations on Chemistry. Part One. (Ramsey.) |
12mo, | 1 50 | ||
Ostwald’s Conversations on Chemistry. Part Two. (Turnbull.) |
12mo, 2 00 | |||
* Penfield’s Notes on Determinative Mineralogy and Record of Mineral Tests |
8vo, paper, | 50 | ||
Pictet’s The Alkaloids and their Chemical Constitution. (Biddle.) |
8vo, | 5 00 | ||
Pinner’s Introduction to Organic Chemistry. (Austen.) |
12mo, | 1 50 | ||
Poole’s Calorific Power of Fuels |
8vo, | 3 00 | ||
Prescott and Winslow’s Elements of Water Bacteriology, with Special Reference to Sanitary Water Analysis |
12mo, | 1 25 | ||
* Reisig’s Guide to Piece-dyeing |
8vo, | 25 00 | ||
Richards and Woodman’s Air, Water, and Food from a Sanitary Standpoint |
8vo, | 2 00 | ||
Richards’s Cost of Living as Modified by Sanitary Science |
12mo, | 1 00 | ||
Cost of Food, a Study in Dietaries |
12mo, | 1 00 | ||
* Richards and Williams’s The Dietary Computer |
8vo, | 1 50 | ||
Ricketts and Russell’s Skeleton Notes upon Inorganic Chemistry. (Part I. Non-metallic Elements.) |
8vo, morocco, | 75 | ||
Ricketts and Miller’s Notes on Assaying |
8vo, | 3 00 | ||
Rideal’s Sewage and the Bacterial Purification of Sewage |
8vo, | 3 50 | ||
Disinfection and the Preservation of Food |
8vo, | 4 00 | ||
Rigg’s Elementary Manual for the Chemical Laboratory |
8vo, | 1 25 | ||
Robine and Lenglen’s Cyanide Industry. (Le Clerc.) |
8vo, | |||
Rostoski’s Serum Diagnosis. (Bolduan.) |
12mo, | 1 00 | ||
Ruddiman’s Incompatibilities in Prescriptions |
8vo, | 2 00 | ||
| * | Whys in Pharmacy |
12mo, | 1 00 | |
Sabin’s Industrial and Artistic Technology of Paints and Varnish |
8vo, | 3 00 | ||
Salkowski’s Physiological and Pathological Chemistry. (Orndorff.) |
8vo, | 2 50 | ||
Schimpf’s Text-book of Volumetric Analysis |
12mo, | 2 50 | ||
Essentials of Volumetric Analysis |
12mo, | 1 25 | ||
| * | Qualitative Chemical Analysis |
8vo, | 1 25 | |
Spencer’s Handbook for Chemists of Beet-sugar Houses |
16mo, morocco, | 3 00 | ||
Handbook for Cane Sugar Manufacturers |
16mo, morocco, | 3 00 | ||
Stockbridge’s Rocks and Soils |
8vo, | 2 50 | ||
* Tillman’s Elementary Lessons in Heat |
8vo, | 1 50 | ||
| * | Descriptive General Chemistry |
8vo, | 3 00 | |
Treadwell’s Qualitative Analysis. (Hall.) |
8vo, | 3 00 | ||
Quantitative Analysis. (Hall.) |
8vo, | 4 00 | ||
Turneaure and Russell’s Public Water-supplies |
8vo, | 5 00 | ||
Van Deventer’s Physical Chemistry for Beginners. (Boltwood.) |
12mo, | 1 50 | ||
* Walke’s Lectures on Explosives |
8vo, | 4 00 | ||
Ware’s Beet-sugar Manufacture and Refining |
Small 8vo, cloth, | 4 00 | ||
Washington’s Manual of the Chemical Analysis of Rocks |
8vo, | 2 00 | ||
Wassermann’s Immune Sera: Hæmolysins, Cytotoxins, and Precipitins. (Bolduan.) |
12mo, | 1 00 | ||
Well’s Laboratory Guide in Qualitative Chemical Analysis |
8vo, | 1 50 | ||
Short Course in Inorganic Qualitative Chemical Analysis for Engineering Students |
12mo, | 1 50 | ||
Text-book of Chemical Arithmetic |
12mo, | 1 25 | ||
Whipple’s Microscopy of Drinking-water |
8vo, | 3 50 | ||
Wilson’s Cyanide Processes |
12mo, | 1 50 | ||
Chlorination Process |
12mo, | 1 50 | ||
Winton’s Microscopy of Vegetable Foods |
8vo, | 7 50 | ||
Wulling’s Elementary Course in Inorganic, Pharmaceutical, and Medical Chemistry |
12mo, | 2 00 | ||
| [Pg 6]CIVIL ENGINEERING. | ||||
| BRIDGES AND ROOFS. HYDRAULICS. MATERIALS OF ENGINEERING. RAILWAY ENGINEERING. |
||||
Baker’s Engineers’ Surveying Instruments |
12mo, | 3 00 | ||
Bixby’s Graphical Computing Table |
Paper 191⁄2 × 241⁄4 inches. | 25 | ||
** Burr’s Ancient and Modern Engineering and the Isthmian Canal. (Postage, 27 cents additional.) |
8vo, | 3 50 | ||
Comstock’s Field Astronomy for Engineers |
8vo, | 2 50 | ||
Davis’s Elevation and Stadia Tables |
8vo, | 1 00 | ||
Elliott’s Engineering for Land Drainage |
12mo, | 1 50 | ||
Practical Farm Drainage |
12mo, | 1 00 | ||
* Fiebeger’s Treatise on Civil Engineering |
8vo, | 5 00 | ||
Folwell’s Sewerage. (Designing and Maintenance.) |
8vo, | 3 00 | ||
Freitag’s Architectural Engineering. 2d Edition, Rewritten |
8vo, | 3 50 | ||
French and Ives’s Stereotomy |
8vo, | 2 50 | ||
Goodhue’s Municipal Improvements |
12mo, | 1 75 | ||
Goodrich’s Economic Disposal of Towns’ Refuse |
8vo, | 3 50 | ||
Gore’s Elements of Geodesy |
8vo, | 2 50 | ||
Hayford’s Text-book of Geodetic Astronomy |
8vo, | 3 00 | ||
Hering’s Ready Reference Tables (Conversion Factors) |
16mo, morocco, | 2 50 | ||
Howe’s Retaining Walls for Earth |
12mo, | 1 25 | ||
Johnson’s (J. B.) Theory and Practice of Surveying |
Small 8vo, | 4 00 | ||
Johnson’s (L. J.) Statics by Algebraic and Graphic Methods |
8vo, | 2 00 | ||
Laplace’s Philosophical Essay on Probabilities. (Truscott and Emory.) |
12mo, | 2 00 | ||
Mahan’s Treatise on Civil Engineering. (1873.) (Wood.) |
8vo, | 5 00 | ||
| * | Descriptive Geometry |
8vo, | 1 50 | |
Merriman’s Elements of Precise Surveying and Geodesy |
8vo, | 2 50 | ||
Merriman and Brooks’s Handbook for Surveyors |
16mo, morocco, | 2 00 | ||
Nugent’s Plane Surveying |
8vo, | 3 50 | ||
Ogden’s Sewer Design |
12mo, | 2 00 | ||
Patton’s Treatise on Civil Engineering |
8vo half leather, | 7 50 | ||
Reed’s Topographical Drawing and Sketching |
4to, | 5 00 | ||
Rideal’s Sewage and the Bacterial Purification of Sewage |
8vo, | 3 50 | ||
Siebert and Biggin’s Modern Stone-cutting and Masonry |
8vo, | 1 50 | ||
Smith’s Manual of Topographical Drawing. (McMillan.) |
8vo, | 2 50 | ||
Sondericker’s Graphic Statics, with Applications to Trusses, Beams, and Arches. |
8vo, | 2 00 | ||
Taylor and Thompson’s Treatise on Concrete, Plain and Reinforced |
8vo, | 5 00 | ||
* Trautwine’s Civil Engineer’s Pocket-book |
16mo, morocco, | 5 00 | ||
Wait’s Engineering and Architectural Jurisprudence |
8vo, | 6 00 | ||
| Sheep, | 6 50 | |||
Law of Operations Preliminary to Construction in Engineering and Architecture |
8vo, | 5 00 | ||
| Sheep, | 5 50 | |||
Law of Contracts |
8vo, | 3 00 | ||
Warren’s Stereotomy—Problems in Stone-cutting |
8vo, | 2 50 | ||
Webb’s Problems in the Use and Adjustment of Engineering Instruments. |
16mo, morocco, | 1 25 | ||
Wilson’s Topographic Surveying |
8vo, | 3 50 | ||
| BRIDGES AND ROOFS. | ||||
Boller’s Practical Treatise on the Construction of Iron Highway Bridges |
8vo, | 2 00 | ||
| * | Thames River Bridge |
4to, paper, | 5 00 | |
Burr’s Course on the Stresses in Bridges and Roof Trusses, Arched Ribs, and Suspension Bridges |
8vo, | 3 50 | ||
| [Pg 7]
Burr and Falk’s Influence Lines for Bridge and Roof Computations |
8vo, | 3 00 | ||
Design and Construction of Metallic Bridges |
8vo, | 5 00 | ||
Du Bois’s Mechanics of Engineering. Vol. II. |
Small 4to, | 10 00 | ||
Foster’s Treatise on Wooden Trestle Bridges |
4to, | 5 00 | ||
Fowler’s Ordinary Foundations |
8vo, | 3 50 | ||
Greene’s Roof Trusses |
8vo, | 1 25 | ||
Bridge Trusses |
8vo, | 2 50 | ||
Arches in Wood, Iron, and Stone |
8vo, | 2 50 | ||
Howe’s Treatise on Arches |
8vo, | 4 00 | ||
Design of Simple Roof-trusses in Wood and Steel |
8vo, | 2 00 | ||
Johnson, Bryan, and Turneaure’s Theory and Practice in the Designing of Modern Framed Structures |
Small 4to, | 10 00 | ||
Merriman and Jacoby’s Text-book on Roofs and Bridges: |
||||
Part I. Stresses in Simple Trusses |
8vo, | 2 50 | ||
Part II. Graphic Statics |
8vo, | 2 50 | ||
Part III. Bridge Design |
8vo, | 2 50 | ||
Part IV. Higher Structures |
8vo, | 2 50 | ||
Morison’s Memphis Bridge |
4to, | 10 00 | ||
Waddell’s de Pontibus, a Pocket-book for Bridge Engineers |
16mo, morocco, | 2 00 | ||
Specifications for Steel Bridges |
12mo, | 1 25 | ||
Wright’s Designing of Draw-spans. Two Parts in one volume |
8vo, | 3 50 | ||
| HYDRAULICS. | ||||
Bazin’s Experiments upon the Contraction of the Liquid Vein Issuing from an Orifice. (Trautwine.) |
8vo, | 2 00 | ||
Bovey’s Treatise on Hydraulics |
8vo, | 5 00 | ||
Church’s Mechanics of Engineering |
8vo, | 6 00 | ||
Diagrams of Mean Velocity of Water in Open Channels |
paper, | 1 50 | ||
Hydraulic Motors |
8vo, | 2 00 | ||
Coffin’s Graphical Solution of Hydraulic Problems |
16mo, morocco, | 2 50 | ||
Flather’s Dynamometers, and the Measurement of Power |
12mo, | 3 00 | ||
Folwell’s Water-supply Engineering |
8vo, | 4 00 | ||
Frizell’s Water-power |
8vo, | 5 00 | ||
Fuertes’s Water and Public Health |
12mo, | 1 50 | ||
Water-filtration Works |
12mo, | 2 50 | ||
Ganguillet and Kutter’s General Formula for the Uniform Flow of Water in Rivers and Other Channels. (Hering and Trautwine.) |
8vo, | 4 00 | ||
Hazen’s Filtration of Public Water-supply |
8vo, | 3 00 | ||
Hazlehurst’s Towers and Tanks for Water-works |
8vo, | 2 50 | ||
Herschel’s 115 Experiments on the Carrying Capacity of Large, Riveted, Metal Conduits |
8vo, | 2 00 | ||
Mason’s Water-supply. (Considered Principally From a Sanitary Standpoint.) |
8vo, | 4 00 | ||
Merriman’s Treatise on Hydraulics |
8vo, | 5 00 | ||
* Michie’s Elements of Analytical Mechanics |
8vo, | 4 00 | ||
Schuyler’s Reservoirs for Irrigation, Water-power, and Domestic Water-supply |
Large 8vo, | 5 00 | ||
** Thomas and Watt’s Improvement of Rivers. |
(Post., 44c. additional.) 4to, | 6 00 | ||
Turneaure and Russell’s Public Water-supplies |
8vo, | 5 00 | ||
Wegmann’s Design and Construction of Dams |
4to, | 5 00 | ||
Water-supply of the City of New York From 1658 to 1895 |
4to, | 10 00 | ||
Williams and Hazen’s Hydraulic Tables |
8vo, | 1 50 | ||
Wilson’s Irrigation Engineering |
Small 8vo, | 4 00 | ||
Wolff’s Windmill as a Prime Mover |
8vo, | 3 00 | ||
Wood’s Turbines |
8vo, | 2 50 | ||
Elements of Analytical Mechanics |
8vo, | 3 00 | ||
| [Pg 8]MATERIALS OF ENGINEERING. | ||||
Baker’s Treatise on Masonry Construction |
8vo, | 5 00 | ||
Roads and Pavements |
8vo, | 5 00 | ||
Black’s United States Public Works |
Oblong 4to, | 5 00 | ||
* Bovey’s Strength of Materials and Theory of Structures |
8vo, | 7 50 | ||
Burr’s Elasticity and Resistance of the Materials of Engineering |
8vo, | 7 50 | ||
Byrne’s Highway Construction |
8vo, | 5 00 | ||
Inspection of the Materials and Workmanship Employed in Construction. |
16mo, | 3 00 | ||
Church’s Mechanics of Engineering |
8vo, | 6 00 | ||
Du Bois’s Mechanics of Engineering. Vol. I. |
Small 4to, | 7 50 | ||
* Eckel’s Cements, Limes, and Plasters |
8vo, | 6 00 | ||
Johnson’s Materials of Construction |
Large 8vo, | 6 00 | ||
Fowler’s Ordinary Foundations |
8vo, | 3 50 | ||
* Greene’s Structural Mechanics |
8vo, | 2 50 | ||
Keep’s Cast Iron |
8vo, | 2 50 | ||
Lanza’s Applied Mechanics |
8vo, | 7 50 | ||
Marten’s Handbook on Testing Materials. (Henning.) 2 vols. |
8vo, | 7 50 | ||
Maurer’s Technical Mechanics |
8vo, | 4 00 | ||
Merrill’s Stones for Building and Decoration |
8vo, | 5 00 | ||
Merriman’s Mechanics of Materials |
8vo, | 5 00 | ||
Strength of Materials |
12mo, | 1 00 | ||
Metcalf’s Steel. A Manual for Steel-users |
12mo, | 2 00 | ||
Patton’s Practical Treatise on Foundations |
8vo, | 5 00 | ||
Richardson’s Modern Asphalt Pavements |
8vo, | 3 00 | ||
Richey’s Handbook for Superintendents of Construction |
16mo, mor., | 4 00 | ||
Rockwell’s Roads and Pavements in France |
12mo, | 1 25 | ||
Sabin’s Industrial and Artistic Technology of Paints and Varnish |
8vo, | 3 00 | ||
Smith’s Materials of Machines |
12mo, | 1 00 | ||
Snow’s Principal Species of Wood |
8vo, | 3 50 | ||
Spalding’s Hydraulic Cement |
12mo, | 2 00 | ||
Text-book on Roads and Pavements |
12mo, | 2 00 | ||
Taylor and Thompson’s Treatise on Concrete, Plain and Reinforced |
8vo, | 5 00 | ||
Thurston’s Materials of Engineering. 3 Parts |
8vo, | 8 00 | ||
Part I. Non-metallic Materials of Engineering and Metallurgy |
8vo, | 2 00 | ||
Part II. Iron and Steel |
8vo, | 3 50 | ||
Part III. A Treatise on Brasses, Bronzes, and Other Alloys and their Constituents |
8vo, | 2 50 | ||
Thurston’s Text-book of the Materials of Construction |
8vo, | 5 00 | ||
Tillson’s Street Pavements and Paving Materials |
8vo, | 4 00 | ||
Waddell’s De Pontibus. (A Pocket-book for Bridge Engineers.) |
16mo, mor., | 2 00 | ||
Specifications for Steel Bridges |
12mo, | 1 25 | ||
Wood’s (De V.) Treatise on the Resistance of Materials, and an Appendix on the Preservation of Timber |
8vo, | 2 00 | ||
Wood’s (De V.) Elements of Analytical Mechanics |
8vo, | 3 00 | ||
Wood’s (M. P.) Rustless Coatings: Corrosion and Electrolysis of Iron and Steel |
8vo, | 4 00 | ||
| RAILWAY ENGINEERING. | ||||
Andrew’s Handbook for Street Railway Engineers |
3×5 inches, morocco, | 1 25 | ||
Berg’s Buildings and Structures of American Railroads |
4to, | 5 00 | ||
Brook’s Handbook of Street Railroad Location |
16mo, morocco, | 1 50 | ||
Butt’s Civil Engineer’s Field-book |
16mo, morocco, | 2 50 | ||
Crandall’s Transition Curve |
16mo, morocco, | 1 50 | ||
Railway and Other Earthwork Tables |
8vo, | 1 50 | ||
Dawson’s “Engineering” and Electric Traction Pocket-book |
16mo, morocco, | 5 00 | ||
| [Pg 9] Dredge’s History of the Pennsylvania Railroad: (1879) |
Paper, | 5 00 | ||
* Drinker’s Tunnelling, Explosive Compounds, and Rock Drills |
4to, half mor., | 25 00 | ||
Fisher’s Table of Cubic Yards |
Cardboard, | 25 | ||
Godwin’s Railroad Engineers’ Field-book and Explorers’ Guide |
16mo, mor., | 2 50 | ||
Howard’s Transition Curve Field-book |
16mo, morocco, | 1 50 | ||
Hudson’s Tables for Calculating the Cubic Contents of Excavations and Embankments |
8vo, | 1 00 | ||
Molitor and Beard’s Manual for Resident Engineers |
16mo, | 1 00 | ||
Nagle’s Field Manual for Railroad Engineers |
16mo, morocco, | 3 00 | ||
Philbrick’s Field Manual for Engineers |
16mo, morocco, | 3 00 | ||
Searles’s Field Engineering |
16mo, morocco, | 3 00 | ||
Railroad Spiral |
16mo, morocco, | 1 50 | ||
Taylor’s Prismoidal Formulæ and Earthwork |
8vo, | 1 50 | ||
* Trautwine’s Method of Calculating the Cube Contents of Excavations and Embankments by the Aid of Diagrams |
8vo, | 2 00 | ||
The Field Practice of Laying Out Circular Curves for Railroads. |
12mo, morocco, | 2 50 | ||
Cross-section Sheet |
Paper, | 25 | ||
Webb’s Railroad Construction |
16mo, morocco, | 5 00 | ||
Wellington’s Economic Theory of the Location of Railways |
Small 8vo, | 5 00 | ||
| DRAWING. | ||||
Barr’s Kinematics of Machinery |
8vo, | 2 50 | ||
* Bartlett’s Mechanical Drawing |
8vo, | 3 00 | ||
* Bartlett’s Mechanical Drawing Abridged Ed. |
8vo, | 1 50 | ||
Coolidge’s Manual of Drawing |
8vo, paper, | 1 00 | ||
Coolidge and Freeman’s Elements of General Drafting for Mechanical Engineers |
Oblong 4to, | 2 50 | ||
Durley’s Kinematics of Machines |
8vo, | 4 00 | ||
Emch’s Introduction to Projective Geometry and its Applications |
8vo, | 2 50 | ||
Hill’s Text-book on Shades and Shadows, and Perspective |
8vo, | 2 00 | ||
Jamison’s Elements of Mechanical Drawing |
8vo, | 2 50 | ||
Advanced Mechanical Drawing |
8vo, | 2 00 | ||
Jones’s Machine Design: |
||||
Part I. Kinematics of Machinery |
8vo, | 1 50 | ||
Part II. Form, Strength, and Proportions of Parts |
8vo, | 3 00 | ||
MacCord’s Elements of Descriptive Geometry |
8vo, | 3 00 | ||
Kinematics; or, Practical Mechanism |
8vo, | 5 00 | ||
Mechanical Drawing |
4to, | 4 00 | ||
Velocity Diagrams |
8vo, | 1 50 | ||
MacLeod’s Descriptive Geometry |
Small 8vo, | 1 50 | ||
* Mahan’s Descriptive Geometry and Stone-cutting |
8vo, | 1 50 | ||
Industrial Drawing. (Thompson.) |
8vo, | 3 50 | ||
Moyer’s Descriptive Geometry |
8vo, | 2 00 | ||
Reed’s Topographical Drawing and Sketching |
4to, | 5 00 | ||
Reid’s Course in Mechanical Drawing |
8vo, | 2 00 | ||
Text-book of Mechanical Drawing and Elementary Machine Design |
8vo, | 3 00 | ||
Robinson’s Principles of Mechanism |
8vo, | 3 00 | ||
Schwamb and Merrill’s Elements of Mechanism |
8vo, | 3 00 | ||
Smith’s (R. S.) Manual of Topographical Drawing. (McMillan.) |
8vo, | 2 50 | ||
Smith (A. W.) and Marx’s Machine Design |
8vo, | 3 00 | ||
Warren’s Elements of Plane and Solid Free-hand Geometrical Drawing |
12mo, | 1 00 | ||
Drafting Instruments and Operations |
12mo, | 1 25 | ||
Manual of Elementary Projection Drawing |
12mo, | 1 50 | ||
Manual of Elementary Problems in the Linear Perspective of Form and Shadow |
12mo, | 1 00 | ||
Plane Problems in Elementary Geometry |
12mo, | 1 25 | ||
| [Pg 10] Warren’s Primary Geometry |
12mo, | 75 | ||
Elements of Descriptive Geometry, Shadows, and Perspective |
8vo, | 3 50 | ||
General Problems of Shades and Shadows |
8vo, | 3 00 | ||
Elements of Machine Construction and Drawing |
8vo, | 7 50 | ||
Problems, Theorems, and Examples in Descriptive Geometry |
8vo, | 2 50 | ||
Weisbach’s Kinematics and Power of Transmission. (Hermann and Klein.) |
8vo, | 5 00 | ||
Whelpley’s Practical Instruction in the Art of Letter Engraving |
12mo, | 2 00 | ||
Wilson’s (H. M.) Topographic Surveying |
8vo, | 3 50 | ||
Wilson’s (V. T.) Free-hand Perspective |
8vo, | 2 50 | ||
Wilson’s (V. T.) Free-hand Lettering |
8vo, | 1 00 | ||
Woolf’s Elementary Course in Descriptive Geometry |
Large 8vo, | 3 00 | ||
| ELECTRICITY AND PHYSICS. | ||||
Anthony and Brackett’s Text-book of Physics. (Magie.) |
Small 8vo, | 3 00 | ||
Anthony’s Lecture-notes on the Theory of Electrical Measurements |
12mo, | 1 00 | ||
Benjamin’s History of Electricity |
8vo, | 3 00 | ||
Voltaic Cell |
8vo, | 3 00 | ||
Classen’s Quantitative Chemical Analysis by Electrolysis. (Boltwood.) |
8vo, | 3 00 | ||
Crehore and Squier’s Polarizing Photo-chronograph |
8vo, | 3 00 | ||
Dawson’s “Engineering” and Electric Traction Pocket-book |
16mo, morocco, | 5 00 | ||
Dolezalek’s Theory of the Lead Accumulator (Storage Battery). (Von Ende.) |
12mo, | 2 50 | ||
Duhem’s Thermodynamics and Chemistry. (Burgess.) |
8vo, | 4 00 | ||
Flather’s Dynamometers, and the Measurement of Power |
12mo, | 3 00 | ||
Gilbert’s De Magnete. (Mottelay.) |
8vo, | 2 50 | ||
Hanchett’s Alternating Currents Explained |
12mo, | 1 00 | ||
Hering’s Ready Reference Tables (Conversion Factors) |
16mo, morocco, | 2 50 | ||
Holman’s Precision of Measurements |
8vo, | 2 00 | ||
Telescopic Mirror-scale Method, Adjustments, and Tests |
Large 8vo, | 75 | ||
Kinzbrunner’s Testing of Continuous-current Machines |
8vo, | 2 00 | ||
Landauer’s Spectrum Analysis. (Tingle.) |
8vo, | 3 00 | ||
Le Chatelier’s High-temperature Measurements. (Boudouard—Burgess.) |
12mo, | 3 00 | ||
Löb’s Electrochemistry of Organic Compounds. (Lorenz.) |
8vo, | 3 00 | ||
* Lyon’s Treatise on Electromagnetic Phenomena. Vols. I. and II. |
8vo, each, | 6 00 | ||
* Michie’s Elements of Wave Motion Relating to Sound and Light |
8vo, | 4 00 | ||
Niaudet’s Elementary Treatise on Electric Batteries. (Fishback.) |
12mo, | 2 50 | ||
* Rosenberg’s Electrical Engineering. (Haldane Gee—Kinzbrunner.) |
8vo, | 1 50 | ||
Ryan, Norris, and Hoxie’s Electrical Machinery. Vol. I. |
8vo, | 2 50 | ||
Thurston’s Stationary Steam-engines |
8vo, | 2 50 | ||
* Tillman’s Elementary Lessons in Heat |
8vo, | 1 50 | ||
Tory and Pitcher’s Manual of Laboratory Physics |
Small 8vo, | 2 00 | ||
Ulke’s Modern Electrolytic Copper Refining |
8vo, | 3 00 | ||
| LAW. | ||||
* Davis’s Elements of Law |
8vo, | 2 50 | ||
| * | Treatise on the Military Law of United States |
8vo, | 7 00 | |
| * | Sheep, | 7 50 | ||
Manual for Courts-martial |
16mo, morocco, | 1 50 | ||
Wait’s Engineering and Architectural Jurisprudence |
8vo, | 6 00 | ||
| * | Sheep, | 6 50 | ||
Law of Operations Preliminary to Construction in Engineering and Architecture |
8vo, | 5 00 | ||
| Sheep, | 5 50 | |||
Law of Contracts |
8vo, | 3 00 | ||
Winthrop’s Abridgment of Military Law |
12mo, | 2 50 | ||
| [Pg 11]MANUFACTURES. | ||||
Bernadou’s Smokeless Powder—Nitro-cellulose and Theory of the Cellulose Molecule |
12mo, | 2 50 | ||
Bolland’s Iron Founder |
12mo, | 2 50 | ||
“The Iron Founder,” Supplement |
12mo, | 2 50 | ||
Encyclopedia of Founding and Dictionary of Foundry Terms Used in the Practice of Moulding |
12mo, | 3 00 | ||
Eissler’s Modern High Explosives |
8vo, | 4 00 | ||
Effront’s Enzymes and their Applications. (Prescott.) |
8vo, | 3 00 | ||
Fitzgerald’s Boston Machinist |
12mo, | 1 00 | ||
Ford’s Boiler Making for Boiler Makers |
18mo, | 1 00 | ||
Hopkin’s Oil-chemists’ Handbook |
8vo, | 3 00 | ||
Keep’s Cast Iron |
8vo, | 2 50 | ||
Leach’s The Inspection and Analysis of Food with Special Reference to State Control |
Large 8vo, | 7 50 | ||
Matthews’s The Textile Fibres |
8vo, | 3 50 | ||
Metcalf’s Steel. A Manual for Steel-users |
12mo, | 2 00 | ||
Metcalfe’s Cost of Manufactures—And the Administration of Workshops |
8vo, | 5 00 | ||
Meyer’s Modern Locomotive Construction |
4to, | 10 00 | ||
Morse’s Calculations used in Cane-sugar Factories |
16mo, morocco, | 1 50 | ||
* Reisig’s Guide to Piece-dyeing |
8vo, | 25 00 | ||
Sabin’s Industrial and Artistic Technology of Paints and Varnish |
8vo, | 3 00 | ||
Smith’s Press-working of Metals |
8vo, | 3 00 | ||
Spalding’s Hydraulic Cement |
12mo, | 2 00 | ||
Spencer’s Handbook for Chemists of Beet-sugar Houses |
16mo, morocco, | 3 00 | ||
Handbook for Cane Sugar Manufacturers |
16mo, morocco, | 3 00 | ||
Taylor and Thompson’s Treatise on Concrete, Plain and Reinforced |
8vo, | 5 00 | ||
Thurston’s Manual of Steam-boilers, their Designs, Construction and Operation |
8vo, | 5 00 | ||
* Walke’s Lectures on Explosives |
8vo, | 4 00 | ||
Ware’s Beet-sugar Manufacture and Refining |
Small 8vo, | 4 00 | ||
West’s American Foundry Practice |
12mo, | 2 50 | ||
Moulder’s Text-book |
12mo, | 2 50 | ||
Wolff’s Windmill as a Prime Mover |
8vo, | 3 00 | ||
Wood’s Rustless Coatings: Corrosion and Electrolysis of Iron and Steel |
8vo, | 4 00 | ||
| MATHEMATICS. | ||||
Baker’s Elliptic Functions |
8vo, | 1 50 | ||
* Bass’s Elements of Differential Calculus |
12mo, | 4 00 | ||
Briggs’s Elements of Plane Analytic Geometry |
12mo, | 1 00 | ||
Compton’s Manual of Logarithmic Computations |
12mo, | 1 50 | ||
Davis’s Introduction to the Logic of Algebra |
8vo, | 1 50 | ||
* Dickson’s College Algebra |
Large 12mo, | 1 50 | ||
| * | Introduction to the Theory of Algebraic Equations |
Large 12mo, | 1 25 | |
Emch’s Introduction to Projective Geometry and its Applications |
8vo, | 2 50 | ||
Halsted’s Elements of Geometry |
8vo, | 1 75 | ||
Elementary Synthetic Geometry |
8vo, | 1 50 | ||
Rational Geometry |
12mo, | 1 75 | ||
* Johnson’s (J. B.) Three-place Logarithmic Tables: |
Vest-pocket size paper, | 15 | ||
| 100 copies for | 5 00 | |||
| * | Mounted on heavy cardboard, 8 × 10 inches, | 25 | ||
| 10 copies for | 2 00 | |||
Johnson’s (W. W.) Elementary Treatise on Differential Calculus |
Small 8vo, | 3 00 | ||
| [Pg 12] Johnson’s (W. W.) Elementary Treatise on the Integral Calculus |
Small 8vo, | 1 50 | ||
Johnson’s (W. W.) Curve Tracing in Cartesian Co-ordinates |
12mo, | 1 00 | ||
Johnson’s (W. W.) Treatise on Ordinary and Partial Differential Equations |
Small 8vo, | 3 50 | ||
Johnson’s (W. W.) Theory of Errors and the Method of Least Squares |
12mo, | 1 50 | ||
* Johnson’s (W. W.) Theoretical Mechanics |
12mo, | 3 00 | ||
Laplace’s Philosophical Essay on Probabilities. (Truscott and Emory.) |
12mo, | 2 00 | ||
* Ludlow and Bass. Elements of Trigonometry and Logarithmic and Other Tables |
8vo, | 3 00 | ||
Trigonometry and Tables published separately |
Each, | 2 00 | ||
* Ludlow’s Logarithmic and Trigonometric Tables |
8vo, | 1 00 | ||
Mathematical Monographs. Edited by Mansfield Merriman and Robert S. Woodward |
Octavo, | each 1 00 | ||
| No. 1. History of Modern Mathematics, by David Eugene Smith. No. 2. Synthetic Projective Geometry, by George Bruce Halsted. No. 3. Determinants, by Laenas Gifford Weld. No. 4. Hyperbolic Functions, by James McMahon. No. 5. Harmonic Functions, by William E. Byerly. No. 6. Grassmann’s Space Analysis, by Edward W. Hyde. No. 7. Probability and Theory of Errors, by Robert S. Woodward. No. 8. Vector Analysis and Quaternions, by Alexander Macfarlane. No. 9. Differential Equations, by William Woolsey Johnson. No. 10. The Solution of Equations, by Mansfield Merriman. No. 11. Functions of a Complex Variable, by Thomas S. Fiske. | ||||
Maurer’s Technical Mechanics |
8vo, | 4 00 | ||
Merriman and Woodward’s Higher Mathematics |
8vo, | 5 00 | ||
Merriman’s Method of Least Squares |
8vo, | 2 00 | ||
Rice and Johnson’s Elementary Treatise on the Differential Calculus |
Sm. 8vo, | 3 00 | ||
Differential and Integral Calculus. 2 vols. in one |
Small 8vo, | 2 50 | ||
Wood’s Elements of Co-ordinate Geometry |
8vo, | 2 00 | ||
Trigonometry: Analytical, Plane, and Spherical |
12mo, | 1 00 | ||
| MECHANICAL ENGINEERING. | ||||
| MATERIALS OF ENGINEERING, STEAM-ENGINES AND BOILERS. | ||||
Bacon’s Forge Practice |
12mo, | 1 50 | ||
Baldwin’s Steam Heating for Buildings |
12mo, | 2 50 | ||
Barr’s Kinematics of Machinery |
8vo, | 2 50 | ||
* Bartlett’s Mechanical Drawing |
8vo, | 3 00 | ||
* Bartlett’s Mechanical Drawing Abridged Ed |
8vo, | 1 50 | ||
Benjamin’s Wrinkles and Recipes |
12mo, | 2 00 | ||
Carpenter’s Experimental Engineering |
8vo, | 6 00 | ||
Heating and Ventilating Buildings |
8vo, | 4 00 | ||
Cary’s Smoke Suppression in Plants using Bituminous Coal. (In Preparation.) |
||||
Clerk’s Gas and Oil Engine |
Small 8vo, | 4 00 | ||
Coolidge’s Manual of Drawing |
8vo, paper, | 1 00 | ||
Coolidge and Freeman’s Elements of General Drafting for Mechanical Engineers |
Oblong 4to, | 2 50 | ||
Cromwell’s Treatise on Toothed Gearing |
12mo, | 1 50 | ||
Treatise on Belts and Pulleys |
12mo, | 1 50 | ||
Durley’s Kinematics of Machines |
8vo, | 4 00 | ||
Flather’s Dynamometers and the Measurement of Power |
12mo, | 3 00 | ||
Rope Driving |
12mo, | 2 00 | ||
Gill’s Gas and Fuel Analysis for Engineers |
12mo, | 1 25 | ||
Hall’s Car Lubrication |
12mo, | 1 00 | ||
Hering’s Ready Reference Tables (Conversion Factors) |
16mo, morocco, | 2 50 | ||
| [Pg 13] Hutton’s The Gas Engine |
8vo, | 5 00 | ||
Jamison’s Mechanical Drawing |
8vo, | 2 50 | ||
| Jones’s Machine Design: | ||||
Part I. Kinematics of Machinery |
8vo, | 1 50 | ||
Part II. Form, Strength, and Proportions of Parts |
8vo, | 3 00 | ||
Kent’s Mechanical Engineers’ Pocket-book |
16mo, morocco, | 5 00 | ||
Kerr’s Power and Power Transmission |
8vo, | 2 00 | ||
Leonard’s Machine Shop, Tools, and Methods |
8vo, | 4 00 | ||
* Lorenz’s Modern Refrigerating Machinery. (Pope, Haven, and Dean.) |
8vo, | 4 00 | ||
MacCord’s Kinematics; or, Practical Mechanism |
8vo, | 5 00 | ||
Mechanical Drawing |
4to, | 4 00 | ||
Velocity Diagrams |
8vo, | 1 50 | ||
MacFarland’s Standard Reduction Factors for Gases |
8vo, | 1 50 | ||
Mahan’s Industrial Drawing. (Thompson.) |
8vo, | 3 50 | ||
Poole’s Calorific Power of Fuels |
8vo, | 3 00 | ||
Reid’s Course in Mechanical Drawing |
8vo, | 2 00 | ||
Text-book of Mechanical Drawing and Elementary Machine Design |
8vo, | 3 00 | ||
Richard’s Compressed Air |
12mo, | 1 50 | ||
Robinson’s Principles of Mechanism |
8vo, | 3 00 | ||
Schwamb and Merrill’s Elements of Mechanism |
8vo, | 3 00 | ||
Smith’s (O.) Press-working of Metals |
8vo, | 3 00 | ||
Smith (A. W.) and Marx’s Machine Design |
8vo, | 3 00 | ||
Thurston’s Treatise on Friction and Lost Work in Machinery and Mill Work |
8vo, | 3 00 | ||
Animal as a Machine and Prime Motor, and the Laws of Energetics |
12mo, | 1 00 | ||
Warren’s Elements of Machine Construction and Drawing |
8vo, | 7 50 | ||
Weisbach’s Kinematics and the Power of Transmission. (Herrmann—Klein.) |
8vo, | 5 00 | ||
Machinery of Transmission and Governors. (Herrmann—Klein.) |
8vo, | 5 00 | ||
Wolff’s Windmill as a Prime Mover |
8vo, | 3 00 | ||
Wood’s Turbines |
8vo, | 2 50 | ||
| MATERIALS OF ENGINEERING. | ||||
* Bovey’s Strength of Materials and Theory of Structures |
8vo, | 7 50 | ||
Burr’s Elasticity and Resistance of the Materials of Engineering. 6th Edition. Reset |
8vo, | 7 50 | ||
Church’s Mechanics of Engineering |
8vo, | 6 00 | ||
* Greene’s Structural Mechanics |
8vo, | 2 50 | ||
Johnson’s Materials of Construction |
8vo, | 6 00 | ||
Keep’s Cast Iron |
8vo, | 2 50 | ||
Lanza’s Applied Mechanics |
8vo, | 7 50 | ||
Martens’s Handbook on Testing Materials. (Henning.) |
8vo, | 7 50 | ||
Maurer’s Technical Mechanics |
8vo, | 4 00 | ||
Merriman’s Mechanics of Materials |
8vo, | 5 00 | ||
Strength of Materials |
12mo, | 1 00 | ||
Metcalf’s Steel. A manual for Steel-users |
12mo, | 2 00 | ||
Sabin’s Industrial and Artistic Technology of Paints and Varnish |
8vo, | 3 00 | ||
Smith’s Materials of Machines |
12mo, | 1 00 | ||
Thurston’s Materials of Engineering 3 vols., |
8vo, | 8 00 | ||
Part II. Iron and Steel |
8vo, | 3 50 | ||
Part III. A Treatise on Brasses, Bronzes, and Other Alloys and their Constituents |
8vo, | 2 50 | ||
Text-book of the Materials of Construction |
8vo, | 5 00 | ||
Wood’s (De V.) Treatise on the Resistance of Materials and an Appendix on the Preservation of Timber |
8vo, | 2 00 | ||
| [Pg 14] Wood’s (De V.) Elements of Analytical Mechanics |
8vo, | 3 00 | ||
Wood’s (M. P.) Rustless Coatings: Corrosion and Electrolysis of Iron and Steel |
8vo, | 4 00 | ||
| STEAM-ENGINES AND BOILERS. | ||||
Berry’s Temperature-entropy Diagram |
12mo, | 1 25 | ||
Carnot’s Reflections on the Motive Power of Heat. (Thurston.) |
12mo, | 1 50 | ||
Dawson’s “Engineering” and Electric Traction Pocket-book |
16mo, mor., | 5 00 | ||
Ford’s Boiler Making for Boiler Makers |
18mo, | 1 00 | ||
Goss’s Locomotive Sparks |
8vo, | 2 00 | ||
Hemenway’s Indicator Practice and Steam-engine Economy |
12mo, | 2 00 | ||
Hutton’s Mechanical Engineering of Power Plants |
8vo, | 5 00 | ||
Heat and Heat-engines |
8vo, | 5 00 | ||
Kent’s Steam boiler Economy |
8vo, | 4 00 | ||
Kneass’s Practice and Theory of the Injector |
8vo, | 1 50 | ||
MacCord’s Slide-valves |
8vo, | 2 00 | ||
Meyer’s Modern Locomotive Construction |
4to, | 10 00 | ||
Peabody’s Manual of the Steam-engine Indicator |
12mo, | 1 50 | ||
Tables of the Properties of Saturated Steam and Other Vapors |
8vo, | 1 00 | ||
Thermodynamics of the Steam-engine and Other Heat-engines |
8vo, | 5 00 | ||
Valve-gears for Steam-engines |
8vo, | 2 50 | ||
Peabody and Miller’s Steam-boilers |
8vo, | 4 00 | ||
Pray’s Twenty Years with the Indicator |
Large 8vo, | 2 50 | ||
Pupin’s Thermodynamics of Reversible Cycles in Gases and Saturated Vapors. (Osterberg.) |
12mo, | 1 25 | ||
Reagan’s Locomotives: Simple Compound, and Electric |
12mo, | 2 50 | ||
Rontgen’s Principles of Thermodynamics. (Du Bois.) |
8vo, | 5 00 | ||
Sinclair’s Locomotive Engine Running and Management |
12mo, | 2 00 | ||
Smart’s Handbook of Engineering Laboratory Practice |
12mo, | 2 50 | ||
Snow’s Steam-boiler Practice |
8vo, | 3 00 | ||
Spangler’s Valve-gears |
8vo, | 2 50 | ||
Notes on Thermodynamics |
12mo, | 1 00 | ||
Spangler, Greene, and Marshall’s Elements of Steam-engineering |
8vo, | 3 00 | ||
Thurston’s Handy Tables |
8vo, | 1 50 | ||
Manual of the Steam-engine |
2 vols., 8vo, | 10 00 | ||
Part I. History, Structure, and Theory |
8vo, | 6 00 | ||
Part II. Design, Construction, and Operation |
8vo, | 6 00 | ||
Handbook of Engine and Boiler Trials, and the Use of the Indicator and the Prony Brake |
8vo, | 5 00 | ||
Stationary Steam-engines |
8vo, | 2 50 | ||
Steam-boiler Explosions in Theory and in Practice |
12mo, | 1 50 | ||
Manual of Steam-boilers, their Designs, Construction, and Operation |
8vo, | 5 00 | ||
Weisbach’s Heat, Steam, and Steam-engines. (Du Bois.) |
8vo, | 5 00 | ||
Whitham’s Steam-engine Design |
8vo, | 5 00 | ||
Wilson’s Treatise on Steam-boilers. (Flather.) |
16mo, | 2 50 | ||
Wood’s Thermodynamics, Heat Motors, and Refrigerating Machines |
8vo, | 4 00 | ||
| MECHANICS AND MACHINERY. | ||||
Barr’s Kinematics of Machinery |
8vo, | 2 50 | ||
* Bovey’s Strength of Materials and Theory of Structures |
8vo, | 7 50 | ||
Chase’s The Art of Pattern-making |
12mo, | 2 50 | ||
Church’s Mechanics of Engineering |
8vo, | 6 00 | ||
Notes and Examples in Mechanics |
8vo, | 2 00 | ||
Compton’s First Lessons in Metal-working |
12mo, | 1 50 | ||
Compton and De Groodt’s The Speed Lathe |
12mo, | 1 50 | ||
| [Pg 15] Cromwell’s Treatise on Toothed Gearing |
12mo, | 1 50 | ||
Treatise on Belts and Pulleys |
12mo, | 1 50 | ||
Dana’s Text-book of Elementary Mechanics for Colleges and Schools |
12mo, | 1 50 | ||
Dingey’s Machinery Pattern Making |
12mo, | 2 00 | ||
Dredge’s Record of the Transportation Exhibits Building of the World’s Columbian Exposition of 1893 |
4to half morocco, | 5 00 | ||
Du Bois’s Elementary Principles of Mechanics: |
||||
Vol. I. Kinematics |
8vo, | 3 50 | ||
Vol. II. Statics |
8vo, | 4 00 | ||
Mechanics of Engineering. Vol. I. |
Small 4to, | 7 50 | ||
Vol. II. |
Small 4to, | 10 00 | ||
Durley’s Kinematics of Machines |
8vo, | 4 00 | ||
Fitzgerald’s Boston Machinist |
16mo, | 1 00 | ||
Flather’s Dynamometers, and the Measurement of Power |
12mo, | 3 00 | ||
Rope Driving |
12mo, | 2 00 | ||
Goss’s Locomotive Sparks |
8vo, | 2 00 | ||
* Greene’s Structural Mechanics |
8vo, | 2 50 | ||
Hall’s Car Lubrication |
12mo, | 1 00 | ||
Holly’s Art of Saw Filing |
18mo, | 75 | ||
James’s Kinematics of a Point and the Rational Mechanics of a Particle. |
Small 8vo, | 2 00 | ||
* Johnson’s (W. W.) Theoretical Mechanics |
12mo, | 3 00 | ||
Johnson’s (L. J.) Statics by Graphic and Algebraic Methods |
8vo, | 2 00 | ||
| Jones’s Machine Design: | ||||
Part I. Kinematics of Machinery |
8vo, | 1 50 | ||
Part II. Form, Strength, and Proportions of Parts |
8vo, | 3 00 | ||
Kerr’s Power and Power Transmission |
8vo, | 2 00 | ||
Lanza’s Applied Mechanics |
8vo, | 7 50 | ||
Leonard’s Machine Shop, Tools, and Methods |
8vo, | 4 00 | ||
* Lorenz’s Modern Refrigerating Machinery. (Pope, Haven, and Dean.) |
8vo, | 4 00 | ||
MacCord’s Kinematics; or, Practical Mechanism |
8vo, | 5 00 | ||
Velocity Diagrams |
8vo, | 1 50 | ||
Maurer’s Technical Mechanics |
8vo, | 4 00 | ||
Merriman’s Mechanics of Materials |
8vo, | 5 00 | ||
| * | Elements of Mechanics | 12mo, | 1 00 | |
* Michie’s Elements of Analytical Mechanics |
8vo, | 4 00 | ||
Reagan’s Locomotives: Simple, Compound, and Electric |
12mo, | 2 50 | ||
Reid’s Course in Mechanical Drawing |
8vo, | 2 00 | ||
Text-book of Mechanical Drawing and Elementary Machine Design |
8vo, | 3 00 | ||
Richards’s Compressed Air |
12mo, | 1 50 | ||
Robinson’s Principles of Mechanism |
8vo, | 3 00 | ||
Ryan, Norris, and Hoxie’s Electrical Machinery. Vol. I. |
8vo, | 2 50 | ||
Schwamb and Merrill’s Elements of Mechanism |
8vo, | 3 00 | ||
Sinclair’s Locomotive-engine Running and Management |
12mo, | 2 00 | ||
Smith’s (O.) Press-working of Metals |
8vo, | 3 00 | ||
Smith’s (A. W.) Materials of Machines |
12mo, | 1 00 | ||
Smith (A. W.) and Marx’s Machine Design |
8vo, | 3 00 | ||
Spangler, Greene, and Marshall’s Elements of Steam-engineering |
8vo, | 3 00 | ||
Thurston’s Treatise on Friction and Lost Work in Machinery and Mill Work |
8vo, | 3 00 | ||
Animal as a Machine and Prime Motor, and the Laws of Energetics |
12mo, | 1 00 | ||
Warren’s Elements of Machine Construction and Drawing |
8vo, | 7 50 | ||
Weisbach’s Kinematics and Power of Transmission. (Herrmann—Klein.) |
8vo, | 5 00 | ||
Machinery of Transmission and Governors. (Herrmann—Klein.) |
8vo, | 5 00 | ||
Wood’s Elements of Analytical Mechanics |
8vo, | 3 00 | ||
Principles of Elementary Mechanics |
12mo, | 1 25 | ||
Turbines |
8vo, | 2 50 | ||
The World’s Columbian Exposition of 1893 |
4to, | 1 00 | ||
| [Pg 16]METALLURGY. | ||||
Egleston’s Metallurgy of Silver, Gold, and Mercury: |
||||
Vol. I. Silver |
8vo, | 7 50 | ||
Vol. II. Gold and Mercury |
8vo, | 7 50 | ||
** Iles’s Lead-smelting. (Postage 9 cents additional.) |
12mo, | 2 50 | ||
Keep’s Cast Iron |
8vo, | 2 50 | ||
Kunhardt’s Practice of Ore Dressing in Europe |
8vo, | 1 50 | ||
Le Chatelier’s High-temperature Measurements. (Boudouard—Burgess.) |
12mo, | 3 00 | ||
Metcalf’s Steel. A Manual for Steel-users |
12mo, | 2 00 | ||
Minet’s Production of Aluminum and its Industrial Use. (Waldo.) |
12mo, | 2 50 | ||
Robine and Lenglen’s Cyanide Industry. (Le Clerc.) |
8vo, | |||
Smith’s Materials of Machines |
12mo, | 1 00 | ||
Thurston’s Materials of Engineering. In Three Parts |
8vo, | 8 00 | ||
Part II. Iron and Steel |
8vo, | 3 50 | ||
Part III. A Treatise on Brasses, Bronzes, and Other Alloys and their Constituents |
8vo, | 2 50 | ||
Ulke’s Modern Electrolytic Copper Refining |
8vo, | 3 00 | ||
| MINERALOGY. | ||||
Barringer’s Description of Minerals of Commercial Value. |
Oblong, morocco, | 2 50 | ||
Boyd’s Resources of Southwest Virginia. |
8vo, | 3 00 | ||
Map of Southwest Virginia |
Pocket-book form, | 2 00 | ||
Brush’s Manual of Determinative Mineralogy. (Penfield.) |
8vo, | 4 00 | ||
Chester’s Catalogue of Minerals |
8vo, | paper, | 1 00 | |
| Cloth, | 1 25 | |||
Dictionary of the Names of Minerals |
8vo, | 3 50 | ||
Dana’s System of Mineralogy |
Large 8vo, half leather, | 12 50 | ||
First Appendix to Dana’s New “System of Mineralogy.” |
Large 8vo, | 1 00 | ||
Text-book of Mineralogy |
8vo, | 4 00 | ||
Minerals and How to Study Them |
12mo, | 1 50 | ||
Catalogue of American Localities of Minerals |
Large 8vo, | 1 00 | ||
Manual of Mineralogy and Petrography |
12mo, | 2 00 | ||
Douglas’s Untechnical Addresses on Technical Subjects |
12mo, | 1 00 | ||
Eakle’s Mineral Tables |
8vo, | 1 25 | ||
Egleston’s Catalogue of Minerals and Synonyms |
8vo, | 2 50 | ||
Hussak’s The Determination of Rock-forming Minerals. (Smith.) |
Small 8vo, | 2 00 | ||
Merrill’s Non-metallic Minerals: Their Occurrence and Uses |
8vo, | 4 00 | ||
* Penfield’s Notes on Determinative Mineralogy and Record of Mineral Tests. |
8vo, paper, | 50 | ||
Rosenbusch’s Microscopical Physiography of the Rock-making Minerals. (Iddings.) |
8vo, | 5 00 | ||
* Tillman’s Text-book of Important Minerals and Rocks |
8vo, | 2 00 | ||
| MINING. | ||||
Beard’s Ventilation of Mines |
12mo, | 2 50 | ||
Boyd’s Resources of Southwest Virginia |
8vo, | 3 00 | ||
Map of Southwest Virginia |
Pocket-book form, | 2 00 | ||
Douglas’s Untechnical Addresses on Technical Subjects |
12mo, | 1 00 | ||
* Drinker’s Tunneling, Explosive Compounds, and Rock Drills |
4to, hf. mor., | 25 00 | ||
Eissler’s Modern High Explosives |
8vo, | 4 00 | ||
| [Pg 17] Fowler’s Sewage Works Analyses |
12mo, | 2 00 | ||
Goodyear’s Coal-mines of the Western Coast of the United States |
12mo, | 2 50 | ||
Ihlseng’s Manual of Mining |
8vo, | 5 00 | ||
* Iles’s Lead-smelting. (Postage 9c. additional.) |
12mo, | 2 50 | ||
Kunhardt’s Practice of Ore Dressing in Europe |
8vo, | 1 50 | ||
O’Driscoll’s Notes on the treatment of Gold Ores |
8vo, | 2 00 | ||
Robine and Lenglen’s Cyanide Industry. (Le Clerc.) |
8vo, | |||
* Walke’s Lectures on Explosives |
8vo, | 4 00 | ||
Wilson’s Cyanide Processes |
12mo, | 1 50 | ||
Chlorination Process |
12mo, | 1 50 | ||
Hydraulic and Placer Mining |
12mo, | 2 00 | ||
Treatise on Practical and Theoretical Mine Ventilation |
12mo, | 1 25 | ||
| SANITARY SCIENCE. | ||||
Bashore’s Sanitation of a Country House |
12mo, | 1 00 | ||
Folwell’s Sewerage. (Designing, Construction, and Maintenance.) |
8vo, | 3 00 | ||
Water-supply Engineering |
8vo, | 4 00 | ||
Fuertes’s Water and Public Health |
12mo, | 1 50 | ||
Water-filtration Works |
12mo, | 2 50 | ||
Gerhard’s Guide to Sanitary House-inspection |
16mo, | 1 00 | ||
Goodrich’s Economic Disposal of Town’s Refuse |
Demy 8vo, | 3 50 | ||
Hazen’s Filtration of Public Water-supplies |
8vo, | 3 00 | ||
Leach’s The Inspection and Analysis of Food with Special Reference to State Control |
8vo, | 7 50 | ||
Mason’s Water-supply. (Considered principally from a Sanitary Standpoint) |
8vo, | 4 00 | ||
Examination of Water. (Chemical and Bacteriological.) |
12mo, | 1 25 | ||
Ogden’s Sewer Design |
12mo, | 2 00 | ||
Prescott and Winslow’s Elements of Water Bacteriology, with Special Reference to Sanitary Water Analysis |
12mo, | 1 25 | ||
* Price’s Handbook on Sanitation |
12mo, | 1 50 | ||
Richards’s Cost of Food. A Study in Dietaries |
12mo, | 1 00 | ||
Cost of Living as Modified by Sanitary Science |
12mo, | 1 00 | ||
Richards and Woodman’s Air, Water, and Food from a Sanitary Standpoint |
8vo, | 2 00 | ||
* Richards and Williams’s The Dietary Computer |
8vo, | 1 50 | ||
Rideal’s Sewage and Bacterial Purification of Sewage |
8vo, | 3 50 | ||
Turneaure and Russell’s Public Water-supplies |
8vo, | 5 00 | ||
Von Behring’s Suppression of Tuberculosis. (Bolduan.) |
12mo, | 1 00 | ||
Whipple’s Microscopy of Drinking-water |
8vo, | 3 50 | ||
Winton’s Microscopy of Vegetable Foods |
8vo, | 7 50 | ||
Woodhull’s Notes on Military Hygiene |
16mo, | 1 50 | ||
| MISCELLANEOUS. | ||||
De Fursac’s Manual of Psychiatry. (Rosanoff and Collins.) |
Large 12mo, | 2 50 | ||
Emmons’s Geological Guide-book of the Rocky Mountain Excursion of the International Congress of Geologists |
Large 8vo, | 1 50 | ||
Ferrel’s Popular Treatise on the Winds |
8vo, | 4 00 | ||
Haines’s American Railway Management |
12mo, | 2 50 | ||
Mott’s Fallacy of the Present Theory of Sound |
16mo, | 1 00 | ||
Ricketts’s History of Rensselaer Polytechnic Institute, 1824-1894 |
Small 8vo, | 3 00 | ||
Rostoski’s Serum Diagnosis. (Bolduan.) |
12mo, | 1 00 | ||
Rotherham’s Emphasized New Testament |
Large 8vo, | 2 00 | ||
| [Pg 18] Steel’s Treatise on the Diseases of the Dog |
8vo, | 3 50 | ||
The World’s Columbian Exposition of 1893 |
4to, | 1 00 | ||
Von Behring’s Suppression of Tuberculosis. (Bolduan.) |
12mo, | 1 00 | ||
Winslow’s Elements of Applied Microscopy |
12mo, | 1 50 | ||
Worcester and Atkinson. Small Hospitals, Establishment and Maintenance; Suggestions for Hospital Architecture: Plans for Small Hospital |
12mo, | 1 25 | ||
| HEBREW AND CHALDEE TEXT-BOOKS. | ||||
Green’s Elementary Hebrew Grammar |
12mo, | 1 25 | ||
Hebrew Chrestomathy |
8vo, | 2 00 | ||
Gesenius’s Hebrew and Chaldee Lexicon to the Old Testament Scriptures. (Tregelles.) |
Small 4to, half morocco, | 5 00 | ||
Letteris’s Hebrew Bible |
8vo, | 2 25 | ||
Transcriber’s Notes:—
The spelling, hyphenation, punctuation and accentuation are as the original, except for apparent typographical errors which have been corrected.
The original table of contents is here:—
| CONTENTS. | ||
|---|---|---|
| PAGE | ||
| CHAPTER | I. INTRODUCTION. | 1 |
| II. CONTINUOUS FILTERS AND THEIR CONSTRUCTION | 5 | |
| Sedimentation-basins | 8 | |
| Size of Filter-beds | 10 | |
| Covers for Filters | 12 | |
| III. FILTERING-MATERIALS | 20 | |
| Sand | 20 | |
| Gravel | 35 | |
| Underdrains | 39 | |
| Depth of Water on Filters | 45 | |
| IV. RATE OF FILTRATION AND LOSS OF HEAD | 47 | |
| Rate of Filtration | 47 | |
| Loss of Head and Apparatus for regulating it | 52 | |
| Limit to the Loss of Head | 60 | |
| V. CLEANING FILTERS | 68 | |
| Scraping | 68 | |
| Frequency of Scraping | 72 | |
| Sand-washing | 76 | |
| VI. THEORY AND EFFICIENCY OF FILTRATION | 83 | |
| Bacterial Examination of Waters | 93 | |
| VII. INTERMITTENT FILTRATION | 97 | |
| The Lawrence Filter | 100 | |
| The Chemnitz Filter | 107 | |
| VIII. TURBIDITY AND COLOR, AND THE EFFECT OF MUD UPON | ||
| SAND FILTERS | 113 | |
| Color | 114 | |
| Turbidity | 117 | |
| Preliminary Processes to remove Mud | 133 | |
| Effect of Mud upon Sand Filters | 137 | |
| IX. COAGULATION OF WATERS | 144 | |
| Substances used for Coagulation | 145 | |
| Amount of Coagulant required to remove Turbidity | 150 | |
| Amount of Coagulant required to remove Color | 153 | |
| Successive Applications of Coagulant | 154 | |
| Amount of Coagulant which Waters will receive | 155 | |
| X. MECHANICAL FILTERS | 159 | |
| Influence of Amount of Coagulant on Bacterial | ||
| Efficiency | 165 | |
| Types of Mechanical Filters | 172 | |
| XI. OTHER METHODS OF FILTRATION | 181 | |
| XII. REMOVAL OF IRON FROM GROUND-WATERS | 186 | |
| Cause of Iron in Ground-waters | 187 | |
| Treatment of Iron-containing Waters | 189 | |
| Iron-removal Plants in Operation | 192 | |
| XIII. TREATMENT OF WATERS | 197 | |
| Cost of Filtration | 200 | |
| XIV. WATER-SUPPLY AND DISEASE | 210 | |
| APPENDIX I. GERMAN OFFICIAL REGULATION IN REGARD TO FILTRATION | 221 | |
| II. EXTRACTS FROM DR. REINCKE’S REPORT UPON THE HEALTH | ||
| OF HAMBURG FOR 1892 | 226 | |
| III. METHODS OF SAND-ANALYSIS | 233 | |
| IV. STATISTICS OF SOME FILTERS | 241 | |
| Results of Operation | 241 | |
| List of Sand Filters in Use | 244 | |
| List of Mechanical Filters in Use | 247 | |
| Notes regarding Sand Filters in America | 251 | |
| Extent of the Use of Filters | 254 | |
| V. WATER-SUPPLY OF LONDON | 255 | |
| VI. WATER-SUPPLY OF BERLIN | 261 | |
| VII. WATER-SUPPLY OF ALTONA | 265 | |
| VIII. WATER-SUPPLY OF HAMBURG | 269 | |
| IX. NOTES ON SOME OTHER EUROPEAN SUPPLIES | 272 | |
| X. LITERATURE OF FILTRATION | 277 | |
| XI. THE ALBANY FILTRATION PLANT | 288 | |
| INDEX | 317 | |
In the table of “ANALYSES OF SANDS USED IN WATER FILTRATION” the place name “Owesty” has been corrested to read “Oswestry”.
On page 270 the statement “the velocity in the drain will reach 0.97 foot” should probably read “0.97 feet per second”.