Transcriber’s Note:
The cover image was created by the transcriber and is placed in the public domain.
A
TEXT-BOOK OF ENTOMOLOGY
INCLUDING
THE ANATOMY, PHYSIOLOGY, EMBRYOLOGY AND METAMORPHOSES
OF
INSECTS
FOR USE IN AGRICULTURAL AND TECHNICAL SCHOOLS AND COLLEGES
AS WELL AS BY THE WORKING ENTOMOLOGIST
PREFACE

In preparing this book the author had in mind the wants both of the student and the teacher. For the student’s use the more difficult portions, particularly that on the embryology, may be omitted. The work has grown in part out of the writer’s experience in class work.
In instructing small classes in the anatomy and metamorphoses of insects, it was strongly felt that the mere dissection and drawing of a few types, comprising some of our common insects, were by no means sufficient for broad, thorough work. Plainly enough the laboratory work is all important, being rigidly disciplinary in its methods, and affording the foundation for any farther work. But to this should be added frequent explanations or formal lectures, and the student should be required to do collateral reading in some general work on structural and developmental entomology. With this aim in view, the present work has been prepared.
It might be said in explanation of the plan of this book, that the students having previously taken a lecture course in the zoölogy of the invertebrates, were first instructed in the facts and conclusions bearing on the relations of insects to other Arthropoda, and more especially the anatomy of Peripatus, of the Myriopoda, and of Scolopendrella. Then the structure of Campodea, Machilis, and Lepisma was described, after which a few types of winged insects, beginning with the locust and ending with the bee, were drawn and dissected; the nymph of the locust, and the larva and pupa of a moth and of a wasp and bee being drawn and examined. Had time permitted, an outline of the embryology and of the internal changes in flies during their metamorphoses would have been added.
viThis book gives, of course with much greater fulness and detail for reference and collateral reading, what we roughly outlined in our class work. The aim has been to afford a broad foundation for future more special work by any one who may want to carry on the study of some group of insects, or to extend in any special direction our present knowledge of insect morphology and growth.
Many of our entomologists begin their studies without any previous knowledge of the structure of animals, taking it up as an amusement. They may be mere collectors and satisfied simply to know the name of their captures, but it is hoped that with this book in their hands they may be led to desire farther information regarding what has already been done on the structure and mode of growth of the common insects. For practical details as to how to dissect, to make microscopic slides, and to mount and preserve insects generally, they are referred to the author’s “Entomology for Beginners.”
It may also be acknowledged that even in our best and latest general treatises on zoölogy, or comparative anatomy, or morphology, the portion related to insects is scarcely so thoroughly done as those parts devoted to other phyla, that of Lang, however, his invaluable Comparative Anatomy, being an exception. On this account, therefore, it is hoped that this hiatus in our literature may be in a degree filled.
The author has made free use of the excellent article “Insecta” of Newport, of Lang’s comprehensive summary in his most useful Text-book of Comparative Anatomy, of Graber’s excellent Die Insecten, of Miall and Denny’s The Structure and Life-History of the Cockroach, and of Sharp’s Insecta. Kolbe’s Einführung has been most helpful. But besides these helps, liberal use has been made of the very numerous memoirs and monographic articles which adorn our entomological literature. The account of the embryology of insects is based on Korschelt and Heider’s elaborate work, Lehrbuch der Vergleichenden Entwicklungsgeschichte der Wirbellosen Thiere, the illustrations of this portion being mainly taken from it, through the Messrs. Swan Sonnenschein & Co., London.
viiProfessor H. S. Pratt has kindly read over the manuscript and also the proofs of the portion on embryology and metamorphoses, and the author is happy to acknowledge the essential service he has rendered.
The bibliographical lists are arranged by dates, so as to give an idea of the historical development of each subject. The aim has been to make these lists tolerably complete and to include the earliest, almost forgotten works and articles as well as the most recent.
Much care has been taken to give due credit either to the original sources from which the illustrations are copied, or to the artist; about ninety of the simpler figures were drawn by the author, many of them for this work. For the use of certain figures acknowledgments are due to the Boston Society of Natural History, to the Division of Entomology, U. S. Department of Agriculture, through the kind offices of Mr. L. O. Howard, and to the Illinois State Laboratory of Natural History, through Professor S. A. Forbes and Mr. C. A. Hart. Professor W. M. Wheeler, of the University of Chicago, has kindly loaned for reproduction several of his original drawings published in the Journal of Morphology. A number are reproduced from figures in the reports of the United States Entomological Commission.
TABLE OF CONTENTS

| PART I. MORPHOLOGY AND PHYSIOLOGY | |||
| PAGE | |||
|---|---|---|---|
| Position of Insects in the Animal Kingdom | 1 | ||
| Relations of Insects to Other Arthropoda | 2 | ||
| The Crustacea | 4 | ||
| The Merostomata | 5 | ||
| The Trilobita | 5 | ||
| The Arachnida | 6 | ||
| Relations of Peripatus to insects | 9 | ||
| Relation of Myriopods to insects | 11 | ||
| Relations of the Symphyla to insects | 18 | ||
| Diagnostic or essential characters of Symphyla | 22 | ||
| Insecta (Hexapoda) | 26 | ||
| Diagnostic characters of insects | 26 | ||
| 1. EXTERNAL ANATOMY | |||
| a. Regions of the body | 27 | ||
| b. The integument (exoskeleton) | 28 | ||
| Chitin | 29 | ||
| c. Mechanical origin and structure of the segments (somites, arthromeres, etc.) | 30 | ||
| d. Mechanical origin of the limbs and of their jointed structure | 35 | ||
| The Head and its Appendages | 42 | ||
| a. The head | 42 | ||
| The labrum | 42 | ||
| The epipharynx and labrum-epipharynx | 43 | ||
| Attachment of the head to the trunk | 46 | ||
| The basal or gular region of the head | 46 | ||
| The occiput | 48 | ||
| The tentorium | 49 | ||
| Number of segments in the head | 50 | ||
| The composition of the head in the Hymenoptera | 55 | ||
| x | b. Appendages of the head | 57 | |
| The antennæ | 57 | ||
| The mandibles | 59 | ||
| The first maxillæ | 62 | ||
| The second maxillæ | 68 | ||
| The hypopharynx | 70 | ||
| Does the hypopharynx represent a distinct segment? | 82 | ||
| The Thorax and its Appendages | 86 | ||
| a. The thorax: its external anatomy | 86 | ||
| The patagia | 89 | ||
| The tegulæ | 89 | ||
| The apodemes | 92 | ||
| The acetabula | 94 | ||
| b. The legs: their structure and functions | 95 | ||
| Tenent hairs | 99 | ||
| Why do insects have but six legs? | 100 | ||
| Loss of limbs by disuse | 101 | ||
| c. Locomotion (walking, climbing, and swimming) | 103 | ||
| Mechanics of walking | 103 | ||
| Locomotion on smooth surfaces | 111 | ||
| Climbing | 116 | ||
| The mode of swimming of insects | 116 | ||
| d. The wings and their structure | 120 | ||
| The veins | 121 | ||
| The squamæ | 123 | ||
| The halteres | 124 | ||
| The thyridium | 124 | ||
| The tegmina and hemelytra | 124 | ||
| The elytra | 124 | ||
| e. Development and mode of origin of the wings | 126 | ||
| Embryonic development of the wings | 126 | ||
| Evagination of the wing outside of the body | 132 | ||
| Extension of the wing; drawing out of the tracheoles | 133 | ||
| f. The primitive origin of the wings | 137 | ||
| The development and structure of the tracheæ and veins of the wing | 144 | ||
| g. Mechanism of flight | 148 | ||
| Theory of insect flight | 150 | ||
| Graber’s views as to the mechanism of the wings, flight, etc. | 153 | ||
| The Abdomen and its Appendages | 162 | ||
| The median segment | 163 | ||
| The cercopoda | 164 | ||
| The ovipositor and sting | 167 | ||
| The styles and genital claspers (Rhabdopoda) | 176 | ||
| Velum penis | 181 | ||
| The suranal plate | 181 | ||
| xi | The podical plates or paranal lobes | 182 | |
| The infra-anal lobe | 183 | ||
| The egg-guide | 183 | ||
| The Armature of Insects: Setæ, Hairs, Scales, Tubercles, Etc. | 187 | ||
| The cuticula | 187 | ||
| Setæ | 188 | ||
| Glandular hairs and spines | 190 | ||
| Scales | 193 | ||
| Development of the scales | 195 | ||
| Spinules, hair-scales, hair-fields, and androconia | 197 | ||
| The Colors of Insects | 201 | ||
| Optical colors | 201 | ||
| Natural colors | 203 | ||
| Chemical and physical nature of the pigment | 206 | ||
| Ontogenetic and phylogenetic development of colors | 207 | ||
| 2. INTERNAL ANATOMY | |||
| The Muscular System | 211 | ||
| Musculature of a caterpillar | 213 | ||
| Musculature of a beetle | 213 | ||
| Minute structure of the muscles | 215 | ||
| Muscular power of insects | 217 | ||
| The Nervous System | 222 | ||
| a. The nervous system as a whole | 222 | ||
| b. The brain | 226 | ||
| The optic or procerebral segment | 231 | ||
| Procerebral lobes | 232 | ||
| The mushroom or stalked bodies | 233 | ||
| Structure of the mushroom bodies | 234 | ||
| The central body | 237 | ||
| The antennal or olfactory lobes (Deutocerebrum) | 237 | ||
| The œsophageal lobes (Tritocerebrum) | 237 | ||
| c. Histological elements of the brain | 238 | ||
| d. The visceral (sympathetic or stomatogastric) system | 238 | ||
| e. The supraspinal cord | 240 | ||
| f. Modifications of the brain in different orders of insects | 240 | ||
| g. Functions of the nerve-centres and nerves | 243 | ||
| The Sensory Organs | 249 | ||
| a. The eyes and insect vision | 249 | ||
| The simple or single-lensed eye (ocellus) | 249 | ||
| The compound or facetted eye (ommateum) | 250 | ||
| The facet or cornea | 250 | ||
| The crystalline lens or cone | 251 | ||
| xii | The pigment | 253 | |
| The basilar membrane | 253 | ||
| The optic tract | 253 | ||
| Origin of the facetted eye | 255 | ||
| Mode of vision by single eyes or ocelli | 255 | ||
| Mode of vision by facetted eyes | 256 | ||
| The principal use of the facetted eye to perceive the movements of animals | 259 | ||
| How far can insects see? | 260 | ||
| Relation of sight to the color of eyes | 260 | ||
| The color sense of insects | 260 | ||
| b. The organs of smell | 264 | ||
| Historical sketch of our knowledge of the organs of smell | 264 | ||
| Physiological experiments | 268 | ||
| Relation of insects to smelling substances before and after the loss of their antennæ | 269 | ||
| Experiments on the use of the antennæ in seeking for food | 270 | ||
| Experiments testing the influence of the antennæ of the males in seeking the females | 270 | ||
| Structure of the organs of smell in insects | 271 | ||
| c. The organs of taste | 281 | ||
| Structure of the taste organs | 282 | ||
| Distribution in different orders of insects | 282 | ||
| Experimental proof | 286 | ||
| d. The organs of hearing | 287 | ||
| The ears or tympanal and chordotonal sense-organs of Orthoptera and other insects | 288 | ||
| Antennal auditory hairs | 292 | ||
| Special sense-organs in the wings and halteres | 293 | ||
| e. The sounds of insects | 293 | ||
| The Digestive Canal and its Appendages | 297 | ||
| a. The digestive canal | 302 | ||
| The œsophagus | 303 | ||
| The crop or ingluvies | 303 | ||
| The “sucking stomach” or food-reservoir | 305 | ||
| The fore-stomach or proventriculus | 306 | ||
| The œsophageal valve | 311 | ||
| Proventricular valvule | 313 | ||
| The peritrophic membrane | 313 | ||
| The mid-intestine | 314 | ||
| Histology of the mid-intestine | 316 | ||
| The hind-intestine | 316 | ||
| Large intestine | 316 | ||
| The ileum | 317 | ||
| The gastro-ileal folds | 317 | ||
| The colon | 317 | ||
| The rectum | 318 | ||
| xiii | The vent (anus) | 319 | |
| Histology of the digestive canal | 320 | ||
| b. Digestion in insects | 324 | ||
| The mechanism of secretion | 326 | ||
| Absorbent cells | 328 | ||
| The Glandular and Excretory Appendages of the Digestive Canal | 331 | ||
| a. The salivary glands | 331 | ||
| b. The silk or spinning glands, and the spinning apparatus | 339 | ||
| The process of spinning | 340 | ||
| How the thread is drawn out | 343 | ||
| Appendages of the silk-gland (Filippi’s glands) | 345 | ||
| c. The cæcal appendages | 347 | ||
| d. The excretory system (urinary or Malpighian tubes) | 348 | ||
| Primitive number of tubes | 353 | ||
| e. Poison-glands | 357 | ||
| f. Adhesive or cement-glands | 360 | ||
| g. The wax-glands | 361 | ||
| h. “Honey-dew” or wax-glands of Aphids | 364 | ||
| i. Dermal glands in general | 365 | ||
| Defensive or Repugnatorial Scent-Glands | 368 | ||
| Eversible coxal glands | 369 | ||
| Fœtid glands of Orthoptera | 369 | ||
| Anal glands of beetles | 372 | ||
| The blood as a repellent fluid | 374 | ||
| Eversible glands of caddis-worms and caterpillars | 375 | ||
| The osmeterium in Papilio larvæ | 377 | ||
| Dorsal and lateral eversible metameric sacs in other larvæ | 377 | ||
| Distribution of repugnatorial or alluring scent-glands in insects | 382 | ||
| The Alluring or Scent-Glands | 391 | ||
| The Organs of Circulation | 397 | ||
| a. The heart | 397 | ||
| The propulsatory apparatus | 401 | ||
| The supraspinal vessel | 403 | ||
| The aorta | 404 | ||
| The pericardial cells | 405 | ||
| Pulsatile organs of the legs | 405 | ||
| b. The blood | 407 | ||
| The leucocytes | 407 | ||
| c. The circulation of the blood | 409 | ||
| Effects of poisons on the pulsations | 412 | ||
| The Blood Tissue | 419 | ||
| a. The fat-body | 419 | ||
| b. The pericardial fat-body or pericardial cells | 420 | ||
| Leucocytes or phagocytes in connection with the pericardial cells | 421 | ||
| xiv | c. The œnocytes | 423 | |
| d. The phosphorescent organs | 424 | ||
| Physiology of the phosphorescence | 426 | ||
| The Respiratory System | 430 | ||
| a. The tracheæ | 431 | ||
| Distribution of the tracheæ | 432 | ||
| b. The spiracles or stigmata | 437 | ||
| The position and number of pairs of stigmata | 439 | ||
| The closing apparatus of the stigma | 441 | ||
| c. Morphology and homologies of the tracheal system | 442 | ||
| d. The spiral threads or tænidia | 444 | ||
| e. Origin of the tracheæ and of the “spiral thread” | 447 | ||
| Internal, hair-like bodies | 451 | ||
| f. The mechanism of respiration and the respiratory movements of insects | 451 | ||
| g. The air-sacs | 456 | ||
| The use of the air-sacs | 457 | ||
| h. The closed or partly closed tracheal system | 459 | ||
| i. The rectal, tracheal gills, and rectal respiration of larval Odonata and other insects | 463 | ||
| j. Tracheal gills of the larvæ of insects | 466 | ||
| Blood-gills | 475 | ||
| k. Tracheal gills of adult insects | 476 | ||
| The Organs of Reproduction | 485 | ||
| a. The male organs of reproduction | 494 | ||
| The testes | 495 | ||
| The seminal ducts | 496 | ||
| The ejaculatory duct | 497 | ||
| The accessory glands | 497 | ||
| The spermatozoa | 497 | ||
| Formation of the spermatozoön | 498 | ||
| b. The female organs of reproduction | 500 | ||
| The ovaries and the ovarian tubes | 500 | ||
| Origin of incipient eggs in the germ of the testes | 504 | ||
| The bursa copulatrix | 505 | ||
| The spermatheca | 506 | ||
| The colleterial glands | 506 | ||
| The vagina or uterus | 507 | ||
| Signs of copulation in insects | 507 | ||
| PART II. EMBRYOLOGY OF INSECTS | |||
| a. The egg | 515 | ||
| Mode of deposition | 518 | ||
| Vitality of eggs | 520 | ||
| xv | Appearance and structure of the ripe egg | 520 | |
| The egg-shell and yolk-membrane | 520 | ||
| The micropyle | 522 | ||
| Internal structure of the egg | 524 | ||
| b. Maturation or ripening of the egg | 525 | ||
| c. Fertilization of the egg | 525 | ||
| d. Division and formation of the blastoderm | 526 | ||
| e. Formation of the first rudiments of the embryo and of the embryonic membranes | 531 | ||
| Formation of the embryonic membranes | 532 | ||
| The gastrula stage | 535 | ||
| Division of the embryo or primitive band into body-segments | 536 | ||
| Differences between the invaginated and overgrown primitive band | 538 | ||
| Revolution of the embryo where the primitive band is invaginated | 540 | ||
| f. Formation of the external form of the body | 542 | ||
| Origin of the body-segments | 542 | ||
| The procephalic lobes | 544 | ||
| Fore-intestine (stomodæum) and hind-intestine (proctodæum), labrum | 547 | ||
| Completion of the head | 548 | ||
| g. The appendages | 548 | ||
| The cephalic appendages | 548 | ||
| The thoracic appendages | 550 | ||
| The abdominal appendages | 550 | ||
| Appendages of the first abdominal segment (pleuropodia) | 551 | ||
| Are the abdominal legs of Lepidoptera and phytophagous Hymenoptera true limbs? | 552 | ||
| The tracheæ | 553 | ||
| h. Nervous system | 554 | ||
| Completion of the definite form of the body | 555 | ||
| i. Dorsal closure and involution of the embryonic membranes | 556 | ||
| j. Formation of the germ-layers | 558 | ||
| k. Farther development of the mesoderm; formation of the body-cavity | 563 | ||
| l. Formation of organs | 566 | ||
| The nervous system | 566 | ||
| Development of the brain | 567 | ||
| Development of the eyes | 567 | ||
| Intestinal canal and glands | 569 | ||
| The salivary glands | 570 | ||
| The urinary tubes | 572 | ||
| The heart | 572 | ||
| The blood-corpuscles | 574 | ||
| Musculature; connective tissue; fat-body | 574 | ||
| The reproductive organs | 575 | ||
| Development of the male germinal glands | 579 | ||
| m. Length of embryonic life | 582 | ||
| n. The process of hatching | 583 | ||
| The hatching spines | 585 | ||
| xviPART III. THE METAMORPHOSES OF INSECTS | |||
| a. The nymph as distinguished from the larval stage | 593 | ||
| b. Stages or stadia of metamorphosis | 594 | ||
| c. Ametabolous and metabolous stages | 594 | ||
| The Larva | 599 | ||
| a. The Campodea-form type of larva | 600 | ||
| b. The eruciform type of larva | 602 | ||
| c. Growth and increase in size of the larva | 608 | ||
| d. The process of moulting | 609 | ||
| The number of moults in insects of different orders | 615 | ||
| Reproduction of lost limbs | 619 | ||
| Formation of the cocoon | 619 | ||
| Sanitary conditions observed by the honey-bee larva, and admission of air within the cocoon | 623 | ||
| The Pupa State | 625 | ||
| a. The pupa considered in reference to its adaptation to its surroundings and its relation to phylogeny | 631 | ||
| b. Mode of escape of the pupa from its cocoon | 632 | ||
| c. The cremaster | 636 | ||
| Mode of formation of the cremaster and suspension of the chrysalis in butterflies | 637 | ||
| Formation of the Pupa and Imago in the Holometabolous Insects (the Diptera excepted) | 640 | ||
| a. The Lepidoptera | 642 | ||
| The changes in the head and mouth-parts | 646 | ||
| The change in the internal organs | 647 | ||
| The wings | 654 | ||
| Development of the feet and of the cephalic appendages | 654 | ||
| Embryonic cells and the phagocytes | 655 | ||
| Formation of the femur and of the tibia; transformation of the tarsus | 656 | ||
| The antennæ | 657 | ||
| Maxillæ and labial palpi | 658 | ||
| Process of pupation | 660 | ||
| b. The Hymenoptera | 661 | ||
| Ocular or oculo-cephalic buds | 665 | ||
| The antennal buds | 665 | ||
| The buds of the buccal appendages | 665 | ||
| The buds of the ovipositor | 665 | ||
| Development of the Imago in the Diptera | 666 | ||
| a. Development of the outer body-form | 668 | ||
| Formation of the imago in Corethra | 668 | ||
| Formation of the imago in Culex | 670 | ||
| xvii | Formation of the imago in Chironomus | 671 | |
| Formation of the imago in Muscidæ | 673 | ||
| b. Development of the internal organs of the imago | 678 | ||
| The hypodermis | 678 | ||
| The muscles | 680 | ||
| The digestive canal | 681 | ||
| The tracheal system | 683 | ||
| The nervous system | 684 | ||
| The fat-body | 685 | ||
| Definitive fate of the leucocytes | 685 | ||
| The post-embryonic changes and imaginal buds in the Pupipara (Melophagus) | 686 | ||
| c. General summary | 687 | ||
| Hypermetamorphism | 688 | ||
| Summary of the Facts and Suggestions as to the Causes of Metamorphism | 705 | ||
| Theoretical conclusions; causes of metamorphosis | 708 | ||

PART I.—MORPHOLOGY AND PHYSIOLOGY

POSITION OF INSECTS IN THE ANIMAL KINGDOM
Although the insects form but a single class of the animal kingdom, they are yet so numerous in orders, families, genera, and species, their habits and transformations are so full of instruction to the biologist, and they affect human interests in such a variety of ways, that they have always attracted more attention from students than any other class of animals, the number of entomologists greatly surpassing that of ornithologists, ichthyologists, or the special students of any other class, while the literature has assumed immense proportions.
Insects form about four-fifths of the animal kingdom. There are about 250,000 species already named and contained in our museums, while the number of living and fossil species in all is estimated to amount to between one and two millions.
In their structure insects are perhaps more complicated than any other animals. This is partly due to the serial arrangement of the segments and the consequent segmental repetition of organs, especially of the external appendages, and of the muscles, the tracheæ, and the nerves. The brain is nearly or quite as complicated as that of the higher vertebrates, while the sense-organs, especially those of touch, sight, and smell are, as a rule, far more numerous and only less complex than those of vertebrates. Moreover, in their psychical development, certain insects are equal, or even superior, to any other animals, except birds and mammals.
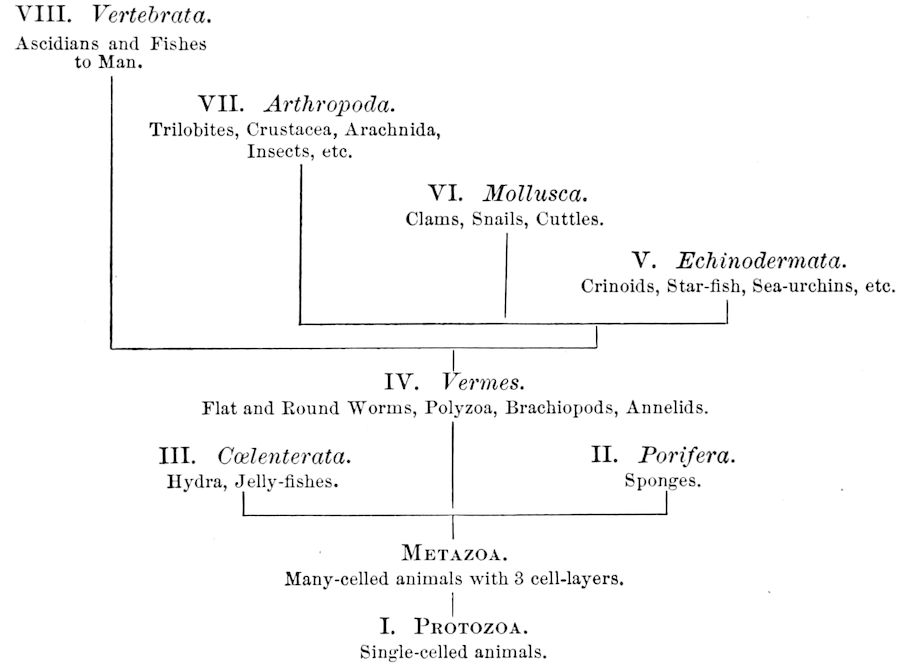
The animal kingdom is primarily divided into two grand divisions, the one-celled (Protozoa) and many-celled animals (Metazoa). In the latter group the cells and tissues forming the body are arranged in three fundamental cell-layers; viz. the ectoderm or outer layer, the mesoderm, and endoderm. The series of branches, or phyla, comprised 2under the term Metazoa are the Porifera, Cœlenterata, Vermes, Echinodermata, Mollusca, Arthropoda, and Vertebrata. Their approximate relationships may be provisionally expressed by the following
Tabular View of the Eight Branches or Phyla of the Animal Kingdom.
RELATIONS OF INSECTS TO OTHER ARTHROPODA
The insects by general consent stand at the head of the Arthropoda. Their bodies are quite as much complicated or specialized, and indeed, when we consider the winged forms, more so, than any other class of the branch, and besides this they have wings, fitting them for an aërial life. It is with little doubt that to their power of flight, and thus of escaping the attacks of their creeping arthropod enemies, insects owe, so to speak, their success in life; i.e. their numerical superiority in individuals, species, and genera. It is also apparently their power of moving or swimming swiftly from one place to another which has led to the numerical superiority in species of fishes to other Vertebrata. Among terrestrial vertebrates, the birds, by virtue of their ability to fly, greatly surpass in number of species the reptiles and mammals.
The Arthropoda are in general characterized by having the body 3composed of segments (somites or arthromeres) bearing jointed appendages. They differ from the worms in having segmented appendages, i.e. antennæ, jaws, and legs, instead of the soft unjointed outgrowths of the annelid worms. Moreover, their bodies are composed of a more or less definite number of segments or rings, grouped either into a head-thorax (cephalothorax) and hind-body, as in Crustacea, or into a head differentiated from the rest of the body (trunk), the latter not being divided into a distinct thorax and abdomen, as in Myriopoda; or into three usually quite distinct regions—the head, thorax, and hind-body or abdomen, as in insects. In certain aberrant, modified forms, as the Tardigrada, or the Pantopoda, and the mites, the body is not differentiated into such definite regions.
In their internal organs arthropods agree in their general relations with the higher worms, hence most zoölogists agree that they have directly originated from the annelid worms.
The position and general shape of the digestive canal, of the nervous and circulatory systems, are the same in Arthropoda as in annelid (oligochete) worms, so much so that it is generally thought that the Arthropoda are the direct descendants of the worms. It is becoming evident, however, that there was no common ancestor of the Arthropoda as a whole, and that the group is a polyphyletic one. Hence, though a convenient group, it is a somewhat artificial one, and may eventually be dismembered into at least three or four phyla or branches.
The following diagram may serve to show in a tentative way the relations of the classes of Arthropoda to each other, and also may be regarded as a provisional genealogical tree of the branch.
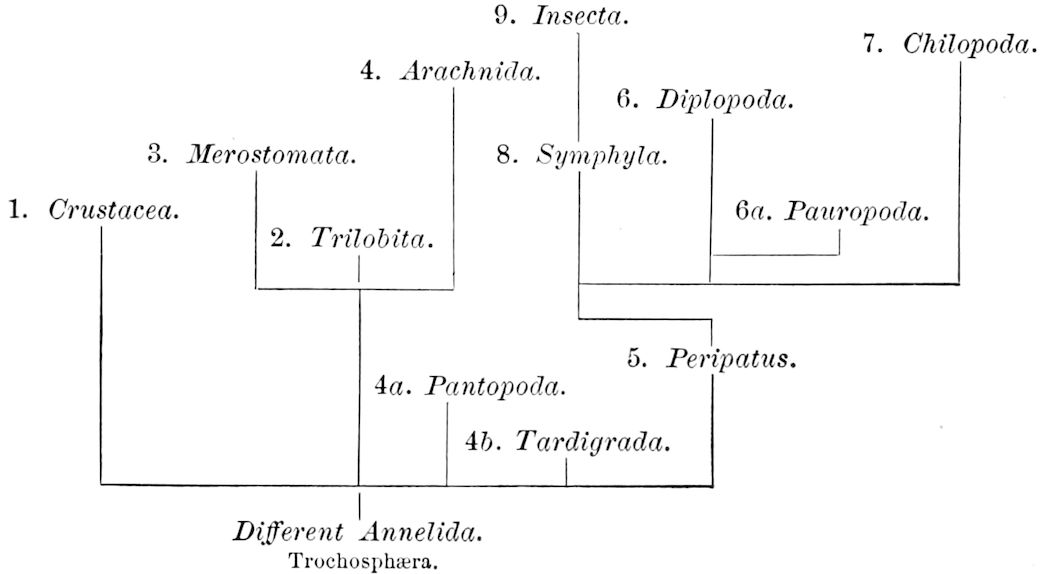
4We will now rapidly review the leading features of the classes of Arthropoda.
The Crustacea.—These Arthropoda are in many most important characteristics unlike the insects; they have two pairs of antennæ, five pairs of buccal appendages, and they are branchiate Arthropoda. They have evidently originated entirely independently, and by a direct line of descent from some unknown annelid ancestor which was either a many-segmented worm, with parapodia, or the two groups together with the Rotifera may have originated from a common appendigerous Trochosphæra. Their segments in the higher forms are definite in number (23 or 24) and arranged into two regions, a head-thorax (cephalothorax) and hind-body (abdomen). Nearly all the segments, both of the cephalothorax and abdomen, bear a pair of jointed limbs, and to them at their base are, in the higher forms, appended the gills (branchiæ). The limbs are in the more specialized forms (shrimps and crabs) differentiated into eye-stalks, two pairs of antennæ, a pair of palpus-bearing jaws (mandibles), two pairs of maxillæ and three pairs of maxillipeds; these appendages being biramose, and the latter bearing gills attached to their basal joints. The legs are further differentiated into ambulatory thoracic legs and into swimming or abdominal legs, and in the latter the first pair of the male is modified into copulatory organs (gonopoda). The male and female reproductive organs as a rule are in separate individuals, hermaphrodites being very unusual, and the glands may be paired or single. The sexual outlets are generally paired, and, as in the male lobster and other Macrura, open in the basal joint of the last pair of legs, and in the female in the third from the last; while originally in all Crustacea the sexual organs were most probably paired (Fig. 3, B).
They are, except a few land Isopoda, aquatic, mostly marine, and when they have a metamorphosis, pass through a six-legged larval stage, called the Nauplius, the shrimps and crabs passing through an additional stage, the Zoëa. Crustacea also differ much from insects in the highly modified nature of the nephridia, which are usually represented by the green gland of the lobster, or the shell-glands of the Phyllopoda, which open out in one of the head-segments; also in the possession of a pair of large digestive glands, the so-called liver.
Intermediate in some respects between the Crustacea and insects, but more primitive, in respect to what are perhaps the most weighty characters, than the Crustacea, are the Trilobita, the Merostomata (Limulus), and, finally, the Arachnida, these being allied groups. In the Trilobita and Merostomata (Limulus), the head-appendages are 5more like feet than jaws, while they have in most respects a similar mode of embryonic development, the larval forms being also similar.
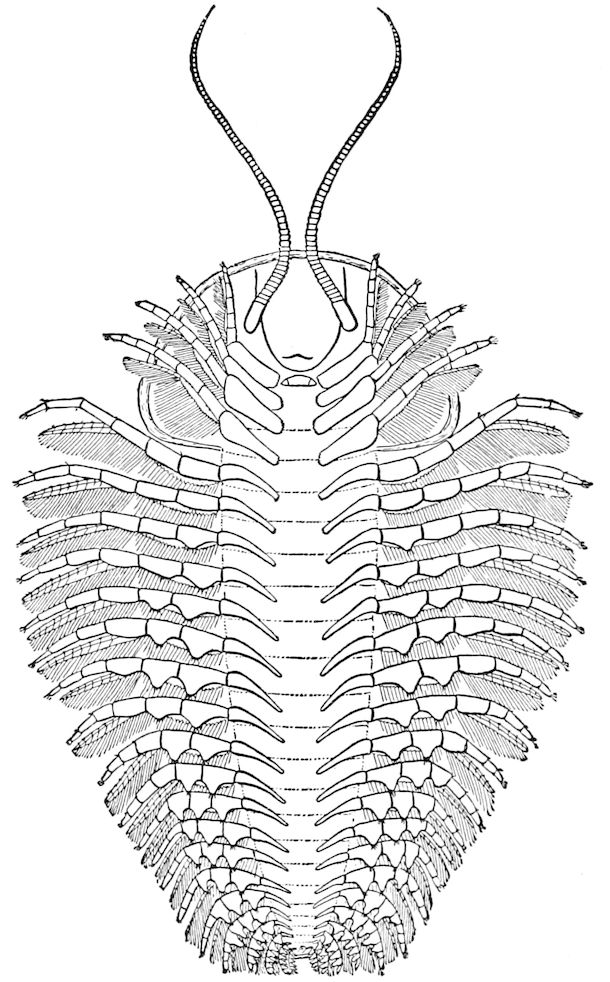
Fig. 1.—Restoration of under side of a trilobite (Triarthrus becki), the trunk limbs bearing small triangular respiratory lobes or gills.—After Beecher.
The Merostomata.—The only living form, Limulus, is undoubtedly a very primitive type, as the genital glands and ducts are double, opening wide apart on the basal pair of abdominal legs (Fig. 3). Moreover, their head-appendages, which are single, with spines on the basal joint, are very primitive and morphologically nearer in shape to those of the worms (Syllidæ, etc.) than even those of the Crustacea. Besides, their four pairs of coxal glands, with an external opening at the base of the fifth pair of head-appendages, and which probably are modified nephridia (Crustacea having but a single pair in any one form, either opening out on the second antennal, green gland, or second maxillary, shell-gland, segment), indicate a closer approximation to the polynephrous worms. Limulus has other archaic features, especially as regards the structure of the simple and compound eyes and the simple nature of the brain.
The Trilobita.—These archaic forms are still more generalized and primitive than the Merostomata and Crustacea, and probably were the first Arthropoda to be evolved from some unknown annelid worm. They had jointed biramose limbs of nearly uniform shape and size on each segment of the body, which were not, as in Crustacea, differentiated into antennæ, jaws (mandibles), maxillæ, maxillipeds, and two kinds of legs (thoracic and 6abdominal), showing that they are a much more primitive type, and nearer to the annelids than any other Arthropoda. Their gills, as shown by the researches of Walcott and of Beecher, were attached to nearly if not every pair of limbs behind the antennæ (Figs. 1, 2). The fact that in Trilobita the first pair of limbs is antenniform does not prove that they are Crustacea, since Eurypterus has a similar pair of appendages.
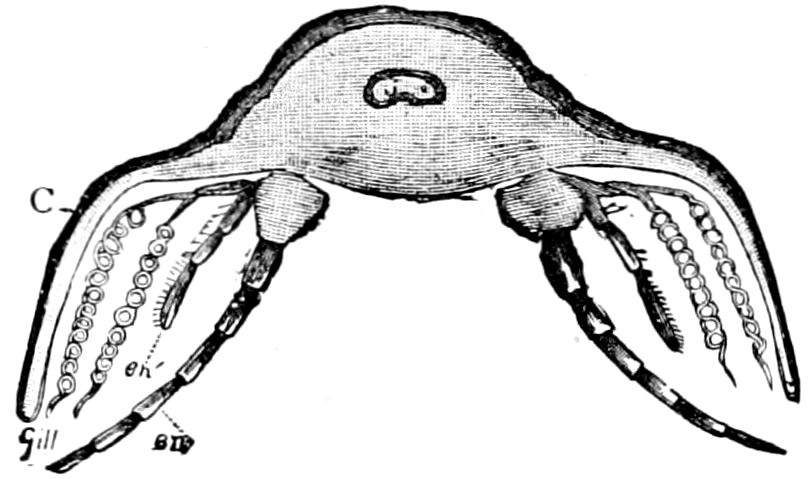
Fig. 2.—Restored section of Calymene: C, carapace; en, endopodite; en′, exopodite; with the gills on the epipodal or respiratory part of the appendage.—After Walcott.
The limbs in trilobites, as well as the abdominal ones of merostomes, and all those of Crustacea, except the first antennæ, are biramose, consisting of an outer (exopodite) and an inner division (endopodite). In this respect the terrestrial air-breathing tracheate forms, Arachnida, Myriopoda, and Insecta, differ from the branchiate forms, as their legs are single or undivided, being adapted for supporting the body during locomotion upon the solid earth. It is to be observed that when, as in Limulus, the body is supported by cephalic ambulatory limbs, they are single, while the abdominal limbs, used as they are in swimming, are biramose, much as in Crustacea.
The Arachnida.—The scorpions and spiders are much less closely allied to the myriopods and insects than formerly supposed. Their embryology shows that they have descended from forms related to Limulus, possibly having had an origin in common with that animal, or having, as some authors claim, directly diverged from some primitive eurypteroid merostome. But they differ in essential respects, and not only in the nature and grouping of their appendages; the first pair instead of antenniform being like mandibles, and the second pair like the maxillæ, with the palps, of insects, the four succeeding segments (thoracic) bearing each a pair of legs. They also have a brain quite unlike that of Limulus, the nervous cord behind the brain, however, being somewhat similar, though that of Limulus differs in being enveloped by an arterial coat. Arachnida respire by tracheæ, besides book-lungs, which, however, are possibly derivatives of the book-gills of Limulus, while they perform the office of excretion by means of the malpighian tubes, and like Limulus possess two large digestive glands (“liver”). Their embryos have, on at least six abdominal segments, rudiments of limbs, three pairs of which form the spinnerets, showing their origin from Limulus-like or eurypteroid forms; their coxal glands are retained from their eurypteroid ancestors. The Arachnida probably descended from marine merostomes, and not from an independent annelid ancestry, hence we have represented them in the diagram on p. 3 as branching off from the merostomatous phylum, rather than from an independent one.
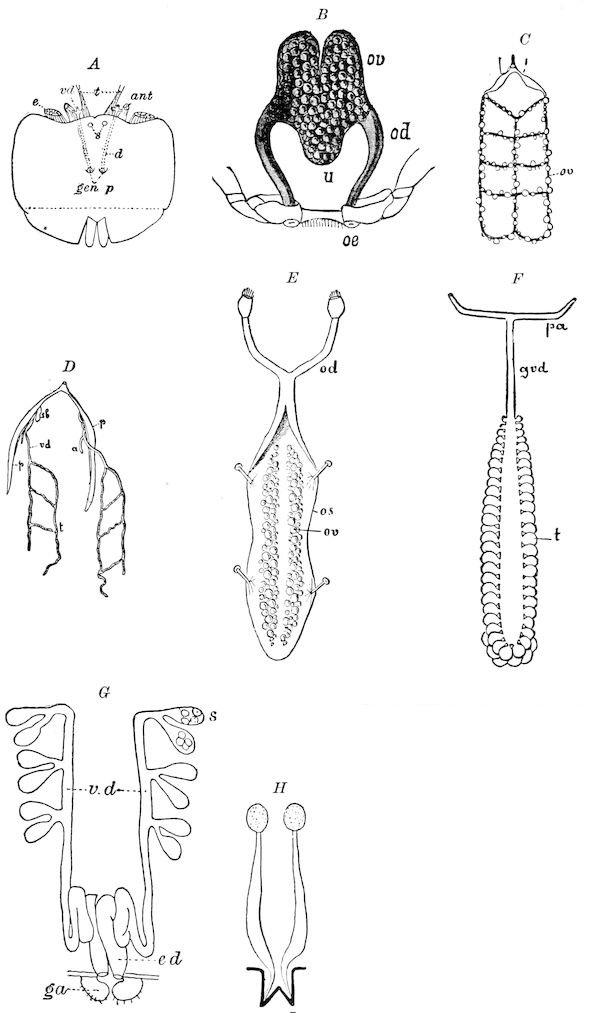
Fig. 3.—Paired genital openings of different classes of arthropods. A, the most primitive, of Limulus polyphemus: gen. p, generative papillæ; d, duct; vd, vas deferens; t, tendinous stigmata; stig, stigmata; e, external branchial muscle; ant, anterior lamellar muscle.—After Benham, with a few changes. B, lobster (Homarus vulgaris), ♀: oe, genital aperture on 3d pair of legs; ov, ovary; u, unpaired portion of the same; od, oviduct. C, ♀, scorpion: ov, ovary, with a single external opening. D, ♂: t, testis; vd, vasa deferentia; sb, seminal vesicle; a, glandular appendage; p, penis.—After Blanchard. E, a myriopod (Glomeris marginata, ♀): os, ovarian sac, laid open; od, paired oviducts. F, ♂: t, testis; gvd, common vas deferens; pa, paired ducts.—After Favre, from Lang. G, Lepisma saccharina, young ♂: vd, vas deferens, ed, ejaculatory duct; ga, external appendages.—After Nassonow. H, Ephemera, ♂, showing the double outlets.—After Palmén.
8The characters in which arachnids approach insects, such as tracheæ and malpighian tubes (none occur, as a rule, in marine or branchiate arthropods), may be comparatively recent structures acquired during a change from a marine to a terrestrial life, and not primitive heirlooms.
Arachnida also show their later origin than merostomes by the fact that their sexual glands are in most cases single, and though with rare exceptions the ducts are paired, these finally unite and open externally by a common single genital aperture in the median line of the body, at the base of the abdomen (Fig. 3, C, D). In this respect Limulus, with its pair of genital male or female openings, situated each at the end of a papilla, placed widely apart at the base of the first abdominal limbs, is decidedly more archaic. Unlike Crustacea and insects, Arachnida do not, except in the mites (Acarina), which is a very much modified group, undergo a metamorphosis.
We see, then, that the insects, with the Myriopoda, are somewhat isolated from the other Arthropoda. The Myriopoda have a single pair of antennæ, and as they have other characters in common with insects, Lang has united the two groups in a single class Antennata; but, as we shall see, this seems somewhat premature and unnecessary. Yet the two groups have perhaps had a common parentage, and may prove to belong to a distinct, common phylum.
Not only by their structure and embryology, as well as their metamorphosis, do the myriopods and insects stand apart from the Arachnida and other arthropods, but it seems probable that they have had a different ancestry, the arthropods being apparently polyphyletic.
There are two animals which appear to connect the insects with the worms, and which indicate a separate line of descent from the worms independent of that of the other classes. These are the singular Peripatus, which serves as a connecting link between arthropods and worms, and Scolopendrella (Symphyla). These two animals are guide-posts, pointing out, though vaguely to be sure, the way probably trod by the forms, now extinct, which led up to the insects.
9Relations of Peripatus to Insects.—We will first recount the characteristics of this monotypic class. Peripatus (Fig. 4) stands alone, with no forms intermediate between itself and the worms on the one hand, and the true Arthropoda on the other. Originally supposed to be a worm, it is now referred to a class by itself, the Malacopoda of Blainville, or Protracheata of Haeckel. It lives in the tropics, in damp places under decaying wood. In general appearance it somewhat resembles a caterpillar, but the head is soft and worm-like, though it bears a pair of antenna-like tentacles. It may be said rather to superficially resemble a leech with clawed legs, the skin and its wrinkles being like those of a leech. There is a pair of horny jaws in the mouth, but these are more like the pharyngeal teeth of worms than the jaws of arthropods. The numerous legs end each in a pair of claws. The ladder-like nervous system is unlike that of annelid worms or arthropods, but rather recalls that of certain molluscs (Chiton, etc.), as well as that of certain flat and nemertine worms. Its annelid features are the large number of segmentally arranged true nephridia, and the nature of the integument. Its arthropodan features, which appear to take it out of the group of worms, are the presence of tracheæ, of true salivary and slime glands, of a pair of coxal glands (Fig. 4, C, cd) as well as the claws at the end of the legs. The tracheæ, which are by no means the only arthropodan features, are evidently modified dermal glands. The heart is arthropodan, being a dorsal tube lying in a pericardial sinus, with many openings. This assemblage of characters is not to be found in any marine or terrestrial worm.
The tracheæ (Fig. 4, D, tr) are unbranched fine tubes, without a “spiral thread,” and are arranged in tufts, in P. edwardsii opening by simple orifices or pores (“stigmata”) scattered irregularly over the surface of the body; but in another species (P. capensis) some of the stigmata are arranged more definitely in longitudinal rows,—on each side two, one dorsally and one ventrally. “The stigmata in a longitudinal row are, however, more numerous than the pairs of legs.” (Lang.)
The salivary glands, opening by a short common duct into the under side of the mouth, in the same general position as in insects, are evidently, as the embryology of the animal proves, transformed nephridia, and being of the arthropodan type explain the origin and morphology of those of insects. It is so with the slime glands; these, with the coxal glands, being transformed and very large dermal glands. Those of insects arose in the same manner, and are evidently their homologues, while those of Peripatus were probably originally derived from the setiparous glands in the appendages (parapodia) of annelid worms.
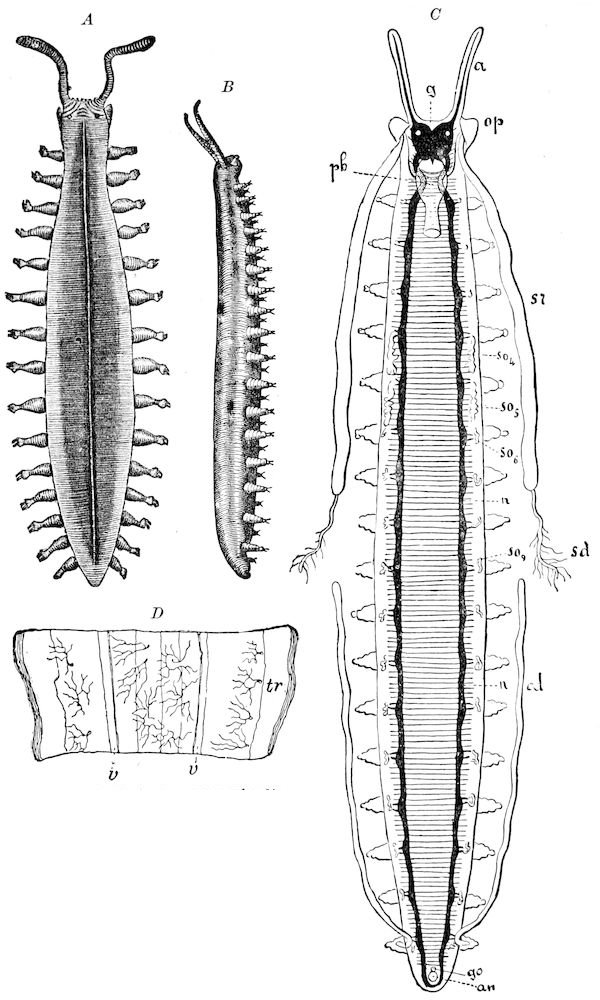
Fig. 4.—A, Peripatus novæ zealandiæ.—After Sedgwick, from Lang. B, Peripatus capensis, side view, enlarged about twice the natural size.—After Moseley, from Balfour. C, Anatomy of Peripatus capensis. The enteric canal behind the pharynx has been removed. g, brain; a, antenna; op, oral or slime papillæ; sd, slime gland; sr, slime reservoir, which at the same time acts as a duct to the gland; so4, so5, so6, so9, nephridia of the 4th, 5th, 6th, and 9th pairs of limbs; cd, elongated coxal gland of the last pair of feet; go, genital aperture; an, anus; ph, pharynx; n, longitudinal trunk of the nervous system.—After Balfour, from Lang. D, Portion of the body of Peripatus capensis opened to show the scattered tufts of tracheæ (tr); v, v, ventral nerve cords.—After Moseley.
11The genital glands and ducts are paired, but it is to be observed that the outlets are single and situated at the end of the body. In the male the ejaculatory duct is single; in its base a spermatophore is formed. It will be seen, then, that Peripatus is not only a composite type, and a connecting link between worms and tracheate arthropods, but that it may reasonably be regarded, if not itself the ancestor, as resembling the probable progenitor of myriopods and insects, though of course there is a very wide gap between Peripatus and the other antennate, air-breathing Arthropoda.
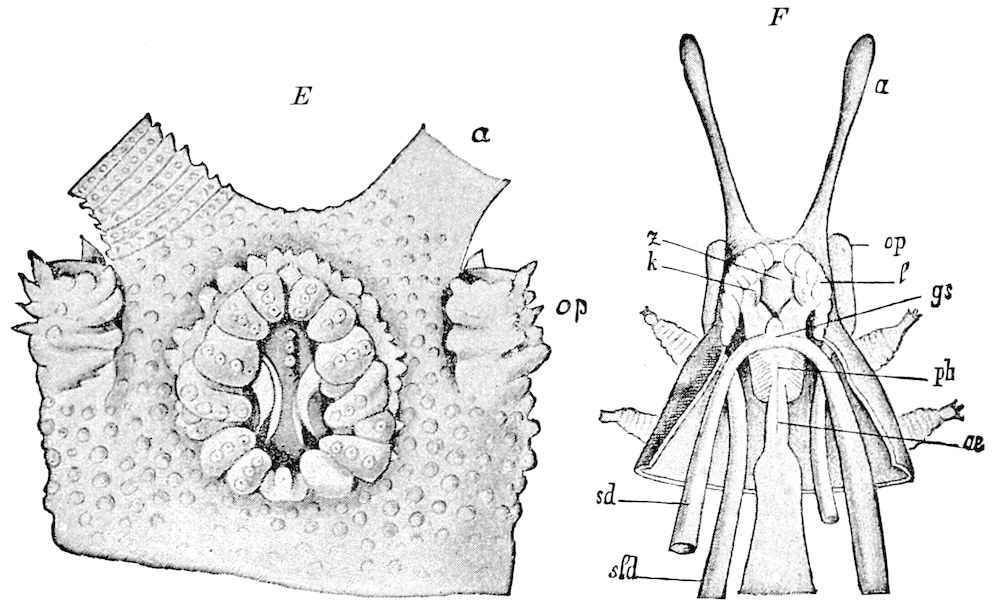
Fig. 4.—E, Peripatus edwardsii, head from the under side: a, base of antenna; op, oral papilla; the figure also shows the papillæ around the mouth, and the four jaws.—After Balfour, from Lang. F, Anterior end of Peripatus capensis, ventral side, laid open: a, antenna; z, tongue; k, jaw; sd, salivary gland; gs, union of the two salivary glands; ph, pharynx; œ, œsophagus; l, lip papillæ around the mouth; op, oral or slime papilla; sld, duct or reservoir of the slime gland.—After Balfour, from Lang.
Relation of Myriopods to Insects.—The Myriopoda are the nearest allies of the insects. They have a distinct head, with one pair of antennæ. The eyes are simple, with the exception of a single genus (Cermatia), in which they are aggregated or compound. The trunk or body behind the head is, as a rule, long and slender, and composed of a large but variable number of segments, of equal size and shape, bearing jointed legs, which invariably end in a single claw.
The mouth-parts of the myriopods are so different in shape and general function from those of insects, that this character, together with the equally segmented nature of the portion of the body behind the head (the trunk), forbids our merging them, as some have been 12inclined to do, with the insects. There are two sub-classes of myriopods, differing in such important respects that by Pocock[1] and by Kingsley they are regarded as independent classes, each equivalent to the insects.
Of these the most primitive are the Diplopoda (Chilognatha), represented by the galley-worms (Julus, etc.).
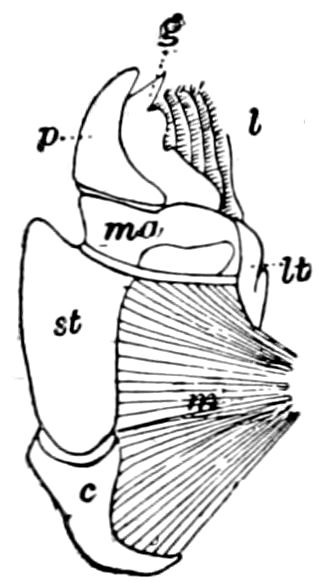
Fig. 5.—Mandible of Julus: l, lacinia; g, galea; p, dens mandibularis; ma, “mala”; lt, lamina tritoria; st, stipes; c, cardo; m, muscle.—After Latzel.
In the typical Diplopoda the head consists of three segments, a preoral or antennal, and two postoral, there being two pairs of jaw-like appendages, which, though in a broad morphological sense homologues of the mandibles and first maxillæ of insects, are quite unlike them in details.
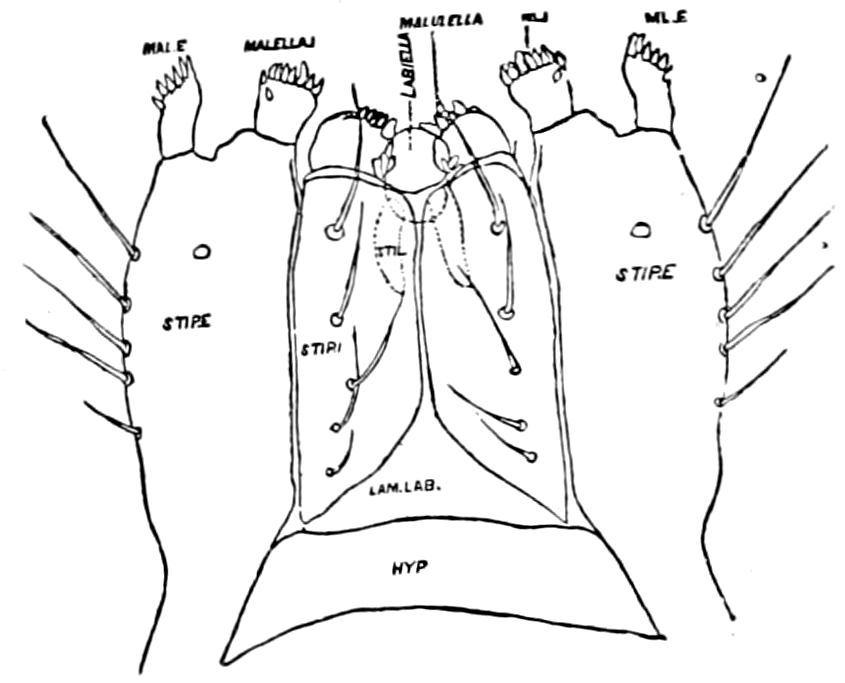
Fig. 6.—Under lip or deutomala of Scoterpes copei: hyp, hypostoma or mentum; lam. lab, lamina labialis; stip. e, stipes exterior; with the malella exterior (mal. e) and malella interior (mal. i); the stipes interior, with the malulella; and the labiella (hypopharynx of Vom Rath) with its stilus (stil.).
As we have previously stated,[2] the so-called “mandibles” of diplopods are entirely different from those of insects, since they appear to be 2– or 3–jointed, the terminal joint being 2–lobed, thus resembling the maxillæ rather than the mandibles of insects, which consist of but a single piece or joint, probably the homologue of the galea or molar joint of the diplopod protomala. The mandible of the Julidæ (Fig. 5, Julus molybdinus), Lysiopetalidæ, and Polydesmidæ consists of three joints; viz. a basal piece or cardo, a stipes, and the mala mandibularis, which supports two lobes analogous to the galea and lacinia of the maxilla of an insect. There is an approach, as we shall see, in the mandible of Copris, to that of the Julidæ, but in insects in general the lacinia is wanting, and the jaw consists of but a single piece.
The deutomalæ (gnathochilarium), or second pair of diplopod jaws, are analogous to the labium or second maxillæ of insects, forming a flattened, plate-like under-lip, constituting the floor of the mouth (Fig. 6). This pair of appendages needs farther study, especially in the late embryo, before it can be fully understood. So far as 13known, judging by Metschnikoff’s work on the embryology of the diplopods, these myriopods seem to have in the embryo but two pairs of post-antennal mouth-parts, which he designated as the “mandibles” and “labium.” Meinert, however, regards as a third pair of mouth-parts or “labium” what in our Fig. 7 is called the internal stipes (stip. i.), behind which is a triangular plate, lamina labialis (lam. lab), which he regards as the sternite of the same segment.
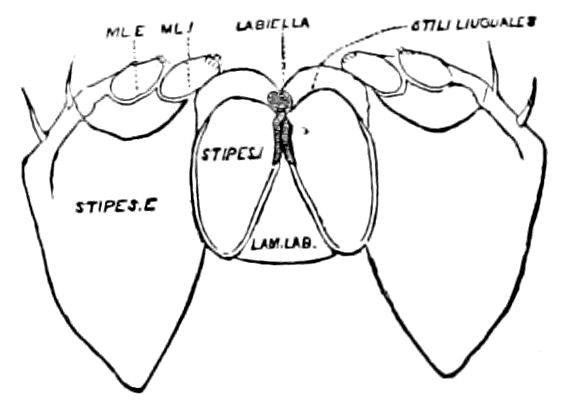
Fig. 7.—Deutomala of Julus, the lettering as in Fig. 6.
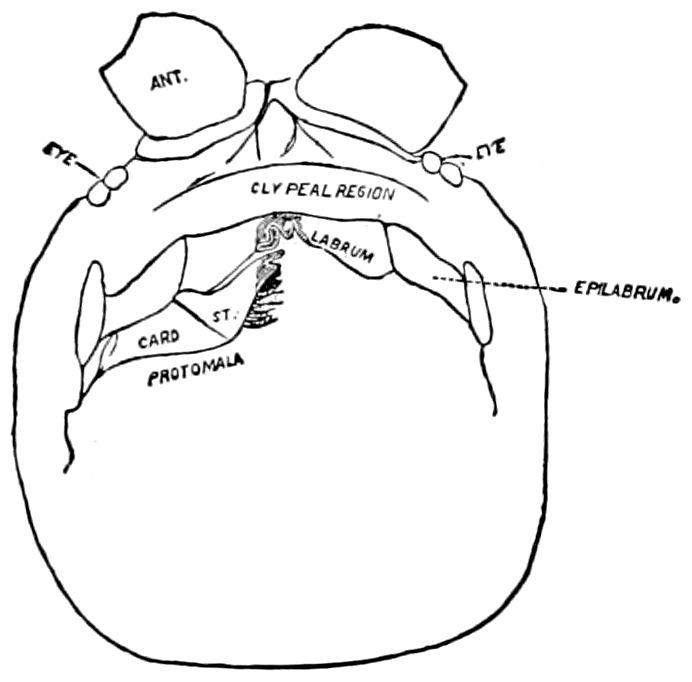
Fig. 8.—Head of Scolopendra, seen from beneath, showing the “mandible” (protomala) with its cardo (card.) and stipes (st.), also the labrum and epilabrum.
The hypopharynx, our “labiella,” (Fig. 6), with the supporting rods or stili linguales (sti. l), of Meinert, are of nearly the same shape as in some insects.
Of the clypeus of insects there is apparently no homologue in myriopods, though in certain diplopods there is an interantennal clypeal region. The labium of insects is represented by a short, broad piece, which, however, unlike that of insects, is immovable, and is flanked by a separate piece called the epilabrum (Fig. 8). Vom Rath has observed an epipharynx, which has the same general relations as in insects.
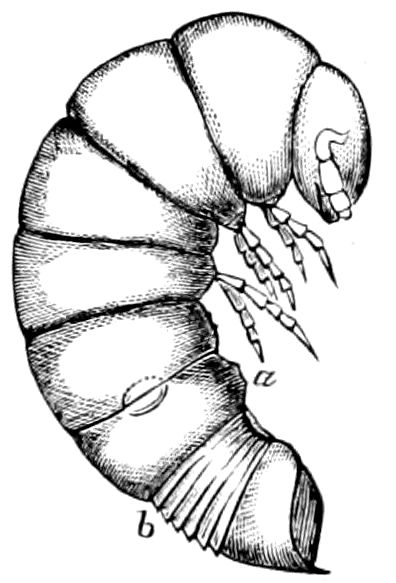
Fig. 9.—Larva of Julus: a, the 3d abdominal segment, with the new limbs just budding out; b, new segments arising between the penultimate and the last segment.—After Newport.
The embryology of myriopods is in many respects like that of insects. The larva of diplopods hatches with but few segments, and with but three pairs of limbs; but these are not, as in insects, appended to consecutive segments, but in one species the third, and in another, Julus multistriatus? (Fig. 10), the second, segment from the head is footless, while Vom Rath represents the first segment of an European Blaniulus as footless, the feet being situated consecutively on segments 2 to 4. The new segments arise at “the growing point” situated between the last and penultimate segment, growing out in groups of sixes (Newport) or in our Julus multistriatus? in fives (Fig. 10). In 14adult life diplopods (Julus) have a single pair of limbs on the three first segments, or those corresponding to the thoracic segments of insects, the succeeding segments having two pairs to each segment.
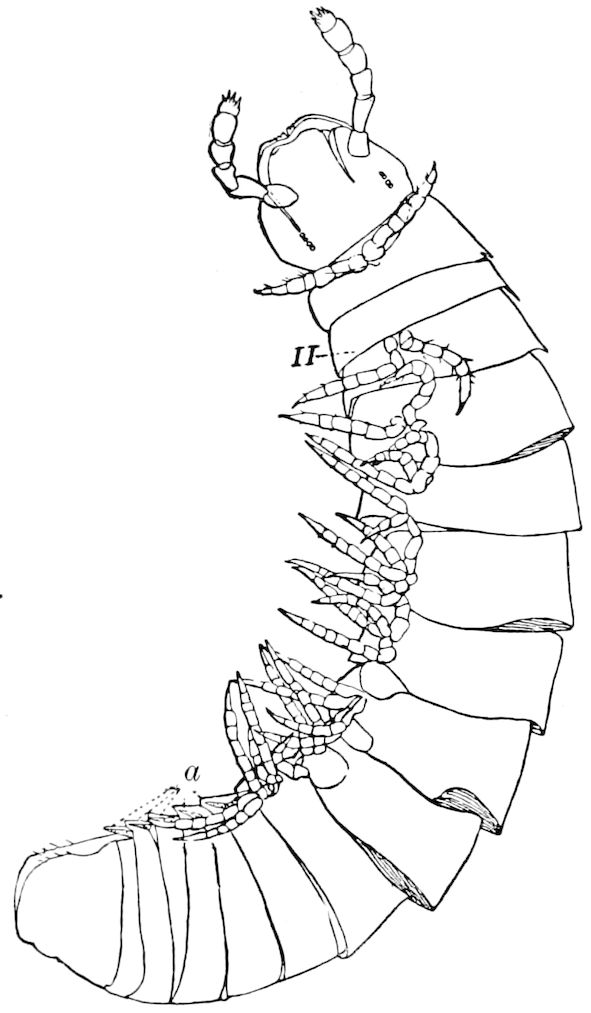
Fig. 10.—Freshly hatched larva of Julus multistriatus? 3 mm. long: a, 5 pairs of rudimentary legs, one pair to a segment.
Sinclair (Heathcote) regards each double segment in the diplopods as not two original segments fused together, nor a single segment bearing two pairs of legs, but as “two complete segments perfect in all particulars, but united by a large dorsal plate which was originally two plates which have been fused together.” (Myriopods, 1895, p. 71.) That the segments were primitively separate is shown, he adds, by the double nature of the circulatory system, the nerve cord, and the first traces of segmentation in the mesoblast. Kenyon believes that from the conditions in pauropods, Lithobius, etc., there are indications of alternate plates (not segments) having disappeared, and of the remaining plates overgrowing the segments behind them, so as to give rise to the anomalous double segments.[3]
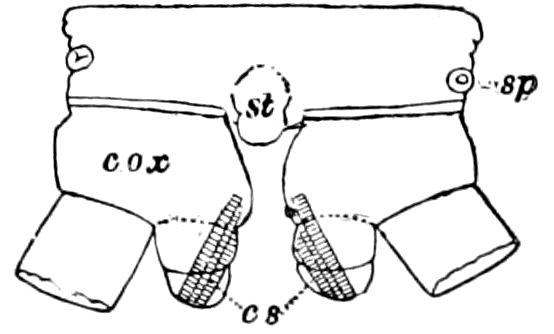
Fig. 11.—Sixth pair of legs of Polyzonium germanicum, ♀: cs, ventral sacs; cox, coxa; st, sternal plate; sp, spiracle.—After Haase.
Diplopods are also provided with eversible coxal sacs, in position like those of Symphyla and Synaptera; Meinert, Latzel, and also Haase having detected them in several species of Chordeumidæ, Lysiopetalidæ, and Polyzonidæ (Fig. 11). In Lysiopetalum anceps these blood-gills occur in both sexes between the coxæ of the third to sixteenth pair of limbs. In the Diplopods the blood-gills appear to be more or less permanently everted, while in Scolopendrella they are usually retracted within the body (Fig. 15, cg).
Diplopods also differ externally from insects in the genital armature, a complicated apparatus of male claspers and hooks apparently arising from the sternum of the sixth segment and being the modified seventh pair of legs. In myriopods 15there are no pleural pieces or “pleurites,” so characteristic of winged insects.
Perhaps the most fundamental difference between diplopods and insects is the fact that the paired genital openings of the former are situated not far behind the head between the second and third pair of legs. Both the oviducts and male ejaculatory ducts are paired, with separate openings. The genital glands lie beneath, while in chilopods they lie above the intestine; this, as Korschelt and Heider state, being a more primitive relation, since in Peripatus they also lie above the digestive canal.
The nervous system of diplopods is not only remarkable for the lack of the tendency towards a fusion of the ganglia observable in insects, but for the fact that the double segments are each provided with two ganglia. The brain also is very small in proportion to the ventral cord, the nervous system being in its general appearance somewhat as in caterpillars.
The arrangement of the tracheæ and stigmata is much as in insects, but in the Diplopoda the tracheary system is more primitive than in chilopods, a pair of stigmata and a pair of tracheal bundles occurring in each segment, while the bundles are not connected by anastomosing branches, branched tracheæ only occurring in the Glomeridæ. The tracheæ themselves are without spiral threads (tænidia). It is noteworthy that the tracheæ arise much later than in insects, not appearing until the animal is hatched; in this respect the myriopods approximate Peripatus.
In the Chilopoda also the parts of the head, except the epicranium, are not homologous with those of insects, neither are the mouth-parts, of which there are five pairs.
The structure of the head of centipedes is shown in part in Fig. 12, compare also Fig. 8. It will be seen that it differs much from that of the diplopods, though the mandibles (protomalæ) are homologous; they are divided into a cardo and stipes, thus being at least two-jointed.
The second pair of postoral appendages is in centipedes very different from the gnathochilarium of diplopods. As seen in Fig. 12 2, they are separate, cylindrical, fleshy, five-jointed appendages, the maxillary appendages of Newport, which are “connected transversely at their base with a pair of soft appendages” (c), the lingua of Newport. The third and fourth pair are foot-jaws, and we have called them malipedes, as they have of course no homology with the maxillipedes of Crustacea. The second pair of these malipedes, forming the last pair of mouth-appendages, is the poison-fangs (4), 16which are intermediate between the malipedes and the feet; Meinert does not allow that these are mouth-appendages.
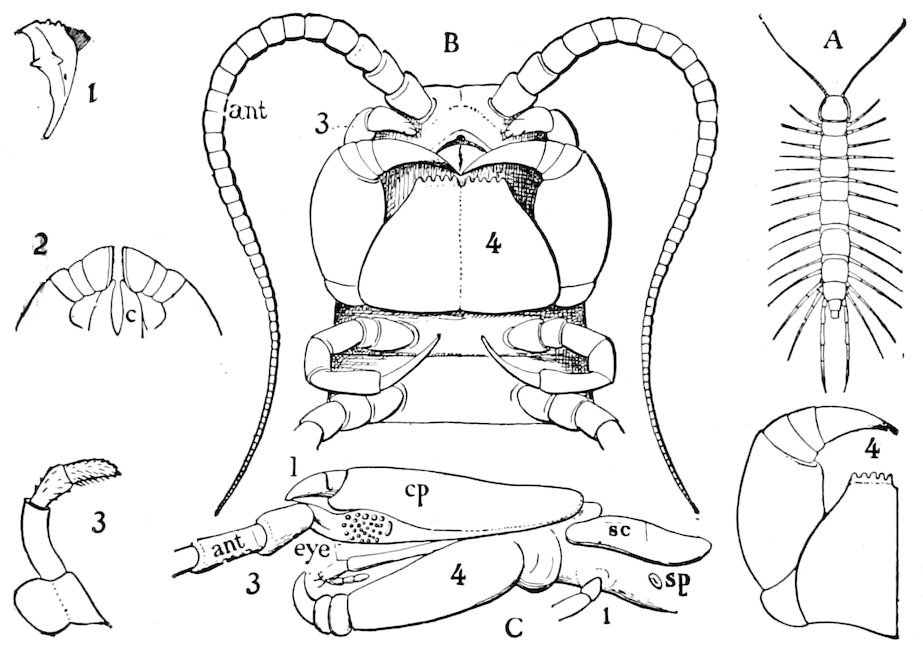
Fig. 12.—Structure of a chilopod. A, Lithobius americanus, natural size. B, under side of head and first two body-segments and legs, enlarged: ant, antenna; 1, jaws; 2, first accessory jaw; c, lingua; 3, second accessory jaw and palpus; 4, poison-jaw. (Kingsley del.) C, side view of head (after Newport): ep, epicranium; l, frontal plate; sc, scute; 1, first leg; sp, spiracle.
The embryology of Geophilus by Metschnikoff shows plainly the four pairs of post-antennal appendages. The embryo Geophilus is hatched in the form of the adult, having, unlike the diplopods, no metamorphosis, its embryological history being condensed or abbreviated. But in examining Metschnikoff’s figures certain primitive diplopod features are revealed. The body of the embryo shortly before hatching is cylindrical; the sternal region is much narrower than in the adult, hence the insertions of the feet are nearer together, while the first six pairs of appendages begin to grow out before the hinder ones. Thus the first six pairs of appendages of the embryo Geophilus correspond to the antennæ, two pairs of jaws, and three pairs of legs of the larval Julus. These features appear to indicate that the chilopods may be an offshoot from the diplopod stem. The acquisition of a second pair of legs to a segment in diplopods, as in the phyllopod Crustacea, is clearly enough a secondary character, as shown by the figures of Newport in his memoir on the development of the Myriopoda (Pl. IV.). Thus the tendency in the Myriopoda, both diplopods and chilopods, is towards the multiplication of segments and the elongation of the body, while in insects the polypodous embryo has the three terminal segments of the 17abdomen well formed, these being, however, before hatching, partly atrophied, so that the body of insects after birth tends to become shortened or condensed. This indicates the descent of insects from ancestors with elongated polypodous hind-bodies like Scolopendrella. Korschelt and Heider suggest that the stem-form of myriopods was a homonomously jointed form like Peripatus, consisting of a rather large number of segments, but we might, with Haase, consider that the great number of segments which we now find indicates a late acquisition of this form.
The genital opening in chilopods is single, and situated in the penultimate segment of the body, as in insects. While recognizing the close relationship of the Myriopoda with the insects, it still seems advisable not to unite them into a single group (as Oudemans, Lang, and others would do), but to regard them as forming an equivalent class. On the other hand, when we take into account the form and structure of the head, antennæ, and especially the shape of the first pair of mouth-appendages, being at least two-jointed in both groups, we think these characters, with the homonomously segmented body behind the head, outweigh the difference in the position of the genital outlet, important as that may seem. It should also be taken into account that while insects are derived from polypodous ancestors, no one supposes, with the exception of one or two authors, that these ancestors are the Myriopoda, the latter having evidently descended from a six-legged ancestor, quite different from that of the Campodea ancestor of insects.[4]
In regard to the sexual openings of worms, though their position is in general in the anterior part of the body, it is still very variable, though, in general, paired. In the oligochete worms the genital zone, with the external openings, is formed by the segments lying between the 9th and 14th rings, though in some the genital organs are situated still nearer the head. The myriopods, which evolved from the worms earlier than insects, appear to have in their most primitive forms (the Diplopoda) retained this vermian position of the genital outlets. In the later forms, the chilopods, the genital openings have been carried back to near the end of the body, as in insects. From observations made by three different observers on the freshly hatched larva of the Julidæ, it appears that the ancestral diplopods were six-footed, or oligopod, the larva of Pauropus 18(Fig. 13) approaching nearest to our idea of the ancestral myriopod, which might provisionally be named Protopauropus.
Relations of the Symphyla to Insects.—Opinions respecting the position of the Symphyla, represented by Scolopendrella (Fig. 14), are very discordant. By most writers since Newport, Scolopendrella has been placed among the myriopods. The first author, however, to examine its internal anatomy was Menge (1851), who discovered among other structures (tracheæ, etc.) the silk-glands situated in the last two segments, and which open at the end of each cercus. He regarded the form as “the type of a genus or family intermediate between the hexapod Lepismidæ and the Scolopendridæ.”
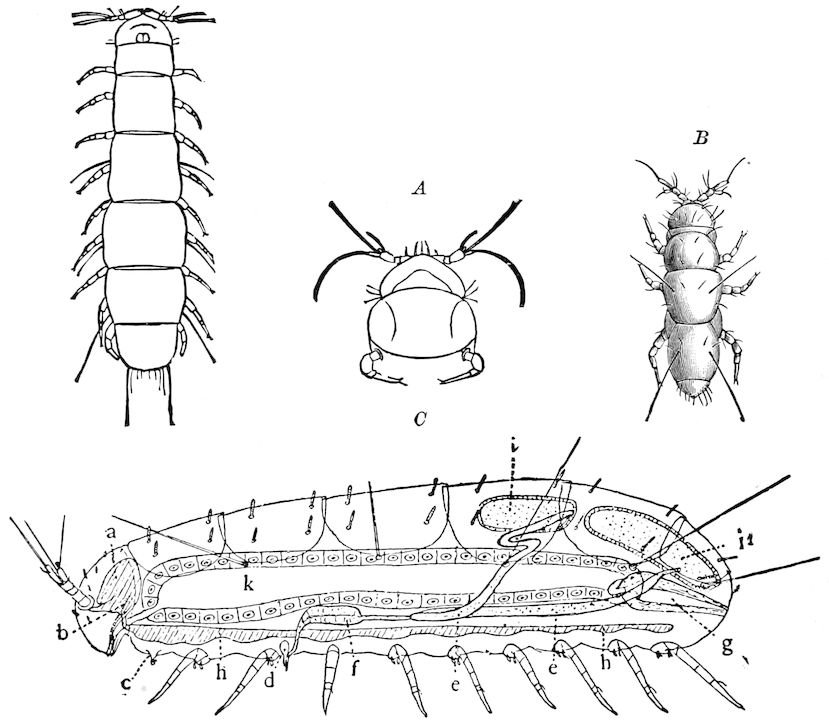
Fig. 13.—Pauropus huxleyi, much enlarged. A, enlarged view of head, antennæ, and first pair of legs (original). B, young.—After Lubbock. C, longitudinal section of Pauropus huxleyi, ♂: a, brain; b, salivary gland; k, mid-intestine; g, rectum; h, ventral nerve-cord; c, bud-like remnants of coxæ; d, penis; e, vesicula seminalis; f, ductus glandularis; i1, divisions of testes.—After Kenyon.
In 1873[5] the writer referred to this form as follows: “It may be regarded as a connecting link between the Thysanura and Myriopoda, 19and shows the intimate relation of the myriopods and the hexapods, perhaps not sufficiently appreciated by many zoölogists.”
In 1880 Ryder regarded it as “the last survival of the form from which insects may be supposed to have descended,” and referred it to “the new ordinal group Symphyla, in reference to the singular combination of myriopodous, insectean, and thysanurous characters which it presents.[6]”
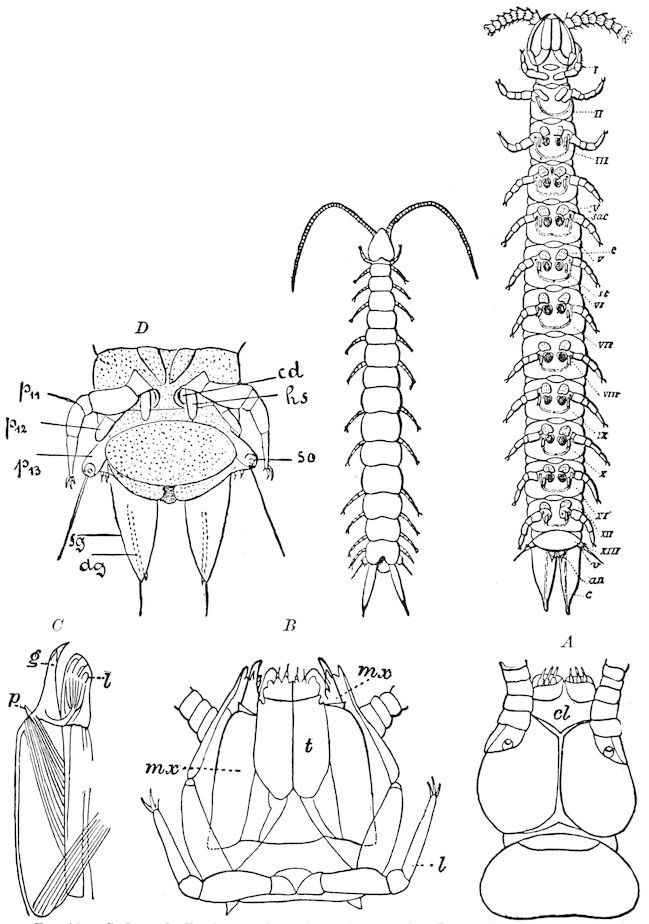
Fig. 14.—Scolopendrella immaculata, from above,—after Lang; also from beneath, the genital opening on the 4th trunk-segment: sac, eversible or coxal sac; an, anus; c, cereopod; v, vestigial leg.—After Haase, from Peytoureau. A B C, head and buccal appendages of Scolopendrella immaculata: A, head seen from above; cl, clypeus. B, head from beneath; l, first pair of legs; mx, 1st maxilla; mx1, 2d maxilla; t, “labial plates” of Latzel, labium of Muhr. C, 1st maxilla; l, lacinia; g, galea; p, rudiment of the palpus.—After Latzel. D, end of the body: p11, eleventh, p12, twelfth undeveloped pair of legs; p13, modified, vestigial legs, bearing tactile organs (so); sg, cercopod, with duct of spinning gland, dg; cd, eversible or coxal gland; h8s, coxal spur of the 11th pair of legs.—After Latzel from Lang.
Wood-Mason considered it to be a myriopod, and “the 20descendant of a group of myriopods from which the Campodeæ, Thysanura, and Collembola may have sprung.” We are indebted to Grassi for the first extended work on the morphology of Scolopendrella (1885). In 1886 he added to our knowledge facts regarding the internal anatomy, and gives a detailed comparison with the Thysanura, besides pointing out the resemblances of Scolopendrella to Pauropus, diplopods, chilopods, as well as Peripatus.
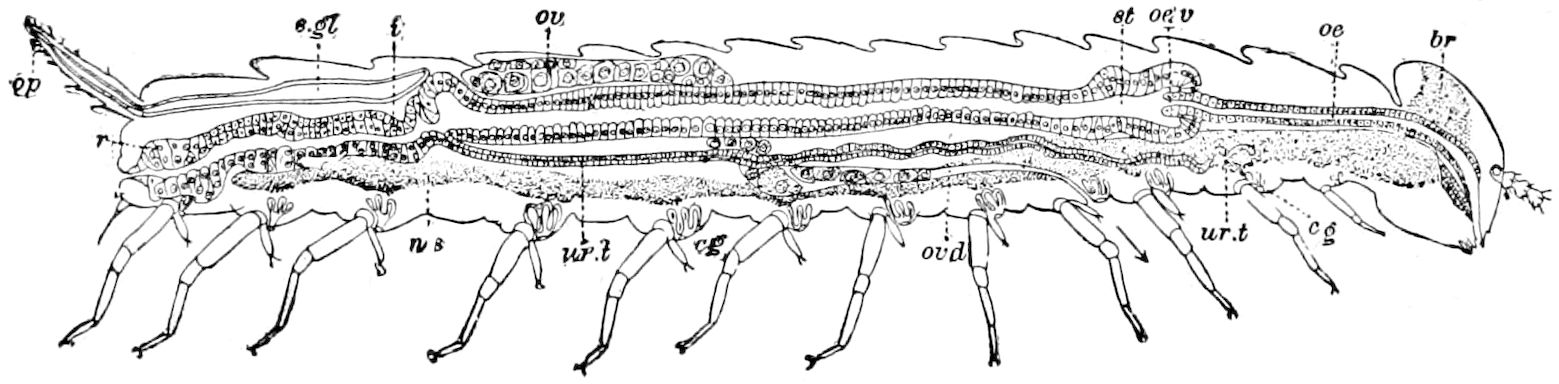
Fig. 15.—Section of Scolopendrella immaculata: œ, œsophagus; oe. v, œsophageal valve entering the mid-intestine (“stomach”); i, intestine; r, rectum; br, brain; ns, abdominal chain of ganglia; ovd, oviduct; ov, ovary; s. gl, silk-gland, and op, its outer opening in cercus, ur. t, urinary tube; cg, coxal glands or blood-gills.—Author del.
In 1888 Grassi expressed his view as to the position of the Symphyla, stating that it should not be included in the Thysanura, since it evidently has myriopod characters; these being the supraspinal vessel, the ventral position of the genital glands; the situation of the genital opening in the fourth segment of the trunk, its ganglionic chain being like that of diplopods, its having limbs on all the segments, etc. On the other hand, Grassi has with much detail indicated the points of resemblance to the Thysanura. The principal ones are the thin integument, the want of sympathetic ganglia, the presence of a pair of cephalic stigmata, like that said to occur in certain Collembola, and in the embryo of Apis; two endoskeletal processes situated near the ventral fascia of the head; the epicranial suture also occurring in Thysanura, Collembola, Orthoptera, and other winged insects, and being absent in diplopods and chilopods. He also adds that the digestive canal both in Symphyla and Thysanura is divided into three portions; the malpighian tubes in Thysanura present very different conditions (there being none in Japyx), among which may be comprised those of Scolopendrella. In both groups there is a single pair of salivary glands. The cellular epithelium of the mid-intestine of Scolopendrella is of a single form as in Campodea and Japyx. The fat-body, dorsal vessel, with its valves and ostia, are alike in the two groups, as are the appendages of the end of the abdomen, the anal cerci (cercopoda) of Scolopendrella being the homologues of the 21multiarticulate appendages of Lepisma, etc., and of the forceps of Japyx. In those of Scolopendrella, we have found the large duct leading from the voluminous silk-gland, a single large sac extending forwards into the third segment from the end of the body (Fig. 15, s. gl). Other points of resemblance, all of which he enumerates, are the slight differences in the number of trunk-segments, the presence in the two groups of the abdominal “false-legs” (parapodia), the dorsal plate, and the mouth-parts. As regards the latter, Grassi affirms that there is a perfect parallelism between those of Scolopendrella and Thysanura. To this point we will return again in treating more especially of those of the Symphyla. Finally, Grassi concludes that there is “a great resemblance between the Thysanura and Scolopendrella.” He, however, believed that the Symphyla are the forerunners of the myriopods, and not of the insects, his genealogical tree representing the symphylan and thysanuran phyla as originating from the same point, this point also being, rather strangely, the point of origin of the arachnidan phylum.
Haase (1889) regarded Scolopendrella as a myriopod, and Pocock (1893) assigned the Symphyla to an independent class, regarding Scolopendrella as “the living form that comes nearest to the hypothetical ancestor of the two great divisions of tracheates.” Schmidt’s work (1895) on the morphology of this genus is more extended and richly illustrated than Grassi’s, his method of research being more modern. He also regards this form as one of the lower myriopods.
In conclusion, it appears to us that, on the whole, if we throw out the single characteristic of the anteriorly situated genital opening, the ovarian tubes being directed toward the end of the body (Fig. 15, ovd, ov), there is not sufficient reason for placing the Symphyla among the Myriopoda, either below or near the diplopods. This is the only valid reason for not regarding Scolopendrella as the representative of a group from which the insects have descended, and which partly fills the wide abyss between Peripatus and insects. With the view of Pocock, that both insects and myriopods have descended from Scolopendrella, we do not agree, because this form has so many insectean features, and a single unpaired genital opening. For the same reason we should not agree with Schmidt in interpolating the Symphyla between the Pauropoda and Diplopoda. In these last two progoneate groups the genital openings are paired, hence they are much more primitive types than Scolopendrella, in which there is but a single opening. It seems most probable that the Symphyla, though progoneate, are more recent forms than the 22progoneate myriopods, which have retained the primitive feature of double sexual outlets. It is more probable that the Symphyla were the descendants of these polypodous forms. Certainly Scolopendrella is the only extant arthropod which, with the sole exception of the anteriorly situated genital opening, fulfils the conditions required of an ancestor of Thysanura, and through them of the winged insects. No one has been so bold as to suggest the derivation of insects from either diplopods or chilopods, while their origin from a form similar to Scolopendrella seems not improbable. Yet Uzel has very recently discovered that Campodea develops in some respects like Geophilus, the primitive band sinking in its middle into the yolk, with other features as in chilopods.[7] The retention of a double sexual opening in the diplopods is paralleled by the case of Limulus with its double or paired sexual outlets, opening in a pair of papillæ, as compared with what are regarded as the generalized or more primitive Crustacea, which have an unpaired sexual opening.
The following summary of the structural features of the Symphyla, as represented by Scolopendrella, is based mainly on the works of Grassi, Haase, and Schmidt, with observations of my own.
Diagnostic or essential characters of Symphyla.—Head shaped as in Thysanura (Cinura), with the Y-shaped tergal suture, which occurs commonly in insects (Thysanura, Collembola, Dermaptera, Orthoptera, Platyptera, Neuroptera, etc.), but is wanting in Myriopoda (Diplopoda and Chilopoda); antennæ[8] unlike those of Myriopoda in being very long, slender, and moniliform. Clypeus distinct. Labrum emarginate, with six converging teeth. Mandibles 2–jointed, consisting of a vestigial stipes and distal or molar joint, the latter with eight teeth. First maxillæ with an outer and inner mala situated on a well-developed stipes; with a minute, 1–jointed palpus. Second pair of maxillæ: each forming two oblong flat pieces, median sutures distinct, with no palpi; these pieces are toothed in front, and appear to be homologous with the two 23median pieces of the gnathochilarium of Diplopoda. Hypopharynx? Epipharynx?
Trunk with from fifteen to sixteen dorsal, more or less free subequal scutes, the first the smallest. Pedigerous segments twelve; also twelve pairs of 5–jointed legs, which are of nearly equal length, the first pair 4–, the others 5–jointed, all ending in two claws, as in Synaptera and winged insects. A pair of 1–jointed anal cerci homologous with those of Thysanura and Orthoptera, into each of which opens a large abdominal silk-gland. Abdominal segments with movable styles or “pseudopods” (“Parapodia” of Latzel and of Schmidt), like those of Campodea and Machilis, and situated on the base of the coxal joint in front of the ventral sac. Within the body near the base of each abdominal style is an eversible coxal sac or blood-gill (Fig. 15, cg). The single genital opening is on the fourth trunk-segment in both sexes (Fig. 15, indicated by the arrow). The malpighian tubes (ur. t) are two in number, opening into the digestive canal at the anterior end of the hind intestine; they extend in front to the third or second segment from the head. They are broad and straight at their origin, becoming towards the end very slender and convoluted.
The three divisions of the digestive tract are as in insects, the epithelium of the mid-gut being histologically as in Campodea and Japyx; rectal glands are present. A pair of very large salivary glands are situated in the first to the fourth trunk-segments, consisting of a glandular portion with its duct, which unite into a common duct opening on the under side of the head, probably in the labium.
But a single pair of stigmata is present, and these are situated in the front of the head, beneath the insertion of the antennæ and within the stipes of the mandibles; the tracheæ are very fine, without spiral threads (tænidia), and mostly contained within the head, two fine branches extending on each side into the second trunk-segment.
After birth the body increases in length by the addition of new segments at the growing point.
In respect to the nervous system, there are no diagnostic characters; there are, however, not as many as two pairs of ganglia to a segment. The brain is well developed, sending a pair of slender nerves to the small eyes. The ganglia of the segment bearing the first pair of legs is fused with the subœsophageal ganglion. Grassi was unable to detect a true sympathetic system, but he suspects the existence of a very small frontal ganglion.
The slender dorsal vessel, provided with ostia and valvules, pulsates along the entire length of the trunk; an aorta passes into the head.
24The internal genital organs of both sexes are paired, and extend along the greater part of the trunk; in either sex they may be compared to two long, slender, straight cords extending from the fourth to the tenth pair of legs. The two oviducts do not unite before reaching the sexual opening (Fig. 15, ovd).
The male sexual organs are more complicated than the feminine. The paired testicular tubes lie in trunk-segments 6 to 12, on each side, and partly under the intestinal canal, communicating with each other by a cross-anastomosis situated under the intestine, and which, like the testes, is filled with sperm. Of the paired seminal ducts (vas deferens) in trunk-segment 4, each unites again into a thick tube, sending a blind tube forward into the third segment. Under the place of union of the two vasa deferentia arise the paired ductus ejaculatorii, which open beneath in the uterus masculinus. The anterior blind ends of the vasa deferentia form a sort of small paired vesiculæ seminales in which a great quantity of ripe sperm is stored. The uterus masculinus is in its structure homologous with the evaginable penis of Pauropus, Polyxenus, and some diplopods, and the sexual opening has without doubt become secondarily unpaired. The sexual opening is rather long and is closed by two longitudinal folds. “In several respects the male sexual organs of Scolopendrella are like those of Pauropus; in the last-named form we have indeed an unpaired testis, but also in Scolopendrella we see the beginning of such a singleness; namely, the presence of an anastomosis uniting the two tubes, their communication by means of a transverse connecting canal and a glandular structure in the epithelium forming them. The male sexual organs of Pauropus differ only through a still greater complication.” (Schmidt.)
Scolopendrella in habits resembles chilopods, being found in company with Geophilus burrowing deep in light sand under leaves, or living at the surface of the ground under sticks or stones. It is very agile in its movements, and is probably carnivorous. It was considered by Haase to be eyeless, but the presence of two ocelli has been demonstrated both by Grassi and by Schmidt. Whether the pigment and corneous facet are present is not certain. The embryology is entirely unknown (although Henshaw reports finding a hexapodous young one), and it need not be said that a knowledge of it is a very great desideratum. It is most probable that the young is hexapodous, since the first pair of limbs are 4–jointed, all the rest 5–jointed; while Newport, and also Ryder, observed specimens with nine, ten, eleven, and twelve pairs, and Wood-Mason confirms their observations, “which prove that a pair of legs 25is added at each moult,” and he concludes that the addition of new segments “therefore takes place in this animal by the intercalation of two at each moult between the antepenultimate and penultimate sterna, as in the Chilognatha, and as also in some of the Chilopoda.”
There is but one family, Scolopendrellidæ, and a single genus, Scolopendrella, which seems to be, like other archaic types, cosmopolitan in its distribution.
Our commonest species is S. immaculata Newport, which occurs from Massachusetts to Cordova, Mexico, and in Europe from England to the Mediterranean and Russia; Mr. O. F. Cook tells me he has found a species in Liberia, West Africa. The other species are S. notacantha Gervais, Europe and Eastern United States; S. nivea Scopoli (S. gratiæ Ryder), Europe and United States; S. latipes Scudder, Massachusetts.
LITERATURE ON SCOLOPENDRELLA
Newport, George. Monograph of the class Myriopoda, order Chilopoda. (Trans. Linn. Soc. xix, pp. 349–439, 1 Pl., 1845.)
Menge, A. Myriapoden der Umgegend von Danzig. (Neuste Schriften der naturforsch. Gesell. Danzig. iv, 1851.)
Ryder, John H. Scolopendrella as the type of a new order of articulates (Symphyla). (Amer. Nat., May, 1880, xiv, pp. 375, 376.)
—— The structure, affinities, and species of Scolopendrella. (Proc. Acad. Nat. Soc. Phil., pp. 79–86, 1881, 2 Figs.)
Packard, A. S. Scolopendrella and its position in nature. (Amer. Nat., 1881, pp. 698–704, Fig.)
Muhr, Jos. Die Mundtheile von Scolopendrella und Polyzonium. Prag, 1882, 1 Pl.
Mason, J. Wood. Morphological notes bearing on the origin of insects. (Trans. Ent. Soc. London, 1879, pp. 145–167, Figs.)
—— Notes on the structure, post-embryonic development, and systematic position of Scolopendrella. (Ann. and Mag. Nat. Hist., July, 1883, pp. 53–63.)
Latzel, Robert. Die Myriapoden der osterreichisch-ungarischen Monarchie, ii, Wien, 1884, pp. 1–39, Pls.
Haase, Erich. Die Abdominalanhänge der Insekten mit Berücksichtigung der Myriapoden. (Morph. Jahrbuch, xv, pp. 331–435, 2 Pls., 1889.)
Schmidt, Peter. Beiträge zur Kenntnis der niederen Myriapoden. (Zeits. f. wissen. Zool., lix, pp. 436–510, 2 Pls., 1895.)
INSECTA (HEXAPODA)
We are now prepared to discuss the fundamental or essential characters of the insects, including the wingless subclass (Synaptera), and the winged (Pterygota).
Diagnostic characters of insects.—Body consisting of not more than twenty-one segments, which are usually heteronomous or of unequal size and shape, arranged in three usually well-defined regions; i.e. a head, thorax, and hind-body or abdomen. Head small and flattened or rounded, composed of not less than six segments, and bearing, besides the eyes, at least four pairs of jointed appendages; i.e. one pair of antennæ, and three pairs of masticatory appendages, the distal or molar portion of which is primarily divided into three divisions, supported on a stipes and cardo, and in certain orders modified into piercing or sucking structures. The head is composed of an epicranium, bearing a distinct clypeus and labrum, with the epipharynx. Mandibles 1–jointed, without a palpus and very generally with no, or uncertain, traces of a lacinia and a stipes. Two pairs of maxillæ; the first pair separate, usually 3–lobed, comprising a lacinia, galea, and palpifer, with a palpus which is never more than 6–jointed. The second pair united to form the labium or under lip, composed of two laciniæ fused together; in the generalized forms with a rudimentary galea; bearing a pair of palpi, never more than 4–jointed; with paraglossæ sometimes present.
(A third pair of mouth-appendages situated between the antennæ and mandibles in the embryo of Anurida, and Apis, and adult Campodea.)
The epipharynx forming the roof of the mouth, and bearing gustatory organs. Hypopharynx usually well developed, lying on the under side of the mouth, just above the labium, and receiving the end of the salivary duct.
Eyes of two kinds: a pair of compound, and from two to three simple eyes (ocelli).
The thorax consisting of three segments, the two latter segments in the winged orders highly differentiated into numerous tergal and lateral pieces and a single sternum; in the Synaptera the segments are undivided. (In the higher Hymenoptera the basal abdominal segment coalesced with the thorax.) Three pairs of legs, each foot ending in a pair of claws. Two pairs of wings (except in the Synaptera), a pair to each of the two hinder thoracic segments; the wings occasionally reduced or wanting in certain adaptive forms, which, however, had winged ancestors.
Abdomen consisting at the most of from ten to twelve segments. No 27functional abdominal legs except in the Thysanura, and in the larvæ of Lepidoptera. A pair of 1– or many-jointed cercopods on the tenth segment; and in certain forms a pair of styles on the ninth segment. In certain orders an ovipositor or sting formed of three pairs of styliform processes; in Collembola a single pair of processes forming the elater.
The genital openings opisthogoneate, usually single, but paired in Thysanura (Lepisma), Dermaptera, and Plectoptera (Ephemeridæ).
The digestive canal in the winged orders is highly differentiated, the fore-intestine being divided into an œsophagus and proventriculus, the hind-intestine into an ileum, colon, and rectum, with rectal glands.
The nervous system consists of a well-developed brain, in the more specialized orders highly complicated; no more than thirteen pairs of ganglia, which may be more or less fused in the more specialized orders. Three frontal ganglia, and a well-developed, sympathetic system present.
Stigmata confined (except possibly in Sminthurus) to the thorax and abdomen, not more than ten pairs in all, and usually but nine pairs. Tracheal system as a rule highly differentiated; invariably with tænidia.
Dorsal vessel with ostia and valvules; no arteries except the cephalic aorta; no veins. After birth there is in the more specialized pterygote orders a reduction in the number of terminal segments of the abdomen.
Development either direct (Synaptera), or with an incomplete (with nymph and winged or imaginal stages), or complete metamorphosis; in the latter case with a larval, pupal, and imago stage.
The insects may be divided into two sub-classes,—the Synaptera, and the winged orders, Pterygota, of Gegenbaur (1877), since the differences between the two groups appear on the whole to be of more than ordinal rank.
1. EXTERNAL ANATOMY
a. The regions of the body
The insects differ from other arthropods in that the body is divided into three distinct regions,—the head, thorax, and abdomen, the latter regions in certain generalized forms not always very distinctly differentiated. The body behind the head may also conveniently be called the trunk, and the segments composing it the trunk-segments.
In insects the head is larger in proportion to the trunk than in other classes, notably the Crustacea; the thorax is usually slightly or somewhat larger than the head, while the hind-body or abdomen 28is much the larger region, as it consists of ten to eleven, and perhaps in the Dermaptera and Orthoptera twelve, segments, and contains the mid- and hind-intestine, as well as the reproductive organs.
When we compare the body of an insect with that of a worm, in which the rings are distinctly developed, we see that in insects ring distinctions have given way to regional distinctions. The segments lose their individuality. It is comparatively easy to trace the segments in the hind-body of an insect, as in this region they are least modified; so with the thorax; but in the head of the adult insect it is impossible to discover the primitive segments, as they are fused together into a sort of capsule, and have almost entirely lost their individuality.
In general it may be said that the head contains or bears the organs of sense and of prehension and mastication of the food; the thorax the organs of locomotion; and the abdomen those of reproduction.
When we compare the body of a wasp or bee with that of a worm, we see that there is a decided transfer of parts headward; this process of cephalization so marked in the Crustacea likewise obtains in insects. Also the two hinder regions of the body are, in a much greater degree than in worms, governed by the brain, the principal seat of the intelligence, which, so to speak, dominates and unifies the functions of the body, both digestive, locomotive, and reproductive, as also those of the muscles moving the different segments and regions of the body. To a large extent arthropodan morphology and class distinctions are based on the regional arrangement of the somites themselves. Thus in the process of grouping of the segments into the three regions, some increase in size, while others undergo a greater or less degree of reduction; one segment being developed at the expense of one or more adjoining ones. This principle was first pointed out by Audouin, and is called Audouin’s law. It is owing to the greater development of certain segments and the reduction of others, both of the body-segments and of the segments of the limbs, that we have the wonderful diversity of form in the species and genera, and higher groups of insects, as well as those of other arthropods.
b. The integument (exoskeleton)
The skin or integument of insects consists, primarily, as in worms and all arthropods, of an epithelial layer of cells called the hypodermis. This layer secretes the cuticle, which is of varying thickness 29and flexibility, and is usually very dense, impermeable, and light, compared with the crust of the Crustacea, where the cuticle becomes heavy and solid by the deposition of the carbonate and phosphate of lime. This is due to the presence of a substance called by Odier chitin.[9] The cuticle is thin, delicate, and flexible between the joints; it is likewise so in such diaphanous aquatic larvæ as that of Corethra, and in the gills of aquatic insects, also in the walls of the tracheæ and of the salivary ducts. The cuticle thus forms a more or less solid crust which is broken into joints and pieces (sclerites), forming supports for the attachments of the muscles and serving to protect the soft parts within.
Chitin.—If we allow an insect to soak for a long time in acids, or boil it in liquid potassa or caustic potash, the integument is not affected. The muscles and the other soft parts are dissolved, leaving the cuticle clear and transparent. This insolubility of the cuticle is due to the presence of chitin, the insoluble residue left after such treatment. It also resists boiling in acids, in any alkalies, alcohol or ether. The chemical formula is C15H26N2O10.[10]
“Chitin forms less than one-half by weight of the integument, but it is so coherent and uniformly distributed that when isolated by chemical reagents, and even when cautiously calcined, it retains its original organized form. The color which it frequently exhibits is not due to any essential ingredient; it may be diminished or even destroyed by various bleaching processes.” (Miall and Denny.)
“The chemical stability of chitin is so remarkable that we might expect it to accumulate like the inorganic constituents of animal skeletons, and form permanent deposits. Schlossberger (Ann. d. chem. u. pharm., bd. 98) has, however, shown that it changes slowly under the action of water. Chitin kept for a year under water partially dissolved, turned into a slimy mass, and gave off a peculiar smell. This looks as if it were liable to putrefaction. The minute proportion of nitrogen in its composition may explain the complete disappearance of chitin in nature.” (Miall and Denny, The Cockroach, p. 29.)
Chitin, or a substance closely similar to it, occurs in worms and in their tubes, especially in the pharyngeal teeth of annelids and in their setæ. The shell of Lingula and the pen of cuttle-fish contain true chitin (Krukenberg). The integument of Limulus, of trilobites, and of Arachnida, as well as Myriopoda, appears to consist of chitin.[11]
The chitin is rapidly deposited at the end of embryonic life, also during the larval and pupal stages. As is well known, insects after 30moulting are white, but in a few hours turn dark, and those which live in total darkness are white, showing that light has a direct effect in causing the dark color of the integument.
Moseley analyzed one pound weight of Blatta, and found plenty of iron with a remarkable quantity of manganese.
Schneider regarded chitin as a hardening of the protoplasm rather than a secretion, and the cuticle is looked upon as an exudation. It is structureless, not consisting of cells, and consists of fine irregular laminæ. “A cross-section of the chitinous layer or ‘cuticle’ examined with a high power shows extremely close and fine lines perpendicular to the laminæ.” In the cockroach the free surface of the cuticle is divided into polygonal, raised spaces or areas which correspond each to a chitinous cell of the hypodermis. (Miall and Denny.)
Numerous pore-canals pass through the cuticle of all the external parts of the body. The larger canals nearly always form the way for the passage of secretions from dermal cells, or connect with the cavities of hairs or setæ; when very fine and not connected with hairs or scales, they are either empty or filled with air, and may possibly serve for respiration.
Vosseler distinguishes in the cuticle two layers of different physical and chemical characters. Besides the external chitinous layer there is an inner layer which entirely agrees with cellulose. (Zool. Centralblatt, ii, 1895, p. 117.)
The reparative nature of chitin is seen in the fact that Verhoeff finds that a wound on an adult Carabus, and presumably on other insects, is speedily closed, not merely by a clot of blood, but by a new growth of chitin.
c. Mechanical origin and structure of the segments (somites, arthromeres, metameres, zonites)
The segments are merely thickenings of the skin connected by folds or duplications of the integument, and not actually separate or individual rings or segments. This is shown by longitudinal (sagittal) sections through the body, and also by soaking or boiling the entire insect in caustic potash, when it is seen that the integument is continuous and not actually subdivided into separate somites or arthromeres, since they are seen to be connected by a thin intersegmental membrane (Fig. 16). But this segmentation or metamerism of the integument is, however, the external indication of the segmentation of the arthropodan body most probably inherited 31from the worms, being a disposition of the soft parts which is characteristic of the vermian type. This segmentation of the integument is correlated with the serial repetition of the ganglia of the nervous system, of the ostia of the dorsal vessel, the primitive disposition of the segmental and reproductive organs, of the soft, muscular dissepiments which correspond to the suture between the segments, and with the metameric arrangement of the muscles controlling the movements of the segments on each other, and which internal segmentation or metamerism is indicated very early in embryonic life by the mesoblastic somites.
Fig. 16.—Diagram of the anterior part of an insect, showing the membranous intersegmental folds, g.—After Graber.
In the unjointed worms, as Graber states, the body forms a single but flexible lever. In the earthworm the muscular tube or body-wall is enclosed by a stiffer cuticle, divided into segments; hence the worm can move in all required directions, but only by sections, as seen in Fig. 16, which represents the thickened integument divided into segments, and folded inward between each segment, this thin portion of the skin being the intersegmental fold. Each segment corresponds to a special zone of the subdivided muscular tube (m), the fascia extending longitudinally. The figure shows the mode of attachment of the fascia of the muscle-tube to the segment. The anterior edge is inserted on the stiff, unyielding, inner surface of each segment: the hinder edge of the muscle is attached to the thin, flexible, intersegmental fold, which thus acts as a tendon on which the muscle can exert its force. (Graber.)
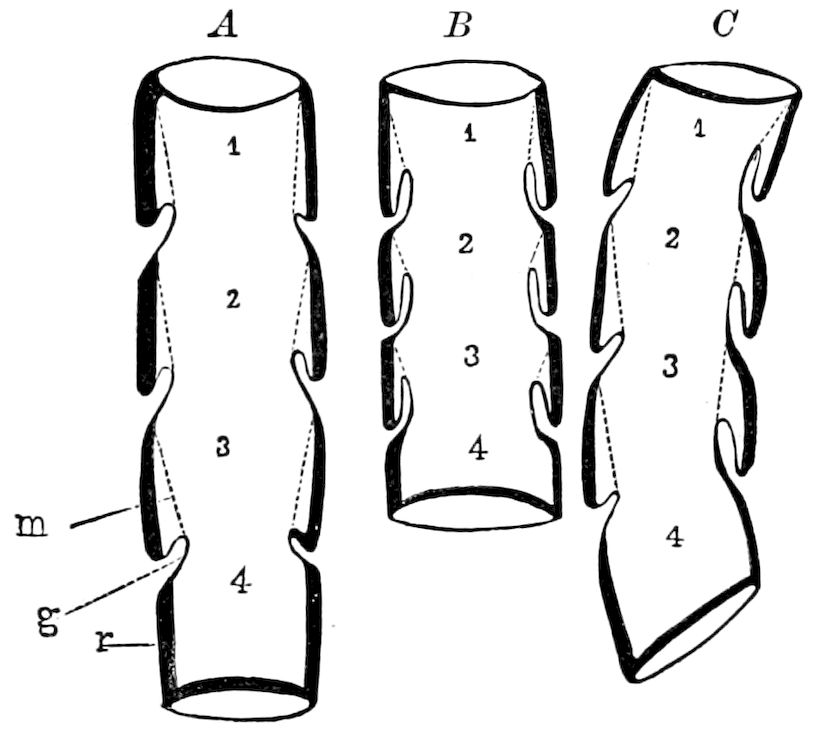
Fig. 17.—Diagram of the integument and arrangement of the segmental muscles: A, relaxed; m, muscle; g, membranous articulation; r, chitinous ring. B, the same contracted on both sides. C, on one side.—After Graber.
“Fig. 17 makes this still clearer. The muscles (m) extend between two segments immediately succeeding each other. Supposing the anterior one (A) to be stationary, what do we then see when the muscle contracts? Does it also become shorter? The intersegmental 32fold is drawn forwards, and hence the entire hinder segment moves forward and is shoved into the front one, and so on with the others, as at B. Afterwards, if the strain of the muscle is relieved by the diminishing action of the tensely stretched, intersegmental membrane, it again returns to a state of rest.” (Graber.)
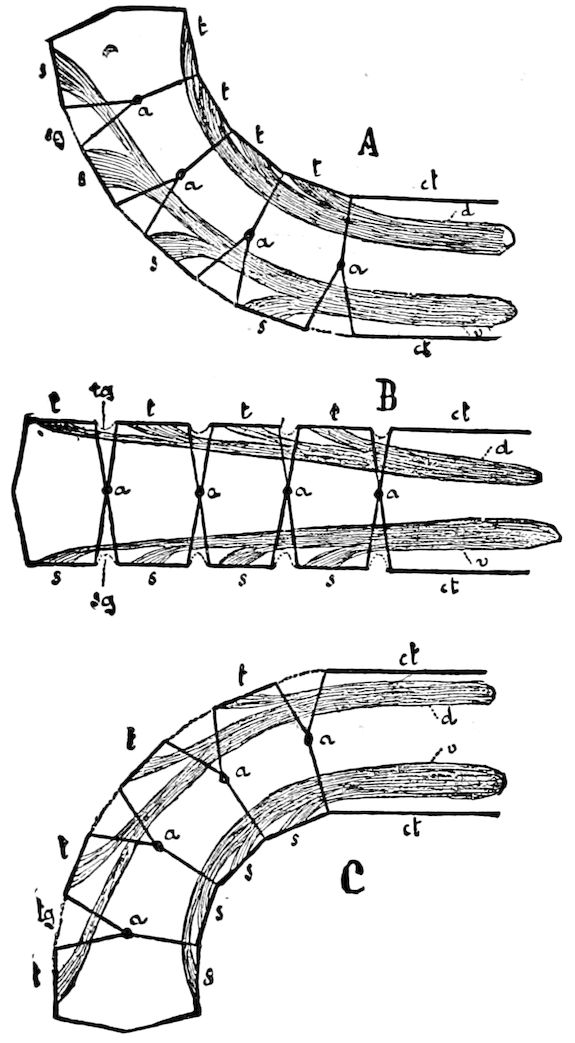
Fig. 18.—Diagrams to demonstrate the mechanism of the motion of the segmented body in the Arthropoda: One larger segment (cf) and 4 smaller. The exoskeleton is indicated by black lines, the interarticular membranes by dotted lines. The hinges between consecutive segments are marked at, tergal (dorsal) skeleton; s, sternal (ventral) skeleton; d, dorsal longitudinal muscles = extensors (and flexors in an upward direction); v, ventral longitudinal muscles = flexors. In B, the row of segments is stretched; in A, by the contraction of the muscles (d) bent upward; in C, downward; tg, tergal; sg, sternal interarticular membranes.—After Lang.
While we look upon the dermal tube of worms as a single but flexible lever, the body of the arthropods, as Graber states, is a linear system of stiff levers. We have here a series of stiff, solid rings, or hooks, united by the intersegmental membrane into a whole. When the muscles, extending from one ring to the next behind contract, and so on through the entire series, the rings approximate each other.
The ectoskeletal segments bend to one side by the contraction of the muscles on one side, the point of the outer segmental fold opposite the fixed point becoming converted into the turning-point (C).
The usual result of the arrangement of the locomotive system is the simple curving of the body (C), and then the alternate bending of the body to right and left, which produces the serpentine movements characteristic of the earthworms, the centipede, and many insect larvæ. The most striking example of the wonderful variety of movements which can be made by an insect are those of the Syrphus larva. When feeding amid a herd of aphides, it is seen to now raise the front part of the 33body erect and stiff, then to bend it down, or rapidly turn it to either side, or move it in a complete circle. (Graber, pp. 23–26.)
The arrangement and mode of working of the muscles, says Lang, is illustrated by Fig. 18, which shows us five segments, one larger (ct) and four smaller, in vertical projection. The thicker portion of the integument is marked by strong outlines, the delicate and flexible interarticular membranes (tg, sg) in dotted lines. The hinges between two consecutive segments are marked a. A dorsal muscle (d) is attached to the larger segment (ct), and runs through the smaller segments, being inserted in the dorsal portion of the crust (t) of each by means of a bundle of fibres. A ventral muscle (v) does the same on the sternal side (s).
“The skeletal segments,” adds Lang, “may be compared to a double-armed lever, whose fulcrum lies in the hinges. If the dorsal muscle contracts, it draws the dorsal arm of the lever (the tergal portion of the skeleton) in the direction of the pull towards the larger segments; the tergal interarticular membranes become folded, the ventral stretched, and the four segments bend upward (Fig. 18, A). If the ventral muscle contracts, while at the same time the dorsal slackens, the row of segments will be bent downwards (Fig. 18, C).”
L. B. Sharp suggests, that in the Crustacea the rings formed by “the regularity and stress of muscular action” would be hardened by the deposition of lime at the most prominent portion, i.e. between what we have called the intersegmental folds. (American Naturalist, 1893, p. 89.) Cope also states that “with the beginning of induration of the integument, segmentation would immediately appear, for the movements of the body and limbs would interrupt the deposit at such points as would experience the greatest flexure. The muscular system would initiate the process, since flexure depends on its contractions, and its presence in animals prior to the induration of the integuments in the order of phylogeny, furnishes the conditions required.” (The Primary Factors of Organic Evolution, p. 268, 1895.)
It is apparent that the jointed or metameric structure of the bodies of insects and other arthropods is an inheritance from the segmented worms. In the worms the body is a continuous dermo-muscular tube, while in arthropods this tube is divided into regions, and the cuticle is thicker and more resistant. To go back to the incipient stages in the process of segmentation of the body, we conceive that the worms probably arose from a creeping gastrula-like form, the gastræa. The act of creeping gradually induced an elongated shape of the body. The movement of such an organism in a 34forward direction would gradually evolve a fore and aft, dorsal and ventral, and bilateral symmetry. As soon as this was attained, as the effect of creeping over rough irregular surfaces there would result mechanical lateral strains intermittently acting during the serpentine movements of the worm. The integument would, we can readily suppose, tend to bend or yield, or become permanently wrinkled, at more or less regular intervals. The arrangement of the muscles would gradually conform to this habit of creeping, and finally the nervous system and other organs more directly connected with the creeping movements of the organism would tend to be correlated in their arrangement with that of the segments. In this way the homonomous segments of the annelid body probably became developed, and their relations and shapes were eventually fixed by inheritance. After this stage was reached, and limbs began to appear, the segments would tend to become heteronomous, and to be grouped into regions.
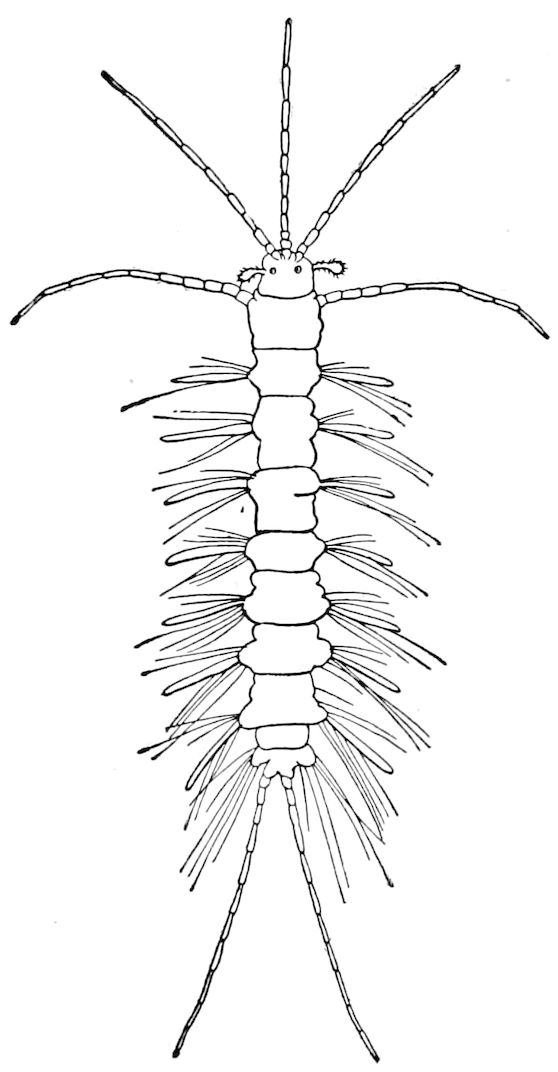
Fig. 19.—Dujardinia rotifera, with jointed tentacles and caudal appendages.—With some changes, after Quatrefages.
The origin of the joints or segments in the limbs of arthropods was probably due to the mechanical strains to which what were at first soft fleshy outgrowths along the sides of the body became subjected. Indeed, certain annelid worms of the family Syllidæ have segmented tentacles and parapodia, as in Dujardinia (Fig. 19). We do not know enough about the habits of these worms to understand how this metamerism may have arisen, but it is possibly due to the act of pushing or repeated efforts to support the body while creeping over the bottom among broken shells, over coarse gravel, or among seaweeds.
It is obvious, however, that the jointed structure of the limbs of arthropods, if we are to attempt any explanation at all of the origin of such structure, was primarily due mainly to lateral strains and 35impacts resulting from the primitive endeavors of the ancestral arthropods to raise and to support the body while thus raised, and then to push or drag it forward by means of the soft, partially jointed, lateral limbs which were armed with bristles, hooks, or finally claws.
On the other hand, by adaptation, or as the result of parasitism and consequent lack of active motion, the original number of segments may by disuse be diminished. Thus in adult wasps and bees, the last three or four abdominal segments may be nearly lost, though the larval number is ten. During metamorphosis the body is made over, and the number, shape, and structure of the segments greatly modified. In the female of the Stylopidæ the thorax loses all traces of segments, and is fused with the head, and the abdominal segments are faintly marked, losing their chitin.
While the maxillæ have several joints, the mandibles are 1–jointed, but there are traces of two joints in Campodea, certain beetles, etc. In the antenna there is a great elasticity in respect to the number of joints, which vary from one or two to a hundred or more. It is likewise so in the thoracic legs, where the number of tarsal joints varies from one to five; also in the cercopoda, the number of joints varying from one or two to twelve or more.
d. Mechanical origin of the limbs and of their jointed structure
We have already hinted at the mode of origin of the limbs of arthropods. Like the body or trunk, the limbs are chitinous dermo-muscular tubes, with a dense solid cuticle, and internal muscles, and were it not for their division at more or less regular intervals into segments, forming distinct sets of levers, set up by the strains in these tubular supports, there would be no power of varied motion.
Even certain worms, as already stated, have their tentacles and parapodia, or certain appendages of their parapodia, more or less jointed, but there are no indications of claws or of any other hard chitinous armature at the extremity, and the skin is thin and soft.
In the most simple though not the most primitive arthropods, such as the Tardigrades, whose body is not segmented, there are four pairs of short unjointed legs, ending each in two claws, which have probably arisen in response to the stimulus of pushing or dragging efforts.
The legs of Peripatus are unjointed, and have a thin cuticle, but end in a pair of claws, which have evidently arisen as a supporting 36armature, the result of the act of moving or pulling the body over the uneven surface of the ground.
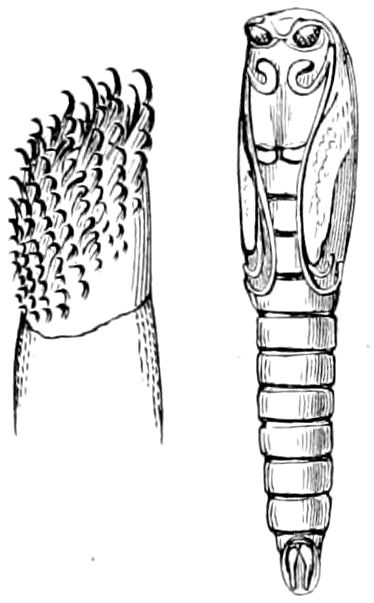
Fig. 20.—A prothoracic leg of Chironomus larva; and pupa.
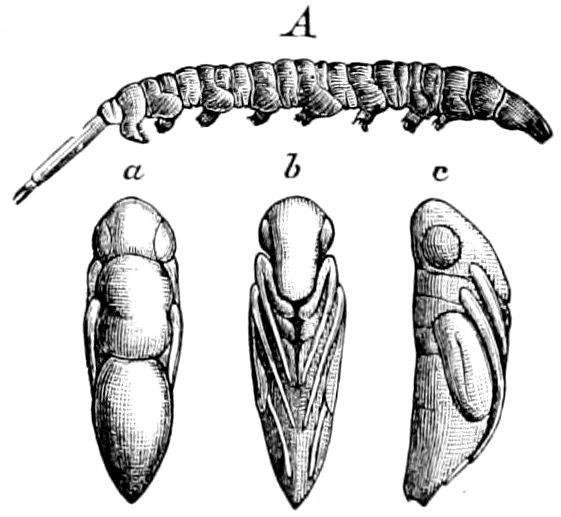
Fig. 21.—A, larva of Ephydra californica: a, b, c, pupa.
There is good reason to suppose that such limbs arose from dynamical causes, similar to those exciting the formation of secondary adaptations such as are to be seen in the prop or supporting legs of certain dipterous larvæ, as the single pair of Chironomus (Fig. 20) and Simulium, or the series of unjointed soft tubercles of Ephydra (Fig. 21), etc., which are armed with hooks and claws, and are thus adapted for dragging the insect through or over vegetation or along the ground.
Now by frequent continuous use of such unjointed structures, the cuticle would tend to become hard, owing to the deposit of a greater amount of chitin between the folds of the skin, until finally the body being elongated and homonomously segmented, the movements of walking or running would be regular and even, and we would have homonomously jointed legs like those of the trilobites, or of the most generalized Crustacea and of Myriopoda.
In the most primitive arthropods,—and such we take it were on the whole the trilobites, rather than the Crustacea,—the limbs were of nearly the same shape, being long and slender and evenly jointed from and including the antennæ, to the last pair of limbs of the abdominal region. In these forms there appear to be, so far as we now know, no differentiation into mandibles, maxillæ, maxillipedes, and thoracic legs, or into gonopoda. The same lack of diversity of structure and function of the head-appendages has survived, with little change, in Limulus. In the trilobites (Fig. 1) none of the limbs have yet been found to end in claws or forceps; being in this respect nearly as primitive as in the worms. Secondary adaptations have arisen in Limulus, the cephalic appendages being forcipated, adapted as supports to the body and for pushing it onward through the sand or mud, while the abdominal legs are broad and flat, adapted for swimming and bearing the broad gill-leaves.
It is thus quite evident that we have three stages in the evolution of the arthropodan limb; i.e. 1, the syllid stage, of simple, jointed, soft, yielding appendages not used as true supports (Fig. 19); 2, the 37trilobite stage, where they are more solid, evenly jointed, but not ending in claws; and by their comparatively great numbers (as in the trilobite, Triarthrus) fully supporting the body on the bottom of the sea. In Limulus they are much fewer in number, thicker, and acting as firm supports, the cephalic limbs of use in creeping, and ending in solid claws. 3, The third stage is the long slender swimming head-appendages of the nauplius stage of Crustacea.
As regards the evolution of limbs of terrestrial arthropods, we have the following stages: 1, the soft unjointed limbs of Tardigrades, ending in two claws, and those of Peripatus, and the pseudo- or prop-legs of certain dipterous larvæ; 2, finally the evolution of the long, solid, jointed limbs of Pauropus and other primitive myriopods, the legs forming solid, firm supports elevating the body, and enabling the insect to drag itself over the ground or to walk or run. When the body is elongated and many-segmented, the legs are necessarily numerous; but when it is short, the legs become few in number, i.e. six, in the hexapodous young of myriopods and in insects, or eight in Arachnida. Whenever the legs are used for walking, i.e. to raise and support the body, they end in a solid point or in a pair of forceps or claws. On the other hand, as in phyllopods, where the legs are used mainly for swimming, they are unarmed and are soft and membranous, or, as in the limbs of the nauplius or zoëa stage of crustaceans, end in a simple soft point, which often bears tactile setæ.
The tarsal joints are more numerous in order to give greater flexibility to the limb in seizing and grasping objects, both to drag the body forwards and to support it.
Unlike those of the Crustacea, the limbs of insects are not primitively biramose, but single, the three-lobed first maxillæ, and secondarily bilobed second maxillæ being the result of adaptation. Embryology on the whole proves the truth of this assumption; the maxillæ of both pairs are at first single buds, afterwards becoming lobed. All the appendages of the body, including the ovipositor or sting, are modified limbs, as shown by their embryological development.
It is noticeable that in the crab, where the body is raised by the limbs above the bottom, it is much shorter and more cephalized than in the shrimps. Also in the simply walking and running spiders, the hind-body is shorter than in scorpions, while in the running and flying insects, such as the Cicindelidæ, and in the swiftly flying flies and bees, there is a tendency to a shortening of the body, especially of the abdomen. The long body of the dragon-fly is an 38impediment to flight, but compensated for by the action of the large wings.
The arthropodan limb is a compound leverage system. It is, says Graber, a lateral outgrowth of the trunk, which repeats in miniature that of the main trunk, its single series of joints or segments forming a jointed dermo-muscular tube. Yet the lateral appendages of an insect differ from the main trunk in two ways: (1) they taper to the end which bears the two claws, and (2) their segments are in the living animal arranged not in a straight line, but at different angles to each other. The basal joint turning on the trunk acts as the first of a whole series of levers. The second joint, however, is connected with the musculature of the first or basal joint, and thus each succeeding joint is moved on the one preceding. Each lever, from the first to the last, is both an active and a passive instrument. (Graber.)
While, however, as Graber states, the limbs possess their own sets of muscles and can move by the turning of the basal joint, the labor is very much facilitated, as is readily seen, by the trunk, though the latter has to a great extent delegated its locomotive function to the appendages, which again divide its labor among the separate joints.
Graber then calls attention to the analogy of the mechanics of locomotion of insects to those of vertebrates. An insect’s and a vertebrate’s legs are constructed on the same general mechanical principles, the limbs of each forming a series of levers.
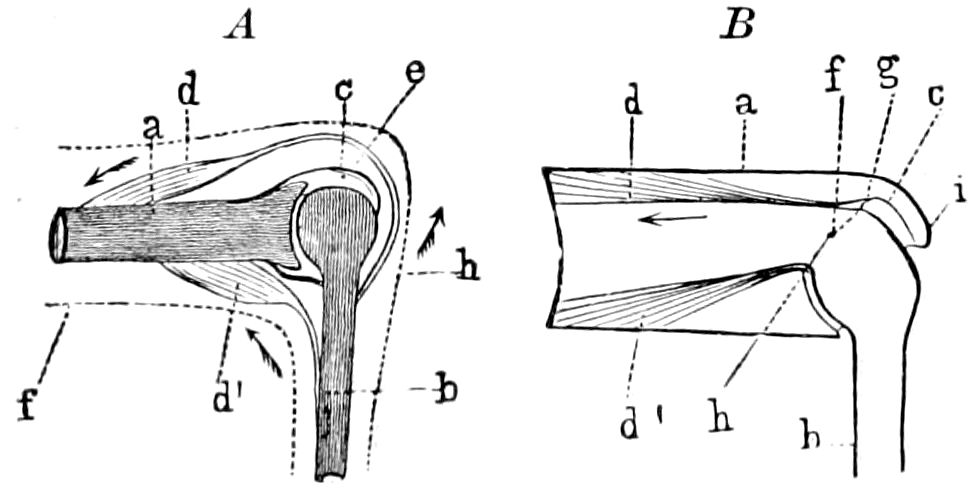
Fig. 22.—Diagram of the knee-joint of a vertebrate (A) and an insect’s limb (B): a, upper; b, lower, shank, united at A by a capsular joint, at B by a folding joint; d, extensor or lifting muscle; d1, flexor or lowering muscle of the lower joint. The dotted line indicates in A the contour of the leg.—After Graber.
Fig. 22, A, represents diagrammatically the knee joint of a vertebrate, and B that of an insect; a, the femur or thigh, and b, the tibia or shank. In the vertebrate the internally situated bones are brought into close union and bend by means of a hinge-joint; so also in the chitinous-skinned insect.
The stiff dermal tube of the insect acts as a lever by means of the thin intersegmental membrane (c) pushed in or telescoped in to the thigh joint, a special joint-capsule being superfluous. The muscles are in general the same in both types; they form a circle. In both the shank is extended by the contraction of the upper muscles (d) and is bent by the contraction of the lower 39(d1). The intersegmental membrane of the insect’s limb is in a degree a two-armed lever, whose pivot (f) lies in the middle. The internal invagination of the intersegmental fold (B, g-h) affords the necessary support to the muscles acting like the tendon in the vertebrate. (Graber.)
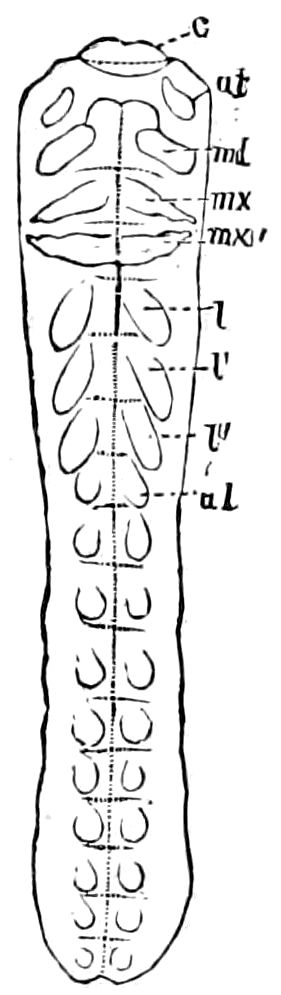
Fig. 23.—Primitive band or germ of a Sphinx moth, with the segments indicated, and their rudimentary appendages: c, upper lip; at, antennæ; md, mandibles; mx, mx′, first and second maxillæ; l, l′, l″, legs; al, abdominal legs.—After Kowalevsky.
Graber also calls attention to the fact that this insect limb differs in one important respect from that of land vertebrates. The leverage system in the last is divided at the end into five parallel divisions or digits. In arthropods, on the contrary, all the joints succeed one another in a linear series.
In insects, as well as in other arthropods, modifications of the limbs usually take the form of a simple reduction in the number of segments. Thus while the normal number of tarsal joints is five, we have trimerous and dimerous Coleoptera, and in certain Scarabæidæ the anterior tarsi are lost.
Savigny was the first, in 1816, in his great work, “Théorie des organes de la bouche des Crustacés et des Insectes,” to demonstrate that not only were the buccal appendages of biting insects homologous with those of bugs, moths, flies, etc., but that they were homologous with the thoracic legs, and that thus a unity of structure prevails throughout the appendages of the body of all arthropods. Oken also observed that “the maxillæ are only repeated feet.”
What was modestly put forth as a theory by the French morphologist has been abundantly proved by the embryology of insects of different orders to be a fact. As shown in Fig. 23 the antennæ and buccal appendages arise as paired tubercles exactly as the thoracic legs. The abdominal region also bears similar embryonic or temporary limbs, all of which in those insects without an ovipositor disappear, except the cercopoda, after birth.
LITERATURE ON THE EXTERNAL ANATOMY
General
Swammerdam, Johann. Biblia naturæ. (In Dutch, German, and English.) 1737–1738, fol., London, 1758, Pls.
Réaumur, Réné Antoine Ferchault, de. Mémoires pour servir à l’histoire des insectes. i-iv, 4º, Paris, 1734–1742.
Lyonet, Pieter. Traité anatomique de la chenille, qui ronge le bois de saule, etc. 4º, pp. xxii, 616. À la Haye, 1732. Tab. 18.
—— Recherches sur l’anatomie et les metamorphoses de differentes espèces d’insectes. Ouvrage posthume, publié par M. W. de Haan. pp. 580, tab. 54, 1832.
Latreille, Pierre André. Des rapports généraux de l’organization extérieure des animaux invertébres articulés, et comparaison des Annelides avec les Myriapodes. (Mémoires du Mus. d’Hist. Nat., 1820, vi, pp. 116–144.)
—— De quelques appendices particuliers du thorax de divers insectes. (Mémoires du Mus. d’Hist. Nat., 1821, vii, pp. 1–21).
—— Observations nouvelles sur l’organization extérieure et générale des animaux articulés et à pieds articulés, et application de ces connoissances à la nomenclature des principales parties des mêmes animaux. (Mèmoires du Mus. d’Hist. Nat., viii, 1822, pp. 169–202.)
Cuvier, George Leopold Christian Dagobert. Rapport sur les recherches anatomiques sur le thorax des animaux articulés et celui des insectes en particulier par M. V. Audouin. 4º, pp. 15, tab. 1, Paris, 1823.
Audouin, Jean Victor. Recherches anatomiques sur le thorax des animaux articulés et celui des insectes hexapodes en particulier. (Annales des Sciences naturelles, i, pp. 97–135, 416–432, 1824.)
Kirby, William, and William Spence. Introduction to entomology. i-iv, 1816–1828, London.
Straus-Durckheim, Hercule. Considérations générales sur l’anatomie comparée des animaux articulés. Paris, 1828, atlas of 19 plates.
MacLeay, William Sharp. Explanation of the comparative anatomy of the thorax in winged insects, with a review of the present state of the nomenclature of its parts. (Zoöl. Journal, v, pp. 145–179, 1830, 2 Pls.)
Burmeister, Hermann. A manual of entomology. Trans. by W. E. Shuckard, 8º, pp. 654, London, 1836, 32 Pl.
Westwood, John Obadiah. An introduction to the modern classification of insects, i, ii, 8º, pp. 462, 587, 158, 1 Pl. and 133 blocks of figs., 1839–1840.
Newport, George. Art. Insecta in Todd’s Cyclopædia of Anatomy and Phys. ii, pp. 853–994, 1839, Figs. 329–439.
Erichson, Wilhelm Ferdinand. Entomographien. Berlin, 1840.
Brullé, Auguste. Recherches sur les transformations des appendices dans les articulés. (Annales des Sciences nat. Sér. 3, ii, pp. 271–374, tab. 1, 1844.)
Winslow, A. P.: son. Om byggnaden af thorax hos Insekterna. Helsingborg, 1862, 1 Pl, pp. 24.
Packard, Alpheus Spring. Guide to the study of insects. 1869.
—— Systematic position of the Orthoptera in relation to other insects. (Third report U. S. Ent. Commission, pp. 286–345, 1883, Pls. xxiii-lxi.)
Graber, Vitus. Die Insekten. 12º, pp. 403, 603, München, 1877, many Figs.
Huxley, Thomas Henry. A manual of the anatomy of invertebrated animals. 12º, pp. 397–451, Figs., London, 1877.
41Hammond, Arthur. Thorax of the blow-fly. (Journ. Linn. Soc., London, xv. Zoöl., 1880, pp. 31.)
Brauer, Friedrich. Ueber das Segment médiaire Latreille’s. (Sitzb. d. k. Akad. d. Wissensch. Wien, 1882, pp. 218–241, 3 tab.)
—— Systematisch-zoologische Studien. (Ibid., 1885, pp. 237–413.)
Gosch, C. C. A. On Latreille’s theory of “Le Segment médiaire.” (Nat. Tidsskrift (3), xiii, pp. 475–531, 1883.)
Miall, L. C., and Denny, Alfred. The structure and life-history of the cockroach (Periplaneta orientalis). An introduction to the study of insects. 8º, pp. 224, London, 1886.
Cheshire, Frank R. Bees and bee-keeping. i, Scientific, London, 1886, Pls. and Figs.
Lang, Arnold. Text-book of comparative anatomy. i, pp. 426–508, 1891, many Figs.
Kolbe, H. J. Einführung in die Kenntniss der Insekten. 8º, pp. 709, 324 figs., Berlin, 1893.
Sharp, David. The Cambridge natural history. Insecta, i, 8º, pp. 83–584, 1895, Figs. 47–371.
Also the works of Bos, Chabrier, Cholodkowsky, Comstock, Dewitz, Eaton, Erichson, Gerstaecker, Girard, Grassi, Hagen, Haase, Kellogg, Knoch, Lacordaire, Latreille, Leuckart, Lendenfeld, Lowne, Lubbock, Mayer, Meinert, F. Müller, Osten-Sacken, Pagenstecher, Reinhard, Schaum, Schiödte, Scudder, J. B. Smith, Spinola, Stein, Weismann, Wood-Mason.
THE HEAD AND ITS APPENDAGES
a. The head
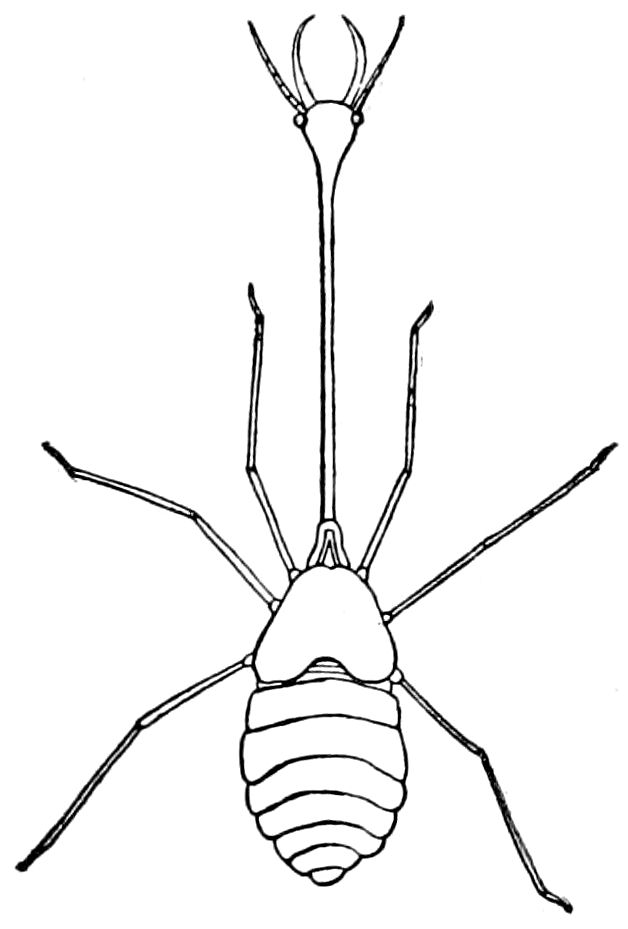
Fig. 24.—Presumed larva of Nemoptera (Necrophilus arenarius), Pyramids of Egypt.—After Roux, from Sharp.
While the head is originally composed of probably not less than six segments, these are in the adult insect fused together into a capsule or hard chitinous box, the epicranium, with no distinct traces of the primitive segments. The head contains the brain and accessory ganglia, the mouth or buccal cavity, also the air-sacs in many winged forms, and gives support to the external organs of sense, the antennæ, and to the buccal appendages, the larger part of the interior being filled with the muscles moving these structures. The solid walls of the head serve as a lever or support for the attachment of these muscles, especially those of the mandibles. Thus there is a correlation between the large size of the mandibles of the soldier white ants and ants, the head being correspondingly large to accommodate the great mandibular muscles. The other extreme is seen in the larva of Necrophilus (Fig. 24), with its long slender neck and diminutive head.
The clypeus.—This is that part of the head situated in front of the epicranium, and anterior to the eyes, forming the roof of the posterior part of the mouth, and is, as embryology shows, probably a tergal sclerite. It varies greatly in shape and size in the different orders of insects. It is often divided into two parts, the clypeus posterior and clypeus anterior, or which may be designated as the post- and ante-clypeus (Figs. 29, B).
The labrum.—The “upper lip” or labrum is an unpaired flap-like piece hinged to the front edge of the clypeus, and may be seen to move up and down when the insect moves its mandibles. It forms the roof of the anterior part of the mouth (Figs. 69, 74), and its inner side is lined with a soft membrane, usually provided with hairs and sense-papillæ or cups, forming the epipharynx.
The labrum is more or less deeply bilobed, especially in caterpillars and in adult Staphylinidæ, and has been thought by some 43writers (Kowalevsky, Carrière, and also Chatin) to represent a pair of appendages, but Heymons (1895) refutes this view, stating as his reason that the labrum arises between the two halves of the nervous system (protocerebrum), while all the true appendages arise on each side of the nervous system. (See also Fig. 34.)
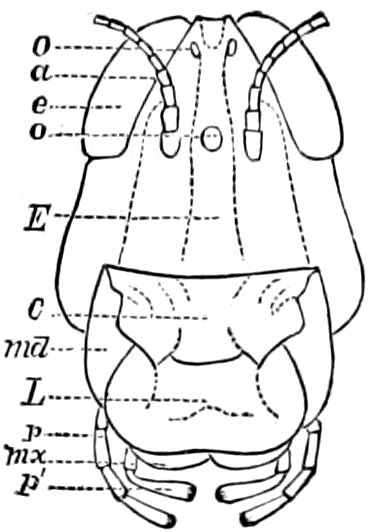
Fig. 25.—Front view of the head of C. spretus: E, epicranium; C, clypeus; L, labrum; O, o, ocelli; e, eye; a, antenna; md, mandible; mx, portion of maxilla uncovered by the labrum; p, maxillary palpus; p′, labial palpus.
In the fleas (Siphonaptera) both the clypeus and labrum are wanting.
While it apparently forms an anterior specialized portion of the procephalic lobes, Viallanes regarded it as belonging to the third, or his tritocerebral, segment, since the labral nerves arise from the tritocerebral ganglia. But since in all the early as well as late stages of embryonic life it appears to be situated in front of the mouth, it would seem to belong to the first segment.
In the embryo of Blatta it first appears as a thick crescentic fold being slightly divided anterior to the mouth, and in Doryphora it appears as a heart-shaped or deeply bilobed prominence situated in front of the mouth (Wheeler).
The epipharynx and labrum-epipharynx.—The epipharynx is the under surface or pharyngeal lining of the clypeus and labrum, forming the membranous roof of the mouth. As it contains the organs of taste and has been generally overlooked by entomologists, we may dwell at some length on its structure in different orders.
Réaumur was, so far as we have been able to ascertain, the first author to describe and figure the epipharynx, which he observed in the honey bee and bumble bee, and called la langue, remarking that it closes the opening into the œsophagus, and that it is applied against the palate. According to Kirby and Spence, De Geer described the epipharynx of the wasp; and Latreille referred to it, calling it the sous labre.
The name epipharynx was bestowed upon this organ by Savigny, who thus speaks of that of the bees: “Ce pharynx est, à la vérité, non seulement caché par la lèvre supérieure, mais encore exactement recouvert par un organe particulier que Réaumur a déjà décrit. C’est une sorte d’appendice membraneux qui est reçu entre les deux branches des mâchoires. Cette partie ayant pour base le bord supérieur du pharynx, peut prendre le nom d’épipharynx ou d’épiglosse.”
44He also describes that of Diptera. What Walter has lately proved to be the epipharynx of Lepidoptera was regarded by Savigny and all subsequent writers as the labrum.
The latest account of the function of this organ is that by Cheshire, who states that the tube made by the maxillæ and labial palpi cannot act as a suction pipe, because it is open above. “This opening is closed by the front extension of the epipharynx, which closes down to the maxillæ, fitting exactly into the space they leave uncovered, and thus the tube is completed from their termination to the œsophagus.”
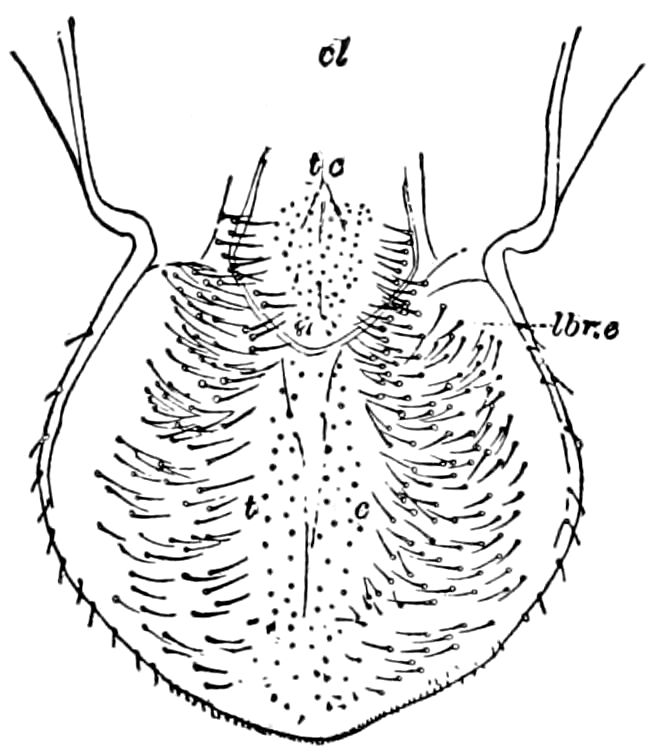
Fig. 26.—Epipharynx of Phaneroptera angustifolia: cl, clypeus; lbr. e, labrum-epipharynx; t c, taste cups, both on the clypeal and on the labral regions.
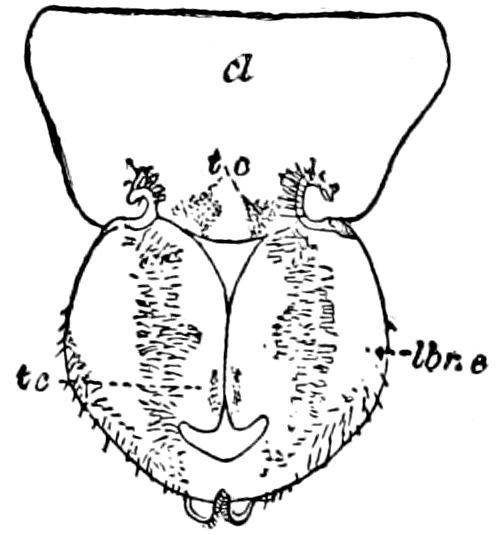
Fig. 27.—Epipharynx of Hadenœcus subterraneus, cave cricket.
It is singular that this organ is not mentioned in Burmeister’s Manual of entomology, in Lacordaire’s Introduction à l’entomologie, or by Newport in his admirable article Insecta in Todd’s Cyclopedia of anatomy. Neither has Straus-Durckheim referred to or figured it in his great work on the anatomy of Melolontha vulgaris.
In their excellent work on the cockroach, Miall and Denny state that “The epipharynx, which is a prominent part in Coleoptera and Diptera, is not recognizable in Orthoptera” (p. 45). We have, however, found it to be always present in this order (Figs. 26, 27).
We are not aware that any modern writers have described or referred to the epipharynx of the mandibulate orders of insects. Although Dr. G. Joseph speaks of finding taste-organs on the palate of almost every order of insects, especially plant-feeding forms, we are unable to find any specific references, his detailed observations being apparently unpublished.
The epipharynx is so intimately associated with the elongated labium of certain Diptera, that, with Dr. Dimmock, we may refer to the double organ as the labrum-epipharynx; and where, as in the lepidopterous Micropteryx semipurpurella, described and figured by Walter, and the Panorpidæ (Panorpa and Boreus), the labrum seems pieced out with a thin, pale membranous fold which appears to be 45an extension of the epipharynx, building up the dorsal end of the labrum, this term is a convenient one to use.
In the lower orders of truly mandibulate insects, from the Thysanura to the Coleoptera, excluding those which suck in liquid food, such as the Diptera, Lepidoptera, and Hymenoptera, and the Mecoptera (Panorpidæ) with their elongated head and feeble, small mandibles, the epipharynx forms a simple membranous palatal lining of the clypeus and labrum. In such insects there is no soft projecting or pendant portion, fitted to close the throat or to complete a partially tubular arrangement of the first and second maxillæ.
In all the mandibulate insects, then, the epipharynx forms simply the under surface or pharyngeal lining of the clypeus and labrum, the surface being uniformly moderately convex, and corresponding in extent to that of the clypeus and labrum, posteriorly merging into the palatal wall of the pharynx; the armature of peculiar gathering-hairs sometimes spreading over its base, being continuous with those lining the mouth and beginning of the œsophagus. The suture separating the labrum from the clypeus does not involve the epipharynx, though since certain gustatory fields lie under the front edge of the clypeus, as well as labrum, one may in describing them refer to certain fields or groups of cups or pits as occupying a labral or clypeal region or position.
The lack of traces of a suture in the epipharynx corresponding to the labral suture above, suggests that the labrum does not represent a pair of coalesced appendages, and that it, with the clypeus, simply forms the solid cuticular roof of the mouth.
The only soft structures seen between the epipharynx and labrum, besides the nerves of special sense, are the elevator muscles of the labrum, and two tracheæ, one on each side.
The structure and armature of the epipharyngeal surface even besides the taste-pits, taste-cups and rods, is very varied, the setæ assuming very different shapes. There seem to be two primary forms of setæ, (1) the normal forms which arise from a definite cell; and (2) soft, flattened, often hooked hairs which are cylindrical towards the end, but arise from a broad triangular base, without any cell-wall. These are like the “gathering hairs” of Cheshire, situated on the bees’ and wasps’ tongue; they also line the walls of the pharynx and extend toward the œsophagus. They are also similar to the “hooked hairs” of Will. The first kind, or normal setæ, are either simply defensive, often guarding the sense-cups or sensory fields on which the sense-cups are situated, or they have a nerve extending to them and are simply tactile in function.
46The surface of the epipharynx, then, appears to be highly sensitive, and to afford the principal seat of the gustatory organs, which are described under the head of organs of taste.
LITERATURE ON THE EPIPHARYNX
Réaumur. Mémoires pour servir à l’histoire des insectes, v, 1740, p. 318, Pl. 28, Figs. 4, 7, 8, 9, 10, 11 l.
Kirby and Spence. Intr. to entomology, iii, 1828, p. 457.
De Geer. ii, 1778; v, 26, Fig. 11, M.
Kirby and Spence. Pl. xii, Fig. 2 K.
Latreille. Organisation extérieure des insectes, p. 184. (Quoted from Kirby and Spence.)
Savigny. Mémoires sur les animaux sans vertèbres. Partie Ire, 1816, p. 12.
Walter, Alfred. Beiträge zur Morphologie der Schmetterlinge. Erster Theil. Zur Morphologie des Schmetterlingsmundtheile. (Jena. Zeits., xviii, 1885, p. 752.)
Cheshire, F. R. Bees and bee-keeping, i, London, 1886, p. 93.
Joseph, Gustav. Zur Morphologie des Geschmacksorganes bei Inseckten. (Amtlicher Bericht der 50 Versammlung deutscher Naturforscher u. Artzte in München. 1877, pp. 227, 228.)
Dimmock, George. The anatomy of the mouth-parts and of the sucking apparatus of some Diptera, 1881. (Also in Psyche, iii, pp. 231–241, Pl. 1, 1882.)
Packard, A. S. On the epipharynx of the Panorpidæ. (Psyche, 1889, v, pp. 159–164.)
—— Notes on the epipharynx and the epipharyngeal organs of taste in mandibulate insects. (Psyche, v, pp. 193–199, 222–228, 1889.)
Attachment of the head to the trunk.—The head is either firmly supported by the broad prothoracic segment in Orthoptera, many beetles, etc., into which it is more or less retracted, or it is free and attached by a slender neck, easily turning on the trunk, as in dragon-flies, flies, etc. In some insects there are several chitinous plates, situated on an island in the membrane on the under side of the neck; these are the “cervical sclerites” of Sharp, occurring “in Hymenoptera, in many Coleoptera, and in Blattidæ.”
The basal or gular region of the head.—At the hinder part of the head is the opening (occipital foramen) into the trunk. The cheek (gena) is the side of the head, and to its inner wall is attached the mandibular muscle; it thus forms the region behind the eye and over the base of the mandibles. In the Termitidæ, where the head is broad and flat, it forms a distinct piece on the under side of the head bounding the gulo-mental region (Fig. 28). In the Neuroptera (Corydalus, Fig. 29, and Mantispa, Fig. 30) it is less definitely outlined.
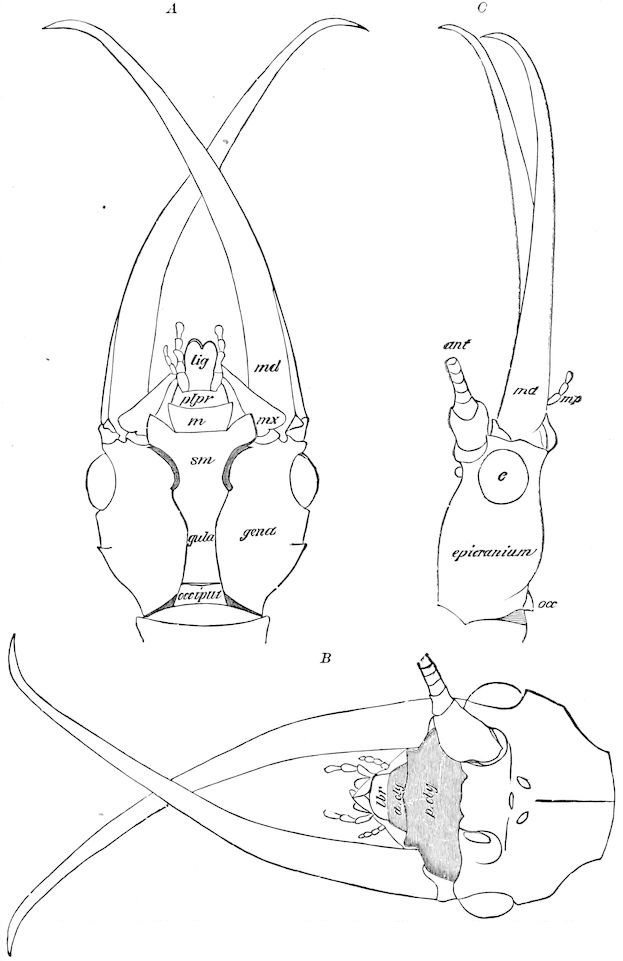
Fig. 29.—Head of Corydalus cornutus, ♂: A, from above. B, from beneath. C, from the side. a. cly, clypeus anterior; p. cly, clypeus posterior; lbr, labrum; md, mandible; mx, base of first maxilla; mp, its palpus; m, mentum; sm, submentum; plpr, palpifer; lig, fused second maxillæ; ant, antenna; occ, occiput.
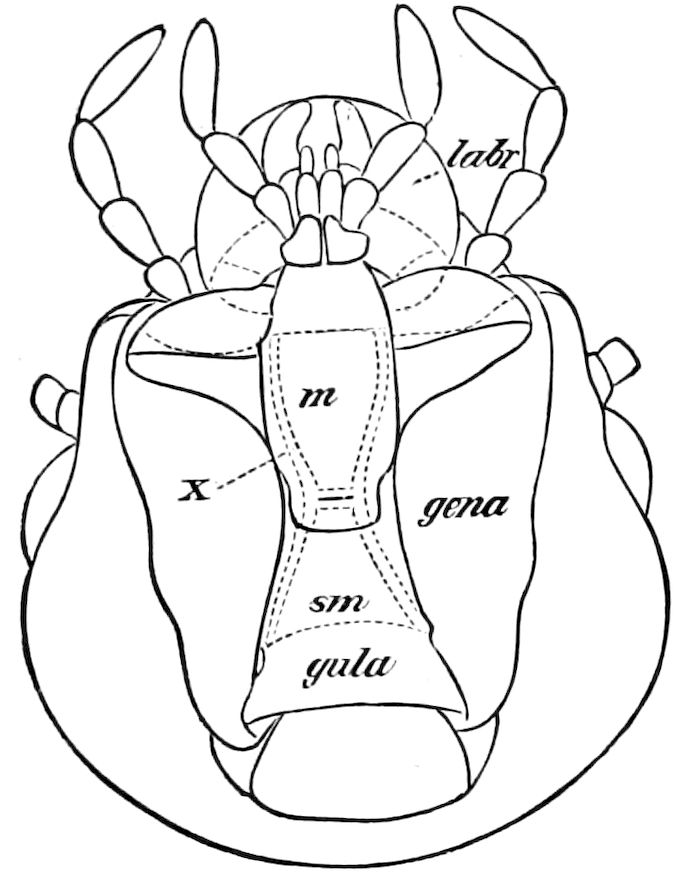
Fig. 28.—Head of Termopsis angusticollis, seen from beneath, showing the gena and gula: m, mentum; sm, submentum; labr, under side of the labrum; x, hypopharyngeal chitinous support.
All the gulo-mental region of the head appears to represent the base of the second maxillæ, and the question hence arises whether the submentum is not the homologue of the cardines of the first maxillæ fused, and the mentum that of the stipites of the latter also fused together. If this should prove to be the case, the homologies between the two pairs of maxillæ will be still closer than before supposed. Where the gula is differentiated, this represents the basal piece of the second maxillæ. In Figs. 28, 29, 30, and 31, these three pieces are clearly shown to belong to the second maxillary segment. It is evident that these pieces or sclerites belong to the second maxillary or labial segment of the head, as does the occiput, which may represent the tergo-pleural portion of the segment. Miall and Denny also regarded the submentum as the basal piece of the second maxillæ.
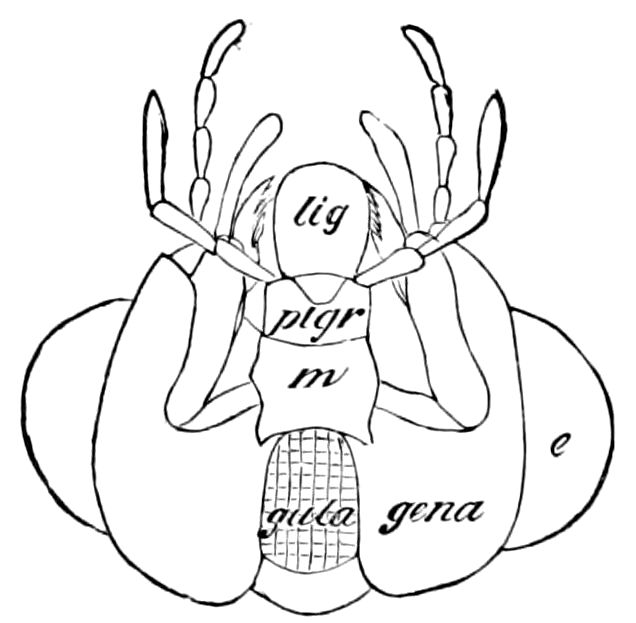
Fig. 30.—Head of Mantispa brunnea, under side: e, eye; other lettering as in Fig. 29.
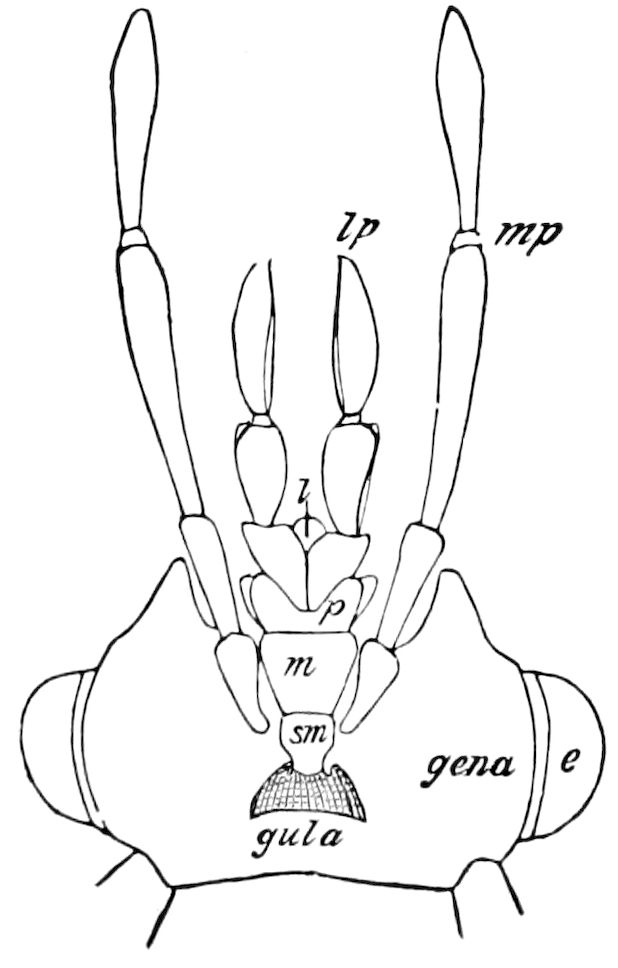
Fig. 31.—Head of Limnephilus pudicus, under side: e, eye; l, ligula; p, palpifer; lp, labial palpi.
The occiput (Fig. 29, B, C), as stated beyond, is very rarely present as a separate piece; in the adult insect we have only observed it in Corydalus. The occipital region may be designated as that part of the head adjoining and containing the occipital foramen. Newport considers the occiput as that portion of the base of the head “which is articulated with the anterior margin of the prothorax. It is perforated by a large foramen, through which the organs of the head are connected 49with those of the trunk. It is very distinct in Hydroüs and most Coleoptera, and in some, the Staphylinidæ, Carabidæ, and Silphidæ is constricted and extended backwards so as to form a complete neck.” (See also p. 51.)
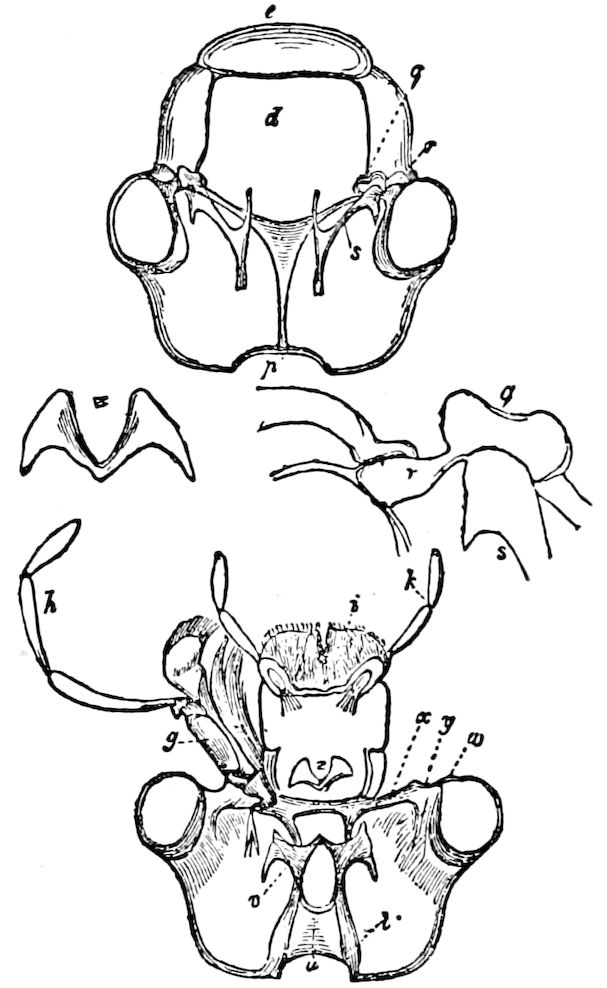
Fig. 32.—Interior and upper and under surface of the head of Hydroüs piceus: d, clypeus; e, labrum; g, maxilla; h, its palpus; i, labium; k, labial palpus; p, sutura epicranii; q, cotyloid cavity; r, torulus; s, v, laminæ squamosa; t, laminaæ posteriores; u, tentorium; w, laminæ orbitales; x, os transversum; y, articulating cavity for the mandible; z, os hypopharyngeum.—After Newport.
The tentorium.—The walls of the head are supported or braced within by two beams or endosternites passing inwards, and forming a solid chitinous process or loop which extends in the cockroach downwards and forwards from the lower edge of the occipital foramen. “In front it gives off two long crura or props, which pass to the ginglymus, and are reflected thence upon the inner surface of the clypeus, ascending as high as the antennary socket, round which they form a kind of rim.” (Miall and Denny.) The œsophagus passes upwards between its anterior crura, the long flexor of the mandible lies on each side of the central plate; the supraœsophageal ganglion rests on the plate above, and the subœsophageal ganglion lies below it, the nerve cords which unite the two passing through the circular aperture. (Miall and Denny.) In Coleoptera (Hydroüs) it protects the nervous cord which passes under it. (Newport, Fig. 32, u.)
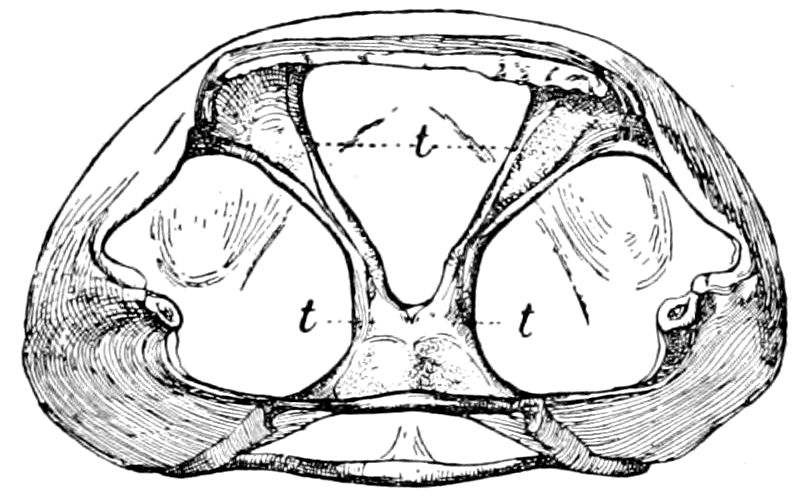
Fig. 33.—Posterior view of head of Anabrus; t, tentorium. Joutel del.
In Anabrus the tentorium is V-shaped, the two arms originating on each side of the base of the clypeus next to the base of each mandible the origin being indicated by two small foramina partly concealed externally and passing inwards and backwards and uniting just before reaching the posterior edge of the large occipital foramen (Fig. 33).
50Palmén regards the tentorium as representing a pair of tracheæ (with the cephalic spiracles) which have become modified for supports or for muscular attachment, since he finds that in Ephemera the tentorium breaks across the middle during exuviation, each half being drawn out of the head like the chitinous lining of a tracheal tube. This view is supported by Wheeler, who has shown that the tentorium of Doryphora originates from five pairs of invaginations of the longitudinal commissures, and which are anterior to those of the second maxillary segment. “These invaginations grow inwards as slender tubes, which anastomose in some places. Their lumina are ultimately filled with chitin.” (Jour. Morph., iii, p. 368.)
This view has also been held by Carrière and Cholodkowsky, but Heymons concludes from his embryological studies on Forficula and Blattidæ (1895) that it is unfounded. That this is probably the case is proved by the fact that the apodemes of the thoracic region are evidently not modified tracheæ, since the stigmata and tracheæ are present.
Number of segments in the head.—While it is taken for granted by many entomologists that the head of insects represents a single segment, despite the circumstance that it bears four pairs of appendages, the more careful, philosophical observers have recognized the fact that it is composed of more than a single segment. Burmeister recognized only two segments in the head; Carus and Audouin recognized three; Macleay and Newman four; Straus-Durckheim even so many as seven. Huxley supposed that there are five segments bearing appendages, remarking, “if the eyes be taken to represent the appendages of another somite, the insect head will contain six somites.” (Manual of Anat. Invert. Animals, p. 398.)
These discordant views were based on the examination of the head in adult insects; but if we confine ourselves to the imago alone, it is impossible to arrive at a solution of the problem.
Newport took a step in the right direction when he wrote: “It is only by comparing the distinctly indicated parts of the head in the perfect insect with similar ones in the larva that we can hope to ascertain the exact number of segments of which it is composed.” He then states that in the head of Hydroüs piceus are the remains of four segments, though still in the next paragraph, when speaking of the head as a whole, he considers it as the first segment, “while,” he adds, “the aggregation of segments of which it is composed we shall designate individually subsegments.”
That the head of insects is composed of four segments was shown on embryological grounds by the writer (1871) and afterwards by Graber (1879). The antennæ and mouth-parts are outgrowths budding out from the four primitive segments of the head; the antennæ grow out from the under side of the procephalic lobes, and these should therefore receive the name of antennal lobes. In like manner 51the mandibles and first and second maxillæ arise respectively from the three succeeding segments.
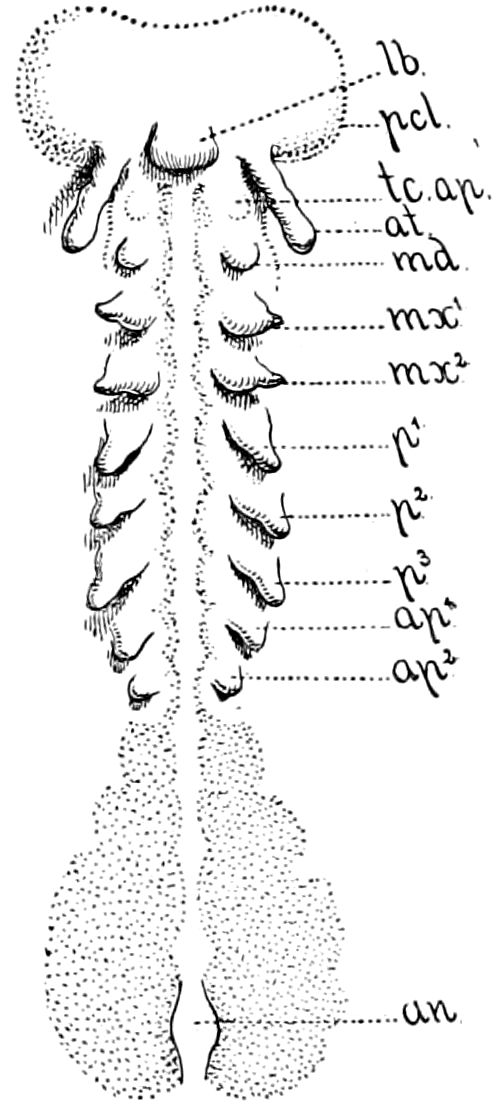
Fig. 34.—Embryo of Anurida maritima: tc. ap, minute temporary appendage of the tritocerebral segment, the premandibular appendage; at, antenna; md, mandible; mx1, first maxilla; mx2, second maxilla; p1–p3, thoracic; ap1, ap2, abdominal appendages; an, anus—After Wheeler.
While the postoral segments and their appendages are readily seen to be four in number, the question arises as to whether the eyes represent the appendages of one or more preoral segments. In this case embryology thus far has not afforded clear, indubitable evidence. We are therefore obliged to rely on the number of neuromeres, or primitive ganglia. In the postoral region of the head, as also in the trunk, a pair of neuromeres correspond to each segment. (See also under Nervous System, and under Embryology.) We therefore turn to the primitive number of neuromeres constituting the procephalic lobes or brain.
From the researches of Patten, Viallanes, and of Wheeler, especially of Viallanes, it appears that the brain or supraœsophageal ganglion is divided into three primitive segments. (See Nervous System, Brain.) The antennæ are innervated from the middle division or deutocerebrum. Hence the ocular segment, i.e. that bearing the compound and simple eyes, is supposed to represent the first segment of the head. This, however, does not involve the conclusion that the eyes are the homologues of the limbs, however it may be in the Crustacea.
The second head-segment is the antennal, the antennæ being the first pair of true jointed appendages.
The third segment of the head is very obscurely indicated, and the facts in proof of its existence are scanty and need farther elucidation.
Viallanes’ tritocerebral lobes or division of the brain is situated in a segment found by Wheeler to be intercalated between the antennal and mandibular segments. He also detected in Anurida maritima, the rudiments of a pair of appendages, smaller than those next to it, and which soon disappear (Fig. 34, tc. ap). He calls this segment the intercalary.[12] Heymons (1895) designates it as the 52“Vorkiefersegment,” and it may thus be termed the premandibular segment.
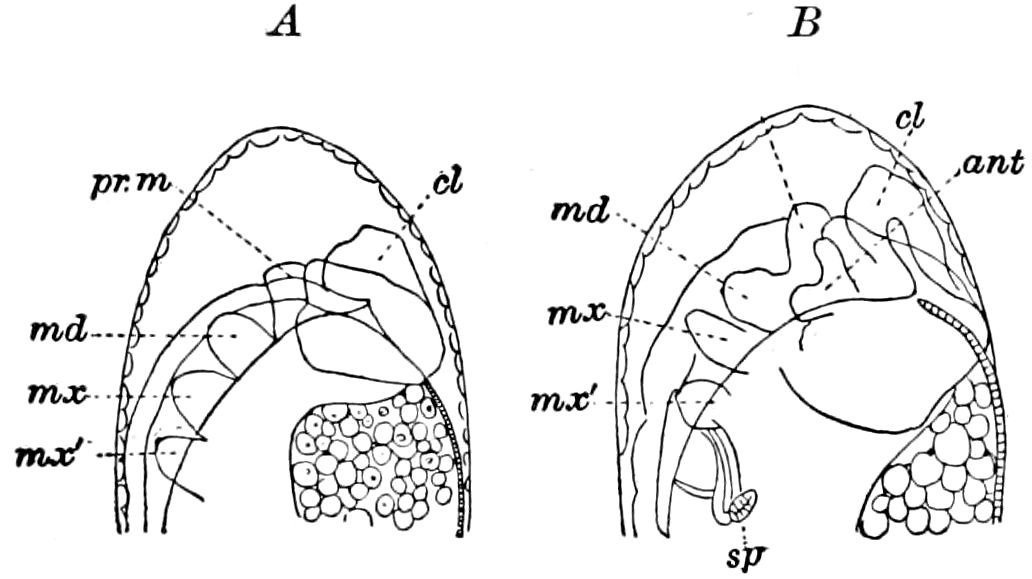
Fig. 35.—Head of embryo of honey bee: B, a little later stage than A. pr.m, premandibular segment; cl, clypeus; ant, antenna; md, mandible; mx, first maxilla; mx′, second maxilla; sp, spiracle.—After Bütschli.
As early as 1870 Bütschli observed in the embryo of the honey bee the rudiments of what appeared to be a pair of appendages between the antennæ and mandibles, but, judging by his figures, nearer to and more like the mandibles than the rudimentary antennæ (Fig. 35); they seemed to him “almost like a pair of inner antennæ.”
“I find,” he says, “in no other insects any indication of this peculiar appendage, which at the time of its greatest development attains a larger size than the antennæ, and which, afterwards becoming less distinct, forms by fusion with that on the other side a sort of larval lower lip. That this appendage does not belong to the category of segmental appendages is indicated by the site of its origin on the upper side of the primitive band.” (Zeitschr. wissen. Zool., xx, p. 538.)
Grassi has also observed it in Apis, and regards it as the germ of a first, but deciduous, pair of jaws. In the embryo of Hylotoma Graber (Figs. 134, 135) found what he calls three pairs of “preantennal projections,” one of which he thinks corresponds to the “inner antennæ” of Bütschli. This subject needs further investigation.
It thus appears that the procephalic lobes of the embryo of insects, with the rudiments of the antennæ, constitute the primitive head, and perhaps correspond to the annelidan head, while gradually the antennal appendages were in the phylogenetic development of the class fused with the two segments of the primary head. That the second maxillary segment, the occiput, was the last to be added, and at first somewhat corresponded in position to the poison-fangs 53of centipedes (Chilopods), is shown by our observations on the embryology of Æschna (Fig. 36).
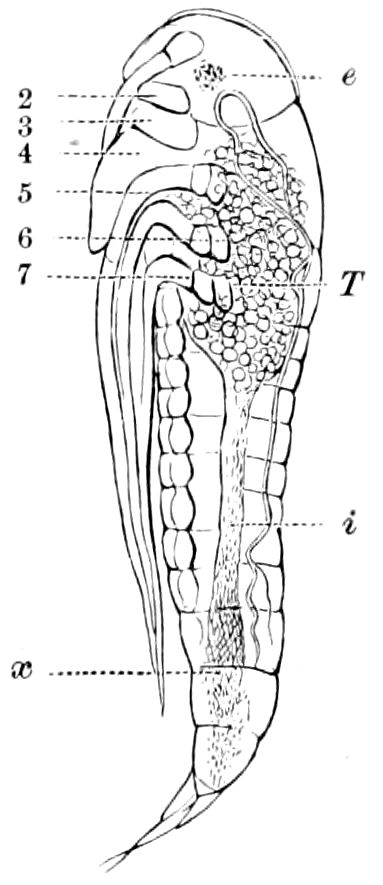
Fig. 36.—Æschna nearly ready to hatch: 4, labium, between T and e the occipital tergite; 5–7, legs.
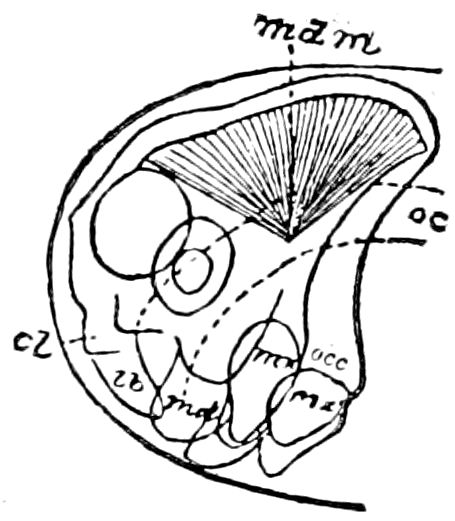
Fig. 37.—Head of embryo Nematus, showing the labial segment: occ, forming the occiput; cl, clypeus; lb, labrum; md, mandible; mdm, muscle of same; mx, maxilla; mx′, second maxilla (labium); oe, œsophagus.
The mandibular segment appears to form a large part of the post-antennal region of the epicranium on account of the great mandibular muscle which arises from so large an area of the anterior region of the head (Fig. 37).
Judging from the embryo of Nematus (Fig. 37), the first maxillary segment is tergally aborted, there being no tergo-pleural portion left.[13]
The second maxillary segment tergally appears to be represented by the occipital region of the head.
All the gular region, including the submentum and mentum, probably represents the base of the labium or second maxillæ.[14] The so-called “occiput” forms the base of the head of Corydalus, a neuropterous insect, which, however, is more distinct in the larva. In most other adult insects the occiput is either obsolete or fused with the hinder part of the epicranium. We have traced the history of this piece (sclerite) in the embryo of Æschna, a dragon-fly, and have found that it represents the tergal portion of the sixth or labial segment. In our memoir on the development of this dragon-fly, Pl. 2, Fig. 9, the head of the embryo is seen to be divided into two regions, the anterior, formed of the antennal, mandibular, and first maxillary segments, and the posterior, formed of the sixth or labial segment. 54This postoral segment at first appears to be one of the thoracic segments, but is afterwards added to the head, though not until after birth, as it is still separate in the freshly hatched nymph (Fig. 4; see also Kolbe, p. 132, Fig. 59, sq. 5). A. Brandt’s figure of Calopteryx virgo (Pl. 2, Fig. 19) represents an embryo of a stage similar to ours, in which the postoral or sixth (labial) segment is quite separate from the rest of the head. The accompanying figure, copied from our memoir, also shows in a saw-fly larva (Nematus ventricosus) the relations of the labial or sixth segment to the rest of the head. The suture between the labial segment and the preoral part of the head disappears in adult life. From this sketch it would seem that the back part of the head, i.e. of the epicranium, may be made up in part of the tergite or pleurites of the mandibular segment, since the mandibular muscles are inserted on the roof of the head behind the eyes. It is this labial segment which in Corydalus evidently forms the occiput, and of which in most other insects there is no trace in larval or adult life, unless we except certain Orthoptera (Locusta), and the larva of the Dyticidæ.
The following table is designed to show the number and succession of the segments of the head, with their respective segments.
| Tabular View of the Segments, Pieces (Sclerites), and Appendages of the Head | |||
|---|---|---|---|
| Name of Segment | Pieces or Regions of the Head-capsule | Appendages, etc. | |
| Preoral, in early embryo. | 1. Ocellar (Protocerebral). | Epicranium, anterior region with the clypeus labrum, and epipharynx. | Compound and simple eyes (Ocelli). |
| Postoral, in early embryo. | 2. Antennal (Deutocerebral). | Epicranium, including the antennal sockets. | Antennæ. |
| 3. Premandibular, or intercalary (Tritocerebral). | Wanting in postembryonic life, except in Campodea. | Premandibular appendages (in Campodea). | |
| 4. Mandibular. | Epicranium behind the antennæ, genæ. | Mandibles. | |
| 5. 1st Maxillary. | Epicranium, hinder edge? Tentorium. | 1st Maxillæ. | |
| 6. 2d Maxillary, or labial. | Occiput. | 2d Maxillæ or Labium. Post-gula, gula, submentum, mentum, hypopharynx (lingua, ligula), paraglossæ, spinneret. | |
Fig. 38.—Larva (a) of a chalcid, about to pupate, with the head, including the eyes and three ocelli, in the prothoracic segment: b, c, pupa.
The composition of the head in the Hymenoptera.—Ratzeburg stated in 1832 that the head in the adult Hymenoptera (Cynips, Hemiteles, and Formica) does not correspond to that of the larva, but is derived from the head and the first thoracic segment of the larva. Westwood and also Goureau made less complete but similar observations, though Westwood afterwards changed his opinion, and the same view was maintained by Reinhard. Our own observations (as seen in Fig. 38) led us to suppose that this was a mistaken view; that the larval head, being too small to contain that of the semipupa, was simply pushed forward, as in caterpillars. Bugnion, however, reaffirms it in such a detailed way that we reproduce his account. He maintains that the views of Ratzeburg are exact and easy to verify in the chalcid genus Encyrtus, except, however, that which concerns the ventral part and the posterior border of the prothoracic segment.
As the time of transformation approaches, the head of the larva, he says, is depressed and soon concealed under the edge of the prothoracic segment; the latter elongates, becomes thicker and more convex, and within can be seen the two oculo-cephalic imaginal buds. The head of the perfect insect is derived not only from the head of the larva, but also from the portion of the prothoracic segment which is occupied by the buds, i.e. almost its entire dorsolateral face. But the hinder and ventral part of this segment (which contains the imaginal buds of the first pair of legs) takes no part in the formation of the head; these parts, according to Bugnion, towards the end of the larval period detaching themselves so as to become fused with the thorax and constitute the pronotum and the prosternum.
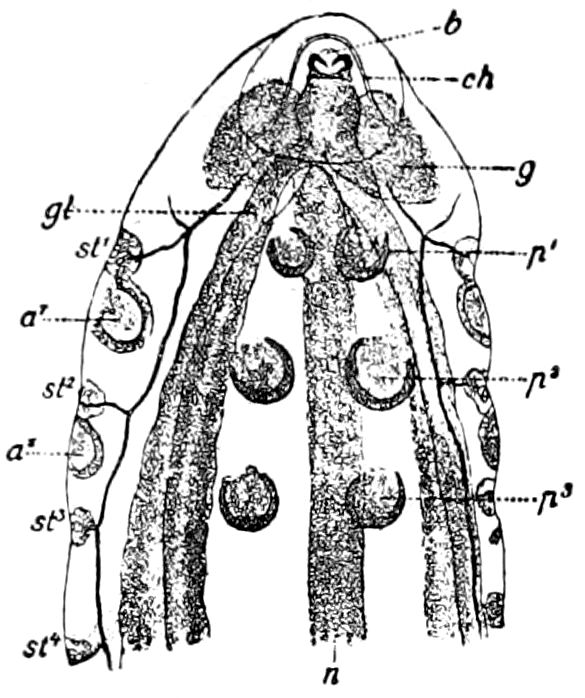
Fig. 39.—Anterior half of larva of Encyrtus, ventral face, showing the upper (wing) and lower (leg) thoracic imaginal buds: b, mouth; ch, chitinous arch; gl, silk gland; g, brain; n, nervous cord; a1, bud of fore, a2, bud of hind, wing; p1–p3, buds of legs; st1–st3, stigmata.
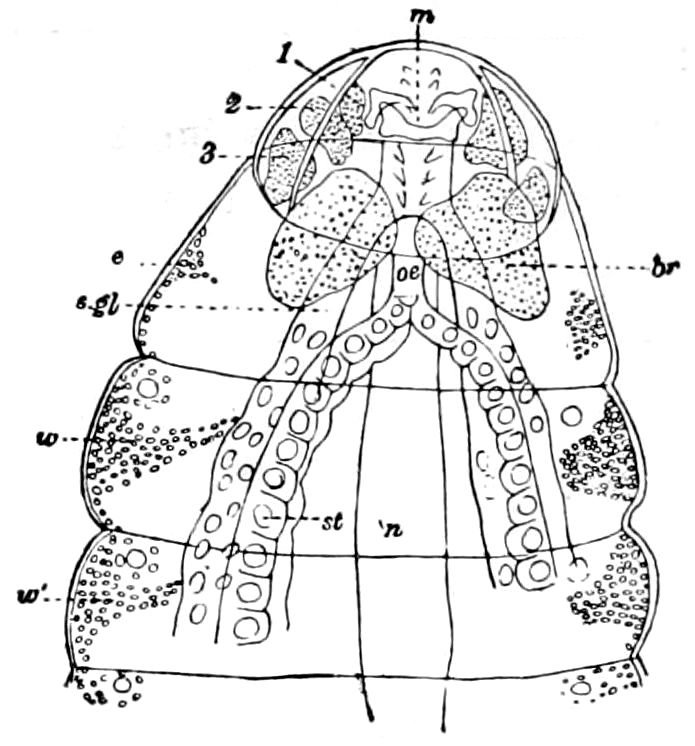
Fig. 40.—Anterior part of Encyrtus larva, 1.2 mm. in length; dorsal face; the cellular masses beginning to form the buds of the wings, eyes, and antennæ: o, eye bud; e, stomach.
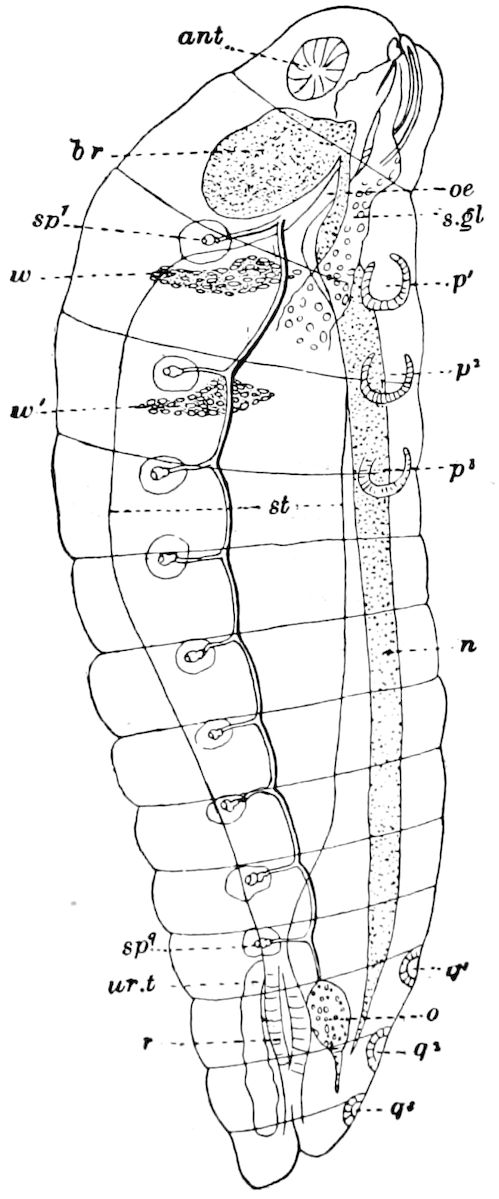
Fig. 41.—Older Encyrtus larva, lateral view, showing the buds of the antennæ (f), legs, and wings; oe, œsophagus; q1, q2, q3, buds of the genital armature; x, rudiment of the sexual gland (ovary or testis); u, urinary tube; i, intestine (rectum); a, anus.
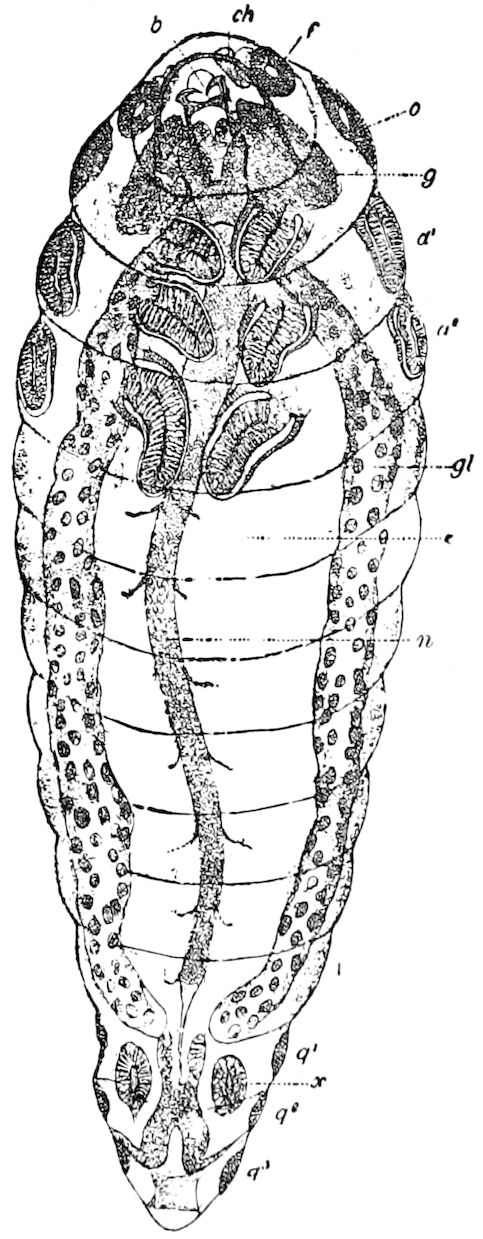
Fig. 42.—A still older larva, ready to transform. The imaginal buds of the antennæ, eyes, wings, and legs have become elongated; lettering as in Fig. 41.—This and Figs. 39–41 after Bugnion.
This mode of formation of the head may be observed still more easily in Rhodites, Hemiteles, and Microgaster, from the fact that their oculo-cephalic buds are much more precocious, and that the eyes are charged with pigment at a period when the insect still preserves its larval form.
“... I believe that this mode of formation of the head occurs in all Hymenoptera with apodous larvæ, in this sense; that a more or less considerable part of the first thoracic segment is always soldered to the head of the larva to constitute the head of the perfect insect. The arrangement of the nervous system is naturally in accord with this peculiarity of development, and the cephalic ganglia of the larva to which the ocular blastems later adapt themselves, are found not in the head, but in the succeeding segment (Figs. 39, 40, 41).
“Relying on these facts, I maintain that the encroachment of the head on the prothorax is a consequence of the preponderance in size of the brain, and indicates the superiority of the Hymenoptera over other insects....”
That the pronotum is derived from the larval prothoracic segment is proved by the fact that the first pair of stigmata becomes what authors call the “prothoracic” stigmata of the perfect insect. But Bugnion thinks that the projection which carries it, and which he calls the shoulder (Figs. 41 and 42), belongs to the mesonotum.
b. Appendages of the head
The antennæ.—These are organs of tactile sense, but also bear olfactory, and in some cases auditory organs; they are usually inserted between or in front of the eyes, and moved by two small muscles at the base, within the head. In the more generalized insects the antennæ are simple, many-jointed appendages, the joints being equal in size and shape. The antennæ articulate with the head by a ball and socket joint, the part on which it moves being called the torulus (Fig. 32, r). In the more specialized forms it is divided into the scape, the pedicel, and a flagellum (or clavola); but usually, as in ants, wasps, and bees, there are two parts, the basal three-jointed one being the scape, and the distal one, the usually long filiform flagellum. The antennæ, especially the flagellum, vary greatly in form in insects of different families and orders, this variation being the result of adaptation to their peculiar surroundings and habits. The number of antennal joints may be one (Articerus, a clavigerid beetle), or two in Paussus and in Adranes cœcus (Fig. 4312), where they are short and club-shaped; in flies (Muscidæ, etc.), they are very short and with few joints, and when at rest lying in a cavity adapted for their reception. In the lamellicorn beetles the flagellum is divided into several leaves, and this condition may be approached in the serrate or flabellicorn antennæ of other beetles. In Lepidoptera, and in certain saw-flies and beetles, they are either pectinate or bipectinate, being in one case at least, that of the Australian Hepialid (Abantiades argenteus), tripectinate (Fig. 44), and in the dipterous (Tachinid) genus Talarocera the third joint is bipectinate (Fig. 45). In Xenos and in Parnus they may be deeply forked, 58while in Otiocerus, two long processes arise from the base, giving it a trifid shape. In dragon-flies and cicadæ, they are minute and hair-like, though jointed, while in the larvæ of many metabolous insects they are reduced to minute three-jointed tubercles. In aquatic beetles, bugs, etc., the antennæ are short, and often, when at rest, bent close to the body, as long antennæ would impede their progress.
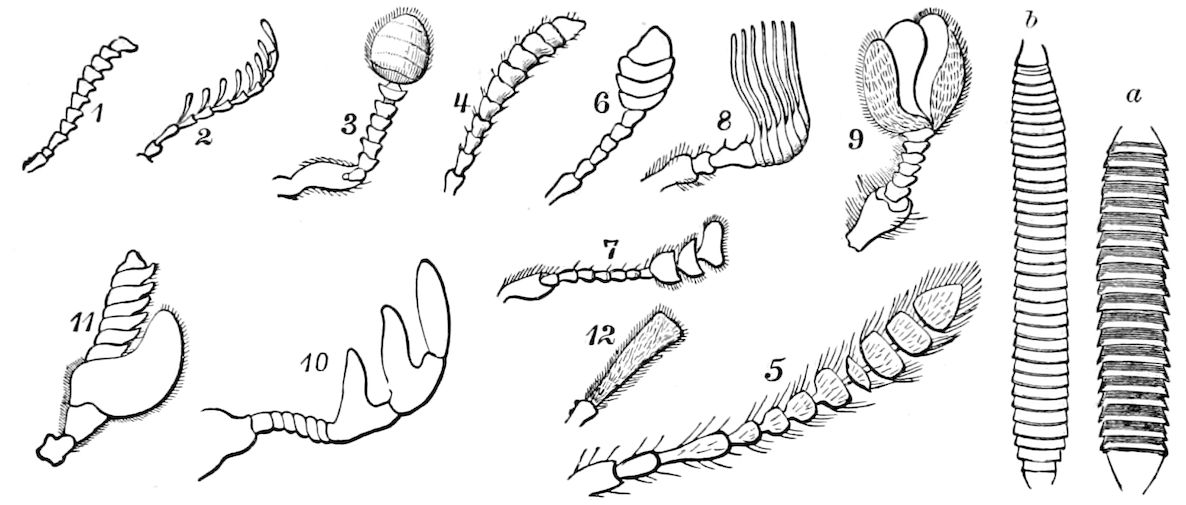
Fig. 43.—Different forms of antennæ of beetles: 1, serrate; 2, pectinate; 3, capitate (and also geniculate); 4–7, clavate; 8, 9, lamellate; 10, serrate (Dorcatoma); 11, irregular (Gyrinus); 12, two-jointed antenna of Adranes cæcus.—After LeConte. a, first joint of flagellum of antenna of Troctes silvarum; b, of T. divinatorius.—After Kolbe.
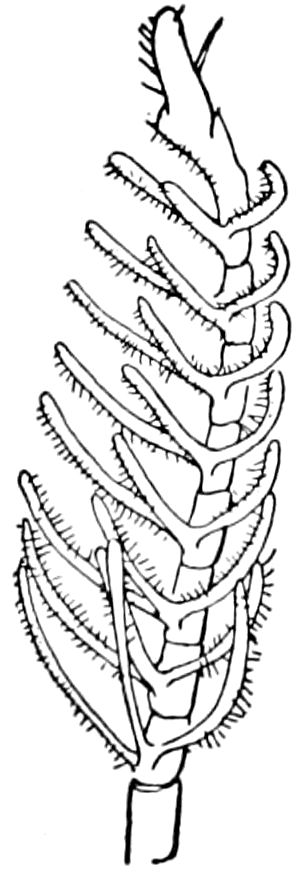
Fig. 44.—Tripectinate antenna of an Australian moth.
While usually more or less sensorial in function, Graber states that the longicorn beetles in walking along a slender twig use their antennæ as a rope-dancer does his balancing pole.
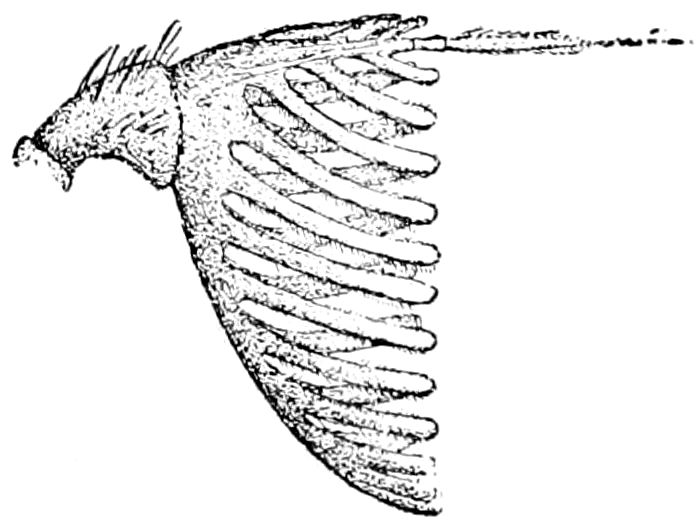
Fig. 45.—Antenna of Talarocera nigripennis, ♂.—After Williston.
Recent examination of the sense-organs in the antennæ of an ant, wasp, or bee enables us, he says, to realize what wonderful organs the antennæ are. In such insects we have a rod-like tube which can be folded up or extended out into space, containing the antennal nerve, which arises directly from the brain and sends a branch to each of the thousands of olfactory pits or pegs which stud its surface. The antenna is thus a wonderfully complex organ, and the insect must 59be far more sensitive to movements of the air, to odors, wave-sounds, and light-waves, than any of the vertebrate animals.
That ants appear to communicate with each other, apparently talking with their antennæ, shows the highly sensitive nature of these appendages. “The honey-bee when constructing its cells ascertains their proper direction and size by means of the extremities of these organs.” (Newport.)
How dependent insects are upon their antennæ is seen when we cut them off. The insect is at once seriously affected, its central nervous system receiving a great shock, while it gives no such sign of distress and loss of mental power when we remove the palpi or legs. On depriving a bee of its antennæ, it falls helpless and partially paralyzed to the earth, is unable at first to walk, but on partly recovering the use of its limbs, it still has lost the power of coördinating its movements, nor can it sting; in a few minutes, however, it becomes able to feebly walk a few steps, but it remains over an hour nearly motionless. Other insects after similar treatment are not so deeply affected, though bees, wasps, ants, moths, certain beetles, and dragon-flies are at first more or less stunned and confused.
The antennæ afford salient secondary sexual differences, as seen in the broadly pectinated antennæ of male bombycine moths, certain saw-flies (Lophyrus), and many other insects.
The mouth-parts, buccal appendages, or trophi, comprise, besides the labrum, the mandibles and maxillæ.
The mandibles.—These are true jaws, adapted for cutting, tearing, or crushing the food, or for defence, while in the bees they are used as tools for modelling in wax, and in Cetonia, etc., as a brush for collecting pollen. They are usually opposed to each other at the tips, but in many carnivorous forms their tips cross each other like shears. They are situated below the clypeus on each side, and are hinged to the head by a true ginglymus articulation, consisting of two condyles or tubercles to which muscles are attached, the principal ones being the flexor and great extensor (Fig. 48). They are solid, chitinous, of varied shapes, and in the form of the teeth those of the same pair differ somewhat from each other (Fig. 46 A). In the pollen-eating beetles (Cetoniæ) and in the dung-beetles (Aphodius, etc.) the edge is soft and flexible. In the males of Lucanus, etc. (Fig. 47), and of Corydalus (Fig. 29), they are of colossal size, and are large and sabre-shaped in the larvæ of water-beetles, ant-lions, Chrysopa, etc. where they are perforated at the tips, through which the blood of their prey is sucked.
While the mandibles are generally regarded as composed of a 60single piece, in Campodea and Machilis there appears to be an additional basal piece apparently corresponding to the stipes of the first maxilla, and separated by a faint suture from the molar or distal joint. In Campodea there is a minute movable appendage figured both by Meinert and by Nassonow, which appears to represent the lacinia of the maxilla (Fig. 48). Wood-Mason has observed in the mandibles of the embryo of a Javanese cockroach, Blatta (Panesthia) javanica, indications of “the same number of joints as in that of chilognathous myriopods, or one less than in that of Machilis.” Also he adds: “In both ‘larvæ’ and adults of Panesthia javanica a faint groove crosses the ‘back’ of the mandible at the base. This groove appears to be the remains of the joint between the third and apical segments of the formerly 4–segmented mandibles.”
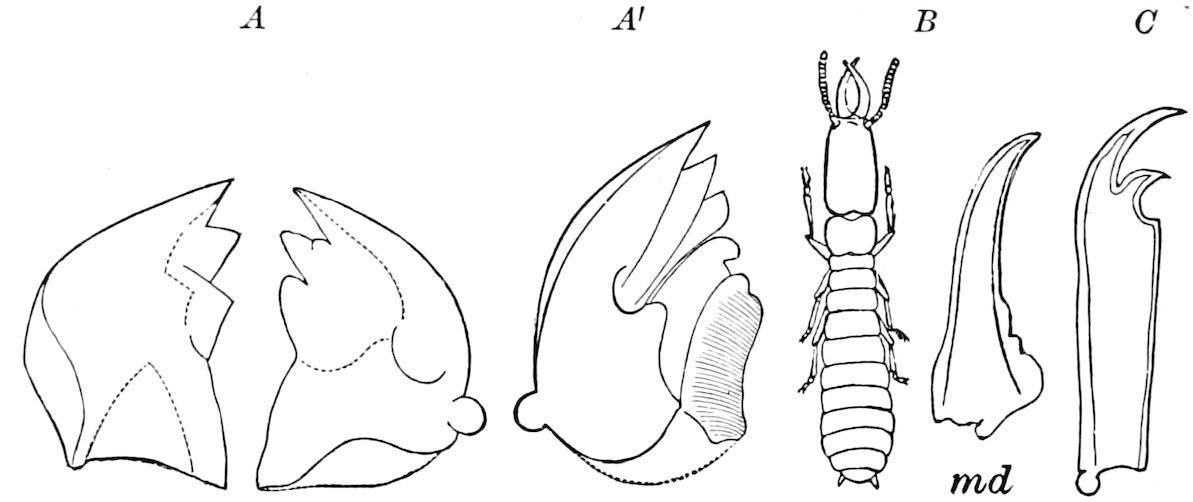
Fig. 46.—Various forms of mandibles. A, right and left of Termopsis. A′, showing at the shaded portion the “molar” of Smith. B, Termes flavipes, soldier; md, its mandible. C, Panorpa.
Fig. 47.—Chiasognathus grantii, reduced. Male.—After Darwin.
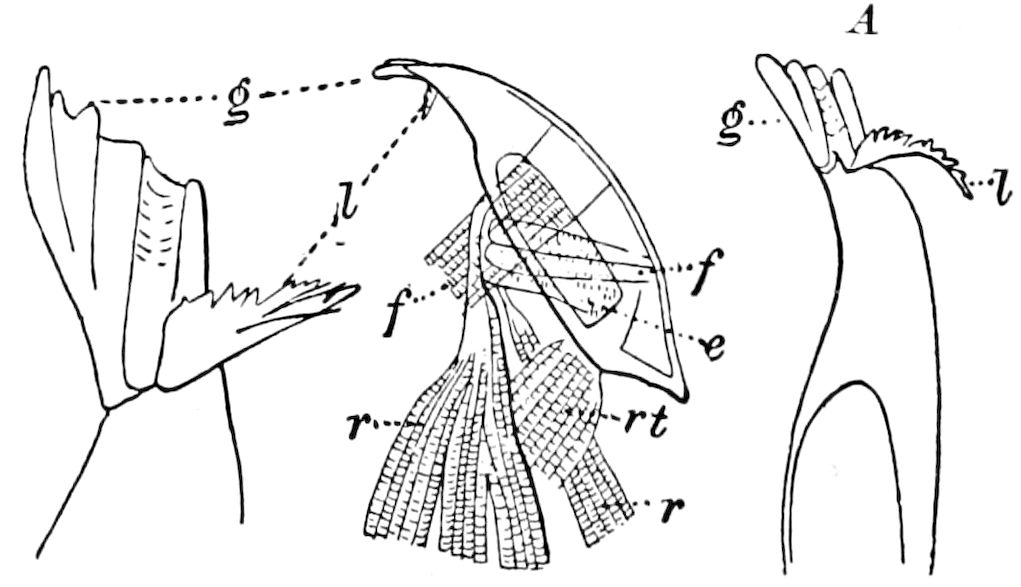
Fig. 48.—Mandible of Campodea: l, prostheca or lacinia; g, galea; f, f, flexor muscles; e, extensor; r, r, retractor; rt, muscle retaining the mandible in its place.—After Meinert. A, extremity of the same.—After Nassonow.
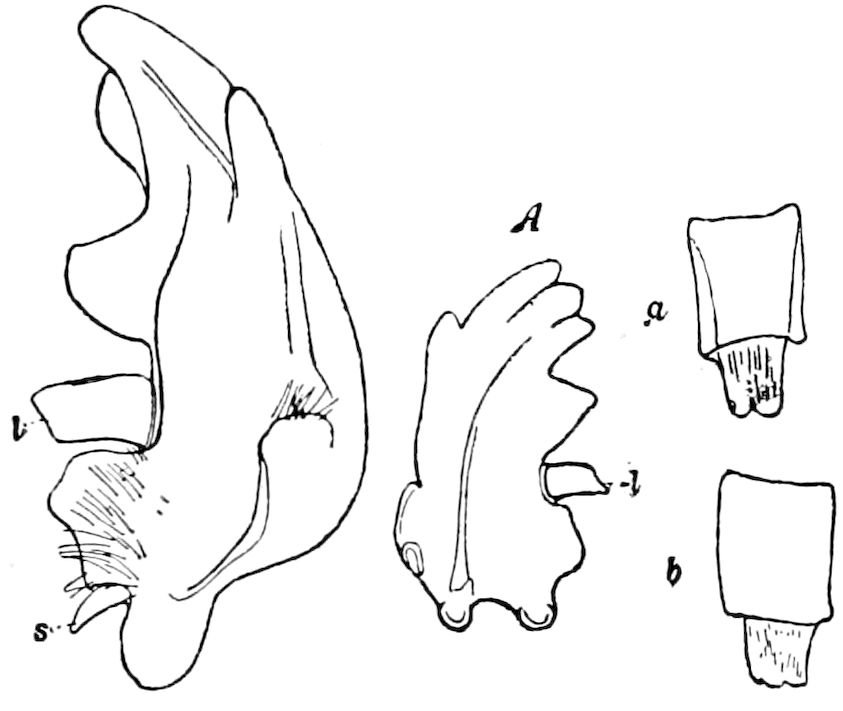
Fig. 49.—Mandible of Passalus cornutus with the prostheca (l): A, that of a Nicaraguan species; a, inside, b, outside view, with the muscle.
He also refers to the prostheca of Kirby and Spence (Fig. 49), which he thinks appears to be a mandibular lacinia homologous with it in Staphylinidæ and other beetles (J. B. Smith also considers it as “homologous to the lacinia of the maxilla”), and on examining it in P. cornutus and a Nicaragua species (Fig. 49), we adopt his view, since we have found that it is freely movable and attached by a tendon and muscle to the galea. In the rove beetles (Goërius, Staphylinus, etc.) and in the subaquatic Heteroceridæ, instead of a molar process, is a membranous setose appendage not unlike the coxal appendages of Scolopendrella, movably articulated to the jaw, which he thinks answers to the molar branch of the jaws in Blatta and Machilis. “It has its homologue in the diminutive Trichopterygidæ in the firmly chitinized quadrant-shaped second mandibular joint, which is used in a peculiar manner in crushing the food”; also in the movable tooth of the Passalidæ, and in the membranous inner lobe of the mandibles of the goliath-beetles, etc.
J. B. Smith has clearly shown that the mandibles are compound in certain of the lamellicorns. In Copris carolina (Fig. 50), he says, the small membranous mandibles are divided into a basal piece (basalis), the homologue of the stipes in the maxilla; another of the basal pieces he calls the molar, and this is the equivalent of the subgalea, while a third sclerite, only observed in Copris, is the conjunctivus, the lacinia (prostheca) being well developed. Smith therefore concludes “that the structure of the mandible is fundamentally the same as that of the labium and maxilla, and that we have an equally complex organ in point of origin. Its usual function, however, demands a powerful and solid structure, and the sclerites are in most instances as thoroughly chitinized and so closely united to the others that practically there is only a single piece, in which the homology is obscured.” (Trans. Amer. Ent. Soc., xix, pp. 84, 85. 1892.) From the studies of Smith and our observations on Staphylinus, Passalus, Phanæus, etc. (Fig. 50, A, B) we fully agree with the view that the mandibles are primarily 3–lobed appendages like the maxillæ. Nymphal Ephemerids have a lacinia-like process. (Heymons.)
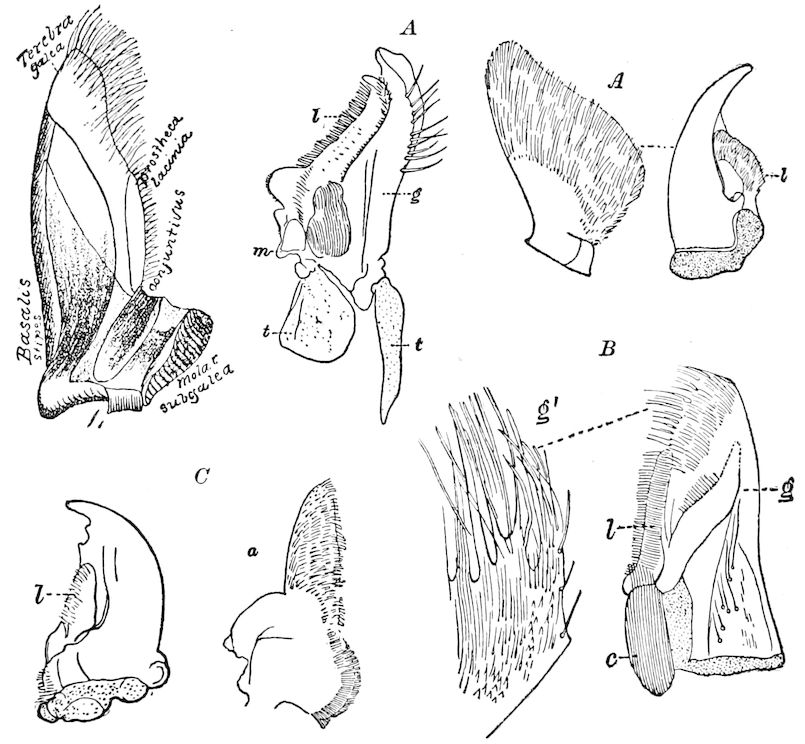
Fig. 50.—Mandible of Copris carolina.—After Smith. A′C. anaglypticus. A (figure to right), do. of Leistotrophus cingulatus; B, of Phanæus carnifex; g′, end of galea,—g, enlarged; c, conjunctivus. C, of Meloë angusticollis: l, lacinia; a, lacinia enlarged.
Mandibles are wanting in the adults of the more specialized Lepidoptera, being vestigial in the most generalized forms (certain Tineina and Crambus), but well developed in that very primitive moth, Eriocephala (Fig. 51). They are also completely atrophied in the adult Trichoptera, though very large and functional in the pupa of these insects (Fig. 52), as also in the pupa of Micropteryx (Fig. 53). They are also wanting in the imago of male Diptera and in the females of all flies except Culicidæ and Tabanidæ.
They are said by Dr. Horn to be absent in the adult Platypsyllus castoris, though well developed in the larva; and functional mandibles are lacking in the Hemiptera.
The first maxillæ.—These highly differentiated appendages are inserted on the sides of the head just behind the mandibles and the mouth, and are divided into three lobes, or divisions, which are supported upon two, and sometimes three basal pieces, i.e. the basal 63joint or cardo, the second joint or stipes, with the palpifer, the latter present in Termitidæ (Fig. 54, plpgr), but not always separately developed (Fig. 55). The cardo varies in shape, but is more or less triangular and is usually wedged in between the submentum and mandible. It is succeeded by the stipes, which usually forms the support for the three lobes of the maxilla, and is more or less square in shape.
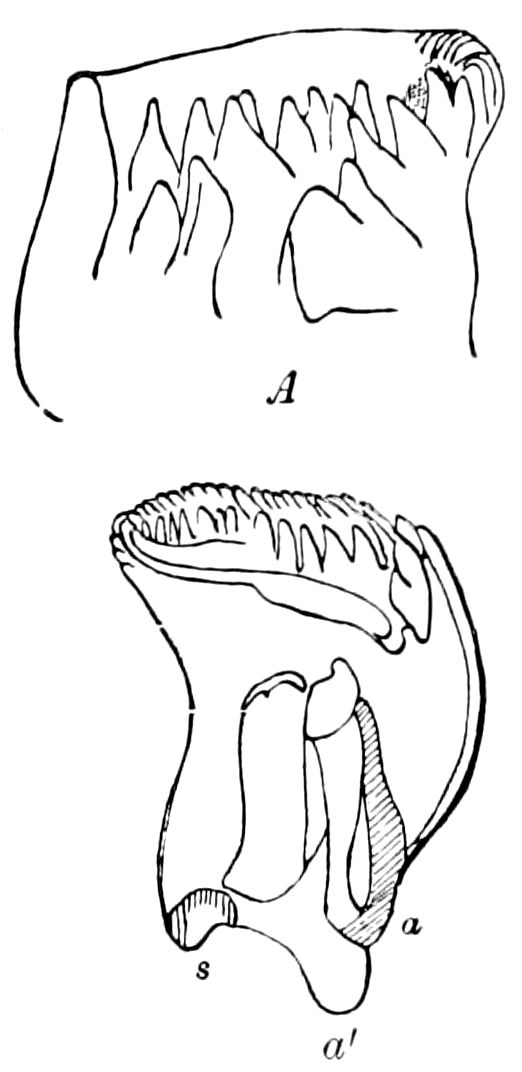
Fig. 51.—Mandible of Eriocephala calthella: a, a′, inner and outer articulation; s, cavity of the joint (acetabulum); A, end seen from one side of the cutting edge.—After Walter.
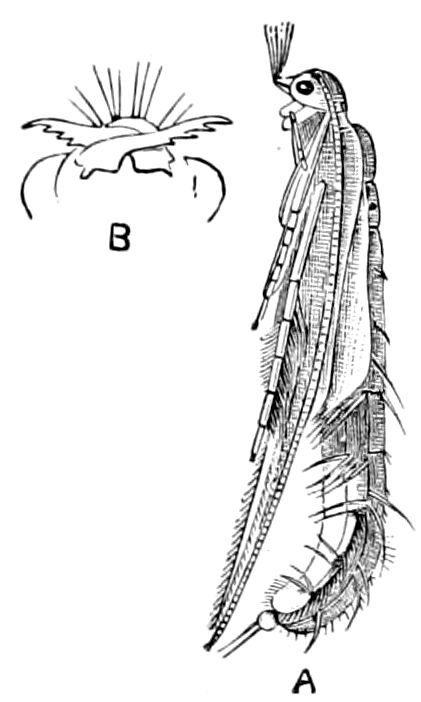
Fig. 52.—A, Pupa of Phryganea pilosa.—After Pictet. B, mandibles of pupa of Molanna angustata.—After Sharp.
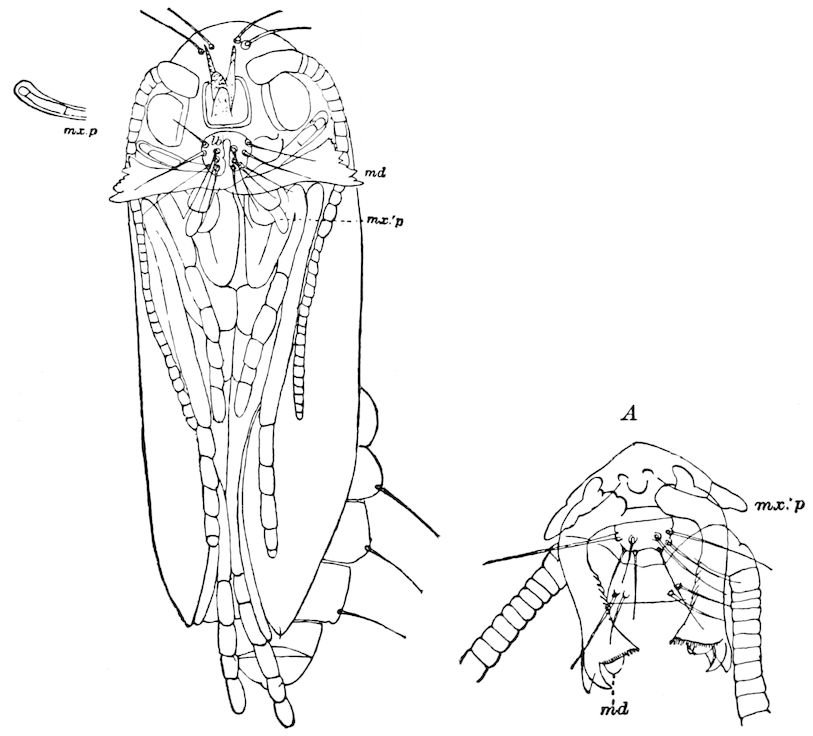
Fig. 53.—Pupa of Micropteryx purpuriella, front view: md, mandibles; mx.p, maxillary palpus, end drawn separately; mx.’p, labial palpi; lb, labrum; A, another view from a cast skin.
The three distal divisions of the maxilla are called, respectively, beginning with the innermost, the lacinia, galea, and palpifer, the latter being a lobe or segment bearing the palpus. The lacinia is more or less jaw-like and armed on the inner edge with either flexible or stiff bristles, spines, or teeth, which are very variable in shape and are of use 64as stiff brushes in pollen-eating beetles, etc. The galea is either single-jointed and helmet-shaped or subspatulate, as in most Orthoptera, or 2–jointed in Gryllotalpa, or lacinia-like in Myrmeleon (Fig. 55, C); or, in the Carabidæ (Fig. 56) and Cicindelidæ, it is 2–jointed and in form and function like a palpus.
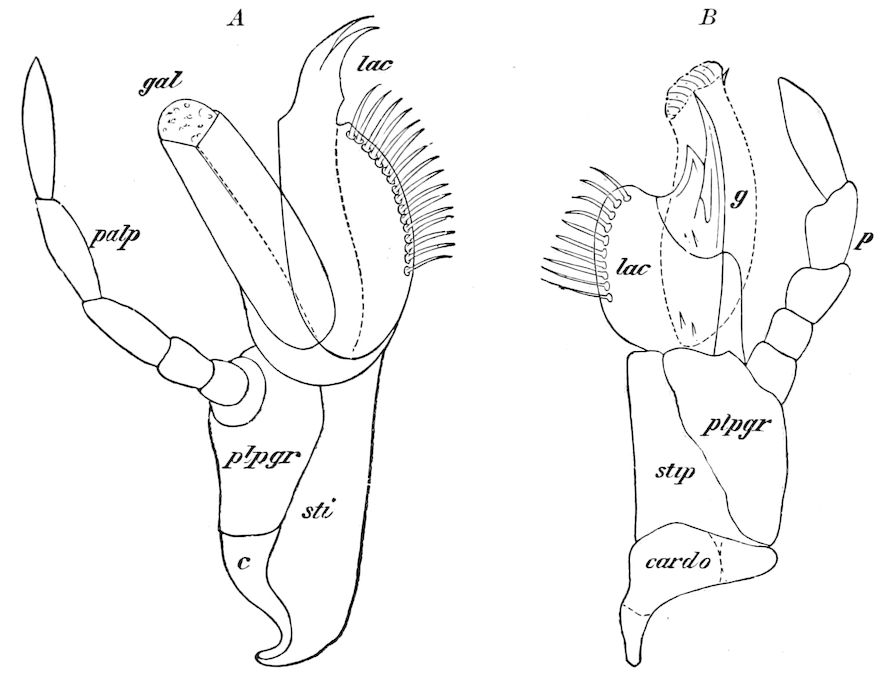
Fig. 54.—A, maxilla of Termopsis angusticollis. B, Termes flavipes: c, cardo; sti, stipes; plpgr, palpiger; palp, palpus; lac, lacinia; g, gal, galea.
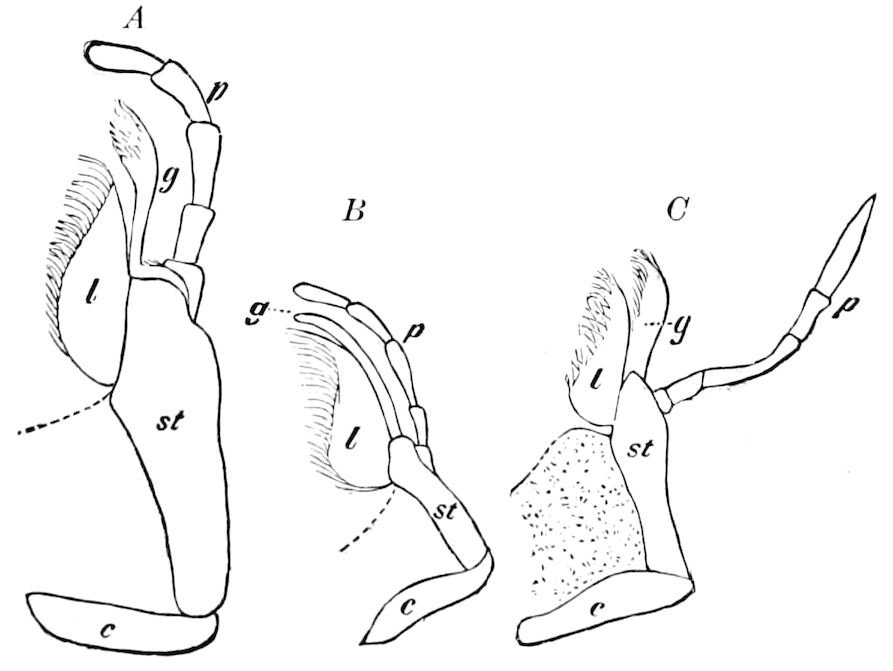
Fig. 55.—A, maxilla of Mantispa brunnea. B, Ascalaphus longicornis. C, Myrmeleon diversum. Lettering as in Fig. 54.
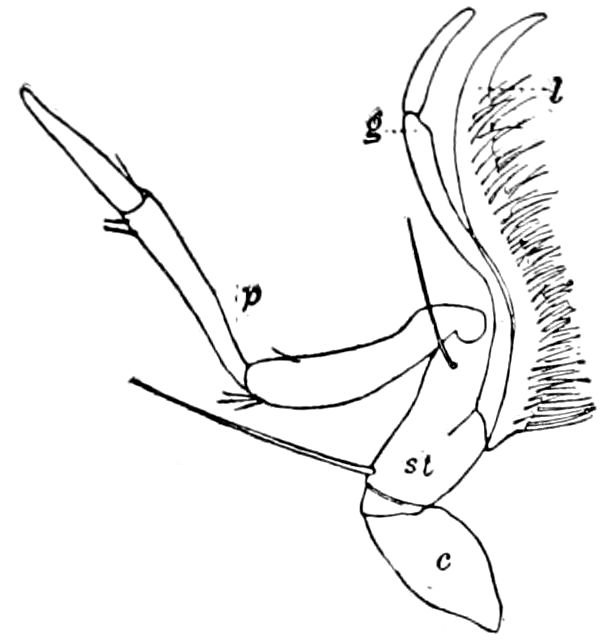
Fig. 56.—Maxilla of a carabid, Anophthalmus tellkampfii: l, lacinia; g, 2–jointed galea; p, palpus; st, stipes; c, cardo.
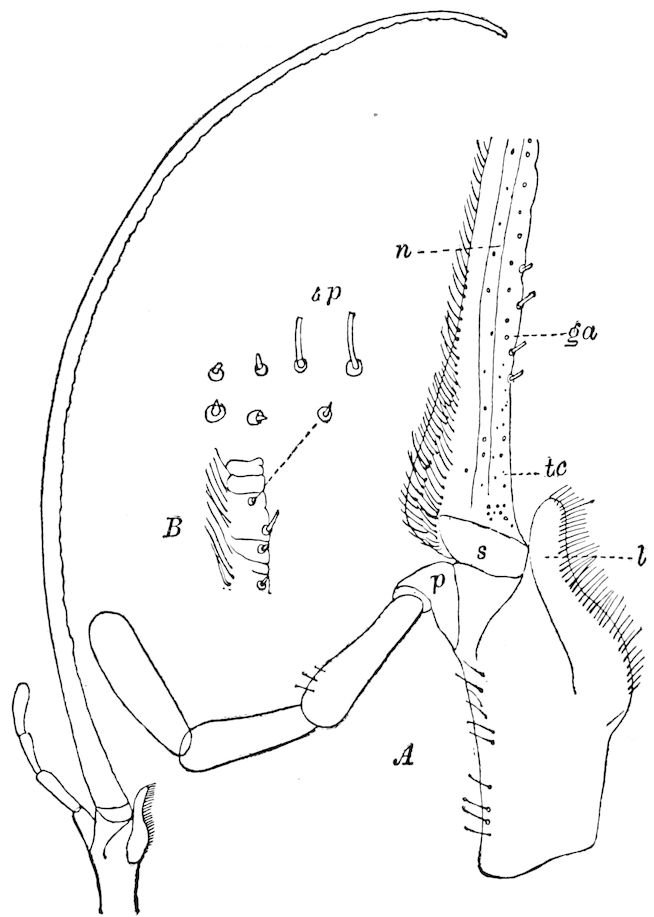
Fig. 57.—Maxilla of Nemognatha, ♀, from Montana. A, base of maxilla enlarged to show the taste-papillæ (tp) and cups (tc), on the galea (ga). B, part of end of galea to show the imperfect segments and taste-organs: n, nerve; a ganglionated nerve supplies each taste-papilla or cup; l, lacinia; p, palpifer; s, subgalea.
Fig. 58.—Maxilla of Panorpa.
Fig. 59.—Maxilla of Limnephilus pudicus: mx, stipes; lac, galea.
The palpus is in general antenniform and is composed of from 1 to 6 joints, being usually 4– or 5–jointed, and is much longer than the galea. In the maxilla of the beetle Nemognatha (Fig. 57), the galea is greatly elongated, the two together forming an imperfect tube or proboscis and reminding one of the tongue of a moth, while the lacinia is reduced. In the Mecoptera the lacinia and galea are closely similar (Fig. 58); in the Trichoptera only one of the lobes is present (Fig. 59), while in the Lepidoptera the galea unites with its mate to form the so-called tongue (Fig. 60). The maxilla of the male of Tegeticula yuccasella is normal, though the galeæ are separate; but in the female, what Smith regards as the palpifer (the “tentacle” of Riley) is 66remarkably developed, being nearly as long as the galea (Fig. 61) and armed with stout setæ, the pair of processes being adapted for holding a large mass of pollen under the head.
Fig. 60.—Tongue of Aletia xylina, with the end magnified.—Pergande del., from Riley. A, much reduced maxilla (mx) of Paleacrita vernata; mx.p, palpus.
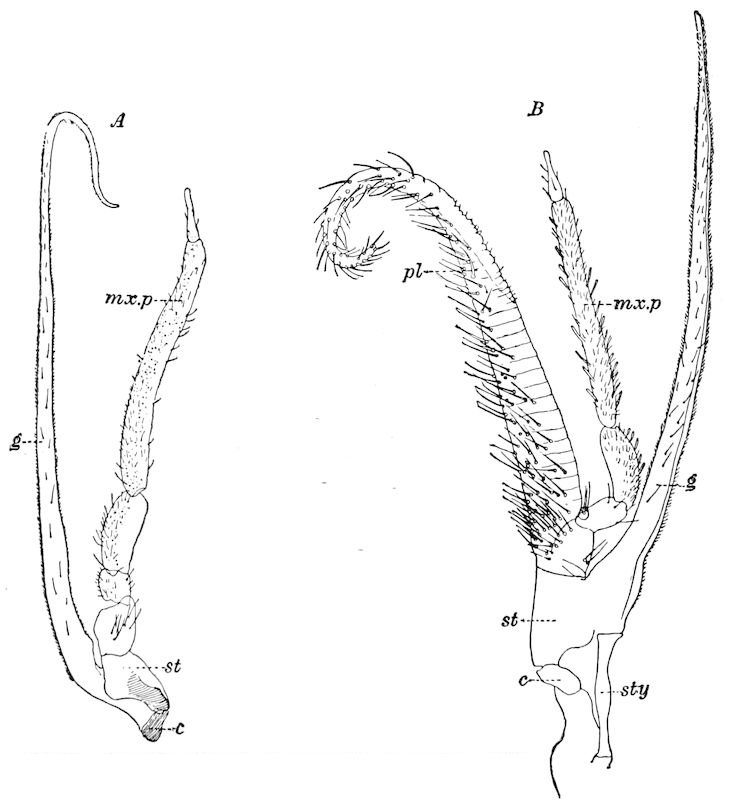
Fig. 61.—A, maxilla of Tegeticula yuccasella, ♂: g, galea. B, ♀: pl, enormously developed palpifer; mx.p, palpus; c, cardo; st, stipes; sty, stylus.
In coleopterous larvæ the maxillæ are 2–lobed 67(Fig. 62), the galea being undifferentiated, but in those of saw-flies the galea is present (Fig. 63, gal).
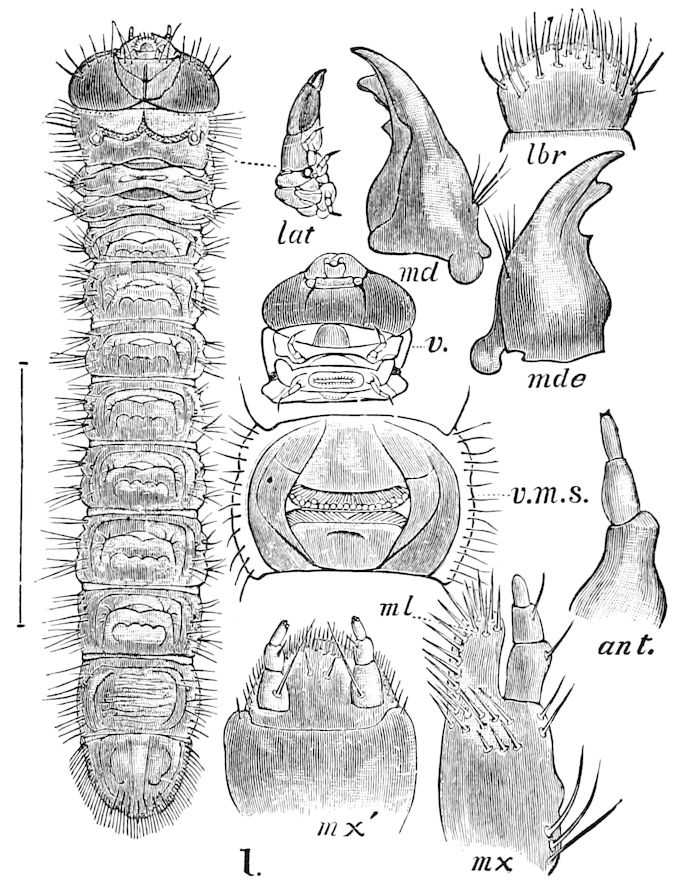
Fig. 62.—Larva of Rhagium lineatum: lat, lateral view of head and thoracic segments; mx, first maxilla; ml, undifferentiated lacinia and galea; v, under side of head and pro- and meso- thoracic segments; v.m.s., one of the middle ventral segments, magnified six times; mx′, 2d maxilla.
It now seems most probable that in the first maxillæ we have the primary form of buccal appendage of insects, the appendage being composed of three basal pieces with three variously modified distal lobes or divisions; and that the mandibles and second maxillæ are modifications of this type.
How wonderfully the maxillæ of the Lepidoptera are modified, and the peculiar shapes assumed in the Diptera, Hymenoptera, and other groups, will be stated in the accounts of those orders, but it is well to recall the fact that in the most primitive and generalized moth, Eriocephala, the lacinia is well developed (Fig. 64).
As Newport remarks, the office of the maxillæ in the mandibulate insects is of a twofold kind; since they are adapted not only for seizing and retaining the food in the mouth, but also as accessory jaws, since they aid the mandibles in comminuting it before it is passed on to the pharynx and swallowed. Hence, as the food varies so much in nature and situation, it will be readily seen that the maxillæ, especially their distal parts, vary correspondingly. Thus far no close observations on the exact use of the first and second maxillæ have been published.
The palpi also are not only organs of touch, but in some cases act as hands and also bear minute sense-organs, the function 68of which is unknown, but would appear to be usually that of smell.
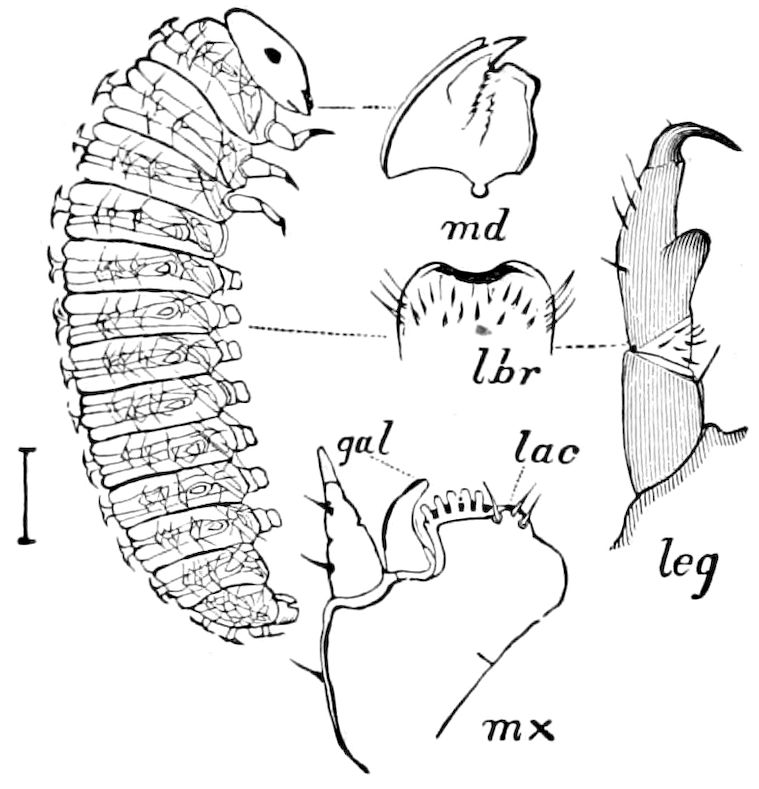
Fig. 63.—Selandria larva, common on Carya porcina, with details of mouth-parts: leg, leg; mx, maxilla; gal, galea; lac, lacinia.
The second maxillæ.—The “under-lip” or labium of insects is formed by the fusion at the basal portion of what in the embryo are separate appendages, and which arise in the same manner as the first maxillæ. They are invariably solidly united, no cases of partial or incomplete fusion being known. The so-called labium is situated in front of the gula or gular region, and is bounded on each side by the gena, or cheek. As already observed, the second maxillæ appear to be the appendages of the last or occipital segment of the head.
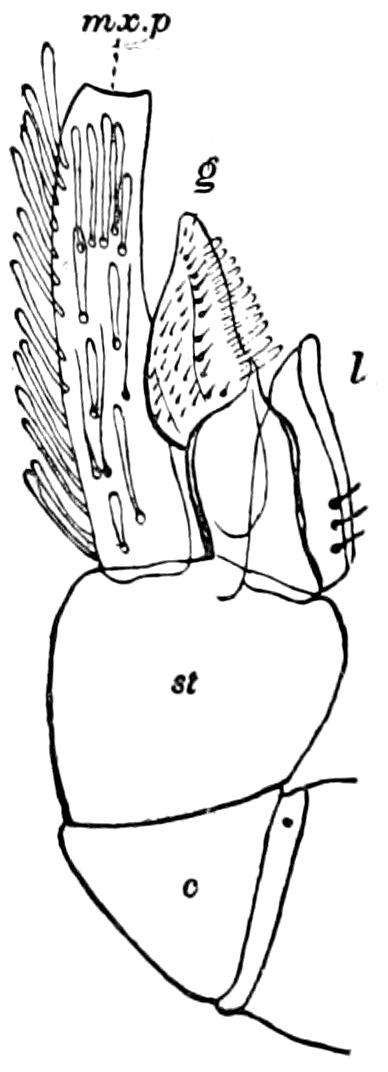
Fig. 64.—Maxilla of Eriocephala calthella: l, lacinia; g, galea; mx.p, maxillary palpus; st, stipes; c, cardo.—After Walter.
The second maxillæ are very much differentiated and vary greatly in the different orders, being especially modified in the haustellate or suctorial orders, notably the Hymenoptera and Diptera. In the mandibulate orders, particularly the Orthoptera, where they are most generalized and primitive in shape and structure, they consist of the following parts: the gula (a postgula is present in Dermaptera), submentum (lora of Cheshire, i, p. 91), mentum, palpifer, the latter bearing the palpi; the lingua (ligula) and paraglossæ, while the hypopharynx or lingua is situated on the upper side. The labial palpi are of the same general shape as those of the first maxillæ, but shorter, with very rarely more than three joints, though in Pteronarcys there are four. Leon has detected vestigial labial palpi in several Hemiptera (Fig. 73). As to the exact nature and limits of the gula, we are not certain; it is not always present, and may be only a differentiation of the submentum, or the latter piece may be regarded as a part of the gula.
We are disposed to consider the second maxillæ as morphologically nearly the exact equivalents of the first pair of maxillæ, and if we 69adopt this view it will greatly simplify our conception of the real nature of this complicated organ. The object of the fusion of the basal portion appears to be to form an under-lip, in order both to prevent the food from falling backwards out of the mouth, and, with the aid of the first pair of maxillæ, to pass it forward to be crushed between the mandibles, the two sets of appendages acting somewhat as the tongue of vertebrates to carry and arrange or press the morsels of food between the teeth or cutting edges of the mandibles.
The spines often present on the free inner edges of the first and second maxillæ (Figs. 54, 62) form rude combs which seem to clean the antennæ, etc., often aiding the tibial combs in this operation.
The submentum and mentum, or the mentum when no submentum is differentiated (with the gula, when present), appear to be collectively homologous with the cardines of the first pair of maxillæ, together with the palpifers and the stipites.[15] These pieces are more or less square, and have a slightly marked median suture in Termitidæ, the sign of primitive fusion or coalescence.
The most primitive form of the second maxillæ occurs in the Orthoptera and in the Termitidæ. The palpifer is either single (Periplaneta, Diapheromera, Gryllidæ) or double (Blatta orientalis, Locustidæ). In Prisopus the single piece in front of the palpifer is in other forms divided, each half (Blatta, Locustidæ, Acrydidæ) bearing the two “paraglossæ,” which appendages in reality are the homologues of the lacinia and galea of the first maxillæ.[16] In the Termitidæ (Fig. 65) the lingua is not differentiated from the palpifer, and the two paraglossæ (or the lamina externa and interna of some authors) with the palpus are easily seen to be the homologues of the three lobes of the first maxillæ. In the Perlidæ (Pteronarcys, Fig. 66) the palpifer is divided, while the four paraglossæ arise, as in Prisopus and Anisomorpha, from an undivided piece, the lingua not being visible from without. In the Neuroptera the lingua or ligula is a large, broad, single lobe, without “paraglossæ,” and the palpifer is either single (Myrmeleon, Fig. 67), or divided (Mantispa, Fig. 68). 70In Corydalus (Fig. 29) the palpifer forms a single piece, and the lingua is undivided, though lobed on the free edge.
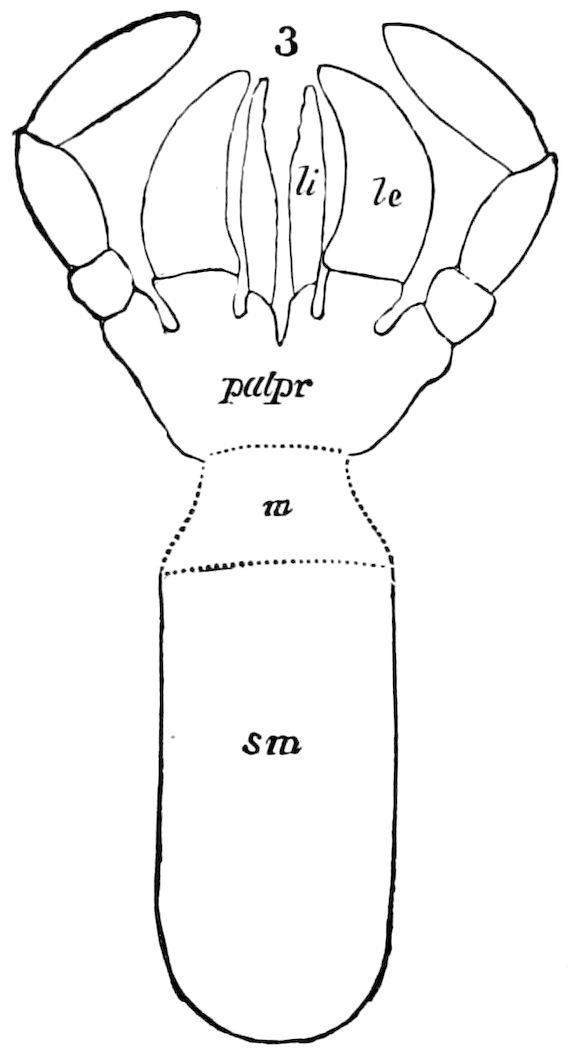
Fig. 65.—Second maxillæ of Termopsis angusticollis: li, the homologue of the lacinia; le, galea.
In the metabolic orders above the Neuroptera the lingua is variously modified, or specialized, with no vestiges of the lacinia or galea, except in that very primitive moth, Eriocephala, in which Walter found a minute free galea, me, and an inner lobe (Figs. 76, 77), the lacinia.
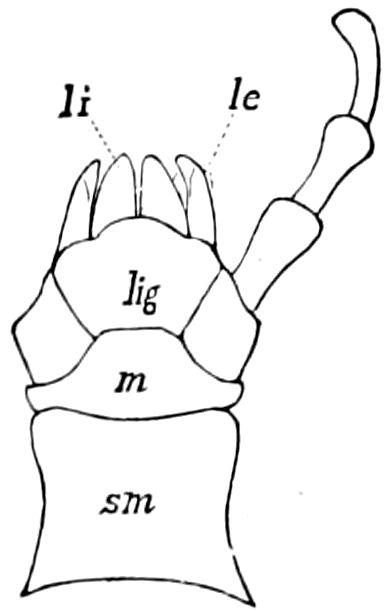
Fig. 66.—Second maxillæ of Pteronarcys californica.
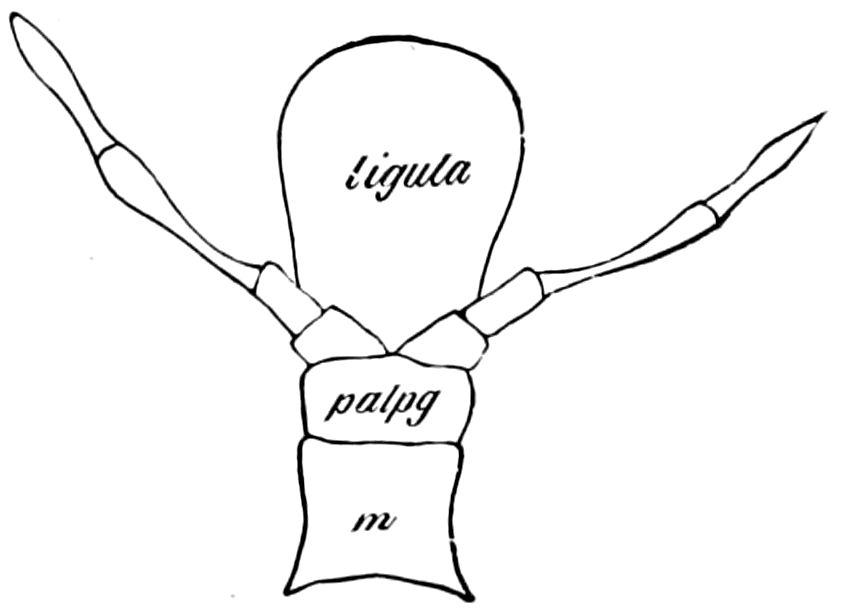
Fig. 67.—Second maxillæ of Myrmeleon diversum.
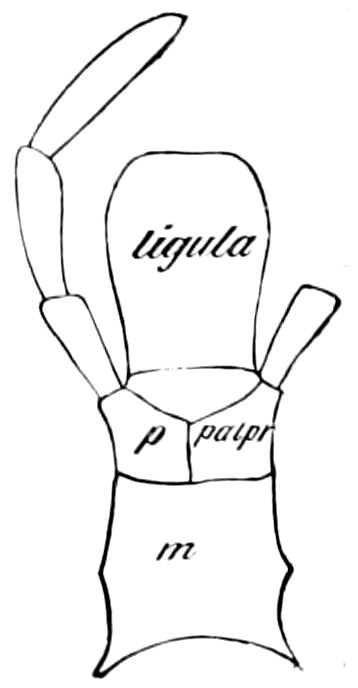
Fig. 68.—Second maxillæ of Mantispa brunnea.
The hypopharynx.—While in its most generalized condition, as in Synaptera, Dermaptera, Orthoptera, and Neuroptera, this anterior median fold or outgrowth of the labium forming the floor of the mouth may retain the designation of “tongue,” lingua, or ligula; in its more specialized form, particularly when used as a piercing or lapping organ, the use of the name hypopharynx seems most desirable. And this is especially the case since, like the epipharynx, it is morphologically a median structure, and while the epipharynx forms the soft, sensitive roof of the mouth, or pharynx; its opposite, the hypopharynx, rises as a fold from the floor of the mouth, forming in its most generalized condition a specialized fold of the buccal integument. In certain cases, as in the honey-bee, the very long slender “tongue” or hypopharynx is evidently, as in the case of the epipharynx, a highly sensitive armature of the mouth.
71In all insects this organ—whether forming a soft, tongue-like, anterior portion or fold of the labium, and “continuous with the lower wall of the pharynx,” or a hard, piercing, awl-like appendage (fleas and flies), or a long, slender, hairy or setose, trough-like structure like the “tongue” of the honey-bee—has a definite location at the end and on the upper side of the labium, and serves to receive at its base the external opening of the salivary duct.
The hypopharynx, as well shown in its lingua condition in Orthoptera, is continuous with and forms the anterior part or fold of the base of the coalesced second maxillæ. It does not seem to be paired, or to represent a pair of appendages.
Opinion regarding the homology of this unpaired piercing organ is by no means settled, and while there is a general agreement as to the nature of the paired mouth-parts, recent observers differ very much as to the morphology of the organ in question.
It is the langue or lingua of Savigny (1816), the ligula of Kirby and Spence (1828), the langue ou languette (lancette médiane du suçoir) of Dugès (1832), the lingua of Westwood (Class, ins., ii, p. 489, 1840), “the unpaired median piercing organ” (“the analogon of the epipharynx of Diptera”) of Karsten (1864), the “tongue” of Taschenberg (1880).
The name hypopharynx was first proposed by Savigny in 1816, who, after naming the membranous plate which has for its base the upper side of the pharynx, the epipharynx, remarks: “Dans quelques genres, notamment dans les Eucères, le bord inférieur de ce même pharynx donne naissance à un autre appendice plus solide que le précédent, et qui s’emboîte avec lui. Je donnerai à ce dernier le nom de langue ou d’hypopharynx. Voilà donc la bouche des Hyménoptères composée de quatre organes impaires, sans y comprendre la ganache ou le menton; savoir, la lèvre supérieure, l’épipharynx, l’hypopharynx, et la lèvre inférieure, et de deux organes paires, les mandibules et les mâchoires.”
As stated by Dimmock: “The hypopharynx is usually present in Diptera (according to Menzbier absent in Sargus), and contains a tube, opening by a channel on its upper surface; this channel extends back, more or less, from the tip, and is the outlet for the salivary secretion. The tip of the hypopharynx may be naked and used as a lance (Hæmatopota, according to Menzbier), or may be hairy (Musca). The upper side of the base of the hypopharynx is continuous with the lower wall of the pharynx; its under surface may entirely coalesce with the labium (Culex, male), may join the labium more or less, anterior to the month (Musca), or, if 72either mandibles or maxillæ are present, its base may join them (Culex, female).” (p. 43.)
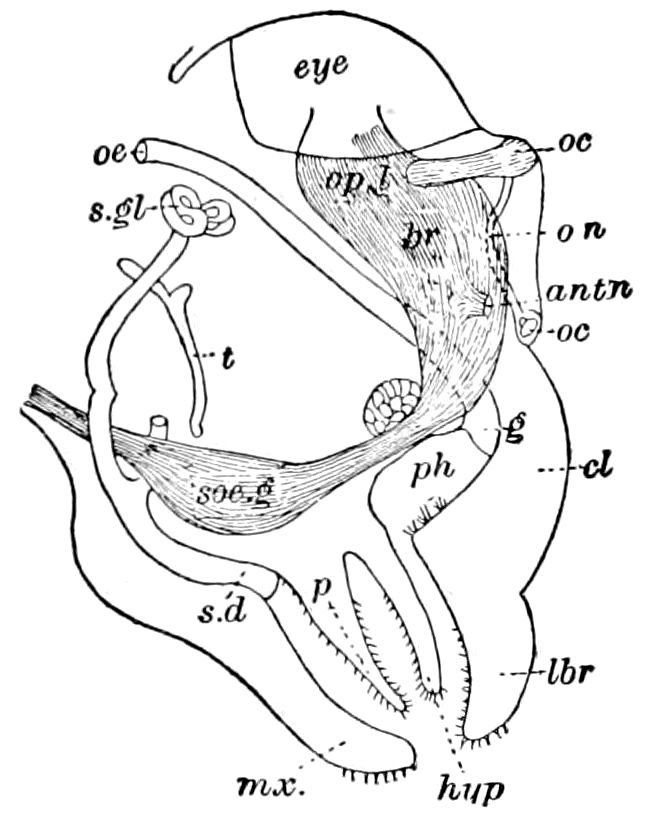
Fig. 69.—Section of head of Machilis maritima: hyp, hypopharynx; lbr, labrum; t, tentorium; ph, room in which the mandibles move on each other; p, paraglossa; mx, labium; sd, salivary duct; s.gl, salivary gland. oe, œsophagus.—After Oudemans.
We will now briefly describe the lingua, first of the mandibulate or biting insects, and then its specialized form, the hypopharynx of the haustellate and lapping insects.
The lingua (hypopharynx) exists in perhaps its most generalized condition in the Thysanura (Fig. 69), where it forms a soft projection, having the same relations as in Anabrus and other Orthoptera.[17]
In the cockroach (Fig. 70), as stated by Miall and Denny, the lingua is a chitinous fold of the oral integument situated in front of the labium, and lying in the cavity of the mouth. The common duct of the salivary glands enters the lingua, and opens on its hinder surface. The lingua is supported by a chitinous skeleton (Figs. 70, B; 82, shp). “The thin chitinous surface of the lingua is hairy, like other parts of the mouth, and stiffened by special chitinous rods or bands.” (Miall and Denny.)
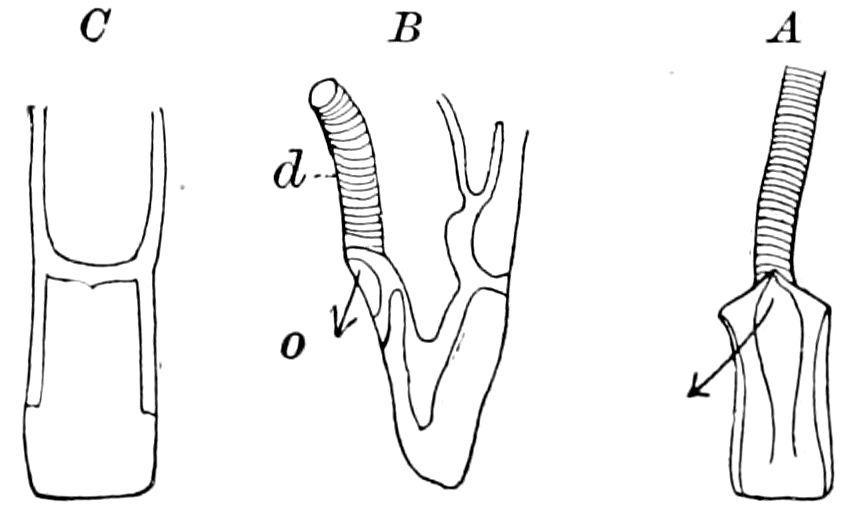
Fig. 70.—Hypopharynx of Periplaneta orientalis; the arrow points out of the opening of the salivary duct: A, origin of salivary duct. B, side view. C, front view.—After Miall and Denny.
In the Acrydiidæ (Melanoplus femur-rubrum) the tongue is a large, membranous, partly hollow expansion of the base of the labium. It may be exposed by depressing the end of the labium, when the opening of the salivary duct may be seen at the bottom or end of the space or gap between the hinder base of the tongue, and the inner anterior base of the labium, as shown by the arrows in Fig. 70. It is somewhat pyriform, slightly keeled above, and bearing fine stiff bristles, which, as they point more or less inwards, probably aid in retaining the food within the mouth. The 73base of the tongue is narrow, and extends back to near the pharynx, there being on the floor of the mouth, behind the tongue, two oblique, slight ridges, covered with stiff, golden-yellow hairs, like those on the tongue. The opening of the salivary duct is situated on the under or hinder side of the hypopharynx, between it and the base of the labium, the base of the former being cleft; the hollow thus formed is situated over the opening, and forms the salivary receptacle.
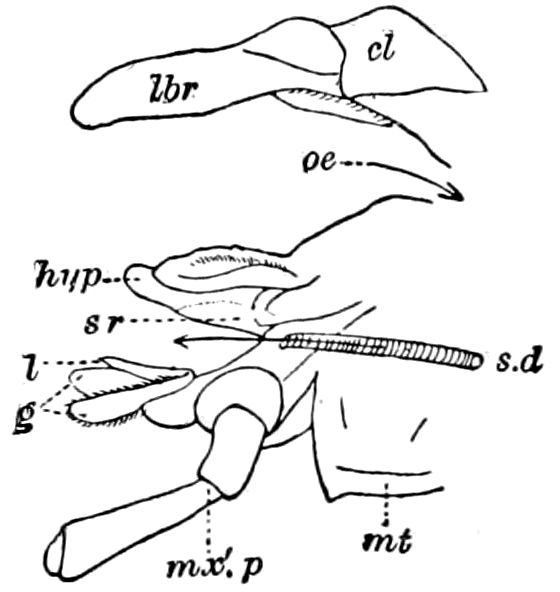
Fig. 71.—Section through the anterior part of the head of Anabrus (the mandibles removed), showing the relations of the hypopharynx (hyp) to the opening of the salivary duct (sd): g, galea; l, lacinia; mt, mentum; oe, œsophagus; lbr, labrum; cl, clypeus.
In the Locustidæ (Anabrus, Fig. 71) the tongue (hypopharynx) is a broad, somewhat flattened lobe arising from the upper part of the base of the mentum and behind the palpifer. This lobe is cavernous underneath, the hollow being the salivary receptacle (sr); the latter is situated over the opening of the salivary duct, which is placed between the base of both the hypopharynx and the labium. The salivary fluid apparently has to pass up and around on each side of the hypopharynx in order to mix with the food.
These relations in the Orthoptera are also the same in the Perlidæ, where the hypopharynx is well developed, forming an unusually large tongue-like mass, nearly filling the buccal cavity.
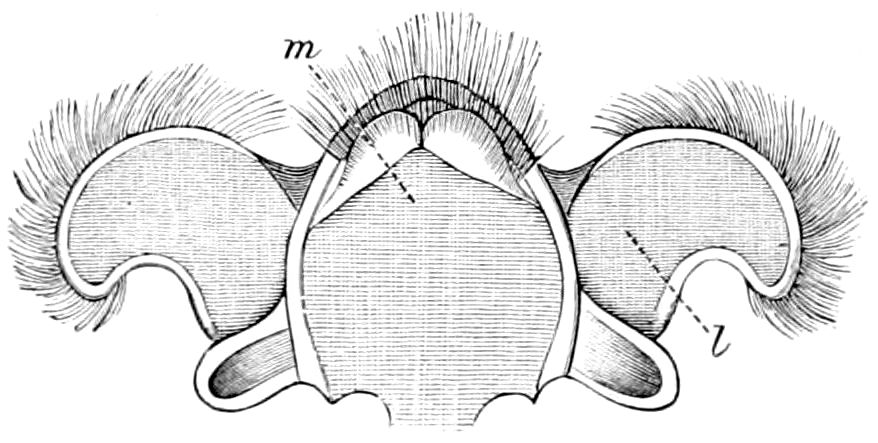
Fig. 72.—Lingua of a May-fly, Heptagenia longicauda, ×16: m, central; l, lateral pieces.—After Vayssière from Sharp.
In the Odonata the lingua is a small, rounded lobe, as also in the Ephemeridæ; in the nymph, however, of Heptagenia (Fig. 72) it is highly developed, according to Vayssière, who seems inclined to regard it as representing a pair of appendages. The tongue in Hemiptera is said by Léon to be present in Benacus griseus (Say) and to correspond to the subgalea of Brullé or hypodactyle of Audouin (Fig. 73), but this appears to correspond to the labium proper, rather than a true lingua, the latter not being differentiated in this order. In the Coleoptera the lingua is rather small. In beetles, as Anopthalmus (Fig. 74), it forms a setose lobe; and a well-developed nerve, the lingual nerve, passes to it, dividing at the end into several branches (n-l). In Sialis the lingua is short, much less developed than usual, being rounded, 74and bears on the edge what appear to be numerous taste-hairs, like those on the ends of the maxillary and labial palpi.
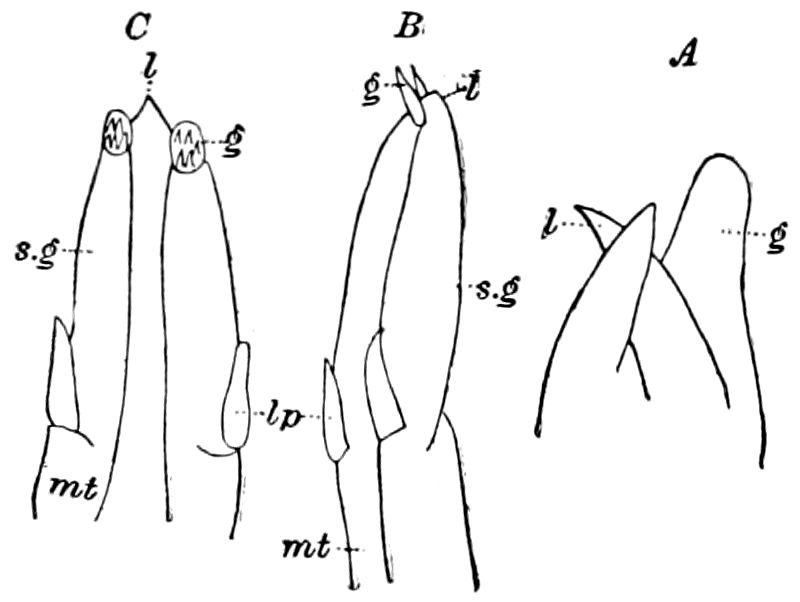
Fig. 73.—A, labium of Zaitha anura. B, of Z. margineguttata. C, of Gerris najas: mt, mentum; lp, labial palpi; sg, subgalea; l, lacinia (= intermaxillare and præmaxillare of Brullé); g, galea.—After Léon.
In the adult Panorpidæ the lingua is a minute, simple lobe.
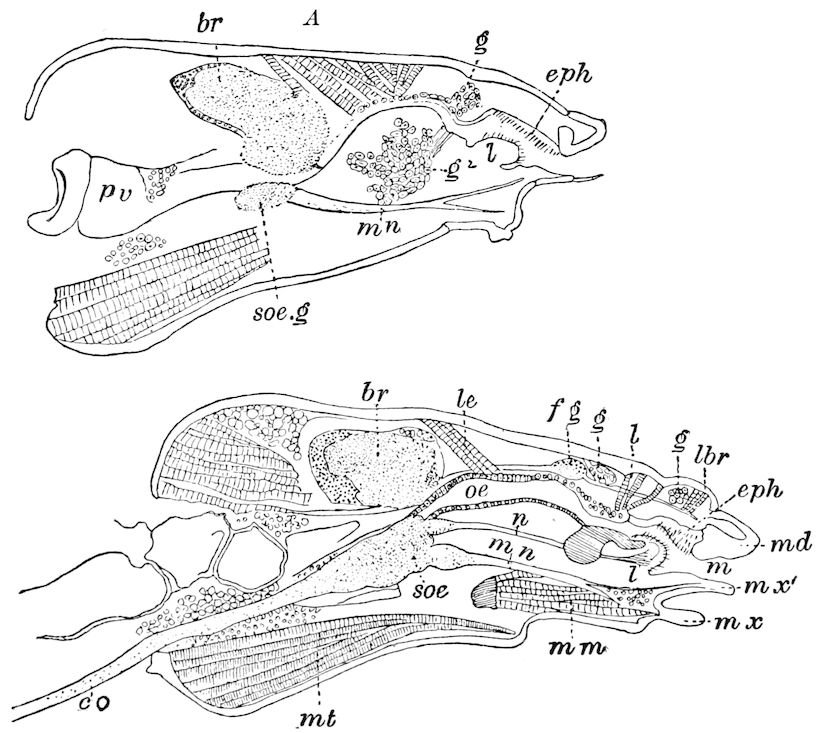
Fig. 74.—Section through head of a carabid, Anopthalmus telkampfii: br, brain; f. g, frontal ganglion; soe, subœsophageal ganglion; co, commissure; n. l, nerve sending branches to the lingua (l); mn, maxillary nerve; mx, 1st maxilla; mm, maxillary muscle; mx′, 2d maxilla; mt, muscle of mentum; le, elevator muscle of the œsophagus; l of the clypeus, and a third beyond raising the labrum (lbr); eph, epipharynx; g, g, salivary glands above; g2, lingual gland below the œsophagus (oe); m, mouth; pv, proventriculus; md, mandible. A, section passing through lingual gland (g2).
In the larval Trichoptera the spinneret is well developed, and in structure substantially like that of caterpillars, and it is plainly the homologue of the hypopharynx, receiving as it does the end of the silk-duct.
In the adult Trichoptera the hypopharynx is a very large, tongue-like, fleshy outgrowth, and is, both in situation and structure, since it contains the opening of the silk-duct, exactly homologous with the hypopharynx of insects of other orders, being somewhat intermediate between the fleshy tongue or lingua of the mandibulate insects, especially the 75Neuroptera, and the hypopharynx of the bees (Fig. 86). Lucas describes and figures it under the name of “haustellum,” but does not homologize it with the hypopharynx. The caddis-flies have been observed to drink water and take in both fluid and fine particles of solid food, and to use the haustellum for this purpose, the end being provided with minute sense-organs like those on the first maxillary lacinia, and possibly of a gustatory nature.
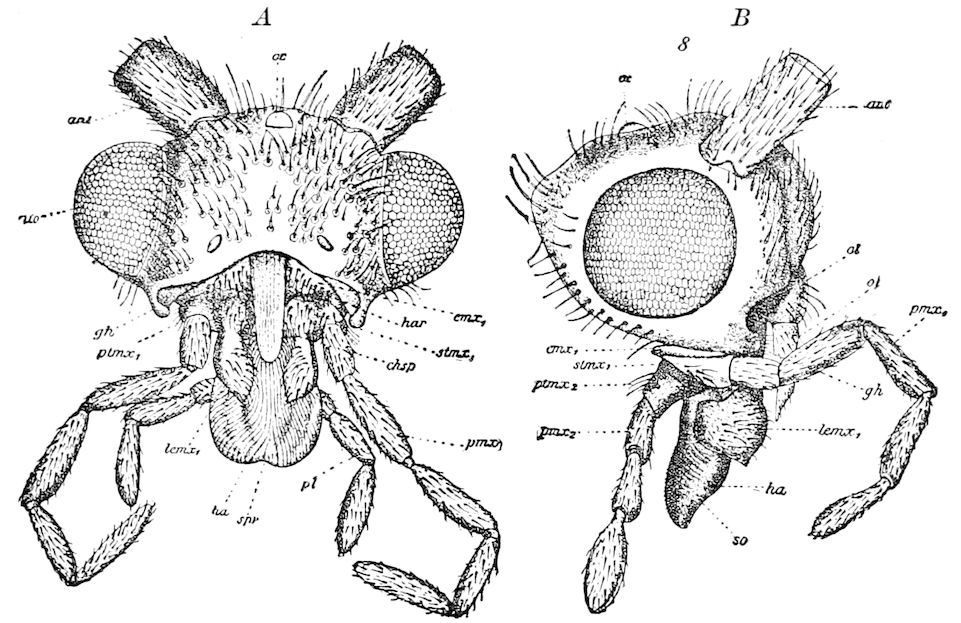
Fig. 75.—Head of Anabolia furcata: A, front view, showing the labrum removed. B, side view; ant, antenna; oc, ocellus; ol, labrum; gh, articulatory process; cmx1, cardo; stmx1, stipes; lemx1, outer lobe (galea); ptmx1, palpus of 1st maxilla; pl, palpus of 2d maxilla; ha, haustellum; so, gustatory pits; spr, opening of salivary duct; chsp, chitinous hook of the clasp; spr, furrow or gutter of the haustellum.—After Lucas.
Fig. 76.—Hypopharynx of Eriocephala calthella: lig, ligula, its membranous hinder edge; lig′, anterior horny edge of the ligula-tube opening outwards; hp, contour of the hypopharynx; mi, mala interior (lacinia); me, mala exterior (galea), of second maxilla; mx′ p, labial palpus.—After Walter.
The spinneret of the larvæ of Lepidoptera is evidently the homologue of the hypopharynx of insects of other orders. It will be seen that the homology of the different parts is identical, the common duct of the silk-glands opening at the end of the hypopharynx, which here forms a complete tube or proboscis extending beyond the end of the labium, in adaptation to its use as a spinning organ.
Walter refers to Burgess’s discovery of a hypopharynx in Danais archippus, 76remarking that this organ in the adult Eriocephalidæ (Fig. 76) exhibits a great similarity to the relations observable in the lower insects, adding:—
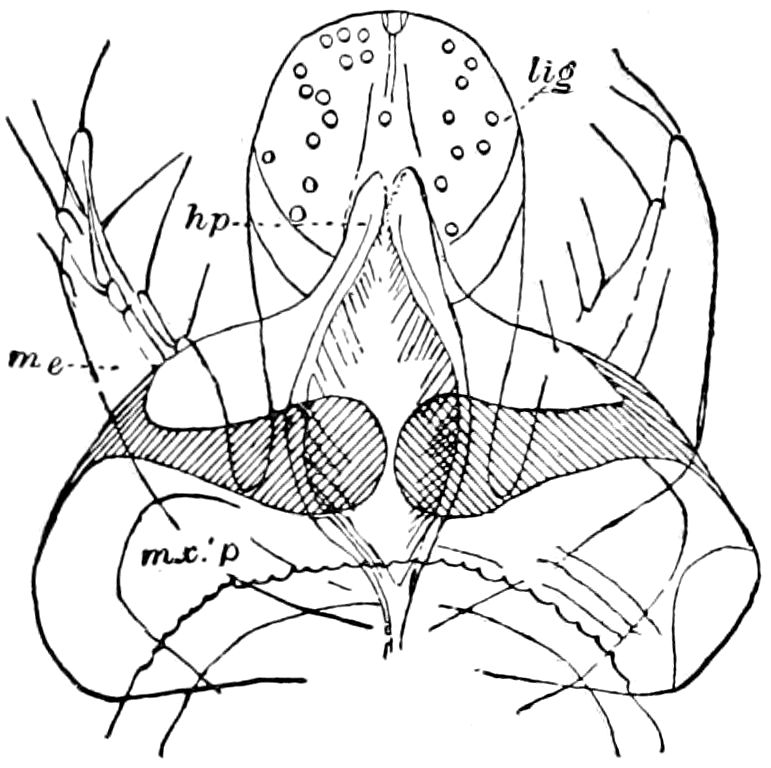
Fig. 77.— Labium of Micropteryx anderschella seen from within (the labial palpi (mx.′ p) removed to their basal joint). Lettering as in Fig. 76.—After Walter.
“The furrow is here within coalesced with the inner side of the labium, and though I see in the entire structure of the head the inner edge of the ligula tube extended under the epipharynx as far as the mandible, I must also accept the fact that here also the hypopharynx extends to the mouth-opening as in all other sucking insects with a well-developed under-lip, viz. the Diptera and Hymenoptera.”
He has also discovered in Micropteryx a paired structure which he regards as the hypopharynx (Fig. 77). As he states:
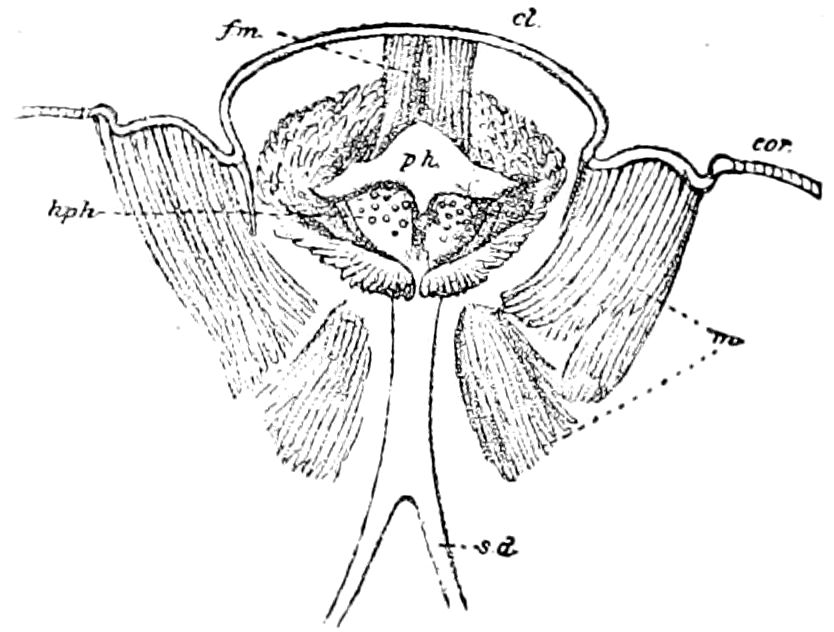
Fig. 78.—Hypopharynx (hph) of Danais: cl, clypeus; sd, salivary duct; m, labial palp muscles; fm, frontal muscle; ph, pharynx; cor, cornea.—After Burgess.
“A portion of the inner surface of the tube-like ligula is covered by a furrow-like band which, close to the inner side, is coalesced with it, and in position, shape, as well as its appendages or teeth on the edge, may be regarded as nothing else than the hypopharynx.”
A hypopharynx is also present in the highest Lepidoptera, Burgess having detected it in Danais archippus. He states that the hypopharynx forms the floor of the pharyngeal cavity; “it is convex on each side of a median furrow (Fig. 78, hph) and somewhat resembles in shape the human breast. The convex areas are dotted over with little papillæ, which possibly may be taste-organs.”
As a piercing organ the hypopharynx reaches its greatest development in the Siphonaptera and Diptera, where the chitinous parts are greatly hypertrophied, the fleshy tongue-like portion so developed in the mandibulate orders being greatly reduced. The chitinous parts are alike on each side of the median organ, being bilaterally symmetrical.
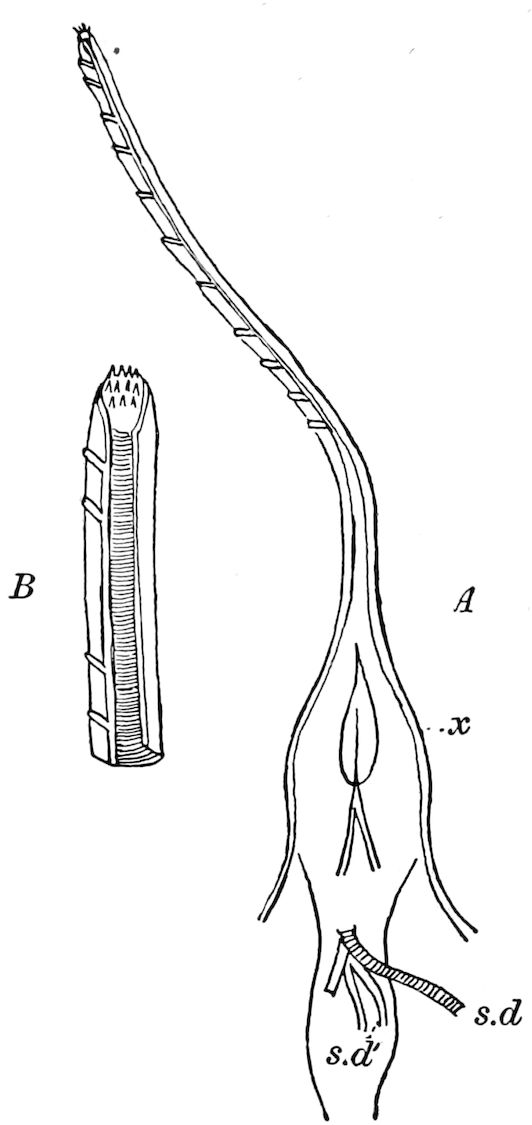
Fig. 79.—A, hypopharynx of Pulex canis: x, basal portion situated within the head; s. d, common duct of the four bladder-shaped salivary glands; s. d′, opening of the tubular salivary glands into the throat. B, end of the hypopharynx, showing the gutter-like structure and teeth at the end.—After Landois.
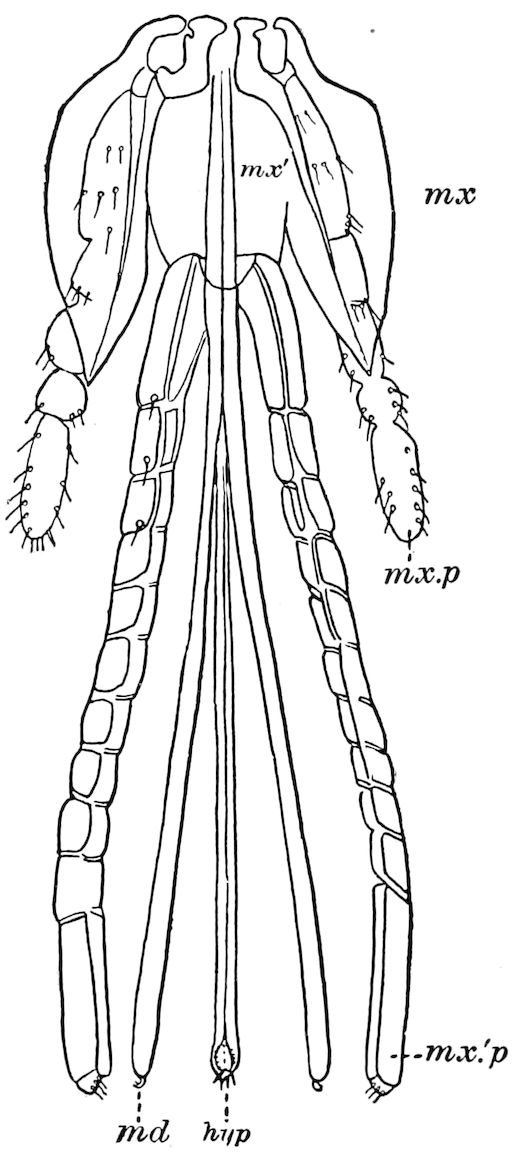
Fig. 80.—Beak of Vermipsylla: hyp, hypopharynx.—After Wagner.
In the fleas the hypopharynx is a large, slender, unpaired, long, chitinous trough, as long as the mandibles, and toothed at the end. Figures 79 and 80 show its relations to the other parts of the mouth; in Fig. 79, x, is seen where the salivary duct opens into the pharynx. Although this organ is not unanimously referred to the hypopharynx, yet from the description of Landois and others, it is evident that this structure does not correspond to the labrum or epipharynx, but belongs to or arises from the floor of the mouth, and, being in close relation to the labium, and also receiving the salivary duct, must be a true hypopharynx.
78In the Diptera the hypopharynx reaches its highest development as a large, stout, awl-like structure.
Meinert, in his detailed and elaborately illustrated work, Trophi Dipterorum (1881), has made an advance on our knowledge of the hypopharynx and its homologies, both by his evidently faithful descriptions and dissections, and by his admirably clear figures.
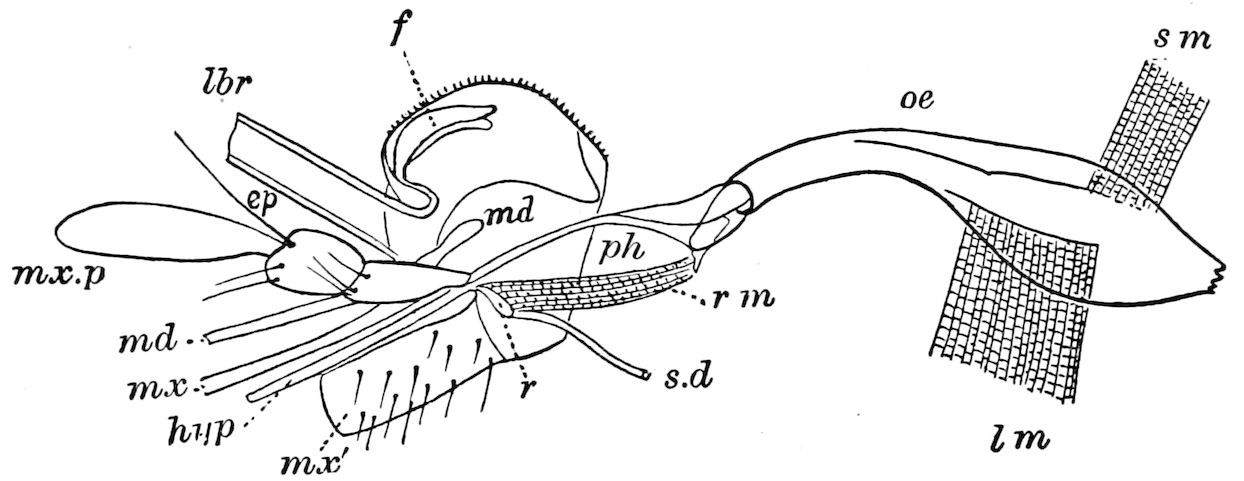
Fig. 81.—Culex pipiens, section of head: oe œsophagus; sm, upper muscle, lm, lower muscle of the œsophagus; ph, pharynx; rm, retractor muscle of the receptacle (r) of the salivary duct (s.d); lbr, labrum; ep, left style of the epipharynx; f, part of front of head.—After Meinert.
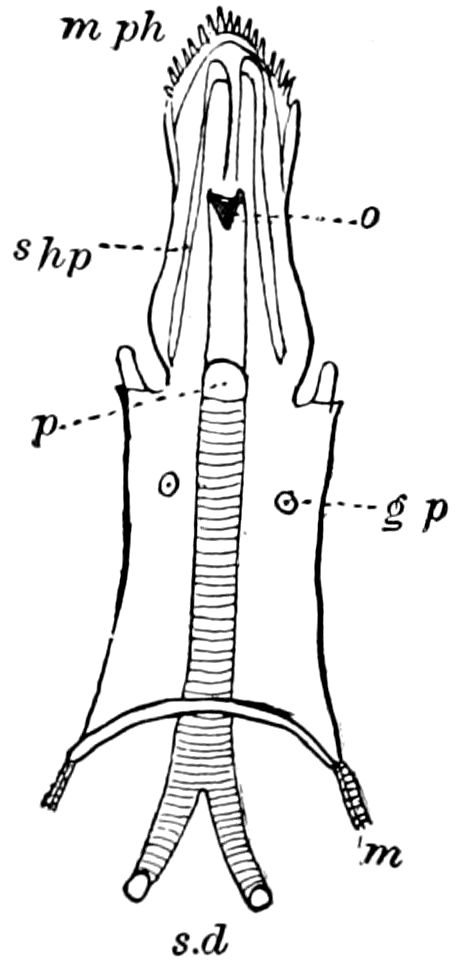
Fig. 82.—Pharynx and hypopharynx of Simulium fuscipes: lph, lower lamina of the pharynx; p, the salivary duct (s.d) perforating the pharynx; o, orifice of the duct; shp, styles of the hypopharynx; mph, membranous edge of the hypopharynx; m, protractor muscle of the pharynx; gp, gustatory papillæ.—After Meinert.
“The hypopharynx, a continuation of the lower edge (lamina) of the pharynx, most generally free, more or less produced, acute anteriorly, forms with the labrum the tube of the pump (antliæ). (The hypopharynx when obsolete, or coalesced with the canal of the proboscis, is the theca; in such a case the siphon or tube is formed by the theca and labrum.) Meanwhile the hypopharynx, the largest of all the trophi (omnium trophorum maximus), constitutes the chief piercing organ (telum) of Diptera. The hypopharynx is moved by protractor, most generally quite or very powerful, and by retractor muscles.
“The efferent duct of the thoracic salivary glands (ductus salivalis) perforates the hypopharynx, more or less near the base, that the saliva may be ejected through the canal into the wound, or that it may be conducted along the labella. Very rarely the salivary duct, perforating the hypopharynx, is continued in the shape of a free, very slender tube.
“The salivary duct behind the base of the hypopharynx forms the receptacle or receptaculum, provided with retractor and levator muscles.”
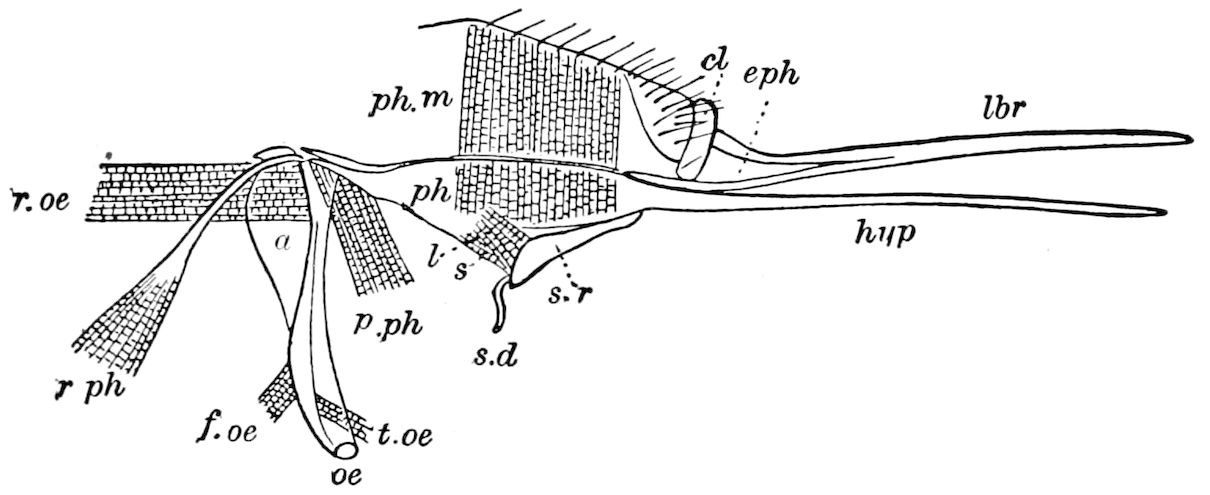
Fig. 83.—Labrum-epipharynx (lbr and eph) and hypopharynx (hyp) of Tabanus brominus: oe, posterior cylindrical portion of the œsophagus; a, anterior swollen portion of the same; ph, pharynx; ph.m, pharyngeal muscle; p.ph, protractor muscle of the pharynx; r.oe, retractor muscle of the œsophagus; r.ph, retractor muscle of the pharynx; f.oe, flexor muscle of the pharynx; t.oe, twisting muscle of the œsophagus; s.r, receptacle of the salivary duct; l, its elevator muscle; s, its retractor muscle; cl, clypeus.—After Meinert.
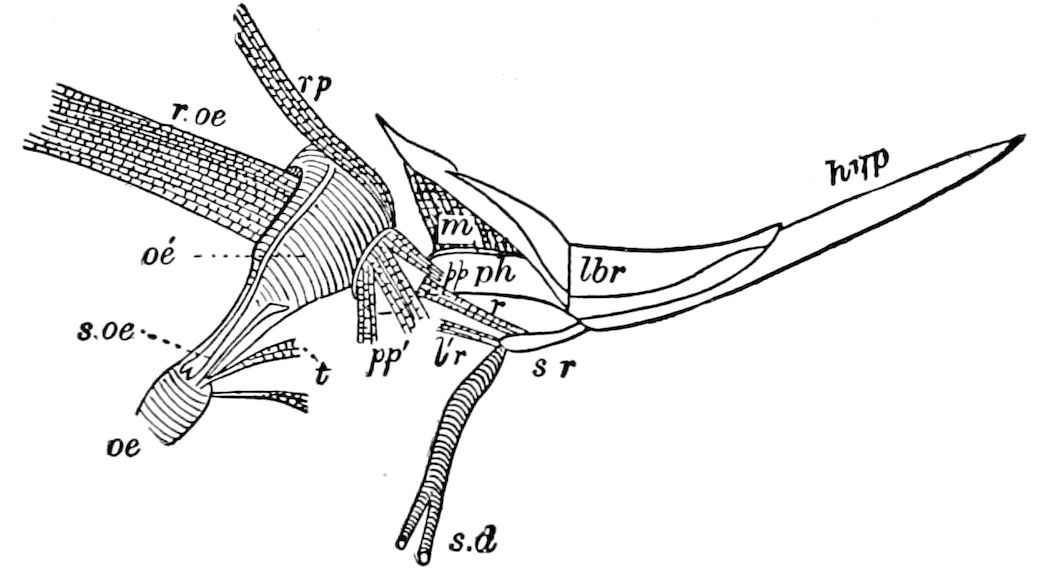
Fig. 84.—Œsophagus (oe), pharynx (ph) with epipharynx and labrum (lbr) of Asilus atricapillus: m, ph, pharyngeal muscle; sr, salivary receptacle; t, twisting; r, l′r, retractor muscles; other lettering as in Fig. 83.—After Meinert.
It has been carefully studied by Meinert in a species of Culex (Fig. 81), Simulium (Fig. 82), Tabanus (Fig. 83), and in Asilus (Fig. 84), where it is seen to attain enormous proportions. In the Hymenoptera, this organ in its most specialized condition is a trough-like rod, adapted for lapping nectar (Fig. 85, 86, hyp). The tongue or hypopharynx of the honey-bee has been elaborately described by Cheshire in his Bees and Bee Keeping.[18] He calls it the tongue or ligula. It is situated in a tube formed by the maxillæ and labial palpi, and can be partially retracted into the mentum. He states that it can move up and down in the tube thus formed, and then describes it as covered by a hairy sheath, its great elasticity being due to a rod running through its centre enabling it to be used as a lapping tongue. The sheath
80“passes round the tongue to the back, where its edges do not meet, but are continuous with a very thin plaited membrane (G, pm) covered with minute hairs. This membrane, after passing towards the sides of the tongue, returns to the angle of the nucleus, or rod, over the under surface of which it is probably continued. The rod passes through the tongue from end to end, gradually tapering towards its extremity, and is best studied in the queen, where I trace many nerve threads and cells. It is undoubtedly endowed with voluntary movement, and must be partly muscular, although I have failed completely in getting any evidence of striation. The rod on the underside has a gutter, or trough-like hollow (cd, the central duct) which is formed into a pseudotube (false tube) by intercrossing of black hairs. It will also be seen that, by the posterior meeting of the sheath, the space between the folded membrane (G, sd) becomes two pseudotubes of larger size, which I shall call the side ducts.
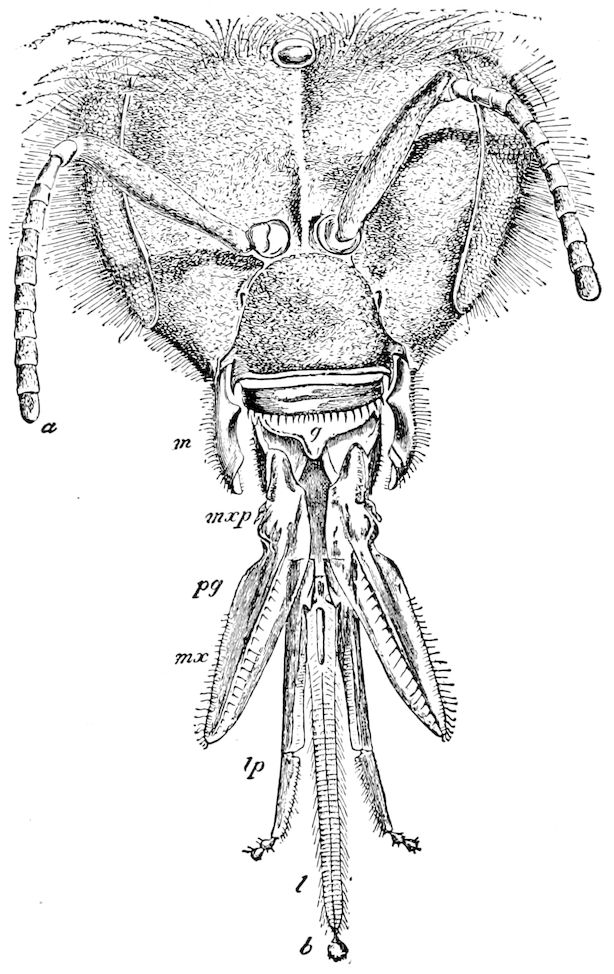
Fig. 85.—Head of honey bee, worker: a, antenna; g, epipharynx; m, mandible; mx, maxilla; mxp, maxillary palpus; pg, paraglossa; lp, labial palpus; l, hypopharynx; b, its spoon.—After Cheshire; from Bull. Div. Ent. U. S. Dept. Agr.
“These central and
side ducts run down to
that part of the tongue
where the spoon, or
bouton (K, Fig. 86)
is placed. This is provided
with very delicate
split hairs (b, Fig. 86)
capable of brushing up
the most minute quantity
of nectar, which by
capillarity is at once
transferred by the gathering
hairs (which are
here numerous, long,
and thin) to two side groove-like forms at the back of the bouton, and which
are really the opened-out extremity of the centre and side ducts, assuming,
immediately above the bouton, the form seen in F, Fig. 86. The central duct,
which is only from 1
600 inch to 1
1000 inch in diameter, because of its smaller
size, and so greater capillary attraction, receives the nectar, if insufficient in
quantity to fill the side ducts. But good honey-yielding plants would bring both
centre and side ducts into requisition. The nectar is sucked up until it reaches
the paraglossæ (pa, B, Fig. 86), which are plate-like in front, but membranous
extensions, like small aprons, behind; and by these the nectar reaches the front
of the tongue, to be swallowed as before described.”
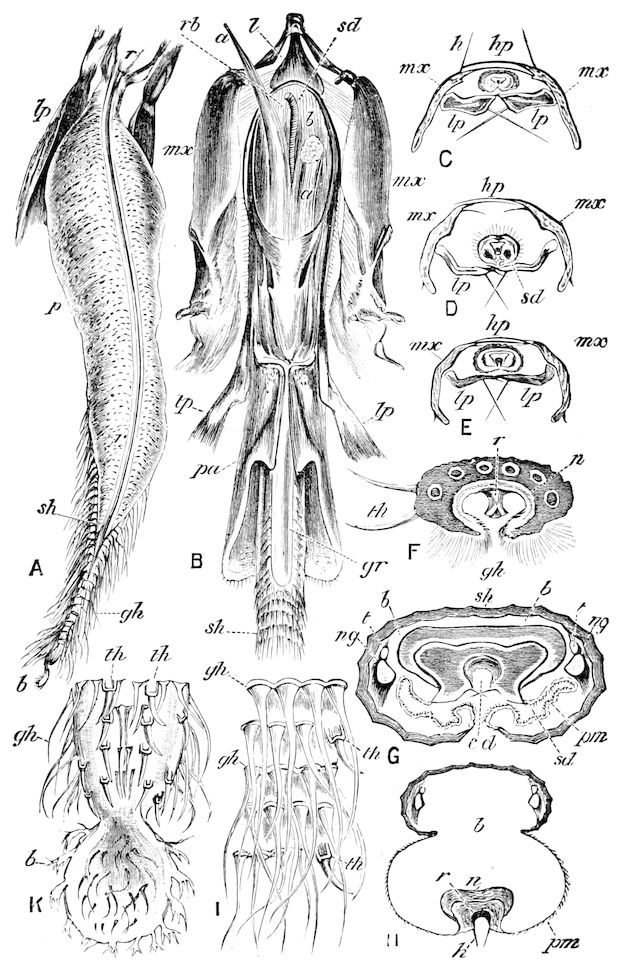
Fig. 86.—Tongue or ligula of the honey bee: A, under side of the tongue; lp, labial palpi; r, r, rod; p, pouch; sh, sheath; gh, gathering hairs; b, bouton or spoon. B, under lip or labium, with appendages, partly dissected; l, lora or submentum; a, a, retractor linguæ longus; sd, salivary duct; rb and b, retractor linguæ biceps; mx, maxillæ; lp, labial palpi; pa, paraglossa; gr, feeding groove; sh, sheath of ligula. C, D, E, sections of ligula; hp, hyaline plate of maxilla; h, hairs acting as stops; mx, maxilla; lp, labial palpi; sd, side duct. F, cross-section of extremity of tongue near the “spoon”; th, tactile hairs; r, rod; n, nucleus; gh, gathering hairs. G, cross-section of tongue without gathering hairs, × 400 times; sh, sheath; b, blood space; t, trachea: ng, gustatory nerve; cd, central duct; sd, lateral duct; pm, plaited membrane. H, same as G, but magnified two hundred times, and with pm, plaited membrane, turned outwards; h, closing hairs; lp, labial palpi; b, blood; n, nucleus; r, rod; h, closing hairs. I, small portion of the sheath; lettering as before. K, extremity of the tongue, with spoon; b, branching hairs for gathering.—After Cheshire.
82Cheshire then settles the question which has been in dispute since the time of Swammerdam, whether the bee’s tongue is solid or tubular. He agrees with Wolff that the duct is a trough and not a tube, and proves it by a satisfactory experiment. He remarks:
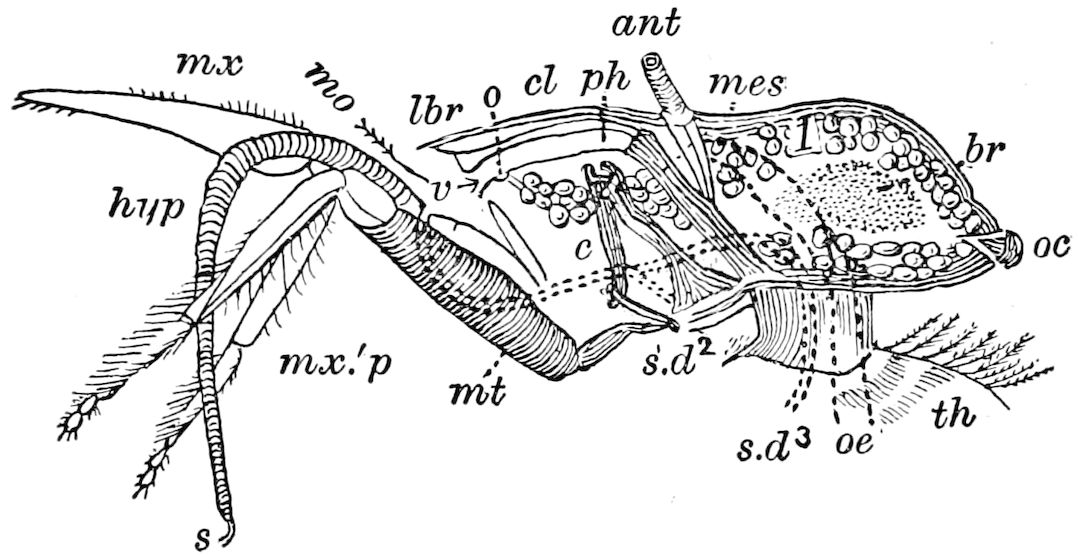
Fig. 87.—Longitudinal section through the head of the honey bee, ♀, just outside of right antenna: ant, antenna with three muscles attached to mes, mesocephalic pillar; cl, clypeus; lbr, labrum; 1, chyle-gland (system no. 1, of Siebold); o, opening of the same; oc, ocellus; br, brain; n, neck; th, thorax; oe, œsophagus; s.d2, s.d3, common salivary ducts of systems 2 and 3; v, salivary valve; c, cardo; ph, pharynx; mx′, labium; mx.′p, labial palpi; mt, mentum; mx, maxilla; hyp, hypopharynx; s, bouton.—After Cheshire.
“Bees have the power, by driving blood into the tongue, of forcing the rod out from the sheath, and distending the wrinkled membrane so that in section it appears as at H, Fig. 86, the membrane assuming the form of a pouch, given in full length at A. It will be seen at once that this disposition of parts abolishes the side ducts, but brings the central duct to the external surface. The object of this curious capability on the part of the bee is, in my opinion, to permit of cleaning away any pollen grains, or other impediment that may collect in the side ducts. The membrane is greasy in nature, and substances or fluids can be removed from it as easily as water from polished metal. If, now, the sides of a needle, previously dipped into clove oil in which rosanilin (magenta) has been dissolved, so as to stain it strongly red, be touched on the centre of the rod, the oil immediately enters, and passes rapidly upwards and downwards, filling the trough.”
Does the hypopharynx represent a distinct segment?—The facts which suggest that the hypopharynx may possibly represent a highly modified pair of appendages, arising from a distinct intermaxillary segment, are these: Heymons plainly shows that, in the embryo of Lepisma, the hypopharynx originates as a transverse segment-like fold in front of the 2d maxillary segment, and larger than it, and though he does not mention it in his text, it appears like the rudiment of a distinct segment; the hypopharynx of Ephemeridæ; arises and remains separate in the nymph from the labium (see Heymons’ Fig. 29, and there are two lateral projections; see also Fig. 72, and Vayssiere’s view that it may represent a pair of appendages; Kolbe also regards it as representing a third pair of maxillæ, his endolabium, p. 213). Though what is called an unpaired 83organ, it is composed of, or supported by, two bilaterally symmetrical styles, both in Myriopods (Fig. 6, labiella, stil) and in insects (Fig. 77, etc.). On the other hand, in the embryo of pterygote insects, an intermaxillary segment has not been yet detected.
LITERATURE OF THE MOUTH-PARTS OR BUCCAL APPENDAGES
a. General
Savigny, Jules-César. Mémoires sur les animaux sans vertèbres. 1re Part. Description et classification des animaux invertébrés et articulés, etc. Fasc. 1re. Mém. 1–2. Théorie des organes de la bouche des crustacés et des insectes. 12 Pl., Paris, 1816, pp. 1–117.
Gerstfeld, Georg. Ueber die Mundteile der saugenden Insekten. Dorpat, 1853.
Olfers, Ernestus V. Annotationes ad anatomiam Podurarum. Berolini, 1862, 4 Pls.
Gerstaecker, Carl Eduard Adolph. Zur Morphologie der Orthoptera amphibiotica. (Festschrift zur Feier des hundertjährigen Bestehens der Gesellschaft naturf. Freunde zu Berlin. 4º, 1873, pp. 39–59, 1 Taf.)
Muhr, Joseph. Die Mundteile der Orthoptera. Ein Beitrag zur vergleichenden Anatomie. (Jahrbuch “Lotos.” Prag, 1877, pp. 40–71, 8 Taf.)
Burgess, Edward. The anatomy of the head and the structure of the maxilla in the Psocidæ. (Proc. Boston Soc. Nat. Hist., xix, 1878, pp. 291–296, 1 Pl.)
Meinert, Fr. Sur la conformation de la tête et sur l’interpretation des organes buccaux chez les insectes, ainsi que sur la systématique de cette ordre. (Ent. Tidsskr., 1. Arg., 1880, pp. 147–150.)
—— Tungens udskydelighed hos Steninerne, en slaegt af Staphylinernes familie. (Vidensk. meddel. fra den naturh. Foren, 1884–1886, pp. 180–207, 2 Pls. Also Zool. Anzeiger, 1887, pp. 136–139.)
Müller, A. Vergleichend-anatomische Darstellung der Mundteile der Insekten. Villach, 1881, 3 Taf.
Kraepelin, Karl. Ueber die Mundwerkzeuge der saugenden Insekten. (Zool. Anzeiger, 1882, pp. 574–579.)
Dewitz, H. Ueber die führung an den Körperanhangen der Insekten. (Berlin. Zeitschr. xxvi., 1882, pp. 51–68, Figs.)
Wolter, Max. Die Mundbildung der Orthopteren mit specieller Berücksichtigung der Ephemeriden. 4 Taf. Greifswald, 1883.
Oudemans, J. T. Beiträge zur Kenntniss der Thysanura und Collembola. (Bijdragen tot de Dierkunde, pp. 149–226. Amsterdam, 1888, 3 Taf.)
Smith, John B. An essay on the development of the mouth-parts of certain insects. (Trans. Amer. Philosophical Soc., xix, pp. 175–198, 3 Pls.)
Also articles by Chatin, McLachlan, Riley, Wood-Mason.
b. Thysanoptera (Physapoda)
Jordan, Karl. Anatomie und biologie der Physapoda. (Zeitschr. f. wissens. Zool., xlvii, pp. 541–620, 3 Taf. 1888.)
Garman, H. The mouth-parts of the Thysanoptera. (Bull. Essex Inst., xxii, 4 pp., Fig. 1890.)
—— The asymmetry of the mouth-parts of Thysanoptera. (Amer. Naturalist, July, 1896, pp. 591–593, Fig.)
Bohls, J. Die Mundwerkzeuge der Physapoden. Dissertation Göttingen, 1891, pp. 1–36.
84Uzel, Heinrich. Monographie der Ordnung Thysanoptera. Königgrätz, 1895, pp. 472, 10 Taf., 9 Figs.
c. Hemiptera
Léon, N. Beiträge zur Kenntniss der Mundteile der Hemipteren. Jena, 1887, pp. 47, 1 Taf.
—— Labialtaster bei Hemipteren. (Zool. Anzeiger, pp. 145–147, 1892, 1 Fig.)
—— Beiträge zur Kenntniss des Labiums der Hydrocoren. (Zool. Anzeiger, März 29, 1897, pp. 73–77, Figs. 1–5.)
Geise, O. Mundteile der Rhynchoten. (Archiv f. Naturgesch., xlix, 1883, pp. 315–373, 1 Taf.)
Wedde, Hermann. Beiträge zur Kenntniss des Rhynchotenrüssels. (Archiv f. Naturgesch., li Jahrg., 1 Bd., 1885, pp. 113–148, 2 Taf.)
Smith, John B. The structure of the hemipterous mouth. (Science, April 1, 1892, pp. 189–190, Figs. 1–5.)
d. Coleoptera
Smith, John B. The mouth-parts of Copris carolina; with notes on the homologies of the mandibles. (Trans. Amer. Ent. Soc., xix, April, 1892, pp. 83–87, 2 Pls.)
e. Lepidoptera
Kirbach, P. Ueber die Mundwerkzeuge der Schmetterlinge. (Zool. Anzeiger, vi Jahrg., 1883, pp. 553–558, 2 Figs.)
—— Ueber die Mundwerkzeuge der Schmetterlinge. (Archiv f. Naturgeschichte, 1884, pp. 78–119, 2 Taf.)
Walter, Alfred. Palpus maxillaris Lepidopterorum. (Jenaische Zeitschr. f. Naturwiss, xviii, 1884, pp. 121–173, Taf.)
—— Beiträge zur Morphologie der Lepidoptera. I, Mundteile. (Jenaische Zeitschr. f. Naturwiss, xviii, 1885, pp. 751–807, 2 Taf.)
Breitenbach, W. Vorläufige Mitteilung über einige neue Untersuchungen an Schmetterlingsrüsseln. (Archiv f. mikroskop. Anatomie, xiv, 1877, pp. 308–317, 1 Taf.)
—— Untersuchungen an Schmetterlingsrüsseln. (Ibid., xv, 1878, pp. 8–29, 1 Taf.)
—— Ueber Schmetterlingsrüssel. (Entomolog. Nachr. 5 Jahrg., 1879, pp. 237–243, 1 Taf.)
—— Der Schmetterlingsrüssel. (Jenaische Zeitschr. f. Naturwiss, 1881.)
f. Siphonaptera
Kräpelin, K. Ueber die systematische Stellung der Puliciden. (Festschrift z. 50 jahr. Jubil. d. Realgymnas. Iohanneum, Hamburg, pp. 17, 1 Taf. 1884.)
Kellogg, V. L. The mouth-parts of the Lepidoptera. (Amer. Nat., xxix, 1895, pp. 546–556, 1 Pl. and Fig.)
g. Diptera
Menzbier, Michael Alexander. Ueber das Kopfskelett und die Mundwerkzeuge der Zweiflügler. (Bull. Soc. Imp. Natur. de Moscou, lv, 1880, pp. 8–71, 2 Taf.)
Dimmock, George. The anatomy of the mouth-parts and of the sucking apparatus of some Diptera. Boston, 1881, pp. 48, 4 Pls.
85Meinert, F. Fluernes Munddele. Trophi Dipterorum. Kjöbenhavn, 1881, 6 Pls.
—— Die Mundteile der Dipteren. (Zool. Anz. 1882, pp. 570–574, 599–603.)
Becher, E. Zur Kenntniss der Mundteile der Dipteren. (Denkschr. Akad. d. Wissensch. Wien., xlv, 1882, pp. 123–162, 4 Taf.)
Hansen, H. J. Fabrica oris dipterorum: Dipterernes mund: anatomisk og systematisk henseende. 1 Tabanidae, Bombyliidae, Asilidae, Thereva, Mydas, Apiocera. (Naturhist. Tidsskrift, 1883, xiv, pp. 1–186, Taf. 1–5.)
Kräpelin, Karl. Zur Anatomie und Physiologie des Rüssels von Musca. (Zeitschr. f. wissensch. Zool., xxxix, 1883, pp. 683–719, 2 Taf.)
McCloskie, George. Kraepelin’s Proboscis of the house-fly. (American Naturalist, xviii, 1884, pp. 1234–1244, Figs.)
Langhoffer, August. Beiträge zur Kenntniss der Mundtheile der Dipteren. Jena, 1888, pp. 1–32.
Smith, John B. A contribution toward a knowledge of the mouth-parts of the Diptera. (Trans. Amer. Ent. Soc., xvii, Nov. 1890, pp. 319–339, Figs. 1–22.)
h. Hymenoptera
Briant, Travers J. On the anatomy and functions of the tongue of the honey-bee (worker). (Journ. Linn. Soc., London, xvii, 1884, pp. 408–416, 2 Pls.)
Breithaupt, P. F. Ueber die Anatomie und die Funktionen der Bienenzunge. (Archiv f. Naturgesch., Jahrg. lii, 1886, pp. 47–112, 2 Taf.)
i. Larval stages
Brauer, F. Die Zweiflügler des kaiserlichen Museums zu Wien. III, Systematische Studien auf Grundlage der Dipterenlarven nebst einer Zusammenstellung von Beispielen aus der Litteratur uber dieselben und Beschreibung neuer Formen. (Denkschr. math.-naturwiss. Cl. k. Akad. Wiss. Wien, 1883, xlvii, pp. 100, 5 Taf.)
Dewitz, H. Ueber die Führung an den Körperanhangen der Insekten speziell betrachtet an der Legescheide der Acridier, dem Stachel der Meliponem und den Mundteilen der Larve von Myrmeleon, nebst Beschreibung dieser Organe. (Berliner ent. Zeitschr., xxvi, 1882, pp. 51–68.)
—— Die Mundteile der Larve von Myrmeleon. (Sitzungsber. d. Ges. naturforsch. Freunde zu Berlin, 1881, pp. 163–166.)
Redtenbacher, Josef. Uebersicht der Myrmeleonidenlarven. (Denkschrift, math.-naturwiss. Cl. k. Akad. Wiss. Wien, 1884, xlviii, pp. 335–368, 7 Taf.)
Schiödte, J. G. De metamorphosi Eleutheratorum. Bidrag til insekternes udviklingshistorie. (Kroyer’s Naturhist. Tidsskrift. Kjöbenhavn. 12 Teile mit 88 Taf., 1862–1883.)
j. Embryonic stages
Heymons, Richard. Grundzüge der Entwicklung und des Körpersbaues von Odonaten und Ephemeridem (Anhang zu den Abhandl. K. Akad. d. Wissens. Berlin, 1896, p. 22, 2 Taf. See Figs. 5, 29.)
—— Entwicklungsgeschichtliche Untersuchungen an Lepisma saccharina L. (Zeitschr. f. Wissens. Zoologie, lxii, 1897, p. 595, 2 Taf. See Fig. 10.)
THE THORAX AND ITS APPENDAGES
a. The thorax; its external anatomy
The middle region of the body is called the thorax, and in general consists of three segments, which are respectively named the prothorax, mesothorax, and metathorax (Figs. 88, 89, 98).
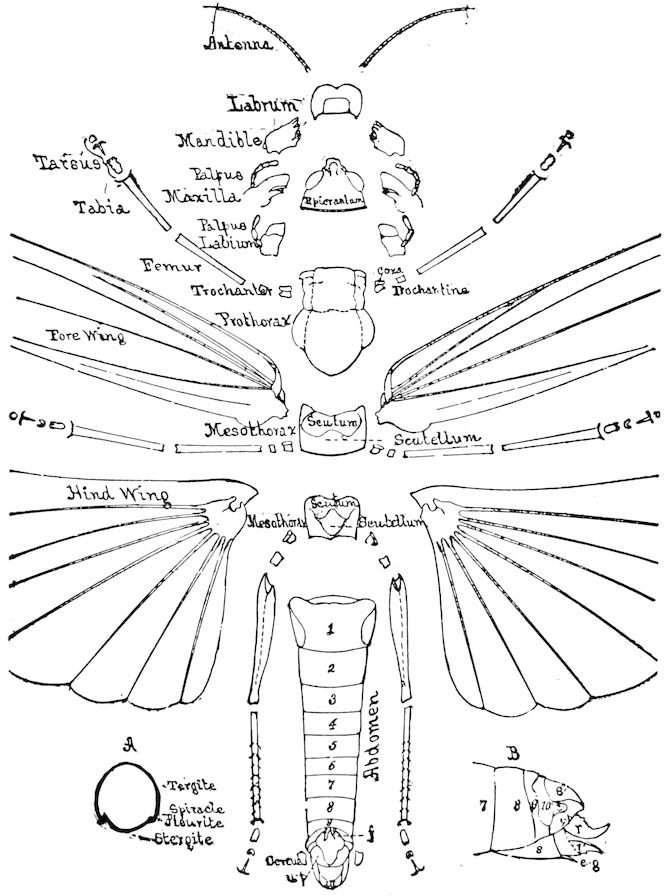
Fig. 88.—External anatomy of Melanoplus spretus, the head and thorax disjointed.
The thorax contains the muscles of flight and those of the legs, besides the fore intestine (œsophagus and proventriculus), as well as, in the winged insects, the salivary glands.
87In the more generalized orders, notably the Orthoptera, the three segments are distinct and readily identified.
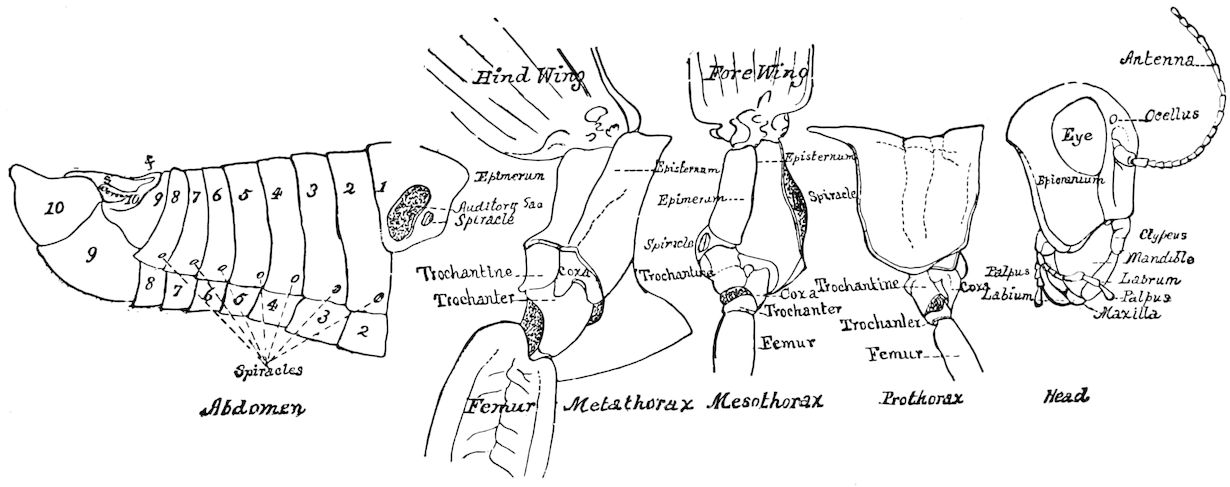
Fig. 89.—Locust, Melanoplus, side view, with the thorax separated from the head and abdomen, and divided into its three segments.
Each segment consists of the tergum, pleurum, and sternum. In the prothorax these pieces are not subdivided, except the pleural; in such case the tergum is called the pronotum. The prothorax is very large in the Orthoptera and other generalized forms, as also in the Coleoptera, but small and reduced in the Diptera and Hymenoptera. In the winged forms the tergum of the mesothorax is differentiated into four pieces or plates (sclerites). These pieces were named by Audouin, passing from before backwards, the præscutum, scutum, scutellum, and postscutellum. In the nymph stage and in the wingless adults of insects such as the Mallophaga, the true lice, the wingless Diptera, ants, etc., these parts by disuse and loss of the wings are not differentiated. It is therefore apparent that their development depends on that of the muscles of flight, of which they 88form the base of attachment. The scutum is invariably present, as is the scutellum. The former in nearly all insects constitutes the larger part of the tergum, while the latter is, as its name implies, the small shield-shaped piece directly behind the scutum.
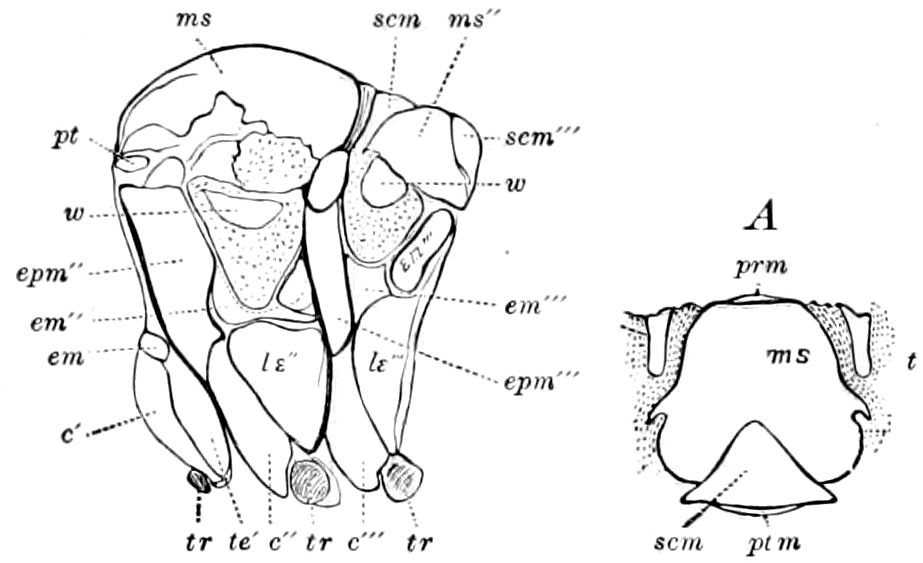
Fig. 90.—Thorax of Telea polyphemus, side view, pronotum not represented: em, epimerum of prothorax, the narrow piece above being the prothoracic episternum; ms, mesoscutum; scm, mesoscutellum; ms″, metascutum; scm‴, metascutellum; pt, a supplementary piece near the insertion of tegulæ; w, pieces situated at the insertion of the wings, and surrounded by membrane; epm″, episternum of the mesothorax; em″, epimerum of the same; epm‴, episternum of the metathorax; em‴ epimerum of the same, divided into two pieces; c′, c″, c‴, coxæ; te′, te″, te‴, trochantines; tr, tr, tr, trochanters. A, tergal view of the mesothorax of the same; prm, præscutum; ms, scutum; scm, scutellum; ptm, postscutellum; t, tegula.
The præscutum and postscutellum are usually minute and crowded down out of sight between the opposing segments. As seen in Fig. 90, the præscutum of most moths (Telea) is a small rounded piece, bent vertically down so as not to be seen from above. In Polystœchotes and also in Hepialus the præscutum is large, well-developed, triangular, and wedged in between the two halves of the scutum. The postscutellum is still smaller, usually forming a transverse ridge, and is rarely used in taxonomy.
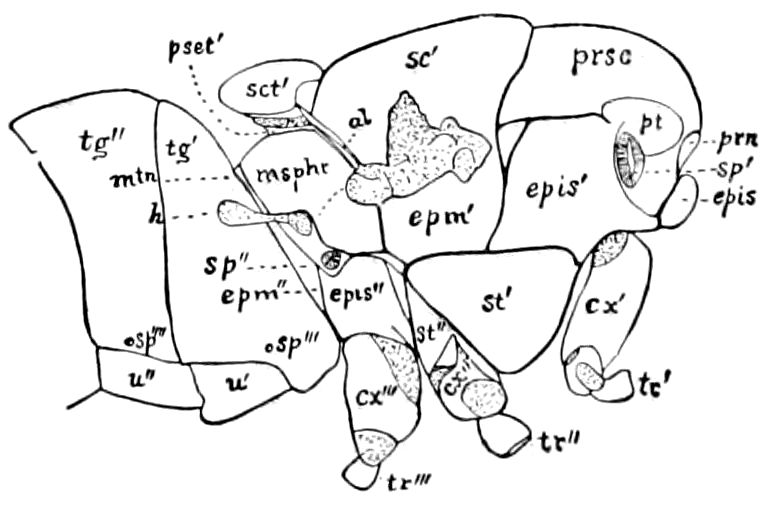
Fig. 91.—Thorax of the house-fly: prn, pronotum; prsc, præscutum; sc′, mesoscutum; sct′, mesoscutellum; psct′, postscutellum; al, insertion of squama, extending to the insertion of the wings, which have been removed; msphr, mesophragma; h, balancer (halter); pt, tegula; mtn, metanotum; epis, epis′, epis″, episternum of pro-, meso-, and metathorax; epm′, epm″, meso- and meta-epimerum; st′, st″, meso- and metasternum; cx′, cx″, cx‴, coxæ; tr′, tr″, tr‴, trochanters of the three pairs of legs; sp′, sp″, sp‴, sp‴′, sp‴″, first to fifth spiracles; tg′, tg″, tergites of first and second abdominal segments; u′, u″, urites.
The metathorax is usually smaller and shorter than the mesothorax, being proportioned to the size of the wings. In certain Neuroptera and in Hepialidæ and some tineoid moths, where the hind wings are nearly as large as those of the anterior pair, the metathorax is more than half or nearly two-thirds as large as the mesothorax. In Hepialidæ the præscutum is large and distinct, while the scutum is divided into two widely separated pieces. The postscutellum is nearly or quite obsolete.
The pleurum in each of the three thoracic segments is divided into two pieces; the one in front is called the episternum, since it 89rests upon the sternum; the other is the epimerum. To these pieces, with the sternum in part, the legs are articulated (Fig. 89).
Between the episterna is situated the breastplate or sternum, which is very large in the more primitive forms, as the Orthoptera, and is small in the Diptera and Hymenoptera.
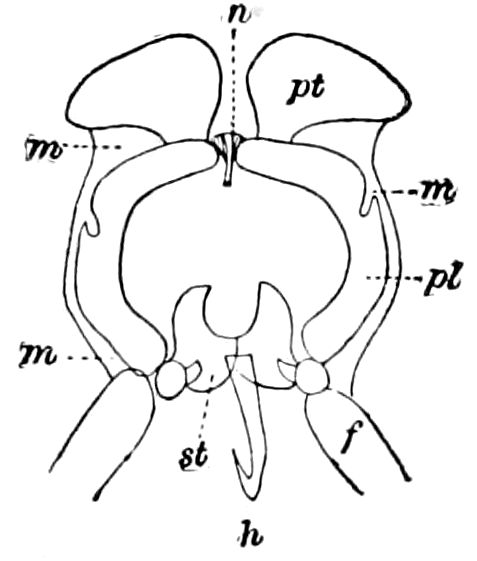
Fig. 92.—Prothorax of Geometra papilionaria: n, notum; p, pleura; st, sternum; pt, patagia; m, membrane; f, femur; h, a hook bent backwards and beneath, and connecting the pro- with the mesothorax.—After Cholodkowsky.
The episterna and epimera are in certain groups, Neuroptera, etc., further subdivided each into two pieces (Fig. 102). The smaller pieces, hinging upon each other and forming the attachments of the muscles of flight, differ much in shape and size in insects of different orders. The difference in shape and degree of differentiation of these parts of the thorax is mentioned and illustrated under each order, and reference to the figures will obviate pages of tedious description. A glance, however, at the thorax of a moth, fly, or bee, where these numerous pieces are agglutinated into a globular mass, will show that the spherical shape of the thorax in these insects is due to the enlargement of one part at the expense of another; the prothoracic and metathoracic segments being more or less atrophied, while the mesothorax is greatly enlarged to support the powerful muscles of flight, the fore wings being much larger than those appended to the metathorax. In the Diptera, whose hinder pair of wings are reduced to the condition of halteres, the reduction of the metathorax as well as prothorax is especially marked (Fig. 91).
The patagia.—On each side of the pronotum of Lepidoptera are two transversely oval, movable, concavo-convex, erectile plates, called patagia (Fig. 92). On cutting those of a dry Catocala in two, they will be seen to be hollow. Cholodkowsky[19] states that they are filled with blood and tracheal branches; and he went so far as to regard them as rudimentary prothoracic wings, in which view he was corrected by Haase,[20] who compares them with the tegulæ, regarding them also as secondary or accessory structures.
The tegulæ.—On the mesothorax are the tegulæ of Kirby (pterygodes of Latreille, paraptera of McLeay, hypoptère or squamule), which cover the base of the fore wings, and are especially developed in the Lepidoptera (Fig. 90, A, t) and in certain Hymenoptera (Fig. 95, c).
90The external opening of the spiracles just under the fore wings, is situated in a little plate called by Audouin the peritreme.
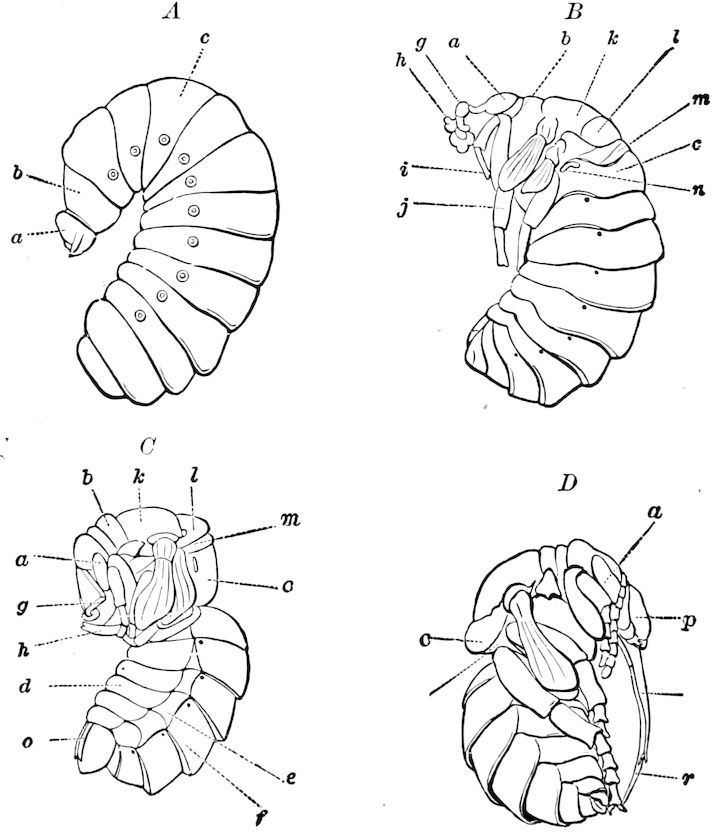
Fig. 93.—Transformation of the bumble bee, Bombus, showing the transfer of the 1st abdominal larval segment (c) to the thorax, forming the propodeum of the pupa (D) and imago; n, spiracle of the propodeum. A, larva; a, head; b, 1st thoracic; c, 1st abdominal segment. B, semipupa; g, antenna; h, maxillæ; i, 1st; j, 2d leg; k, mesoscutum; l, mesoscutellum; m, metathorax; d, urite (sternite of abdomen); e, pleurite; f, tergite; o, ovipositor; r, lingua; q, maxilla.
In the higher or aculeate Hymenoptera, besides the three segments normally composing the thorax, the basal abdominal segment is during the change from the larva to the pupa transferred to this region, making four segments. This first abdominal is called “the median segment” (Figs. 93–95). In such a case the term alitrunk has been applied to this region, i.e. the thorax, as thus constituted. Latreille wrongly stated that in the Diptera the first abdominal segment also entered into the composition of the thorax; but Brauer has fully disproved that view, as may be seen by an examination of his sketches which we have copied (Fig. 94).
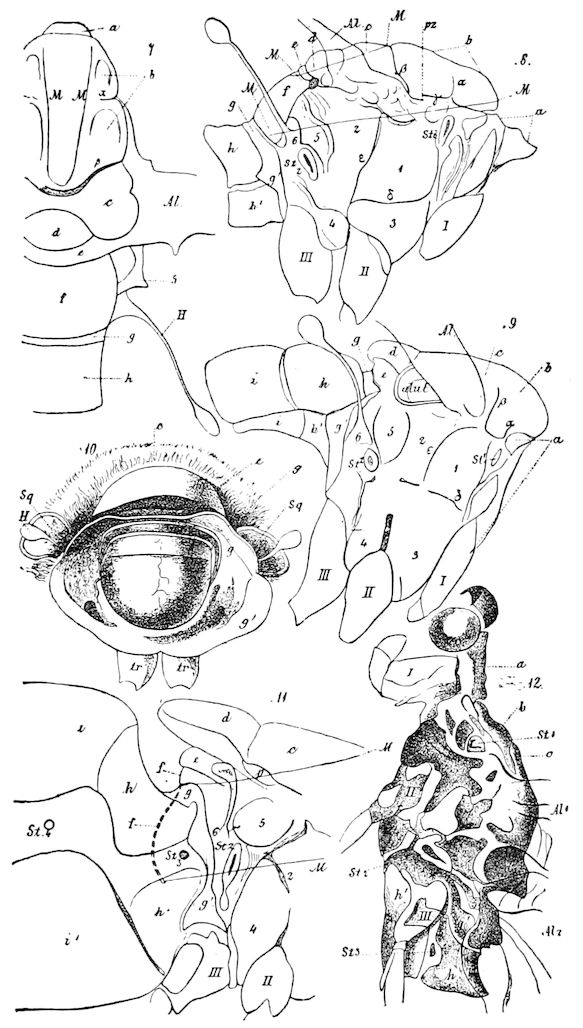
Fig. 94.—7, 8, thorax of Tipula gigantea; 9, of Leptis; 10, thorax of Tabanus bromius after the removal of the abdomen, in order to bring into view the inner mesophragma (f), and to show the extension of the metathorax g and g′; tr, trochanter; 11, hind end of the mesothorax, the entire metathorax, and the 1st and 2d abdominal segments of Volucella zonaria, seen from the side. The internal mesophragma (f), and the position of the muscle inserted in it, are indicated by the two lines M. p, Callus postalaris; pr (pz in 8), callus præalaris Osten Sacken (= “patagium” of some authors); g, metanotum; g′, metepimerum, “segment médiaire” of Latreille (wrongly considered by him to be the 1st abdominal segment); 4, metasternum (hypopleura of Osten Sacken); 5 (? “episternum of metathorax” (Brauer) = metapleura of Osten Sacken); 6, and also H, halter; st1, mesothoracic stigma; st2, metathoracic stigma; st3, first abdominal stigma; γ, dorsopleural; δ, sternopleural; ε, mesopleural sutures; h, 1st, i, 2d, abdominal segment; al, wing; alul, alula. 12, the head and the three thoracic rings, and the 1st abdominal segment of Ephemera vulgata, the connecting membranes are in white: a, prothorax; b, præscutum; c, scutum; d, scutellum; e, postscutellum; ps, postscutellum of mesothorax.—After Brauer.
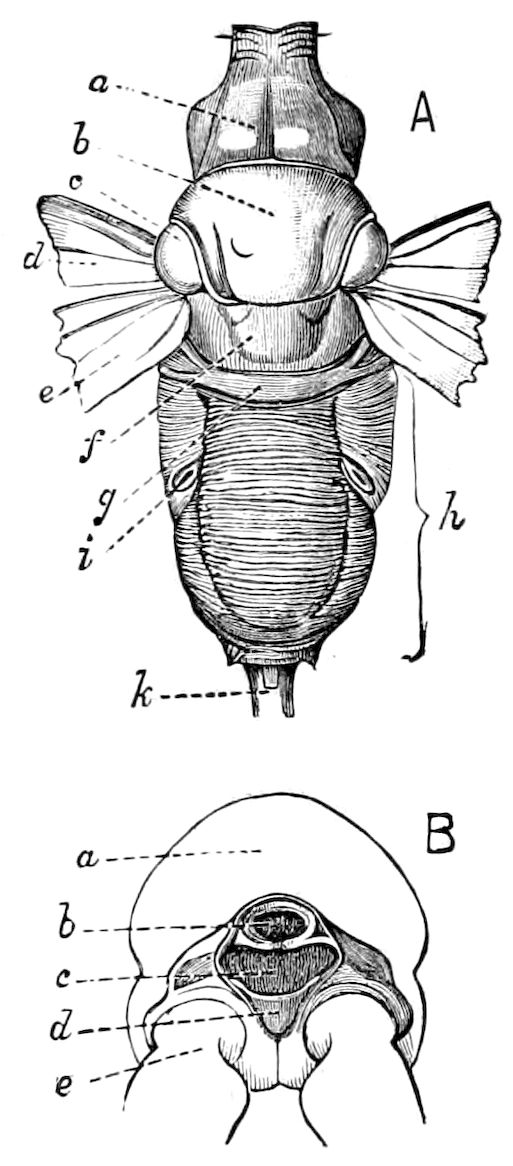
Fig. 95.—Alitrunk of Sphex chrysis: A, dorsal aspect; a, pronotum; b, mesonotum; c, tegula; d, base of fore,—e, of hind, wing; f, g, divisions of metanotum; h, median (true first abdominal) segment; i, its spiracle; k, second abdominal segment, usually called the petiole or first abdominal segment. B, posterior aspect of the median segment; a, upper part; b, superior,—c, inferior, abdominal foramen; d, ventral plate of median segment; e, coxa.—After Sharp.
The sternum is in rare cases subdivided into two halves, as in the meso- and metathorax of the cockroach; in Forficula the prosternum is divided into four pieces besides the sternum proper (Fig. 96); and in Embia, also, the sternites, according to Sharp, are complex.
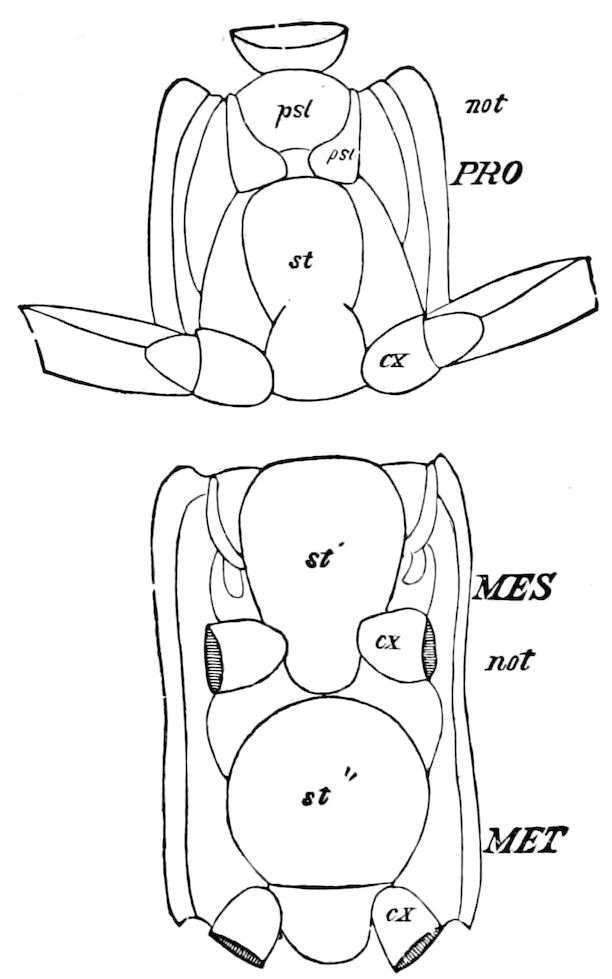
Fig. 96.—Sternal view of pro-, meso-, and metathorax of Forficula tæniata: pst, præsternum, divided into 4 pieces; st, pro-, st′, meso-, st″, metasternum; cx, coxa; not, notum.
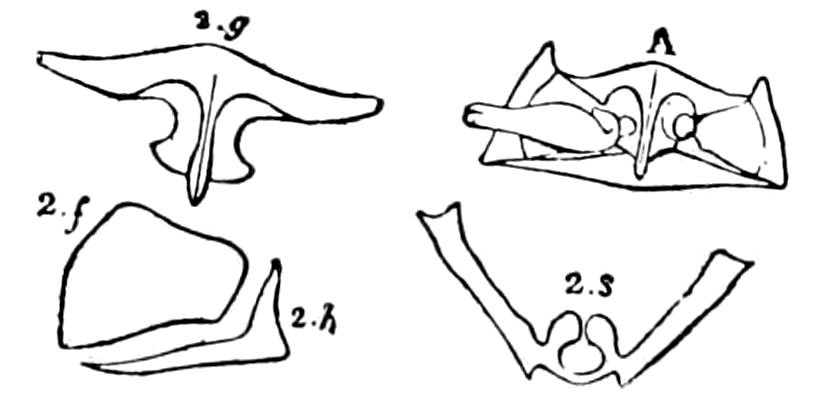
Fig. 97.—A, under surface of prothorax, or prosternum, of Dyticus circumflexis: 2.g, prosternum; 2.f, episternum; 2.h, epimerum; 2.s, antefurca or entothorax.
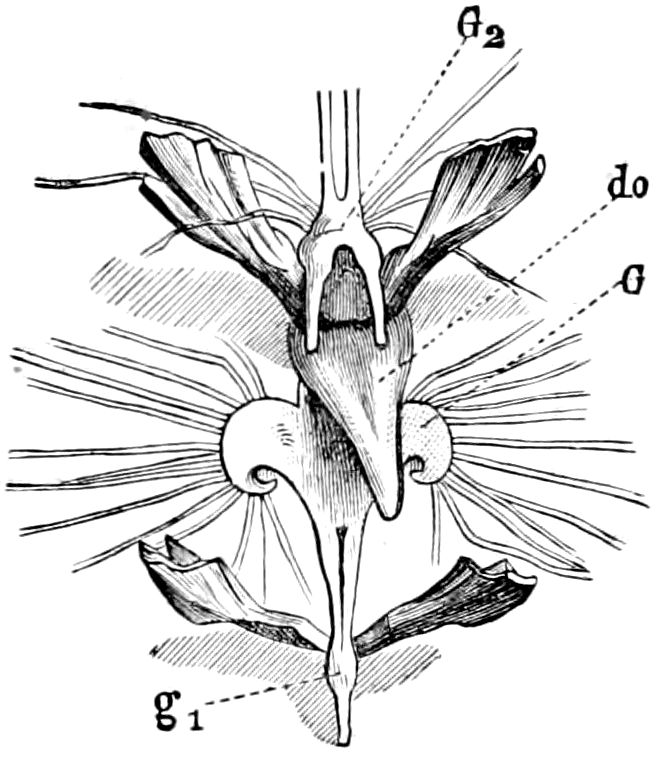
Fig. 98.—Meso- (G2) and metathoracic ganglia (G1), with the apodemes of Gryllotalpa.—After Graber.
Fig. 99.—Parts of the mesothorax of Dyticus: A, mesosternum; 3.a, præscutum; 3.b, scutum; 3.c, scutellum; 3.d, postscutellum; 3.e, parapteron; 3.g, mesosternum; 3.f, episternum; 3.h, epimerum; 3.s, medifurca or entothorax.
Fig. 100.—Parts of the metathorax of Dyticus: A, metasternum; 4.a, præscutum; 4.b, scutum; 4.c, scutellum; 4.d, postscutellum; 4.e, parapteron; 4.f, episternum; 4.g, metasternum; 4.h, epimerum; 4.s, postfurca.—This and Figs. 97 and 99 from Audouin, after Newport.
The apodemes.—The thorax is supported within by beam-like processes, or apodemes, which pass inward and also form attachments for the muscles. Those passing up from the sternum form the entothorax of Audouin, and the process of each thoracic segment is called respectively the antefurca, medifurca, and postfurca. In the Orthoptera (Caloptenus and Anabrus), the antefurca is large, thin, flattened, directed forward, and bounds each side of the prothoracic ganglion. In the Coleoptera two plates (Fig. 97, 2.s) arise from the inside of the sternum and “form a collar or leave a circular hole between them for the passage of the nervous cord” (Newport). The medifurca is a pair of flat processes which diverge and bridge the commissure, while the postfurca is situated under the commissure. In beetles (Dyticus) Newport states that it is expanded into two broad plates, to which the muscles of the posterior legs are attached. Graber also notices in the mole cricket between the apodemes of the meso- and metathorax, a flattened spine (Fig. 98, do) with two perforations through which pass the commissures connecting the ganglia. Besides these processes there are large, thin, longitudinal partitions passing down from the tergum (or dorsum), called phragmas; they are most developed in those insects which fly best, i.e. in Coleoptera (Figs. 97–101), Lepidoptera, Diptera, and Hymenoptera, none being developed in the prothorax. (The term phragma has also been applied to a partition formed by the inflexed hinder edge of this segment, and is present only in those insects in which the prothorax is movable.—Century Dictionary.) All these ingrowths 94may be in general termed apodemes. There are similar structures in Crustacea and also in Limulus; but Sharp restricts this term to minute projections in beetles (Goliathus) situated at the sides of the thorax near the wings. (Insecta, p. 103, Fig. 57.) The internal processes arising from the sternal region have been called endosternites.
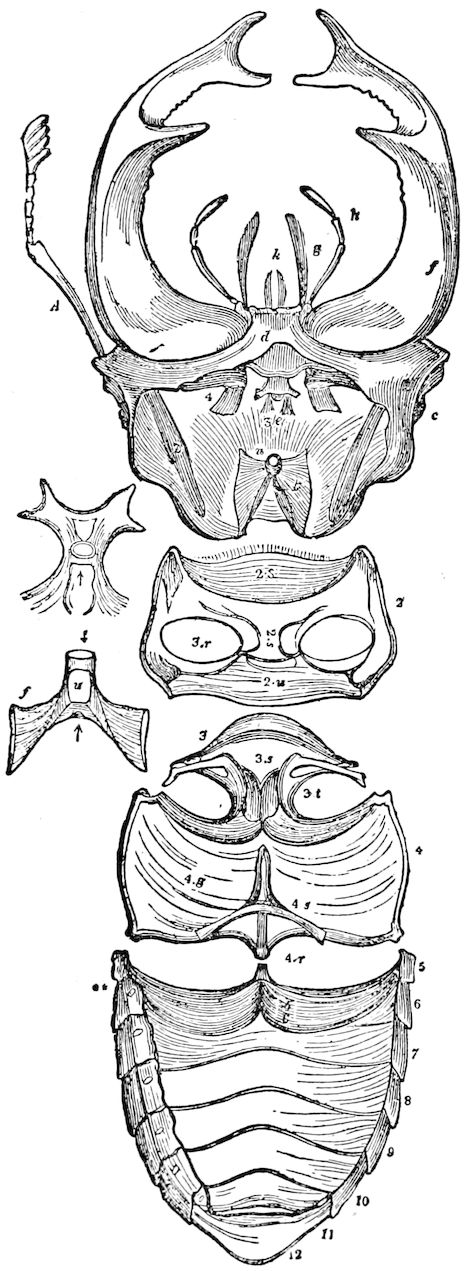
Fig. 101.—Internal skeleton of Lucanus cervus, ♂, head: A, antenna; f, mandible; d, mentum; 2, 4, tendons of mandible; f, u, t, parts of the tentorium; 3 e, labial muscles. Thorax: 2, prothorax; 3, 4, meso- and metathorax fused solidly together; 3 r, acetabulum of prothorax, into which the coxa is inserted; 2 s, sternum; 3t, acetabulum of mesothorax, 4r, of metathorax; 3 s, mesothoracic sternum fused with that of the metathorax (4g); 4 s, apodeme.—After Newport.
The acetabula.—These are the cavities in which the legs are inserted. They are situated on each side of the posterior part of the sternum, in each of the thoracic segments. They are, in general, formed by an approximation of the sternum and epimerum, and sometimes, also, of the episternum, as in Dyticus (Fig. 97, A). This consolidation of parts, says Newport, gives an amazing increase of strength to the segments, and is one of the circumstances which enables the insect to exert an astonishing degree of muscular power.
| 95 | ||
| Tabular View of the Segments, Pieces, and Appendages of the Thorax | ||
| Name of Segment | Pieces (Sclerites) | Appendages |
|---|---|---|
| 1. Prothorax | Pronotum, sometimes differentiated into | |
| Scutum | 1st pair of legs | |
| Scutellum | Patagia | |
| Episternum | ||
| Epimerum | ||
| Sternum | ||
| Antefurca | ||
| 2. Mesothorax | Præscutum | |
| Scutum | 2d pair of legs | |
| Scutellum | 1st pair of wings | |
| Proscutellum | Tegulæ | |
| Episternum | Squamæ (Alulæ) | |
| Epimerum | Peritreme | |
| Sternum | ||
| Mesofurca | ||
| Mesophragma | ||
| Apodemes | ||
| 3. Metathorax | Præscutum | |
| Scutum | 3d pair of legs | |
| Scutellum | 2d pair of wings | |
| Postscutellum | (Halteres of Diptera) | |
| Episternum | ||
| Epimerum | ||
| Sternum | ||
| Postfurca | ||
| Metaphragma | ||
| Apodemes | ||
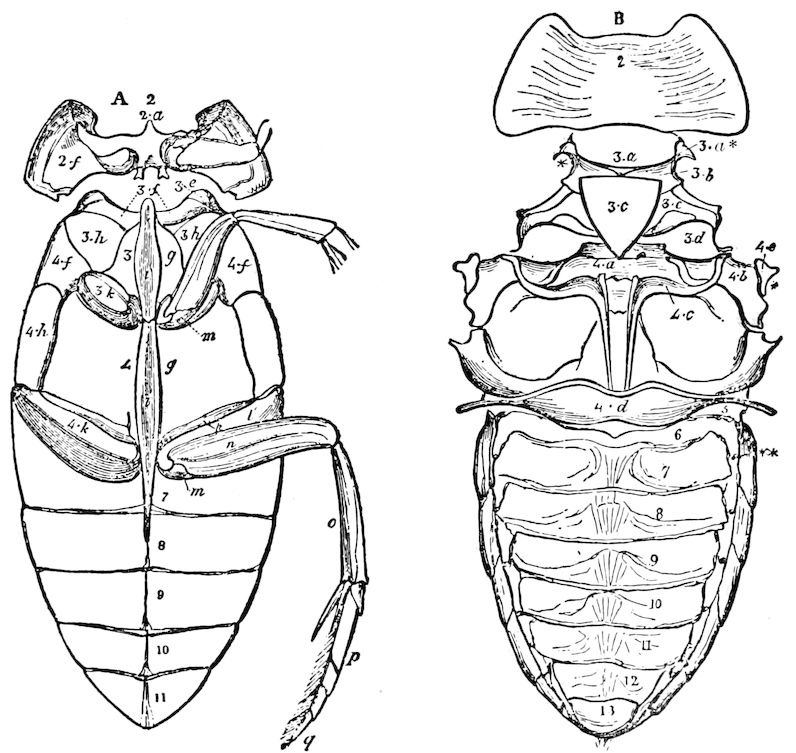
Fig. 102.—External anatomy of the trunk of Hydröus piceus: A, sternal—B, tergal aspect; 2, pronotum; 2 a, prosternum; 2 f, episternum; 3 a, præscutum; 3 b, scutum; 3 c, scutellum; 3 d, postscutellum; 3 g, mesosternum; 3 h, episternum; 3 f, epimerum; 3 i, crest of the mesosternum; 3 a, parapteron; 3 k, coxa; 4 a, metapræscutum; 4 b, metascutum; 4 c, metascutellum; 4 d, postscutellum; 4 e, tegula; 4 f, episternum; 4 h, epimerum; 4 g, metasternum; 4 i, crest of metasternum; 4 k and l, coxa; 4 m, trochanter; n, femur; o, tibia; p, tarsus; q, unguis; 7–11, abdominal segments.—After Newport.
b. The legs: their structure and functions
The mode of insertion of the legs to the thorax is seen in Figs. 90, 97, 101, and 103. They are articulated to the episternum, epimerum, and sternum, taken together, and consist of five segments. The basal segment or joint is the coxa, situated between the episternum and trochanter. The coxa usually has a posterior subdivision or projection, the trochantine; sometimes, as in Mantispa (Fig. 103), the trochantine is obsolete. We had previously supposed that the trochantine was a separate joint, but now doubt whether it represents a distinct segment of the leg, and regard it as only a subdivision of the coxa. It is attached to the epimerum, and is best developed in Panorpidæ, Trichoptera, and Lepidoptera. In the Thysanura the trochantine is wanting, and in the cockroach it merely forms a subdivision of the coxa, its use being to support the latter. The second segment is the trochanter, a more or less short spherical joint on which the leg proper turns; in the parasitic groups (Ichneumonidæ, etc., Fig. 104) it is usually divided into two pieces, though there are some exceptions. The trochanter is succeeded by the femur, tibia, and tarsus, the latter consisting of from one to five segments, the normal number being five. Tuffen West believed that the pulvillus is the homologue of an additional tarsal joint, “a sixth tarsal joint.” The last tarsal segment ends in a pair of freely movable claws (ungues), which are modified setæ; between the claws is a cushion-like 97pad or adhesive lobe, called the empodium or pulvillus (Fig. 105, also variously called arolium, palmula, plantula, onychium, its appendage being called paronychium and also pseudonychium). It is cleft or bilobate in many flies, but in Sargus trilobate. All these parts vary greatly in shape and relative size in insects of different groups, especially Trichoptera, Lepidoptera, Diptera, and Hymenoptera. In certain flies (e.g. Leptogaster) the empodium is wanting (Kolbe). By some writers the middle lobe is called the empodium and the two others pulvilli.
Fig. 103.—Side view of meso- and metathorax of Mantispa brunnea, showing the upper and lower divisions of the epimerum (s. em′, s. em″, i. em′, i. em″); s. epis, i. epis″, the same of the episternum.
Fig. 104.—Divided (ditrochous) trochanter of an ichneumon: cx, coxa; tr, the two divisions of the trochanter; f, femur.—After Sharp.
The fore legs are usually directed forward to drag the body along, while the middle and hind legs are directed outward and backward to push the body onwards. While arachnids walk on the tip ends of their feet, myriopods, Thysanura, and all larval insects walk on the ends of the claws, but insects generally, especially the adults, are, so to speak, plantigrade, since they walk on all the tarsal joints. In the aquatic forms the middle and hind tarsi are more or less flattened, oar-like, and edged with setæ. In leaping insects, as the locusts and grasshoppers, and certain chrysomelids, the hind femora are greatly swollen owing to the development of the muscles within. The tibia, besides bearing large, lateral, external spines, occasionally bears at the end one or more spines or spurs called calcaria. The fore tibia also in ants, etc., bear tactile hairs, and chordotonal organs, as well as other isolated sense-organs (Janet), and, in grasshoppers, ears.
In the Carabidæ the legs are provided with combs for cleaning the antennæ (Fig. 107), and in the bees and ants these cleansing organs are more specialized, the pectinated spine (calcar) being opposed by a tarsal comb (Fig. 106, d; for the wax-pincers of bees, see g). In general the insects use their more or less spiny legs for cleansing the head, antennæ, palpi, wings, etc., and the 98adaptations for that end are the bristles or spinules on the legs, especially the tibiæ.
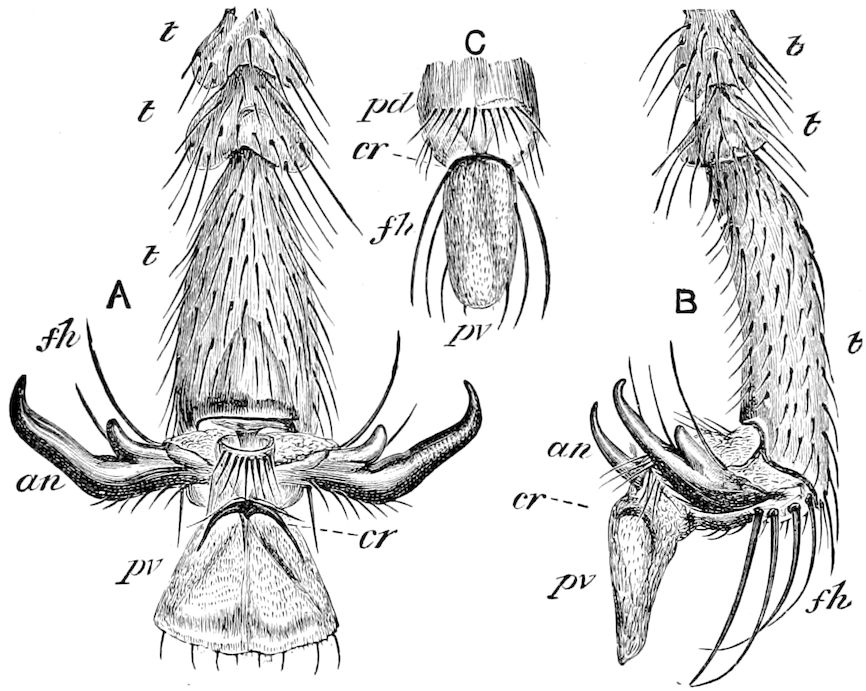
Fig. 105.—Foot of honey-bee, with the pulvillus in use: A, under view of foot; t, t, 3d–5th tarsal joints; a n, unguis; f h, tactile hairs; p v, pulvillus; cr, curved rod. B, side view of foot. C, central part of sole; pd, pad; cr, curved rod; pv, pulvillus unopened.—After Cheshire.
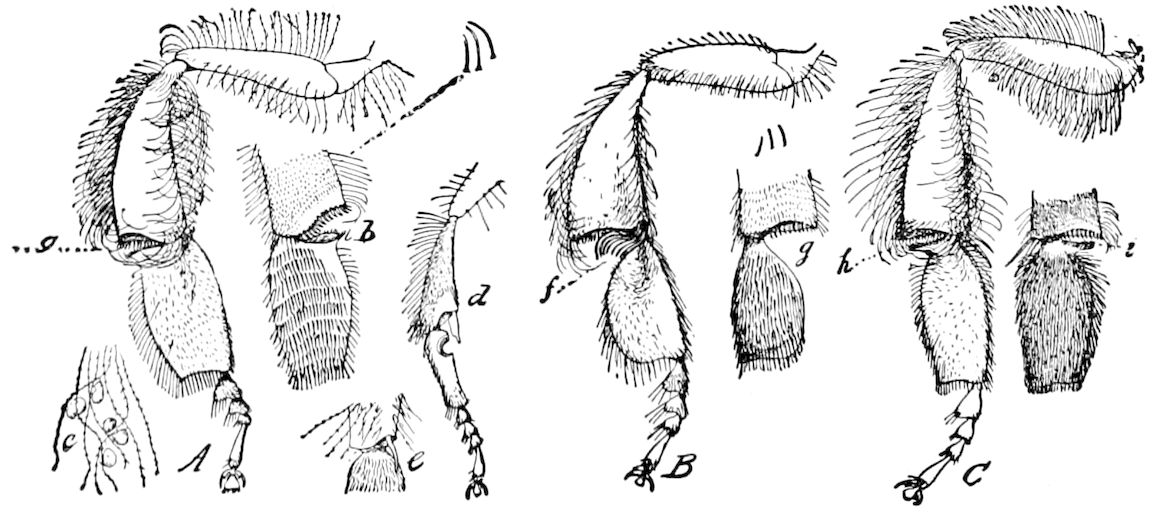
Fig. 106.—Modifications of the legs of different bees. A, Apis: a, wax-pincer and outer view of hind leg; b, inner aspect of wax-pincer and leg, with the nine pollen-brushes or rows of hairs; c, compound hairs holding grains of pollen; d, anterior leg, showing antenna-cleaner; e, spur on tibia of middle leg. B, Melipona: f, peculiar group of spines at apex of tibia of hind leg; g, inner aspect of wax-pincer and first tarsal joint. C, Bombus: h, wax-pincer; i, inner view of the same and first tarsal joint, all enlarged.—From Insect Life, U. S. Div. Ent.
Osten Sacken states that among Diptera the aerial forms (Bombylidæ, etc.) with their large eyes or holoptic heads, which carry with them the power of hovering or poising, have weak legs, principally fit for alighting. On the other hand, the pedestrian or walking Diptera (Asilidæ, etc.) “use the legs not for alighting only, but for running, and all kinds of other work, seizing their prey, carrying 99it, climbing, digging, etc.; their legs are provided not only with spines and bristles, but with still other appendages, which may be useful, or only ornamental, as secondary sexual characters.”
Fig. 107.—End of tibia and tarsal joints of Anophthalmus; c, comb.
Tenent hairs.—Projecting from the lower surface of the empodium are the numerous “tenent hairs,” or holding hairs, which are modified glandular setæ swollen at the end and which give out a minute quantity of a clear adhesive fluid (Figs. 108, 109, 130, 134). In larval insects, and the adults of certain beetles, Coccidæ, Aphidæ, and Collembola, which have no empodium, there are one or more of these tenent hairs present. They enable the insect to adhere to smooth surfaces.
Fig. 108.—Transverse section through a tarsal joint of Telephorus, a beetle: ch, cuticula of the upper side; m, its matrix; ch′, the sole; m′, its matrix; h, adhesive hair; h′, tactile hair, supplied with a nerve (n′), and arising from a main nerve (n); n″, ganglion of a tactile hair; t, section of main trachea, from which arises a branch (t′); dr, glands which open into the adhesive hairs, and form the sticky secretion; e, chitinous thickening; s, sinew; b, membrane dividing the hollow space of the tarsal joint into compartments. See p. 111.—After Dewitz.
Striking sexual secondary characters appear in the fore legs of the male Hydrophilus, the insect, as Tuffen West observes, walking on the end of the tibia alone and dragging the tarsus after it. The last tarsal joint is enlarged into the form of an irregular hollow shield. The most completely suctorial feet of insects are those of the anterior pair of Dyticus (Fig. 132). The under side of the three basal joints is fused together and enlarged into a single broad and nearly circular shield, which is convex above and fringed with fine branching hairs, and covered beneath with suckers, of which two are exceptionally large; by this apparatus of 100suckers the male is enabled to adhere to the back of its mate during copulation. The line branching hairs around the edge prevent the water from penetrating and thus destroying the vacuum, “while if the female struggle out of the water, by retaining the fluid for some time around the sucker, they will in like manner under these altered conditions equally tend to preserve the effectual contact.” (Tuffen West.)
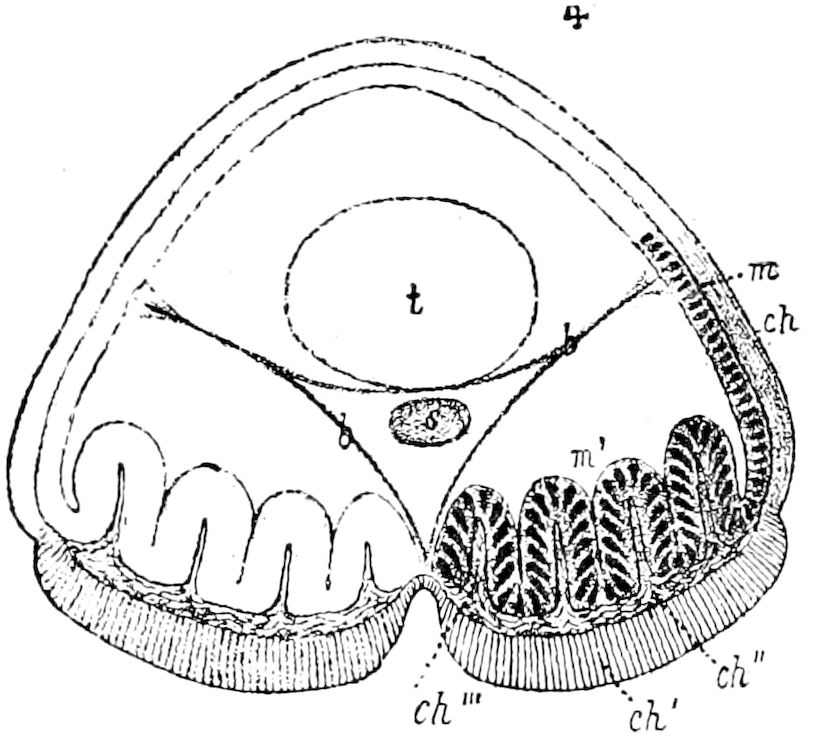
Fig. 109.—Cross-section through tarsus of a locust: ch, cuticula of upper side,—ch′, ch″, ch‴, of sole; ch, tubulated layer; ch″, lamellate layer; ch‴, inner projections of ch″. Other lettering as in Fig. 101. See p. 113.—After Dewitz.
In the saw-flies (Uroceridæ and Tenthredinidæ) and other insects, there are small membranous oval cushions (arolia, Figs. 109 and 131) beneath each or nearly each tarsal joint.
The triunguline larvæ of the Meloidæ are so called from apparently having three ungues, but in reality there is only a single claw, with a claw-like bristle on each side.
Why do insects have but six legs?—Embryology shows that the ancestors of insects were polypodous, and the question arises to what cause is due the process of elimination of legs in the ancestors of existing insects, so that at present there are no functional legs on the abdomen, these being invariably restricted (except in caterpillars) to the thorax, and the number never being more than six. It is evident that the number of six legs was fixed by heredity in the Thysanura, before the appearance of winged insects. We had thought that this restriction of legs to the thorax was in part due to the fact that this is the centre of gravity, and also because abdominal legs are not necessary in locomotion, since the fore legs are used in dragging the insect forwards, while the two hinder pairs support and push the body on. Synchronously with this elimination by disuse of the abdominal legs, the body became shortened, and subdivided into three regions. On the other hand, as in caterpillars, with their long bodies, the abdominal legs of the embryo persist; or if it be granted that the prop-legs are secondary structures, then they were developed in larval life to prop up and move the abdominal region.
The constancy of the number of six legs is explained by Dahl as being in relation to their function as climbing organs. One leg, he says, will almost always be perpendicular to the plane when the animal is moving up a vertical surface; and, on the other hand, we know that three is the smallest number with which stable equilibrium is possible; an insect must therefore have twice this number, and the great numerical superiority of the class may be associated with this mechanical advantage. (This numerical superiority of insects, however, seems to us to be rather due to the acquisition of wings, as we have already stated on pages 2 and 120.)
101Loss of limbs by disuse.—Not only are one or both claws of a single pair, or those of all the feet atrophied by disuse, but this process of reduction may extend to the entire limb.
In a few insects one of the claws of each foot is atrophied, as in the feet of the Pediculidæ, of many Mallophaga, all of the Coccidæ, in Bittacus, Hybusa (Orthoptera), several beetles of the family Pselaphidæ, and a weevil (Brachybamus). Hoplia, etc., bear but a single claw on the hind feet, while the allied Gymnoloma has only a single claw on all the feet. Cybister has in general a single immovable claw on the hind feet, but Cybister scutellaris has, according to Sharp, on the same feet an outer small and movable claw. In the water bugs, Belostoma, etc., the fore feet end in a single claw, while in others (Corisa) both claws are wanting on the fore feet. Corisa also has no claws on the hind feet; Notonecta has two claws on the anterior four feet, but none on the hind pair. In Diplonychus, however, there are two small claws present. (Kolbe.)
Fig. 110.—Last tarsal joint of Melolontha vulgaris, drawn as if transparent to show the inner mechanism: un, claws; str, extensor plate; s, tendon of the flexor muscle; vb, elastic membrane between the extensor plate and the sliding surface u; krh, process of the ungual joint; emp, extensor spine, and th, its two tactile hairs.—After Ockler, from Kolbe.
Among the Scarabæidæ, the individuals of both sexes of the fossorial genus Ateuchus (A. sacer) and eight other genera, among them Deltochilum gibbosum of the United States, have no tarsi on the anterior feet in either sex. The American genera Phanæus (Fig. 111), Gromphas, and Streblopus have no tarsal joints in the male, but they are present in the female, though much reduced in size, and also wanting, Kolbe states, in many species of Phanæus. The peculiar genus Stenosternus not only lacks the anterior feet, but also those of the second and third pair of legs are each reduced to a vestige in the shape of a simple, spur-like, clawless joint. The ungual joint is wanting in the weevil Anoplus, and becomes small and not easily seen in four other genera.
Ryder states that the evidence that the absence of fore tarsi in Ateuchus is due to the inheritance of their loss by mutilation is uncertain. Dr. Horn suggests that cases like Ateuchus and Deltochilum, etc., “might be used as an evidence of the persistence of a character gradually acquired through repeated mutilation, that is, a loss of the tarsus by the digging which these insects perform.” On the other hand, the numerous species of Phanæus do quite as much digging, and the anterior tarsi of the male only are wanting. “It is true,” he adds, “that many females are seen which have lost their anterior tarsi by digging; have, in fact, worn them off; but in recently developed specimens the 102front tarsi are always absent in the males and present in the females. If repeated mutilation has resulted in the entire disappearance of the tarsi in one fossorial insect, it is reasonable to infer that the same results should follow in a related insect in both sexes, if at all, and not in the male only. It is evident that some other cause than inherited mutilation must be sought for to explain the loss of the tarsi in these insects.” (Proc. Amer. Phil. Soc., Philadelphia, 1889, pp. 529, 542.)
Fig. 111.—Fore tibia of Phanæus carnifex, ♂, showing no trace of the tarsus.
Fig. 112.—Fore leg of the mole-cricket: A, outer, B, inner, aspect; e, ear-slit.—After Sharp.
The loss of tarsi may be due to disuse rather than to the inheritance of mutilations. Judging by the enlarged fore tibiæ, which seem admirably adapted for digging, it would appear as if tarsi, even more or less reduced, would be in the way, and thus would be useless to the beetles in digging. Careful observations on the habits of these beetles might throw light on this point. It may be added that the fore tarsi in the more fossorial Carabidæ, such as Clivina and Scarites, as well as those of the larva of Cicada and those of the mole crickets (Fig. 112), are more or less reduced; there is a hypertrophy of the tibiæ and their spines. The shape of the tibia in these insects, which are flattened with several broad triangular spines, bears a strong resemblance to the nails or claws of the fossorial limbs of those mammals which dig in hard soil, such as the armadillo, manis, aardvark, and Echidna. The principle of modification by disuse is well illustrated in the following cases.
In many butterflies the fore legs are small and shortened, and of little use, and held pressed against the breast. In the Lycænidæ the fore tarsi are without claws; in Erycinidæ and Libytheidæ the fore legs of the males are shortened, but completely developed in the females, while in the Nymphalidæ the fore legs in both sexes are shortened, consisting in the males of one or two joints, the claws being absent in the females. Among moths loss of the fore tarsi is less frequent. J. B. Smith[21] notices the lack of the fore tarsi in the male of a deltoid, Litognatha nubilifasciata (Fig. 113), while the hind feet of 103Hepialus hectus are shortened. In an aphid (Mastopoda pteridis, Esl.) all the tarsi are reduced to a single vestigial joint (Fig. 114).
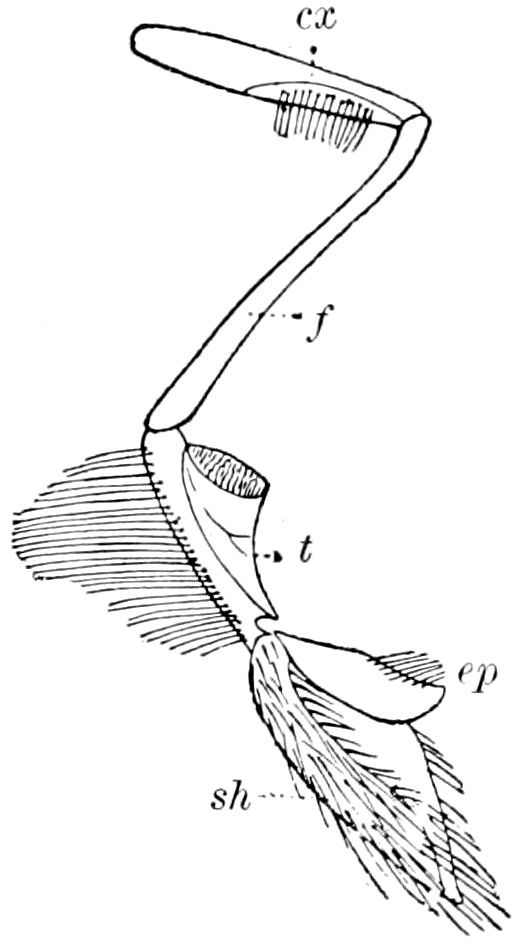
Fig. 113.—Leg of Litognatha: cx, coxa; f, femur; t, tibia; ep, its epiphysis, and sh, its shield-like process. The tarsus entirely wanting.—After Smith.
Entirely legless adult insects are rare, and the loss is clearly seen to be an adaptation due to disuse; such are the females of the Psychidæ, the females of several genera of Coccidæ (Mytilaspis, etc.), and the females of the Stylopidæ.
Apodous larval insects are common, and the loss of legs is plainly seen to be a secondary adaptive feature, since there are annectant forms with one or two pairs of thoracic legs. All dipterous and siphonapterous larvæ, those of all the Hymenoptera except the saw-flies, a few lepidopterous larvæ, some coleopterous, as those of the Rhyncophora, Buprestidæ, Eucnemidæ, and other families, and many Cerambycidæ are without any legs. In Eupsalis minuta, belonging to the Brenthidæ, the thoracic legs are minute.
The legs of larvæ end in a single claw, upon the tips of which the insect stands in walking.
c. Locomotion (walking, climbing, and swimming)
Mechanics of walking.—To Graber we owe the best exposition of the mechanics of walking in insects.
“The first segment of the insect leg,” he says, “upon which the weight of the body rests first of all, is the coxa. Its method of articulation is very different from that of the other joints. The enarthrosis affords the most extensive play, particularly in the Hymenoptera and Diptera.”
In the former the development of their social conditions is very closely connected with the freest possible use of the legs, which serve as hands. In the beetles, however, which are very compactly built, there exists a solid articulation whereby the entire hip rests in a tent-like excavation of the thorax, and can only be turned round a single axis, as may be seen in Fig. 115, where c represents the imaginary revolving axis and d the coxa. In the case we are supposing, therefore, only a backward and forward movement of the coxa is possible, the extent of the play of which depends on the size of the coxal pan, as well as certain groin or bar-like structures which limit further rotation. In the very dissimilar arrangement which draws in the fore, middle, and hind legs toward the body it is self-evident that their extent of action is also different. This arrangement seems to be most yielding on the fore legs, where the hips, to confine ourselves to the stag-beetles, can be turned backward and forward 60° from 104the middle or normal position, and therefore describe on the whole a curve of 120°. The angle of turning on the middle leg hardly exceeds a legitimate limit, yet a forward as well as a backward rotation takes place. The former is entirely wanting in the hind hips; they can only be moved backward.
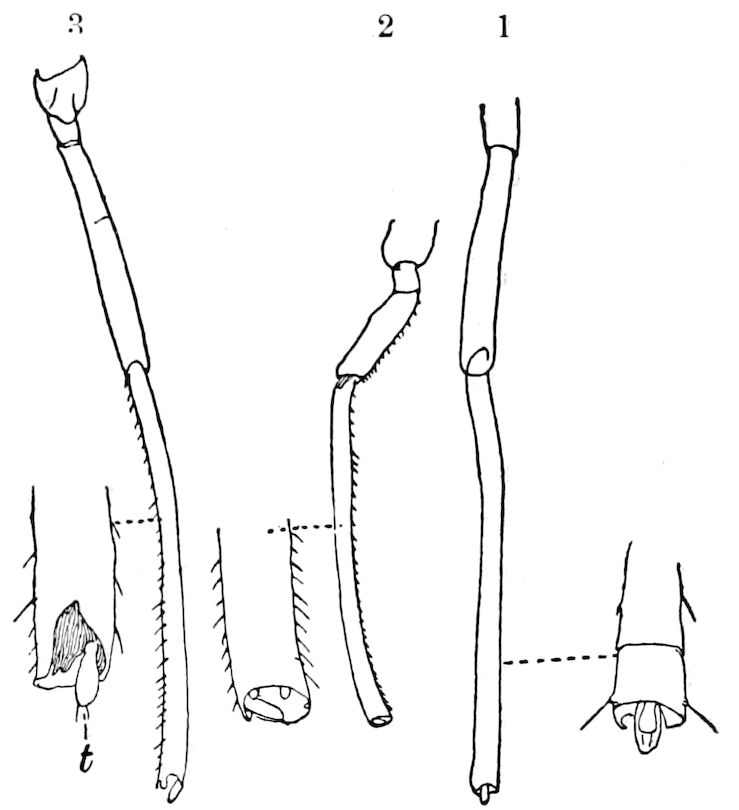
Fig. 114.—Leg of an Aphid, with the tarsus (t) much reduced: 1, 2, 3, legs of 1st, 2d, and 3d pairs.
The number and strength of the muscles on which the rotation of the hips depends, correspond with these varying movements of the individual legs. Thus, according to Straus Durckheim, the fore coxa of many beetles possesses five separate muscles and four forward and one backward roll; the middle coxa a like number of muscles but only two forward rolls, while the hind hips succeed in accomplishing each of the motions named with a single muscle.
One can best see how these muscles undertake their work, and above all how they are situated, if he lays bare the prothorax of the stag beetle (Fig. 116). Here may be seen first the thick muscle which turns to the front the rotating axis in its cylindrical pan, and thus helps to extend the leg, while two other tendons, which take the opposite direction, are fitted for reflex movements.
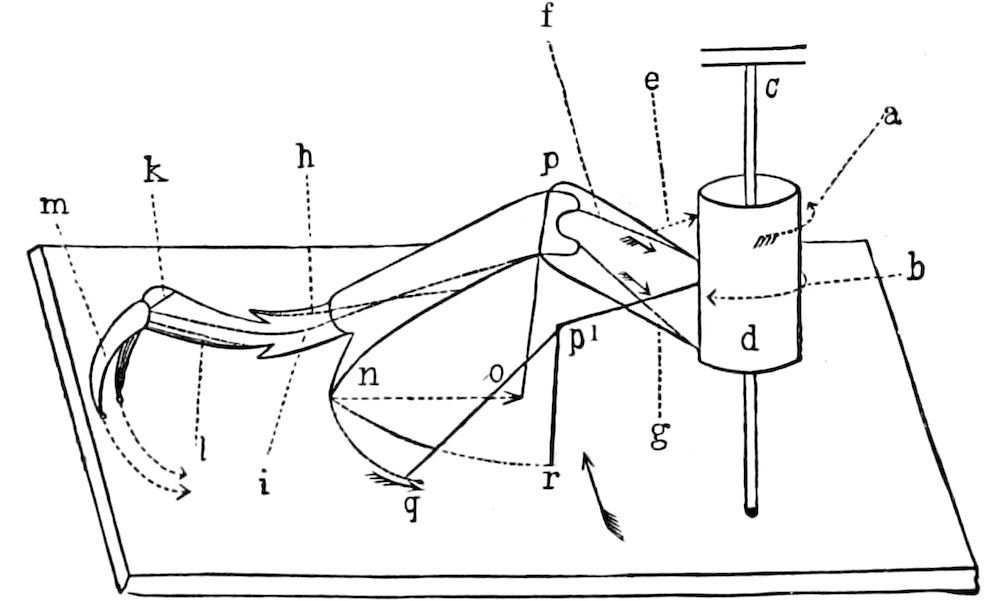
Fig. 115.—Mechanics of an insect’s leg: d, coxa,—c, axis of revolution; a and b, the coxal muscles; e, trochanter muscle (elevator of the femur); f, extensor,—g, flexor, of the tibia (pn); n, tibial spine; h, flexor.—i, extensor, of the foot; k, extensor,—l, flexor, of the claw; po, place of flexure of the tibia; p1q, leg after being turned back by the coxa.—p1r, by the simultaneous flexure of the tibia. The resulting motion of the end of the tibia, through the simultaneous movement (no) and revolution (nq), indicates the curve nr.—After Graber.
In Fig. 115 the muscles mentioned above, and their modes of working, may be distinguished by the arrows a and b.
In order to simplify matters, we will imagine the second component part 105of the normal insect leg, i.e. the trochanter (Figs. 116, 117, r), as grown together with the third lever, i.e. the femur, as the movement of both parts mostly takes place uniformly.
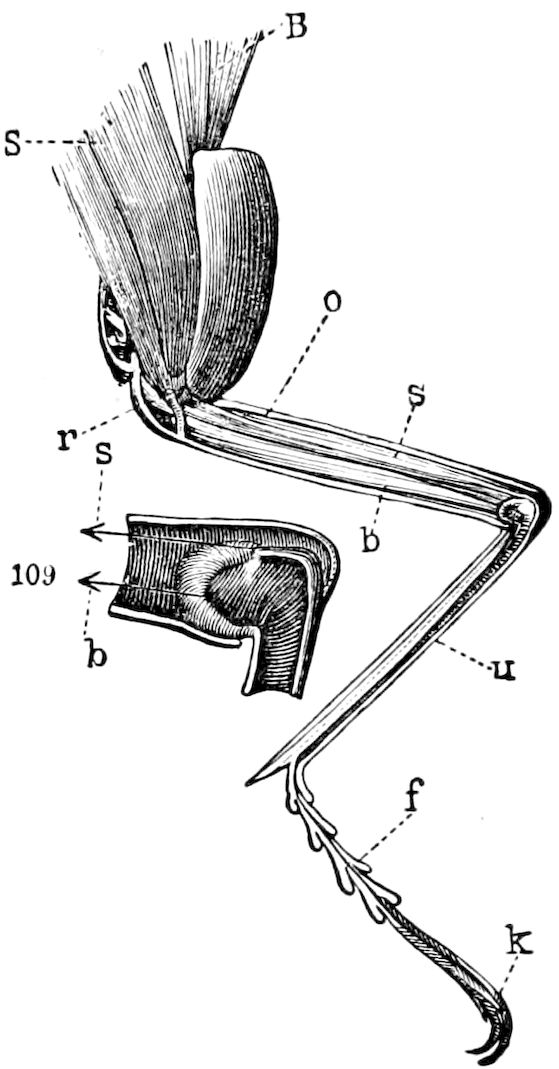
Fig. 116.—Section of the fore leg of a stag-beetle, showing the muscles: S, extensor,—B, flexor, of the leg; s, extensor,—b, flexor, of the femur; o, femur; u, tibia; f, tarsus; k, claw; 109, s, extensor,—b, flexor, of the femoro-tibial joint, both enlarged.—After Graber.
The pulling of the small trochanter muscle works against the weight of the body when this is carried over on to the trochanter by means of the coxa, as seen at the arrow e in Fig. 115. It may be designated as the femoral lever.
The plane of direction in which the femur, as seen by the rotation just mentioned, is moved, exactly coincides in insects with that of the tibia and the foot, while all can be simultaneously raised or dropped, or, as the case may be, stretched out or retracted. Therein, therefore, lies an essential difference from the fully developed extremities of vertebrates among which, even on the lever arms which are stationary at the end, an extensive turning is possible.
The muscles which move the tibia, and indirectly the femur, also consist of an extensor muscle which is situated in the upper side of the femur (Fig. 116, s, Fig. 115, f), and of a flexor (Fig. 116, b, Fig. 115, g), which lies under the former.
The stilt-like spines on the point (Figs. 115 and 118, L3n) on which this segment is directly supported are important parts of the tibia. (Graber.)
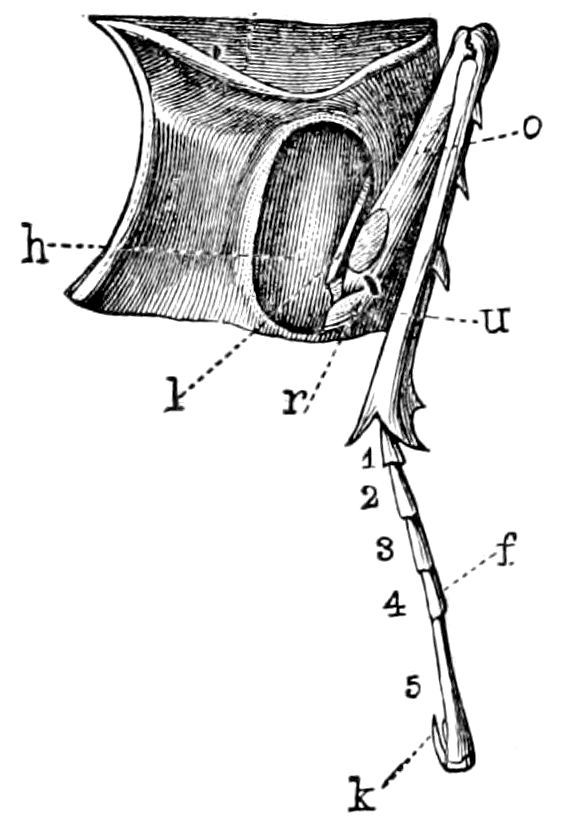
Fig. 117.—Left fore leg of a cerambycid beetle: h, coxa; r, trochanter; o, femur; u, tibia; f, tarsus; k, claw.—After Graber.
Considering the respective positions of the individual levers of the leg and the nature of the materials of which they are made, the legs of insects may be likened, as Graber states, to elastic bows, which, when pressed down together from above, their own indwelling elasticity is able to raise again and thus keep the body upright.
This is very plainly shown in certain stilt-legged bark-beetles, in which, as in a rubber doll, as soon as the body is pressed down on the ground, the organs of motion extend again without the intervention of muscles; indeed this experiment succeeds even with dead, but not yet wholly stiff, insects.
Graber then turns to the analysis of the movements of insect legs when in motion, 106and the mode of walking of these insects in general. This subject had been but slightly investigated until Graber made a series of observations and experiments, of which we can give only the most important results.
The locomotion of insects is an extremely complicated subject.
Let us consider, Graber says, first, a running or carabid beetle, when walking merely with the fore and hind legs. The former will be bent forward and the latter backward.
“Let us begin with the left fore leg (Fig. 118, L1). Let the same be extended and fixed on the ground by means of its sharp claws and its pointed heel. Now what happens when the tibial flexors draw together? As the foot, and therefore the tibia also, have a firm position, then the contraction of the muscles named must cause the femur to approach the tibia, whereby the whole body is drawn along with it. This individual act of motion may be well studied in grasshoppers when they are climbing on a twig by stretching out their long fore leg directly forward, and then drawing up the body through the shortening of the tibial flexors until the middle leg also reaches the branch.
“But while the fore legs advance the body by drawing the free lever to the fixed leg-segment, the hind legs do this in exactly the opposite way. The hind leg, namely, seeks to stretch out the tibia, and thus to increase the angle of the knee (R3), thereby giving a push on the ground, by means of which the body is shoved forward a bit.
“Though it might be supposed that the feet would remain stationary during the extension or retraction of the limbs, this never occurs in actual walking. Not merely the upper, but also the lower, thigh is either drawn in or stretched out, as the case may be. The latter then describes a straight line with its point during this scraping or scratching motion (Fig. 115, no), which is obviously the chord to that quadrant which would be drawn by the tibia or foot in a yielding medium, as water, for instance. But even this motion results extremely rarely, and never in actual walking. If we fix our eye anew upon the fore leg at the very moment when it is again retracted, after the resultant ‘fixing,’ we shall then observe that the hip also is simultaneously turned backward in a definite angle. The tibia would describe the arc nq (Fig. 115) by means of the latter alone.
“This plane, in conjunction with the rectilinear ‘movement’ (no) obtained by the retraction of the tibia, produces a path (nr), and this is what is actually described by a painted foot upon a properly prepared surface, as a sheet of paper;[22] supposing, however, that the body in the meantime is not moved forward by other forces. In the last case, and this indeed always takes place in running, the trunk is moved a bit forward, together with the leg which is just describing its curve with a rapidity corresponding to the momentum obtained; the result of this is that the curve of the foot from its beginning (n) to its end (a) bends round close to itself, just as a man who, when on board a ship in motion, walks across it diagonally, and yet on the whole moves forward, because his line of march, uniting with that of the ship, results in a change of position in space.
107“The case is the same in the middle and hind legs, which must make a double course also, yet in such a way that the straight line is drawn, not during the retraction, but during the extension; during which, however, quite as in the fore leg, the members mentioned (R3) gradually approach the body.
“When the legs have reached the maximum of their retraction, or of their extension, as the case may be, and therefore the end of their active course for that time, then begins the opposite or backward movement; that is, the fore legs are again extended, while their levers draw the remaining legs together again.
Fig. 118.—A Carabus beetle in the act of walking or running: three legs (L1, R2, L3) are directed forward, while the others (R1, L2, R3), which are directed backward toward the tail, have ended their activity; ab, cd, and ef are curves described by the end of the tibiæ, and passing back to the end of the body; bh, di, and fg are curves described by the same legs during their passive change of position.—After Graber.
“At the same time, as we may see by the uniting leg, the limb is either a little raised, that there may be no unnecessary friction, or it remains during the passive step also, with its means of locomotion in slight contact with the ground.
“The curve of two steps, as inscribed by the end of the tibia of the left fore leg of a stag-beetle, affords an instructive summary of the conditions of which we have been speaking (Fig. 121, B). We see two curves. The thick one (ab), directed toward the axis of the body, corresponds to the effective act of a single 108walking function, which brings the body a bit forward; the thinner, on the other hand, or we might say the hair line (bc), which, however, is but rarely made quite clearly, is produced by the ineffectual backward movement, by which the insect again approaches its working posture (c). It is at first placed at some distance from the body, in order that (like c also) it may draw near to the body again; but in such a way, naturally, that it coincides with the starting-point of the following active curve (cd). It is evident that even the passive curve is not the imprint of the movement accomplished exclusively by the leg, for this latter, while struggling to reach its resting-place, is really involuntarily carried forward with the rest of the body.
“The scroll-like lines drawn by the swimming beetle (Dyticus), with the large, sharp points of its hind tibia, are also very instructive (Fig. 119, A).
Fig. 119.—A, trail curves described by the tibial spines of the right and left hind limb of Dyticus. B, the same made by the right hind leg (r3) alone. Natural size.—After Graber.

Fig. 120.—The same by the two hind legs of Melolontha: a, the active and thickened section of the curve. Natural size.
Fig. 121.—A, track curves of two of the tibial spines of the left, middle legs of a stag-beetle. Natural size. B, the same enlarged; fg, the longitudinal axis of the trunk; cd and ab, the active curve passing inward,—bc and de, the passive going outward. C, two curves described by the left hind legs; in this case, the curves are not inwards or backwards, but partly directly inward (b), and in part obliquely forwards (a).
“The diversions and modifications in the course of the active step, as furnished by the moving factor of the remaining legs, are already clearly illustrated by the curves shown by the joints of the hind tibia of a May-beetle (Fig. 120) and a stag-beetle (Fig. 121, c). The actual faint line in this case does not run from the front toward the back, as would correspond to the active leg-motion, but either directly inward (Fig. 121, cb), or even somewhat to the front. In the May-beetles, and even more in the running garden-beetle, the curves of the hind legs present themselves as screw-like lines (Fig. 122, l3), while the scrawling of the remaining members (l1, l2) is much simpler.
“Inasmuch as we now have a cursory knowledge of the movements made by each individual leg for itself,—movements, however, which plainly occur very differently according to the structure of these appendages,—the question now is of the combined play, the total effect of all the legs taken together, and therefore of the walk and measure of the united work of the foot.
“In opposition to the caterpillars and many other crawling animals which extend their legs in pairs and really swing them by the worm-like mode of contraction of the dermo-muscular tube, the legs of fully grown insects are moved in the contrary direction and in no sense in pairs, but alternately—or, more strictly speaking, in a diagonal direction.
109“For an examination of the gait of insects, we choose, for obvious reasons, those which have very long legs and which at the same time are slow walkers.
“Insects may be called ‘double-three-footed,’ from the manner in which they alternately place their legs. There are always three legs set in motion at the same time, or nearly so, while in the meantime the remaining legs support the body, after which they change places.

Fig. 122.—The same by the left fore (l1), middle (l2), and hind, leg (l3) of a Carabus. Natural size.
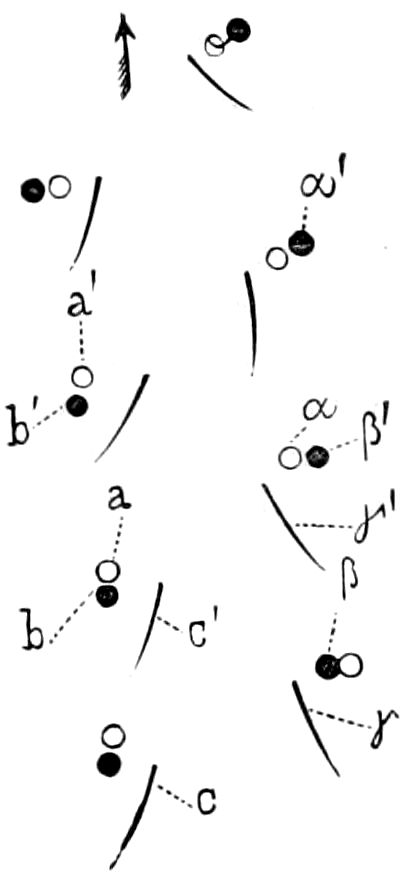
Fig. 123.-Tracks of a Blaps mortisaga marked by the differently painted tibial points: ●, tracks of fore, —○, middle, —/, hind leg. Natural size.
Fig. 124.—Tracks of Necrophorus vespilio. Natural size.
“To be more exact, it is usually thus: At first (Fig. 118) the left fore leg (L1) steps out, then follows the right middle leg (R2), and the left hind leg (L3). Then while the left fore leg begins to retract and thus make the backward movement, the right fore leg is extended, whereupon the left middle leg and the right hind leg are raised in the same order as the first three feet.”
Graber[23] painted the feet of beetles and let them run over paper, and goes on to say:
“Let us first pursue the tracks of the Blaps, for example (Fig. 123). Let the insect begin its motion. The left fore leg stands at a, the right middle leg at β, and the left hind leg at c. The corresponding number of the other set of three feet at α, b, γ. At the first step the three feet first mentioned advance to a′β′c′, the second set on the other hand to α′b′γ′. Thereby the tracks made by the successive steps fall quite, or almost quite, on each other, as appear also in the tracks of a burying beetle (Fig. 124).
“As the fore legs are directed forward and the hind legs backward, while the middle legs are placed obliquely, the reason of the more marked impressions of the latter is evident.
“The highest testimony to the precise exactitude and accuracy of the walking mechanism of insects is furnished by the fact that in most insects, and particularly in those most fleet of foot, which, whether they are running away or chasing their prey, must be able to rely entirely upon their means of locomotion;—the fact, we say, that whether they desire to move slowly or more 110quickly, the distances of the steps, measured by the length as well as by the cross-direction, hardly differ a hair’s breadth from one another, and this is also the case when the tarsi are cut off and the insects are obliged to run on the points of their heels (tibiæ).
“Thence, inasmuch as the trunk of insects is carried by two legs and by one on each side alternately, it may surely be concluded a priori that when walking it is inclined now to the right and now to the left, and that the track, too, which is left behind by a precise point of the leg, can in no wise be a straight line; and in reality this is not the case.
“A plainly marked regular curve, which approaches a sinuous line, as seen in Fig. 125, is often obtained by painting many insects, for example Trichodes, Meloë, etc., which, when running, either bring the end of their hind body near to the ground or into contact with it.
Fig. 125.—Tracks of Trichodes; the middle sinuous line is made by the tip of the abdomen. Natural size.
Fig. 126.—Tracks of another insect which, in running, can only use three legs (r1, l4, r3) which become indicated differently from normal conditions. Natural size.
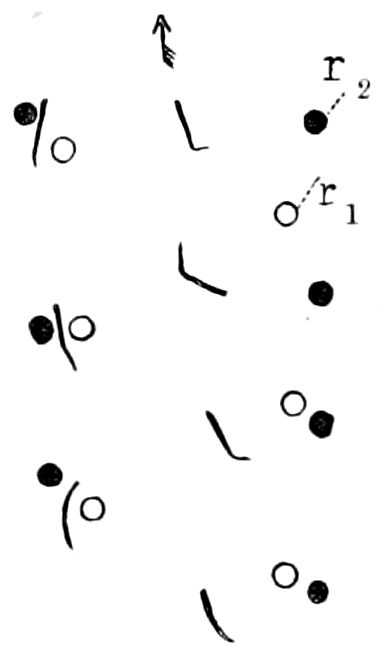
Fig. 127.—The same of an insect crossing over a surface inclined 30° from the horizon, whereby the placing of the feet becomes changed. Natural size.—This and Figs. 120–126 after Graber.
“The locomotive machine of insects may be called, to a certain extent, a double set of three feet each, as most insects, and particularly those provided with a broad trunk, are able to balance themselves with one of these two sets of feet, and indeed when walking, as well as when standing still, can move about even better with one set of these feet than with four legs. In the latter case, that is, if one cuts off a pair of legs from an insect, the trunk can balance itself only with extreme difficulty, and there is therefore little prospect that insects will ever become four-footed.
“But if one compels insects to run on three legs, he will thus make the interesting discovery that to make up the deficiency they place the remaining feet and bring them to the ground somewhat differently than when the second set of feet is active. Figs. 124 and 126 may be compared for this purpose. The former shows the footprints of a burying beetle running with all six legs, the latter the track of the same insect, which, however, has at its disposal only the right fore leg, the left middle leg, and the right hind leg. One may plainly see here that the track of the hind leg on the right side (r3) approaches the track of the middle leg on the left side, and then further, that the right fore leg (r1) steps out more to the right to make up for the deficiency of the middle leg.
111“A similar adaptation of the position of the legs, which is entirely dependent on the choice of the insect, may also be observed there, if one compels insects which are not provided with corresponding adhesive lobes to run away over crooked surfaces. Fig. 123 shows the footprints of a Blaps when running upon a horizontal plane. Fig. 127, on the contrary, shows the tracks of the legs when going diagonally over a gradually inclined surface. Here, also, the insect holds on with his fore and middle legs (r1, r2) stretched upward, whereby also the impressions on both sides come to lie farther apart than in the normal mode of walking.
“It will not surprise the reader who is familiar with the gait of crabs, to hear that many insects also understand the laudable art of going backward, wherein the hind legs simply change places with the fore legs.
“The jumping motion of insects may be best studied in grasshoppers. When these insects are preparing for a jump, they stretch out the upper thigh horizontally, clap the tibiæ together, and also retract the foot-segment. After a slight pause for rest, during which they are getting ready for the jump, they then jerk the tibiæ suddenly backward and against the ground with all their strength by means of the extensor muscles.”
The correctness of Graber’s views has been confirmed by Marey by instantaneous photographs (Figs. 128, 129).
Locomotion on smooth surfaces.—How flies and other insects are able to walk up, or run with the body inverted, on hard surfaces has been lately discovered by Dewitz, Dahl, and others. All authors are agreed that this power is due to the presence of the specialized empodium of each tarsus.
Dewitz confirmed the opinion of Blackwell, that a glutinous liquid is exuded from the apices of the tenent hairs which fringe the empodium. By fastening insects feet uppermost on the under side of a covering glass which projects from a glass slide, the hairs which clothe the empodia of the foot of a fly (Musca erythrocephala) may be seen to be tipped with drops of transparent liquid. On the leg being drawn back from the glass, a transparent thread is drawn out, and drops are found to be left on the glass. In cases where these hairs are wanting, as in the Hemiptera, the adhesive fluid exudes directly from pores in the foot. In the beetles (Telephorus dispar) and other insects the tenent hairs on the foot end in sharp points, below which are placed the openings of the canals. The glands, Dewitz states, are chiefly flask-shaped and unicellular, situated in the hypodermis of the chitinous coat; each gland opening into one of the hairs (Fig. 108); they are each invested by a structureless tunica propria, and contain granular protoplasm, a nucleus placed at the inner side, and a vesicle, prolonged into a tube which, traversing the neck of the gland, is attached to the root of the hair; the vesicle receiving the secretion. Each gland is connected with a fine nerve-twig, and secretion is probably voluntary. Among the tenent 112hairs of the empodium are others which must be supplied with a nerve, forming tactile hairs, as they each proceed from a unicellular ganglion (Fig. 108, n″). The secretion is forced out of the gland by the contraction of the protoplasm, Dewitz having seen the secretion driven out from the internal vesicle into its neck.

Fig. 128.—The walk of an orthopterous insect: series to be followed from right to left.—After Marey.

Fig. 129.—Beetle walking: series to be followed from left to right.—After Marey.
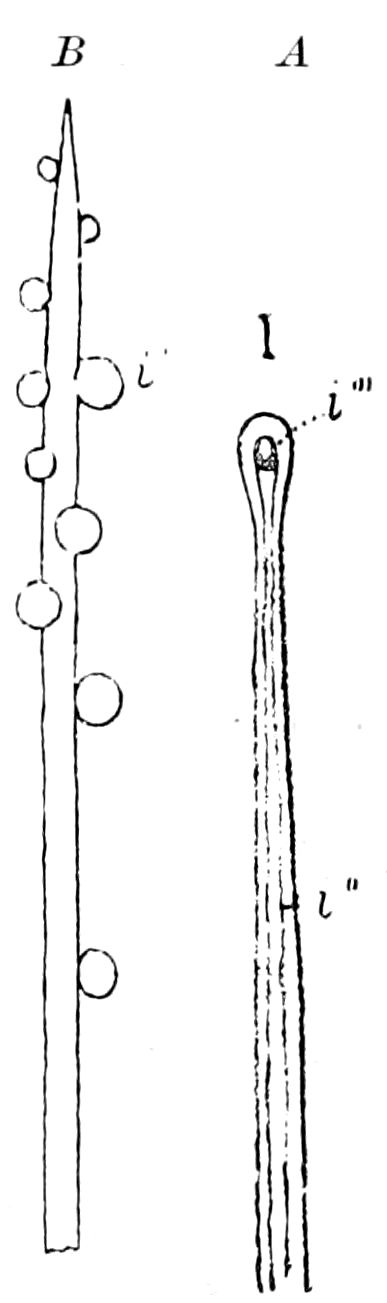
Fig. 130.—A, end of an adhesive hair of a weevil (Eupolus): i′, canal: i‴, its external opening at the end of the hair. B, end of a similar hair of Telephorus with drops of the secretion.—After Dewitz.
In the spherical last tarsal joint of Orthoptera (Fig. 109), which is without these tenent hairs, nearly all the cells of the hypodermis are converted into unicellular glands, each of which sends out a long, fine, chitinous tubule, which is connected with its fellows by very fine hairs and is continuous with the chitinous coat of the foot and opens through it. The sole of the foot is elastic and adapts itself to minute inequalities of surfaces, while the anterior of each tarsal joint is almost entirely occupied by an enlargement of the trachea, which acts on the elastic sole like an air chamber, rendering it tense and at the same time pliant. Dewitz adds that the apparatus situated on the front legs of the male of Stenobothrus sibiricus (Fig. 131) must have the function of causing the legs to adhere closely to the female by the excretion of an adhesive material. The hairs of the anterior tarsi of male Carabi also appear to possess the power of adhesion. In the house-fly the empodia seem to be only called into action when the insect has to walk on vertical smooth surfaces, as at other times they hang loosely down.
Burmeister observed the use of a glutinous secretion for walking in dipterous larvæ, and Dewitz found that the larva of a Musca used for this purpose a liquid ejected from the mouth. The larvæ of another fly (Leucopis puncticornis) perform their loop-like walk by emitting a fluid from both mouth and anus. A Cecidomyia larva is able to leap by fixing its anterior end by means of an adhesive fluid. The larva of the leaf-beetle, Galeruca, moves by drawing up its hinder end, fixing it thus, and carrying the anterior part of the body forward with its feet until fully extended, when it breaks the glutinous adhesion. The abdominal legs of some saw-fly larvæ have the same power.
Dahl could not detect in the foot of the hornet (Vespa crabro) any space which could be considered as a vacuum.
Fig. 131.—Stenobothrus sibiricus pairing: A. the ♂, fore tarsus (t) greatly enlarged; ar, arolia; p, pulvillus.—After Pagenstecher.
Simmermacher states that in most cases of climbing beetles the tubular tenent hairs pour out a secretion (Figs. 133, 134), “and it is probable that we have here to do with the phenomena not of actual attachment by, as it were, gluing, but of adhesion; the orifice of the tubes is divided obliquely, and the tubes are, at this point, extremely delicate and flexible, so as to adhere by their lower surface; 114in this adhesion they are aided by the secreted fluid.” In the case of the Diptera he does not accept the theory by which the movement of the fly along smooth surfaces is ascribed to an alternate fixation and separation, but believes in a process of adhesion, aided by a secretion, as in many Coleoptera. (In the Cerambycidæ there is no secretion, and the tubules are merely sucking organs, like those observed in the male Silphidæ.) “The attaching lobes, closely beset with chitinous hairs, are enabled, in consequence of the pressure of the foot, to completely lie along any smooth surface; this expels the air beneath the lobes, which are then acted on by the pressure of the outer air.” (Journ. Roy. Micr. Soc., 1884, p. 736.) Another writer (Rombouts) thinks this power is due to capillary adhesion.
Fig. 132.—Fore leg of ♂ Dyticus, under side, with sucker, formed of 3 enlarged tarsal joints: with a small cupule highly magnified. × 120.—After Miall.
The action of the pulvillus and claws when at rest or in use by the honey-bee is well shown by Cheshire (Fig. 135, B). In ascending a rough surface, “the points of the claws catch (as at B) and the pulvillus is saved from any contact, but if the surface be smooth, so that the claws get no grip, they slide back and are drawn beneath the foot (as at A), which change of position applies the pulvillus, so that it immediately clings. It is the character of the surface, then, and not the will of the bee, that determines whether claw or pulvillus shall be used in sustaining it. But another contrivance, equally beautiful, remains to be noticed. The pulvillus is carried folded in the middle (as at C, Fig. 105), but opens out when applied to a surface; for it has at its upper part an elastic and curved rod (cr, Figs. 105 and 135), which straightens 115as the pulvillus is pressed down; C and D, Fig. 135, making this clear. The flattened-out pulvillus thus holds strongly while pulled, by the weight of the bee, along the surface, to which it adheres, but comes up at once if lifted and rolled off from its opposite sides, just as we should pull a wet postage stamp from an envelope. The bee, then, is held securely till it attempts to lift the leg, when it is freed at once; and, by this exquisite yet simple plan, it can fix and release each foot at least twenty times per second.” (Bees and Bee-keeping, p. 127.)
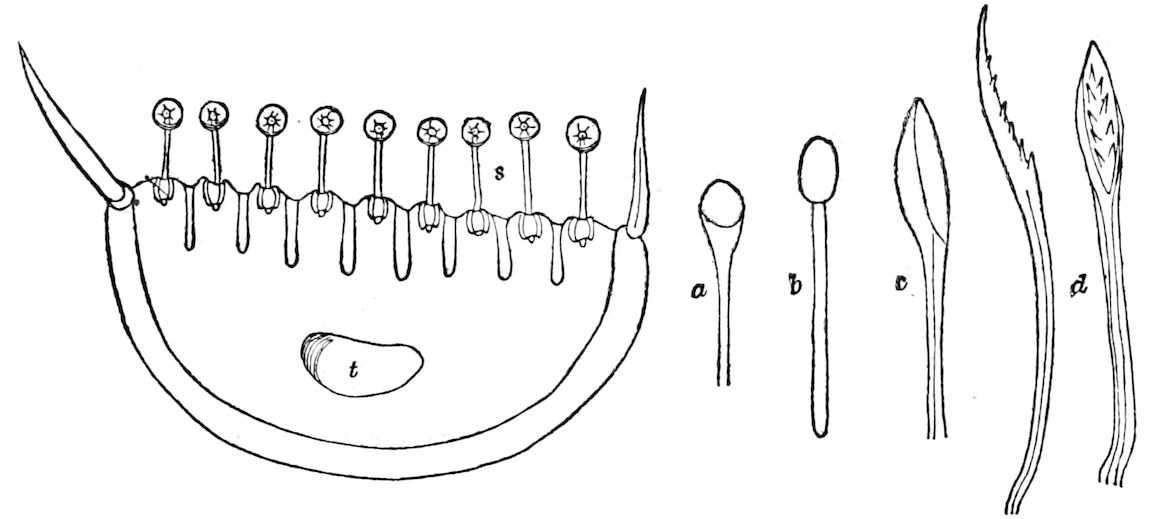
Fig. 133.—Cross-section through a tarsal joint of fore leg of Dyticus, ♂, showing the stalked chitinous suckers (s), with a marginal bristle on each side: t, trachea; a, an isolated tubule or sucker of Loricera,—b, of Chlænius,—c, of Cicindela; d, two views of one of Necrophorus germanicus, ♂.
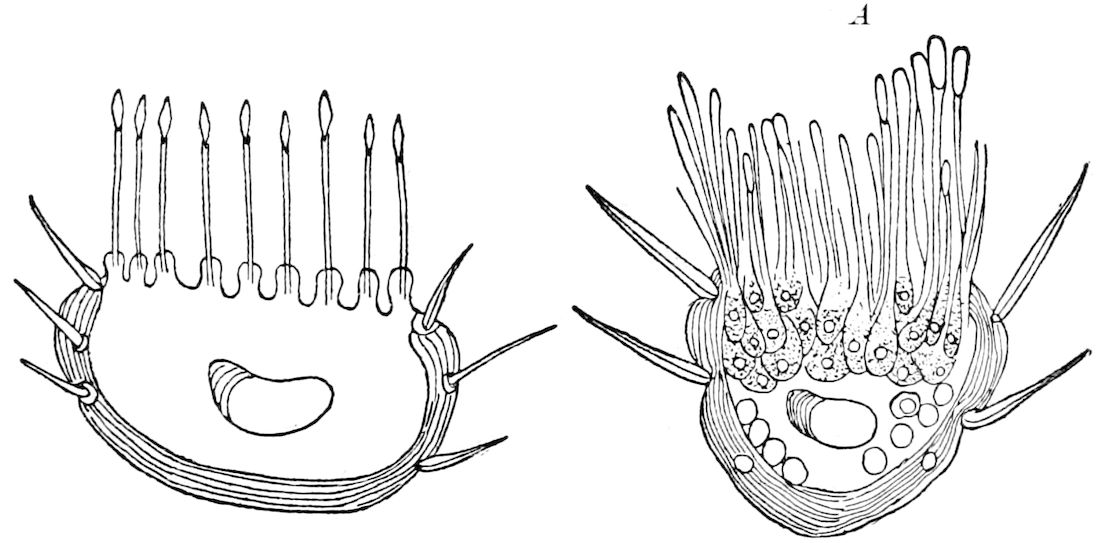
Fig. 134.—Section through the tarsus of a Staphylinid beetle; the glandular or tenent hairs arising from chitinous processes. A, section through the tarsal joint of the pine weevil, Hylobius abietis, showing the crowded, bulbous, glandular, or tenent hairs arising from unicellular glands.—This and Fig. 133 after Simmermacher.
Ockler divides the normal two-clawed foot into three subtypes: 116(1) with an unpaired median empodium; (2) with two outer lateral adhesive lobes; (3) with two adhesive lobes below the claws; the latter is the chief type and forms either a climbing or a clasping foot. The amount of movement possessed by the claws is limited, and what there is, is effected by means of an elastic membrane and the extensor plate (Fig. 110). The “extensor sole” which is always present in insects with an unpaired median fixing or adhesive organ (empodium) is to be regarded as a modification of the extensor seta. The extensor plate is peculiar to an insect’s foot. Ockler states that the so-called “pressure plate” of Dahl is only a movably articulated, skeletal, supporting plate for the median fixing lobule.
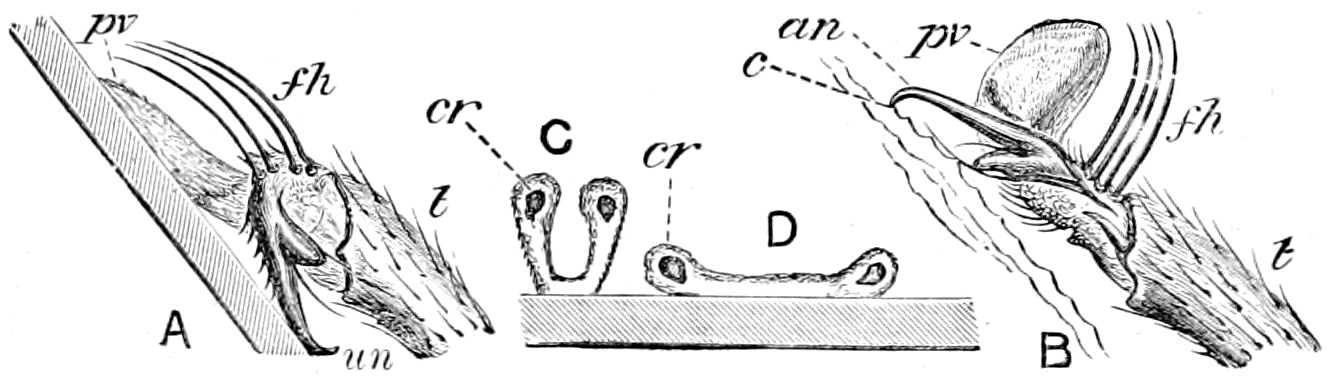
Fig. 135.—Honey-bee’s foot in the act of climbing, showing the automatic action of the pulvillus, × 30: A, position of foot in climbing on a slippery surface, or glass; pv, pulvillus; fh, tactile hairs; un, unguis; t, last tarsal joint. B, position of foot in climbing rough surface. C, section of pulvillus just touching flat surface; cr, curved rod. D, the same applied to the surface.—After Cheshire.
Climbing.—In certain respects the power of climbing supplies the want of wings, and even exists often in house-flies among which there is shown a many-sided motion that is quite unheard of in other groups of insects.
The best climbers are obviously those insects which live on trees and bushes, as, for example, longicorn beetles and grasshoppers. These may be accurately called the monkeys of the insect kind, even if their movements take place less gracefully, and indeed rather stiffly and woodenly. We already know what are the proper climbing organs; that is, the sharp easily movable claws on the foot. With the help of these claws certain insects, May-beetles for example, can hang upon one another like a chain; indeed, bees and ants in this manner bind themselves together into living garlands and bridges. There are still added to the chitinous hooks flaps and balls of a sticky nature, by help of which likewise the insects glue themselves together. To facilitate the spanning of still thicker twigs, the climbing foot of insects has a greater movability even than when it only serves as a sole. (Graber.)
The mode of swimming of insects.—To study the swimming movements of insects, let us examine a Dyticus. It will appear, as Graber states, to be wonderfully adapted to its element.
117“The body resembles a boat. There is nowhere a projecting point or a sharp corner which would offer unnecessary resistance to motion; bulging out in the middle and pointed at the end, it cuts through the resistance of the water like a wedge. The movable parts, the oars, seem to be as well fitted for their purpose as the burden to be moved by them. That the hind legs must bear the brunt of this follows from their position exactly in the middle of the body, where it is widest. In other insects also these legs are used for the same purpose as soon as the insects are put in the water. But the swimming legs of water-beetles are oars of quite peculiar construction. They are not turned about in the coxæ, as are other legs, but at the foot-joint. The coxa, namely, has grown entirely together with the thoracic partition. The muscles we have mentioned, exceeding in strength all the soft parts taken together, take hold directly of the large wing-shaped tendons of the upper thigh, and extend and retract the leg in one of the planes lying close to the abdominal partition. The foot forms the oar, however. It is very much lengthened and still more widened, and can be turned and bent in by separate muscles in such a way that in the passive movement, that is, the retraction, the narrow edge is turned to the fore, and therefore to the medium to be dislodged; however, as soon as the active push is to be performed and the leg is extended with greater force, it cuts down through the water with its whole width. These effective oar-blades are still considerably enlarged by the hairs arising on the side of the foot, which spread out at the decisive moment.
“Every one knows that the oar-blades of swimming beetles always go up and down simultaneously and in regular time. On the other hand, as soon as one puts a Dyticus on the dry land, i.e. on an unyielding medium, it uses its hind legs entirely after the manner of other land insects; that is, they are drawn in and extended again alternately, as takes place clearly enough from the footsteps in Fig. 119, A. We learn from this that water insects have not yet, from want of practice, forgotten the mode of walking of land insects.
“The forcing up of the water as a propelling power is added to the repulsion produced by the strong strokes of the oars. If the beetle stood up horizontally in the water, he would be lifted up.
“As the trunk, however, assumes an oblique position when the insect wishes to swim, one can then imagine the driving up of the water as being divided into two forces, one of which drives the body forward in a horizontal direction, while the other, that is, the vertical component, is supplied by the moving of the oars. The swimming insect is thus, as it were, a snake flying in the water.
“The long streamer-like hind legs of many water-bugs, for example Notonecta, approach more nearly our artificial oars. These legs are turned out from the bottom.
“There is no doubt but that the legs of insects, as regards the many-sidedness and exactitude of their locomotive actions, place the similar contrivances of other animals far in the shade. We shall be forced to admire these ingenious levers still more, however, when we take into consideration their energy and strength. That the force with which the locomotive muscles of insects is drawn together is enormous compared with that of vertebrates, we may learn if we try to subdue the rhythmical movements of the thorax of a large butterfly by the pressure of our finger or to open against the insect’s will the closed jumping leg of a grasshopper, or the fossorial shovel of a mole-cricket.”
LITERATURE ON LEGS AND FEET
MacLeay, W. S. On the structure of the tarsus in the tetramerous Coleoptera of the French entomologists. (Trans. Linn. Soc. London, xv, 1825, pp. 63–73.)
Speyer, O. Untersuchung der Beine der Schmetterlinge. (Isis, 1843, pp. 161–207, 243–264.)
Pokorsky Joravko, A. von. Quelques remarques sur le dernier article du tarse des Hyménoptères. (Bull. Soc. imp. Natur. Moscou, 1844, xvii, pp. 140–159. Ref. in Isis, 1848, v, p. 347.)
Rossmassler, E. A. Das Bein der Insekten. (Aus der Heimath, 1860, 3 kap., pp. 327–334, Fig.)
West, Tuffen. The foot of the fly; its structure and action; elucidated by comparison with the feet of other insects, etc. Part I. (Trans. Linn. Soc. London, xxiii, 1861, pp. 393–421, 1 Pl.)
Sundevall, C. On insektenas extremiteter samt deras hufoud och munddelar. (Kongl. Vetenskaps Akad. Handlingar. iii, Nr. 9, 1861.)
Lindemann, C. Notizen zur Lehre vom ausseren Skelete der Insekten (Gelenke und Muskeln der Füsse). 1 Taf. (Bull. Soc. imp. d. Natur. Moscou, xxxvii, 1864, pp. 426–432.)
Liebe, O. Die Gelenke der Insekten. Chemnitz, 1873. 4º. 1 Taf.
Canestrini, J. Ueber ein sonderbares Organ der Hymenopteren. (Zool. Anzeiger, 1880, pp. 421, 422.)
Dahl, F. Beiträge zur Kenntnis des Baues und der Funktionen der Insektenbeine. (Archiv f. Naturgesch. 1 Jahrg., 1884, pp. 146–193, 3 Taf. Sep., 48 pp. Vorlauf. Mitteil, in Zool. Anz., 1884, pp. 38–41.)
Langer, K. Ueber den Gelenkbau bei den Arthrozoen. Vierter Beitrag zur vergleichenden Anatomie und Mechanik der Gelenke. (Denkschriften der Akad. d. Wissensch. Wien, xviii, Bd. Physikal.-mathem. Classe, pp. 99–140. 3 Taf.)
Graber, Vitus. Ueber die Mechanik des Insektenkörpers. (Biolog. Centralbl., iv, 1884, pp. 560–570.)
—— Die ausseren mechanischen Werkzeuge der Tiere, ii Teil. Wirbellose Tiere, 1886, pp. 175–182, 208–210.
Dewitz, H. Ueber die Fortbewegung der Tiere an senkrechten glatten Flächen vermittelst eines Sekretes. 3 Taf. (Pflüger’s Archiv f. d. ges. Physiologie, xxxiii, 1884, pp. 440–481.)
Ockler, A. Das Krallenglied am Insektenfuss. (Archiv f. Naturgesch., 1890, pp. 221–262, 2 Taf.)
LITERATURE OF LOCOMOTION (WALKING, ETC.)
Carlet, G. Sur le mode de locomotion des chenilles. (Compt, rend. Acad. Paris, 1888, cvii, pp. 131–134. Naturwiss. Rundschau, iii Jahrg., 1888, No. 42, p. 543.)
—— De la marche d’un insecte rendu tetrapode par la suppression d’une paire de pattes. (Ibid., pp. 565, 566.)
—— Sur la locomotion des insectes et des arachnides. (Ibid., 1879, T. 89, pp. 1124, 1125.)
—— Ueber den Gang eines vierfüssig gemachten Insekts. (Naturwiss. Rundschau, viii Jahrg., 1888, pp. 666–667; Compt. rend. 1888, cvii.)
119Demoor, J. Recherches sur la marche des insectes et des arachnides. Étude experimentale d’Anatomie et de Physiologie comparées. (Archiv de Biologie, Liège, 1880, 42 pp. 3 Pls.)
—— Ueber das Gehen der Arthropoden mit Berücksichtigung der Schwankungen des Körpers. (Compt. rend. Acad. d. Sc. Paris, 1890, cxi, pp. 839–840.)
Osten-Sacken, C. R. von. Ueber das Betragen des kalifornischen flügellosen Bittacus (apterus McLachl.). (Wiener Ent. Zeit., 1882, pp. 123.)
Dixon, H. H. Preliminary note on the walking of some of the Arthropoda. (Proc. R. Dublin Soc. vii, pp. 574–578, 1892. Also Nature, 1897.)
Also the works of Graber, Marey, Cheshire, etc.
LITERATURE OF WALKING ON SMOOTH SURFACES
Blackwell, J. Remarks on the pulvilli of insects. (Trans. Linn. Soc. London, xvi, 1831, pp. 487–492, 767–770.)
Lowne, B. T. On the so-called suckers of Dytiscus and the pulvilli of insects. (Trans. Roy. Micr. Soc., pp. 267–271, 1871, 1 Pl.)
West, Tuffen. On certain appendages to the feet of insects subservient to holding or climbing. (Journ. of the Proceed. Linn. Soc. London, Zoölogy, vi, 1862, pp. 26–88.)
Dewitz, H. Ueber die Fortbewegung der Tiere an senkrechten, glatten Flächen vermittelst eines Sekrets. (Pflüger’s Archiv f. d. ges. Physiologie, xxxiii, 1884, pp. 440–481. 3 Taf. Also Zool. Anzeiger, 1884, pp. 400–405.)
—— Wie ist es den Stubenfliegen und anderen Insekten möglich, an senkrechten Glaswanden emporzulaufen. (Sitzungsb. Ges. naturf. Freunde zu Berlin, 1882, pp. 5–7.)
—— Weitere Mitteilungen über den Klettern der Insekten (Ibid., 1882, pp. 109–113).
—— Die Befestigung durch einen klebenden schleim beim springen gegen senkrechte Flächen. (Zool. Anzeiger, 1883, pp. 273, 274.)
—— Ueber die Wirkung der Haftlappchen toter Fliegen. (Ent. Nachr., x Jahrg., 1884, pp. 286, 287.)
—— Weitere Mitteilungen über das Klettern der Insekten an glatten senkrechten Flächen. (Zoolog. Anzeiger, 1885. viii Jahrg., pp. 157–159.)
—— Richtigstellung der behauptungen des Herrn F. Dahl. (Archiv f. mikroskop. Anat., 1885, xxvi, pp. 125–128.)
Rombouts, J. E. Ueber die Fortbewegung der Fliegen an glatten Flächen. (Zool. Anzeiger, 1884, pp. 619–623.)
—— De la faculté qu’out les mouches de se mouvoir sur le verre et sur les autres corps polis. (Archiv Museum Teyler (2), 4 Part, pp. 16. Fig.)
Simmermacher, G. Untersuchungen über Haftapparate an Tarsalgliedern von Insekten. (Zeitschr. f. wissensch. Zool. xl, 1884, pp. 481–556. 3 Taf., 2 Figs. Also Zoolog. Anzeiger, vii Jahrg., 1884, pp. 225–228.)
—— Antwort an Herrn Dr. H. Dewitz. (Ibid., pp. 513–517.)
Dahl, F. Die Fussdrüsen der Insekten. (Archiv f. mikroskop. Anat., 1885, xxv, pp. 236–263. 2 Taf. See also p. 118.)
Emery, C. Fortbewegung von Tieren an senkrechten und überhangenden glatten Flächen. (Biolog. Centralbl., 1884, 4 Bd., pp. 438–443.)
Léon, N. Disposition anatomique des organes de succion chez les Hydrocores et les Géocores. (Bull. Soc. des Medec. et Natur, de Jassy., 1888.)
d. The wings and their structure
The insects differ from all other animals except birds in possessing wings, and as we at the outset have claimed, it is evidently owing to them that insects are numerically so superior to any other class of animals, since their power of flight enables them to live in the air out of reach of many of their enemies, the greatest destruction to insect life occurring in the wingless larval and pupal stages.
The presence of wings has exerted a profound influence on the shape and structure of the body, and it is apparently due to their existence that the body is so distinctly triregional, since this feature is least marked in the synapterous insects. The wings are thin, broad leaf-like folds of the integument, attached to the thorax and moved by powerful muscles which occupy the greater part of the thoracic cavity. The two pairs of wings are outgrowths of the middle and hinder part of the thorax, the anterior pair being attached to the mesothoracic and the hinder pair to the metathoracic segment. The larger pair is developed from the middle segment of the thorax. The differentiation of the tergites into scutum, scutellum, etc., is the result of the appearance of wings, because these sclerites are more or less reduced or effaced in wingless insects, such as apterous Orthoptera and moths, ants, etc.
The size of the hinder thoracic segments is closely related to that of the wings they bear. In those Orthoptera which have hind wings larger than those of the fore pair, the metathorax is larger than the mesothorax. In such Neuroptera as have the hind wings nearly or quite as large as the anterior pair, or in the Trichoptera and in the Hepialidæ, the metathorax is nearly as large as the mesothorax, while in Coleoptera the metathorax is as large and often much larger. In the Ephemeridæ, Diptera, and Hymenoptera, which have either only rudimentary (halteres) or small hind wings, the metathorax is correspondingly reduced in size.
The wings morphologically, as their development shows, are simple sac-like outgrowths of the integument, i.e. of the free hinder edge of the tergal plates, their place of origin being apparently above the upper edge of the epimera or pleural sclerites. Calvert[24] however, regards the upper lamina of the wing as tergal, and the lower, pleural.
The wings in most insects are attached to the thorax by a membrane containing several little plates of chitin called by Audouin articulatory epidemes.
121The wings, then, are simple, very thin chitinous lamellate expansions of the integument, which are supported and strengthened by an internal framework of hollow chitinous tubes.
The veins.—The so-called “veins” or “nervures,” which are situated between the upper and under layers of the wing are so disposed as to give the greatest lightness and strength to the wings. Hagen has shown that in the freshly formed wings these two layers can be separated, when it can be seen that the veins pass through each layer.
These veins are in reality quite complex, consisting of a minute central trachea enclosed within a larger tube which at the instant the insect emerges from the nymph, or pupa, as the case may be, is filled with blood (Fig. 136). Since these tubes at first contain blood, which has been observed to circulate through them, and since the heart can be most easily injected through them, they may more properly be called veins than nervures. The shape and venation of the wings afford excellent ordinal as well as family and generic characters, while they also enable the systematist to exactly locate the spots and other markings of the wings. The spaces enclosed by the veins and their cross-branches are called cells, and their shape often affords valuable generic and specific characters.
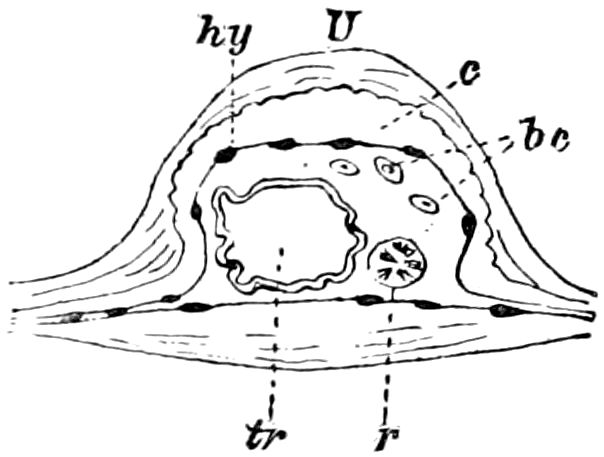
Fig. 136.—Cross-section of wing of Pronuba.—After Spuler.
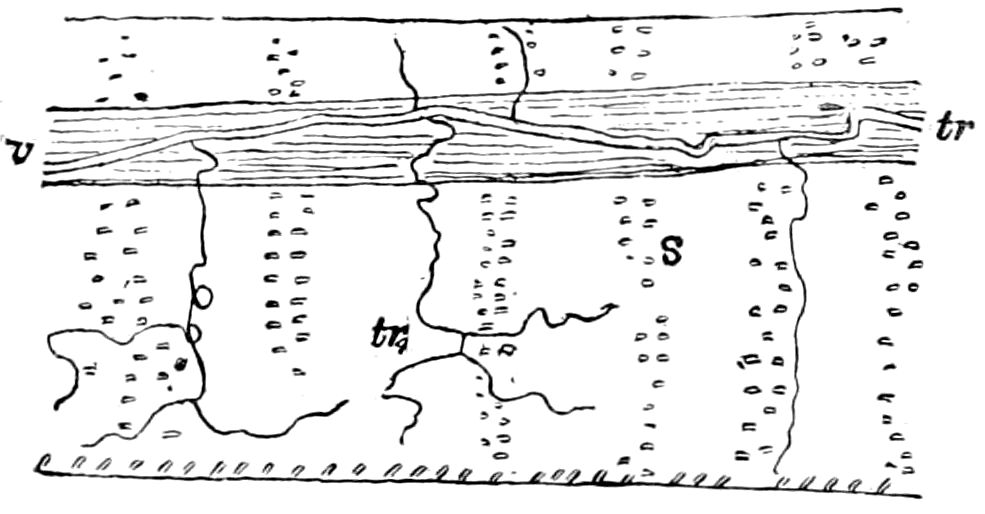
Fig. 137.—Cross-section of wing of Pieris: s, insertions of scales.-After Spuler.
The structure of a complete vein is described by Spuler. In a cross-section of a noctuid moth (Triphæna pronuba, Fig. 136) the chitinous walls are seen to consist of two layers, an outer (U) and inner (c), the latter of which takes a stain and lies next to the hypodermis (hy). In the cavity of the vein is the trachea (tr), which shows more or less distinctly the so-called spiral thread; within the cavity are also Semper’s “rib” (r) and blood-corpuscles (bc), which proves that the blood circulates in the veins of the completely formed wing, though this does not apply to all Lepidoptera with hard mature wings. We have been able to observe the same structure in sections of the wing of Zygæna.
122A cross-section of a vein of Pieris brassicæ shows that the large trachea is first formed, and that it extends along the track between the protoplasmic threads connecting the two hypodermal layers.
The main tracheæ throw off on both sides a number of secondary branches showing at their end a cell with an intracellular tracheal structure; these accessory tracheæ afterwards branch out. The accessory or transverse tracheæ often disappear, though in some moths they remain permanently. Fig. 137 tr2 represents these secondary veins in the edge of the fore wing of Laverna vanella, arising from a main trachea (tr) passing through vein I (v), two of the twigs extending to the centre, showing that the latter has no homology with a true vein. Only rarely and in strongly developed thick folds are the transverse tracheæ provided with a chitinous thickening, as for example in Cossus ligniperda. Since from such accessory tracheæ the transverse veins in lepidopterous wings are developed, we can recognize in them the homologies of the net-veins in reticulated venations. There is no sharply defined difference between reticulated and non-reticulated venations; no genetic difference exists between the two kinds of venation, since there occur true Blattidæ both with and without a reticulated venation (Spuler).
In the fore wings of Odonata, Psocina, Mantispidæ, and most Hymenoptera is an usually opaque colored area between the costal edge and the median vein, called the pterostigma.
In shape the wings are either triangular or linear oval, and at the front edge the main veins are closer together than elsewhere, thus strengthening the wings and affording the greatest resistance to the air in making the downward stroke during flight. It is noticeable that when the veins are in part aborted from partial disuse of the wings, they disappear first from the hinder and middle edge, those on the costal region persisting. This is seen in the wings of Embiidæ (Oligotoma), Cynipidæ, Proctotrupidæ, Chalcids, ants, etc.
The front edge of the wing is called the costal, its termination in the outer angle of the wing is called the apex; the outer edge (termen) is situated between the apex and the inner or anal angle, between which and the base of the wing is the inner or internal edge.
While in Orthoptera, dragon-flies, Termitidæ, and Neuroptera the wings are not attached to each other, in many Lepidoptera they are loosely connected by the loop and frenulum, or in Hymenoptera by a series of strong hooks. These hooks are arranged, says Newport, “in a slightly twisted or spiral direction along the margin of the wing, so as to resemble a screw, and when the wings are expanded attach themselves to a little fold on the posterior margin 123of the anterior wing, along which they play very freely when the wings are in motion, slipping to and fro like the rings on the rod of a window curtain.”
At the base of the hind wings of Trichoptera and in the lepidopterous Micropteryx there is an angular fold (jugum) at the base of each wing (Fig. 138); that of the anterior wings is retained in Eriocephala and Hepialidæ.
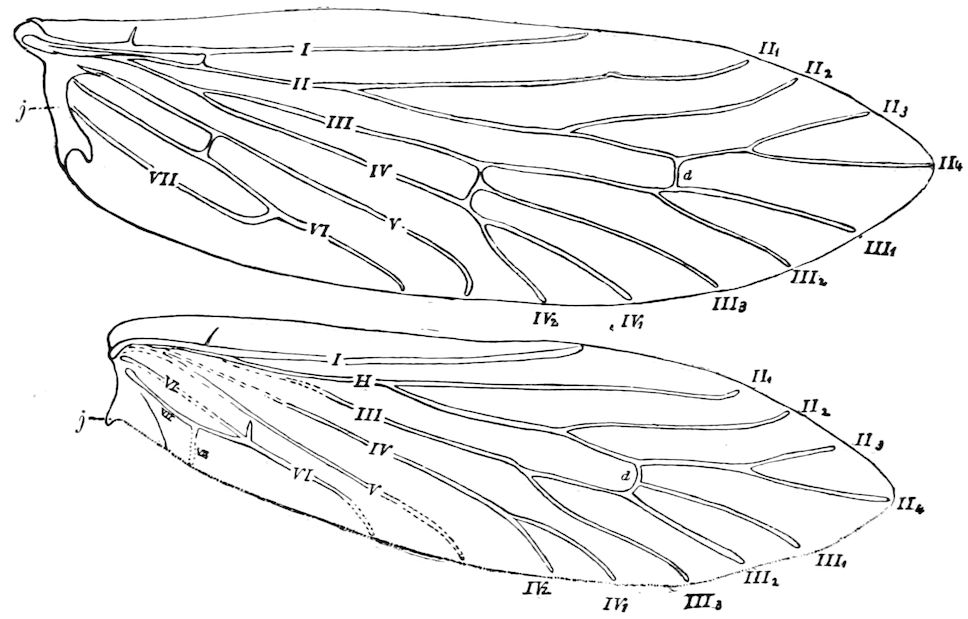
Fig. 138.—Venation of fore and hind wings of Micropteryx purpurella: j, jugum, on each wing; d, discal vein; the Roman numerals indicate veins I.-VIII. and their branches.
In the wings of Orthoptera as well as other insects, the fore wings, especially, are divided into three well-marked areas, the costal, median, and internal; of these the median area is the largest, and in grasshoppers and crickets is more or less modified to form the musical apparatus, consisting of the drum-like resonant area, with the file or bow.
The squamæ.—In the calyptrate Muscidæ, a large scale-like membranous broad orbicular whitish process is situated beneath the base of the wing, above the halter; (Fig. 94, 10 sq.) it is either small or wanting in the acalyptrate muscids. Kirby and Spence state that when the insect is at rest the two divisions of this double lobe are folded over each other, but are extended during flight. Their exact use is unknown. Kolbe, following other German authors, considers the term squama as applicable to the whole structure, restricting the term alula to the other lobe-like division.
124More recently (1890 and 1897) Osten-Sacken recommends “squamæ; in the plural, as a designation for both of these organs taken together; squama, in the singular, would mean the posterior squama alone, and antisquama the anterior squama alone;” the strip of membrane running in some cases between them, or connecting the squama with the scutellum, should be called the post-alar membrane. By a mistake Loew, and others following him, used the word tegula for squama, but this term should be restricted to the sclerite of the mesothorax previously so designated (Fig. 90, A, t). The squama or its two subdivisions has also by various authors been termed alula, calypta, squamula, lobulus, axillary lobe, aileron, cuilleron, schuppen, and scale. (Berlin Ent. Zeitschrift, xli, 1896, pp. 285–288, 328, 338.)
The halteres.—In the Diptera the hind wings are modified to form the halteres or balancers, which are present in all the species, even in Nycteribia, but are absent in Braula.
Meinert finds structures in the Lepidoptera which he considers as the homologues of the halteres of Diptera. “In the Noctuidæ,” he remarks, “I find arising from the fourth thoracic segment (segment médiaire), but covered by hair, an organ like the halter of Diptera.” (Ent. Tidskrift., i, 1880, p. 168.) He gives no details.
In the Stylopidæ, on the contrary, the fore wings are reduced to little narrow pads, while the hind wings are of great size.
The thyridium is a whitish spot marking a break in the cubital vein of the fore wing of Trichoptera; these minute thyridia occur in the fore wings of the saw-flies; there is also an intercostal thyridium on the costal part of the wings of Dermaptera.
The fore wings of Orthoptera are thicker than the hinder ones, and serve to protect the hind-body when the wings are folded; they are sometimes called tegmina. It is noteworthy, that, according to Scudder, in all the paleozoic cockroaches the fore wings (tegmina) were as distinctly veined as the hinder pair, “and could not in any sense be called coriaceous.” (Pretertiary Insects of N. A., p. 39.) Scudder also observes that in the paleozoic insects as a rule the fore and hind wings were similar in shape and venation, “heterogeneity making its appearance in mesozoic times.” In the heteropterous Hemiptera, also, the basal half of the fore wings is thick and coriaceous or parchment-like, and also protects the body when they are folded; these wings are called hemelytra. In the Dermaptera the small short fore wings are thickened and elytriform.
The elytra.—This thickening of the fore wings is carried out to its fullest extent in the fore wings of beetles, where they form the sheaths, shards, or elytra, under which the hind wings are folded. The indexed costal edge is called the epipleurum, being wide in the Tenebrionidæ. During flight “the elytra are opened so as to form an 125angle with the body and admit of the free play of the wings” (Kirby and Spence). In the running beetles (Carabidæ), also in the weevils and in many Ptinidae, the hind wings are wanting, through disuse, and often the elytra are firmly united, forming a single hard shell or case. The firmness of the elytra is due both to the thickness of the chitinous deposit and to the presence of minute chitinous rods or pillars connecting the upper and lower chitinous surfaces.
Fig. 139.—Longitudinal section through the edge of the elytrum of Lina ænea: gl, glands; r, reservoir; fb, fat-body; m, matrix; u, upper,—l, lower, lamella.—After Hoffbauer.
Hoffbauer finds that in the elytra of beetles of different families the venation characteristic of the hind wings is wanting, the main tracheæ being irregular or arranged in closely parallel longitudinal lines, and nerve-fibres pass along near them, sense-organs being also present. The fat-bodies in the cavity of the elytra, which is lined with a matrix layer, besides nerves, tracheæ, and blood, contain secretory vesicles filled with uric-acid concretions such as occur in the fat-body of Lampyris. There are also a great many glands varying much in structure and position, such occurring also in the pronotum (Fig. 139).
Meinert considers the elytra of Coleoptera to be the homologues of the tegulæ of Lepidoptera and of Hymenoptera. He also calls attention to the alula observed in Dyticus, situated at the base of the elytra, but which is totally covered by the latter. The alulæ of these beetles he regards as the homologues of the anterior wings of Hymenoptera and Diptera. No details are given in support of these views. (Ent. Tidskrift, i, 1880, p. 168.)
Hoffbauer (1892) also has suggested that the elytra are not the homologues of the fore wings of other insects, but of the tegulæ.
Kolbe describes the alula of Dyticus as a delicate, membranous lobe at the base of the elytra, but not visible when they are closed: its fringed edge in Dyticus is bordered by a thickening forming a tube which contains a fluid. The alula is united with the inner basal portion and articulation of the wing-cover, forming a continuation of them. Dufour considered that the humming noise made by these beetles is produced by the alulets.
Hoffbauer finds no structural resemblances in the alulæ of Dyticus to the elytra. He does not find “the least trace of veins.” They are more like appendages of the elytra. Lacordaire considered that their function is to prevent 126the disarticulation of the elytra, but Hoffbauer thinks that they serve as contrivances to retain the air which the beetle carries down with it under the surface, since he almost always found a bubble of air concealed under it; besides, their folded and fringed edge seems especially fitted for taking in and retaining air. Hoffbauer then describes the tegulæ of the hornet and finds them to be, not as Cholodkowsky states, hard, solid, chitinous plates, but hollow. They are inserted immediately over the base or insertion of the fore wings, being articulated by a hinge-joint, the upper lamella extending into a cavity of the side of the mesothorax, and connected by a hinge-like, articulating membrane with the lower projection of the bag or cavity. The lower lamella becomes thinner towards the place of insertion, is slightly folded, and merges without any articulation into the thin, thoracic wall at a point situated over the insertion of the fore wing. The tegulæ also differ from the wings in having no muscles to move them, the actual movements being of a passive nature, and due to the upward and downward strokes of the wings.
Comstock adopts Meinert’s view that the elytra are not true fore wings, but gives no reasons. (Manual, p. 495.)
Dr. Sharp,[25] however, after examining Dyticus and Cybister, affirms that this structure is only a part of the elytron, to which it is extensively attached, and that it corresponds with the angle at the base of the wing seen in so many insects that fold their front wings against the body. He does not think that the alula affords any support to the view that the elytra of beetles correspond with the tegulæ of Hymenoptera rather than with the fore wings.
That the elytra are modified paraptera (tegulæ) is negatived by the fact that the latter have no muscles, and that the elytra contain tracheæ whose irregular arrangement may be part of the modified degenerate structure of the elytra. Kolbe finds evidences of veins. The question may also be settled by an examination of the structure of the pupal wings. A study of a series of sections of both pairs of wings of the pupa of Doryphora and of a Clytus convinces us that the elytra are the homologues of the fore wings of other insects.
e. Development and mode of origin of the wings
Embryonic development of the wings.—The wings of insects are essentially simple dorsal outgrowths of the integument, being evaginations of the hypodermis. They begin to form in the embryo before hatching, first appearing as folds, buds, or evaginations, of the hypodermis, which lie in pouches, called peripodal cavities. They are not visible externally until rather late in larval life, after the insect, such as a grasshopper, has moulted twice or more times; while in holometabolous insects they are not seen externally until the pupa state is attained.
The subject of their origin is in a less satisfactory state than desirable from the fact that at the outset the development of the wings of the most generalized insects, such as Orthoptera, Termes, etc., was not first examined, that of the most highly modified of any insects, i.e. the Muscidæ, having actually been first studied.
127In the course of his embryological studies on the Muscidæ (Musca comitoria and Sarcophaga carnaria) Weismann (1864) in examining the larvæ of these flies just before pupation, found that the wings, as well as the legs and mouth-appendages, developed from microscopic masses of indifferent cells, which he called “imaginal discs.” From the six imaginal discs or buds in the lower part of the thorax arise the legs, while from four dorsal discs, two in the meso- and two in the metathoracic segment, arise the fore and hind wings (Fig. 141.) These imaginal buds, as we prefer to call these germs, usually appear at the close of embryonic life, being found in freshly hatched larvæ.
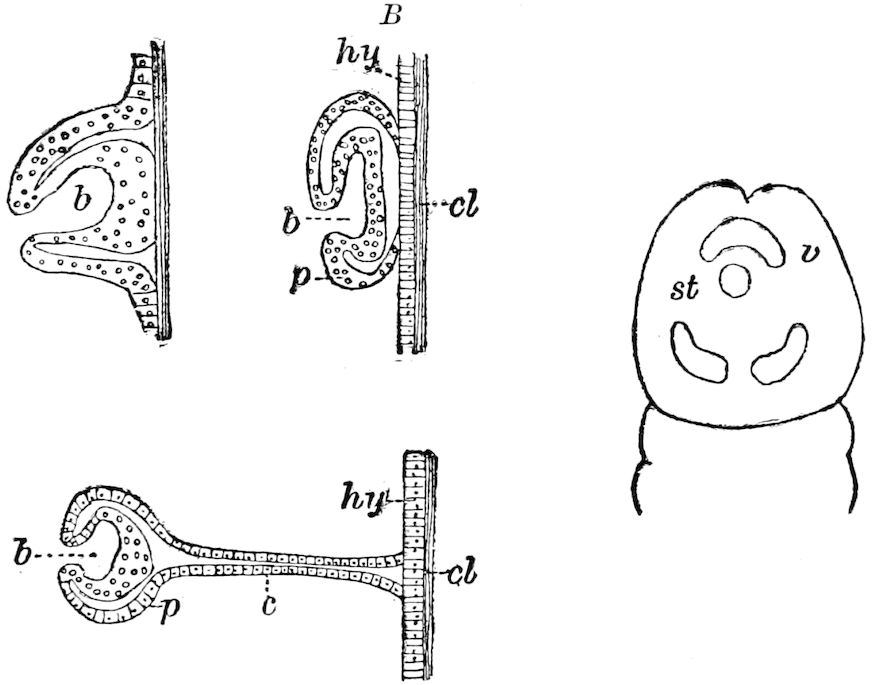
Fig. 140.—Imaginal buds in Musca,—A, in Corethra,—B, in Melophagus,—C, in embryo of Melophagus; dorsal view of the head; b, bud; p, peripodal membrane; c, cord; hy, hypodermis; cl, cuticula; st, stomodæum; v, ventral cephalic, behind are the two dorsal cephalic buds.—After Pratt.
As first observed by Weismann, the buds are, like those of the appendages, simply attached to tracheæ and sometimes to nerves, in the former case appearing as minute folds or swellings of the peritoneal membrane of certain of the tracheæ. In Volucella the imaginal buds were, however, found by Künckel d’Herculais to be in union with the hypodermis. Dewitz detected a delicate thread-like stalk connecting the peripodal membrane with the hypodermis, and Van Rees has since proved in Musca, and Pratt in Melophagus, the connection of the imaginal buds with the hypodermis (Fig. 140). These tracheal enlargements increase in size, and become differentiated into a solid mass which corresponds to the upper part of the mesothorax, while a tongue-shaped continuation becomes the rudiment of the wing. During larval life the rudiments of the wings 128crumple, thus forming a cavity. While the larva is transforming into the pupa, the sheath or peripodal membranes of the rudimentary wings are drawn back, the blood presses in, and thus the wings are everted out of the peripodal cavities.
Due credit, however, should be given to Herold, as the pioneer in these studies, who first described in his excellent work on the development of Pieris brassicæ (1815) the wing-germs in the caterpillar after the third moult. This discovery has been overlooked by recent writers, with the exception of Gonin, whose statement of Herold’s views we have verified. Herold states that the germs of the wings appear on the inside of the second and third thoracic segments, and are recognized by their attachment to the “protoplasmic network” (schleimnetz), which we take to be the hypodermis, the net-like appearance of this structure being due to the cell-walls of the elements of the hypodermal membrane. These germs are, says Herold, also distinguished from the flakes of the fat-body by their regular symmetrical form. Fine tracheæ are attached to the wing-germs, in the same way as to the flakes of the fat-body. It thus appears that Herold in a vague way attributes the origin of these wing-germs, and also the germs of the leg, to the hypodermis, since his schleimnetz is the membrane which builds up the new skin. Herold also studied the later development of the wings, and discovered the mode of origin of the veins, and in a vague way traced the origin of the scales and hairs of the body, as well as that of the colors of the butterfly.
Herold also says that as the caterpillar grows larger, and also the wing-germs, “the larval skin in the region under which they lie hidden is spotted and swollen,” and he adds in a footnote: “This is the case with all smooth caterpillars marked with bright colors. In dark and hairy caterpillars the swelling of the skin through the growth of the underlying wing-germs is less distinct or not visible at all” (pp. 29, 30).
It should be added that Malpighi, Swammerdam, and also Réaumur had detected the rudiments of the wings in the caterpillar just before pupation under the old larval skin. Lyonet (1760) also describes and figures the four wing-germs situated in the second and third thoracic segments, but was uncertain as to their nature. Each of these masses, he says, is “situated in the fatty body without being united to it, and is attached to the skin in a deep fold which it makes there.” He could throw no certain light on their nature, but says: “their number and situation leads to the supposition that they may be the rudiments of the wings of the moth” (pp. 449, 450).
During the transformation into the pupa the imaginal buds unite and grow out or extend along their edges, while the enveloping membrane disappears. The rudimentary wings are now like little sacs, and soon show a fusion of the two wing-membranes or laminæ with the veins, while the tracheæ disappear, the places occupied by the tracheæ becoming the veins. “Very early, as soon as the scales are indicated, begin in a very peculiar way the fusion of the wing-laminæ. There occur openings in the hypodermis into which the cells extend longitudinally and then laterally give way to each other. Hence no complete opening is found, but the epithelium appears by sections through a straight line sharply bordered along the wingcavity. 129It is a continuous membrane formed of plasma which I will call the ground membrane of the epithelium. Through this ground membrane pass blood-corpuscles as well as blood-lymph.” (Schaeffer.)
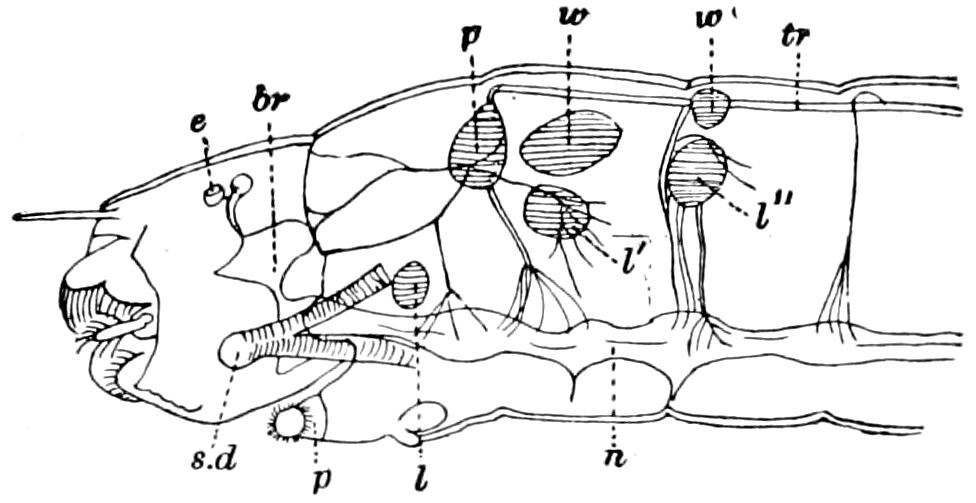
Fig. 141.—Anterior part of young larva of Simulium sericea, showing the thoracic imaginal buds: p, prothoracic bud (only one not embryonic); w, w′, fore and hind wing-buds; l, l′, l″, leg-buds; n, nervous system; br, brain; e, eye; sd, salivary duct; p, prothoracic foot.—After Weismann.
Afterwards (1866) Weismann studied the development of the wings in Corethra plumicornis, which is a much more primitive and generalized form than Musca, and in which the process of development of the wings is much simpler, and, as since discovered, more as in other holometabolous insects. He also examined those of Simulium (Fig. 141).
In Corethra, after the fourth and last larval moulting, there arises at first by evagination and afterwards by invagination a cup-shaped depression on each side in the upper part of the mesothoracic segment within which the rudiment of the wings lies like a plug. The wings without other change simply increase in size until, in the transformation into the pupa by the withdrawal of the hypodermis, the wings project out and become filled with blood, the tracheæ now being wholly wanting, and other tissues being sparingly present.
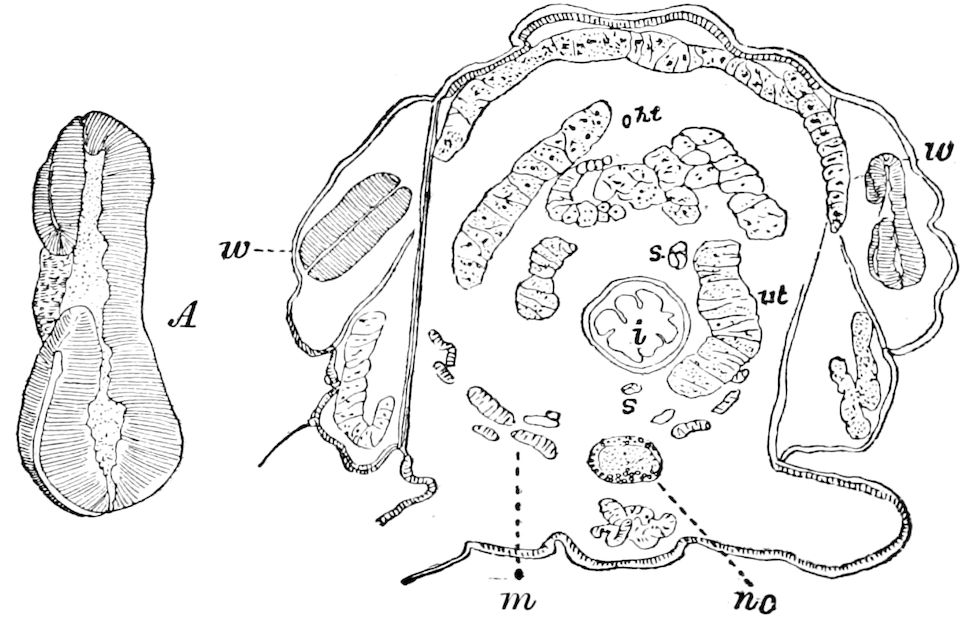
Fig. 142.—Section through thorax of a Tineid larva on sycamore, passing through the 1st pair of wings (w): ht, heart; i, œsophagus; s, salivary gland: ut, urinary tube; nc, nervous cord; m, recti muscles; a part of the fat body overlies the heart. A, right wing-germ enlarged.
130These observations on two widely separate groups of Diptera were confirmed by Landois, and afterwards by Pancritius, for the Lepidoptera, by Ganin for the Hymenoptera, by Dewitz for Hymenoptera (ants) and Trichoptera; also for the Neuroptera by Pancritius. In the ant-lion (Myrmeleon formicarius) Pancritius found no rudiments of the wings in larvæ a year old, but they were detected in the second year of larval life, and do not differ much histologically or in shape from those of Lepidoptera. In the Coleoptera and Hymenoptera the imaginal buds appear rather late in larval life, yet their structure is like that of Lepidoptera. In Cimbex the rudiments of the wings are not found in the young larva, but are seen in the semipupa, which stage lasts over six weeks.
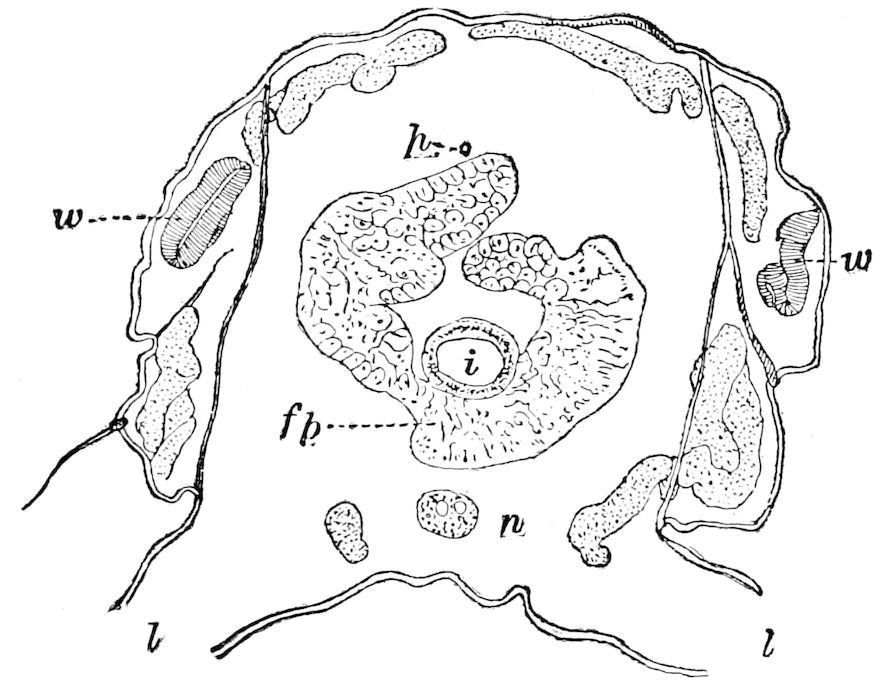
Fig. 143.—Section of the same specimen as in Fig. 142, but cut through the second pair of wings (w): i, mid-intestine; h, heart; fb, fat-body; l, leg; n, nervous cord.
The general relation of the rudiments (imaginal buds) of the wings of a tineid moth to the rest of the body near the end of larval life may be seen in Figs. 142, 143 (Tinea?), the sections not, however, showing their connection with the hypodermis, which has been torn away during the process of cutting. That the wing is but a fold of the hypodermis is well seen in Fig. 144, of Datana, which represents a much later stage of development than in Figs. 142 and 143, the larva just entering on the semipupa stage.
In caterpillars of stage I, 3 to 4 mm. in length, Gonin found the wing-germs as in Fig. 145, A being a thickening of the hypodermis, with the embryonic cells, i.e. of Verson, on the convex border. The two leaves, or sides of the wing, begin to differentiate in stage II (C, D), and in stage III the envelope is formed (E), while the tracheæ begin to proliferate, and the capillary tracheæ or tracheoles at this time arise (Fig. 145, tc). The wall of the principal trachea 131appears to be resolved into filaments, and all the secondary branches assume the appearance of bundles of twine. Landois regarded them as the product of a transformation of the nuclei, but Gonin thinks they arise from the entire cells, stating that from each cell arises a ball (peloton) of small twisted tubes.
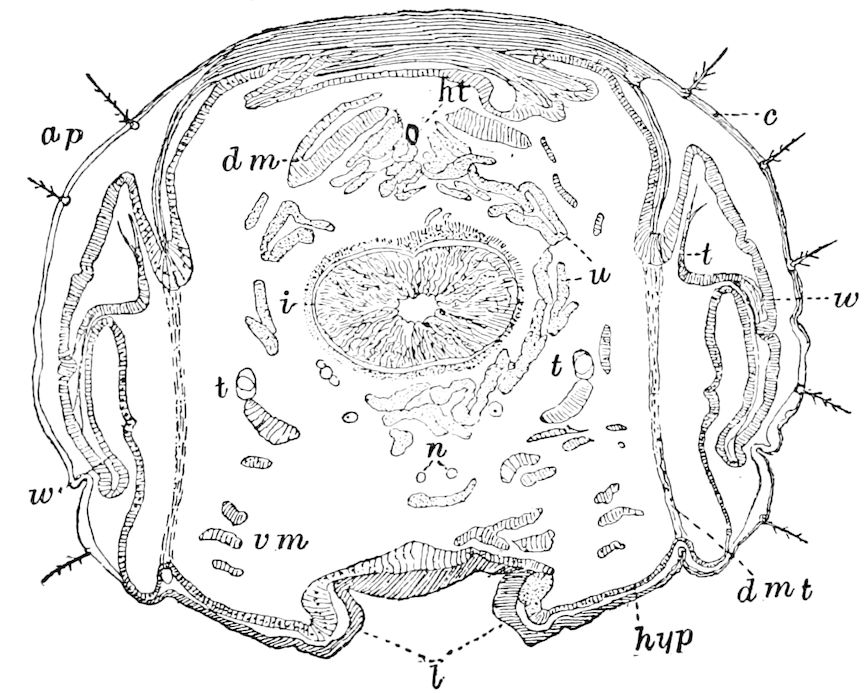
Fig. 144.—Section through mesothoracic segment of Datana ministra, passing through the wings (w): c, cuticula; hyp, hypodermis: ap, apodeme; dm, dorsal longitudinal.—vm, ventral longitudinal. muscles; dmt, depressor muscle of tergum; t, trachea; n, nerve cords; i, intestine; u, urinary tubes; l, insertion of legs.
As the large branches penetrate into the wing, the balls (pelotons) of fine tracheal threads tend to unroll, and each of the new ramifications of the secondary tracheal system is accompanied in its course by a bundle of capillary tubes. This secondary system of wing-tracheæ, then, arises from the mother trachea at the end of the third stage, when we find already formed the chitinous tunic, which will persist through the fourth stage up to pupation. It differs from the tracheoles in not communicating with the air-passage; it possesses no spiral membrane at the origin, and takes no part in respiration.
Gonin thus sums up the nature of the two tracheal systems in the rudimentary wing, which he calls the provisional and permanent systems. “The first, appearing in the second stage of the larva, comprises all the capillary tubes, and arising from numerous branches passes off from the lateral trunk of the thorax before reaching the wing; the second is formed a little later by the direct ramification of the principal branch.
132“These two systems are absolutely independent of each other within the wing. Their existence is simultaneous but not conjoint. One is functionally active after the third moult; the other waits the final transformation before becoming active.”
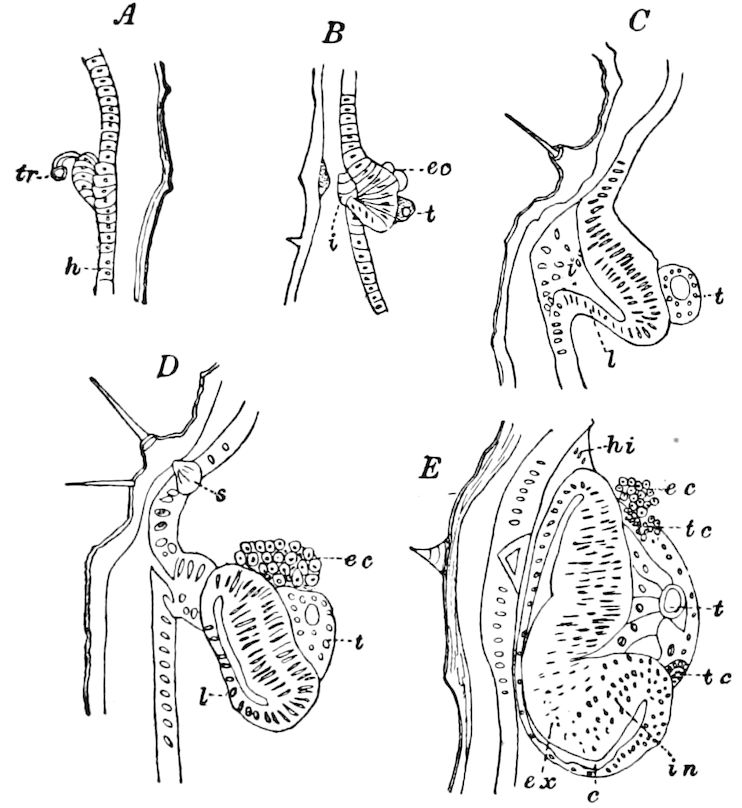
Fig. 145.—A, section of wing-bud of larva of Pieris brassicæ of stage I, in front of the invagination pit. B, section passing through the invagination pit. C, section of same in stage II, through the invagination pit;—D, behind it, making the bud appear independent of the thoracic wall. E, wing-bud at the beginning of the 3d larval stage, section passing almost through the pedicel or hypodermic insertion, the traces of which appear at hi; h, hypodermis; t or tr, trachea; i, opening of invagination; ec, embryonic cells; l, external layer or envelope; in, internal wall of the wing; ex, external wall; s, cell of a tactile hair; tc, capillary tubes; c, cavity of invagination.—After Gonin.
Evagination of the wing outside of the body.—We have seen that the alary germs arise as invaginations of the hypodermis; we will now, with the aid of Gonin’s account, briefly describe, so far as is known, the mode of evagination of the wings. During the fourth and last stage of the caterpillar of Pieris, the wings grow very rapidly, and undergo important changes.
Six or seven days after the last larval moult the chitinous wall is formed, the wing remaining transparent. It grows rapidly and its lower edge extends near the legs. It is now much crumpled on the edge, owing to its rapid growth within the limits of its own segment. Partly from being somewhat retracted, and partly owing to the irregularity of its surface, the wing gradually separates from its envelope, and the cavity of invagination (Fig. 145, c) becomes more like a distinct or real space. The outer opening of the alary sac enlarges quite plainly, though without reaching the level of the edge of the wing.
This condition of things does not still exactly explain how the wing passes to the outside of the body. Gonin compares these conditions to those exhibited by a series of sections of the larva, made forty-eight hours later, on a caterpillar which had just spun its girdle of silk. At this time the wings have become entirely external, but, 133says Gonin, we do not see the why or the how. The partition of the sac has disappeared, and with it the cavity and the leaf of the envelope.
It appears probable that the partition has been destroyed, because the space between the two teguments is strewn with numerous bits, many of which adhere to the chitinous integument, while others are scattered along the edges of the wings, in their folds, or between the wings and the wall of the thorax.
Another series of sections showed that the exit of the fore wings had been accomplished, while the hinder pair was undergoing the process of eversion. In this case the partition showed signs of degeneration: deformation of the nuclei, indistinct cellular limits, pigmentation, granular leucocytes, and fatty globules.
After the destruction of the partition, what remains of the layer of the envelope is destined to make a part of the thoracic wall and undergoes for this purpose a superficial desquamation. The layer of flattened cells is removed and replaced by a firmer epithelium like that covering the other regions. It is this renewed hypodermis which conceals the wing within, serves to separate it from the cavity of the body, and gives the illusion of a complete change in its situation. Other changes occur, all forming a complete regeneration, but which does not accord with the description of Van Rees for the Muscidæ. Finally, Gonin concludes that the débris scattered about the wing comes from the two layers of the partition of the sac, from the flattened hypodermis of the renewed envelope, from the chitinous cuticle of the wing, and from the inner surface of the chitinous integument.
He thinks that the metamorphosis of Pieris is intermediate between the two types of Corethra and of Musca, established by Weismann, as follows:
Corethra.—The wing is formed in a simple depression of the hypodermic wall. No destruction.
Pieris.—The rudiment is concealed in a sac attached to the hypodermis by a short pedicel. Destruction of the partition and its replacement by a part of the thoracic wall by means of the imaginal epithelium.
Musca.—The pedicel is represented by a cord of variable length, whose cavity may be obliterated (Van Rees). The imaginal hypodermis is substituted for the larval hypodermis, which has completely disappeared, either by desquamation (Viallanes), or by histolytic resorption (Van Rees).
Extension of the wing; drawing out of the tracheoles.—When it is disengaged from the cavity, the wing greatly elongates and the creases on its surface are smoothed out; the blood penetrates between the two walls, and the cellular fibres, before relaxed and sinuous, are now firmly extended.
Of the two tracheal systems, the large branches are sinuous, and they are rendered more distinct by the presence of a spiral membrane; but the two tunics are not separated as in the other tracheæ of the thorax; moreover, the mouth choked up with débris does not yet communicate with that of the principal trunk. The bundles of tracheoles on their part form straight lines, as if the folds of the organ had had no influence on them. As they have remained bound together, apart from the chitinous membrane of the tracheal trunk, 134they become drawn out with this membrane, at the time of exuviation, i.e. of pupation, and are drawn out of the neighboring spiracle.
Fig. 146.—Full-grown larva of Pieris brassicæ, opened along the dorsal line: d, digestive canal; s, silk-gland; g, brain; st I, prothoracic stigma; st IV, 1st abdominal stigma; a, a′, germs (buds) of fore and hind wings; p, bud of prothoracic segment;—those of the third pair are concealed under the silk-glands; I–III, thoracic rings.—After Gonin.
“This is a very curious phenomenon, which can be verified experimentally: if we cut off the wing, while sparing the larval integument around the thoracic spiracles, we preserve the two tracheal systems; the same operation performed after complete removal of the larval skin does not give the secondary tracheal system.” (Gonin.) Deceived by the appearance of the tracheoles while still undeveloped, Landois and Pancritius, who have not mentioned the drawing out of the capillaries of the larva, affirm that they are destroyed by resorption in the chrysalis.
135“The study of the tracheæ is closely connected with that of the veins (nervures). It is well to guard against the error of Verson, who mistakes for these last the large tracheal branches of the wing. This confusion is easily explained; it proves that Verson had, with us, recognized that the secondary system is, in the larva, exempt from all respiratory function. Landois thought that the pupal period was the time of formation of the veins. It seems to me probable that they are derived from the sheath of the peritracheal spaces.” (Gonin, pp. 30–33.)
Fig. 147.—Left anterior wing of a larva 3 days before pupation. The posterior part is rolled up: st, prothoracic stigma; tr. i., internal tracheal trunk; tr. e., tr. e.′, external tracheal trunk; p, cavity of a thoracic leg, with the imaginal bud b.—After Gonin.
The appearance of the wing-germs in the fully grown caterpillar, as revealed by simple dissection, is shown at Fig. 146; Fig. 147 136represents a wing of a larva three days before pupation, with the germ of a thoracic leg.
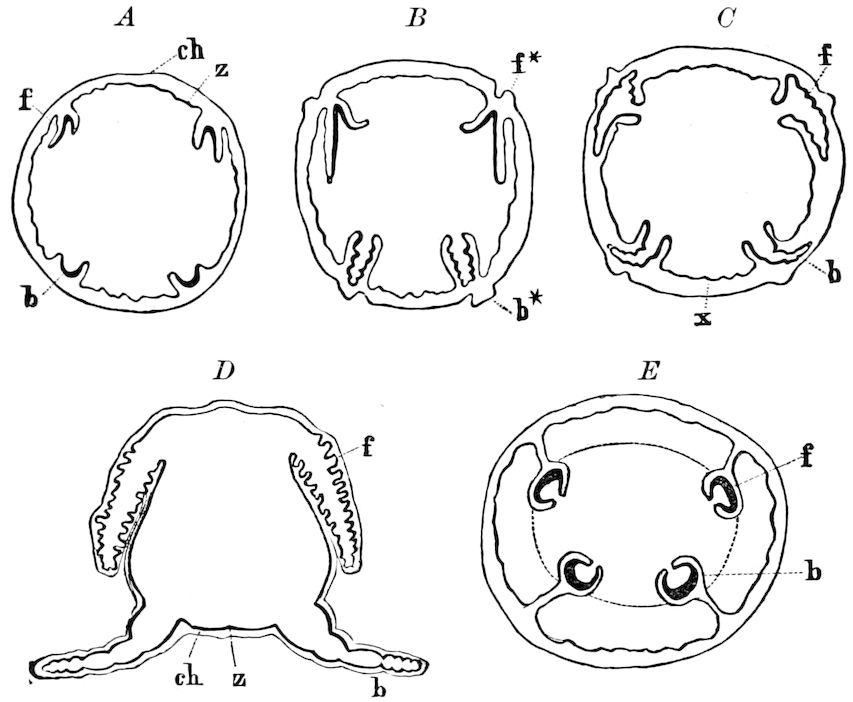
Fig. 148.—Graber’s diagrams for explaining the origin and primary invagination of the hypodermis to form the germs of the leg (b), and wings (f, A-C), and afterwards their evagination D, so that they lie on the outside of the body. E, stage B, showing the hypodermal cavities (f) and stalks connecting the germs with the hypodermis (z).—After Graber.
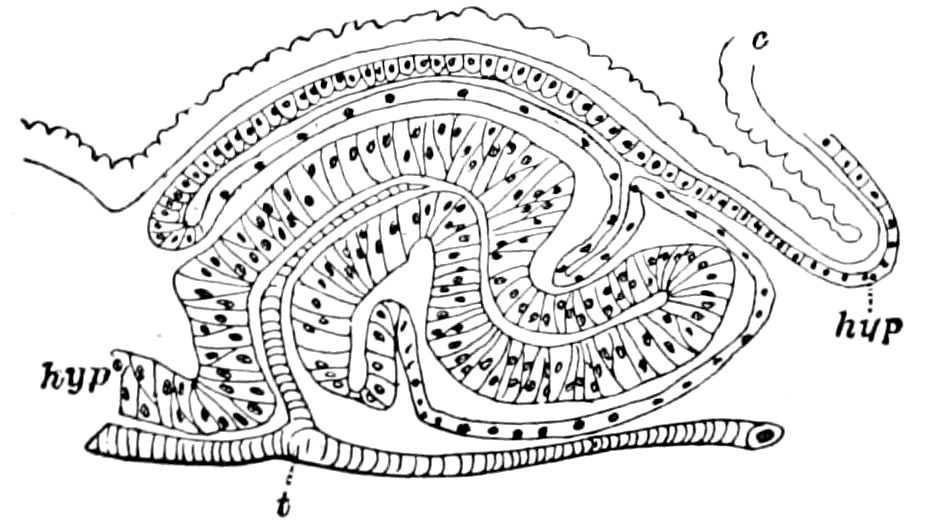
Fig. 149.—Section lengthwise through the left wing of mature larva in Pieris rapæ: t, trachea; hyp, hypodermis; c, cuticula.—After Mayer.
A. G. Mayer has examined the late development of the wings in Pieris rapæ. Fig. 149 represents a frontal section through the left wing of a mature larva and shows the rudiment of the wing, lying in its hypodermal pocket or peripodal cavity. How the trachea passes into the rudimentary wing, and eventually becomes divided into the branches, around which the main veins afterwards form, is seen in Figs. 144, 147, 159.
The histological condition of the wing at this time is represented by Fig. 151, the spindle-like hypodermal cells forming the two walls being separated by the ground-membrane of Semper.
“While in the pupa state,” says Mayer, “the wing-membrane is thrown into a very regular series of closely compressed folds, a single scale being inserted upon the crest of each fold. When the butterfly 137issues from the chrysalis, these folds in the pupal wings flatten out, and it is this flattening which causes the expansion of the wings.... It is evident that the wings after emergence undergo a great stretching and flattening. The mechanics of the operation appears to be as follows. The hæmolymph, or blood, within the wings is under considerable pressure, and this pressure would naturally tend to enlarge the freshly emerged wing into a balloon-shaped bag; but the hypodermal fibres (h) hold the upper and lower walls of the wing-membrane closely together, and so, instead of becoming a swollen bag, the wing becomes a thin flat one. And thus it is that the little thick corrugated sac-like wings of the freshly emerged insect become the large, thin, flat wings of the imago.... The area of the wing of the imago of Danais plexippus is 8.6 times that of the pupa. Now, as the wing of the young pupa has about 60 times the area of the wing in the mature larva, it is evident that in passing from the larval state to maturity the area of the wings increases more than 500 times.”
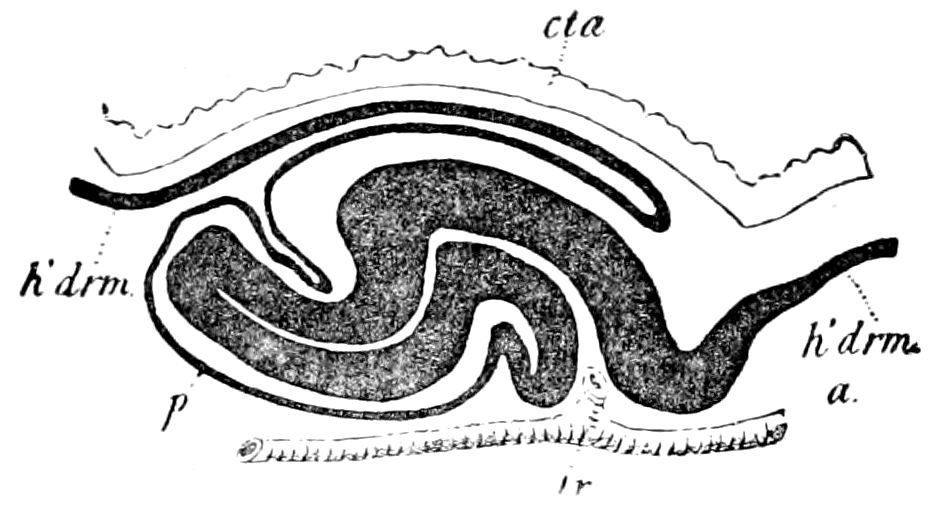
Fig. 150.—Diagrammatic reproduction of Fig. 149 showing the wing-germ in its peripodal cavity (p): h’drm, hypodermis; tr, trachea; cta, cuticula; a, anterior end.—After Mayer.
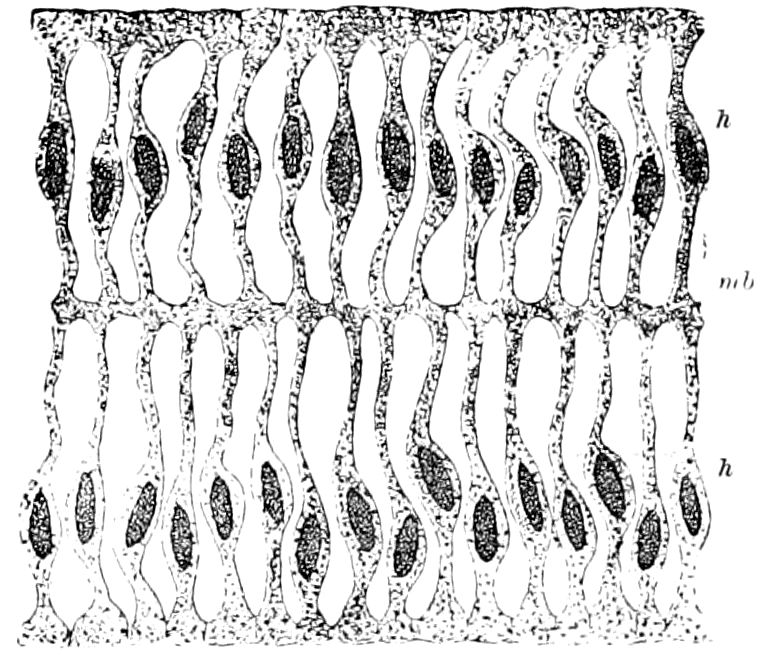
Fig. 151.—Section of the wing-germ, the upper and lower sides connected by spindle-like hypodermic cells (h), forming the rods of the adult wing; mbr, ground-membrane of Semper.—After Mayer.
f. The primitive origin of the wings
Farther observations are needed to connect the mode of formation of the wings in the holometabolous insects with the more primitive mode of origin seen in the hemimetabolous orders, but the former mode is evidently inherited from the latter. Pancritius remarks that the development of the rudiments of the wing in a hypodermal cavity is in the holometabolic insects to be regarded as a later inherited character, the external conditions causing it being unknown.
138Fritz Müller was the first to investigate the mode of development of the wings of the hemimetabolic insects, examining the young nymphs of Termites. He regards the wings as evaginations of the hypodermis, which externally appear as thoracic scale-like projections, into which enter rather late in nymphal life tracheæ which correspond to the veins which afterward arise.
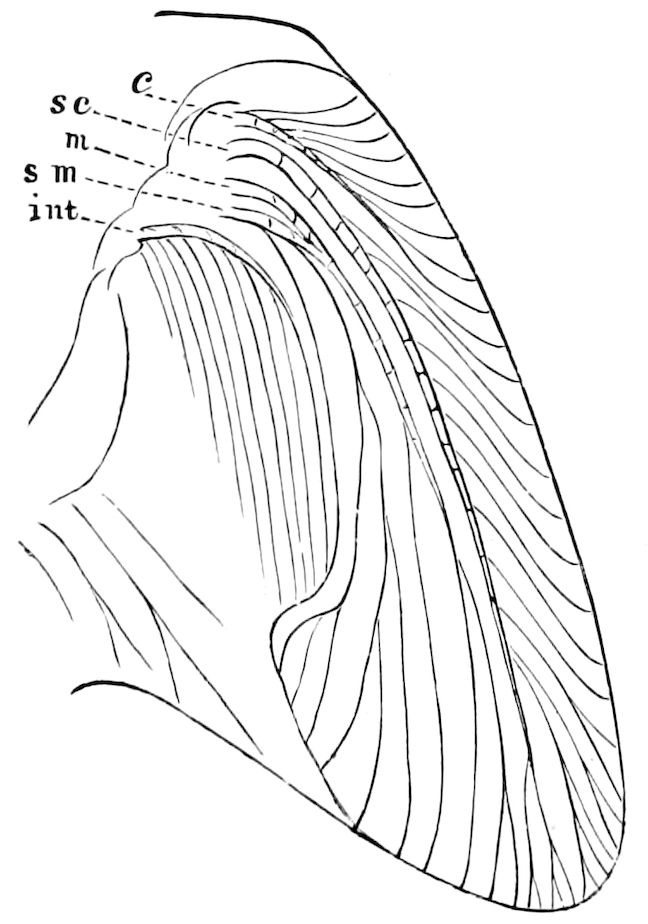
Fig. 152.—Rudimentary wing of young nymph of Blatta, with the five principal veins developed.
The primitive mode of origin of the wings may, therefore, be best understood by observing the early stages of those insects, such as the Orthoptera and Hemiptera, which have an incomplete metamorphosis. If the student will examine the nymphs of any locust in their successive stages, he will see that the wings arise as simple expansions downward and backward of the lateral edges of the meso- and metanotum. In the second nymphal stage this change begins to take place, but it does not become marked until the succeeding stage, when the indications of veins begin to appear, and the lobe-like expansion of the notum is plainly enough a rudimentary wing.
Graber[26] thus describes the mode of development of the wings in the nymph of the cockroach:
“If one is looking only at the exterior of the process, he will perceive sooner or later on the sides of the meso- and metathorax pouch-like sacs, which increase in extent with the dorsal integument and at the same time are more and more separated from the body. These wing-covers either keep the same position as in the flat-bodied Blattidæ, or in insects with bodies more compressed the first rudiments hang down over the sides of the thorax. As soon as they have exceeded a certain length, these wing-covers are laid over on the back. However, if we study the process of development of the wings with a microscope, by means of sections made obliquely through the thorax, the process appears still more simple. The chief force of all evolution is and remains the power of growth in a definite direction. In regard to the skin this growth is possible in insects only in this way; namely, that the outer layer of cells is increased by the folds which are forced into the superficial chitinous skin. These folds naturally grow from one moult to another in proportion to the multiplication of the cells, and are not smoothed out until after the moulting, when the outer resistance is overcome.
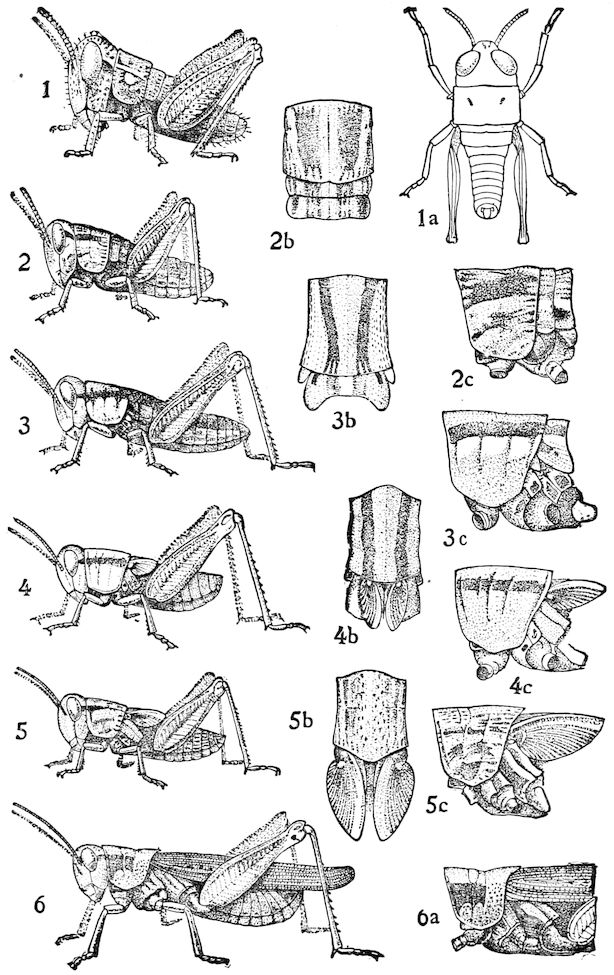
Fig. 153.—Partial metamorphosis of Melanoplus femur-rubrum, showing the five nymph stages, and the gradual growth of the wings, which are first visible externally in 3, 3b, 3c.—Emerton del.
“As, however, the first wing-layers depend upon the wrinkling of the general integument of the body through the increase in the upper layer, the further growth of the wings depends in the later stages upon the wrinkling of the epidermis of the wing-membrane even, which fact we also observe under the microscope when the new wings drawn forth from the old covers appear at 140first to be quite creased together. These wing-like wrinkles in the skin are not empty pouches, but contain tissues and organs within, which are connected with the skin, as the fat of the body, the network of tracheæ, muscles, etc. Alongside the tracheæ, running through the former wing-pouches and accompanied by the nerves, there are canals through which the blood flows in and out.
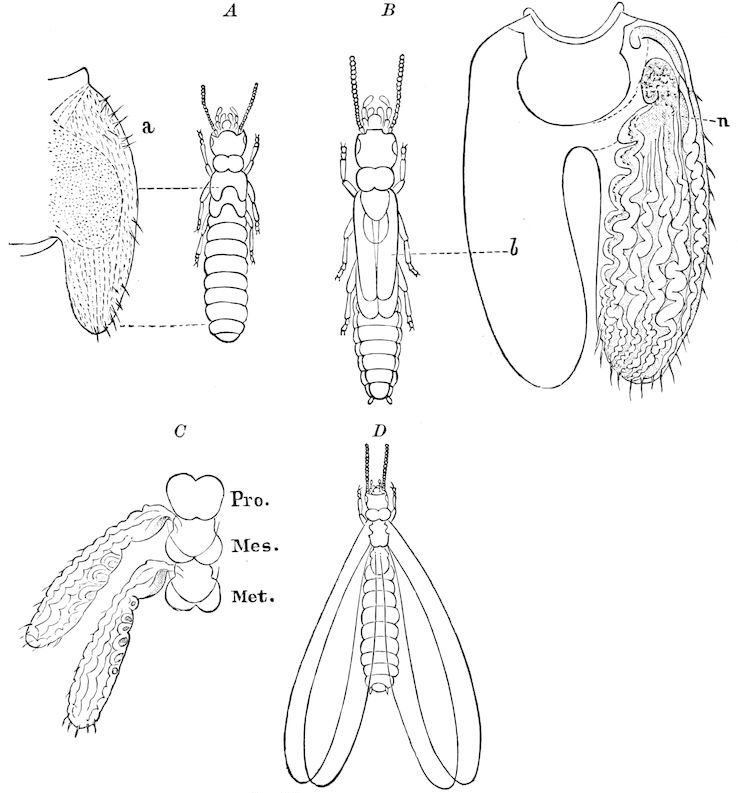
Fig. 154.—Stages in the growth of the wings of the nymph of Termes flavipes: A, young; a, a wing enlarged. B, older nymph; b, fore wing; n, a vein. C, wings more advanced;—D, mature.
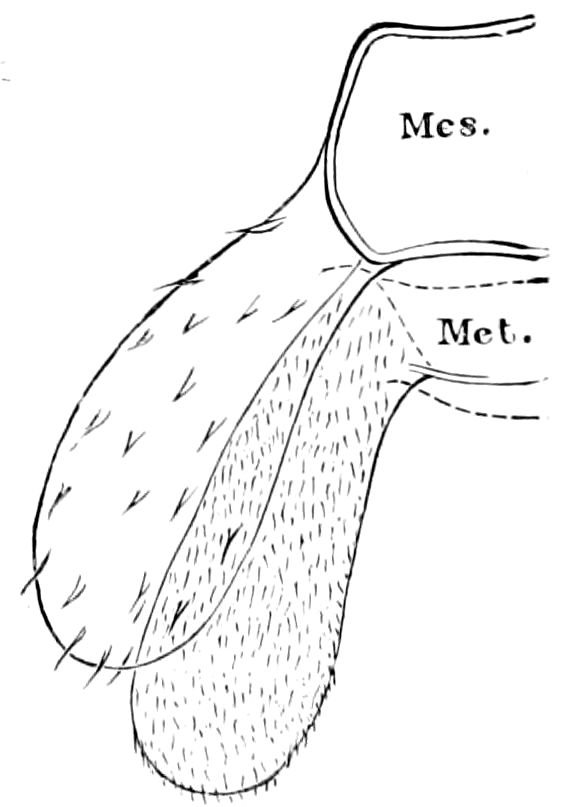
Fig. 155.—Wings of nymph of Psocus.
“After the last moult, however, when the supply of moisture is very much reduced in the wing-pouches, which are contracted at the bottom, their two layers become closely united, and afterward grow into one single, solid wing-membrane.
“These thick-walled blood-tubes arising above and beneath the upper and lower membrane of the wing are the veins of the wings; the development of 141the creased wings in the pupa of butterflies is exactly like that of cockroaches and bugs. The difference is only that the folds of integument furnishing the wings with an ample store of material for their construction reach in a relatively shorter time, that is the space of time between two moults, the same extent that they would otherwise attain only in the course of several periods of growth in the ametabolous insects.”
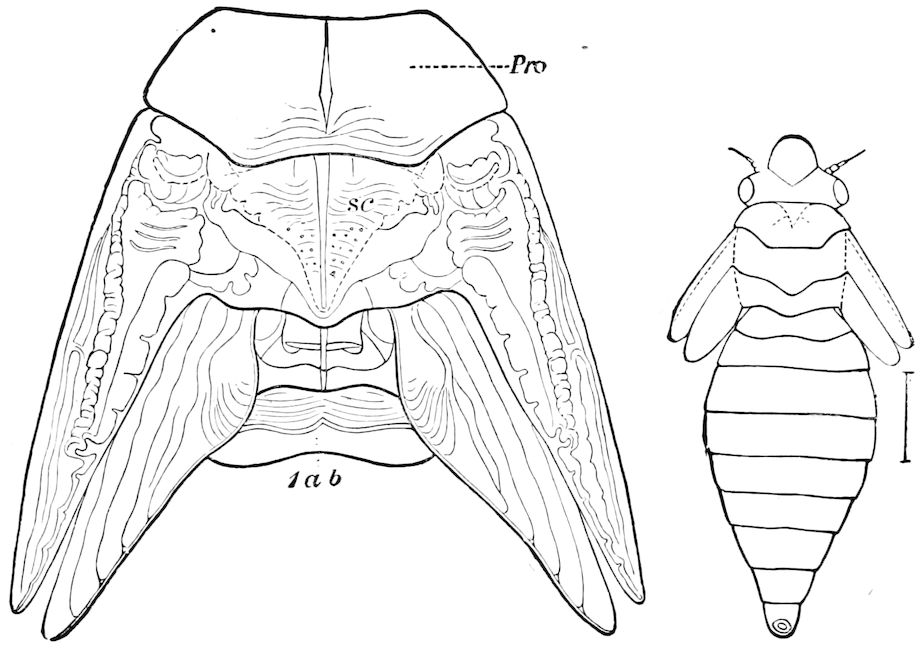
Fig. 156.—Nymph of Aphrophora permutata, with enlarged view of the wings and the veins: pro, pronotum; sc, mesoscutum; 1ab, 1st abdominal segment.
Ignorant of Graber’s paper, we had arrived at the same result, after an examination of the early nymph-stages of the cockroach, as well as the locusts, Termites, and various Hemiptera. In all these forms it is plainly to be seen that the wings are simply expansions, either horizontal or partly vertical (where, as in locusts, etc., the body is compressed, and the meso- and metanota are rounded downwards), of the hinder and outer edge of the meso- and metanotum. As will be seen by reference to the accompanying figures, the wings are notal (tergal) outgrowths from the dorsal arch of the two hinder segments of the thorax. At first, as seen in the young pupal cockroach (Fig. 152) and locust (Fig. 153, also Figs. 154 and 156) the rudiments of the wings are continuous with the notum. Late in nymphal life a suture and a hinge-joint appear at the base of the wing, and thus there is some movement of the wing upon the notum; finally, the tracheæ are well developed in the wings, and numerous small sclerites are differentiated at the base of the wing, to which the 142special muscles of flight are attached, and thus the wings, after the last nymphal moult, have the power of flapping, and of sustaining the insect in the air; they thus become true organs of flight.
It is to be observed, then, that the wings in all hemimetabolous insects are outgrowths from the notum, and not from the flanks or pleurum of the thorax. There is, then, no structure in any other part of the body with which they are homologous.
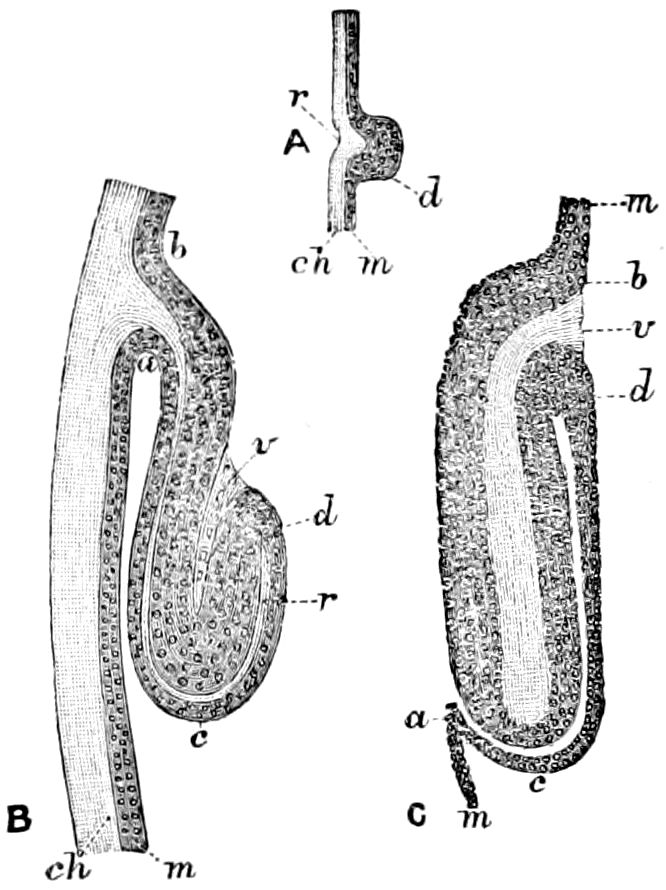
Fig. 157.—Development of wings of Trichoptera: A, portion of body-wall of young larva of Trichostegia; ch, cuticula, forming at r a projection into the hypodermis, m; r, and d, forming thus the first rudiment of the wing. B, the parts in a larva of nearly full size; a, c, d, b, the well-developed hypodermis of the wing-germ separated into two parts by r, the penetrating extension of the cuticula; v, mesoderm, C, wing-pad of another Phryganeid freed from its case at its change to the pupa: b, d, outer layer of the hypodermis (m) of the body-wall; v, inner layer within nuclei.—After Dewitz, from Sharp.
The same may be said of the true Neuroptera, Trichoptera (Fig. 157), the Coleoptera, and the Diptera, Lepidoptera, and Hymenoptera. As we have observed in the house fly,[27] the wings are evidently outgrowths of the meso- and metanotum; we have also observed this to be most probably the case in the Lepidoptera, from observations on a Tortrix in different stages of metamorphosis. It is also the case with the Hymenoptera, as we have observed in bees and wasps;[28] and in these forms, and probably all Hymenoptera, the wings are outgrowths of the scutal region of the notum.
With these facts before us we may speculate as to the probable origin of the wings of insects. The views held by some are those of Gegenbaur, also adopted by Lubbock, and originally by myself.[29] According to Gegenbaur:
“The wings must be regarded as homologous with the lamellar tracheal gills, for they do not only agree with them in origin, but also in their connection with the body, and in structure. In being limited to the second and third thoracic segments they point to a reduction in the number of the tracheal gills. It is quite clear that we must suppose that the wings did not arise as such, but were developed from organs which had another function, such as the tracheal gills; I mean to say that such a supposition is necessary, for we cannot imagine that the wings functioned as such in the lower stages of their development, and that they could have been developed by having such a function.”
Fig. 158.—Changes in external form of the young larva of Calotermes rugosus, showing, in A and B, the mode of origin of the wing-pads: A, newly hatched, with 9 antennal joints, × 8. B, older larva, with 10 joints, × 8. C, next stage, with 11 joints, × 8. D, larva, with twelve joints; the position of the parts of the alimentary canal are shown: v, crop; m, stomach; b, “paunch”; e, intestine; r, heart, × 16
3.—After Fritz Müller, from Sharp.
If we examine the tracheal gills of the smaller dragon-fly (Agrion), or the May-flies, or Sialidæ, or Perlidæ, or Phryganeidæ, we see that they are developed in a very arbitrary way, either at the end of the abdomen, or on the sternum, or from the pleurum; moreover, in structure they invariably have but a single trachea, from which minute twigs branch out;[30] in the wings there are five or six main tracheæ, which give rise to the veins. Thus, in themselves, irrespective of their position, they are not the homologues of the gills. The latter are only developed in the aquatic representatives of the Neuroptera and Pseudoneuroptera, and are evidently adaptive, secondary, temporary organs, and are in no sense ancestral, primitive structures from which the wings were developed. There is no good reason to suppose that the aquatic Odonata or Ephemerids or Neuroptera were not descendants of terrestrial forms.
To these results we had arrived by a review of the above-mentioned facts, before meeting with Fritz Müller’s opinions, derived from a study of the development of the wings of Calotermes (Fig. 158). Müller[31] states that “(1) The wings of insects have not originated from ‘tracheal gills.’ The wing-shaped continuations of the youngest larvæ are in fact the only parts in which air tubes are completely wanting, while tracheæ are richly developed in all other parts of the body.[32] (2) The wings of insects have arisen from 144lateral continuations of the dorsal plates of the body-segments with which they are connected.”
Now, speculating on the primary origin of wings, we need not suppose that they originated in any aquatic form, but in some ancestral land insect related to existing cockroaches and Termes. We may imagine that the tergites (or notum) of the two hinder segments of the thorax grew out laterally in some leaping and running insect; that the expansion became of use in aiding to support the body in its longer leaps, somewhat as the lateral expansions of the body aid the flying squirrel or certain lizards in supporting the body during their leaps. By natural selection these structures would be transmitted in an improved condition until they became flexible, i.e. attached by a rude hinge-joint to the tergal plates of the meso- and metathorax. Then by continued use and attempts at flight they would grow larger, until they would become permanent organs, though still rudimentary, as in many existing Orthoptera, such as certain Blattariæ and Pezotettix. By this time a fold or hinge having been established, small chitinous pieces enclosed in membrane would appear, until we should have a hinge flexible enough to allow the wing to be folded on the back, and also to have a flapping motion. A stray tracheal twig would naturally press or grow into the base of the new structure. After the trachea running towards the base of the wing had begun to send off branches into the rudimentary structure, the number and direction of the future veins would become determined on simple mechanical principles. The rudimentary structures beating the air would need to be strengthened on the front or costal edge. Here, then, would be developed the larger number of main veins, two or three close together, and parallel. These would be the costal, subcostal, and median veins. They would throw out branches to strengthen the costal edge, while the branches sent out to the outer and hinder edges of the wings might be less numerous and farther apart. The net-veined wings of Orthoptera and Pseudoneuroptera, as compared with the wings of Hymenoptera, show that the wings of net-veined insects were largely used for respiration as well as for flight, while in beetles and bees the leading function is flight, that of respiration being quite subordinate. The blood would then supply the parts, and thus respiration or aëration of the blood would be demanded. As soon as such expansions would be of even slight use to the insect as breathing organs, the question as to their permanency would be settled. Organs so useful both for flight and aëration of the blood would be still further developed, until they would become permanent structures, genuine wings. They would thus be readily transmitted, and being of more use in adult life during the season of reproduction, they would be still further developed, and thus those insects which could fly the best, i.e. which had the strongest wings, would be most successful in the struggle for existence. Thus also, not being so much needed in larval life before the reproductive organs are developed, they would not be transmitted except in a very rudimentary way, as perhaps masses of internal indifferent cells (imaginal discs), to the larva, being the rather destined to develop late in larval and in pupal life. Thus the development of the wings and of the generative organs would go hand in hand, and become organs of adult life.[33]
The development and structure of the tracheæ and veins of the wing.—The so-called veins (“nervures”) originate from fine tracheal 145twigs which pass into the imaginal discs. A single longitudinal trachea grows down into the wing-germ (Fig. 147), this branch arising through simple budding of the large body-trachea passing under the rudiment of the wing.
Fig. 159.—Germ of a hind wing detached from its insertion, and examined in glycerine: i, pedicel of insertion to the hypodermis; tr, trachea; b, semicircular pad; e, enveloping membrane; c, bundle of capillary tracheoles; the large tracheæ of the wing not visible; they follow the course of the bundles of tracheoles.—After Gonin.
Gonin states that before the tracheæ reach the wing they divide into a great number of capillary tubes united into bundles and often tangled. This mass of tracheæ does not penetrate into the wing-germ by one of its free ends, but spreading over about a third of the surface of the wing, separates into a dozen bundles which spread out fan-like in the interior of the wing. (Fig. 159). These ramifications, as seen under the microscope, are very irregular; they form here and there knots and anastomoses. They end abruptly in tufts at a little distance from the edge of the wing. A raised semicircular ridge (b) surrounds the base of the wing, and within this the capillaries are formed, while on the other side they are covered by a cellular layer.
Landois, he says, noticed neither the pedicel of the insertion of the wing (i) nor the ridge (b). Herold only states that the tracheæ pass like roots into the wing. Landois believed that they formed an integral part of it. Dewitz and Pancritius used sections to determine their situation.
Fig. 160 will illustrate Landois’ views as to the origin of the tracheæ and veins. A represents the germ of a hind wing attached 146to a trachea; c the elongated cells, in which, as seen at B, c, a fine tangled tracheal thread (t) appears, seen to be magnified at C. The cell walls break down, and the threads become those which pass through the centre of the veins.
Fig. 160.—Origin of the wings and their veins.—After Landois.
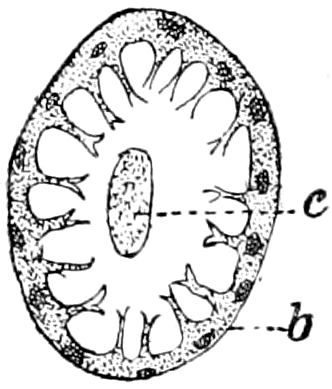
Fig. 161.-Section of the “rib” of a vein: c, cord; b, twig.—After Schaeffer.
The wing-rods.—Semper discovered in transverse sections of the wings, what he called Flügelrippen; one such rib accompanying the trachea in each vein. He did not discover its origin, and his description of it is said to be somewhat erroneous. Schaeffer has recently examined the structure, remarking: “I have surely observed the connection of this cellular tube with the tracheæ. It is found in the base of the wing where the lumen of the tracheæ is much widened. I only describe the fully formed rib (rippe). In a cross-section it forms a usually cylindrical tube which is covered by a very thin chitinous intima which bears delicate twigs (Fig. 161). These twigs are analogous to the thickened ridge of the tracheal intima. I can see no connection between the branches of the different twigs. Through the ribs (rippen) extend a central cord (c) which shows in longitudinal section a clear longitudinal streaking. Semper regarded it as a nerve. But the connection of the tube with the trachea contradicts this view. I can only regard the cord as a separation-product of the cells of the walls.”
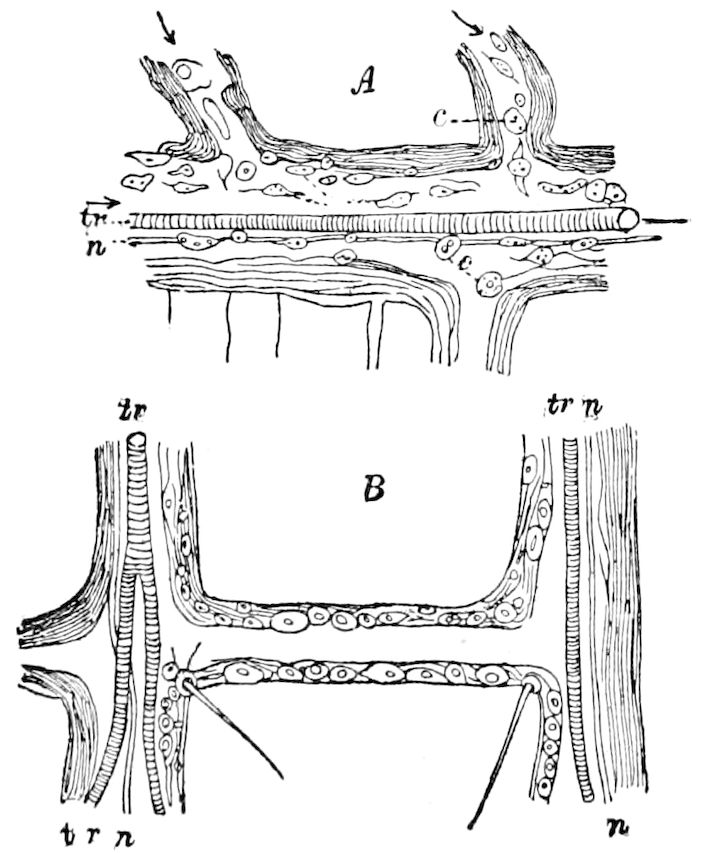
Fig. 162.—Parts of a vein of the cockroach, showing the nerve (n) by the side of the trachea (tr); c, blood-corpuscles.—After Moseley.
Other histological elements.—These are the blood-lymph, corpuscles, blood-building masses, and nerves. Schaeffer states that in the immature pupal wings we find besides the large tracheæ, which are more or less branched, 147and in the wing-veins at a later period, blood-corpuscles which are more or less gorged with nutritive material, and also the “balls of granules” of Weismann, which are perhaps the “single fat-body cells” detected by Semper. Schaeffer also states that into the hypodermal fold of the rudiments of the wings pass peculiar formations of the fat-body and tracheal system, and connected with the fat-body are masses of small cells which by Schaeffer are regarded as blood-building masses.
Fine nerves have also been detected within the veins, Moseley stating that a nerve-fibre accompanies the trachea in all the larger veins in the insects he has examined (Fig. 162), while it is present in Melolontha, where the trachea is absent.
LITERATURE ON THE WINGS
Jurine, L. Nouvelle méthode de classer les Hyménoptères et les Dipterès. Genève, 1807, 4º pp. 319, 14 Pls.
—— Observations sur les ailes des Hyménoptères. (Mém. acad. Turin, 1820, xxiv, pp. 177–214.)
Latreille, P. A. De la formation des ailes des Insectes. (Mém. sur divers sujets de l’histoire naturelle des Insectes, etc. Paris, 1819. Fasc. 8.)
—— De quelques appendices particuliers du thorax de divers Insectes. (Mém. du Mus. d’Hist. nat., 1821, vii, pp. 1–21, 354–363.)
Chabrier, J. Essai sur le vol des insectes. (Mém. du Mus. d’Hist. nat., 1820, vi, pp. 410–476; 1821, vii, pp. 297–372; 1822, viii, pp. 47–99, 349–403.) Separate, pp. 328, 13 Pls.
Burmeister, Hermann. Handbuch der entomologie, i, 1832, pp. 96–106, 263–267, 494–505.
—— Untersuchungen über die Flügeltypen der Coleopteren. (Abhandl. d. naturf. Ges. Halle, 1854, ii, pp. 125–140, 1 Taf.)
Romand, B. E. de. Tableau de l’aile supérieure des Hyménoptères, 1 Pl. Paris, 1839. (Revue Zool., ii, pp. 339; Bericht von Erichson für 1839, pp. 54–56.)
Lefebure, A. Communication verbale sur la ptérologie des Lépidoptères. (Annal. Soc. Ent. France, 1842, i, pp. 5–35, 3 Pls. Also Revue Zool. Paris, 1842, pp. 52–58, 1 Pl.)
Deschamps, B. Recherches microscopiques sur l’organisation des élytres des Coléoptères. (Ann. sc. nat., sér. 3, iii, 1845, pp. 354–363.)
Heer, Oswald. Die Insektenfauna der Tertiärgebilde von Oeningen und Radaboj., 1847, 1. Teil, pp. 75–94.
Newman, E. Memorandum on the wing-rays of insects. (Trans. Ent. Soc. London, ser. 2, iii, 1855, pp. 225–231.)
Westwood, J. O. Notes on the wing-veins of insects. (Trans. Ent. Soc. London, ser. 2, iv, 1857, pp. 60–64.)
Loew, H. Die Schwinger der Dipteren. (Berlin, Entom. Zeitschr., 1858, pp. 225–230.)
Saussure, H. de. Études sur l’aile des Orthoptères. (Ann. scienc. nat., 5 sér. x, p. 161.)
Schiner, J. R. Ueber das Flügelgeäder der Dipteren. (Verhdl. k. k. Zool.-bot., Ges. Wien, 1864, pp. 193–200, 1 Taf.)
148Hagen, H. A. Ueber rationelle Benennung des Geäders in den Flügeln der Insekten. (Stettin. Ent. Zeitung, 1870, xxxi, pp. 316–320, 1 Taf.)
—— Kurze Bemerkungen über das Flügelgeäder der Insekten. (Wiener Entom. Zeit., 1886, v, pp. 311, 312.)
Plateau, F. Qu’est-ce que l’aile d’un insecte? (Stett. Ent. Zeit. Jahrg. 32, 1871, pp. 33–42, 1 Taf. Journal d. Zool., ii, 1873, pp. 126–137.)
Moseley, H. N. On the circulation in the wing of Blatta orientalis and other insects, etc. (Quart. Journ. Micr. Sc. 1871, xi, pp. 389–395, 1 Pl.)
Roger, Otto. Das Flügelgeäder der Kafer. Erlangen, 1875, 90 p.
Rade, E. Die westfalischen Donacien und ihre nachsten Verwandten. 3 Taf. (Vierter Jahresber. d. Westfal Prov.-Vereins f. Wiss. u. Kunst, 1876, pp. 52–87; Flügel, pp. 61–68.)
Katter, F. Ueber Inseckten, speziell Schmetterlingsflügel. (Entom. Nachr., iv, 1878, pp. 279–281, 293–298, 304–309, 321–323.)
Hofmann, Georg v. Ueber die morphologische Deutung der Insektenflügel. (Jahresber. d. akad.-naturwiss. Vereins, Graz, v Jahrg., 1879, pp. 63–68.)
Kolbe, H. J. Das Flügelgeäder der Psociden und seine systematische Bedeutung. (Stettin. Entom. Zeitung, 1880, pp. 179–186, 1 Taf.)
—— Die Zwischenraume zwischen den Punktstreifen der punktiertgestreiften Flügeldecken der Coleoptera als rudimentare Rippen aufgefasst. (Jahresber. zool. Sektion d. Westfal. Prov.-Ver. f. Wiss. u. Kunst. Münster, 1886, pp. 57–59, 1 Taf.)
Lee, A. Bolles. Les balanciers des Diptères, leurs organes sensifères et leurs histologie. (Recueil Zool. Suisse, i, 1885, pp. 363–392, 1 Pl.)
Poppius, Alfred. Ueber das Flügelgeäder der finnischen Dendrometriden. 1 Taf. (Berl. Entom. Zeitschr., 1888, pp. 17–28.)
Comstock, J. H. On the homologies of the wing-veins of insects. (American Naturalist, xxi, 1887, pp. 932–934.)
Brauer, F. Ansichten über die paläozoischen Insekten und deren Deutung. (Annal. d. k. k. naturhist. Mus. Wien, Bd. i, 1886, pp. 86–126, 2 Taf.)
Brauer, F., und J. Redtenbacher. Ein Beitrag zur Entwicklung des Flügelgeäders der Insekten. (Zool. Anz. 1888, pp. 443–447.)
Redtenbacher, J. Vergleichende Studien über das Flügelgeäder der Insekten. 12 Taf. (Annalen d. k. k. naturhist. Hofmuseums zu Wien, 1886, i, pp. 153–231.)
Schoch, G. Miscellanea entomologica. I. Das Geäder des Insektenflügels; II. Prolegomena zur Fauna dipterorum Helvetiae, Wissenschaftl. (Beilage z. Programm d. Kantonsschule Zurich, 1889, 4º, 40 p.)
Bondsdorff, A. von. Ueber die Ableitung der Skulpturverhältnisse bei den Deckflügeln der Coleopteren. (Zool. Anz., 1890, xiii Jahrg., pp. 342–346.)
Spuler, Arnold. Zur Phylogenie und Ontogenie des Flügelgeäders der Schmetterlinge. (Zeitschr. wissens. Zool., liii, 597–646, 2 Taf., 1892.)
Also the writings of Adolph, Bugnion, Calvert, Comstock, Diez, Giraud, Gonin, Graber, Kellogg, Packard, Pratt, Scudder, Walsh.
g. Mechanism of flight
Marey’s views on the flight of insects.—As we owe more to Marey than to any one else for what exact knowledge we have of the theory of flight of insects, the following account is condensed from his work entitled “Movement.” The exceedingly complicated 149movements of the wings would lead us, he says, to suppose that there exists in insects a very complex set of muscles of flight, but in reality, he claims, there are only the two elevator and depressor muscles of each wing.[34] And Marey says that when we examine more closely the mechanical conditions of the flight of insects, we see that an upward and downward motion given by the muscles is sufficient to produce all these successive acts, so well coordinated with each other; the resistance of the air effecting all the other movements. He also refers to the experiments of Giraud which prove that the insect needs for flight a rigid main-rib and a flexible membrane.

Fig. 163.—The two upper lines are produced by the contacts of a drone’s wing on a smoked cylinder. In the middle are recorded the vibrations of a tuning-fork (250 vibrations per second) for comparison with the frequency of the wing movements. Below are seen the movements of the wing of a bee.—After Marey.
If we take off the wing of an insect, and holding it by the small joint which connects it with the thorax, expose it to a current of air, we see that the plane of the wing is inclined more and more as it is subjected to a more powerful impulse of the wind. The anterior nervure resists, but the membranous portion which is prolonged behind bends on account of its greater pliancy.
The wings of insects may be regarded simply as vibrating wires, and hence the frequency of their movements can be calculated by the note produced. Their movements can be recorded directly on a revolving cylinder, previously blackened with smoke, the slightest touch of the tip of the wing removing the black and exposing the white paper beneath; Fig. 163 was obtained in this way. By this method it was calculated that in the common fly the wings made 330 strokes per second, the bee 190, the Macroglossus 72, the dragon-fly 15028, and the butterfly (Pieris rapæ) 9. Thus the smaller the species, the more rapid are the movements of the wings.
Fig. 164.—Appearance of a wasp flying in the sun: the extremity of the wing is gilded.—After Marey.
The path or trajectory made by the tip of the wing is like a figure 8. Marey obtained this by fastening a spangle of gold-leaf to the extremity of a wasp’s wing. The insect was then seized with a pair of forceps and held in the sun in front of a dark background, the luminous trajectory shaping itself in the form of a lemniscate (Fig. 164).
To determine with accuracy the direction taken by the wing at different stages of the trajectory, a small piece of capillary glass tubing was blackened in the smoke of a candle, so that the slightest touch on the glass was sufficient to remove the black coating and show the direction of movement in each limb of the lemniscate. This experiment was arranged as shown in Fig. 165. Different points on the path of movement were tested by the smoked rod, and from the track along which the black had been removed the direction of movement was deduced. This direction is represented in the figure by means of arrows.
Fig. 165.—Experiment to test the direction of movement of an insect’s wing: a, a′, b, b′, different positions of the smoked rod.
Theory of insect flight.—“The theory of insect flight,” says Marey, “may be completely explained from the preceding experiments. The wing, in its to-and-fro movement, is bent in various directions by the resistance of the air. Its action is always that of an inclined plane striking against a fluid and utilizing that part of the resistance which is favorable to its onward progression.
“This mechanism is the same as that of a waterman’s scull, which as it moves backwards and forwards is obliquely inclined in opposite directions, each time communicating an impulse to the boat.”
The mechanism in the case of the insect’s wing is far simpler, 151however, than in the process of sculling, since “the flexible membrane which constitutes the anterior part of the wing presents a rigid border, which enables the wing to incline itself at the most favorable angle.”
“The muscles only maintain the to-and-fro movement, the resistance of the air does the rest, namely, effects those changes in surface obliquity which determine the formation of an 8–shaped trajectory by the extremity of the wing.”

Fig. 166.—Bee flying about in the chamber of the apparatus.—After Marey.
Lendenfeld has applied photography
to determine the position of the wings
of a dragon-fly, and Marey has carried
chronophotography farther to indicate
the normal trajectory of the wing, and
to show the position in flight. Fig.
166 shows a bee in various phases of
flight. “The insect sometimes assumes
almost a horizontal position, in which
case the lower part of its body is much
nearer the object-glass than is its head,
and yet both extremities are equally
well defined in the photograph. The
successive images are separated by an
interval of 1
20 of a second (a long time
when compared to the total time occupied
by a complete wing movement,
i.e. 1
190 of a second). And hence it is
useless to attempt to gain a knowledge
of the successive phases of movement
by examining the successive
photographs of a consecutive series representing
an insect in flight. Nevertheless
an examination of isolated
images affords information of extreme
interest with regard to the mechanism
of flight.
“We have seen that owing to the resistance of the air the expanse of wing is distorted in various directions by atmospheric resistance. Now, as the oscillations during flight are executed in a horizontal plane, the obliquity of the wing-surface ought to diminish the apparent breadth of the wing. This appearance can be seen in Fig. 167. There is here a comparison between two Tipulæ: the one in the act of flight, the other perfectly motionless and resting against the glass window.

Fig. 167.—Illustration to show two Tipulæ, one of them remaining motionless on the glass, and the other moving its limbs in different directions, and setting its body at various inclinations: the illustration only represents a small part of a long series.—After Marey.
“The motionless insect maintains its wings in a position of vertical extension; the plane is therefore at right angles to the axis of the object-glass. The breadth of the wing can be seen in its entirety; the nervures can be counted, and the rounding off of the extremities of the wings is perfectly obvious. On the other hand, the flying insect moves its wings in a horizontal direction, and owing to the resistance of the air the expanse of the wings is obliquely disposed, and only the projection of its surface can be seen in the photograph. This is why the extremity of the wings appears as if it were pointed, while the other parts look much narrower than normal. The extent of the obliquity can be measured from the apparent alteration in width, for the projection of this plane with the vertical is the sine of the angle. From this it may be gathered that the right wing (Fig. 168, third image) was inclined at an angle of about 50° with the vertical, say 40° with the horizontal. This inclination necessarily varies at different points of the trajectory and must augment with the rapidity 153of movement; the obliquity reaching its maximum in those portions of the wings which move with the greatest velocity, namely, towards the extremities. The result is that the wing becomes twisted at certain periods of the movement.” (See the fourth image in Fig. 168.) The position of the balancers seems to vary according to that of the wings. (Marey’s Movement, pp. 253–257.)

Fig. 168.—Tipula in the act of flying, showing the various attitudes of the wings and the position of the balancers.
Graber’s views as to the mechanism of the wings, flight, etc.—Although in reality insects possess but four wings, nature, says Graber, evidently endeavors to make them dipteral. This end is attained in a twofold manner. In the butterflies, bees, and cicadas, the four wings never act independently of each other, as two individual pairs, but they are always joined to a single flying plate by means of peculiar hooks, rows of claws, grooved clamps, and similar contrivances proceeding from the modified edges of the wings; indeed, this connection is usually carried so far that the hind wings are entirely taken in tow by the front, and consequently possess a relatively weak mechanism of motion. The other mode of wing reduction consists in the fact that one pair is thrown entirely out of employment. We observe this for instance in bugs, beetles, grasshoppers, etc.
In the meantime, then, we may not trust to appearances. As their development indeed teaches us, the wings as well as the additional members must be regarded as actual evaginations of the common sockets of the body, and in order especially to refute the prevalent opinion that these wing-membranes are void of sensation, it should be remembered that Leydig has proved the existence, as well as one can be convinced by experiment, of a nerve-end apparatus in certain basal or radical veins 154of the wing-membrane, which is very extensive and complicated, and therefore indicates the performance of an important function, perhaps of a kind of balancing sense, and also that these same insect wings, with their delicate membrane, are very easily affected by different outside agents, as, for instance, warmth, currents of air, etc.
Usually in their inactive or passive state the wings are held off horizontally from the body during flight, and are laid upon the back again when the insect alights; but an exception occurs in most butterflies and Neuroptera, among which the wing-joint allows only one movement round the oblique and long axis of the wings. From this cause, too, the insects just mentioned can unfold their wings suddenly.
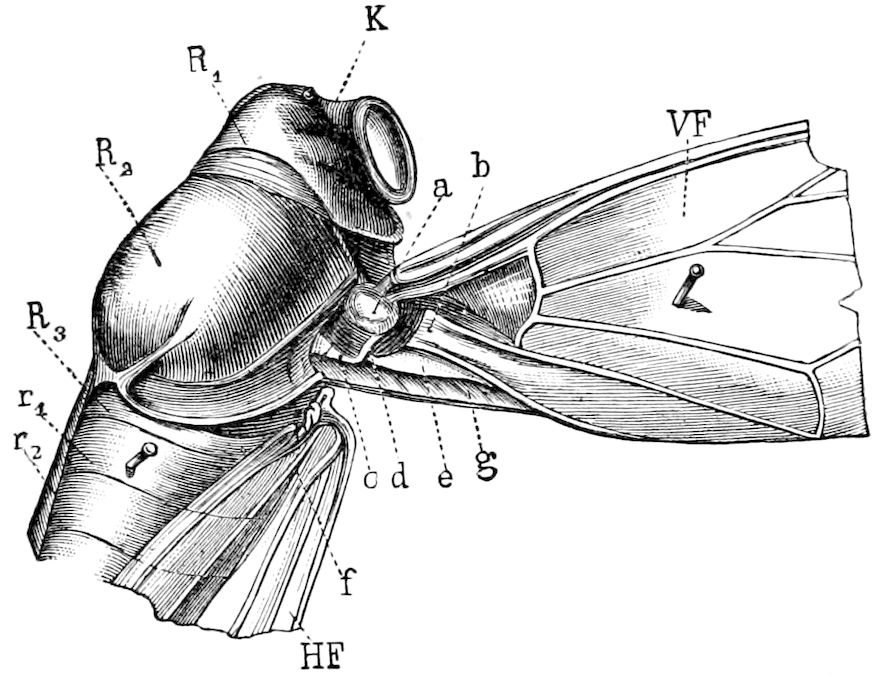
Fig. 169.—Anterior part of a Cicada for demonstrating the mechanism of the articulation of the fore wing: a, articular head; b, articular pan, frog, or cotyla; g, elastic band; c, d, e, system of elastic rods; r1, r2, 1st and 2d abdominal segments. HF, hind wings.—After Graber.
The transition of the wings from the active to the resting condition seems to be by way of a purely passive process, which, therefore, usually gives no trouble to the insect. The wing being extended by the tractive power of the muscles, flies back, when this ceases, to its former or resting posture by means of its natural elasticity, like a spiral spring disturbed from its balance. The structure of this spring joint is very different, however.
It usually consists (Fig. 169) of two parts. The wing can move itself up and down in a vertical plane by means of the forward joint, and at the same time can rotate somewhat round its long axis, because the chitinous part mentioned above is ground off after the fashion of a mandrel.
The hinder joint, at a greater distance from the body, virtually consists of a rounded piece (a) capitate towards the outside, and of a prettily hollowed socket (b) formed by the union of the thick ribs of the hind wings, which slides round 155the head joint when the wings snap back upon the back. The mechanism which causes this turning is, however, of a somewhat complicated nature. The most instrumental part of it is the powerful elastic band (g) which is stretched over from the hinder edge of the mesothorax (R2) towards that of the wings. This membrane is extended by the expansion of the wings, and draws them towards the body as soon as the contraction of the muscles relaxes. This closing band of the wings is assisted by a leverage system consisting of three little chitinous rods (c, d, e), which at its joining presses inwards on the body on one side, and on the hinder edge and head-joint of the wing on the other.
We must, however, lay great stress on a few more kinds of wing support.
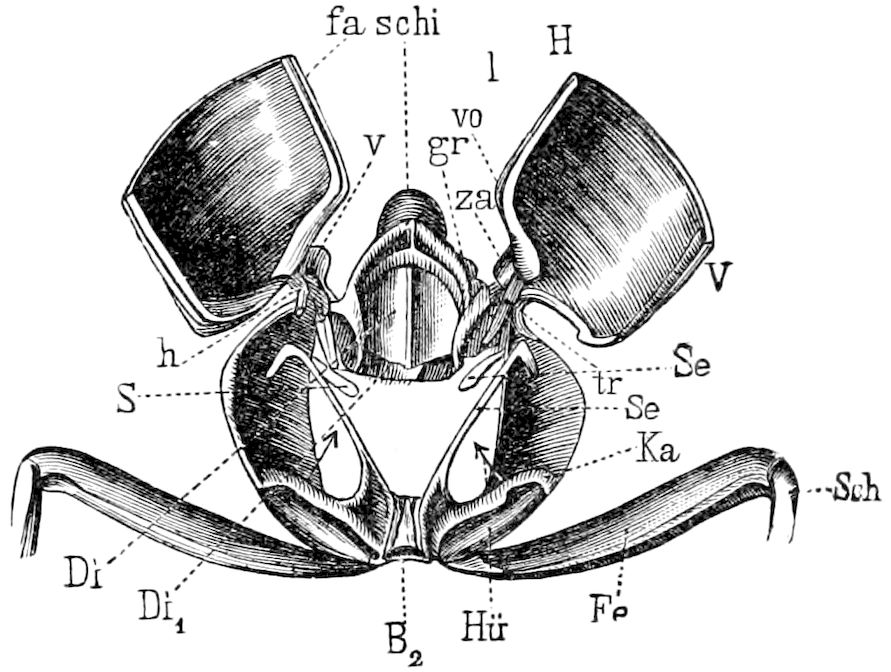
Fig. 170.—Mesothoracic skeleton of a stag beetle: schi, scutellum, on each side of which is the articulation of the fore wing (V), consisting of two small styliform processes (v, h) of the base of the wing; za, tooth which fits into the cavity of the wing-lock (gr); l, edge of the right wing, passing into the corresponding groove (fa) of the left; Di, diaphragm for the attachment of the tergal muscle of the metasternum; Di1 (not explained by author); Ka, acetabulum of the coxa (Hü); Se, chitinous process for the attachment of the coxal muscle; Fe, femur; Sch, tibia; B2, sternum.—After Graber.
The wing-cases of beetles at their return from flight are joined together like the shells of a mussel on the inside as well as to the wedge-shaped plate (Fig. 170, schi) between their bases. There is even a kind of clasp at hand for this purpose. The base of the wing, that is, bears a pair of tooth-like projections (za), which fit into the corresponding hollows of the little plate.
The commissure arising from the joining of the inner edges is characteristic. Usually the wings on both sides interlock by means of a groove, as in stag-beetles, but sometimes even, as in Chlamys, after the manner of two cog-wheels, so that we have here also an imitation of the two most prevalent methods which the cabinet-maker uses in joining boards together.
The act of folding the broad hind wings among beetles is not less significant than the arrangement of the fore wing. If we forcibly spread out the former in a beetle which has just been killed and then leave it to its own resources again, we observe the following result: According to its peculiar mode of joining, the costal vein on the fore edge approaches the mid or discoidal vein of the basal half as well as the distal half of the wing, whence arises a longitudinal fold which curves in underneath. Then the distal half snaps under like the blade of a pocket knife and lies on the plane of the costal edge of the wing, while it also draws after it the neighboring wing-area. The soft hinder-edge portion turns in simultaneously when this wing-area remains fixed to the body while the costal portion is moving towards the middle line of the body.
The wing-membranes of almost all insects have, moreover, the capability of folding themselves somewhat, and this power of extending or contracting the wing-membrane at will is of great importance in flight.
Yes, but how is the folded wing spread out again? The fact may be shown more simply and easily than one might suppose, and may be most plainly demonstrated even to a larger public by making an artificial wing exactly after the pattern of the natural one, in which bits of whalebone may take the place 156of veins and a piece of india rubber the membrane spread out between them. The reader will be patient while we just explain to him the act of unfolding of the membranous wing of the beetle. The actual impulse for this unfolding is due to the flexor muscles which pull on, and at the same time somewhat raise the vein on the costal edge. By this means the membranous fold lying directly behind the costal vein is first spread out. But since this fold is connected with the longitudinal fold of the distal end of the wing which closes like a blade, the wing-area last mentioned which is attached to the middle fold of the wing by the elastic spring-like diagonal vein becomes stretched out. The hinder rayed portion adjacent to the body is, on the other hand, simply drawn along when the wing stands off from the body.
In order to properly grasp the mechanism of the insect wing we must again examine its mode of articulation to the body somewhat more accurately.
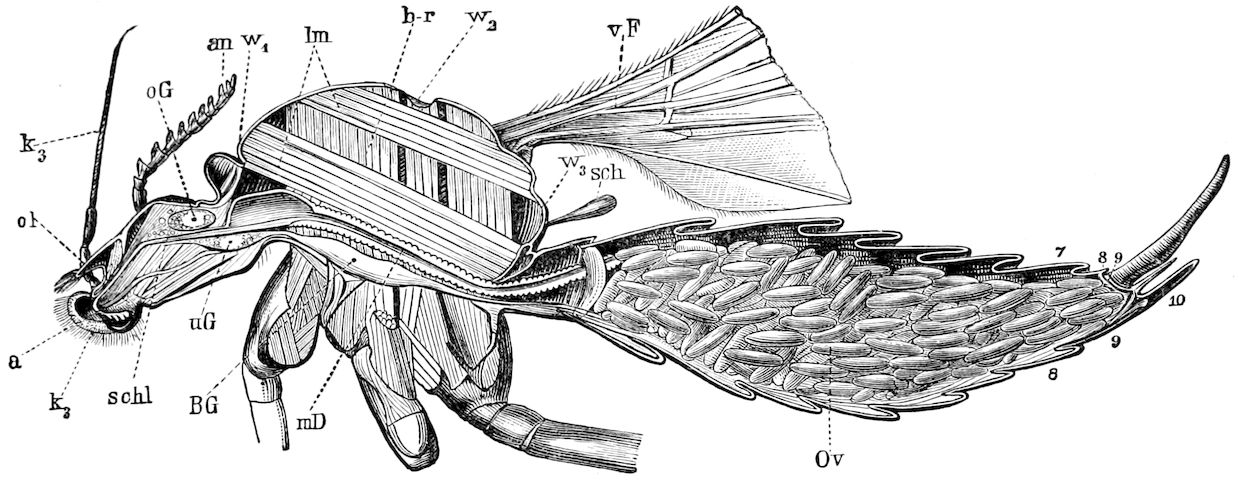
Fig. 171.—Longitudinal section through a Tipula: a, mouth; an, antenna; k3, maxillary palpus; ol, labrum; oG, brain; uG, subœsophageal ganglion; BG, thoracic ganglion; schl, œsophagus; mD, digestive canal; Ov, ovary; vF, fore wing; sch, halter; lm, longitudinal—b-r, lateral muscles.—After Graber.
If we select the halteres of a garden gnat (Tipula) at the moment of extension, we shall find them to be formed almost exactly after the pattern of our oars, since the oblong oar-blade passes into a longitudinal handle. The pedicel of the balancer is formed by the thick longitudinal primary veins of the wing-membrane. This pedicel (Fig. 171) is implanted in the side of the thorax in such a manner that the wing may be compared to the top of a ninepin. One may think, and on the whole it is actually the fact, that the stiff pedicel of the wing is inserted in the thoracic wall, and that a short portion of it (Fig. 172), projects into the cavity of the thorax. It is true there is no actual hole to be found in the thoracic wall, as the intermediate 157space between the base or pedicel of the wing and the aperture in the thorax is lined with a thin yielding membrane, on which the wing is suspended as on an axle-tree. According to this, therefore, the insect wing, as well as any other appendage of arthropods, acts as a lever with two arms. The reader can then conjecture what may be the further mechanism of the wing machine. We only need now two muscles diametrically opposed to each other and seizing on the power arm of the wing, one of which pulls down the short wing arm, thereby raising the oar, while the other pulls up the power arm. And indeed the raising of the wing follows in the manner indicated, since a muscle (hi) is attached to the end of the wing-handle (a) which projects freely into the breast cavity by the contraction of which the power arm is drawn down.
Fig. 172.—Scheme of the flying apparatus of an insect: mnl, thoracic walls; ab, wings; c, pivot; d, point of insertion of the depressor muscle of the wing (kd);—a, that of the elevator of the wing (ai); rs, muscle for expanding,—ml, for contracting, the walls of the thorax.—After Graber.
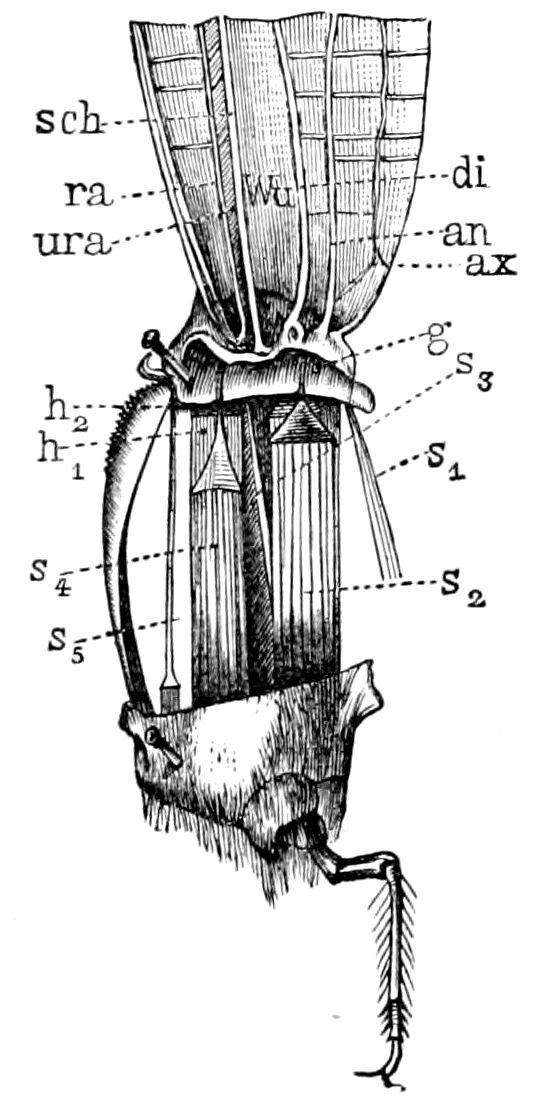
Fig. 173.—Muscles of the fore wing of a dragon-fly (an, ax), exposed by removing the thoracic walls: h1, h2, elevators,—s1-s5, depressors, of the wings (s1, s2, rotators).—After Graber.
On the other hand, we have been entirely mistaken in reference to the mechanism which lowers the wings. The muscle concerned, that is kd, is not at all the antagonist of the elevator muscle of the wing, since it is placed close by this latter, but nearer to the thoracic wall. But then, how does it come to be the counterpart of its neighbor? In fact, the lever of the wing is situated in the projecting piece alone. The extensor muscle of the wing does not pull on the power arm, but on the resistant arm on the other side of the fulcrum (c). The illustration shows, however, how such a case is possible. The membrane of the joint fastening the wing-stalk to the thorax is turned up outwards below the stalk like a pouch. The tendon of the flexor of the wing passes through this pouch to its point of attachment (c) lying on the other side of the fulcrum (d). Thus it is very simply explained how two muscles which act in the same direction can nevertheless have an entirely contrary working power.
This is in a way the bare physical scheme of the flying machine by the help of which we shall more easily become acquainted with its further details.
Dragon-flies are unquestionably the most suitable objects for the study of the muscles pulling directly on the wing itself. If the lateral thoracic wall (Fig. 173) be removed or the thorax opened lengthwise there appears a whole storehouse of muscular cords which are spread out in an oblique direction between the base of the wing and the side of the thoracic plate. There is first to be ascertained, by the experiment of pulling the individual muscles in the line with a pincers, which ones serve for the lifting and which for the lowering 158of the wings. In dragon-flies the muscles are arranged in two rows and in such a way that the flexors or depressors (s, 1 bis) cling directly to the thoracic wall (compare also the muscle dk in Fig. 172 and se in Fig. 174), while the raiser or extensor (h 1, to h 2, Fig. 172, hi and Fig. 174 he) lie farther in. The form of the wing-muscles is sometimes cylindrical, sometimes like a prism, or even ribbon-like. However, the contracted bundles of fibres do not come directly upon the joint-process we have described, but pass over often indeed at a very considerable distance from them, into peculiar chitinous tendons. These have the form of a cap-like plate, often serrate on the edge, which is prolonged into a thread, which should be considered as the direct continuation of the base of the wings. The wings, therefore, sink down into the thoracic cavity as if they were a row of cords ending in handles where the strain of the muscles is applied.
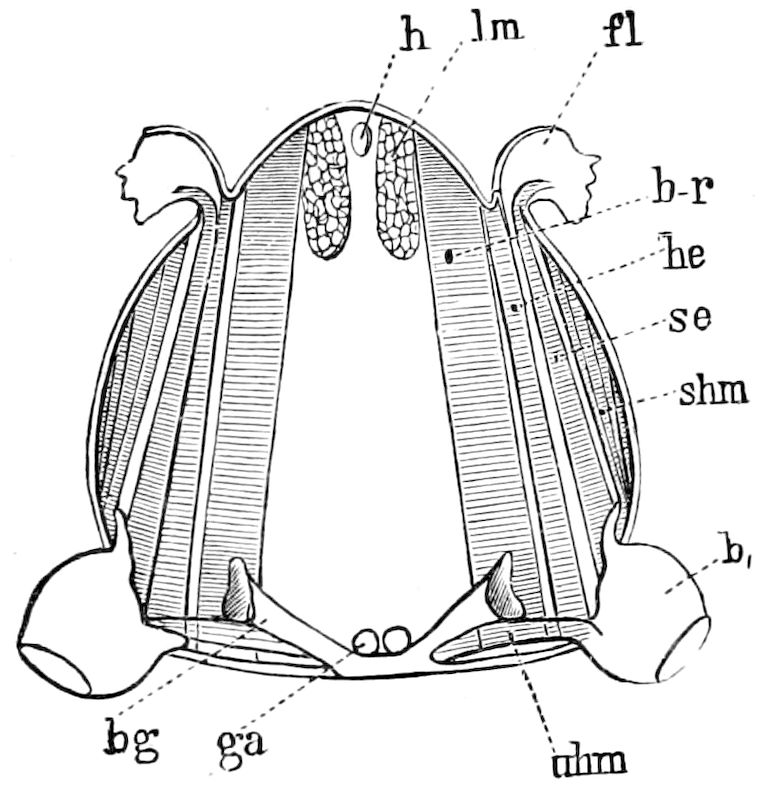
Fig. 174.—Transverse section through the thorax of a locust (Stenobothrus): b1, leg; h, heart; ga, ventral cord; se, depressor,—he, elevator, of the wing (fl); b-r, lateral muscles which expand the thoracic walls;—lm, longitudinal muscles which contract them; shm, uhm, muscles to the legs; bg, apodemes.—After Graber.
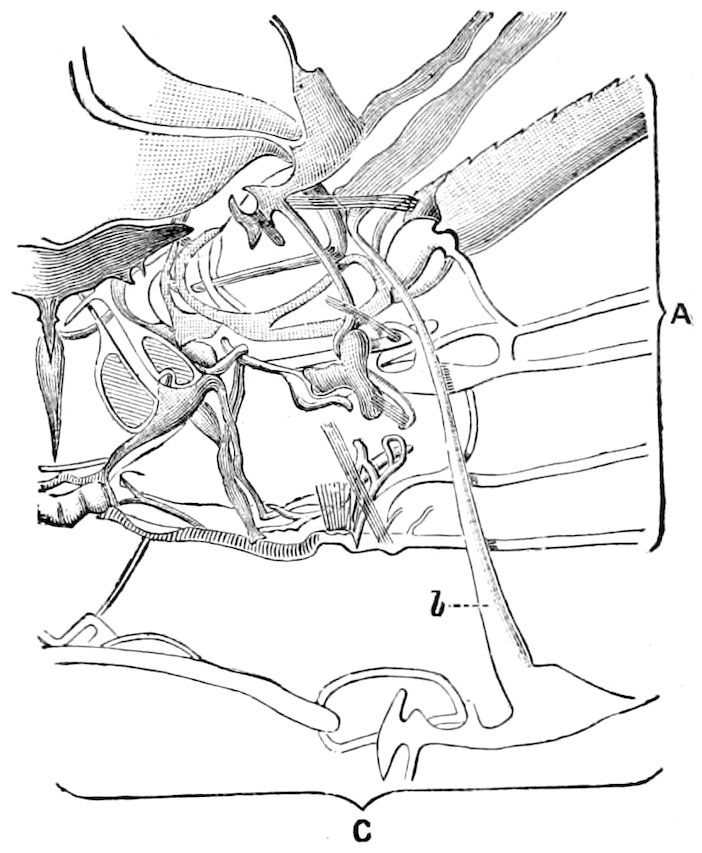
Fig. 175.—Inner view of a portion of the left side of body of Libellula depressa, showing a part of the mechanism of flight, viz., some of the chitinous ridges at base of the upper wing, and some of the insertions of the tendons of muscles: A, line of section through the base of the upper wing, the wing being supposed to be directed backwards. C, upper portion of mechanism of the lower wing; b, lever extending between the pieces connected with the two wings.—After von Lendenfeld, from Sharp.
As may be seen in Fig. 173, the contractile section of several of the muscles of the wing (s5) is extraordinarily reduced, while its thread-like tendon is proportionately longer. This gradation being almost like that of the pipes of an organ in the length of the wing-muscles, as may so easily be observed in the large dragon-flies, plainly indicates that the strain of the individual muscles is quite different in strength, since, as the phenomenon of flight demands it, the different parts of the base of the wing become respectively relaxed in very dissimilar measure.
We have thus far discussed only the elevator and depressor muscles. Other groups (s1s3) are yet to be added, however, crossing under the first at acute 159angles, which when pulling the wing sidewise, bring about in union with the other muscles a screw-like turning of the wings.
While in dragon-flies all the muscles which are principally influential in moving the wing are directly attached to it, and thus evidently assert their strength most advantageously, the case is essentially different with all other insects. Here, as has already been superficially mentioned above, the entire set of muscles affecting the wing is analyzed into two parts of which the smaller only is usually directly joined to the wings, while the movement is indirectly influenced by the remainder (Graber).
In the dragon-fly the two wings are “brought into correlative action by means of a lever of unusual length existing amongst the chitinous pieces in the body wall at the base of the wings (Fig. 175, b). The wing-muscles are large; according to von Lendenfeld there are three elevator, five depressor, and one abductor muscles to each wing. He describes the wing-movements as the results of the correlative action of numerous muscles and ligaments, and of a great number of chitinous pieces connected in a jointed manner” (Sharp).
If again we take the longitudinal section of the thoracic cavity of gnats in Fig. 171, we shall perceive a compactly closed system of muscular bars intersecting each other almost at right angles and interlaced with a tangled mass of tracheæ, some of which muscles extend (lm) longitudinally, that is from the front to the back, while others (b-r) stretch out in a vertical direction, that is between the plates of the abdomen and back.
In order that we may more easily comprehend this important muscular apparatus we will illustrate the thoracic cavity of insects by an elastic steel ring (Fig. 172), to which we may affix artificial wings. If this ring be pressed together from above downward, along the line rs, thus imitating the pulling of the vertical or lateral thoracic muscles, then the wings on both sides spring up. This is to be explained by the fact that through this manipulation a pressure is exerted on the lifting power arm of the wings. If, on the other hand, the ring be compressed on the sides (ml), which is the same thing as if the longitudinal muscles contracted the thorax from before backward, and thus arched it more, then the wings are lowered.
Agrioninæ, according to Kolbe, can fly with the fore pair of wings or with the hind pair almost as well as with both pairs together. Also the wings of these insects can be cut off before the middle of their length without injuring their power of flight. Butterflies, Catocalæ, and Bombycidæ fly after the removal of the hind wings. Also the balancers of the Diptera must be useful in flying, since their removal lessens the power of flight.
Chabrier regarded the under sides of the shell-like extended wing-covers of the beetles as wind-catchers, which, seized by wind currents, carry the insect through the air. We may also consider the wing-covers as regulators of the centre of gravity of flight.
The observations of insects made by Poujade (Ann. Soc. Ent., France, 1887, p. 197) during flight teaches us, says Kolbe, that in respect to the movement during flight of both pairs of wings, they may be divided into two categories:—
1. Into those where both pairs of wings (together), either united, and also when separated from each other, perform flight. Such are the Libellulidæ, Perlidæ, Sialidæ, Hemerobiidæ, Mymeleonidæ, Acridiidæ, Locustidæ, Blattidæ, Termitidæ, etc.
2. Into those whose fore and hind wings act together like one wing, since they are connected by hooks (hamuli), as in certain Hymenoptera, or are attached in other ways. Here belong Hymenoptera, Lepidoptera, Trichoptera, Cicadidæ, Psocidæ, etc.
160The musculature of the mesothorax and metathorax is similar in those insects both of whose pairs of wings are like each other, and act independently during flight, viz. in the Libellulidæ. On the other hand, in the second category, where the fore and hind wings act as a single pair and the fore wings are mostly larger than the hinder (except in most of the Trichoptera), the musculature of the mesothorax is more developed than that of the metathorax.
To neither category belong the beetles, whose wing-covers are peculiar organs of flight, and not for direct use, and the Diptera, which possess but a single pair of wings. In the beetles the hind wings, in the Diptera the fore wings, serve especially as organs of flight. It may be observed that the Diptera are the best fliers, and that those insects which use both pairs of wings as a single pair fly better than those insects whose two pairs of wings work independently of each other. An exception are the swift-flying Libellulidæ, whose specially formed muscles of flight explain their unusual capabilities for flying (Kolbe).
LITERATURE ON FLIGHT
Marey, E. J. La machine animale. Locomotion terrestre et aërienne. Paris, 1874.
—— Mémoire sur le vol des insectes et des oiseaux. (Annal. Scienc. natur., 5 sér., Zool. xii, 1869, pp. 49–150; 5 sér., Zool. xv, 1872, 42 Figs.)
—— Note sur le vol des insectes. (Compt. rend. et Mém. Soc. d. Biol. Paris, 4 sér., v, 1869, C. R. pp. 136–139.)
—— Recherches sur le mécanisme du vol des insectes. (Journal de l’Anatomie et de la Physiologie, 6 Année, 1869, pp. 19–36, 337–348.)
—— Animal mechanism. New York, 1879, pp. 180–209.
—— Movement. New York, 1895, pp. 239–274.
Hartings. Ueber den Flug. (Niederland. Archiv f. Zoologie, iv, Leiden, 1877–78.)
Lucy. Le vol des oiseaux, chauvesouris et insectes. Paris.
Tatin, V. Expériences physiologiques et synthétiques sur le mécanisme du vol. (Ecole prat. d. haut. étud. Physiol. expérim. Trav. du laborat. de Marey, 1877, pp. 293–302.)
—— Expériences sur le vol mécanique. (Ibid., 1876, pp. 87–108.)
Bellesme, Jousset de. Recherches expérimentales sur les fonctions du balancier chez les insectes Diptères, Paris, 1878, 96 pp., Figs.
—— Sur une fonction de direction dans le vol des insectes. (Compt. rend., lxxxix, 1879, pp. 980–983.)
Pettigrew, J. Bell. On the mechanical appliances by which flight is attained in the animal kingdom. (Trans. Linn. Soc., 1868, xxvi, Pt. I, pp. 197–277, 4 Pl.)
—— On the physiology of wings. (Trans. Roy. Soc. Edinburgh, 1871, xxvi, pp. 321–446.)
Krarup-hansen, C. J. L. Beitrag zu einer Theorie des Fluges der Vogel, Insekten und Fledermause. (Copenhagen u. Leipzig, Fritsch, 1869, 48 pp.)
Lendenfeld, R. V. Der Flug der Libellen. (Sitzungsber. d. kais. Akad. d. Wiss. Wien, lxxxiii, 1881, pp. 289–376, 7 Taf.; Zool. Anz., 1880, p. 82.)
Girard, M. Note sur diverses expériences relatives à la fonction des ailes chez les insectes. (Ann. Soc. Ent. France, 4 sér., ii, 1862, pp. 154–162.)
Mühlhäuser, F. A. Ueber das Fliegen der Insekten. (22. bis 24., Jahresb. d. Pollichia, Dürkheim, 1866, pp. 37–42.)
Plateau, Félix. Recherches expérimentales sur la position du centre de gravité chez les insectes. (Archiv d. Scienc. phys. et natur. d. Genève, Nouv. période, xliii, 1872, pp. 5–37.)
161Plateau, Félix. Ueber die Lage des Schwerpunktes bei den Insekten. Auszug. (Naturforscher v. Sklarek, v. Jahrg., 1872, pp. 112–113.)
—— Recherches physico-chimiques sur les articulés aquatiques. (Bull. d. l’Acad. Roy. Belg., xxxiv, 1872, pp. 1–50, Fig.)
—— Qu’est-ce que l’aile d’un insecte? (Stett. Ent. Zeit., 1871, pp. 33–42, Pl.)
—— L’aile des insectes. (Journ. d. Zool., ii, 1873, pp. 126–137.)
Perez, J. Sur les causes de bourdonnement chez les insectes. (Comptes rend., lxxxvii, p. 535, Paris, 1878.)
Strasser, Hans. Mechanik des Fluges. (Archiv f. Anat. u. Phys., 1878, p. 310–350, 1 Taf.)
—— Ueber die Grundbedingungen der aktiven Locomotion. (Abhandl. d. naturf. Gesellsch., Halle, 1880, xv, pp. 121–196, Figs.)
Moleyre, L. Recherches sur les organes du vol chez les insectes de l’ordre des Hemiptères. (Compt. rend. de l’Acad. d. Scienc. de Paris, 1882, xcv, pp. 349–352.)
Amans, P. Essai sur le vol des insectes. (Revue d. Sc. Nat. Montpellier, 3 sér., ii, 1883, pp. 469–490, 2 Pl.; iii, 1884, pp. 121–139, 3 Pl.)
—— Étude de l’organe du vol chez les Hyménoptères. (Ibid., iii, pp. 485–522, 2 Pl.)
—— Comparaisons des organes du vol dans la série animale. Des organes du vol chez les insectes. (Annal. d. Scienc. nat. Zool., 6 sér., xix, pp. 1–222, 8 Pl.)
Mullenhoff, K. Die Grosse der Flugflächen. (Pflüger’s Archiv f. d. ges. Physiologie, 1884, xxxv, pp. 407–453.)
—— Die Ortsbewegungen der Tiere. (Wissensch. Beil. z. Programm d. Andreas-Realgymnas. Berlin, 1885, 19 pp.)
Poujade, G. A. Note sur les attitudes des insectes pendant le vol. (Ann. Soc. Ent. France, 1884, 6 sér., iv, pp. 197–200, 1 Pl.)
Krancher, O. Die Töne der Flügelschwingungen unserer Honigbiene. (Deutscher Bienenfreund, 1882, 18. Jahrg., pp. 197–204.)
Landois, H. Ueber das Flugvermögen der Insekten. (Natur. und Offenbarung, vi, 1860, pp. 529–540.)
Ungern-Sternberg, von. Betrachtungen über die Gesetze des Fluges. (Zeitschr. d. Deutschen Vereins z. Förderung d. Luftschiffahrt-Naturwissensch. Wochenschrift v. Potonie, iv, 1889, p. 158.)
Baudelot, E. Du mecanisme suivant lequel s’effectue chez les Coléoptères le retract des ailes inférieures sous les élytres au moment du passage a l’état de repos. (Bull. Soc. d. Scienc. nat., Strasbourg, 1 Année, 1868, pp. 137–138.)
Ris, Fr. Die schweizerischen Libellen. Schaffhausen, 1885. (Beiheft der Mitteil. d. Schweiz. Ent. Ges., vii, pp. 35–84.)
THE ABDOMEN AND ITS APPENDAGES
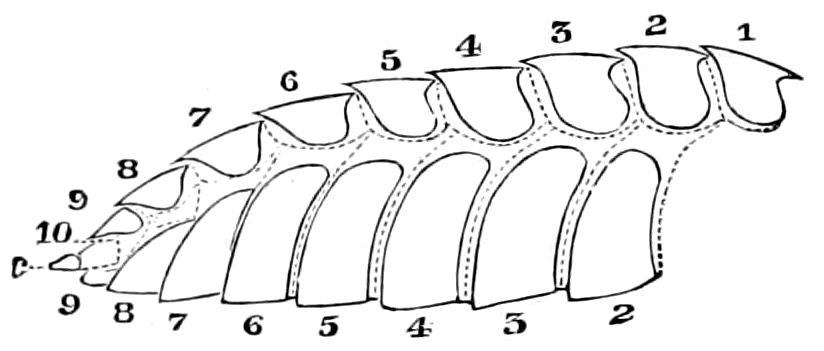
Fig. 176.—Abdomen of Termes flavipes: 1–10, the ten tergites; 1–9, the nine urites; c, cercopod.
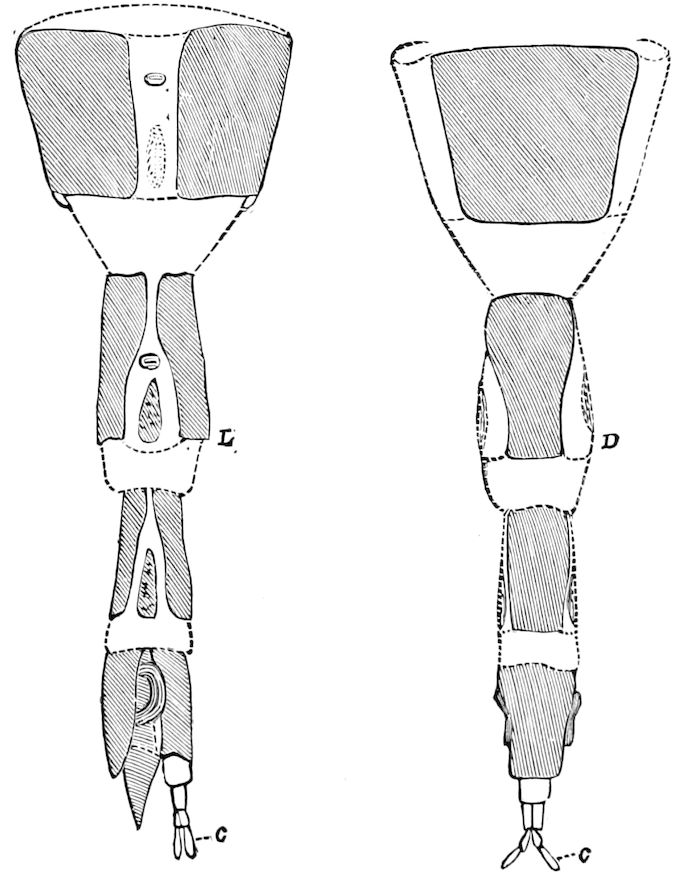
Fig. 177.—End of abdomen of Panorpa debilis drawn out, the chitinous pieces shaded: L, lateral, D, dorsal view; c, jointed cercopoda.—Gissler del.
In the abdomen the segments are more equally developed than elsewhere, retaining the simple annular shape of embryonic life, and from their generalized nature their number can be readily distinguished (Fig. 176). The tergal and sternal pieces of each segment are of nearly the same size, the tergal often overlapping the sternal (though in the Coleoptera the sternites are larger than the tergites), while there are no pleural pieces, the lateral region being membranous when visible and bearing the stigmata (Fig. 177, L). In the terminal segments beyond the genital outlet, however, there is a reduction in and loss of segments, especially in the adults of the metabolous orders, notably the Panorpidæ (Fig. 177), Diptera, and aculeate Hymenoptera; in the Chrysididæ only three or four being usually visible, the distal segments being reduced and telescoped inward.
The typical number of abdominal segments (uromeres), i.e. that occurring in each order of insects, is ten; and in certain families of Orthoptera, eleven. In the embryos, however, of the most generalized winged orders, Orthoptera (Fig. 199), Dermaptera, and Odonata, eleven can be seen, while Heymons has recently detected twelve in blattid and Forficula embryos, and he claims that in the nymphs of certain Odonata there are twelve segments, the twelfth being 163represented by the anal or lateral plates. It thus appears that even in the embryo condition of the more generalized winged insects, the number of uromeres is slightly variable.
We have designated the abdomen as the urosome; the abdominal segments of insects and other Arthropods as uromeres, and the sternal sclerites as urosternites, farther condensed into urites. (See Third Report U. S. Entomological Commission, 1883, pp. 307, 324, 435, etc.)
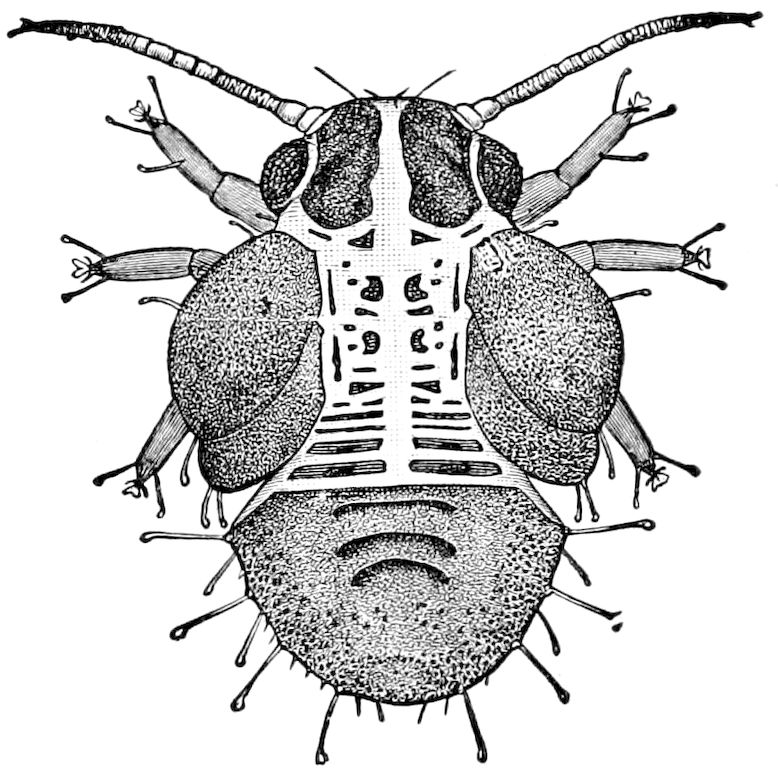
Fig. 178.—Nymph of the pear tree Psylla, with its glandular hairs.—After Slingerland. Bull. Div. Ent. U. S. Dep. Agr.
The reduction takes place at the end of the abdomen, and is usually correlated with the presence or absence of the ovipositor. In the more generalized insects, as the cockroaches, the tenth segment is, in the female, completely aborted, the ventral plate being atrophied, while the dorsal plate is fused during embryonic life, as Cholodkowsky has shown, with the ninth tergite, thus forming the suranal plate.
In the advanced nymph of Psylla the hinder segments of the abdomen appear to be fused together, the traces of segmentation being obliterated, though the segments are free in the first stage and in the imago (Fig. 178). It thus recalls the abdomen of spiders, of Limulus, and the pygidium of trilobites.
The median segment.—There has been in the past much discussion as to the nature of the first abdominal segment, which, in those Hymenoptera exclusive of the phytophagous families, forms a part of the thorax, so that the latter in reality consists of four segments, what appearing to be the first abdominal segment being in reality the second.
Latreille and also Audouin considered it as the basal segment of the abdomen, the former calling it the “segment médiaire,” while Newman termed it the “propodeum.” This view was afterward held by Newport, Schiödte, Reinhard, and by the writer, as well as Osten-Sacken, Brauer, and others. The first author to attempt to prove this by a study of the transformations was Newport in 1839 (article “Insecta”). He states that while the body of the larva is in general composed of thirteen distinct segments, counting the head as the first, “the second, third, fourth, and, as we shall hereafter see, in part also the fifth, together form the thorax of the future imago” (p. 870). Although at first inclined to Audouin’s opinion, he does not appear to fully accept it, yet farther 164on (p. 921) he concludes that in the Hymenoptera the “fifth” segment (first abdominal) is not in reality a part of the true thorax, “but is sometimes connected more or less with that region, or with the abdomen, being intermediate between the two. Hence we have ventured to designate it the thoracico-abdominal segment.” Had he considered the higher Hymenoptera alone, he would undoubtedly have adopted Latreille’s view, but he saw that in the saw-flies and Lepidoptera the first abdominal segment is not entirely united with the thorax, being still connected with the abdomen as well as the thorax. Reinhard in 1865 reaffirmed Latreille’s view. In 1866 we stated from observations on the larvæ made three years earlier, that during the semipupa stage of Bombus the entire first abdominal segment is “transferred from the abdomen to the thorax with which it is intimately united in the Hymenoptera,” and we added that we deemed this to be “the most essential zoölogical character separating the Hymenoptera from all other insects.” (See Fig. 93, showing the gradual transfer and fusion of this segment with the thorax.) In the saw-flies the fusion is incomplete, as also in the Lepidoptera, while in the Diptera and all other orders the thorax consists of but three segments. (See also pp. 90–92.)
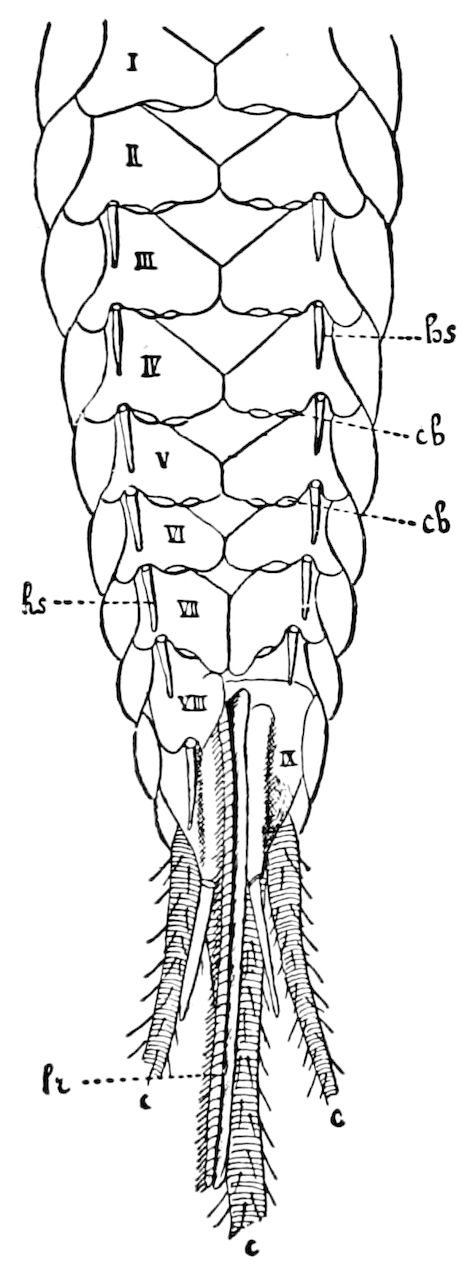
Fig. 179.—Abdomen of Machilis maritima, ♀, seen from beneath: the left half of the 8th ventral plate removed; I-IX, abdominal segments; c, cercopoda; cb, coxal glands; hs, coxal stylets; lr, ovipositor.—After Oudemans, from Lang.
The cercopoda.—We have applied this name to the pair of anal cerci appended to the tenth abdominal segment, and which are generally regarded as true abdominal legs. As is now well known, the embryos of insects of different orders have numerous temporary pairs of abdominal appendages which arise in the same manner, have the same embryonic structure, and are placed in a position homologous with those of the thorax. In the embryo of Œcanthus rudimentary legs appear, as shown by Ayers, on the first to tenth abdominal segment, the last or tenth pair becoming the cercopoda; and similar rudimentary appendages have been detected in the embryos of Coleoptera, Lepidoptera, and Hymenoptera (Apidæ). Cholodkowsky has observed eleven pairs of abdominal appendages in Phyllodromia.
They are very long and multiarticulate in the Thysanura (Fig. 179). In the Dermaptera they are not jointed and are forcep-like. It should also be observed that in the larva or Sisyra (Fig. 181) there are seven pairs of 5–jointed abdominal appendages, though these may be secondary structures or tracheal gills. In the Perlidæ 165and the Plectoptera (Ephemeridæ), they are very long, sometimes over twice as long as the body, and composed of upward of 55 joints; they also occur in the Panorpidæ (Fig. 177). In the dragon-flies the cerci are large, but not articulated, and serve as claspers or are leaf-like[35] (Fig. 180). In a few Coleoptera, as the palm-weevil (Rhynchophorus phœnicis), Cerambyx, Drilus, etc., the so-called ovipositor ends in a hairy, 1–jointed, palpiform cercus. Short 25–jointed cercopoda are present in Termitidæ, and 2–jointed ones in Embiidæ.
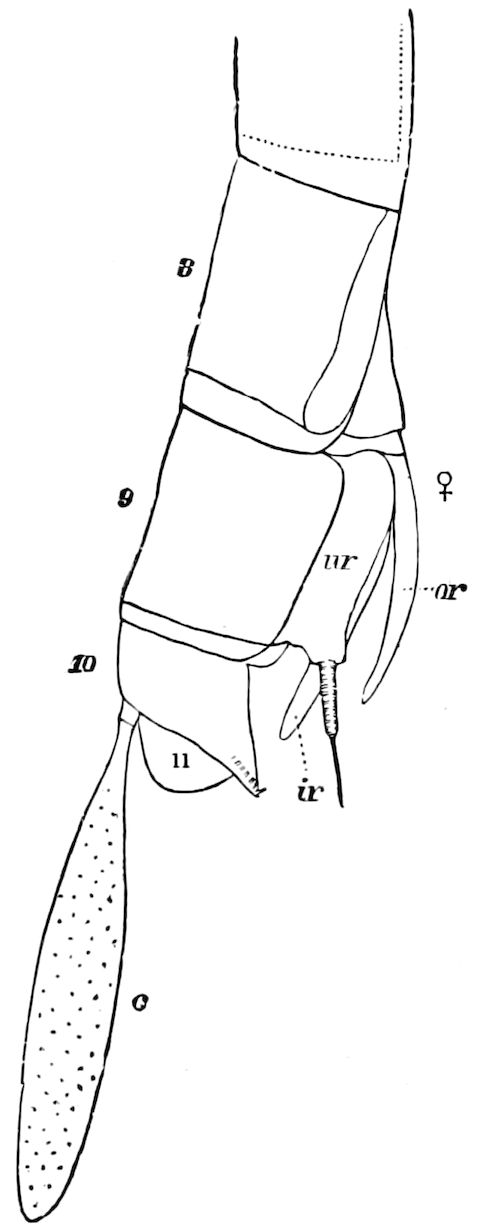
Fig. 180.—End of abdomen of Æschna heros, ♀: ur, urosternite; or, outer, ir, inner styles of the ovipositor; 11, 11th abdominal segment; c, cercopod.
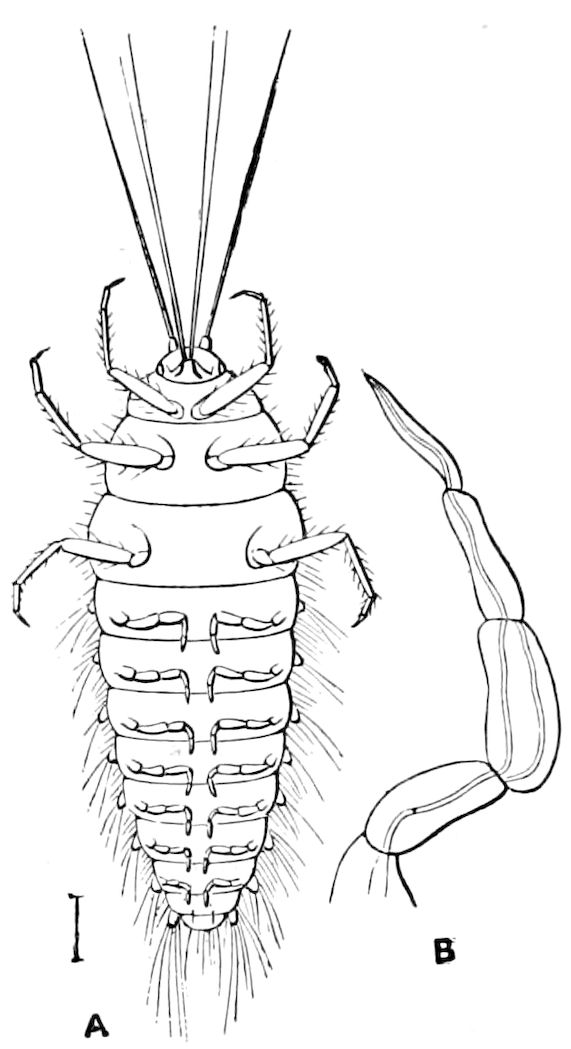
Fig. 181.—Larva of Sisyra, from beneath. B, an abdominal appendage.—After Westwood, from Sharp.
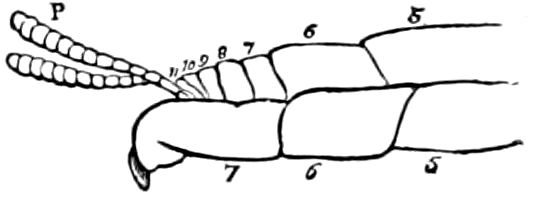
Fig. 182.—Cercopoda (P) of Mantis.—After Lacaze-Duthiers.
The anal cerci are present in the Orthoptera and, when multiarticulate, function as abdominal antennæ. They are longest in the Mantidæ (Fig. 182); they also occur in the larva of the saw-fly, Lyda (Fig. 183). Dr. A. Dohrn has stated that the cerci of Gryllotalpa are true sensory organs, and we have called those of the cockroach abdominal antennæ, having detected about ninety sacs on the upper side of each joint of the stylets, which are supposed to be olfactory in nature, and which are larger and more numerous than similar sacs or pits in the antennæ 166of the same insect.[36] From his experiments upon decapitated cockroaches, Graber concluded that these cerci were organs of smell.
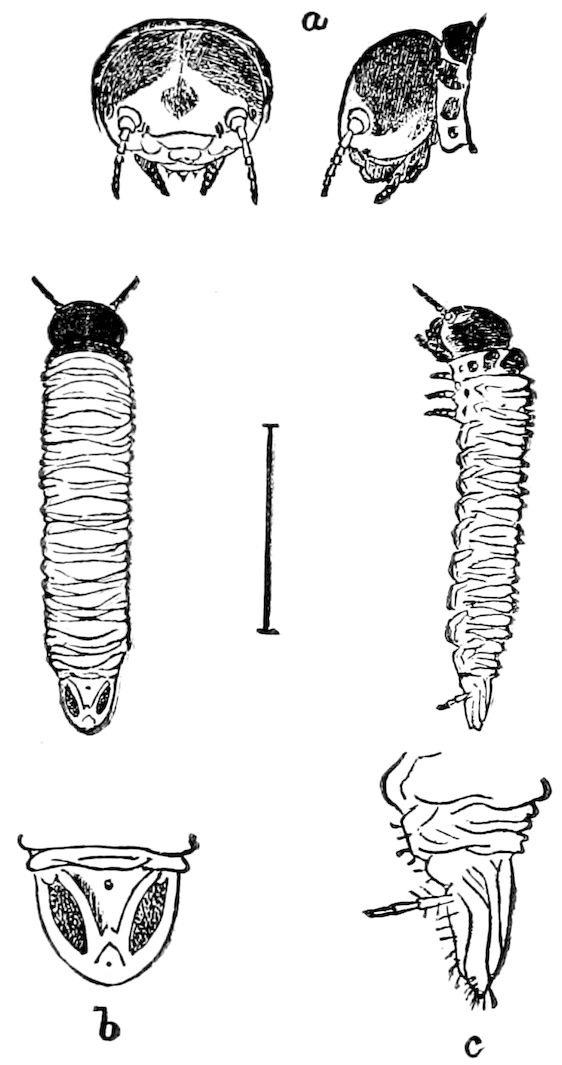
Fig. 183.—Lyda larva: a, head; b, end of body seen from above; c, from side, with cercopod.
Haase regarded these appendages, from their late development and frequent reduction, as old inherited appendages which are approaching atrophy through disuse.
Cholodkowsky states that Tridactylus, a form allied to Gryllotalpa, bears on the tenth abdominal segment two pairs of cerci (ventral and dorsal), and that the ventral pair may correspond to the atrophied appendages of the tenth embryonic segment of Phyllodromia, with which afterward the eleventh segment becomes fused.
The cercopods are not necessarily confined to the eleventh or to the tenth segment, for when there are only nine segments, with the vestige of a tenth, as in Xiphidium, they arise from the ninth uromere, and in the more modern cockroaches, as Panesthia, in which there are but seven entire segments, they are appended to the last or eighth uromere.
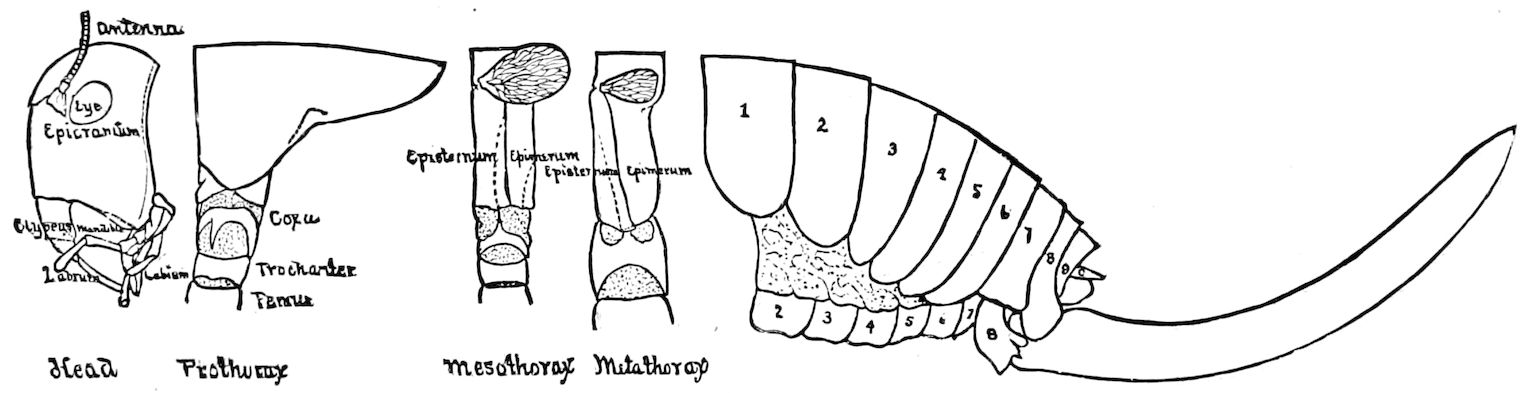
Fig. 184.—Anabrus, ♀, side-view, dissected; showing the relative size of the ovipositor: c, the minute cercopod.—Kingsley del.
167As to the homology and continuity of these cercopods with the ventral outgrowths of the embryo, several embryologists, notably Wheeler, are emphatic in regarding them as such. It thus appears that either the embryonic appendages of the seventh or eighth, ninth or tenth uromere may persist, and form the cercopoda of the adult.
The ovipositor.—The end of the oviduct is guarded by three pairs of chitinous, unjointed styles closely fitted together, forming a strong, powerful apparatus for boring into the ground or into leaves, stems of plants, the bodies of insects, or even into solid wood, so that the eggs may be deposited in a place of safety. In the ants, wasps, and bees the ovipositor also functions as a sting, which is further provided with a poison-sac.
Morphologically, the ovipositor is composed of three pairs of unjointed styles (rhabdites of Lacaze-Duthiers, gonapophyses of Huxley), which are closely appressed to or sheathed within each other, the eggs passing out from the end of the oviduct, which lies, as Dewitz states, between the two styles of the lowest or innermost pair, and under the cross-bars or at the base of the stylets mentioned; the styles or blades spreading apart to allow of the passage of the egg.
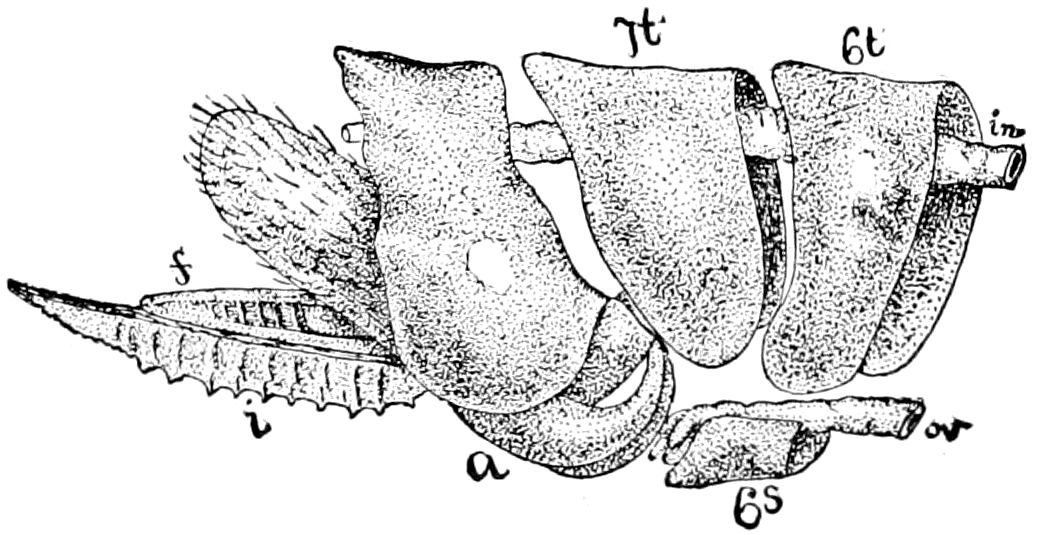
Fig. 185.—Saw of Hylotoma: a, lateral scale; i, saw; f, gorget; 7t, 7th tergite; 6s, 6th sternite; ov, oviduct; in, intestine.—After Lacaze-Duthiers.
The ovipositor is best developed in the Thysanura (Fig. 179, Campodea excepted), in Orthoptera (Fig. 184), in the Odonata, Hemiptera, certain Physapoda, Rhaphiidæ, and in the phytophagous Hymenoptera, where it is curiously modified to form a rather complicated saw for cutting slits in wood or leaves (Fig. 185). It is wanting or quite imperfect in Coleoptera, Diptera, and Lepidoptera.
Morphologically, the ovipositor appears to be formed out of the abdominal appendages of the seventh, eighth, and ninth segments of the female, which, instead of disappearing in the orders first mentioned, persist as permanent styles.
Wheeler asserts from his study of the embryonic development of Xiphidium “there can be no doubt concerning the direct continuity of the embryonic appendages with the gonapophyses.” He goes on to say:—
168“One embryo, which had just completed katatrepis, still showed traces of all the abdominal appendages. The pairs on the eighth, ninth, and tenth segments were somewhat enlarged. In immediately succeeding stages the appendages of the second to sixth segments disappear; the pair on the seventh disappear somewhat later. Up to the time of hatching the gonapophyses could be continuously traced, since in Xiphidium there is no flexure of the abdomen, as in other forms, to obscure the ventral view of the terminal segments. From the time of hatching Dewitz has traced the development of the ovipositor in another locustid (Locusta viridissima), so that now we have the complete history of the organ.”
Heymons, however, is inclined to believe that they are simply hypodermal outgrowths.
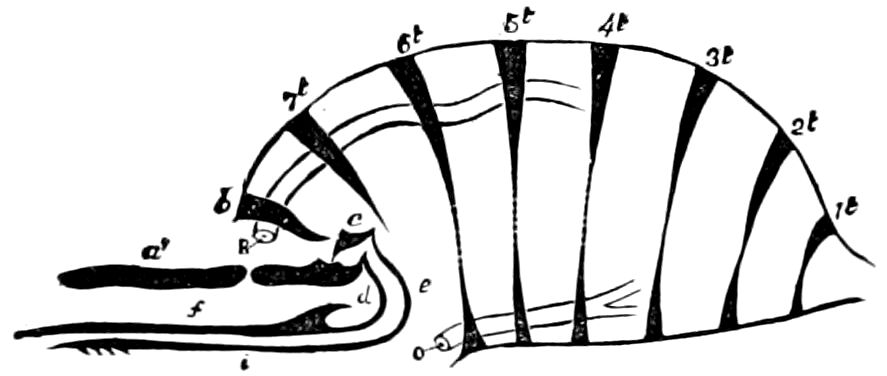
Fig. 186.—Ideal plan of the structure of the ovipositor to illustrate Lacaze-Duthiers’ view: b, 8th tergite; c, epimerum; a′, a, two pieces forming the outer pair of rhabdites; i, the 2d pair, or stylets; and f, the inner pair, or sting; d, support of sting; e, piece supporting the stylet; R, anus; o, outlet of oviduct. The 7th, 8th, and 9th sternites are aborted.—After Lacaze-Duthiers.
The first to study the morphology of the ovipositor was Lacaze-Duthiers, who referred their origin to the partially atrophied dorsal or ventral sclerites of one of the last abdominal segments; a view accepted by Gerstaecker[37] (Figs. 186, 187). The present writer (1866), however, showed that the sting of Bombus was not formed of the reduced pieces of the segments themselves, but arose from special outgrowths on the ventral side of the eighth and ninth abdominal segments. These appendages he did not at first regard as the homologues of the limbs, until in 1871, after studying the origin of the spring of the Podurans (Isotoma), he found that it was a true jointed appendage and therefore a homologue of a pair of the styles forming the ovipositor of the winged insects, and that the three pairs of styles of the latter were homologues of the thoracic legs and cephalic appendages. The view was stated in the Guide to the Study of Insects. (See also Amer. Nat., March, 1871, p. 6.) Kraepelin also affirms that the styles of the ovipositor are segmental appendages and homologues of the antennæ, wings (sic), and legs.
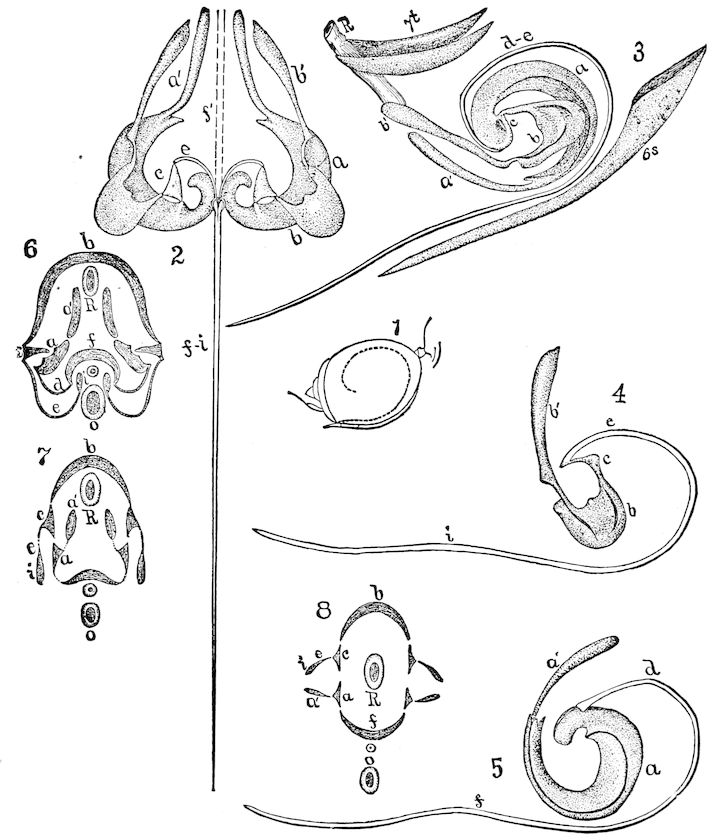
Fig. 187.—1, abdomen of Cynips, showing the great dorsal segment, the peduncle, and the position of the ovipositor within; 2, the entire ovipositor; a, lateral scale; a′, its valve; b, anal scale; b′, stylet; c, support of the stylet; e, base or support of sting (fi); 3, profile showing the relation of the genital armature to the rest of the abdomen, the 6th sternite having been drawn to show its full size; 4, anal scale (b) and stylet; e, i, supports and body of the stylet; c, piece uniting the two scales; 5, lateral scale (a), and a′ sheath; d, support of the sting (f); 6, transverse section of the body through the sting (diagrammatic); R, internal armature; o, oviduct; a, lateral scale; a′ its valve; e, support of the stylets (i); b, anal scale; c, piece uniting two scales; f, sting; d, its support; 7, a second section simpler and more theoretical than the first; 8, diagrammatic, all the elements of the sting have been reduced to pieces of the same form.—After Lacaze-Duthiers.
170An objection to this view is the fact that the posterior pairs of styles appear to arise both from one and the same segment,—the ninth. Dewitz questions whether the four appendages of the ninth segment represent two pairs of limbs, or one pair split into two branches, and prefers the latter view, but leaves it as a point to be settled by future investigations. As will be seen below, both Kraepelin and Bugnion observed a pair of rudiments to each of the three penultimate segments, those of the middle pair splitting in two. Wheeler maintains, erroneously we think, that the inner of the two pairs on the ninth segment represents the tenth pair of abdominal appendages; but in reality this latter pair become the cercopods. That there are probably originally in insects of all the orders provided with an ovipositor three distinct pairs of appendages, one to each segment, is proved, or at least strongly suggested, by Ganin’s researches on the three pairs of abdominal imaginal discs of the third larva of Platygaster and Polynema (Fig. 188), which are transformed into the ovipositor. He remarks that these imaginal discs have the same origin and pass through the same changes as those in front, i.e. those destined to form the thoracic legs. Dewitz has shown that the germs of the ovipositor of the honey-bee arise as buds on the two segments before the last (Fig. 189).
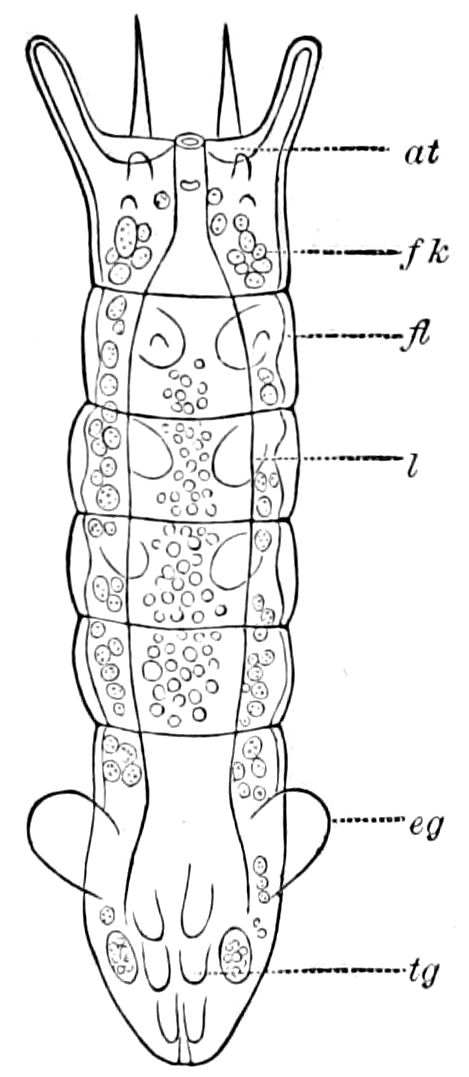
Fig. 188.—Third larva of Polynema: at, antenna; fl, imaginal buds of the wing; l, of the legs; tg, buds of the middle pair of stylets of the ovipositor; fk, fat-body; eg, ear-like process.—After Ganin.
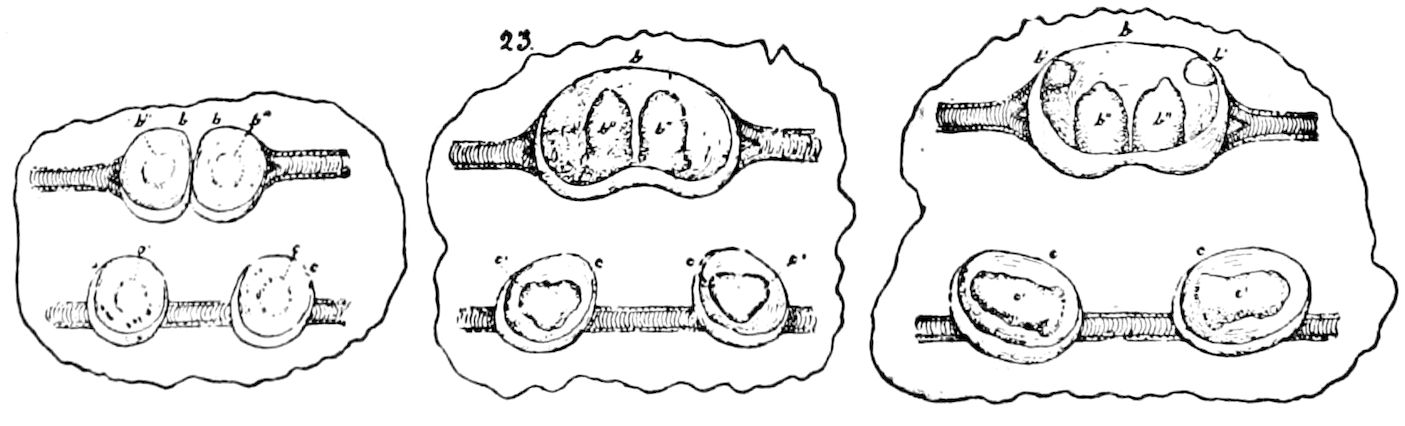
Fig. 189.—Imaginal buds and papillæ of the ovipositor of the honey-bee attached to tracheæ; at different stages: b′, 1st; b″, 2d or middle; and c′, 3d pair of papillæ.—After Dewitz.
Kraepelin also detected in the larva of the honey-bee a pair of what he regarded as genuine imaginal buds on abdominal segments eight, nine, and ten; the buds on the tenth segment are divided each in two; of these four appendages the two median ones form the barbed sting (gorgeret or stachelrinne), and the two lateral stylets, the valves (stachelscheiden). The two buds of the ninth segment give rise to the vagina and to the oviducts, and these unite secondarily with the posterior end of the ovaries. The genital appendages 171of the male correspond to those of the female, and arise from four imaginal buds situated on the under side of the tenth abdominal segment.
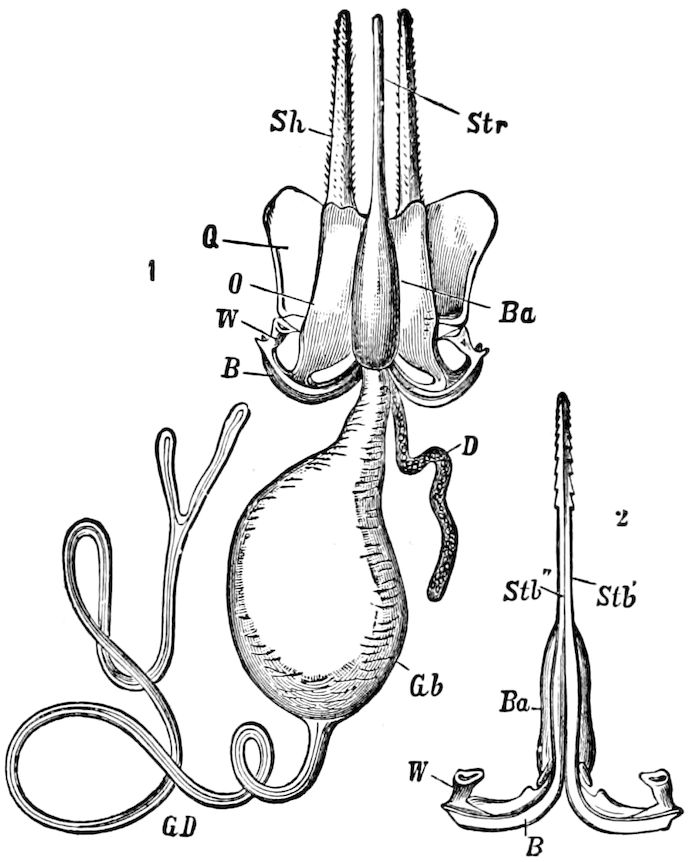
Fig. 190.—1, sting and poison sac of the honey-bee: GD, poison gland; Gb, poison reservoir; D, accessory gland; sh, sheathing style or sting-“feeler”; Str sting; Ba, sheath; Q, quadrate plate; O, oblong piece; W, angular piece; B, base of the sting and stylets; Stb′, Stb″, the two barbed stylets or darts. 2, sting seen from the ventral face: lettering as in the other figure.—After Kraepelin, from Perrier.
In the ants, according to Dewitz, the genital armature is derived from imaginal buds situated on the under side of the seventh, eighth, and ninth abdominal segments. Bugnion has observed the formation of six imaginal buds of the genital armature in the larva of a chalcid (Encyrtus, Figs. 41, 42, 191, q1, q2, q3), the transformation of the central part of these structures into small digitiform pads, then the division of the two intermediate buds into four (?) (Fig. 191, B, q2), but was unable to trace their farther development.
The subject still needs farther investigation, since certain observers, as Haase, and, more recently, Heymons, do not believe that they are homologues of the legs, but integumental structures, though of somewhat higher value than the style of the base of the legs of Scolopendrella and Thysanura; but it is to be observed that as yet we know but little of the embryological history of these styles.
Those authors who have examined the elements of the ovipositor, and regard them as homologues (homodynamous) of the limbs, are Weismann (1866), Ganin (1869), Packard (1871), Ouljanin (1872), Kraepelin, Kowalevsky (1873), Dewitz (1875), Huxley (1877), Cholodkowsky, Bugnion (1891), and Wheeler (1892).
As shown, then, by our observations and those of Dewitz (Figs. 189 and 192), the rudiments of the ovipositor consist of three pairs of tubercles, arising, as Kraepelin and also Bugnion (Fig. 191) have shown, from three pairs of imaginal discs, situated respectively on the seventh, eighth, and ninth uromeres, or at least on the three penultimate segments of the abdomen. With the growth of the 172semipupa, the end of the abdomen decreases in size, and is gradually incurved toward the base (Fig. 193), and the three pairs of appendages approach each other so closely that the two outer ones completely ensheath the inner pair, until a complete extensible tube is formed, which, by the changes in form of the muscles within, is gradually withdrawn entirely within the body.
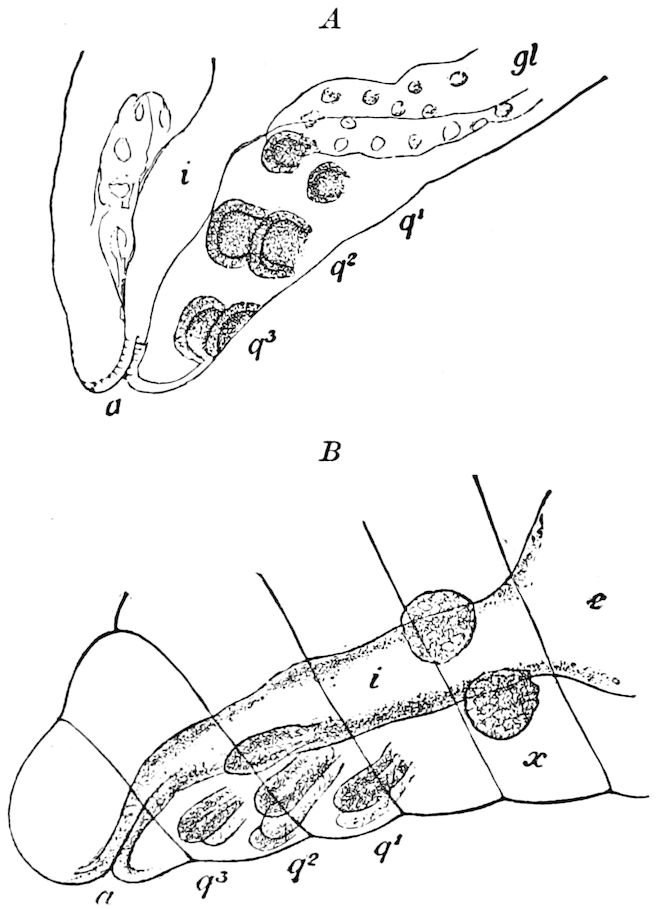
Fig. 191.—A, end of larva of Encyrtus of 2d stage, showing the three pairs of imaginal buds of the ovipositor q1, q2, q3. B, the same in an older larva ready to transform; i, intestine; x, genital gland; a, anus.—After Bugnion.
174An excellent account of the honey-bee’s sting is given by Cheshire (Figs. 194, 195). The outermost of the three pairs of stylets forming the apparatus is the two thick, hairy “palpi” or feelers (P), these being freer from the sting proper than in the ovipositor of Orthoptera. The sting itself is composed of the two inner pairs of stylets; one of these pairs is united to form the sheath (sh), while the other pair form the two barbed darts. The sheath has three uses: first, to open the wound; second, to act as an intermediate conduit for the poison; and third, to hold in accurate position the long barbed darts. The sheath does not enclose the darts as a scabbard, but is cleft down the side presented in Fig. 194, which is below when the sting points backward. But, says Cheshire, as the darts move up and down, they would immediately slip from their position, unless prevented by a mechanical device, exhibited by B and C, giving in cross-section sheath and darts near the end, and at the middle of the former. “The darts (d) are each grooved through their entire length, while upon the sheath (sh) are fixed two guide rails, each like a prolonged dovetail, which, fitted into the groove, permits of no other movement than that directly up and down.” The darts are terminated by ten barbs of ugly form (D, Fig. 194), and much larger than those of the sheath, and as soon as the latter has established a hold, first one dart and then the other is driven forward by successive blows. These in turn are followed by the sheath, when the darts again more deeply plunge, until the murderous little tool is buried to the hilt. But these movements are the result of a muscular apparatus yet to be examined, and which has been dissected away to bring the rigid pieces into view. The dovetail guides of the sheath are continued far above its bulbous portion, as we see by E, Fig. 195; and along with these the darts are also prolonged upward, still held to the guides by the grooved arrangement before explained; but both guides and darts, in the upper part of their length, curve from each other somewhat like the arms of a Y, to the points c, c′ (A, Fig. 194), where the darts 175make attachment to two levers (i, i′). The levers (k, l and k′, l′) are provided with broad muscles, which terminate by attachment to the lower segments of the abdomen. These, by contraction, revolve the levers aforesaid round the points f, f′, so that, without relative movement of rod and groove, the points 176c, c′ approach each other. The arms of the Y straighten and shorten, so that the sheath and darts are driven from their hiding-place together and the thrust is made by which the sheath produces its incision and fixture. The sides being symmetrical, we may, for simplicity’s sake, concentrate our attention on one, say the left in the figure. A muscular contraction of a broad strap joining k and d (the dart protractor) now revolves k on l, so that a is raised, by which clearly c is made to approach d; i.e. the dart is sent forward, so that the barbs extend beyond the sheath and deepen the puncture. The other dart, and then the sheath, follow, in a sequence already explained, and which G, Fig. 195, is intended to make intelligible, a representing the entrance of the sheath, b the advance of the barbs, and c the sheath in its second position. The barb retractor muscle is attached to the outer side of i, and by it a is depressed and the barbs lifted. These movements, following one another with remarkable rapidity, are entirely reflex, and may be continued long after the sting has been torn, as is usual, from the insect. By taking a piece of wash-leather, placing it over the end of the finger, and applying it to a bee held by the wings, we may get the fullest opportunity of observing the sting movements, which the microscope will show to be kept up by continued impulses from the fifth abdominal ganglion and its multitudinous nerves (n, Fig. 194, A), which penetrate every part of the sting mechanism, and may be traced even into the darts. These facts, together with the explanation at page 49, will show why an abdomen separated many hours may be able to sting severely, as I have more than once experienced.
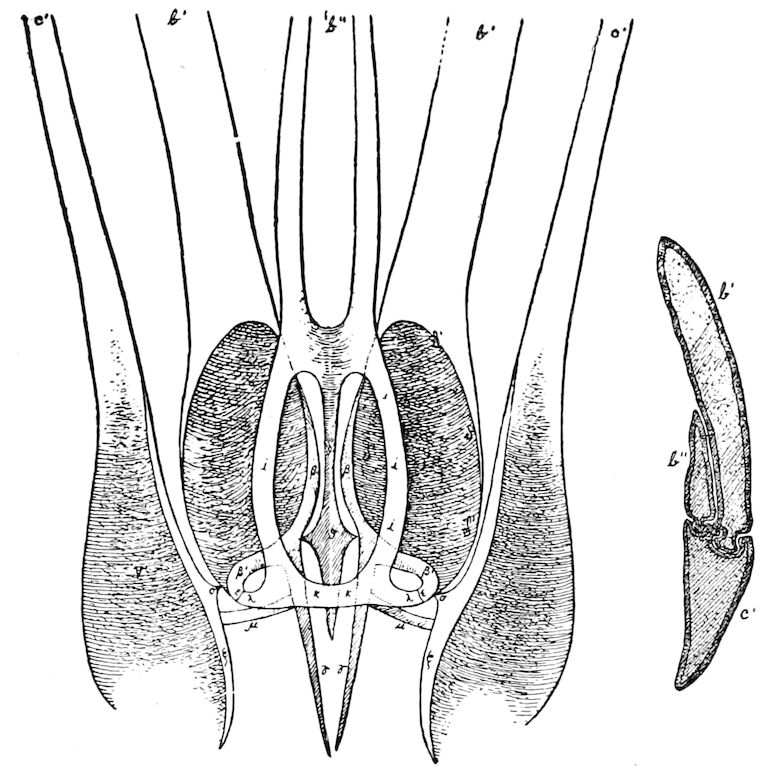
Fig. 192.—Base of the ovipositor of Locusta viridissima seen from beneath: c′, sheath, or outer and lower pair of stylets turned to one side to show the others; b′, upper and inner pair; b″, third or innermost, smallest pair of stylets. A, the same on one side, in section. The shaded parts show the muscular attachments. The muscles which extend the apparatus and are attached to ν, δ, and η, as also the membranes which unite the pieces from η; to γ with each other and the body, are removed, so that only the chitinous parts remain.—After Dewitz.
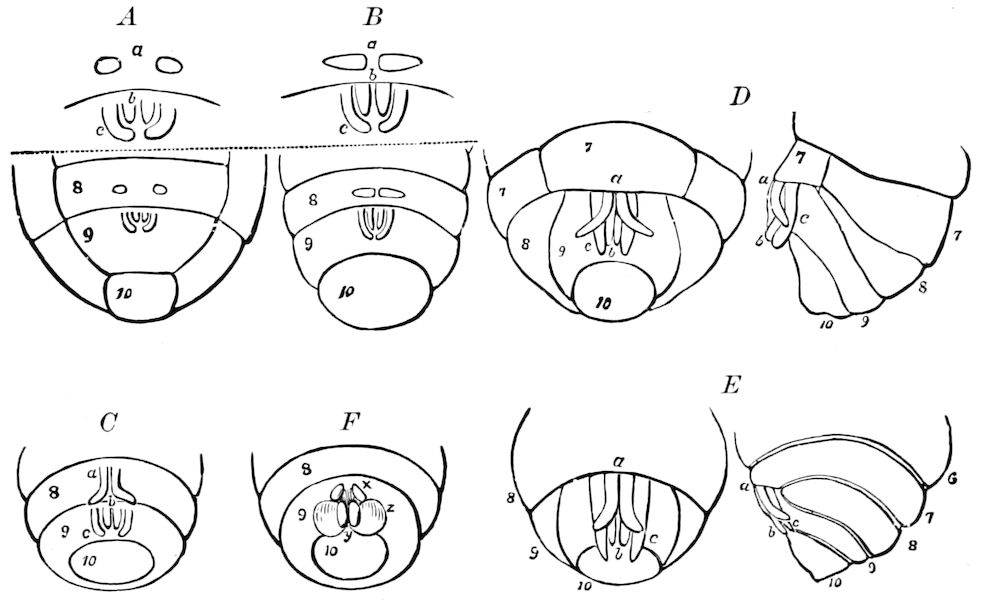
Fig. 193.—Development of the sting in Bombus: A, a, 1st pair on 8th sternite; b, 2d inner pair forming the darts; c, outer pair. B-E, more advanced stages. F, x, y, z, three pairs of tubercles, the germs of the male organs.
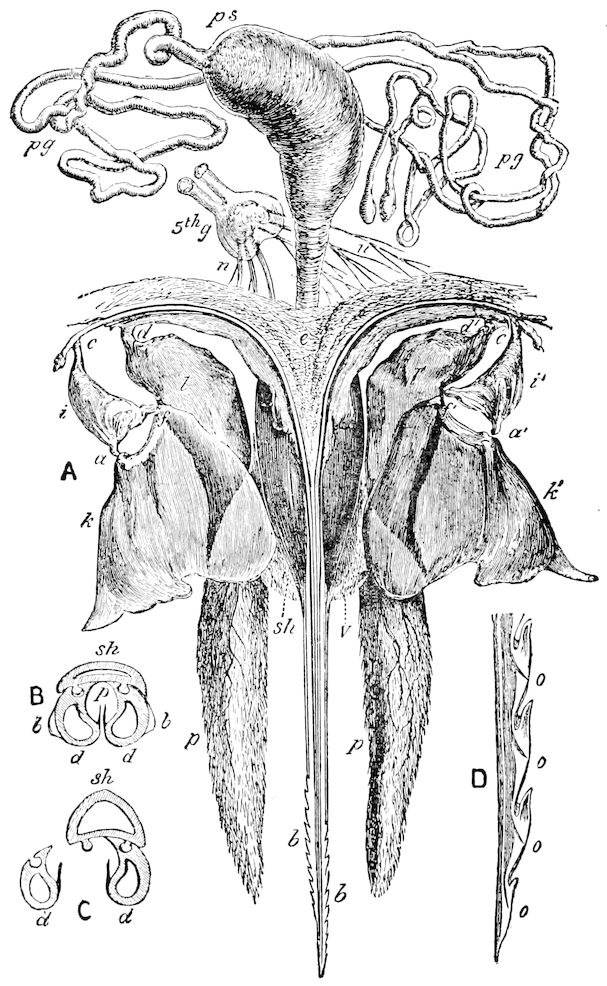
Fig. 194.—Sting of bee × 30 times: A, sting separated from its muscles; ps, poison sac; pg, poison gland; 5th g, 5th abdominal ganglion; n, n, nerves; e, external thin membrane joining sting to last abdominal segment; i, k, l, and i′, k′, l′, levers to move the darts; sh, sheath; v, vulva; p, sting-palpus or feeler, with tactile hairs and nerves. B and C, sections through the darts and sheath, × 300 times: sh, sheath; d, darts; b, barbs; p, poison-channel. D, end of a dart, × 200: o, o, openings for poison to escape into the wound.—After Cheshire.
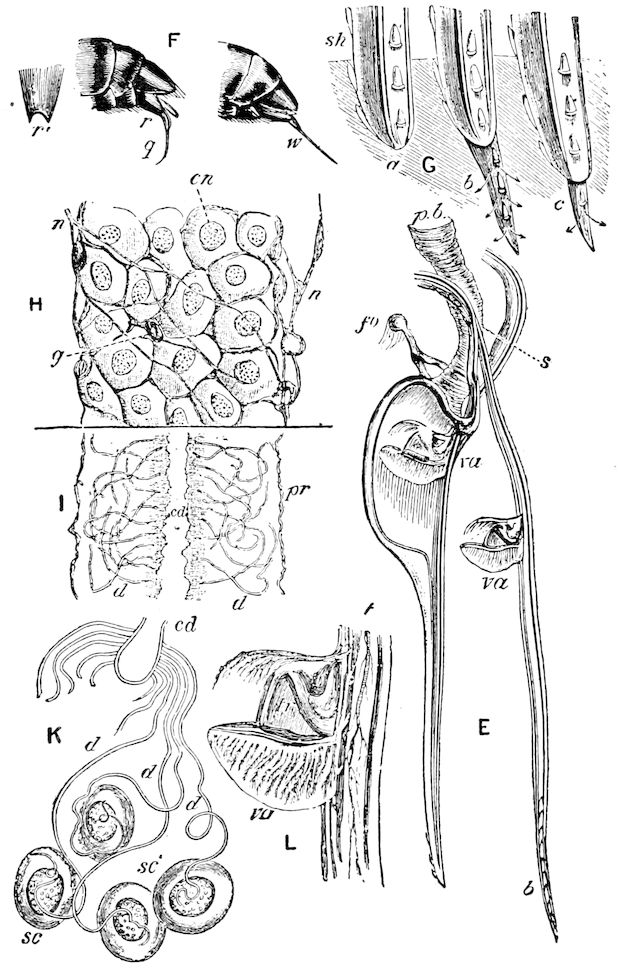
Fig. 195.—Details of sting of bee: E, darts, sheath, and valves; pb, poison-bag duct; fo, fork; s, slide piece; va, valve; b, barbs. F, terminal abdominal segments; w, worker’s sting; q, queen’s sting; r, r′, anal plate; G, sting entering skin; sh, sheath; a, b, c, positions in first, second, and third thrusts with the sting. H, portion of poison gland, × 300; cn, cell nucleus; n, nerve; g, ganglionic cell. I, portion of the poison gland, cells removed; cd, central duct; d, individual small ducts; pr, tunica propria. K, gland of Formica rufa; cd, central duct; d, small ducts; sc, secreting cells. L, valve and support; t, trachea; va, valve; tr, truss or valve-prop.—After Cheshire.
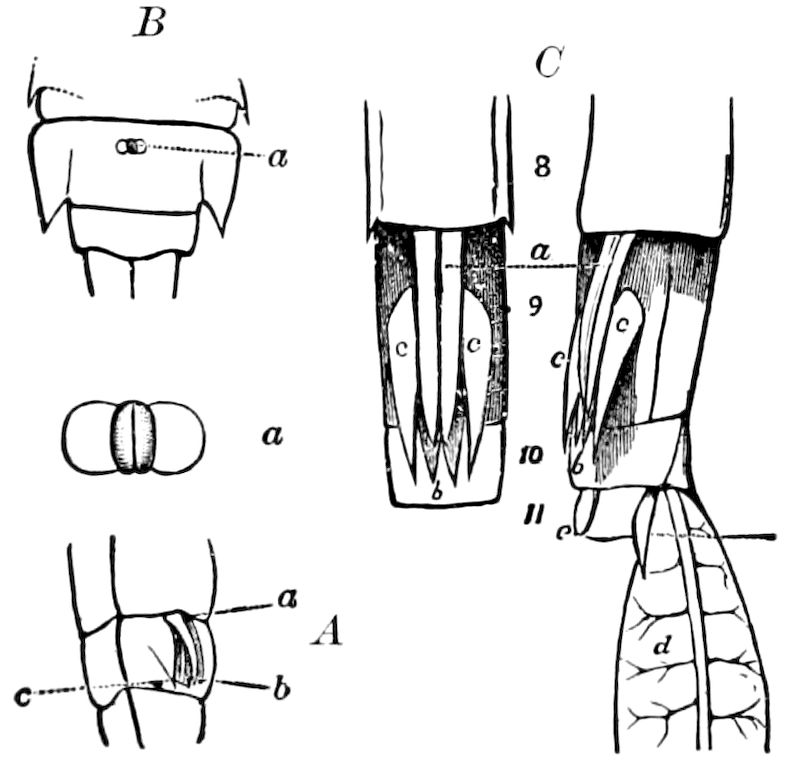
Fig. 196.—A, rudimentary ovipositor of nymph of Æschna. B, the corresponding ♂ structures; a, enlarged. C, ovipositor of nymph of Agrion; d, gill.
The male genital armature in the bees is originally composed of three pairs of tubercles, homologous with those of the female, all originally arising from three abdominal segments, two afterward being anterior, and the third pair nearer the base of the abdomen.
The ovipositor of the dragon-flies (Odonata) is essentially like that of the Orthoptera and Hymenoptera. Thus in Æschna (Fig. 196), Agrion (Fig. 196, C), and also in Cicada it consists of a pair of closely appressed ensiform processes which grow out from under the posterior edge of the eighth uromere and are embraced between two pairs of thin lamelliform pieces of similar form and structure.
The styles and genital claspers (Rhabdopoda).—Other appendages of the end of the abdomen of pterygote insects, and generally, if not always, arising from the ninth segment, are the clasping organs, or rhabdopoda as we may call them, of Ephemeridæ (Fig. 197), Neuroptera (Corydalus [Fig. 198], Myrmeleon, Rhaphidia), Trichoptera, 177Lepidoptera, Diptera, and certain phytophagous Hymenoptera. They do not appear to occur in insects which are provided with an ovipositor. In Thysanura the styles are present on segments 1–9 (Fig. 179). Those of the male Ephemeridæ, of which there are two pairs arising from the ninth segment, are remarkable, since they are jointed, and they serve to represent or may be the homologues of two of the pairs of stylets composing the ovipositor of insects of other orders. The lower pair (Fig. 197, rh) are either 2–, 3–, or 4–jointed (in Oniscigaster 5–jointed), while those of the upper pair are 2–jointed (rh′). These rhabdopods in the ephemerids are evidently very primitive structures, since they approach nearest in shape and in being jointed to the abdominal legs of Scolopendrella and the Myriopoda. The styles of the Orthoptera are survivals of the embryonic appendages of the ninth segment (Wheeler, etc.). In Mantis they are seen to have the same relations as the cerci, as shown by Heymons (Fig. 200).
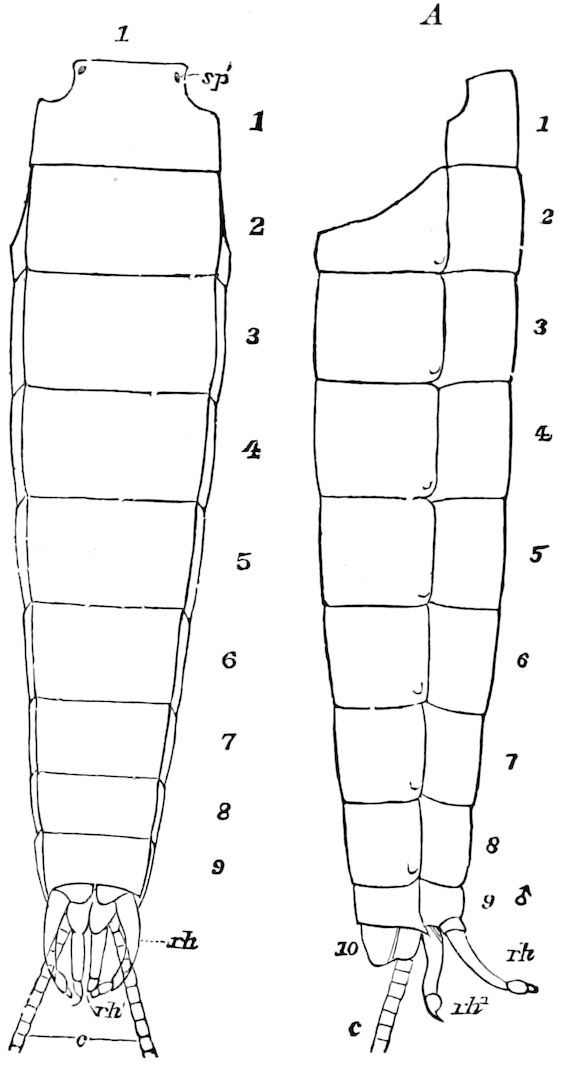
Fig. 197.—Abdomen of Ephemera (Leptophlebia) cupida, ♂: c, base of cercopoda; rh, outer 3–jointed claspers or rhabdopods; rh′, inner pair. A, side view.
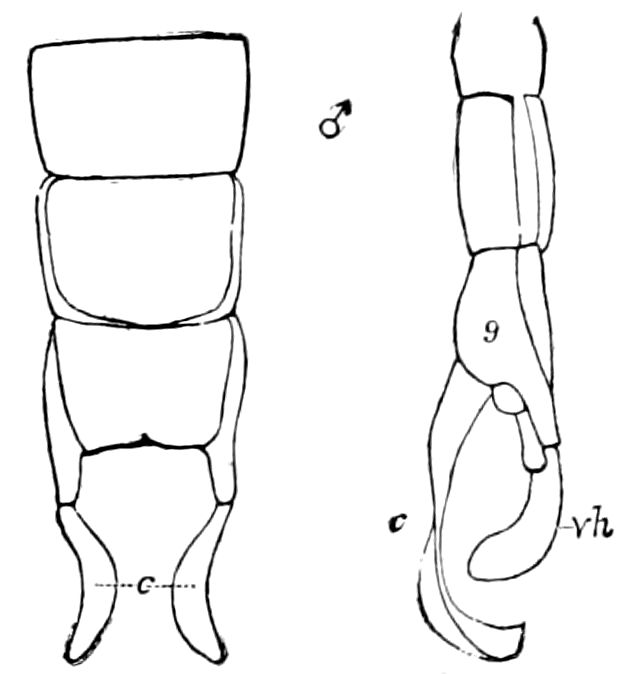
Fig. 198.—End of abdomen of Corydalus cornutus, ♂: vh, rhabdopod; c, cercopod.
In the Phasmidæ, in Anabrus, and in the Odonata the cercopods, which are not jointed, are converted into claspers, and in the Odonata the claspers are spiny within, so as to give a firmer hold. The suranal plate is apparently so modified as to aid in grasping the female. In nearly all the Trichoptera there are, besides the suranal plate, which is sometimes forked (Nosopus), a pair of superior and of inferior claspers, and in certain genera 178(Ascalaphomerus, Macronema, Rhyacophila, Hydropsyche, Amphipsyche, Smicridea, and Ganonema) the lower pair are 2–jointed like those of Ephemeridæ. The number of abdominal segments in the adult Trichoptera is nine, and McLachlan states that the genital armature consists of three pairs of appendages, i.e. a superior, inferior, and intermediate pair, besides the suranal plate (vestige of a tenth segment) and the penis. Judging by his figures, these three pairs of appendages arise from the last or ninth uromere, and the upper pair seem to be the homologue of the cercopoda of ephemerids. It needs still to be ascertained whether the intermediate pair is a separate set, or merely subdivisions of the upper or lower, and whether one of the latter may not arise from the penultimate segment, because we should not expect that the last segment should bear more than one pair of appendages, as we find to be the case in arthropods in general, and in the Neuroptera, from which the Trichoptera may have originated.
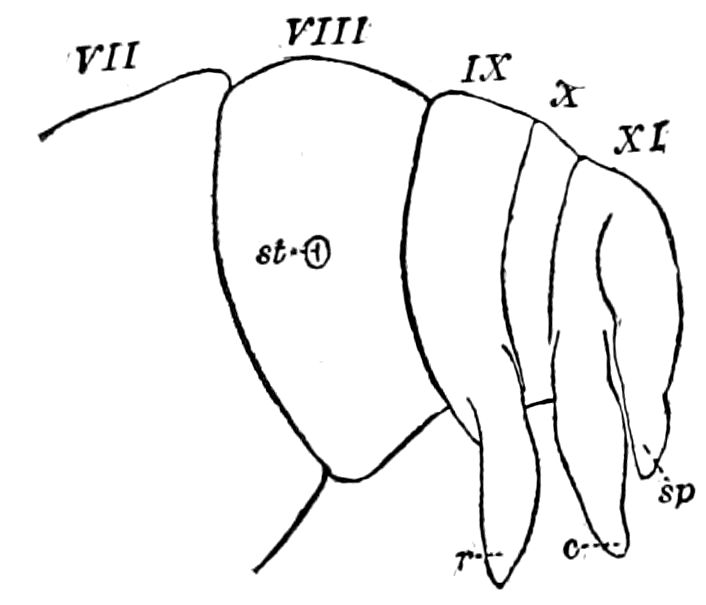
Fig. 199.—End of abdomen of embryo of Mantis: r, rhabdopod; c, cercopod; sp, suranal plate; st, stigma on 8th segment.—After Heymons.
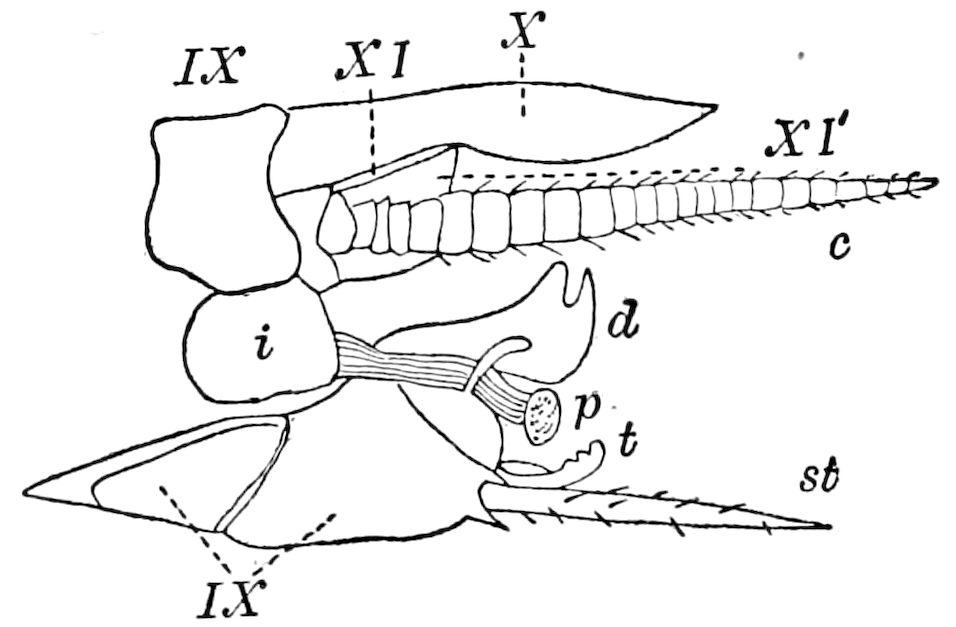
Fig. 200.—End of abdomen of Periplaneta americana, ♂, side view: c, cercopod; st, stilus; p, penis; t, titillator; d, “bird’s head” (clasper?); i, “oblong plate”; IX-XI, terminal segments; X, suranal plate; XI′, 11th sternite.—After Peytoureau.
In most larvæ of the Trichoptera, especially the Rhyacophilidæ and Hydropsychidæ, the last abdominal segment bears a pair of 2–jointed legs (cercopoda), ending in either one or two claws, which under various forms, sometimes forming long processes, persist in the pupa; and there appears to be a suranal plate, the vestige of the tenth uromere. In the pupa, judging by Klapálek’s figure of Leptocerus (248,9, 25), a pair of lateral spines arise which may in the imago form one of the pairs of appendages or styles. In the pupa of Œcetis furva his figure 289 shows two pairs of 1–jointed appendages arising from the last segment; whether the long dorsal or upper styles arise from the vestige of a more distal 179segment is not distinctly shown in Klapálek’s sketch. The origin of these elements of the genital armature evidently needs further study.
Whether the abdominal legs or so-called false or prop-legs of lepidopterous larvæ are genuine legs, homologous with those of the thorax and with the cephalic appendages, or whether they are secondary adaptive structures, is a matter still under discussion. That, however, they are true legs is shown by the embryology of the Lepidoptera, where there is a pair to each abdominal segment. It may also be asked whether the anal legs of lepidopterous larvæ are not the homologues of the 2–jointed anal appendages of caddis-worms.
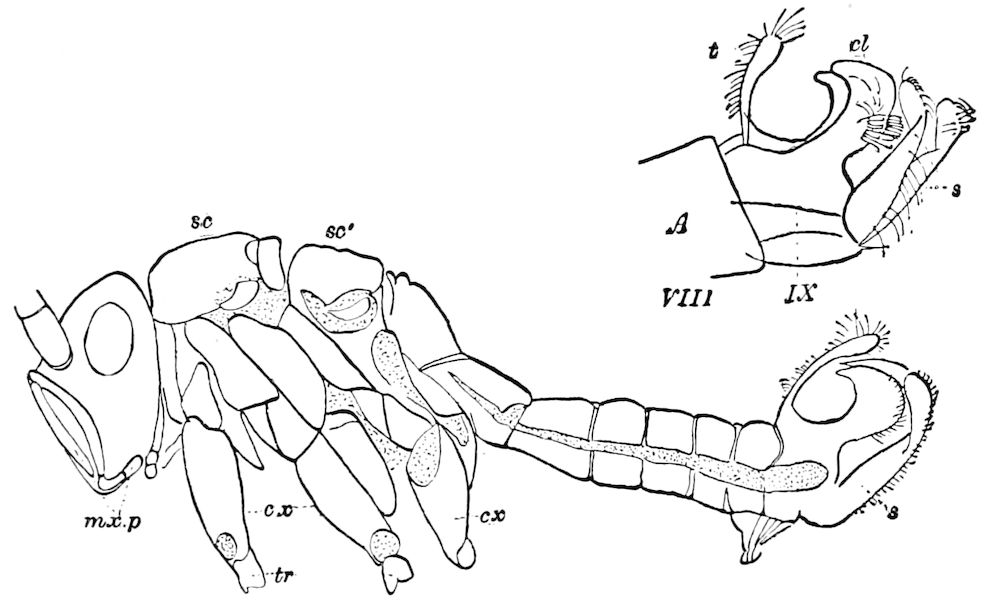
Fig. 201.—Eriocephala calthella, ♂, side view: t, palpiform suranal plate; cl, claspers; s, inferior claspers; mxp, maxillary palpi; cx. coxa; tr, trochanter; sc, scutum; sc′, scutellum.
In Lepidoptera, notably the male of the very generalized Eriocephala calthella (Fig. 201), besides the broad unjointed claspers, which are curved upward and provided with a brush of stiff hooked setæ (this upper pair being perhaps modified cercopods), there is an accessory lower slenderer pair, while the suranal plate (t) is palpiform or clavate and also adapted to aid in the action of the claspers. The examination of the cercopods and rhabdopods in the Trichoptera and in a generalized lepidopterous form like this enables one to understand the morphology of the genital armature, since it consists, besides the suranal plate, which is often deeply forked (in Sphingidæ, Smith), of a pair of modified hook-like cercopoda, and in some cases (Eriocephala) of an additional pair of claspers which 180may be the homologues of the ephemerid rhabdopods. A pair of hooks, often strong and claw-like (harpes), are situated, one near the base on the inside of each clasper; they are especially developed in the Noctuidæ (Smith), and appear to be present in certain Trichoptera, but this remains to be proved. This complicated apparatus of claspers and hooks is utilized by those insects which pair while on the wing, and is wanting in such forms as Coleoptera and Hemiptera. Besides the forceps of Panorpa, there are two pairs of slender filiform appendages which need farther examination. In the Diptera, especially Tipulidæ, there is a pair of 2–jointed appendages or forceps, as in Limnophila (Osten Sacken). The male genital armature of Diptera appears to be on the same general plan as in Lepidoptera, but more complicated.
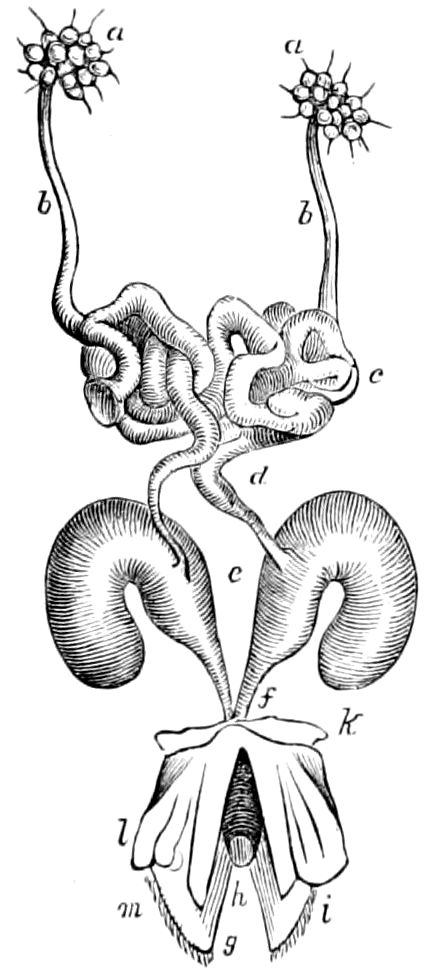
Fig. 202.—Male organs of generation of Athalia.—After Newport.
Notice should also be taken of the paired uncinate hooks which are modifications of the penis-sheath of the male of cockroaches (Phyllodromia), which Haase states appear to originate on the tenth ventral plate, and which probably “serve to open and dilate the vagina of the female, especially as a perforated penis, which is highly developed in Machilis, seems to be wanting in the Blattidæ.” (Haase.)
The penis.—This is a single or double median style-like structure either hollow and perforated, or solid, very variable in shape, receiving the end of the ejaculatory duct. It is usually enclosed between two lateral plates, the homologues perhaps of the inner pair of sheaths of the ovipositor. In the Coleoptera, as in Carabidæ and Melolonthidæ, the penis is a long chitinous tube, “retractile within the abdomen on the under surface as far as the anterior segments.” (Newport.) In the Hymenoptera, of which that of the saw-flies is a type, Newport states that it “consists of a short valvular projectile organ, covered externally by two pointed horny plates (i) clothed with soft hairs.” Above these are two other irregular double-jointed plates (Fig. 202, l) surrounded at their base by a chitinous ring (k); they are edged with prehensile hooked spines (i). Between these in the middle line are two elongated muscular parts (m) which enclose the penis (h), and which, like those in beetles, perhaps aid in dilating the vulva of the female.
An examination of Figs. 203–207 will aid in understanding the various modifications in beetles, etc., of this organ.
181A general study of the anatomy and homologies of the male genital armature, from a developmental point of view, together with a comparison of them with the corresponding female organs, is still needed.
Fig. 203.—End of abdomen situated under the anal lobes of Hydrophilus piceus, drawn out, seen from the ventral side: 6, sternal region of 6th segment; 7, 8, 9, segments telescoped, when retracted, in 6th segment; zw, membrane connecting 6th and 7th segments; G, intromittent apparatus; vl, external lobes; vlu, inner lobes; pn, penis.
Fig. 204.—The same as in Fig. 203, seen from the side: 6, the free 6th segment; 7–10, the four last, when at rest, retracted and telescoped within the 6th segment, with the copulatory apparatus (g); vl, outer, vlu, inner lobe; 10s, tergite of 10th segment; 10i, sternite of the same; an, anal opening.
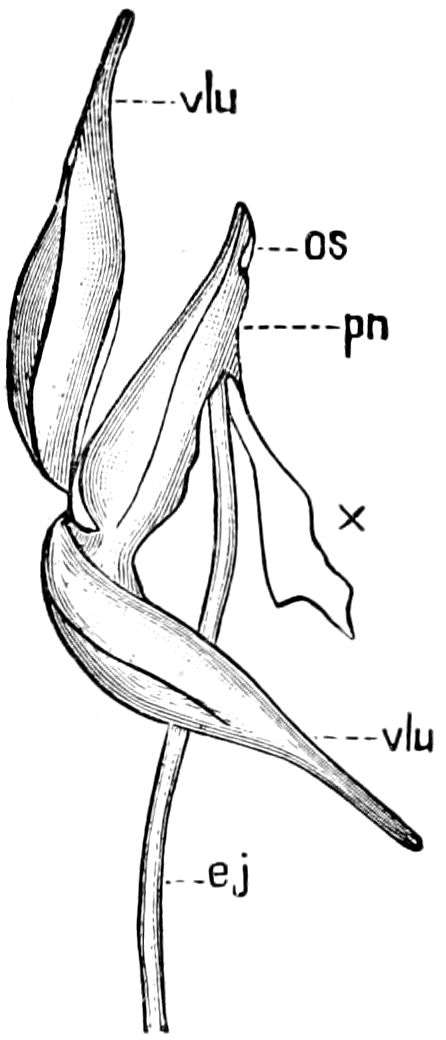
Fig. 205.—Terminal parts of the male copulatory apparatus of Hydrophilus piceus, torn apart: vlu, the two inner lodes; pn, penis; x, membrane torn from under side of penis; ej, ejaculatory duct; os, its opening on the under side of the penis, directly under its tip. The muscles, tracheæ, and nerves are not drawn.
Velum penis.—In the locusts (Acrydiidæ) the penis is concealed by a convex plate, flap, or hood, free anteriorly and attached posteriorly and on the sides to the ridge forming the upper edge of the tenth sternite. When about to unite sexually, the tip of the abdomen is depressed, the hood is drawn backward, uncovering the chitinous penis.
The suranal plate.—This is a triangular, often thick, solid plate or area, the remnant of the tergum of the last, usually tenth, segment of the abdomen, the supra-anal or suranal plate, or anal operculum (lamina supra-analis) of Haase. In most lepidopterous larvæ this plate is well marked; in those of the Platypteridæ it is remarkably elongated, forming an approach to a flagellum-like terrifying 182appendage, and in that of Aglia tau it forms a long, prominent, sharp spine. In the cockroach, both Cholodkowsky and Haase maintain that the tenth abdominal segment is suppressed in the female, the tergal portion being fused with the suranal plate (the latter in this case, as we understand it, being the remnant of the eleventh segment of the embryo). As to the nature of the middle jointed caudal appendage in Thysanura and May-flies Heymons has satisfactorily shown that it is a hypertrophied portion of the suranal plate, being in Lepisma but a filamental elongation of the small eleventh abdominal tergite.
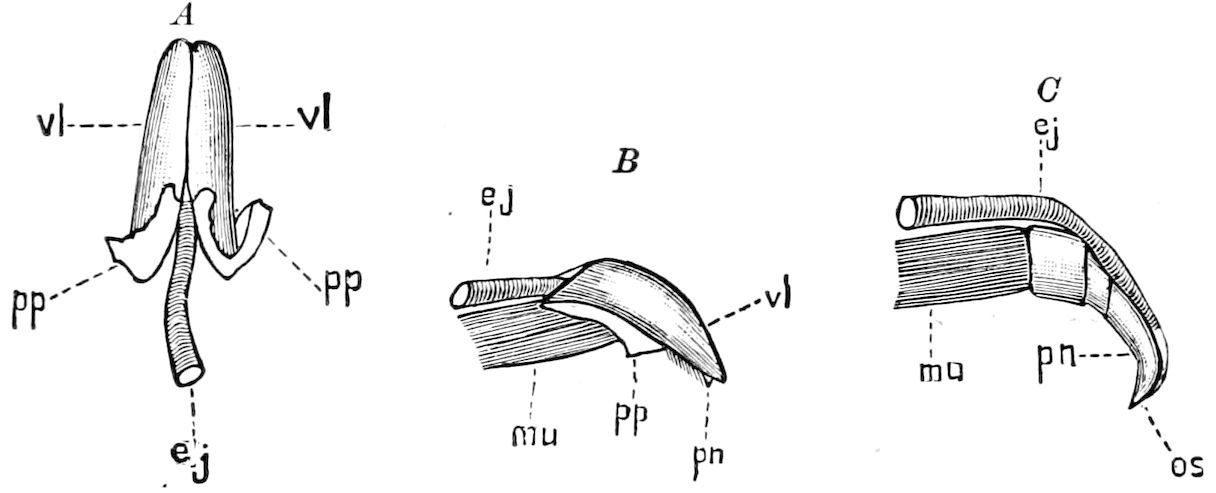
Fig. 206.—Copulatory organ of a weevil, Rhychophorus phœnicis, seen from above. A, vl, the lobes united into a capsule; pp, torn membrane which connects the capsule with the 9th abdominal segment; ej, ejaculatory duct. B, the same seen from the side; mu, end of the muscle of the penis. C, the same as B, without the capsule; os, opening of the ejaculatory duct (ej). Other letters as in A.
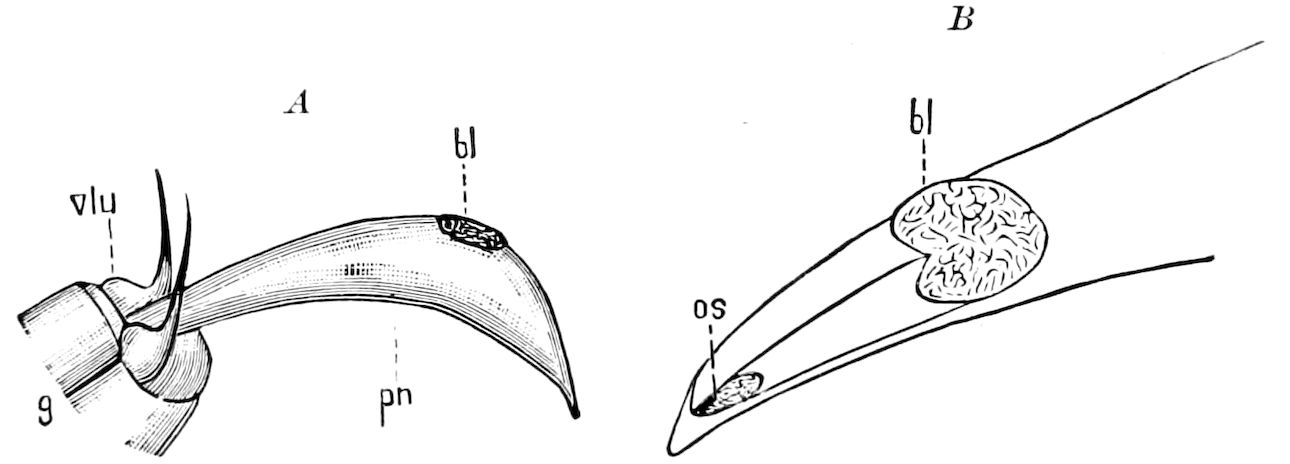
Fig. 207.—A, penis (pn) of Carabus hortensis: bl, wrinkled membranous vesicle; vlu, the valves; g, part of 9th segment. B, end of penis of the same, enlarged; os, cleft-like opening; also a wrinkled vesicle, as at bl.—This and Figs. 203–205 after Kolbe.
At the base of the suranal plate of locusts (Acrydiidæ) is the suranal fork or suranal furcula (furcula supra-analis, as we have called it) (Fig. 88, 89, f).
The podical plates or paranal lobes.—In the cockroach and other insects, also in the nymphs of Odonata, the anus is bounded on each side by a more or less triangular plate, the two valves being 183noticeable in lepidopterous larvæ. They are the valvulæ of Burmeister, and podical plates of Huxley, who also regarded them as the tergites of an eleventh abdominal segment;[38] and the subanal laminæ of Heymons. They are wanting in Ephemeridæ.
The infra-anal lobe.—Our attention was first called to this lobe or flap, while examining some geometrid larvæ. It is a thick, conical, fleshy lobe, often ending in a hard, chitinous point, and situated directly beneath the vent. Its use is evidently to aid in tossing the pellets of excrement away so as to prevent their contact with the body. The end may be sharp and hard or bear a bristle. Whether this lobe is the modified ventral plate of the ninth urite, we will not undertake at present to say.
The egg-guide.—In the Acrydiidæ the external opening of the oviduct is bounded on the ventral side by a movable, triangular, acute flap, the egg-guide (Fig. 88, B, eg). Whether this occurs in other orders needs to be ascertained.
LITERATURE ON THE ABDOMEN AND ITS APPENDAGES
a. General (including the cerci, stili, etc.)
Cornelius, C. Beiträge zur naheren Kenntnis von Palingenia longicauda. (Programm d. Real- u. Gewerbeschule zu Elberfeld, 1848, pp. 1–38, 4 Taf.)
Schiödte, J. G. Bemerkungen über Myrmecophilen. Ueber den Bau des hinterleibes bei einigen Käfergattungen. (Germar’s Zeits. f. Ent., 1844.)
—— De metamorphosi Eleutheratorum observationes. (Naturhist. Tidsskr. i-xiii, 1861–1883.)
Meinert, F. Anatomia Forficularum. Anatomisk undersogelse af de Danske Orentviste, i. (Naturhist. Tidsskr. 3 raekke, ii, 1863, pp. 427–482, 1 Pl.)
—— Om dobbelte Saedgange hos insecter. (Naturhist. Tidsskrift, 3 raekke, v, 1868.)
Tullberg, Tycho. Sveriges Podurider. (Svenska Vetensk.-Akad. Handl., 1872, x, Nr. 10, 12 Pls.)
Davis, H. Notes on the pygidia and cerci of insects. (Journ. R. Micr. Soc., 1879, ii.)
Westhoff, F. Ueber den Bau des Hypopygiums der Gattung Tipula Meig. (Münster, 1882, pp. 1–62, 6 Taf.)
Saunders, Edward. Further notes on the terminal segments of aculeate Hymenoptera. (Trans. Entom. Soc. London, 1884, pp. 251–267, 1 Pl.)
Palmén, J. A. Ueber paarige Ausführungsgange der Geschlechtsorgane bei Insekten. (Helsingfors, 1884, 5 Taf.)
Haase, E. Die Abdominalanhänge der Insekten mit Berücksichtigung der Myriopoden. (Morpholog. Jahrbuch, 1889, xv, pp. 331–435, 2 Taf.)
—— Abdominalanhänge bei Hexapoden. (Sitzungsber. d. Gesellsch. naturforsch. Freunde, 1889, pp. 19–29.)
184Wheeler, William M. On the appendages of the first abdominal segment of embryo insects. (Trans. Wis. Acad. Sc., viii, 1890, pp. 87–140, 3 Pls.)
Janet, Charles. Études sur les fourmis, 5e note, sur la morphologie du squelette des segments post-thoraciques chez les Myrmicides. (Mém. Soc. Acad, de l’Oise, xv, pp. 591–611, Figs. 1–5, 1894.)
Heymons, Richard. Die Segmentirung des Insektenkörpers. (Anh. Abh. Akad. Berlin, Phys. Abh., pp. 39, Taf., 1895. See also Die Embryonalent, von Dermapteren und Orthopteren, under Embryology.)
—— Grundzüge der Entwicklung und des Körperbaues von Odonaten und Ephemeriden. (Abhandl. k. Preuss. Akad. d. Wissens. Berlin, 1896, 2 Taf., pp. 1–66.)
—— Zur Morphologie der Abdominalanhänge bei den Insecten. (Morphol. Jahrb., iv, 1896, pp. 178–203, 2 Taf.)
Peytoureau, S. A. Contribution à l’étude de la morphologie de l’armure génital des insectes. (Bordeaux, 1895, pp. 248, 22 Pls., 43 Figs. in text.)
Verhoeff, C. Cerci und styli der Tracheaten. (Ent. Nachr., xxi, pp. 166–168, 1895.)
Also the writings of Oudemans, Packard, etc.
b. The ovipositor
Lacaze-Duthiers, Henri. Recherches sur l’armure génitale femelle des insectes. (Ann. d. sc. natur., 1849, xii, pp. 353–374, 1 Pl.; 1850, xiv, pp. 17–52, 1 Pl. (Hyménoptères); 1852, xvii, pp. 206–251, 1 Pl. (Orthoptères); 1853, xix, pp. 25–88, 4 Pl. (Neuroptères, Thysanures, Coléoptères, Diptères); pp. 203–237 (Lépidoptères, Aphaniptères en general. Also separate.))
Sollmann, A. Der Bienenstachel. (Zeitschr. f. Wissens. Zool., xiii, 1863, pp. 528–540, 1 Taf.)
Fengger, H. Anatomie und Physiologie des Giftapparates bei den Hymenopteren. (Archiv f. Naturgesch., 1863, pp. 139–178, 1 Taf.)
Eaton, A. E. Remarks upon the homologies of the ovipositor. (Trans. Ent. Soc. London, 1868, pp. 141–144.)
Packard, A. S. On the structure of the ovipositor and homologous parts in the male insects. (Proc. Boston Soc. Nat. Hist., xi, 1868, pp. 393–399, Figs. 1–11.)
Lambrecht, A. Samtliche Teile des Stechapparates in Bienenkörper und ihre Verwendung zu technischen und vitalen Zwecken. (Bienemwirtsch. Centralbl., 7 Jahrg., 1871, pp. 5–11.)
Kraepelin, K. Untersuchungen über den Bau, Mechanismus und die Entwicklung des Stachels der bienenartigen Tiere. (Zeitschr. f. wiss. Zoologie, xxiii, 1873.)
Dewitz, H. Vergleichende Untersuchungen über Bau und Entwicklung des Stachels der Honigbiene und der Legescheide der grünen Heuschrecke. (Königsberg, 1874.)
—— Ueber Bau und Entwicklung des Stachels und der Legescheide einiger Hymenopteren und der grünen Heuschrecke. (Zeitschr. f. wissen. Zoologie, xxv, 1874, pp. 174–200, 2 Taf.)
—— Ueber Bau und Entwicklung des Stachels der Ameisen. (Zeitschr. f. wiss. Zoologie, 1877, xxviii, pp. 527–556, 1 Taf.)
Adler, H. Legeapparat und Eierlegen der Gallwespen. (Deutsche Entom. Zeitschr., 1877, xxi Jahrg., pp. 305–332, 1 Taf.)
Cholodkowsky, N. Ueber den Hummelstachel und seine Bedeutung für die Systematik. (Zool. Anzeiger, vii, 1884, pp. 312–316.)
185Ihering, H. von. Der Stachel der Meliponen. (Ent. Nachr., 1886, xii, Jahrg., pp. 177–188, 1 Taf.)
Meinert, F. Bidrag til de danske Myrers Naturhistorie. (Kjöbenhavn, 1890, 68 s.u. Danske Vidensk. Selsk. Skrifter, 5 Raek, v, 3 Pls.)
Beyer, O. W. Der Giftapparat von Formica rufa ein reduziertes Organ. (Jena. Zeitschr. Naturw., 1890, xxv, pp. 26–112, 2 Taf.)
Carlet, G. Mémoire sur le venin et l’aiguillon de l’abeille. (Ann. d. sc. nat. Zool., 7 sér., ix, 1890, pp. 1–17, 1 Pl.)
Künckel d’Herculais, J. Méchanisme physiologique de la ponte chez les insectes orthoptères de la famille des Acridides.—Rôle de l’air comme agent mécanique et fonctions multiples de l’armure génitale. (Compt. Rend., cxix, pp. 244–247, 1894.)
Also the writings of Verhoeff, Heymons.
c. The external genital armature
Klug, Johann C. F. Versuch einer Berichtigung der Fabriciusschen Gattungen Scolia u. Tiphia. (Ueber u. Mohr Beiträge zur Naturkunde, i, pp. 8–40, 1805, 1 Taf.)
Audouin, J. V., and Lachat. Observations sur les organes copulateurs males des Bourdons. (Annal. général, d. sc. phys., 1821, viii, pp. 285–289.)
Audouin, J. V. Lettre sur la génération des insectes. (Ann. des sc. nat., sér. 1, ii, 1824.)
Suckow, F. W. L. Geschlechtsorgane der Insekten. (Heusinger, Zeitschr. organ. Physik., 1828, ii, pp. 231–264, 1 Taf.)
Rathke, H. De Libellularum partibus genitalibus. Regiomonti, 1832, pp. 6 + 38, 3 Pls.
Siebold, C. Th. E. von. Ueber die Fortpflanzungsweise der Libellulinen. (Germar’s Zeitschr. f. Ent., 1840, ii, pp. 421–438.)
Selys-Longchamps, E. de. Monographie des Libellulidees d’Europe. 4 Pls., 1840.
Bassi, C. A. Studi sulle funzioni degli organi genitali degl’ insetti da lui osservati piu specialmente nella Bombyx mori. (Atti della 5 Riun. d. Scienz. Ital. Lucca, 1844, pp. 39–94.)
Stein, F. Vergleichende Anatomie und Physiologie der Insekten, i. Die weiblich. Geschlechtsorgane der Käfer. Berlin, 1847, 9 Taf.
Ormancey, P. Recherches sur l’étui penial considéré comme limite de l’espèce dans les coléoptères. (Ann. sc. nat., 1849, 3 sér., Zool., xii, pp. 227–242.)
Roussel, C. Recherches sur les organes génitaux des insectes coléoptères de la famille des Scarabéides. (Compt. rend. Acad. d. sc. Paris, 1860, l, pp. 158–161.)
MacLachlan, R. A monographic revision and synopsis of the Trichoptera of the European fauna. London, 1874–80, 59 Pls.
—— On the sexual apparatus of the male Acentropus. (Trans. Ent. Soc. London, 1872, pp. 157–162.)
Thomson, C. G. Nagra anmarkningar ofver arterna af slagtet Carabus. (Thomson’s Opuscula Entomologica, vii, 1857, pp. 615–729, 1 Pl.)
Dufour, L. Sur l’appareil génital male du Coræbus bifasciatus. (Thomson, Archiv ent., 1857, i, pp. 378–381.)
White, F. Buchanan. On the male genital armature in the Rhopalocera. (Trans. Linn. Soc., ser. 1, Zool., i, pp. 357–369, 1876, 3 Pls.)
Graber, V. Die Aehnlichkeit im Baue der ausseren weiblichen Geschlechtsorgane bei den Lokustiden und Akridiern auf Grund ihrer Entwicklungsgeschichte. (Sitzber. k. Akad. d. Wissensch. Wien., 1870, lxi, pp. 1–20, 1 Taf.)
186Scudder, Samuel H., and Edward Burgess. On asymmetry in the appendages of hexapod insects, especially as illustrated in the lepidopterous genus Nisoniades. (Proc. Boston Soc. Nat. Hist., 1871, xiii, pp. 282–306.)
Chadima, J. Ueber die Homologie zwischen den männlichen und weiblichen ausseren Sexualorganen der Orthoptera Saltatoria Latr. (Mitteil. d. naturwiss. Vereins f. Steiermark, 1872, pp. 25–33, 1 Taf.)
Hagens, von. Ueber die Genitalien der männlichen Bienen, besonders der Gattung Sphecodes. (Berlin Ent. Zeitschr., 1874, pp. 25–43.)
—— Ueber die männlichen Genitalien der Bienengattung Sphecodes. (Deutschen Entom. Zeitschr., 1882, pp. 209–228, 2 Taf.)
Lindenman, C. Vergleichend-anatomische Untersuchung ueber das männliche Begattungsglied der Borkenkäfer. (Bull. Soc. Imp. d. Natural. Moscou, 1875–77.)
Forel, A. Der Giftapparat und die Analdrüsen der Ameisen. (Zeitschr. f. wiss. Zoologie, xxx, suppl., 1878.)
Kraatz, G. Ueber die Wichtigkeit der Untersuchung des männlichen Begattungsgliedes der Käfer für die Systematik und Artunterscheidung. (Deutschen Entom. Zeitschr., 1881, xxv, pp. 113–126.)
—— Ueber das männliche Begattungsglied der europaischen Cetoniiden und seine Verwendbarkeit für deren scharfe spezifische Unterscheidung. (Ibid., pp. 129–149.)
Gosse, Ph. H. On the clasping-organs ancillary to generation in certain groups of the Lepidoptera. (Trans. Linn. Soc., 1882, Ser. 2, Zool., ii, pp. 265–345, 8 Pls.)
—— The prehensors of male butterflies of the genera Ornithoptera and Papilio. (Proc. Roy. Soc. London, 1881, xxxiii, pp. 23–27.)
Radoszkowski, O. Revision des armures copulatrices des males du genre Bombus. (Bull. Soc. Natur. Moscou, 1884, xlix, pp. 51–92, 4 Pls.)
—— Revision des armures copulatrices des males de la tribu Philérémides. (Ibid., 1885, lxi, pp. 359–370, 2 Pls.)
—— Revision des armures copulatrices des males de la famille des Mutillidæ. (Horæ Soc. Ent. Ross., 1885, xix, pp. 3–49, 9 Pls.)
—— Revision des armures copulatrices des males de la tribu des Chrysides. (Horæ Soc. Ent. Ross, xxiii, 1890, pp. 3–40, 6 Pls.)
Hofmann, O. Beiträge zur Kenntnis der Butaliden. (Stett. Ent. Zeit., 1888, pp. 335–347, 1 Taf.)
Driedzichi, H. Revue des espèces européennes du genre Phronia Winn. (Horæ Soc. Ent. Ross., 1889, xxiii, pp. 404–532, 10 Pls.)
Sharp, David. On the structure of the terminal segment in some male Hemiptera. (Trans. Ent. Soc. London, 1890, pp. 399–427, 3 Pls.)
Escherich, K. Die biologische bedeutung der Genitalanhänge der Insekten. (Verhandl. d. zool. bot. Ges. Wien., 1892.)
—— Anatomische Studien über das männliche Genitalsystem der Coleopteren. (Zeits. f. wissens. Zool., lvii, pp. 620–641, 1 Taf., 3 figs.)
Verhoeff, C. Zur vergleichenden Morphologie der “Abdominalanhänge” der Coleopteren. (Ent. Nachr., xx, pp. 93–96, 1894. Compare also O. Schwarz and J. Weise’s criticisms in D. Ent. Zeit., pp. 153–157; also pp. 101–109, 155–157. Zool. Anzeiger, pp. 100–106, 1894.)
—— Vergleichende—morphologische Untersuchungen ueber das Abdomen der Endomychiden, etc., und über die Musculature des Copulationsapparates von Triplax. (Archiv f. Naturg., lxi, pp. 213–287, 2 Taf., 1895.)
—— Vergleichende Untersuchungen über die abdominal Segmente der weiblichen Hemiptera-Heteroptera und Homoptera. (Verh. Nat. Ver. Bonn, l, pp. 307–374, 1894.)
187Verhoeff, C. Beiträge zur vergleichenden Morphologie des Abdomens der Coccinelliden, etc. (Archiv f. Naturg., lxi, pp. 1–80, 6 figs., 1895.)
Boas, J. E. V. Organe copulateur et accouplement der hanneton. (Oversigt over det K. Danske Vidensk. Selskab Forhand, 1892, Copenhagen, 1893, 1 Pl., pp. 239–261.)
Pérez, J. De l’organe copulateur mâle des Hyménoptères et de sa valeur taxonomique. (Ann. Soc. Ent. France, lxiii, pp. 74–81, Figs., 1894.)
Goddard, Martha Freeman. On the second abdominal segment in a few Libellulidæ. (Proc. Amer. Philos. Soc., xxxv, pp. 205–212, January 11, 1897, 2 Pls.)
Also the writings of Eaton, Emery, Fischer, Forel, Géhin, Godart, Hagen, Joly, Koletani, Loew, Meinert, Mik, Nicolet, Osten Sacken, Pictet, Roussel, Schaeffer (1754), Schaum, Schenk, J. B. Smith, Thompson, Buchanan-White, Brunner von Wattle-Wyll, Weise, Wyenbergh.
The subject of copulation has been treated by Hoffer, Hartig, Schiedeknecht, Verhoeff, etc.
THE ARMATURE OF INSECTS: SETÆ, HAIRS, SCALES, TUBERCLES, ETC.
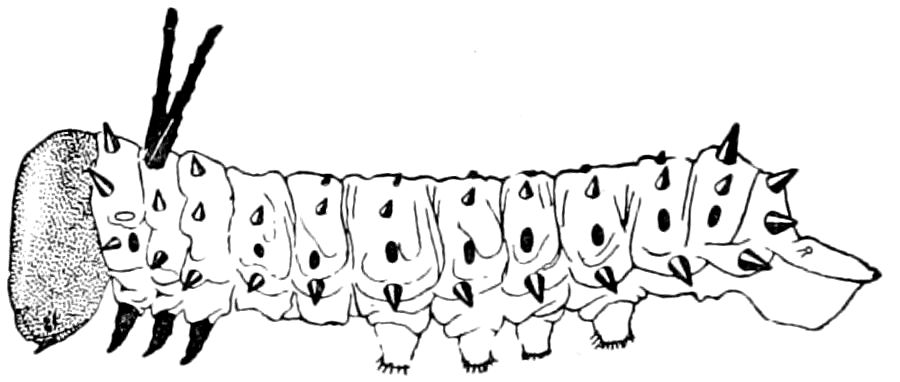
Fig. 208.—Larva of Dryocampa rubicunda, stage II.—Bridgham del.
The cuticula.—The integument is externally either smooth and shining or variously punctured, granulated, tuberculated, striated, or hairy. In certain orders the skin is clothed with flattened setæ or scales, while many forms, as some caterpillars (Figs. 208, 209), beetles (Fig. 210), etc., are protected by spines, horns, etc., these in adult insects often forming secondary sexual characters, usually being more developed in the males than in the females.

Fig. 209.—Larva of Hyperchiria io, on hatching.
The cuticula is not always smooth, but is often finely granulated or even minutely spinulated. On the abdominal segments of Anabrus, as observed by Minot, the cuticula is armed with microscopic conical nodules scattered irregularly over it. They do not correspond, he says, in any way to hairs; for they do not rest over pores, nor did he see any specially modified cells underlying them. “As far as I have observed, they are mere local irregularities, each nodule being apparently supported by some four or six unmodified epidermal cells.” Minot adds that the whole of the cuticula, except the cones just described and the hairs, is divided into numerous minute fields, each of which corresponds 188to a single cell of the underlying hypodermis. Each field is bounded by a distinct polygonal outline, and its surface is either covered by a large number of extremely minute projecting points, as on the dorsal arch of the segment, or is smooth, as upon the articular membrane and ventral arch. Upon the sides of the dorsal arch and upon the spiracular membrane each field has a projecting spine or sometimes two or even three. (See also pp. 28, 30.)
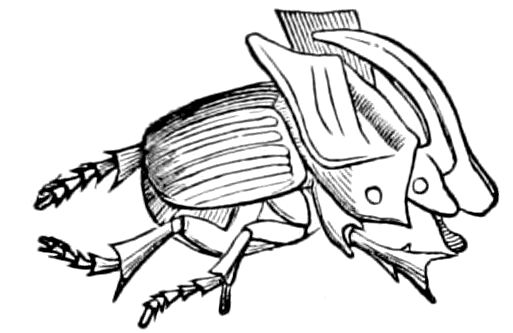
Fig. 210.—Phanæus pegasus, ♂, from Mexico.—After Graber.
Fig. 211.—Section of integument of Datana ministra: c, cuticula; hyp, hypodermis; p, outer pigmented nodulated layer.
The cuticle of lepidopterous larvæ has also been described and figured by Minot. In the caterpillars of different groups investigated by him, the cuticle was found to be rough with microscopic teeth or spinules, erect or flattened and scale-like, and either densely crowded or scattered, and affording excellent generic and specific characters. In the slug-worms (Limacodids) we have observed that the cuticula is unusually rough, especially on the spiniferous tubercle of Empretia, Parasa, etc. (Fig. 213, c). The skin of the body between the tubercles is seen to be finely shagreened, due to the presence of fine teeth, which are more or less curved and bent, these teeth arising from a very finely granulated surface (d). The cuticle of neuropterous, trichopterous, and tenthredinid larvæ will probably afford similar cases. The integument of the larva of Datana is, on the black bands, rough and nodulated, the irregular nodules being filled with a black pigment, and forming a layer (p) external to the true cuticula (Fig. 211).
Fig. 212.—Hairs of Datana: f, formative hair-cell; c, cuticula; p, pigmented layer; hy, hypodermis.
The integument of many insects contains fine canals passing through the chitinous layers and opening externally in minute pores. Certain of the pore-canals communicate with hollow setæ which sit directly over the pores; other pores form the external openings of dermal glands, but in many cases they are empty or only filled with air, and do not have any hairs connected with them. Each of these pores communicates with a hair-forming hypodermal cell, called by Graber a trichogen.
Setæ (“hairs” and bristles).—The setæ of insects are, as in worms, processes of the cuticle originating from certain of the hypodermal cells. They arise either from a ring-like pit, or from a minute tubercle, and are usually situated at the outlet of a pore-canal, which connects with an underlying cell of the hypodermis (Fig. 212). They are, then, bristle or hair-like processes arising from the hypodermis. Where the hairs or setæ are rubbed off, their site is indicated by a minute ring like a follicle in the 189chitinous integument. The cuticular hair, says Leydig, is in its first condition the secretion of the cellular element of the skin, and a thread-like continuation of the cell-body may rise up through the pore-canal into the centre of the hair, remaining there permanently.
While the setæ are usually simple, they are often branched, plumose, or spinulose, as in larval Hemerobiidæ, Anthrenus, and Dermestes, the larvæ of certain coccinellid beetles, notably Epilachna, and of Cassida, the larvæ of arctians, etc., and in bees (Anthophila, Megachile, Osmia, Colletes, Apis, etc.).
The use of these spinulose, plumose, and twisted hairs in the bees is clearly shown by J. B. Smith, who states that as these insects walk over flowers, the pollen grains adhere to the vestiture, “and this also accounts for the fact, probably noticed by every observant fruit-grower, that bees frequently bury themselves completely in the blossoms, or roll over every part of them. Such insects are after pollen, not honey, and by so rolling about, the pollen grains are brought into contact with and adhere to the surface of the insect.” The syrphid flies also pollenize flowers, the pollenizing of chrysanthemums being effected, as Smith states, by Eristalis tenax, and he adds that the body vestiture of the syrphids “is often composed of spurred and branched hairs.” (For reference to gathering hairs, see p. 45.)
Fig. 213.—Cuticular spinules of larva of Adoneta: a, b, c, d, different forms; e, e′, caltrops.
Certain remarkable spines occur in limacodid larvæ, notably Empretia and Adoneta. These we have called caltrops spines, from their resemblance to the caltrops formerly used in repelling the attacks of cavalry. They are largely concerned in producing the poisonous and irritating effects resulting from contact with the caterpillars of these moths, and are situated in scattered groups near the end of the tubercles. A group of three is represented at Fig. 213, e. They are not firmly embedded in the cuticle, but on the contrary 190appear to become very easily loosened and detached, and they probably, when brought into contact with the skin of any aggressor, burrow underneath, and are probably in part the cause of the continual itching and annoyance occasioned by these creatures. It will be seen by reference to Fig. 213, e′, that the body of the spine is spherical, with one large, elongated, conical spine arising from it, the spherical base being beset with a number of minute, somewhat obtuse spinules.
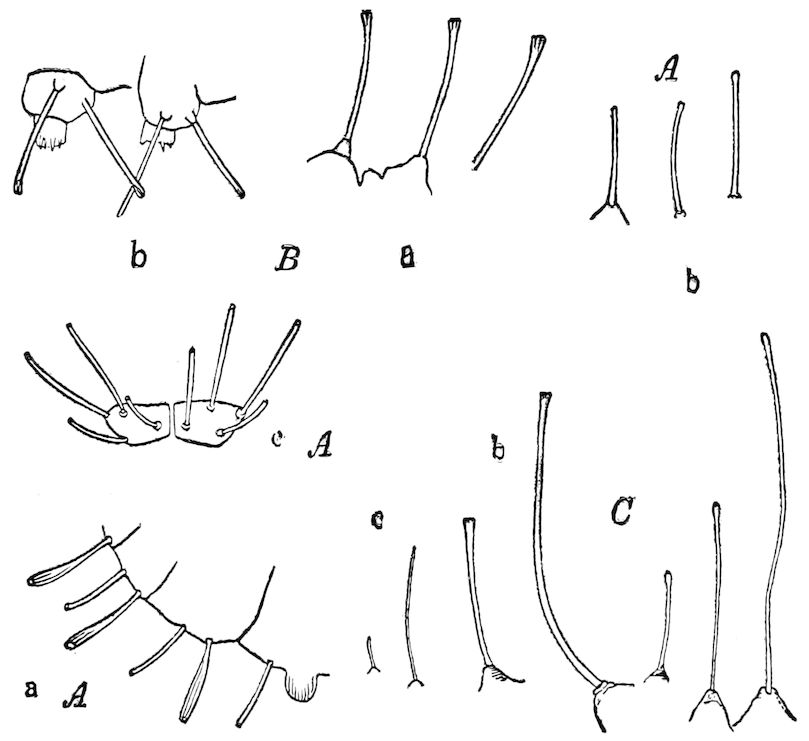
Fig. 214.—Glandular hairs of caterpillars. A, Dasylophia anguina: a, of body; b, of head; c, of prothoracic shield. B, Ceratosia tricolor: a, on body; b, on abdominal legs. C, Schizura ipomeæ: a, from third thoracic segment; b, from larva stage II; c, simple setæ from minute warts.
Glandular hairs and spines.—In some insects occur fine, minute, hollow setæ from which exude, perhaps through pore-canals of extreme fineness, droplets of a clear watery or plasma-like sticky fluid. The club-shaped tenent hairs of the feet of Collembola, and the hairs fringing the feet of Diptera, are modified glandular hairs. Here they serve to give out a sticky fluid enabling the insect to walk on smooth surfaces; they end in a vesicle-like bulbous expansion, which may contain numerous pore-canals. Those of caterpillars were first noticed by Zeller, and Dimmock has particularly described those of 191the larvæ of Pterophoridæ. They are either club-shaped, or variously forked at the end (Fig. 214, B, a). They are usually replaced after the first larval moult by ordinary, simple, solid, pointed setæ, and their use in caterpillars is as yet unknown. Whether these hairs, as seems most probable, arise from a specialized glandular hypodermal cell, or not, has not yet been discovered.
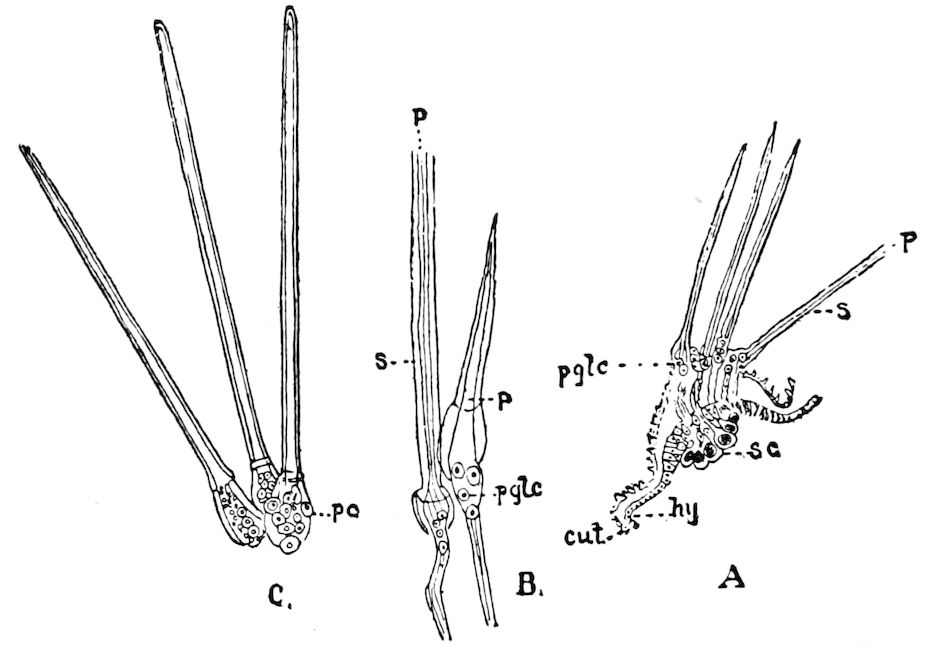
Fig. 215.—A, group of setæ arising from a subdorsal tubercle: cut, the cuticle; hy, the hypodermis; sc, the enlarged and specialized cells of the hypodermis which secrete the spines themselves; pglc, the nuclei which secrete the venomous fluid which fills the cavity of the seta (s), seen at p in a broken spine. B, a short entire, and a long broken seta (s-p); pgle, four poison cells; p, the poison in the hollow of the spine.
These temporary fine glandular hairs are probably the homologues of the larger true glandular bristles and spines of the later stages of certain lepidopterous larvæ, which are brightly colored and lead an exposed life, living through a large part of the summer. In these structures the bristles or spines are hollow, filled with a poisonous secretion formed in a single large, or several smaller specialized hypodermal cells situated under the base of the spine. In the venomous spines of Lagoa crispata the poisonous fluid in the larger spines (Figs. 215, C, 216, b) is secreted in several large cells situated at the base of the spine, and this is the usual form. In the finer spines of a large tubercle (Figs. 215, A, 216) there appears to be a differentiation of the hypodermal cells into two kinds, the large, basal deep-seated, setigenous cells (216, sc) and the poison-secreting nuclei (216, pglc) situated nearer the base of the setæ. The spines being filled with poison and breaking into bits in the skin of the hands or neck, cause great irritation and smarting. These nettling or poisonous hairs or spines are especially venomous in the larva of 192Orgyia, Empretia stimulea, Hyperchiria io, the larvæ of the saturnians (Fig. 217) and lasiocampids, etc. They rarely occur in insects of other orders, though the skin of Telephorus is said by Leydig to bear glandular hairs.
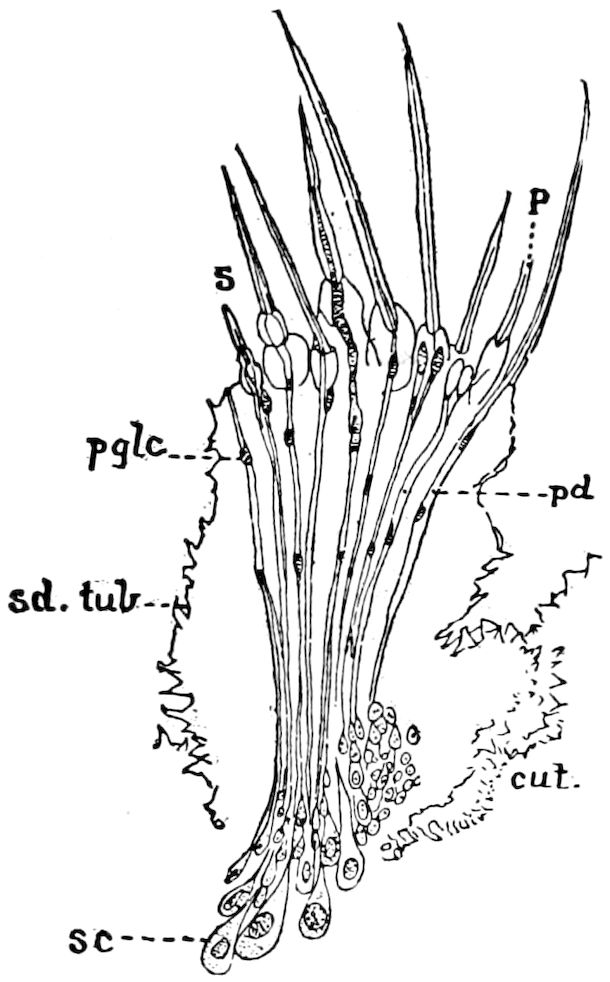
Fig. 216.—Section of a subdorsal tubercle from a larva in stage 1: sc, the setigenous cells, one for each seta; pglc, nuclei by which the poison is secreted; s, seta; p, poison in middle of a broken spine; cut, cuticle; sd, tub, spinulated surface of the subdorsal tubercle.
Leydig states that in the stout bristles of Saturnia there is, as in the integument of the body, a homogeneous cuticula, under which is the cellular matrix (hypodermis), and the clear contents (hyaloplasma) are secreted from the blood. The cell-structure of the hairs consist, as in the cells of the body, of spongioplasma and hyaloplasma. Leydig has observed the droplets of the secretion of the caterpillar of Saturnia carpini oozing through distinctly observable pores, and states that there are similar openings in the hairs and scales. Dewitz found easily observable openings at the end of the hair of a large exotic weevil (Fig. 130).
The advanced nymph of Psylla is also armed with clavate glandular hairs (Fig. 178).
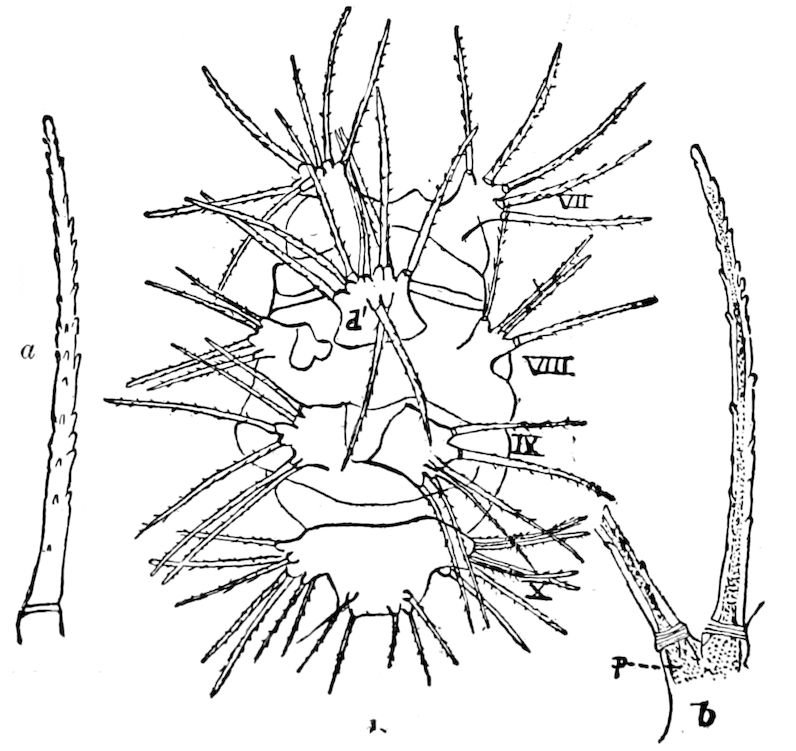
Fig. 217.—Armature of last four segments of Callosamia promethea: a, a dorsal seta; b, one showing the poison (p) within.
The tubercles are outgrowths of the body-walls; they are either smooth, warty, or spiny, as in many caterpillars. While the armature of insects is of little morphological 193importance, it is evidently of great biological importance, the welfare or even the life of the insect depending upon it; and it varies in each species of insect, especially in Diptera, where the position of even a single seta characterizes the species.
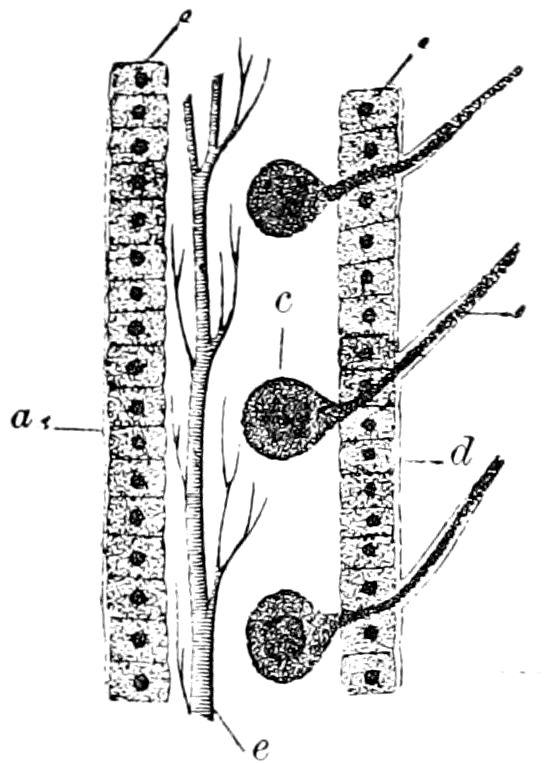
Fig. 218.—Section through an antennal pectination of Saturnia carpini: a, hypodermis, formative cells of the hairs (c); d, cuticula; e, trachea.—After Semper.

Fig. 219.—Flattened hairs from the lateral tufts of larva of Gastropacha americana: A, three from the lateral tuft of Heteropacha rileyana.
The mode of development of the hairs was first described by Semper. In the pectination of the antenna of Saturnia carpini he observed that the hairs arise, like the scales of the wings, from large round formative-cells lying in the cavity, which send out through the hypodermis and cuticle a long slender process which finally becomes the hair (Fig. 218).
Tactile hairs are those setæ arising over nerve cells or nerve terminations and will be discussed under the organs of sense.
Fig. 220.—The same in G. quercifolia: a, a small hair ending in two minute processes.
Scales.—In very rare cases the hairs of caterpillars (Fig. 219) are flattened and scale-like, and this passage in the same insect of cylindrical hairs into flattened scale-like ones, shows that the scales are only modified hairs. Also, as we shall see farther on, Semper has proved that their mode of origin is identical. While true scales are characteristic of Synaptera (Thysanura and Colembola), as well as Lepidoptera and Trichoptera, they also occur in the Psocidæ (Amphientomum), in 194many Coleoptera (Curculionidæ, Cleridæ, Ptinidæ, Dermestidæ, Byrrhidæ, Scarabæidæ, Elateridæ, and Cerambycidæ), and in the Culicidæ, and a few other Diptera, though they are especially characteristic of the Lepidoptera, not a species of this great order being known to be entirely destitute of them.
Fig. 221.—Flattened and spinulated hairs of tufts of larva of Acronycta hastulifera.
Fig. 222.—Scales from dorsal tuft, on second thoracic segment of larva of Gastropacha quercifolia.
The scales vary much in shape, but are more or less tile-like, attached to the surface of the body or wing by a short slender pedicel, and are more loosely connected with the integument than the hairs, which are thicker at the base or insertion than beyond.
The markings of the scales, both of Synaptera and Lepidoptera, are very elaborate, consisting of raised lines, ridges, or striæ with transverse ridges between. “The striæ of the transparent scales of Micropteryx are from about 500 to 300 to the millimetre, varying in different species. The 195opaque scales of Morpho, which show metallic reflections, have about 1400 striæ to the millimetre.” (Kellogg.)
The primary use of scales, as observed by Kellogg, is to protect the body, as seen in Synaptera and Lepidoptera. A nearly as important use is the production of colors and patterns of colors and markings, while in certain butterflies certain scales function as the external openings of dermal scent-glands, and they afford in some cases (as first claimed by Kettelhoit in 1860) generic and specific characters. Spuler has shown that the scales are strengthened by internal chitinous pillars. Burgess has observed in the scales of Danais plexippus that the under surface of the scales is usually smooth, or provided with few and poorly developed ridges, and this has been confirmed by Spuler and by Mayer (Fig. 226).
In the irised and metallic scales the ridges, says Spuler, are not divided into teeth, and they converge at the base to the pedicel and also toward the end of the scale (Micropteryx), or end in a single process beyond the middle (the brass-colored scales of Plusia chrysitis).
The arrangement of the scales on the wings is, in the generalized moths, irregular; in the more specialized forms they are arranged in bands forming groups, and in the most specialized Lepidoptera they are more thickly crowded, overlapping each other and inserted in regular rows crossing the wings, these rows either uniting with each other or running parallel. (Spuler.) The scattered irregular arrangement seen in Micropteryx is also characteristic of the Trichoptera and of Amphientomum.
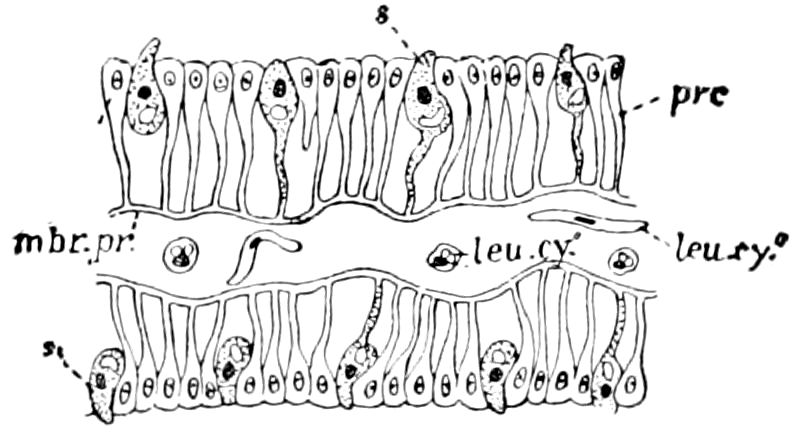
Fig. 223.—Portion of a longitudinal section through one of the young pupal wings of a summer pupa of Vanessa antiopa: s, young scale; leu. cy., leucocyte; mbr. pr., ground membrane; prc, hypodermis-cells.
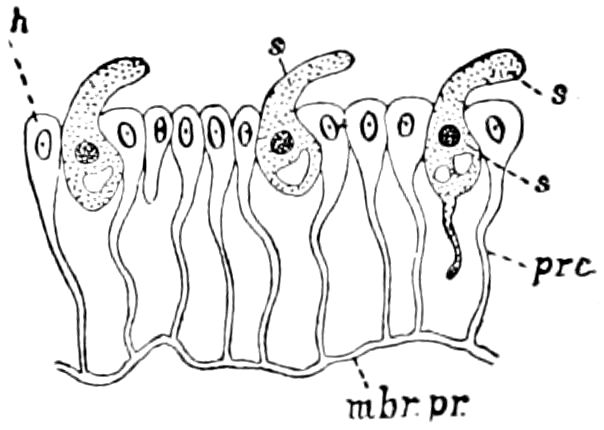
Fig. 224.—Portion of a longitudinal section through one wall only of the pupal wing of a specimen slightly older than that of Fig. 223; s, older scale.
Development of the scales.—The mode of origin of the scales was first worked out by Semper in 1886, who stated that in the wing of the pupal Sphinx and Saturnia they are seen, in sections, to arise from large roundish cells just under the hypodermis and which have a projection which passes out between the hypodermis (his “epidermis”) cells, expanding into a more or less spherical vesicle, the latter being the first indication of the future scale. He 196observed that the scales are not all formed at once, but arise one after another, so that on one and the same wing the scales are in different stages of development.
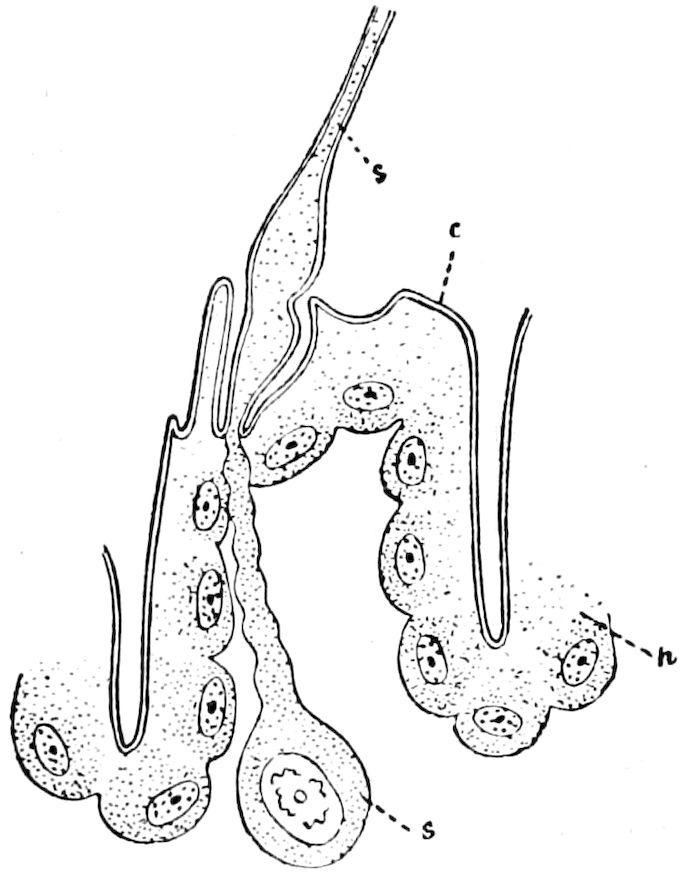
Fig. 225.—Portion of a longitudinal section through a pupal wing about eight days before emergence: s, formative scale-cell; upper s, a scale.
More recently Schaeffer has stated that the scales and also the hairs are evaginations of greatly enlarged hypodermis cells, and still more complete evidence has been afforded by A. G. Mayer (1896). In the wings of Lepidoptera, about three weeks before the imago emerges, certain of the hypodermis cells, which occur at regular intervals, begin to increase in size and to project slightly above the level of the hypodermis; these are Semper’s “formative cells,” and are destined to secrete the scales. They increase in length, and appear as in Fig. 223. In the next stage observed, the projections are much longer (Fig. 224). The hypodermis is now thrown up into a regular series of ridges, which run across the wing. Each ridge, says Mayer, corresponds in position with a row of formative cells, and each furrow with the interval between two adjacent rows. The scales always project from the tops of these ridges. The ground or basal membrane has not participated in this folding, and the deep processes of the hypodermis (prc) that once extended to this membrane have largely disappeared. Figure 225 represents a more advanced stage almost eight days before the emergence of the imago.
The scales are originally filled with protoplasm, which gradually withdraws, leaving behind it little chitinous bars or pillars which serve to bind together the upper and lower surfaces of the scales, and finally the scales become “merely little flattened hollow sacs containing only air.” As Mayer shows (Figs. 226, 227), from the study of scales examined four days before emergence of the butterfly (Danais), “the striations upon the upper surface of the scale are due to a series of parallel longitudinal ridges,” while the under side is usually smooth.
The mode of insertion is seen in Fig. 227. The narrow cylindrical pedicel of the scale is merely, according to Semper, inserted into a minute close-fitting socket, which perforates the wing-membrane, 197and not into a tube, as Landois supposed. Spuler describes a sort of double sac structure or follicle (Schuppenbalg) which receives the hollow pedicel of the scale. This was originally (1860) observed by F. J. Carl Mayer, but more fully examined by Spuler (Fig. 228) though not detected by A. G. Mayer.
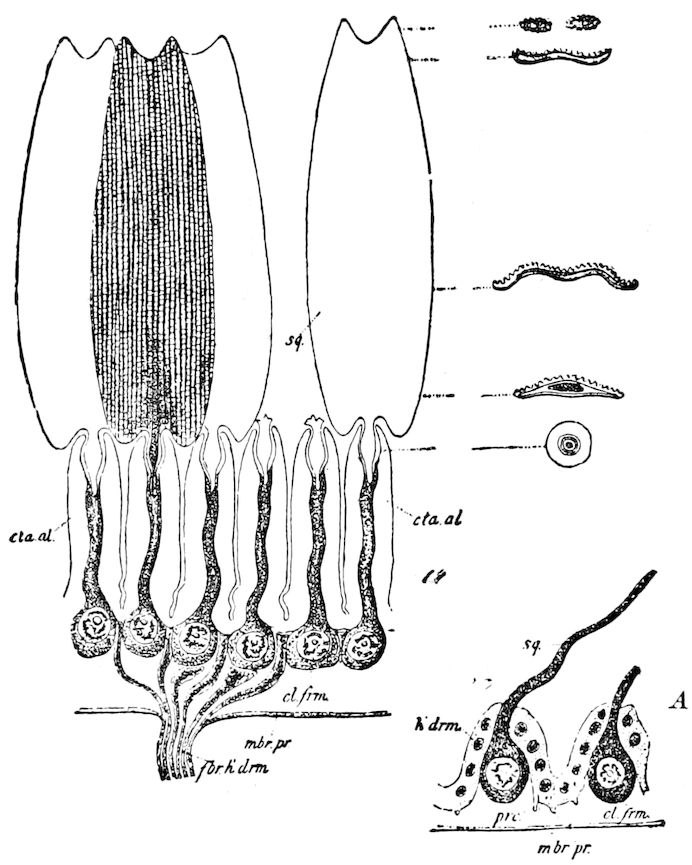
Fig. 226.—Portion of a cross-section through the pupal wing of Danais plexippus, about six days before emergence: sg, scale; cta.al, wing-membrane; cl.frm, formative cell of the scale; mbr.pr, ground-membrane; fbr.h′drm, hypodermal fibres of pupal wings. A, portion of a longitudinal section through the pupal wing, eight or nine days before emergence; prc, processes of young hypodermis scales.—This and Figs. 223–225 after Mayer.
Spinules, hair-scales, hair-fields, and androconia.—Besides the scales, fine spinules occur on the thickened veins of the wings of the Blattidæ, where they resemble fir-cones; also in the Perlidæ, in the Trichoptera, and in the more generalized Lepidoptera (Micropterygidæ and Hepialidæ), occur, as indicated by Spuler, delicate chitinous hollow spinules scarcely one-tenth as long as, and more numerous than, the scales, which sometimes form what he calls “Haftfelds,” or holding areas. These spinules have also been noticed by Kellogg, and by myself in Micropteryx; Kellogg, and also Spuler, have observed them in certain Trichoptera (Hydropsyche). 198These also occur on the veins, and detached ones near large one-jointed hairs, or hair-scales, said by Kellogg to be striated. Kellogg has detected these scale-hairs, as he calls them, in Panorpa.
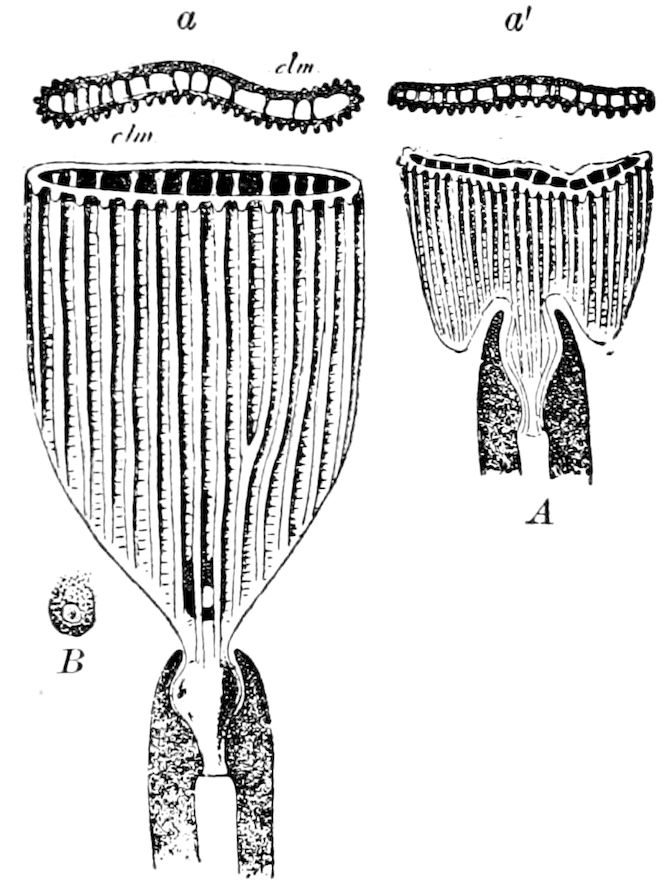
Fig. 227.—View looking down upon the upper (i.e. exposed) surface of one of the large scales situated on the veins of Danais plexippus, about four days before emergence: clm, chitinous pillars found in scales. A, a smaller scale, a, a′, sections of the scales. B, leucocyte found in the larger scale.—After Mayer.
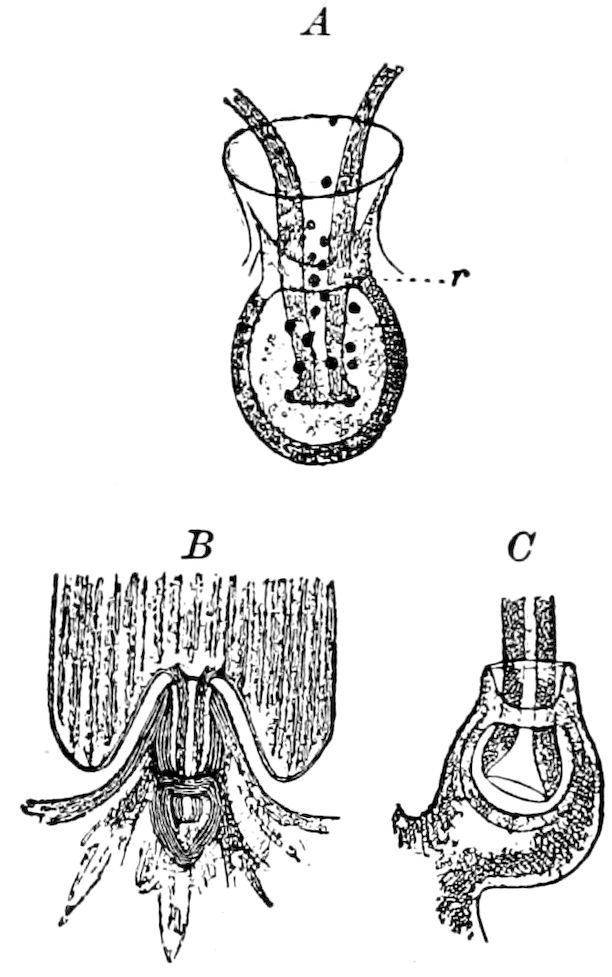
Fig. 228.—Scale-follicles: A, of a scale of Galleria mellonella: r, neck-ring. B, the same of Polyommatus phlæas. C, the same of a hair on inner edge of hind wing of Lycæna alexis ♀.—After Spuler.
Fig. 229.—A, portion of wing of a caddis-fly (Mystacides). B, enlarged, showing the androconia and hair-scales. C, a separate androconium.—After Kellogg.
The “hair-scales” of the phylogenetically older Trichoptera correspond to certain scales of Lepidoptera, especially the Psychidæ (Spuler), variously called “plumules” (Deschamps), “battledore scales,” also certain minute cylindrical hairs. To these scent-scales 199is applied the term androconia. They are found, almost without exception, on the upper side of the fore wings, occurring in limited areas, such as the discal spots, or on folds of the wings. Fritz Müller has shown that they function as scent-scales, and are confined to the males. Kellogg has detected androconia-like scales on the wings of a caddis-fly, Mystacides punctata (Fig. 229).
Fig. 280.—Cross-section of androconia surface on wing of Thecla calanus; a, androconia; gl, gland of base; s, ordinary scales; w, wing in section.—After Thomas.
Thomas has proved by sections of the wing of Danais, etc., that the androconia arise from glands situated in a fold of the wing (Fig. 230), and he states that the material elaborated by the local glands, and distributed upon the surface of the wing by the androconia, is that which gives to many of the Lepidoptera their characteristic odor. On comparing these “glands,” it is evident that they are groups of specialized formative cells of Semper (trichogens), which secrete an odorous fluid, issuing perhaps from extremely fine pore-canals at the ends of the androconia. They thus correspond to the glandular hairs, poison-hairs, and spines of caterpillars, the formative cells of which contain either a clear lymph or poison.
LITERATURE
a. Hairs, bristles, cleaning spines, calcaria, combs, etc.
Leydig, Franz. Zum feineren Bau der Arthropoden. (Müller’s Archiv f. Anat. und Phys., 1855, pp. 376–480.)
Fobel, Auguste. Les fourmis de la Suisse. Bâle, 1874.
Saunders, Edward. Remarks on the hairs of some of our British Hymenoptera. (Trans. Ent. Soc. London, 1878, pp. 169–171.)
Perez, J. Notes d’apiculture. (Bull. Soc. d’Apic. de la Gironde, Bordeaux, 1882.)
Osten Sacken, C. R. von. An essay on comparative chætotaxy, or the arrangement of characteristic bristles of Diptera. (Trans. Ent. Soc. London, 1884, pp. 497–517.)
—— Preliminary notice of a subdivision of the suborder Orthorrhapha Brachycera (Diptera) on chætotactic principles. (Berlin Ent. Zeitschr., 1896, pp. 365–373.)
200Janet, Charles. Études sur les fourmis. 8e Note. Sur l’organe de nettoyage tibio-tarsien de Myrmica rubra. (Ann. Soc. Ent. France, 1895, pp. 691–704, 6 Figs.)
See also J. B. Smith’s Economic Entomology, 1896, hairs of bees. Also the writings of De Geer, Huber, Fenger, Mayr, Forel, Canestrini, and Berlese (1880); Dahl, Cheshire, etc.
b. Glandular and poisonous setæ and spines
Ratzeburg, J. Th. Ch. Ueber entomologische Krankheiten. (Stettin Ent. Zeit., 1846, vii, pp. 35–41.)
Zeller, P. C. Revision der Pterophoriden. (Linnæa Ent., vi, pp. 319–416, 1852, at p. 356 speaks of “Drüsenhärchen.”)
Dimmock, George. On some glands which open externally on insects. (Psyche, iii, pp. 387–401, 1882.)
Goossens, Th. Des chenilles urticants. (Ann. Soc. Ent. France, 1881, pp. 231–236.) Des chenilles vésicants. (Ibid., 1886, pp. 461–464.)
Packard, A. S. Notes on some points in the external structure and phylogeny of insects. (Proc. Boston Soc. Nat. Hist., xxv, 1890, pp. 83–114, 2 Pls.)
—— A study of the transformations and anatomy of Lagoa crispata, a bombycine moth. (Proc. Amer. Phil. Soc., xxxii, pp. 275–292, 7 Pls., 1894.)
Holmgren, Emil. Studier öfner hudens och de körtelartade hudorganens morfologi hos Skandinaviska macrolepidopterlarver. (K. Svenska Vetenskaps-Akad. Handl., xxvii, pp. 1–83, Stockholm, 1895, 9 Pls.)
Also the writings of Leydig, Keller, Bach, Karsten, Scribner, Riley, etc.
(See also Literature of repugnatorial glands.)
c. Androconia
Deschamps, Bernard. Récherches microscopiques sur l’organisation des ailes der Lépidoptères. (Ann. des Sc. nat. [?], iii, pp. 111–157, 1835.)
Waufor, T. W. On certain butterfly scales characteristic of sex. (London, 1867–68.)
McIntire, S. J. Notes on the minute structure of the scales of certain insects. (London, 1871.)
Anthony, J. The markings on the battledore scales of some of the Lepidoptera. (London, 1872.)
Scudder, S. H. Antigeny or sexual dimorphism in butterflies. (Proc. Amer. Acad. Arts and Sc., xii, 1877, pp. 150–158.) Also Butterflies, etc. (New York, 1881, pp. 192–206, figs.)
Müller, Fritz. A prega costal das Hesperideas. (Archivas do Museo nac. do Rio de Janeiro, iii, pp. 41–50, 2 Pls., 1878.)
Thomas, M. B. The androconia of Lepidoptera. (Amer. Nat., xxvii, pp. 1018–1021, 2 Pls., 1893.)
d. Scales
Leydig, Franz. Zum feineren Bau der Arthropoden. (Archiv f. Anat. und Phys., 1855, pp. 376–480, 1 Taf.)
Semper, Carl. Beobachtungen über die Bildung der Flügel, Schuppen, und Haare bei den Lepidopteren. (Zeitschrift f. wissensch. Zoologie, 1857, pp. 326–339, 1 Taf.)
Mayer, F. T. Karl. Ueber den Staub der Schmetterlingsflügel. (Allgem. mediz. Centralzeitung, 1860, pp. 772–774.)
201Landois, H. Beiträge zur Entwicklungsgeschichte der Schmetterlingsflügel in der Raupe und Puppe. (Zeitschr. f. wissensch. Zoologie, xxi, 1871, pp. 305–316, 1 Taf.)
Weismann, August. Ueber Duftschuppen. (Zool. Anzeiger, i, 1878, pp. 98–99.)
Dimmock, George. Scales of Coleoptera. (Psyche, iv, pp. 1–11, 23–27, 43–47, 63–71, 1883.)
Schaeffer, Cäsar. Beiträge zur Histologie der Insekten. (Zool. Jahrbücher, Abth. f. Anat. u. Ontog., iii, pp. 611–652, 2 Pls., 1889.)
Kellogg, Vernon L. The taxonomic value of the scales of the Lepidoptera. (Kansas Univ. Quart., iii, pp. 45–89, figs. 1–17, 9 Taf., 1894.)
Mayer, Alfred G. The development of the wing-scales and their pigment in butterflies and moths. (Bull. Mus. Comp. Zool., xxix., 1896, pp. 209–236, 7 Pls.)
Spuler, Arnold. Beiträge zur Kenntniss des feineren Baues und der Phylogenie der Flügeltedeckung der Schmetterlings. (Zool. Jahrb. Abth. f. Anat. u. Ontog., viii, pp. 520–543, 1 Taf., 1895.)
—— Ueber das Vorhandensein von Schuppenbalg bei den Schmetterlingen. (Biol. Centralblatt, xvi, Sept. 15, 1896, pp. 677–679, 3 figs.)
THE COLORS OF INSECTS
The colors and bright markings of insects, especially those of butterflies, render them the most brilliant and beautiful creatures in existence, rivalling and even excelling the gay hues of our most splendidly colored birds. The subject has been but recently taken up and is in a somewhat crude condition, but the leading features have been roughly sketched out by the work of a few observers from a physical, chemical, and biological point of view.
The colors of insects, as of all other animals, are primarily due to the action of light and air; other factors are, as Hagen observes, heat and cold, moisture and dryness, as recently shown by the experiments on butterflies by Dorfmeister, Weismann, W. H. Edwards, and later observers. They have their seat in the integument. Hagen divides colors into optical and natural.
Optical colors.—“These,” says Hagen, “are produced by the interference of light, and are by no means rare among insects, but they are solely optical phenomena. Colors by the interference of light are produced in two different ways: either by thin superposed lamellæ, or by many very fine lines or small impressions in very close juxtaposition.
“1. There must be present at least two superposed lamellæ to produce colors by interference. The naked wings of Diptera, of dragon-flies, and of certain Neuroptera often show beautiful interference colors. The wings of Chrysopa and Agrion show interference colors only for a certain time, viz., as long as the membranes of the wings are soft and not firmly glued together. Afterwards such wings become simply hyaline.
202“The scales of Entimus and other Curculionidæ are well known for their brilliancy, and it is interesting to remark that when dry scales are examined with the microscope, many are found partly injured, which give in different places different colors, according to the number of layers which remain. The elytra of some Chrysomelina and other beetles with iridescent colors probably belong to the same category.
“2. When there are scales with many fine lines or small impressions close to each other, we have the second mode of producing colors.
“The fine longitudinal and transversal lines of lepidopterous scales seem to serve admirably well to produce the brilliant effect of color-changing butterflies. But there must be something more present, as most of the scales of Lepidoptera are provided with similarly fine lines, and only comparatively few species change colors. I remark purposely that the lines in the color-changing scales are not in nearer juxtaposition.” (Hagen.)
“The colors of butterflies change mostly from purple to blue, sometimes to yellow. The splendid violet color at the end of the wings of Callosune ione is brought out by a combination of the natural with interference colors. Originally the scales are colored lake-red; but a blue interference color is mixed with it; hence the violet hue results. The blue tones, i.e. the splendid varying blue of the Morpho butterflies, Schatz claims, owe their hue less to the interference of light than to a clouded layer of scales situated over the dark ground, through which the light becomes reflected on the same. The scales of the Morphids are in reality brown, as we see by transmitted light; moreover, only the upper side of the scales sends off blue reflections—the under side is simply brown. But the blue scales of Urvilliana are also shining blue beneath; by transmitted light they appear as if clear yellow. The smaragd-green scales of Priamus show by transmitted light a bright red-orange, and the orange-yellow of Crœsus a deep grass-green.” (Schatz in Kolbe.)
“Krukenberg presumes the golden-green color of Carabus auratus to be an interference color. It is not changed by the interference of light, nor was he able to extract from the elytra any green pigment with ether, benzol, carbon of sulphur, chloroform, or alcohol, even after having previously submitted the elytra to the influence of muriatic acid or ammonia. Chlorophyll is not present, whether free or combined with an acid.” (Hagen.)
Leydig has shown that the interference colors of the hairs of certain worms (Aphrodite and Eunice) may be produced by very small impressions in juxtaposition, which bring about the same effect as striæ. Such an arrangement occurs on the feathers of birds, i.e. on the necks of pigeons and elsewhere, and Hagen suggests that this kind of interference colors occurs more frequently among insects than is commonly known. At least the limbs of certain forms appear yellow, but when held in a certain position change to brown or blackish. “I know of no other explanation of this not uncommon fact on the legs of Diptera, of Hymenoptera, and of Phryganidæ.” Interference colors, he adds, may occur in the same place together with natural colors. “The mirror spots of Saturnia pernyi show besides the interference colors a white substance in the cells of the matrix, which Leydig believes to be guanin. But this fact is denied by Krukenberg for the same species and also for Attacus mylitta and Plusia chrysitis.”
203Natural colors.—These are divided by Hagen into dermal (cuticular) and hypodermal. The dermal colors are due to pigment deposited in the form of very small nuclei in the cuticula. Hagen considers them as “produced mostly by oxidation or carbonization, in consequence of a chemical process originating and accompanying the development and the transformations of insects.”
“To a certain extent the dermal colors may have been derived from hypodermal colors, as the cuticula is secreted by the hypodermis, and the colors may have been changed by oxidation and air-tight seclusion. The cuticula is in certain cases entirely colorless,—so in the green caterpillar of Sphinx ocellata; but the intensely red and black spots of the caterpillar of Papilio machaon belong to the cuticula, and only the main yellow color of the body to the hypodermis.” (Leydig, Histiol., p. 114.)
“The dermal colors are red, brown, black, and all intermediate shades, and all metallic colors, blue, green, bronze, copper, silver, and gold. The dermal colors are easily to be recognized as such, because they are persistent, never becoming obliterated or changed after death.” (Hagen.)
Minot and Burgess refer to the cuticular colors of the cotton-worm (Aletia), the dark brown color belonging to the cuticula or crust. “Upon the outside of the crust is a very thin but distinct layer, which in certain parts rises up into a great number of minute, pointed spines that look like so many dots in a surface view. Each spine is pigmented diffusely, and together they produce the brown markings. The spines are clustered in little groups, one group over each underlying hypodermal cell.” (U. S. Ent. Comm., 4th Report, p. 46.) Minot also shows that in caterpillars generally a part of the coloration is caused by pigmentation of the cuticula.
In a dull-colored insect, such as the Mormon cricket (Anabrus), the coloration, as Minot states, depends principally upon the pigment of the hypodermis shining through the cuticula. “Most of the cells contain dull, reddish-brown granules, but scattered in among them are patches of cells bright green in color. I have observed no cells intermediate in color; on the contrary, the passage is abrupt, a brown or red cell lying next a green one. Indeed, I have never seen any microscopic object more bizarre than a piece of the epidermis of Anabrus spread out and viewed from the surface.” (2d Report U. S. Ent. Comm., p. 189.)
The pigment may extend through the entire cuticula, but it is usually confined to the outermost layers, and occurs there in union with a peculiar modelling of the upper surface into microscopic figures which are of interest not only from their delicacy, but because they vary with each species. (See p. 184.)
The hypodermal colors, situated in the hypodermis, are, according to Hagen, the result of a chemical process, generating color out of substances contained in the body. They are easily recognized, since 204they fade, change, and disappear after death. But where these colors are preserved after death and enclosed in air-tight sacs, as in the elytra and scales and hairs of the body, they persist, though, as we well know, they may fade after exposure to light.
The hypodermal colors are mostly brighter and lighter than the dermal ones, being light blue or green in different shades, yellow to orange, and the numerous shades of these colors combined with white; exceptionally they are metallic, as in Cassida, and are then obliterated after death.
“The fact that such metallic colors can be retained in dead specimens by putting a drop of glycerine under the elytra, leads us to conclude that those colors are based upon fat substances. The hypodermal colors are never glossy, as far as I know; the dermal colors frequently.
“As the wings, elytra, and hairs all possess a cuticula, dermal colors are frequently to be found, together with hypodermal ones, chiefly in metallic colors. In the same place both colors may be present, or one of them alone. So we find hypodermal colors in the elytra of Lampyridæ. In the elytra of the Cicindelidæ the main metallic color is dermal, the white lines or spots are hypodermal, by which arrangement the variability in size and shape of those spots is explained.
“There occur in a number of insects external colors, that is, colors upon the cuticula, which I consider to be in fact displaced hypodermal colors: the mealy pale blue or white upon the abdomen of some Odonata, the white on many Hemiptera, the pale gray on the elytra and on the thorax of the Goliath beetle, and the yellowish powder on Lixus. Some of these colors dissolve easily by ether or melt in heat, and some of them are a kind of wax. I believe that those colors are produced in the hypodermis, and are exuded through the pore-canals.” (Hagen.)
The white colors are simply for the most part due to the inclusion of air in scales. The white mother-of-pearl spots of Argynnis are produced by a system of fine transverse pore-canals filled with air; in Hydrometra the white ventral marks have the same origin. (Leydig.)
The further statements and criticisms of Hagen regarding the relation of color to mimicry, sexual selection, and the origin of patterns are of much weight and will be referred to under those heads. Indeed, these subjects cannot well be discussed without reference to the fundamental facts stated in the masterly papers of Leydig and of Hagen, and much of the theorizing of these latter days is ill-founded, because the colors of insects and animals are attributed to natural selection, when they seem really the result of the action of the primary factors of organic evolution, such as changes of light, heat, cold, and chemical processes dependent on the former.
As to the chemical nature of color, Hagen, after quoting the results of Krukenberg and others, thinks that the colors of insects are chemically produced by a combination of fats or fat-acids with other 205acids or alkalis under the influence of air, light, and heat. He concludes:—
1. That some colors of insects can be changed or obliterated by acids.
2. That two natural colors, madder-lake and indigo, can be produced artificially by the influence of acid on fat-bodies.
3. As protein bodies in insects are changed into fat-bodies, and may be changed by acids contained in insects into fat-acids, the formation of colors in the same manner seems probable.
4. That colors can be changed by different temperatures.
5. That the pattern is originated probably by a combination of oxygen with the integument.
6. That mimicry of the hypodermal colors may be effected by a kind of photographic process.
7. Finally, color and pattern are produced by physiological processes in the interior of the bodies of insects.
Krukenberg concludes that change of color (in perfectly developed insects) is a consequence of the change of food, and can be explained by the alteration of the pigment through heat and light. His experiments were made in order to ascertain the cause of the turning of green grasshoppers in autumn into yellow and pink. He tried to answer two questions: First, does the pigment of grasshoppers originate directly out of the food, and does it consist of pure chlorophyll or of a substance containing chlorophyll, or is it to be accepted as a peculiar product of the organism? Second, is the color the consequence of only one pigment, or of several? Special analysis proves that the green color has no connection with chlorophyll. He concludes: “It is evident that the green color of the grasshopper is the consequence of several different pigments which can be separated by a chemical process.” Krukenberg believes that light has a marked influence on the color of insects and that light turns to red or pink the insects which were green during the summer. It would seem, however, more probable that cold was the agent, the change being due to the colder autumn weather.
Here we might refer to the results of the studies of Buckton and Sorby, on the changes in color of Aphides:—
“1. The purple coloring matter appears to be a quasi-living principle, and not a product of a subsequent chemical oxidizing process. Mounted in balsam or other preserving fluids, the darker species stain the fluid a fine violet.
“2. As autumn approaches and cold weather reduces the activity of the Aphides, the lively greens and yellows commonly become converted into ferruginous red, and even dark brown, which last hue in reality partakes more or less of intense violet or purple. These changes have some analogy with the brilliant hues assumed by maple and other leaves during the process of slow decay.
“3. Aqueous solutions of crushed dark brown and yellow-green varieties of Aphides originate different colors with acids and alkalies.
“4. In the generality of cases coloring-matters, such as indigo, Indian yellow, madder-lake, and the like, do not separately exist in the substance of vegetables, but the pigments are disengaged through fermentation or oxygenation. Again, alizarin itself is reddish yellow, but alkaline solutions strike it a rich 206violet just as we find them to act towards the substance which Mr. Sorby calls aphidilutein.
“5. Mr. Sorby’s four stages of the changes effected by the oxidation of aphideine produce four different substances.”
Chemical and physical nature of the pigment.—Researches in this difficult field of inquiry have been made by Landois (1864), Sorby (1871), Meldola (1871), by Krukenberg (1884), and more recently by Coste, Urech, Hopkins, and Mayer, and the subject is of fundamental importance in dealing with mimicry and protective coloration, the primary causes of which appear to be due to the action of physical and chemical agents.
Over twenty years ago Meldola observed that the yellow pigment of the sulphur-yellow butterfly (Gonopteryx rhamni) was soluble in water, and showed that its aqueous solution had an acid reaction.
Besides the yellow uranidin found by Krukenberg in different beetles and lepidopterous pupæ, still other coloring-matters, which are very constant in different species are readily recognized by the spectroscope. “Thus there appear in the brownish yellow lymph of Attacus pernyi, Callosamia promethea and Telea polyphemus, after saponification of the precipitated soap readily effected by ether, or incompletely or not removed by benzine, a chlorophane-like lipochrome; and in the yellowish green lymph of Saturnia pyri and of Platysamia cecropia besides this pigment still another whose spectrum shows a broad band on D, but which disappears with the addition of acetic acid or ammonia, as also after a long heating of the lymph up to 66° C.”
Coste, and more especially Urech, have shown that many of the pigments may be dissolved out of the scales by means of chemical reagents, giving colored solutions, and leaving the scales white or colorless. They have also shown that some of these pigments may be changed in color by the action of reagents, and then restored to their original color by other reagents. They have proved that reds, yellows, browns, and blacks are always due to pigments, and in a few cases greens, blues, violets, purples, and whites, and not, as is usually the case, to structural conditions, such as striæ on the scales (Mayer). They confined themselves solely to the chemical side of the problem, not considering the structure of the scales themselves.
Urech has also discovered a beautiful smaragd-green coloring-matter in the wings (not in the scales) of the pupa of Pieris brassicæ. It is not chlorophyll, and Urech suggests that it may be either the germinal substance of the pigments of the scales or its bearer. It is not the pigment of the blood.
Urech has also demonstrated that in many Lepidoptera the color of the urine which is voided upon emergence from the chrysalis is similar to the principal color of the scales.
207Hopkins has worked on the pigments within the scales of butterflies. The yellow pigment in Gonopteryx rhamni is a derivation of uric acid, and he calls it lepidotic acid. Its aqueous solution is strongly acid to litmus, and must be bad-tasting to birds.
Hopkins has dissolved the red pigment from the border of the hind wing of Delias eucharis, an Indian butterfly, in pure water, finding as the result a yellow solution; but if the solution be evaporated to dryness, the solid residue of pigment is red once more. He has obtained from this pigment of eucharis a silver compound which contains a percentage of metals exactly equal to that from the pigment of G. rhamni. (Nature, April 2, 1892.)
“The scales of the wings of the white butterflies (Pieridæ) are also shown by Hopkins to contain uric acid, this substance practically acting as a white pigment in these insects. A yellow pigment, widely distributed in the same family, is shown to be a derivative of uric acid, and its artificial production as a by-product of the hydrolysis of uric acid is demonstrated. That this yellow pigment is an ordinary excretory product of the butterfly is indicated by the fact that an identical substance is voided from the rectum on emergence from the pupa. These excretory pigments, which have well-marked reactions, are apparently confined to the Pieridæ, and are not found in other Rhopalocera. This fact shows that when a Pierid mimics an insect belonging to another group, the pigments of the mimicked and mimicking insects, respectively, are chemically quite distinct. Other pigments existing, not in the scales, but between the wing-membranes, are shown to be of use for ornament.” (Proc. Royal Soc., London, 1894.)
Griffiths (1892) claims that the green pigment found in several species of Papilio, Hesperia, and Limenitis, also in Noctuidæ, Geometridæ, and Sphingidæ likewise consists of a derivative of uric acid, which he calls lepidopteric acid. By prolonged boiling in HCl it is converted into uric acid.
Spuler, however, finds that green does not depend on pigmentation, but is an optical color. As remarked by Spuler, either the chitin of the scales itself is colored reddish (yellow grayish), or the pigment is secreted in the nuclei.
A. G. Mayer believes that the pigments of the scales are derived from the hæmolymph or blood of the pupa, for the following reasons: (1) He is unable to find anything but blood within the scales during the time when the pigment is formed. (2) In Lepidoptera generally the first color to appear upon the pupal wings is a dull ochre-yellow, or drab, and this is also the color assumed by the blood when it is removed from the pupa and exposed to the air. (3) He has succeeded by artificial means in manufacturing several pigments from the blood which are similar in color to various markings upon the wing of the imago; chemical reagents have the same effect upon these manufactured pigments that they do upon the similarly colored pigments of the wings. “It should be here noted,” he says, “that in 1866 Landois pointed out the fact that the color of the dried blood of many caterpillars is similar to the ground color of the wings of the mature insect.”
Ontogenetic and phylogenetic development of colors.—The colors of the wings of Lepidoptera, as is well known, are acquired at the end 208of the pupal state. The order of development of the colors in the pupal wings has been observed by Schaeffer, Van Bemmelen, Urech, Haase, Dixey, Spuler, and A. G. Mayer. The immature wings are at first transparent and full of protoplasm. The transparent condition of the wings corresponds to the period before the scales are formed, and when they are full of protoplasm; they then become whitish as the scales develop; the latter are at first filled with protoplasm, and afterwards turn whitish, being little hollow sacks filled with air. After the protoplasm has completely withdrawn from the scales, the blood of the pupa enters them, and then the coloring-matter forms. (Mayer.) He adds that “about twenty-four hours after the appearance of the dull yellow suffusion the mature colors begin to show themselves. They arise, faint at first, in places near the centre of the wings, and are distinguished by the fact that they first appear upon areas between the nervures, never upon the nervures themselves. Indeed, the last place to acquire the mature coloration are the outer and costal edges of the wings, and the nervures.”
The faint color of the scales gradually increases in intensity. “For example, if a scale be destined to become black, it first becomes pale grayish brown, and this color gradually deepens into black.”
Urech states that in Vanessa io first a white, and in V. urticæ a pale reddish hue, are spread over the entire wings, and then successively arise other colors in the following order: yellow, yellow to brown, red, brown and black.
Spuler, however, claims that the differentiation of colors and markings do not follow one another, but arise simultaneously, and that his view is confirmed by Fischer. This may be the case with the highly specialized and diversely marked butterflies, but certainly taking the Lepidoptera as a whole the yellows and drabs must have been the primitive hues, the other colors being gradually added in the later more specialized forms.
It is noticeable that the most generalized moths, such as the species of Micropteryx, Tinea, Psychidæ, Hepialidæ (in general), etc., are dull brown or yellow-drab without bars, stripes, or spots of bright hues. These shades prevail in others of the more primitive Lepidoptera, such as many bombycine moths, and they even appear to a slight extent in certain caddis-flies. The authors mentioned, especially Mayer, whom we quote, claim that “dull ochre-yellows and drabs are, phylogenetically speaking, the oldest pigmental colors in the Lepidoptera; for these are the colors that are assumed by the hæmolymph upon mere exposure to the air. The more brilliant pigmental colors, 209such as bright yellow, reds, greens, etc., are derived by more complex chemical processes. We find that dull ochre-yellow and drabs are at the present day the prevalent colors among the less differentiated nocturnal moths. The diurnal forms of Lepidoptera have almost a monopoly of the brilliant colorations, but even in these diurnal forms one finds that dull yellow or drab colors are still quite common upon those parts of their wings that are hidden from view.”
The more primitive moths being more or less uniformly yellowish or drab, the next step was the formation of bars, stripes, finally spots, and eyed spots, these markings in the later forms appearing simultaneously in one and the same species of certain highly specialized moths and butterflies. All that has been said will prepare the reader for the consideration of the subject of insect coloration. The origin of such markings has been discussed by Weismann, Eimer, Haase, Dixey, Fischer, and others.
LITERATURE
Heer, O. Einfluss des Alpenklimas auf die Farbe der Insecten. (Froebel u. Heer, Mitth. aus dem Gebiete der theoret. Erdkunde, 1836, i, pp. 161–170.)
Goureau. Mémoire sur l’irisation des ailes des insectes. (Ann. Soc. Ent. France, 2 sér., i, 1848, pp. 201–215.)
Laboulbène, A., et M. Follin. Note sur la matière pulvérulente qui recouvre la surface du corps des Lixus et de quelques autres insectes. (Ann. Soc. Ent. de France, 1848, vi, pp. 301–305, Fig.)
Coquerel, Ch. Note sur la prétendue poussière cryptogamique qui recouvre le corps de certains insectes. (Ann. Soc. Ent. France, 1850, viii, pp. 13–15.)
Brauer, F. Beobachtungen in Bezug auf den Farbenwechsel bei Chrysopa vulgaris. (Verhandl. k. k. zool.-botan. Gesellsch. Wien., 1852, pp. 12–14.)
Prittwitz, O. F. W. v. Bemerkungen über die geographische Farbenverteilung unter den Lepidopteren. (Stett. Ent. Zeit., 1855, xvi, pp. 175–185.)
Latham, A. G. The causes of the metallic lustre of the scales on the wings of certain moths. (Proc. Lit. and Phil. Soc. Manchester, iii, 1864, pp. 198–199. Quart. Journ. Micr. Sc., new ser., iv, 1864, pp. 48–49.)
Sorby, H. C. On the coloring matter of some Aphides. (Quart. Journ. Micr. Sc., new ser., xi, 1871, pp. 352–361.)
Leydig, Franz. Bemerkungen über Farben der Hautdecke und Nerven der Drüsen bei Insekten. (Archiv f. mikr. Anatomie, xii, 1876, pp. 536–550, 1 Taf.)
Weismann, A. Studien zur Descendenz-Theorie, ii, 1876.
Hemmerling, Hermann. Ueber die Hautfarbe der Insecten. (Bonn, 1878, p. 27.)
Buckton, C. B. Monograph of the British Aphides. (London, 1879, ii, p. 167.)
Cameron, P. Notes on the coloration and development of insects. (Trans. Ent. Soc. London, 1880, pp. 69–79.)
Hagen, Hermann A. On the color and pattern of insects. (Proc. Amer. Acad. Arts and Sc., 1882, pp. 234–267.)
210Poulton, Edward Bagnall. The essential nature of the colouring of phytophagous larvæ (and their pupæ), etc. (Proc. Roy. Soc. London, xxxviii, pp. 269–315, 1884–1885.)
—— An inquiry into the cause and extent of a special colour-relation between certain exposed lepidopterous pupæ and the surfaces which immediately surround them. (Phil. Trans. Roy. Soc. London, clxxviii, pp. 311–441, 1 Pl., 1887.)
Krukenberg, C. Fr. W. Grundzüge einer vergleichenden Physiologie der Farbstoffe und der Farben. (Heidelberg, 1884, pp. 102.)
McMunn, C. A. Krukenberg’s chromatological speculation. (Nature, xxxi, p. 217, 1885.)
Müller, Fritz, and Dr. H. A. Hagen. The color and pattern of insects. (Kosmos, xiii, 1886, pp. 466–469.)
Slater, J. W. On the presence of tannin in insects and its influence on their colors. (Trans. Ent. Soc. London, 1887, iii, Proceed., pp. 32–34.)
Bemmelen, J. F. van. Ueber die Entwicklung der Farben und Adern auf den Schmetterlingsflügeln. (Tijdschrift der nederland. Dierkundige Vereeniging, ser. 2, pp. 235–247, 1889.)
Hopkins, F. G. Uric acid derivatives functioning as pigments in butterflies. (Proc. Chem. Soc. London, 1889, p. 117; also Nature, xl, p. 335.)
—— Pigment in yellow butterflies. (Nature, xlv, p. 197, 1891.)
—— The pigments of the Pieridæ. (Proc. Roy. Soc. London, lvii, No. 340, pp. 5, 6, 1894. Phil. Trans. Roy. Soc. London, clxxxvi, pp. 661–682, 1896.)
Coste, F. H. P. Contributions to the chemistry of insect colors. (The Entomologist, xxiii, 1890; xxiv, 1891, pp. 9–15, etc. Nature, xlv., pp. 513–517, 541–542, 605.)
Urech, F. Beobachtungen über die verschiedenen Schuppenfarben und die zeitliche Succession ihres Auftretens. (Zool. Anzeiger, xiv, pp. 466–473, 1891; Ibid., August 1, 1892.)
—— Beiträge zur Kenntniss der Farbe von Insektenschuppen. (Zeits. f. Wissens. Zool., lvii, pp. 306–384, 1893.)
Griffiths, A. B. Recherches sur les couleurs de quelques insectes. (C. R. Acad. Sc. Paris, cxv, pp. 958, 959.)
Mayer, Alfred Goldsborough. On the color and color-patterns of moths and butterflies. (Proc. Bost. Soc. Nat. Hist., xxvii., March, 1897, pp. 243–330, 10 Pls. See also p. 201 under Mayer.)
Also the writings of Bates, Beddard, Belt, Butler, Darwin, Dimmock, Dixey, Eimer, Haase, Higgins, Müller, Poulton, Seitz, Wallace, Weismann.
2. INTERNAL ANATOMY
THE MUSCULAR SYSTEM
In its general arrangement the muscular system of insects corresponds to the segmented structure of the body. Of the muscles belonging to a single segment, some extend from the front edge of one segment to that of the next behind it, and others to the hinder edge; there are also sets of dorsal and ventral muscles passing in an oblique or vertical course (Figs. 16–18). As Lang observes, “the greater part of the muscles of the body can be traced back to a paired system of dorsal and ventral intersegmental longitudinal muscles.” The muscular system is simplest in larval insects, such as caterpillars, where the musculature is serially repeated in each segment.
In the larva of Cossus Lyonet found on one side of the body 217 dorsal, 154 lateral, 369 ventral, and in the thoracic legs 63, or 803 muscles in all. “Adding to this number the 12 small muscles of the second segment, and 8 others of the third, which he did not describe, there would be for all the muscles on one side of the caterpillar 823. This would make for the entire body 1646, without counting a small single muscle which occurs in the subdivision of the last segment,” and also those of the internal organs as well as those of the head, so that the total number probably amounts to about 2000, not 3000, as usually stated in the books. Lubbock admits that Lyonet was right in his mode of estimating the number. In the larva of Pygærci bucephala he found that “the large muscles scarcely vary at all,” though certain smaller ones are very variable. Lubbock observed that certain of the longitudinal muscles in the caterpillar of Diloba split up into numerous, not less than ten, separate fascicles. “This separation of the fibres composing a muscle into separate fascicles is carried on to a much greater extent in the larvæ of Coleoptera. Of course in the imago the number of thoracic muscles is greatly increased, or at least in Dyticus and the wood-feeding Lamellicorns, which alone I have examined. In these two groups each of the larger muscles is represented by at least twenty separate fascicles, which makes it far more difficult to distinguish the arrangement of the muscles.”
The muscles are whitish or colorless and transparent, those in the thorax being yellowish or pale brown; and of a soft, almost gelatinous consistence. In form they are simply flat and thin, straight, band-like, or in rare cases pyramidal, barrel or feather shaped. They act variously as rotators, elevators, depressors, retractors, protractors, flexors, and extensors.
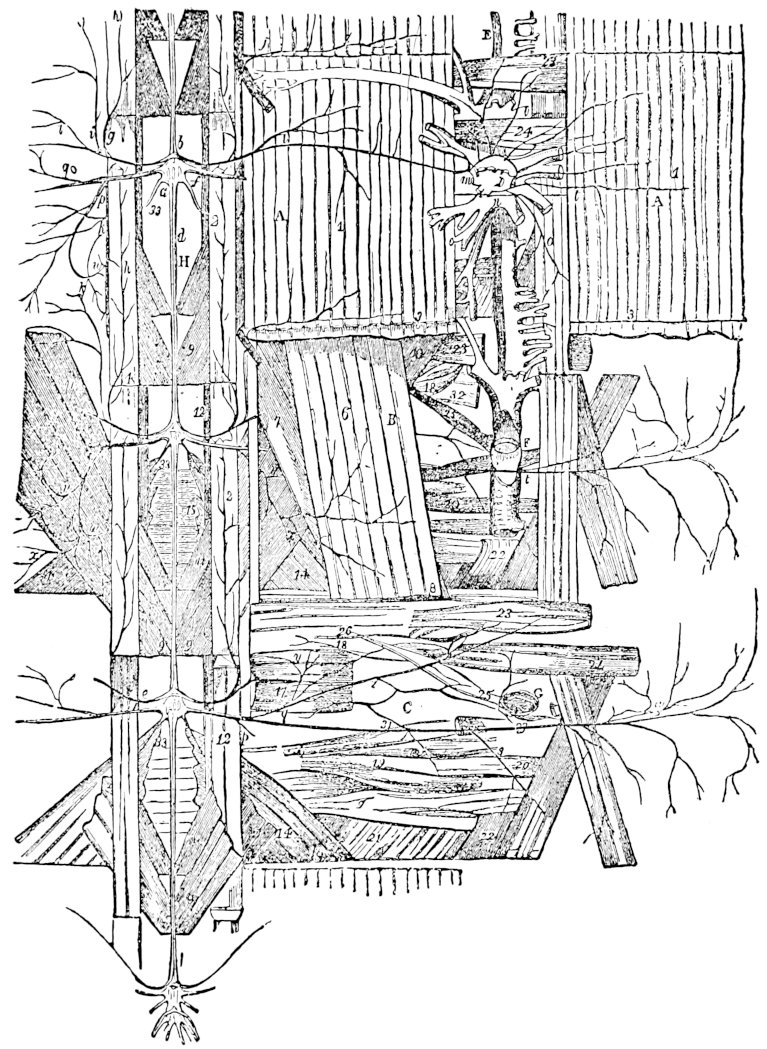
Fig. 231.—Diagram of the muscles and nerves of the ventral surface of the segments in the larva of Sphinx ligustri: A, A, recti muscles; 1, 2, ventral recti muscles (1, recti majores; 2, recti minores); 3, ridge giving origin to recti muscles of one segment, and insertion to the same of the adjoining segment; 4, ridge for attachment of muscle; 5, retractor ventriculi, connecting the mid-intestine with the outer integument of the body. B, 6, first oblique,—7, second oblique,—9, 10, third oblique, muscles; 11, fourth oblique,—12. third rectus,—13, fifth oblique,—14, triangularis, muscle; 15, transversus medius; 16, transverse ridge; 17, transversi abdominales; 18, abdominales anteriores; 19, 20, abdominales laterales, some (20) longer than others; 21, obliquus posterior; 22, postero-laterales obliqui; 23, transversus lateralis; 24, second transversus lateralis; 25, retractor spiraculi, or constrictor of the spiracles, attached by a long
tendon (26); 27, retractor valvulæ.
Nerves: a, ganglion,—c, transverse nerves, of which p is the first, q the second, r the third,
and s the fourth branch; t, the main trunk, which crosses the great longitudinal trachea, receives a
filament from the transverse nerve (n), and divides into two branches (t);—some of these branches
form a small plexus (u); the nerve t divides in two divisions (p and v). The second division ends
in w and x; the branch q divides into y and z. For other explanations, see Newport, art. Insecta.—After
Newport.
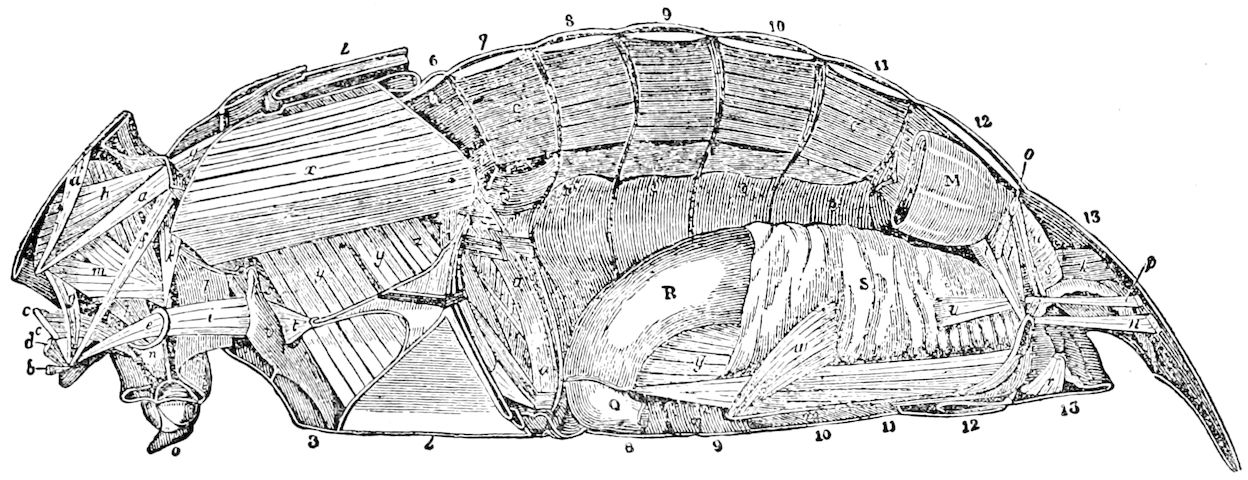
Fig. 232.—Musculature of the European cockchafer, Melolontha vulgaris: a, a, levatores capitis; b, depressores capitis; c, rotatores capitis; d, depressors externi; e, retractor or flexor of the jugular plate; f, oblique extensor of the jugular plate; g, the other retractor of the jugular plate; h, retractor prothoracis superior; i, inferior retractor, the proper depressor of the prothorax; k, elevator prothoracis; l, one of the rotatores prothoracis; m, n, o, flexors of the coxa; x, great depressor muscle of the wing; y, y, elevators and protractors attached to the metaphragma and base of the postfurca; z, second flexor of hind leg; a, a, extensors of hind leg; c, c, dorsal recti of abdomen. Q, ejaculatory duct; R, penis; S, its prepuce. M, rectum.—After Straus-Durckheim, from Newport.
Our knowledge of the muscular system of insects is still very imperfect. To work it out thoroughly one should begin first with that of Scolopendrella, then some generalized synapterous form, as Japyx or Lepisma, then passing to that of a caterpillar, and ending with some of the more highly specialized forms, such as a beetle, etc. Thus far our knowledge is confined to that of the caterpillars (Lyonet, Newport, and Lubbock) and the beetle (Straus-Durckheim) and ants (Forel, Lubbock, and Janet).
Musculature of a caterpillar.—Newport’s account of that of the larva of Sphinx ligustri is the most useful (Fig. 231). The muscles here present, he says, great uniformity of size and distribution in every segment, the motions of each of these divisions of the body being almost precisely similar, especially in the 4th to 9th trunk segments. In these segments the first layer seen on removing the fat and viscera are the flat straight recti muscles. They are the most powerful of all the trunk muscles, and are those which are most concerned in shortening the body, in effecting the duplicature of the external teguments during the changes of the insect, and which during the larval state mainly assist in locomotion. There are four sets, two dorsal and two ventral (Fig. 231, A, A). Without entering into farther details, the reader is referred to the works of Newport and to Fig. 231.
Musculature of a beetle.—The best general account of the musculature of a perfect insect is that of Straus-Durckheim in his famous work on the Melolontha. 214We will copy the summary of Newport, who adopted the nomenclature applied to these parts by Burmeister:—
“The muscles that connect the head with the thorax are contained within the prothorax (Fig. 232, 2), and are of three kinds, extensors, flexors, and retractors. The extensors, levatores capitis (a, a), consist of two pairs, one of which arises from the middle line of the pronotum, and diverging laterally from its fellow of the opposite side, passes directly forwards, and is inserted by a narrow tendon into the anterior superior margin of the occipital foramen. The other arises further back from the prophragma. It is a long, narrow muscle that passes directly forwards through the prothorax, and is inserted by a tendon near the superior median line of the foramen; so that, while this muscle and its fellow of the opposite side elevate the head almost in a straight line, the one first described, when acting alone or singly, draws the head a little on one side; but when the whole of these muscles act in unison, they simply elevate the head upon the prothorax. The depressors or flexors, depressores capitis (b), are exceedingly short muscles, which arise from the jugular plate, or, when that part does not exist, from the border of the prosternum, and are attached to the inferior margin of the occipital foramen. They simply flex the head on the prothorax. The lateral flexors, depressores externi (d), are two little muscles that arise from the same point as the preceding, and are attached to the lateral inferior margin of the occipital foramen. The rotatory muscles, rotatores capitis (c), are two flat muscles like the elevators, which arise, one at the side of the antefurca and the other from the posterior jugular plate, and passing upwards and outwards are attached to the lateral margin of the occipital foramen. The retractor or flexor of the jugular plate is a small muscle (e) that arises from the margin of the antefurca, and passing directly forwards is inserted by a small tendon into the middle of the jugular piece. The oblique extensor of the jugular plate is a long, slender muscle (f) that arises from the external margin of the pronotum, and passing obliquely downwards and forwards traverses the prothorax and is inserted by a narrow tendon to the jugular plate immediately before the retractor. The other retractor (g) arises from the anterior superior boundary of the pronotum, and passing downwards is inserted into the jugular plate between the larger levator and flexor capitis.
“The muscles proper to the prothorax consist of four pairs, by which it is united to the succeeding segments. The first of these, the superior retractor, retractor prothoracis superior (h), arises by a broad, fleshy head from the anterior external margin of the pronotum, and passing directly backwards is inserted by a tendon into the prophragma, a little on one side of the median line. The next muscle of importance, the inferior retractor (i), arises from the anterior border of the medifurca, and is united to the posterior of the antefurca, thus forming with that muscle part of the great recti of the larva. This muscle must be considered as the proper depressor of the prothorax. The elevator prothoracis (k) is narrow, pyramidal, and arises fleshy from the lateral surface of the prophragma. It passes downwards and is attached by a narrow tendon to the superior portion of the antefurca. The rotatores prothoracis are the largest of all the muscles of this segment. They arise, one on each side (l), by a narrow head from the posterior part of the pronotum, and passing beneath the prophragma are considerably enlarged and attached to the tegument between the two segments, and also to the anterior portion of the mesothorax. The remaining muscle proper to the prothorax is the closer of the spiracle, an exceedingly small muscle not shown in the drawing.
“The other muscles of this segment are those of the legs, which are of considerable size. There are three distinct flexors of the coxa (m, n, o). The first of 215these arises from the superior lateral border of the pronotum, the second from the superior posterior border, the third from the sides of the prothorax, and the fourth a little nearer posteriorly, and the whole of them are attached by narrow tendons to the sides of the coxa. But there is only one extensor muscle to this part. In like manner, the extensor of the trochanter is formed of three portions (Fig. 233, a, b, c); but there is only one flexor (d), and one abductor (e). In the femur, there is one extensor (f),—a long penniform muscle that occupies the superior part of the thigh, and is attached by a tendon to the anterior-posterior margin of the joint formed by the end of the tibia. There is also but one flexor (g) in the femur, which, like the preceding muscle, is penniform, and occupies the inferior portion of the femur, and its tendon is attached to the inferior border of the tibia. In the tibia itself there is also one flexor and one extensor. The flexor (i) occupies the superior portion of the limb, and ends in a long tendon (l) that passes directly through the joints of the tarsus, on their inferior surface, and is attached to the inferior margin of the claw (g). The extensor (h) occupies the inferior portion of the tibia, and is shorter than the preceding muscle, like which it ends in a long tendon that is attached to the upper margin of the claw. Besides these muscles, which are common to the joints of the tarsus, there are two others belonging to the claw, situated in the last joint. The first of these, the extensor (m), is short, and occupies the superior portion of the last phalanx of the tarsus, and the other, the flexor (n), is a much longer penniform muscle, which occupies nearly the whole of the upper and under surface of the posterior part of the phalanx, and is attached, like the long flexor of the tarsus, to the inferior part of the claw.”
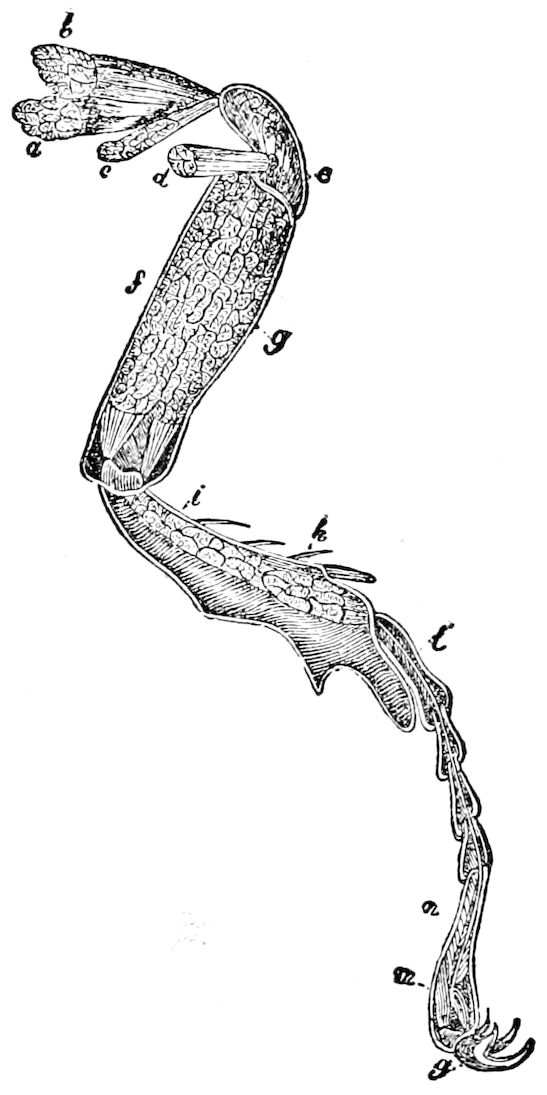
Fig. 233.—Muscles of the fore leg of Melolontha vulgaris: a, b, c, three divisions of the extensor of the trochanter; d, flexor,—e, abductor, of the trochanter; f, extensor of the femur; g, flexor of the femur; h, extensor of the tibia; i, flexor of the tibia; l, tendon attached to the lower edge of the claw (g); m, extensor,—n, flexor, of the claw.—After Straus-Durckheim, from Newport.
These are the muscles of the prothorax, and its organs of locomotion. The reader is referred for a further account of the muscles of the hinder thoracic and of the abdominal segments to Straus-Durckheim’s original work.
Minute structure of the muscles.—The muscular fibres of insects are striated (Figs. 235–238), even those of the alimentary canal; the only notable exception being the alary muscles of the pericardial septum, while Lowne states that certain of the thoracic muscles of the blow-fly are not striated (Miall and Denny).
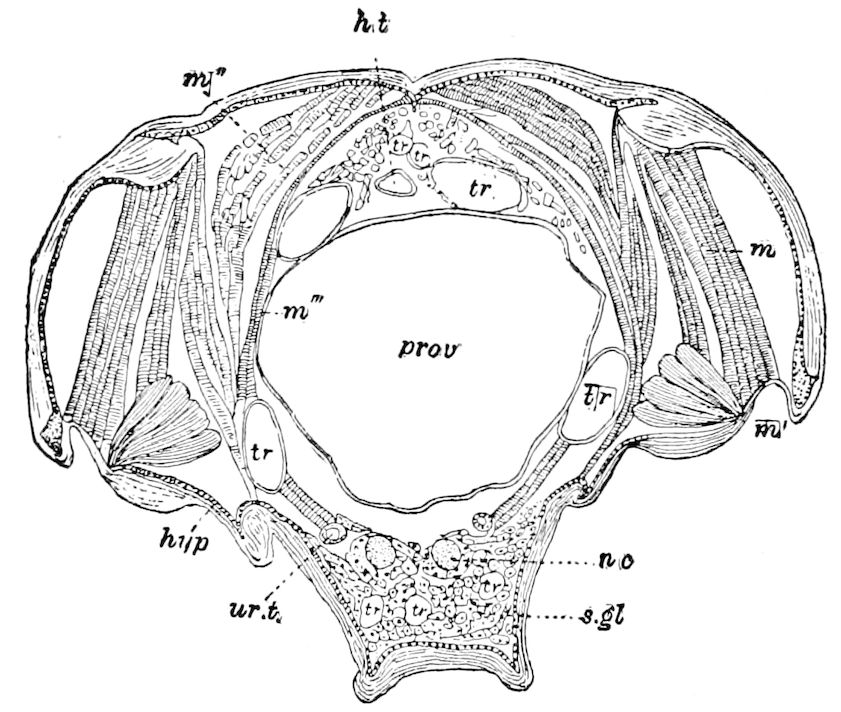
Fig. 234.-Section through the prothorax of Diapheromera femoratum: prov, proventriculus; tr, trachea; n. c, nervous cord; s. gl, salivary gland; hyp, hypodermis; ur. t, urinary tube; ht, heart; m, m″, m‴, muscles for lowering and raising the tergum; m′, another muscle, its use unknown.
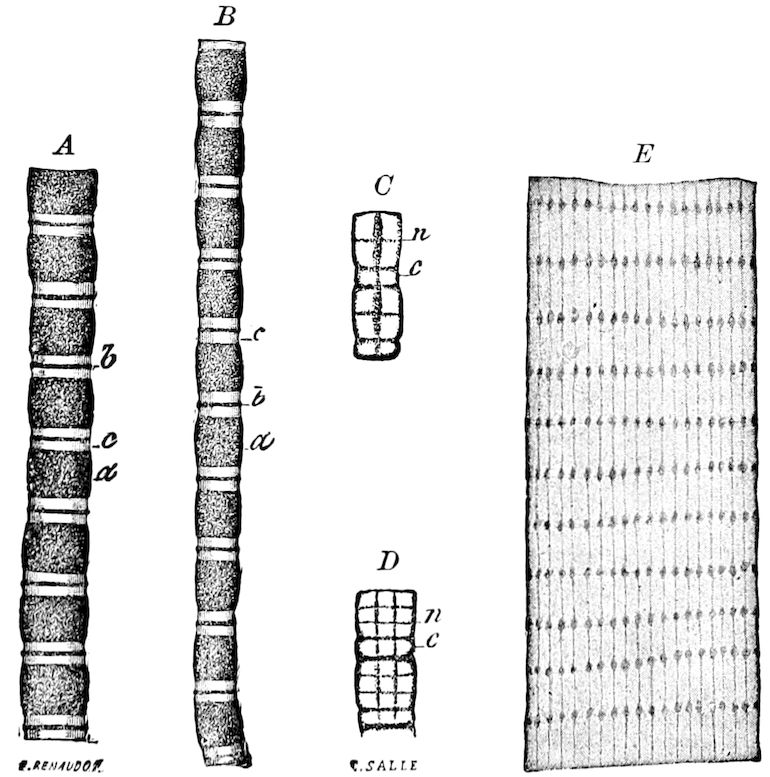
Fig. 235.—Striated muscular fibre of Hydrophilus: A and B, two fibrillæ in a state of extension; a, thick disk; b, thin disk; c, intermediate space. C, D, portion of the same fibrillæ seen by moving the objective farther away and using a small diaphragm; n, thick; c, thin disk. × 2000 diam.—After Ranvier, from Perrier. E after Gehuchten, from Lang.
In describing the minute structure of the muscles of ants, wasps, and bees, C. Janet states that each consists of a group of fibres diverging from a tendon, which is an integumentary invagination (Fig. 236). Each fibre may be regarded as a multinucleate cell; 217the sarcolemma represents the cell-membrane. It forms a resistant and extremely elastic tube. The longitudinal (Fig. 236, E) and radiating filaments or reticulum (spongioplasm of Gehuchten) lie in a nutritive filling substance (the hyaloplasm of Gehuchten). The radiating filaments are formed of an exceedingly elastic substance, and serve to sustain the longitudinal filaments, to transmit the nervous stimulus to them, and to bring them back into position after contraction. Janet’s account agrees on the whole with that of Gehuchten.
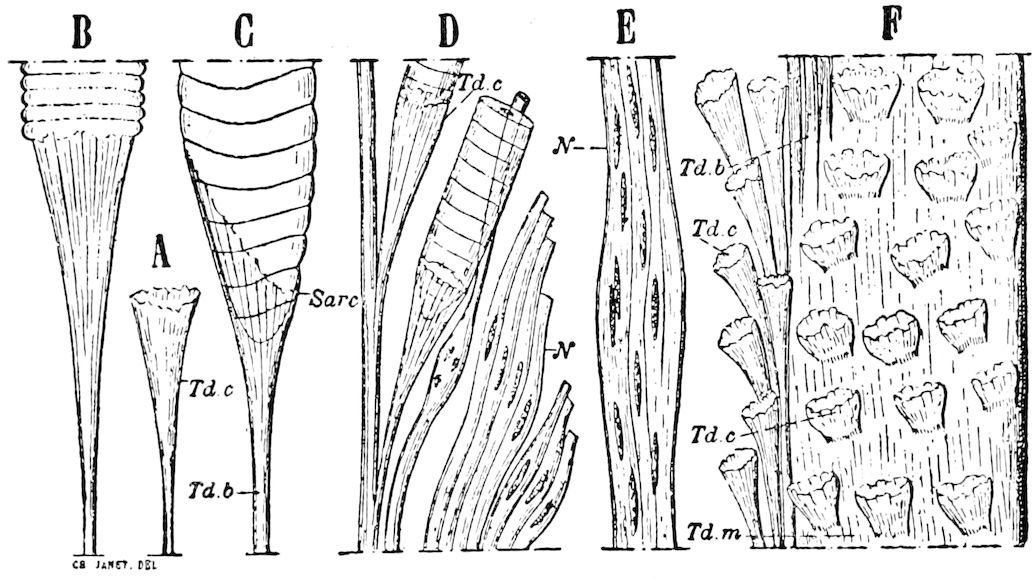
Fig. 236.—Preparations from the adductor muscle of the mandible of Vespa crabro, worker, fixed by heat and alcohol several hours after leaving its cell. A to E × 425; F × 212: A, terminal cupule of the tendon of a fibre. B, C, union of the fibres with their tendon. D, branch of the tendon of a muscle sending out tendons of some of the fibres; this branch is accompanied with numerous nervous ramifications (N). E, fragment of a nerve which furnishes the ramifications of Fig. D. F, fragment of the tendon of the adductor muscle of the mandible; at the left are seen the terminal cupules of the fibres (td, c); on the right, on the body of the tendons, some sessile cupules, each of which forms the attachment of a fibre; td, b, tendons of the fibres.—After Janet.
The muscles of flight are said to be penetrated by fine tracheal branches, probably to supply a greater amount of oxygen, as the most energetic movements of the insect are made in moving the wings during flight; while the other muscles of the body are only surrounded by the air-tubes. (Sharp.)
Without entering into tedious details, the reader is referred to figures or references to the more important systems of muscles, such as those of the legs and other appendages, of the wings, of respiration, etc., to the sections treating of those organs or functions; also to Figs. 16, 17, 18, 22, 48, 74, 81, 83, 84, 115, 116, 172, 173, 174, etc.
Muscular power of insects.—The most detailed and careful experiments are those of Plateau. His experiments prove that even the 218weakest insects pull at least five times their own weight; many of them, however, get the better of a burden twelve to twenty fold as heavy as themselves, while a strong man or a draught horse, for example, is not even able to pull a burden which is equal to the weight of his body. Plateau came to the following results as to the relation of the weight of the body to the load drawn (1 and 2 are to be compared with each other, 1 being the larger, and 2 the smaller insect; it will be seen that the smaller insect is the stronger).
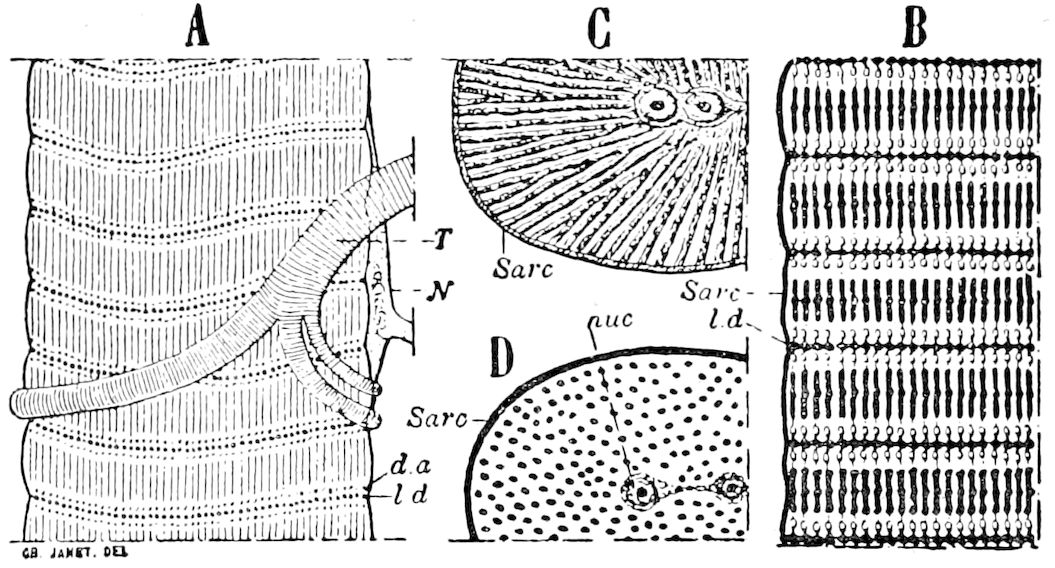
Fig. 237.—Vespa crabro, worker, fixed by heat and alcohol some hours after leaving its cell. A × 425; B to D × 850 times: A, muscular fibre of the motor muscles of the mandibles treated, for ten minutes, by 1 per cent potassium to bring out the reticulum; the nodes of union of the rayed filaments with the longitudinal filaments are indicated by distinct granulations (l.d), and these longitudinal filaments present accessory thickenings (d.a); T, trachea; N, junction of a nervous filament with the muscular fibres. B, fibre of the same muscle, not treated with potassium, stained by hæmatoxylin; C, transverse section of a disk at the level of a layer of rayed filaments; Sarc, sarcolemma. D, transverse section of a disk at the level of the rods; nuc, nucleus.—After Janet.
As regards the pushing power, the relation of the load to the size of the body in different large beetles, gave the following figures:—
219The leaping force of locusts was found by Straus-Dürckheim to be in Œdipoda grossa as 1.6, in Œ. parallela as 3.3 of their weight.
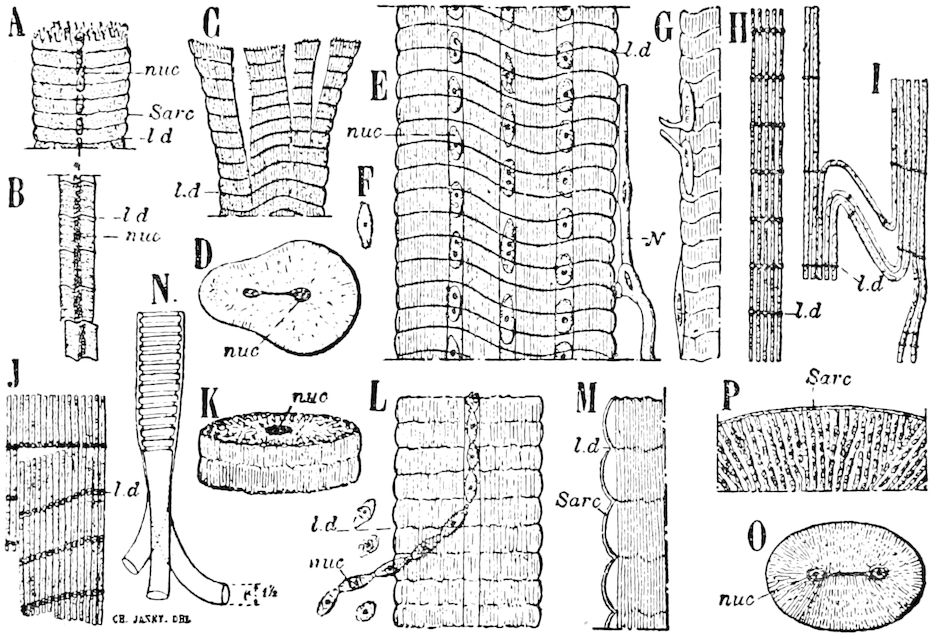
Fig. 238.—Vespa crabro, fixed and stained as in the subjects of the other figures. I, N, P × 1700; H, J, M × 850; the others × 425 times: A-C, motor muscles of the antennal scape. D-P, motor muscles of the 3d coxa. A, B, the two ends, in very different states of contraction, of the same fibre; on one side the transverse striæ are near together, on the other very far apart. C, a crushed and split fibre showing a fibrous appearance, owing to the rupture of the radiated filaments, and the separation of the longitudinal filaments. D, muscular disk seen in section, with two rows of nuclei. E, a muscular fibre with three rows of nuclei. F, a nucleus, accompanied with coagulated protoplasm, oozing from a previous break of the muscular fibre. G, nerve-terminations very near each other on the same muscular fibre. H, longitudinal filaments, evenly covered with the coagulated substance, and forming, throughout the mass of the fibre, continuous filaments. I, filaments widely separated. J, longitudinal filaments showing the beginning of one of the transverse breaks which isolate some of the disks. K, oblique view of a disk obtained by such a break, and of a fibre in circular section, with an axial row of nuclei; this piece comprises three stages of radiated filaments. L, muscular fibre with a row of nuclei; at the lower part, the nuclei have issued from a longitudinal fissure in the fibre, and have remained attached in a chain. M, edge of fibre in which there is quite a large, clear space between the sarcolemma and the rods. N, passage of the trachea, with the spiral thread, into three capillaries with a smooth cuticula. O, elliptical disk from a fibre, with two rows of nuclei, and showing a layer of radiated filaments. P, fragment (highly magnified) of the edge of a disk seen in section.—After Janet.
A humble bee (Bombus terrestris) can carry while flying a load 0.63 of its own weight, and a honey bee 0.78; here, as usual, the smaller insect is the stronger.[39]
LITERATURE ON THE MUSCLES
a. General
Lyonet, P. Traité anatomique de la chenille. La Haye, 1762.
Cornalia, E. Monographia del Bombyce del gelso. (Mem. R. Instituto Lombardo Sc. Lett. ed Arte, 1856.)
Basch, S. Skelett und Muskeln des Kopfes von Termes. (Zeitschr. f. wissens. Zool., xv, 1865, pp. 55–75, 1 Taf.)
Lubbock, John. Arrangement of the cutaneous muscles of the larva of Pygæra bucephala. London, 1858. 2 Pls.
—— On some points in the anatomy of ants. (Month. Micr. Journ., xviii, pp. 121–142, 1877, 4 Pls.)
—— On the anatomy of ants. (Trans. Linn. Soc., Ser. 2; Zool., ii, 1879, pp. 141–154, 2 Pls.)
Poletajeff, N. Du développement des muscles d’ailes chez les Odonates. (Horæ Soc. Ent. Ross., xvi, 1879, pp. 10–37, 5 Pls.)
—— Die Flugmuskeln der Lepidopteren und Libelluliden. (Zool. Anzeiger, 1880, pp. 212, 213.)
—— Ueber die Flugmuskeln der Rhopaloceren. (Arbeiten d. Russ. Ent. Ges., 1881, xiii, p. 9, 1 Taf., in Russian.)
Lendenfeld, R. von. Der Flug der Libellen. (Sitzb. k. Akad. Wissens., 1 Abth. Wien, 1881, lxxxiii, pp. 289–376, 7 Taf.)
Luks, Constantine. Ueber die Brustmuskulature der Insekten. (Jena. Zeitschr. f. Naturwissen., xvi, N. Folge IX, 1883, pp. 520–552, 2 Taf.)
Carlet, G. Sur les muscles de l’abdomen de l’abeille. (Comptes rend., 1884, xcviii, pp. 758, 759.)
Janet, Charles. Sur les muscles des fourmis, des guêpes et des abeilles. (Comptes rend., cxxi, p. 610, 1 Fig., 1895.)
Also the writings of Straus-Durckheim, Newport, Graber, Burgess, Leydig, Dahl, Ockler, Dogiel, Dimmock, Kraepelin, Becher, Langer, Kolbe.
b. Histology
Aubert, H. Ueber die eigenthümliche Struktur der Thoraxmuskeln der Insekten. (Zeitschr. f. wissens. Zool., iv, 1853, pp. 388–399, 1 Taf.)
Verson, E. Zur Insertionsweise der Muskeln. (Sitzsb. Akad. d. wiss. math. naturw. Cl. Wien., lvii, 1 Abth., pp. 63–66, 1868.)
Künckel d’Herculais. Sur le développement des fibres musculaires striées chez les insectes. (Compt. rend. de l’Acad. Sc. Paris, lxxv, 1872.)
Grunmach, Emil. Ueber die Structur der quergestreiften Muskelfaser bei den Insekten. Berlin, 1872. pp. 47.
Fredericq, L. Note sur la contraction des muscles striés de l’Hydrophile. (Bull. Acad. Roy. Belgique, xli, p. 583, 2 Pls.)
Gehuchten, A. van. Étude sur la structure intime de la cellule musculaire striée. (La Cellule, ii, pp. 289, 293–453, 1886, 6 Pls.)
Janet, Charles. Études sur les fourmis, les guêpes et les abeilles. 12e note. (Structure des membranes articulaires des tendons et des muscles, Limoges, 1895, pp. 25, 11 Figs.)
Also the writings of Burmeister, Chabrier, Leydig, Meckel, Lebert, Wagner, Wagener, Amici, Krause, Heppner, Retzius, Rollet, G. Elias Müller, F. Merkel, Hensen, Kölliker, Dogiel, Dönitz, Hagen, Vosseler, Bütschli u. Schewiakoff, Lowne, Ciaccio, Biedermann, Cohnbeim, Brücke, Haycraft, Melland, Bowman.
c. Muscular power of insects
Plateau, Félix. Sur la force musculaire des insectes. (Bull. Acad. Roy. Belgique, 2 Sér. xx, 1865, pp. 732–757; xxii, 1866, pp. 283–308.)
—— Recherches sur la force absolue des muscles des invertébrés. 1884.
Radan, R. La force musculaire des insectes. (Revue de deux mondes, 2 Sér., lxiv, 1866, pp. 770–777.)
Bibiakoff, Paul von. Zur Muskelkraft der Insekten. (Natur, xvii, 1868, p. 399.)
Delbœuf. Nains et géants, Étude comparative de la force des petits et des grands animaux. Bruxelles. (Also in Kosmos, xiii, 1883, pp. 58–62.)
Camerano. Mem. Acc. Torino (2), xliii, 1893, p. 229.
Also Newport, Art. Insecta, p. 76. Kirby and Spence, Burmeister, Graber, Kolbe, pp. 375, 376.
THE NERVOUS SYSTEM
a. The nervous system as a whole
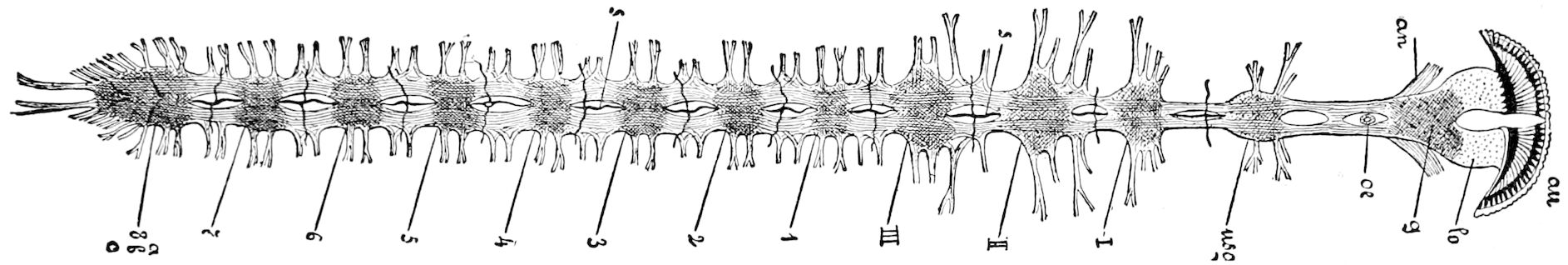
Fig. 239.—Central nervous system of Machilis maritima: au, eye; lo, optic tract; g, brain; an, antennal nerve; oe, œsophagus passing between the œsophageal commissures; usg, infraœsophageal ganglion; I-III, thoracic ganglia; 1–8, abdominal ganglia, the last (Sabc) consisting of three fused ganglia; s, sympathetic nervous system of the ventral cord.—After Oudemans, from Lang.
The nervous system of insects consists of a double series or chain of ganglia connected by nervous cords or commissures. The first of these is the brain or supraœsophageal ganglion; it is situated in the upper part of the head, above the gullet or œsophagus, while the rest of the system, called the ventral cord, lies on the floor of the body, under the digestive canal.
A ganglion or nerve-centre consists of a mass of ganglion-cells, from each of which a process or fibre passes off, uniting with others to form a nerve; by means of these nerves the ganglia are connected with other ganglia, and with the sensory cells and muscle-fibres. The ganglia may be simple, and arranged in pairs, corresponding to each segment of the body, or they may be compound, the result of the fusion of several pairs of ganglia, which in the early stages of the embryo are separate. Thus the brain of insects is a compound ganglion, or ganglionic mass.
The nerves are of two kinds: 1. Sensory, which transmit sensations from the peripheral sense-cells to the ganglion, or brain; 2. Motor, which send stimuli from the brain or any other ganglion to the muscles.
Of ganglion cells, some are tactile, and others give rise to nerves of special sense, being distributed to the eyes, or to the organs of hearing, smell, taste, or touch.
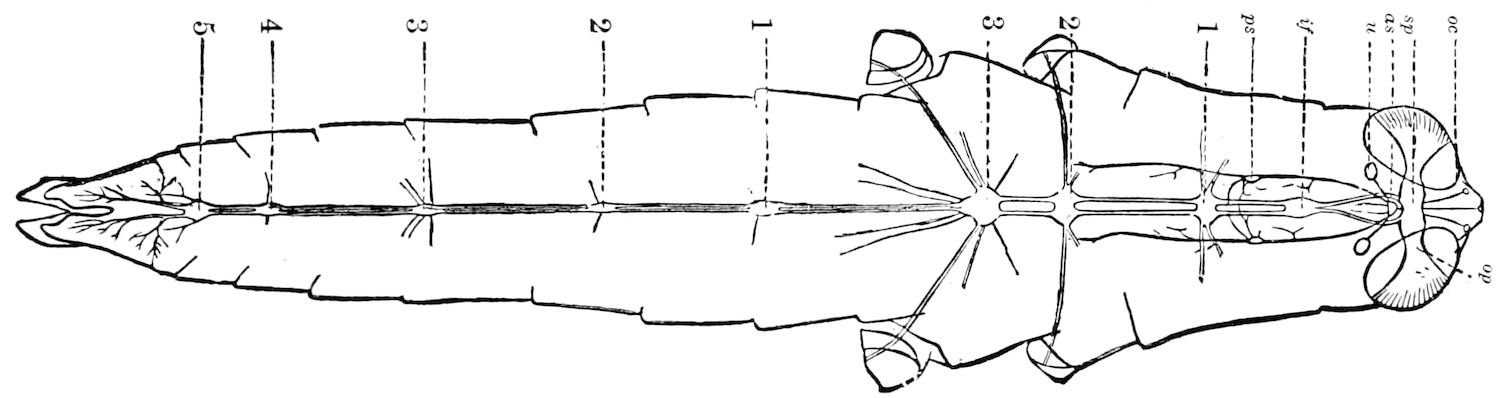
Fig. 240.—Nervous system of Melanoplus spretus: sp, supraœsophageal ganglion, sending off the large optic nerve (op) to the eyes, and an ocellar nerve to each ocellus (the dotted line oc stops short of the left ocellus); if, infraœsophageal ganglion; 1, 2, 3, thoracic ganglia; 1–5, five abdominal ganglia (the fifth the largest, and sending branches to the ovipositor, etc.) The sympathetic nerve and ganglia are represented by the two main nerves which arise from the medio-cephalic (as) resting on and above the œsophagus, and two ganglia (ps) on the under side of the crop. From each of these ganglia, two nerves are sent under the crop, and a larger nerve on each side to as far as the stomachal cæca, ending the figure at the dotted line 2, near the second thoracic ganglion. u, a round, shining body, connected by a nerve with the medio-cephalic ganglion, its nature unknown.
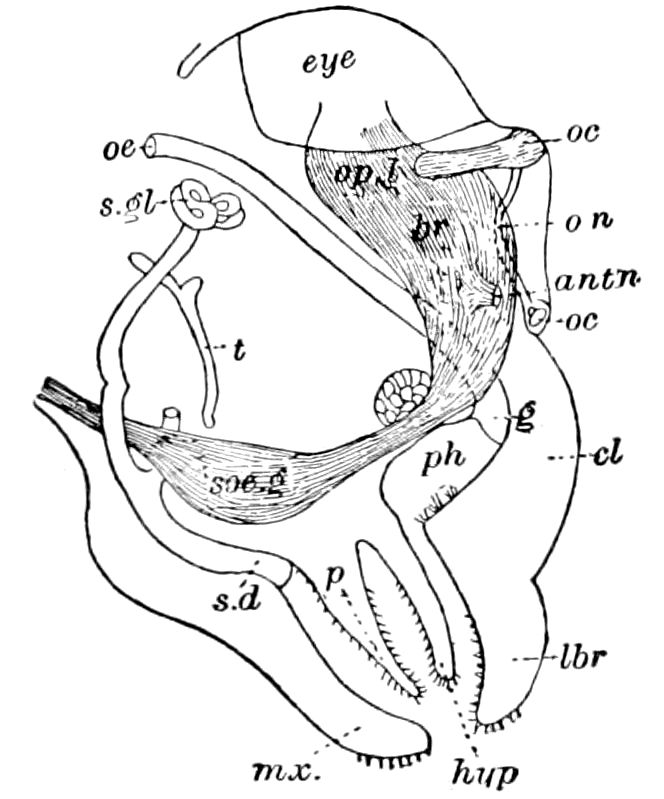
Fig. 241.—Section through the head of Machilis, showing the brain (br), and subœsophageal ganglion (soe. g); cl, clypeus; lbr, labrum; oc, ocellus.
While the supraœsophageal ganglion, or “brain,” of the insect is much more complex than any other ganglion, consisting more exclusively both of sensory as well as motor ganglia and their nerves, it should be borne in mind that the subœsophageal ganglion also receives nerves of special sense, situated on the palpi and on the tongue, as in the bee and other insects; hence this ganglion is probably complex, consisting of sensory and motor cells. The third thoracic ganglion is also, without doubt, a complex one, as in the locusts the auditory nerves pass into it from the ears, which are situated at the base of the abdomen, while in the green grasshoppers, such as the katydids and their allies, whose ears are situated in their fore legs, the first thoracic ganglion is a complex one. In the cockroach and in Leptis (Chrysopila), a common fly, the caudal appendages bear what are probably olfactory organs, and as these parts are undoubtedly supplied from the last abdominal ganglion, this is probably composed of sensory and motor ganglia; so that we have in the ganglionated cord of insects a series of brains, as it were, running from head to tail, and thus in a still stronger sense 224than in vertebrates the entire nervous system, and not the brain alone, is the organ of the mind of insects.
The simplest, most primitive form of the nervous system of insects is seen in that of the Thysanura. That of Campodea has not yet been fully examined, but in that of the more complicated genus, Machilis (Fig. 239), we see that there is a pair of ganglia to nearly each segment, while the brain (Fig. 241) is composed of three lobes, viz. the optic, the cerebral (Fig. 239, g), behind which is the antennal lobe, from which the antennal nerve takes its origin. Behind the opening for the throat (oe) is situated the first ganglion of the ventral cord, the subœsophageal ganglion, which gives rise to the nerves supplying the jaws and other mouth-parts.
Fig. 242, A-D.—The nervous systems of 4 genera of Diptera, to demonstrate their various degrees of fusion of ganglia: A, non-concentrated more primitive nervous system of Chironomus plumosus, with 3 thoracic and 6 abdominal ganglionic masses. B, nervous system of Empis stercorea, with 2 thoracic and 5 abdominal ganglionic masses. C, nervous system of Tabanus bovinus, with 1 thoracic ganglionic mass, and the abdominal ganglia closely approximated. D, highly modified nervous system of Sarcophaga carnaria, in which all the ganglia of the ventral cord behind the subœsophageal ganglion are fused into a single ganglionic mass.—After Brandt, from Lang.
In the Collembola, which are retrograde Thysanura, there are from one (Smynthurus), to three or four ventral ganglia.
In the winged insects, where the ganglia are more or less fused, the fusion taking place in the head and at the end of the abdomen; there are in the more simple and generalized forms, such as Ephemera, the grasshopper, locusts (Fig. 240), etc., thirteen ganglia besides the two pairs of compound ganglia in the head, three pairs of thoracic 225ganglia, and usually from five to eight pairs of ganglia in the abdomen.
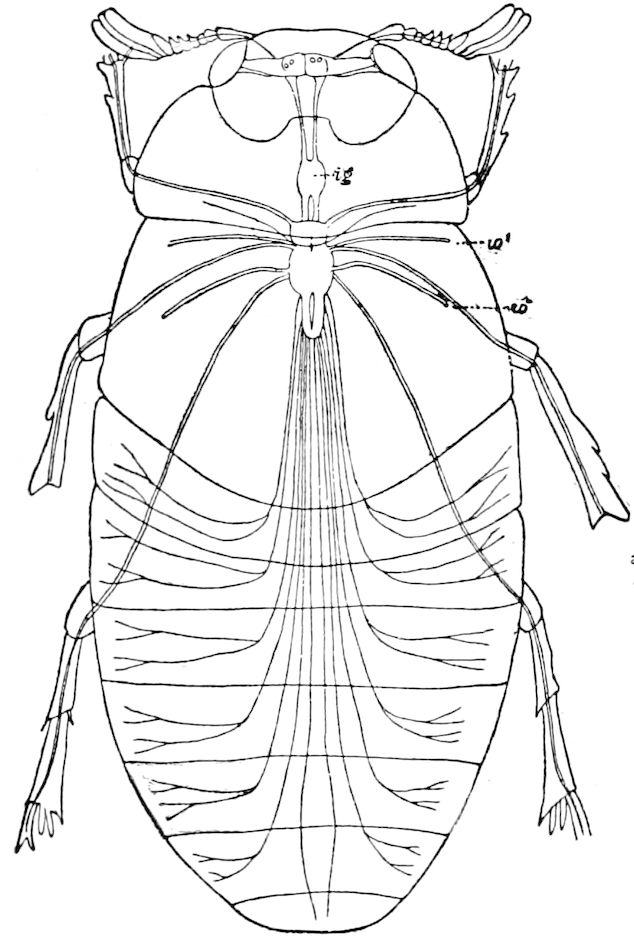
Fig. 243.—Nervous system of the May beetle, Lachnosterna fusca: w1, nerve to 1st,—w2, nerve to 2d, pair of wings; ig, infraœsophageal ganglion.
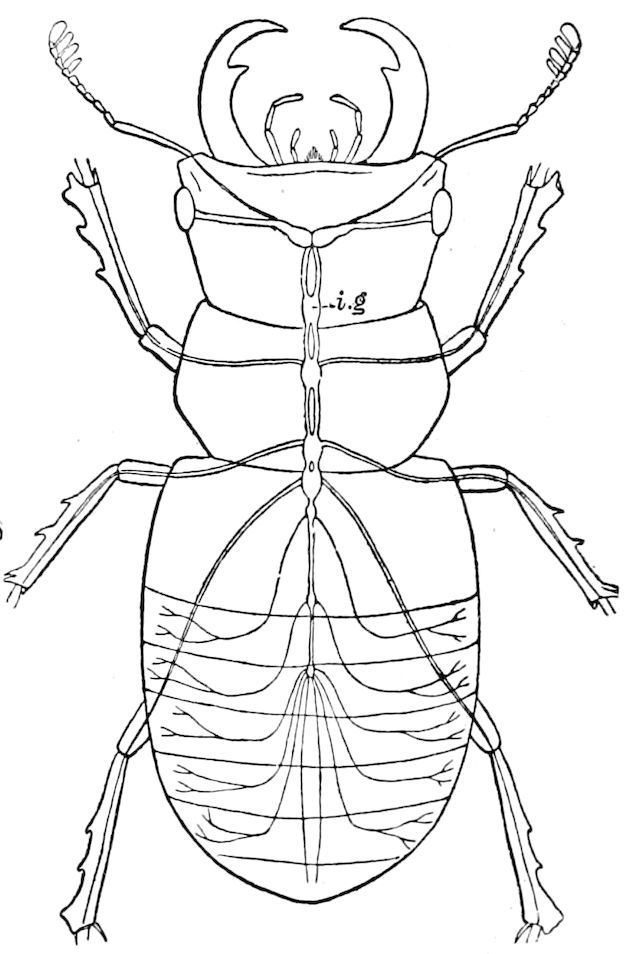
Fig. 244.—The same of the stag-beetle, Lucanus dama, where there are 3 thoracic, and 3 separate abdominal ganglia.
In certain winged insects the process of fusion or degeneration is carried to such an extreme that there are either no abdominal ganglia (Fig. 242, D), or their vestiges are situated in the thorax and partially fused with the thoracic ones, as in the May beetle, in which the prothoracic pair of ganglia is separate, while the two other thoracic ganglia are fused with the abdominal, the latter being situated in the thorax; this fusion is carried to a further extent than in any other Coleoptera yet examined. In many Diptera and Hemiptera the abdominal ganglia are either absent or the vestiges are fused with the thoracic ganglia.
Rhizotrogus, which is allied to our May beetle, as also Hydrometra and the Stylopidæ are said to lack the subœsophageal ganglion (Brandt).
In numerous Coleoptera (Acilius, Gyrinus, Necrophorus, Melolontha, Bostrichus, Rhynchænus); in many Diptera (Culex, Tipula, 226Asilus, Xylophaga, and Phora); and in the higher Hymenoptera (Crabronidæ, Vespidæ, and Apidæ), as well as in many Lepidoptera (Vanessa, Argynnis, and Pontia), two of the thoracic ganglia are fused together, while all three are partially fused into a single mass in many brachycerous Diptera (Conops, Syrphus, Pangonia, and the Muscidæ); in certain Hemiptera (Pentatoma, Nepa, and Acanthia); also in a beetle (Serica brunnea). Sometimes the subœsophageal ganglion is fused with the first thoracic, as in Acanthia, Nepa, and Notonecta. The greatest amount of variation is seen in the number of abdominal ganglia, all being fused into a single one or from one to eight. The fusion is usually greatest where the abdomen is shortened, due to the partial atrophy and modification of the terminal segments which bear the ovipositor, where present, and the genital armature.
There is only one pair of abdominal ganglia in Gyrinus and in certain flies (Conops, Trypeta, Ortalis, and Phora); two in Rhynchænus, a weevil, and in the flies, Syrphus and Volucella; three in Crabro and Eucera; four in Sargus, Stratiomys and in butterflies, five in the beetle, Silpha, and in the fly, Sciara, and the moth, Hepialus.
The nervous system in the larvæ of the metabolous orders is not concentrated, though in that of the neuropterous Myrmeleo it has undergone fusion from adaptation to the short compressed form of this insect.
b. The brain
The brain of insects appears to be nearly, if not quite, as complex as that of the lower vertebrates. As in the latter, the pair of supraœsophageal ganglia, or brain, is the principal seat of the senses, the chief organ of the insect’s mind.
It is composed of a larger number of pairs of primitive ganglia than any of the succeeding nerve-centres, and is, structurally, entirely different from and far more complicated than the other ganglia of the nervous system. It possesses a central body in each hemisphere, a “mushroom body,” optic lobes and optic ganglia and olfactory lobe, with their connecting and commissural nerve-fibres, and a number of other parts not found in the other ganglia.
In the succeeding ganglia the lobes are in general motor; the fibres composing the œsophageal commissures, and which arise from the œsophageal commissural lobes, extend not only to the subœsophageal ganglion, but pass along through the succeeding ganglia to the last pair of abdominal nerve-centres.[40] Since, then, there is a 227direct continuity in the fibres forming the two main longitudinal commissures of the nervous cord, and which originate in the brain, it seems to follow that the movements of the body are in large part directed or coördinated by the brain.[41] Still, however, a second brain, so to speak, is found in the third thoracic ganglion of the locust, which receives the auditory nerves from the ears situated in the base of the abdomen; or in the first thoracic ganglion of the green grasshoppers (katydids, etc.), whose ears are situated in their fore legs; while even the last pair of abdominal ganglia in the cockroach and mole cricket, is, so to speak, a secondary brain, since it distributes sensory nerves to the caudal stylets, which are provided with organs probably olfactory in nature.
It is impossible to understand the morphology of the brain unless we examine the mode of origin of the nervous system in the early life of the embryo. The head of an embryo insect consists of six segments, i.e. the ocular, antennal, premandibular, mandibular, and the 1st and 2d maxillary segments, so named from the appendages they bear. Of these the first three in the larva and adult are preoral, and the last three are postoral. The antennal segment was probably either postoral in the progenitors of insects, or the antennæ were inserted on the side of the mouth, the latter finally moving back.[42]
The nervous system in the early embryonic condition, as shown by Wheeler (Fig. 245), at first consists of nineteen pairs of primitive ganglia, called neuromeres. Those of the head, which later in embryonic life fuse together to form the brain, are the first three, corresponding to the protocerebrum, deutocerebrum, and tritocerebrum 228of Viallanes. The first pair of primitive ganglia, and which is situated in front of the mouth, is divided into three lobes.
Fig. 245, A-D.—Diagrams of four consecutive stages in the development of the brain and nerve-chain of the embryo of Xiphidium: I, cephalic,—II, thoracic,—III, abdominal, region; st, stomodæum or primitive mouth; an, anus; e, optic plate; pc(og), 1st protocerebral lobe, or optic ganglion; pc2, pc3, 2d and 3d protocerebral lobes; dc, deutocerebrum; tc, tritocerebrum; 1–16, the 16 postoral ganglia; po. c, postoral commissure; fp, furcal pit; ac, anterior,—pc, posterior, ganglionic commissure; ag, anterior,—pg, posterior,—cg, central,—lg, lateral gangliomeres.—After Wheeler.
The first or outermost lobe, according to Wheeler, forms the optic ganglion of the larva and imago, while the second and third lobes. (pc2, pc3) ultimately form the bulk of the brain proper, or the protocerebral lobes. The second (primitively postoral) brain-segment or pair of ganglia gives origin to the antennæ, while the third brain, or premandibular (intercalary) segment, gives origin to a temporary embryonic pair of appendages found in Anurida and Campodea (the premandibular ganglia), and also to the nerves supplying the labrum. These three pairs of ganglia later on in embryonic life become preoral, the mouth moving backwards. The three pairs of primitive ganglia, behind, i.e. the mandibular and 1st and 2d maxillary ganglia, become fused together to form the subœsophageal ganglion, and which in larval and adult life is postoral.
229If the tongue (ligula, or hypopharynx) represents a distinct pair of appendages, then there are seven segments in the head.
Fig. 246.—Section through head of a carabid, Anopthalmus telkampfii: br, brain; fg, frontal ganglion; soe, subœsophageal ganglion; co, commissure; n. l, nerve sending branches to the lingua (l); mn, maxillary nerve; mx, 1st maxilla; mm, maxillary muscle; mx′ 2d maxilla; mt, muscle of mentum; le, elevator muscle of the œsophagus; l′ of the clypeus, and a third beyond raising the labrum (lbr); eph, epipharynx; g. g′, salivary glands above; g2, lingual gland below the œsophagus (œ); m, mouth; pv, proventriculus; md, mandible.
The brain, then, supplies nerves to the compound and simple eyes, and to the antennæ, and gives origin to the sympathetic nerves; it is thus the seat of the senses, also of the insect’s mind, and coördinates the general movements of the body.
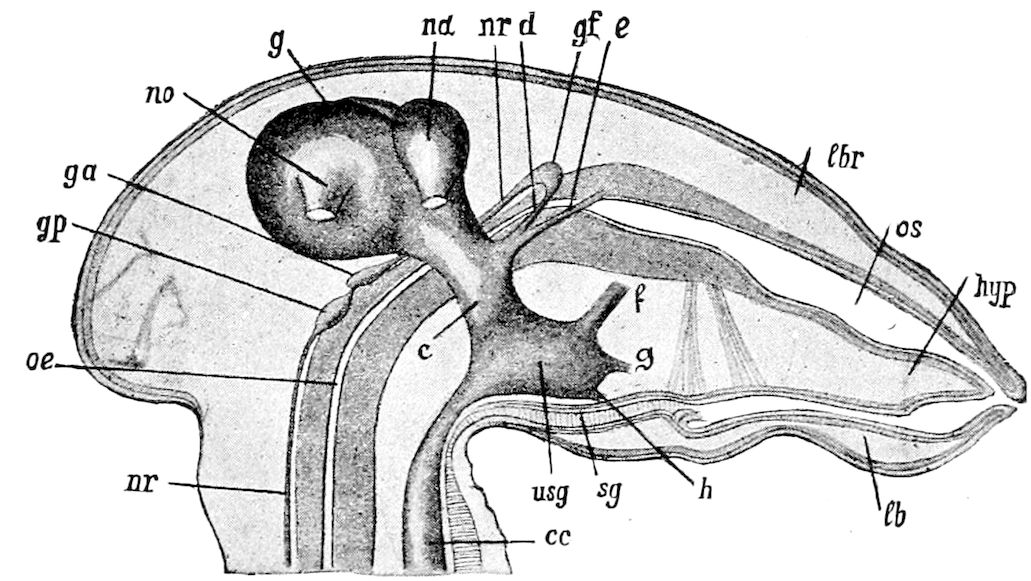
Fig. 247.—Median longitudinal section through the head of Blatta orientalis. The nervous system of the head is drawn entire. hyp, hypopharynx; os. oral cavity; lbr, upper lip; gf, frontal ganglion; g, brain; na, root of the antennal nerve; no, root of the optic nerve; ga, anterior,—gp, posterior ganglion of the paired visceral nervous system; œ, œsophagus; c, œsophageal commissure; usg, infraœsophageal ganglia; cc, longitudinal commissure between this and the first thoracic ganglion; sg, common duct of the salivary glands; lb, labium (2d maxillæ); nr, recurrent nerve; d, nerve uniting the frontal ganglion with the œsophageal commissure; e, nerve from this commissure to the labrum; f, nerve from the infraœsophageal ganglion to the mandible, —g, to the 1st maxillæ, —h, to the lower lip (2d maxillæ).—After Hofer, from Lang.
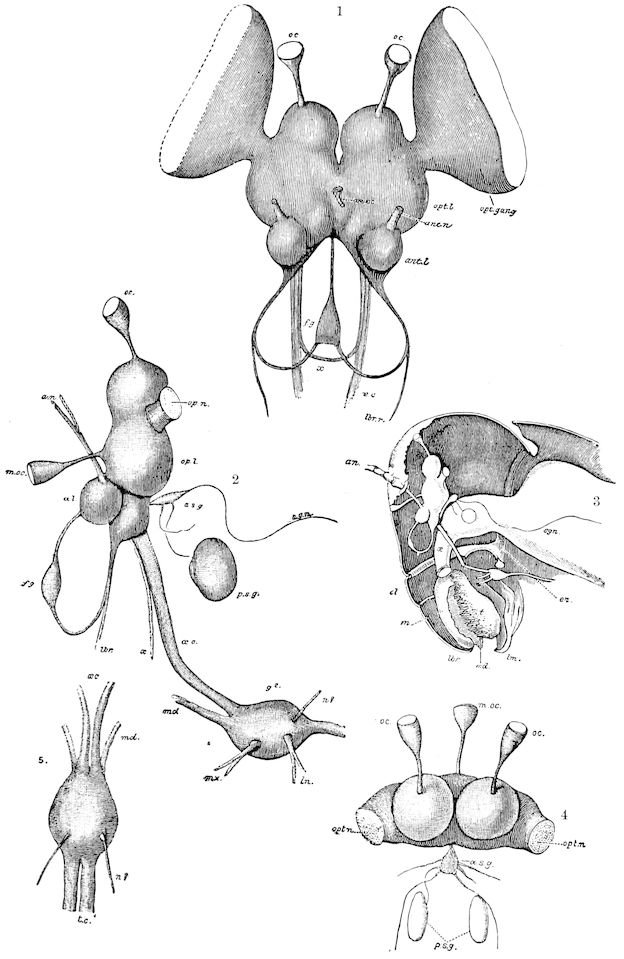
Fig. 248.—1, front view of the brain of Melanoplus femur-rubrum: opt. gang, optic ganglion; oc, ocelli and nerves leading to them from the two hemispheres, each ocellar nerve arising from the region containing the calices; m. oc, median ocellar nerve; opt. l, optic lobe sending off the optic nerve to the optic ganglion; ant. l, antennal or olfactory lobe; ant. n, antennal nerve; f. g, frontal ganglion of sympathetic nerve; lbr. n, nerve to labrum; x, cross-nerve or commissure between the two hemispheres; œ. c, œsophageal commissure to subœsophageal ganglion. 2, side view of the brain and subœsophageal ganglion (lettering of brain as in 1): s. g, stomatogastric or sympathetic nerve; a. s. g, anterior, and p. s. g, posterior, sympathetic ganglia; g2, subœsophageal ganglion; md, nerve to mandible; mx, maxillary nerve; ln, labial nerve; nl, unknown nerve,—perhaps salivary. 3, interior view of the right half of the head, showing the brain in its natural position: an, antenna; cl, clypeus; lbr, labrum; m, mouth-cavity; md, mandible; t, tongue; œ, œsophagus; c, crop; en, right half of the endocranium or X-shaped bone, through the anterior angle of which the œsophagus passes, while the great mandibular muscles play in the lateral angles. The moon-shaped edge is that made by the knife passing through the centre of the X. 4, view of brain from above (letters as before). 5, subœsophageal ganglion from above: t. c, commissure to the succeeding thoracic ganglion (other letters as before). Fig. 3 is enlarged 8 times; all the rest 25 times.—Drawn from original dissections, by Mr. Edward Burgess, for the Second Report of the U. S. Entomological Commission.
The pair of subœsophageal ganglia distributes nerves to the mandibles, to the 1st and 2d maxillæ, and to the salivary glands (Fig. 248).
Its general shape and relations to the walls and to the outer organs of the head is seen in Figs. 247, 248. In all the winged insects (Pterygota) its plane is situated more or less at right angles to the horizontal plane of the ventral cord. On the dorsal and anterior sides are situated the ocular lobes, and below these the antennal lobes.
Viallanes first, independently of embryonic data, divided the brain of adult insects into three regions or segments; i.e. the “protocerebron,” “deutocerebron” and “tritocerebron,” which he afterwards found to correspond with the three primitive elements (neuromeres) of the brain and with the segments of the head of the embryo.
The brain of the locusts (Melanoplus and Œdipoda) being best known will serve as the basis of the following description, taken mainly from Viallanes, with minor changes in the name of the three segments, and other modifications.
I. The optic or procerebral segment is composed of a median portion, i.e. two fused procerebral lobes (median protocerebrum), and of two lateral masses, the optic ganglia (protocerebrum), and comprises the following regions fused together and forming the median procerebral mass (Viallanes):—
Fig. 249.—Diagram of an insect’s brain: cc, central body; cg, ganglionic cells; che, external, chi, internal chiasma; cœ, œsophageal commissure; cp, mushroom body; ctc, tritocerebral commissure; fpr, postretinal fibres; goc, ocellar ganglion; goc1, œsophageal ganglion, the dotted ring the œsophagus; gv1, gc2, gv3, 1st, 2d, 3d, unpaired visceral ganglion; gvl, lateral visceral ganglion; ld, dorsal lobe of the deutocerebrum; lg, ganglionic plate; lo, olfactory lobe; lpc, protocerebral lobe; me, external, mi, internal medullary mass; na, olfactory or antennal nerve; nl, nerve to labrum; no, ocular nerve; nt, tegumentary nerve; œ, œsophagus; plp, bridge of the protocerebral lobes; rvd, visceral root arising from the deutocerebrum; rvt, visceral root arising from the tritocerebrum; tr, tritocerebrum; to, optic nerve or tract.—After Viallanes.
Optic ganglia.—Each of the two optic ganglia is formed of a series of three ganglionic masses situated between the compound eyes and the median procerebral mass, i.e. the ganglionic plate (Fig. 249, lg), the external medullary mass (me), and the internal medullary mass (mi).
The postretinal fibres (fpr) arising from the facets or single eyes of the compound eye (ommatidia) pass into the ganglionic plate (lg), which is united within by the chiasmatic fibres (che, external chiasma) of the external medullary mass (me). The last is attached to the internal medullary mass (mi) by fibres (chi), some of which are chiasmatic, and others direct. Finally, the internal medullary mass connects with the median part of the protocerebrum by direct fibres forming the optic nerve or tract (to).
Procerebral lobes.—The median procerebral lobes are fused together on the median line, forming a single central mass. From each side 233or lobe arises the mushroom or stalked body. In the middle of the mass is the central body, and directly in front is the procerebral bridge (plp). The latter is a band uniting the two halves of the brain.
The procerebral lobes also give origin to the nerves to the ocelli (no).
Fig. 250.—Transverse section through the brain of the locust (Œdipoda and Caloptenus): c′, lower part of the wall of c, calyx;—st, stalk of the same; bpcl, bridge of the protocerebral lobes; mo, nerve of median ocellus; ch, transverse fascia of the optico-olfactory chiasma; fcb, fibrous region of the central body; lcb, tubercle of the central body; fch, descending fascia of the optico-olfactory chiasma; choo, superior fascia of the optico-olfactory chiasma; pt, protocerebral lobes; ld, dorsal lobe of the deutocerebrum; lt, tritocerebral lobe; gcld, gc, ganglion cells.—After Viallanes.
The mushroom or stalked bodies.—These remarkable organs were first discovered by Dujardin, who compared them to mushrooms, and observed that they were more highly developed in ants, wasps, and bees than in the lower insects, and thus inferred that the higher intelligence of these insects was in direct relation to the development of these bodies. We will call them the mushroom bodies.
These two bodies consist of a rounded lobular mass (the trabecula) of the procerebral lobe, from which arises a double stalk (Fig. 253), the larger called the cauliculus, the smaller the peduncle (or pedicel); these support the cap or calyx. The calices of the bee were compared by Dujardin to a pair of disks on each side of the brain as seen from above, “each disk being folded together and bent downwards before and behind, its border being thickened, and the inner 234portion radiated.” In the locust there are but two divisions of the calyx; in the cockroach, ants, wasps, and bees, four.
The shape and relation of the mushroom bodies are represented in Figs. 252 and 253. The bodies are connected by commissural fibres, and are connected with the optic ganglion of the same side, and with the central body; while they are connected with the antennal lobes by the optico-olfactory chiasma.
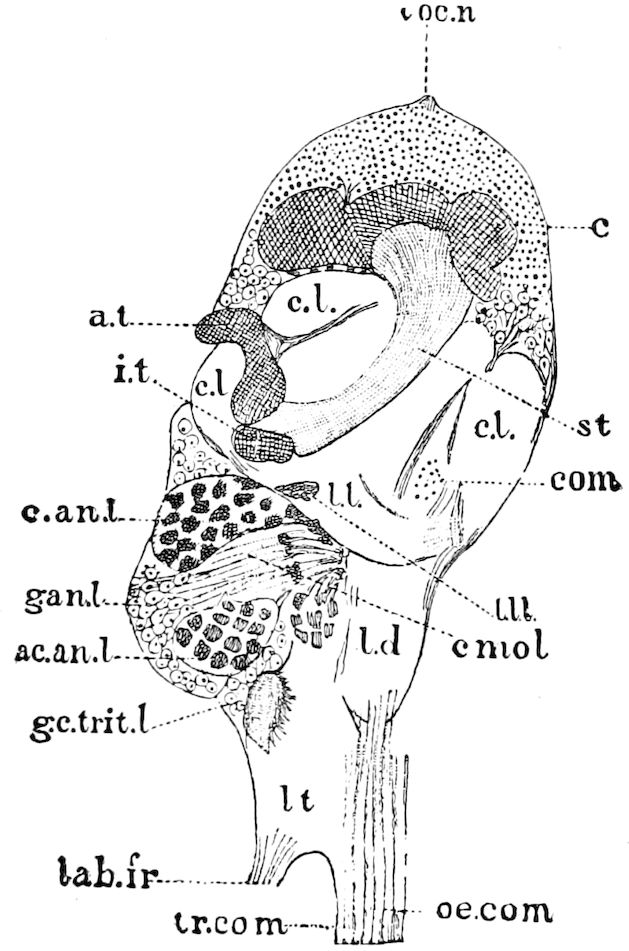
Fig. 251.—Sagittal section through the brain of the locust: l. oc. n, lateral ocellus nerve; a. t, anterior tubercle of the mushroom body; i. t, internal tubercle of the mushroom body; c. l, cerebral lobes; l. l, lateral lobe of the middle protocerebrum; com, commissural cord; c. mol, central mass of the olfactory lobe; ac. an. l, fibres uniting the median lobe of the middle protocerebrum with dorsal lobes of the deutocerebrum; gc. trit. l, ganglionated cortex of the tritocerebral lobe; c. an. l, cortex of antennal (olfactory) lobe; lab. fr, labrofrontal nerve; oe. com, œsophageal commissure; tr. com, transverse commissure of œsophageal ring; other letters as in Fig. 250.—After Viallanes.
The stalked bodies are enveloped by the cortical layers of ganglion-cells, those filling the hollow of the calyx having little or no protoplasm around the nucleus.
Structure of the mushroom bodies.—By staining the brain of the honey bee with bichromate of silver, Kenyon has worked out the structure of the mushroom bodies, with their cells. The cup-shaped bodies or calyces are composed of fibrillar substance (punktsubstanz). Each of these cups, he says, is “filled to overflowing with cells having large nuclei and very little cytoplasm.” From the under surface of each of these cups there descends into the general fibrillar substance of the brain “a column of fibrillar substance, which unites with its fellow of the same side to send a large branch obliquely downward to the median line of the brain, and an equally large or larger branch straight forwards to the anterior cerebral surface.”
The cells of the mushroom bodies, observes Kenyon, “stand out in sharp contrast to all other nerve cells known, though they recall to some extent the cells of Purkinje in the higher mammals. Each of the cells contained within the fibrillar cup sends a nerve-process into the latter, where it breaks up into a profusely arborescent system of branchlets, which often appear with fine, short, lateral processes, such as are characteristic of the dendrites of some mammalian nerve-cells.” Just before entering the fibrillar substance, a fine branch is given off that travels along the inner surface of the cup along with others of the same nature, forming a small bundle to the stalk of the mushroom body, down which it continues until it reaches the origin 235of the anterior and the inner roots above mentioned. “Here it branches, one branch continuing straight on to the end of the anterior root, while the other passes to the end of the inner root. Throughout its whole course the fibre and its two branches are very fine. Nearly the whole stalk and nearly the whole of each root is made up of these straight, parallel fibres coming from the cells within the cup of the mushroom bodies. What other fibres there are enter these bodies from the side, and branch between the straight fibres very much as the dendrites of the cells of Purkinje branch among the parallel fine fibres from the cells of the granular layer in the mammalian cerebellum. These fibres are of the nature of association fibres.”
Viallanes showed that from the olfactory or antennal lobes, as well as from the optic ganglia, there are tracts of fibres which finally enter the cups of the mushroom bodies, and Kenyon has confirmed this observation. Kenyon has also, by the Golgi method, detected another tract, before unknown, “passing down the hinder side of the brain, from the cups to the region above the œsophagus, where it bends forward and comes in contact with fibres from the ventral cord, which exists, although Binet was unable to discover any growth of fibres connecting the cord with the brain.
“The fibres entering the cups from the antennal lobe, the optic ganglia, and the ventral region, spread out and branch among the arborescent endings of the mushroom-body cells. The fibres branching among the parallel fibres of the roots and the stalk lead off to lower parts of the brain, connecting with efferent or motor-fibres, or with secondary association fibres, that in their turn make such connections. This portion of the circuit has not been perfectly made out, though there seems to be sufficient data to warrant the assumption just made.
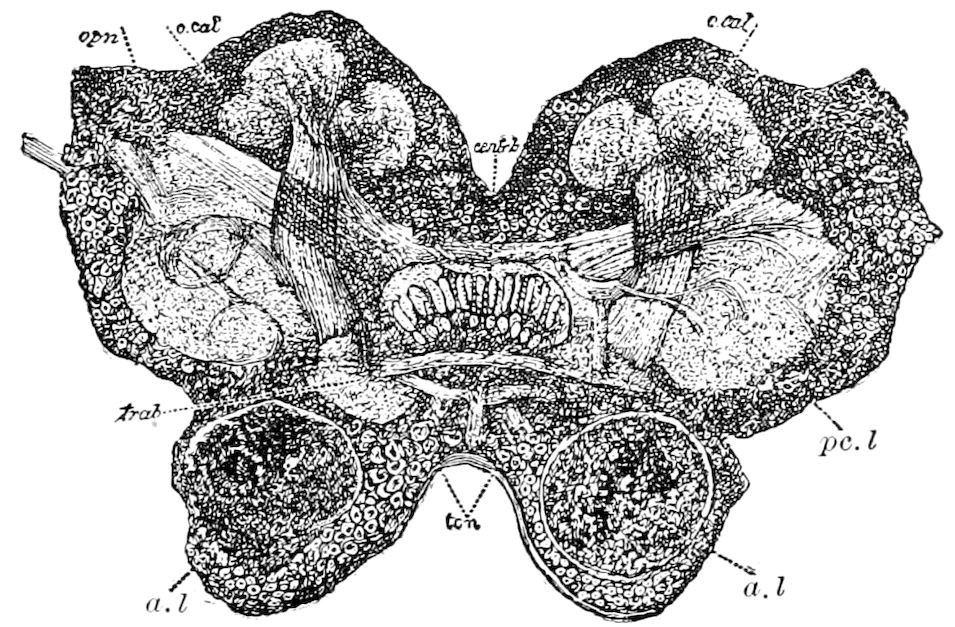
Fig. 252.—Section 17, showing the central body (centr. b) and mushroom body, optic and antennal lobes (a. l), and procerebral lobes (pc. l); o. cal, outer division of the calyx; op. n, optic nerve; trab, trabeculum; tc. n, transverse nerve.
“Such fibres existing as described, there is then a complete circuit for sensory stimuli from the various parts of the body to the cells of the mushroom bodies. The dendritic or arborescent branches of these cells take them up and pass them on out along the parallel fibres or neurites in the roots of the mushroom bodies as motor or other efferent impulses.
“This, however, is not all. For there are numerous fibres evident in my preparations, the full courses of which I have not been thus far able to determine, but which are so situated as to warrant the inference that they may act as 236association fibres between the afferent fibres from the antennæ, optic ganglia, and ventral system, and the efferent fibres. There is then a possibility of a stimulus entering the brain and passing out as a motor impulse without going into the circuit of the fibres of the mushroom bodies; or, in other words, a possibility of what may be compared to reflex action in higher animals.”
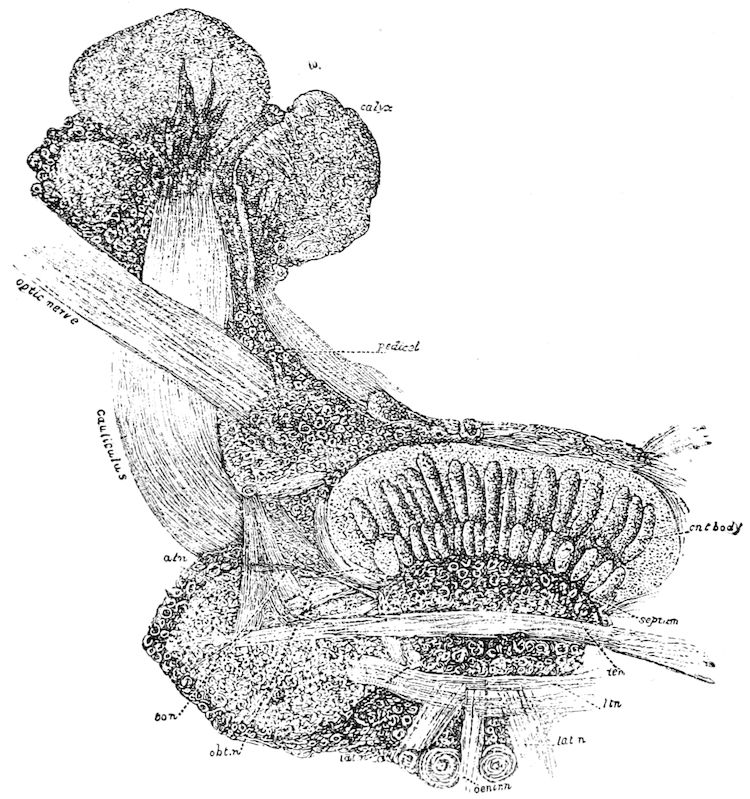
Fig. 253.—Enlarged view of the trabeculum (the dotted lines tcn and obt. n pass through it) and its nerves, of the mushroom body,—its calices and stalk, and the origin of the optic nerve × 225 diameters: atn, ascending trabecular nerve; obt. n, oblique trabecular nerve; tcn, transverse nerve; lat. n, lateral nerve; cent. n, central nerve.
The mushroom bodies have not yet been found to be present in the Synaptera, but occur in the larvæ, at least of those of most metamorphic insects (Lepidoptera and Hymenoptera), though not yet found in the larvæ of Diptera. The writer has found these bodies in the nymphs of the locust (Melanoplus spretus), but not in the embryo just before hatching. They occur in the third larval or nymph stage of this insect. It is evident that by the end of the first larval stage the brain attains the development seen in the third larval state of the two-banded species (C. bivittatus).
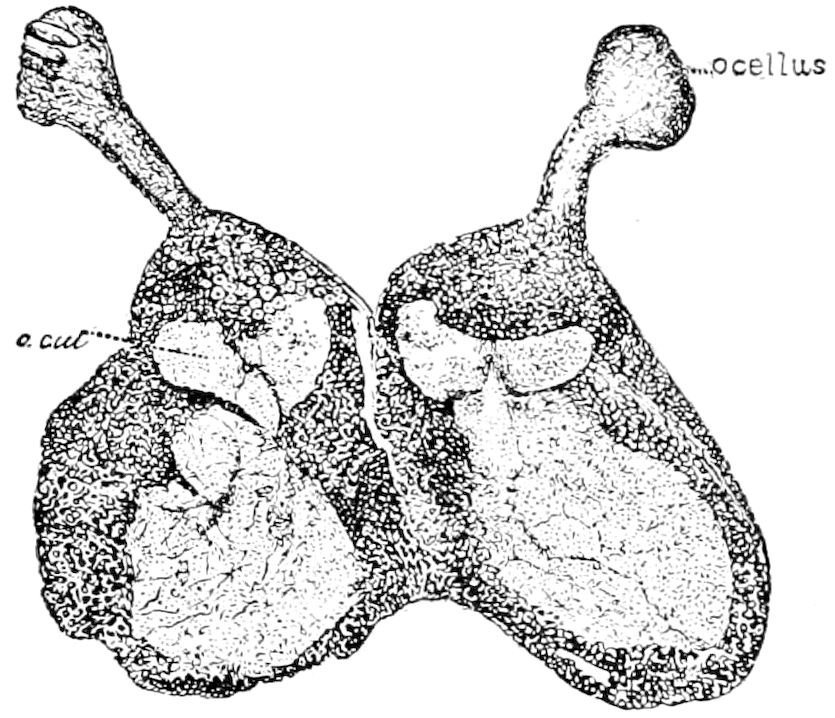
Fig. 254.—Section through the brain of Caloptenus bivittatus in the third larval stage, showing the two hemispheres or sides of the brain, and the ocelli and ocellar nerves, which are seen to arise from the top of the hemispheres directly over the calices (compare Fig. 251): o. cal, outer division of calyx of left mushroom body.
The result of our studies on the brain of the embryo locust was that from the embryonic cerebral lobes are eventually developed the central body and the two mushroom bodies. Fig. 254 shows the early condition of the mushroom bodies and their undoubted origin from the cerebral ganglia. Hence these bodies appear to be differentiations of the cerebral ganglia or lobes, having no connection with the optic or antennal lobes.
The central body (Fig. 252, centr. b).—This is the only single or unpaired organ in the brain. Dietl characterizes it as a median commissural system. Viallanes describes it as formed entirely of a very fine and close fibrillar web, like a thick hemispherical skull-cap, situated on the median line and united with the cerebral lobes. “It is like a central post towards which converge fibres passing from all points of the brain; being bound to the cerebral lobes, to the stalked bodies, to the optic ganglia, and to the olfactory lobes by distinct fibrous bundles.”
The antennal or olfactory lobes (Deutocerebrum).—This portion of the brain consists of two hemispherical lobes, highly differentiated for special sensorial perceptions, and connected by a slightly differentiated medullary mass, the dorsal lobe (Figs. 248, 249 lo), from which arise the motor fibres and those of general sensibility. The antennal lobes are in part attached to the optic ganglia, and partly to the stalked body on the same side, by the optic olfactory chiasma (Fig. 250 fch, choo), a system of fibres partially intercrossed on the median line.
The œsophageal lobes (Tritocerebrum) (Figs. 249, 250).—From this region the labrum and viscera are innervated, the nerves to the latter being called the visceral, sympathetic, or stomatogastric system. As Viallanes remarks, though plainly situated in front of the mouth, they are in fact post-œsophageal centres. The two lobes are situated far apart, and are connected by a bundle of fibres passing behind the œsophagus, called the transverse commissure of the œsophageal ring (Lienard). The œsophageal ganglia, besides giving 238rise to the labral nerves, also give origin to the root of the frontal ganglion.
c. Histological elements of the brain
The brain and other ganglia are composed of two kinds of tissue.
1. The outer slightly darker, usually pale grayish white portion consists of cortical or ganglion-cells differing in size. This portion is stained red by carmine, the cells composing it readily taking the stain.
The large ganglion cells (represented in Figs. 252 and 253) are oval, and send off usually a single nerve-fibre; they have a thin fibrous cell-wall, and the contents are finely granular. The nucleus is very large, often one-half the diameter of the entire cell, and is composed of large round refractive granules, usually concealing the nucleolus.
2. The medullary or inner part of the brain consists of matter which remains white or unstained after the preparation has remained thoroughly exposed to the action of the carmine. It consists of minute granules and interlacing fibres. The latter often forms a fine irregular network inclosing masses of finely granulated nerve matter.
This is called by Dietl “marksubstanz.” Leydig, in his Vom Bau des thierischen Körpers, p. 89, thus refers to it:—
“In the brain and ventral ganglia of the leech, of insects, and in the brain of the gastropods (Schnecken) I observe that the stalks (stiele) of the ganglion-cells in nowise immediately arise as nerve-fibres, but are planted in a molecular mass or punktsubstanz, situated in the centre of the ganglion, and merged with this substance. It follows, from what I have seen, that there is no doubt that the origin of the nerve-fibres first takes place from this central punktsubstanz.”
“This relation is the rule. But there also occur in the nerve-centres of the invertebrates single, definitely situated ganglion-cells, whose continuations become nerve-fibres without the intervention of a superadded punktsubstanz.” We may, with Kenyon, call it the fibrillar substance.
Leydig subsequently (p. 91) further describes this fibrillar substance, stating that the granules composing it form a reticulated mass of fibrillæ, or, in other words, a tangled web of very fine fibres:—
“We at present consider that by the passage of the continuation of the ganglion-cells into the punktsubstanz this continuation becomes lost in the fine threads, and on the other side of the punktsubstanz the similar fibrillar substance forms the origin of the axis-cylinders arranged parallel to one another; so it is quite certain that the single axis-cylinder derives its fibrillar substance as a mixture from the most diverse ganglion-cells.”
d. The visceral (sympathetic or stomatogastric) system
This system in insects is composed (1) of a series of three unpaired ganglia (Fig. 249, gv1, gv2, gv3), situated over the dorso-median line of the œsophagus, and connected by a median nervous cord or recurrent 239nerve (nr, vagus of Newport). The first of these ganglia is the frontal ganglion, which is connected with the œsophageal ganglia by a pair of roots (rvt), which have an origin primitively common with that of the labral nerves (Fig. 248, fg and lbr).
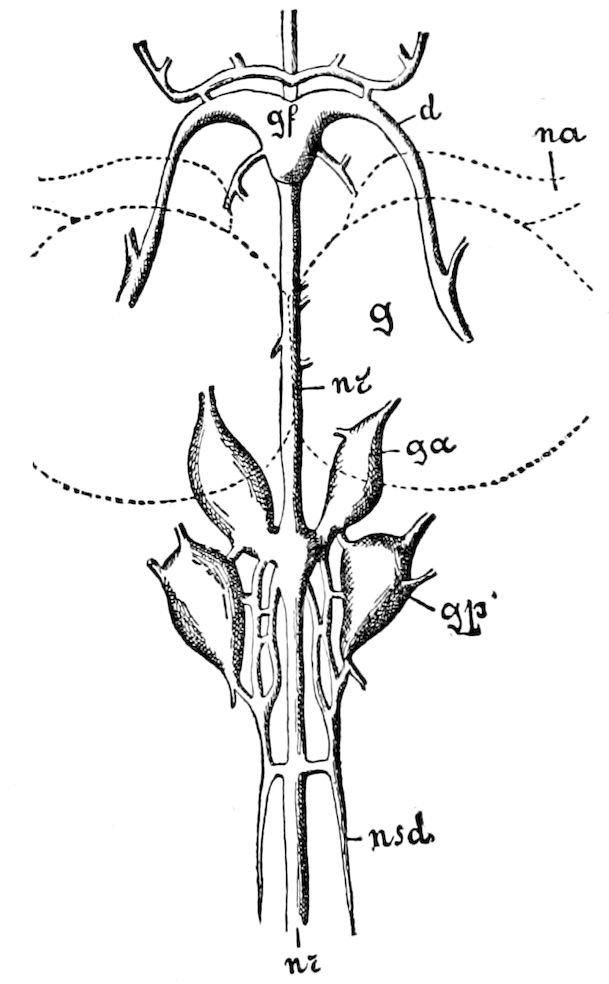
Fig. 255.—Anterior portion of the paired and unpaired visceral nervous system of Blatta orientalis seen from above. The outlines of the brain (g) and the roots of the antennal nerve (na), which cover a portion of the sympathetic nervous system, are given by dotted lines. Lettering as in Fig. 247. nsd, nerve to salivary gland. The nervus recurrens (nr) enters an unpaired stomach ganglion farther back.—After Hofer, from Lang.
2. Of two pairs of lateral ganglia (Fig. 255, ga, gp) situated two on each side of the œsophagus. They are connected both with the antennal lobes by a nerve (rvd), and to the chain of unpaired ganglia by a special connective. The first pair of these ganglia sends nerves to the heart and aorta; the second pair to the tracheæ of the head.
The unpaired median or recurrent nerve (nr) extends back from under the brain along the upper side of the œsophagus, and (in Blatta), behind the origin of the nerves to the salivary glands, enters an unpaired ganglion, called the stomachic ganglion (ganglion ventriculare), situated in front of the proventriculus. The number of these stomachic ganglia varies in different orders of insects.
In Blatta, Küpffer and also Hofer have shown (Fig. 255) (Müller, Brandt, ex Kolbe) that the nerve to each salivary gland arises from three different centres: the anterior end situated under the œsophagus is innervated by the paired visceral nerves from the hinder paired ganglia; the remaining part by nerves arising from each side of the recurrent nerve; and thirdly by a pair of nerves arising from the subœsophageal ganglion which accompanies the common salivary duct, and ends in branches which partly innervate the salivary glands and in part their muscles.
Hofer considers that the function of this complex system of paired and unpaired ganglia, with their nerves, is a double one, viz. serving both as a centre for the peristaltic action of the œsophagus, and as innervating the salivary glands.
Besides these a second portion of the visceral system arises from the thoracic and abdominal ventral cord. It may be seen in the 240simplest condition yet known in the nervous system of Machilis (Fig. 239 s). It consists of a fine, slender nerve, which extends along the surface of the ventral chain of ganglia, and sending off a pair of branches (accessory transverse nerves) in front of each ganglion. These accessory nerves receive nerve-twigs from the upper cord of the ventral chain, dilating near their origins into a minute elongated ganglion, and then passing partly outwards to the branches of the tracheæ and the muscles of the spiracles, uniting in the middle line of each segment of the body behind the head, i.e. of those segments containing a pair of ganglia.
e. The supraspinal cord
In the adult Lepidoptera has been detected, continuous with and on the upper side of the abdominal portions of the ventral cord, a longitudinal cord of connective tissue forming a white or yellowish band, and which seems to be an outgrowth of the dorsal portion of the neurilemma of the ventral cord. Muscles pass from it to the neighboring ventral portions of the integument. Its use is unknown, and attention was first called to it by Treviranus, who called it “an unknown ventral vessel” (Bauchgefäss). Afterwards it was re-discovered by Newport, who described it as “a distinct vascular canal.” But Burger has proved by cross-sections that it is not tubular, but a comparatively solid cord composed, however, of loose connective tissue. Newport found it in the larva of Sphinx ligustri, but Cattie states that it is not present in that of Acherontia atropos. It has not yet been observed in insects of other orders, but its homologue exists in the scorpion and in the centipede, and it may prove to correspond with the far more complete arterial coat which, with the exception of the brain, envelops the nervous system of Limulus.
f. Modifications of the brain in different orders of insects
There are different grades of cerebral development in insects, and Viallanes claimed that it was no exaggeration to say that the brain of the locust (Melanoplus) differs as much from that of the wasp as that of the frog differs from that of man. He insists that the physiological conditions which determine the anatomical modifications of the brain are correlated with 1, the food; 2, the perfection of the senses; and 3, with the perfection of the psychic faculties. For example, in those which feed on solid food and whose œsophagus is large (Orthoptera and Coleoptera), the connectives are elongated, the subœsophageal commissure free in all 241its extent, and the tritocerebrum is situated quite far from the preceding segment of the brain.
On the other hand, in insects which feed on fluid food (Hymenoptera, Lepidoptera, Diptera, Hemiptera), the œsophagus is slender and the nervous centres which surround them are very much condensed; the connectives are short, and the tritocerebrum is closely fused, partly to a portion of the antennal lobes (deutocerebrum) and partly to the mandibular ganglion.
As regards the perfection of the senses, where, as in dragon-flies, the eyes are very large, the optic ganglia are correspondingly so, and in the same insects the antennæ being very small, the antennal lobes are almost rudimentary. The ants exhibit inverse conditions; in their brain the antennal lobes are well developed, while the optic ganglia are reduced, and where, as in Typhlopone, the eyes are wanting, they are completely atrophied.
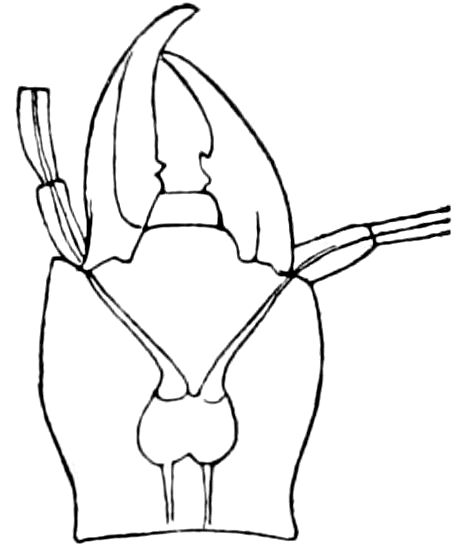
Fig. 256.—Head of Anophthalmus tellkampfii, showing the brain,—the optic ganglia, nerves, and eyes totally atrophied.
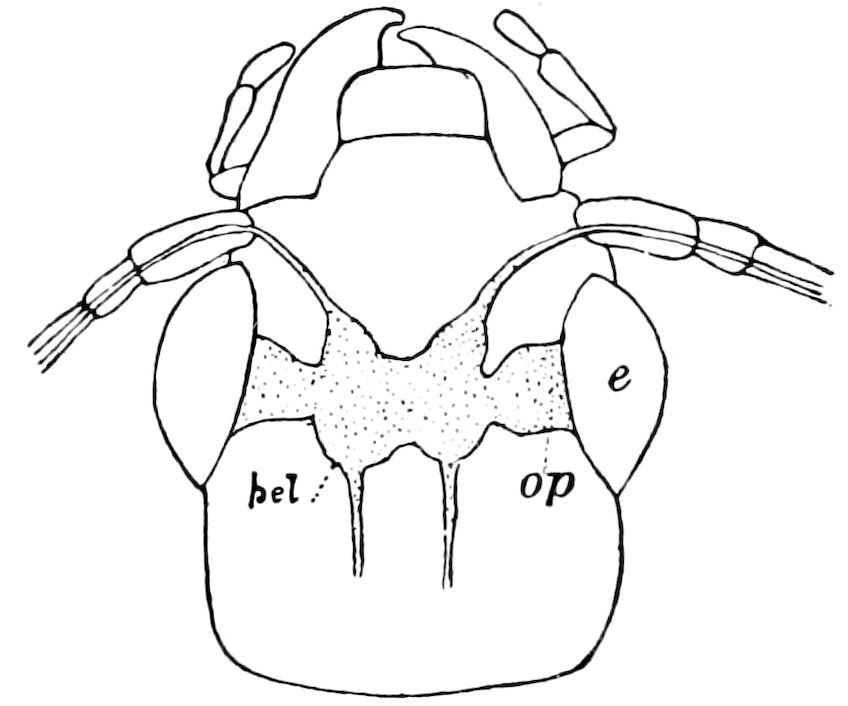
Fig. 257.—Head of another Carabid, with the brain and eyes normal: op, optic ganglion; pcl, brain.
In certain cave insects where the eyes are wanting, the optic ganglia are also absent. In the eyeless cave species of Anophthalmus the optic ganglia and nerves are entirely atrophied, as they are in Adelops, which, however, has vestiges of the facets (ommatidia). Fig. 257 represents the brain of Chlænius pennsylvanicus, a Carabid beetle, with its eyes and optic ganglia (op) which may be compared with Anopthalmus, in which these parts are totally atrophied.
Dujardin claimed that the degree of complication of the stalked body of the Hymenoptera was in direct relation with their mental powers. This has been proved by Forel, who has shown that in the honey bee and ants the mushroom bodies are much more developed in the workers than in the males or females and Viallanes adds that these bodies are almost rudimentary in the dragon-flies, whose eyes are so large; while on the contrary in the blind ants (Typhlopone), these bodies are as perfect and voluminous as in the ants with eyes.
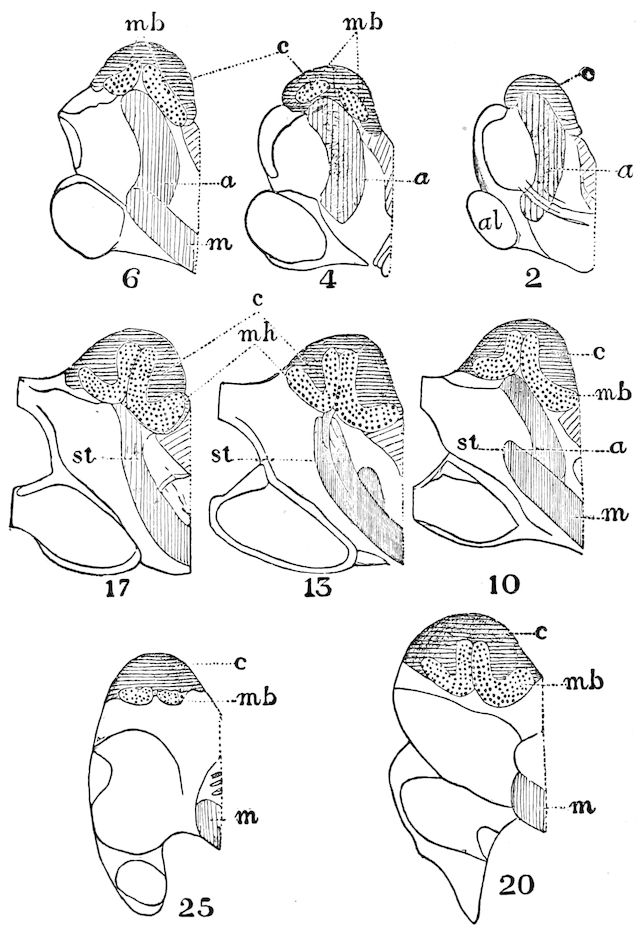
Fig. 258.—Diagrammatic outlines of sections of the upper part of the brain of a cockroach. Only one side of the brain is here represented. The numbers indicate the position in the series of 34 sections into which this brain was cut. mb, mushroom bodies, with their cellular covering (c) and their stems (st); a, anterior nervous mass; m, median nervous mass.—After Newton.
243Within the limits of the same order the stalked bodies are most perfect in the most intelligent forms. Thus in the Orthoptera, says Viallanes, the Blattæ, Forficulæ, and the crickets, the mushroom bodies are more perfect than in the locusts, which have simpler herbivorous habits. This perfection of the mushroom bodies is seen not only in the increase in size, but also in the complication of its structures. Thus in the groups with lower instincts (Tabanus, Æschna) the stalk does not end in a calyx projecting from the surface of the brain, but its end, simply truncated, is indicated externally only by an accumulation of the ganglionic nuclei which cover it.[43]
In types which Viallanes regards as more advanced, i.e. Œdipoda and Melanoplus, the end of the stalk projects and is folded into a calyx.
The brain of the cockroach (Periplaneta, Fig. 258) is a step higher than that of the locusts, each calyx being divided into two adjacent calices, although the cockroaches are an older and more generalized type than locusts.
The stalked bodies of cockroaches are thus complex, like those of the higher Hymenoptera, the calices in Xylocopa, Bombus, and Apis being double and so large as to cover almost the entire surface of the brain.
Finally, in what Viallanes regards as the most perfect type (Vespa), the sides of the calices are folded and become sinuous, so as to increase the surface, thus assuming an appearance which, he claims, strongly recalls that of the convolutions of the brain of the mammals.
Cheshire also calls attention to a progression in the size of these appendages,
as well as in mental powers as we rise from the cockchafer (Melolontha vulgaris)
to the cricket, up to the ichneumon, then to the carpenter bee, and finally
to the social hive bee, “where the pedunculated bodies form the ⅕ part of the
volume of the cerebral mass, and the 1
870 of the volume of the entire creature,
while in the cockchafer they are less than 1
2300 the part. The size of the brain
is also a gauge of intelligence. In the worker bee the brain is 1
174 of the body;
in the red ant, 1
296; in the Melolontha, 1
3500; in the Dyticus beetle, 1
4400.”
(Bees and bee-keeping, p. 54.)
g. Functions of the nerve-centres and nerves
As we have seen, the central seat of the functions of the nervous system is not the brain alone (supraœsophageal ganglion), but each ganglion is more or less the seat of vital movements, those of the 244abdomen being each a distinct motor and respiratory centre. The two halves of a ganglion are independent of each other.
According to Faivre, the brain is the seat of the will and of the power of coördinating the movements of the body, while the infraœsophageal ganglion is the seat of the motive power and also of the will.
The physiological experiments of Binet, which are in the line of those of Faivre, but more thorough, demonstrate that an insect may live for months without a brain, if the subœsophageal ganglion is left intact, just as a vertebrate may exist without its cerebrum. As Kenyon says: “Faivre long ago showed that the subœsophageal ganglion is the seat of the power of coördination of the muscular movements of the body. Binet has shown that the brain is the seat of the power directing these movements. ‘A debrained hexapod will eat when food is placed beneath its palpi, but it cannot go to its food even though the latter be but a very small space removed from its course or position. Whether the insect would be able to do so if the mushroom bodies only were destroyed, and the antennal lobes, optic lobes, and the rest of the brain were left intact, is a question that yet remains to be answered’” (Kenyon).
In insects which are beheaded, however readily they respond to stimulation of the nerves, they are almost completely wanting in will power. Yet insects which have been decapitated can still walk and fly. Hymenoptera will live one or two days after decapitation, beetles from one to three days, and moths (Agrotis) will show signs of life five days after the loss of their head.
That the loss of will power is gradual was proved by decapitating Polistes pallipes. A day after the operation she was standing on her legs and opening and closing her wings; 41 hours after the operation she was still alive, moving her legs, and thrusting out her sting when irritated. Ichneumon otiosus, after the removal of its head, remained very lively, and cleaned its wings and legs, the power of coördination in its wings and legs remaining. A horse-fly, a day after decapitation, was lively and flew about in a natural manner.[44]
When the abdomen is cut off, respiration in that region is not at first interrupted. The seat of respiratory movements was referred by Faivre to the hinder thoracic ganglion, but Plateau says that this view must be entirely abandoned, remarking: “All carefully performed experiments on the nervous system of Arthropoda have shown that each ganglion of the ventral chain is a motor centre, and 245in insects a respiratory centre, for the somite to which it belongs” (Miall and Denny’s The Cockroach, p. 164).
The last pair of abdominal ganglia serve as the nervous centre of the nerves sent to the genital organs.
The recurrent or stomatogastric nerve, which, through the medium of the frontal ganglion, regulates digestion, has only a slight degree of sensibility; the insect remains quiet even when a powerful allurement is presented to the digestive tract (Kolbe).
Faivre states that the destruction of the frontal ganglion, or a section of the commissures connecting it with the brain, puts an end to swallowing movements; on the other hand, stimulation results in energetic movements of this nature.
Yersin, by cutting through the commissure in different places, and thus isolating the ganglia of the nervous cord of Gryllus campestris, arrived at the following results:—
1. The section of a nerve near its origin rendered the organ supplied by this nerve incapable of performing its functions.
2. If the connectives between two ganglia, i.e. the second and third thoracic ganglia, are cut through, the fore as well as hinder parts of the body retain their power of motion and sensation; but a stimulus applied to the anterior part of the body does not pass to the hinder portion.
3. Insects with an incomplete metamorphosis after section of the connectives are not in every case unable to moult and to farther develop.
4. If only one of the two connectives be cut through, the appendages of the side cut through which take their origin between the place injured and the hinder end of the body, often lose sensation and freedom of motion, or the power of coördination of movements becomes irregular. Sometimes this is shown by an unsteadiness in the gait, so that the insect walks around in a circle; after a while these irregularities cease, and the movements of the limbs on the injured side are only slightly restrained. By a section of both connectives in any one place the power of coördination of movements is not injured.
5. The section of the connectives appear to have no influence on nutrition, but affects reproduction, the attempt at fertilization on the part of the male producing no result, and the impregnated female laying no eggs.
6. Injury to the brain, or to the subœsophageal, or one of the thoracic ganglia, is followed by a momentary enfeeblement of the ganglion affected. Afterwards there results a convulsive trembling, 246which either pervades the whole body or only the appendages innervated by the injured ganglion.
7. As a result of an injury to the brain there is such a lack of steadiness in the movements that the insect walks or flies in a circle; for instance, a fly or dragon-fly thus injured in flying describes a circle or spiral. Steiner, in making this experiment, observed that the insect circled on its uninjured side. The brain is thus a motor centre.
8. By injuring a thoracic ganglion, one or all the organs which receive nerves from the ganglion are momentarily weakened. Afterwards the functions become restored. Sometimes, however, the insect walks in a circle. Faivre observed that after the destruction of the metathoracic ganglion of Dyticus marginalis the hind wings and hind legs were partially paralyzed (Kolbe, ex Yersin).
LITERATURE ON THE NERVOUS SYSTEM
a. General
Newport, George. On the nervous system of the Sphinx ligustri L., and on the changes which it undergoes during a part of the metamorphoses of the insect. (Phil. Trans. Roy. Soc., London, 1832, pp. 383–398; 1834, pp. 389–423, Pls.)
Helmholtz, H. L. F. De fabrica systematis nervosi evertebratorum. Diss. in aug. Berolini, 1842.
Blanchard, E. Recherches anatomiques et zoologiques sur le système nerveux des animaux sans vertèbres. Du système nerveux des insectes. (Annales des Sciences nat., Sér. 3, v, 1846, pp. 273–379, 8 Pls.)
—— Du système nerveux chez les invertèbres dans ses rapports avec la classification de ces animaux. Paris, 1849.
—— in Cuvier’s Règne animal. (Edition accompagnée de planches gravées. Insectes. Pl. 3, 3a, and 4.)
Leidy, Joseph. History and anatomy of the hemipterous genus Belostoma. (Memoirs Amer. Acad. Arts and Sc., N. S. iv, 1849, pp. 57–67, 1 Pl.)
Scheiber, S. H. Vergleichende Anatomie und Physiologie der Œstridenlarven. (Sitzungsb. k. Akad. wiss. Wien. Math.-Naturwiss. Cl., xli, 1860, pp. 439–496; xlv, 1862, pp. 7–68; 5 Taf.)
Tullberg, Tycho. Sveriges Podurider. (K. Svenska vet. Akad. Handl. x, 1872, pp. 1–70, 12 Taf.)
Berlese, A. Osservazione sulla anatomia descrittiva del Gryllus campestris L. (Atti della soc. Veneto-Trentina, 1880, vii, pp. 200–299.)
Baudelot, E. Contributions à la physiologie der système nerveux des insectes. (Revue d. sc. nat., i, pp. 269–280, 1872.)
Studer, Th. Ueber Nervenendigung bei Insekten. Kleine Beiträge zur Histologie der Insekten. (Mitt. Naturf. Ges., Bern, 1874, pp. 97–104, 1 Taf.)
Brandt, E. Recherches anatomiques et morphologiques sur le système nerveux des insectes Hyménoptères. (Compt. rendus de l’Acad. Sc., Paris, 1875.)
—— Ueber das Nervensystem der Apiden. (Sitzungsb. d. naturf. Ges., in Petersbourg, vii, 1876.)
—— Ueber das Nervensystem der Schmetterlingsraupen. (Verhandl. der Russ. 247Ent. Gesellsch., x, 1877. Also 16 other articles with plates, in Horæ Soc. Ent. Ross., 1878–1882.)
Mark, E. L. The nervous system of Phylloxera. (Psyche, ii, pp. 201–207, 1879.)
Riley, Charles Valentine. The nervous system and salivary glands of Phylloxera. (Psyche, ii, pp. 225, 226, 1879.)
Cholodkowsky, N. Zur Frage über den Baue und über die Innervation der Speicheldrüsen der Blattiden. (Horæ Soc. Ent. Ross., 1881, xvi, pp. 6–9, 2 Taf.)
Liénard, V. Constitution de l’anneau œsophagien. (Archives de Biologie, i, pp. 381–391, 1880, 1 Taf.)
Michaels, H. Nervensystem von Oryctes nasicornis im Larven-, Puppen-, und Käferzustande. (Zeits. f. wissens. Zool., xxxiv, 1880, pp. 641–702, 4 Taf.)
Rossi, A. Sul modo di terminare dei nervi nei muscoli dell’ organo sonoro della Cicala commune (Cicada plebeja). (Mem. accad. sc. Bologna, 1880, 4 Ser., i, pp. 661–665.)
Foettinger, A. Sur le termination des nerfs dans les muscles des insectes. (Archiv de Biologie, i, 1880.)
Binet. Contribution à l’étude der system nerveux sous intestinal des insectes. (Journ. l’anat. et phys., xxx, pp. 449–580, 1894.)
Paulowa. Zum Bau des Eingeweide Nervensystems der Insekten. (Zool. Anzeiger., xviii, Feb. 25, 1895, pp. 85–87.)
Also the writings of Lyonet, Cuvier, Rolando, Straus-Durckheim, Leydig, Newport, Graber, Viallanes, Grassi, Oudemans.
b. The brain
Dujardin, F. Mémoires sur le système nerveux des insectes. (Annales des Sciences nat, Sér. 3, 1850, xiv, pp. 195–206, Pl. 1, 1850.)
Rabl-Rückhard. Studien über Insectengehirne. (Archiv für Anatomie, Physiologie, etc., herausg. von Reichert u. R. du Bois-Raymond, 1876, p. 480, Taf. i.)
Dietl, M. J. Die Organization des Arthropodengehirns. (Zeitschr. wissens. Zool., xxvii, 1876, p. 488, Taf. xxxvi.-xxxviii.)
Flogel, T. H. L. Ueber den einheitlichen Bau des Gehirns in den verschiedenen Insectenordnungen. (Zeitschr. wissens. Zool., xxx, Suppl., 1878, p. 556, Taf. xxiii, xxiv.)
Newton, E. T. On a new method of constructing models of the brains of insects, etc. (Journ. Quekett Microscopical Club, pp. 150–158, 1879.)
—— On the brain of the cockroach, Blatta orientalis. (Quart. Journ. Microscopical Science, July, 1879, p. 340, Pl. xv, xvi.)
Packard, A. S. The brain of the locust. (Chapter xi, Second Report of the U. S. Entomological Commission, pp. 223–242, Pls. ix-xv, 1880.)
Cuccati, Giovanni. Sulla stuttura del ganglio sopraesofageo di alcuni ortotteri. (Acrydium lineola, Locusta viridissima, Locusta (species?), Gryllotalpa vulgaris, Bologna, 1887, 4º, pp. 1–27, Pl. i-iv.)
—— Intorno alla struttura del cervello della Sonomya erythrocephala, nota preventiva. Bologna, 1887.
—— Ueber die Organization des Gehirns des Sonomya erythrocephala. (Zeitschr. f. wissens. Zool., 1888, xlvi, pp. 240–269, 2 Taf.)
Viallanes, H. Études histologiques et organologiques sur les centres nerveux et les organes des sens des animaux articulés.
1. Mémoire. Le ganglion optique de la langouste (Palinurus vulgaris). (Annal. d. Sc. Nat. Zool., 1884, 6e Sér., xvii, Art. 3, pp. 1–74, 5 Pls.)
2482. Mémoire. Le ganglion optique de la Libellule (Æschna maculatissima). (Ibid., 1885, 6e Sér., xviii, Art. 4, pp. 1–34, 3 Pls.)
3. Mémoire. Le ganglion optique de quelques larves de Diptères (Musca, Eristalis, Stratiomys). (Ibid., 1886, 6e Sér., xix, Art. M. 4, pp. 34, 2 Pls.)
4. Mémoire. Le cerveau de la guêpe (Vespa crabro et vulgaris). (Ibid., 1887, 7e Sér., ii, pp. 5–100, 6 Pls.)
5. Mémoire. 1. Le cerveau du criquet (Œdipoda cœrulescens et Caloptenus italicus). 2. Comparaison du cerveau des Crustacés et des Insectes. 3. Le cerveau et la morphologie du squelette céphalique. (Ibid., 1888, 7e Sér., iv, pp. 1–120, 6 Pls.)
—— Sur la structure interne du ganglion optique de quelques larves de Diptères. (Bull. Soc. Phil., Paris, 1885, 7e Sér., ix, pp. 75–78.)
—— La structure du cerveau des Hyménoptères. (Bull. Soc. Philomat., Paris, 1886, 7e Sér., x, pp. 82, 83.)
—— La structure du cerveau des Orthoptères. (Bull. Soc. Philomat., Paris, 1886, 7e Sér., xi, pp. 119–126.)
—— Sur la morphologie comparée du cerveau des Insectes et des Crustacés. (Compt. rend. Acad. Sc. Paris, 1887, civ, pp. 444–447.)
Kenyon, F. C. The meaning and structure of the so-called “mushroom bodies” of the hexapod brain. (Amer. Naturalist, xxx, 1896, pp. 643–650, 1 fig.)
—— The brain of the bee. (Journ. Comp. Neurology, vi, fasc. 3, 1896, pp. 133–210.)
—— The optic lobes of the bee’s brain in the light of recent neurological methods. (Amer. Nat., xxxi, 1897, pp. 369–376, 1 Pl.)
With the embryological works of Graber, Heider, Korscheldt, Patten, Wheeler, etc.
c. Histology of the nervous System
Helmholtz. De fabrica systematis nervosi evertebratorum. Diss. Berolini, 1842.
Remak. Ueber d. Inhalt d. Nervenprimitivröhren. (Archiv f. Anat. u. Phys., 1843.)
Leydig. Lehrbuch der Histologie der Menschen und der Thiere. 1857.
—— Vom Bau des thierischen Körpers. i. 1864.
—— Tafeln zur vergleichenden Anatomie. i. Tübingen, 1864.
—— Zelle und Gewebe, neue Beiträge zur Histologie des Tier-Körpers. Bonn, 1885, pp. 219, 6 Taf.
Walter. Mikroscopische Studien über das Centralnervensystem wirbelloser Thiere. 1863.
Dietl, M. J. Die Gewebselemente des Centralnervensystems bei wirbellosen Thieren. (Aus den Berichten des naturw.-medic. Vereins in Innsbruck.) Innsbruck, 1878.
Berger. Untersuchungen über den Bau des Gehirns und der Retina der Arthropoden. (Arbeiten des zool. Instituts zu Wien, Heft 2, p. 173, 1878.)
—— Nachtrag zu den Untersuchungen über den Bau des Gehirns und der Retina der Arthropoden. (Ibid., Heft 3.)
Viallanes, H. Recherches sur l’histologie des insectes, etc. Paris, 1882. (Annales des Sciences nat., pp. 1–348, Pls. 1–18.)
—— Sur la structure de la substance ponctuée des insectes. Paris, 1885.
Haller, B. Ueber die sogenannte Leydig’sche Punktsubstantz im Centralnervensystem. (Morp. Jahrb., xi, 1886.)
Nansen, F. The structure and combination of the histological elements of the central nervous system. (Bergen’s Museum Aarsberetning for 1886. Bergen, 1887.)
Also the writings of Benedicenti, Holmgren.
THE SENSORY ORGANS
a. The eyes and insect vision
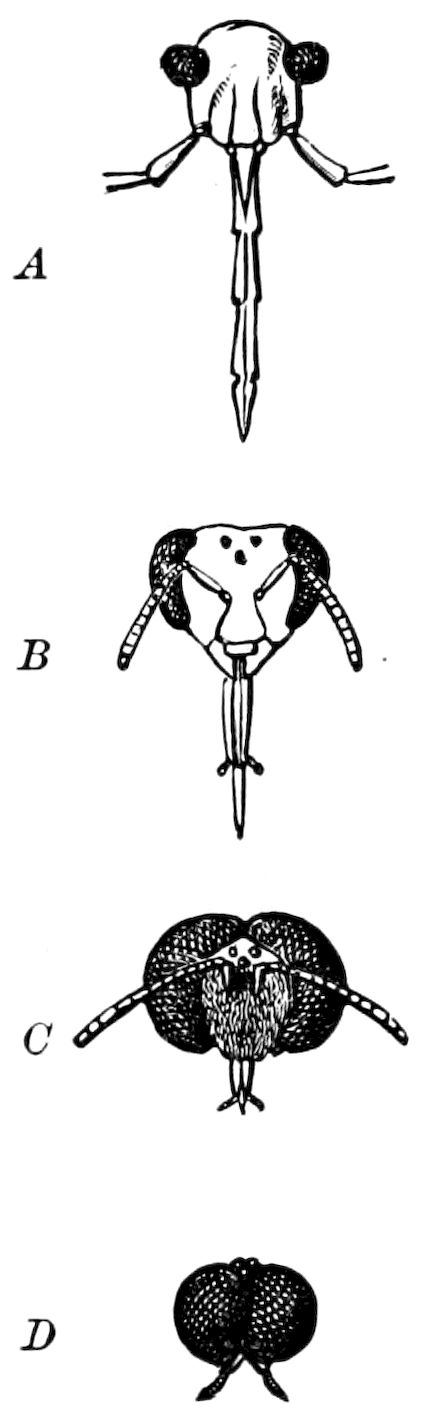
Fig. 259.—Different forms of compound eyes. A, a bug (Pyrrhocoris). B, worker bee. C, drone. D, male Bibio, a holoptic insect.—From Judeich and Nitsche.
Of the eyes of insects there are two kinds, the simple and the compound. Of the former there are usually three, arranged in a triangle near the top of the head, between the compound eyes (Fig. 259, B). The compound or facetted eyes, which are usually round and prominent, differ much in size and in the number of facets.
The number of facets varies from 12 in Lepisma,—though
in a Brazilian beetle (Lathridius) there are only
seven unequal facets,—to 50 in the ant, and up to 4000
in the house-fly, 12,000 in Acherontia atropos, 17,000 in
Papilio, 20,000 in the dragon-fly (Æschna), 25,000 in a
beetle (Mordella), while in Sphinx convolvuli, the number
reaches 27,000. The size of the facets seems to bear some
relation to that of the insect, but even in the smallest
species none have been observed less than 1
2000 of an inch
in diameter. Day-flying Lepidoptera have smaller facets
than moths (Lubbock).
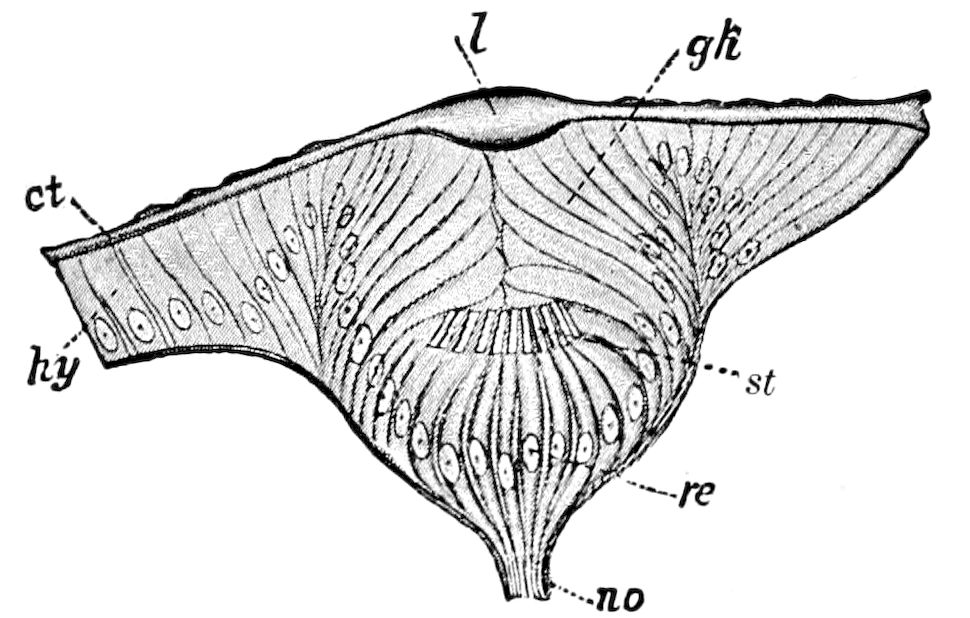
Fig. 260.—Section through the ocellus of a young Dyticus larva: ct, cuticula; l, corneal lens; gh, cells of the vitreous body, being modified hypodermal cells (hy); st, rods; re, retinal cells; no, optic nerve.—After Grenacher, from Lang.
The simple, or single-lensed eye (ocellus).—Morphologically the simple eye is a modified portion of the ectoderm, the pigment enclosing the retinal cells arising from specialized hypodermal cells, and covered by a specialized transparent portion of the cuticula, forming the corneal lens. The apparatus is supplied with a nerve, the fibres of which end in a rod or solid nerve-ending, as in other sensory organs.
As seen in the ocellus of Dyticus (Fig. 260), under the corneal lens the hypodermis forms a sort of pit, and the cells are modified 250to form the vitreous body (vitrella) and retina. Each retinal cell (re) is connected with a fibre from the optic nerve, contains pigment, and ends in a rod directed outwards towards the lens. The cells at the end of the pit or depression are, next to the lens, without pigment, and, growing in between the retina and the lens, fill it up, and thus form a sort of vitreous body.
The ocellus appears to be a direct heirloom from the eyes of worms, while the many-facetted compound eye of the crustaceans and of insects is peculiar to these classes. The compound eye of the myriopod Scutigera differs structurally in many respects from the compound eye of insects, and that of Limulus still more so.
It should be observed that in the young nymph of Ephemera, as well as in the semipupa of Bombus, each of the three ocelli are situated on separate sclerites. In Bombus the anterior ocellus has a double shape, being broad, transversely ovate, and not round like the two others, as if resulting from the fusion of what were originally two distinct ocelli.
The ocelli are not infrequently wanting, as in adult Dermaptera, in the Locustidæ, and in certain Hemiptera (Hydrocora). In Lepidoptera there are but two ocelli; in geometrid moths they are often atrophied, and they are absent in butterflies (except Pamphila).
The compound or facetted eye (ommateum).—The facetted arthropod eye is wonderfully complex and most delicately organized, being far more so than that of vertebrates or molluscs. The simplest or most primitive facetted eye appears to be that of Lepisma. As stated by Watase, the compound eye of arthropods is morphologically “a collection of ectodermic pits whose outer open ends face towards the sources of light, and whose inner ends are connected with the central nervous system by the optic nerve fibres.”
The facetted eye is composed of numerous simple eyes called ommatidia, each of which is complicated in structure. The elements which make up an ommatidium are the following: (1) The facet or cornea, which is a specialized portion of the cuticula; and (2), the crystalline lens or cone; (3), the nerve-ending or retinula, which is formed out of the retinula cells and the rhabdom or rod lying in its axis; and (4) of the pigment enclosing the lens and rod; the last three elements are derived from the hypodermis. The single eyes are separated from each other by pigment cells.
The facet or cornea.—This is biconvex, clear, transparent, usually hexagonal in outline, and refracts the light. The corneal lenses are cast in moulting.
The corneal lenses are circular in most cases where they are very convex, as in Lathridius and Batocera. The hexagonal ones are very irregular. When they are very convex the eye has a granular appearance, but when not greater than the convexity of the eye itself, the eye appears perfectly smooth (Bolbocerus, 251etc.). The facets in the lower part of the eye of Dineutes are a trifle larger than in the upper part (about nine to ten). In many insects the reverse is the case, the upper facets being larger than the lower, a notable instance being Anax. The intervening lines between the facets are often beset with hairs, sometimes very long and dense, as in the drone bee and Trichophthalmus; and the modifications of the hairs into scales which takes place on the body occurs on the eyes also, the scales on the eyes of some beetles of the family Colydiidæ being very large, arranged in lines over the eyes like tombstones (Trachypholis).[45]
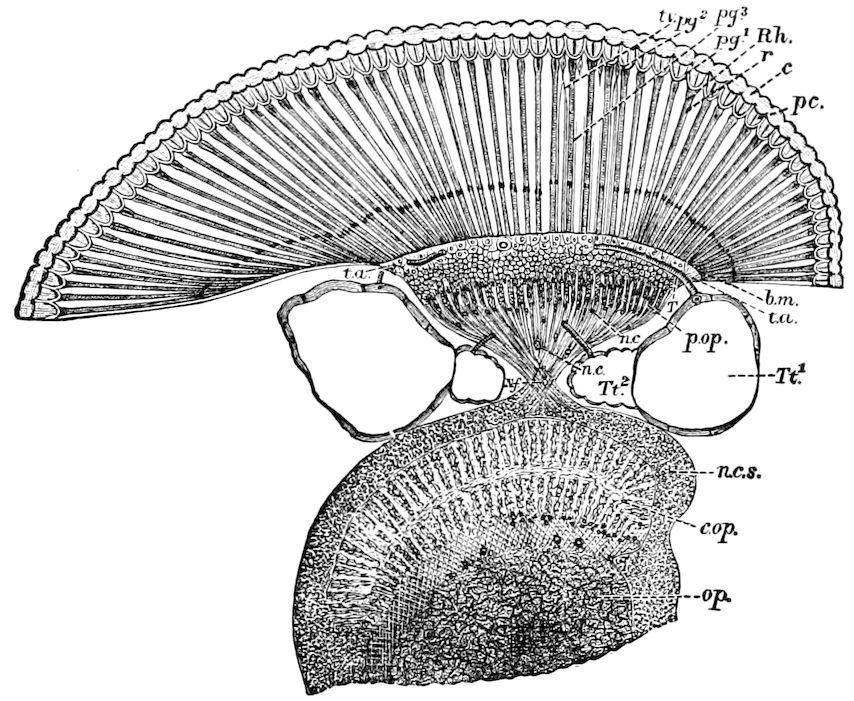
Fig. 261.—Section through the eye of a fly (Musca vomitoria): c, cornea, or facet; pc, pseudocone; r, retinula; Rh, rhabdom; pg1, pg2, pg3, pigment cells; b.m, basilar membrane; T, Tt1, Tt2, trachea; tv, tracheal vesicle; t.a, terminal anastomosis; op, opticon; c.op, epiopticon; p.op, periopticon; n.c, nuclei; n.c.s, nerve-cell sheath; N.f, decussating nerve-fibres.—After Hickson, from Lubbock.
The crystalline lens or cone.—Behind or within the facets is a layer composed of the cones, behind which are the layers of retinulæ and rhabdoms, and which correspond to the layer of rods and cones, but not the retina as a whole, of vertebrate animals.
The crystalline lens is, when present, usually more or less conical, and consists of four or more hypodermis-cells.
The cones are of various shapes and sizes in insects of different groups, or are entirely wanting, and Grenacher has divided the eyes of insects into eucone, pseudocone, and acone. As the pseudocone seems, however, to be rather a modification of the eucone eye, the following division may be made:—
1. Eucone eyes, comprising those with a well-developed cone. They occur in Lepisma, Blatta (Fig. 262), and other Orthoptera, in Neuroptera, in Cicadidæ, in those Coleoptera with five tarsal 252joints, in the dipterous genus Corethra, and in the Lepidoptera and Hymenoptera (Fig. 263).
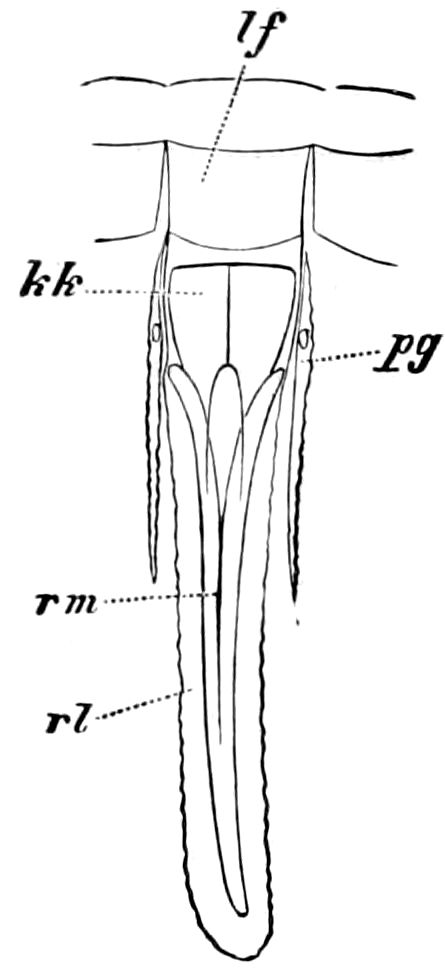
Fig. 262.—Ommatidium of cockroach (Periplaneta): lf, cornea; kk, crystalline cone; pg′ pigment cell; rl, retinula; rm, rhabdom.—After Grenacher, from Lubbock.
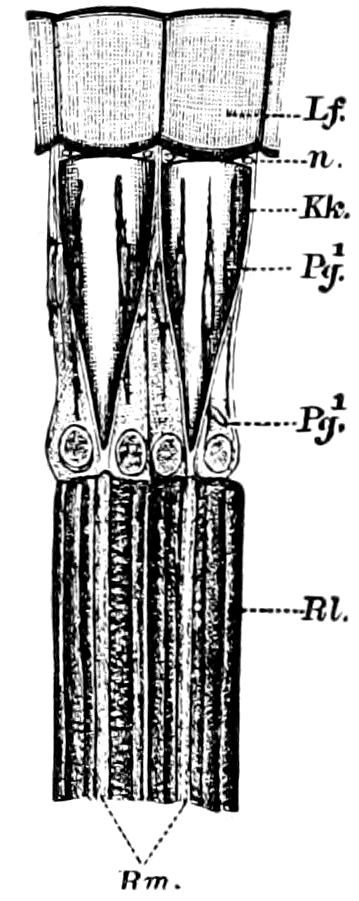
Fig. 263.—Two separate elements of the eucone eye of a bee; Lf, cornea; n, nucleus of Semper; Kk, crystalline cone; Pg, pigment cells; Rl, retinula; Rm, rhabdom.—After Grenacher, from Lubbock.
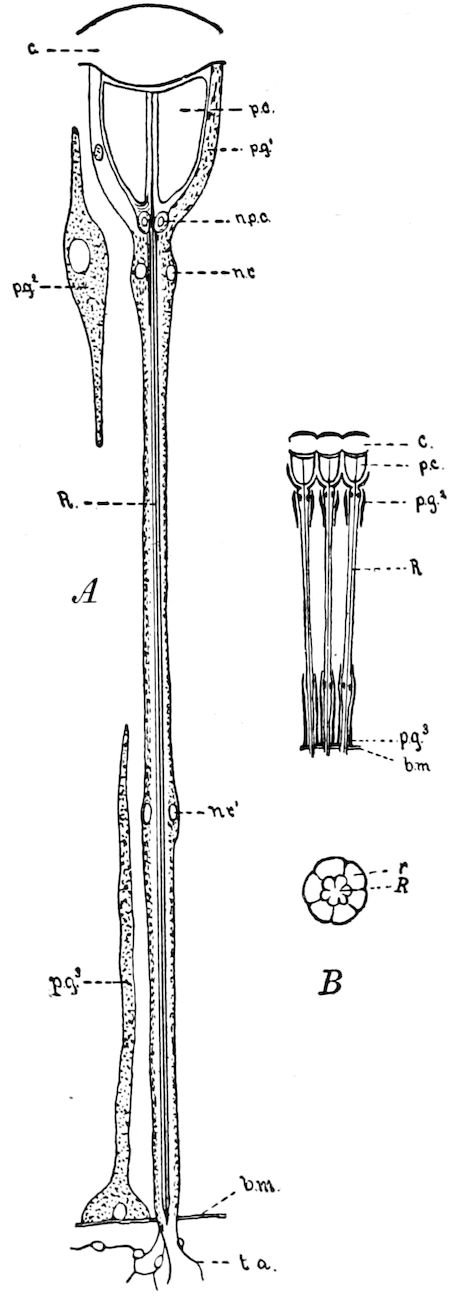
Fig. 264.—Three ommatidia of a pseudocone eye, diagrammatic: A, a separate ommatidium of Musca vomitoria, semi-diagrammatic: c, cornea; p.c, pseudocone; pg′, pigmented cells surrounding the pseudocone; p.g2, additional pigment cells; p.g3, basal pigment cells; n.p.c, nuclei of pseudocone; r, retinulæ; n.r, n.r′, nucleus of retinulæ; R, rhabdom; b.m, basal membrane; t.a, terminal anastomosis sending nerve-fibrils to the retinulæ. B, section through a retinula and rhabdom near the basal membrane, the six retinulæ (r) fused into a tube ensheathing the rhabdom (R).—After Hickson.
a. Pseudocone eyes; in which, instead of the crystalline lens or cone, there are four cells filled with a transparent fluid medium, and a smaller protoplasmic portion containing a nucleus (Muscidæ, Fig. 264, pc). Hickson states that the difference between the eucone and pseudocone eyes lies in the fact that in the pseudocone eye “the refracting body formed by the cone-cell lies behind the nuclei,” and in the eucone eye in front of it.
2. Acone eyes, where the cone or refracting body is wanting, but is represented by the four primitive cone-cells. Acone eyes occur in Forficulidæ, Hemiptera (except Cicadidæ), the nematocerous Diptera (Tipula, etc.), and those Coleoptera which have less than five tarsal joints.
253The retinula and rod.—The retinula is morphologically a nerve-end cell, situated at the end of a nerve-fibril arising from the optic nerve. The elements of the retinula of Musca are six in number and surround the rhabdom (Fig. 264), which consists of a bundle of six long, delicate chitinous rods, more or less firmly united together (Fig. 264, R).
The six elements of the retinula of Musca are in their outer or distal portion free from one another, but towards their base are fused into a sheath (Fig. 264, r). They are true nerve-end cells, as shown by Müller and by Max Schultze, their views having been confirmed by Grenacher and by Hickson. The relations of the nerves to the rods after passing through the basal membrane is seen in Fig. 266.
The pigment.—The cones or pseudocones are mostly buried in pigment, as well as the rods; and the pigment forms two layers. The outer of the two layers is called the iris pigment (Fig. 265, e, iris tapetum), and the inner (f) the retinal pigment.
Between the ommatidia internally there occur, according to Hickson, pigment cells (Fig. 264, p.g3), each of which stands on the basilar membrane and sends a fine process outwards towards the internal process of the external pigment-cell (p.g2). A long, slender tracheal vesicle also passes in between the retinulæ.
Fig. 265.—Two ommatidia from the eye of Colymbetes fuscus, × 160: a, cornea; b, cone; c, rhabdom; d, basal membrane, with nerve filaments below it: e, iris pigment; f, retina pigment.—After Exner, from Sharp.
The basilar membrane.—This is a thin fenestrate membrane (Fig. 261) separating the cones and rods from the optic tract (Fig. 264, b.m). It is perforated for the passage of tracheal diverticula and of the optic nerve fibrils. It separates the dioptric or instrumental portion of the eye from the percipient portion, i.e. the optic tract.
The optic tract.—This is the optic ganglion of earlier writers, and appears to be the percipient portion of the eye, as opposed to the dioptric portion. If the reader will examine Figs. 249 and 261, he will see that it consists of three distinct ganglionic swellings, i.e. the opticon, epiopticon, and periopticon, whose structure is very complicated. In Musca (Fig. 261) the first ganglionic swelling (opticon) is separated from the brain by a slight constriction, which Berger regards as the homologue of the optic nerve of the other arthropods. It consists of a very fine granular matrix traversed throughout by a fine meshwork 254of minute fibrillæ, the neurospongium of Hickson. In the young cockroach (Periplaneta) the optic nerve separating the cerebral ganglion from the opticon is much longer in proportion than it is in the adult blow-fly.
Fig. 266.—Periopticon and terminal anastomosis of Agrion, showing the character of the elements of the periopticon (p.op) and the structure of the terminal anastomosis (t.a). 1. The first layer of the terminal anastomosis, consisting of a plexus of fibrils and nerve-cells (n.c). 2. The second layer, in which the fibrils are collected together in bundles. 3. The final optic plexus and nerve-cells. 4. The layer in which the optic fibrils are collected in bundles to be distributed to the retinulæ (r); b.m, basal membrane.—After Hickson.
The second ganglionic swelling (epiopticon, Fig. 261, c.op) is separated from the opticon by a tract of fine nerve-fibrils, which partially decussate; at the decussation two or three larger nerve-cells may be seen. It also contains a few scattered nerve-cells (n.c). The third ganglionic swelling (periopticon, p.op) is separated from the others by a bundle of long optic nerve-fibrils, which cross one another. It is composed of a number of cylindrical masses of neurospongium arranged side by side (Fig. 261, p.op). Between these elements of the periopticon, which do not seem to bear any relation to the number of ommatidia, a single nerve-cell is very frequently seen. The periopticon does not occur in Periplaneta and Nepa (Hickson). The three optic ganglia thus described, together with the cerebral ganglia, are surrounded by a sheath of densely packed nerve-cells.
Bearing in mind the fact that the retinulæ are the nerve-end cells of the fibres passing through the periopticon, it will be well to read the following account, by Hickson, of the terminal anastomosis of the optic fibrils in the periopticon of Agrion bifurcatum, and to examine his sketch (Fig. 266):
“The terminal anastomosis of Agrion may be conveniently divided into four regions. First the region (1) lying nearest to the periopticon in which the nerve-cells are numerous, and the fibrils leaving the periopticon form a complicated plexus; the region (2) next to this, in which the fibrils have collected into bundles separated by spaces occupied by very thin-walled tracheæ in which there are no spiral markings, and lymph-spaces; next, the region (3) in which the fibrils form a final plexus, and in which there are again a considerable number of nerve-cells; and, lastly, the region (4) in which the fibrils are again collected into bundles, separated by spaces containing tracheæ, which perforate the basement membrane to supply the retinulæ.”
It would seem as if the decussation of the optic nerve-fibrils were a matter of 255primary importance, as it so generally occurs, but in the young of that most generalized of all pterygote insects, the cockroach (Periplaneta), Hickson states that the optic nerve-fibrils which leave the periopticon pass without decussating to the ommateum, and in the adult there is only a partial decussation. In Nepa there is no decussation, but the anastomosis is complicated by the presence of looped and transverse anastomoses.
Looking at the eye as a whole, Hickson regards all the nerve structure of the eye lying between the crystalline cone-layer and the true optic nerve to be analogous with the retina of other animals. With Ciaccio, Berger, and others, he does not regard the layer composed of the retinulæ and rhabdoms as the equivalent of the retina of vertebrates, etc.
Origin of the facetted eye.—The two kinds of eye, the simple and the compound, are supposed to have been derived from a primitive type, resembling the single eye (ommatidium) of the acone eye of Tipula. As stated by Lang, “an increase of the elements of this primitive eye led to the formation of the ocellus; an increase in number of the primitive eyes, and their approximation, led to the formation of the compound facet eye.” This view is suggested, he says, by the groups of closely contiguous single eyes of the myriopods, considered in connection with the compound eye of Scutigera. Grenacher looks upon simple (ocelli) and compound eyes as “sisters,” not derived from one another, but from a common parentage.
Immature insects rarely possess compound eyes; they are only known to occur in the nymphs of Odonata and Ephemeridæ, and in the larvæ and pupa of Corethra.
Mode of vision by single eyes or ocelli.—In their simplest condition, the eyes of worms and other of the lower invertebrates, probably only enable those animals to distinguish light from darkness. The ocelli of spiders and of many insects, however, probably enable them, as Lubbock remarks, to see as our eyes do. The simple lens throws on the retina an image, which is perceived by the fine terminations of the optic nerve. The ocelli of different arthropods differ, however, very much in degree of complexity.
Müller considered that the power of vision of ocelli “is probably confined to the perception of very near objects.”
“This may be inferred,” Müller states, “partly from their existing principally in larvæ and apterous insects, and partly from several observations which I have made relative to the position of these simple eyes. In the genus Empusa the head is so prolonged over the middle inferior eye that, in the locomotion of the animal, the nearest objects can only come within the range. In Locusta cornuta, also, the same eye lies beneath the prolongation of the head.... In 256the Orthoptera generally, also, the simple eyes are, in consequence of the depressed position of the head, directed downwards towards the surface upon which the insects are moving.”[46] Lowne considers that in the ocellus of Eristalis, the great convexity of the lens must give it a very short focus, and the comparatively small number of rods render the picture of even very near objects quite imperfect and practically useless for purposes of vision, and that the function of the ocelli is “the perception of the intensity and the direction of light, rather than of vision, in the ordinary acceptation of the term.”
Réaumur, Marcel de Serres, Dugès, and Forel have shown by experiment, that in insects which possess both ocelli and compound eyes, the former may be covered over without materially affecting the movements of the animals, while if the facetted eyes are covered, they act as if in the dark (Lubbock).
While Plateau regards the ocelli as of scarcely any use to the insect, and Forel claims that wasps, humble bees, ants, etc., walk or fly almost equally well without as with the aid of their ocelli, Lubbock demurs to this view, and says the same experiments of Forel’s might almost be quoted to prove the same with reference to the compound eyes. Indeed, the writer has observed that in caves, eyeless beetles apparently run about as freely and with as much purpose, as their eyed relatives in the open air.
Plateau has recently shown that caterpillars which have ocelli alone are very short-sighted, not seeing objects at a distance beyond one or two centimetres, and it has been fully proved by Plateau and others, that spiders, with their well-formed ocelli, are myopic, and have little power of making out distinctly the shape of the objects they see.
On the whole, we are rather inclined to agree with Lubbock and Forel, that the ocelli are useful in dark places and for near vision. They are, as Lubbock states, especially developed in insects, such as ants, bees, and wasps, which live partly in the open light and partly in the dark recesses of nests. Moreover, the night-flying moths nearly all possess ocelli, while with one known exception (Pamphila) they are wanting in butterflies.
Finally, remarks Lubbock, “Whatever the special function of ocelli may be, it seems clear that they must see in the same manner as our eyes do—that is to say, the image must be reversed. On the other hand, in the case of compound eyes, it seems probable that the vision is direct, and the difficulty of accounting for the existence in the same animal of two such different kinds of eyes is certainly enhanced by the fact that, as it would seem, the image given by the medial eyes is reversed, while that of the lateral ones is direct” (p. 181).
Mode of vision by facetted eyes.—The complexity of the facetted eyes of insects is amazing, and difficult to account for unless we accept the mosaic theory of Müller, who maintained that the distinctness of the image formed by such an eye will be greater in proportion to the number of separate cones. His famous theory is thus stated: “An image formed by several thousand separate points, of which each corresponds to a distinct field of vision in the external world, will resemble a piece of mosaic work, and a better idea cannot be conceived of the image of external objects which will be depicted on the retina of beings endowed with such organs of vision, than by comparing it with perfect work of that kind.”
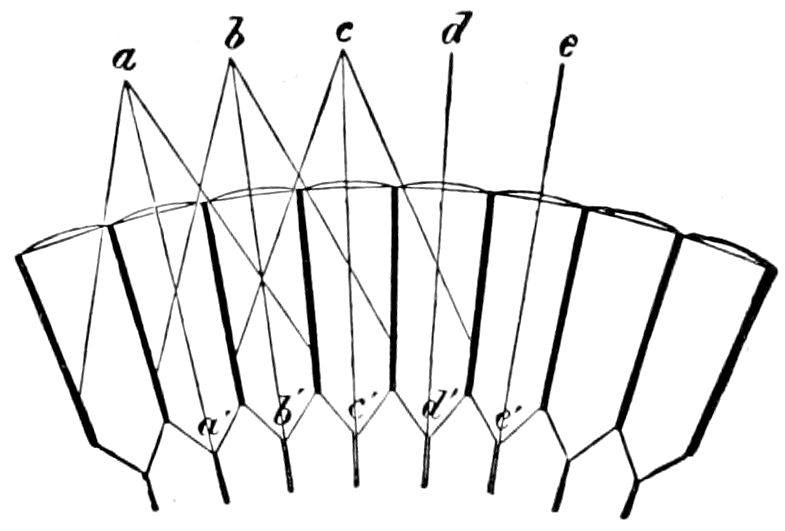
Fig. 267.—From Lubbock.
How vision is effected by a many-facetted eye is thus explained by Lubbock: “Let a number of transparent tubes, or cones with opaque walls, be ranged side by side in front of the retina, and separated from one another by black pigment. In this case the only light which can reach the optic nerve will be that which falls on any given tube in the direction of its axis.” For instance, in Fig. 267, the light from a will pass to a′, that from b to b′, that from c to c′, and so on. The light from c, which falls on the other tubes, will not reach the nerve, but will impinge on the sides and be absorbed by the pigment. Thus, though the light from c will illuminate the whole surface of the eye, it will only affect the nerve at c′.
According to this view those rays of light only which pass directly through the crystalline cones, or are reflected from their sides, can reach the corresponding nerve-fibres. The others fall on, and are absorbed by, the pigment which separates the different facets. Hence each cone receives light only from a very small portion of the field of vision, and the rays so received are collected into one spot of light.
It follows from this theory that the larger and more convex the eye, the wider will be its field of vision, while the smaller and more numerous are the facets, the more distinct will be the vision (Lubbock).
The theory is certainly supported by the shape and size and the immense number of facets of the eye of the dragon-fly, which all concede to see better, and at a longer range, than probably any other insect.
Müller’s mosaic theory was generally received, until doubted and criticised by Gottsche (1852), Dor (1861), Plateau, and others. As Lubbock in his excellent summary states, Gottsche’s observation (previously made by Leeuwenhoek) that each separate cornea gives a separate and distinct image, was made on the eye of the blow-fly, which does not possess a true crystalline cone. Plateau’s objection loses its force, since he seems to have had in his mind, as Lubbock states, Gottsche’s, rather than Müller’s, theory.
Müller’s theory is supported by Boll, Grenacher, Lubbock, Watase, and especially by Exner, who has given much attention to the subject of the vision of insects, and is the weightiest authority on the subject.
Gottsche’s view that each of the facetted eyes makes a distinct image which partially overlaps and is combined with all the images made by the other facets, 258was shown by Grenacher to be untenable, after repeating Gottsche’s experiments with the eyes of moths, in which the crystalline cones are firm and attached to the cornea. He was thus able to remove the soft parts, and to look through the cones and the cornea. When the microscope was focussed at the inner end of the cone, a spot of light was visible, but no image. As the object-glass was moved forward, the image gradually came into view, and then disappeared again. Here, then, the image is formed in the interior of the cone itself.
Exner attempted to make this experiment with the eye of Hydrophilus, but in that insect the crystalline cones always came away from the cornea. “He, however, calculated the focal length, refraction, etc., of the cornea, and concluded that, even if, in spite of the crystalline cone, an image could be formed, it would fall much behind the retinula.”
“In these cases, then,” adds Lubbock, “an image is out of the question. Moreover, as the cone tapers to a point, there would, in fact, be no room for an image, which must be received on an appropriate surface. In many insect eyes, indeed, as in those of the cockchafer, the crystalline cone is drawn out into a thread, which expands again before reaching the retinula. Such an arrangement seems fatal to any idea of an image.”
Lubbock thus sums up the reasons which seem to favor Müller’s theory of mosaic vision, and to oppose Gottsche’s view: “(1) In certain cases, as in Hyperia, there are no lenses, and consequently there can be no image; (2) the image would generally be destroyed by the crystalline cone; (3) in some cases it would seem that the image would be formed completely behind the eye, while in others, again, it would be too near the cornea; (4) a pointed retina seems incompatible with a clear image; (5) any true projection of an image would in certain species be precluded by the presence of impenetrable pigment, which only leaves a minute central passage for the light-rays; (6) even the clearest image would be useless, from the absence of a suitable receptive surface, since both the small number and mode of combination of the elements composing that surface seem to preclude it from receiving more than a single impression; (7) no system of accommodation has yet been discovered; finally (8), a combination of many thousand relatively complete eyes seems quite useless and incomprehensible.”
In his most recent work (1890) on the eyes of crustacea and insects, Exner states that the numerous simple eyes which make up the compound eye have each a cornea, but it is more or less flat, and the crystalline part of the eye has not the shape of a lens, but of a “lens cylinder,” that is, of a cylinder which is composed of sheets of transparent tissue, the refracting powers of which decrease toward the periphery of the cylinder. If an eye of this kind is removed and freed of the pigment which surrounds it, objects may be looked at through it from behind; but its field of vision is very small, and the direct images received from each separate eye are either produced close to one another on the retina (or rather the retinulæ of all the eyes) or superposed. In this last case no less than thirty separate images may be superposed, which is supposed to be of great use to night-flying insects. Exner claims that many other advantages result from the compound nature of an insect’s eye. Thus the mobile pigment, which corresponds to our iris, can take different positions, either between the separate eyes or behind the lens cylinders, in which case it acts as so many screens to intercept the over-abundance of light. Exner finds that with its compound eyes the common glow-worm (Lampyris) is capable of distinguishing large signboard letters at a distance of ten or more feet, as well as extremely fine lines engraved one-hundredth of an inch apart, if they are at a distance of less than half an inch from the eye. Exner substantiates the truth of the results of Plateau’s 259experiments, and claims that while the compound eye is inferior to the vertebrate eye for making out the forms of objects, it is superior to the latter in distinguishing the smallest movements of objects in the total field of vision.
More recently Mallock has given some optical reasons to show that Müller’s view is the true one. He concludes, and thus agrees with Plateau, that insects do not see well, at any rate as regards their power of defining distant objects, and their behavior certainly favors this view. It might be asked, What advantage, then, have insects with compound eyes over those with simple eyes? Mallock answers, that the advantage over simple-eyed animals lies in the fact that there is hardly any practical limit to the nearness of the objects they can examine. “With the composite eye, indeed, the closer the object the better the sight, for the greater will be the number of lenses employed to produce the impression; whereas, in the simple eye the focal length of the lens limits the distance at which a distinct view can be obtained.” He gives a table containing measures of the diameters and angles between the axes of the lenses of various insect eyes, and states that the best of the eyes would give a picture about as good as if executed in rather coarse woodwork and viewed at a distance of a foot, “and although a distant landscape could only be indifferently represented on such a coarse-grained structure, it would do very well for things near enough to occupy a considerable part of the field of view.”
The principal use of the facetted eye to perceive the movements of animals.—Plateau adopts Exner’s views as to the use of the facetted eye in perceiving the movements of other animals. He therefore concludes that insects and other arthropods with compound eyes do not distinguish the form of objects; but with Exner he believes that their vision consists mainly in the perception of moving bodies.
Most animals seem but little impressed by the form of their enemies or of their victims, though their attention is immediately excited by the slightest displacement. Hunters, fishermen, and entomologists have made in confirmation of this view numerous and demonstrative observations.
Though the production of an image in the facetted eye of the insect seems impossible, we can easily conceive, says Plateau, how it can ascertain the existence of a movement. Indeed, if a luminous object is placed before a compound eye, it will illuminate a whole group of simple eyes or facets; moreover, the centre of this group will be clearer than the rest. Every movement of the luminous body will displace the centre of clearness; some of the facets not illuminated will first receive the light, and others will reënter into the shade; some nervous terminations will be excited anew, while those which were so formerly will cease to be. Hence the facetted eyes are not complete visual organs, but mainly organs of orientation.
Plateau experimented in the following way: In a darkened room, with two differently shaped but nearly equal light-openings, one square and open, the other subdivided into a number of small holes, and therefore of more difficult egress, he observed the choices of opening made by insects flying from the other end of the room. Careful practical provisions were made to eliminate error; the light-intensity of the two openings was as far as possible equalized or else noted, and no trees or other external objects were in view. The room was not darkened beyond the limit at which ordinary type ceases to be readable, otherwise the insects refused to fly (it is well known that during the passage of a thick cloud insects usually cease to fly). These observations were made on insects 260both with or without ocelli, in addition to the compound eyes, and with the same results.
From repeated experiments on flies, bees, etc., butterflies and moths, dragon-flies and beetles, Plateau concludes that insects with compound eyes do not notice differences in form of openings in a half-darkened room, but fly with equal readiness to the apparently easy and apparently difficult way of escape; that they are attracted to the more intensely lighted opening, or to one with apparently greater surface; hence he concludes that they cannot distinguish the form of objects, at least only to a very slight extent, though they readily perceive objects in motion.
One result of his experiments is that insects only utilize their eyes to choose between a white luminous orifice in a dark chamber, or another orifice, or group of orifices, equally white. They are guided neither by odorous emanations nor by differences of color. He thinks that bees have as bad sight and act almost exactly as flies.
From numerous experiments on Odonata, Coleoptera, Lepidoptera, Diptera, and Hymenoptera Plateau arrives provisionally at the following conclusions:
1. Diurnal insects have need of a quick strong light, and cannot direct their movements in partial obscurity.
2. Insects with compound eyes do not notice differences of form existing between two light orifices, and are deceived by an excess of luminous intensity as well as by the apparent excess of surface. In short, they do not distinguish the form of objects, or if they do, distinguish them very badly.
Lubbock, however, does not fully accept Plateau’s experiments with the windows, and thinks they discern the form of bodies better than Plateau supposes.
How far can insects see?—It is now supposed that no insects can perceive objects at a greater distance than about six feet. On an average Lepidoptera can see the movements of rather large bodies 1.50 meters, but Hymenoptera only 58 cm., and Diptera 68 cm.; while the firefly (Lampyris) can see tolerably well the form of large objects at a distance of over two meters.
Until further experiments are made, it seems probable, then, that few if any insects have acute sight, that they see objects best when moving, and on the whole—except dragon-flies and other predaceous, swiftly flying insects, such as certain flies, wasps, and bees, which have very large rounded eyes—insects are guided mainly rather by the sense of smell than of sight.
Relation of sight to the color of eyes.—It appears from the observations of Girschner that those Diptera with eyes of a uniform color see better than those with brightly banded or spotted eyes. Thus those flies (Asilidæ, Empidæ, Leptidæ, Dolichopidæ) whose predaceous habits requires good or quick sight have uniformly dark eyes, as have also such flies as live constantly on the wing, i.e., the holoptic Bombyliidæ, Syrphidæ, Pipunculidæ, etc., whose eyes are also very large.
Those flies whose larvæ are parasitic on other animals have eyes of a uniform color that they may readily detect the most suitable host for their young; such are the Bombyliidæ, Conopidæ, Pipunculidæ, and Tachinidæ.
Certain flies which live in the clear sunlight, as many Dolichopidæ, some Bombyliidæ, and certain Tabanidæ (Tabanus, Chrysops, Hæmatopota), and which are often easily caught with the hand, have eyes spotted or banded with bright or metallic colors. This is also a sexual trait, as the males of some horse-flies visiting flowers have eyes of a single color, the spots and bands surviving only on the lower and hinder parts of the eye, while their voracious blood-sucking females have the entire eye spotted or banded (Kolbe).
The color-sense of insects.—Insects, as Spengel first suggested, appear to be 261able to distinguish the color of objects. Lubbock has experimentally proved that bees, wasps, and ants have this power, blue being the favorite color of the honey-bee, and violet of ants, which are sensitive to ultra-violet rays.
It is well known that butterflies will descend from a position high in the air, mistaking white bits of paper for white flowers; while, as we have observed, white butterflies (Pieris) prefer white flowers, and yellow butterflies (Colias) appear to alight on yellow flowers in preference to white ones.
The late Mr. S. L. Elliott once informed us that on a red barn with white trimmings he observed that white moths (Spilosoma, Hyphantria, and Acronycta oblinita) rested on the white parts, while on the darker, reddish portions sat Catocalæ and other dark or reddish moths. Gross observed that house-flies would frequent a bluish green ring on the ceiling of his chamber; but if it were covered by white paper, the flies would leave the spot, though they would return as soon as the paper ring was removed (Kolbe). We have observed that house-flies prefer green paper to the yellowish wall of a kitchen, but were not attracted to sheets of a Prussian blue paper, attached to the same wall and ceiling.
It is generally supposed that the shape and high colors of flowers attract insects; but Plateau has made a number of ingenious experiments which tend to disprove this view. He used in his investigations the dahlia, with its central head of flowerets, which contrast so strongly with the corolla. He finds (1) that insects frequent flowers which have not undergone any mutilation, but whose form and colors are hidden by green leaves. (2) Neither the shape nor lively colors of the central head (capitulum) seem to attract them. (3) The gayly colored peripheral flowerets of simple dahlias and, consequently, of the heads of other composite flowers, do not play the rôle of signals, such as has been attributed to them. (4) The insects are evidently guided by another sense than that of sight, and this sense is probably that of smell.
LITERATURE ON THE EYES AND VISION
a. General
Serres, Marcel de. Mémoires sur les yeux composés et les yeux lisses des insectes. Montpellier, 1813.
Müller, Johannes. Zur vergleichenden Physiologie des Gesichtssinnes der Menschen und der Tiere. 8 Taf. Leipzig, 1826.
—— Ueber die Augen des Maikäfers. (Meckel’s Archiv f. Anat. u. Phys., 1829, pp. 177–181; Ann. d. Sc. nat., 1829, sér. 1, xviii, pp. 108–112.)
Dujardin, F. Sur les yeux simples ou stemmates des animaux articulés. (C. R. Acad. Sci., Paris, 1847, xxv, pp. 711–714.)
Gottsche, C. M. Beitrag zur Anatomie und Physiologie des Auges der Krebse und Fliegen. (Müller’s Archiv für Anat. u. Phys., 1852, pp. 483–492. Figs.)
Murray, Andrew. On insect vision and blind insects. (Edinburgh New Phil. Jour., new ser. vi, 1857, pp. 120–138.)
Claparède, Édouard. Zur Morphologie der zusammengesetzten Augen bei den Arthropoden. (Zeitschr. f. wissensch. Zool., 1859, x, pp. 191–214, 3 Taf.)
Dor, H. De la vision chez les Arthropodes. (Archives Sci. Phys, et Nat., 1861, xii, p. 22, 1 Pl.)
Landois, H. Die Raupenaugen (Ocelli compositi mihi). (Zeitschr. f. wissensch. Zool., xvi, 1866, pp. 27–44, 1 Taf.)
—— und W. Thelen. Zur Entwicklungsgeschichte der fasettierten Augen von Tenebrio molitor L. (Zeitschr. f. wissensch. Zool., xvii, 1867, pp. 34–43, 1 Taf.)
262Schultze, Max. Untersuchungen über die zusammengesetzten Augen der Krebsen und Insecten. Bonn, 1868.
Schmidt, Oscar. Die Form der Krystallkegel in Arthropodenauge. (Zeitschr. f. wissensch. Zool., xxx, Suppl., 1878, pp. 1–12, 1 Taf.)
Grenacher, H. Untersuchungen ueber das Sehorgan der Arthropoden, insbesondere Spinnen, Insecten und Crustaceen. (Göttingen, 1879, 4º, pp. 1–188, 11 Taf.)
Reichenbach, H. Wie die Insekten sehen. Fig. (Daheim, xvi Jahrg., 1880, pp. 284–286.)
Poletajew, N. Ueber die Ozellen und ihr Sehvermögen bei den Phryganiden. (Horæ Soc. Ent. Ross., 1884, xviii, p. 23, 1 Taf. In Russian.)
Hickson, S. J. The eye and optic tract of insects. (Quart. Journ. Micr. Sc., ser. 2, xxv, 1885, pp. 215–221, 3 Pls.)
Notthaft, Jul. Ueber die Gesichtswahrnehmungen vermittelst des Fazettenauges. (Abhandl. Senckenberg. naturf. Ges., xii., 1880, pp. 35–124, 5 Taf.)
—— Die physiologische Bedeutung des fazettierten Insektenauges. (Kosmos, 1886, xviii, pp. 442–450, Fig.)
Mark, E. L. Simple eyes in arthropods. (Bull. Mus. Comp. Zool., 1887, xiii, pp. 49–105, 5 Pls.)
Girschner, E. Einiges über die Färbung der Dipterenaugen. (Berlin. Ent. Zeitschr., 1888, xxxi, pp. 155–162, 1 Taf.)
Graber, V. Das unicorneale Tracheatenauge. (Archiv f. Mikroskop. Anat., xvii, 1879, pp. 58–93, 3 Taf.; Nachtrag, p. 94.)
—— Fundamentalversuche über die Helligkeits- und Farbenempfindlichkeit augenloser und geblendeter Tiere. (Sitzgs.-Ber. Akad. Wissensch., Wien, 1883, lxxxvii, pp. 201–236.)
Dahl, Fr. Die Insekten können Formen unterscheiden. (Zool. Anz., xii, 1889, pp. 243–247.)
Ciaccio, G. V. Figure dichiarative della minuta fabbrica degli occhi de’ Ditteri. Bologna, 1884, 12 Taf., 30 pp.
—— Della minuta fabbrica degli occhi de’ Ditteri. (Mem. Accad. Bologna, 1886, ser. 4, vi, pp. 605–660.)
—— Sur la forme et la structure des facettes de la cornée et sur les milieux refringents des yeux composés des Muscidés. (Journ. Micr., Paris, 1889, xiii Année, pp. 80–84.)
Carrière, J. On the eyes of some invertebrata. (Quart. Journ. Micr. Sc. 1884, ser. 2, xxiv, pp. 673–681, 1 Pl.)
—— Ueber die Arbeiten von Viallanes, Ciaccio und Hickson. (Biolog. Centralblatt, v, 1885, pp. 589–597.)
—— Die Sehorgane der Tiere vergleichend anatomisch dargestellt. München u. Leipzig, 1885, 205 pp., 147 Figs., 1 Taf.
—— Kurze Mitteilungen aus fortgesetzten Untersuchungen über die Sehorgane. (Zool. Anz., ix Jarhg., 1886, pp. 141–147, 479–481, 496–500.)
Forel, A. Les fourmis de la Suisse. (Neue Denkschriften der schweiz. naturforsch. Gesellsch. xxvi. 1874, pp. 480, 2 Pls.) Separate. pp. iv u. 457. Genève.
—— Beitrag zur Kenntnis der Sinnesempfindungen der Insekten. (Mitteil. d. Münchener Ent. Vereins, ii Jahrg., 1878, pp. 1–21.)
—— Sensations des insectes. (Recueil Zool. Suisse, iv, 1886 et 1887.)
Plateau, F. L’instinct chez les insectes mis en défaut par les fleurs artificielles? (Assoc. française avancement des sciences. Congrès de Clermont. Ferrand, 1876.)
263Plateau, F. Recherches expérimentales sur la vision chez les insectes. Les insectes distinguent-ils la forme des objets? (Bull. Acad. Belg. 3 Sér. x, 1885, pp. 231–250.)
—— Recherches expérimentales sur la vision chez les insectes.
1. Part, a. Résumé des travaux effectués jusqu’en 1887 sur la structure et le fonctionnement des yeux simples. b. Vision chez les Myriapodes. (Ibid. Sér. 3, xiv, 1887, pp. 407–448, 1 Pl.)
3. Part, a. Vision chez les chenilles, b. Rôle des ocelles frontaux chez les insectes parfaits. (Ibid. Sér. 3, xv, 1888, pp. 28–91.)
4. Part. Vision à l’aide des yeux composés. a. Résumé anatomo-physiologique. b. Expériences comparatives sur les insectes et sur les vertébrés. (Mém. cour. et autres Mém. Acad. Belg. 1888, xliii, pp. 1–91, 2 Pls.)
5. Part, a. Perception des mouvements chez les insectes. b. Addition aux recherches sur le vol des insectes avenglés. c. Résumé général. (Bull. Acad. Belg. 1888, sér. 3, xvi, pp. 395–457, 1 Pl.)
—— Recherches expérimentales sur la vision chez les Arthropodes, 2 Pls. (Mém. couronn. et autres Mém. publ. p. l’Acad. Roy. d. Sciences, etc., de Belgique, xliii, Bruxelles, 1889.)
Watase, S. On the morphology of the compound eyes in the Arthropoda. (Studies from biol. laborat. Johns-Hopkins Univ., 1890, pp. 287–334, 4 Pls.)
Stefanowska, M. La disposition histologique du pigment dans les yeux des Arthropodes. (Recueil Zool. Suisse, 1890, pp. 151–200, 2 Pls.)
Pankrath, O. Das Auge der Raupen und Phryganiden larven. (Zeitschr. f. wissensch. Zool., 1890, xlix, pp. 690–708, 2 Taf.)
Lowne, B. Th. On the modifications of the simple and compound eyes of insects. (Philos. Trans. Roy. Soc., London, clxix, 1878, pp. 577–602, 3 Pls.)
—— On the structure and functions of the eyes of Arthropoda. (Proc. Roy. Soc., London, 1883, xxxv, pp. 140–145.)
—— On the compound vision and the morphology of the eye in insects. (Trans. Linn. Soc., London, 1884, ii, pp. 389–420, 4 Pls.)
—— On the structure of the retina of the blow-fly (Calliphora erythrocephala). (Jour. Linn. Soc., London, 1890, xx, pp. 406–417, 1 Pl.)
Patten, W. Eyes of molluscs and arthropods. (Journal of Morphol., Boston, 1887, i, pp. 67–92, 1 Pl.; Mitteil. Zool. Stat. Neapel, vi, 1886, pp. 542–756, 5 Taf.)
—— Studies on the eyes of arthropods.—1. Development of the eyes of Vespa, with observations on the ocelli of some insects. (Ibid., pp. 193–226, 1 Pl.)—2. Eyes of Acilius. (Ibid., 1888, ii., pp. 190–97, 7 Pls.)
—— On the eyes of molluscs and arthropods. (Zool. Anzeiger, 1887, x Jahrg., pp. 256–261.)
—— Is the ommatidium a hair-bearing sense-bud? (Anatom. Anzeiger, 1890, v, pp. 353–359, 4 Figs.)
Exner, S. Ueber das Sehen von Bewegungen und die Theorie des zusammengesetzten Auges. (Sitzgsber. d. math. naturwiss. Cl. kais. Akad. d. Wissens. Wien, lxxii Jahrg., 1875, 3 Abt. Physiologie, pp. 156–190, 1 Taf.)
—— Die Frage von der Funktionsweise der Fazettenauges. (Biolog. Centralblatt, i, 1881, pp. 272–281.)
—— Das Netzhautbild des Insektenauges. (Sitzgsber. kais. Akad. d. Wissensch. Wien, 1889, xcviii, 3 Abt., pp. 13–65, 2 Taf. u. 7 Figs.)
—— Durch Licht bedingte Verschiebungen des Pigmentes im Insektenauge und deren physiologische Bedeutung. (Ibid., pp. 143–151, 1 Taf.)
264Exner, S. Die Physiologie der fazettierten Augen von Krebsen und Insekten, 7 Taf., 1, Lichtdruck u. 23 Holzschn. pp. 206. Wien, F. Deuticke, 1891.
Lubbock, John. On the senses, instincts, and intelligence of animals, with special reference to insects. London, 1888, pp. 292.
Mallock, A. Insect sight and the defining power of composite eyes. (Proc. Roy. Soc., London, 1894, lv, pp. 85–90, 3 Figs.)
b. The color-sense
Nussli, J. Ueber den Farbensinn der Bienen. (Schweiz. Bienenzeitung, N. F., ii Jahrg., 1879, pp. 238–240.)
Kramer. Der Farbensinn der Bienen. (Ibid., iii Jahrg., 1880, pp. 179–198.)
Gross, Wilhelm. Ueber den Farbensinn der Tiere, insbesondere der Insekten. (Isis v. Russ., v Jahrg., 1880, pp. 292–294, 300–302, 308–309.)
Lubbock, John. Ants, bees, and wasps. London, 1882, pp. 448. Also On the senses, etc., of animals, 1889.
Graber, Vitus. Grundlinien zur Erforschung des Helligkeits und Farbensinnes der Tiere. Prag u. Leipzig, 1884, pp. 322. (See also p. 262.)
Forel, Auguste. Les Fourmis perçoisent-elles l’ultra-violet avec leurs yeux ou avec leur peau? (Arch. Sci. Phys. Nat. Genève, 1886, 3 sér., xvi, pp. 346–350.)
Also the works of Darwin, Wallace, F. Müller, Grant Allen’s The Color Sense (1879), Beddard’s Animal Coloration, etc.
b. The organs of smell
The seat of the organs of smell is mainly in the antennæ, and they may be regarded as the principal olfactory organs. For our present knowledge of the anatomy and physiology of the olfactory organs of insects we are mainly indebted to the recent investigations of Hauser and of Kraepelin. The following historical and critical remarks are translated from Kraepelin’s able treatise:
Historical sketch of our knowledge of the organs of smell.—In the first half of the last century began the inquiries as to the seat of the sense of smell in the arthropods. Thus Réaumur, in his Mémoires (i, p. 283; ii, 224), expressed the view that in the antennæ was situated a special organ which might be an organ of smell.
Lesser, Roesel, Lyonet, Bonnet, and others expressed the same opinion. Before this Sulzer suggested that an “unknown sense” might exist in the antennæ; others regarded the stigmata as organs of smell, as these were considered the natural passages for the olfactory currents. Duméril, in two special treatises as well as in his Considérations générales, sought to prove the theory as to the seat of the organs of smell in the stigmata.
Against both of these leading views as to the seat of the sense of smell were expressed, in the last century, different opinions. Thus Comparetti thought that the sense of smell might be localized in very different points of the head, in the antennal club of lamellicorns, in the sucking-tube of Lepidoptera, in 265special frontal holes of flies and Orthoptera, etc., while Bonsdorf considered the palpi as organs of smell.
Thus four different views, confused, were held at the opening of this century; the Hamburg zoölogist, M. C. S. Lehrman, in three different treatises, brought together all the hitherto known observations and arguments, treated them critically, and completed them by his own extended studies. Lehrman adopted the opinions of Reimarus, Baster, Duméril, and Schelver, that the stigmata presented the most convenient place for the site of the organs of smell. Cuvier followed throughout the lead of Lehrman, but Latreille returned to the view of the perception of smell by the antennæ, while Treviranus considered the mouth of arthropods as the probable site of the sense of smell, an opinion which, before his time, Huber, in his experiments on bees, had thought to be correct. Marcel de Serres (1811) returned again to the palpi, and asserted—at least in the Orthoptera—their functions to be olfactory, while Blainville, ten years later, again expressed anew the old opinion that the antennæ, or at least their terminations, were organs of smell. Up to that date there was an uncertainty as to the seat of the organs both of smell and hearing. Fabricius, indeed, had already, in 1783, thought he had found an organ of hearing at the base of the outer antenna. In 1826 J. Müller mentioned an already well-known organ in the abdomen of crickets as an organ of hearing. Müller, however, was doubtful, from the fact that the nerve passing to this organ arose, not from the brain, but from the third thoracic ganglion; but, notwithstanding, he remarks: “Perhaps we have not found the organ of hearing in insects because we sought for it in the head.” This discovery was afterwards considerably broadened and extended by Siebold’s work, for the views of these naturalists on the seat of both organs had a definite influence, especially in Germany. For awhile, indeed, Müller’s hypothesis stood in complete contradiction, so that during the following decennial was presented anew the picture of opposing observations and opinions as to the nature of the organs of smell. While Robineau-Desvoidy, at the end of the twentieth year, and also later, in different writings, strove energetically for the olfactory nature of the antennæ, Straus-Dürckheim held fast to the view that the tracheæ possessed the function under discussion. At the same period Kirby and Spence, in their valuable Introduction to Entomology, maintained that “two white cushions on the under side of the upper lip” in the mouth of biting insects formed a nose or “rhinarium” peculiar to insects. This opinion was afterwards adopted by Lacordaire (Introduction à Entomologie), and also by Oken in his Lehrbuch der Naturphilosophie, while Burmeister, rejecting all the views previously held, believed that insects might perhaps smell “with the inner upper surface of the skin.” Müller’s locust’s ear he regarded as a vocal organ.
Besides these occasional expressions of opinion, the French literature of the thirtieth and fortieth years of this century recorded a long series of special works, with weighty experimental and physiological contents, on this subject. Thus Lefebre, in 1838, described the experiments which he made on bees, and which seemed to assign the seat of the sense of smell to the antennæ. Dugès reported similar researches on the Scolopendræ, and Pierret thought that the great development of the antennæ in the male Bombycidæ might be similarly interpreted. Driesch sought to give currency to the views of Bonsdorf, Lamarck, and Marcel de Serres, that the sense of smell was localized in the palpi, though Duponchel went back to the old assertion of æroscepsis of Lehrman, i.e. of the air-test through the antennæ, and Goureau again referred the seat of the sense of smell to the mouth. In England, Newport at this period put forth a work in which he considered the antennæ as organs of touch and hearing, and the palpi 266as organs of smell—a view which, as regards the antennæ, was opposed by Newman.
Thus the contention as to the use of the antennæ and the seat of the organs of smell and hearing fluctuated from one side to the other, and when in 1844 Küster, by reason of his experiments on numerous insects, again claimed that “the antennæ are the smelling organs of insects,” he argued on a scientific basis; yet v. Siebold and Stannius (1848), in their valuable Lehrbuch der vergleichenden Anatomie (p. 581), remarked that “organs of smell have not yet with certainty been discovered in these animals.”
The following decennial was of marked importance in the judgment of many disputed questions. Almost contemporaneously with Siebold and Stannius’ Lehrbuch appeared an opportune treatise by Erichson, in which this naturalist first brought forward certain anatomical data as to the structure of the antennæ of insects. In a great number of insects Erichson described on the upper surface of the antennæ peculiar minute pits, “pori,” which, according to him, were covered by a thin membrane, and to which he ascribed the perception of smell. A still more thorough work on this subject was published in the following year by Burmeister, who recognized in the pits of lamellicorns many small tubercles and hairs; and about the same time Slater, as also Pierret and Erichson before him had done, out of the differences of the antennal development in the males and females in flesh and plant-eating insects, brought together the proof of the olfactory function of the antennæ. But the most valuable work of this period is that of Perris, who, after a review of previous opinions, by exact observations and experiments, a model of their kind, sought to discover the seat of the sense of smell. He comes to the conclusion that the antennæ, and perhaps also the palpi, may claim this sense, and finds full confirmation of Dufour’s views, and adopts as new the physiological possibility expressed by Hill and Bonnet, that the antennæ might be the seat of both senses—those of smell and hearing.
The beautiful works of Erichson, Burmeister, and Perris could not remain long unnoticed. In 1857 Hicks published complete researches on the peculiar nerve-endings which he had found in the antennæ, also in the halteres of flies and the wings of all the other groups of insects, and which he judged to be for the perception of smell. But Erichson’s and Burmeister’s “pori” were by Lespès, in 1858, explained to be so many auditory vesicles with otoliths. This view was refuted by Claparède and Claus without their deciding on any definite sense. Leydig first made a decided step in advance. In different writings this naturalist had busied himself with the integumental structures of arthropods, and declared Erichson’s view as to the olfactory nature of the antennal pits as the truest, before he, in his careful work on the olfactory and auditory organs of crabs and insects, had given excellent representations of the numerous anatomical details which he had selected from his extensive researches in all groups of arthropods. Besides the pits which were found to exist in Crustacea, Scolopendræ, beetles, Hymenoptera, Diptera, Orthoptera, Neuroptera, and Hemiptera, and which had only thus far been regarded as sense-organs, Leydig first calls attention to the widely distributed pegs and teeth, also considering them as sense-organs. “Olfactory teeth,” occurring as pale rods, perforated at the end, on the surface of the antennæ of Crustacea, Myriopoda, Hymenoptera, Lepidoptera, Coleoptera, are easily distinguished, and besides the “olfactory pegs” of the palpi, may be claimed as organs of smell. The nerve-end apparatus first discovered by Hicks in the halteres and wings, Leydig thinks should be ranked as organs of hearing.
There was still some opposition to Leydig’s opinion that in the insects the 267sense of smell is localized in the antennæ (teeth and pits), and here the work of Hensen might be mentioned, which in 1860 had a decided influence upon the conclusion of some inquiries.
Thus Landois denied that the antennæ had the sense of smell, and declared that the pits in the antennæ of the stag beetle were auditory organs. So, also, Paasch rejected Leydig’s conclusion, while he sought to again reinstate the old opinion of Rosenthal as to the olfactory nature of the frontal cavity of the Diptera. In spite of the exact observations and interesting anatomical discoveries of Forel in ants, made in 1874, there appeared the great work of Wolff on the olfactory organs of bees, in which this observer, with much skill and acuteness, sought to give a basis for the hypothesis of Kirby and Spence that the seat of the sense of smell lay in the soft palatine skin of the labrum within the mouth (i.e. the epipharynx). Joseph, two years later, drew attention to the stigmata as olfactory organs, referring to the olfactory girdle, and Forel sought by an occasional criticism of Wolff’s conclusions to prove experimentally the olfactory function of the antenna; but Graber, in his widely read book on insects, defended the Wolffian “nose” in the most determined way, and denied to the antennæ their so often indicated faculty of smell. In 1879 Berté thought he had observed in the antenna of the flea a distinct auditory organ, and Lubbock considered the organs of Forel in the antennæ of ants as a “microscopic stethoscope.” In 1879 Graber described a new otocyst-like sense-organ in the antennæ of flies, which was accompanied by a complete list of all the conceivable forms of auditory organs in arthropods. In this work Graber described in Musca and other Diptera closed otocysts with otoliths and auditory hairs, as Lespès had previously done. But Paul Mayer, in two essays, refuted this view in a criticism of the opinion of Berté, referring the “otocysts with otoliths” to the well-known antennal pits into which tracheæ might pass. Mayer did not decide on the function of the hairs which extend to the bottom of the pits; while in the most recent research, that of Hauser, the author again energetically contended for the olfactory function of the antennæ. Both through physiological experiments and detailed anatomical investigations Hauser sought to prove his hypothesis, as Pierrot, Erichson, Slater, and others had done before him, besides working from an evolutional point of view. In a purely anatomical aspect, especially prominent are his discovery of the singularly formed nerve-rods in the pits and peg-like teeth of the Hymenoptera and their development, as well as the assertion that numerous hairs in the pits described by Leydig, Meyer, etc., should be considered as direct terminations of nervous fibres passing into the pits. In the pits he farther, with Erichson, notices a serous fluid, which may serve as a medium for the perception of smells. Among the latest articles on this subject are those of Künckel and Gazagnaire, which are entirely anatomical, while the latest treatise of Graber on the organs of hearing in insects opposes Hicks’s theory of the olfactory function of the nerve-end apparatus in the halteres, wings, etc., and argues for the auditory nature of these structures. Finally, according to Voges, the sense of smell is not localized, but spread over the whole body.
My own observations on different groups of insects agree, in general, with those of Perris, Forel, and Hauser, without being in a position to confirm or deny the varying relations of the Hemiptera. That irritating odorous substances (chloroform, acetic acid) cause the limbs to move in sympathy with the stimulus, I have seen several times in Acanthosoma; still it may be a gustatory rather than olfactory stimulus.
Turning now from speculation and simple observation to exact anatomical and histological data, the nerve-end apparatus seems to have a distinct reference to 268the perception of odors. It comprises a structure composed of nervous substances which are enclosed in a chitinous tube, and either only stand in relation to the surrounding bodies by the perforated point, or pass to the surface as free nerve-fibrillæ.
In insects there is a remarkable and fundamental difference in the structures of the parts supposed to be the organs of smell. Erichson was acquainted only with the “pori” covered by a thin membrane; but Burmeister, in his careful work on the antennæ of the lamellicorns, distinguished pits at the bottom of which hairs rise from a cup-like tubercle, from those which were free from hairs. Leydig afterwards was the first to regard as olfactory organs the so-called pegs (kegel), a short, thick, hair-like structure distinctly perforated at the tip, which had already, by Lespès in Cercopis, etc., been described as a kind of tactile papilla. Other very peculiar olfactory organs of different form, Forel (Fourmis de la Suisse) discovered in the antennæ of ants, which Lubbock incorrectly associated with the nerve-end apparatus found by Hicks in other insects.
As the final result of his researches Kraepelin states that the great variety of antennal structures previously described may be referred to a single common fundamental type of a more or less developed free or sunken hair-like body which stands in connection by means of a wide pore-canal with a many-nucleated ganglion-cell. The latter sends only a relatively slender nerve-fibre (axial cord) through the pore-canal into the hair; but the same is enclosed by epithelial cells which surround the pore-canal.
Hauser’s researches on the organs of smell in insects were so carefully made and conclusive that our readers will, we feel sure, be glad to have laid before them in detail the facts which prove so satisfactorily that the antennæ of most insects are olfactory rather than auditory in their functions.
Physiological experiments.—First of all one should observe as exactly as possible the normal animal in its relation to certain odorous substances, whose fumes possess no corrosive power or peculiarities interfering with respiration; then remove the antennæ and try after several days to ascertain what changes have taken place in the relation of the animal to the substance. In order to come to no false results it is often necessary to let the insects operated upon rest one or two days, for immediately after the operation they are generally so restless that a careful experiment is impossible.
The extirpation of the antennæ is borne by different insects in different ways; many bear it very easily, and can live for months after the operation, while others die in the course of a few days after the loss of these appendages. The animals seem to be least injured if the operation is performed at a time when they are hibernating. Pyrrhocoris apterus, and many other insects, afforded a very striking proof of this relation.
269Experiments made by placing the antennæ in liquid paraffine so as to cover them with a layer of paraffine, thus excluding the air, gave the same result as if the antennæ had been removed.
The experiments may be divided, according to their object, into three groups. Experiments of the first kind were made on insects in their relation to strong-smelling substances, as turpentine, carbolic acid, etc., before and after extirpation of the antennæ. The second group embraces experiments on the relation of animals as regards their search for food; and finally the third group embraces experiments on the relation of the sexes relative to reproduction before and after the extirpation of the antennæ.
Relation of insects to smelling substances before and after the loss of their antennæ.—Taking a glass rod dipped in carbolic acid and holding it within 10 cm. of Philonthus œneus, found under stones at the end of February, it was seen to raise its head, turn it in different directions, and to make lively movements with its antennæ. But scarcely had Hauser placed the rod close to it when it started back as if frightened, made a sudden turn, and rushed, extremely disturbed, in the opposite direction. When he removed the glass rod, the creature busied itself for some time with its antennæ, while it drew them, with the aid of its fore limbs, through its mouth, although they had not come into direct contact with the carbolic acid. There was the same reaction against oil of turpentine, and it was still more violent against acetic acid.
After having many times carefully tested the relations of the normal animal to the substances mentioned, the antennæ were removed from the socket-cavity.
On the second day after Hauser experimented with the insects, they exhibited no reaction either against the carbolic acid, the oil of turpentine, or even against the acetic acid, although he held the glass rod which had been dipped into it for one or two minutes before and over the head. The creatures remained completely quiet and immovable, at the most slightly moving the palpi. They showed otherwise no change in their mode of life and their demeanor; they ate with great eagerness flesh which had been placed before them, or dead insects, and some were as active as usual as late as May. These beetles had, as proved by the experiments, lost the sense of smell alone; how far the sense of touch was lost Hauser could not experimentally decide.
The same results followed experiments with species of the genus Ptinus, Tenebrio, Ichneumon, Formica, Vespa, Tenthredo, Saturnia, Vanessa, and Smerinthus; also many species of Diptera and Orthoptera, 270besides Julus and Lithobius, while many larvæ reacted in the same manner.
Less satisfactory were the experiments with Carabus, Melolontha, and Silpha; there is no doubt that the species of these genera, through the extirpation of their antennæ, become more or less injured as to the acuteness of their powers of smelling; but they never show themselves wholly unable to perceive strong-smelling substances.
The allurement of the substance acts for a longer time on those deprived of their antennæ, then they become restless, then they wander away from the glass tube held before them; still all their movements are but slightly energetic, and the entire reaction is indeterminate and enfeebled.
Experiments with the Hemiptera gave still more unfavorable results; after the loss of their antennæ they reacted to smells as eagerly as those did which were uninjured.
Experiments on the use of the antennæ in seeking for food.—Under this head experiments were made with Silpha, Sarcophaga, Calliphora, and Cynomyia.
Silpha and its larva were treated in the following manner: they were placed in large boxes whose bottoms were covered with moss, etc.; in a corner of the box was placed a bottle with a small opening, in which was placed strong-smelling meat. So long as the beetles were in possession of their antennæ they invariably after a while discovered the meat exposed in the bottle, while after the loss of their antennæ they did not come in contact with it.
In a similar way acted the species of Sarcophaga, Calliphora, and Cynomyia. Hauser, in experimenting with these, placed a dish with a large piece of decayed flesh on his writing-table. In a short time specimens of the flies referred to entered through the open window of the room. The oftener he drove them away from the meat would they swarm thickly upon it. Then closing the window and catching all the flies, he deprived them of their antennæ and again set them free. They flew about the room, but none settled upon the flesh nor tried to approach it. Where a fly had alighted on a curtain or other object, the decayed flesh was placed under it so that the full force of the effluvium should pass over it, but even then no fly would settle upon it.
Experiments testing the influence of the antennæ of the males in seeking the females.—For this purpose Hauser chose those kinds in which the male antennæ differ in secondary sexual characters from those of the female, and in which it is known that they readily couple in confinement, as Saturnia pavonia, Ocneria dispar, and Melolontha vulgaris. The two first-named insects did not couple after the extirpation of their antennæ. Of Melolontha vulgaris 271twenty pairs were placed in a moderately sized box. On the next morning twelve pairs of them were found coupling. Hauser then, after removing the first lot, placed a new set of thirty pairs in the same box, cut off all the antennæ of the males and those of a number of females. On the following morning only four pairs were found coupling, and at the end of three days five others were observed sexually united.
From these experiments Hauser inferred that those insects deprived of their antennæ were placed in the most favorable situation, such as they would not find in freedom; for the space in which the insects moved about was so limited that the males and females must of necessity meet. But at the same time the results of the experiments cannot absolutely be regarded as proving that the males, after the loss of their antennæ, were then not in condition to find the females, because in the case of the above-mentioned moths, under similar conditions, after the extirpation of the antennæ no sexual union took place. If, however, the experiments made do not all lead to the results desired, Hauser thinks that the results agree with those of his histological researches, that in the greater number of insects the sense of smell has its seat in the antennæ. His results also agree with those of Perris.
Structure of the organs of smell in insects.—The olfactory organs consist, in insects,—i.e., all Orthoptera, Termitidæ, Psocidæ, Diptera, and Hymenoptera, also in most Lepidoptera, Neuroptera, and Coleoptera,—
1. Of a thick nerve arising from the brain, which passes into the antennæ.
2. Of a sensitive apparatus at the end, which consists of staff-like cells, which are modified hypodermis cells, with which the fibres of the nerves connect.
3. Of a supporting and accessory apparatus, consisting of pits, or peg- or tooth-like projections filled with a serous fluid, and which may be regarded as invaginations and outgrowths of the epidermis.
Hauser adds a remark on the distribution of the pits and teeth in the larvæ of insects, saying that his observations are incomplete, but that it appears that in the larvæ the teeth are most generally distributed, and that they occur not on the antennæ alone, but on the palpi; but in very many larvæ neither pits nor teeth[47] occurred. In the Myriopoda teeth-like projections occur on the ends of the antennæ. In Lithobius they form very small, almost cylindrical, pale organs.
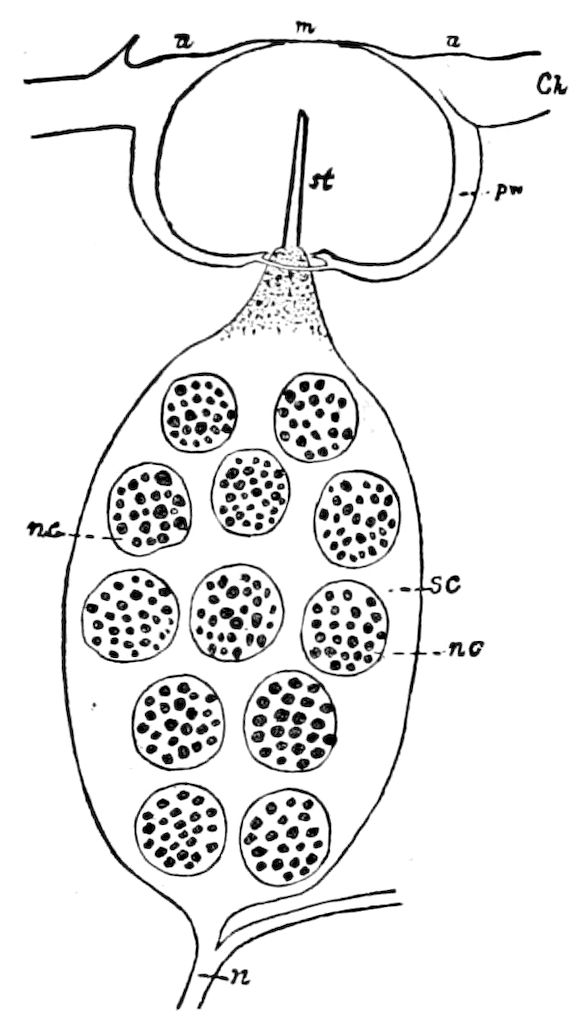
Fig. 268.—Olfactory organ of Caloptenus.
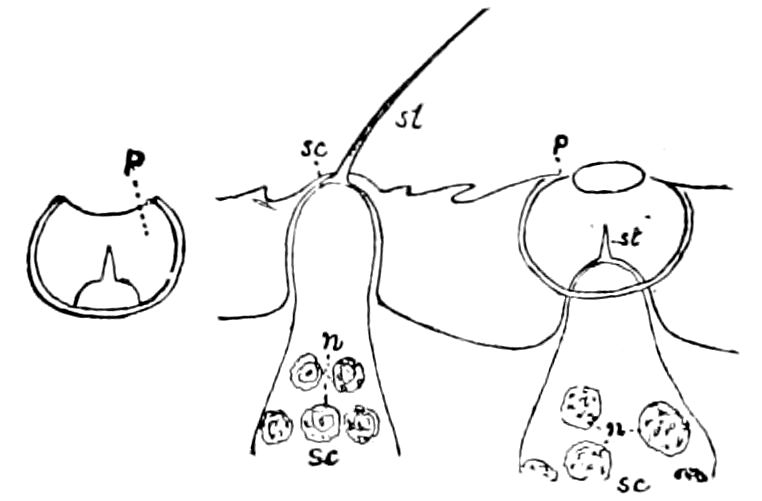
Fig. 269.—Olfactory pits of the antenna of Stenobothrus. This and Fig. 268 after Hauser.
Lettering for Figs. 268, 269, 273, 275, 276, 278–281.—a, a, circular thickening of the skin surrounding the opening of the olfactory pit; ax, thread-like continuation of the nerve-cell; b, vesicle-like bottom of the olfactory pit, through which the olfactory style passes; br bristle in Fig. 283, stout, and protecting the olfactory pit; bs, bent bristle or seta; ch, chitinous integument of the antennæ; d, seen in section; f, invaginated pit; Fv, Forel’s flask-shaped organ; Fvo, its opening seen from the surface; gl, gland-like mass of cells; hyc, hypodermic cells; i, entrance into the canal belonging to the pit; m, olfactory membrane; m′, m″, mc, membrane-forming cell; n, nerve of special sense; nc, nucleus of the sense- or ganglion-cell; o, opening into the olfactory pit; p, olfactory pit; cp, compound pits; pw, wall of the pit; s, a large seta; sc, sense- or ganglion-cell; st, olfactory or sense-style, sometimes peg-shaped; tb, tactile bristle.

Fig. 270.—A, b, sense-organ on the abdominal appendages of a fly (Chrysopila); c, sense-organ on the terminal joint of palpus of Perla.
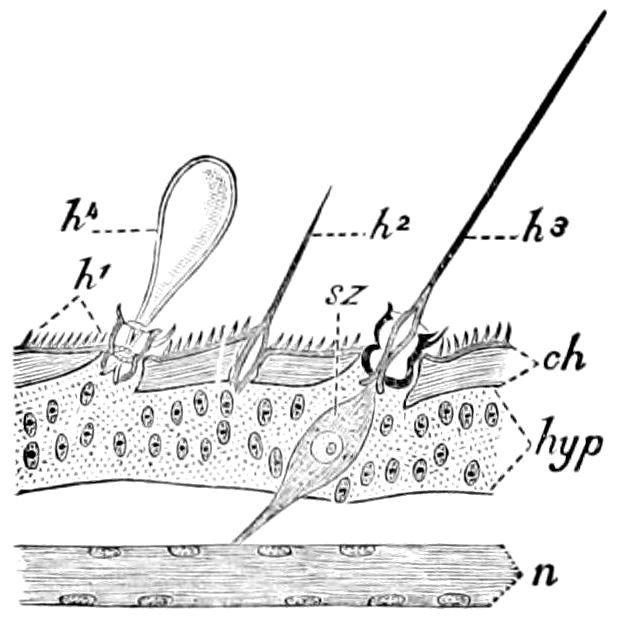
Fig. 271.—Longitudinal section of part of cercus of Acheta domestica: ch, cuticula; hyp, hypodermis; n. nerve; h′1, integumental hairs, not sensory; h2, ordinary hair; h3, sensory hair; h4, bladder-like hair; sz, sense-cell.—After Vom Rath, from Sharp.
In the course of a special description of these sense-organs in the Orthoptera, Hauser describes at length those of Œdipoda cœrulescens and Caloptenus italicus. On one antennal joint of Caloptenus (Fig. 268) was often counted 50 pits; on the anterior joints the number diminishes to about 30. Hauser thinks that in all Orthoptera whose antennæ are like those of Caloptenus occur similar pits, as he found them in Stenobothrus (Fig. 269) as well as in Œdipoda. Gryllotalpa possesses similar pits,—four to six on each antennal joint, making between 300 and 400 pits on each antenna.[48] In Mantis 273religiosa the pits were not detected, but on each joint, except the eighth basal, there are about 200 small, hollow, curved teeth with a fine opening in front.
In the Neuroptera (Chrysopa) there occur on the antennæ, besides numerous very long tactile bristles, small pale, transparent teeth. No pits could be detected.
In the Hemiptera (two species of Pyrrhocoris only were examined) only two kinds of tactile bristles occurred, but Hauser detected no pits, though Lespès states that they are present.
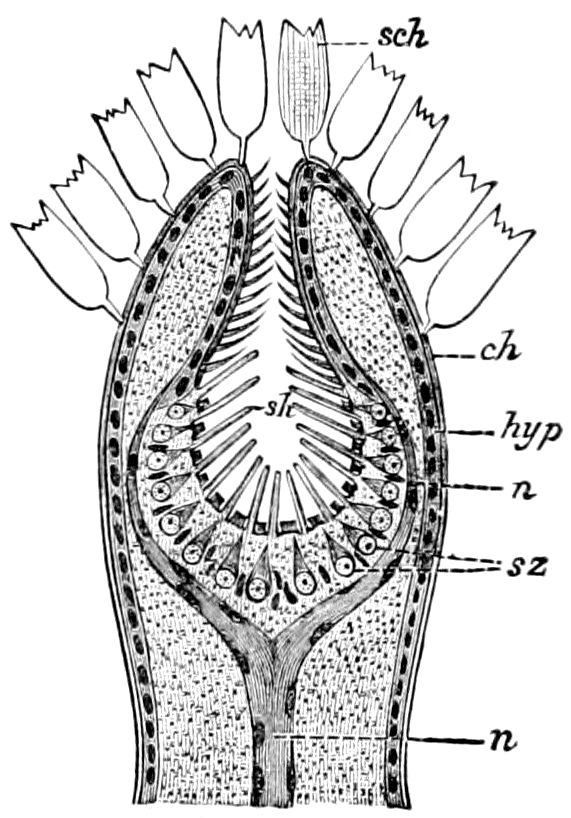
Fig. 272.—Longitudinal section of apex of palpus of Pieris brassicæ: sch, scales; ch, cuticula; hyp, hypodermis; n, nerve; sz, sense-cells; sh, sense-hairs.—After Vom Rath, from Sharp.
Of the Diptera, Hauser examined more than 60 species. The pits in the Diptera brachycera (Muscidæ, etc.) are unexceptionally confined to the third antennal joint. Their number varies extraordinarily in the different species. Helophilus florens has on each antennal disk only a single pit, while Echinomyia grossa possesses 200 of them. In flies of certain families the pits are compound, and contain 10, 20, and often 100 olfactory hairs, partly arising from the coalescence of several pits. Such pits are usually divided by lateral walls into several chambers, whose connection is only indicated by their common outlet. Simple olfactory pits with a single olfactory style were observed only in the Tabanidæ, Asilidæ, Bombylidæ, Leptidæ, Dolichopidæ, Stratiomyidæ, and Tipulidæ. In the last the compound forms do not occur at all, but in the other families mentioned also occur compound pits, receiving from two to ten nerve-terminations.
The antennal pits of flies are always sac-like invaginations of the external chitinous integument, of manifold shapes, opening externally and never closed by a membrane. The pits differ but slightly in the different species, and that of Cyrtoneura stabulans (Fig. 273) is described at length as typical of those of brachycerous flies in general.
The olfactory pits of the Tipulidæ seem to have a somewhat different structure, since the external passage is closed. It is circular, surrounded with a slight chitinous wall, and not covered with bristles. Such pits in their external appearance are like 274those of the locust (Caloptenus) and many Hymenoptera. They are situated usually on the third antennal joint. Pachyrhina pratensis L. has about 60 of them, as have Tipula oleracea L. and Ctenophora.
In the Lepidoptera, olfactory pits are much like those of flies. Hauser describes in detail those of Vanessa io. Those of the moths were not examined, but they can be readily and satisfactorily proved to be the site of the olfactory sense.
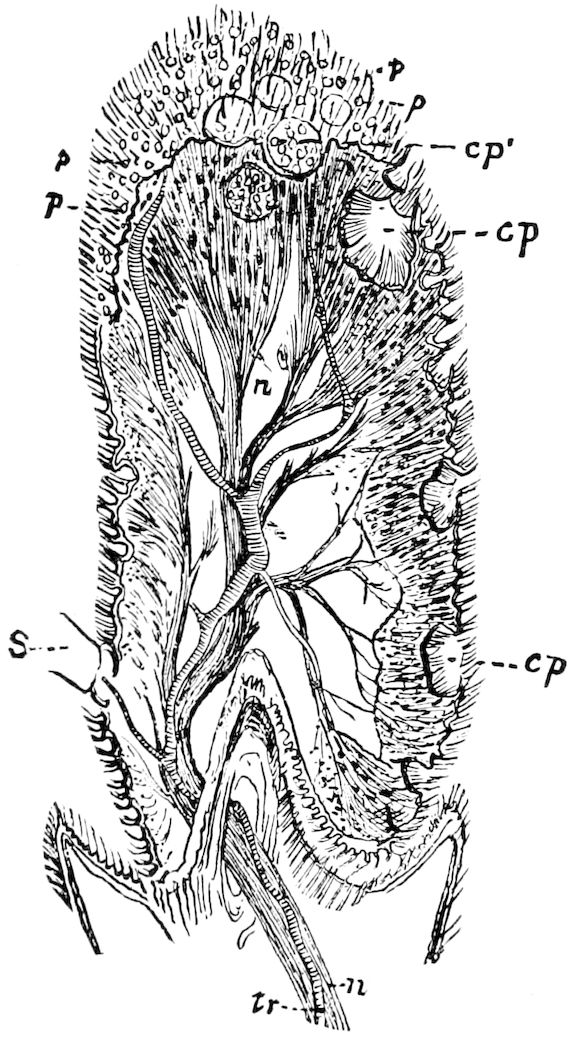
Fig. 273.—Longitudinal section through the third antennal joint of a fly (Cyrtoneura stabulans), showing the compound pits from above and in section.—After Hauser.
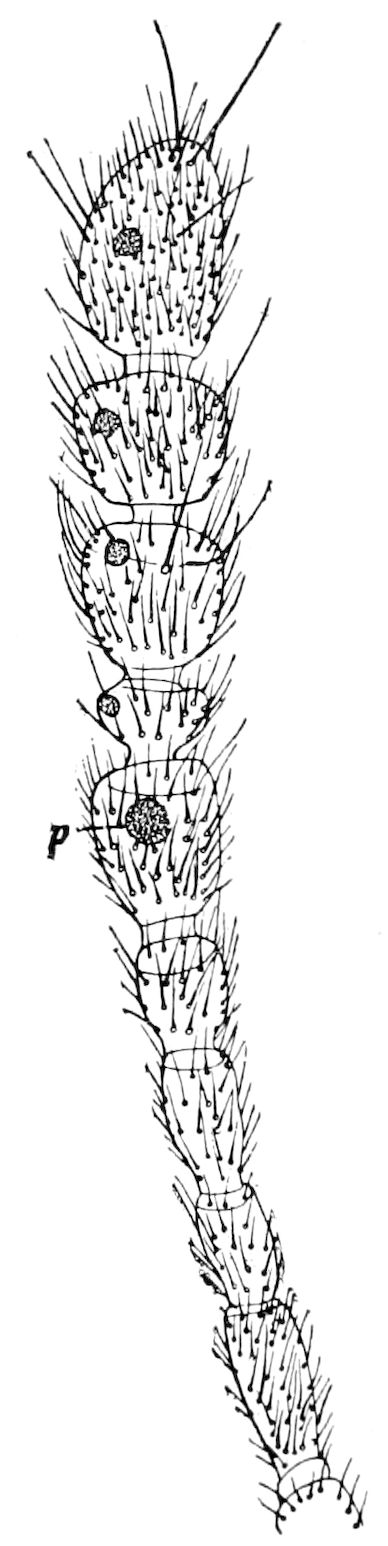
Fig. 274.—Antenna of Adelops, showing the olfactory organs (p) in the five last joints.
Historical researches in respect to the Coleoptera generally gave a very unfavorable result, contrary to Lespès’s views. That author states that in the Carabidæ the pits are found on the four first joints, but Hauser could discover them in none which he examined. Usually only tactile bristles occur, so also in the Cerambycidæ, Curculionidæ, Chrysomelidæ, and Cantharidæ. In a blind silphid beetle 275(Adelops hirtus) of Mammoth Cave we have found well-marked olfactory organs (Fig. 274). Similar organs occur in the antennæ of the Panorpidæ.
Olfactory pits, however, without doubt occur in Silpha, Necrophorus, Staphylinus, Philonthus, and Tenebrio. The openings of the pits are small and surrounded with a small chitinous ring; in Silpha, Necrophorus, and Tenebrio they cannot easily be distinguished from the insertion-cavities of the bristles, but in Philonthus and Staphylinus they are less like them, being distinguished by their somewhat larger size and their often more oval form. In Philonthus æneus about 100 such small pits occur irregularly on the terminal joints; besides, in this species on each side of the terminal joint is an apparatus which is like the compound pit generally occurring in the Diptera.
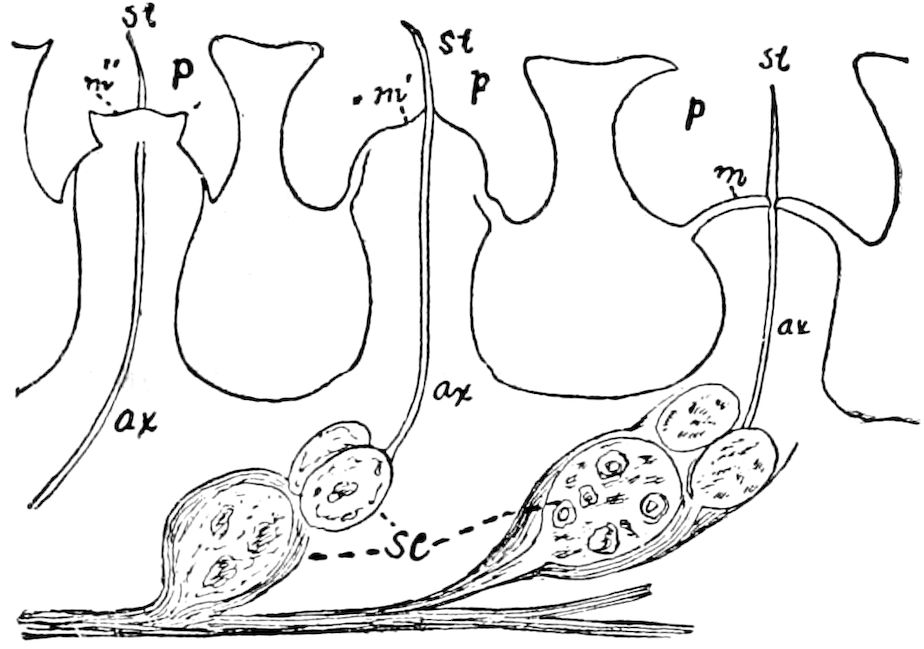
Fig. 275.—Olfactory pits of the antenna of Melolontha vulgaris.—After Kraepelin.
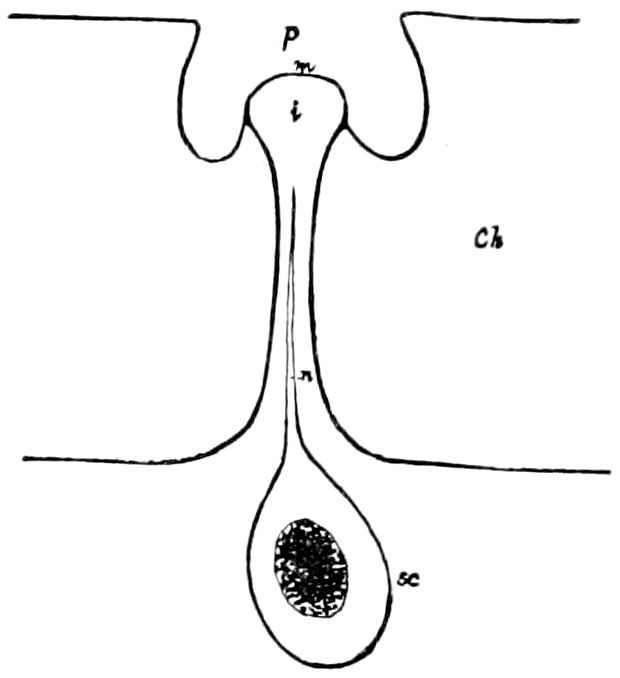
Fig. 276.—Antennal pit of Melolontha vulgaris, seen in vertical section.—After Hauser.
Very remarkable pits occur in the antennal lamellæ of Melolontha vulgaris (Fig. 275) and other lamellicorns. On the outer surface of the first and seventh (in the female the sixth) antennal leaf, as also on the edges of the other leaves, only arise scattered bristles; on the inner surface of the first and seventh leaves, as also on both surfaces of the second to sixth leaves, are close rows of rather shallow depressions of irregular form, some circular, others regularly hexagonal. Their number is enormous: in the males 39,000, in the females about 35,000, occur on each antenna.
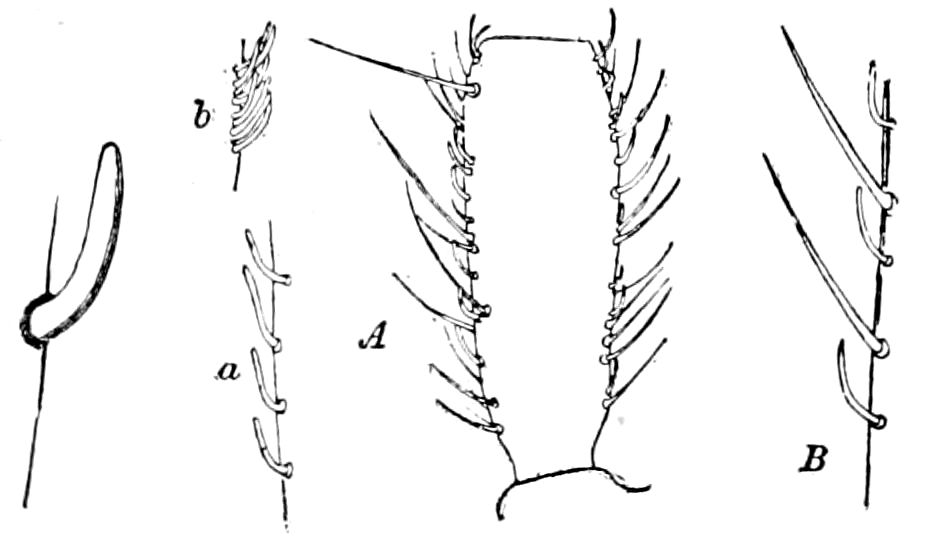
Fig. 277.—Organ of smell of Anophthalmus.—After Hauser. A, a, b, the same in A. tenuis, B in A. tellkampfii.
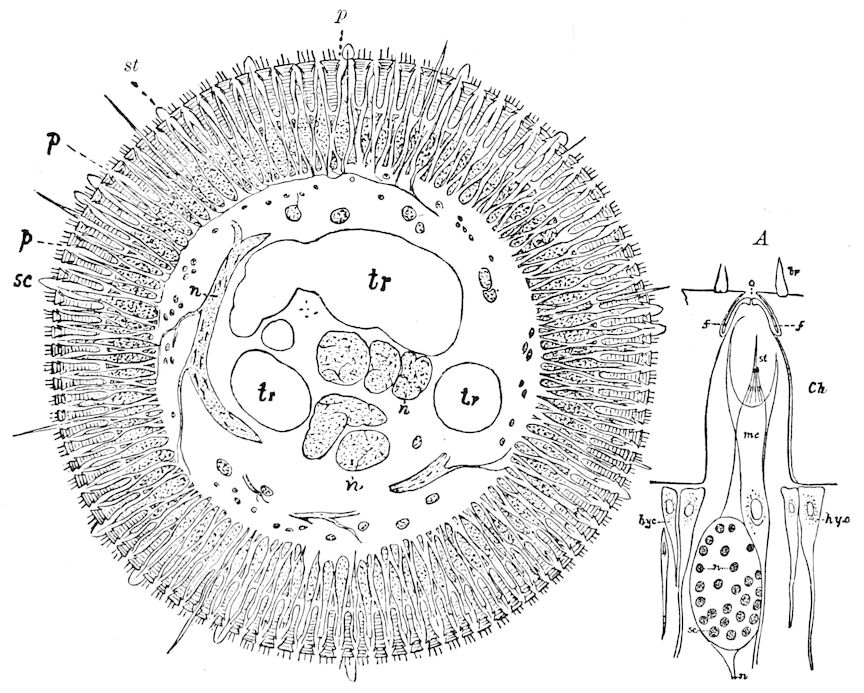
Fig. 278.—Section through antennal joint of Vespa crabro, showing the great number of olfactory pits, olfactory and tactile bristles. A, section through an olfactory pit of Vespa crabro.—After Hauser.
The antennal pits and teeth of Dyticus marginalis are morphologically and physiologically identical with those of bees and wasps. In Anophthalmus bilimekii, Hauser found on the last antennal joints about 60 teeth, which essentially differ in form from those previously described; they are very pale, transparent, cylindrical, elongated, and bent elbow-shaped on the first third, so that the last two-thirds run parallel with the antenna. The length of these remarkable teeth is 0.035 mm., their breadth 0.005 mm. He only found them in Anophthalmus, and in no other species of Carabidæ; they must resemble the teeth described in Chrysopa. Our species possesses similar processes (Fig. 277). Similar teeth occur on the maxillary and labial palpi of beetles. Dyticus marginalis possesses at the end of each terminal palpal joint a group of very small teeth, which were also detected in Anophthalmus bilimekii, Melolontha vulgaris, etc. In 277Carabus violascens were detected on the maxillary palpi large, plainly microscopical, white disks, which are surrounded with a great number of extremely small teeth.
Whether the above-described organs on the palpi of beetles should be considered as olfactory or gustatory in their nature can only be determined by means of physiological experiments; they probably receive taste-nerve terminations.
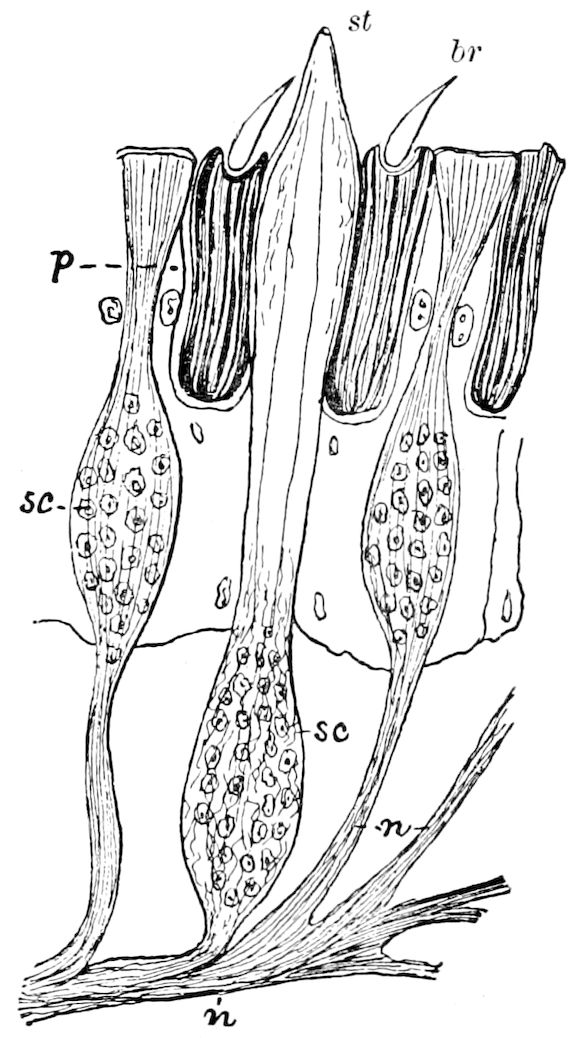
Fig. 279.—Olfactory pits of the antenna of Vespa vulgaris.—After Kraepelin.
The Hymenoptera furnished very good material for histological purposes, so that Hauser could not only study the terminal apparatus of the olfactory nerves in the perfect insect, but also in three different stages of the pupa. These are described at length, as regards the distribution of the pits and teeth, in Vespa crabro; each joint of the antenna (flagellum) possesses between 1300 and 1400 pits, nearly 60 teeth, and about 70 tactile hairs; on the terminal joint there are more than 200 teeth, so that each antenna has between 13,000 and 14,000 olfactory pits and about 700 teeth (Kegeln). Fig. 278 represents a cross-section through the penultimate antennal joint of Vespa crabro; we can see how thick are the series of openings on the surface of the antennæ, and how regular is the distribution of the teeth.
The distribution of the olfactory pits and olfactory teeth is thus seen to be very general; the deviations are so insignificant that there is no reason for the establishment of more than one type.
Antennal pits with a small crevice-like opening occur in genera nearly allied to Vespa and also in most Ichneumonidæ, Braconidæ, and Cynipidæ. But the crevice-like openings in these families are considerably longer and often are of a somewhat twisted shape. In all the species with translucent antennæ we can recognize the inner mouth of the pit as a round or nearly round disk situated usually under the middle of the opening. The antennal pits of Apis mellifica, as well as those of Bombus (Fig. 280) and allied genera, differ 278from those of the Ichneumonidæ in being not like crevices, but circular openings.
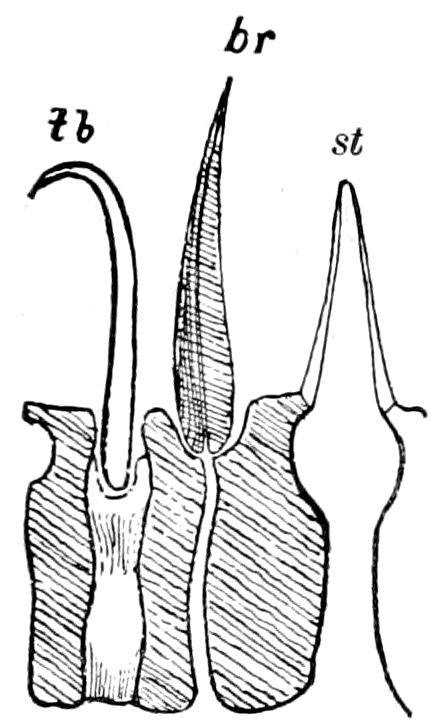
Fig. 280.—Olfactory pits of the antenna of Bombus.—After Kraepelin.
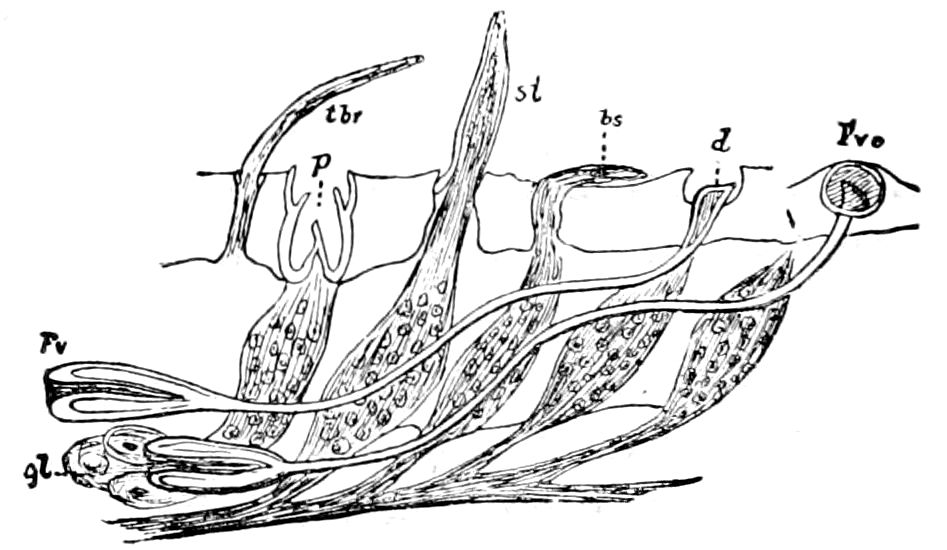
Fig. 281.—Olfactory pits of the antenna of Formica: Fv, Hicks’ “bottle,” Forel’s flask-shaped organ, Fvo, its opening.—After Kraepelin.
Fig. 282.—Supposed olfactory organs at end of antenna of Campodea: A, C. staphylinus. B, C. cookei, from Mammoth Cave.
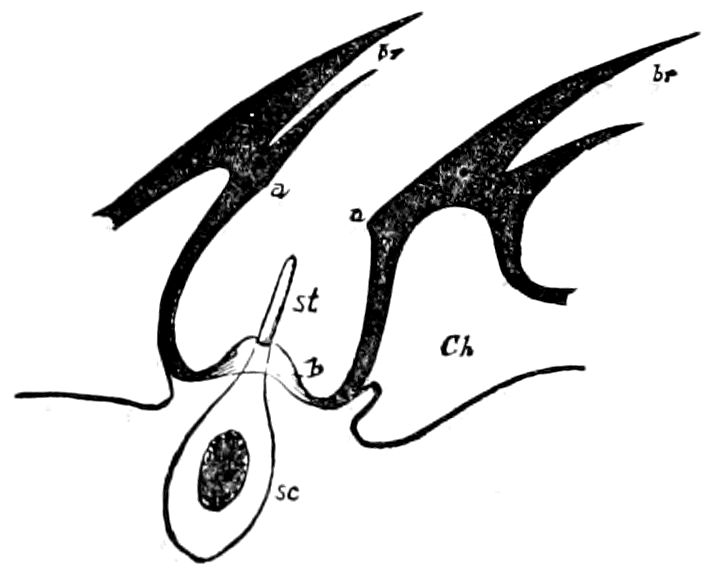
Fig. 283.—Vertical section through a single olfactory pit in the antenna of the horse-fly (Tabanus bovinus). For lettering see p. 272.—After Hauser.
The distribution of the olfactory peg or tooth-like projections seems to be much more limited than that of the pits in the Ichneumonidæ. Hauser could not find any. Apis mellifica possesses on each antennal joint only about twenty slender pale teeth, scarcely a third as many as in Vespa crabro; on the other hand, Formica, of which genus several species were examined, seems to have far more teeth than pits; they are relatively long, pale, transparent, and somewhat clavate; they are not unlike those of Chrysopa; on the terminal joint only occur the round openings (Fvo), which lead into a bottle-shaped invagination of the integument (Fv) and contain an olfactory style (Fig. 281). In the Tenthredinidæ only teeth and no pits were to be detected. Sirex has on the under side of the nine last joints of each antenna a group of from 200 to 300 small teeth, which resemble those of Vespa crabro; Lyda has on the terminal joints about 100 279teeth. We may add that supposed organs of smell occur on the antennæ of Campodea (Fig. 282).
Kraepelin also thus briefly summarizes Hauser’s statements as to the forms of the different organs of smell.
The manifold nature of the antennal organs has, by Hauser, from thorough studies of the nerve-elements belonging to them, been not simplified but rendered more complicated. According to this naturalist we may distinguish the following forms which the olfactory organs may assume: 1. “Pale, tooth-like chitinous hairs on the outer surface of the antennæ, which are perforated at the end; nothing is known as to the relation of the nerve passing into it (Chrysopa, Anophthalmus). 2. In pit-like depressions of the antennæ arise nerve-rods (without a chitinous case) which stand in direct relation with a ganglion-cell lying under it. These pits are either simple, viz. with only an ‘olfactory rod’ (Tabanus, Fig. 283, and other Diptera, Vanessa), or compound (Muscidæ, and most other Diptera, and Philonthus). It is important that these pits are partly open (in the above-named groups of insects), and partly closed and covered with a thin membrane, under whose concavity the olfactory rods end (Orthoptera, Melolontha, and other lamellicorns). 3. Short, thick pits sunken slightly into the surface of the antennæ, and over this a chitinous peg perforated at the end, in whose base, from the interior, projects a very singular nerve-peg, which is situated over an olfactory ganglion-cell, and provided with a slender crown of little rods, and flanked on each side by a flagellum-cell (Hymenoptera). 4. Round or crevice-like pits covered over by a perforated chitinous membrane with nerve-rods like those in 3, but in place of the flagellum-cell with ‘membrane-forming’ cells spread before it. Hauser finally mentions further differences in the ganglion-cells sent out into the nerve-end apparatus. These exhibit in Diptera and Melolontha only one nucleus, in Hymenoptera a single very large one (with many nucleoli) and three small ones, in Vanessa six, in Orthoptera a very large number of nuclei, etc.”
LITERATURE OF THE ORGANS OF SMELL
Réaumur, Réné Ant. de. Mémoires pour servir à l’histoire des insectes. Paris, 1734–42. (i, 283; ii, 224).
Lesser, F. C. Insecto-theologia, 1740, p. 262.
Roesel, A. J. Insektenbelustigungen, 1746, ii, p. 51.
Reimarus, H. S. Allgemeine Betrachtung ueber die Triebe der Thiere hauptsächlich ihre Kunsttriebe (Instinct). Hamburg, 1760, p. 355.
Sulzer, J. H. Die Kenntzeichen der Insecten. Zürich, 1761.
Lyonet, P. Traité anatomique de la chenille, 1762, pp. 42, 96, 195.
Comparetti, A. De aure interna comparata. Patavii, 1769.
Bonnet, C. Œuvres complètes, 1779–1783, ii, p. 36.
—— Contemplation de la nature, Ch. iii, p. 18.
Scarpa, Ant. Anatomicæ disquisitiones de auditu et olfactu. Ticini, 1789.
Huber, F. Nouvelles observations sur les abeilles, 1792, ii, p. 475.
Lehrmann, M. C. G. De antennis insectorum. Londini, Hamburgi, 1799, p. 48, Diss. posterior; Hamburg and London, 1800, p. 80 (especial sense, ærocepsis).
Latreille, P. A. Histoire naturelle des crustacés et des insectes, 1806–1809, ii, 50.
Blainville, M. H. D. Principes d’anatomie comparée, 1822, i, p. 339.
Dugès, A. L. Traité de physiologie comparée, 1838, i, p. 161.
280Newport, G. On the use of the antennæ of insects. (Trans. Ent. Soc., London, ii, 1840, pp. 229–248.)
Robineau-Desvoidy, A. J. B. Sur l’usage réel des antennes chez les insectes. (Ann. Soc. Ent. France, 1842, xi Bull., pp. 23–27.)
Erichson, W. F. De fabrica et usu antennarum in insectis. Berlin, 1847, 1 Tab., p. 13.
Perris, E. Mémoire sur le siège de l’odorat dans les articulés. (Ann. sc. nat., Sér. 3, 1850, xiv, pp. 159–178.)
Dufour, L. Quelques mots sur l’organe de l’odorat et sur celui de l’ouie dans les insectes. (Actes d. l. Soc. Linn., Bordeaux, 1850, xvii, Ann. sc. nat., Sér. 3, Zool., xiv, 1850, pp. 179–184.)
Leydig, F. Zum feineren Bau der Arthropoden. (Müller’s Archiv, 1855, pp. 376–480); Lehrbuch der Histologie, 1857, p. 220; Zur Anatomie der Insekten (Archiv für Anatomie, 1859, pp. 35–89 and 149–183).
—— Ueber Geruchs- und Gehörorgane der Krebse und Insekten. (Archiv f. Anat. u. Phys., 1860.)
—— Die Hautsinnesorgane der Arthropoden. (Zool. Anzeiger, 1886, pp. 284–291, 308–314, 265–314.)
Landois, H. Das Gehörorgan des Hirschkäfers. (Archiv f. mikrosp. Anat., 1868, iv, pp. 88–95.)
Troschel, H. Ueber das Geruchsorgan der Gliedertiere. (Verhandl. d. naturhist. Vereins d. preuss. Rheinlande u. Westfal., xxvii Jahrg., 1870, pp. 160–161.)
Packard, A. S. The caudal styles of insects sense-organs, i.e., abdominal antennæ. (Amer. Naturalist, 1870, pp. 620, 621. Also Proc. Bost. Soc. Nat. Hist., 1868, xi, p. 398.)
Paasch, A. Von den Sinnesorganen der Insekten im Allgemeinen, von Gehörund Geruchsorganen im Besondern. (Archiv für Naturgesch., xxxix Jahrg., i, 1873, pp. 248–275.)
Chadima, Jos. Ueber die von Leydig als Geruchsorgane bezeichneten Bildungen bei den Arthropoden. (Mitteil. d. naturwiss. Ver. f. Steiermark, 1873, pp. 36–44.)
Forel, A. Les Fourmis de la Suisse. (Neue Denkschr. Allg. Schweiz. Gesellsch. f. d. ges. Naturw., xxvi, 1874, pp. 118, 144.)
—— Études myrmécologiques en 1884, avec une description des organes sensoriels des antennes. (Bull. Soc. Vaud. sc. nat., 1885, Sér. 2, xx, pp. 316–380.)
Graber, V. Die Insekten. München, 1877.
—— Neue Versuche über die Functionen der Insektenfühler. (Biol. Centralb., vii, 1887, pp. 13–19.)
Trouvelot, L. The use of the antennæ in insects. (Amer. Naturalist, xi, 1877, pp. 193–196.)
Mayer, P. Sopra certi organi di senso nelle antenne dei Ditteri. (Atti R. Accad. d. Lincei, Roma, Ser. 3, Mem. Cl. sc. fis., mat. e natur., iii, 1879, p. 11.)
Hauser, Gustav. Physiologische und histiologische Untersuchungen über das Geruchsorgan der Insekten. (Zeitschr. f. wissens. Zool., xxxiv, 1880, pp. 367–403, 3 Taf.)
Kraepelin, Karl. Ueber die Geruchsorgane der Gliedertiere (Oster-Programm der Realschule des Johanneums. Hamburg, 1883, pp. 48, 3 Taf.)
Schiemenz, Paulus. Ueber das Herkommen des Futtersaftes und die Speicheldrüsen der Bienen nebst einem Anhange über das Riechorgan. (Zeitschr. f. wissens. Zool., xxxviii, pp. 71–135, 3 Taf.)
Plateau, F. Expériences sur le rôle des palpes chez les arthropodes maxillés, I., Palpes des insectes broyeurs. (Bull. Soc. Zool. France, x, 1885, pp. 67–90.)
281Plateau, F. Une expérience sur la fonction des antennes chez la Blatte (Periplaneta orientalis). (Comptes rend. Soc. Ent. de Belgique, 1886, pp. 118–122, 1 Fig.)
Lubbock, J. On some points in the anatomy of ants. (Monthly Micr. Journ., 1887, pp. 121–142.)
Ruland, Franz. Beiträge zur Kenntniss der antennalen Sinnesorgane der Insekten. Diss. Marburg, 1888, p. 31, 1 Taf.
Wasmann, E. Die Fühler der Insekten. (Stimmen aus Maria-Laach. Freiburg i. B., 1891, p. 37.)
Sergi, G. Ricerche su alcuni organi di senso nelle antenne delle formiche. (Riv. Filos. Sci. Milano, 1891, p. 10, 3 Figs.)
Nagel, Wilibald. Die niederen Sinne der Insekten, 19 Figs., Tübingen, 1892, pp. 67.
With the writings of Baster, Lamarck, Cuvier, Treviranus, Oken, Lefebure, Duméril, Schelver, Bousdorf, Rosenthal, Burmeister, Slater, Balbiani, Marcel de Serres, Garnier, Berté, Porter, Sazepin, Reuter, Pierret, Duponchel, Driesch, Küster, Peckham, Lubbock, A. Dohrn, Lespès.
c. The organs of taste
The gustatory organs of insects are microscopic pits or setae, either hair-like or resembling short pegs, which form the ends of ganglionated nerves. They are difficult to distinguish morphologically from certain olfactory structures, and it is owing to their position at or very near the mouth that they are supposed to be gustatory in nature.
Meinert was the first (1860) to suggest that organs of taste occurred in ants. He observed in the maxillæ, and tongue of these insects a series of canals in the cuticula of these organs connected with ganglion-cells, and through them with the nerves, and queried whether they were not organs of taste. Forel afterwards (1874) confirmed these observations. Wolff in an elaborate work (1875) described a group of minute pits (Fig. 284) at the base of the tongue of the honey bee, which he supposed to possess the sense of smell, but Forel and also Lubbock attributed to these sensory pits the function of taste. Ten years afterward Will showed conclusively, both by anatomical studies and by experiments, that Diptera and Hymenoptera possess gustatory organs. He, however, denied that the organs of Wolff were gustatory, and maintained that organs of smell were confined to the maxillæ, paraglossæ, and tongue. As we shall see, however, what appear to be with little doubt taste-pits, with hairs or pegs arising from them, are most numerous on the epipharynx of nearly all insects, and situated at a point where they 282necessarily must come in contact with the food as it enters the mouth and passes down the throat.
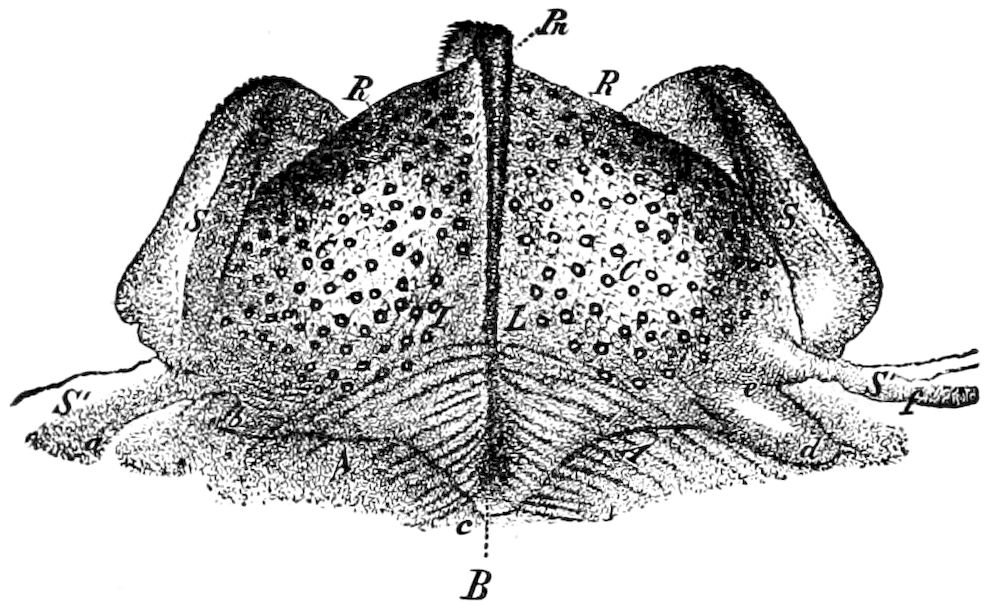
Fig. 284.—Taste-pits on the epipharynx (C) of the honey-bee: B, horny ridge; R, R, taste-pits; L, A, A, muscular fibres; S, S′, a b c d e f, section of skin of œsophagus.—After Wolff.
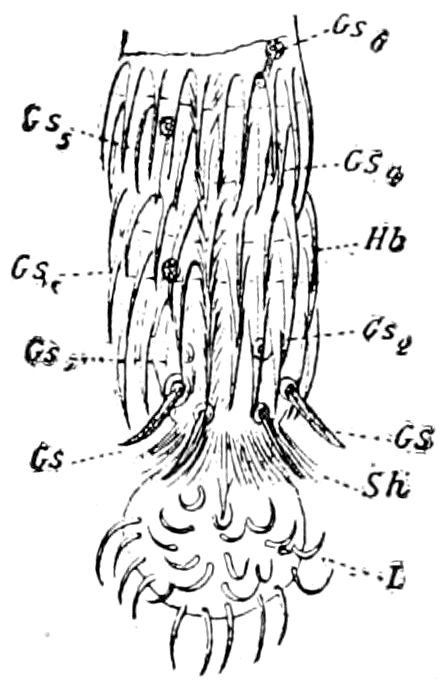
Fig. 285.—Tip of the proboscis of the honey bee, × 140: L, terminal button or ladle: Gs taste-hairs; Sh, guard-hairs; Hb, hooked hairs.—After Will.
Kraepelin (1883) discovered taste-organs on the proboscis of the fly, and taste-hairs at the end of the tongue of the humble-bee (Fig. 285), and afterwards Lubbock critically discussed the subject, and concluded that the organs of taste in insects are situated “either in the mouth itself, or on the organs immediately surrounding it.”
Structure of the taste-organs.—The organs have been best studied by Will, who, besides describing and figuring the chitinous structures, such as the pits or cups, hairs and the pegs, showed that they were the terminations of ganglionated nerves.
Figure 286 represents the taste-cups on the maxilla of a wasp, and Fig. 287 the taste-cone or peg projecting from the cup or pit. The cell out of which the pit and projecting hair or peg are formed is a modified hypodermis cell; and the seta is apparently a modification of a tactile hair, situated at the end of a nerve, which just beneath the chitinous structures passes into a ganglion-cell, which sends off a nerve-fibre to the main nerve.
Will detected on the tongue of the yellow ant (Lasius flavus) from 20 to 24, and in Atta from 40 to 52, of these structures. The number of pits on the maxillæ vary much, not always being the same on the two sides of the same insect. We have observed these taste-cups in the honey and humble bee, not only at the base of the second maxillæ (Fig. 288, g), but also on the paraglossæ (pg).
Distribution in other orders of insects.—The writer has detected these taste-cups in other orders than Diptera and Hymenoptera. They very generally occur in mandibulate insects on the more exposed surface of the epipharynx 283(compare pp. 43–46). We have not observed them in the Synaptera (Lepisma and Machilis).
In the Dermaptera the taste-cups appear to be undeveloped in the nymph, while in the adult they are fewer in number than in any other pterygote order yet investigated.
In a species of Forficula from Cordova, Mexico, the taste-pits are few in number, there being only about a dozen on each side in all; most of them being situated on the anterior half, and a few near the base. The taste-pits are provided each with a short fine seta, as usual arising from the centre.
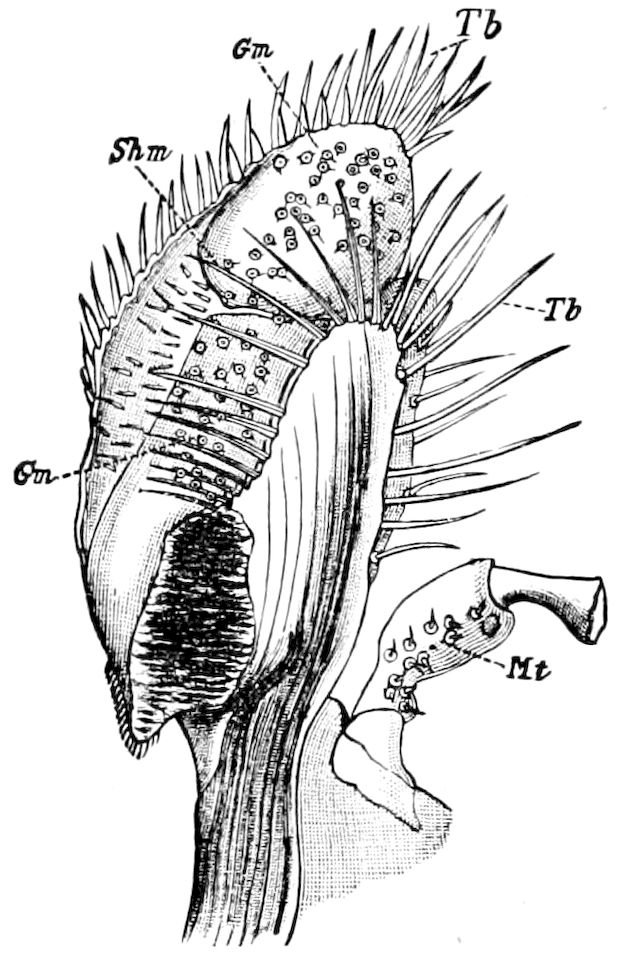
Fig. 286.—Under side of left maxilla of Vespa: Gm, taste-cups; Shm, protecting hairs; Tb, tactile hairs; Mt, base of maxillary palpus.—After Will.
In the order Platyptera (including Perla, Pteronarcys, Psocus, Termes, Eutermes, and Termopsis) we have been unable to detect any organs of taste.

Fig. 287.—Section through a taste-cup: SK, supporting cone; N, nerve; SZ, sense-cell.—After Will. This and Figs. 284–286 from Lubbock.
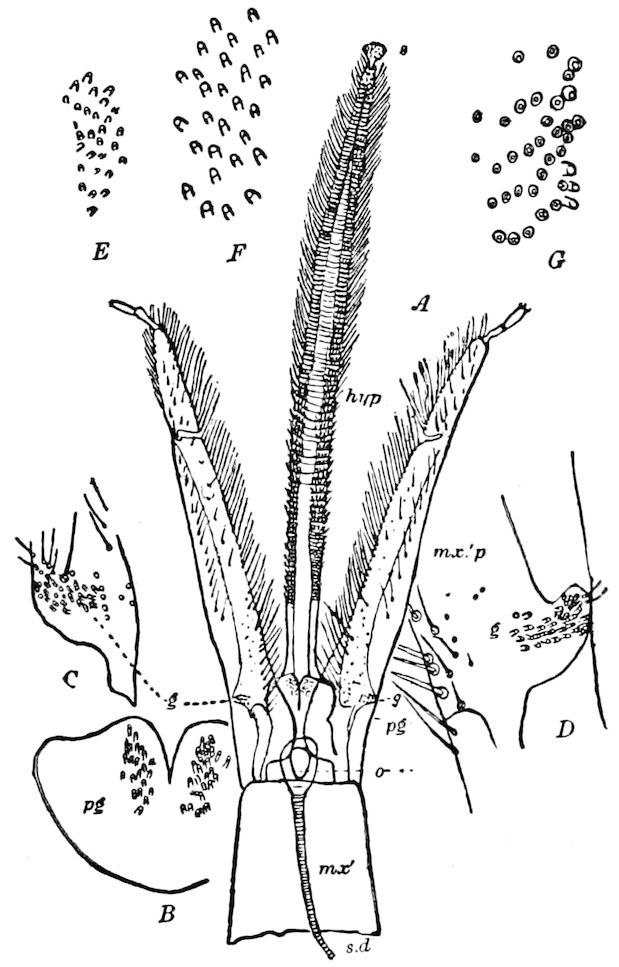
Fig. 288.—Tongue of worker honey-bee: pg, paraglossæ; B, the same enlarged, showing the taste-papillæ; C, D, base of a labial palpus (mx.′p) with the taste-papillæ; E, taste-cups on paraglossæ of Bombus; F, group of same on left, G, on right side, at base of labial palpus.
In the Odonata, however, they are fairly well developed; in Calopteryx, about 50 taste-cups were discovered; in a species of Diplax about 28, there being a group of 14 at the base of the epipharynx on each side of the median line, while in Æschna heros there are two groups of from 25 to 30 taste-cups, situated as in the two aforenamed genera.
In the Orthoptera the gustatory cups are numerous, well developed, and present in all the families except the Phasmidæ, where, however, they may yet be found to occur.
284In a large cockroach (Blabera) from Cuba they are well developed. On each side of the middle of the epipharynx is a curved row of stiff, defensive spines, and at the distal end of each row is a sensory field, containing 20 taste-cups on one side and 23 on the other. Near the front edge of the clypeal region are two more sensory fields, situated on each side of the median line, there being 35 taste-cups in each field. The taste-cups in this form are rather smaller than usual in the order.
In the Acrydiidæ they are more numerous than in the Blattidæ. For example, in Camnula pellucida, near what corresponds to the front edge of the clypeus are two gustatory fields, each bearing about 35 taste-pits. Just in front, under the clypeo-labral suture, are two similar fields, each containing from 40 to 42 taste-pits. There are none in front of these. There are thus about 140–150 sense-cups in all.
The members of the Locustidæ (Fig. 26) appear to be better provided with the organs of taste than any other Orthoptera, those of the katydid numbering from 170 to 180. There are from 50 to 60 taste-cups in the front region; behind the middle a group of 25 on each side, and over an area corresponding to the base of the labrum and front edge of the clypeus is a sensory field with about 70 taste-cups on each side. They are true cups or beaker-like papillæ, some with a fine, others with a short, stout, conical seta.
The gustatory organs in the cave cricket (Hadenœcus subterraneus, Fig. 27), from Mammoth Cave, are highly developed, being rounded papillæ with the nucleus at the top or end. They are grouped on each side of the middle near the front edge, there being 25 on each side. An irregular row of these beaker-like organs extends along each side; some occur under the base of the labrum, but they are most numerous in a field corresponding to the front edge of the clypeus, there being 50 on each side, or 100 in all, where in Ceuthophilus there are only 9 or 10. It would thus appear as if the sense of taste were much more acute in the cave-dweller than in the out-of-doors form.
In the Coleoptera taste-cups and setæ are very generally distributed, though we were unable to detect them in Dendroctonus or in Lucanus dama. As seen in Fig. 57, we have observed numerous taste-pegs along the maxilla of Nemognatha lurida, but otherwise taste-organs have only been detected in the epipharynx. They not only occur in the adult beetles, but we have found them in the larvæ of cerambycid, scarabæid, and other beetles. In the adults taste-cups appear to be about as well developed in the carnivorous forms (Carabidæ) as in the phytophagous or lignivorous groups.
In Chlænius tomentosus there are about half as many of these organs as in Harpalus, while in Calosoma there are 90 taste-cups, 45 on each side, under the base of the labrum. The cups are papilliform, being rather high, with a seta arising from each.
In the Cicindelidæ, the epipharynx bears a sensory field quite different from that of the Carabidæ. There are no normal taste-cups, except a few situated on two large, round, raised areas which are guarded in front by a few very long setæ. On the surface of each area are numerous very long setæ which may, if not tactile, have some other sense, as they arise from cup-like bases or cells. Those on the outside are like true taste-cups, with a bristle but little larger than normal in taste-cups generally. We are disposed to regard this sensory field as a highly specialized gustatory apparatus.
In the Dyticidæ the taste-cups are nearly as described in the Carabidæ.
The Staphylinidæ are not well provided with taste-organs. Under the clypeus of Staphylinus violaceus, on each side near the middle, is a bare rounded area, in which are situated 4 or 5 papilliform taste-cups, and at the base behind them 285is another linear group of about 7 slenderer, somewhat curved, taste-cups. In the Elateridæ these organs are scantily developed.
In the Buprestidæ (Buprestis maculiventris alone examined) no true taste-cups were detected. On the other hand, the Lampyridæ are well supplied with them. Under the clypeus is situated a sensory field bearing 26 taste-cups, which are rather smaller than usual. Over the epipharyngeal surface are scattered a few taste-cups, but they are small and perhaps not gustatory. Under the clypeus of Lucidota punctata Lec. is a group of 12 taste-cups, and in the middle region of the epipharynx, situated in a field extending from near the base to near the front edge, are about 40 taste-cups, which, however, are not, as is usual, arranged on each side of the median line, but are scattered among the hairs of the pilose surface of the epipharynx. In the Cleridæ the taste-cups are few in number.
In the great family of Scarabæidæ, the presence of gustatory organs is variable. None occur in Lucanus dama, though in the June beetle (Lachnosterna fusca Fröhl.) they are abundantly developed. The epipharynx bears on each side outside of a spiny area a group of about 50 taste-cups, each bearing a long seta, those on the outside of the area passing into a few high, rather slender, papillæ, without a seta. On the under side of the clypeus is a median group of 10 taste-cups of singular form, the cups being large, with broad bases, which posteriorly bear three spines, of which the median one is the largest.
Taste-cups occur without any known exception in the longicorn beetles. In Leptura canadensis they are numerous; in Euryptera lateralis they are abundant along and near the middle of the anterior half of the labral region, and in Cyllene robiniæ Forst. (or pictus Drury) they are more numerous than usual, extending in an unbroken sensory field from near the front margin of the clypeal region to near the front edge of the epipharynx. The cups vary much in size, some being one-half as large as others; and those on the sides of the sensory field bear short, and a few others rather long, bristles, showing that the taste-cups are modified tactile bristles.
The Tenebrionidæ are fairly well endowed with taste-cups, their number in Eleodes obsoleta Say amounting to 30 or 40.
Those of the Meloidæ especially are unusual in size and number.
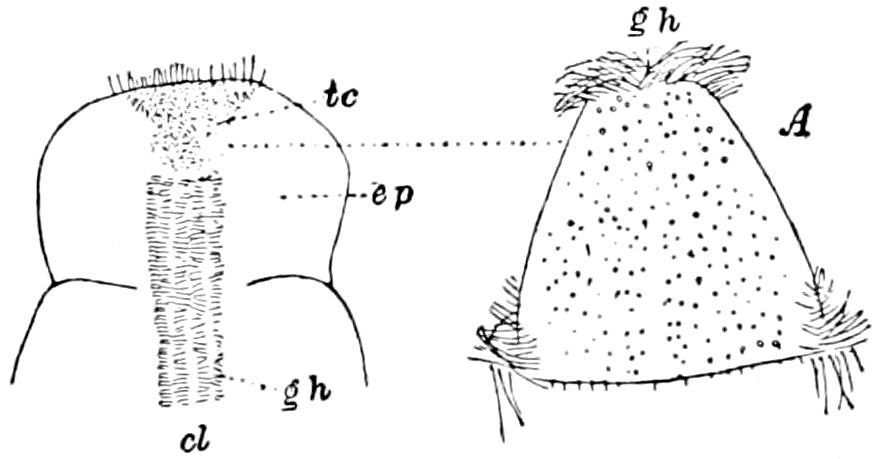
Fig. 289.—Epipharynx (ep) of Nemognatha: cl, clypeus; gh, gathering hairs; tc, triangular sensory field dotted with taste cups; A, the field enlarged.
In Nemognatha lurida Lec. (Fig. 289) the front edge of the epipharynx contains about 80 remarkably small taste-cups, arranged irregularly in a triangular sensory space, and not more than ¼ to ⅙, as large as those on the maxillæ of the same beetle. Unless the former structures are gustatory it is difficult to account for their presence here, and it will be observed that the taste-cups in Epicauta are unusually abundant. Thus in the middle and near the front of the epipharynx of the blister-beetle over 100 gustatory cups were counted. They are conical, papilliform, and truncated at the end as if open, the edge of the opening being ragged, though bearing no bristle, except in a few cases. Around the edge of the sinus, on the under side of the labrum, is a regular marginal row of large, longer, more distinctly chitinized taste-cups, whose walls are streaked up and down by chitinous thickenings. In E. callosa Lec. there are about 55 taste-cups under 286the labrum, besides about 10 cells, which may be gustatory structures, situated on either side of a median setose ridge which passes back under the clypeal region.
The taste-cups of the leaf-beetles are fairly numerous, judging from an examination of Diabrotica vittata. The surface of the epipharynx is pilose, but the median region is naked, and on the anterior half bears from 11 to 12 taste-cups, arranged each side of the median line in a rude Y. On each side, at the base of the labial region, are two sensitive fields, each bearing about 25 to 26 taste-cups. More were seen under the clypeus.
In the Neuroptera unmistakable taste-cups are not always present. In Sialis infumata along the median line of the epipharynx and near the front are about 20 scattered gustatory pegs, which are minute, but longer and more acute than usual. In Chauliodes maculatus there are one or two taste-cups under the front edge of the clypeus; others are scattered along the middle from the base of the labrum to the front, but are not arranged in definite order. In Corydalis cornutus no sense-cups, pits, or rods are present. In Chrysopa there are scattered cups armed with a short acute bristle, which are possibly gustatory in function. In Myrmeleon diversum also the presence of sense-pits or of taste-cups is doubtful, though a group of about 12 pits on each side of the clypeal region of the epipharynx, and a few situated at the base of the labral region, may be endowed with the sense of taste. In Mantispa brunnea, however, along the middle of the epipharynx are scattered about 30 unmistakable taste-cups, each bearing a short, fine hair.
In the Mecoptera (Panorpa debilis?) taste-cups, giving rise to a minute hair, occur on the labium in two regions, and also on the maxillæ situated on the stipes near the base of the palpi, and on the lacinia and galea. They are also to be found on the maxillæ of Boreus californicus, but were not detected on the labium.
They were first detected by Reuter in various microlepidoptera, and occur on the “basal spot” of the palpi of many butterflies. In a Tineid moth (Coleophora coruscipennella) we have detected what we suppose to be a group of four taste-pits on the inner side of the basal joint of the labial palpi.
Experimental proof.—No one, says Lubbock, who has ever watched a bee or wasp can entertain the slightest doubt as to their possession of the sense of taste. “Forel mixed morphine and strychnine with some honey, which he offered to his ants. Their antennæ gave them no warning. The smell of the honey attracted them, and they began to feed; but the moment the honey touched their lips they perceived the fraud.”
Will at first fed wasps with sugar, so that they frequently visited it; afterwards he substituted alum for the sugar. Eagerly flying to it, they had scarcely touched it when they drew back from the distasteful substance with the most comical gestures, and cleaned their tongues by frequently running them in and out, repeatedly stroking them with their fore feet. He noticed a great repugnance to quinine in nearly all the insects experimented on. Bees and wasps were observed to have a more delicate gustatory sense than flies, etc., which are more omnivorous in their tastes.
LITERATURE ON THE ORGANS OF TASTE
Meinert, F. Bidrag til de danske myrers naturhistorie. (Kgl. Dansk Vidensk. selsk. skrifter. Kjoebenhavn. Raekke 5 Naturvid. og math. Afd., v, 1861, pp. 273–340.)
Wolff, O. J. B. Das Riechorgan der Biene. (Nova acta d. K. Leop.-Carol. Akad., xxxviii, 1875, pp. 1–251, 8 Taf.)
Joseph, G. Zur Morphologie des Geschmacksorganes bei Insekten. (Amtlicher Bericht der 50. Versammlung deutscher Naturforscher und Aerzte in München, 1877, pp. 227–228.)
Künckel et Gazagnaire. Du siège de la gustation chez les insectes Diptères. Constitution anatomique et physiologique de l’epipharynx et l’hypopharynx. (Comptes-rend. Acad. Sc., Paris, 1881, xcv, pp. 347–350.)
—— Recherches sur l’organisation et le développement des Diptères et en particulaire des Volucelles, i, 1875, ii, 1881, 26 Pls.
Kraepelin, K. Zur Kenntniss der Anatomie und Physiologie des Rüssels von Musca. (Zeitschr. f. wissens. Zool., xxxix, 1883, pp. 683–719, 2 Taf.)
Kirbach, P. Ueber die Mundwerkzeuge der Schmetterlinge. (Zool. Anzeiger, 1883, pp. 553–558, 2 Figs.)
Will, F. Das Geschmacksorgan der Insekten. (Zeitschr. f. wissens. Zool., 1885, xlii, pp. 674–707, 1 Taf.)
Gazagnaire, J. Du siège de la gustation chez les insectes Coléoptères. (Comptes-rend. Acad. Sc., Paris, 1886, cii, pp. 629–632; Ann. Soc. Ent. France, Sér. 6, Bull., pp. 79–80.)
Forel, A. Expériences et remarques critiques sur les sensations des insectes. 2me Part. (Recueil Zool. Suisse, iv, 1887, pp. 161–240.)
Reuter, Enzio. Ueber den “Basalfleck” auf den Palpen der Schmetterlinge. (Zool. Anzeiger, 1888, pp. 500–503.)
—— Ueber die Palpen der Rhopaloceren, etc., 6 Taf. Helsingfors, 1896, pp. 1–578. (Acta Soc. Sc. Fennicæ, xxii, 1896.)
Packard, A. S. On the occurrence of organs, probably of taste, in the epipharynx of the Mecoptera (Panorpa and Boreus). (Psyche, 1889, v, pp. 159–164.)
—— Notes on the epipharynx, and the epipharyngeal organs of taste in mandibulate insects. (Psyche, 1889, v, pp. 193–199, 222–228.)
Also Lubbock’s Senses, etc., of animals, and the writings of Briant, Breithaupt (titles on p. 85).
d. The organs of hearing
Although it has been denied by Forel that insects have the sense of hearing, yet the majority of writers and experimenters agree that insects are not deaf. On general grounds if, as we know, many insects produce sounds, it must follow that they have ears to hear, for there is every reason to suppose that the sounds thus made are, as in other animals, either for attracting the sexes, for a means of communication, or to express the emotions. We will begin by briefly describing the structures now generally supposed to be auditory in 288function, and about which there can be no reasonable doubt, and then consider the more problematical organs, closing with an account of the extremely various means of producing sounds and cries.
The ears or tympanal and chordotonal sense-organs of Orthoptera and other insects.—The ears or tympana of locusts (Acrydiidæ) are situated one on each side, on the basal joint of the abdomen, just behind the first abdominal spiracle. That this is a true ear was first suggested by J. Müller, and his opinion was confirmed by Siebold, Leydig, Hensen, Graber, Schmidt, Lubbock, etc.[49]
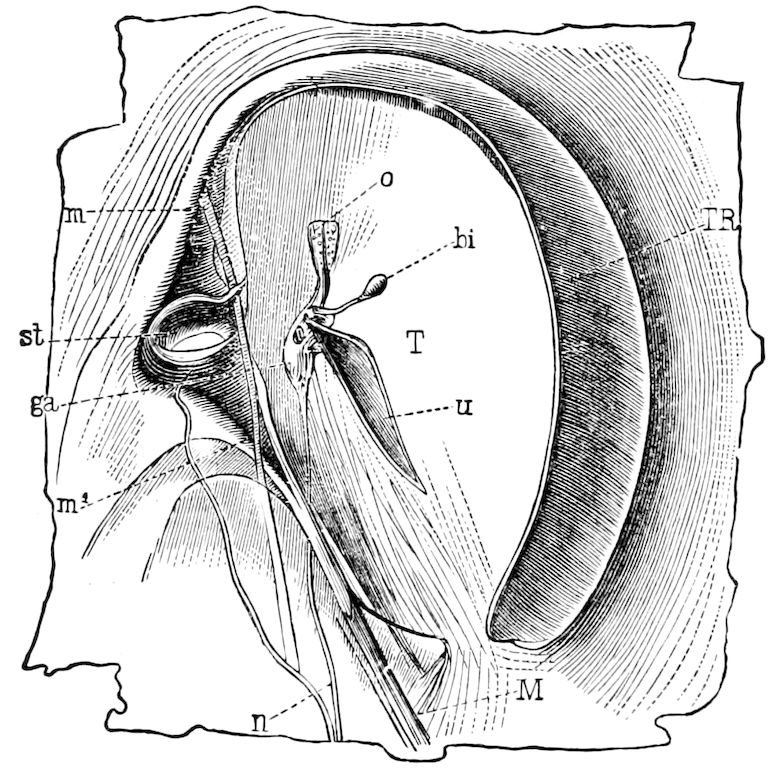
Fig. 290.—Ear of a locust (Caloptenus italicus), seen from the inner side: T, tympanum; TR, its border; o, u, two horn-like processes; bi, pear-shaped vesicle; n, auditory nerve; ga, terminal ganglion; st, stigma; m, opening, and m′, closing, muscle of the same; M, tensor muscle of the tympanum membrane.—After Graber.
The apparatus consists of a tense membrane, the tympanum, surrounded by a horny ring (Fig. 290). “On the internal surface of this membrane are two horn-like processes (o, u), to which is attached an extremely delicate vesicle (bi) filled with a transparent fluid, and representing a membranous labyrinth. This vesicle is in connection with an auditory nerve (n) which arises from the third thoracic 289ganglion, forms a ganglion (ga) upon the tympanum, and terminates in the immediate neighborhood of the labyrinth by a collection of cuneiform, staff-like bodies, with very finely pointed extremities (primitive nerve-fibres?), which are surrounded by loosely aggregated ganglionic globules” (Siebold’s Anatomy of the Invertebrates).
Fig. 291.—Fore tibia of Locusta viridissima. td, cover of the drum; tr, fissure between the drum and its cover.—After Graber, from Lang.
In the green grasshoppers, katydids, and their allies, the ears are situated on the fore tibiæ, where these organs can be found after a careful search (Figs. 291, 292).
The presence of the structure is indicated by the oval disc, the drum, which is a thin tense membrane covering the auditory apparatus of nerves, ganglion cells, and auditory rods beneath.
The tympana, or drums, are not present in all Locustidæ and Gryllidæ, and, as Lubbock states, it is an additional reason for regarding them as auditory organs, that in those species which possess no stridulating organs the tympana are also wanting. In many of the Locustidæ the tympana are covered or protected by a fold of the skin projecting over them. These covered ones are, Graber thinks, derived from the open ones.
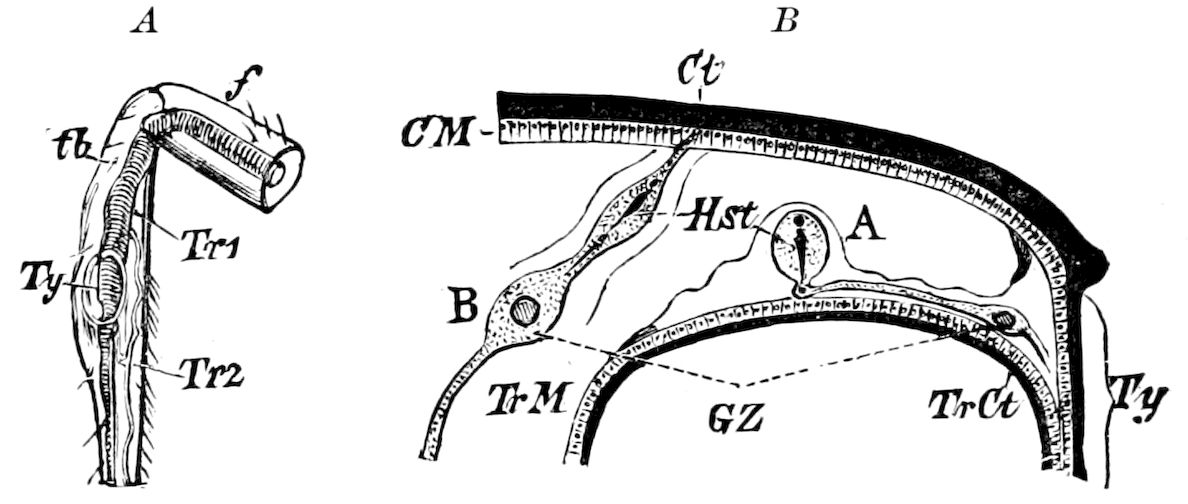
Fig. 292.—A, fore tibia of a European grasshopper (Meconema), containing the ear: Ty, tympanum or outer membrane; Tr 1, Tr 2, tracheæ. B, diagrammatic cross-section through the tibia and ear of the same; Ty, tympanum; Ct, cuticula; CM, hypodermis: A, the auditory organ connecting with the tympanum; B, supra-tympanal auditory organ; GZ, the ganglion-cell belonging to them; Hst, the auditory rod connecting with the ganglion-cells.—After Graber, from Judeich and Nitsche.
On examining the apparatus within the leg under the drum, it is seen to consist of the trachea, the auditory vesicles and rods, ganglion cells, and acoustic nerve. The trachea is greatly modified (Fig. 292, Tr 1). On passing into the tibia the trachea enlarges and divides into two branches, which reunite lower down. The spiracles supplying the air to this enlarged trachea are considerably enlarged, while in the dumb species it is of the normal size. The enlarged 290trachea passes close to the tympanum, which thus has air on both sides of it: the open air on the outer, the air of the trachea on its inner surface. In fact, as Lubbock states, “the trachea acts like the Eustachian tube in our own ear; it maintains an equilibrium of pressure on each side of the tympanum, and enables it freely to transmit the atmospheric vibrations.”
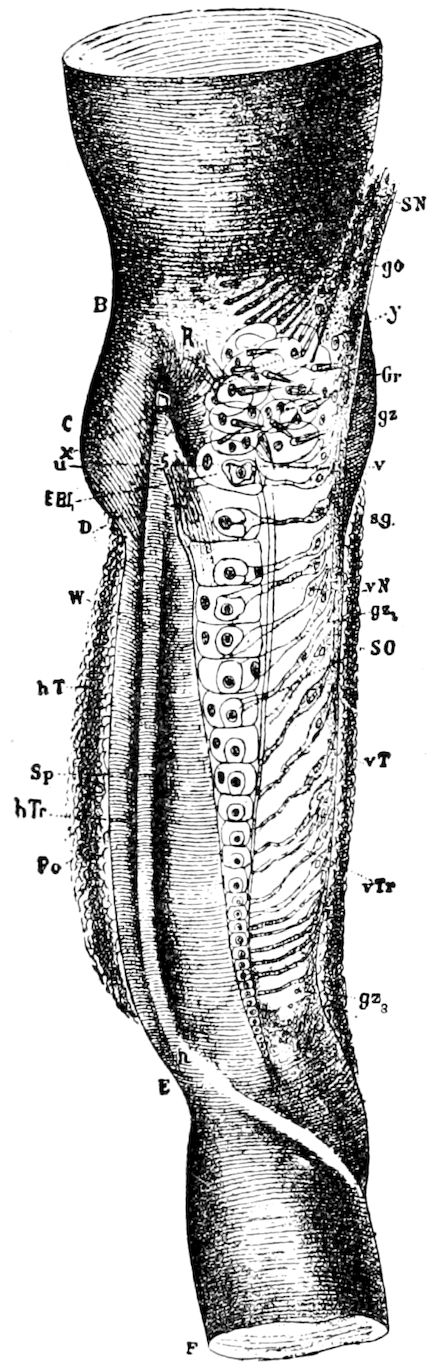
Fig. 293.—The auditory apparatus in the tibia of a grasshopper, showing the tympanal nerve-endings in situ: EBI, terminal vesicles of Siebold’s organ; SN, nerve of the organ of Siebold; Gr, group of vesicles of same; SO, nerve-endings of the same; vT, front tympanum; vTr, front branch of the trachea; hT, hinder tympanum; hTr, hinder branch of the trachea; Sp, space between the tracheæ; go, supra tympanal ganglion; rN, connecting nerve-fibrils between the ganglion cells and the terminal vesicles; R, upper, n-S, lower, root of the transparent covering membrane. (Other lettering not explained by author.)—After Graber.

Fig. 294.—Auditory rod of Gryllus viridissimus: fd, auditory rod; ko, terminal piece.—After Graber, from Lubbock.
These tracheæ, says Graber, though formed on a similar plan, present many variations, corresponding to those of the tympana, and showing that the tympana and the tracheæ stand in intimate connection with one another. For instance, in those species where the tympana are equal, the tracheæ are so likewise; in Gryllotalpa, where the front tympanum only is developed, though both tracheal branches are present, the front one is much larger than the other; and where there is no tympanum, the trachea remains comparatively small, and even in some cases undivided (Lubbock, ex Graber).
The acoustic nerve, which next to the optic is the thickest in the body, divides soon after entering the tibia into two branches, one almost immediately forming a ganglion, the supra-tympanal ganglion, the other passing down to the tympanum, where it expands into an elongated flat ganglion, the organ of Siebold (Fig. 293), and closely applied to the anterior tracheæ.
At the upper part of the ganglion is a group terminating below in a single row of vesicles, the first few of which are approximately equal, but which subsequently diminish regularly in size. Each 291of these vesicles is connected with the nerve by a fibril (Fig. 293, vN), and contains an auditory rod (Fig. 294). They are said by Graber to be brightly refractive, hollow (thus differing from the retinal rods, which are solid), and terminate in a separate end-piece (ko). The rods were first discovered by Siebold, and, as Lubbock remarks, may be regarded as specially characteristic of the acoustic organs of insects.
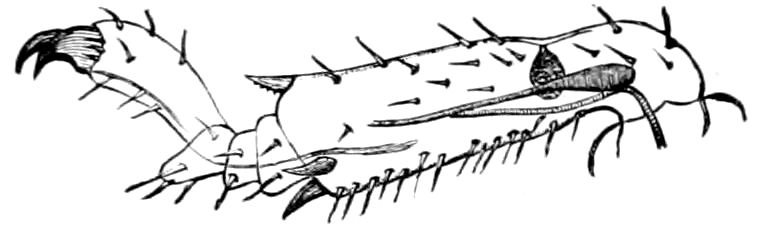
Fig. 295.—Chordotonal organ in nymph of a white ant.—After Müller, from Sharp.
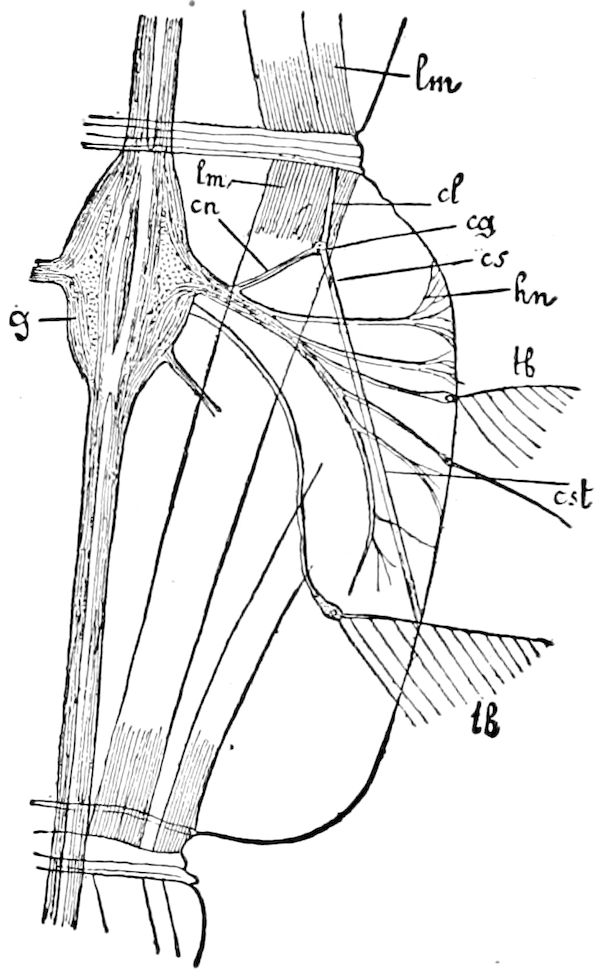
Fig. 296.—Right half of 8th body-segment of Corethra plumicornis: g, ganglion of ventral cord; lm, longitudinal muscle; cn, chordotonal nerve; cl, chordotonal ligament; cg, chordotonal ganglion; cs, rod of chordotonal organ; cst, terminal cord; tb, tactile setæ; hn, out-going fibres of the integumental nerves.—After Graber, from Lang.
As will be seen in Fig. 293, at the upper part of the tibial organ of Ephippigera there is a group of cells, and below them a single row of cells gradually diminishing in size from above downwards. “One cannot but ask oneself,” says Lubbock, “whether the gradually diminishing size of the cells in the organ of Siebold may not have reference to the perception of different notes, as is the case with the series of diminishing arches in the organ of Corti of our own ears.”
These organs were supposed to be restricted to the Orthoptera, but in 1877 Lubbock discovered what seems to resemble the supra-tympanal auditory organ of Orthoptera in the tibia of the yellow ant (Lasius flavus). Graber confirmed Lubbock’s account, and also discovered these organs in the tibia of a Perlid (Isopteryx apicalis), and Fritz Müller has detected them in the fore tibiæ of the nymph of Calotermes rugosus (Fig. 295). To these structures Graber gave the name of chordotonal organs.
He has also detected these organs in all the legs of other insects (Trichoptera, Pediculidæ), and auditory rods have been discovered in the antennæ of Dyticus and of Telephorus by Hicks, Leydig, and Graber. Graber classifies the chordotonal organs into truncal and membral. In Coleoptera and Trichoptera they may occur on several joints of the leg; others are more localized,—thus he distinguishes femoral (Pediculidæ), tibial (Orthoptera, Perlidæ, Formicidæ), and tarsal organs (Coleoptera).
A type of chordotonal organ, observed in the body-segments of the larvæ of several insects by Leydig, Weismann, Graber, Grobben, and Bolles Lee, is to be seen in the transparent larva of Corethra (Fig. 296), where the auditory organ extends to the skin. It contains at the point cs two or three auditory 292rods. In the opposite direction a fine ligament (cl) passes from cg to the skin; in this way the auditory organ is suspended in a certain state of tension, and is favorably situated to receive even very fine vibrations. A similar apparatus has been detected in the larva of Ptychoptera.
Antennal auditory hairs.—It is not at all improbable that the antennæ of different insects contain auditory as well as olfactory structures. Lubbock has suggested that the singular organs which have only been found in the antennæ of ants and certain bees, and to which he gives the name of “Hicks’ bottles” (Fig. 281), may act as microscopic stethoscopes, while Leydig also regards them as chordotonal organs.
That, however, some of the antennal hairs of the mosquito, as first suggested by Johnson and afterwards proved experimentally by Mayer, are auditory, seems well established. Fastening a male mosquito down on a glass slide, Mayer then sounded a series of tuning-forks. With an Ut4 fork of 512 vibrations per second, some of the hairs were seen to vibrate vigorously, while others remained comparatively at rest. The lower (Ut3) and higher (Ut5) harmonics of Ut4 also caused more vibration than any intermediate notes. These hairs, then, are specially tuned so as to respond to vibrations numbering 512 per second. Other hairs vibrated to other notes, extending through the middle and next higher octave of the piano.
Mayer then made large wooden models of these hairs, the one corresponding to the Ut3 hair being about a metre in length, and on counting the number of vibrations they made when they were clamped at one end and then drawn on one side, he found that it “coincided with the ratio existing between the numbers of vibrations of the forks to which covibrated the fibrils,” or hairs. It should be observed that the song of the female mosquito corresponds nearly to this note, and would consequently set the hairs in vibration. Mayer observed that the song of the female vibrates the hairs of one of the antennæ more forcibly than those of the other. Those auditory hairs are most affected which are at right angles to the direction from which the sound comes. Hence from the position of the antennæ and the hairs a sound will be loudest or most intense if it is directly in front of the head. If, then, the song of the female affects one antenna more than another, the male turns his head until the two antennæ are equally affected, and is thus able to fly straight towards the female. From his experiments Mayer found that the male can thus guide himself to within 5° of the direction of the female. Hence he concludes that “these insects must have the faculty 293of the perception of the direction of sound more highly developed than in any other class of animals.” (Also see Child’s work.)
Special sense-organs in the wings and halteres.—Organs of a special sense, which Hicks supposed to be those of smell, were found by him near or at the base of the wings of Diptera, Coleoptera, and less perfect ones in Lepidoptera, Neuroptera, and Orthoptera, with a trace of them in Hemiptera; but these were considered by Leydig to be auditory organs, since he found the nerves to end in club-shaped rods, like those of Orthoptera.
Hicks found, as to the halteres and their sense-organs, that the nerve in the halter is the largest in the insect, except the optic nerve; and that at the base of the halteres is a number of vesicles arranged in four groups, to each of which the nerve sends a branch. Afterwards Bolles Lee discovered that the vesicles, undoubtedly perforated, contain a minute hair, those of the upper groups being protected by hoods of chitin. He regarded them as olfactory organs, while Lubbock seems inclined to consider them as auditory structures. Graber also regards the vesicles of Hicks as chordotonal organs.
In his elaborate account of the balancers, Weinland concludes that the organs of sense of varying structure occurring at the base of these appendages allow the perception of movements which the halteres perform and which enable the fly to steer or direct its course. The halteres can thus cause differences in the direction of the flight of a fly in the vertical plane. If the balancers act unequally, there is a change in direction.
e. The sounds of insects
Insects have no true voice; but sounds of different intensity, shrill cries, and other noises are produced mechanically by insects, either being love-songs to attract the sexes, to give signals, to communicate intelligence, or perhaps to express the emotions. The loud, shrill cry of the Cicada, or chirp of the cricket, is evidently a love-call, and results in the mating of individuals of separate broods more or less widely scattered, thus preventing too close interbreeding.
The simplest means of making a noise is that of the death-watch (Anobium), which strikes or taps on the wall with its head or abdomen. Longicorn beetles make a sharp sound by the friction of the mesoscutellum against the edge of the prothoracic cavity, the head being alternately raised and lowered, Burying-beetles (Necrophorus) rub the abdomen against the hinder edges of the elytra. Weevils make a loud noise by rapidly rubbing the tips of the abdomen on the ends of the elytra.
Landois offers the following summary of the kinds of noises produced by beetles:
- 1.
- Tapping sounds (Bostrycinæ, Anobium).
- 2.
- Grating sounds (Elateridæ).
294
- 3.
- Friction without special rasping organs (Euchirus longimanus).
- 4.
- Rasping sounds produced by friction:
- a. Rubbing of the pronotum on the mesonotum (Cerambycidæ except Spondyli and Prionus).
- b. Friction of the prosternum on the mesosternum (Omaloplia brunnea).
- c. Elytra with a rasp at the end (Curculionidæ, Dyticidæ, Pelobius).
- d. With a coxal rasp (Geotrupes, Ceratophyus). The male of Ateuchus stridulates to encourage the female in her work, and from distress when she is removed. (Darwin.)
- e. Friction of the edge of the elytra against the femur (Chiasognathus grantii).
- f. Pygidium with two rasps in the middle (Crioceris, Lema, Copris, Oryctes, Necrophorus, Tenebrionidæ).
- g. Abdomen with a grating ridge and four grating plates (Trox).
- h. Abdomen with two toothed ridges rubbing on a rasp on edge of wing-cover (Elaphus, Blethisa, Cychrus).
- i. Rubbing the elytra on a rasp on the hind wings (Pelobius hermanni).
- j. Friction of the wing against the abdominal segments (Melolontha fullo).
Mutilla makes a rather sharp noise by rubbing one abdominal segment against another. Ants (Ponera) have a stridulating apparatus, and other genera numerous (20) ridges between the segments.
Even certain moths and butterflies emit a rasping or crackling noise. The death’s-head moth and other sphinges cause it by rubbing the palpi against the base of the proboscis. These and certain butterflies are provided with parallel ridges forming a rasp on the “basal spot” of the inner side of the basal joint of each palpus (Reuter). A South American butterfly (Ageronia feronia) can be heard for several yards as it flies with a crackling sound. Hampson finds that the cause of the clicking sound is due to a pair of strong chitinous hooks attached to the thorax, against which play the spatulate ends of a pair of hooks attached to the fore wings. An Australian moth (Hecatesia) flies with a whizzing sound; Vanessa is said to be sonorous.
The males of Orthoptera produce their shrill cries or chirping noises, 1, by rubbing the thighs against the sides of the body (Acrydiidæ); 2, by the friction of the base of the fore wings on each other (Locustidæ); 3, by rubbing the base of the upper on the base of the hinder or under pair (Gryllidæ), in the two last there being a shrilling apparatus consisting of a file on the hind wings, which rubs on a resonant surface on the fore wings. The females are not invariably dumb, both sexes of the European Ephippigera being able to faintly stridulate. Corixa also produces shrill chirping notes. (Carpenter.)
Certain insects also hum, and have what may perhaps be called a voice. The cockchafer, besides humming with the wings, produces a sound almost like a voice. In the large trachea, just behind each spiracle, is a chitinous process, which is thrown into vibrations by the air during respiration, and thus produces a humming noise. (Lubbock.) Such is also the case with flies, the mosquito, dragon-flies, and bees. In flies and dragon-flies the “voice” is caused by the air issuing from the thoracic spiracles; while in the humble-bee the abdominal spiracles are also musical. The sound made by the spiracles bears no relation to that caused by the wings. Landois 295tells us that the wing-tone of the honey-bee is A′; its voice, however, is an octave higher, and often goes to B″ and C″.
The sounds produced by the wings are constant in each species, except where, as in Bombus, there are individuals of different sizes; in these the larger ones generally give a higher note. Thus the comparatively small male of Bombus terrestris hums on A′, while the large female hums an entire octave higher.
From the note produced the rapidity of the vibrations can be calculated. For example, the house-fly, which produces the sound of F, vibrates its wings 21,120 times in a minute, or 335 times in a second; and the bee, which makes a sound of A′, as many as 26,400 times, or 440 times in a second. On the contrary, a tired bee hums on E′, and therefore, according to theory, vibrates its wings only 330 times in a second. Marey has confirmed these numbers graphically, and found by experiment that the fly actually makes 330 strokes in a second. (Lubbock.)
A different kind of musical apparatus is that of the cicada, which has been elaborately described by Graber. The shrill, piercing notes issue from a pair of organs on the under side of the base of the abdomen of the male, these acting somewhat as two kettle-drums, the membrane covering the depressions being rapidly vibrated.
LITERATURE ON THE ORGANS OF HEARING
a. The auditory organs
Siebold, C. Th. E. von. Ueber das Stimm- und Gehörorgan der Orthopteren. (Archiv f. Naturgesch., 1844, x, pp. 52–81.)
Johnston, Christopher. Auditory apparatus of the culex mosquito. (Quart. Journ. Micr. Soc., 1855, iii, pp. 97–102, 1 Fig.)
Hicks, Braxton. On a new organ in insects. (Journ. Linn. Soc. Zool., London, 1857, pp. 130–140, 1 Pl.)
—— Further remarks on the organ found on the bases of the halteres and wings of insects. (Trans. Linn. Soc., London, 1857, xxii, pp. 141–145, 2 Pls.)
Hensen, V. Ueber das Gehörorgan von Locusta. (Zeitschr. f. wissens. Zool., xvi, 1866, pp. 190–207.)
Graber, V. Bemerkungen über die Gehör- und Stimmorgane der Heuschrecken und Cicaden (Wiener Sitzungsber. Math.-natur-wiss. Cl., lxvi, 1 Abt., 1872, pp. 205–213, 2 Figs.)
—— Die tympanalen Sinnesapparate, der Orthopteren. (Denkschr. d. k. Akad. d. wissens. Wien, xxxvi, 1876, 2 Abt., pp. 1–140, 10 Taf.)
—— Die abdominalen Tympanalorgane der Cicaden und Gryllodeen. (Ibid., 1870, xxxvi, pp. 273–290, 2 Taf.)
—— Ueber neue, otocystenartige Sinnesorgane der Insekten. (Archiv f. mikroskop. Anat., 1878, pp. 35–57, 2 Taf.)
—— Die chordotonalen Sinnesorgane und das Gehör der Insekten. (Archiv f. mikroskop. Anat., 1882, xx, pp. 506–640; 1883, xxi, pp. 65–145, Taf.)
296Mayer, Alfred Marshall. Researches in Acoustics No. 5. 3. Experiments on the supposed auditory apparatus of the culex mosquito. (Amer. Jour. Sc. and Arts, Ser. 3, viii, 1874, pp. 81–103; also Amer. Naturalist, viii, pp. 577–592.)
Schmidt, Oscar. Die Gehörorgane der Heuschrecken. (Archiv f. mikroskop. Anat., xi, 1875, pp. 195–215, 3 Taf.)
Ranke, J. Beiträge zu der Lehre von den Uebergangssinnesorganen, das Gehörorgan der Acridier und das Sehorgan der Hirudineen. (Zeitschr. f. wissens. Zool., xxv, 1875, pp. 143–164, 1 Taf.)
Lee, A. Bolles. Les balanciers des Diptères, leurs organes sensifères et leur histologie. (Recueil Zool. Suisse, ii, 1885, pp. 363–392, 1 Pl.)
—— Bemerkungen über der feineren Bau der Chordotonalorgane. (Archiv f. mikroskop. Anat., 1883, xxiii, pp. 133–140, 1 Taf.)
—— Les organes chordotonaux des Diptères et la méthode du chlorure d’or. (Observations critiques.) (Recueil Zool. Suisse, 1884, ii, pp. 685–689, 1 Pl.)
Weinland, E. Ueber die Schwinger (Halteren) der Dipteren (Zeitschr. f. wissens. Zool., 1890, li, pp. 55–166, 5 Taf.)
Adelung, N. v. Beiträge zur Kenntnis des tibialen Gehörapparates der Locustiden. Inaug. Diss., Leipzig, 1892, 2 Taf.
Child, Ch. M. Ein bisher wenig beachtetes antennales Sinnesorgan der Insekten, mit besonderer Berücksichtigung der Culiciden und Chironomiden. (Zeitschr. f. wissens. Zool., lviii, 1894, pp. 475–528, 2 Taf.; also, Zool. Anzeiger, xvii Jahrg., pp. 35–38, and in Annals and Mag. Nat. Hist., 1894 (6), xiii, pp. 372–374).
Also the writings of J. Müller, Kirby and Spence, Burmeister, Gilbert White, Westwood, Guilding, Meinert, Paasch, Leydig, Viallanes, Minot, Forel, Mayer, Darwin (Descent of Man, i, ch. x.), F. Müller, Lubbock (Senses of animals), Westring, Köppen, Bates, Vom Rath, Peckham, Jourdan, Nagel, etc.
b. The sounds made by insects
Scudder, S. H. Notes on the stridulation of grasshoppers. (Proc. Bost. Soc. Nat. Hist., xi, 1868, pp. 306–313 and 316.)
—— The songs of the grasshoppers. (Amer. Naturalist, ii, 1868, pp. 113–120, 5 Figs.)
Riley, C. V. The song notes of the periodical Cicada. (Proc. Amer. Assoc. Adv. Science, xxxiv, 1885, pp. 330–332; also in Kansas City Rev., October, 1885, pp. 173–175.)
Swinton, A. H. (Ent. Month. Mag., 1877.) Sound produced in Ageronia by a modification of the hook and bristle of the wings.
Hampson, G. F. On stridulation in certain Lepidoptera, etc. (Proc. Zool. Soc. London, 1892, ii, pp. 188–193, Fig; also Psyche, vi, p. 491, 1 Fig.)
With the writings of Landois, Lubbock, Graber, Kolbe, Carpenter (Nat. Science), Bruyant, and others.
THE DIGESTIVE CANAL AND ITS APPENDAGES
Fig. 297.—Transverse section through an abdominal segment of larva of Megalopyge crispata, showing the relations of the digestive canal to the other organs: int, hind-intestine, with its mucous or epithelial layer (ep), and ml its outer or muscular layer; ng, ventral ganglion; ht, heart; mp, urinary tubes; f, fat-body; sc, thickened portion of the hypodermis (hy) containing the setigenous cells; m, muscles; m′, a pair of retractor muscles inserted near the base of the lateral glandular process (lgp); cut, cuticula; l, legs. Also compare Figs. 142–144 and 234.
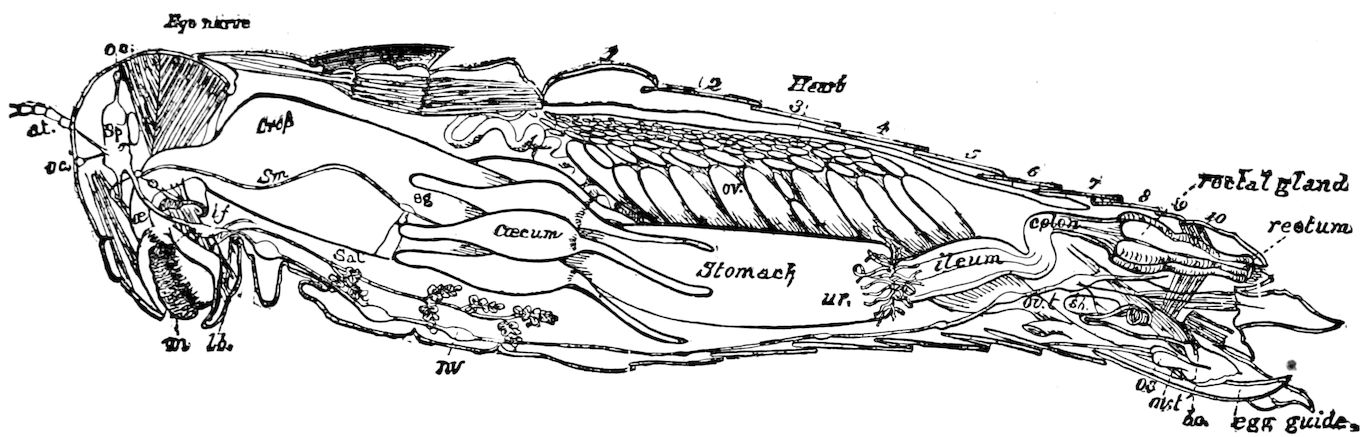
Fig. 298.299
The alimentary or digestive canal of insects is a more or less
straight tube, which connects the mouth and anus, the latter invariably
situated in the last segment of the body, under the last
tergite or suranal plate. It lies directly over the ventral nervous
cord and under the dorsal vessel, passing through the middle of the
body (Fig. 297). It is loosely held in place by delicate retractor
muscles (retractores ventriculi, found by Lyonet in the larvæ of
Lepidoptera, and occurring in those of Diptera), but is principally
supported by exceedingly numerous branches of the main tracheæ.
Fig. 298.—Internal anatomy of Melanoplus femur-rubrum: at, antenna and nerve leading
to it from the “brain” or supraœsophageal ganglion (sp); oc, ocelli, anterior and vertical ones,
with ocellar nerves leading
to them from the brain; œ, œsophagus; m, mouth; lb, labium or under
lip; if, infraœsophageal ganglion, sending three pairs of nerves to the mandibles, maxillæ, and
labium respectively (not clearly shown in the engraving); sm, sympathetic or vagus nerve, starting
from a ganglion resting above the œsophagus, and connecting with another ganglion (sg) near the
hinder end of the crop; sal, salivary glands (the termination of the salivary duct not clearly
shown by the engraver); nv, nervous cord and ganglia; ov, ovary; ur, origin of urinary tubes;
ovt, oviduct; sb, sebaceous gland; bc, bursa copulatrix; ovt, site of opening of the oviduct (the
left oviduct cut away); 1–10, abdominal segments. The other organs labelled in full.—Drawn from
his original dissections by Mr. Edward Burgess.
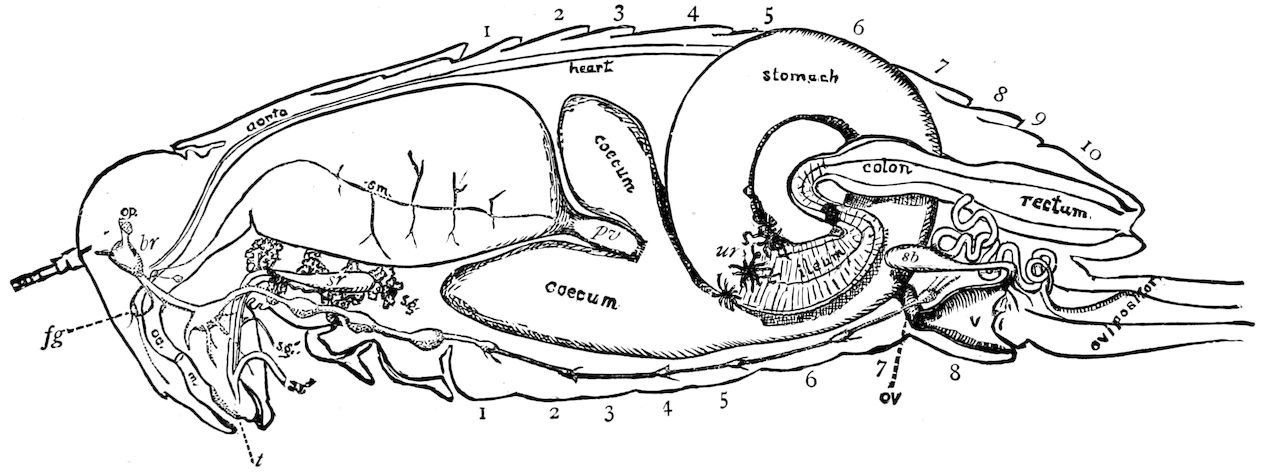
Fig. 299.—Digestive canal of Anabrus: m, mouth: œ, œsophagus; sm, the sympathetic nerve passing along the crop; t, tongue; fg, frontal ganglion; br, brain, the nervous cord passing backward from it; sr, salivary reservoir; sg, salivary gland; pv, proventriculus; ur, origin of urinary tubes; sb, sebaceous gland; 1–10, the ten abdominal segments.—Burgess del.
It is in the higher adult insects differentiated into the mouth and pharynx, the œsophagus or gullet, supplementary to which is the crop (ingluvies) or “sucking stomach” of Lepidoptera, Diptera, and Hymenoptera; the proventriculus or gizzard; the ventriculus, “chyle-stomach,” or, more properly, mid-intestine, and the hind-intestine, which is divided into the ileum, or short intestine, the long intestine, often slender and coiled, with the colon and the rectum. Morphologically, however, the digestive or enteric canal is divided into three primary divisions, which are indicated in the embryo insect; i.e., the fore-intestine (stomodæum of the embryo), mid-intestine or “chyle-stomach,” and hind-intestine or proctodæum (Fig. 300). The three primary regions, with their differentiations, may be tabulated thus:—
| Fore-intestine (Stomodæum). | Mouth and pharynx. |
| Pumping apparatus of Hemiptera, Lepidoptera, and Diptera. | |
| Œsophagus. | |
| Crop or ingluvies, food reservoir, or “sucking stomach.” | |
| Proventriculus. | |
| Mid-intestine (Mesenteron). | Mid-intestine, “chylific stomach,” or ventriculus (with cœcal glands). |
| Hind-intestine (Proctodæum). | Ileum, or short intestine (with the urinary tubes). |
| Long intestine. | |
| Colon. | |
| Rectum (with rectal glands). | |
| Anus (with anal glands). |
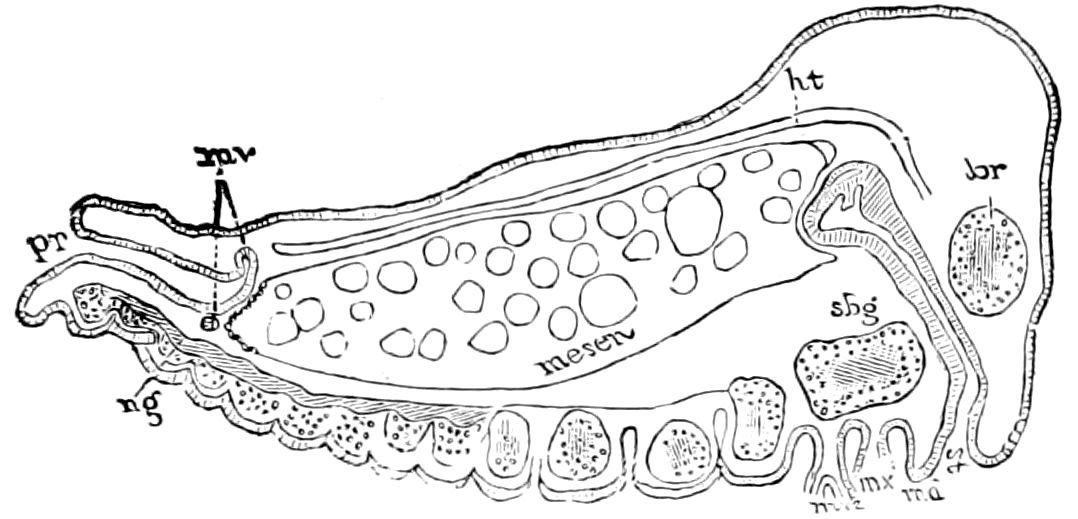
Fig. 300.—The three primary divisions of the alimentary canal of an embryonic orthopterous insect: br, brain; sbg, subœsophageal ganglion; ng, nervous cord; st, stomodæum; pr, proctodæum; mv, malphigian tubes; mesen, mid-intestine; ht, heart; md, mandibles; mx, mx′, 1st and 2d maxillæ.—After Ayers, with some changes.
The appendages of the alimentary canal are: (1) the salivary and poison glands, which arise from the stomodæum in embryonic life; 300(2) while to the chylific stomach a single pair of cœcal appendages (Orthoptera and larval Diptera, e.g. Sciara), or many cœca may be appended; (3) the urinary tubes, also the rectal glands and the paired anal glands. In a Hemipter (Pyrrhocoris apterus) appendages arise from the intestine in front of the origin of the urinary tubes. In certain insects a single cœcal appendage (Nepa, Dyticus, Silpha, Necrophorus, and the Lepidoptera) arises from the proctodæum.
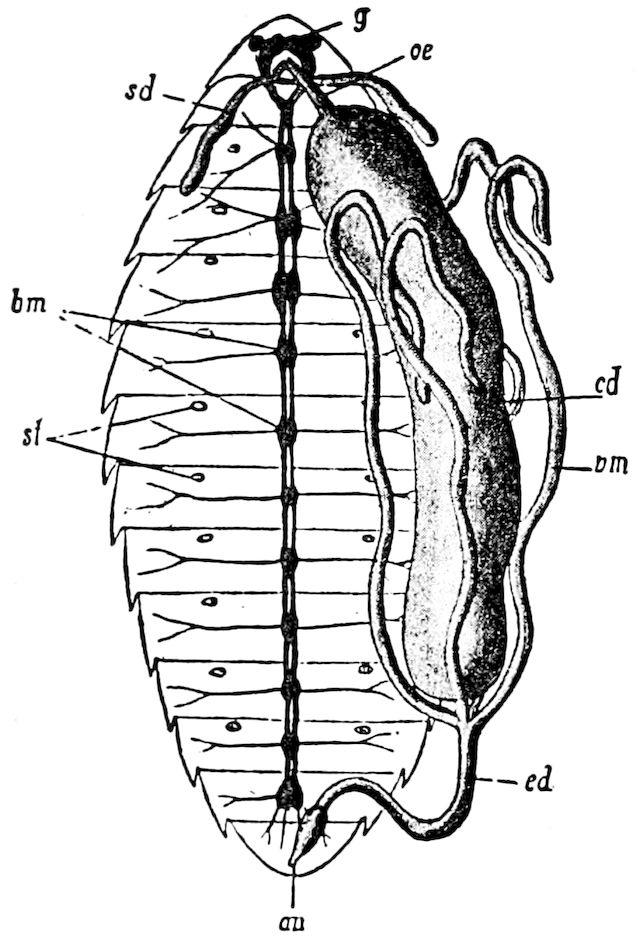
Fig. 301—Larva of honey-bee: g, brain; bm, ventral nervous cord; œ, œsophagus; sd, spinning-gland; cd, mid-intestine or chyle-stomach; ed, hind-intestine, not yet connected with the mid-intestine; vm, urinary tube; an, anus; st, stigmata.—After Leuckart, from Lang.
In certain larval insects, as those of the Proctotrypidæ (first larval stage), the higher Hymenoptera (ichneumons, ants, wasps, and bees, Fig. 301), in the Campodea-like larvæ of the Meloidæ and Stylopidæ, the larva of the ant-lion (Myrmecoleo), and those of Diptera pupipara (Melophagus), the embryonic condition of the separation of the proctodæum and mid-gut (mesenteron) persists, the stomach ending in a blind sac; in such cases the intestine, together with the urinary tubes, is entirely secretory.
The anus is wanting in the larva of the ant-lion, as also in the wasps (in which there is a rudimentary colon) and in freshly hatched bees, though it becomes perfectly formed in the fully grown larvæ (Newport, art. Insecta, p. 967, and H. Müller).
In the larvæ of lamellicorn Coleoptera (Melolontha vulgaris) the digestive tube is nearly as simple as in bees, though there is a large colon, which at its beginning forms an immense cœcum, and has also one anal aperture (Newport).
The length and shape of the digestive canal is dependent on the nature of the food and also on the mode of life, especially the ease or difficulty with which the food is digested.
301Newport, while stating that the length of the alimentary canal in larvæ is not in general indicatory of the habits of the species, makes this qualification after describing the digestive canal of Calandra as compared with that of Calosoma: “The length and complication of the intestines, therefore, appear to have some reference to the quality of the food to be digested, since it is well known that the food of these latter insects (weevils) is of difficult assimilation, being as it is chiefly the hard ligneous fibres of vegetable matter; but they cannot be received as always indicatory of a carnivorous [or] vegetable feeder, since, as above remarked, the length of the canal is considerable in one entirely carnivorous larva, while it is much shorter in some herbivorous, and particularly in pollenivorous larvæ, as in the Melolontha and the apodal Hymenoptera.”
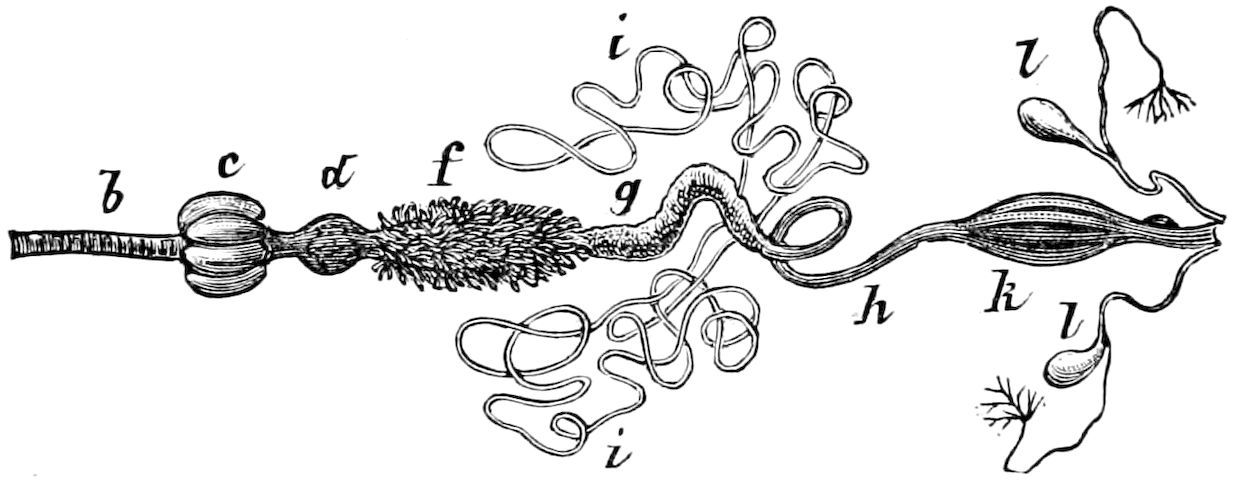
Fig. 302.—Digestive canal of a carabid beetle: b, œsophagus; c, crop; d, proventriculus; f, mid-intestine, or “chyle-stomach,” with its cœeca; g, posterior division of the stomach; i, the two pairs of urinary tubes; h, intestine; k, rectum; l, anal glands.—After Dufour, from Judeich and Nitsche.
Newport also contends that the length of the alimentary canal is not more indicative in the perfect insect of the carnivorous or phytophagous habits of the species than in the larva. It is nearly as long (being from two to three times the length of the whole body), and is more complicated, in the rapacious Carabidæ (Fig. 302) than in the honey-sipping Lepidoptera, whose food is entirely liquid. Referring to the digestive canal of Cicindelidæ, which is scarcely longer than the body, he claims that “we cannot admit that the length of the digestive organs, and the existence of a gizzard and gastric vessels, are indicatory of predacity of habits in the insect, because a similar conformation of parts exists often in strictly vegetable feeders. The existence and length of these parts seem rather to refer to the comparative digestibility of the food than to its animal or vegetable nature.” Newport then refers to the digestive canal of Forficulidæ (in which the gizzard is present, the canal, however, passing in an almost direct line through the body, making but one slight convolution), “a farther proof that the length of the canal must not be taken as a criterion whereby to judge of the habits of a species.” He adds this will apply equally well to the omnivorous Gryllidæ, in which there exists a short alimentary canal, but a gizzard of more complicated structure than that of the Dytiscidæ.
In larval insects and others (Synaptera, Orthoptera, etc.), in which the digestive canal is simplest, it is scarcely longer than the body, and passes through it as a straight tube.
In the caterpillar, which is a voracious and constant feeder, the digestive canal is a large straight tube, not clearly differentiated into fore-stomach, stomach, and intestine; but in the imago, which only 302takes a little liquid food, it is slender, delicate, and highly differentiated. In the larva the mid-gut forms the largest part of the canal; in the imago, the intestine becomes very long and coiled into numerous turns; at the same time the food-reservoir (the “sucking stomach”) develops, and the excretory tubes are longer.
a. The digestive canal
Fig. 303.—Interior view of the bottom of the head of Danais archippus, the top having been cut away, showing, in the middle, the pharyngeal sac with its five muscles: the frontal (f.m), dorsal pair (d.m), and the lateral pair (l.m); cl, clypeus; cor, cornea; œ, œsophagus; p.m, one of the large muscles which move the labial palp.—After Burgess.
It will greatly simplify our conception of the anatomy of the digestive canal if we take into account its mode of origin in the embryo, bearing in mind the fact that during the gastrula condition the ectoderm is invaginated at each pole to form the primitive mouth and fore-gut (stomodæum) and hind-gut (proctodæum). The cells of the ectoderm secrete a chitinous lining (intima), which forms the continuation of the outer chitinous crust, and thus the lining of each end of the digestive canal is cast whenever the insect molts; while the mid-intestine (mesenteron), arising independently of the rest of the canal much later in embryonic life from the mesoderm, is not the result of any invagination, being directly derived from the mesoderm, and is not lined with chitin.
The mouth, or oral cavity, and pharynx.—This is the beginning of the alimentary bounded above by the clypeus, and labrum, with the epipharynx, and below by the hypopharynx, or tongue, as well as the labium. Into it pour the secretion of the salivary glands, which passes out through an opening at the base of the tongue or hypopharynx. On each side of the mouth are the mandibles and first maxillæ.
The sucking or pharyngeal pump.—This organ has been observed by Graber in flies and Hemiptera, but the fullest account is that by Burgess, who was the first to discover it in Lepidoptera. In the milk-weed butterfly (Danais archippus) the canal traversing the proboscis 303opens into a pharynx enclosed in a muscular sac (Figs. 303, 304, and 310).
The pharyngeal sac, says Burgess, serves as a pumping organ to suck the liquid food through the proboscis and to force it backwards into the digestive canal.
Fig. 304.—Longitudinal section through the head of Danais, showing the interior of the left half: mx, left maxilla, whose canal leads into the pharynx; hph, floor of the latter, showing some of the taste-papillæ; oe, œsophagus; ep, epipharyngeal valve; sd, salivary duct; d.m, f.m, and cl, as in Fig. 302.—After Burgess.
Meinert (“Trophi Dipterorum”) has made elaborate dissections of the mouth and its armature, including the pharynx of several types of Diptera, with its musculature. He describes the pharynx as the principal, and in most Diptera, as the only part of the pump (antlia), and says: “By the muscles of the pump (musculis antliæ) the superior lamina of the pharynx is varied that the space between the two laminæ may be increased, and the liquid is thus led through the siphon formed by the mouth-parts into the mouth” (Fig. 81).
The œsophagus.—This is a simple tube, largest in those insects feeding on solid, usually vegetable, food, and smallest in those living on liquid food. It usually curves upwards and backwards, passing directly under the brain, and merges into the crop or proventriculus either at the back part of the head or in the thorax, its length being very variable. Its inner walls longitudinally are folded and lined with chitin.
According to Newport, in the œsophagus of the Gryllidæ, of the two layers of the mucous lining the second is distinctly glandular and secretory, and in it there are many thousands of very minute granular glandular bodies, which probably secrete the “molasses” or repellent fluid often ejected by these and other insects when captured.
The crop or ingluvies.—This, when present, is an enlargement of the end of the œsophagus, and lined internally with a muscular coat. It is very large in locusts (Fig. 298), Anabrus (Fig. 299), and other Orthoptera (the Phasmidæ excepted), in the Dermaptera, and most 304adult Coleoptera. A crop-like dilatation in front of a spherical gizzard is also present in the Synaptera (Poduridæ and Lepismidæ), as well as in the Mallophaga (Nirmidæ).
Fig. 305.—Digestive canal of Calandra: H, pear-shaped œsophagus; I, crop; K, gastric cœca L, ilium; MN, colon; P, urinary tubes.—After Newport.
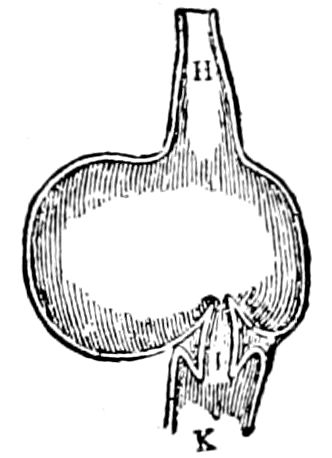
Fig. 306.—Section of the crop (H), gizzard (I), and stomach (K) of Athalia.-After Newport.
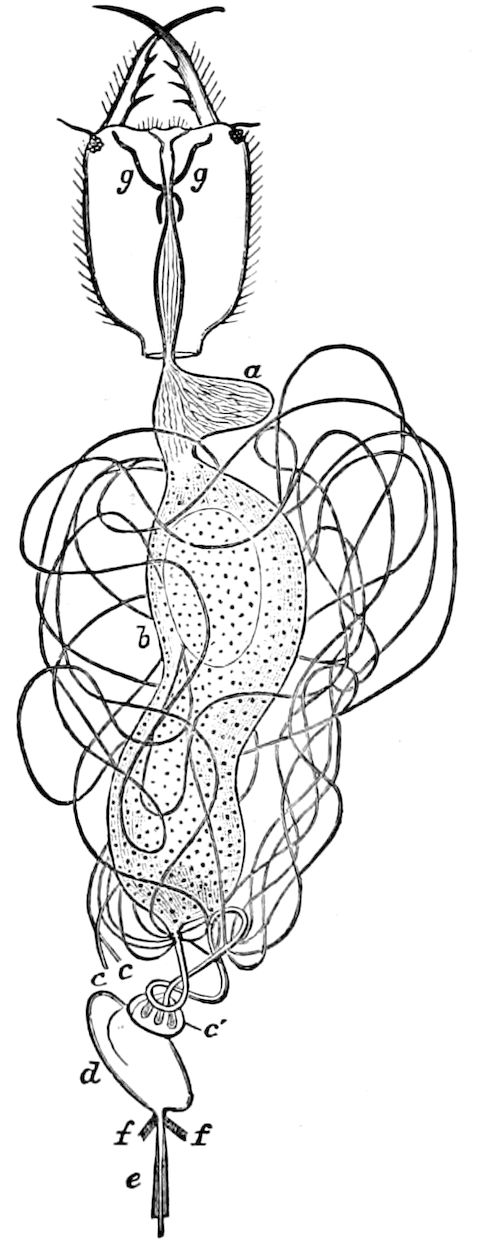
Fig. 307.—Upper side of head and digestive canal of Myrmeleon larva: a, crop; b, “stomach”; c, free ends of two urinary tubes; c′, common origin of other six tubes; d, cœcum; e, spinneret; ff, muscles for protruding its sheath; gg, maxillary glands.—After Meinert, from Sharp.
In the larvæ of weevils (Calandra sommeri) there is a crop (Fig. 305), but not in the larva of Calosoma; also, according to Beauregard, in the pollen-eating beetles Zonitis, Sitaris, and Malabris it is wanting, while in Meloe it is highly developed (Kolbe).
The crop forms a lateral dilatation of the end of the œsophagus in the larvæ of weevils and of saw-flies (Athalia centifoliæ, Fig. 306).
305The “sucking stomach” or food-reservoir.—This is a thin muscular pouch connected by a slender neck with the end of the œsophagus or the crop, when the latter is present. There is no such organ in Orthoptera, except in Gryllotalpa. It is wanting in the Odonata and in the Plectoptera (Ephemeridæ); in Platyptera (Perlidæ and Termitidæ), in Trichoptera, and in Mecoptera (Panorpidæ). In most adult Neuroptera (Myrmeleonidæ, Hemerobiidæ, and Sialidæ), but not in Rhaphidiidæ, the long œsophagus is dilated posteriorly into a kind of pouch or crop, and besides there is often a long “food-reservoir” arising on one of its sides, that of Myrmeleon (Fig. 307) and Hemerobius being on the right side.
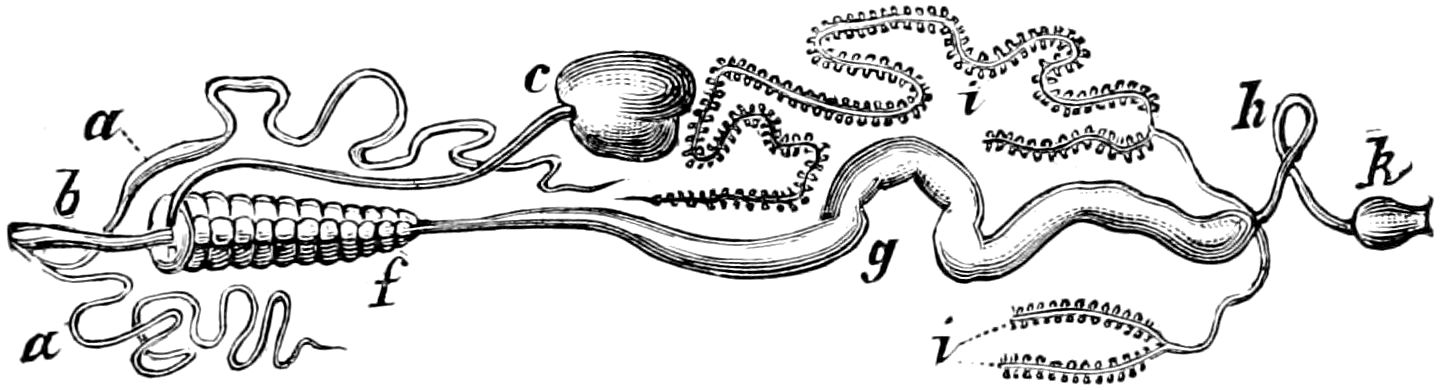
Fig. 308.—Digestive canal of Sarcophaga carnaria: a, salivary gland; b, œsophagus; c, food reservoir; f-g, stomach; h, intestine; i, urinary tubes; k, rectum.—From Judeich and Nitsche.
A true food-reservoir is present in most Diptera (Fig. 308) as well as in the larvæ of the Muscidæ, but according to Dufour it is wanting in some Asilidæ and in Diptera pupipara, and according to Brauer in the Œstridæ. The food-reservoir in Diptera is always situated on the left side of the digestive canal; there is usually a long neck or canal, while the reservoir is either oval or more usually bilobed, and often each lobe is itself curiously lobed.
In Lepidoptera (Figs. 309, 310) the so-called “sucking stomach” is, as Graber has proved, simply a reservoir for the temporary reception of food; though generally found to contain nothing but air, Newport has observed that in flies it is filled with food after feeding. He has found this to be the case in the flesh fly, and in Eristalis he has found it “partially filled with yellow pollen from the flowers of the ragwort upon which the insect was captured,” the pollen grains also occurring in the canal leading to the bag, in the gullet, and in the stomach itself. Graber has further proved by feeding flies with a colored sweet fluid that this sac is only a food-receptacle. As he says: “It can be seen filling itself fuller and fuller with the colored fluid, the sac gradually distending until it occupies half the hind-body.”
The food-reservoir of the Hymenoptera is a lateral pouch at the end of the long, slender œsophagus, and has been seen in the bee to be filled with honey.
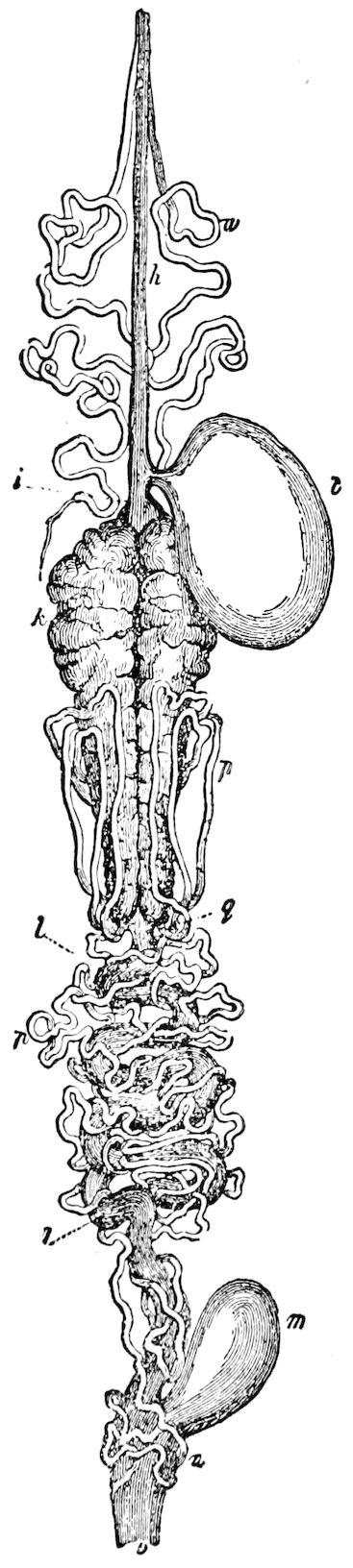
Fig. 309.—Digestive canal of Sphinx ligustri: h, œsophagus; i, rudiment of the gizzard; k, “stomach”; q, its pyloric end; t, food reservoir; p, urinary tubes; l, ilium; m, cœcum of colon; n, rectum; v, vent.—After Newport.
In the mole-cricket the hinder part of the crop is armed within with hook-like bristles directed backwards so as not to prevent the energetic pressure of the food backwards into the proventriculus, and to obviate the possibility of a regurgitation. (Eberli.)
The fore-stomach or proventriculus.—This is especially well developed in the Dermaptera, in the Orthopterous families Locustidæ, Gryllidæ, and Mantidæ, while in the Thysanura (Lepisma) there is a spherical gizzard provided with six teeth. It also occurs in many wood-boring insects, and in most carnivorous insects, notably the Carabidæ, Dyticidæ, Scolytidæ, in the Mecoptera (scorpion-flies), in the fleas, and in many kinds of ants, as well as Cynips, Leucospis, and Xyphidria. It is very muscular, lined within with chitin, which is usually provided with numerous teeth arising from the folds. These folds begin in the œsophagus or crop, and suddenly end where the mesenteron (“chylific stomach”) begins. It has been compared with the gizzard of birds, and is usually called by German authors the chewing or masticating stomach. (Kaumagen.)
The proventriculus is best developed in the Gryllidæ (Acrida viridissima), where the six folds at the end of the crop close together to form a valve between the crop and proventriculus. “They are each armed with five very minute hooked teeth; and, continued into the gizzard, develop many more in their course through that organ. These first teeth are arranged around the entrance to the gizzard, and seem designed to retain the insufficiently comminuted food and to pass it on to that organ.
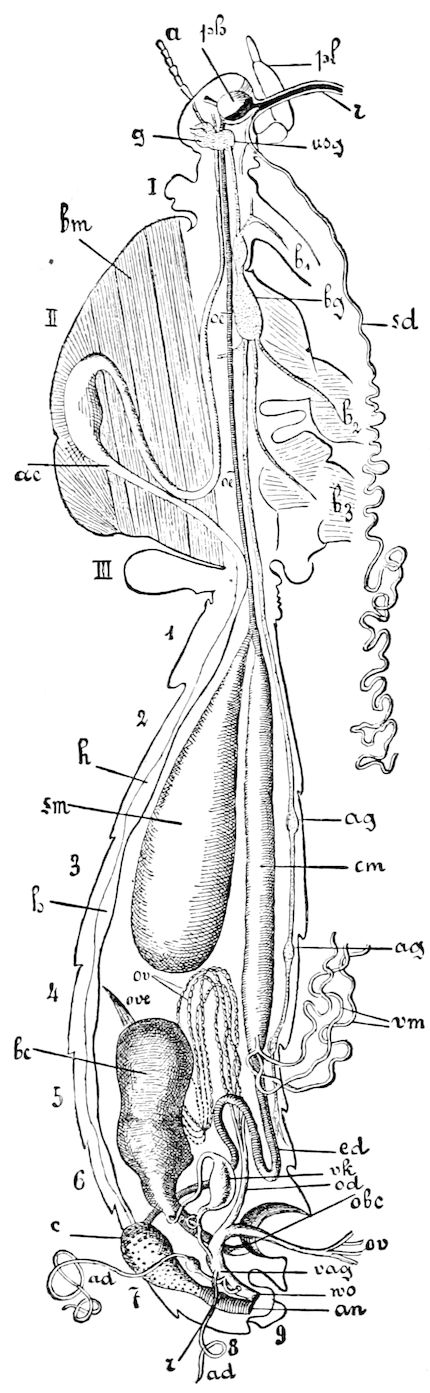
Fig. 310.—Anatomy of Danais archippus after removal of right half of the body. Lettering of the head: a, antenna; ph, pharynx; pl, labial palpi; r, proboscis; g, brain; usg, subœsophageal ganglion. Lettering of the thorax: I. II. III. thoracic segments; b1, b2, b3, the coxal joints of the three pairs of legs; bm, muscles of the wings; ac cephalic aorta with its swelling; œ, œsophagus; bg, thoracic ganglia of the ventral cord; sd, salivary glands of one side, those of the other side cut off near their entrance into the common salivary duct. Lettering of the abdomen: 1–9. abdominal segments; h, heart; sm, so-called sucking-stomach (food-reservoir); cm, chyle-stomach; ag, abdominal ganglia: ed, hind intestine with colon (c) and rectum (r); rm, urinary vessels; ov, ovarial tubes, those of the right side cut off; ove, terminal filaments of the ovaries; bc, bursa copulatrix; obc, its outer aperture; od, oviduct; vag, vagina; wo, its outer aperture; ad, glandular appendages of the vagina partly cut away; vk, connective canal between the vagina and bursa copulatrix with swelling (receptaculum seminis); an, anus.—After Burgess, from Lang.
Fig. 311.—Transverse section of the proventriculus of Gryllus cinereus: muc, muscular walls; r, horny ridge between the large teeth (sp).—After Minot.

Fig. 312.—Transverse section of the proventriculus of the cockroach.—After Miall and Denny.
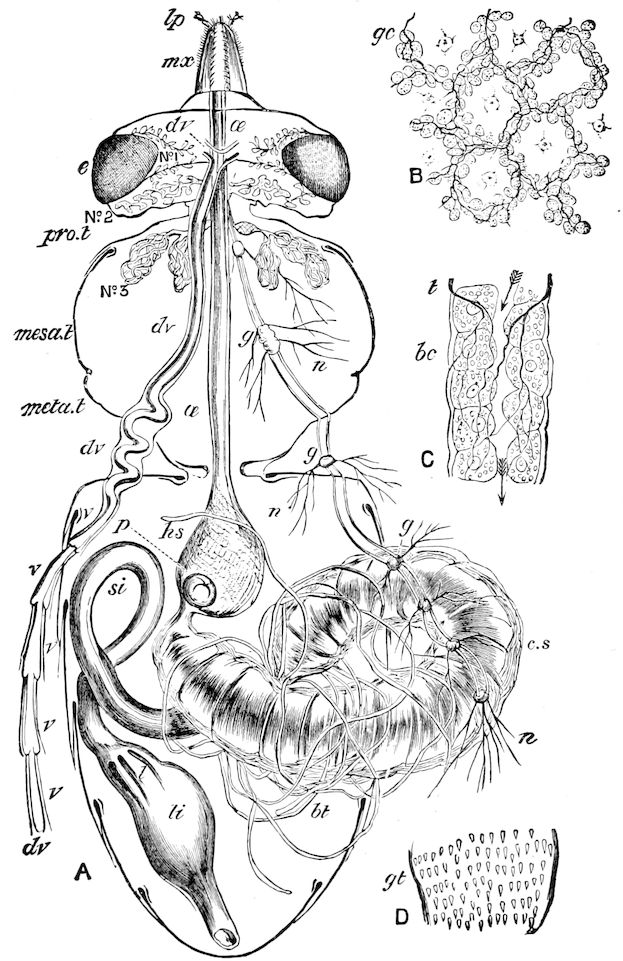
Fig. 313.—Digestive canal of the honey-bee: A, horizontal section of the body; lp, labial palpus; mx, maxilla; e, eye; pro. t, prothorax; mesa. t, mesothorax; meta. t, metathorax; dv, dorsal vessel; v, v, ventricles of the same; No. 1, No. 2, No. 3, salivary gland systems; œ œsophagus; g, g, ganglia of chief nerve-chain; n, nerves; hs, honey-sac; p, petaloid stopper or calyx of honey-sac or stomach-mouth; c. s, chyle stomach; bt, urinary tubes; si, small intestine (ilium); l, “lamellæ or gland-plates of colon,” rectal glands; li, rectum. B, cellular layer of stomach; gc, gastric cells, × 200. C, urinary tube; bc, cells; t, trachea. D, inner layer, with gastric teeth (gt).—After Cheshire.
“Next to these in succession on each of the longitudinal ridges are four flat, broad, somewhat quadrate teeth, each of which is very finely denticulated along its free margin. These extend about half-way through the gizzard. They appear to be alternately elevated and depressed during the action of the gizzard, and to serve to carry on the food to the twelve cutting teeth, with which each ridge is also armed, and which occupy the posterior part of the organ. These 309teeth are triangular, sharp-pointed, and directed posteriorly, and gradually decrease in size in succession from before backward. Each tooth is very strong, sharp-pointed, and of the color and consistence of tortoise shell, and is armed on each side by a smaller pointed tooth. These form the six longitudinal ridges of the gizzard, between each two of which there are two other rows of very minute teeth of a triangular form, somewhat resembling the larger one in structure, occupying the channels between the ridges. The muscular portion of the gizzard is equally interesting. It is not merely formed of transverse and longitudinal fibres, but sends from its inner surface into the cavity of each of the large teeth other minute but powerful muscles, a pair of which are inserted into each tooth. The number of teeth in the gizzard amounts to 270, which is the same number in these Gryllidæ as found formerly by Dr. Kidd in the mole-cricket. Of the different kinds of teeth there are as follows: 72 large treble teeth, 24 flat quadrate teeth, 30 small single-hooked teeth, and 12 rows of small triangular teeth, each row being formed of 12 teeth. This is the complicated gizzard of the higher Orthoptera.” (Newport.)
In the more generalized cockroach, there are six principal folds, the so-called teeth, which project so far inwards as to nearly meet (Fig. 312). The entire apparatus of muscles and teeth is, as Miall and Denny state, “an elaborate machine for squeezing and straining the food, and recalls the gastric mill and pyloric strainer of the crayfish. The powerful annular muscles approximate the teeth and folds, closing the passage, while small longitudinal muscles, which can be traced from the chitinous teeth to the cushions, appear to retract these last, and open a passage for the food.”
As in the fore-stomach or proventriculus of the lobster, the solid, rounded teeth do not appear to triturate the solid fragments found in the organ, but act rather as a pyloric strainer to keep such bodies out of the chylific stomach. We accept the view of Plateau that this section of the digestive canal in insects, which he compares to the psalterium of a ruminant, is a strainer rather than a masticatory stomach, and both Forel and Emery, as well as Cheshire, take this view.
The proventriculus of the honey-bee (Fig. 313, hs) is called by apiarians the “honey-sac” or “honey-stomach.” Cheshire states that if it be carefully removed from a freshly killed bee, its calyx-like “stomach-mouth” may be seen to gape open and shut with a rapid snapping movement. The entrance to the stomach is guarded by four valves, each of which is strongly chitinous within, and fringed along its edge with downward-pointing fine stiff bristles. By the contraction of the longitudinal muscles (lm), the valves open to allow the passage of food from the honey-sac to the “chyle-stomach.” It is closed at will by circular muscles (tm). Then the bee can carry food for a week’s necessities, either using it rapidly in the production of wax, or eking it out if the weather is unfavorable for the gathering of a new store.
Fig. 314.—“Honey-sac stopper,” “stomach-mouth,” or calyx-bell of honey-bee, × 50. A, front view of one of the lobes of the calyx-bell; l, lip-like point, covered by down-turned bristles (b); sm, side membrane. B, longitudinal section of the stomach-mouth, with continuations into entrance of chyle-stomach; l, l, lip-like ends of leaflets; s, setæ; lm, longitudinal muscles; tm, transverse muscles in cross-section; cl, cell-layer of honey-sac; LM, TM, longitudinal and transverse muscles of same; nc, nucleated cells of tubular extension of stomach-mouth into chyle-stomach; lm′, tm′, longitudinal and transverse muscles of chyle stomach; c, c, cells covered within by an intima. C, cross-section of stomach-mouth; m, cross-section of muscles seen at lm in B; tm, transverse muscles surrounding stomach-mouth. D, cross-section through small intestine; a and m, longitudinal and surrounding muscles.—After Cheshire.
Cheshire also shows that when bees suck up from composite and other flowers nectar together with much pollen, the outside wrinkled membrane (sm, A, Fig. 314) “is seen to continually run up in folds, and gather itself over the top of the stomach-mouth, bringing with it, by the aid of its setæ, the large pollen-grains the nectar contains.” The lips (l, l, B, Fig. 314), now opening, take in this pollen, which is driven forwards into the cavity made between the separating lips by an inflow of the fluid surrounding the granules. The lips in turn close, but the down-pointing bristles are thrown outwards from the face of the leaflet, in this way revealing their special function, as the pollen is prevented from receding while the nectar passes back into the honey-sac, strained through between the bristles aforesaid, the last parts escaping by the loop-like openings seen in the corners of C, Fig. 314. The whole process is immediately and very rapidly repeated, so that the pollen collects and the honey is cleared. “Three purposes, in addition to those previously enumerated, are thus subserved by this wondrous mechanism. First, the bee can either eat or drink from the mixed diet she carries, gulping down the pollen in pellets, or swallowing the nectar as her necessities demand. Second, when the collected pollen is driven 311forwards into the chyle-stomach, the tube extension, whose necessity now becomes apparent, prevents the pellets forming into plug-like masses just below p, Fig. 313, for, by the action of the tube, these pellets are delivered into the midst of the fluids of the stomach, to be at once broken up and subjected to the digestive process. And third, while the little gatherer is flying from flower to flower, her stomach-mouth is busy in separating pollen from nectar, so that the latter may be less liable to fermentation and better suited to winter consumption. She, in fact, carries with her, and at once puts into operation, the most ancient, and yet the most perfect and beautiful, of all ‘honey-strainers.’”
Forel’s experiments on the proventriculus of ants prove that through its valvular contrivance it closes the passage from the crop to the mid-intestine (“chylific stomach”), and allows the contents of the former to pass slowly and very gradually into the latter. Emery confirms this view, and concludes that the organ in the Camponotidæ and in the Dolichoderidæ provided with a calyx-bell, usually regarded as a triturating stomach (Kaumagen), but more correctly as a pumping stomach, consists of parts which perform two different functions. Under the operation of the muscles of the crop the entrance to the pumping stomach becomes closed, in order by such spasmodic contraction to prevent the flow of the contents of the crop into the proventriculus. By the pressure of the transverse muscles of the proventriculus its contents are emptied into the mid-intestine, while simultaneously a regurgitation into the crop is prevented. In the Dolichoderidæ and Plagiolepidinæ the closure in both cases is effected by the valves. In the true Camponotidæ there are two separate contrivances for closing; the calyx belonging to the crop-musculature, while the valves essentially belong to the proventricular pumping apparatus.
Opinions vary as to the use of this portion of the digestive canal. Graber compares it to the gizzard of birds, and likens the action of the rosette of teeth to the finer radiating teeth of the sea-urchin, and styles it a chopping machine, which works automatically, and allows no solid bits of food to pass in to injure the delicate walls of the stomach (mid-gut).
He also states that the food when taken from the proventriculus is very finely divided, while that found in the œsophagus contains large bits.
Kolbe says that this view has recently been completely abandoned, and that the teeth are used to pass the food backwards into the chylific stomach. “But Goldfuss had denied the triturating action of the proventriculus of the Orthoptera (Symbolæ ad Orthopterorum quorundam Œconomiam, 1843), stating that the contents of the same are already fluid in the gullet, so that the fore-stomach (Kaumagen) does not need to comminute the food” (Kolbe). In the Gryllidæ and Locustidæ, just before the posterior opening of the proventriculus into the stomach the chitinous lining swells into a ring and projects straight back as the inner wall of the cylindrical chylific stomach. The muscular layer forms two sac-like outgrowths or folds, which separate on the circular fold from the chitinous membrane. This apparatus only allows very finely comminuted food to pass into the stomach.
In the Acrydiidæ (Eremobia muricata) at the end of the proventriculus, where it passes into the stomach, is a small circular fold which hangs down like a curtain in the stomach.
The œsophageal valve.—Weismann[50] states that the origin of the proventriculus in the embryo of flies (Muscidæ) shows that it should 312be regarded as an intussusception of the œsophagus. While in the embryo the invaginated portion of the œsophagus is short, after the hatching of the larva it projects backwards into the mid-intestine. Kowalevsky also observed in a young muscid larva, 2.2 mm. in length, that the œsophagus, shaped like a tube, extends back into the expanded portion (proventriculus) and opens into the stomach (Fig. 315, A). In a larva 10 mm. long the funnel is shorter, the end being situated in the proventriculus (Fig. 315, B, pr). In the cavity between the outer (o) and inner wall (i) no food enters, and the use of this whole apparatus seems to be to prevent the larger bits of food from passing into the chylific stomach (Kowalevsky).
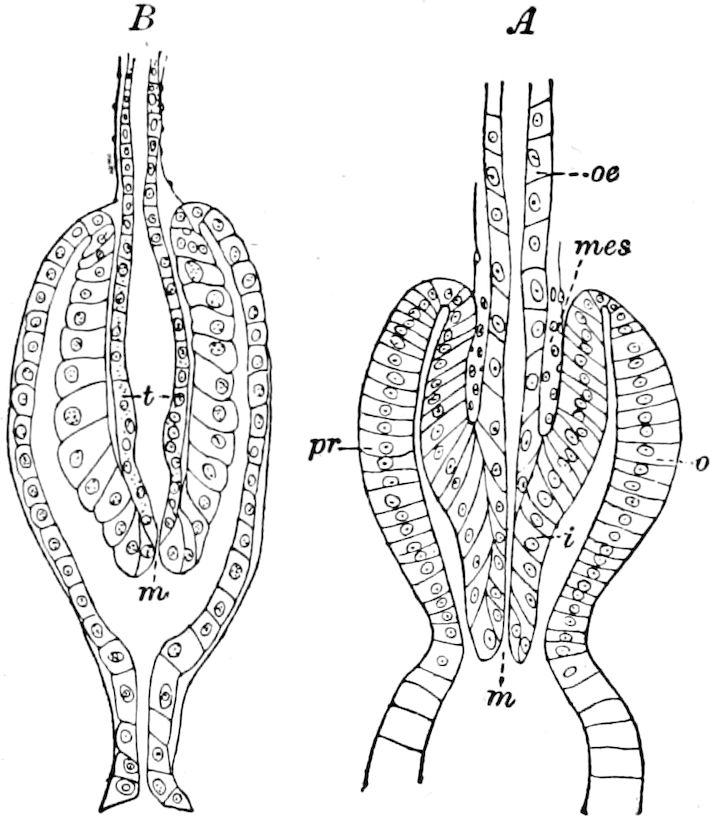
Fig. 315.—Œsophageal valve of young muscid larva: m, its opening: t, thickening of the cells; mes, mesoderm.—After Kowalevsky.
Beauregard has found a similar structure in the Meloidæ, and calls it the “cardiac valvule” (Fig. 318, Kl). It was observed by Mingazzini in the larvæ of phytophagic lamellicorn beetles, and Balbiani described it in a myriopod (Cryptops) under the name of the “œsophageal valvule.”
Gehuchten describes a homologous but more complicated structure in a tipulid larva (Ptychoptera contaminata), but differing in containing blood-cavities, as a tubular prolongation of the posterior end of the œsophagus which passes through the proventriculus and opens at various positions in the anterior part of the chylific stomach (Fig. 316).
The three layers composing this funnel are distant from each other and separated by blood-cavities, the whole forming “an immense blood-cavity extended between the epithelial proventricular lining and the muscular coat.”
According to Schneider the longitudinal muscular fibres of the fore and hind gut in insects pass into the stomach (mid-gut). The anterior part of the fore-gut has generally only circular fibres. When, however, the longitudinal fibres arise behind the middle, then they separate from the digestive canal and are inserted a little behind the beginning of the chylific stomach. Hence there is formed an invagination of the proventriculus, which projects into the cavity of the stomach.
Schneider describes this process, which he calls the “beak,” as an invagination of the fore-stomach which projects into the cavity of the stomach. The two layers of the invagination in growing together form a beak varying in shape, being either simple or lobed and armed with bristles or teeth. This beak is tolerably large in Lepisma, Dermaptera (Forficula), Orthoptera, and in the larvæ and adults of Diptera, but smaller in the Neuroptera and Coleoptera, while in other insects it is wanting.
313Proventricular valvule.—Gehuchten also describes in Ptychoptera what he calls “the proventricular valvule,” stating that it is “a circular fold of the intestinal wall” (Fig. 310, vpr). He claims that it has not before been found, the “proventricular beak” of Schneider being regarded by him as the œsophageal valvule.
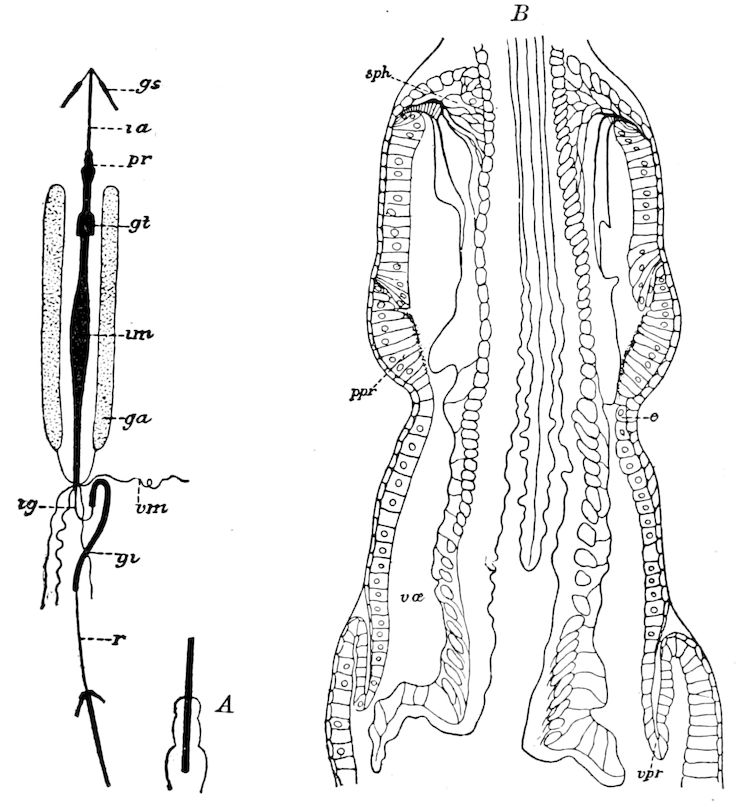
Fig. 316.—Digestive canal of Ptychoptera contaminata: gs, salivary glands; ra, œsophagus; pr, proventriculus; gt, crown of eight small tubular glands; im, mid-intestine; ga, two accessory white glands; vm, urinary vessels; ig, small intestine; gi, large intestine; r, rectum; A, the proventriculus in which the hinder end of the œsophagus extends as far as the chyle-stomach. B, longitudinal section of the proventricular region; sph, muscular ring or œsophageal sphincter; ppr, wall of the proventriculus; e, circular constriction dividing the cavity of the proventriculus in two; vpr, circular fold of the wall of the mid-intestine forming the proventricular valve; vœ, œsophageal valve.—After Gehuchten.
The peritrophic membrane.—This membrane appears first to have been noticed by Ramdohr in 1811 in Hemerobius perla. It has been found by Schneider, who calls it the “funnel.” On the hinder end of the fore-stomach, he says, the cuticula forms a fold enclosing the outlet of the fore-stomach, and extending back like a tube to the anus. This “funnel,” he adds, occurs in a great number of insects. It has been found in Thysanura, but is wanting in Hemiptera. In the Coleoptera it is absent in Carabidæ and Dyticidæ. It is generally present in Diptera and in the larvæ of Lepidoptera, but not in 314the adults. In Hymenoptera it has been found in ants and wasps, but is absent in Cynipidæ, Ichneumonidæ, and Tenthredinidæ. All those insects (including their larvæ) possessing this funnel eat solid, indigestible food, while those which do not possess it take fluid nourishment. It is elastic, and firmly encloses the contents of the digestive tract. Until Schneider’s discovery of its general occurrence, it had only been known to exist in the viviparous Cecidomyia larvæ (Miastor). Wagner, its discoverer, noticed in the stomach of this insect a second tube which contained food. Pagenstecher was inclined to regard the tube as a secretion of the salivary glands. Metschnikoff, however, more correctly stated that the tube consisted of chitin, but he regarded it as adapted for the removal of the secretions. (Schneider.) Plateau, however, as well as Balbiani, the latter calling it the “peritropic membrane,” considers this membrane as a secretion of the chylific stomach, and that it is formed at the surface of the epithelial cells. It surrounds the food along the entire digestive tract, forming an envelope around the fæcal masses. On the other hand, Gehuchten states that in the larva of Ptychoptera its mode of origin differs from that described by Plateau and by Schneider, and that it is a product of secretion of special cells in the proventriculus.
The mid-intestine.—This section of the digestive canal, often, though erroneously, called the “chylific stomach” or ventriculus, differs not only in its embryonic history, but also in its structure and physiology from the fore and hind intestine of arthropods, and also presents no analogy to the stomach of the vertebrate animals. In insects it is a simple tube, not usually lined with chitin, since it is not formed by the invagination of the ectoderm, as are the fore and hind intestine, the absence of the chitinous intima promoting the absorption of soluble food. Into the anterior end either open two or more large cœcal tubes (Fig. 299), or its whole outer surface is beset with very numerous fine glandular filaments like villi (Fig. 317 and Fig. 329).
The mid-intestine varies much in size and shape; it is very long in the lamellicorn beetles (Melolontha and Geotrupes), and while in Meloë it is very large, occupying the greatest part of the body-cavity, in the longicorn beetles and in Lepidoptera it is very small. The pyloric end consists of an internal circular fold projecting into the cavity. In the Psocidæ (Cæcilius) the pyloric end is prolonged into a slender tube nearly as long as the larger anterior portion.
The limits between the mid and hind intestine are in some insects difficult to define, the urinary tubes sometimes appearing to open 315into the end of the mid-intestine (“stomach”). The latter also is sometimes lined with an intima. The limits are also determined by a circular projection, directly behind which is an enlargement of the intestine in the shape of a trench (rigole), or circular cul-de-sac (the “pyloric valvule” of some authors, including Beauregard), while the walls of the small intestine contract so as to produce a considerable constriction of the cavity of the canal. This constriction exactly coincides with the beginning of the double layer of circular muscles in the wall of the small intestine. An internal layer, which is the continuation of the circular muscles of the chylific stomach, and an external layer much more developed probably belong to this part of the alimentary canal. Since the homologue of the circular fold occurs in the locust as well as in Diptera, it is probably common to insects in general.
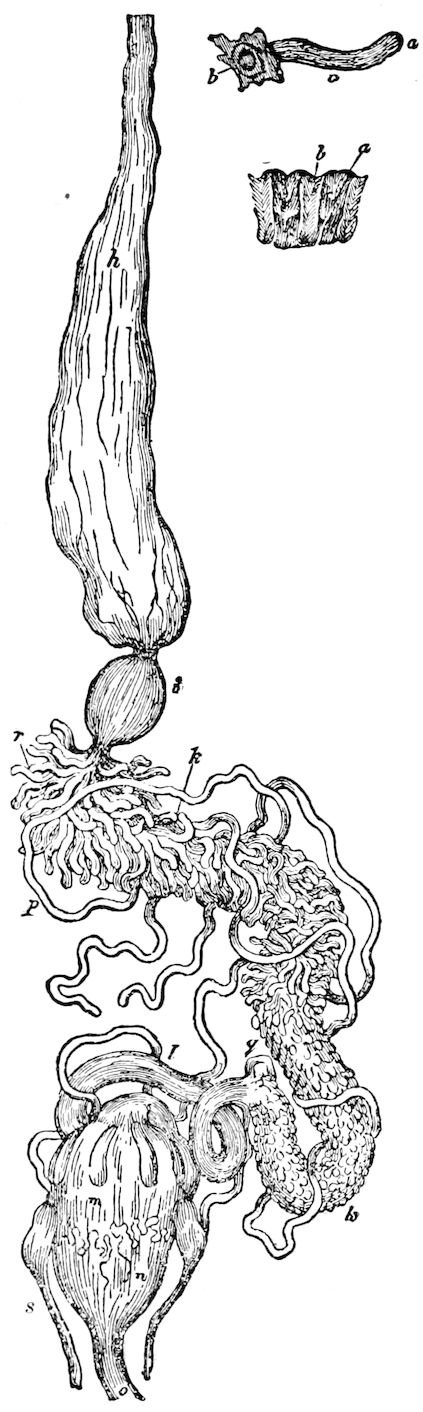
Fig. 317.—Digestive canal of Carabus monilis: h, œsophagus; i, gizzard or proventriculus; k, “stomach,” with its cœca (r); p, urinary tubes; q, their point of insertion; m, n, colon, with cœcal glands; s, anal glands; a, b, c, a gastric cœcum; a, b, portion of lining of gizzard.—After Newport.
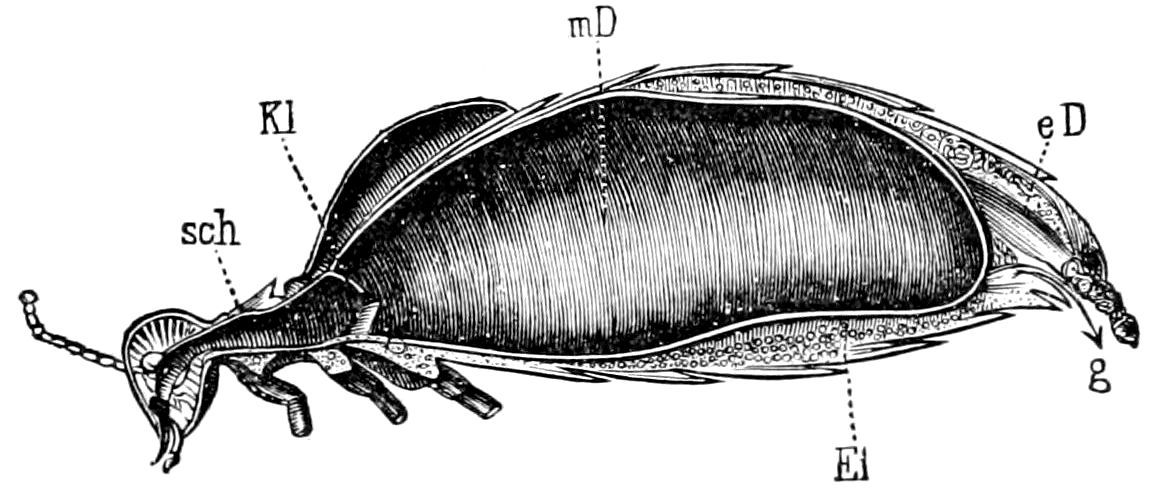
Fig. 318.—Digestive canal of Meloe: sch, œsophagus; Kl, œsophageal valve; mD, mid-intestine; eD, hind-intestine; Ei, eggs; g, sexual opening.—After Graber.
Gehuchten adds that the limit set by the circular projection does not exactly coincide with the opening into the intestine of the urinary tubes and the two annexed glands. He shows by a section (his Fig. 133) that the tubular glands open into the alimentary canal 316in front of the circular fold. It is the same with the Malpighian tubes. They are not, therefore, he claims, dependences of the terminal intestine, but of the mid-intestine. Beauregard has observed the same thing in the vesicating insects (Meloidæ). The Malpighian tubes, he says, open into the “chylific stomach” before the valvular crown. This arrangement does not seem to be general, because, according to Balbiani, the Malpighian vessels open into the beginning of the intestine in Cryptops. Compare also Minot’s account of the valve in locusts separating the stomach from the intestine, and in front of which the urinary or Malpighian tubes open.
Histology of the mid-intestine.—The walls of the stomach are composed of an internal epithelium, a layer of connective tissue, with two muscular layers, the inner of which is formed of unstriated circular muscular fibres, and the outer of striated longitudinal muscular fibres.
In the cockroach short processes are given off from the free ends of the epithelial cells, as in the intestine of many mammals and other animals. “Between the cells a reticulum is often to be seen, especially where the cells have burst; it extends between and among all the elements of the mucous lining, and probably serves, like the very similar structure met with in mammalian intestines, to absorb and conduct some of the products of digestion.” (Miall and Denny.)
Gehuchten shows that the epithelial lining of the mesenteron (chylific stomach) of the dipterous larva Ptychoptera is composed of two kinds of cells, i.e. secreting or glandular cells and absorbent cells, the former situated at each end of the stomach, and the absorbent cells occupying the middle region. The part played by these cells in digestion will be treated of beyond in the section on digestion. (See p. 327.)
The hind-intestine.—In many insects this is divided into the ileum, or short intestine, and the long intestine. The limit between the intestine and stomach is externally determined by the origin of the urinary tubes, which are outgrowths of the anterior end of the proctodæum. Like the fore-intestine the hind-intestine is lined with a thick muscular layer, and, as Gehuchten states, the passage from the epithelial lining of the stomach (mid-intestine) to the muscular lining of the intestine is abrupt.
Large intestine.—In Ptychoptera, as described by Gehuchten, there are no precise limits between the small and large intestine; the epithelium of the large intestine has a special character, and its constituents present a close resemblance to the absorbed cells of the chylific stomach, being like them large and polygonal. The muscular layer is not continuous, and is formed of longitudinal and circular fibres, the latter being the larger.
317The ileum—Though in most insects slender, and therefore called the small intestine, the ileum is in locusts (Fig. 298) and grasshoppers (Anabrus, Fig. 299) as thick as the stomach. In many carnivorous beetles (Dyticus, Fig. 320, il, and Necrophorus) it is very long, but rather slender and short in the Carabidæ and Cicindelidæ, as well as those insects whose food is liquid, such as Diptera. In the Lepidoptera it varies in length, being in Sphinx quite long and bent into seven folds (Fig. 309), while it is very short in the Psocidæ, Chrysomelidæ, and Tenthredinidæ.
In the locust the ileum is traversed by six longitudinal folds with intervening furrows; outside of each furrow is a longitudinal muscular band. Seen from the inner surface the epithelium has an unusual character, the cells in the middle of each of the flat folds being quite large, polygonal in outline, while towards the furrows the cells become very much smaller. The walls are double when seen in transverse section, the inner layer consisting of epithelial cells resting on connective tissue, the outer layer formed of circular muscles. The cuticula is thin, but probably chitinous; it resembles that on the gastro-ileal folds, except that there are no spinules, but unlike the cuticula of the stomach it extends equally over the folds and the furrows. (Minot.) In the cockroach the junction of the small intestine with the colon is abrupt, a well-developed annular fold assuming the nature of a circular valve. (Miall and Denny.)
The gastro-ileal folds.—In the locust the intestine is separated from the chylific stomach by what Minot calls “the gastro-ileal folds,” which form a peculiar valve. The urinary vessels open just underneath and in front of this valve. In Melanoplus, and probably in the entire family of Acrydiidæ, they are indicated as “dark spots, round in front and lying at the anterior end of the ileum so as to form a ring around the interior of the intestine.” They are 12 in number, and all alike. They are pigmented and round in front where they are broadest and stand up highest; they narrow down backwards, the pigment disappears, and they gradually fade out into the ileal folds. Directly beneath them, and just at the posterior end of the stomach, there is a strong band of circular striated muscular fibres. The epithelium of these folds is covered with minute conical spines, which are generally, but not always, wanting between the folds. (Minot.)
The colon.—This section of the intestine (Fig. 319) is sometimes regarded as a part of the rectum. In the locust the six longitudinal folds of the ileum are continued into the colon, but their surface, instead of being smooth as in the ileum, is thrown up into numerous 318irregular curved and zigzag secondary folds. The cells of the epithelium are of uniform size, and the layer is covered by a highly refringent cuticula without spines; and, like that in the ileum, it rests on a layer of connective tissue, beyond which follows (1) an internal coat of longitudinal, and (2) an external coat of circular striated muscular fibres. (Minot.)
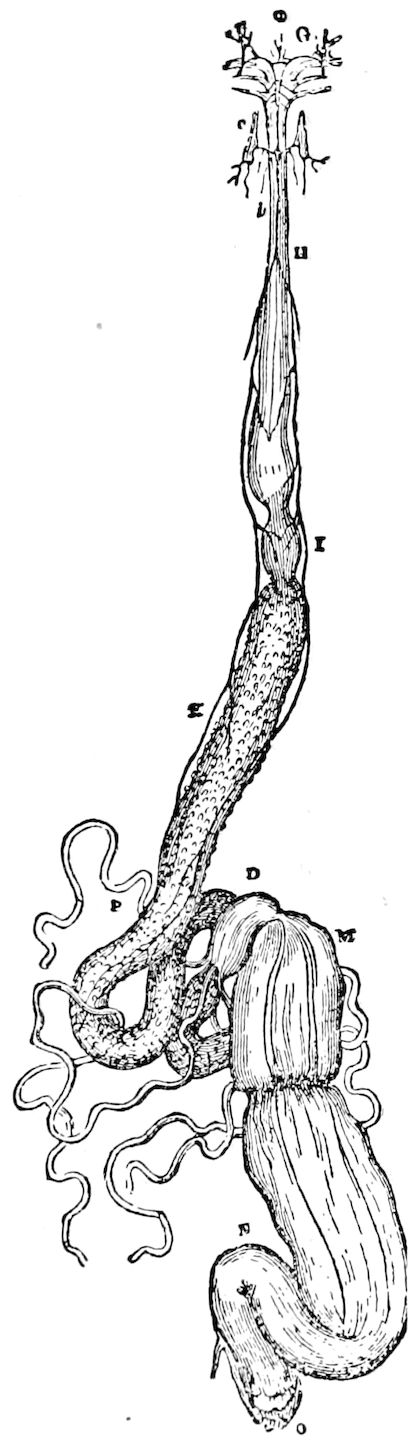
Fig. 319.—Digestive canal of Lucanus cervus: G, anterior muscles of the pharynx; H, œsophagus; I, gizzard; K, chyle-stomach; L, ilium; M, colon (cœcal part of); N, colon; O, rectum; a, frontal ganglion of the vagus; b, vagus nerve; c, anterior lateral ganglion connected with the vagus.—After Newport.
In butterflies (Pontia brassicæ), in Sphinx ligustri, and probably in most Lepidoptera the colon is distinct from the rectum, and is anteriorly developed into a very large more or less pyriform or bladder-like cæcum (Figs. 309, 310), which in certain Coleoptera (Dyticus, Fig. 320, d; Silpha, Necrophorus, etc.) is of remarkable length and shape; it also occurs in Nepidæ (Fig. 327). In the cockroach a lateral cæcum “is occasionally, but not constantly, present towards its rectal end,” and a constriction divides the colon from the rectum. (Miall and Denny.)
The rectum.—The terminal section of the hind-gut varies in length and size, but is usually larger than the colon, and with thick, muscular walls. In Lepidoptera it is narrow and short.
The rectum is remarkable for containing structures called rectal glands (Fig. 298). Chun describes those of Locusta viridissima as six flat folds, formed by a high columnar epithelium and a distinct cuticula; there is a coat of circular bands corresponding to the furrows between the glands. Minot states that this description is applicable to the locusts (Acrydiidæ) he has investigated, the only difference being in the structural details of the single layers. He claims that the rectal folds “do not offer the least appearance of glandular structure,” neither is their function an absorbent one, as Chun supposed. From their structure and position Fernald regards the rectal glands of Passalus as acting like 319a valve, serving to retain the food in the absorptive portions of the digestive tract till all nutriment is extracted.
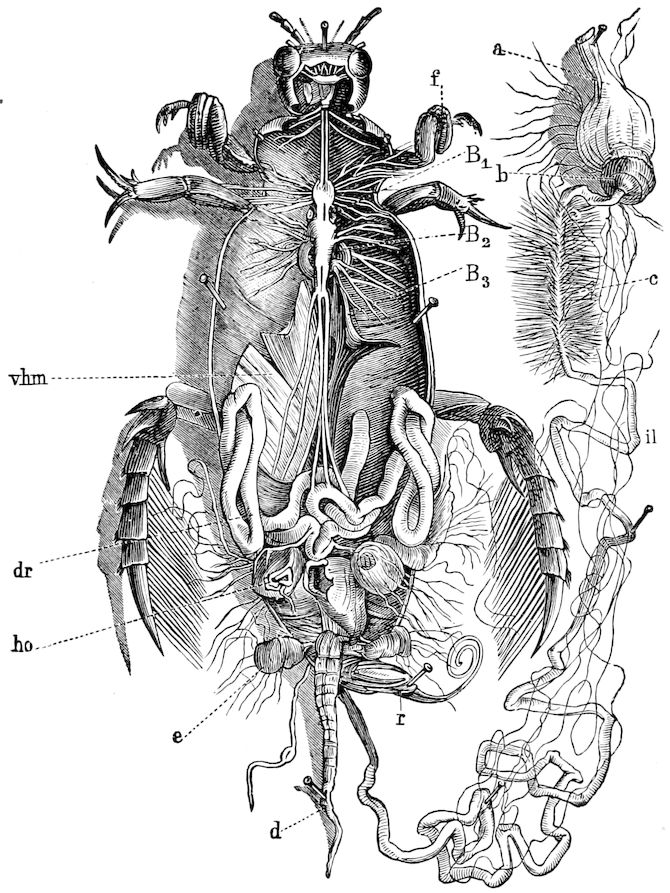
Fig. 320.—Dyticus marginalis, ♂ opened from the back: a, crop; b, proventriculus; c, mid-intestine beset with fine cœcal glands; d, long cœcal appendage of the colon; apodemes; B1-B3, apodemes; vhm, coxal extensor muscle, moving the hind leg; ho, testis; dr, accessory gland; r, penis; e, reservoir of the secretion of the anal gland.—After Graber.
The epithelial folds of the larvæ of dragon-flies serve as organs of respiration, the water being admitted into this cavity, and when forcibly expelled serving to propel the creature forward. Paired and single anal glands (repugnatorial) enter the rectum of certain Coleoptera (Figs. 302, l; 317, s; 320, e).
The vent (anus).—The external opening of the rectum is situated in the end of the body, in the vestigial 10th or 11th abdominal segment, 320and is more or less eversible. It is protected above in caterpillars, and other insects with 10 free abdominal segments, by the suranal plate. It is bounded on the sides by the paranal lobes, while beneath is the infra-anal lobe.
The anus is wanting in certain insects, and where this is the case the hind-gut, owing to a retention of the embryonic condition, is usually separated from the mid-intestine. (See p. 300.)
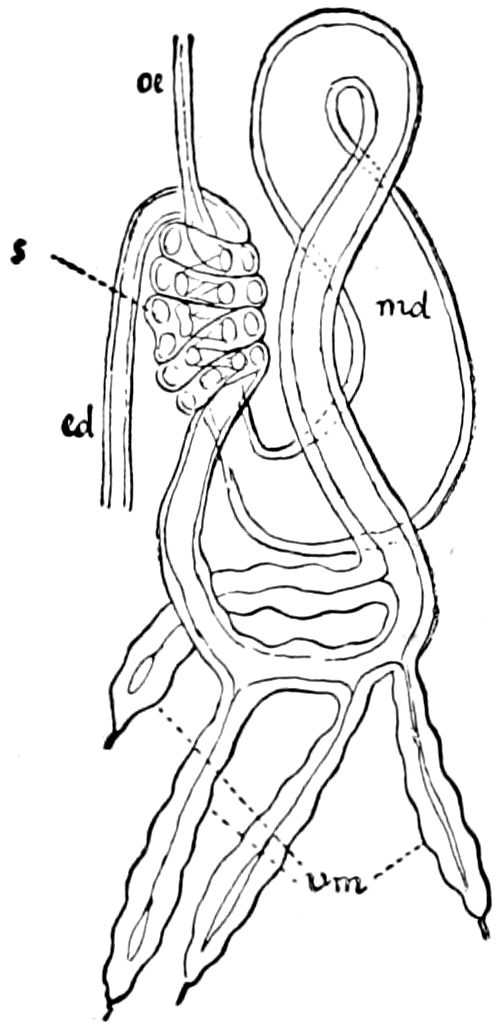
Fig. 321.—Enteric canal of Psyllopsis fraxinicola: œ, œsophagus; md, mid-intestine; ed, hind-intestine; vm, urinary vessels; s, the coil formed by the hind-intestine and the most anterior part of the mid-intestine.—After Witlaczil, from Lang.
Some remarkable features of the digestive canal in hemipterous insects are noteworthy. In the Coccidæ, according to Mark, the anterior end of the long mid-intestine forms, with the hinder end of the œsophagus, a small loop, whose posterior end is firmly grown to the wall of the rectum, and forms a cup-like invagination of the latter. Then the rest of the tube-like stomach turns sidewise and forms a large loop, which turns back on itself and occupies a large part of the body-cavity. This loop receives on the anterior end, near the œsophagus, the two urinary vessels, and forms just below the opening into the rectum a short cæcum.
In other homopterous genera (Psyllidæ and some Cicadidæ) Witlaczil describes nearly the same peculiarity, the mid-gut and part of the intestine forming a loop growing together for a certain distance and winding round each other (Fig. 321).
Histology of the digestive canal.—In all the divisions of the digestive canal of insects the succession of the cellular layers composing it is the same: 1st, a cuticula; 2d, an epithelium; 3d, connective tissue; 4th, muscular tissue. In the locust, the first division of the canal (fore-gut), there are two muscular coats, an internal longitudinal and an external circular coat; the fibres are all striated. The lining epithelium is not much developed, but forms a thick, hard, and refringent cuticula, which is thrown up into spiny ridges. In the second division (mid-gut, “stomach”) the epithelium is composed of very high columnar cells, which make up the greater part of the thickness of the walls, while the cuticula is very delicate, slightly refringent, with no ridges, and is probably not chitinous; the fibres of the muscular coats are not striated, while this division is also distinguished by the presence of glandular follicles and folds. The stomach and the cæcal appendages have all these peculiarities in common, while no other part of the canal is thus characterized.
321The third division (intestine and rectum) is composed of an epithelium, the cells of which are intermediate in size between those of the fore and mid gut. The cells are often pigmented, and they are covered by a much thicker cuticula than that of the stomach, but which is not so thick and hard as that of the œsophagus and proventriculus. The very refringent cuticula is not thrown up into ridges, though in some parts it is covered with delicate conical spines, which are very short. “The epithelium and underlying connective tissue (tunica propria) are thrown up into six folds, which run longitudinally, being regular in the ileum and rectum (as the rectal glands), but very irregular in the colon. Outside the depression between each two neighboring folds there is a longitudinal muscular band, these making six bands. This peculiar disposition of the longitudinal muscles does not occur in any other part of the canal; it is, therefore, especially characteristic of the third division.” (Minot.)
LITERATURE ON THE ORGANS OF DIGESTION
Treviranus, G. R. Resultate einiger Untersuchungen über den inneren Bau der Insekten. (Verdauungsorgane von Cimex rufipes.) (Annal. d. Wetterau. Gesells., 1809, i, pp. 169–177, 1 Taf.)
Ramdohr, C. A. Abhandlungen über die Verdauungswerkzeuge der Insekten. 1811, vii, pp. 221, 30 Taf.
Dutrochet, R. J. H. Mémoire sur les métamorphoses du canal alimentaire dans les insectes. (Journal de Physique, 1818, lxxxvi, pp. 130–135, 189–204; Meckel’s Archiv, 1818, iv, pp. 285–293.)
Suckow, F. W. L. Verdauungsorgane der Insekten. (Heusinger’s Zeitschr. f. organ. Physik., 1828, iii, pp. 1–89.)
Doyère, L. Note sur le tube digestif des Cigales. (Ann. Sc. nat. Zool., 1839, 2 Sér., xi, pp. 81–85.)
Grube, A. E. Fehlt den Wespen- oder Hornissenlarven ein After oder nicht? 1 Taf. (Müller’s Archiv für Physiol., 1849, pp. 47–74.)
Sirodot. Recherches sur les sécrétions chez les insectes. (Ann. Sc. nat. Zool., 1858, 4 Sér., x, pp. 141–189, 251–328, 12 Pls.)
Milne-Edwards, H. Leçons sur la physiologie et l’anatomie comparée, v, 1859, pp. 498–536, 581–638.
Dufour, L. Recherches anatomiques sur les Carabiques et sur plusieurs autres insectes Coléoptères. Appareil digestif. (Ann. Sc. nat., ii, 1824, pp. 462–482, 2 Pls.; iii, 1824, pp. 215–242, 5 Pls., pp. 476–491, 3 Pls.; iv, 1824, pp. 103–125, 4 Pls.; iv, 1825, pp. 265–283.)
—— Recherches anatomiques sur l’Hippobosque des chevaux. (Ann. Sc. nat., 1825, vi, pp. 299–322, 1 Pl.)
—— Description et figure de l’appareil digestif de l’Anobium striatum. (Ibid., xiv, 1828, pp. 219–222, 1 Pl.)
—— Recherches anatomiques sur les Labidoures. Appareil de la digestion. (Ibid., xiii, 1828, pp. 348–354, 2 Pls.)
322—— Recherches anatomiques et considerations entomologiques sur quelques insectes Coléoptères, compris dans les familles des Dermestins, des Byrrhiens, des Acanthopodes et des Leptodactyles. Appareil digestif. (Ibid., Sér. 2, Zool., i, 1834, pp. 67–76, 2 Pls.)
—— Résumé des recherches anatomiques et physiologiques sur les Hémiptères. (Ibid., pp. 232–239.)
—— Mémoire sur les métamorphoses et l’anatomie de la Pyrochroa coccinea. Appareil digestif. (Ibid., Sér. 2, Zool., xiii, 1840, pp. 328–330, 334–337, 2 Pls.)
—— Histoire comparative des metamorphoses et de l’anatomie des Cetonia aurata et Dorcus parallelepipedus. Appareil digestif. (Ibid., Sér. 2, Zool., 1824, xviii, pp. 174–176, 2 Pls.)
—— Anatomie générale des Diptères. Appareil digestif. (Ibid., Sér. 3, Zool., i, 1814, pp. 248, 249.)
—— Histoire des métamorphoses et de l’anatomie du Piophila petasionis. Appareil digestif. (Ibid., Sér. 3, Zool., i, 1844, pp. 372–377, 1 Pl.)
—— Études anatomiques et physiologiques sur les insectes Diptères de la famille des Pupipares. Appareil digestif. (Ibid., Sér. 3, Zool., iii, 1845, pp. 67–73, 1 Pl.)
—— Recherches sur l’anatomie et l’histoire naturelle de l’Osmylus maculatus. Appareil digestif. (Ibid., Sér. 3, Zool., ix, 1848, pp. 346–349, 1 Pl.)
—— Études anatomiques et physiologiques, et observations sur les larves des Libellules. Appareil digestif. (Ibid., Sér. 3, Zool., xvii, 1852, pp. 101–108, 1 Pl.)
—— Recherches anatomiques sur les Hyménoptères de la famille des Urocerates. Appareil digestif. (Ibid., Sér. 4, Zool., i, 1854, pp. 212–216, 1 Pl.)
—— Fragments d’anatomie entomologique. Sur l’appareil digestif du Nemoptera lusitanica. (Ibid., Sér. 4, viii, 1857, pp. 6–9, 1 Pl.)
—— Recherches anatomique et considerations entomologiques sur les Hémiptères du genre Leptopus. Appareil digestif. (Ibid., Sér. 4, Zool., 1858, x, pp. 352–356, 1 Pl.)
—— Recherches anatomiques sur l’Ascalaphus meridionalis. Appareil digestif. (Ibid., Sér. 4, xiii, 1860, pp. 200–202, 1 Pl.)
Leydig, F. Zur Anatomie von Coccus hesperidum. (Zeitschr. f. wissens. Zool., v, 1853, pp. 1–12, 1 Taf.)
Lubbock, J. On the digestive and nervous System of Coccus hesperidum. (Proc. Roy. Soc., ix, 1886, pp. 480–486; also Ann. Mag. Nat. Hist., 1859, Ser. 3, iii, pp. 306–311.)
Scheiber, S. H. Vergleichende Anatomie und Physiologie der Œstriden-Larven. V. Das chylo- und uropœtische System. (Sitzber. d. k. Akad. d. Wissens. Wien. Math.-naturwiss. Cl., 1862, xlv, pp. 39–64, 1 Taf.)
Gerstaecker, A. Bronn’s Klassen und Ordnungen des Tierreichs. V. Gliederfüssler. (Ernährungsorgane, pp. 87–105.)
Graber, V. Zur naheren Kenntnis des Proventriculus und der Appendices ventriculares bei den Grillen und Laubheuschrecken. (Sitzber. d. k. Akad. d. Wissensch. Wien. Mathem.-naturwiss. Cl., lix, 1869, pp. 29–46, 3 Taf.)
—— Ueber die Ernährungsorgane der Insekten und nächstverwandten Gliederfüssler. (Mitteil. d. naturwiss. Vereins für Steiermark. Graz, 1871, ii, pp. 181, 182.)
—— Verdauungssystem des Prachtkäfers. (Ibid., Graz, 1875.)
—— Die Insekten., i, 1877. (Verdauungsapparat, pp. 308–328.)
Wilde, K. F. Untersuchungen über den Kaumagen der Orthopteren. (Archiv f. Naturgesch., xliii Jahrg., 1877, pp. 135–172, 3 Taf.)
323Simroth, H. Ueber den Darmkanal der Larven von Osmoderma eremita mit seinen Anhängen. (Giebel’s Zeitschr. f. d. ges. Naturwiss., 1878, li, pp. 493–518, 3 Taf.)
Müller, H. Ueber die angebliche Afterlösigkeit der Bienenlarven. (Zool. Anzeiger, 1881, pp. 530, 531.)
Schiemenz, Paulus. Ueber das Herkommen des Futtersaftes und die Speicheldrüsen der Bienen, nebst einem Anhänge über das Riechorgan. (Zeitschr. f. wissens. Zool., xxxviii, 1883, pp. 71–135, 3 Taf.)
Rovelli, G. Alcune ricerche sul tubo digerente degli Atteri, Ortotteri e Pseudo-Neurotteri. (Como, 1884, p. 15.)
Beauregard, H. Structure de l’appareil digestif des Insectes de la tribu des Vésicants. (Compt. rend. Acad. Paris, 1884, xcix, pp. 1083–1086.)
—— Recherches sur les Insectes vésicants., 1 Part, Anatomie. (Journ. Anat. Phys. Paris, 1885, xxi Année, pp. 483–524, 4 Pls.; 1886, xxii Année, pp. 85–108, 242–284, 5 Pls.)
—— Les Insectes vésicants, Paris, 1890, Chap. III, Appareil digestif, pp. 63–99; (Phénomènes digestifs, pp. 161–170; Pls. 6–9.)
Wertheimer, L. Sur la structure du tube digestif de l’Oryctes nasicornis. (Compt. rend. Soc. Biol. Paris, 1887, Sér. 8, iv, pp. 531, 532.)
Kowalevsky, A. Beitrage zur Kenntniss der nachembryonal Entwicklung der Musciden. (Zeitschr. f. wissens. Zool., xlv, 1887, pp. 542–594, 5 Taf.)
Schneider, A. Ueber den Darm der Arthropoden, besonders der Insekten. (Zool. Anzeiger, 1887, x Jahrg., pp. 139, 140.)
—— Ueber den Darmkanal der Arthropoden. (Zool. Beiträge von A. Schneider, ii, 1887, pp. 82–96, 3 Taf.)
Fritze, A. Ueber den Darmkanal der Ephemeriden. (Berichte der Naturforsch.-Gesellsch. zu Freiburg i. Br., 1888, iv, pp. 59–82, 2 Taf.)
Emery, C. Ueber den sogenannten Kaumagen einiger Ameisen. (Zeitschr. f. wissens. Zool., 1888, xlvi, pp. 378–412, 3 Taf.)
Meinert, F. Contribution à l’anatomie des Fourmilions. (Overs. Danske Vidensk. Selsk. Forhandl. Kjöbenhavn, 1889, pp. 43–66, 2 Pls.)
Mingazzini, P. Richerche sul canale digerente dei Lamellicorni fitofage (Larve e Insetti perfetti). (Mitteil. Zool. Station zu Neapel, ix, 1889–1891, pp. 1–112, 266–304, 7 Pls.)
Fernald, Henry T. Rectal glands in Coleoptera. (Amer. Naturalist, xxiv, pp. 100, 101, Jan., 1890.)
Visart, O. Digestive canal of Orthoptera. (Atti Soc. Toscana Scient. Natur., vii, 1891, pp. 277–285.)
Eberli, J. Untersuchungen an Verdauungstrakten von Gryllotalpa vulgaris. (Vierteljahresschr. d. Naturforsch. Gesells. Zurich, 1892, Sep., p. 46, Fig.)
Holmgren, Emil. Histologiska studier öfver några lepidopterlarvers digestionskanal och en del af deras Körtelartade bildningar. (Ent. Tidskr. Årg. xiii, pp. 129–170, 1892, 6 Pls.)
Ris, F. Untersuchung über die Gestalt des Kaumagens bei den Libellen und ihren Larven. (Zool. Jahrb. Abth. Syst., ix, 1896, pp. 596–624, 13 Figs.)
See also the works of Straus-Dürckheim, Newport, Mark, Witlaczil, Vayssière, Landois, Jordan, Oudemans, Berlese, List, Grassi, Verson, Miall and Denny, Leidy, Cheshire, Kowalevsky, Gehuchten, Locy, etc.
b. Digestion in insects
For the most complete and reliable investigation of the process of digestion, we are indebted to Plateau, whose results we give, besides the conclusions of later authors:
In mandibulate or biting insects, the food is conducted through the œsophagus by means of the muscular coating of this part of the digestive canal. Suctorial insects draw in their liquid food by the contractions followed by the dilatations of the mid-intestine (chylific stomach). Dragon-flies, Orthoptera, and Lepidoptera swallow some air with their food.
Where the salivary glands are present, the neutral alkaline fluid secreted by them has the same property as the salivary fluid of vertebrates of rapidly transforming starchy foods into soluble and assimilable glucose. In such forms as have no salivary glands, their place is almost always supplied by an epithelial lining of the œsophagus, or, as in the Hydrophilidæ, a fluid is secreted which has the same function as the true salivary fluid.
Nagel states that the saliva of the larva of Dyticus is powerfully digestive, and has a marked poisonous action, killing other insects, and even tadpoles of twice the size of the attacking larva, very rapidly. The larvæ not only suck the blood of their victims, but absorb the proteid substances. Drops of salivary juice seem to paralyze the victim, and to ferment the proteids. The secretion is neutral, the digestion tryptic. Similar extra-oral digestion seems to occur in larvæ of ant-lions, etc. (Biol. Centralbl., xvi, 1896, pp. 51–57, 103–112; Journ. Roy. Micr. Soc., 1896, p. 184.)
In carnivorous insects and in Orthoptera, the œsophagus dilates into a crop (ingluvies) ended by a narrow, valvular apparatus (or gizzard of authors). The food, more or less divided by the jaws, accumulates in the crop, which is very distensible; and, when the food is penetrated by the neutral or alkaline liquid, there undergoes an evident digestive action resulting, in carnivorous insects, in the transformation of albuminoid substances into soluble and assimilable matter analogous to peptones, and, in herbivorous insects, an abundant production of sugar from starch. This digestion in the crop, a food-reservoir, is very slow, and, until it is ended, the rest of the digestive canal remains empty.
“Any decided acidity found in the crop is due to the injection of acid food; but a very faint acidity may occur, which results from the presence in the crop of a fluid secreted by the cæcal diverticula of the mesenteron.” (Miall and Denny.)
325When digestion in the crop is accomplished, the matters are subjected to an energetic pressure of the walls through peristaltic contractions, and then, guided by the furrows and chitinous teeth, pass along or gradually filter through the valvular apparatus or proventriculus, whose function is that of a strainer.
At the beginning of the “chyle-stomach” (mesenteron) of Orthoptera are glandular cæca which secrete a feebly acid fluid. This fluid emulsifies fats, and converts albuminoids into peptones. It passes forwards into the crop, and there acts upon the food.
In the mesenteron (mid-intestine) the food is acted upon by an alkaline or neutral fluid, never acid, either secreted, as in Orthoptera, by local special glands, or by a multitude of minute glandular cæca, as in many Coleoptera, or by a simple epithelial layer. It has no analogy with the gastric juices of vertebrates; its function differs in insects of different groups; in carnivorous Coleoptera it actively emulsionizes greasy matters; in the Hydrophilidæ it continues the process of transformation of starch into glucose, begun in the œsophagus. In the Scarabæidæ, it also produces glucose, but this action is local, not occurring elsewhere; in caterpillars, it causes a production of glucose, and transforms the albuminoids into soluble and assimilable bodies analogous to peptones, and also emulsionizes greasy matters. Finally, in the herbivorous Orthoptera there does not seem to be any formation of sugar in the stomach itself, the production of glucose being confined to the crop (jabot).
When digestion in the crop is finished, the proventriculus relaxes, and the contents of the crop, now in a semi-fluid condition, guided by the furrows and teeth, passes into the mesenteron, which is without a chitinous lining, and is thus fitted for absorption.
The contents of the mid-intestine (chylific stomach) then slowly and gradually pass into the intestine, the first anterior portion of which, usually long and slender, is the seat of an active absorption. The epithelial lining observed in certain insects seems, however, to indicate that secondary digestion takes place in this section. The reaction of the contents is neutral or alkaline.
The second and larger division of the intestine only acts as a stercoral reservoir. (The voluminous cæcum occurring in Dyticidæ, Nepa, and Ranatra, whether full or empty, never contains gas, and it is not, as some have supposed, a swimming-bladder.) The liquid product secreted by the Malpighian tubes accumulates in this division, and, under certain circumstances, very large calculi are often formed. In his subsequent paper on the digestion of the cockroach, Plateau states that in the intestine are united the residue of 326the work of digestion and the secretion of the urinary or Malpighian tubes, this secretion being purely urinary.
These organs are exclusively depuratory and urinary, freeing the body from waste products of the organic elements. The liquid they secrete contains urea (?), uric acid and abundant urates, hippuric acid (?), chloride of sodium, phosphates, carbonate of lime, oxalate of lime in quantity, leucine, and coloring-matters.
The products of the rectal or anal glands vary much in different groups, but they take no part in digestion, nor are they depuratory in their nature.
Insects have nothing resembling chylific substances.[51] The products of digestion, dissolved salts, peptones, sugar in solution, emulsionized greasy matters, pass through the relatively delicate walls of the digestive canal by osmose, and mingle outside of the canal with the blood.
Whatever substances remain undigested are expelled with the excrements; such are the chitin of the integuments of insects, vegetable cellulose, and chlorophyll, which is detected by the microspectroscope all along the digestive canal of phytophagous insects.
In his experiments in feeding the larvæ of Musca with lacmus, Kowalevsky found that the œsophagus, food-reservoir, and proventriculus, with its cæcal appendages, always remained blue, and had an alkaline reaction; the mid-intestine, also, in its anterior portion, remained blue, but a portion of its posterior half became deep red, and also exhibited a strong reaction. The hind-intestine, however, always remained blue, and also had an alkaline reaction. (Biol. Centralbl., ix, 1889, p. 46.)
The mechanism of secretion.—Gehuchten describes the process of secretion in insects, the following extract being taken from his researches on the digestive apparatus of the larva of Ptychoptera. The products of secretion poured into the alimentary canal are more or less fluid; for this reason, it is impossible to say when an epithelial cell at rest contains these products. For the secreting nature of these cells is only apparent at the moment when they are ready 327for excretion; then the cellular membrane swells out, and a part of the protoplasmic body projects into the intestinal cavity.
Before going farther, the terms secretion and excretion should, he says, be defined. With Ranvier, he believes that the elaboration in the protoplasm of a definite fluid substance is, par excellence, the secretory act, while the removal of this substance is the act of excretion.
Fig. 322.—Different phases of the mechanism of secretion and of excretion.—After Gehuchten.
A glandular cell of the chylific stomach, when at rest, is always furnished with a striated “platform,” or flat surface, or face, on the side facing the cavity of the stomach, and the free edge of the platform, or plateau, is provided with filaments projecting into the digestive cavity (Fig. 322, f). These glandular cells, when active, differ much in appearance. In a great number, the platform (plateau) has disappeared, and is replaced by a simple, regular membrane. During the process of secretion, a finely granular mass, in direct continuity with the protoplasm, swells, and raises the membrane over the entire breadth of the cell, causing it to project into the intestinal cavity (Fig. 322, A, B). These vesicles, or drops of the secretion, whether free or still attached by a web to the cells, are clear and transparent in the living insect, but granular in the portions of the digestive canal fixed for cutting into sections. Gehuchten then asks: “How does a cell gorged with the products of secretion empty itself?” Both Ranvier and also Heidenhain believe that one and the same glandular cell may secrete and excrete several times without undergoing destruction, but their researches 328made on salivary glands have not answered the question. Gehuchten explains the process thus: when the epithelial cell begins to secrete, the clear fluid elaborated in the protoplasm of the cell increases the intracellular tension, until, finally, the fluid breaks through certain weak places in the swollen basal membrane of the platform, and then easily passes through the closely crowded filaments, and projects out into the intestinal cavity as a pear-shaped vesicle of a liquid rich in albumens at first attached to the free face of the cell, but finally becoming free, as at Fig. 322, A, B.
When the elaboration of the substance to be secreted is more active, the mechanism of the secretion is modified. The basal membrane of the platform may then be raised at several places at once; instead of a single vesicle projecting into the intestinal cavity, each cell may present a great number more or less voluminous. If all remain small and rapidly detach themselves from the glandular cell, the filaments of the platform are simply separated from each other at different points of the free face, as in Fig. 322, C. On the other hand, when the different vesicles of a single cell become larger, the filaments of the platform are compressed and crowded against each other in the spaces between the vesicles remaining free, and the undisturbed portions of the platform appear homogeneous (Fig. 322, D). After the excretion of the secretory products by this process of strangulation, the cell then assumes the aspect of a glandular cell at rest, and may begin again to form a new secretion.
To sum up: The process of excretion may occur in two ways:
1. Where the membrane ruptures and the substances secreted are sent directly out into the digestive cavity. 2. Where the vesicles become free by strangulation, floating in the glandular or intestinal cavity, and ending by rupturing and coming into contact with the neighboring vesicles or with the food.
Absorbent cells.—Besides the glandular or secreting cells in Ptychoptera, there is between the two regions of the chyle-stomach lined with these cells a region about a centimetre long composed of absorbent cells. The absorbent cells are very large, polygonal, and contain a large nucleus, in which is a striated convoluted chromatic cord.
The food on entering the chyle-stomach is brought into contact with the products secreted in the proventriculus, in the first part of the chyle-stomach, and in the tubular glands. These products of secretion act on the food, extracting from them useful substances which they render soluble. These substances, after having been absorbed by the absorbent cells in the middle region of the stomach, 329undergo special modifications, and are transformed into solid products, which are situated at the bottom of these cells. Afterwards the alimentary substances freed from a portion of their useful substances are again placed in contact with the products of secretion in the distal part of the chylific ventricle, and reach the terminal part of the intestine.
“The products of secretion,” adds Gehuchten, “diverted into the intestinal canal do not come into immediate contact with the alimentary substances; they are separated from it by a continuous, structureless, quite thick membrane (the peritrophic membrane), which directly envelops the cylinder of food matters, extending from the orifice of the œsophageal valvule to the end of the intestine. Between this membrane and the free face of the epithelial lining there exists a circular space, into which are thrown and accumulate the excreted substances. The latter then cannot directly mingle with the aliments; but when they are liquid they undoubtedly pass through this membrane by osmose, and thus come into contact with the nutritive substances. It is the same with the products of absorption. The absorption of soluble products of the intestinal cavity is not then so simple a phenomenon as it was at first thought to be, since these products are nowhere brought into immediate contact with the absorbent cells” (pp. 90, 91).
The most recent authority, Cuénot, states that absorption of the products of digestion takes place entirely in the mid-intestine, and in its cæca when these are present. The mid-intestine exercises a selective action on the constituents of the food comparable to the action of the vertebrate liver.
LITERATURE ON THE PHYSIOLOGY OF DIGESTION
Davy, J. Note on the excrements of certain insects, and on the urinary excrement of insects. (Edinburgh New Phil. Journ., 1846, xl, pp. 231–234, 335–340; 1848, xlv, pp. 17–29.)
—— Some observations on the excrements of insects, in a letter addressed to W. Spence. (Trans. Ent. Soc. London, Ser. 2, iii, 1854, pp. 18–32.)
Bouchardat, A. De la digestion chez le ver à soie. (Revue et Mag. de Zool., Sér. 2, 1851, iii, pp. 34–40.)
Lacaze-Duthiers, H., et A. Riche. Mémoire sur l’alimentation de quelques insects gallicoles et sur la production de la graisse. (Ann. Scienc. natur., 1854, Sér. 4, ii, pp. 81–105.)
Basch, S. Untersuchungen über das chylopoetische und uropoetische System von Blatta orientalis. (Sitzungsber. d. math.-naturwiss. Classe d. Akad. d. Wissensch. Wien., 1858, xxxiii, pp. 234–260, 5 Taf.)
Lambrecht, A. Der Verdauungsprozess der stickstoffreichen Nährmittel, welche unsere Bienen geniessen, in den dazu geschaffenen Organen derselben. (Bienenwirtschaftl. Centralbl., viii Jahrg., 1872, pp. 73–78, 83–89.)
330Plateau, F. Recherches sur les phénomènes de la digestion chez les insectes, (Mém. Acad. roy. de Belgique, Sér. 2, xli, 1 Part, 1873, pp. 124, 3 Pls.)
—— Note additionelle au mémoire sur les phénomènes de la digestion chez les insectes. (Bull. Acad. roy. de Belgique, Sér. 2, xliv, 1877, pp. 710–733.)
Tursini, G. Fr. Un primo passo nella ricerca dell’ assorbimento intestinale degli artropodi. (Rend. d. R. Accadem. di Sc. fis. e matemat. di Napoli, 1877, xvi, pp. 95–99, 1 Pl.)
Jousset de Bellesme, Physiologie comparée. Recherches expérimentales sur la digestion des insectes et en particulier de la blatte. Paris, 1876, vii, and 96 pp., 3 Pls.
—— Recherches sur les fonctions des glandes de l’appareil digestif des Insectes. (Compt. rend., lxxxii, Paris, 1876, pp. 97–99.)
—— Travaux originaux de physiologie comparée. (i, Insectes, Digestion, Métamorphoses.) Paris, 1878, 5 Pls.
Simroth, H. Einige Bemerkungen über die Verdauung der Kerfe. (Zeitschr. f. d. gesammten Naturwiss., xli, 1878, pp. 826–831.)
Krukenberg, C. Fr. W. Versuche zur vergleichenden Physiologie der Verdauung und vergleichende physiologische Beiträge zur Kenntnis der Verdauungsvorgange. (Untersuch, a. d. physiolog. Institut d. Universität Heidelberg, 1880, i, 4, pp 327, Figs.; ii, 1, p. 1, Figs.)
Metschnikoff, E. Untersuchungen über die intrazelluläre Verdauung bei wirbellosen Tieren. (Arb. d. zool. Instit. Wien., 1883, v, pp. 141–168, 2 Taf.)
Locy, William A. Anatomy and physiology of the family Nepidæ. (Amer. Naturalist, xviii, 1884, pp. 250–255, 353–367, 4 Pls.)
Vangel, E. Beiträge zur Anatomie, Histiologie, und Physiologie des Verdauungsapparates des Wasserkäfers, Hydrophilus piceus. (Termész. Füzet., x, 1886, pp. 111–126 (in Hungarian); pp. 190–208 (in German), 1 Pl.)
Schönfeld. Die physiologische Bedeutung des Magenmundes der Hönigbiene. (Archiv f. Anat. u. Physiol., Physiol. Abt., 1886, pp. 451–458.)
Faussek, V. Beiträge zur Histiologie des Darmkanals der Insekten. (Zeitschr. f. wiss. Zool., 1887, xl, pp. 694–712, 1 Taf.; Abstract in Zool. Anz., Jahrg. x, pp. 322, 323, 1 Taf.)
Frenzel, J. Ueber Bau und Thätigkeit des Verdauungskanals der Larve des Tenebrio molitor, mit Berücksichtigung anderer Arthropoden (Berlin. Ent. Zeitschr., 1882, pp. 267–316, 1 Taf.); Inaug.-Diss. Göttingen, 1882.
—— Einiges über den Mitteldarm der Insekten, sowie über Epithel-regeneration. (Archiv f. Mikrosk. Anat., 1885, xxvi, pp. 229–306, 3 Taf.)
—— Zum feineren Bau des Wimperapparates. (Ibid., 1886, xxviii, pp. 53–80, 1 Taf.)
—— Die Verdauung lebenden Gewebes und die Darmparasiten. (Archiv f. Anat., 1891.)
Gehuchten, A. van. Recherches histologiques sur l’appareil digestif de la Ptychoptera contaminata, I Part. Étude du revêtment épithélial et recherches sur la sécrétion. (La Cellule, 1890, vi, pp. 183–291, 6 Pls.)
Cuénot, L. Études physiologiques sur les Orthoptères. (Arch. Biol., xiv, 1895, pp. 293–341, 2 Pls.)
Needham, James G. The digestive epithelium of dragon-fly nymphs. (Zool. Bull., i, 1897, Chicago, pp. 104–113, 10 Figs.)
With the writings of Mingazzini (see p. 323), Kowalevsky, Ranvier, Haidenhain, Beauregard (p. 323), Sadones.
THE GLANDULAR AND EXCRETORY APPENDAGES OF THE DIGESTIVE CANAL
Into each primary division of the digestive canal open important glands. The salivary and silk-glands are offshoots of the œsophagus (stomodæum); the cœcal appendages open into the stomach (mesenteron), while the urinary tubes grow out in embryonic life from the primitive intestine (proctodæum), and there are other small glands which are connected with the end of the hind-intestine.
a. The salivary glands
We will begin our account of these glands with those of the Orthoptera, where they are well developed. In the cockroach a large salivary gland and accompanying reservoir lie on each side of the œsophagus and crop. The gland is a thin, leaf-like, lobulated mass, divided into two principal lobes. These open into a common trunk, which after receiving a branch from a small accessory lobe, and from the salivary reservoir, unites with its fellow to form the unpaired salivary duct which opens into the under side of the lingua. Each salivary reservoir is a large oval sac with transparent walls. (Miall and Denny, also Figs. 299, sr, and 327.) The ducts and reservoirs have a chitinous lining, and the ducts are, like the tracheæ, surrounded by a so-called spiral thread, or by separate, incomplete, hooplike bands, which serve to keep the duct permanently distended. In the locust (Fig. 298) the lobules are more scattered, forming small separate groups of acinose glands. In the embryo of Forficula Heymons has observed a pair of salivary glands opening on the inner angle of the mandibles, a second pair opening in the second maxillæ, while a third pair of glands, whose function is doubtful, is situated in the hinder part of the head, opening to the right and left on the chitinous plate (postgula) behind the submentum. In Perla, there are two pairs segmentally arranged (Fig. 343).
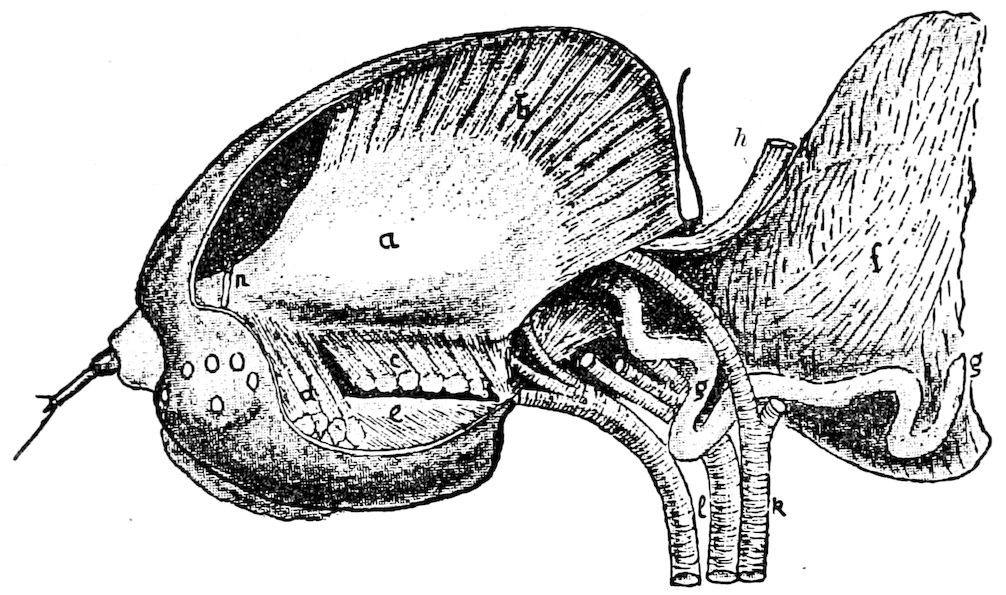
Fig. 323.—Left side of the head of the silkworm: a, adductor muscle of the mandible, from which the muscular fibres have been removed; b, upper fibres of the same; c, lower fibres cut away to show the adductor muscle (e); d, fibres inserted on the accessory adductor lamella; f, œsophagus, much swollen; g, salivary gland; h, dorsal vessel; i, l, tracheæ of the mandibular muscles; k, trachea; n, optic nerve.—After Blanc.
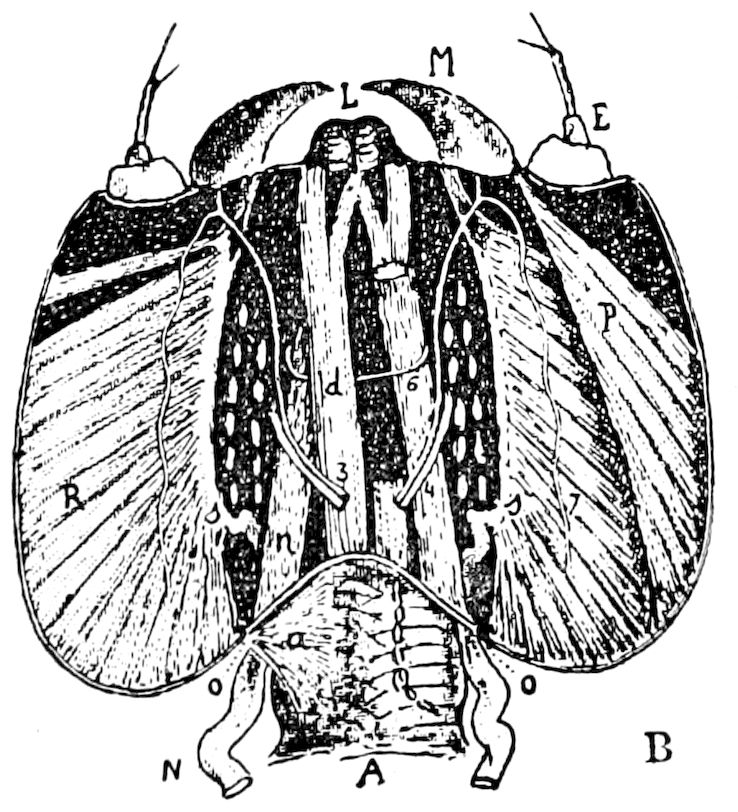
Fig. 324.—Lower side of the head of the silkworm exposed, the spinning apparatus, the œsophageal ganglion, and the adductor of the left mandible removed: M, mandible; P, abductor of the mandible; R, adductor; N, salivary gland attached at O to the edge of the adductor muscle; o, o, transverse portion of the “hyoid”; 3, masticator nerve and its recurrent branch (7); L, tongue cut horizontally.—After Blanc.
Here we might refer to a pair of glands regarded by Blanc as the true salivary glands. They do not appear to be the homologues of the salivary glands of other insects, though probably functioning as such. The functional salivary glands of lepidopterous larvæ have been overlooked by most entomotomists, and the spinning glands have been, it seems to us, correctly supposed to be modified salivary glands. Lucas also regards those of case-worms (Trichoptera) as morphologically salivary glands. Those of the silkworm were figured by Réaumur (Tom. i, Pl. v, Fig. 1), but not described; while those of Cossus, which are voluminous, were regarded by Lyonet as “vaisseaux dissolvans.” Dr. Auzoux (1849), in his celebrated model of the silkworm, represented them accurately, while Cornalia briefly described them as opening into the mouth. The first satisfactory description is that of Blanc (1891), who states that in the silkworm “the two salivary glands” are small, flexuous, yellow tubes, which occupy a variable position on the sides of the œsophagus (Fig. 323). The glandular portion passes into the head, ending at the level of the adductor plate of the mandibles (Fig. 324, o), and entering the buccal cavity at the base of the mandible, as seen in Fig. 323. It is plain, when we recognize the direct homology of the silk-glands of the caterpillars with the salivary glands of other insects, and of the spinneret with the hypopharynx, that 333these so-called “salivary glands” in lepidopterous larvæ are different structures. They are probably modified coxal glands, belonging to the mandibular segment.
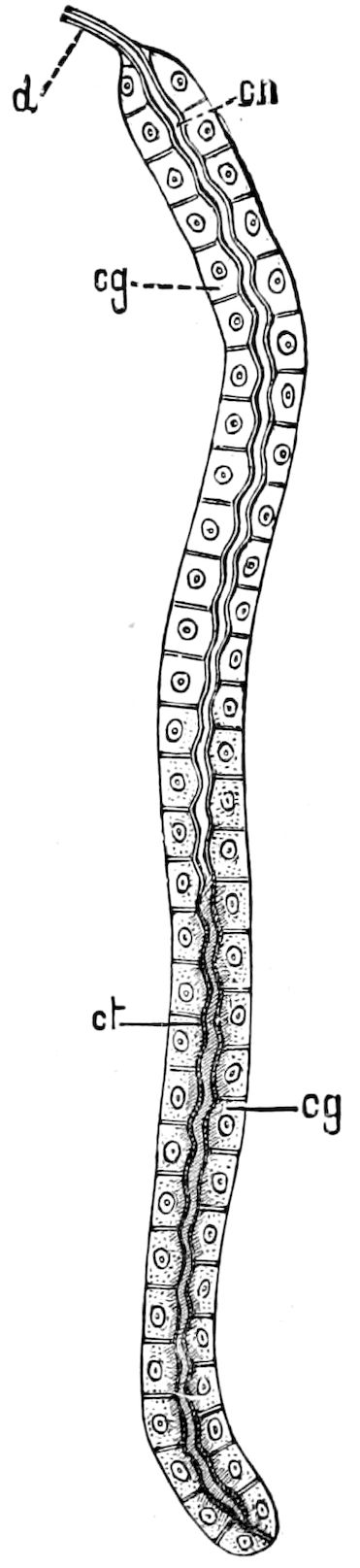
Fig. 325.—One of the two salivary glands of Cæcilius burmeisteri: d, excretory duct; cn, the lumen or canal; cg, gland-cells; ct, salivary fluid.—After Kolbe.
The polygonal epithelial cells of these glands contain branched nuclei, recalling those of the spinning-glands. In those caterpillars which feed on leaves, the salivary glands are slightly developed, but in such as bore into and eat wood, as the Cossidæ, the glands are, as figured by Lyonet, very large, forming two sausage-shaped bodies passing back to the beginning of the mid-intestine, each ending in a long convoluted filament. The salivary glands of the imago are very long and convoluted (Fig. 310, sd).
In the Panorpidæ these glands differ in the sexes, the males having three pairs of very long tortuous tubes, while, in the females, they are reduced to two indistinct vesicles. (Siebold.)
In the Diptera in general there are two pairs, one situated in the beak, the other in the thorax. In the larvæ there is a single pair (Fig. 341). Kraepelin describes a third pair in the Muscidæ at the point of transition from the fulcrum to the œsophagus, but Knüppel has apparently found only what may be fat cells at this point, so that the supposed presence of a third pair in Diptera needs confirmation. In the Psocidæ there are two salivary glands, of simple tubular shape (Fig. 325).
In the Nepidæ the salivary glands are four in number, and of conglomerate structure, two being long and extending back into the beginning of the abdomen, while the other two are about one-fourth as long. (Figs. 327, 328.) In Cicada, besides a pair of simple tortuous tubes, there is in the head another pair of glands, each composed of two tufts of short lobes, situated one behind the other. (Dufour.) In many Hemiptera (Pyrrhocoris, Capsus, etc.) there is but a single pair, each gland consisting of four lobes; in the Coccidæ each gland is divided into two lobes (Fig. 326); in the Aphidæ, according to Witlaczil, they consist of two lobes grown together. In the Psyllidæ they are said to be absent.
In Phylloxera vastatrix the saliva is forced through a salivary 334passage out of the duct and into the mouth by a pumping apparatus furnished with special muscles. (Krassilstschik.)
In the Odonata acinose glands are present in the imago, but not in the nymph until in its last stage, Poletaiew accounting for their absence in the earlier stages by the fact that the larva swallows more or less water while taking its food.
In the Coleoptera, as we have observed in Anopthalmus, there are three pairs of salivary glands (Fig. 74). In the Blapsidæ these glands consist of many ramifying tubes united on each side of the œsophagus into a single duct; in others they are but slightly developed, while in still others they are wanting.
The salivary glands are most highly differentiated in the Hymenoptera, and especially in the bees (Bombus and Apis), where Schiemenz found not less than five systems of glands (Fig. 329; also 87), of which four systems are paired. One pair of these glands lies in the tongue, three in the head, and one in the thorax.
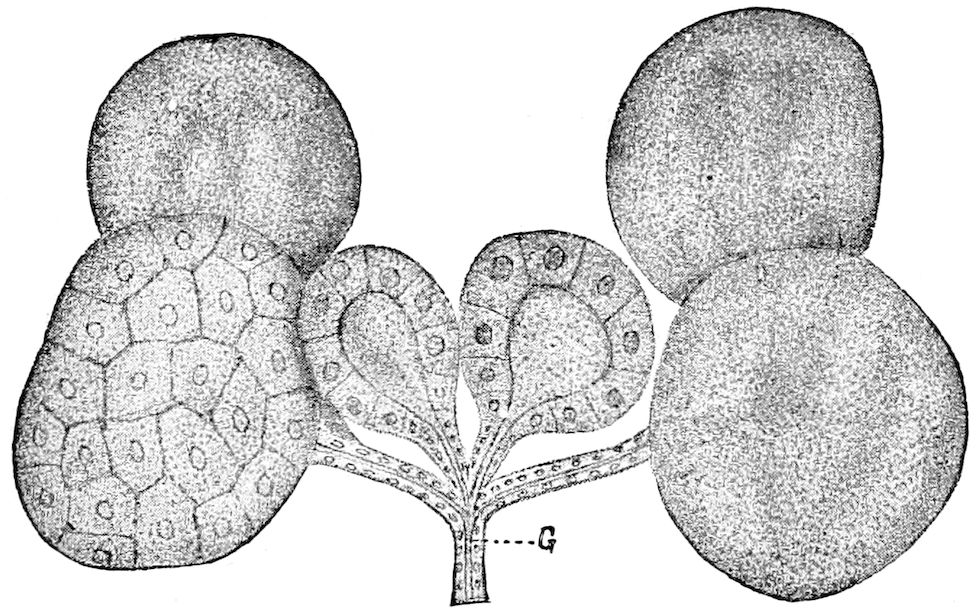
Fig. 326.—Acinous salivary glands of Orthezia cataphracta. In some acini the nuclei and boundaries of the cells are shown.—After List, from Field’s Hertwig.
System I is situated in the head, and consists of unicellular glands; the duct from each cell leads into a common, strongly chitinized duct, opening into the gullet.
System II, composed of acinose glands, lies also in the head; its duct is united with that of System III, situated in the thorax. (Fig. 329, 2, 3.)
System IV is situated at the base of the upper surface of the mandibles, and forms a delicate sac lined within with glandular cells; its duct opens at the insertion of the mandibles.
System V lies in the beak, and is a single gland consisting of unicellular glands; it opens into the common opening of Systems II and III. This system is wanting in the honey-bee, but occurs in Bombus and other genera.
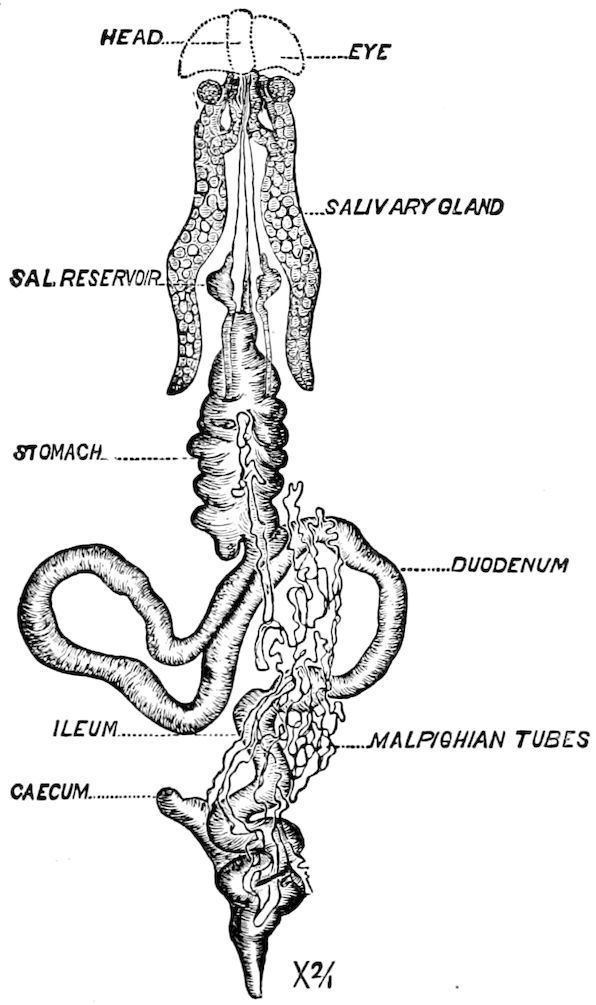
Fig. 327.—Appendages of digestive canal of Belostoma.—After Locy.
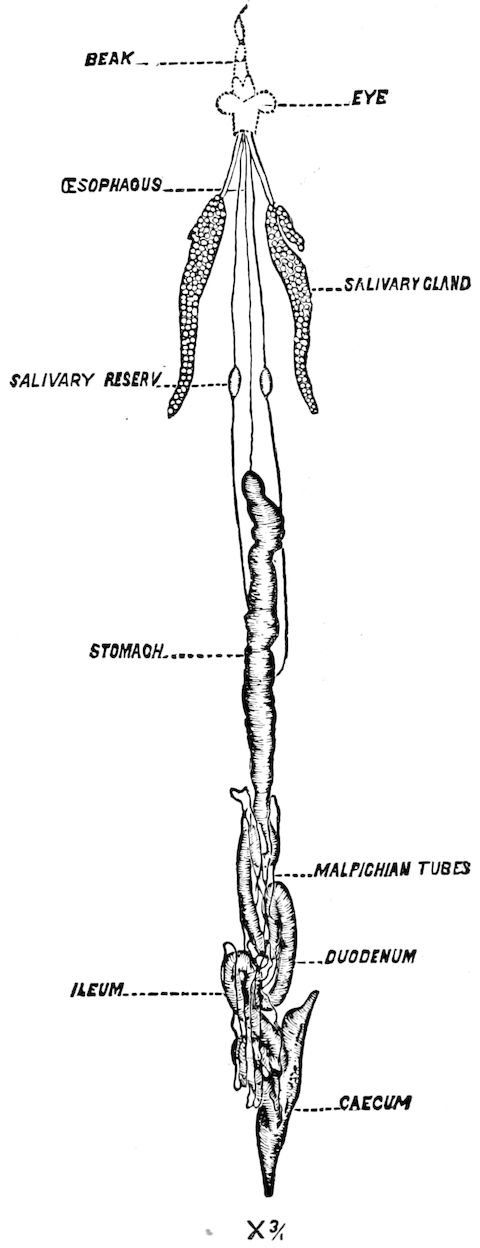
Fig. 328.—Salivary and other glands of Ranatra.—After Locy.
In all the five systems there constantly occur three cellular layers: the intima, epithelial, and propria. As regards their origin Schiemenz states that Systems I and IV are new structures, that System III arises in part, and Systems II and V wholly, from the silk-glands of the larva. As the glands differ much in the sexes, and in different species and genera, Schiemenz believes that their function is very manifold.
In addition to those previously discovered by Schiementz, Bordas has detected two additional pairs of salivary glands in the worker and male honey-bee, i.e. the internal mandibular and sublingual glands, so that in Apis there are in all six pairs, and apparently one unpaired.
The delicate chitinous external layer of the gland is perforated by many very fine pores through which the salivary fluid secreted 336by the epithelial cells passes into the salivary duct. The glands are externally bathed by the blood.
In many insects, including lepidopterous larvæ, the single median opening of the salivary duct is converted into a spraying apparatus.
In the adult Lepidoptera, according to Kirbach:—
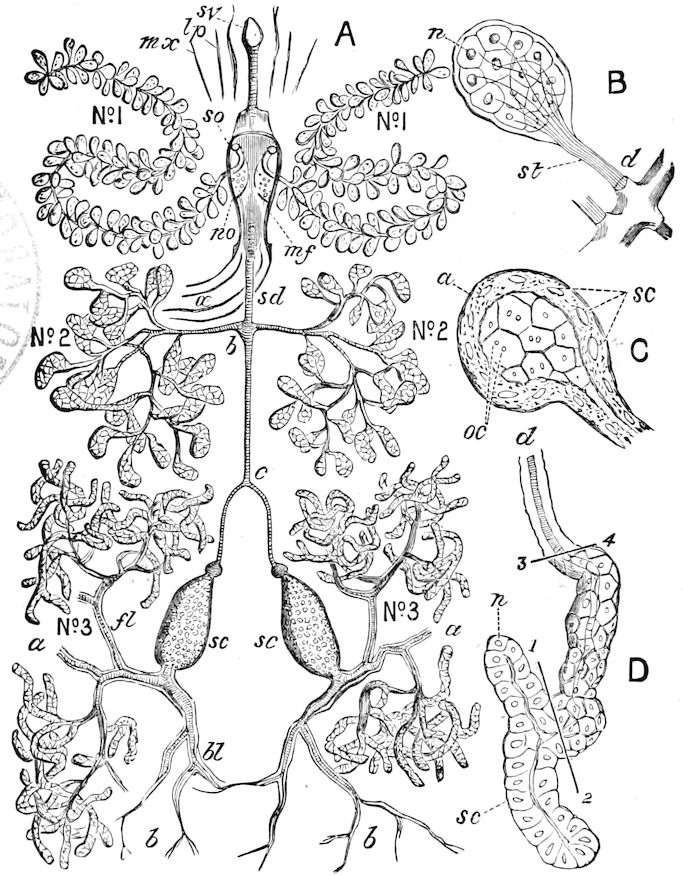
Fig. 329.—Salivary glands of the honey-bee: systems No. 1–3, × 15: sv, salivary valve (of systems 2 and 3) at base of tongue; lp, labial palpus; mx, maxilla; so, salivary opening of system 1 in hypopharyngeal plate; no, openings in plate for termination of taste-nerve; œ, œsophagus; sd, salivary duct; b, junction of ducts of system No. 2; c, junction of ducts of system No. 3; sc, sc, salivary sacs; fl, front lobe; bl, back lobe; a, chitinous duct, with spiral thread. B, single acinus of system No. 1, × 70: n, nucleus; st, salivary tract; d, large duct. C, single pouch, or acinus, from system No. 2: a, propria or outer membrane; sc, secreting cells. D, termination of system No. 3:1,2,3,4, lines marking end of section; d, duct in section; sc, secreting cells in section; n, nucleus.—After Cheshire.
“Its lower half forms a thick chitinous gutter, with a concave cover above, in which the similarly shaped upper half lies encased, so that between the two only a small semicircular opening remains. Powerful muscles extend from the cover to the lower side and to the two ridges of the bottom plate; through their contraction the upper channel is elevated, and presses out of the hinder part of the ducts into the space thus formed a great quantity of the saliva, which by allowing the contraction of the cover-muscle through the crevice-like opening, which is situated in the lower edge of the mouth-opening, becomes 337squeezed out in order either to mix with the fluid where the 2d maxillæ fuse, passing up into the canal in the proboscis, or to penetrate into and thus dilute the semi-fluid or solid substances taken, into the proboscis.”
The morphology and general relations of the salivary glands have been sketched out by Hatschek, Patten, and by Lucas, from observations on those of the case-worms or larval Trichoptera.
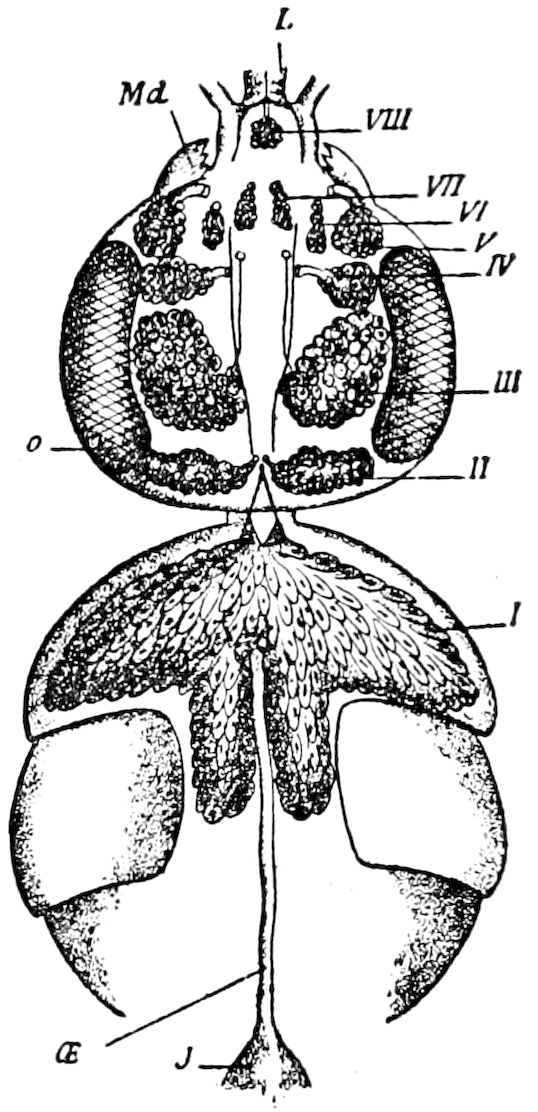
Fig. 330.—Eight pairs of glands of Andrena: I, thoracic; II, postcerebral; III, supracerebral; IV, lateropharyngeal; V, mandibular; VI, internomandibular; VII, sublingual; VIII, lingual; Md, mandible; L, tongue; o, eye; œ, œsophagus; J, honey-sac.—After Bordas.
Patten states that the spinning-glands in Neophylax are formed by a pair of ectodermal invaginations on the ventral side of the embryo, between the base of the 2d maxillæ and the nervous cord. They increase rapidly in length, and “they also unite to form a common duct, which opens at the end of the upper lip.”
The salivary glands in the same insect are “formed by invagination of the ectoderm on the inner sides of the mandibles, in the same manner as are the spinning glands.”
Lucas has shown that in trichopterous larvæ (Anabolia) there are three pairs of salivary glands in the head, which are serially arranged. The first pair belong to the mandibular, the second pair to the 1st maxillary, and the third pair, or spinning glands, to the 2d maxillary segment. The first or mandibular glands open into the mouth at the base of the mandibles directly behind the dorsal condyle. The second pair open between the 1st and 2d maxillæ; at the base of the latter, near the ventral condyle of the mandibles. The third pair open into the hypopharynx, which is modified to form the spinneret. Lucas agrees with Korschelt in regarding them as modified coxal glands, Schiemenz having previously regarded the headglands of the imago of the bee as belonging to the segments bearing the three pairs of buccal appendages, so that each segment originally contained a pair of glands. It is thus proven that the silk-glands are modified salivary glands adapted to the needs of spinning larvæ, and indeed in the imago the sericteries revert to their primitive shape and use as salivary glands.
The serial arrangement of the salivary glands in the Hymenoptera, where the number varies from five to ten pairs, is clearly proved by Bordas. He has detected five more pairs than were previously known, and names the whole series as follows:—1, the thoracic salivary glands, which are larger than the others, and nine other pairs, which are all contained in the head as follows: 2, postcerebral; 3, supracerebral; 4, lateropharyngeal; 5, mandibular; 6, internomandibular, situated on the inner side of the base of mandible; 7, sublingual; 8, lingual (these and 1 to 7 common to all Hymenoptera); 9, paraglossal (in Vespidæ); 10, maxillary (very distinct in most wasps). These glands do not all occur in the same species, being more or less atrophied.
Bordas further shows the segmental arrangement of the cephalic glands by stating that the supracerebral glands correspond to the antennal segment, the sublingual glands to the labial, the mandibular glands (external and internal) 338to the mandibular segment, the maxillary glands to the 1st maxillary segment, the lingual glands to the 2d maxillary segment, while the thoracic and postcerebral salivary glands, he thinks, correspond to the ocular segment, a view with which we are indisposed to agree, although conceding that each of the six segments of the head has in it at least one pair of salivary glands.
Functions of the different salivary glands in Hymenoptera.—The secretion of the thoracic glands is feebly alkaline. The postcerebral salivary glands, considered by Ramdohr to be organs of smell, secrete, like the preceding, a distinctively alkaline fluid, which mingles with the products of the thoracic glands. The supracerebral glands, also equally well developed in all Hymenoptera, though much atrophied in the females and especially the males of Apis mellifica, also in the Vespinæ and Polistinæ, secrete an abundant, feebly acid liquid, which is actively concerned in digestion.
As to the mandibular glands, which Wolf supposed to be olfactory organs, their acid secretion, though smelling strongly, acts energetically on the food as soon as introduced into the mouth.
The sublingual glands, atrophied in most Apidæ, but relatively voluminous in Sphegidæ, Vespinæ, Polistinæ, Crabronidæ, etc., empty their secretion into a small prebuccal excavation, where accumulate vegetable and earthy matters collected by the tongue, and the saliva secreted by these glands, acts upon them before they pass into the pharynx. The lingual glands secrete a thick, sticky liquid, which causes foreign bodies to adhere to the tongue, and also agglutinates alimentary substances. The uses of the other glands, maxillary and paraglossal, are from their minuteness undetermined. (Bordas.)
LITERATURE ON THE SALIVARY GLANDS
Leydig, F. Zur Anatomie der Insekten. (Archiv Anat. und Phys. 1859.)
—— Untersuchungen zur Anatomie und Histiologie der Tiere. Bonn, 1883, pp. 174, 8 Taf.
—— Intra- und interzellulare Gänge. (Biolog. Centralblatt, x, 1890, pp. 392–396.)
Dohrn, A. Zur Anatomie der Hemipteren. (Stettin. Entom. Zeit., 1866, salivary glands, pp. 328–332.)
Kupffer, C. Die Speicheldrüsen von Periplaneta orientalis und ihr Nervenapparat. (Beiträge zur Anatomie und Physiol., 1875.)
Schiemenz, P. Ueber das Herkommen des Futtersaftes und die Speicheldrüsen der Biene. (Zeitschr. f. wissens. Zool., xxxviii, 1883, pp. 71–135, 3 Taf.)
Korschelt, E. Ueber die eigentümlichen Bildungen in den Zellkernen der Speicheldrüsen von Chironomus plumosus. (Zool. Anzeiger, 1884, pp. 189–194, 221–225, 241–246.)
Hofer, B. Untersuchungen über den Bau der Speicheldrüsen und des dazu gehörenden Nervenapparates von Blatta. (Nova Acta d. Kais. Leopold.-Carol. Deutsch. Akad. d. Naturforscher, li, 1887, pp. 345–395, 3 Taf.)
Knüppel, A. Ueber Speicheldrüsen von Insekten. (Archiv für Naturg., 1887, Jahrg. 52, pp. 269–303, 2 Taf.)
Blanc, Louis. La tête du Bombyx mori à l’état larvaire, anatomie et physiologie. (Extrait des Travaux du Laboratoire d’Études de la Soie, 1889–1890; Lyon, 1891, p. 180, many figs.)
Bordas, L. Anatomie des glandes salivaires des Hyménoptères de la famille des Ichneumonidæ. (Zool. Anzeiger, 1894, pp. 131–133.)
—— Glandes salivaires des Apides, Apis mellifica. ♂ and ♀. (Comptes rendus Acad. Sc., Paris, cxix, pp. 363, 483, 693–695, 1894; also two articles in Bull. Soc. Philomath. Paris, 1894, pp. 5, 12, 66.)
339—— Appareil glandulaire des Hyménoptères. (Ann. Sc. Nat. Zool., xix, Paris, 1894, pp. 1–362, 11 Pls.) (See also p. 366.)
Berlese, Antonio. Le cocciniglie Italiane viventi sugli agrumi. Firenze, 1896, 12 Pls. and 200 Figs.
With the writings of Mark, Minot, Locy, List, Krassilstschik, Nagel (1896).
b. The silk or spinning glands, and the spinning apparatus
The larvæ of certain insects, chiefly those of the Lepidoptera, possess a pair of silk or spinning glands (sericteries) which unite to form a single duct opening in the upper lip at the end of the lingua, which is modified to form the spinneret. (See pp. 71, 75.) All caterpillars possess them, and they are best developed in the silkworms, which spin the most complete cocoon. Silk-glands also occur in the larvæ of the Tenthredinidæ, in the case-worms or larval Trichoptera, also in certain chrysomelid beetles (Donacia, Hæmonia), and in a weevil (Hypera). In a common caddis-worm (Limnophilus) the glands are of a beautiful pale violet-blue tint, and two and a half times as long as the larva itself; viz. the body is 20 mm. and the glands 55 mm. in length.
In caterpillars the glands are of tubular shape, shining white, and much like the ordinary simple tubular salivary glands of the imago. When only slightly longer than the body they are twice folded, the folds parallel and situated partly beneath and partly on the side of the digestive canal; not usually, when folded in their natural position, extending much behind the end of the stomach; but in the silkworms they are so long and folded as to envelop the hinder part of the canal. In geometrid caterpillars the glands when stretched out only reach slightly beyond the end of the body; in Datana they are half again as long as the body. Helm thus gives their relative length in certain Eurasian caterpillars, and we add that of Telea polyphemus:—
| Vanessa io | length of body | 32 mm.; of the silk glands | 26 mm. |
| Smerinthus tiliæ | length of body | 63 mm.; of the silk glands | 205 mm. |
| Bombyx mori | length of body | 56 mm.; of the silk glands | 262 mm. |
| Antheræa yamamaya | length of body | 100 mm.; of the silk glands | 625 mm. |
| Telea polyphemus | length of body | 60 mm.; of the silk glands | 450 mm. |
Thus in Telea the silk-glands are about 18.50 inches in length, being about seven times as long as the body.
For the most complete accounts of the spinning glands of Lepidoptera and their mechanism we are indebted to Helm and to Blanc, and for that of the Trichoptera to Gilson.
340The unpaired portion, or spinning apparatus (filière of Lyonet), is divided into two portions; the hinder half being the “thread-press,” the anterior division the “directing tubes.” The silk material, stored up in the thickest portion of the glands, passes into the thread-press (Fig. 334, A), which is provided with muscles which force the two double ribbon-like threads through the directing tube, as wire is made by molten iron being driven through an iron plate perforated with fine holes. The entire spinning apparatus, or filator, as we may call it, is situated in the tubular spinneret. The opening of the spinneret is directed anteriorly, and the anterior end of the directing tube passes directly into this opening so that the directing tube may be regarded as an invagination of the lingua.
The silk thread which issues from the mouth of the spinneret is, as Leeuwenhoek discovered, a double ribbon-like band, as may be seen in examining the silk of any cocoon.
The process of spinning.—Since the appearance of Helm’s account, Gilson, and also Blanc, have added to our knowledge of the way in which the silk is spun and of the mechanism of the process. Gilson has arrived, in regard to the function of the press or filator, at the following conclusions: 1, the press regulates the thread, it compresses it, gives it its flattened shape; 2, it regulates the layer of gum[52] (grès) which surrounds the thread; 3, it may render the thread immovable by compressing it as if held by pincers.
The process of spinning in the silkworm, says Blanc, comprises all the phenomena by which the mass of silk contained in the reservoir is transformed into the silk fluid of which the cocoon is spun. The excretory canals each contain a cylindrical thread of silk having a mean diameter of 0.2 mm. and surrounded by a layer of gum (grès) which in the fresh living organ exactly fills the annular space situated between the fibroin cylinder and the wall. Arrived within the common duct, the two threads receive the secretion of Filippi’s gland, where the silken fluid is formed, but has not yet assumed its definite external characters. The two threads press through the common canal and arrive at the infundibulum (Fig. 334, c) of the press, at 341the bottom of which is situated the orifice of the spinning canal, almost completely divided into two by the sharp edge of the rachis (Figs. 334, a, 335, l). The threads each pass into one of the two grooves, and the layer of gum (grès) fills the rest of the canal of the press or filator.
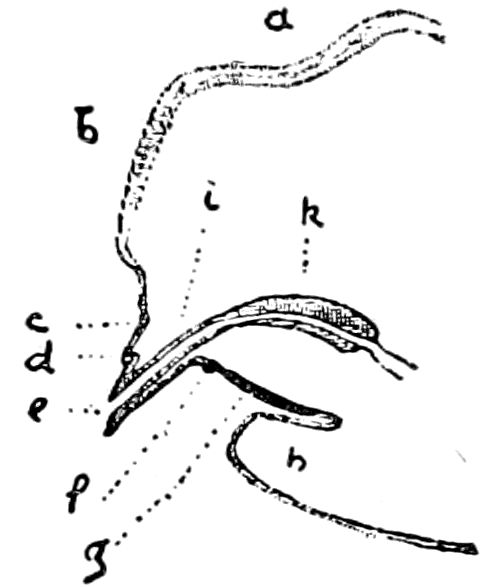
Fig. 331.—Longitudinal section of the spinneret: a, horizontal portion of the tongue; b, vertical portion; c, f, circle of the tongue; d, tongue-pad; e, orifice of the spinneret; g, body of the lyre; h, prebasilar membrane forming a fold; i, internal canal of the spinneret; k, filator.—After Blanc.
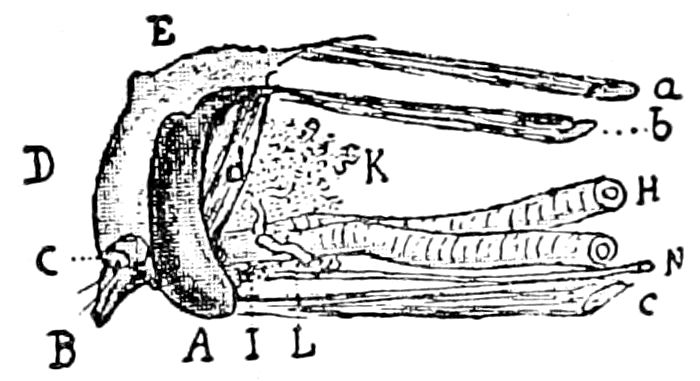
Fig. 332.—The lower lip (labium) of Bombyx mori, isolated, seen from the left side: A, lyre; B, spinneret; C, labial palpus; D, vertical part of the labium; E, horizontal part of the same; H, L, silk-canal; K, right gland of Filippi; L, canal of the left gland; N, labial nerve; a, oblique fibre of the elevator of the labium; b, right fibre of the same; c, depressor of the labium; d, superior spinning muscles.
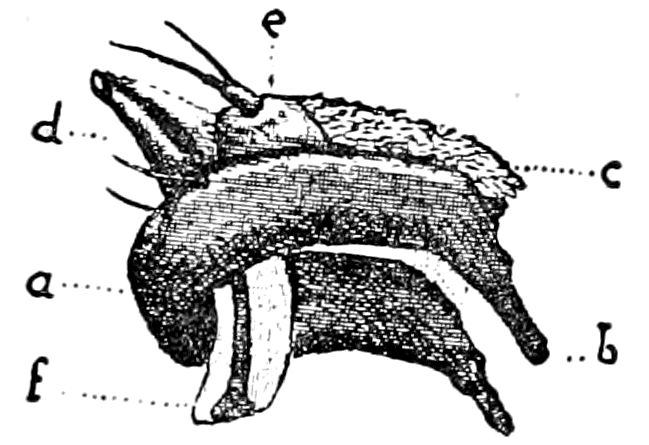
Fig. 333.—The labium in a horizontal position, seen from the side: f, the filator or press situated under the external part of the spinneret (d), between the branches (b), of the lyre (a); e, labial palp; c, tongue.
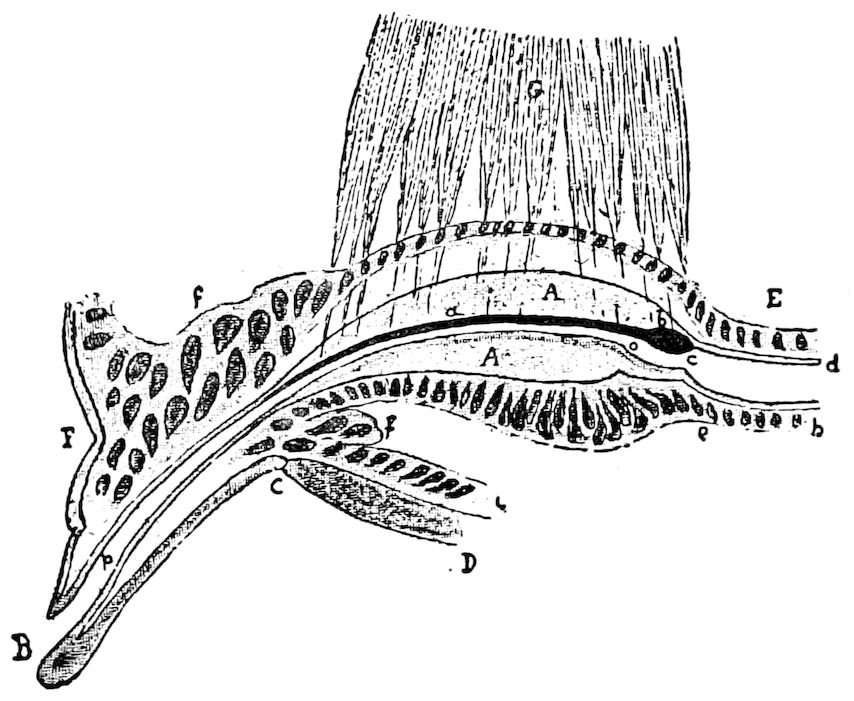
Fig. 334.—Longitudinal section of the spinneret and press (filator): A, filator or press; B, spinneret; C–D, body of the lyre; F, lower part of the labium; E, common canal; eh, its epithelium; G, superior muscle of the press; a, rachis; b, its posterior enlargement; c, infundibulum; d, cuticle; o, orifice of the spinning canal; op, central canal of the lyre and of the spinneret; fi, hypodermis of the lyre; f, f, hypodermic pad of the lyre.
The silken substance is then pressed by the more or less powerful contractions of the muscles of the filator, so 342that the passage of the threads is facilitated. If the muscles totally contract, the spinning canal is opened wide, the threads pass easily upwards and assume the form of a triangular prism (Fig. 336).
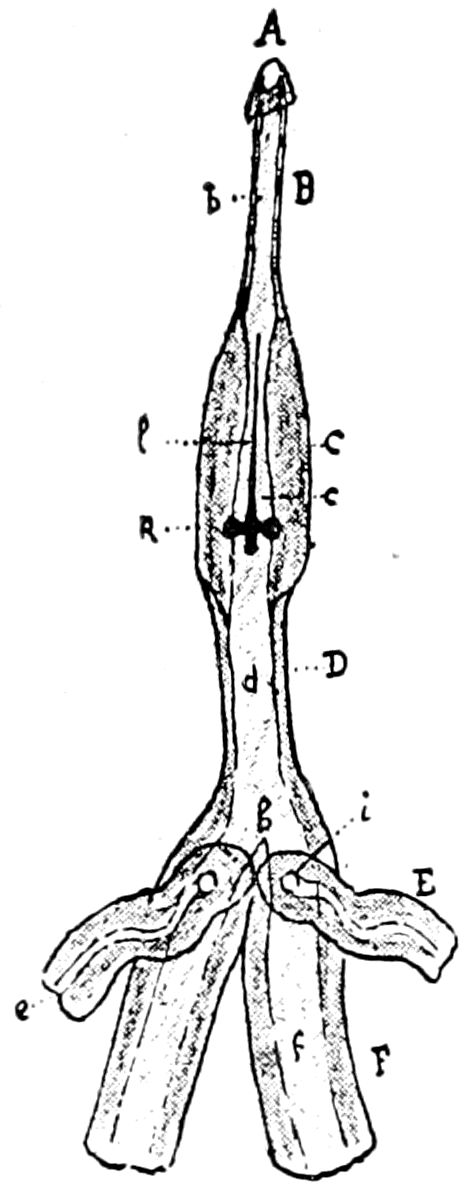
Fig. 335.—Spinning apparatus, seen from above: A, opening of the spinneret; B, central canal of the spinneret (C); D, common canal; E, canal of Filippi; F, excretory canal of a silk-gland; i, orifice of the canal of Filippi’s gland; l, rachis; k, ring of the infundibulum; b, c, d, e, f, cavity of the different canals; h, spur which separates the two excretory canals.—This and Figs. 331–334 after Blanc.
If this contraction diminishes, the chitinous wall of the spinneret comes together, owing to its elasticity; the ceiling of the canal approaches the floor; the cavity tends to take the form of a semicircular slit, and the threads are compressed, flattened. As each mass or thread of silk is much more voluminous than the canal, except when the latter is extremely dilated, it follows that the two threads are always compressed, or squeezed together, and that each of them is compelled to mould itself in the groove it occupies and to take its shape. Hence the variations in the appearance of the two masses or divided portions of silk, which as stated present all grades between the form of an isosceles-triangular prism and that of a nearly flat ribbon; but this last case is quite rare. The use of the spinneret, then, is to compress the thread and to change its form more or less considerably, at the same time as it diminishes its diameter.
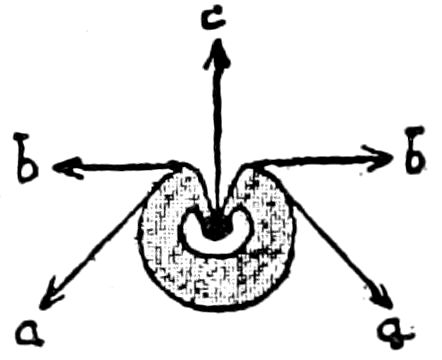
Fig. 336.—Diagram of the press and its muscles: a, lower; b, lateral; c, upper muscles of the press.—After Blanc.
Moreover, this constant compression of the thread as it passes through the press keeps it in a certain state of tension so as to allow the caterpillar while spinning to firmly hold its thread.
Finally, when the worm suspends the contraction of its spinning muscles, the press flattens, vigorously compresses the thread, and arrests its motion, in such a way that if there was a strain on the silken fluid (bave), it would break rather than oblige the caterpillar to let go any more of it.
The press does not act directly on the silken thread, but through the gummy layer (grès) which transmits over the whole surface of the silken fluid (brin) the pressure exerted on it. After having overcome this difficult passage, the silk thread has acquired its definite form; it rapidly passes out of the spinneret.
343How the thread is drawn out.—Having seen, says Blanc, how the two masses of silk (brins), in passing through the spinning apparatus (or press), join each other, constituting the frothy silken fluid, thus becoming modified in form, it remains to examine the way in which the thread is drawn out of the spinneret. If we examine a caterpillar while spinning, it will be seen that in moving its head it draws on the frothy mass of silk fixed to the web of the cocoon. This traction certainly aids very much the exit of the thread, but it is not the only cause.
The silk, Blanc affirms, is pushed out by a force a tergo, developed by different agents, such as the pressure of the distended cuticle or the silky mass contained in the reservoir, as seen in the section of a worm which has spun its cocoon. But if we consider a caterpillar before it has begun to spin, it is difficult to explain the mechanism of spinning. As Blanc has often observed, in making sections of the heads of silkworms, two cases arise. Sometimes the worm has already spun a little, and a certain length of the frothy silk (bave) issues from the orifice of the spinneret, where it forms a small twisted bundle. At other times the worm has not spun since its last moult or the frothy mass of silk has broken within the head, and we find the end in the common tube. In the first of these two cases, the worm, dilating its press, is able by a general contraction to discharge a little of the gritty material (grès) which lines the ball of silk hanging at the end of the spinneret. It can also reject a certain quantity of the secretion of Filippi’s glands and thus soften the gritty substance. The little plug of silk can then adhere to the body with which it comes in contact.
In the same case it is necessary that the two bits or portions of silk traverse the press, and this normally has a calibre less than their diameter. The worm should then distend the spinning tube as much as is practicable, so as to make the openings as large as possible. It has been stated that the press is, in this condition, at least as large as the mass of frothy silk. This Blanc believes (although Gilson thinks otherwise) is pushed by a force a tergo, and reaches the funnel of the spinning canal; its two bits of silk (brins) unite there, penetrate into the canal itself, and, owing to successive impulses produced by the general contractions of the worm, press through and pass out of the spinneret.
While the silkworm is engaged in spinning its cocoon, the spinneret and press execute very varied movements, determined by the elevator, depressor, retractor, and protractor muscles of the labium, as well as those of the press. These movements, originally very 344numerous, may combine among themselves, so that the spinneret is susceptible of assuming during the process of spinning still more diverse positions.
Fig. 337.—Portion of the silk-gland of Bombyx mori: p, tunica propria; i, tunica intima; s, secretion-cell with branched nuclei; a, separate secretion-cell from the anterior part of the silk-gland of Amphidasis betularia; b, the same of Vanessa urticæ; c, the same in Smerinthus tiliæ.—After Helm.
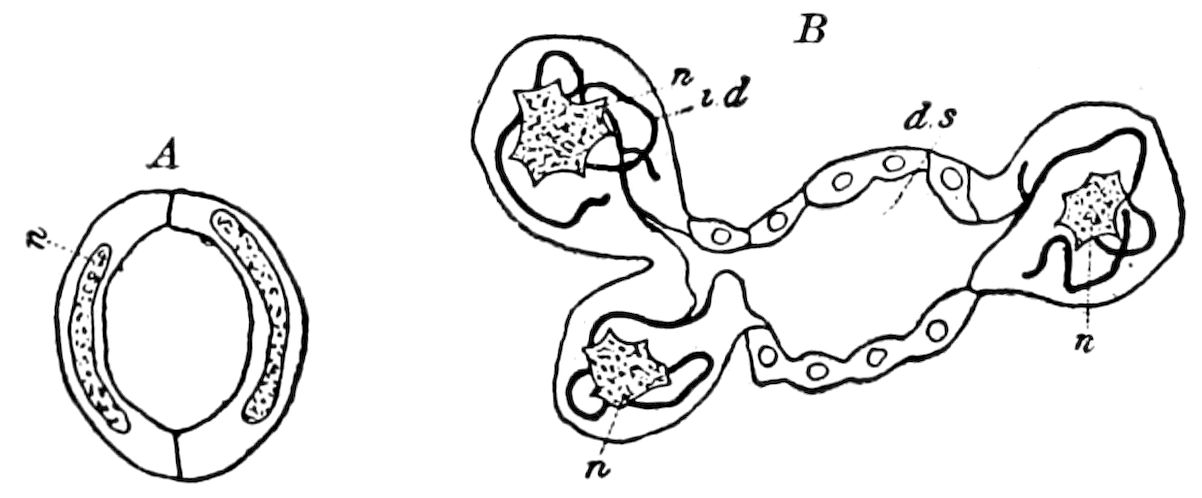
Fig. 338.—A, section of gland of lepidopter: B, section of silk-gland of a saw-fly larva; n, nucleus; i.d, canals; d.s, cavity.—After Gilson.
Histologically the silk-glands are composed of three layers,—the outer, or tunica propria (Fig 337); the inner, the tunica intima; the middle layer being composed of extraordinarily large epithelial cells which can be seen with the naked eye, and are also remarkable for the branched shape of the nuclei (a, b, c, 337), the branches being 345more or less lobed, and the larger the cells the more numerous are the branches of the nucleus. Gilson[53] finds that those of Trichoptera, Lepidoptera, Diptera, and Hymenoptera ordinarily consist of a small number of cells; and it is quite common, he says, to find only two cells in a transverse section (Fig. 338, A). In the Tenthredinidæ, however, “the organ still consists of a tube, the wall of which is composed of flat cells, but in addition to that, two series of spheroidal cells are attached to the sides. Each of these cells contains a system of tiny canals running through their cytoplasm (B, i. d). These cells are the secreting elements; they continually cast the silk substance into the tube.” A peculiarity of the tunica intima is its distinct transverse striation.
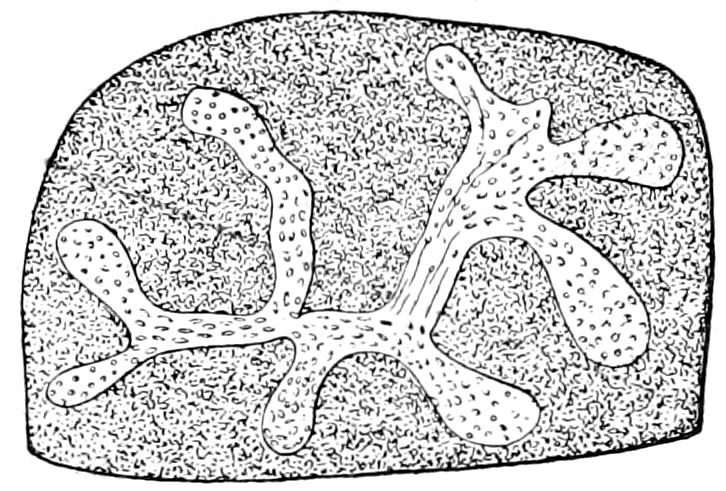
Fig. 339.—Branching nucleus of spinning gland of Pieris larva.—After Korschelt, from Wilson.
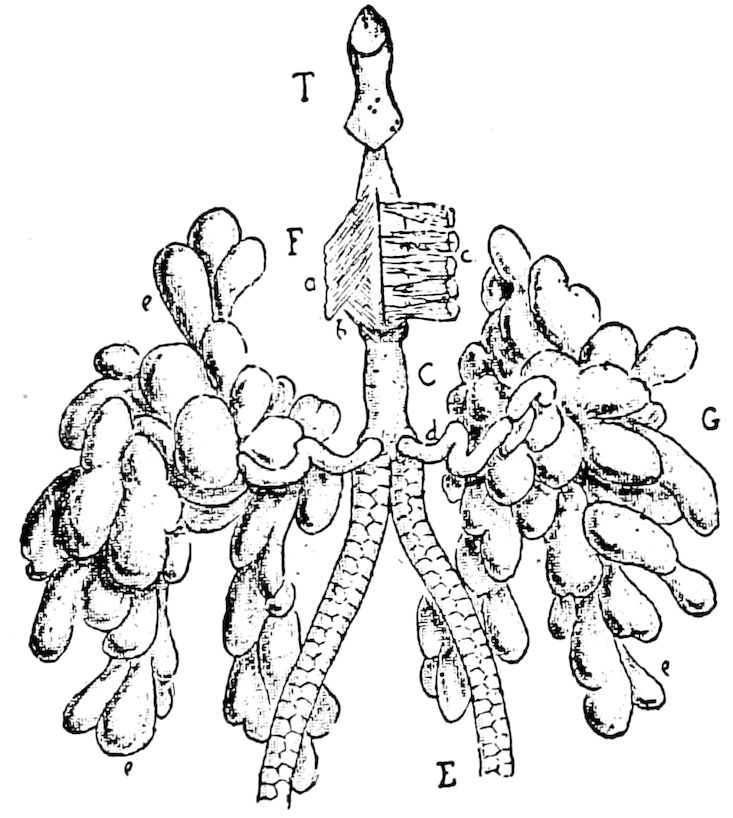
Fig. 340.—Filippi’s glands (G) isolated and seen from above: e, e, its lobules; d, its excretory canal; E, silk-duct; C, common canal; c, upper spinning muscle; b, lower muscle; a, lateral muscle; T, spinneret.—After Blanc.
The lining of the glands and of their common duct is moulted when the caterpillar casts its skin, and this, as well as the mode of development, shows that the glands are invaginations of the ectoderm. Gilson finds that the silk-glands and silk-apparatus of Trichoptera are very similar to those of caterpillars, and that the silk is formed in the same way.
Appendages of the silk-gland (Filippi’s glands).—In most larvæ there is either a single or a pair of secondary glands which open into the spinning glands near their anterior end. They are outgrowths of the gland provided with peculiarly modified excretory 346cells or evaginations of the entire glandular epithelium. Those of Bombyx mori (Fig. 340) are very well developed, and, according to Blanc, form two whitish, lobulated masses in the labium on each side of the common duct of the spinning gland. Externally they appear to be acinose; but their structure, as described by Blanc and by Gilson, is very peculiar. Helm thinks, with Cornalia, that the function of these glands is to secrete the adhesive fluid which unites the silk threads, and also to make the silk more adhesive in the process of spinning, but Blanc states that this is done before the thread passes into the common excretory tubes, and he is inclined to think that the secretion serves to lubricate the spinneret, and thus to facilitate the passage of the thread. On the other hand, in certain caterpillars these glands are situated quite far from the spinning apparatus.
The silk-glands in the pupa state undergo a process of degeneration, and finally completely disappear. They are specific larval organs evolved in adaptation to the necessity of the insect’s being protected during its pupal life by a cocoon. (Helm.)
Morphologically the silk-glands are by Lang regarded as modified coxal glands, and homologues of the setiparous parapodial glands of chætopod worms, the coxal glands of Peripatus, and the spinning glands of spiders.
In Scolopendrella, spinning glands are situated in the two last segments of the body, opening out at the end of the cercopods (Fig. 15, s.gl), and the larvæ of the true Neuroptera (Chrysopa, Myrmeleon, etc.) which spin cocoons, have spinning glands opening into the rectum. The silk forming the cocoon of the ant-lion, as Siebold and the older observers have stated, is secreted by the walls of the rectal or anal sac. Siebold (Anatomy of the Invertebrates, p. 445) states that in the larva of Myrmeleon, the silk-apparatus is very remarkable, “for the rectum itself is changed into a large sac and secretes this substance which escapes through an articulated spinneret projecting from the opening of the anus”[54] (Fig. 307, e). The larvæ of the Mycetophilidæ have spinning glands at the hinder end of the body, as also the imago of the female of the tineid moth Euplocamus. (Kennel.) The larvæ of ichneumons, wasps, bees, of Cecidomyia, and other Diptera, spin silken cocoons, but their glands have not yet been examined.
It should also be observed that during the process of pupation the larvæ of butterflies, of certain flies (Syrphus), and beetles (Coccinellidæ and some Chrysomelidæ) attach themselves by silk spun from the anus, so that the pupa is suspended by its tail; such glands are probably homogenetic with the coxal glands.
The silk in its fluid or soft state is mucilaginous, and according to Mulder, in the silkworm consists of the following substances, varying somewhat in their relative proportions by weight:
| Silk-fibre material | 53.67 |
| Glue (Leim) | 20.66 |
| Protoplasm | 24.43 |
| Wax | 1.39 |
| Coloring matter | 0.05 |
| Fat and resin | 0.10 |
LITERATURE ON THE SPINNING GLANDS
Helm, E. Anatomische und histiologische Darstellung der Spinndrüsen der Schmetterlingsraupen. (Zeitschr. f. wissens. Zool., xxvi, 1876, pp. 434–469, 2 Taf.)
Lidth de Jeude, Th. W. van. Zur Anatomie und Physiologie der Spinndrüsen der Seidenraupe. (Zool. Anzeiger, 1878, pp. 100–102.)
Engelmann, W. Zur Anatomie und Physiologie der Spinndrüsen der Seidenraupe. (Onderz. Phys. Lab. Utrecht, iii, 1880, pp. 115–119.)
Joseph, G. Vorläufige Mitteilung über Innervation und Entwickelung der Spinnorgane bei Insekten. (Zool. Anzeiger, 1880, pp. 326–328.)
Poletajew, N. Ueber die Spinndrüsen der Blattwespen. (Zool. Anzeiger, 1885, pp. 22–23.)
Meinert, Fr. Contribution à l’anatomie des fourmilions. (Overs. Danske Vidensk. Selsk. Forh. Kjöbenhavn, 1889, pp. 43–66, 2 Pls.)
Blanc, Louis. Étude sur la sécrétion de la soie et la structure du brin et de la bave dans le Bombyx mori. Lyon, 1889, pp. 48, 4 Pls.
—— La tête du Bombyx mori à l’état larvaire. Anatomie et physiologie. (Extrait du volume des Travaux du Laboratoire d’Études de la Soie. Années 1889–1890, Lyon, 1891, pp. 180, 95 figs.)
Gilson, G. Recherches sur les cellules sécrétantes. La soie et les appareils séricigènes: I. Lépidoptères. (La Cellule, 1890, vi, pp. 115–182, 3 Pls. I, Lépidoptères (suite); II, Trichoptères. Ibid., x, pp. 71–93, 1893, 1 Pl.)
Garman, H. Silk-spinning dipterous larvæ (Science, xx, 1893, p. 215).
Also the writings of Meckel, Pictet, Duméril, Klapálek, Wistinghausen, Loew, Hagen, Fritz Müller, Kolbe, McLachlan, de Selys-Longchamps.
c. The cæcal appendages.
These diverticula of the mid-intestine (“stomach”) are appended to the anterior end, and in the living, transparent larva of Sciara, which has two large, long, slender cœca (Fig. 341), the partly digested food may be seen oscillating back and forth from the anterior end of the stomach into and out of the base of each cæcum. In the Locustidæ (Anabrus, Fig. 299) and Gryllidæ (Fig. 344, e) there are two large, short cæca, and in the locusts (Caloptenus) there are six cæca, while cockroaches have eight. In the Coleoptera (Carabidæ and Dyticidæ) these large cæca appear to be replaced by very numerous slender, minute villi or tubules, which arise from the anterior part of the stomach (Figs. 317, r, also 342).
These cæca differ in structure from the stomach, as shown by Graber, as well as by Plateau and by Minot. The latter states that a single transverse section of one of the diverticula of the locust demonstrates at once that its structure is entirely different from that of the stomach.
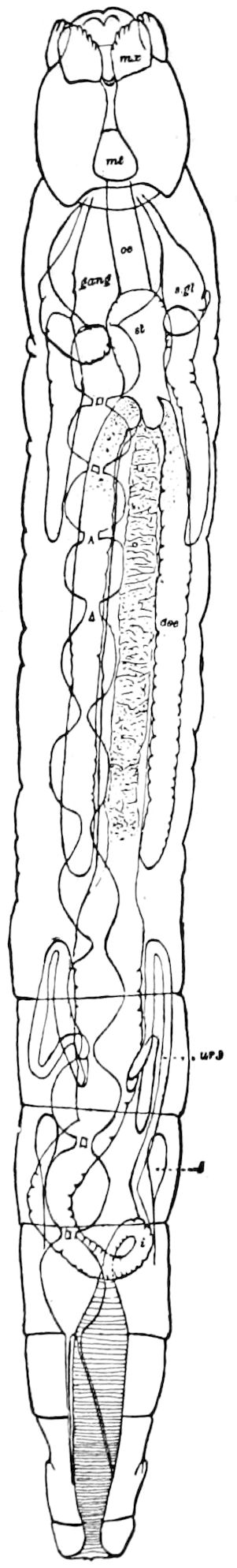
Fig. 341.—Larva of Sciara: s.gl, salivary gland; ur.t, urinary tubes; i intestine; st, stomach; cae cæcal appendages; t, testis.
Its inner surface is thrown up into longitudinal folds, generally twelve in number. These folds shine through the outer walls, and are accordingly indicated in the drawings of Dufour, Graber, and others. The entire cæcum has an external muscular envelope, outside of which are a few isolated longitudinal muscular bands. The folds within are formed mainly by the high cylindrical epithelium which lines the whole interior of the cavity. Tracheæ ramify throughout all the layers outside the epithelium. There are appearances of glandular follicles in the bottom of the spaces between the folds. (Minot.)
Burmeister supposed that these cæca were analogous to the pancreas, and this view has been confirmed by Hoppe Seyler, Krukenberg, Plateau, and others, who claim that the digestive properties of the fluid secreted in them agrees with the pancreatic fluid of vertebrates.

Fig. 342—Cross-section of mid-intestine of Acilius sulcatus, showing the arrangement of the cæca, two tracheæ passing into each cæcum.—After Plateau.
d. The excretory system (urinary or Malpighian tubes)
The excretory matters or waste products of the blood tissue of worms are carried out of the body by segmentally arranged tubes called nephridia. As a rule they arise in the blood sinuses of the body and open externally through minute openings in the skin. As there is a pair to each segment (in certain oligochete worms two or three pairs to a segment), they are often called segmental organs. In the annulate worms each segment of the body, even the cephalic or oral segment, originally contains a pair of these excretory organs. These 349vessels may have survived in myriopods and perhaps do exist in insects as urinary tubes, and also occur in many of the Arachnida, and thus are characteristic of each important class of land arthropods, but are either wanting or are very rudimentary or much modified in the marine classes, notably the Crustacea and Merostomata (Limulus), where they are represented by the shell-glands of Copepoda, green glands of the lobster, and the brick-red glands of Limulus.
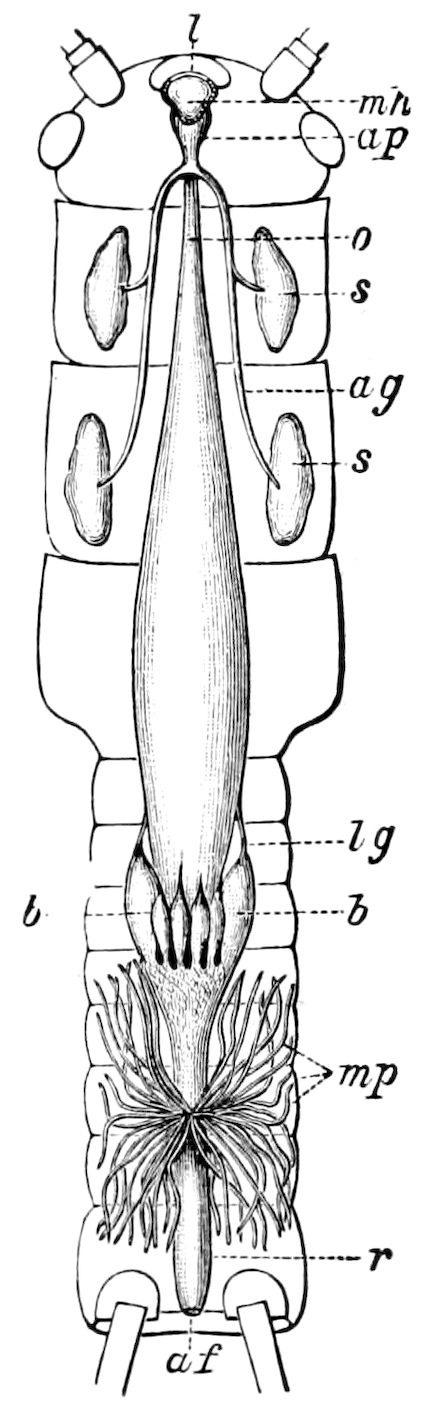
Fig. 343.—Digestive canal of Perla maxima: l, upper lip; mh, buccal cavity; ap, common end of salivary ducts (ag); o, œsophagus; s, s, salivary glands, arranged segmentally; b, cæca of chyle-stomach; lg, their ligaments of attachment; mp, urinary tubes; r, rectum; af, anal orifice.—After Imhof, from Sharp.
In the earliest tracheate arthropod, Peripatus, these tubes are well developed and are highly characteristic, each segment behind the head bearing a pair (Fig. 4, so4-so9). It has been suggested by some, but not yet proved, that the urinary tubes of insects are morphologically the same as the segmental organs of worms and of Peripatus; but there are no facts directly supporting this view, and, as Sograff states, it is a pure hypothesis and can only be confirmed or disproved by very detailed researches on the development of the urinary tubes of myriopods and of insects. Others regard them as probably homologous with the tracheæ, since they have a similar origin. As, however, they arise in the embryo as outgrowths of the proctodæum they may have arisen in myriopods and insects independently, and not be vermian heirlooms.
While in worms and in Peripatus a pair of these segmental organs occur in each segment, in insects this serial arrangement is not apparent; those with a purely excretory function are not segmentally arranged, with outlets opening externally, but arise as outgrowths of the hind-intestine or proctodæum of the embryo, not being segmentally arranged. The place of their origin is usually the dividing line between the mid and hind intestine (Fig. 343, mp); this applies to Scolopendrella (Fig. 15, urt) as well as to insects.
The urinary tubes are usually long, slender, blind, tubular glands varying in number from two to over a hundred, which generally arise at the constriction between the mid and hind intestine, and which lie loosely in the cavity of the body, often extending towards the head, and then ending near the rectum (Figs. 301, 310, vm). 350They were first discovered by the Italian anatomist Malpighi, after whom they were called the Malpighian tubes. While at first generally regarded as “biliary” tubes, they are now universally considered to be exclusively excretory organs, corresponding to the kidneys of the higher animals.
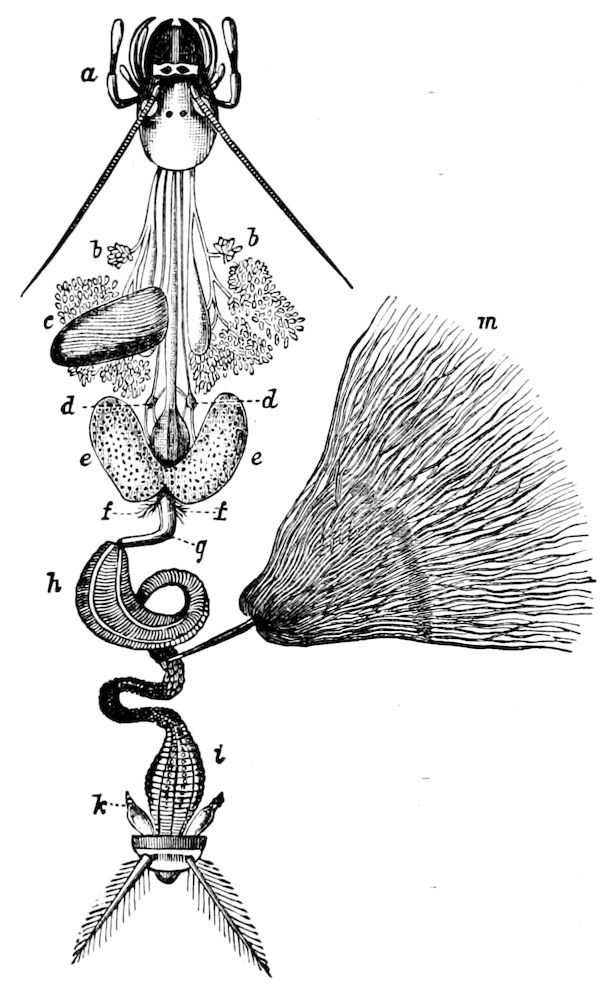
Fig. 344.—Digestive canal and appendages of the mole-cricket; a, head: b, salivary glands and receptacle; c, lateral pouch; d, stomatogastric nerves; e, anterior lobes of stomach; f, peculiar organ; g, neck of stomach; h, plicate part of same; i, rectum; k, anal gland; m, urinary tubes.—After Dufour, from Sharp.
Usually arising from the anterior end of the hind-intestine where it passes into the mid-intestine, in certain forms they shift their position, in some Hemiptera (Lygæus, Cimex) opening into the rectum, while in the Psyllidæ they arise from the slender hinder part of the mid-intestine, being widely separated at their origin. (Fig. 321.)
The length varies in different groups; where they are few in number (two to four, six to eight), they are very long, but where very numerous they are often short, forming dense tufts, each tuft connecting with the intestine by a common duct (ureter), or, as in the mole-cricket, the numerous tubes empty into a single duct (Fig. 344); in the locusts (Acrydiidæ), however, they are arranged in 10 groups, each group consisting of about 15 tubes, making about 150 in all; and are much convoluted and wound irregularly around the digestive canal, and when stretched out being about as long as the entire body.
The urinary tubes occur in twos, or in multiples of two, though a remarkable exception is presented in the dipterous genera Culex and Psychodes, in which there are five tubes; the young and fully grown larvæ, as well as the pupa and imago of Culex, having this number (Fig. 433, mg.)
351In many insects (Pentatoma, Cimex, Velia, Gerris, Haltica, Donacia, and often in caterpillars), the vessels open into a sort of urinary bladder connecting with the intestine on one side.
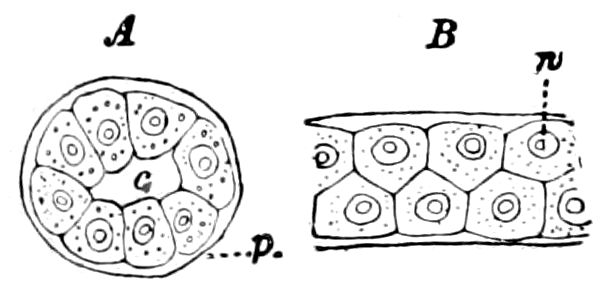
Fig. 345.—A, section of urinary tube of Periplaneta; B, part of tube of Perla; p, peritoneal membrane; c, cavity or lumen; n, nucleus of a secreting cell.—After Schindler.
In the larvæ of some insects the blind ends of the tubes are often externally bound to the rectum, in the silkworms being attached by fine threads to the intestine, while in some flies (Tipula and Ctenophora), two vessels may unite to form a loop. In all larval Cecidomyiæ, the two tubes are united to form a loop which curves backward, opening near the vent, the proctodæum being very short. (Giard.)
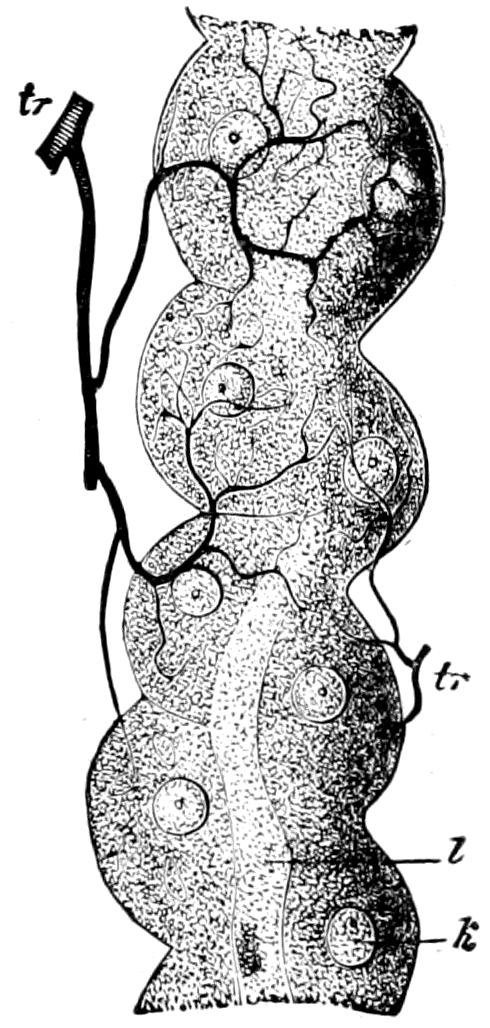
Fig. 346.—Portion of a urinary tube of Calliphora vomitoria: tr, trachea; l, lumen; k, nucleus.—After Gegenbaur.
While usually the urinary vessels form simple tubes, in many species of Lepidoptera and Diptera they are branched, thus resembling those of spiders and scorpions. Moreover, in many Lepidoptera and Diptera (Fig. 308), the tubes are not simple, but are lobulated, and in some Hemiptera (Pentatoma, Notonecta, and Tettigonia) are twisted or lace-like. In rare cases there are two kinds of urinary tubes; in Melolontha vulgaris, two of them are partly lobulated and yellow, while the other two are simple and white. Their color in beetles varies, some being whitish or yellowish; in Geotrupes, Dyticidæ, Hydrophilidæ, etc., reddish brown; in Gryllotalpa as well as Locusta viridissima, there are two different kinds of vessels, differing in contents and in color (white or yellow), as well as histologically. (Schindler.)
The exterior of the tubes is richly provided with tracheæ, which often form a web around them, and the fine branches often seem to attach them to the intestine. In Acheta they are enveloped by a very delicate, loose network of muscular fibres. (Schindler.)
The urinary tubes consist, according to Schindler, of at least three cellular layers (Fig. 345):—
1. An external, connective, nucleated membrane, the peritoneal membrane.
2. A very delicate homogeneous basal membrane, the tunica propria.
3. A single layer of large polygonal excretory cells.
4. Lining the internal canal a chitinous layer penetrated by pore-canals, the intima often wanting.
The secretory cells are usually of the same size, but in many cases are relatively small; sometimes four to six or more form the periphery of the canal, sometimes three or only two. In some insects the cells are so very large that a single cell forms the entire periphery. The nuclei in the Lepidoptera (Papilio, Pontia, Cossus) are large and irregularly branched.
The excretions of the Malpighian vessels, derived from the blood and from the fat-body, are more or less fluid and granular, sometimes pulpy. From the cells they pass into the canal, thence into the intestine, and thence out of the body. How, says Kolbe, the secretion passes into the intestine, whether by the contraction of the fine fibrillæ of the peritoneal membrane, or by the external pressure of the other organs, or by the pressure of the secretory matter behind, 352is not yet known. Grandis observed in living Hydrophilus that the urinary tubes moved, without the muscles seeming to show what caused the motion. Moreover, the cells incessantly changed their form. At a lower temperature such motions ceased. The tracheæ, ending freely in the cells, did not anastomose. (Kolbe.)
The different colors of the tubes (white, yellow, red, brown, or green) is due to the hue of the excretions, and is independent of the color of the blood and of the urinary substances held in the secreted matter.
Schindler found that insects of different stages, collected in winter, differed very much in their urinary secretions, the tubes in the adults being entirely empty, while in the larvæ they were filled full, so that he concluded that in the former the process of excretion during the winter hibernation is very slow, but in the latter very rapid.
As to the activity of the urinary vessels the following experiments will throw some light. Tursini fed a Pimelia with fuchsin; its urinary tubes were consequently colored red. Schindler fed insects with indigo-carmine, which was excreted by the urinary tubes; Kowalevsky arrived at the same results, which seems to prove that these vessels are analogous to the kidneys of vertebrates. Moreover, Schindler injected through the side of the first abdominal segment into the cavity of the body of a Gryllotalpa a concentrated solution of sodium salt of indigotin-disulphonic acid. After one or two hours the external portion of the epithelium of the urinary vessels was stained deep blue, while the inner portion remained of the normal transparency; the nuclei being for the most part deeply stained. Between one and two days after, the staining matter had not yet wholly passed through the central canal, the surface recently stained still appearing light blue.
The solid contents of the urinary tubes consist partly of crystals, which occur singly in the epithelial cells, or form scattered masses when situated in the central canal. Besides tabular rhombic crystals, there occur concretions which contain uric acid, and probably consist of urate of soda, also octahedral crystals of chloride of soda, and quadro-pyramidal crystals of oxalate of lime. Also acicular prisms occur; besides chloride of soda, phosphates, carbonate of lime, oxalate of lime in quantity, leucine, coloring matters, etc.; while the fluid secretion also contains urea (?), uric acid, and abundant urates; uric acid crystals were precipitated by the addition of acetic acid, and by adding hydrochloric acid crystals belonging to the dimetric system were formed. The often numerous spheroidal small granules are biurate of soda and biurate of ammonia. Pale, concentrically banded concretions are leucine pellets.
According to Kölliker the contents of the urinary vessels[55] in general are: (1) round granules of urate of soda and urate of ammonia; (2) oxalate of lime; 353and (3) pale transparent concretions of leucine. Crystals of taurin are also said to occur. (Claus’ Zoölogy, p. 531.)
Although uric acid is characteristic of the urinary tubes, yet sometimes it is wanting in them, while uric acid substances in quantity occur in the fat-body or in the mid-intestine.
In the living insect the urinary tubes remove urates from the blood; “the salts are condensed and crystallized in the epithelial cells, by whose dehiscence they pass into the central canals of the tubules and thence into the intestine.” (Miall and Denny.)
The process of excretion is carried on not only by the urinary tubes, but also, as Cuénot has recently shown (1896) in Orthoptera, by the pericardial cells and certain cells of the fat-bodies. In the last-named cells urates are stored throughout life; the pericardial cells apparently secrete but do not store waste products, which are finally eliminated by the urinary tubes, the latter constantly eliminating waste.
Primitive number of tubes.—Wheeler considers the primitive number of urinary tubules to be six, other authors regarding two pairs as the primary or typical number; and while Wheeler agrees that the more ancestral tracheate arthropods had but a single pair, Cholodkowsky supposes the primitive number in insects themselves to be a single pair. This view is strengthened by the fact that Scolopendrella has but a single pair (Fig. 15).
While Peripatus has no urinary tubes, in Myriopods a single pair arises, as in insects, from the hind-intestine.
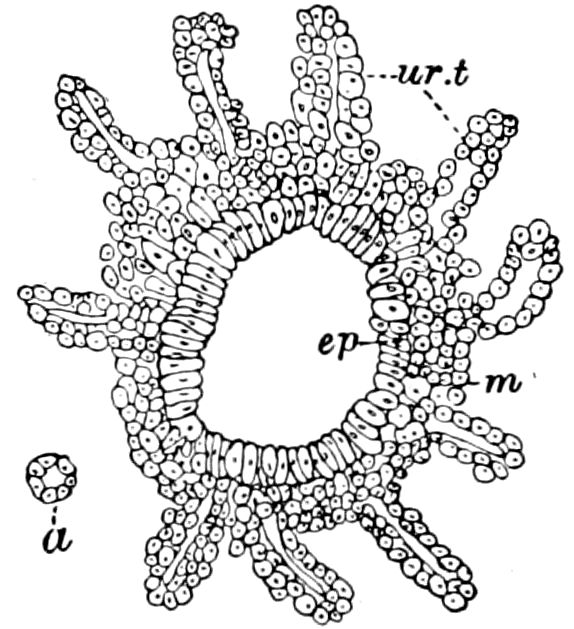
Fig. 347.—Section of proctodæum of embryo locust, showing origin of urinary tubes (ur.t); ep, epithelial or glandular layer; m, cells of outer or muscular layer; a, section of a tube.
When in insects the number of these tubes is few, they are, with rare exceptions, arranged in pairs, so that Gegenbaur and others have considered this paired arrangement as the primitive one. When the tubules are very numerous in the adult, as in Orthoptera, the embryos and larvæ have a much smaller number, Wheeler stating that “in no insect embryo have more than three pairs of these vessels been found.” We have observed 10 primary tubes in the embryo of Melanopus (Fig. 347), from each of which afterwards arise 15 secondary tubules. In the Termites, only, do the young forms have more urinary tubes than the adults.
In Campodea there are about 16 urinary tubes and in Machilis either 12 (Grassi) or 20 (Oudemans); but in other Thysanura the number is much less, Lepisma having either four, six, or eight, according to different authors, and both Nicoletia and Lepismina having six, opening separately into the hind-intestine. On the other hand, these organs have not yet been detected in Japyx. Whether they exist at all in the Collembola, which are degenerate forms, is doubtful. The weight of opinion denies their existence, though they may yet be found existing in a vestigial condition. They are said by Tullberg and by Sommer to exist in Podura, but are of peculiar shape.
Coming now to the winged insects, in what on the whole is perhaps the lowest or most generalized order, the Dermaptera, the number is over 30, and their insertions regularly encircle the intestine. (Schindler.) In the most ancient and generalized family of Orthoptera, the Blattidæ, Schindler detected from 60 to 70 tubes, but in a nymph of Periplaneta not quite 10 mm. in length he 354found from 16 to 18, and in nymphs 4 to 5 mm. long there were only eight vessels; while Wheeler has found in the embryo of Phyllodromia germanica but four tubes. In the adult Acrydiidæ there are as many as 150, in the Locustidæ between 40 and 50, and in the Gryllidæ about 100.
The Ephemeridæ with about 40, the Odonata with 50 to 60 tubules, the Perlidæ with from 50 to 60, are polynephrious; while the Termitidæ and Psocidæ are oligonephrious, the former having from six to eight and the Psocidæ only four tubes. So also all the other orders not mentioned, except the Hymenoptera, have few of these tubes. The Hemiptera, with none in Aphidæ, a single pair in the Coccidæ, and two in all the rest of the order, have the fewest number.
In the Neuroptera there are from six to eight, while in a larva, possibly that of Chauliodes, Wheeler finds the exceptional number of seven.
The closely allied order Mecoptera (Panorpidæ), and also the Trichoptera, agree with the Neuroptera (Sialis) in having six. According to Cholodkowsky all Lepidoptera have six of these vessels, except Galleria, which has but four. He finds that in Tinea biselliella (also T. pellionella and Blabophanes rusticella) the larva has six vessels, which, however, undergo histolysis during pupation, a single pair arising in their stead. On this account he regards the primitive number of urinary tubes as two, or a single pair, this return from six vessels in the larva to two in the imago being considered a case of atavism.
In the Coleoptera, the number of urinary tubes is from four to six; in what few embryo beetles have been examined (Doryphora, Melolontha), there are six vessels, but in the embryo of Dyticus fasciventris, Wheeler has detected only four, this number being retained in the adult. He thinks that in beetles in general, a pair of vessels must be “suppressed during post-embryonic development, presumably in early larval life.”
In Diptera and Siphonaptera, the number four is very constant, there being, however, a fifth one in Culex and Psychoda (Fig. 400.)
The number of these vessels is very inconstant in the Hymenoptera, varying from six (Tomognathus, an ant, worker) to 12 (Myrmica), and in Apis reaching the number of 150.
In the embryo of the honey-bee and wall-bee (Chalicodoma), there are only four; we still lack any knowledge of the number in embryo saw-flies.
The following is a tabular view of insects with few urinary tubes (Oligonephria) and many (Polynephria). It will be seen that the number has little relation to the classification or phylogeny, insects so distantly related as the Orthoptera and Hymenoptera being polynephrious:—
Here should be mentioned the singular fact discovered by Koulaguine that in the larva of Microgaster, the urinary tubes have no connection with the intestine, but open dorsally on the outside of the body on each side of the anus. Ratzeburg had stated that the last segment of the body was in the form of a vesicle. Koulaguine now shows that this vesicle is in reality the end of the intestine opening upwards; as the result of this dorsal opening of the intestine the Malpighian vessels open on the sides of the oval vent, and have no connection with the intestinal canal. Whether this is of morphological import, or is only a secondary adaptation, Koulaguine does not state, his paper being a preliminary abstract.
Wheeler thus sums up our present knowledge regarding the number and homologies of the Malpighian or urinary tubes:
1. It is very probable that the so-called Malpighian vessels of Crustacea and Arachnida are not the homologues of the vasa Malpighi of the Eutracheata (insects and myriopods).
2. The Malpighian vessels of the Eutracheata arise as paired diverticula of the hind-gut and are, therefore, ectodermal.
3. In no insect embryo are more than six vessels known to occur; although frequently only four are developed.
4. The number six occurs either during embryonic or post-embryonic life in members of the following groups: Apterygota, Orthoptera, Corrodentia; Neuroptera, Panorpata, Trichoptera, Coleoptera, Lepidoptera, and Hymenoptera.
5. The number four seems to be typical for the Corrodentia, Thysanoptera, Aphaniptera, Rhynchota, Diptera, and Hymenoptera.
6. The embryonic number in Dermaptera, Ephemeridea, Plecoptera, and Odonata has not been ascertained, but will probably be found to be either four or six.
7. There is evidence that in at least one case (Melolontha), the tetranephric is ontogenetically derived from the hexanephric condition by the suppression of one pair of tubules.
3568. It is probable that the insects which never develop more than four Malpighian vessels have lost a pair during their phylogeny.
9. The post-embryonic increase in the number of Malpighian vessels in some orders (Orthoptera, Odonata, Hymenoptera) is secondary and has apparently arisen to supply a demand for greater excreting surface.[56]
LITERATURE ON THE EXCRETORY (URINARY) ORGANS
Malpighi, M. Dissertatio epistolica de Bombyce, Societati regiæ Londini ad scientiam naturalem promovendam institutæ dicata. (Londini, 1669, 12 Pls.)
Herold, M. J. D. Entwicklungeschichte der Schmetterlinge. 1815.
Rengger, J. R. Physiologische Untersuchungen über den tierischen Haushalt der Insekten. Tübingen, 1817, pp. 82.
Wurzer. Chemische Untersuchungen des Stoffes in den Gallgefässen von Bombyx mori. (Meckel’s Archiv f. Physiol., iv, 1818, pp. 213–215.)
Gaede, H. M. Physiologische Bemerkungen über die sogenannten Gallgefässe der Insekten. (Nova Acta Acad. Caes. Leopold.-Carolin., 1821, x, Pars II, pp. 186–196.)
Meckel, J. F. Ueber die Gallen- und Harnorgane der Insekten. (Meckel’s Archiv, i, 1826, pp. 21–36.)
Audouin, J. V. Calculs trouvés dans les canaux biliaires d’un cerf volant. (Ann. sc. nat., 2 Sér., 1836, v, pp. 129–137.)
Frey und Leuckart. Anatomie und Physiologie der Wirbellosen. 1843.
Dufour, L. Mémoire sur les vaisseaux biliaires ou le foie des Insectes. (Ann. sc. nat., 1848, Sér. 2, xix, pp. 145–182, 4 Pls.)
Karsten, H. Harnorgane von Brachinus complanatus. (Müller’s Archiv f. Anat. und Physiol., 1848, pp. 367–374.)
Fabre, J. L. Étude sur l’instinct et les metamorphoses des Sphégiens. (Ann. d. sc. nat., 4 Sér., 1856, vi, pp. 137–189.)
—— Étude sur le rôle du tissu adipeux dans la sécrétion urinaire chez les Insectes. (Ibid., 4 Sér., xix, pp. 351–382.)
Schlossberger, J. E. Untersuchungen über das chemische Verhalten der Krystalle in den Malpighischen Gefässen der Raupen. (Archiv f. Anat. und Physiol., 1857, pp. 61–62.)
Leydig, F. Lehrbuch der Histiologie. 1857.
Sirodot, S. Recherches sur les sécrétions chez les Insectes. (Ann. sc. nat., 4 Sér., Zool., 1858, x, pp. 141–189, 251–334, 12 Pls.)
Kölliker, A. Zur feineren Anatomie der Insekten (Ueber die Harnorgane, u.s.w.) (Verhandl. d. Physikal.-medizin. Gesellsch. in Würzburg, viii, 1858, pp. 225–235.)
Schindler, E. Beitrage zur Kenntnis der Malpighischen Gefässe der Insekten. 3 Taf. (Zeitschr. f. wiss. Zool., xxx, 1878, pp. 587–660.)
Chatin, G. Note sur la structure du noyau dans les cellules marginales des tubes de Malpighi chez les Insectes et les Myriapodes. (Ann. d. sc. nat., 6 Sér., xiv., 1882, pp. 7, 1 Pl.)
Witlaczil, E. Zur Anatomie der Aphiden. (Arbeiten a. d. Zool. Instit. d. Univers. Wien., iv, 1882, pp. 397–441, 3 Taf.)
357Cholodkowsky, N. Sur les vaisseaux de Malpighi chez les Lépidoptères. (Compt. rend. Acad. d. Sc., Paris, xcix, 1884, pp. 631–633.)
—— Sur la morphologie de l’appareil urinaire des Lépidoptères. (Archives de Biologie, 1887, vi, pp. 497–514, 1 Pl.)
Loman, J. C. C. Ueber die morphologische Bedeutung der sogenannten Malpighischen Gefässe der echten Spinnen. (Tijdschr. Nederl. Dierk. Ver. (2) Deel 1, 1887, pp. 109–113, 4 Fig.)
Marchal, P. Contribution à l’étude de la désassimilation de l’azote. L’acide urique et la fonction rénale chez les Invertébrés. (Mém. Soc. Zool. de France, 1889, iii, pp. 42–57.)
Kowalevsky, A. O. Ein Beitrag zur Kenntnis der Exkretionsorgane. (Biol. Centralbl., ix, 1889–90, pp. 33–47, 65–76, 127–128.)
—— Sur les organes excréteurs chez les arthropodes terrestres. (Congrès international de Zool., 2me Session à Moscou, 1892, Pt. I, pp. 186–235, 4 Pls.)
Griffiths, A. B. On the Malpighian tubules of Libellula depressa. (Proc. Roy. Soc., Edinburgh, 1889, xv, pp. 401–403, Figs.)
Grandis, V. Sulle modificazioni degli epitelii ghiandolari durante la secrezione. (Atti Accad. Torino, 1890, xxv, pp. 765–789, 1 Pl.; Archiv Ital. Biol., 1890, xiv, pp. 160–182, 1 Pl.)
Koulaguine, N. Notice pour servire à l’histoire du développement des hyménoptères parasites. (Congrès internat. de Zool., 2me Session à Moscou, 1892, Pt. I, pp. 253–277.)
Sograff, Nicolas. Note sur l’origine et les parentés des Arthropodes, principalement des Arthropodes trachéates. (Congrès internat. de Zool., 2me Session à Moscou, 1892, Pt. I, pp. 278–302.)
Giard, Alfred. (Note on the urinary tubes of larval Cecidomyia. Annals Ent. Soc., France, lxii, 1893, pp. lxxx-lxxxiv, 1 Fig.)
Wheeler, William M. The primitive number of Malpighian vessels in insects. (Psyche, vi, May-December, 1893, Parts 1–6, pp. 457–460, 485–486, 497–498, 509–510, 539–541, 545–547, 561–564.)
Metalnikoff, C. K. Organes excréteurs des insectes. (Bull. Acad. imp. Sci. St. Pétersbourg, 1896, iv, pp. 57–72, in Russian, 1 Pl.)
See also the works of Straus-Dürckheim, Will (Müller’s Archiv. 1848, p. 502), Brugnatelli, Leidy, Dufour, Ramdohr, Basch, Davy, Grassi, Minot, Berlese, Adlerz, Marchal (Bull. Ent. Soc. France, 1896, p. 257); Bordas (Appareil glandulaire des Hyménoptères, 1894), also C. R. Acad. Sc. Paris, 1897.
e. Poison-glands
Poison-glands are mainly confined to the stinging Hymenoptera, i.e. certain ants, and the wasps and bees, but also occur in the mosquito, while many, if not most bugs, seem to instil a drop of poison into the punctured wounds they make.
In the honey and other bees the poison apparatus consists of two poison-glands whose secretion passes by a single more or less convoluted efferential duct into the large poison-sac, and thence by the excretory duct, which is enlarged at the base of the sting (Figs. 194, 195), out through the sting by the same passage as the eggs. According to Carlet, the poison apparatus of bees consists of two kinds of glandular organs, of which one kind secretes a feebly 358alkaline fluid, the other an acid product. The poison is only effective when both fluids are mixed. The resultant venom is always acid. The action of this venom upon some animals, as rabbits, frogs, and certain beetles, is slight; but the domestic fly and the flesh-fly are immediately killed by it. The inoculation of a fly with the secretion of one of the glands does not produce death until after a considerable time, but death follows very quickly if the same fly is subjected to a second inoculation, this time with the secretion of the other gland. The alkaline glands are in bees and all poisonous Hymenoptera strongly developed, but become vestigial in those forms which sting their prey to serve as food for their larvæ. The poison which the solitary sand and wood wasps and Pompilidæ inject into their victims only paralyzes them.
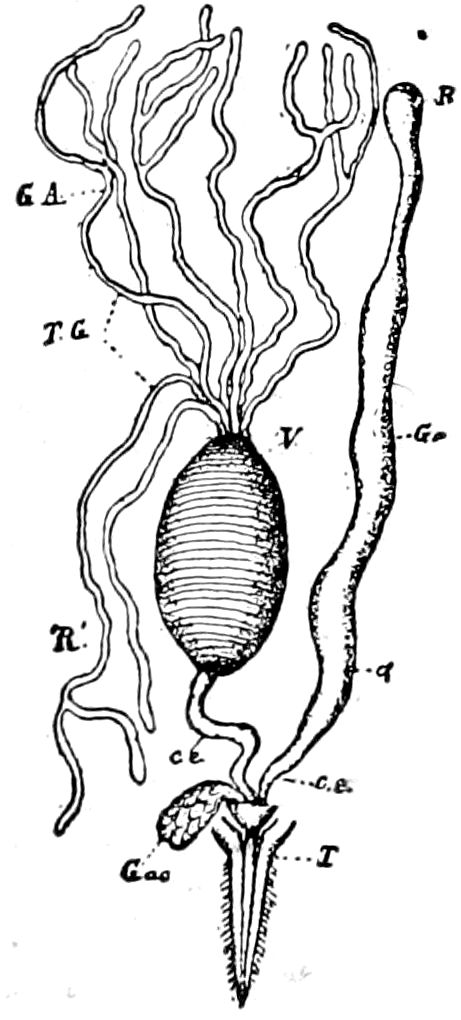
Fig. 348.—The poison apparatus of Ichneumon: T, sting; GA acid gland; TG, R′, its tubes opening into the common poison-sac or reservoir; ce, its efferent canal; Ga, the tubular alkaline gland; R, the glandular end; a, the reservoir; ce, its duct; Gac, the accessory gland—After Bordas.
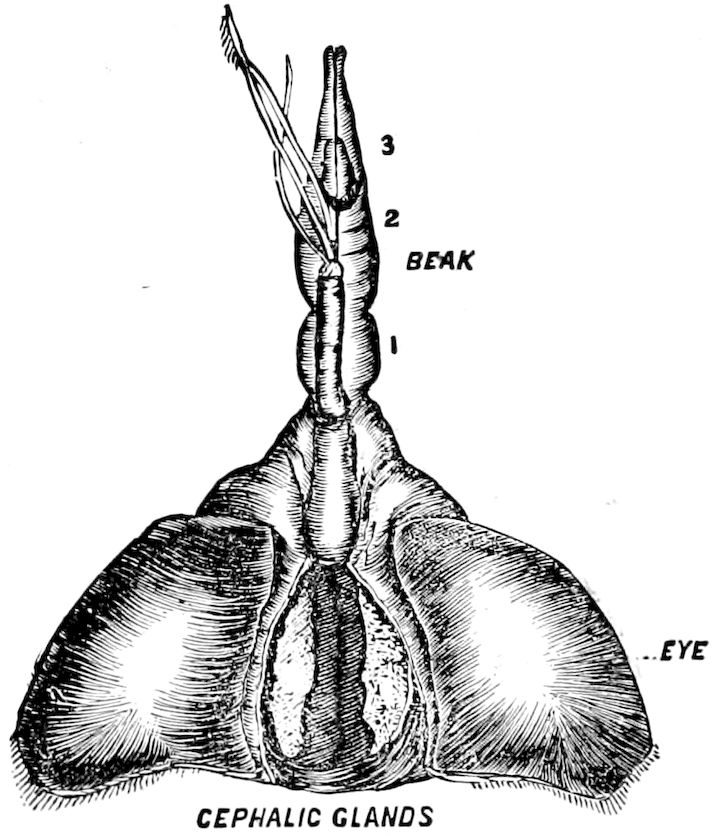
Fig. 349.—Cephalic gland of Belostoma.
Bordas has found both the alkaline gland (gland of Dufour) and the acid gland to occur in a hundred species of Hymenoptera, including not only Aculeata, but also Ichneumonidæ (Fig. 348), Tenthredinidæ, and they may be safely said to be of general occurrence. The acid gland consists of three parts, the glandular portion, the reservoir for the poison, and the secretory canal. The alkaline gland is an irregular tube, with a striated surface and without a reservoir. In most Hymenoptera there is still a third gland, which is unpaired, granular, rectangular or lanceolate, with a short filamentous duct which opens beside the orifice of the alkaline glands.
The poison in ants, wasps, and bees consists of two substances, i.e. formic acid and a whitish, fatty, bitter residue in the secretion 359of the glands; the corroding active formic acid is the essential part of the poison. (Will.)
In Melipona the sting and poison-glands are aborted; in certain ants (Formica, Lasius, etc.) the sting is wanting, but the poison-sac is extraordinarily large.
Bordas finds in various species of Ichneumon three kinds of glands opening into the base of the sting. The first two correspond to the acid (Fig. 348, G.A) and alkaline (G.A) glands of bees and wasps (Vespidæ, etc.), and the third (G.ac) is situated between the two lateral muscular bundles which attach the base of the sting to the last abdominal segment. The poison-reservoir (Fig. 348, V) is recognized by its yellow color and diaphanous and striated appearance. It is situated on the left of the hind-intestine, a little in front of the rectum. The tubular gland (Ga) or alkaline gland of aculeate Hymenoptera is remarkably large; it is situated on the left side of the body. The accessory gland (G.A) is elongated, triangular, flat, its duct opening at the base of the alkaline gland; it is formed of small spherical cells. Bordas has met with well-developed poison-glands in forty species belonging to the Terebrantia, including that of Tenthredo, Emphytus, as well as various genera of Ichneumonidæ, but in all these species the accessory gland was wanting.
Fig. 350.—View from above of the cephalic gland of Belostoma, × 20.—This and Fig. 349 after Locy.
Under the name of cephalic glands (Fig. 349), Locy describes a pair of glands in the head of Nepidæ. The epithelial or secreting cells are 8–sided (Fig. 350). “When these insects are irritated,” he says, “a secretion is freely thrown out around the base of the beak, which produces death very quickly when introduced on a needle point into the body of an insect.” He infers that the cephalic glands may be the source of this poisonous secretion. The poisonous salivary fluid of the larva of Dyticus is referred to on p. 324.
That the mosquito injects poison into the wound it makes has been proved by Macloskie, who discovering fine droplets of a yellow oily-looking fluid escaping from the end of the hypopharynx, afterwards detected the poison-glands. It appears that the two salivary glands are subdivided, each into three lobes, the middle of which (Fig. 351, pg) differs from the others in having evenly granulated contents and staining more deeply than the others. Having examined the preparations, we agree with the discoverer that these lobes 360secrete the poison. The poison is diluted by the secretion of the salivary lobes, and the two efferent ducts, one from each set of glands, “carry forward and commingle the venomo-salivary products in the main duct; and the stream is then carried by the main duct to the reservoir at the base of the hypopharynx.”
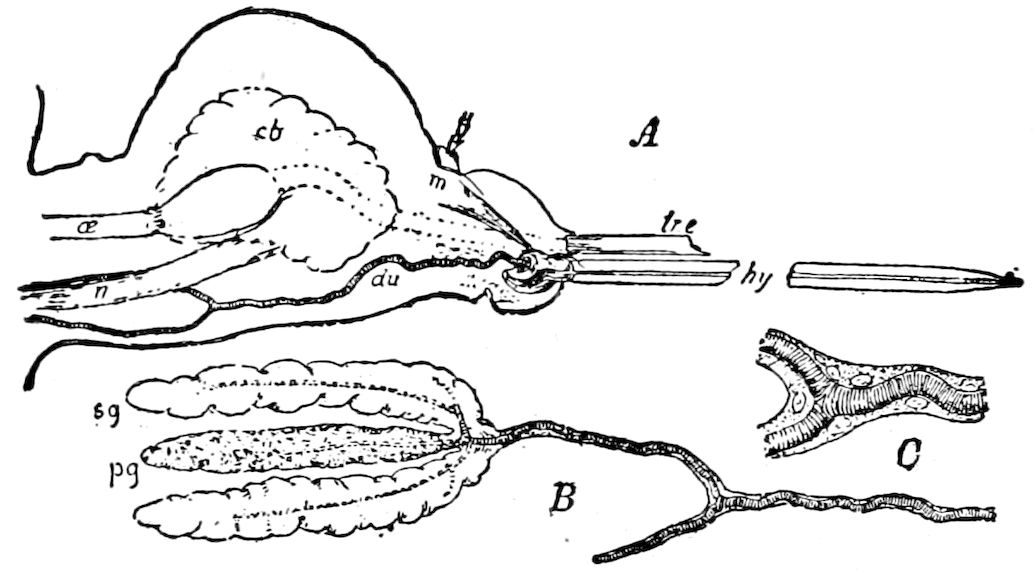
Fig. 351.—A, median section of head, showing (du) the venomo-salivary duct, with its insertion in (hy) the hypopharynx; cb, brain; below is the pharyngeal pump, leading from (œ) the œsophagus; lre, base of labrum-epipharynx; m, muscle; n, commissure (other parts removed). B, the venomo-salivary duct, showing its bifurcation, and the three glands on one of its branches; pg, poison gland; sg, the upper of the two salivary glands. C, the bifurcation of the duct, with its nucleated hypodermis.—After Macloskie.
f. Adhesive or cement-glands
Dewitz has discovered in ants and bees, in close connection with the poison-glands, and like them discharging their secretion through the sting, cement-glands. They arise by budding at the base of the poison-glands.
The two glands in these Hymenoptera correspond to the tubular glands of the Orthoptera, which open at the base of the inner sheath of the ovipositor (Fig. 299, sb), so that the secretion flows out through it as the poison of bees, etc., out of the sting. The use of the secretion of these glands is either to glue the eggs together, or to afford material for the egg-case of cockroaches and Mantidæ and the gummy egg-case of the locusts, etc. The contents of the cement-glands serves for the fixture of the eggs after deposition. In the stinging Hymenoptera one of the cement-glands is an accessory gland; the other becomes the poison-sac. The cement-glands are in the Hemiptera only short blind sacs, in the Lepidoptera and Diptera long convoluted tubes, tubular and branched in the Coleoptera, or richly branched in the Ichneumonidæ and Tenthredinidæ. In the cockroach there are two cement-glands, but the right one is probably of no functional importance. The left one is filled with a milky 361substance, containing many crystals and a coagulable fluid, out of which the egg-capsule (oötheca) is formed. (Miall and Denny.) In the locusts the sebific or cement-gland (Fig. 298, sb) secretes a copious supply of a sticky fluid, which is poured out as the eggs pass out of the oviduct and agglutinates the eggs into a mass, forming a thin coating around each egg, which from the mutual pressure of the eggs causes the tough coating to be pitted hexagonally. In other insects also (Trichoptera, Chrysopidæ, Lepidoptera, etc.) there are similar secretions for the protection or fastening of the eggs when laid.[57] The Trichoptera lay their eggs either in or on the surface of the water in bunches or in strings or in annular gelatinous masses on stones or on plants. This jelly-like substance is secreted by two highly developed paired anal glands. (Weltner, in Kolbe, p. 621.) Also in certain dragon-flies (Libellula, Diplax, and Epitheca) the eggs are laid in jelly-like masses.
With a similar secretion, spun from the end of the abdomen, the Psocidæ cover their little bunches of eggs laid on the under side of leaves; and the silk thread forming the egg-sac of the great water-beetle (Hydrophilus) is secreted from such anal glands.
g. The wax-glands
Besides the honey-bee, which secretes wax in little scales on the under side of the abdomen, the bodies of many other insects, such as the plant and bark lice, as well as the Psyllidæ, Cicadidæ (especially Flata and Lystra), are covered with a waxy powder, or as in Chermes, Schizoneura, Flata, etc., with wool-like filaments of wax.

Fig. 352.—Under side of worker honey-bee, carrying wax scales, × 3.—After Cheshire.
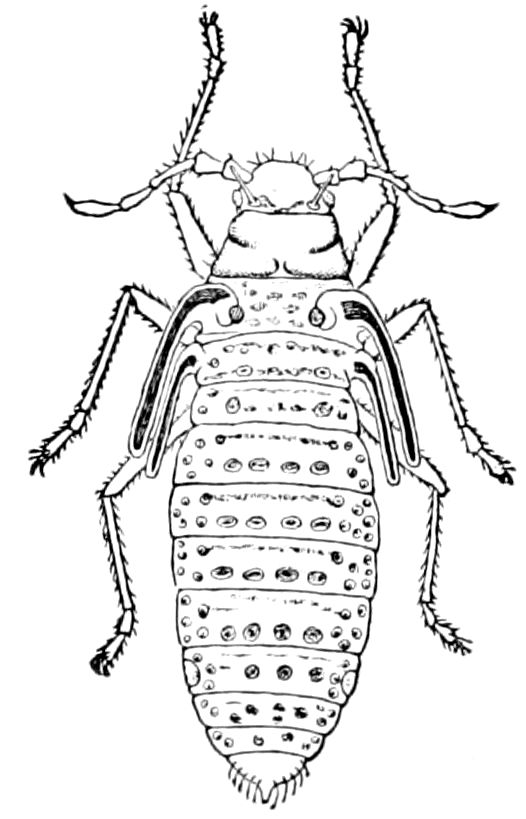
Fig. 353.—Nymph of Lachnus, showing position of wax-glands.—Gissler del.
362The wax is secreted by minute unicellular dermal glands, which in the lower insects (Hemiptera) are distributed nearly all over the body, but in the bees are restricted either to the under (Apis, Fig. 352) or upper side (Trigona) of the end of the abdomen.
The wax-glands of Pemphigus, Chermes, etc., lie under the little warts, seen in Lachnus strobi, the white-pine aphis, to be distributed in transverse lines across the back and sides of the abdominal segments (Fig. 353). These warts are surrounded by a chitinous ring, and divided into delicately marked areas. Through the delicate numerous pits in the chitinous membrane of these areas the little waxen threads project, since under each area ends a duct leading from a large glandular cell, which is a specially modified hypodermis cell (Claus). The wax threads are hollow, and all those arising from a single glued cell form a bundle, whose threads separate from each other and form a white woolly down or bloom covering the body. Witlaczil also shows that gall-forming Aphids secrete a wax-like substance, which, during the movements of the insects in the gall, is rubbed off, becoming a watery layer mixed with the fluid excrement, which forms a spherical impervious layer lining the gall, and thus rendering possible the mode of life of the gall-lice.
In the Psyllidæ Witlaczil has discovered wax-glands which also secrete slender waxen threads. They are situated in groups of two or three at the end of the abdomen near the anus, and arise from hypodermis cells. The wax threads surround the liquid excrement as it passes out of the vent, covering it with a continuous layer of wax. The excrement accordingly is discharged very slowly and gradually, in sausage-shaped masses slightly strung together and rolled into close spirals. The body becomes unavoidably smeared with the sticky excrement, since it is not entirely covered by the waxy layer. Moreover, in the larvæ of many Psyllidæ waxen threads are formed on the upper side of the abdomen; they are for the most part tightly curled or frizzly, like wool, and form, though partly torn, a waxen coat, chiefly on the side and back of the thorax and abdomen. The insects appear therefore as if covered with dust. The mature animals of many species are also covered with a waxen down. The wax threads rapidly dissolve and disappear in alcohol. From a wax-like substance more or less easily dissolved in alcohol arise peculiar hair-like structures which, in the larvæ of Psyllidæ, are situated on the side and end of the body and also on the rudiments of the wings. They are readily distinguished from ordinary hairs, as they arise from glandular cells, and are of very different lengths, more or less like bristles, but hollow, and very brittle. 363They are leaf-like in the first nymphal stages of Trioza rhamni, but in following stages become narrow and form a row around the entire periphery of the body.
The waxen dorsal shield which protects the body of bark-lice (Coccidæ) is a similar product.
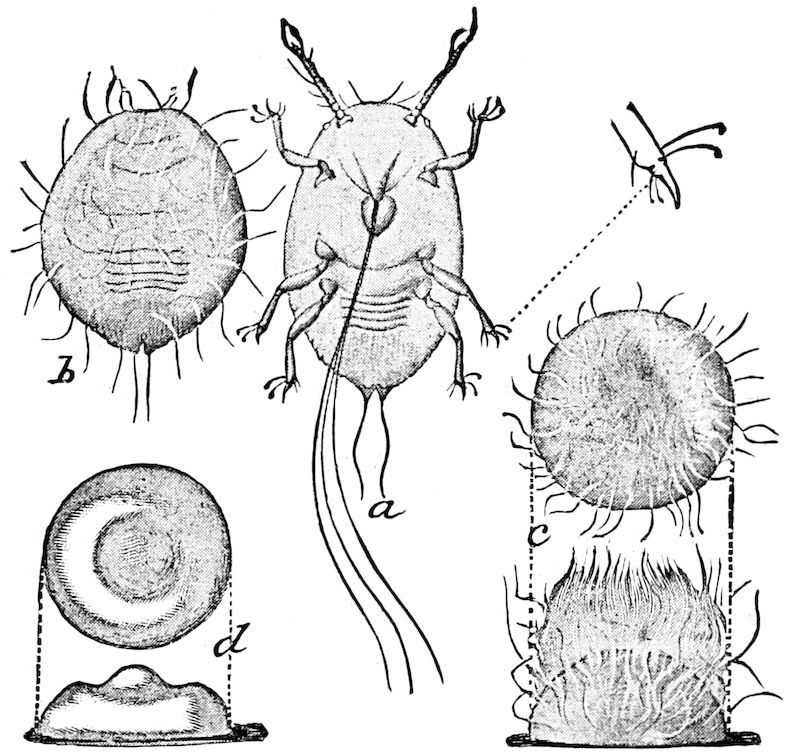
Fig. 354.—Young nymph and developing scale of Aspidiotus perniciosus: a, ventral view of nymph, showing sucking beak with setæ separated, with enlarged tarsal claw at right; b, dorsal view of same, somewhat contracted, with the first waxy filaments appearing; c, dorsal and lateral views of same, still more contracted, illustrating further development of wax secretion; d, later stage of same, dorsal and lateral views, showing matting of wax secretions and first form of young scale; all greatly enlarged.—After Howard and Marlatt, Bull. 3, N. S., Div. Ent., U. S. Dept. of Agr.
Witlaczil has described the way it is formed in Aspidiotus and Leucaspis. The freshly hatched nymph shows no signs of a waxy secretion. But eventually waxen threads arise first on the hinder and anterior end of the body, and then over the whole surface. These threads interlace into a sort of felting and thus form the shield, which is usually much larger than the body and lies closely upon it. The shield is formed after the first moult. It is noteworthy that these threads are matted together to form as thick a tissue as that of the shield itself. The shield is whitish or gray and rather thin. On the thinnest part of the edge the single threads may be drawn out. The growth of the shield advances with the increase in size of the nymph around the entire edge, but is greatest behind. The first two larval skins are retained on the back under the shield. Also a very thin waxen pellicle remains on the resting place of the insect when it is raised. The wax-glands open in the pitted fields, and appear as clear brownish cells which are distinguished from the ordinary hypodermis cells by their greater size. (Witlaczil. Compare also Fig. 354.)
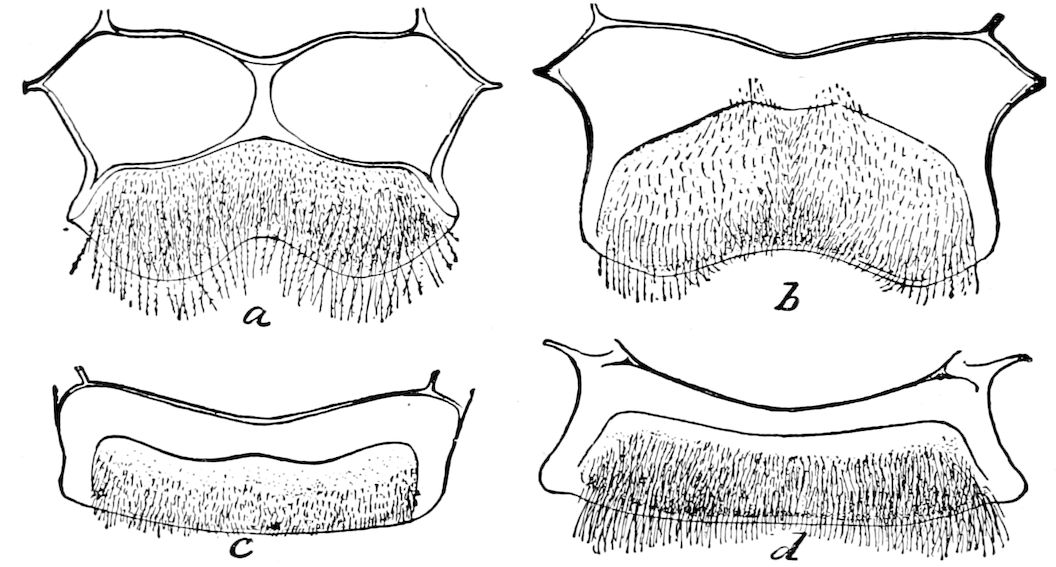
Fig. 355.—Wax disks of social bees: a, Apis mellifica, worker; b, do., queen; c, Melipona, worker; d, Bombus, worker.—From Insect Life, U. S. Dept. Agr.
The wax-glands in the honey-bee are scale-shaped organs situated on the under side of the four last abdominal segments (Fig. 355). These secrete the wax, which appears as whitish scales, and secretion is only possible when the bees have sufficient honey and pollen. The wax is secreted by the hypodermal cells rather than by glands within the abdominal cavity; the wax traverses the cuticular layer, and accumulates on its outer surface (Carlet). According to Fritz Müller, in the stingless bees (Trigona) which he observed, the wax-glands are situated on the back of the abdomen, but Ihering states that in many species of Trigona and Melipona there are also slightly developed wax-organs on the ventral side.
It has been found that certain caterpillars secrete wax. Thus the cells of the Tortrix of the fir (Retinia resinella) formed of resin are lined with wax, as on dissolving away the resin with alcohol, Dr. Knaggs found a slight film of wax; also a secretion of wax has been detected in the larva of a butterfly (Parnassius apollo). The bodies of certain saw-fly larvæ are covered with a white powdery secretion, while the remarkable larva of a Selandria is clothed with snow-white, long, flocculent, waxy masses, nearly concealing the body (Fig. 356).
h. “Honey-dew” or wax-glands of Aphids
The so-called “honey-dew” of Aphids which oozes from two wart-like tubercles or tubes situated near the end of the body, is secreted by hypodermal unicellular glands which open into a modification of a pore-canal, the tube itself being an outgrowth of the cuticula.
Witlaczil states that both in the “honey” tubes and in the body beneath, the sugary matter exists in cells of the connective tissue in the form of granules. “These large ‘sugar-cells’ in contact with the air undergo destruction, while the sugar crystallizes into needles, and thus each cell is transformed into a radiated crystalline mass.”
365“A muscle extends from a horseshoe-shaped place (a valve?) in the middle of the flat terminal plate of the honey tube, through this and down through the abdomen to the ventral surface. By this muscle the honey tube is at times erected, and we then find, as also when we lightly press the body of the insect, lumps of crystallized sugar which have been expressed through the tips of the honey tubes.” (Zool. Anzeiger, 1882, p. 241.)

Fig. 356.—Wax-secreting larva of a saw-fly.
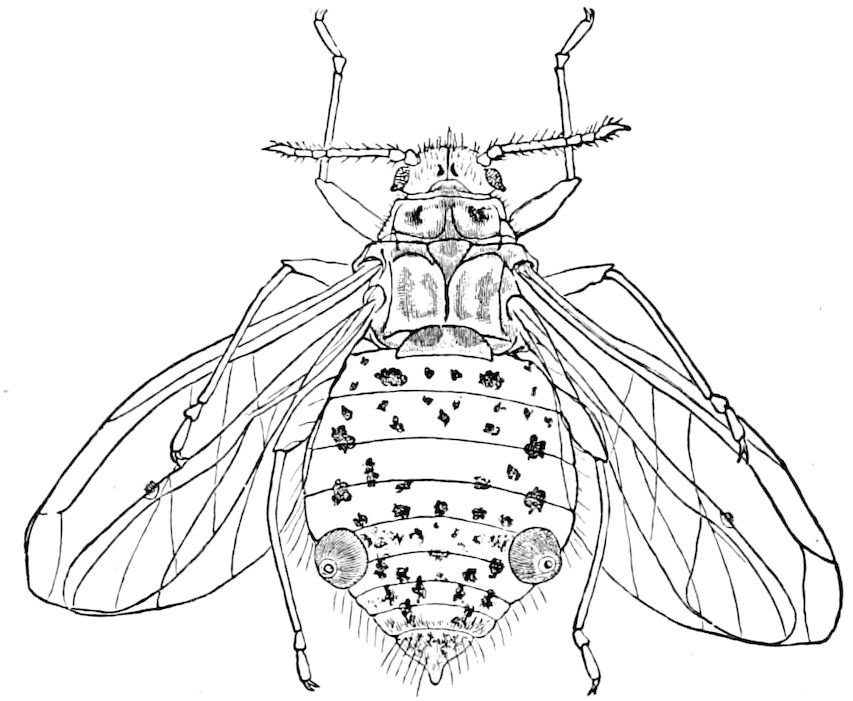
Fig. 357.—Lachnus strobi, and its two “honey” warts.—Gissler del.
Busgen, after careful research, denies that this is a sugar, but claims as the result of chemical analysis, that it is more like wax. He observed that on reaching the air the drops issuing from the “nectary” or “honey” tube stiffened almost instantly into a wax-like mass, which was easily crushed between the teeth, and had no taste at all. No sugar-like substance or urea could be detected. He therefore concludes that the secretion in question should be regarded as a wax-like mass, which agrees well with Witlaczil’s anatomical observations, and confirms the statements of previous observers. Thus, as early as 1815, Kyber stated that the Aphides expelled an excrementitious substance through the “sap tubes.” Burmeister states that the tubes give out a fluid which “dries gumlike, but, so far as I have observed, has no peculiar taste.” Réaumur, and also Kaltenbach, state that the “honey” does not issue from the tubes, but from the anus. Lastly, Forel emphatically states that “the two dorsal tubes of Aphides do not secrete a sweet fluid, but a gluey wax, which is not sought by the ants. Moreover the shield-lice and many leaf-lice have no such tubes, but yet are often sought by ants. The drops of sugar which the ants lick up are rather the excrement of the insects in question.” Hence the opinion first stated by Linné, that a sweet fluid is secreted by Aphides, must be abandoned.
On the other hand, Busgen, after careful observations, finds that the use of the sticky, waxen secretion is in reality a protective one, as he observed that when a larval Chrysopa rudely attacks the Aphides, they smear its face with the sticky wax, causing at least a momentary interruption in its attacks. He also observed that Aphides when invaded by coccinellid larvæ set their tubes in motion and besmear their heads and front part of the body. He thus seems to establish the fact that these tubes secrete a protective, sticky fluid.
i. Dermal glands in general
We have seen that certain of the hypodermal cells may be modified or specialized to form secretory unicellular glands. Such are 366those (trichogens) which secrete chitinous setæ, hairs, and spines, certain setæ in some insects being hollow and containing a poison (p. 187); others secrete wax, certain ones in Aphids “honey-dew”; in some cases dermal glands may excrete protective, sticky, or otherwise offensive matters, or may be depuratory, or facilitate the process of moulting.
There are other minute, unicellular, or compound dermal glands whose function is unknown.
Dermal glands may be segmentally or serially arranged. Thus Verson has detected a series of one or two pairs of unicellular glands near the stigmata in each thoracic, and the first eight abdominal segments of the silkworm (B. mori). In the earliest stages of growth of the caterpillar they give out oxalate of lime, and in later stages uric acid. They thus appear to act interchangeably with the urinary tubes, as excretory organs. They do not, however, carry their products directly outwards, but leave them between the hypodermis and cuticula, in order to facilitate the sloughing off of the latter in the process of moulting.
LITERATURE ON THE SECRETORY GLANDS
a. General
Sirodot, S. Recherches sur les sécrétions chez les Insectes. (Ann. sc. nat., 4 Sér., Zool., 1858, x, pp. 141–189, 251–328, 12 Pls.)
Gazagnaire, G. Des glandes chez les Insectes. (Compt. rend. Acad. Sc., Paris, 1886, cii, pp. 1501–1503; Annal. Soc. Ent. France, 1886, Bull., pp. 104–106.)
Leydig, F. Beitrage zur Anatomie und Histiologie der Insekten. 1887.
Hanow, Karl. Ueber Kerfabsonderungen und ihre Benutzung im eigenen Haushalte. (Programm des Realprogymnasiums zu Delitzsch für das Schuljahr 1889, xc; Delitzsch, 1890, pp. 3–22.)
Verson, E. Di una serie di nuovi organi escretori scoperti nel filugello. (Publ. R. Stazione Bacologica di Padova, v, 1890, pp. 30. 4 Pls.)
—— Altre cellule glandulari di origine postlarvale. (Ibid., vii, 1892, pp. 16, 1 Pl.)
—— ed E. Bisson. Cellule glandulari ipostigmatiche nel Bombyx mori. (Ibid., vi, 1891.)
Borgert, H. Die Hautdrüsen der Tracheaten. Jena, 1891, pp. 80.
Batelli, Andrea. Di una particolarità nell’ integumento dell’ Aphrophora spumaria. (Monitore Zool. Ital., 1891, Anno ii., pp. 30–32, dermal gland in last segment.)
Koschewnikow, G. A. On a new compound dermal gland found in the sting of the bee. (Journal of the Zoological Section of the Society of the Friends of Natural Science. Moscow, ii, Nos. 1, 2, 1892, p. 36. Preliminary notice. In Russian.)
Willem, V., et H. Salbe. Le tube ventral et les glandes céphaliques des Sminthurus. (Ann. Soc. Ent. Belg., 1897, xli, pp. 130–132.)
Henseval, Maurice. Les glandes à essence der Cossus ligniperda. (La Cellule, 1897, xii, pp. 19–26, 27, 29.)
367—— Recherches sur l’essence der Cossus ligniperda. (La Cellule, 1897, xii, pp. 169–181, 183.)
b. Poison-glands
Macloskie, George. The poison-apparatus of the mosquito. (Amer. Nat., xxii, 1888, pp. 884–888. 1 Fig. Also in Science, 1887, p. 106.)
Beyer, Otto W. Der Giftapparat von Formica rufa, ein reduziertes Organ. (Jena. Zeitschr. f. Wissens, xxv, 1891, pp. 26–112, 2 Taf.)
Bordas, L. Sur l’appareil venimeux des Hyménoptères. (Comtes rend., cxviii, 1894, pp. 296–299 and 873–874; also Zool. Anzeiger, xvii Jahrg., 1894, pp. 385–387, Figs.)
—— Appareil glandulaire des Hyménoptères. (Glandes salivaires; Tubes de Malpighi et glandes venimeuses.) Paris, 1894, pp. 362, 11 Pls.
—— Description anatomique et étude histologique des glandes à venin des insectes Hyménoptères. Paris, 1897, pp. 53, 2 Pls.
See Forel (p. 186); also the standard authors, Kolbe, etc.; also Locy (Amer. Nat., xviii, 1884, p. 355), Nagel (p. 324), Fenger.
c. Wax-glands
Brandt und Ratzeburg. Medicinische Zoologie, ii, 1830, p. 179. Taf. xxv, Fig. 18.
Treviranus, George R. Ueber die Bereitung des Wachses durch die Bienen. (Zeitschrift für Physiologie, etc., iii, 1832, pp. 62, 225.)
Dufour, L. Note anatomique sur la question de la production de la cire des abeilles. (Comptes rend. Acad. Sc., Paris, 1843, xvii, pp. 809–813, 1248–1253; Revue Zool. 1843, l’Institut, 1843, xi.)
Dujardin, F. Mémoire sur l’étude microscopique de la cire, etc. (Ann. Sc. Nat., xii, 1849, pp. 250–259.)
Tarzione-Tozzetti, H. Studii sulle cocciniglie. Milano, 1867, 7 Pls.
—— Sur la cire qu’on peut obtenir de la cochenille du figuier (Coccus caricæ). (Comptes rend. Acad. Sc., Paris, lxv, 1867, pp. 246–247.)
Claus, C. Ueber die wachsbereitenden Hautdrüsen der Insekten. (Sitzungsber. Gesells. z. Beförd. d. Gesammt. Naturw. zu Marburg, June, 1867, No. 8, pp. 65–72.)
Witlaczil, E. Die Anatomie der Psylliden. (Zeitschr. wissens. Zool., xlii, 1885, pp. 582–586.)
—— Zur Morphologie und Anatomie der Cocciden. (Zeitschr. wissens. Zool., xliii, 1886, pp. 149–174, 1 Taf.)
Ihering, H. von. Der Stachel der Meliponen. (Ent. Nachrichten, xii Jahrg., 1886, p. 185.)
Carlet, G. Sur les organes sécréteurs et la sécrétion de la cire chez l’Abeille. (Comptes rendus, cx, pp. 361–363, 1890.)
—— La cire et ses organes sécréteurs. (Le Naturaliste, 1890, pp. 149–151, 2 Figs.)
Also the works of Siebold, Cheshire, Kolbe, Howard and Marlatt (Bull., 3, N. S., Div. Ent. U. S. Dept. Agr., 1896, p. 40), Knaggs, Berlese.
d. Wax-like glands of Aphides
Huber et Forel, A. Études myrmécologiques, 1875.
Witlaczil, E. Zur Anatomie der Aphiden. (Zool. Anzeiger, v Jahrg., 1882, pp. 239–241; Arbeiten a. d. Zool. Institut der Univ. Wien., iv, 1882, pp. 397–441, 3 Taf.)
Busgen, M. J. Der Honigtau. Biol. Studien an Pflanzen u. Pflanzenlause. (Jena. Zeitschrift, xxv, 1891, pp. 339–428.)
DEFENSIVE OR REPUGNATORIAL SCENT-GLANDS
While these eversible glands are not found in marine or aquatic arthropods such as Crustacea or Merostomata (Limulus), they are often present in the air-breathing forms, especially insects. In the winged insects they are of frequent occurrence, existing under great variety of form, varying greatly in position, and appearing usually to be in immediate relation with their active volant habits. Their presence is in direct adaptation to the needs and habits of their possessors, and being repellent, warning, or defensive structures, the odors they secrete being often exceedingly nauseous, they appear to have been called into existence in direct response to their biological environment. The fact that these singular organs do not exist in marine or aquatic Crustacea suggests that the air-breathing, aërial, or volant insects by these eversible glands, usually in the form of simple evaginable hypodermic pouches, are enabled to protect themselves by emitting an infinitesimal amount of an offensively odorous fluid or ether-like spray which charges the air throughout an extent of territory which may be practically illimitable to the senses of their enemies. The principle is the same as in the mephitic sulphuretted oil ejected by the skunks, the slight quantity these creatures give out readily mixing with and charging the atmosphere within a radius of many miles of what we may call the centre of distribution.
As is now well known, the very delicate, attenuated highly volatile
odors exhaled are perceived by insects with extreme ease and
rapidity, the degree of sensitiveness to such scents being enormously
greater than in vertebrates, their organs of sense being developed in
a corresponding degree. Professors Fischer and Penzoldt, of Erlangen,
have recently established the fact that the sense of smell is
by far the most delicate of the senses. They find that the olfactory
nerve is able to detect the presence of 1
2,760,000,000 of a grain of mercaptan.[58]
The smallest particle of matter that can be detected by the
eye is sodium, when observed by the spectroscope, and this particle
is 250 times coarser than the particle of mercaptan which can be
detected by the human nose.
In those Arachnida which are provided with poison-glands, these scent-glands are absent, but in certain Acarina and Linguatulidæ, which have no poison-glands, there are various oil-glands, stigmatic glands, as well as scent-glands, 369and in seizing a Thelyphonus with the forceps we have observed it to send out from each side of the body a jet of offensive spray.
We not infrequently find in myriopods (Polydesmidæ, Julidæ, and Glomeris) repugnatorial or the so-called cyanogenic glands, which are either paired, opening on the sides of the body, or form a single row along the median line of the under side of the body. Leidy describes and figures the spherical glands of Julus marginatus, of which there are 50 pairs. These glands have been regarded as modified nephridia, but are more probably coxal glands, and the homologues of the parapodial glands of annelid worms.
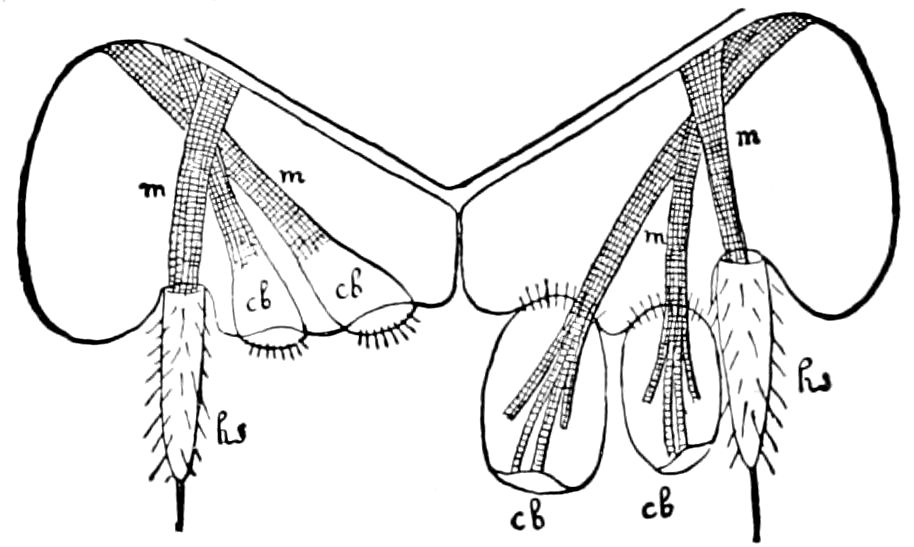
Fig. 358.—Sternite of Machilis maritima, with the pair of coxal sacs (cb) on the right side everted; hs, coxal appendages; m, retractor muscles.—After Oudemans, from Lang.
Eversible coxal glands.—True coxal glands occur in Scolopendrella immaculata on the 2d to 11th segment, on the inner side of the base of the legs (Fig. 15, c.g.). Homologous glands also occur in the same position in Campodea staphylinus (also in C. cookei and C. mexicana) on the 1st to 8th abdominal segments, and Oudemans has described a pair of eversible sacs on each side of segments one to seven of Machilis. These eversible sacs in the synapterous insects are evidently modified coxal glands, and are probably repugnatorial as well as respiratory in function.
The apparatus consists of an eversible gland, composed of hypodermic cells, usually retracted by a slender muscle and with an efferent passage, but the glands vary greatly in shape and structure in different insects. In some cases these fœtid glands appear not to be the homologues of the coxal glands, but simply dermal glands.
These repugnatorial glands are of not infrequent occurrence in the lower or more generalized winged insects, and in situation and appearance are evidently the homologues of the coxal glands of the Symphyla and Synaptera.
Fœtid glands of Orthoptera.—In the ear-wigs (Forficula and Chelidura) Meinert has detected a pair of what he calls fœtid glands at the posterior margin of the dorsal plates of the 2d and 3d abdominal segments.
Vosseler also describes the same glands as consisting of a retort-shaped sac, in whose walls are numerous small hypodermal cells and large single glandular cells provided with an efferent passage, the fluid being forced out by the pressure of the dermal muscles, one acting specially to retract the gland. The creature can squirt to a 370distance of 5 and even 10 cm. (4 inches) a yellowish-brown liquid or emulsion with the odor of a mixture of carbolic acid and creosote.
The large eversible dorsal glands of the Blattidæ, since they contain numerous hairs, which, when everted, are fan-like or like tufts, serve, as in the spraying or scent apparatus, to disseminate the odor, and might be classified with the alluring unicellular scent-glands or duftapparat of other insects, as they are by some authors; but as the glands are large and compound they may prove to be the homologues of the coxal glands rather than of the dermal glands.
Evaginable organs in the Blattids were first observed by Gerstæcker in both sexes of Corydia; they are yellowish white, covered with hairs, and are thrust out from between the dorsal and ventral plates of the 1st and 2d abdominal segments.
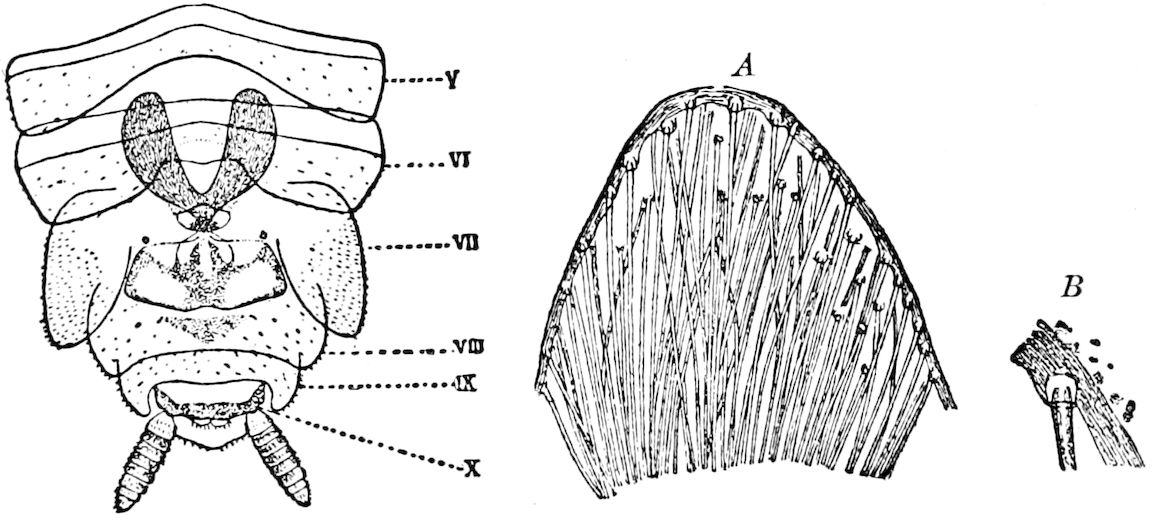
Fig. 359.—Under side of end of Aphlebia, showing the two eversible sacs; V-X, five last abdominal segments; A, portion showing the hairs; B, showing origin of a hair in its follicle.—After Krauss.
In the cockroach (P. orientalis) Minchin detected two pouch-like invaginations of the cuticle, lying close on each side of the middle line of the body between the 5th and 6th tergites of the abdomen. They are lined by a continuation of the cuticle, which forms, within the pouches, numerous stiff, branched, finely pointed bristles, beneath which are a number of glandular epithelial cells. In the male nymph of P. decorata he also found beside these glandular pouches “an additional gland, opening by a tubular duct under the intersegmental membrane between the 5th and 6th terga above the glandular pouch of each side, and extending forward into the body cavity. The gland and its duct are proliferations of the hypodermis, and there is no invagination of the cuticle.” These eversible glands are most complicated in Phyllodromia germanica. While it is absent in the female, in the male it is relatively of enormous size, extending over the 6th and 7th somites, as well as projecting far into the body cavity (Minchin). 371Haase states that these glands become everted by blood-pressure and give out the well-known disagreeable smell of these insects. He states that in the male of P. germanica the dorsal glands in the 6th and 7th abdominal segments are without hairs and produce an oily secretion; they function as odoriferous organs in sexual union.
In the male of another Blattid (Aphlebia bivittata) of the Canary Islands, Krauss has detected two yellowish dorsal sacs 1.5 mm. in length, opening out on the 7th abdominal segment, and filled full of long yellowish hairs, the ends directed towards the opening, where they form a thick tuft. These eversible glands lined with hairs appear to be closely similar to the long slender eversible hairy appendages or scent organs of certain Arctian and Syntomid moths. (Fig. 359.)
Fig. 360.—External flaps (gl) of glands of Platyzosteria.
We have found the external median wart with lateral lids or flaps in between the 5th and 6th tergites of Platyzosteria ingens Scudder, a large wingless Blattid living under the leaf scars of the cocoanut tree in Southern Florida (Fig. 360), but were unable to detect them in Polyzosteria or in the common Blabera of Cuba, or in another genus from Cordova, Mexico.
In another group of Orthoptera, the Phasmidæ, occur a pair of dorsal prothoracic glands, each opening by a pore and present in both sexes. In the walking-stick, Anisomorpha buprestoides, ♂ and ♀, these openings are situated on each side of the prothorax at its upper anterior extremity, situated at the bottom of a large deep pit. When seized it discharged a “milky white fluid from the pores of the thorax, diffusing a strong odor, in a great measure like that of the common Gnaphalium or life everlasting” (Peale in Say’s American Entomology, i, p. 84). Boll states that the females when captured “spurt from the prothorax, somewhat after the manner of bombardier beetles, a strong vapor, which slightly burnt the skin; when the females were seized by the males a thick fluid oozed from the same spot.” Scudder describes these glands in another Phasmid (Autolyca pallidicornis) as two straight, flattened, ribbon-like bodies, with thick walls, broadly rounded at the end, lying side by side and extending to the hinder end of the mesothorax. In Anisomorpha buprestoides the glands are of the same size and shape (Scudder). In Diapheromera femorata the repugnatorial foramina are very minute, and the apparatus within consists of a pair of small obovate or subfusiform sacs, one on each side of the prothorax, about 1 mm. 372in length, with a short and very slender duct opening externally at the bottom of the pit (Scudder).
In the Mantidæ these seem to be genuine coxal glands, as there is a pair situated between the coxæ of the first pair of legs. An evaginable organ like a wart, with a glandular appearance, occurs on the hind femora of the Acrydiidæ in a furrow on the under side, into which the tibia fits, about one-fourth from the base (Psyche, iii, p. 32).
In the male cricket, the anal odoriferous glands are small lobes opening into a reservoir on each side of the rectum (Dufour). Homologous glands also occur in the Coleoptera (Fig. 302, l and 317, s).
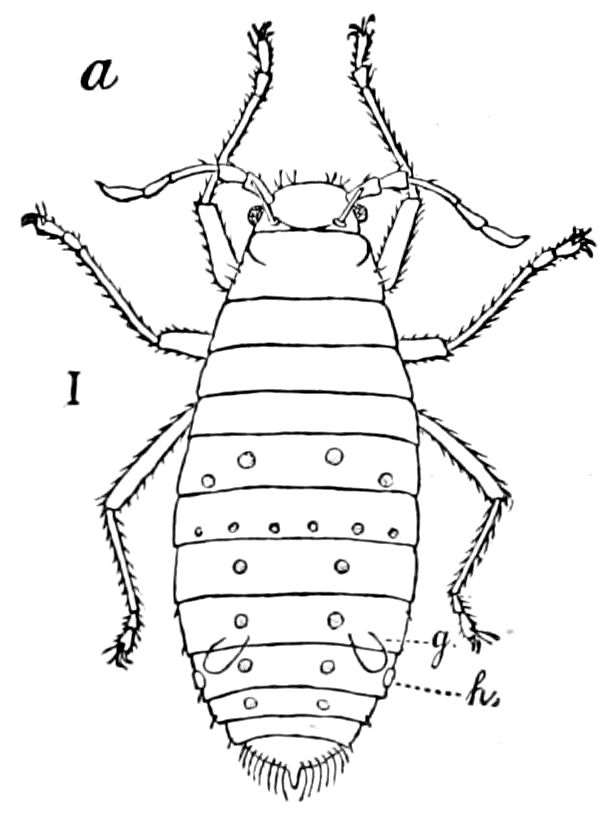
Fig. 361.—Glands (g) of Lachnus; h, “honey” wart.—Gissler del.
Most Hemiptera or bugs send out a fœtid or nauseous odor due to a fluid secreted by a single or double yellow or red pear-shaped gland, situated in the middle of the mesothoracic segment, and opening between the hinder or third pair of coxæ. In Belostoma Leidy describes these glands as consisting of two rather long cœcal tubes situated in the metathorax, beneath the other viscera, extending backwards into the abdomen, and opening between the coxæ of the third pair of legs. Locy states that the smell arising from these glands is pleasant, resembling that of well ripened pears or bananas. Other bugs, moreover, emit an agreeable odor, that of Syromastes resembling that of a fine bergamot pear. (Siebold.) The fluid given out by the European fire-bug (Pyrrhocoris apterus) has a sweetish smell, like ether. In the nymph there are three pairs of dorsal glands, on abdominal segments 2–5, which are atrophied in the mature insect. In the bed-bug, the nymph has three odoriferous glands each with paired openings in the three basal abdominal segments respectively, and situated on the median dorsal line, being arranged transversely at the edge of the tergites; but after the last moult these are aborted, and replaced by the sternal metathoracic glands (Künckel). Gissler has detected a pair of glands in Lachnus strobi (Fig. 361).
Anal glands of beetles.—Certain beetles are endowed with eversible repugnatorial glands. Eleodes gigantea and E. dentipes of both sexes are said by Gissler to possess these glands. When teased “they stand on their anterior and middle legs, holding the abdomen 373high up and spurting the contents of the glands right and left.” The glands (Fig. 366, 1) are two reddish brown, somewhat bilobed sacs, and extend from the base of the last up to the middle of the 2d abdominal segment, with an average length of 6.5 mm. The liquid stains the human skin, has an acid reaction, with a peculiar, “intensely penetrant odor, causing the eye to lachrymate. It is soluble in water, alcohol, and ether. Boiled with concentrated sulphuric acid and alcohol an ethereal aromatic vapor is produced, indicating the presence of one or more organic acids, though neither formic or acetic acid could be detected.” Williston has observed the same habits in seven other species of Eleodes, all ejecting a pungent vile-smelling liquid, one species (E. longicollis) ejecting a stream of fluid from the anal gland, backwards sometimes to the distance of 10 cm. or more, and he regards these beetles as “the veritable skunks of their order.” Leidy briefly describes the odoriferous glands of Upis pennsylvanica.
The anal glands consist, according to Meckel and also Dufour, of two long, simple, flexuous cœca with reservoirs having two short excretory ducts situated near the anus (Siebold).
Glands like those of Eleodes found in Blaps mortisaga are described in detail by Gilson (Fig. 366, 2). They form two pouches or cuticular invaginations situated in the end of the abdomen on the sides of the end of the intestine and unite on the median line underneath the genital organs, forming a very short tube with a chitinous wall, continuous with the cuticle of the last abdominal segment. Into each pouch open a large number of fine slender lobules varying in shape, giving a villous aspect to the surface. These lobules are composed of as many as fifty unicellular glands, each of which is composed of four parts: (1) A radiated vesicle, (2) a central sac, giving rise (3) to a fine excretory tube, and (4) a sheath near the origin of the excretory tube. These are all modifications of the cytoplasm of the cell with its reticulum; the nucleus with its chromosomes is also present, but situated on one side of the central sac. The fine excretory tubules form a bundle passing down into the mouth of each lobule.
Similar glands, though usually smaller, which have not been carefully examined, occur in Carabus (Fig. 300, 3) and Cychrus, which eject from the vent a disagreeable fluid containing butyric acid (Pelouse). The bombardier beetle Brachinus, with its anal glands, ejects a jet of bluish vapor accompanied with a considerable explosion, which colors the human skin rust red; it is caustic, smells like nitrous acid, and turns blue paper red. Westwood states that individuals of a large South American Brachinus on being seized “immediately began to play off their artillery, burning and staining the flesh to such a degree 374that only a few specimens could be captured with the naked hand, leaving a mark which remained for a considerable time.” The fluid ejected by another species, in Tripoli, blackened the fingers of the collector. “It is neither alkaline nor acid, and it is soluble in water and in alcohol.” (Kirby and Spence, iv, p. 149.)
Species of other genera (Agonum, Pheropsophus, Galerita, Helluo, Paussus, Ozæna) are also bombardiers, though less decidedly so than Brachinus. A Paussid beetle (Cerapterus) ejects explosively a fluid containing free iodine (Loman), while Staphylinus, Stenus, Ocypus olens, Lacon, etc., have similar anal fœtid glands, the liquid being more or less corrosive. The secretion of Mormolyce phyllodes is so corrosive that it is said to paralyze the fingers for 24 hours after. (Cuénot.)
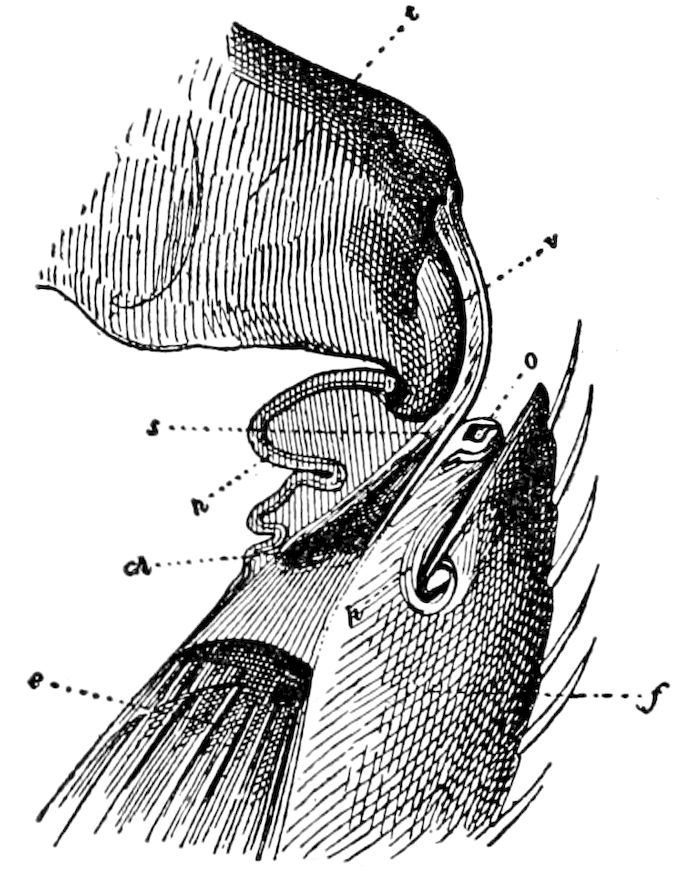
Fig. 362.—Median section through the femoro-tibial joint of leg of Coccinella, showing at o the opening through which the blood oozes out; f, femur; t, tibia; e, extensor muscle of the tibia: s, sinew of the same; at ch, chitinized; h, articular membrane; v, tibial process.—After Lutz.
The two pairs of remarkably large, soft, eversible, forked, orange-yellow glands of the European genus Malachius, are thrust out from the side of the 1st and the 3d thoracic segments. They are everted by blood-pressure, and retracted by muscles. The larva of Hydrophilus piceus ejects by the anus a black, fœtid fluid.
Claus has shown that the larva of Lina populi and other Chrysomelidæ possess numerous minute, eversible glands in each of the warts on the upper surface of the body, each gland containing a whitish, repellent fluid smelling like the oil of bitter almonds and containing salicylic acid derived from its food-plant, which issues as pearl-like drops. Candèze thinks the fluid may contain prussic acid. The fluid is secreted by a variable number of glandular cells, each provided with an efferent duct. The larvæ of saw-flies, notably Cimbex americana, also eject droplets of a clear fluid from non-evaginable glands situated near each stigma (Chlolodkovsky).
The blood as a repellent fluid.—In this connection it may be mentioned that though there are no special glands present, many beetles emit drops of blood from the femoro-tibial joints of their legs as a means of defence. Such are the oil-beetles (Meloë), Cantharis, Lytta. The cantharadine secreted by these beetles, according to Beauregard, is an efficient means of defence, as birds, reptiles, and carnivorous insects will not usually attack them. This substance 375is formed in the blood and also in the genital organs, and is so extremely caustic that scavenger insects which feed upon their dead bodies will leave untouched the parts containing cantharadine, and if May-beetles or mole-crickets are washed with the blood of Meloë or with cantharidate of potassa, it will for several days render them safe from the attacks of the carabids which usually prey upon them. The eggs even after deposition are strongly vesicant, and are thus free from the attacks of egg-eating insects (Cuénot). The Coccinellidæ are also protected by a yellow, mucilaginous, disagreeable fluid oozing out of the ends of the femora; in our common, two-spotted lady-bird (C. bipunctata) the yellow fluid is disagreeable, smelling like opium. Lutz has found that the blood in Coccinellidæ passes out through a minute opening situated at the end of each femur (Fig. 362). The blood is very repellent to insectivorous animals.
The Dyticidæ eject from the anus a colorless, disagreeable fluid, while these beetles, and especially the Gyrinidæ, when captured send out a milky fluid which appears to issue from the joints of the body. The Silphidæ throw out both from the mouth and vent a fœtid liquid with an ammoniacal odor. They possess but a single anal gland, the reservoir opening on one side of the rectum (Dufour).
Other malodorous insects have not yet been investigated; such are the very persistent odors of lace-winged flies (Chrysopa).
More agreeable secretions, but probably formed by similar glands, is the odor of rose or hyacinth given out by Cicindelæ, or the rose fragrance exhaled by the European Aromia moschata.
Eversible glands of caddis-worms and caterpillars.—Gilson, while investigating the segmentally disposed thoracic glands of larval Trichoptera, has found in the larva of Limnophilus flavicornis that the sternal prothoracic tubercle gives exit to an underlying tubular gland. In Phryganea grandis each thoracic sternum affords an exit to an eversible gland. Many caterpillars, as our subjoined list will show, are very well protected by eversible repugnatorial glands situated either in the under or upper side of the body. Since the time of De Geer (1750) the fork-tailed larva of Cerura has been known to throw out a secretion, which was described by Bonnet in 1755 as a true acid, sharp, sour, and biting. This spraying apparatus in Cerura (Harpyia) vinula has been well described by Klemensiewicz (Fig. 366, 4), though Rengger in 1817 noticed the general form of the secretory sac, and that it opens out in two muscular eversible tubes, out of which the secretion is ejected.
The fork-tailed larva of Macrurocampa marthesia, which is much like that of Cerura, when teased sends out a jet of spray to the 376distance of nearly an inch from each side of the neck. While examining the very gayly-colored and heavily-spined caterpillars of Schizura concinna we observed that when a fully-grown one was roughly seized with the forceps or fingers it sent out a shower of spray from each side of the prothoracic segment, exactly like that of Cerura and Macrurocampa.
In the European Cerura vinula the apparatus consists of a single sac, which opens by a narrow transverse slit on the under side of the neck, out of which is rapidly everted four lateral tubes, two on each side (Fig. 366, 4, t), which are withdrawn within the opening by the contraction of several fine muscles. The apparatus in the American C. multiscripta is as in the European C. vinula. In a living specimen the large secretory sac was seen to be of the same size and shape as in Macrurocampa, and of the color of raw silk. The sac when distended extends back to a little behind the middle pair of legs, and is nearly two-thirds as wide as the body. The caterpillar sent out the fluid when handled, but we could not make it spray.
In the larva of Macrurocampa marthesia the cervical or secretory gland (Fig. 366, 5) is situated in the 1st and 2d thoracic segments, extending to the hinder edge of the latter and lying between the nervous cord and the œsophagus and proventriculus, and when empty the bulk of it lies a little to one side of the median line of the body. It is partly held in place by small tracheæ, one quite large branch being sent to it from near the prothoracic spiracle. The short, large duct, leading from it to the transverse opening in the membrane between the head and prothoracic segment, is a little narrower than this opening, and is kept distended by tænidia, or a series of short, spiral threads which are pale, not honey-yellowish, in color. This duct lies on one side of the prothoracic ganglion, resting just under the commissures passing up to the brain; it is also situated between the two silk ducts.
The very distensible sac (Fig. 366, 5) is rendered elastic by a curious arrangement of the cuticle, the tænidia of the duct itself being represented by very thickly-scattered, irregular, separate, sinuous, chitinous ridges, which stand up from the cuticular lining of the wall of the sac (Fig. 366, 6). The secretory cells of the walls of this sac in Cerura vinula are said by Klemensiewicz to be large hexagonal cells, resembling those of silk-glands, having like them large branched nuclei.
The fluid thrown out is said by Poulton to be formic acid; it causes violent effervescence when allowed to fall upon sodium-bicarbonate, 377and colors blue litmus paper red. It also appears from the researches of Latter that these creatures in the imago state secrete free potassium hydroxid, a substance for the first time known to exist in the animal kingdom.
In the caterpillar of Astyanax archippus (Limenitis disippus) a dark, bladder-like sac is everted, but the lateral tubes appear to be wanting, and no spray is sent out; it occurs in the larvæ of many Nymphalidæ and other butterflies and moths.
These glands are functionally active in Perophora, but obsolete (at least the external openings) in Lacosoma.
The osmeterium in Papilio larvæ.—The caterpillars of the swallowtailed butterflies (Papilio, Doritis, and Thais), as is well known, when irritated thrust out from a transverse slit on the upper part of the prothoracic segment a large orange-yellow V-shaped fleshy tubular process (the osmeterium), from which is diffused a more or less melonlike but disagreeable, in some cases insufferable, odor; the secretion is acid and reddens litmus paper. The mechanism has been described and figured by Klemensiewicz.
When at rest, or retracted, the osmeterium lies in the upper part of the body in the three thoracic segments, and is crossed obliquely by several muscular bundles attached to the walls of the body, and by the action of these muscles the evagination of the osmeterium is strongly promoted. After eversion the tubes are slowly retracted by two slender muscles inserted at the end of each fork or tube, and arising from the sides of the 3d segment behind the head, crossing each other in the median line (Fig. 366, 7 r.m.). The secretion is formed by an oval mass of glandular cells at the base of the forks; in the glandular mass is a furrow-like depression about which the secretory cells are grouped. The secretion collects in very fine drops on the side of each furrow opposite the glandular cells.
According to C. D. Ash the larva of an Australian Notodontid (Danima banksii Lewin) protrudes from the under side of the prothoracic segment a Y-shaped red organ like that of Papilio; no fluid or odor is given out.
Dorsal and lateral eversible metameric sacs in other larvæ.—The showy caterpillars of Orgyia and its allies have a conspicuous coral-red tubercle on the back of the 6th and also the 7th abdominal segment, which on irritation are elongated, the end of the tubercles being eversible. When at rest the summit is crateriform, but on eversion the end becomes rounded and conical. These osmeteria are everted by blood pressure, and retracted by a muscle. Fig. 366, 9, represents a section of an osmeterium of Orgyia leucostigma when 378retracted by the muscle (m); at the bottom of the crater are the secreting or glandular cells (gc), being modified hypodermal cells. These doubtless serve as terrifying organs to ichneumons and other insect enemies, and though we have been unable to detect any odor emanating from the tubercles, yet possibly they give out a scent perceived by and disagreeable to their insect assailants.

Fig. 363.—Freshly hatched larva of Hyperchiria io, with its two pairs of eversible glands (g).
Fig. 364.—Young larva of Megalopyge crispata, enlarged, showing the seven pairs of lateral processes (lp): sp, spiracle; abl′, abl6, six pairs of abdominal legs besides the anal pair.
In the Hemileucidæ there is a pair of lateral osmeteria, on the 1st and on the 7th abdominal segments, which however, are not highly colored (Figs. 363, 366, 10). In Megalopyge (Lagoa, Fig. 364) there is a lateral row of singular pale permanently everted processes which appear to be the homologues of the osmeteria of larvæ of other lepidopterous families. As these are repeated on seven segments, their metameric arrangement is obvious. The relation of these curious glands to the viscera is seen in Fig. 297, lgp, and their minute structure in Fig. 365.
At A, the lumen (l) is a deep narrow cavity, with the secretion (secr.), collected at the mouth of the cavity, composed of a thin, mucus-like, coagulated fluid, containing granules of varying degrees of fineness, which take the stain readily. Outside of these are collected fine nuclei (bc), stained dark, and enveloped in a slight, transparent, pale, protoplasmic envelope, which may be blood corpuscles. The glandular cells themselves are simply modified hypodermal 379cells, as seen at C. In some of the nuclei, indistinct nucleoli are seen, and deeply stained granules, especially around the periphery of the nuclei. At B is represented a section on one side of the middle, but still showing the spacious lumen. In the section represented by C, the knife passed through the process still nearer the outer edge, and near the base; at C1, three of the glandular cells, with their large, deeply stained nuclei, are drawn. A transverse section at D shows the large lumen or cavity (l).
As to the function and homologies of these structures, it is difficult to decide. We have never noticed that they give off any odor, though they may prove to be repugnatorial; they are not visible in the fully grown, living insect, being concealed by the long, dense hairs clothing the body; they are not spraying organs, as they are imperforate at the end, not ending as the lateral, eversible glands of Hyperchiria io, etc., in a crateriform orifice.
They may be permanently everted glands, or osmeteria, which have, by disuse, lost their power of retraction and their crateriform opening, as well as the power of secreting a malodorous fluid.
Fig. 365.—Section of lateral processes of larva of Megalopyge.
In certain of the butterflies, the Heliconidæ (Colænis, Heliconius, Euides, and Dione), there is thrust from the end of the abdomen a pair of large, irregular, rounded, eversible glands, which give out a disagreeable odor, and are consequently repellent, and which seem to be the homologues of the odoriferous glands of other butterflies.
The large, soft, rounded, eversible glands, looking like puff-balls or a rounded pudding (Fig. 366, 12), are everted, when the butterflies are roughly seized, from the dorsal side of the penultimate segment of the abdomen. The males possess two smaller tubercles on the inside of the anal claspers or lobes. Müller also detected, in the females of various species of the Heliconidæ enumerated above, a pair of club-shaped processes like the balancers of flies, which are thrust out on each side of and under the odoriferous puff-balls of the hinder edge of the penultimate segment (Fig. 366, 13). The club or head is armed with hairs or bristles, which, in Heliconius, are like the scales of a butterfly.
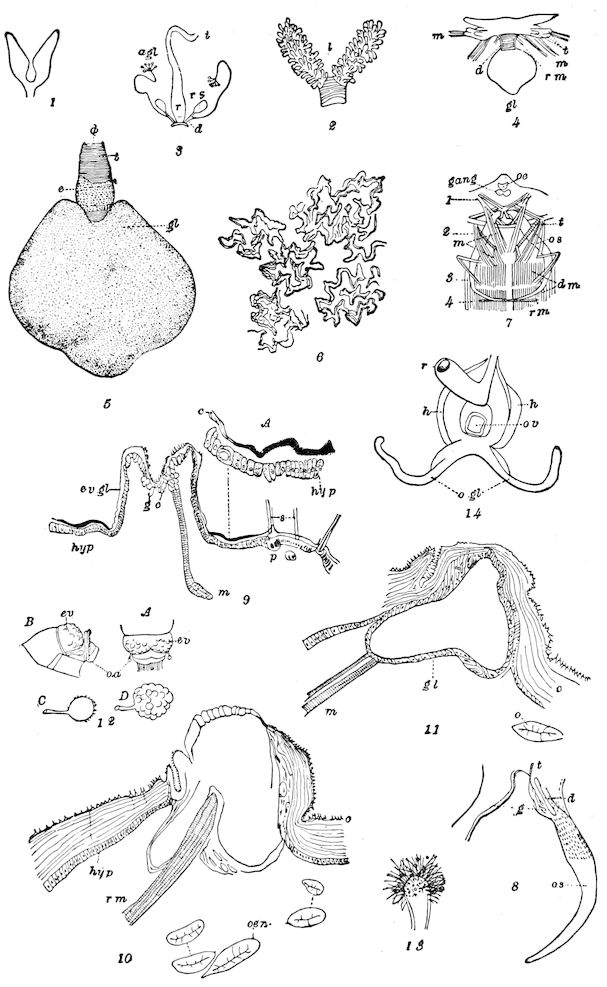
Fig. 366.—Scent-glands of insects: 1. Anal eversible glands of Eleodes.—After Gissler. 2. Anal eversible glands of Blaps.—After Gilson. 3. Anal glands (agl) of Carabus hortensis: rs, reservoir; d, excretory duct; i, intestine; r, rectum.—After Kolbe. 4. Prothoracic spraying apparatus of Cerura vinula: gl, the gland; d, its duct, with tænidia; t, the spraying tubes; m, muscles; rm, retractor muscles.—After Klemensiewicz. 5. The thoracic glandular sac of Macrurocampa marthesia: gl, the glandular sac; d, its duct; e, peritracheal epithelium; t, the spiral threads or tænidia. 6. Irregular separate masses of chitinous ridges on the cuticular lining of the wall of the sacs of Macrurocampa marthesia. 7. Osmeterium (os) of the larva of Papilio machaon at rest: rm, the retractor muscles at the ends; m, the numerous oblique muscles; dm, dorsal longitudinal muscles; t, trachea; oe, œsophagus; gang, brain; 1, head; 2, 3, 4. thoracic segments. 8. Osmeterium (os) of one side, enlarged: g, glandular portion at the base; d, depressions in the cuticula of the glandular portion; t, trachea.—This and Fig. 7 after Klemensiewicz. 9. Eversible dorsal glands (ev. gl) of larva of Orgyia leucostigma in Stage II: gc, glandular cells at bottom of the crater-like depression; m, retractor muscle; p, poison gland-cells of the root of the seta (s); c, cuticula; hyp, hypodermis; A, portion of the cuticle and hypodermis enlarged. 10. Lateral eversible gland of Hyperchiria io, Stage II: rm, retractor muscle; oen, œnocytes. 11. The same as Fig. 10, but representing a section through one side of the eversible gland. 12. A, end of body of Colænis julia; ev, eversible anal gland; oa, odoriferous appendages; B, the same in Heliconius apseudes, side view; C, odoriferous appendages of Colænis dido in fresh condition; D, tested with alcohol and benzine. 13. Odoriferous appendages of Heliconius eucrate, head cleansed.—Figs. 12, 13, after F. Müller. 14. Odoriferous glands (ogl) in the pupa of Vanessa io: r, rectum; h, the folds of hypodermis which forms the terminal papilla of the abdomen; ov, oviduct.—After Jackson.
In the caterpillars of certain blue butterflies (Lycænidæ) is an internal osmeterium, being a very minute sac which is everted from a transverse slit on the top of the 7th abdominal segment. Its function is quite the opposite of those of the caterpillars of other families, since the sac exudes a sweet fluid very attractive to ants, which may be diffused more widely by the delicate spinulose bristles crowning the summit. W. H. Edwards states that in several species of Lycæna, besides that on the 7th abdominal segment, there is on the 8th segment a pair of minute dorsal evaginable tubercles.
A pair of small ramose odoriferous glands are said by Siebold, who regarded them as alluring glands, to occur in Argynnis, Melitæa, and Zygæna, to be situated near the orifice of the oviduct, and Scudder has detected them near the anus of the female pupa of Danais archippus. The appearance of the odoriferous glands in the pupa of Vanessa io is well shown by Jackson (Fig. 366, 14). They develop as two tubular ingrowths of the hypodermis, perfectly distinct one from the other, each having its own separate aperture to the exterior. In Fig. 366, 14 the condition of parts is nearly as in the imago, the glands being situated below the rectum and opening of the oviduct. In both sexes of another Brazilian butterfly (Didonis biblis) on the median line of the abdomen between the 4th and 5th segments are two roundish vesicles covered with short gray hairs, which emit a disagreeable smell.
It is possible that the dark-green fluid in Parnassius, secreted by an evaginable gland, and which is moulded into shape by the scimetar-shaped peraplast (Scudder), is formed by the homologues of the anal glands of other butterflies.
Distribution of repugnatorial or alluring scent-glands in insects[59]
A. Larval Insects
a. Each thoracic segment; sternal. Phryganea grandis.
b. Prothoracic, sternal, discharging a lateral jet of spray; with a single large internal sack.
LEPIDOPTERA
Family Tineidæ
Hyponomeuta evonymella.
Family Noctuidæ
Bryophila, Cucullia formosa, C. scrophulariæ, Habrostola, Cleophana linariæ, Catocala (sp.), Aporia cratægæ, Aplecta nebulosa, Leucania staminea, L. hispanica, L. nonagrioides, Plusia gamma.
Family Notodontidæ
Pheosia rimosa, Schizura concinna, Danima Banksii (Australia), Macrurocampa marthesia, Heterocampa pulverea, Cerura vinula, C. furcula, C. borealis, C. multiscripta.
Family Nymphalidæ
Probably all the species.
c. Prothoracic, dorsal; sending out a V-shaped odoriferous organ (osmeterium).
Family Papilionidæ
All the species as a rule.
d. Thoracic sternal, evaginable glands.
Family Perophoridæ
Lacosoma chirodota, Perophora melsheimerii.
Family Nolidæ
In three, and probably in all the species of Nola.
e. Lateral, abdominal, non-eversible glands, one near each spiracle, emitting a clear fluid.
Family Tenthredinidæ
Cræsus septentrionalis, C. varus, Cimbex americana, C. betulæ, Trichiosoma.
f. Lateral, abdominal, partly eversible glands emitting neither moisture nor odor, but flesh-colored.
Family Tineidæ
Phyllocnistis? (eight pairs.)
Family Hemileucidæ
Hyperchiria io (two pairs, viz. on 1st and 7th segments), H. sp. (Mexico), Hemileuca yavapai, pamina, H. maia, H. artemis, Pseudohazis eglanterina.
g. Lateral, abdominal, permanently everted, metameric glands, not known to secrete a fluid, nor to be odoriferous.
Family Megalopygidæ
Megalopyge crispata.
h. Medio-dorsal, partly eversible glands, emitting a spray of liquid but no odor(?), and colored coral-red or orange-yellow (P. auriflua), but usually in the European species yellowish.
Family Liparidæ
All the species except those of Demas.
i. A single, median, abdominal, dorsal gland, emitting a fluid attractive to ants, on 7th segment; with a pair of minute, index glands on the 8th segment.
Family Lycænidæ
All the species.
j. Protrusile organs near the anus.
Myrmeleon larva (Hagen? Dimmock).
B. Nymph of Heterometabolous Insects
a. Paired, dorsal glands, on abdominal segments 1, 2, and 3.
Cimex lectularius (Künckel).
b. The same on abdominal segment 5.
Lachnus strobi.
C. Pupa of Certain Bombyces
At anterior end of certain pupæ, internal glands to moisten threads of the cocoon for exit of moth.
D. Adult Insects
a. Occurring on the prothorax only; strongly repugnatorial, best developed in ♂.
Anisomorpha buprestoides, Autolyca pallidicornis, Phasma putidum, Phyllium (sp.), Heteropteryx (sp.), Diapheromera femoratum (probably in all the species of the family), Mantis carolina.
b. Occurring on the pro- and mesothorax, and on the middle of the abdomen, orange-yellow, fleshy tubercles or evaginations.
Malachius bipustulatus, Anthocomus equestris, Evæus thoracicus.
c. Segmental, eversible glands, homologues of the coxal glands of other Arthropods, occurring on all, or nearly all, the abdominal segments.
Scolopendrella immaculata (coxal glands on 3d to 11th pair of legs), Campodea staphylinus (a pair of coxal glands on 1st to 8th abdominal segments), Machilis maritima (eversible, coxal glands on segments 1–7).
384d. Occurring in the abdomen.
d1. In the two first abdominal segments.
Corydia carunculigera ♂ and ♀.
d2. Alluring (?) organs situated on the dorsal side of the abdomen, in the 6th, or 6th and 7th, abdominal segment.
Periplaneta americana ♂, P. orientalis (nymph), P. decorata ♂ (nymph), Ectoblatta germanica ♂, Ectobia lapponica ♂, Phyllodromia ♂, Aphlebia bivittata ♂, Platyzosteria ingens (on seventh segment).
e. At the end of the body.
Colænis julia ♀ (F. Müller), Heliconius apseudes (F. Müller).
LITERATURE ON DEFENSIVE OR REPUGNATORIAL GLANDS
Aldrovandus, U. De animalibus insectis libri septem cum singulorum iconibus ad vivum expressis. (Denuo impress: Bonon. apud Clementem Ferronium, 1638, p. 273. The first edition was in 1602.)
Moufet, T. Insectorum sive minimorum animalium theatrum, etc. London, 1634, pp. 185, 186.
Gœdart, J. Metamorphosis naturalis sive insectorum historia, etc. Amstelodami, 1700, Pars ii, p. 136. (French ed. of 1700, ii, p. 162; Lister’s Latin ed., London, 1685, p. 60.)
Réaumur, R. A. F. Mémoires pour servir à l’histoire des insectes, etc. Paris, 1736, ii, pp. 266–269, Pls. 21, 22. [ii, partie ii, pp. 21–23, of the Amsterdam ed. of 1737–1748.]
De Geer, C. Observation sur la propriété singulière qu’ont les grandes chenilles à quatorze jambes et à double queue, du saule, de seringuer de la liqueur. (Mém. sav. étrang, Paris, 1750, i, pp. 530, 531, Pl.; Goetze und Bonnet, etc., Auserlesene Abhandlungen, 1774, p. 220.)
Schaeffer, J. C. Neuendeckte Theile an Raupen und Zweyfaltern, etc. Regensburg, 1754.
Sulzer, J. H. Die Kennzeichen der Insekten, etc. Zürich, 1761, pp. 65–67, Taf. 5, Fig. 34.
Müller, O. F. Pile-larven med dobbelt Hale, og dens Phalæne, etc. Kjöbenhavn, 1772, pp. 53–56, Pl. 2, Figs. 3–5.
Bonnet, C. Mémoire sur une nouvelle partie commune à plusieurs espèces de chenilles. (Mém. math. d. savants étrang., Paris, 1755, ii, pp. 44–52; Collection complète des œuvres de C. Bonnet, 1779, ii, pp. 3–16.)
—— Mémoire sur la grande chenille à queue fourchue du saule, dans lequel on prouve, que la liqueur que cette chenille fait jaillir, est un véritable acide, et un acide très-actif. (Mém. math, de savants étrang., Paris, 1755, ii, pp. 267–282; Collection complète des œuvres de C. Bonnet, 1779, ii, pp. 17–24.)
Amoreux, P. J. Notice des insectes de la France, réputés venimeux, etc. Paris, 1789, pp. 282–285.
Schwarz, C. Neuer Raupenkalender. Nürenberg, 1791, Abth. i, p. 59.
Petzhold, C. P. Lepidopterologische Beyträge. (L. G. Scriba’s Beiträge zu der Insekten-geschichte, Frankfurt am Main, 1793, Heft 3, pp. 230–251.)
Nouvelle Dictionnaire d’Hist. Nat., xv, p. 487. (Larva of Hydrophilus ejects with a slight noise a fœtid and blackish fluid.)
385Rengger, Johann Rudolph. Physiologische Untersuchungen über die thierische Haushaltung der Insecten. Tübingen, 1817. (In the chapter entitled Abgesonderte Säfte bei den Raupen, he speaks of the glandular apparatus of the larva of B. vinula, noticing the general form of the secretory sac, that it opens out in two muscular evertible points, out of which the secretion is ejected.)
Dufour, L. Mémoire anatomique sur une nouvelle espèce d’insecte du genre Brachinus. (Ann. de mus. d’histoire nat., xviii, 1811, pp. 70–81.)
—— Recherches anatomiques sur les carabiques et sur plusieurs autres coléoptères. (Ann. d. Sci. Nat., 1826, viii, pp. 5–54.)
—— Mémoire sur les métamorphosis et l’anatomie de la Pyrochroa coccinea. Glande odorifique. (Ibid., ii sér. Zoologie, xiii, 1840, pp. 340, 341.)
—— Recherches anatomiques sur les Diptères. 1851, pp. 195, 313. (Alimentary canal of Sepsis contains the seat of a “glande odorifique.”)
Kirby and Spence. Introduction to entomology, etc. (2d ed., i, 1815; London, 1818, ii, pp. 238, 239.)
Lyonet, P. Recherches sur l’anatomie et les metamorphoses de différentes espèces d’insectes. Ouvrage posthume. Paris, 1832.
Morren, C. Mémoire sur l’émigration du puceron du pêcher (Aphis persicæ), et sur les caractères et l’anatomie de cette espèce. (Ann. Sci. Natur. Zool., 1836, vi, pp. 65–93, Pls. 6, 7.)
Ratzeburg, J. T. C. Die Forstinsekten, etc. (Theil i, Die Käfer, etc., 1837, p. 246.)
Aubé, C. Note sur une sécrétion fétide d’Eumolpus pretiosus. (Ann. Soc. Ent. Fr., 1837, i, vi; Bull., p. 58.)
Lacordaire, J. S. Introduction à l’entomologie. 1838, ii, p. 45.
Meckel, von Hemsbach, Johann Friedrich. Mikrographie einiger Drüsenapparate der niederen Thiere. (Anat. Phys. u. wiss. Med., 1846, pp. 1–73, Taf. 1–3; p. 46, Müller’s Archiv.)
Stein, Friedrich. Vergleichende Anatomie und Physiologie der Insekten. Berlin, 1847.
Leidy, Joseph. History and anatomy of the hemipterous genus Belostoma. (Journ. Acad. Nat. Sci. Philadelphia, Ser. 2, 1847, i, Part i, pp. 57–67, Pl. 1.)
—— Odoriferous glands of invertebrata. (Proc. Acad. Philadelphia, 1849, iv, 234–236, 1 Pl.; Ann. and Mag. N. H., Ser. 2, 1850, v, pp. 154–156.)
Chapuis et Candèze. Catalogue des larves des coléoptères, etc. (Mém. Soc. Sci. de Liège, 1853, viii, pp. 351–653, Pls. 1–9, pp. 611, 612.)
Siebold, Carl Theodor. Lehrbuch der vergleichenden Anatomie der wirbellosen Thiere, 1848. (Burnett’s transl., Boston, 1854.)
Burnett, Waldo Irving. Translation of Siebold’s Anatomy of the Invertebrates, 1854. (Note on the osmeteria of Papilio asterias, which he regards as an odoriferous and defensive, rather than tactile, organ, p. 415.)
Karsten, H. Bemerkungen über einige shaarfe und brennende Absonderungen verschiedener Raupen. (Müller’s Archiv für Anat. Phys. u. wiss. Med., 1848, pp. 375–382, Taf. 11, 12.) Describes the poison-glands at the base of the spines of Saturnia larvæ.
—— Harnorgane des Brachinus complanatus. (Müller’s Archiv, 1848, pp. 367–376, Taf.)
Laboulbéne, Alexandre. Note sur les caroncules thoraciques du Malachius. (Annales de la Soc. Ent. de France, 3e Sér., vi, 1858, pp. 521–528.)
Saussure, Henri de. Recherches zoologiques de l’Amerique centrale et du Mexique. (6e Partie, Études sur les Myriopodes et les Insectes, Paris, 1860.)
386Gerstaecker, C. E. A. Ueber das vorkommen von ausstülpbaren Hautanhängen am Hinterleibe an Schaben. (Archiv f. Naturgesch., 1861, xxi, pp. 107–115.)
Liegel, Hermann. Ueber den Ausstülpungsapparat von Malachius und verwandten Formen. Inaug. Diss., Göttingen, pp. 31, 1 Taf. (n. d., since 1858 and before 1878.)
Leydig, F. Zur Anatomie der Insecten. (Archiv f. Anat. Phys. u. wiss. Med., 1859, pp. 33–89, 149–183, Taf. 2–4, pp. 35 and 38.)
—— Ueber bombardier Käfer. (Biolog. Centralbl., x, 1890, pp. 395, 396.)
Claus, C. Ueber die Seitendrüsen der Larve von Chrysomela populi. (Zeits. f. wissens. Zool., xi, 1861, pp. 309–314, Taf. xxv.)
—— Ueber Schutzwassen der Raupen des Gabelschwanzes. (Würzburger Naturw. Zeitschrift, 1862, iii, xiv; Sitz. am., 28 Juni, 1862.)
Rogenhofer, Alois. Drei Schmetterlingsmetamorphosen. (Verhandlungen der k. k. zoolog.-bot. Gesellschaft, Wien, xiii, 1862, pp. 1224–1230.)
Fitch, Asa. Eighth report on the noxious and other insects of ... New York. (Trans. N. Y. State Agric. Soc., 1862, xxii, pp. 657–684), p. 677. (Separate.)
Guenée, Achille. D’une organe particulier que présente une chenille de Lycæna. (Annales Soc. Ent. de France, Sér. 4, 1867, pp. 665–668, Pl. 13.)
Landois, L. Anatomie der Bettwanze, Cimex lectularius, mit berücksichtigung verwandter Hemipterengeschlechter. (Zeitsch. f. wissens. Zool., 1868, xvii, pp. 206–224, 218–223, Taf. 11, 12.)
Studer, Theodor. Mittheilungen der naturforsch. Gesellschaft in Bern, 1872–1873, No. 792–811, p. 101.
Candèze, E. Les moyens d’attaque et de défense chez les insectes. (Bull. Acad. royale de Belgique, 2 Sér., xxxviii, 1874, pp. 787–816.)
Mayer, Paul. Anatomie von Pyrrhocoris apterus. (Reichert und du Bois-Reymond’s Archiv f. Anat. Phys., etc., 1874, pp. 313–347, 3 Taf.)
Scudder, Samuel Hubbard. Odoriferous glands in Phasmidæ. (Psyche, i, pp. 137–140, Jan. 14, 1876; Amer. Nat., x, p. 256, April, 1876.)
—— Prothoracic tubercles in butterfly caterpillars. (Psyche, i, pp. 64, 168, 1876.)
—— Organs found near the anus of the ♀ pupa of Danais, which recall the odoriferous organs mentioned by Burnett, transl. Siebold’s Comp. Anat. as occurring in Argynnis and other genera. (Psyche, iii, p. 278, 1882, p. 453, note 22.)
—— Glands and extensile organs of larvæ of blue butterflies. (Proc. Bos. Soc. Nat. Hist., xxxiii, pp. 357, 358, 1888.)
—— Butterflies of Eastern United States. i-iii, 1889.
—— New light on the formation of the abdominal pouch in Parnassius. (Trans. Ent. Soc. London for 1892, January, 1893, pp. 249–253.)
Müller, Fritz. Die Stinkkölbchen der weiblichen Maracujáfalter. (Zeitschr. f. wissens. Zool., 1877, xxx, pp. 167–170, Taf. 9.)
Plateau, Félix. Note sur une sécrétion propre aux coléoptères dytiscides. (Ann. Soc. Ent. Belg., 1876, v, xix, pp. 1–10.)
Edwards, William H. Notes on Lycæna pseudargiolus and its larval history. (Can. Ent., x, Jan., 1878, pp. 1–14. Fig.)
—— On the larvæ of Lycæna pseudargiolus and attendant ants. (Can. Ent., x, July, 1878, pp. 131–136.)
—— Butterflies of North America, i-iv. Many Pls. Phil., 1868—.
Voges, Ernst. Beiträge zur Kenntniss der Juliden. (Zeitsch. f. wissens. Zoologie, xxxi, p. 127, 1878. The scent-glands are retort-shaped bodies, the necks of which open into foramina repugnatoria.)
387Rye, E. C. Secretion of water-beetles. (Ent. Month. Mag., xiv, 1877–1878, pp. 232, 233.)
Forel, A. Der Giftapparat und die anal Drüsen der Ameisen. (Zeits. f. wissens. Zool., 1878, xxx, Suppl., pp. 28–68, Taf. 3, 4.)
Saunders, William. Notes on the larva of Lycæna scudderi. (Can. Ent., x, Jan., 1878, p. 14.)
Weismann, A. Ueber Duftschuppen. (Zool. Anzeiger, 26th Aug., 1878, Jahrg., i, pp. 98, 99.)
Gissler, Carl Friedrich. On the repugnatorial glands in Eleodes. (Psyche, ii, Feb., 1879, pp. 209, 210.)
—— Odoriferous glands on the 5th abdominal segment in nymph of Lachnus strobi. (Fig. 273, p. 804, of Packard’s Report on Forest and Shade Tree Insects, 1890.)
Brunner von Wattenwyl, K. Ueber ein neues Organ bei den Acridiodeen. (Verhandl. k. k. Zool. Bot. Gesells. Wien., 1879, xxix; Sitzungsber., pp. 26, 27.)
—— Verhandl. k. k. Zool. Bot. Gesells. Wien. (A peculiar organ on hind femora of Acridiidæ.)
Rougement, P. Observations sur l’organe détonnant du Brachinus crepitans Oliv. (Bull. Soc. Sci. Nat. Neuchâtel, 1879, xi, pp. 471–478, Pl.)
Goossens, Th. Sur une organe entre la tête et la première paire de pattes de quelques chenilles. (Ann. Soc. Ent. France, ix, p. 4, 1809; Bull., pp. 60, 61.)
—— Des chenilles vésicantes. (Ann. Ent. Soc. France, vi, pp. 461–464, 1887.)
Coquillett, D. W. On the early stages of some moths. (Can. Ent., March, 1880, xii, pp. 43–46.)
Chambers, Victor Tousey. Notes upon some Tineid larvæ. (Psyche, iii, July, 1880, p. 67. Certain retractile processes “from the sides of certain segments of the larva.”)
—— Further notes on some Tineid larvæ. (Psyche, iii, p. 135, Feb. 12, 1881. Larva of Phyllocnistis has eight pairs of lateral pseudopodia on first eight abdominal segments.)
French, G. H. Larvæ of Cerura occidentalis Lint, and C. borealis Bd. (Can. Ent., July, 1881, xiii, pp. 144, 145.)
Passerini, N. Sopra i due tubercoli abdominali della larva della Porthesia chrysorrhœa. (Bull. Soc. Ent. Ital., 1881, xiii, pp. 293–296.)
Klemensiewicz, Stanislaus. Zur näheren Kenntniss der Hautdrüsen bei den Raupen und bei Malachius. (Verhandlungen d. Zool. Bot. Gesellsch. Wien., xxxii, pp. 459–474, 1882, 2 Taf.)
Weber, Max. Ueber eine Cyanwasserstoffsäure bereitende Drüse. (Archiv für Mikr. Anat., xxi, pp. 468–475, xxiv, 1882.)
Bertkau, Philip. Ueber den Stinkapparat von Lacon murinus L. (Archiv f. Naturg., 1882, Jahrg., xlviii, pp. 371–373.)
Dimmock, George. Organs, probably defensive in function, in the larva of Hyperchiria varia Walk. (Saturnia io Harris). (Psyche, iii, pp. 352, 353, Aug. 19, 1882. Account of lateral eversible glands on 1st and 7th abdominal segments; they emit neither moisture nor odor.)
—— On some glands which open externally on insects. (Psyche, iii, pp. 387–399, Jan. 15, 1883. Treats of poison-glands, glandular hairs, eversible glands of Cerura, etc.)
Coleman, N. Notes on Orgyia leucostigma. (Papilio, November-December, 1882, Jan., 1883, ii, pp. 164–166.)
Müller, F. Der Anhang am Hinterleibe der Acræa-weibchen. (Zool. Anzeiger, 6th Aug., 1883, Jahrg., vi, pp. 415, 416.)
388Dewitz, H. Ueber das durch die Foramina repugnatoria entleerte Secret bei Glomeris. (Biol. Centralblatt, iv, pp. 202, 203, 1884.)
Williston, S. A. Protective secretion of Eleodes ejected from anal gland. (Psyche, iv, p. 168, May, 1884.)
Poulton, Edward Bagnall. Notes in 1885 upon lepidopterous larvæ and pupæ, including an account of the loss of weight in the freshly-formed lepidopterous pupæ. (Trans. Ent. Soc., London, June, 1886, pp. 156, 157, 159.)
—— Notes in 1886 upon lepidopterous larvæ, etc. (Trans. Ent. Soc., London, Sept., 1887, pp. 295–301.)
—— Notes in 1887 upon lepidopterous larvæ, etc. (Trans. Ent. Soc., London, 1888, p. 597.)
Künckel-d’Herculais, J. La punaise de lit et ses appareils odoriférants. (Comptes rendus, ciii, 1886, pp. 81–83; Annals & Mag. Nat. Hist., 5th Ser., xviii, 1886, pp. 167, 168.)
—— Étude comparée des appareils odorifiques dans les differents groupes d’Hemiptères hétéroptères. (Compt. rend. Acad. Sc., Paris, cxx, pp. 1002–1004.)
Packard, A. S. The fluid ejected by notodontian caterpillars. (Amer. Nat., 1886, xx, pp. 811, 812.)
—— An eversible “gland” in the larva of Orgyia. (Amer. Nat., 1886, xx, p. 814.)
—— Fifth Rep. U. S. Ent. Comm. Insects injurious to forest and shade trees, p. 136, 1890.
—— Hints on the evolution of the bristles, spines, and tubercles of certain caterpillars. (Proc. Boston Soc. Nat. Hist., xxiv, 1890, p. 551.)
—— Notes on some points in the external structure and phylogeny of lepidopterous larvæ. (Proc. Bost. Soc. Nat. Hist., xxv, 1890, pp. 83–114.)
—— A study of the transformations and anatomy of Lagoa crispata, a bombycine moth. (Proc. Amer. Phil. Soc., Philadelphia, xxxii, 1893, pp. 275–292, 7 Pls.)
—— The eversible repugnatorial scent glands of insects. (Journ., N. Y. Ent. Soc. iii, 1895, pp. 110–127; iv, p. 896; pp. 26–32, 1 Pl.)
Loman, J. C. C. Freies Jod als Drüsensecret. (Tijdschr. Neder. Dierk. Ver. Deel 1, 1887, pp. 106–108.)
Riley, Charles Valentine. Proc. Ent. Soc., Washington, March 13, 1888, i, pp. 87–89.
—— Notes on the eversible glands of larvæ of Orgyia and Parorgyia leucopæa and P. clintonii (achatina). (See 5th Rep. U. S. Ent. Comm., p. 137.)
Denham, Ch. S. The acid secretion of Notodonta concinna. (Insect Life, i, p. 147, 1888; hydrochloric acid.)
Michin, Edward A. Note on a new organ, and on the structure of the hypodermis, in Periplaneta orientalis. (Quart. Journ. Micros. Sc., Dec., 1888, xxiv, 1 Pl.)
—— Further observations on the dorsal gland in the abdomen of Periplaneta and its allies. (Zool. Anz., 27 Jan., 1890, pp. 41–44.)
Maynard, C. L. The defensive glands of a species of Phasma, Anisomorpha buprestoides. (Contributions to Science, i, April, 1889.)
Schaeffer, Cæsar. Beiträge zur Histologie der Insekten. (Zool. Jahrb. Morph. Abth. iii, pp. 611–652, Taf. xxix, xxx, 1889; treats of the ventral glands in prothorax of caterpillars; scales and hairs are secretions from the very greatly enlarged hypodermic cells.)
Gilson, G. Les glandes odorifères der Blaps mortisaga et de quelques autres espèces. (La Cellule, v. pp. 1–21, 1 Pl., 1889.)
—— The odoriferous apparatus of Blaps mortisaga. (Rep. 58th Meeting Brit. Assoc. Adv. Sc., 1889, pp. 727, 728.)
389Haase, Erich. Ueber die Stinkdrüsen der Orthoptera. (Sitzgsber. Ges. Naturf. Freunde, Berlin, pp. 57, 58, 1889.)
—— Zur Anatomie der Blattiden. (Zool. Anz., xii Jahrg., pp. 169–172, 1889.)
Herbst, Curt. Anatomische Untersuchungen an Scutigera coleoptrata. Ein Beitrag zur vergleichenden Anatomie der Articulaten. Dissert., Jena, pp. 36 (Hautdrüsen, Coxal-Organ.); p. 1, 1889.
Wheeler, William M. Hydrocyanic acid secreted by Polydesmus virginiensis Drury. (Psyche, v, p. 422.)
—— New glands in the hemipterous embryo. (Amer. Nat., Feb. 1890, p. 187; odorous(?) glands.)
Jackson, W. Hatchett. Studies in the morphology of the Lepidoptera, Pt. i. (Trans. Linn. Soc., London, 2 Ser., Zoöl., v, May, 1890.)
Krauss, Hermann. Die Duftdrüse der Aphlebia bivittata Brullé (Blattidæ) von Teneriffa. (Zool. Anz., xiii Jahrg., 1890, pp. 584–587, 3 Figs.)
Fernald, H. T. Rectal glands in Coleoptera. (Amer. Nat., xxiv, pp. 100, 101, Pls. 4, 5, 1890.)
Verson, E. Hautdrüsen system bei Bombyciden (Seidenspinner). (Zool. Anzeiger, 1890, pp. 118–120.)
Vosseler, Julius. Die Stinkdrüsen der Forficuliden. (Arch. Mikr. Anat., xxxvi, 1890, pp. 565–578, Taf. 29.)
Carrière, J. Die Drüsen am ersten Hinterleibsringe der Insektenembryonen. (Biol. Centralblatt, xi, pp. 110–127, 1891.)
Borgert, Henry. Die Hautdrüsen der Tracheaten. Inaugural Diss., Jena, 1891, pp. 1–80.
Lang, Arnold. Lehrbuch der vergleichende Anatomie, English Trans. by Henry M. and Matilda Bernard, 1891, pp. 458, 459.
Kennel, J. von. Die Verwandtschaftverhältnisse der Arthropoden. (Schriften herausgegeben von der Naturforscher Gesellschaft bei der Universität Dorpat, vi, Dorpat, 1891.)
Patton, W. H. Scent-glands in the larva of Limacodes. (Can. Ent., xxiii, Feb. 1891, pp. 42, 43; eight pairs of glands with pores along the edges of the back.)
Batelli, Andrea. Di una particolarita nell integumento dell’ Aphrophora spumaria. (Monitore Zoöl. Ital. Anno 2, pp. 30–32, 1891. Dermal glands in the hindermost segment.)
Ash, C. D. Notes on the larva of Danima banksii Lewin. (Ent. Month. Mag., Sept. 1892, p. 232, Fig.) notodontian larva protrudes from under side of prothoracic segment a Y-shaped, red organ like that of Papilio; no odor or fluid given out.
Bernard, Henry M. An endeavor to show that the tracheæ of the Arthropoda arose from setiparous sacs. (Spengel’s Zool., Jahrbuch, 1892, pp. 511–524, 3 Figs.)
Latter, Oswald. The secretion of potassium hydroxide by Dicranura vinula, and the emergence of the imago from the cocoon. (Trans. Ent. Soc. London, 1892, 287, also xxxii; Prof. Meldola adds that the larva of D. vinula secretes strong, formic acid, and is the only animal known to secrete a strong, caustic alkali.)
—— Further notes on the secretion of potassium hydroxide by Dicranura vinula (imago), and similar phenomena in other Lepidoptera. (Trans. Ent. Soc. London; Nature, 1895, p. 551, March 20, 1895.)
Zograff, Nicolas. Note sur l’origine et les parentes des Arthropodes, principalement des Arthropodes trachèates. (Congrès Internationale de Zoologie, 2e Session à Moscow, Aug. 1892; Part i, Moscow, 1892, pp. 278–302, 1892; cyanogenic glands in Myriopods, p. 287.)
390Swale, H. Odor of Olophrum piceum. (Ent. Month. Mag., v, Jan. 1896, pp. 1, 2.)
Cuénot, L. Moyens de défense dans la série animale, Paris, n. d. (1892); the ejection of blood as a means of defence by some Coleoptera. (Comptes rendus, Acad. Sc. France, April 16; Nature, April 26, 1894.)
—— Sur la saignée réflexe et les moyens de défense de quelques insectes. (Arch. Zool. expér. (3), 1897, iv, pp. 655, 679, 680.)
Holmgren, Emil. Studier öfverhudens och de körtelartade hudorganens morfologi hos skandinaviska macrolepidopterlarver. (K. Svenska Vetenskaps-Akademiens Handlingar, xxvii, No. 4, Stockholm, 4º 1895, pp. 82, 9 Pls.)
Lutz, K. G. Das Blut der Coccinelliden. (Zool. Anzeiger, 1895, pp. 244–255, 1 Fig.)
Gilson, Gustav. Studies in insect morphology. (Proc. Linn. Soc. London, March 5, 1896; Nature, p. 500.)
—— On segmentally disposed thoracic glands in the larvæ of the Trichoptera. (Journ. Linn. Soc., London, xxv. 1897.)
Cholodkowsky, N. Entomotomische Miscellen, v, Ueber die Spritzapparate der Cimbiciden Larven, pp. 135–143, 2 Taf. Ibid., vi. Ueber das Bluten der Cimbiciden Larven, pp. 352–357, 1 Fig. (Horæ Soc. Ent. Rossicæ, xxx, 1897.)
Also the writings of Darwin, Wallace, Poulton, Weir, Beddard, Butler, Busgen, (pp. 365–367), Girard, Kolbe, Locy.
THE ALLURING OR SCENT-GLANDS
It is difficult to draw the line between repelling and alluring glands. Attention was first definitely called to the alluring odors of Lepidoptera by Fritz Müller, who showed that the males of certain butterflies are rendered attractive to the other sex by secreting odorous oils of the ether series. He pointed out that the seat of the odor is the androconia (see p. 199), while either repellent or pleasant odors are exhaled from abdominal glands.
Those of Pieris napi yield a scent like that of citrons, Didonis biblis gives off three different odors from different parts of the body, besides having a distinctly odorous spot on the hind wings. Both sexes have a sac between the fourth and fifth abdominal segments which exhales a very unpleasant (protective) odor, while the males have on the succeeding segment a pair of glands from which proceeds an agreeable odor like that of the heliotrope. Callidryas argante throws off a musky odor. In Prepona laertes the odor is like that of a bat, in Dircenna xantho it is vanilla-like, the androconia being situated on the front edge of the hind wings. In Papilio grayi the odor is said to be as agreeable and intense as in flowers. Certain sphingids are known to exhale a distinct odor, which Müller has traced to a tuft of hair-like scales at the base of the abdomen, and which fits into a groove in the first segment, so as to be ordinarily invisible.
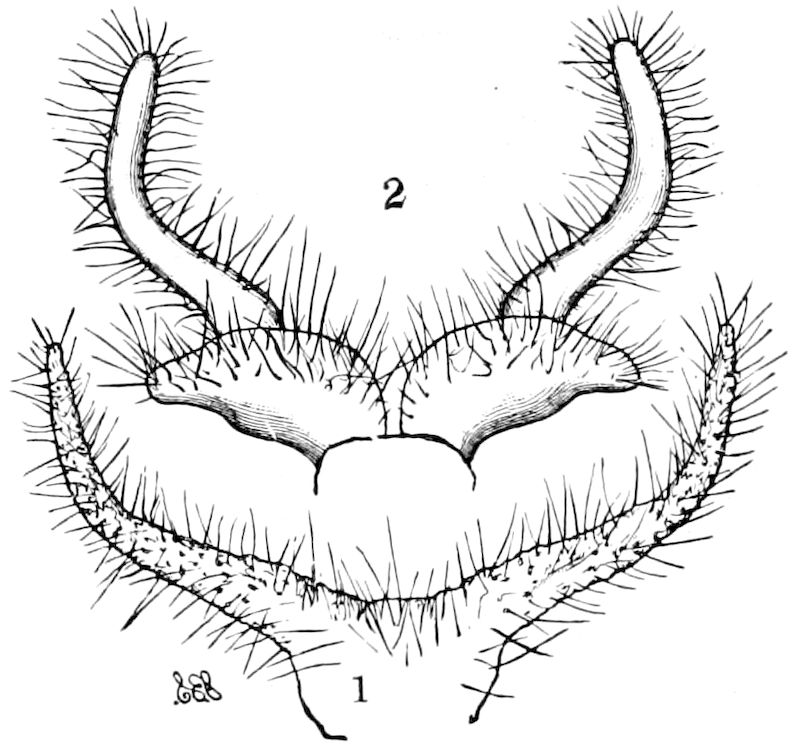
Fig. 367.—Scent-tufts: 1, of Leucarctia acræa; 2, of Pyrrarctia isabella.—After Smith.
In the noctuid genus, Patula, the costal half of the hind wing is modified to form a large scent-gland, and in consequence the venation has been modified. The still greater distortion of the veins in the allied genus, Argida, was attributed by the author to its once having possessed a similar scent-gland, now become rudimentary by disuse. (Hampson.)
Peculiar white or orange-colored, hairy, thread-like processes have been found protruding from narrow openings near the tip of the abdomen of Arctian moths (Fig. 367), which throw off, according to J. B. Smith, “an intense odor, somewhat like the smell of laudanum.” We have perceived the same unpleasant odor emanating from the males of Spilosoma virginica and Arctia virgo, as well as Leucarctia acræa.
We are informed by C. Dury that similar but longer hairy appendages are thrust out by the male of Haploa clymene. Many glaucopid moths protrude similar glandular processes. Thus Müller tells us that on seizing a glaucopid female by the wings, nearly the whole body became enveloped in a large cloud of snow-white wool which came out of a sort of pouch on the ventral side of the abdomen.
The male of a glaucopid was seen to dart out a pair of long hollow hairy retractile filaments which in some species exceed the whole body in length. The 392apparatus secretes a peculiar odor, probably serving to allure the female (Nature), and certain Zygænidæ have on the inner side of the paranal lobes (Afterklappen) glands filled with a sweetly scented fluid. Smith has detected a peculiar brush of hair-like scales in a groove between the dorsal and ventral parts of the basal two segments of the abdomen of Schinia marginata (family Noctuidæ), and when removed it exhaled a laudanum-like smell.
The pupa of Citheronia regalis gives out from the end of the abdomen a scent reminding us of laudanum.

Fig. 368.—Scent-tufts on middle legs of Catocala concumbens.—After Bailey.
Another mode of disseminating pleasant, alluring odors is that of the males of certain moths, which bear pencils and tufts on their fore or hind legs, and in the case of an Indian butterfly on the greatly elongated palpi. Those on the legs are ordinarily concealed in cavities or furrows in the leg, and may be thrust out and expanded so as to widely diffuse their odor. Such are those of the males of Catocala (Fig. 368), which resemble an artist’s fitch brush. In Hepialus hecta, where the arrangements for protecting the tufts are quite abnormal, Bertkau has detected the cells which secrete the odorous fluid. In the male of another Hepialus (H. humuli) a peculiar scent proceeds from the curiously aborted and altered hind tibiæ. (Barrett.) In one case, that of a geometrid moth (Bapata dichroa of New Guinea), these pencils occur on all the legs. (Haase). In many species a distinct odor is perceptible when the leg bearing the pencil or tuft is crushed.
These eversible scent-glands have been supposed to be mostly restricted to the Lepidoptera, and to a single known case in the Trichoptera, but similar alluring male glands also occur in the Orthoptera (Locustidæ). H. Garman has described and figured in the cave cricket (Hadenœcus subterraneus) “a pair of white fleshy appendages protruding from slits between the terga of the 9th and 10th abdominal somites, the nature of which is not clear,” adding, “the slits through which the organs appear are situated one on each side anterior to and a little within the cerci. When fully protruded, the glands are white, cylindrical, a little tapering, and are about one-eighth of an inch long.” He believes that they are protruded during the period of sexual excitement, and suggests that “the sense of smell is certainly the one best calculated to bring the sexes together in the darkness of caves.” We had previously noticed these organs in alcoholic specimens, but supposed that they were fungous growths. 393On dissecting and making microscopic sections of them, the gland is, when extended (Fig. 369), seen to be a long, ensiform, sharp, band-like process, with numerous retractor muscular fibres. When at rest each gland is folded about five times, forming a bundle lying on each side of the end of the intestine. The walls are formed of a single layer of epithelium, as seen in Fig. 369, B.
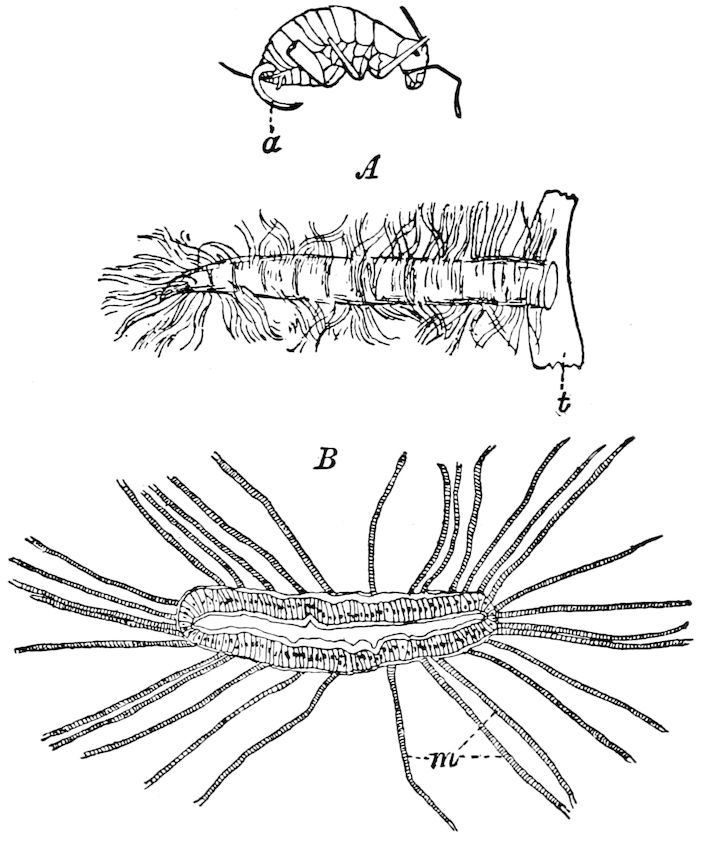
Fig. 369.—Eversible scent-glands (a) of Hadenœcus, nat. size: Kingsley, del.; A, a gland outstretched, with the retractor muscular fibres; t, part of the tergite. B, section of the gland, showing the single layer of epithelial cells, and the muscular fibres (m).—Author del.
In the male of the common wingless cricket, Ceuthophilus maculatus, we have discovered what appears to be a pair of scent-glands lying directly over the last abdominal ganglion. They form two large white sacs situated close together, with a short common duct which passes back and opens externally upwards by a transverse slit on the under side of the last segment of the body.
LITERATURE ON ALLURING GLANDS
Watson, J. On the microscopical examination of plumules, etc. (Ent. Month. Mag., ii, 1865, p. 1.)
—— On certain scales of some diurnal Lepidoptera. (Mem. Lit. and Phil. Soc. Manchester, Ser. 3, ii, 1868, p. 63.)
—— On the plumules or battledore scales of Lycænidæ. (Mem. Lit. and Phil. Soc. Manchester, Ser. 3, iii, 1869, p. 128.) Further remarks, etc. (Ibid., p. 259.)
Anthony, J. Structure of battledore scales. (Month. Microsc. Journ., vii, 1872, p. 250; see also p. 200.)
Morrison, Herbert Knowles. On an appendage of the male Leucarctia acræa. (Psyche, i, pp. 21–22, October, 1874.)
Müller, Fritz. The habits of various insects. (Nature, June 11, 1874, pp. 102–103.)
—— Ueber Haarpinsel, Fitzflecke und ähnliche Gebilde auf den Flügeln männlicher Schmetterlinge. (Jena. Zeitschr. f. Naturw., 1877, xi, pp. 99–114.)
—— Beobachtungen an brasilianischen Schmetterlingen, ii. I. Die Duftschuppen der männlichen Maracujáfalter. (Kosmos, 1877, i, pp. 391–395, Figs. 5, 6.) II. Die Duftschuppen des männchens von Dione vanillæ. (Kosmos, ii, 1877, pp. 38–42, 7 Taf.)
—— As maculas sexuaes dos individuos masculinos das especies Danais erippus e D. gilippus. (Arch. Mus. Nac. Rio Janeiro, ii, 1877 (1878), pp. 25–29, 1 Pl.)
—— Die Duftschuppen der Schmetterlinge (nach dem “Kosmos” in Ent. Nachr., 1878, pp. 29–32, 109).
—— Wo hat der Moschusduft der Schwärmer seinen Sitz? (Kosmos, ii Jahrg., 1878, pp. 84, 85.)
—— Os orgaos odoriferos dos especias Epicalia acontius, Lin. e de Myscelia orsis, Dru. (Arch. Mus. Nac. Rio Janeiro, ii, 1879, pp. 31–35.)
—— Os orgaos odoriferos nas pernas de certos Lepidopteres. (Arch. Mus. Nac. Rio Janeiro, ii, 1879, pp. 37–46, 3 Pls.)
—— Os orgaos odoriferos da Antirrhœa archœa. (Arch. Mus. Nac. Rio Janeiro, iii, 1878, pp. 1–7, 1 Pl.)
—— A prega costal das Hesperideas. (Arch. Mus. Nac. Rio Janeiro, iii, 1880, pp. 41–50, 2 Pls.)
Weismann, August. Ueber Duftschuppen. (Zool. Anzeiger, i, 1878, pp. 98, 99.)
Arnhart, L. Sexundäre Geschlechtscharaktere von Acherontia atropos. (Verh. d. k. k. zool. bot. Ges. Wien, xxix, 1879, p. 54.)
Bertkau, Philipp. Duftapparat an Schmetterlingsbeinen. (Ent. Nachrichten, 1879, Jahrg., pp. 223, 224.)
—— Ueber den Duftapparat von Hepialus hecta. (Archiv f. naturg., xlviii Jahrg., 1882, pp. 363–370, Figs.; also in Biol. Centralblatt., ii Jahrg., 1882, pp. 500–502.)
—— Ergänzung (Duftvorrichtungen bei Lepidopteren). (Ent. Nachr., 1880, p. 206.)
—— Entomologische Mizellen. 1. Ueber Duftvorrichtungen einiger Schmetterlinge. (Verh. d. naturhist. Ver. d. preuss. Rheinlande und Westf., 1884, pp. 343–350.)
Reichenau, W. von. Der Duftapparat von Sphinx ligustri. (Ent. Nachr., 1880, p. 141; also Kosmos, iv Jahrg., 1880, pp. 387–390.)
395Fügner, R. Duftapparat bei Sphinx ligustri. (Ent. Nachr., 1880, p. 166.)
Lelievre, Ernest. (Note in Le Naturaliste, June 1, 1880. Both sexes of Thais polyxena emit an odorous exhalation. Notes on exhalation from Spilosoma fuliginosa.)
Hall, C. G. Peculiar odor emitted by Acherontia atropos. (Entomologist, London, xvi, p. 14.)
Åurivillius, Christopher. Ueber secundäre Geschlechtscharactere nordischer Tagfalter. (Stockholm, 1880, Bihang till K. Svensk. Vet. Akad. Handl., v, pp. 56, 3 Taf.)
—— Des caractères sexuels secundaires chez les papillons diurnes. (Ent. Tidskrift, 1880, pp. 163–166.)
—— Anteckningar om några skandinaviska fjärilarter. (Ent. Tidskr., iv, Årg., 1884, pp. 33–37; Résumé (French), ibid., pp. 55–57.)
Kirby, W. F. Fans on the fore legs of Catocala fraxini. (Papilio, ii, p. 84, 1882.)
Bailey, James S. Femoral tufts or pencils of hair in certain Catocalæ. (Papilio, ii, 1882, pp. 51, 52, 146; also in Stettin Ent. Zeitung, xliii, p. 392.)
Edwards, Henry. Fans on the feet of Catocaline moths. (Papilio, ii, p. 146, 1882.)
Stretch, R. H. Anal appendages of Leucarctia acræa. (Papilio, iii, pp. 41, 42, 1883, 1 Fig.)
Weed, Clarence M. Appendages of Leucarctia. (Papilio, iii, 1883, p. 84.)
Grote, Aug. R. Appendages of Leucarctia acræa. (Papilio, iii, 1883, p. 84.)
Haase, Erich. Ueber sexuelle Charactere bei Schmetterlingen. (Zeitschr. f. Ent., Breslau, N. F., 1885, pp. 15–19, 36–44; also Ent. Nachr., xi Jahrg., pp. 332, 333.)
—— Duftapparate indo-australischer Schmetterlinge. (Corresp. Blatt. Ent. Ver. Iris, Dresden, 1886, pp. 92–107, 1 Taf.; ibid., 1887, pp. 159–178; ibid., 1888, pp. 281–336.)
—— Ueber Duftapparate bei Schmetterlingen. (Sitzgsber. Nat. Ges. Iris, Dresden, 1886, pp. 9–10; Abstr. in Journ. R. Micr. Soc., vi, pp. 969–970, 1886.)
—— Der Duftapparate von Acherontia. (Zeitschr. f. Ent., Breslau, N. F., 1887, pp. 5–6.)
—— Dufteinrichtung indischer Schmetterlinge. (Zool. Anzeiger, 1888, pp. 475–481.)
Dalla Torre, K. W. von. Die Duftapparate der Schmetterlinge. (Kosmos, 1885, ii, pp. 354–364, 410–423; Abstr. by J. B. Smith in Proc. Ent. Soc., Washington, i, pp. 38, 1888.)
Smith, John B. Cosmosoma omphale. (Entomologica Americana, i, pp. 181–185, 1886. Describes and figures cavities in under side of 2d–4th abdominal segments of male, filled with a silky substance. This may be for display to attract ♀, as the whole mass must be very conspicuous when protruded. No odor noticed.)
—— Scent organs in some Bombycid moths. (Entomologica Americana, ii, No. 4, pp. 79–80, 1886. Describes and figures long, slender, forked hairy, orange or white, eversible glands, everted from between 7th and 8th segments of abdomen of ♂ of Leucarctia acræa, Pyrrharctia isabella, Scepsis fulvicollis, and Cosmosoma omphale.)
—— [Notes on odors and odoriferous structures of various moths and a note by L. O. Howard on odor of Dynastes.] (Proc. Ent. Soc., Washington, i, pp. 40, 55, 56.)
Müller, W. Duftorgane der Phryganiden. (Archiv f. Naturgesch., 1887, Jahrg. liii, pp. 95–97.)
396Pollack, W. Duftapparate der Hadena atriplicis und Litargyria. (xv Jahrb. Westphäl. Prov. Ver. Münster, 1887, p. 16.)
Patton, W. H. Scent-glands in the larva of Limacodes. (Can. Ent., 1891, xxiii, pp. 42, 43.)
Garman, H. On a singular gland possessed by the male Hadenœcus subterraneus. (Psyche, 1891, p. 105, 1 Fig.)
Barrett, C. G. Scent of the male Hepialus humuli. (Ent. Month. Mag., Ser. 2, iii, 1892, p. 217. Arises from the curiously aborted and altered hind tibiæ.)
Also the writings of Baillif, Duponchel, F. Müller, Scudder (Psyche, iii, p. 278, 1881), Burgess, Keferstein, Alpheraky, Plateau, Marshall and Nicéville, Wood-Mason, White, Hampson.
THE ORGANS OF CIRCULATION
Although Malpighi was the first to discover the heart in the young silkworm, it was not until 1826 that Carus proved that there was a circulation of blood in insects, which he saw flowing along each side of the body, and coursing through the wings, antennæ, and legs of the transparent larva of Ephemera, though three years earlier Herold demonstrated that the dorsal vessel of an insect is a true heart, pulsating and impelling a current of blood towards the head. This discovery was extended by Straus-Dürckheim, who discovered the contractile and valvular structures of the heart. It is noteworthy that both Cuvier and Dufour denied that any circulation, except of air, existed in insects; and so great an anatomist as Lyonet doubted whether the dorsal vessel was a genuine heart, though he pointed out the fact that there are no arteries and veins connected with this vessel. Another French anatomist, Marcel de Serres, thought that the dorsal vessel was merely the secreting organ of the fat-body.
The so-called peritracheal circulation claimed by Blanchard and by Agassiz has been shown by McLeod to be an anatomical impossibility, the view having first been refuted by Joly in 1849.
Except the aorta-like continuation in the thorax and head which divides into two short branches, there are, with slight exceptions (p. 405), no distinct arteries, such as are to be found in the lobster and other Crustacea, and no great collective veins, such as exist in Crustacea and in Limulus. This is probably the result of a reduction by disuse in the circulatory system, since in myriopods (Julidæ and Scolopendridæ) lateral arteries are said to diverge near the ostia.
a. The heart
The heart or “dorsal vessel” is a delicate, pulsating tube, situated just under the integument of the back, in the median line of the body, and above the digestive canal. It can be partially seen without dissection in caterpillars. It is covered externally and lined within by membranes which are probably elastic; and between these two membranes extends a system of delicate muscular fibres, which generally have a circular course, but sometimes cross each other. The heart is divided by constrictions into chambers, separated by valvular folds. The internal lining membrane referred to forms the valvular folds separating the chambers. Each of these chambers has, at the anterior end, on each side, a valvular orifice (Fig. 370, ostium, i) which can be inwardly closed.
398Miall and Denny thus describe the different layers of the wall of the heart of the cockroach:
“There are: (1) a transparent, structureless intima, only visible when thrown into folds; (2) a partial endocardium, of scattered, nucleated cells, which passes into the interventricular valves; (3) a muscular layer, consisting of close-set, annular, and distant, longitudinal fibres. The annular muscles are slightly interrupted at regular and frequent intervals, and are imperfectly joined along the middle line above and below, so as to indicate (what has been independently proved) that the heart arises as two half-tubes, which afterwards join along the middle. Elongate nuclei are to be seen here and there among the muscles. The adventitia (4), or connective tissue layer, is but slightly developed in the adult cockroach.”
Fig. 370.—Part of the heart of Lucanus cervus: a, the posterior chambers (the anterior ones are covered by a part of the ligaments which hold the heart in place); i, auriculo-ventricular openings; g, g, the lateral muscles fixed by the prolongations h, h, to the upper side of the abdomen.—After Straus-Dürckheim.
Graber says that the heart of insects may be regarded not as an organ de novo, but only as the somewhat modified contractile dorsal vessel of the annelids, in which, however, the transverse arteries arising on each side became, with the gradual development of the tracheæ, superfluous and finally abortive. He describes it as a muscular tube composed of very delicate annular fibres, which within and without is covered by a relatively homogeneous, strong, elastic membrane.
The division into separate chambers is effected by means of a folding inwards and forwards of the entire muscular wall. “A portion of each side of the heart is first extended inwards so as very nearly to meet a corresponding portion from the opposite side, and then, being reflected backwards, forms, according to Straus (Consid., etc., p. 356), the interventricular valve which separates each chamber from that which follows it. Posteriorly to this valve, at the anterior part of each chamber, is a transverse opening or slit (Fig. 371, b), the auriculo-ventricular orifice, through which the blood passes into each chamber, and immediately behind it is a second, but much smaller, semilunar valve (c), which, 399like the first, is directed forwards into the chamber. It is between these two valves on each side that the blood passes into the heart, and is prevented from returning by the closing of the semilunar valve. When the blood is passing into the chamber, the interventricular valve is thrown back against the side of the cavity, but is closed when, by the contraction of the transverse fibres, the diameter of each chamber is narrowed, and the blood is forced along into the next chamber.” (Newport.)
Fig. 371.—A, heart of Lucanus cervus: a, valves or chambers; bb, alary muscles; c, supposed auricular space around the heart. B, division into arteries of the end of the aorta in larva of Vanessa urticæ. C, interior of the chamber, showing the transverse fibres; b, auriculo-ventricular opening and valve into the chambers; c, semilunar valve; d, interventricular valve.—After Straus-Dürckheim, from Newport.
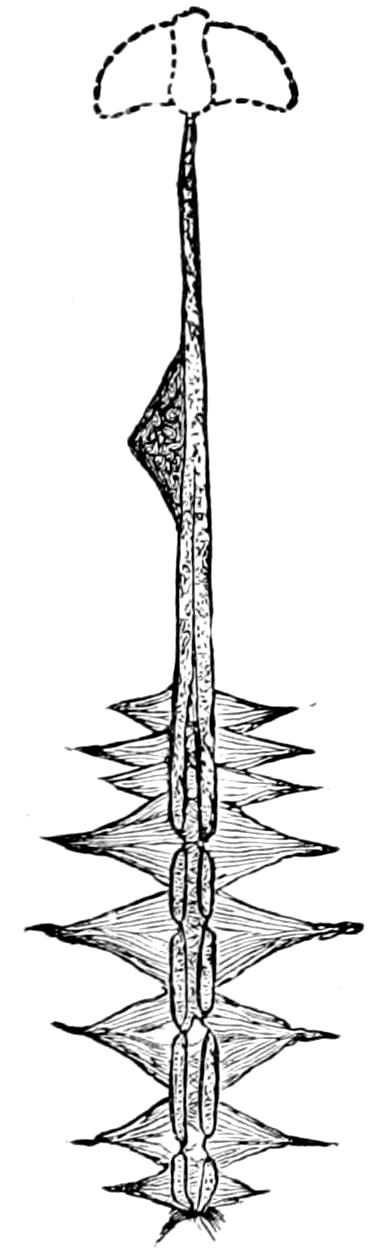
Fig. 372.—Heart of Belostoma.—After Locy.
According to Müller, there is but a single pair of ostia in Phasma, and, in the larva of Corethra, the heart is a simple, unjointed tube, not divided into chambers, and Viallanes states that, in the very young larva of Musca, there are no ostia (Kolbe). In the larva of Ptychoptera, Grobben found a short oval heart, with one pair of ostia situated in the 6th abdominal segment; a long aorta proceeds from it, the thoracic portion of which pulsates; from behind the heart arises a pulsating pouch, which connects with the hinder aorta, which does not pulsate, and ends at the base of two tracheal gills. Burmeister was able to find only four pairs of openings in the larva of Calosoma. Newport states that, while Straus figures nine chambers in Melolontha, and, consequently, eight pairs of openings, he has not been able to observe more than seven pairs of openings in Lucanus cervus. He has invariably found eight pairs of openings both in the larva and imago of Sphinx ligustri, as well as in other Lepidoptera. According to Béla-Dezso, the number of pairs of ostia corresponds to that of the pairs of stigmata.
There also occur, on each side of each chamber, two so-called pear-shaped bodies which are separated from the tubular portion of the heart itself, but, by 400means of muscular fibres, are united with the chamber and with their valves. These pyriform bodies appear as vesicles or cells with granular contents, besides some nuclei with nucleoli. They are of very small size. According to the measurements of Dogiel, in the larva of Corethra plumicornis, they are 0.02 to 0.1 mm. long, and 0.06 to 0.08 mm. broad. He regards these peculiar bodies as apolar nerve-cells of the heart. (Kolbe.)
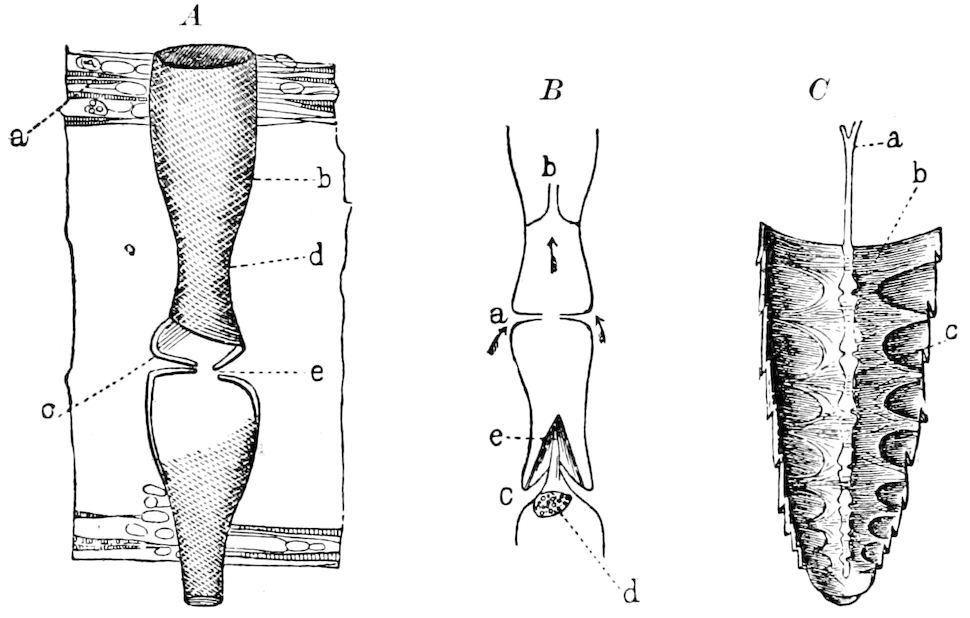
Fig. 373.—A, part of the heart of Dyticus marginalis, showing the spiral arrangement of the muscular fibres; c, closed, e, open, valve; a, dorsal diaphragm with interwoven muscular fibres; b, arrangement of fibres, recalling the screw-like features of the fibres of the human heart; d, narrow end. B, diagrammatic figure of the valvular openings, with the terminal flap (e), and the cellular valve, of a May beetle; a, valvular opening of a dipterous larva, with the interventricular valve (b). C, abdomen of a mole-cricket, ventral view; c, the segmented heart; a, aorta; b, segmented diaphragm under it.—After Graber.
Besides the venous openings of the heart which open into the pericardial region, Kowalevsky has discovered, in the heart of some Orthoptera (Caloptenus, Locusta, etc.), five pairs of openings by which the cardiac chambers receive the blood of the peri-intestinal region. Graber had divided the cœlom of insects into three regions (pericardial, peri-intestinal, and perineural regions), and hitherto only a union of the heart with the pericardial region by slit-like openings was known. These openings are symmetrically distributed on five abdominal segments; each section of the heart in this region has, therefore, four openings, which are all of a truly venous nature. These openings, called cardio-cœlomic apertures, are visible to the naked eye, being situated on conical papillæ of the walls of the heart. These papillæ pass through the outer diaphragm, and open into the peri-intestinal part of the cœlom, in the Acrydiidæ directly, in the Locustidæ through special canals. The cells of the papillæ are spongy, possessing large nuclei, and similar, as a whole, to glandular cells. (Comptes rendus, cxix, 1894.)
The mechanism by which the ostia are closed consists, according to Graber, of an ∞-shaped muscle passing around the two openings, and which, being interlaced, is sufficient to close the openings. But this is not all. The fore and hinder edge of the ostia project, leaf-like, into the cavity of the heart, and thus form, with the outer walls, two valves which, during the systole, filled with the blood rushing in, not only hermetically close the lateral openings, but also, by the simultaneous closure of the entire chamber by the circular muscles in the middle of the same, the two valves, simultaneously approaching each other, so 401nearly touch that they form a transverse partition wall in the chamber. But, for the last purpose, i.e. for the separation of the chambers from one another, there is a very special contrivance. In the May beetle, we find, besides a valve (Fig. 373, B, e), opening into the middle of the chambers, a large, stalked cell (d), which, in the diastole, i.e. in the expansion of the heart, hangs down free on the walls of the heart; but, in the systole or contraction, like a cork, closes the middle of the valve, but does not wholly close the cavity. He has observed, in the larva of Corethra, formal, interventricular valves, which also are not in the middle, but are separated from one another in the interlaced ends. They consist of two longitudinally membranous flaps which move against each other like two valves (Fig. 373, B, b).
“But what is the necessity for such a complicated mechanism? All the blood from behind passes into the heart, and, for its propulsion a simple muscular tube, whose circular fibres would draw together and contract it, would be thought to be sufficient. But the heart, except in some larvæ, ends posteriorly in a blind sac, and the blood can only pass into it by a series of pairs of lateral openings. Now, as regards the reception and the propulsion of the blood forwards, two modes are conceivable. The simplest way would be that the tubular heart should, along its whole length, contract or expand; that, moreover, the blood should be simultaneously sucked in through all the openings, and that then, also, the contraction, or systole, should take place in every part of the heart at the same moment. But this would, plainly, in so long and thin-walled a vessel, be highly impracticable, since, through such a manipulation, the mass of blood enclosed in the heart would be crowded together rather than really impelled forwards. Only the second case could be admissible, and that is this, that each chamber pulsates, one after another, from behind forwards. But, then, each segmental heart must be separated from the others by a valve. To make the matter wholly clear, we may observe an insect heart pulsating, and this is best seen in one of its middle chambers. This chamber expands (simply by the relaxation of its circular muscles), the ostia, also, consequently open, and a given quantity of blood is drawn in from the pericardial cavity. What now would happen after the succeeding contraction if there were no valves between? The blood would not flow forwards, but seek a way out backwards.
“But, in fact, the valve of the hinder chamber, at this time, closes itself, while, by the simultaneous expansion of the anterior ones, their door opens, and this section of the heart, at the same time, causes a sucking in of the contents of the posterior chamber. This phenomenon is repeated, in the same way, from chamber to chamber, which also acts alternately as ventricle and auricle, or by a sucking and pumping action. One is involuntarily reminded of the ingenious manipulation by which, by the alternate opening and shutting of the flood-gates, a vessel is carried along a canal.
“This wave-like motion of an insect’s heart also has the advantage that, just before a pulse-wave has reached the chambers farthest in front, the hinder ones are already prepared for the production of a second, for, as a matter of fact, often 60, and even 100, and, in very agile insects, 150, waves pass, in a single minute, through the series of chambers, which make it very difficult to follow the flowing of their waves.” (Graber.)
The propulsatory apparatus.—But the heart itself is only a part of the entire propulsatorial apparatus to which belongs the following contrivance, the nature of which has been worked out by Graber.
Under the dorsal vessel is stretched a sort of roof-like diaphragm, i.e. a membrane, arched like the dorsal wall of the hind-body which is attached, in a peculiar way, to the sides of the body. The best idea can be gained by a cross-section 402through the entire body (Fig. 374): H is the true dorsal vessel; S, the diaphragm. A surface view is seen at 373, C, b, where it appears as a plate with the edge regularly curved outwards on each side. Its precise mode of working is thus: from each dorsal band of the sides of the abdomen arises a pair of muscles spreading out fan-like, and extending to the heart, so that the fibres of one side pass directly over to those of the other, often splitting apart, or, between the two, extends outwards a perforated, thin web, like an elastic, fibrous sheet (Fig. 373, A, a), with numerous perforations, forming a diaphragm.
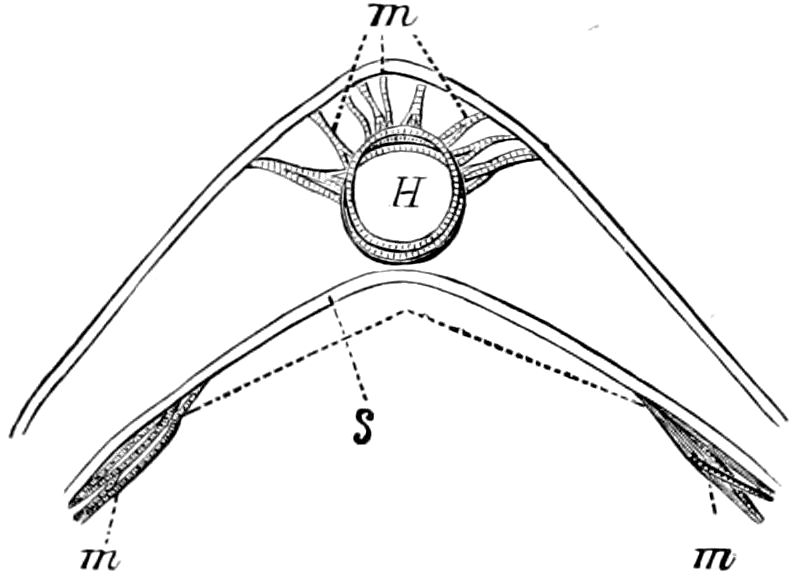
Fig. 374.—Diagram of transverse section of pericardial sinus of Ædipoda cœrulescens: H, heart; s, septum; m, muscles,—the upper suspensory, the lower alary.—After Graber, from Sharp. (See also Fig. 377.)
Graber has thus explained the action of the pericardial diaphragm and chamber, as freely translated by Miall and Denny: “When the alary muscles contract, they depress the diaphragm, which is arched upwards when at rest. A rush of blood towards the heart is thereby set up, and the blood streams through the perforated diaphragm into the pericardial chamber. Here it bathes a spongy or cavernous tissue (the fat-cells), which is largely supplied with air-tubes, and having been thus aerated, passes immediately forwards to the heart, entering it at the moment of diastole, which is simultaneous with the sinking of the diaphragm.”
In the cockroach, however, Miall and Denny think that the facts of structure do not altogether justify this explanation: “The fenestræ of the diaphragm are mere openings without valves. The descent of a perforated non-valvular plate can bring no pressure to bear upon the blood, for it is not contended that the alary muscles are powerful enough to change the figure of the abdominal rings.... The diaphragm appears to give mechanical support to the heart, resisting pressure from a distended alimentary canal, while the sheets of fat-cells, in addition to their proper physiological office, may equalize small local pressures, and prevent displacement. The movement of the blood towards the heart must (we think) depend, not upon the alary muscles, but upon the far more powerful muscles of the abdominal wall, and upon the pumping action of the heart itself.”
“The peculiar office,” says Graber, “performed by the heart has already been stated. It is nothing more than a regulator; than an organ for directing the blood in a determinate course in order that this may not wholly stagnate, or only be the plaything of a force acting in another way, as, for example, through that afforded by the body-cavity and the inner digestive canal. At regular intervals a portion of the blood is sucked through the same, and then by means of the anterior supply tube it is pushed onward into the head, whence it passes into the cavities of the tissues. The different conditions of tension under which the mass of blood stands in the different regions of the body then causes a farther circulation. Besides this, the blood passes through separate smaller pumping apparatuses, and through vessel-like modifications of cavities, also through hollow spaces between the muscles, as, for example, in the appendages where a regular backward and forward flow of the blood, especially in the limbs, wings, antennæ, and certain abdominal appendages takes place. Here and there may occasionally occur a narrow place where the flow of blood is obstructed by the accumulation of the blood corpuscles, causing a considerable stagnation.” (Graber.)
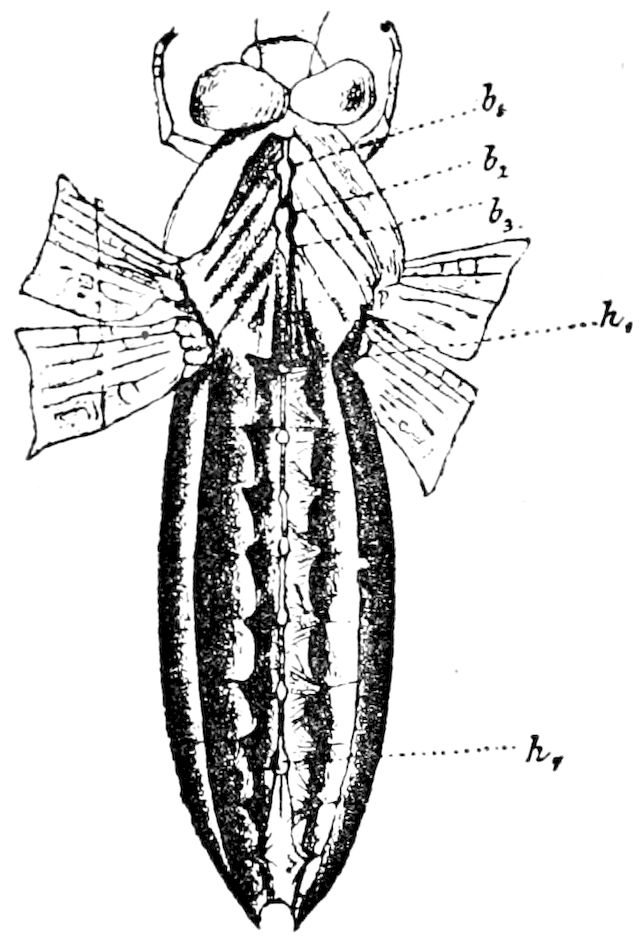
Fig. 375.—Libellula depressa, opened from the back, showing the nervous cord (b1-b3, thoracic, h1-h7, abdominal, ganglia), also the furrow-like ventral sinus closed by a muscular diaphragm.
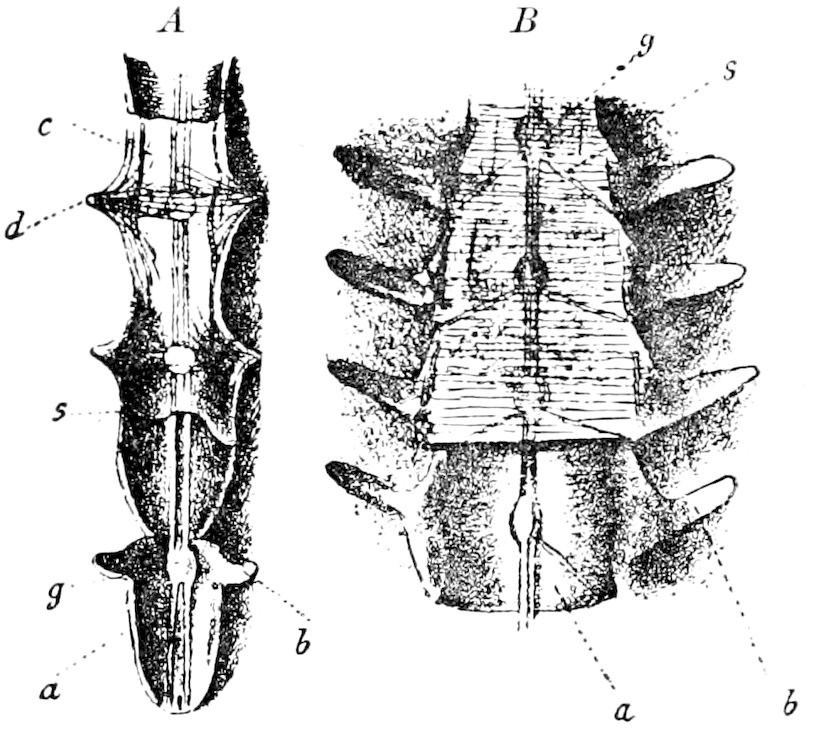
Fig. 376.—A, part of the ventral furrow of Libellula depressa more highly magnified: a, a sternal plate (urite); c, the septum stretched over it, at s in a relaxed or collapsed state; b and d, the wing-like, sternal processes from which the muscular bundles of the diaphragm arise. B, same in Acridium.
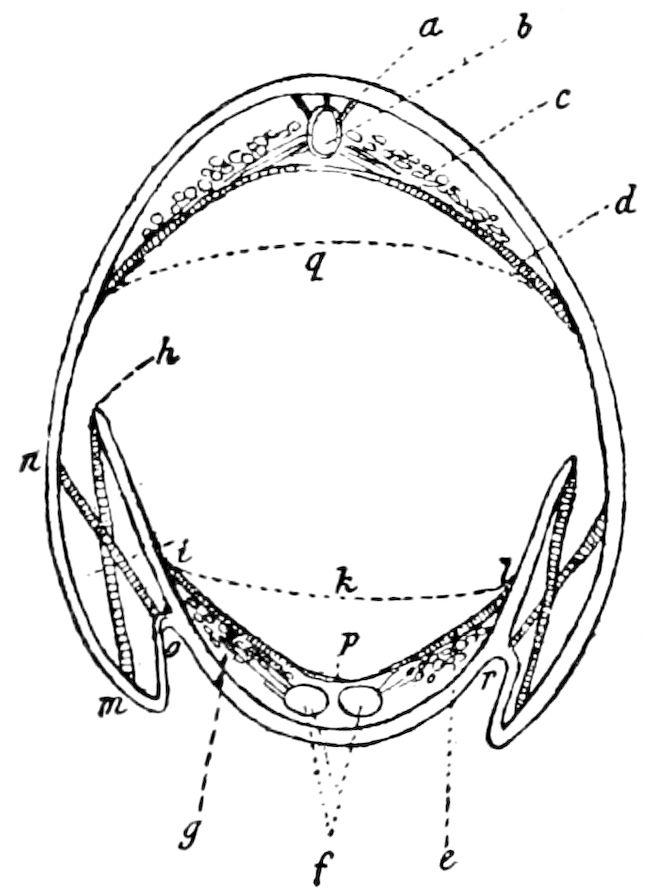
Fig. 377.—Diagrammatic section of the abdomen of Acridium tartaricum, showing the ventral septum (i, p, l) contracted, and (i, k, l) stretched out; oh, rib-like lateral processes of the urite; f, ganglia; b, heart, with its suspensorium (a); c, fat tissue in the pericardial tissue sinus; d, dorsal septum or diaphragm contracted. q, extended; g, fat-body; e, muscular part of diaphragm; no, expiration, hm, inspiration, muscle.—This and Figs. 375, 376, after Graber.
The supraspinal vessel.—In many insects there is a ventral heart acting on the heart’s blood as an aspirator, or more correctly a ventral sinus lying on the nervous cord, and closed by a pulsating diaphragm. This was discovered by Réaumur in the larva of a fly, and by Graber in the dragon-fly and locusts (Acrydiidæ). A glance at Figs. 375 and 376 will save a long description. The ventral wall forms a furrow, and between its borders (Fig. 377, e) extends the diaphragm. During the contraction of the muscles—and this, here, acts from before backwards—the membrane rises up and makes a cavity for the blood, which passes backwards over the nervous cord. The dorsal and ventral sinuses together thus bring about a closed circulation.
It thus appears that the insects are well provided with the means of distribution of their nutritive fluid, and that the blood is kept continually fresh and rich in oxygen. (Graber.)
404The aorta.—While the heart is mostly situated within the abdomen, it is continued into the thorax and the head as a simple, non-pulsating tube, called the aorta. In Sphinx the aorta, as described by Newport, begins at the anterior part of the 1st abdominal segment, where it bends downwards to pass under the metaphragma and enter the thorax; it then ascends again between the great longitudinal dorsal muscles of the wings, and passes onwards until it arrives at the posterior margin of the pronotum; it then again descends and continues its course along the upper surface of the œsophagus, with which it passes beneath the brain, in front of which and immediately above the pharynx, it divides into two branches, each of which subdivides. Newport, however, overlooked a thoracic enlargement of the aorta called by Burgess the “aortal chamber” (Fig. 310, a, c).
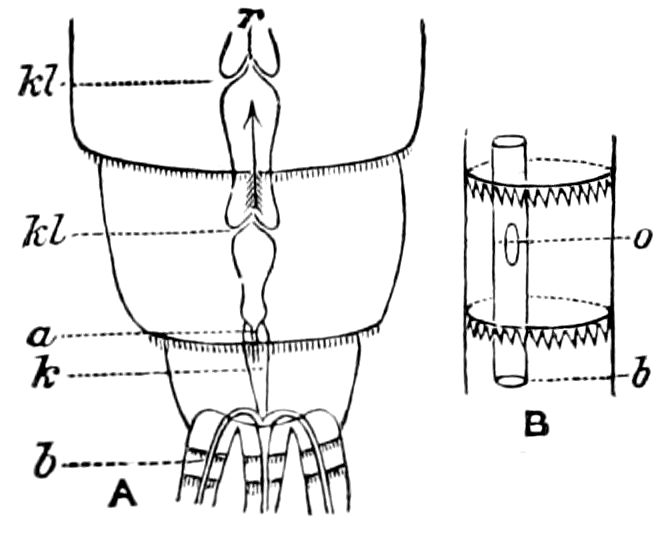
Fig. 378.—A, last three abdominal segments and bases of the three caudal processes of Cloëon dipterum: r, dorsal vessel; kl, ostia; k, special terminal chamber of the dorsal vessel with its entrance a; b, blood-vessel of the left caudal process. B, 26th joint of the left caudal appendage from below: b, a portion of the blood-vessel; o, orifice in the latter.—After Zimmermann, from Sharp.
“In Sphinx and Vanessa urticæ, immediately after the aorta has passed beneath the cerebrum, it gives off laterally two large trunks, which are each equal in capacity to about one-third of the main vessel. These pass one on each side of the head, and are divided into three branches which are directed backwards, but have not been traced farther in consequence of their extreme delicacy. Anterior to these trunks are two smaller ones which appear to be given to the parts of the mouth and antennæ, and nearer the median line are two others which are the continuations of the aorta. These pass upwards, and are lost in the integument. The whole of these parts are so exceedingly delicate that we have not, as yet, been able to follow them beyond their origin at the termination of the aorta, but believe them to be continuous, with very delicate, circulatory passages along the course of the tracheal vessels. It is in the head alone that the aorta is divided into branches, since, throughout its whole course from the abdomen, it is one continuous vessel, neither giving off branches, nor possessing lateral muscles, auricular orifices, or separate chambers.” (Newport, art. Insecta, p. 978.)
Dogiel observed in the transparent larva of Corethra plumicornis that the aorta extends only to the hinder border of the brain. Here it divides into two lamellæ, each of which independently extends farther on. One lamella is seen under the brain and under the eye, the other reaches near the eye. The lamellæ are tied to the integument by threads. At the point of division of the aorta is an opening. (Kolbe.)
True blood-vessels appear to exist in the caudal appendages of the May-flies, as the heart appears to divide and pass directly into them (Fig. 378). The last 405chamber of the heart diminishes in size at the end of the body, and then divides into three delicate tubular vessels which pass into the three caudal appendages, and extend to the end of each one, along the upper side. While the valves of the heart, in all insects, are directed anteriorly because the blood flows from behind, in the larva of the Ephemeridæ the valves of the last chamber of the heart are directed backwards, because from this chamber the blood flows in the opposite direction, i.e. into the caudal appendages. During the contraction of the heart, the elongated section of the same in the last abdominal segment receives a part of the mass of blood contained in the last chamber, which is driven by independent contractions into the caudal appendages. These vessels have openings before the end through which the blood enters into the cavity of the appendages, and can also pass back, in order to be taken up by the body cavity. It is possible that these blood-vessels stand in direct relation to respiration. (Zimmermann, Creutzburg, in Kolbe, p. 544.)
The pericardial cells.—Along the heart, on both sides, occur the so-called pericardial cells, which differ from the fat-cells, and also the peritracheal cells of Frenzel, and are mostly arranged in linear series, which have a close relation to the circulation of the blood. In the larva of Chironomus, they lie in groups; in that of Culex, they are arranged segmentally. In caterpillars, these pericardial cells are not situated in the region of the heart, but are arranged linearly on the side, and form a network of granulated cells situated between the fat-bodies. Other rows of these cells are situated near the stigmata and the main lateral tracheæ. (Kolbe.)
According to Kowalevsky, the pericardial cells, and the garland-shaped, cellular cord consist of cells, whose function it is to purify the blood, and to remove the foreign or injurious matters mingled with the blood.
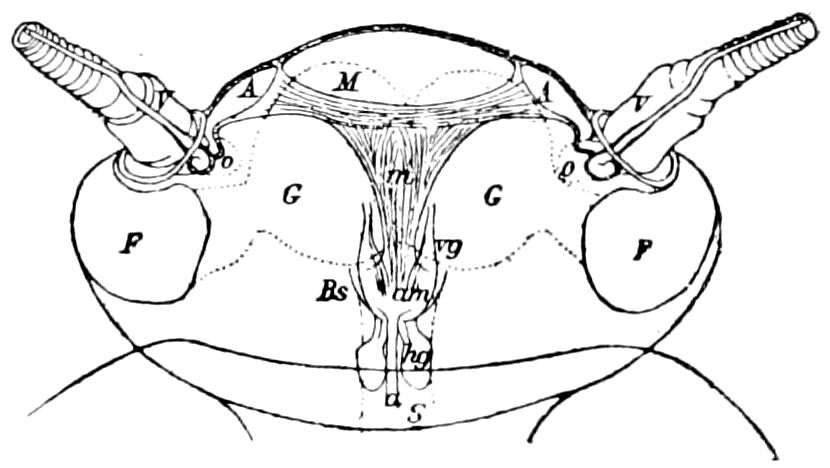
Fig. 379.—Diagram of the circulatory organs in the head of the cockroach, seen from above: A, ampulla; V, antennal vessel; M, chief muscular cord; m, muscular band; Bs, wall of the blood sinus; am, opening of the aorta (a); rg, anterior sympathetic or visceral ganglion; hg, hinder visceral ganglion; F, F, facetted eyes; o, vestigial ocellus; G, G, brain; S, œsophagus.—After Pawlowa.
Ampulla-like blood circulation in the head.—In the head of the cockroach occurs, according to Pawlowa, a contractile vascular sac at the base of each antenna. The cavity has a valvular communication with the blood space below and in front of the brain, and muscle-fibres effect systole and diastole. Each sac is beyond doubt an independently active part of the circulatory system. These organs also occur in Locusta and other Acrydiidæ, and Selvatico has described similar structures in Bombyx mori and certain other Lepidoptera.
Pulsatile organs of the legs.—Accessory to the circulation is a special system of pulsatile organs in the three pairs of legs of Nepidæ, generally situated in the tibia just below its articulation with the femur, but in the fore legs of Ranatra, in the clasp-joint or tarsus, just below its articulation with the tibia. First observed by Behn (1835), Locy has studied the organ (Fig. 380) in Corixa, Notonecta, Gerris, besides the Nepidæ. It is a whip-like structure attached at both ends, with fibres extending upward and backward to the integument of the leg, separate from the muscular fibres and does not involve them in its motions, and is not affected by the muscles themselves. “As the blood-corpuscles flow near the pulsating body they move faster, and around the organ itself there is a whirlpool of motion.” The beating of these organs aids the circulation in both directions, and when the motion ceases, the blood-currents in the legs stop; the rate of the pulsating organ is always faster than that of the heart, and the action is automatic.
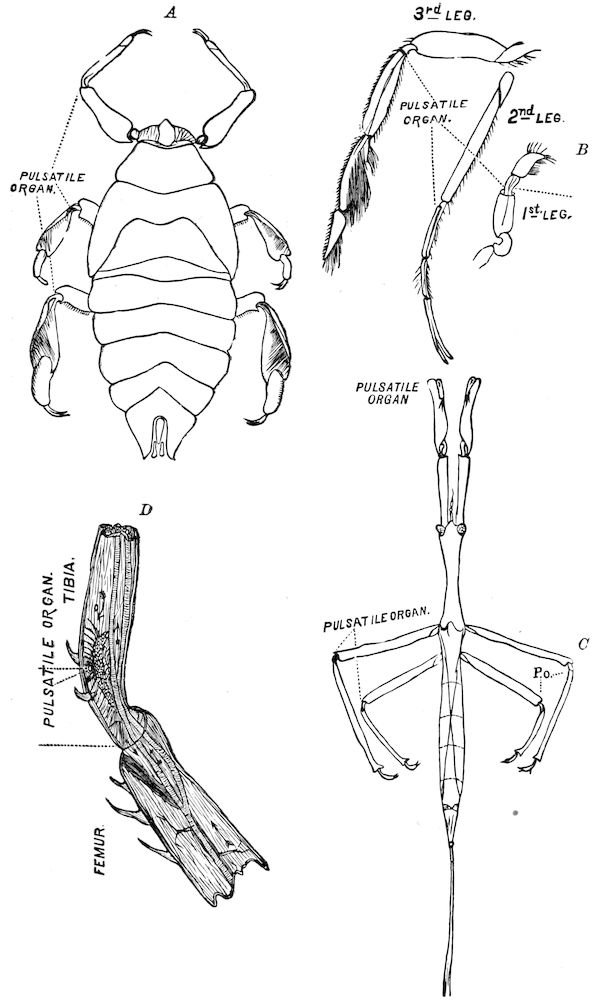
Fig. 380.—Pulsating organs in Hemiptera: A, Belostoma nymph, B, legs of Corixa. C, Ranatra, adult, to show the exceptional position of the pulsating organ in the fore legs. D, pulsatile organ in tibia of Ranatra.—After Locy.
b. The blood
The blood of insects, as in other invertebrates, differs from that of the higher animals in having no red corpuscles. It is a thin fluid, a mixture of blood (serum) and chyle, usually colorless, but sometimes yellowish or reddish, which contains pale amœboid corpuscles corresponding to the white corpuscles (leucocytes) of the vertebrates, though they are relatively less numerous in the blood of insects. The yellow fluid expelled from the joints of certain beetles (Coccinella, Timarcha, and the Meloidæ) is, according to Leydig, only the serum of the blood. In phytophagous insects the blood is colored greenish by the chlorophyll set free during digestion. The blood of Deilephila euphorbia is colored an intense olive-green, and that of Cossus ligniperda is pale yellow. (Urech.) The blood of case-worms (Trichoptera) is greenish. In some insects it is brownish or violet. The serum is the principal bearer of the coloring material, yet Graber has shown that in certain insects the corpuscles are more or less beset with bright yellow or red fat-globules, so as to give the same hue to the blood.
The leucocytes.—The corpuscles are usually elongated, oval, or flattened oat-shaped, with a rounded nucleus, or are often amœbiform; and they are occasionally seen undergoing self-division. When about to die the corpuscles become amœbiform or star-shaped. (Cattaneo.) Their number varies with the developmental stage of the insect, and in larvæ increases as they grow, becoming most abundant shortly before pupation. The blood diminishes in quantity in the pupal stage, and becomes still less abundant in the imago. (Landois.) The quantity also varies with the nutrition of the insect, and after a few days’ starvation nearly all the blood is absorbed. Crystals may be obtained by evaporating a drop of the blood without pressure; they form radiating clusters of pointed needles. The freshly drawn blood is slightly alkaline. (Miall and Denny.)
The size of the corpuscles has been ascertained by Graber, who found that the diameter of the circular blood-disks of the leaf-beetle, Lina populi, is 0.006 mm.; of Cetonia aurata and Zabrus gibbus, 0.008 to 0.01 mm.; and those of certain Orthoptera (Decticus verrucivorus, Ephippiger vitium and Œdipoda cœrulescens), 0.011 to 0.014 mm. The longest diameter of the elongated corpuscles of Carabus cancellatus is 0.008 mm.; of Gryllus campestris, Locusta viridissima, Cossus ligniperda, Sphinx ligustri (pupa), and others, 0.008 to 0.01 mm.; of Caloptenus 408italicus, Saturnia pyri, Anax formosus, and others, 0.011 to 0.014 mm.; of Ephippiger vitium, Œdipoda cœrulescens, Pezotettix mendax, Zabrus gibbus, Phryganea, and others, 0.012 to 0.022 mm.; in Stenobothrus donatus and variabilis, 0.012 to 0.035 mm. The largest known are those of Melolontha vulgaris, which measure from 0.027 to 0.03 mm.
Fig. 381.—Blood corpuscles, or leucocytes, of insects: A, a-g, of Stenobothrus dorsatus (the same forms occur in most Orthoptera and in other insects). B, a, leucocyte of the same insect with the nucleus brought out by ether; b, another of serpentine shape. C, leucocytes of the same insect after a longer stay in ether. D, leucocytes of the same after being in glycerine 14 days.—After Graber.
As regards the nature of the corpuscles, Graber concludes that they are more like the cells of the fat-bodies than genuine cells. That they are not true cells is shown by the fact that after remaining in their normal condition a long time they finally coalesce and form cords. After shrivelling, or after the blood has been subjected to different kinds of treatment, the nucleus is clearly brought out (Fig. 381).
Besides the blood corpuscles there have been detected in the blood round bodies which are regarded as fat-cells. They are circular, and for the most part larger than the blood corpuscles, have a sharp, even, dark outline, and an invariably circular nucleus. (Kolbe.)
The blood of Meloe, besides the amœboid corpuscles, according to Cuénot, contains abundant fibrinogen, which forms a clot; a pigment (uranidine), which is oxidized and precipitated when exposed to the air; a dissolved albuminoid (hæmoxanthine), which has both a respiratory and nutritive function; and, finally, dissolved cantharidine.
The corpuscles arise from tissues which are very similar to the fat-bodies, and which, at given times, separate into cells. The position of these tissues is not always the same in different insects. In caterpillars, they occur in the thorax, near the germs of the wings; in the saw-flies (Lyda), in all parts of the thorax and abdomen; in larval flies (Musca), in the end of the abdomen, just in front of the large terminal stigmata. The place where the blood corpuscles are formed is usually near, or in relation with, the fat-bodies. But while the fat-bodies 409mostly serve as the material for the formation of the blood-building tissues, in caterpillars the tracheal matrix also, and, in dipterous larvæ, the hypodermis serve this purpose. (Cæsar Schaeffer in Kolbe. See also Wielowiejski, Ueber das Blutgewebe.)
Other substances occur in the blood of insects. Landois (1864) demonstrated the existence of egg albumen, globulin, fibrin, and iron in the blood of caterpillars. Poulton found that the blood of caterpillars often contained chlorophyll and xanthophyll derived from their food plants. A. G. Mayer has recently found that the blood (hæmolymph) of the pupæ of Saturniidæ (Callosamia promethea) contains egg albumin, globulin, fibrin, xanthophyll, and orthophosphoric acid, and Oenslager has determined that iron, potassium, and sodium are also present. (Mayer.)
c. The circulation of the blood
Every part of the body and its appendages is bathed by the blood, which circulates in the wings of insects freshly emerged from the nymph or pupal state, and even courses through the scales of Lepidoptera, as discovered by Jaeger (Isis, 1837).
In describing the mechanism of the heart we have already considered in a general way the mode of circulation of the blood.
The heart pumping the blood into the aorta, the nutritive fluid passes out and returns along each side of the body; distinct, smaller streams passing into the antennæ, the legs, wings (of certain insects), and into the abdominal appendages when they are present. All this may readily be observed in transparent aquatic insects, such as larval Ephemeræ, dragon-flies, etc., kept alive for the purpose under the microscope in the animalcule box.
Carus, in 1827, first discovered the fact of a complete circulation of the blood, in the larva of Ephemera. He saw the blood issuing in several streams from the end of the aorta in the head and returning in currents which entered the base of the antennæ and limbs in which it formed loops, and then flowing into the abdomen, entered the posterior end of the heart. Wagner (Isis, 1832) confirmed these observations, adding one of his own, that the blood flows backward in two venous currents, one at the sides of the body and intestine, and the other alongside of the heart itself, and that the blood not only entered at the end of the heart, but also at the sides of each segment, at the position of the valves discovered by Straus-Dürckheim.
Newport maintains that the course of the blood is in any part of the body, as well as in the wings, almost invariably in immediate connection with the course of the tracheæ, for the reason that “the currents of blood in the body of an insect are often in the vicinity of the great tracheal vessels, both in their longitudinal and transverse direction across the segments.”
410The circulation of the blood in the wings directly after the exuviation of the nymph or pupa skin, and before they become dry, has been proved by several observers. As stated by Newport, the so-called “veins” or “nervures” of the wings consist of tracheæ lying in a hollow cavity, the peritracheal space being situated chiefly under and on each side of the trachea.
Fig. 382.—Circulation of the blood in hind wing of Periplaneta orientalis: the arrows indicate the usual direction of the blood currents.—After Moseley.
Newport gives the following summary of the observations of the early observers, to which we add the observations of Moseley. “A motion of the fluids has been seen by Carus in wings of recently developed Libellulidæ, Ephemera lutea and E. marginata, and Chrysopa perla; among the Coleoptera, in the elytra and wings of Lampyris italica and L. splendidula, Melolontha solstitialis and Dytiscus.” Ehrenberg saw it in Mantis, and Wagner in the young of Nepa cinerea and Cimex lectularius. Carus detected a circulation in the pupal wings of some Lepidoptera, and Bowerbank witnessed it in a Noctuid (Phlogophora meticulosa); Burmeister observed it in Eristalis tenax and E. nemorum, and Mr. Tyrrel in Musca domestica, but it has not been observed in the wings of Hymenoptera.
Bowerbank observed
that in the lower wing
of Chrysopa perla the
blood passes from the
base of the wing along
the costal, post-costal,
and externo-medial veins,
outwards to the apex of
the wing, giving off smaller currents in its course, and that it returns along
the anal vein to the thorax. He found that the larger veins, 1
408 in. in diameter,
contained tracheæ which only measured 1
2222 of an inch in diameter;
but in others the tracheæ measured 1
1340, while the cavity measured only 1
500
of an inch. He states, also, that the tracheæ very rarely give off branches
while passing along the main veins, and that they lie along the canals in a
tortuous course. (Newport, art. Insecta, p. 980.)
Bowerbank, also, in his observations on the circulation in the wings of Chrysopa, 411“used every endeavor to discover, if possible, whether the blood has proper vessels, or only occupied the internal cavities of the canals; and that he is convinced that the latter is the case, as he could frequently perceive the particles not only surrounding all parts of the tracheæ, and occupying the whole of the internal diameter of the canals, but that it frequently happens that globules experienced a momentary stoppage in their progress, occasioned by their friction against the curved surface of the tracheæ, which sometimes gave them a rotatory motion.”
Fig. 383.—Parts of a vein of the cockroach, showing the nerve (n) by the side of the trachea (tr); c, blood corpuscles.—After Moseley.
Moseley found, owing to the large size and number of the corpuscles, that the circulation of the blood in the wings of insects is most easily observed in the cockroach, especially the hind wings. As seen in Moseley’s figure, the blood flows outward from the body through the larger veins (I and II) of the front edge of the wings, which he calls the main arteries of the wings, and more generally returns to the body through the veins in the middle of the wing; the blood also flows out from the body through the inner longitudinal veins (those behind vein IV), and the blood is also seen to flow through some of the small cross-veins. Fig. 383 shows one of the main trunks during active circulation. The corpuscles change their form readily, “the spindle-shaped ones doubling up in order to pass crossways through a narrow aperture.... In the irregularly formed corpuscles, which seem to represent leucocytes amœboid movements were observed.... Corpuscles pass freely above and under the tracheæ, showing that these latter lie free in the vessels.” The hypodermis lining the vessels is best seen in the small transverse veins.
The pulse or heart-beat of insects varies in rapidity in different insects, rising at times of excitement, as Newport noticed in Anthophora retusa, to 142 beats in a minute.
When an insect, as, for example, a tineid caterpillar, has been enclosed in a tight box for a day or more, the pulsations of the heart are very languid and slow, but soon, on giving it air, the pulsations will, as we have observed, rise in frequency to about 60 a minute, Herold observed 30 to 40 in a minute in a fully-grown silkworm, and from 46 to 48 in a much younger one. Suckow observed but 30 a minute in a full-grown caterpillar of Gastropacha pini, and 18 only in its pupa.
In a series of observations made by Newport on Sphinx ligustri from the fourth day after hatching from the egg until the perfect insect was developed, he found that before the larva cast its first skin the mean number of pulsations, 412in a state of moderate activity and quietude, was about 82 or 83 a minute; before the second moult 89, while before the third casting it had sunk down to 63; and before its fourth to 45, while, before leaving its fourth stage, and before it had ceased to feed, preparatory to pupating, the pulse was not more than 39. “Thus the number gradually decreases during the growing larva state, but the force of the circulation is very much augmented. Now when the insect is in a state of perfect rest, previously to changing its skin, the number is pretty nearly equal at each period, being about 30. When the insect has passed into the pupa state it sinks down to 22, and subsequently to 10 or 12, and after that, during the period of hibernation, it almost entirely ceases. But when the same insect which we had watched from its earliest condition was developed into the perfect state in May of the following spring, the number of pulsations, after the insect had been for some time excited in flight around the room, amounted to from 110 to 139; and when the same insect was in a state of repose, to from 41 to 50. When, however, the great business of life, the continuation of the species, has been accomplished, or when the insect is exhausted, and perishing through want of food or other causes, the number of pulsations gradually diminishes, until the motions of the heart are almost imperceptible.” Insects, then, he remarks, do not deviate from other animals in regard to their vital phenomena, though it has been wrongly imagined that the nutrient and circulatory functions are less active in the perfect than in the larval condition.
The heart of a larval Gastrus equi taken the day previous from a horse’s stomach beat from 40 to 44 times a minute (Scheiber); while Schröder van der Kolk observed only 30 beats in the same kind of maggot.
In the larva of Corethra, while at rest, the heart contracts from 12 to 16 or 18 times a minute, but when active the number rises to 22. The systole and diastole last from 5 to 6 minutes. (Dogiel.)
Temperature also affects the pulsations, as they increase in frequency with a rise and decrease with a fall in temperature.
Influence of electricity.—The influence of electricity on the action of the insect’s heart, from Dogiel’s experiments, is such as to cause an acceleration in the frequency of the beats, while an increase in the strength of the electric currents either diminishes the frequency of the beats or entirely stops the heart’s action. A violent excitation with the induction current causes a systole when the heart’s action has stopped for a long time; and if the excitation lasts uninterruptedly, then the contractions after a while become noticeable, according to the strength of the current. In such a case there are, however, interruptions in the regularity, strength, and order of the contractions. (Kolbe.)
Effects of poisons on the pulsations.—Dogiel has also experimented on the influence of poisons in the form of vapor or as liquid solutions on the pulsations of insects, which is much as in vertebrates. The application of carbonic oxide to the larva of Corethra, whose heart one minute previous to the poisoning beat 15 times a minute, accelerated the heart-beats in about 55 minutes to 25 pulsations in a minute. Afterwards there was a retardation in the pulse to the normal beat. Carbonic acid had a similar effect.
The following results obtained by Dogiel are somewhat as tabulated by Kolbe:—
I. Substances which cause the pulsations of the heart to accelerate.
II. Substances retarding the heart’s action.
III. Substances whose action is indifferent.
The above-named substances comprise those which in the vertebrates effect a change in the activity of the motor nerve-ganglia of the heart and the muscular fibres. Hence it follows that the heart of the larval Corethra consists of muscular fibres provided with ganglia, and that the contractions of the muscular fibres are provoked through the agency of the ganglia. But since muscarine, atropine, and curare, whose influence in stopping the heart’s action of vertebrates is known, in insects either have no action or only make the pulsations slower; it seems to follow that the heart of the larval Corethra possesses no similar apparatus for lessening the heart’s action, and this is also confirmed by anatomical studies. On the contrary, aconite acts, as we must from observations conclude, exclusively on the motor centres and the muscles, but not on the apparatus for lessening the heart’s action, which, as has been remarked, is not present in the larval Corethra. (Kolbe ex Dogiel.)
Dewitz has discovered an onward movement of the blood corpuscles, somewhat independent of the general circulation. This independent motion of the blood corpuscles is not only a creeping one like the amœboid motion of the white corpuscles of vertebrates, but they have besides a peculiar swimming movement. Dewitz noticed this in the hind wings of a recently emerged meal-worm beetle (Tenebrio molitor), still white and soft, after they had been cut off. The tissues forming the matrix within the wings constitute a network filled with blood. The current of blood within the wing thus cut off may be stopped flowing by a tap on the firmly clamped object-bearer on which the wing is placed, or by drawing it by an apparatus described by the same author, to incite in one way or another the blood corpuscles to swim forwards. When a corpuscle is disposed to move, we see it first stirring restlessly, or wabbling about, in this way changing its form; then it moves forwards, and does not come to a standstill. If it remains still there, after a while, by tapping, it begins again its movements.
“Should one yet doubt the fact of this spontaneous movement of the blood corpuscles, he will surely be convinced of its correctness by observing the so-to-speak reluctantly springing motion of a blood corpuscle in the wing of Tenebrio molitor with the simultaneous change of appearance and shape of the corpuscle.”
This spontaneous or independent motion of the blood corpuscles is also produced by the heating apparatus. As soon as the corpuscles lie still in the severed wing and they are warmed, the corpuscles begin to pass through the meshes of the tissue. When cooled, the motion ceases, but as soon as the temperature rises to a certain grade, the corpuscles again move onwards.
To explain this independent motion Dewitz thinks that they take up and then expel the blood-fluid, and in this way cause their motion. This independent motion is necessitated, in order that the stream of blood may become so regulated, 414that the blood corpuscles shall not be arrested in their course, but even turn back again out of the farther end of the antennæ and limbs. The chief mechanical power for the blood circulation must go on independently of the propulsatorial apparatus and of the heart. (Kolbe.)
LITERATURE ON THE HEART AND ON THE CIRCULATION OF THE BLOOD
a. Anatomy of the organs
Meckel, J. F. Ueber das Rückengefäss der Insekten. (Meckel’s Archiv, i, 1815, pp. 469–476.)
Müller, J. G. De vasi dorsali Insectorum. Berolini, 1816, pp. 22.
Serres, P. Marcel de. Observations sur les usages du vaisseau dorsal ou sur l’influence que le cœur exerce dans l’organisation des animaux articulés, etc. (Ann. du Mus. d’hist. nat., 1818, iv, pp. 149–192, 313–380, 2 Pls.; v, 1819, pp. 59–147, 1 Pl.)
Herold. Physiologische Untersuchungen über das Rückengefäss der Insekten. (Schriften d. Gesellsch. z. Beförderung d. Naturk. in Marburg, 1823, i, pp. 41–107.)
Carus, C. G. Entdeckung eines einfachen vom Herzen aus beschleunigten Blutkreislaufes in den Larven netzilügliger Insekten. Leipzig, 1827, pp. 40, 3 Taf.
—— Fernere Untersuchungen über Blutlauf in Kerfen. (Acta Acad. Leopold. Carol., 1831, xv, pp. 1–18, 1 Taf.)
Stadelmayr, L. Ansichten vom Blutlauf nebst Beobachtungen über das Rückengefäss der Insekten. Diss. München, 1829, pp. 24.
Berthold. Beitrage zur Anatomie, Zoologie und Physiologie. Göttingen, 1831.
Treviranus, G. R. Ueber das Herz der Insekten, dessen Verbindung mit den Eierstocken und ein Bauchgefäss der Lepidopteren. (Zeitschr. f. d. Physiologie, von F. Tiedemann. G. R. u. L. C. Treviranus, 1832, iv, pp. 181–184, 1 Taf.)
—— Beobachtungen aus der Zootomie und Physiologie. Bremen, 1839.
Wagner, R. Beobachtungen über Kreislauf des Blutes und den Bau des Rückengefässes bei den Insekten. (Isis, 1832, iii, p. 30; vii, pp. 320–331, 778–783, Fig.)
Bowerbank, J. S. Observations on the circulation of the blood in insects. (Ent. Mag., 1833, i, pp. 239–244, 1 Pl; also in Müller’s Archiv f. Physiolog., 1834, i, pp. 119–120.)
—— Observations on the circulation of the blood and the distribution of the tracheæ in the wing of Chrysopa perla. (Ent. Mag., 1837, iv, pp. 179–185.)
Jaeger. Ueber die Entdeckung von einer Bewegung in den Schuppen des Schmetterlingsflügel. (Isis, 1837, v, p. 512.)
Behn, W. Découverte d’une circulation de fluide nutritif dans les pattes de plusieurs insectes hémipteres. (Ann. Sc. nat., 1835, Sér. 2, iv, pp. 1–12.)
Newport, G. Insecta, in Todd’s Cyclopædia of Anatomy and Physiology, 1839. London, pp. 853–994. On the circulation of the blood, p. 976, Figs.
Duvernoy, G. L. Résumé sur le fluide nourricier, ses réservoirs et son mouvement dans tout règne animal. (Ann. Sc. nat., 1839, Sér. 2, xii, pp. 300–346.)
Dufour, L. Études anatomiques et physiologiques sur une mouche dans le but d’eclairer l’histoire des metamorphoses et de la prétendue circulation dans les insectes. (Ann. Sc. nat. Zool., Sér. 2, 1841, xvi, pp. 5–14.)
415—— Note sur la prétendus circulation dans les insectes. (Compt. rend. Acad., Paris, 1844, xix, pp. 188–189.)
—— Études anatomiques et physiologiques sur une mouche, dans le but d’éclaircir l’histoire des metamorphoses et de prétendue circulation des insectes. (Mém. mathémat. des Savants étrangers, Paris, 1846, ix, pp. 545–628, 1 Pl.)
—— Sur la circulation dans les insectes. Bordeaux, 1849, 8º, pp. 40. (Compt. rend. Acad. Sci., Paris, 1849, xxviii, pp. 28–33, 101–104, 163–170.)
—— De la circulation du sang et de la nutrition chez les insectes. Bordeaux, 1851. (Act. Soc. Linn., Bordeaux, 1851, xvii, p. 9.)
—— Études anatomiques et physiologiques et observations sur les larves des Libellules, Appareil circulatoire. (Annal. Sci. nat., Sér. 3, Zool., xvii, 1852, pp. 98–101, 1 Pl.)
Schröder van der Kolk, J. S. C. Mémoire sur l’anatomie et physiologie du Gastrus equi. (N. Verhandl. Kl. Nederl. Instit., 11, 1845, pp. 1–155, 13 Pl.)
Nicolet, H. Note sur la circulation du sang chez les Coléoptères. (Ann. Sc. nat., 1847, Sér. 3, vii, pp. 60–64.)
Verloren, C. Mémoire en réponse à la question suivante: éclaircir par des observations nouvelles le phénomène de la circulation dans les insectes, en recherchant si on peut la reconnaître dans les larves de différents ordres de ces animaux. (Mém. couronn. et Mém. d. savants étrang. de l’Acad. Roy. Belgique, xix, 1847.)
Blanchard, E. De la circulation dans les insectes. (Ann. Sc. nat., 1848, Sér. 3, ix, pp. 359–398, 5 Pls.)
—— Sur la circulation du sang chez les insectes et sur la nutrition. (Compt. rend. Acad. Sc., Paris, 1849, xxviii, pp. 76–78; 1851, xxxiii, pp. 367–370.)
—— Nouvelles observations sur la circulation du sang et la nutrition chez les insectes. (Ibid., pp. 371–376.)
Joly, N. Mémoire sur l’existence supposée d’une circulation péritrachéenne chez les insectes. (Ann. Sc. nat. Zool., Sér. 3, 1849, xii, pp. 306–316.)
Bassi, C. A. Rapporto alla sezione di zoologia, anatomia comparata e fisiologia del congresso di Venezia, sul passagio delle materie ingerite nel sistema tracheale degli insetti. (Gazette di Milano, 1847, vi; also Ann. Sc. nat. Zool., Sér. 3, 1851, xv., 362–371.)
Agassiz, Louis. On the circulation of the fluids in insects. (Proceed. Amer. Assoc. Adv. Sc., 1849, pp. 140–143; Ann. Sc. nat. Zool., Sér. 3, xv, 1851, pp. 358–362.)
Leydig, F. Anatomisches und Histiologisches über die Larve von Corethra plumicornis. (Zeitschr. f. wissen. Zool., iii, 1851, pp. 435–451.)
Wedl, C. Ueber das Herz von Menopon pallidum. (Sitzungsber. der k. Akad. d. Wissensch. Wien., 1855, xvii, pp. 173–180.)
Scheiber, S. H. Vergleichende Anatomie und Physiologie der Oestriden Larven. (Sitzungsber. d. k. Akad. d. Wiss. Wien. Math.-naturwiss. Cl., xli, 1860, pp. 409–496, 2 Taf.; The circulatory system, pp. 463–490.)
Brauer, Fr. Beitrag zur Kenntnis des Baues und der Funktion der Stigmenplatten der Gastrus-Larven. (Verhdl. d. k. k. zool.-bot. Gesellsch. Wien., xiii, 1863, pp. 133–136.)
Moseley, H. N. On the circulation in the wings of Blatta orientalis and other insects, and on a new method of injecting the vessels of insects. (Quart. Jour. of Micr. Science, xi, n. s., pp. 389–395, 1871, 1 P1.)
Graber, V. Ueber die Blatkörperchen der Insekten. (Sitzber. Akad. Wien. Math.-naturw. Classe, lxiv, 1871, pp. 9–44, 1 Taf.)
416—— Vorläufiger Bericht über den propulsatorischen Apparat der Insekten. (Sitzber. d. k. Ak. d. Wiss. Wien., lxv, 1872, pp. 16, 1 Taf.)
—— Ueber den propulsatorischen Apparat der Insekten. (Archiv f. mikroskop. Anatomie, ix, 1873, pp. 129–196, 3 Taf.)
—— Ueber den pulsierenden Bauchsinus der Insekten. (Archiv f. mikroskop. Anat., xii, 1876, pp. 575–582, 1 Taf.)
Grobben, Carl. Über bläschenförmige Sinnesorgane und eine eigenthümliche Herzbildung der Larve von Ptychoptera contaminata L. (Sitzb. k. Akad. Wissensch. Wien., 1875, lxxii, p. 22, 1 Taf.)
Liebe, Otto. Ueber die Respiration der Tracheaten, besonders über den Mechanismus derselben und über die Menge der ausgeatmeten Kohlensäure. Inaug.-Diss. Chemnitz, 1872, pp. 28.
Dogiel, John. Anatomie und Physiologie des Herzens der Larve von Corethra plumicornis. (Mém. Acad. imp. St. Petersbourg, 7 Sér., xxiv, 1877, Nr. 10, pp. 37, 2 Pls.) Separate, Leipzig, Voss.
Bütschli, O. Ein Beitrag zur Kenntnis des Stoffwechsels, insbesondere der Respiration bei den Insekten. (Reichert’s und Du Bois-Reymond’s Archiv f. Anatomie u. Physiologie, 1874, pp. 348–361.)
Béla-Dezso. Ueber den Zusammenhang des Kreislaufs und der respiratorischen Organe bei den Arthropoden. (Zool. Anzeiger, i Jahrg., 1878, p. 274.)
Plateau, F. Communication préliminaire sur les mouvements et l’innervation de l’organe central de la circulation chez les animaux articulés. (Bull. Acad. roy. de Belgique, Sér. 2, xlvi, 1878, pp. 203–212.)
Jaworovski, Ant. Ueber die Entwicklung des Rückengefässes und speziell der Muskulatur bei Chironomus und einigen anderen Insekten. (Sitzgsber. d. k. Akad. d. Wissensch. Wien. Math.-naturwiss. Cl., lxxx, 1879, pp. 238–258.)
Zimmermann, O. Ueber eine eigentümliche Bildung des Rückengefässes bei einigen Ephemeridenlarven. (Zeitschr. f. wissens. Zool., 1880, xxxiv, pp. 404–406.)
Burgess, E. Note on the aorta in lepidopterous insects. (Proc. Bost. Soc. Nat. Hist., xxi, 1881, pp. 153–156, Figs.)
—— Contributions to the anatomy of the milk-weed butterfly, Danais Archippus F. (Anniversary Memoirs Boston Soc. Nat. Hist., 1880, pp. 16, 2 Pls.)
Vayssière, A. Recherches sur l’organisation des larves des Éphémérines. (Ann. Sc. nat. Zool., Sér. 6, xiii, 1882, pp. 1–137, 11 Pls.)
Viallanes, H. Recherches sur l’histologie des insectes et sur les phénomènes histologiques qui accompagnent le développement post-embryonnaire de ces animaux. (Ann. Sc. nat., Sér. 6, xiv, 1882, pp. 1–348, 18 Pls.)
Schimkewitsch, W. Ueber die Identität der Herzbildung bei den wirbel und wirbellosen Tieren. (Zool. Anzeiger, 1885, viii Jahrg., pp. 37–40, Fig.)
—— Noch etwas über die Identität der Herzbildung bei den Metazoen. (Zool. Anzeiger, 1885, pp. 384–386.)
Creutzburg, N. Ueber den Kreislauf der Ephemerenlarven. (Zool. Anzeiger, 1885, pp. 246–248.)
Poletajewa, Olga. Du cœur des insectes. (Zool. Anzeiger, 1886, ix Jahrg., pp. 13–15.)
Selvatico, S. L’aorta nel corsaletto e nel capo della farfalla del bombice del gelso. (Padova, 1887, p. 19, 2 Pls.)
—— Die Aorta im Brustkasten und im Kopfe des Schmetterlings von Bombyx mori. (Zool. Anzeiger, 1887, x Jahrg., pp. 562–563.)
Kowalevsky, A. Ein Beitrag zur Kenntnis der Excretionsorgane. (Biol. Centralbl., 1889, ix, pp. 33–47, 65–76, 127–128.)
417Tosi, Alessandro. Osservazioni sulla valvola del cardias in varii generi della famiglia delle Apidi. (Ricerche Lab. Anat. R. Univ. Roma, v, 1895, pp. 5–26, 16 Figs., 3 Pls.)
Pawlowa, Mary. Ueber ampullenartige Blutcirculationsorgane im Kopfe verschiedener Orthopteren. (Zool. Anzeiger, xviii Jahrg., 1895, pp. 7–13, 1 Fig.)
Also the writings of Kolbe.
b. The blood, blood corpuscles, leucocytes, and blood tissue
Wagner, R. Ueber Blutkörperchen bei Regenwürmern, Blutegeln und Dipterenlarven. (Müller’s Archiv f. Anatomie u. Physiologie, 1835, pp. 311–313.)
—— Nachtrage zur vergleichenden Physiologie des Blutes. (Archiv f. Anat. u. Physiologie, 1838.)
Newport, G. On the structure and development of the blood. First series. The development of the blood corpuscle in insects and other invertebrata, and its comparison with that of man and the vertebrata. (Abstr. of the paper in Roy. Soc., 1845, v, pp. 544–546: also in Ann. Mag. Nat. Hist., Ser. 3, 1845, iii, pp. 364–367.)
Landois, H. Beobachtungen über das Blut der Insekten. (Zeitschr. f. wissens. Zool., xiv, 1864, pp. 55–70, 3 Pl.)
——, and L. Landois. Ueber die numerische Entwicklung der histiologischen Elemente d. Insektenkörpers. (Ibid., xv, 1865, pp. 307–327.)
Rollett, A. Zur Kenntnis der Verbreitung des Hämatins. (Sitzgsber. d. k. Akad. d. Wiss. Wien., lxiv, 1871.)
Wielowiejski, H. v. Ueber das Blutgewebe der Insekten. Eine vorläufige Mitteilung. (Zeitschr. f. wissens. Zool., 1886, xliii, pp. 512–536.)
MacMunn, C. A. Researches on myohæmatin and the histohæmatins. (Proc. Roy. Soc. London, 1886, xxxix, pp. 248–252.)
Peyron, J. Sur l’atmosphere interne des insectes comparée à celle des feuilles. (Compt. rend. Acad. Sc., Paris, 1886, cii, pp. 1339–1341.)
Cuénot, L. Études sur le sang, son rôle et sa formation dans la série animale. Part 2, invertébrés; note préliminaire. (Arch. Zool. Expériment., 1888, Sér. 2, v, pp. xliii-xlvii. See also ibid., Sér. 3, 1897, pp. 655, 679–680.)
Dewitz, H. Die selbstandige Fortbewegung der Blutkörperchen der Gliedertiere. (Naturwiss. Rundschau. Braunschweig, 1889, iv Jahrg., pp. 221–222.)
—— Eigenthätige Schwimmbewegung der Blutkörperchen der Gliedertiere. (Zool. Anzeiger, 1889, xii Jahrg., pp. 457–464, Fig.)
Schäffer, C. Beitrage zur Histiologie der Insekten. II, Ueber Blutbildungsherde bei Insektenlarven. (Spengel’s Zool. Jahrbücher Abt. f. Anat. u. Ontogenie, iii, 1889, pp. 626–636, 1 Taf.)
Cattaneo, G. Sulla morfologia delle cellule ameboidi dei Molluschi e Artropodi. (Boll. Sc. Pavia. Anno 11, 1889, p. 59, 2 Pls.)
Wagner, W. A. Ueber die Form der körperlichen Elemente des Blutes bei Arthropoden, Würmern und Echinodermen. (Biolog. Centralblatt, 1890, x, p. 428.)
Preyer, W. Zur Physiologie des Protoplasma. II, Die Funktionen des Stoffwechsels. Die Saftströmung. (Patanie’s Naturwiss. Wochenschr., 1891, vi, pp. 1–5.)
Cholodkowsky, N. Ueber das Bluten der Cimbiciden-Larven. (Entomologische Miscellen, vi, Horæ Soc. Ent. St. Petersburg, 1897, pp. 352–357, 1 Fig. The fluid thrown out through pores or fissures in the skin is the blood.)
With the writings of Korotaiev, Tichomeroff, Pékarsky, Balbiani, Korotneff, Cuénot, and others.
c. The fat-bodies
Dufour, L. Recherches anatomiques sur les Carabiques et sur plusieurs autres insectes Coléoptères. Du tissu adipeux splanchnique. (Ann. Sc. nat., viii, 1826, pp. 29–35.)
—— Histoire comparative des métamorphoses et de l’anatomie des Cetonia aurata et Dorcus parallelepipedus. Tissu adipeux splanchnique. (Ann. Sc. nat., Zoologie, Sér. 2, 1842, xviii, pp. 178–179.)
Meyer, H. Ueber die Entwicklung des Fettkörpers, der Tracheen und der keimbereitenden Geschlechtsteile bei den Lepidopteren. (Zeitschr. f. wissens. Zool., 1849, i, pp. 175–179, 4 Taf.)
Fabre, J. H. Étude sur le rôle du tissu adipeux dans la sécretion urinaire chez les insectes. (Ann. Sc. nat., Sér. 4, xix, 1862, pp. 351–382.)
Leydig, Fr. Einige Worte über Fettkörper der Arthropoden. (Reichert, u. du Bois-Reymond’s Archiv f. Anat., 1863, pp. 192–203.)
Lindemann, K. Zoologische Skizzen. 1. Struktur des Fettkörpers. (Bull. Soc. Imp. d. Natural. Moscou, 1864, pp. 521–526, 1 Pl.)
Landois, Leonh. Ueber die Funktion des Fettkörpers. (Zeitschr. f. wissens. Zoologie, xv, 1865, pp. 371–372.)
Wielowiejski, H. v. Ueber den Fettkörper von Corethra plumicornis und seine Entwicklung. (Zool. Anzeiger, 1883, vi Jahrg., pp. 318–322.)
—— Ueber das Blutgewebe der Insekten. (Zeitschr. f. wissens. Zool., 1886, xliii, pp. 512–536.)
Kowalevsky, A. O. Sur les organes excréteurs chez les arthropodes terrestres. (Congrès internat. Zool., 2me Sess., pp. 196–205, Moscou, 1892.)
THE BLOOD TISSUE
Under this name Wielowiejski has included several important tissues or cellular bodies intimately concerned with the nutrition of the insect. These are:—
a. The fat-body
In the body cavity of winged insects and of their larvæ occur yellowish masses of large cells filled with small drops of fat, and forming the “fat-body.” It is of various shapes, more or less lobulated or net-like, and covers or envelops parts of the viscera, also forming a layer under the integument (Fig. 143). The tracheal endings are usually enveloped by the fat-body. It is larger in the larvæ than in the adults, especially in Lepidoptera, in them forming a reserve of nutrition, used during metamorphosis and during the formation and ripening of the eggs and male cells.
Wielowiejski has shown that there is a regular arrangement of the fat-body in the general cavity of the body. For example, in the larva of Chironomus occur the following forms of this tissue. Around the periphery, on each side of the body cavity, is a loose network of lobes with large meshes constituting the peripheral layer or external lobular fat-body; these lobular masses are segmentally arranged.
Within these segmental lobes, on each side of and along the digestive tract, extending along through almost the entire body, is an unbroken strand of this tissue, forming the internal fat-body cords. From the first larval stage, and even before hatching, its cells are so unusually large, being filled with large, clear, mostly colorless fat-drops, that their limits cannot be defined, and their nuclei can only with great difficulty be detected. Only in some large larvæ of Chironomus has Wielowiejski found clearly defined cells; the protoplasm of these cells contain almost no fat-drops.
The fat-body is of mesodermal origin, and as Wheeler insists, is not derived from the œnocytes, as supposed by Graber. Formed from the mesoderm, it is a differentiation of portions of the cœlomic walls, and therefore metameric in origin. That the fat-body gives origin to the blood corpuscles Wheeler is doubtful.
420The fat-cells are distinct, spherical, and as a rule possess only one nucleus, though in those of Apis and Melophagus there are two nuclei, and in Musca several. Sometimes the cells contain a substance like the white of an egg, and concretions of uric acid, or these take the place of the fat-drops. The presence of uric acid shows that a very active metabolism goes on in the fat-body. “In some cases it has been proved that the fat-body in the larva is rich in fat and poor in concretions of uric acid, while in the imago it is poor in fat and rich in concretions of uric acid” (Lang).
Leydig, in 1857 (Lehrbuch der Histiologie), spoke of the presence of dark concretions in the fat-body, and afterwards (1864) showed that there was a wide distribution of uric acid salts and concretions. Witlaczil, also, has detected concretions in the fat-body of the Psyllidæ, in larval Cecidomyiidæ, in the larvæ and pupæ of ants, and in the pupa of Musca.
The physiological processes which take place in the fat-bodies are obscure. Graber regarded the whole system of the fat-bodies as “a single, many-lobed lung,” while before him Landois, taking into account the intimate relation existing between the finer tracheal branches and the fat-body, considered that the latter was concerned in respiration. Marchal thinks that the fat-body is a urinary organ, as the urates are formed within the cells of this body.
Moreover, Schäffer maintains that a special kind of fat-body cell has the important function of taking up and giving out nutritious matters during the internal processes of metamorphosis, while he also believes that there is a genetic connection between the fat-body and the blood corpuscles—a view combated by Wheeler.
Kowalevsky finds that the fat-body remains absolutely insensible to the action of the substances which stained the Malpighian tubes (p. 352). So long as the cells are healthy and living they are not stained and do not absorb the colors in question; and this insensibility persists, even when the cells are of a different nature, as those of the fly (adipose and “intercalary” cells).
b. The pericardial fat-body or pericardial cells
We have already, on p. 405, called attention to these organs, but they also have an intimate relation to the fat-body.
Kowalevsky (1892) remarks that the disposition of these cells varies much in different insects and even in the same animal. Thus, in the Diptera and the ordinary flies there are found around the lower part of the dorsal vessel 13 pairs of large pericardial cells which lie next to a crowded bed of small cells forming a compact mass around the anterior part of the dorsal vessel. In caterpillars, notably silkworms, from the compact layer of pericardial cells which surround the heart, pass off trunks which are directed towards the lateral walls of the body, also forming close networks around the tracheæ and then passing down into the abdominal cavity of the body of the larva.
In the larvæ of certain Hymenoptera, the trunks which pass off from the pericardial region form a loose cord, a sort of fatty tissue covering the entire body cavity.
421This tissue, adds Kowalevsky, entirely differs from œnocytes, or from the so-called glandular body whose formation in Gryllotalpa has been described by Korotaiev, and in Bombyx mori by Tichomiroff. In a recent work wherein has been collected everything known regarding these last-named cells, Pékarsky proves that they are unique in nature and cannot be regarded either as fat-cells, or as pericardial cells, or even as formative leucocytes.
As to the structure of the pericardial cells, Kowalevsky adds that they are always attached to muscular fibres passing off from the heart, and that they lie, so to speak, upon them. In the locusts the muscular fibres supporting the pericardial cells appear distinctly like little staves or sticks. The attachment of the pericardial cells to the muscular fibres has been observed by Cuénot and reproduced by him in his work, but his description somewhat differs from that observed by Kowalevsky in the locust (Acrydium migratorium).
As to the nature of the acid excretions which are formed in the pericardial cells, in spite of his attempts to solve the problem, Kowalevsky has been unsuccessful. The only observations in this direction are those of Letellier on the pericardial glands of lamellibranch molluscs, which he found to contain hypouric acid, and it is probable, says Kowalevsky, that the acidity of the pericardial cells in insects is due to the presence of the same acid.
Leucocytes or phagocytes in connection with the pericardial cells.—It is thought by Schäffer that the leucocytes or phagocytes may be free or wandering fat-body cells. They play an important part in metamorphosis, while they absorb or feed upon the remains of the larval organs, and thus prove of use in the building up of the organs of the adult insects.
While the faculty of phagocytosis is wanting in the urinary tubes, Balbiani and more recently Cuénot have expressed the opinion that the pericardial cells of insects may have the power of absorbing hard bodies, “acting as a phagocytic gland.” This, however, is called in question by Kowalevsky, from studies made on different insects. On introducing powdered carmine into the body of an insect it has not been absorbed by the pericardial cells, as they have not been colored red. It is the leucocytes which absorb the grains of carmine, and which, after having dissolved them, transmit them to the pericardial cells. Hence, then, the pericardial cells have not the phagocytic power of which Cuénot speaks.
Returning to his own observations on hard bodies introduced into insects, or large globules introduced under the form of a milk emulsion, Kowalevsky has found that these bodies were absorbed in the first place by the free-swimming leucocytes, and in the second place by whole groups or nests of leucocytes situated in different parts 422of the body, principally on the threads of the adipose body. In the Orthoptera the absorption is immediately effected by means of the cells of the membrane which separates the pericardium from the cavity of the body underneath the heart. The regions where the hard bodies are absorbed in great number coincide with the regions of formation of the blood corpuscles. In his researches on the larvæ of Hyponomeuta and other Lepidoptera, Schäffer describes these regions as forming a sort of island. The nests where the blood globules are formed are the most active centres of phagocytosis.
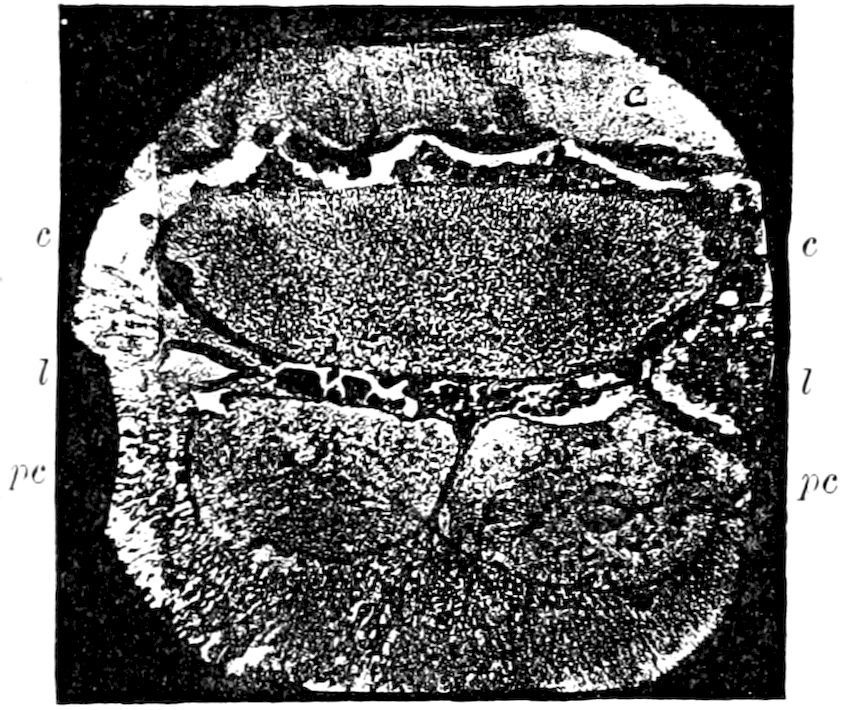
Fig. 384.-Section of the heart (c) and pericardial cells (pc, pc) from the posterior part of the heart of a fly: l, l, nests of leucocytes situated between the heart and pericardial cells.—From a microphotograph, after Kowalevsky.
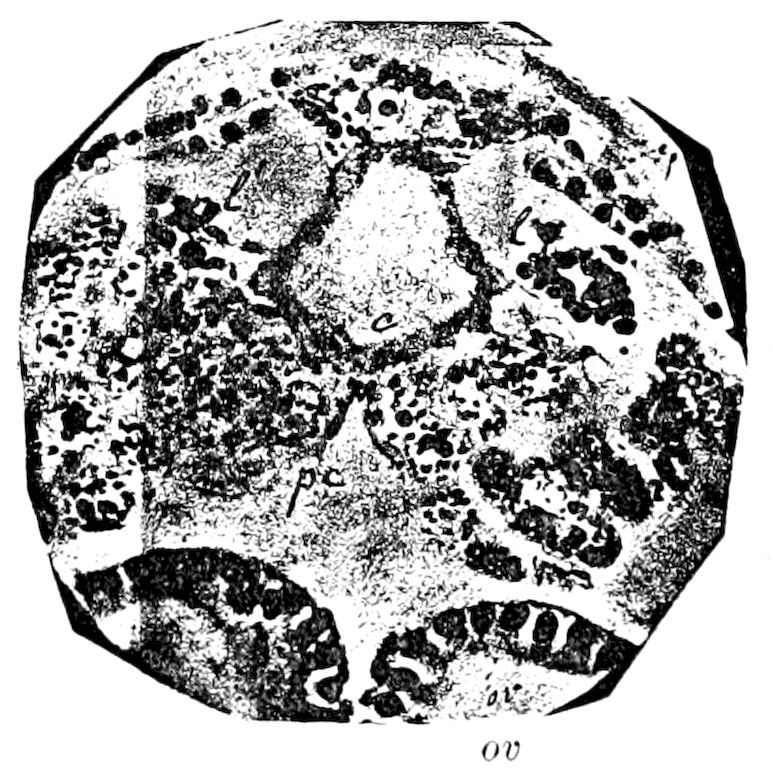
Fig. 385.—Cross-section of the heart of Truxalis nasata and of the structures around it: c, heart: ep, epithelium under the cuticula (hvpodermis); or, ovarian tubes; pc, pericardial cells, with one or two nuclei containing a deposit of carmine; l and l′, group of leucocytes, which have absorbed granules of India ink.—After Kowalevsky.
Balbiani, and also Cuénot, have supposed that the formation of the blood corpuscles takes place in the pericardial cells, but Kowalevsky insists that these cells cannot form the leucocytes, which “are probably formed in different parts of the body, notably in the special nests [Herde of Jäger] situated near the heart, but outside of the pericardial cells.”
In Fig. 384, where the nests of leucocytes (l) are shown, it is evident that they are formed where observed, and “could not have come from the pericardial cells, which have their own structure and their special function,” these cells being very large and characteristic.
In Kowalevsky’s preparations of Truxalis, the pericardial cells with deposits of carmine and the groups of leucocytes (Fig. 385, l and l′) stained with India ink, we have to deal with elements absolutely different. If the formation of leucocytes was caused by the pericardial cells, these last would be obliged to free themselves from their contents and to modify their essential nature.
c. The œnocytes
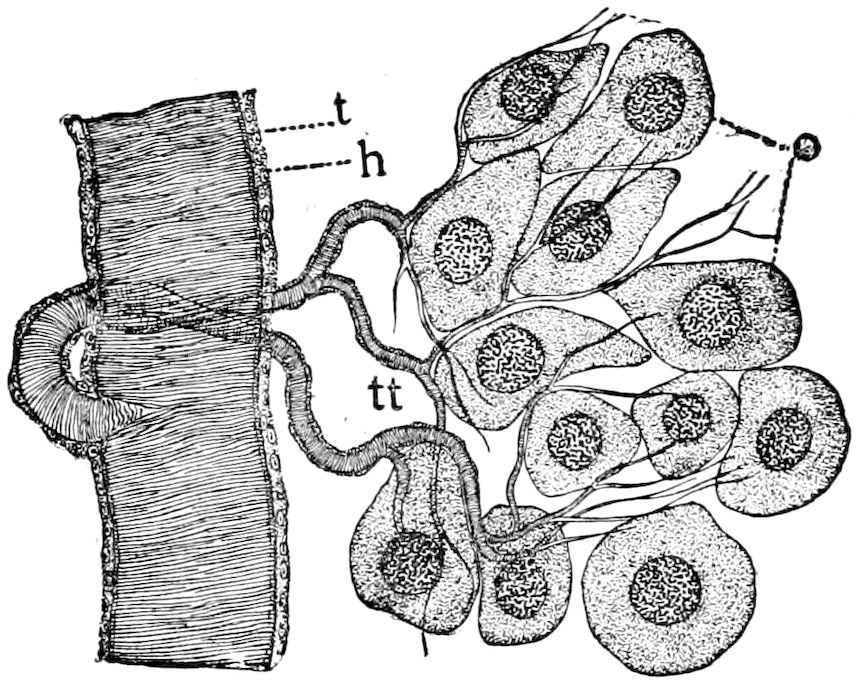
Fig. 386.—Cluster of œnocytes from a nearly mature Phryganeid larva: o, œnocytes; t, large tracheal branch; tt, smaller tracheal ramifications; h, tracheal hypodermis.
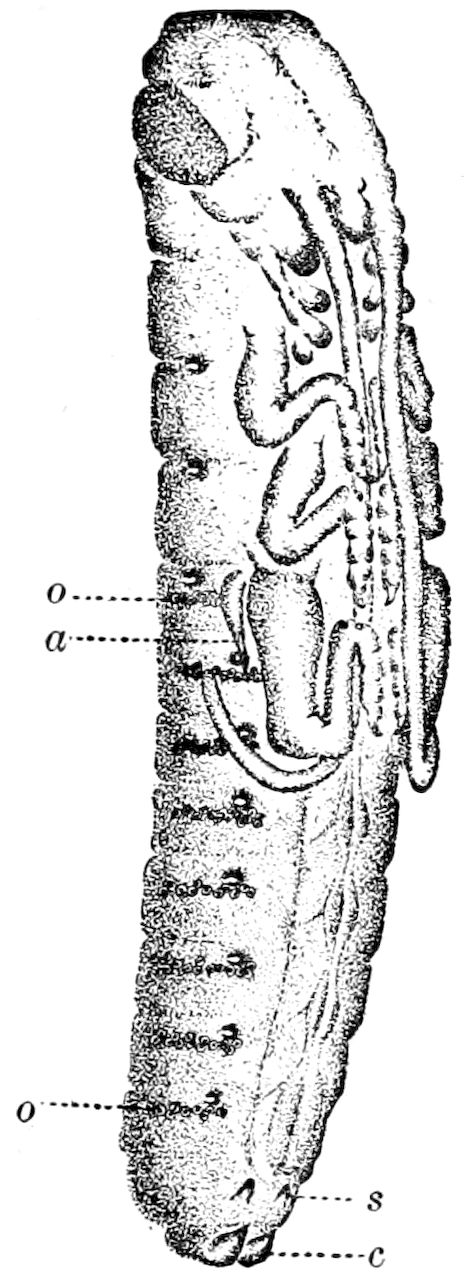
Fig. 387.—A nearly mature embryo of Xiphidium ensiferum: o, o, œnocyte clusters seen from the surface through the integument; a, pleuropodium of the right side (appendage of the first abdominal segment); s, styli; c, cercopods.—This and Fig. 386 after Wheeler.
These cells (Fig. 386), with the exception of the eggs, are the largest in the body, and occur in most if not all winged insects. They were called œnocytes (oinos, wine; kustis, cyst), by Wielowiejski in allusion to their wine-yellow color. These cells are arranged segmentally (Fig. 387) in clusters, held in place by tracheæ, and are situated mostly on each side of the abdomen, rarely being found in the adjoining parts of the thorax. They are more or less intimately associated with the blood and fat-body. Unlike the fat-body, however, they arise in embryonic life from the ectoderm, either by delamination or by immigration, just behind the tracheal involutions.
The separate cells of each cluster are usually separate, but in rare cases may fuse in pairs or form smaller clusters. In shape they are round or oval, often sending out pseudopodia-like processes, by which they are attached to the tracheal twigs or to each other. “The cytoplasm, which is very abundant, is full of yellowish granules and is sometimes radially situated towards its periphery. The large spherical or oval nucleus contains a densely wound and delicate chromatic filament.” (Wheeler.)
Graber first pointed out the identity of these clusters of cells with certain metameric cell-masses in insect embryos, observed by Tichomiroff in those of the silkworm, and by Korotneff in the embryo mole-cricket.
Although they resemble the blood corpuscles in some insects, they are always much larger, and do not seem to be amœboid, while they are never seen to 424undergo self-division, or to exhibit any appearance of giving rise to the blood-cells (Wheeler). They have not yet been detected in Thysanura (Synaptera) or in Myriopoda.
d. The phosphorescent organs
Phosphorescence is not infrequent in the Protozoa, cœlenterates, worms, and has been observed in the bivalve Pholas, in a few abyssal Crustacea, in myriopods (Geophilus), in an ascidian, Pyrosoma, and in certain deep-sea fishes.
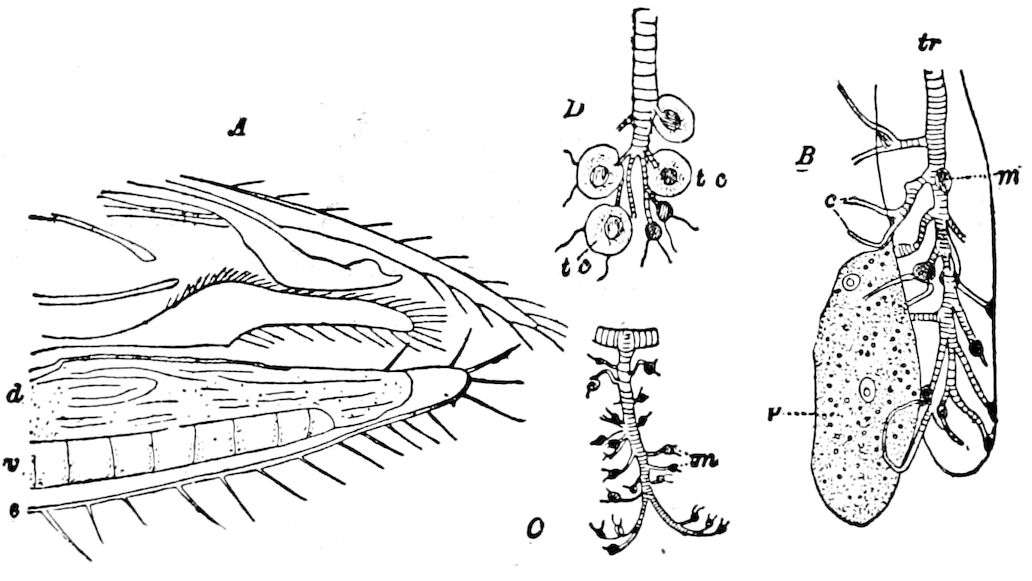
Fig. 388.—A, sagittal section through the hinder end of a male Luciola, the organs above the phosphorescent plate only drawn in outline: s, integument of the last segment, somewhat removed by the section-knife from the phosphorescent tissues; d, dorsal layer of the phosphorescent plate penetrated by irregular tracheal branches, and rendered opaque by numerous urate concretions imbedded in it; v, ventral phosphorescent layer of the plate, with perpendicular tracheal stems whose branches, where they pass into capillaries, bear lumps which stain brown with osmic acid; n, structureless substance (coagulum?) filling the end of the last ventral segment. B, isolated portion of the ventral layer of the phosphorescent plate; tr, tracheal stem surrounded by a cylindrical lobe: p, parenchym cell attached to the cylinder; c, capillary, without the spiral threads; m, coagulum stained brown. C, a tracheal stem of the ventral layer: at the fork of the brown-stained capillaries are lumps stained brown with osmic acid. D, a part of C, more highly magnified, showing the remains of the tracheal end-cells (tc) enveloping the brown lumps (m).—After Emery.
In insects luminosity is mostly confined to a few Coleoptera, and besides the well-known fireflies, an Indian Buprestid (Buprestis ocelata) is said to be phosphorescent; also a telephorid larva. Other luminous insects are the Poduran Anurophorus, Fulgora, certain Diptera (Culex, Chironomus[60] and Tyreophora), and an ant (Orya).
The seat of the light is the intensely luminous areas situated either in the head (Fulgora), in the abdomen (Lampyridæ), or in the thorax (in a few Elateridæ of the genus Pyrophorus). The luminous or photogenic organ is regarded by Wielowiejski and also by Emery as morphologically a specialized portion of the fat-body, being a plate consisting of polygonal cells, situated directly under the integument, and supplied with nerves and fine tracheal branches.
In Luciola as well as in other fireflies, including Pyrophorus, the 425phosphorescent organ or plate consists, as first stated by Kölliker, of two layers lying one over the other, a dorsal one (Fig. 388, d) which is opaque, chalky white, and non-photogenic, and a lower one (v), the active photogenic layer, which is transparent. Through the upper or opaque layer and on its dorsal surface extend large tracheæ and their horizontal branches, from which arise numerous very fine branches which pass down perpendicularly into the transparent or photogenic layer of the organ. Each tracheal stem, together with its short branches, is enveloped by a cylindrical mass of transparent tissue, so that only the short terminal branches or very fine tracheal capillaries project on the upper part of the cylinder. These finest tracheal capillaries are not in Luciola filled with air, but with a colorless fluid, as was also found by Wielowiejski and others in Lampyris.
These transparent cylinders, with the tracheæ within, forming longitudinal axes, resemble lobules. These lobules are so distributed that they appear on a surface section of this plate as numerous round areas in which circular periphery the tracheal capillaries are arranged with the axially disposed tracheal end-cells. These “tracheal end-cells” are only membranous enlargements at the base of the tracheal capillaries (Wielowiejski). The cylindrical lobules are separated from each other by a substance consisting of abundant large granular cells (parenchym cells) among which project the tracheal capillaries. The cylindrical lobules extend to the hypodermis and come in contact only by their lateral faces with the parenchym.
The structure of the upper opaque chalky white layer of the phosphorescent organ is, compared with that of the photogenic lower portion, very simple. In its loose, pappose, mass are no cellular elements, but when treated with different reagents it is seen to be filled with countless urate granules (guanine) swimming in the fluid it contains, the cell plasma appearing to be dissolved, the cells having lost their cohesion.
In comparing the phosphorescent plate or organ of Luciola with that of Lampyris, the general structure, including the clear cell elements of the cylindrical lobules, which envelop the perpendicular tracheal twigs and their branches, and also the granular parenchymatous cells are alike in both, though the arrangement and distribution of the elements in Luciola is more regular, in Lampyris the tracheal stems being irregularly scattered through the parenchym.
Wielowiejski found in the larval and female Lampyris a higher degree of differentiation than in the male, and Luciola has a more differentiated photogenic organ than Lampyris, as seen in the more regular structure of the lobules.
426As regards the light-apparatus of Pyrophorus, or the cucujo, Heinemann shows that, as in the Lampyridæ, it consists of distinct cells, and may be regarded as a glandular structure. It is rich in tracheæ and the other parts already described. In still later researches on a Brazilian Pyrophorus Wielowiejski shows that the phosphorescent plate consists of two layers, the upper usually being filled with crystalline urate concretions, and entirely like those of the Lampyridæ, consisting of distinct polygonal cells, among which are numerous tracheal stems, with tænidia, coursing in different directions, when freshly filled with air, and sending capillaries into the underlying photogenic layer. The latter shows in its structure a striking difference in the cellular arrangement from that of Lampyrids. In the upper or non-photogenic layer are tracheal capillaries which pass down into the underlying cellular plate and which are in the closest possible relations with the single cells—a point overlooked by Heinemann.
Physiology of the phosphorescence.—As is well known, the phosphorescence of animals is a scintillating or glowing light emitted by various forms, the greenish light or luminous appearance thus produced being photogenic, i.e. without sensible heat.
Langley rates the light of the firefly at an efficiency of 100 per cent, all its radiations lying within the limits of the visible spectrum. “Langley has shown that while only 2.4 per cent of luminous waves are contained in the radiation of a gas-flame, only 10 per cent in that of the electric arc, and only 35 per cent in that of the sun, the radiation of the firefly (Pyrophorus noctilucus) consists wholly of visible wave-frequencies.” (Barker’s Physics, p. 385.)
The spectrum of the light of the cucujo was found by Pasteur to be continuous. (C. R. French Acad. Sc. 1864, ii, p. 509.) A later examination by Aubert and Dubois showed that the spectrum of the light examined by the spectroscope is very beautiful, but destitute of dark bands. When, however, the intensity diminishes, the red and orange disappear, and the green and yellow only remain.
Heinemann studied the cucujo at Vera Cruz, Mexico. At night in a dark room it radiates a pale green light which shows a blue tone to the exclusion of any other light. The more gas or lamp light there is present, the more apparent becomes the yellowish green hue, which in clear daylight changes to an almost pure very light yellow with a very slight mixture of green. “In the morning and evening twilight, more constantly and clearly in the former, the cucujo light, at least to my eyes, is an intensely brilliant yellow with a slight mixture of red. In a dark room lighted with a sodium light the yellow 427tone entirely disappears; on the other hand, the blue strikingly increases.” As regards the spectrum he found that almost exactly half of the blue end is wanting and that the red part is also a little narrower than in the spectrum of the petroleum flame.
Professor C. A. Young states that the spectrum given by our common firefly (Photinus?) is perfectly continuous, without trace of lines either bright or dark. “It extends from a little above Fraunhofer’s line C, in the scarlet, to about F in the blue, gradually fading out at the extremities. It is noticeable that precisely this portion of the spectrum is composed of rays which, while they more powerfully than any others affect the organs of vision, produce hardly any thermal or actinic effect. In other words, very little of the energy expended in the flash of the fire is wasted. It is quite different with our artificial methods of illumination. In the case of an ordinary gaslight the best experiments show that not more than one or two per cent of the radiant energy consists of visible rays; the rest is either invisible heat or actinism; that is to say, over 98 per cent of the gas is wasted in producing rays that do not help in making objects visible.”
Panceri also remarks that while in the spectroscope the light of some Chætopteri, Beroë, and Pyrosoma exhibit one broad band like that given by monochromatic light, that of Lampyris and Luciola is polychromatic. (Amer. Nat., vii, 1873, p. 314.)
The filtered rays of Lampyris pass (like Röntgen and uranium rays) through aluminium (Muraoka).
The physiology of insect phosphorescence is thus briefly stated by Lang: “The cells of this luminous organ secrete, under the control of the nervous system, a substance which is burnt during the appearance of the light; this combustion takes place by means of the oxygen conveyed to the cells of the luminous body by the tracheæ, which branch profusely in it and break up into capillaries.”
Emery states that the males of Luciola display their light in two ways. When at night time they are active or flying, the light is given out at short and regular intervals, causing the well-known sparkling or scintillating light. If we catch a flying Luciola or pull apart one resting in the day time, or cut off its hind body, it gives out a tolerably strong light, though not nearly reaching the intensity of the light waves of the sparkling light. In this case the light is constant, yet we notice, especially in the wounded insect, that the phosphorescent plate in its whole extent is not luminous, but glows at different places as if phosphorescent clouds passed over it.
It is self-evident that a microscopic observation of the light of the glow-worm or firefly is not possible, but an animal while giving out 428its light, or a separated abdomen, may readily be placed under the microscope and observed under tolerably high powers. By making the experiment in a rather dark room Emery saw clear shining rings on a dark background. “All the rings are not equally lighted. Comparing this with the results of anatomical investigation, it is seen that the rings of light correspond with the previously described circular tracheal capillaries, i.e. the limits between the tracheal-cell cylinder and the parenchym-cells. The parenchym-cells are never stained of a deep brown; this proves that its plasma may be the seat of the light-producing oxidation. Hence this process of oxidation takes place in the upper surface of the parenchym-cells, but outside of their own substance. The parenchym-cells in reality secrete the luminous matter; this is taken up by the tracheal end-cells and burnt or oxidized by means of the oxygen present in the tracheal capillaries. Such a combustion can only take place when the chitinous membrane of the tracheæ is extraordinarily fine and easily penetrable, as is the case in the capillaries of the photogenic plate; therefore the plasma of the tracheal cells only oxidizes at the forking of the terminal tracheal twigs and in the capillaries.” (Emery.)
The color of the light of Luciola is identical in the two sexes, and the intensity is much the same, though that of the female is more restricted. The rhythm of the flashes of light given out by the male is more rapid, and the flashes briefer, while those of the female are longer, more tremulous, and appear at longer intervals.
Emery then asks: What is the use of this luminosity? Is it only to allure the females of Luciola, which are so much rarer than the males? Contrary to the general view that it is an alluring act, he thinks that phosphorescence is a means of defence, or a warning or danger-signal against insectivorous nocturnal animals. If we dissect or crush a Luciola, it gives out a disagreeable cabbage-like smell, and perhaps this is sufficient to render it inedible to bats or other nocturnal animals. An acrid taste they certainly do not possess.
It has long been known that the eggs of fireflies, both Lampyridæ and Pyrophorus, are luminous. Both Newport and more recently Wielowiejski attributes the luminosity not to the contents of the egg, but to the portions of the fat-body cells or fluid covering on the outside of the eggs, due to ruptures of the parts within the body of the female during oviposition. The larvæ at different ages are also luminous.
The position of the luminous organs changes with age. In the larvæ of Pyrophorus before moulting, according to Dubois, the luminous 429organs are situated only on the ventral side of the head and prothoracic segment. In larvæ of the second stage there are added three shining spots on each of the first eight abdominal segments, and a single luminous spot on the last segment. These spots are arranged in a linear series and thus form three luminous cords. In the adult beetles there is a luminous spot in the middle of the first abdominal sternite, but the greatest amount of light is produced by the two vesicles on the hinder part of the prothorax, the position of which varies according to the species.
LITERATURE ON PHOSPHORESCENCE
Peters, W. Ueber das Leuchten der Lampyris italica. (Müller’s Archiv f. Anatomie, 1841, pp. 229–233.)
Kölliker, A. Die Leuchtorgane von Lampyris, eine vorläufige Mittheilung. (Verhandl. d. phys. medizin. Gesellsch. Würzburg, 1857, viii, pp. 217–224.)
Schultze, Max. Ueber den Bau der Leuchtorgane der Männchen von Lampyris splendidula. (Sitzber. d. niederrhein. Gesellsch. f. Natur. u. Heilkunde zu Bonn, 1864, Sep. p. 7.)
—— Zur Kenntniss der Leuchtorgane von Lampyris splendidula. (Archiv f. mikroskop. Anat., 1865, i, pp. 124–137, 2 Taf.)
Wielowiejski, H. Ritter von. Studien über die Lampyriden. (Zeits. f. wissens. Zool., 1882, xxxvii, pp. 354–428, 2 Taf.)
Emery, Carlo. Untersuchungen über Luciola italica L. (Zeits. f. wissens. Zool., 1884, xl, pp. 338–355, 1 Taf.)
—— La luce della Luciola italica osservata col microscopio. (Bull. Soc. Ent. Ital. Anno xvii, 1885, pp. 351–355, 1 Taf.)
Wheeler, William Morton. Concerning the blood tissue of the Insecta, III; Psyche, vi, 1892, p. 255. (Structure of the light organ of Photuris pennsylvanica.)
Heinemann, C. Zur Anatomie und Physiologie der Leuchtorgane Mexikanischer Cucujos, Pyrophorus. (Archiv f. mikroskop. Anat., 1886, xxvii, pp. 296–383.)
Dubois, R. Contribution à l’étude de la production de la lumière par les êtres vivants. Les Elaterides lumineux. (Bull. Soc. Zool. France, 1886, Année ii, pp. 1–275, 9 Pls.)
Wielowiejski, H. Ritter von. Beiträge zur Kenntniss der Leuchtorgane der Insekten. (Zool. Anzeiger, 1889, Jahrg. xii, pp. 594–600.)
Hudson, G. V. The habits and life-history of the New Zealand glowworms. (Trans. N. Zealand Inst., xviii, 1890, pp. 43–49, 1 P1.)
Dubois, R. Sur le mechanisme de la production de la lumière chez l’Orya barbarica d’Algérie. (Comptes rend. Acad. Sc. France, 1892, cxvii, pp. 184–186. Also in Ann. and Mag. Nat. Hist., 1893 (6), xii, pp. 415–416.)
Schmidt, P. Ueber das Leuchten der Zuckmücken (Chironomidæ). (Zool. Jahrb. Morph., Abth. viii, 1894, pp. 191–216, 2 Taf. Also Ann. and Mag. Nat. Hist., xv, pp. 133–141, 1895. The light is due to bacteria. Nature, 1897.)
Chun, C. Leuchtorgane und Facettenaugen. (Biol. Centralbl., 1896, pp. 315–320.)
Muraoka, H. (On the filtered rays of Lampyris, Wiedemann’s Annalen, Dec., 1896.)
See also the writings of Audouin, Carus, D. Turner, Thompson.
THE RESPIRATORY SYSTEM
While land vertebrates breathe by inhaling the air through the mouth into the lungs, insects respire by internal air-tubes (tracheæ), which ramify throughout every part of the body and its appendages. The air enters these tubes through a few openings, called spiracles or stigmata, arranged segmentally in the sides of the body. These tracheæ are everywhere bathed by the blood, and thus the latter is constantly aërated or kept fresh; the blood not, as in vertebrates or as in molluscs, seeking the lungs or gills, or any specialized respiratory portion of the body where the oxygen combines with the hæmoglobin, but the respiratory tubes, so to speak, themselves seek out the blood and the blood-tissue in every part of the insect body, penetrating to the tips of the antennæ and of the legs, entering the most delicate tissues, even perhaps passing through the walls of epithelial cells. As Lang remarks, the want of an arterial vascular system is compensated for as well as conditioned by the extremely profuse branching of the tracheæ.
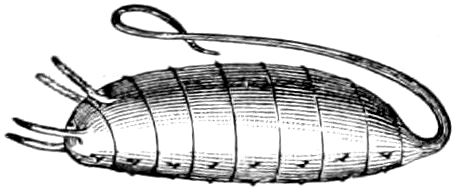
Fig. 389.—Rat-tailed larva of Eristalis.
The aquatic larvæ of certain dragon-flies (Agrionidæ), may-flies, case-worms, etc., respire by means of tracheal gills or branchiæ, which are either filamental or leaf-like appendages containing tracheæ. Somewhat similar structures appended to the thorax of pupal aquatic Diptera, as in the mosquito and its allies, enable them to breathe while stationed a little beneath the surface of the water. Other larvæ, as the rat-tail larva of Eristalis, etc., lying at the bottom of shallow pools or in ditches, etc., can breathe by raising slightly above the surface a long appendage with two spiracles at the end, through which the air enters the tracheal system. (See p. 461.)
Although Aristotle, as well as the natural philosophers of the Middle Ages, supposed that insects did not breathe, one can easily see that they do by holding a grasshopper or dragon-fly in one’s hand and observing the rhythmical rise and fall of the upper and lower walls of the abdomen, during which the air enters and passes out of the air-openings or spiracles on each side of the body.
It is plain that insects consume very little air, since caterpillars may be confined in very small, almost air-tight tin boxes, and continue to eat and undergo their transformations without suffering from the confinement. According to H. Müller an insect placed in 431a small, confined space absorbs all the oxygen. Insects can survive for many hours when placed in an exhausted receiver, or in certain irrespirable gases. “Cockroaches in carbonic acid speedily become insensible, but after twelve hours’ exposure to the pure gas they survive and appear none the worse.” (Miall and Denny, p. 165.) Insects of the swiftest flight breathe most rapidly, their great muscular activity requiring the absorption of an abundance of oxygen.
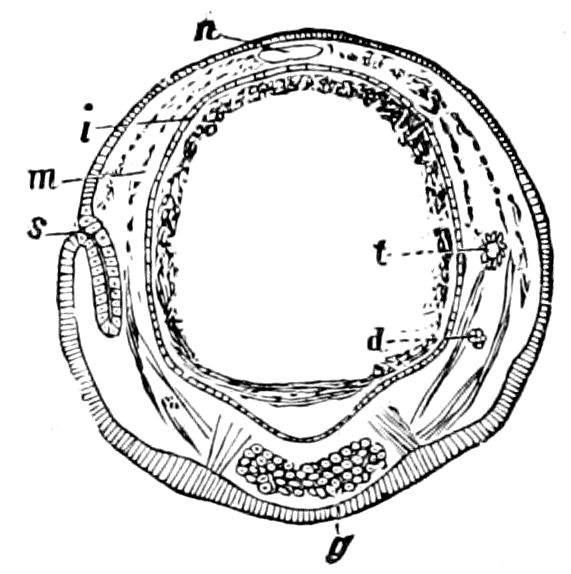
Fig. 390.—Section of Sphinx embryo, showing at s the ectoderm invaginated, and forming the germ of a stigma and trachea (t).—After Kowalevsky.
Warmth, plenty of food, besides muscular activity, increases the demand for oxygen and the quantity of carbonic acid exhaled.
a. The tracheæ
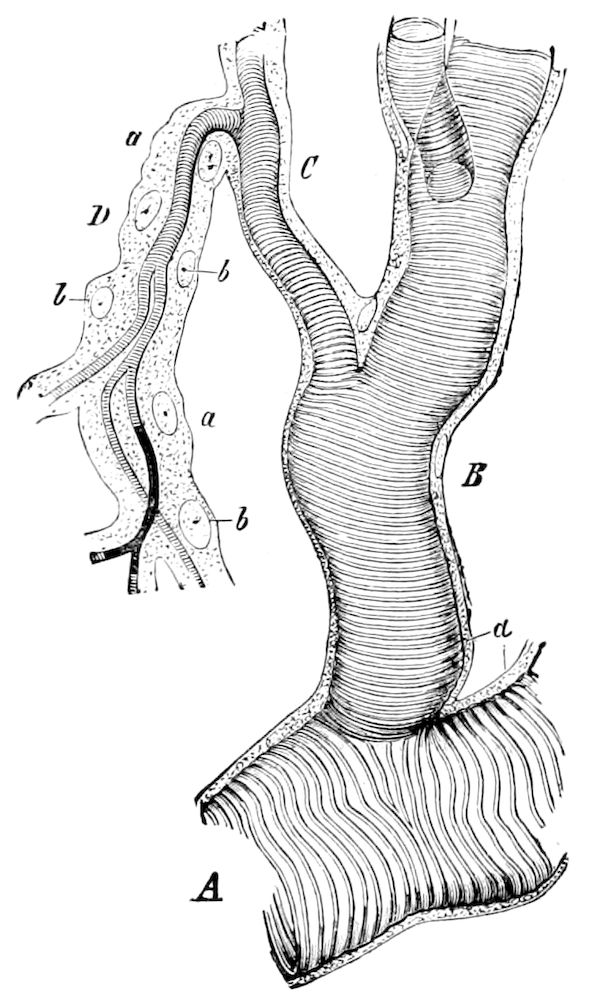
Fig. 391.—Portion of a trachea of a caterpillar, with its branches B, C, D: a, peritracheal membrane; b, nucleus.—After Leydig, from Gegenbaur.
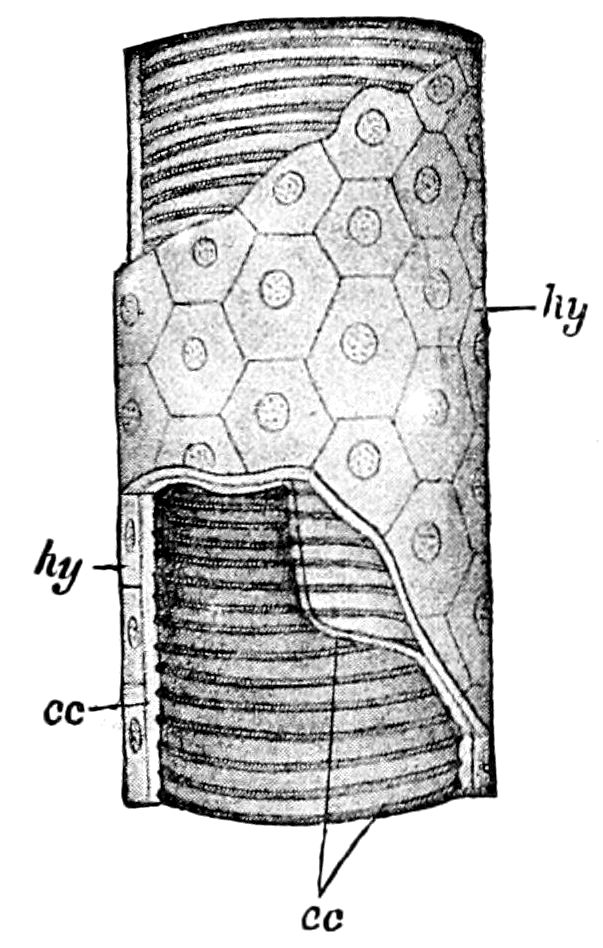
Fig. 392.—Structure of a trachea, diagrammatic: portions of the peritracheal membrane (hy) and chitinous intima (cc) removed to show the structure; in the chitinous intima or endotrachea (cc) can be seen the spiral thickenings or tænidia.—After Lang.
It will much simplify our conception of the nature of the air-tubes when we learn that they originate in the embryo as tubular ingrowths of the integument (ectoderm), these branching and finally reaching every part of the interior of the body. They are elastic tubes, and being filled with air are silvery in color, though at their origin near the spiracles they are reddish or violet bluish; or, in 432the larva of Æschna, reddish brown, this tint being due to a finely granular pigment situated in the peritoneal membrane.
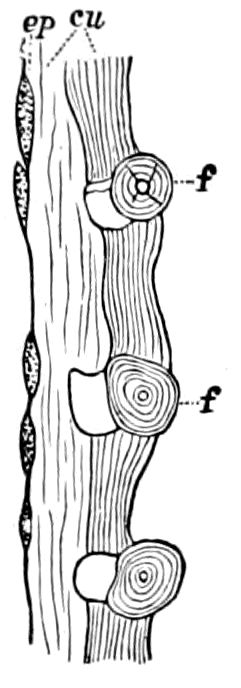
Fig. 393.—Longitudinal section of the trachea of Hydrophilus piceus: ep, epithelium; cu, cuticula; f, spiral threads.—After Minot.
In their essential structure the tracheæ consist of the chitinous intima, which is a continuation of the cuticle of the integument, and of a cellular membrane or outer layer of cells (a continuation of the hypodermis) called the peritoneal membrane, or ectotrachea (Figs. 392, 393).
Leydig discovered that the spiral filaments are not distinct and separate, but intimately connected with the inner membrane (intima), and he detected the outer or peritoneal membrane, which Chun afterwards found to be epithelial in its nature, Minot stating that it is a true pavement epithelium.
Figure 393 represents a longitudinal section of a large trachea of Hydrophilus, showing the peritoneal membrane (ectotrachea, ep) and the intima or endotrachea, divided into the cuticula (cu), with the darker colored inner layer, in which are embedded the dark-colored tænidia (f).

Fig. 394.—Testis of Anabrus, showing the ramifications of the tracheæ.—After Minot.
Distribution of the tracheæ.—The distribution of the air-tubes, as Lubbock and also Minot state, depends first upon the shape of the 433organs, and upon the size of those whose size is variable. Around the large, hollow organs (digestive canal, sexual organs) the tracheæ ramify in all directions, forking so that the branches diverge at a wide angle. In the organs which have muscular walls, like the oviduct, the tracheæ run straight when the walls are distended, but have a sinuous course when the walls are contracted. (Minot.)
“Around the organs of more elongated form the branches of the tracheæ run more longitudinally, as is shown by the air-tubes of the muscles, which also present some peculiarities worthy of especial notice.
“A short, thick trunk arrives at the muscular bundle, and dividing very rapidly, breaks up into a large number of delicate tubes, which penetrate between the muscular fibres, then terminating in tubes of exceeding fineness, which at first sight seem to form a network that might well be called a rete mirabile. A closer examination, however, reveals that it is not a real network, but rather an interlacing confusing to the eye. The longitudinal direction of the tracheæ of the muscles presents a striking contrast to the system of divarication represented in Figs. 13 and 14. The course of the tracheæ of the Malpighian tubes is also very curious. There is one large trachea which winds around the tube in a long spiral, giving off numerous small branches which run to the surface of the tube, upon which they form delicate ramifications. Each tube has but a single main trachea, and I think the trachea continues the whole length of the tube, but of this last point I am not quite sure.” (Minot.)
While in the nymphs of Orthoptera the tracheæ very closely resemble those of the adult, in larvæ of insects with a complete metamorphosis the tracheæ differ very much in distribution from those of the adult. The larval tracheæ are also more generalized and more like those of the original type than the tracheæ of perfect insects. (Lubbock.)
In general there are two main tracheæ, one passing along each side of the body, near the digestive canal, connected with its mate by a few transverse anastomosing branches, and sending off a branch to each spiracle, this arrangement being most simple and apparent in the maggots of Diptera. From these two main branches smaller twigs branch off into every part of the body with its appendages, passing among the different organs, often serving as cables to hold them loosely in place; they also penetrate into the component parts of the organ themselves, passing into the fat-bodies, and among the fibres of muscles, where they become finely attenuated and refined like the capillaries of the vascular system of vertebrates. (Figs. 395, 396.)
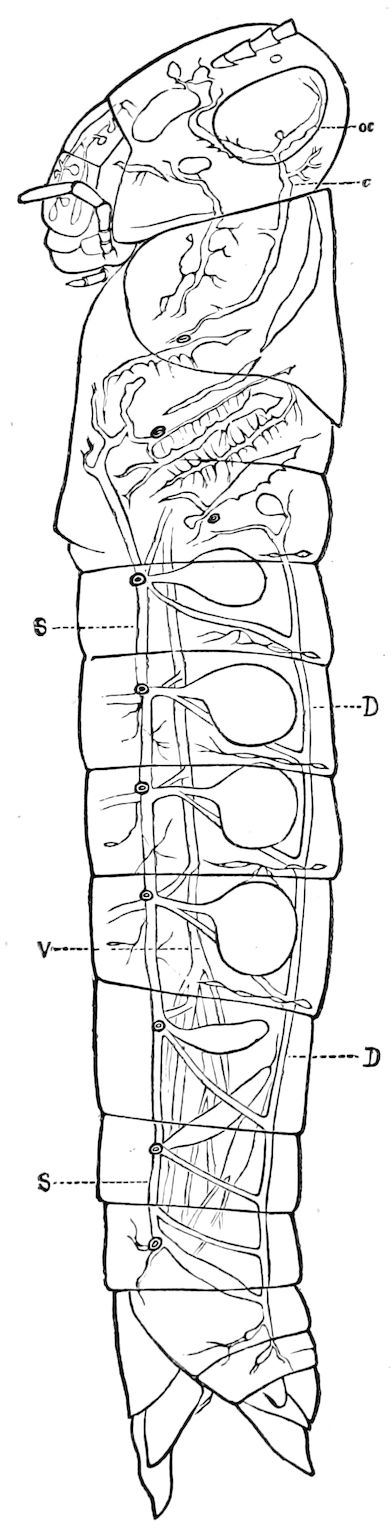
Fig. 395.—Melanoplus femur-rubrum, showing distribution of air-tubes (tracheæ) and air-sacs; V, main ventral trachea (only one of the two shown); S, left stigmatal trachea, connecting by vertical branches with D, the left main dorsal trachea; c, left cephalic trachea; oc, ocular dilated trachea. From the first, second, third, and fourth spiracles arise the first four abdominal air-sacs, which are succeeded by the plexus of three pairs of dilated tracheæ, I, II, III, in Fig. 396. Numerous air-sacs and tracheæ are represented in the head and thorax. The two thoracic spiracles are represented, but not lettered.
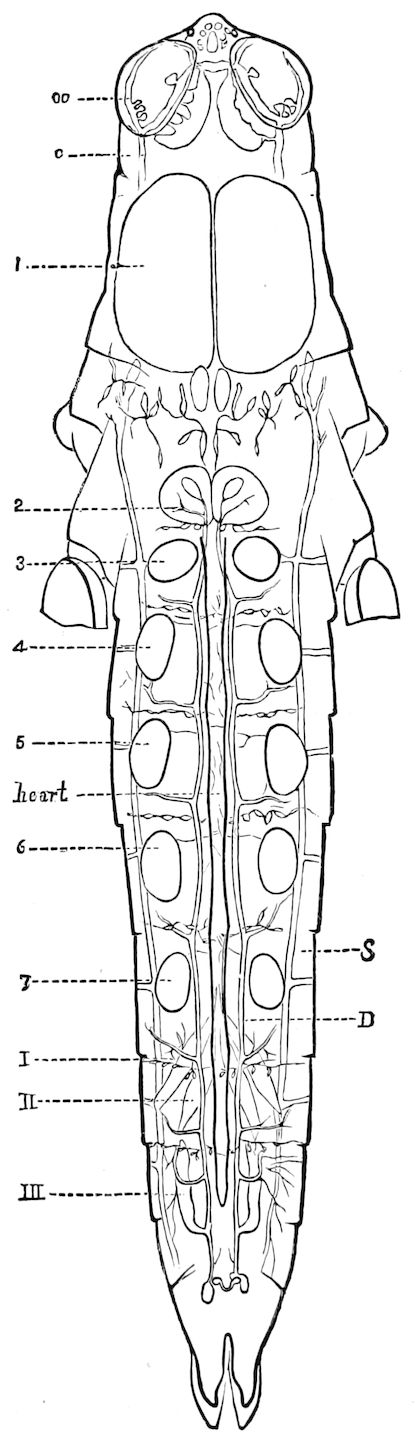
Fig. 396.—D, left dorsal trachea; S, left stigmatal trachea; I, II, III, first, second, and third pairs of abdominal dilated tracheæ, forming a plexus behind the ovaries; 1, pair of enormous thoracic air-sacs; 2, pair of smaller air-sacs; 3–7, abdominal air-sacs; oc, ocular dilated trachea and air-sacs; c, cephalic trachea. The relations of the heart to the dorsal tracheæ are indicated.—Drawn by Emerton from dissections by the author.
In the youngest larva of Corethra plumicornis Weismann ascertained the thickness of the longitudinal stem to be 0.0017 mm. That of the finest tracheal endings in the silk-glands of the silkworm was found by Von Wistinghausen to be 0.0016 mm. (Zeits. f. Wiss. Zool. xlix, 1890, p. 575.) Weismann states that in the larvæ of Corethra and Chironomus the tracheal system is only incompletely developed; the tracheæ are not united with each other, and in the youngest larvæ they do not contain air.
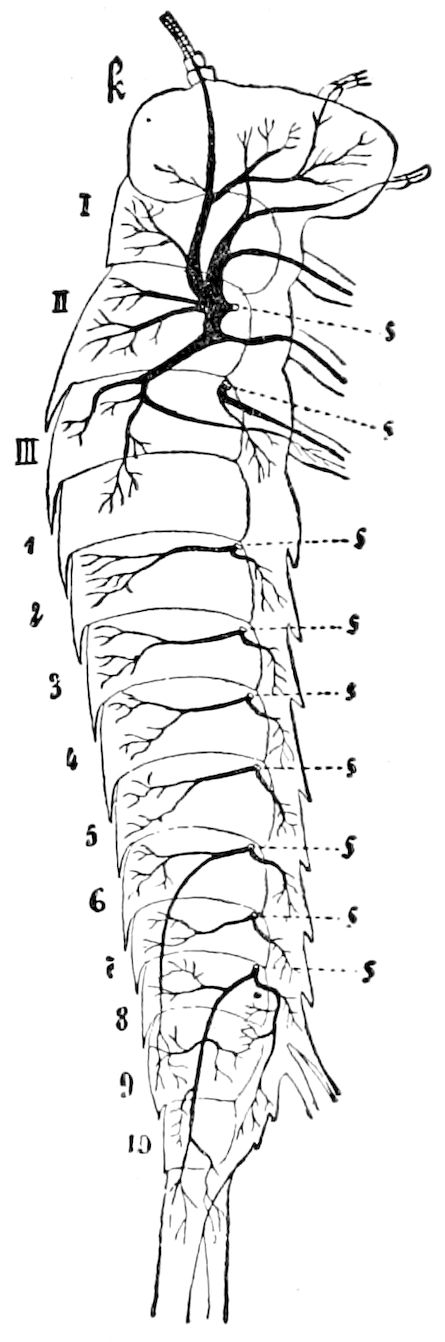
Fig. 397.—Tracheal system of the right side of Machilis maritima: k, head; I, II, III, thoracic segments; 1–10, abdominal segments; s, stigma.—After Oudemans, from Lang.
Each of the two main tracheæ, as Kolbe states, sends off into each segment of the body three branches.
1. An upper or dorsal branch, which supplies the muscles of the dorsal region.
2. A middle (visceral) branch, whose twigs pass to the digestive canal and back to the organs of reproduction.
3. A lower (ventral) branch, whose twigs are distributed to the ganglia and to the muscles of the ventral region.
In certain Thysanura, as a species of Machilis (Fig. 397), we probably have the primitive condition of the tracheal system, the longitudinal and transverse anastomoses being absent, but in other Thysanura (Japyx, Nicoletia, Lepisma, and a few species of Machilis) they are present.
As Kolbe remarks, whether the fine ends of the tracheæ are closed or open, whether after the analogy of the blood capillaries of vertebrates they anastomose with each other, whether the ends of the air-tubes pass between the cells or penetrate into them, these questions are not fully settled. According to Leydig’s[61] latest views the tracheæ penetrate into the cells and unite with the hyaloplasma. Hence the process of respiration in the last instance takes place in the hyaloplasma. This assumption accords with the fact that in the tracheate Arthropods the terminations of the tracheæ carry the atmospheric air into the space bounded by the cellular network, also to the hyaloplasma filling the spaces. Leydig[62] also thinks that the finest tracheal endings penetrate into the muscular tissue and unite with the primitive muscular fibres.
Kupffer is likewise of the opinion that the fine tracheæ penetrate into the cells, and Lidth de Jeude asserts that they enter the epithelial cells, “each cell containing several branches.” Kölliker, Emery, etc., maintain, however, 436that the tracheal endings lie between the cells. Wielowiejski,[63] in describing the line tracheæ of the phosphorescent organs, thinks that the tracheal endings (tracheal capillaries) rarely end blindly, but anastomose with one another, forming an irregular network. The latest observer, Gilson (1893), asserts that tracheal twigs penetrate deeply into the epithelial cells of the silk glands of larval Trichoptera as well as of caterpillars, passing through their protoplasm.
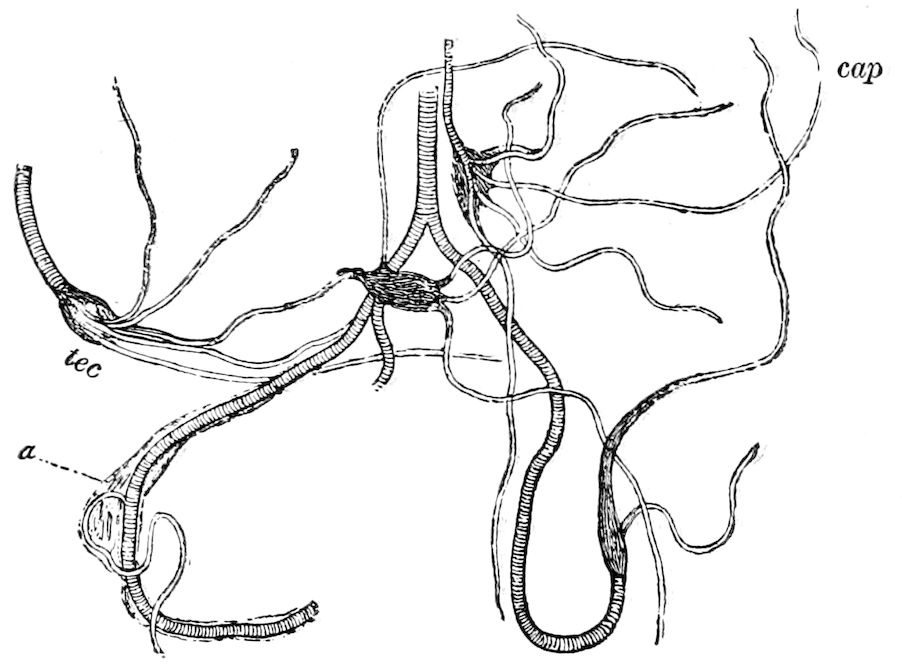
Fig. 398.—Tracheal network of the male glands of Lampyris splendidula: tec, tracheal end-cells; cap, tracheal capillaries; at a, an expanded matrix.—After Wielowiejski.

Fig. 399.—Tracheal capillary end-network (tr. c. n.) of silk glands of Ocneria dispar: p, peritoneal (peritracheal) membrane.—After Wistinghausen.
A late investigator, C. von Wistinghausen, finds in the tracheæ of the spinning-glands of caterpillars a completely formed network between the terminal branches of two or several tracheal groups. The tracheal tubes of this series of 437terminal branches pass into this network, which he calls the tracheal capillary end-network (Figs. 398, 400). This last varies in thickness and spreads out under the membrana propria of the glandular mass over the entire surface of the large gland-cells and on a level with the tracheal capillaries. The tracheal endings do not penetrate into the cells, but are separated from the plasma of the cells by a thin membrane. The tracheal capillary end-network appears as a system of fine tubes like the tracheal capillaries, consisting of a peritoneal layer and a chitinous intima (Fig. 400). The walls of these tubes are homogeneous, not porous, though readily permeable by the parenchymatous fluid. The interchange of gases consequently may go on easier and more vigorously in a system of richly anastomosing tubules of the net-like mass of tracheal capillaries, than in tubes ending blindly.
While the diameter of the tracheal capillaries is 0.0016 mm. or 1 µ, that of the tubules composing the tracheal capillary end-network is scarcely measurable, but is less than 1 µ.
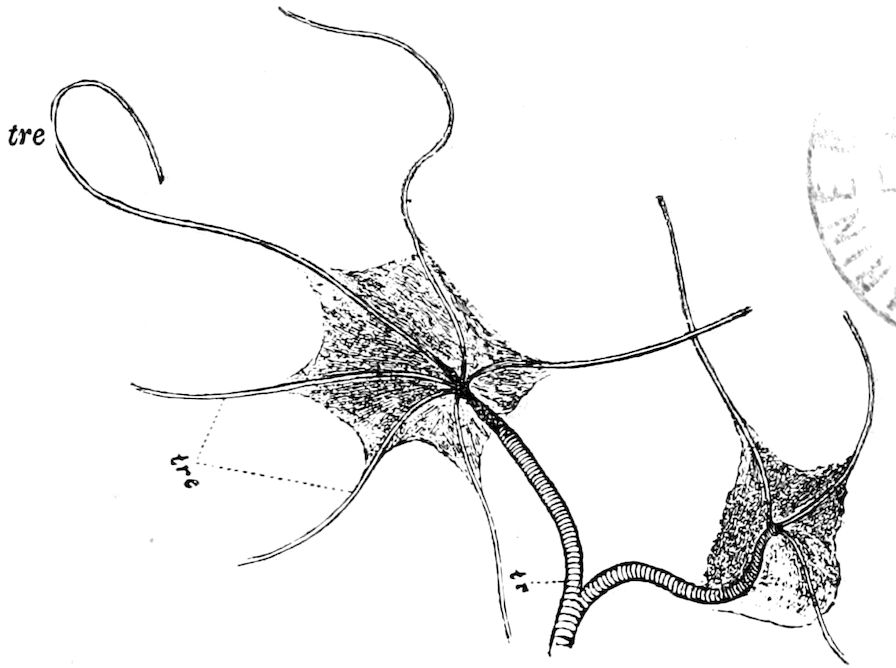
Fig. 400.—Tracheal end-cells of Lampyris splendidula: tr, trachea with tænidia; tre, tracheal capillaries.—After Wielowiejski.
These tracheal capillaries also occur on the seminal and other sexual tubes, on the intestine, on the urinary tubes, on the fat-bodies, but are most easily detected on the silk-glands.
The latest researches are those of E. Holmgren, who has studied the branching of the tracheæ in the spinning-glands of caterpillars. He prefers to call the end-cells “transition cells,” as they lead from the tracheal tubes proper to the capillary network. This latter is formed by slender nucleated cells, often with an intracellular lumen, and, according to the author, probably constituting a respiratory epithelium. He finds that both large and small tracheæ may penetrate the gland-cells. (Anat. Anzeiger, xi, 1895, pp. 340–6, 3 figs.; Jour. Roy. Micr. Soc., 1896, p. 182.)
b. The spiracles or stigmata
The spiracles are segmentally arranged openings in the sides of the thorax and abdomen, through which the air passes into the air-tubes. 438In its essential structure a spiracle, or stigma, is a slit-like opening surrounded by a chitinous ring, the lips or edges of the opening being membranous and closed by a movable valve of the spiracle attached by its lower edge, which is closed by an occlusor muscle (Fig. 401). The aperture when open forms a narrow oval slit; and in most insects the slit is within guarded by a row of projecting spines or setæ, which form a lattice work or grate to keep out dust, dirt, fluids, etc.
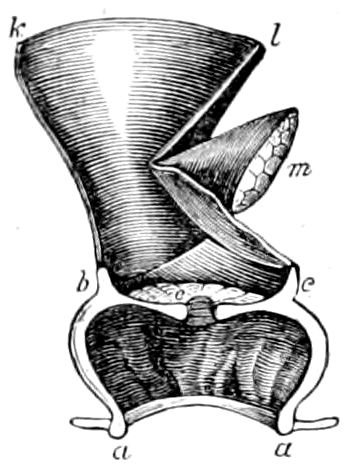
Fig. 401.—Horizontal section of left third stigma and trachea of Melolontha vulgaris, showing the chamber or drum leading into the trachea: a, a, external frame or valve protecting the outer opening of the stigma; b, c, c, inner frame closing the entrance into the trachea (l, k); m, occlusor muscle closing the inner orifice.—After Straus-Dürckheim.
Krancher[64] has described five leading types of stigmata, not, however, taking into account those of the Synaptera.
I. Stigmata without lips (Primitive or generalized stigmata).
a. The simplest stigma is an aperture which is kept open by a chitinous ring (Acanthia). The opening may be round or elliptical. There are no lips nor any movement of the edges to be observed. Such air-holes occur in the abdomen of bugs (Hemiptera) and beetles (Coleoptera); within the opening of the stigmata in the same insects is a funnel-like contraction. Also in the Diptera the abdominal stigmata are of the same type.[65] The stigmata of the Pulicidae (Siphonaptera) are more complicated, as the edges of the openings are provided with setæ (Fig. 402).
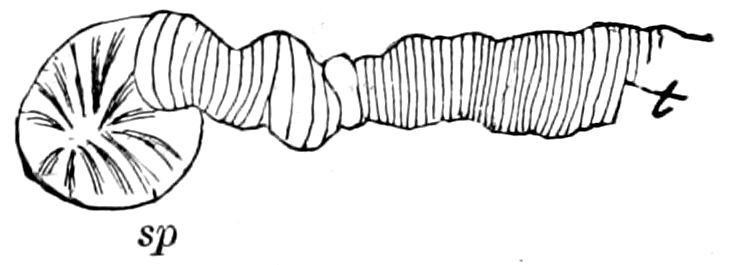
Fig. 402.—First abdominal spiracle with a part of the trachea of the cat-flea: sp, spiracle; t, trachea.

Fig. 403.—Stigma of Melolontha larva, seen from without: b, bulla; s, sieve-like plate; o, curved slit-like opening.—After Boas.
b. The stigma consists of a series of minute single stigmata, which are usually surmounted by a common chitinous ring, and whose tubular continuations unite within in a common trachea, so that the single tubes pass off from the stigma like the fingers on the hand. This form is found in the larvæ and puparia of Diptera.
II. Stigmata with lips (Secondary more specialized stigmata).
c. The lips are represented by a single chitinous ring, with sparse spines. One side of the stigma is a little higher, and partly overlaps the other posteriorly; this form is peculiar to the Orthoptera and Libellulidae.
d. The lips are roof-like, bent inwards and densely hairy, forming a peculiar kind of felting. The setæ of the lips are in most beetles and many Lepidoptera 439separate, and more or less branched. In caterpillars, the setæ are so finely branched as to form a loose felt, or sieve-like arrangement.
e. The stigmata are round, with a very broad border and a concentric middle portion, the structure being complicated. The concentric middle portion is pouch-like and bears the occlusor muscle. This form occurs in the larvæ of lamellicorn beetles, and can be seen with the naked eye, or with a lens, in Oryctes, Cetonia, and Melolontha (Fig. 403).
f. Over the outer opening of the spiracle is an incurved chitinous projection, on one side of which the trachea takes its origin. It is thus in the Hymenoptera.
The remarkable grate-like stigma of the lamellicorn larvæ has the appearance as if the outer closing plate or valve were impenetrable. The earlier observers considered these stigmata to be open, but Meinert regards them as closed; Schiödte, however, has observed by pressing a preserved specimen of a Melolontha larva the alcohol within passing out in drops, through the grate-like plate, and hence he considers this a proof that the stigma is permeable (Kolbe).
More recently (1893) Boas has examined the same structure in the same species of larva as examined by Schiödte, and he finds it to be open only during the process of moulting. He finds that on each side of the larva there are nine short and wide stigmatic branches, each of which is shut off from the exterior by a brown plate; this consists of a reniform sieve-plate, and of a curved bulla which fits into the cavity of the plate. The stigmatic branch, however, is provided with a large external opening, which is homologous with the stigma, but which is usually closed by the plate and bulla, and is only open during the moulting; at first it is circular, but later becomes a cleft. A transverse section shows that the bulla is a simple tegumentary fold, the outer chitinous layer of which has become especially firm. The plate forms a horizontal half-roof, which springs from one side of the tracheal orifice, and is supported by obliquely set bases, which spring from the adjoining part of the inner side of the tracheæ. The plate and bars are purely cuticular structures. (Zool. Anz., 1893; also Journ. Roy. Micr. Soc., p. 54.)
The tracheal system of libellulid nymphs is not closed; on the other hand, in the fully-grown nymphs the anterior stigmata occurring on the dorsal side are large, and the tracheæ arising from them are thick. These stigmata are permeable by the air. In half-grown and still younger stages of Æschna the two anterior thoracic stigmata are undeveloped. In order to breathe, the fully-grown nymph either rises up on the upper side and elevates the end of the body to the surface in order to take the air into the rectum, or it rests with the back of the thorax at the surface in order to breathe through the large stigmata. The young nymphs take in air only through the rectum. The young nymphs of Libellula and its allies, on the other hand, possess large thoracic stigmata, but they prefer to breathe through the rectum. The fully-grown nymphs of Agrion breathe through the thoracic stigmata. (Dewitz, in Kolbe.)
The position and number of pairs of stigmata.—The spiracles are usually situated in the soft membrane between the tergites and pleurites, but their exact position varies in different groups. In the Coleoptera they occupy on the thorax a more ventral position, and on the abdomen are placed near the edge of the dorsal side, under the elytra. In the dragon-flies, the first pair is situated much more dorsally than the second and third pairs; the following seven pairs are almost wholly ventral and lie concealed in the membranous fold 440near the external plate. In the Hemiptera, also, the abdominal stigmata, though entirely free and visible, are situated ventrally.
Primarily, in the embryo a pair of stigmata appear on each segment of the thorax and abdomen, except the 10th and 11th, and even possibly in the head, for a pair of stigmata are said to occur in the head of Podurids (Smynthurus) (Lubbock), though this statement needs confirmation. Scolopendrella, however, is known to possess a pair of cephalic spiracles.
From the foregoing statement it will be seen that while in existing winged insects no more than 10 (in Japyx 11) pairs of stigmata are to be found in any one species, yet that 12 segments of the body, in different groups taken collectively, bear them. The primitive number of pairs of spiracles, therefore, in winged insects, was 12, i.e. a pair in each thoracic segment, and a pair in each of the first nine abdominal segments. Insects were originally all holopneustic, and gradually as the type became differentiated into the different orders they became peripneustic or amphipneustic, and, in certain aquatic forms, apneustic. (See pp. 459, 461.)
In the still more primitive, probably wingless, ancestors of insects there was a larger number of stigmata. Hatschek, in 1877, discovered a pair of tracheal invaginations in each of the three posterior head-segments of the embryo of a moth, with stigmatal openings in the 1st and 2d maxillary segments.
Thus early in embryonic life every segment of the body, except those bearing the eyes and the last abdominal, bore a pair of stigmata, so that the primitive insect had at least 15, and perhaps more, pairs of stigmata.
The position of the stigmata is subject to much variation, the result of adaptation to this or that mode of life. Examples are those insects which live in dusty situations or usually more or less concealed in the earth, as in most beetles, and in the Hymenoptera. In such beetles, the stigmata are situated in the thin membrane between the segments; in the Hymenoptera, on the upper edge of the segments. In the Siphonaptera, Pediculina, bed-bug, and similar forms, which breathe an air freer from dust, the spiracles lie free on the outside of the body.
“When the stigmata are free and without any protection on the abdomen, there are other ways by which the entrance of foreign bodies into the tracheæ is prevented. In such cases the body is covered with dense hairs, as in most Diptera and Neuroptera, as well as many Lepidoptera; or there is situated in front of the stigma either a small fissure which is covered over by a number of hairs arising from the edge, as in many Orthoptera; or, as in most insects, a luxurious 441growth of hairs on the inside of the stigma forms a thick filter for the air. Thus we see that also in this respect each species of insect is completely adapted to its surroundings.” (Krancher.)
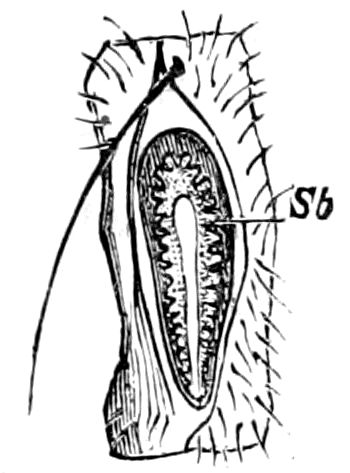
Fig. 404.—A, thoracic stigma of the house-fly: Sb, valve which closes the opening.
Fig. 405.—Diagrammatic figures of the internal apparatus which closes the trachea, in the stag-beetle: A, trachea open; in B, closed; St, the stigma, with its grated lips; Ct, cuticula of the body-walls; Vk, closing pouch; Vbü, closing bow; Vba, closing band; M, occlusor muscle.—From Judeich and Nitsche.
The closing apparatus of the stigma.—Whether the external opening of the stigma is permanently open or closed, communication with the tracheæ may be cut off at pleasure during respiration by an internal apparatus of elastic chitinous bands and rods and the occlusor muscle.
The parts concerned in this operation are: 1. The closing bow; 2. The closing lever or peg; 3. The closing band; 4. The occlusor muscle (Figs. 405, 406).
Fig. 406.—Stigma, with the closing apparatus, of Smerinthus populi (imago), seen from within: b, closing bow; c, closing band: o, stigmatic opening; r, external chitinous ring; l, closing lever; m, occlusor muscle; s, scales which lie like roofing tiles over the stigma.—After Krancher.
“The first three parts are chitinized; they form a ring around the stigmatic opening, and are united to each other by joints. The bow is usually crescentic and as a rule surrounds one-half of the trachea. On the other side is the closing band which, by different contrivances, representing the closing lever or peg, becomes closely pressed against the closing bow. This lever is usually of the shape of a slender chitinous rod, which causes the closure; but it can also bend rectangularly, become converted into a typical lever as in the Lepidoptera, or it may assume the form of two peg-like processes, which press with their base against the closing bow.” (Krancher.)
“The closure of the spiracular opening is effected by the contraction of the muscles, while the opening is due to the elasticity of the chitinous parts. When at rest the spiracle is naturally open, 442so that the air in the trachea can directly communicate with the external air. Usually one end of the muscle is attached to the closing peg, and the other end to the closing bow. Where, as in Melolontha, the closing apparatus is provided with two levers, then naturally the muscle binds these two together and brings about by powerful contractions a firm closure of the trachea”; but, remarks Krancher, “this is not the only kind; there are numerous modifications. Besides the form just described, the levers assume the form of valves (Sirex), or of a brush (Pulex); or of a ring (larvæ of Diptera) with a circular muscle attached to it; or of a ring which simply becomes compressed (thoracic stigmata of Diptera).”
c. Morphology and homologies of the tracheal system
As first shown by Bütschli, the tracheal system is a series of segmentally arranged tubular invaginations of the ectoderm; a pair of stigmata primitively occurring on every segment of the body except perhaps the most anterior, and the last two or last one, a reduction in their number having since taken place, until in the Podurans none have survived. In the supposed ancestor of myriopods and insects, Peripatus, there are tracheæ; but they are very fine, simple, not-branched chitinous tubes which are united into tufts at the base of a flask-shaped depression of the integument, the outer aperture of which depression is regarded as a stigma. In one species (P. edwardsii) these tufts and their openings are scattered irregularly over the body; but in another kind (P. capensis) some of the stigmata at least show traces of a serial arrangement, being disposed in longitudinal rows—two on each side, one dorsally and one ventrally, those of each row, however, being more numerous than the pairs of legs. (See p. 9 and Fig. 4, D.)
It should be observed that in Peripatus, which does not possess urinary tubes, the segmental organs or nephridia are well developed, hence the tracheal tubes coexisting with them cannot be their homologues. We are therefore compelled to regard the tracheal system as of independent origin, arising in the earliest terrestrial air-breathing arthropod, and not indebted for its origin to any structure found in worms, unless perhaps, as both Kennell and Lang suggest, to dermal glands, since, according to Kennell, certain Hirudinea and many Turbellarian worms possess long, mostly unicellular, glands which spread far through the parenchyma of the body. (Kennell.)
Thus Kennell supposes that the ancestors of the Tracheates had spiracles on every segment of the body where the internal organization allowed them to exist. 443“The reduction of the breathing holes to a smaller number, and their restriction of a pair only to a single segment, was brought about partly by adaptation to a peculiar mode of life,—as insect larvæ especially teach us,—partly also—I may say mechanically—as a result of the obstruction to their development made by the growth or excessive development of other organs.” Among these he reckons the thick, dense cuticula of the integument, the internal fusion of several segments to form body-regions, and the arrangement and great development of the muscles in the head and thorax, etc. (p. 29.)
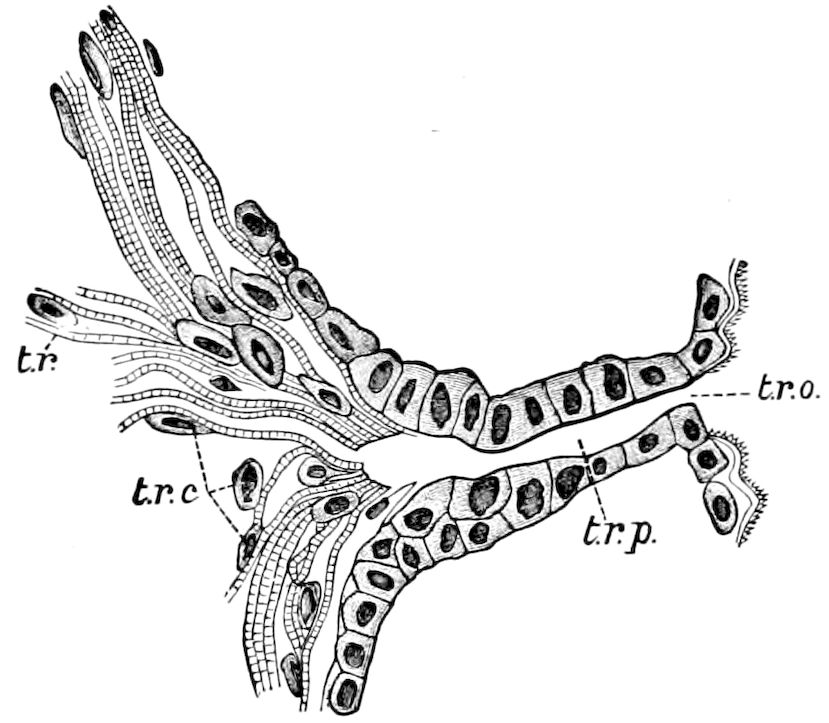
Fig. 407.—Section through a tracheal pit and diverging bundles of tracheal tubes taken transversely to the long axis of the body: tr, tracheæ, showing rudimentary spiral fibre; tr. c, cells resembling those lining the tracheal pits, which occur at intervals along the course of the tracheæ; tr. o, tracheal stigma; tr. p, tracheal pit.—After Balfour, from Sedgwick.
Kennell has suggested the origin of the tracheæ of Peripatus from the unicellular dermal glands of annelidan ancestors, since he has found glands in certain land-leaches of tropical America, which are provided with enormously long tubular passages united into bundles and opening externally, these tubes appearing to be slightly chitinized. Fig. 407 will show the appearance of a bundle of fine tracheal tubes of Peripatus ending at the bottom of a follicle formed by a deep invagination of the integument, which may be regarded as a primitive spiracle. (See Kennell, Ueber einige Landblutegel des tropical America, Zool. Jahrb. ii, 1886; also Die Verwandtschaftsverhältnisse der Arthropoden, 1891, p. 25.) We may add that Carrière supposes from his study of the embryology of the wall-bee (Chalicodoma muraria), published in 1890, that not only the salivary glands, but also the tentorium, are homologues of the tracheæ, while other structures than tracheæ may have evolved from unicellular dermal glands, which are widely distributed. It may in this connection be observed that some authors derive the book-lungs or book-leaf tracheæ of Arachnida from the gills of Limulus; hence if those of Arachnida arose from quite different and more specialized organs than dermal glands, it is not impossible that the tracheæ of Peripatus, Myriopods, and insects arose de novo, and then we need not look for any primitive structures in worms from which they arose.
Although Bütschli in 1870 in his embryology of the honey-bee called attention to the “great similarity which the eleven pairs of invaginations in the eleven first trunk-segments in their first indication (anlage) have with the spinning-glands, and also with the segmental organs of Annelids,” he did not go further than this, and it is now known that in the 2d maxillary segment open not only spinning-glands, but in the embryo a pair of stigmata.
Paul Mayer, however, regarded the tracheæ and urinary tubes as homodynamous structures, and this view was advocated by Grassi (1885) for the reason that while in the embryo honey-bee there are ten pairs of stigmata, the first thoracic and two last abdominal segments wanting them, the germs of the urinary tubes arise in a corresponding situation on the two last abdominal segments. To this view Emery (Biol. Centralb., 1886, p. 692) objects that in Peripatus the nephridia and tracheæ “have nothing to do with the segmental organs,” as Peripatus besides nephridia possesses both coxal glands and tracheæ.
444Both Kennell and Lang derive the coxal glands of Arthropoda from the setiparous or parapodial glands of annelid worms, and the recent endeavor of Bernard to show that the tracheæ arose from setiparous glands seems to be disproved by the fact that in insects as well as in other Arthropoda coxal glands with their outlets exist in the same segments as those bearing stigmata. Reasoning by exclusion, we are led to regard Kennell’s original view as the soundest.
Patten, however, regards the tracheæ as modified ends of nephridia, remarking: “Since in Acilius some of the abdominal tracheæ at first communicate with the cavities of the mesoblastic somites, it is probable that all the tracheæ represent the ectodermic portions of the nephridia.” (Origin of Vertebrates from Arachnids, p. 355.)
It is probable, therefore, that the tracheæ first arose as modifications of dermal glands, as in mites and Peripatus, and that at first they were not provided with tænidia (as in Chilopoda), while in later forms tænidia were developed. In the earliest tracheate forms the stigmata were not segmentally arranged, probably appearing irregularly anywhere in the body, but afterwards in the myriopods and insects became serially arranged.
d. The spiral threads or tænidia
It is generally supposed that the so-called “spiral thread” forms a continuous thread from one end of a tracheal branch to the other. This was first shown not to be the case by Platner in 1844. Minot has proved that “there is not a single spiral thread, but several, which run parallel to one another and end after making a few turns around the trachea.”
The tænidia we have found to be in some cases separate, independent, solid rings, though when there is more than one turn the thread necessarily becomes spiral. The tænidia of a main branch stop at the origin of the smaller branches, and a new set begins at the origin of each branch. The tænidia at the origin of the branch do not pass entirely around the inside of the peritoneal membrane; in the axils they are short, separate, spindle-shaped bands (Fig. 409).
At one point in the main trachea of the larva of Datana the tænidia were seen to end singly on one side (at a considerable distance from any branch or axil) at intervals, with a tænidium situated between them, making four or five turns; then there is only one band situated between two ends; this band or thread is succeeded by a set with five turns between the two ends, this set being succeeded by one complete ring situated between two ends; in all cases the ends vary in length, some threads being short and others long, so that they apparently end anywhere along the circumference of the trachea, and this arrangement is seen to apparently extend along the whole length of the trachea. Hence it is seen that as a rule the tænidia vary much in length, and never, as generally supposed, pass continuously from one end to another of a tracheal branch, for there are many spirals in a branch, each making only from one to five turns, 445most usually four turns. Fig. 408, part of a trachea of Dyticus marginatus, shows that at a slight bend in a trachea the tænidia is interrupted, and short, incomplete, wedge-shaped tænidia (e) are interpolated; at A, d is seen a split in one of the tænidia (compare also MacLeod, Pl. 1, Fig. 9). The threads are quite irregular in width. In the axils of the branches there is, as seen in Fig. 409, a basketwork of independent, short, often spindle-shaped tænidia; these are succeeded by longer ones, until we have threads passing entirely around near the base of each new branch; these being succeeded by others which make from two to five spiral turns.
Fig. 408.—Tænidia of Dyticus: d, a split tænidium; e, e, ends of tænidia.
The shape of the tænidia appears to vary to a great extent. In lepidopterous insects we have observed them to be in their general shape rather flat and slightly concavo-convex, the hollow looking towards the centre of the trachea. Minot’s section (Fig. 393) shows that in Hydrophilus they are cylindrical and solid, and Chun states that those of Stratiomys are round, while in Eristalis they are round, with a ridge projecting into the cavity of the trachea; in Æschna the thread is quadrangular. MacLeod states that sometimes it is cylindrical, in other cases flat, likewise prismatic; Macloskie believes that the spiral threads of the centipede are “fine tubules, externally opening by a fissure along their course.”
Fig. 409.—Tænidia of Dyticus in an axil of two branches: e, e, ends of tænidia.
Stokes confirms Macloskie’s statements, stating that in the hemipterous Zaitha fluminea “the tænidia are fissured tubules formed within and from chitinized folds of the intima, the convexity of the 446folds looking towards the lumen of the tracheæ.” In Fig. 414, 1, are represented portions of several tænidia showing the fissure, which is sometimes interrupted; at 2 are seen “the formation of what may be called apertures in a chitinous bridge.” Stokes regards the tænidia as “inwardly directed folds of the membrane.” Near the spiracles the tracheal membrane is externally studded with minute papillæ, as shown at 3, where are represented three broad and incomplete tænidia, with the tapering end, or the beginning, of another. Stokes adds, “Here they are only broad grooves, with no appearance of the narrow fissure of the completed tænidium. At 4 is figured a portion of the internal surface of a large trachea near the external orifice, the tænidia being in an incipient stage, evidently forming more or less of a network, as is usually the case next to the stigma” (compare p. 451, and Fig. 414).
Fig. 410.—End of salivary duct in base of proboscis of Stomoxys calcitrans: a, incomplete and irregular tænidia; b, two tænidia making incomplete rings near the distal end of the duct.
The tracheæ of chilopod myriopods appear to be like those of insects. A number of authors have failed to detect the spiral threads in the Julidæ. As to the Arachnida, several observers, including Menge and Bertkau, have denied the existence of the spiral thread in the spiders with the exception of the Attidæ; and MacLeod finds them “scarcely visible” in Argyroneta.
Besides the tracheæ, the salivary duct is kept permanently distended by tænidia, which, however, are not spiral. They usually form incomplete rings, as in Stomoxys, arranged as shown in Fig. 410.
The labella (proboscis) of flies are supported by incomplete chitinous tubes or “pseudo-tracheæ,” the ends of which form the scraping teeth, this being, according to Dimmock, their primary function. Dimmock describes them as cylindrical channels opening on the surface in zigzag slits. These channels are held open by incomplete rings, one end of which is forked. “These rings are apparently 447arranged so that one has its fork on one side of the opening of the channel, the next ring the fork on the opposite side of the channel, and so on, in alternation. Their true structure is revealed when flattened out.”
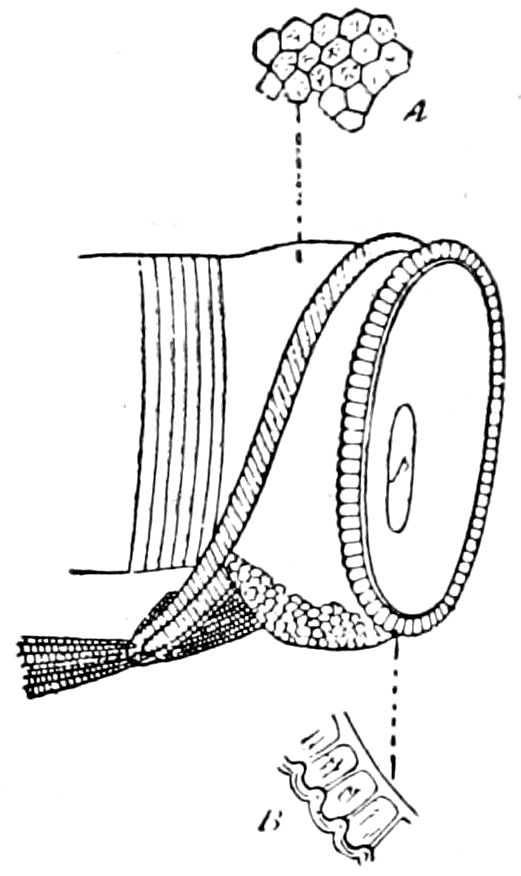
Fig. 411.—Abdominal spiracle (left side) of cockroach (P. americana), side view, showing the bow: p, lateral pouch of spiracle (in centre) seen from within. The tessellated structure of spiracle and trachea shown at A, and the margin of the external aperture at B.—After Miall and Denny.
The use of the elastic tænidia is to render the tracheæ elastic, and to keep them permanently open, as is the case with the parallel rings of the trachea of the higher vertebrates. The tracheæ are thus rendered firm and solid, at the least expense of chitinous material. The spiral thread, as MacLeod remarks, “is the realization in nature of what engineers call a form of the greatest resistance.”
The tænidia are wanting in the fine endings of the tracheæ (tracheal capillaries); also in the cockroach, according to Miall and Denny, they are not developed in the large tracheæ close to the spiracles, and the intima or wall of the tube has a tessellated instead of a spiral marking (Fig. 411). The same structure is seen in the Perlidæ (Nemoura, Gerstaecker, Zeit. f. wissen. Zool. xxiv, Taf. xxiii, Figs. 5 and 7); also in Æschna (Hagen, Zool. Anz. 1880, p. 159). In certain fine tracheæ of the eyes of the fly no spiral threads are developed. (Hickson.) The air-sacs or dilated tracheæ are also without tænidia.
While in the living insect the main and smaller tracheæ are filled, with air, it is stated by Von Wistinghausen that the fine capillary ends contain a fluid.
e. Origin of the tracheæ and of the “spiral thread”
While we owe to Bütschli the discovery of the mode of origin and morphology of the tracheæ, which as he has shown[66] arise by invaginations of the ectoblast; there being originally a single layer of epiblastic cells concerned in the formation of the tracheæ; we are indebted to Weismann[67] for the discovery of the mode of origin of the “intima,” from the epiblastic layer of cells forming the primitive foundation of the tracheal structure.
448Weismann did not observe the earliest steps in the process of formation of the stigma and main trunk of the tracheæ, which Bütschli afterwards clearly described and figured.
Weismann, however, thus describes the mode of development of the intima; after describing the cells destined to form the peritoneal membrane, he says: “The lumen is filled with a clear fluid and already shows a definite border in a slight thickening of the cell-wall next to it.
“Very soon this thickening forms a thin, structureless intima, which passes as a delicate double line along the cells, and shows its dependence on the cells by a sort of adherence to the rounded sides of the cells (Taf. vii, 97 A, a b c). Throughout the mass, as the intima thickens, the cells lose their independence, their walls pressing together and coalescing, and soon the considerably enlarged hollow cylinder of the intima is surrounded by a homogeneous layer of a tissue, whose origin from cells is recognized only by the regular position of the rounded nuclei (Taf. vii, Fig. 97, B).
“Then as soon as the wavy bands of the intima entirely disappear, and it forms a straight, cylindrical tube, a fine pale cross-striation becomes noticeable (vii, 97, B, int), which forms the well-known ‘spiral thread,’ a structure which, as Leydig has shown, possesses no independence, but arises merely from a partial thickening of the originally homogeneous intima.
“Meyer’s idea that the spiral threads are fissures in the intima produced by the entrance of air is disproved by the fact that the spiral threads are present long before the air enters. Hence the correctness of Leydig’s view, based on the histological structure of the tracheæ, is confirmed by the embryological development, and the old idea of three membranes, which both Meyer and Milne-Edwards maintain, must be given up.”
Weismann also contends that the elastic membrane bearing the “spiral thread” is in no sense a primary membrane, not corresponding histologically to a cellular membrane. On the contrary, the “peritoneal membrane comprises the primary element of the trachea; it is nowhere absent, but envelops the smallest branches, as well as the largest trunks, only varying in thickness, which in the embryo and the young larva of Musca stands in relation to the thickness of the lumen.”
The trachea, then, consists primarily of an epithelial layer, the “peritoneal membrane,” or the invaginated epiblast; from this layer an intima is secreted, just as the skin or cuticle is secreted by the hypodermis. We may call the peritoneal membrane the ectotrachea, the intima or inner layer derived from the ectotrachea the endotrachea. The so-called “spiral threads” are a thickening of the endotracheal membrane, sometimes arranged in a spiral manner. For these chitinous bands we have proposed the name tænidia (Greek, little bands).
As to the origin of the spiral thread our observations[68] have been made on the caterpillar of a species of Datana, which was placed in alcohol, just before pupation, when the larva was in a semipupal condition, and the larval skin could be readily stripped off. At this time the ectotrachea of the larva had undergone histolysis, nothing 449remaining but the moulted endotrachea, represented by the tænidia, which lay loosely within the cavity of the trachea. The ectotrachea or peritoneal membrane of the pupa is meanwhile in process of formation; the nuclear origin of the tænidia is now very apparent.
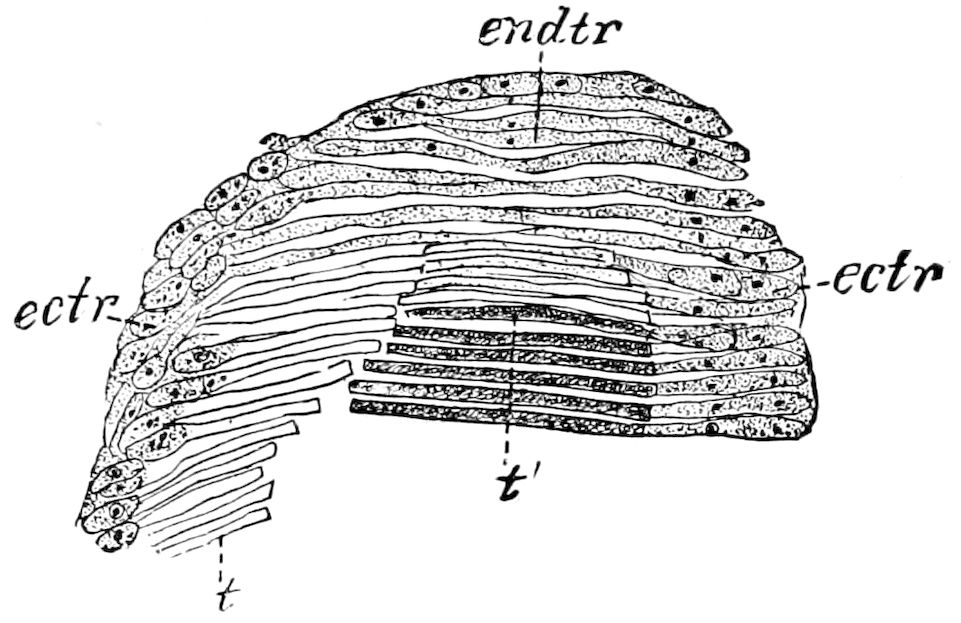
Fig. 412.—Longitudinal section of a trachea, showing the origin of the tænidia.
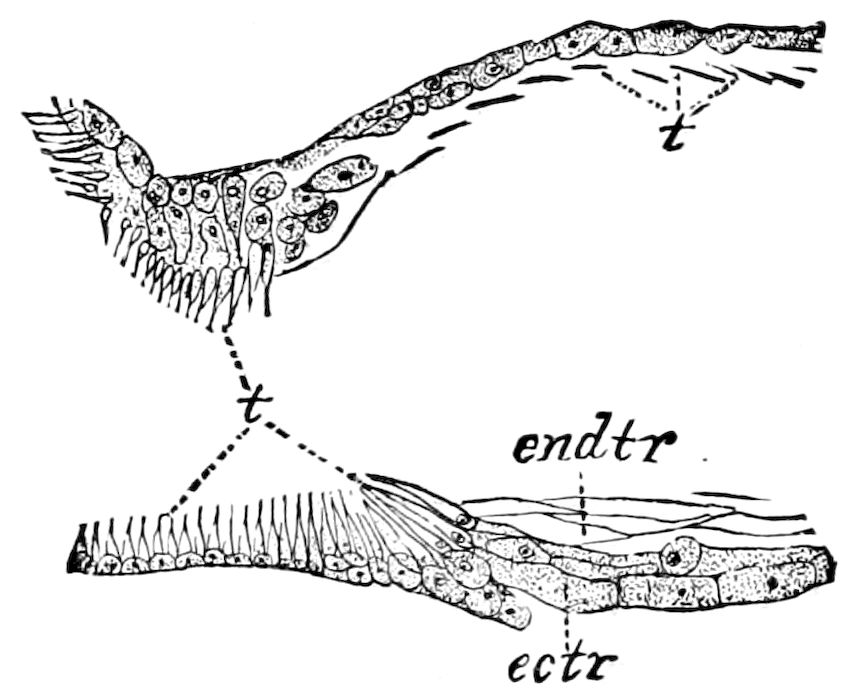
Fig. 413.—Origin of the tænidia from nuclei.
Fig. 412 represents a longitudinal section through a secondary tracheal branch, showing the origin of the chitinous bands, or tænidia. At t′ are pieces of six tænidia which have been moulted; ectr indicates the nuclei forming the outer cellular layer, the ectotrachea or peritoneal membrane. These nuclei send long slender prolongations around the inside of the peritoneal membrane; these prolongations, as may be seen by the figure, become the tænidia. The tænidia, being closely approximate, grow together more or less, and a thin endotracheal membrane is thus produced, of which the tænidia are the thickened band-like portions. The endotracheal membrane is thus derived from the ectotrachea, or primitive tracheal membrane, and the so-called “spiral thread” is formed by thickenings of the nuclei composing the secondary layer of nuclei, and which become filled with the chitin secreted by these elongated nuclei. The middle portion of the tænidia, immediately after the moult, is clear and transparent, with obscure minute granules, while the nuclear base of the cell is filled as usual with abundant granules, and contains a distinct nucleolus.
Fig. 414.—Tænidia and internal hairs of Zaitha.—After Stokes.
The origin of the tænidia is also well shown by Fig. 413, which is likewise a longitudinal section of a trachea at the point of origin of a branch. The peritracheal membrane or ectotrachea (ectr) is composed of large granulated nuclei; and within are the more transparent endotracheal cells; at t′ are fragments of the moulted tænidia. The new tænidia are in process of development at t; at base they 451are seen to be granulated nuclei, with often a distinct nucleolus, each sending a long, slender, transparent, pointed process along the inside of the trachea. These unite to form the chitinous bands or spiral threads.
Internal hair-like bodies.—In the large tracheæ of Lampyris very fine chitinous bristles project free into the cavity of the tube (Gerstaecher), while according to Leydig there are similar chitinous points in the tracheæ of the Carabid beetle Procrustes. Dugardin had previously (1849) called attention to such hairs, giving a list of the insects in which he observed them. Emery figures a section of the tracheæ of Luciola, “in wendig behaart.”[69] Stokes has described those of Zaitha fluminea (Fig. 414) as “internal chitinous, hair-like bodies arising from the fold of the tænidia and projecting into the lumen of the tubes.” They are hollow, their minute cavity distinctly communicating with that of the tænidium, from which they arise by an enlarged base. They end in an exceedingly fine point which is occasionally bifid or trifid. In Fig. 414, 4, several are shown attached to the wrinkles of the tracheæ near a spiracle, and at 5 is represented a transverse section of a trachea with three hairs projecting into its cavity.[70]
Stokes has also described “certain minute, elliptical bodies in the tænidia,
each with an internal, presumably glandular, appendage, to all appearance
forming part of the tænidium from which it springs.” These are shown in
Fig. 414, at 1, 3, and, more in detail, at 6; those at 7, whose thickness is about
1
8000 of an inch, appear as collections of exceedingly minute, rounded apertures
in a cushion-like mass. Although not commonly occurring on the tracheal
membrane between the tænidia, they may be found there, as at 4.
f. The mechanism of respiration and the respiratory movements of insects
By holding a locust in the hand one may observe the ordinary mode of breathing in insects. During this act the portion of the side of the body between the stigmata and the pleurum contracts and expands; the contraction of this region causes the spiracles to open. The general movement is caused by the sternal moving much more decidedly than the tergal portion of the abdomen. When the pleural portion of the abdomen is forced out, the soft pleural membranous region under the fore and hind wings contracts, as does the tympanum, or ear, and the membranous portions at the base of the hind legs. When the tergum or dorsal portion of the abdomen falls, and 452the pleurum contracts, the spiracles open; their opening is nearly but not always exactly coördinated with the contractions of the pleurum, but as a rule they are. There were 65 contractions in a minute in a locust which had been held between the fingers about ten minutes. It was noticed that when the abdomen expanded, the air-sacs in the first abdominal ring contracted.
For expanding the abdomen no special muscles are required, since it expands by the elasticity of the parts. For contracting its walls there are two sets of muscles, viz., special vertical expiratory muscles serving to compress or flatten the abdomen (Figs. 415–418), and other muscles which draw together or telescope the segments.
It was formerly supposed that when the abdomen contracted the air was expelled from the body and the tracheæ emptied; that, when the abdomen again expanded by its own elasticity, the air-tubes were refilled, and that no other mechanism was needed. But Landois insisted that this was not enough; as Miall and Denny state: “Air must be forced into the furthest recesses of the tracheal system, where the exchange of oxygen and carbonic acid is effected more readily than in tubes lined by a dense intima. But in these fine and intricate passages the resistance to the passage of air is considerable, and the renewal of the air could, to all appearance, hardly be effected at all if the inlets remained open. Landois accordingly searched for some means of closing the outlets, and found an elastic ring or spiral, which surrounds the tracheal tube within the spiracle.” By means of the occlusor muscle this ring compresses the tube, “like a spring clip upon a flexible gas-pipe.” “When the muscle contracts, the passage is closed, and the abdominal muscles can then, it is supposed, bring any needful pressure to bear upon the tracheal tubes, much in the same way as with ourselves, when we close the mouth and nostrils, and then, by forcible contraction of the diaphragm and abdominal walls, distend the cheeks or pharynx.”
Thus an important point in the respiration of tracheate animals, whether insects, myriopods, or arachnids, is, as Landois claimed, the closure of the spiracles, in order that pressure may be brought upon the air in the tubes, so that it may pass onward into the finest terminations.
The injection of air by muscular pressure into a system of very fine tubes may, as Miall and Denny remark, appear extremely difficult or even impossible. Graham (Researches, p. 44) applies the law of diffusion of gases to explain the respiration of insects, but until physical experiments have been made, we may, with Miall and Denny, “be satisfied that an appreciable quantity of air may be made by 453muscular pressure to flow along even the finer air-passages of an insect.”
As to the respiratory movements of insects, Plateau is the principal authority, and the following account of the process is taken from his elaborate memoir, and from the statements afterwards contributed by him to Miall and Denny’s “The Cockroach.”
Although many observers have superficially described the respiratory movements of various insects, Rathke was the first one to state precise views as to the mechanism of respiration. His posthumous work, treating of the respiratory movements of the movable chitinous plates of the abdomen, and of the respiratory muscles characteristic of all the principal groups, filled an important blank in our knowledge. But, notwithstanding the skill displayed in this research, many questions still remain unanswered which require more exact methods than mere observations with the naked eye or the simple lens.
Plateau, who was followed a year later by Langendorff, conceived the idea of studying, by such graphic methods as are now familiar, the respiratory movements of perfect insects.
“He has made use of two modes of investigation. The first, or graphic method, in the strict sense of the term, consisted in recording, upon a revolving cylinder of smoked paper, the respiratory movements, transmitted by means of very light levers of Bristol board attached to any part of the insect’s exoskeleton. Unfortunately, this plan is only applicable to insects of more than average size. A second method, that of projection, consisted in introducing the insect, carried upon a small support, into a large magic lantern fitted with a good petroleum lamp. When the amplification does not exceed 12 diameters, a sharp profile may be obtained, upon which the actual displacements may be measured, true to the fraction of a millimetre. Placing a sheet of white paper upon the lantern screen, the outlines of the profile are carefully traced in pencil so as to give two superposed figures, representing the phases of inspiration and expiration respectively. By altering the position of the insect so as to obtain profiles of transverse sections, or of the different parts of the body, and, further, by gluing very small paper slips to parts whose movements are hard to observe, the successive positions of the slips being then drawn, complete information is at last obtained of every detail of the respiratory movements; nothing is lost.”
“This method, similar to that employed by the English physiologist, Hutchinson,[71] is valuable, because it enables us, with a little practice, to investigate readily the respiratory movements of very small arthropods, such as flies or lady-birds. It has this advantage over all others, that it leaves no room for errors of interpretation.”
“Not satisfied with mere observation by such means as these, of the respiratory movements of insects, the writer has also studied the muscles concerned, and, in common with other physiologists (Faivre, Barlow, Luchsinger, Dönhoff, and Langendorff), has examined the action of the various nervous centres upon 454the respiratory organs. The result at which he has arrived may be summarized as follows:—
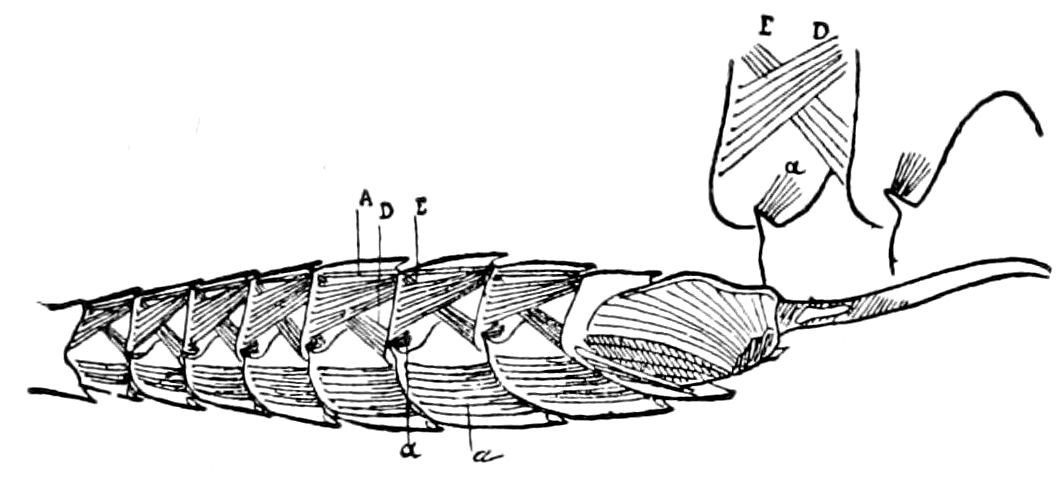
Fig. 415.—Muscles of right half of the abdomen of Forficula auricularia: A, a, longitudinal tergal and sternal muscles; D, E, oblique muscles; a (in upper figure) vertical expirator muscles.
“1. There is no close relation between the character of the respiratory movements of an insect and its systematic position. Respiratory movements are similar only when the arrangement of the abdominal segments, and especially when the disposition of the attached muscles, are almost identical. Thus, for example, the respiratory movements of the cockroach are different from those of other Orthoptera, resembling those of the heteropterous Hemiptera. Those of the Trichoptera are like those of the aculeate Hymenoptera, while the Locustidæ ally themselves in respect to these movements with the Neuroptera and Lepidoptera.
“2. The respiratory movements of insects, when at rest, are localized in the abdomen. As graphically stated by Graber, in insects the chest is placed at the hinder end of the body. If thoracic respiratory movements exist, they do not depend on the action of special muscles.
“3. In most cases the thoracic segments do not share in the respiratory movements of an insect at rest. The respiratory displacements of the posterior segments of the thorax are, however, less rare than Rathke believed. Plateau has observed them in certain Coleoptera (Staphylinus, Chlorophanus, Corymbites), and they are more feebly manifested in Hydrophilus, Carabus, and Tenebrio. Among the singular exceptions to this rule is the cockroach (Periplaneta orientalis), in which the terga of the meso- and metathoracic segments perform movements exactly opposite in direction to those of the abdomen (Fig. 419).
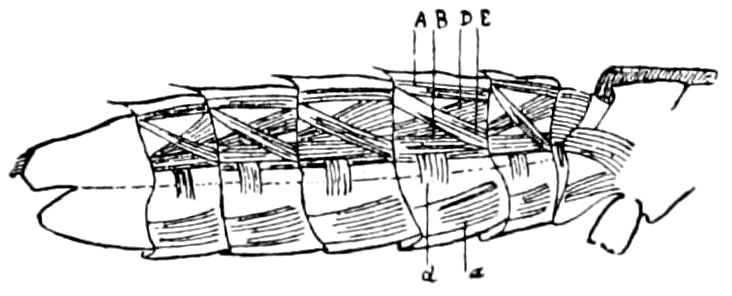
Fig. 416.—Muscles of the left half of abdomen of Staphylinus olens; A, B, longitudinal dorsal muscles; D, E, oblique fascia; a, longitudinal sternal muscles; d, respiratory muscles (vertical expirators).
“4. Leaving out of account all details and all exceptions, the respiratory movements of insects may be said to consist of the alternate contraction and recovery of the figure of the abdomen in two dimensions, viz. vertical and transverse. During expiration both diameters are reduced, while during inspiration they revert to their previous amounts. The transverse expiratory contraction is often slight, and may be imperceptible. On the other hand, the vertical expiratory contraction is never absent, and usually marked. In the cockroach (P. orientalis) it amounts to one-eighth of the depth of the abdomen (between segments 2 and 3); in Eristalis tenax to one-ninth (at the 2d segment).
“5. Three principal types of respiratory mechanism occur in insects, and these admit of further subdivision:
“a. Sterna usually short and very convex, yielding but little. Terga mobile, rising and sinking appreciably. To this class belong all Coleoptera, heteropterous Hemiptera, and Blattina (Fig. 420).
“In the cockroach (Periplaneta), the sterna are slightly raised during expiration (Fig. 421).
455“b. Terga well developed, overlapping the sterna on the sides of the body, and usually concealing the pleural membrane, which forms a sunken fold. The terga and sterna approach and recede alternately, the sterna being almost always the more mobile. To this type belong Odonata, Diptera, aculeate Hymenoptera, and acrydian Orthoptera (Fig. 422).
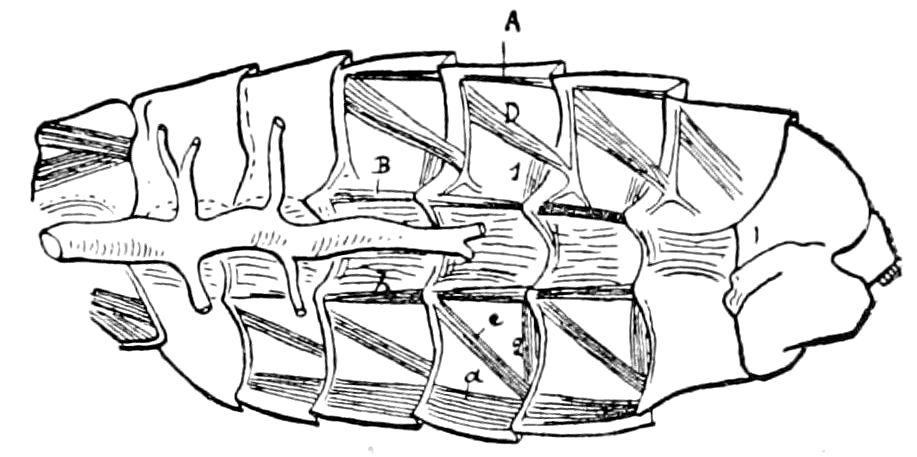
Fig. 417.-Muscles of right half of abdomen of Phryganea striata, ♀: A, B, longitudinal dorsal muscles; a, b, longitudinal sternal muscles; D, e, oblique muscles; 1, 2, inspirator muscles.
“c. The pleural membrane, connecting the terga with the sterna, is well developed and exposed on the sides of the body. The terga and sterna approach and recede alternately, while the pleural zone simultaneously becomes depressed, or returns to its original figure. To this type, Plateau assigns the Locustidæ, Lepidoptera, and the true Neuroptera (excluding Trichoptera) (Fig. 423).
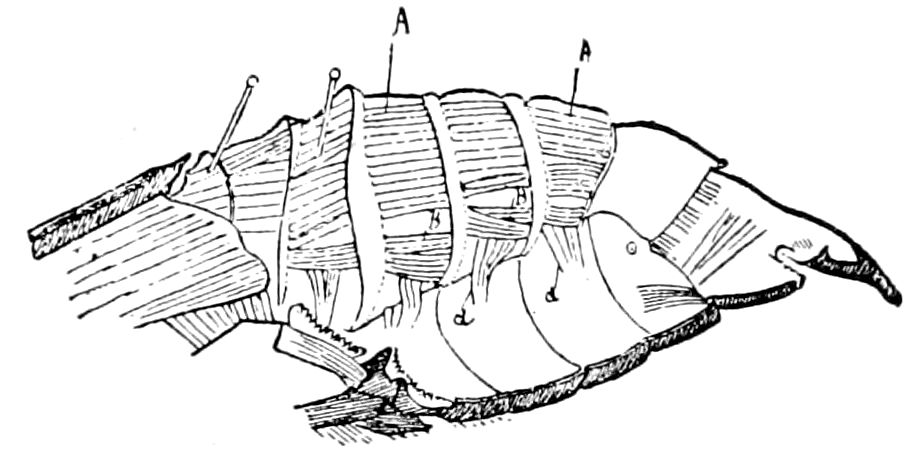
Fig. 418.—Muscles of left half of abdomen of Melolontha, ♀: A, B, longitudinal muscles (prétracteurs of Straus); a, a, true respiratory muscles (expirators).—This and Figs. 415–417, after Plateau.
“6. Contrary to the opinion once general, changes in length of the abdomen, involving protrusion of the segments and subsequent retraction, are rare in the normal respiration of insects. Such longitudinal movements extend throughout one entire group only, viz. the aculeate Hymenoptera. Isolated examples occur, however, in other zoölogical groups.
“7. Among insects, such as large beetles, Locustidæ, dragon-flies, etc., sufficiently powerful to give good graphic tracings, it can be shown that the inspiratory movement is slower than the expiratory, and that the latter is often sudden.
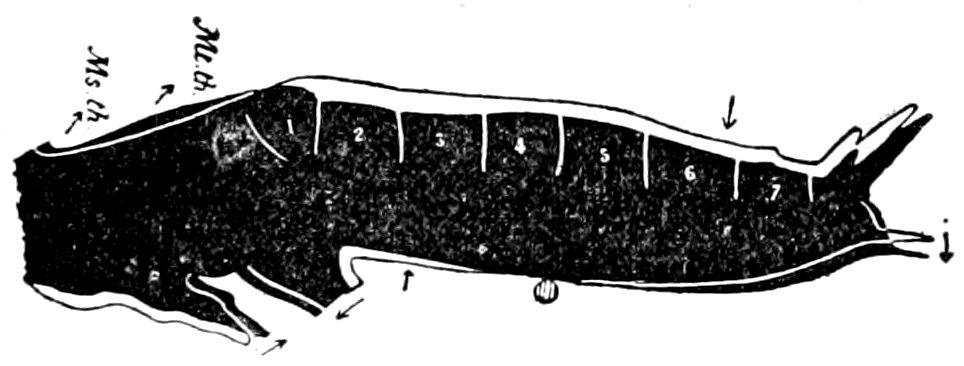
Fig. 419.—Profile of trunk of cockroach (P. orientalis). The black surface represents the expiratory contour, while the inspiratory is indicated by a thin line. The arrows show the direction of the expiratory movement: Ms. th, mesothorax; Mt. th, metathorax. Reduced from a magic-lantern projection.—After Plateau.
“8. In most insects, contrary to what obtains in mammals, only the expiratory movement is active; inspiration is passive, and effected by the elasticity of the body-wall.
“9. Most insects possess expiratory muscles only. Certain Diptera (Calliphora vomitoria and Eristalis tenax) afford the simplest arrangement of the expiratory muscles. In these types, they form a muscular sheet of vertical fibres, connecting the terga with the sterna, and underlying the soft, elastic membrane which unites the hard parts of the somites. One of the most frequent complications arises by the differentiations of this sheet of vertical fibres into distinct muscles, repeated in every segment, and becoming more and more separated as the sterna increase in length. Special inspiratory muscles occur in Hymenoptera, Acridiidæ, and Trichoptera.
456“10. The abdominal, respiratory movements of insects are wholly reflex. Like other physiologists who have examined this side of the question, Plateau finds that the respiratory movements persist in a decapitated insect, as also after destruction of the cerebral ganglia or œsophageal connectives; further, that in insects whose nervous system is not highly concentrated (e.g. Acridiidæ and dragon-flies), the respiratory movements persist in the completely detached abdomen; while all external influences which promote an increased respiratory activity in the uninjured animal, have precisely the same action upon insects in which the anterior, nervous centres have been removed, upon the detached abdomen, and even upon isolated sections of the abdomen.
“The view formerly advocated by Faivre, that the metathoracic ganglia play the part of special, respiratory centres, must be entirely abandoned. All carefully performed experiments on the nervous system of Arthropoda have shown that each ganglion of the ventral chain is a motor centre, and, in insects, a respiratory centre, for the somite to which it belongs. This is what Barlow calls the ‘self-sufficiency’ of the ganglia.” (Miall and Denny.)
Fig. 420.—Transverse section of abdomen of a lamellicorn beetle. The position of the terga and sterna after an inspiration is indicated by the thick line; the dotted line shows their position after an expiration; and the arrow marks the direction of the expiratory movement.
Fig. 421.—Cross-section of abdomen of cockroach.
Fig. 422.—Cross-section of abdomen of bee (Bombus).
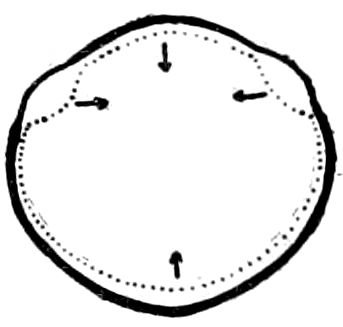
Fig. 423.—Cross-section of abdomen of Sphinx.—This and Figs. 420–422 after Plateau.
Plateau has made similar observations upon the respiration of spiders and scorpions; but, to his great surprise, he was unable, either by direct observation, or by the graphic method, or by projection, to discover the slightest respiratory movement of the exterior of the body. This can only be explained by supposing that inspiration and expiration in pulmonate Arachnida are “intrapulmonary,” and affect only the proper, respiratory organs. The fact is less surprising because of the wide zoölogical separation between Arachnida and insects.
g. The air-sacs
In flying insects the tracheæ are in certain parts of the body enlarged into sacs of various sizes. These air-sacs were first observed by Swammerdam in a beetle (Geotrupes) and afterwards by Sir John Hunter in the bee, Sprengel subsequently discovering them in other insects. Those of the cockroach were described and illustrated in a very elaborate and detailed way by Straus-Dürckheim (Figs. 424 and 425). These vesicles are without tænidia. In the locust (M. femur-rubrum) there is a pair of very large vesicles in the prothorax (Fig. 396). The five pairs of large abdominal air-sacs arise, independently of the main tracheæ, directly from branches 457originating from the spiracles. All these large sacs are superficial, lying directly beneath the hypodermis, while the smaller ones are buried among the muscles. We have detected 53 of these vesicles in the head.
In the honey-bee (Fig. 426) and humble bee (Fig. 427) as well as the flies there are two enormous air-sacs at the base of the abdomen. In larval and wingless insects these sacs are entirely absent.
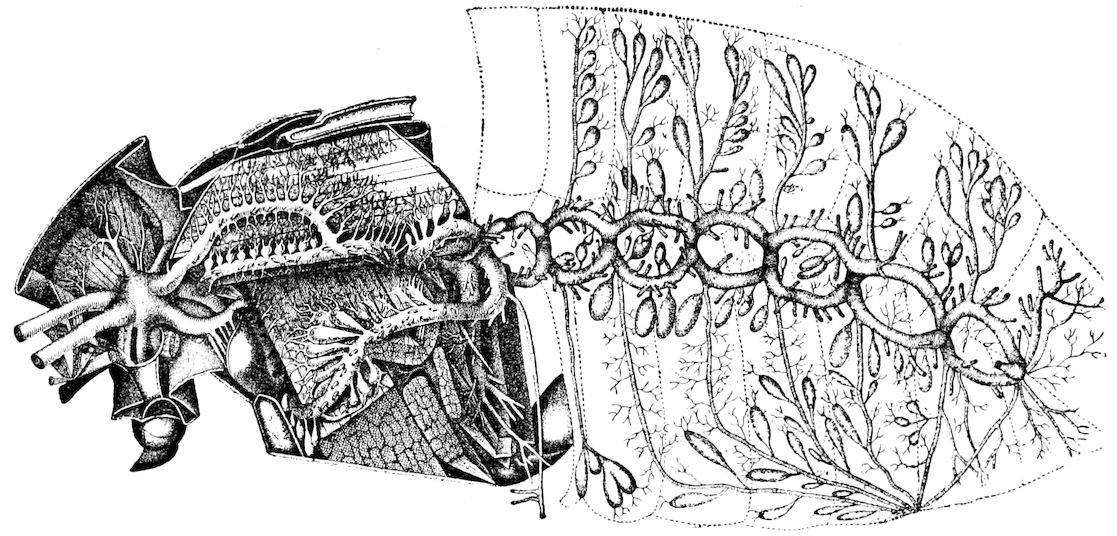
Fig. 424.—Thorax and abdomen of the cockchafer (Melolontha vulgaris), showing the tracheæ and air-sacs.—This and Fig. 425 after Straus-Dürckheim.
The use of the air-sacs.—It was supposed by Hunter as well as by Newport, and the view has been generally held, that the use of these sacs is to lighten the weight, i.e. lessen the specific gravity of the body during flight. It has, however, been suggested to us by A. A. Packard that this view from the standpoint of physics is incorrect. It is evident that the wings have to support just as much weight when the insect is flying, whether the tracheæ and vesicles are filled with air or not, the body of the insect during flight not being lightened by the air in the sacs. The use of these numerous sacs, some of them very spacious, is to afford a greater supply of 458air or oxygen than that contained in the air-tubes alone, and thus to afford a greater breathing capacity. The sacs are largest in dragon-flies, moths, flies, and bees, which are swift of flight. When we compare the active movements of these insects on the wing with those of a caterpillar or maggot, it will be seen that the far greater muscular exertions of the volant insect create a demand for a sudden and abundant supply of air to correspond to the increased rapidity of respiration; and the enlargements of the air-tubes, rapidly filled with air at each inspiration, render it possible to supply the demand.
Fig. 425.—Head of Melolontha vulgaris, showing the numerous air-sacs, represented only on the left side, front view.
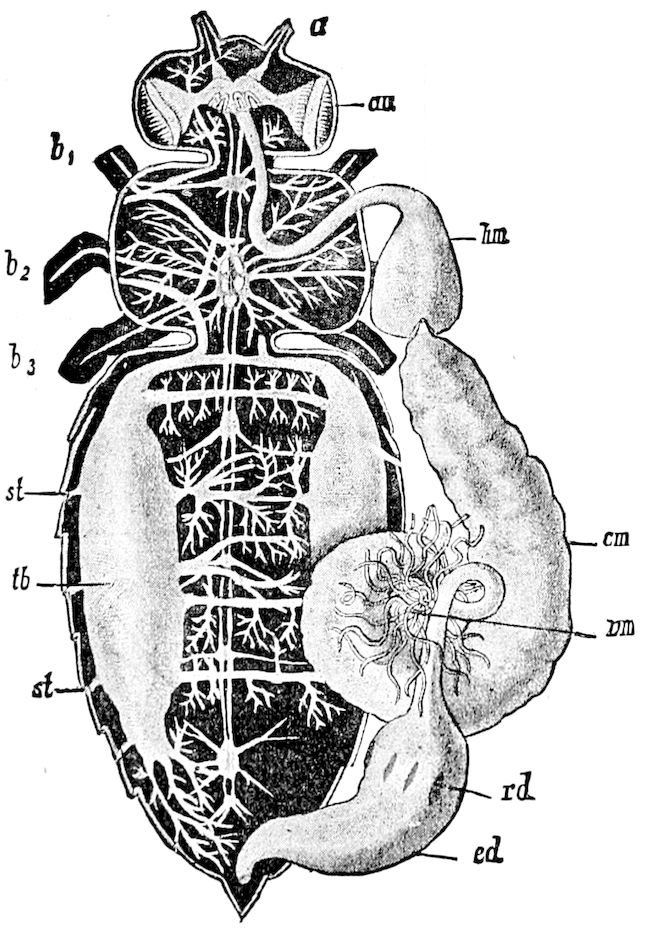
Fig. 426.—Tracheal, nervous, and digestive systems of the honey-bee (the tracheal system on the right side only partially drawn): tb, the large vesicles in the abdomen; st, stigmata; hm, honey stomach; cm, chyle stomach; vm, urinary tubes; rd, rectal glands; ed, rectum; a, antenna; an, eye; b1-b3, legs.—After Leuckart, from Lang.
The case is thus seen to be very different from that of those fishes which, having a swimming-bladder, can in the water change the specific gravity of their bodies. The case of insects is almost exactly paralleled by that of birds, where, as stated by Wiedersheim, the air-sacs appear to form integral parts of the respiratory apparatus: “a greater amount of air can by their means pass in and out during inspiration and expiration, especially through the larger bronchi, and consequently there is less necessity for the expansion of the lung parenchyma.” In other words, the supply of air in these sacs, as in insects, increases the breathing capacity of the bird during flight. Wiedersheim’s retention of the old idea that the specific gravity of the body is lessened (p. 262) seems, however, to be incorrect, as the weight of the bird’s body is not diminished by the air contained in the sacs.
h. The closed or partly closed tracheal system
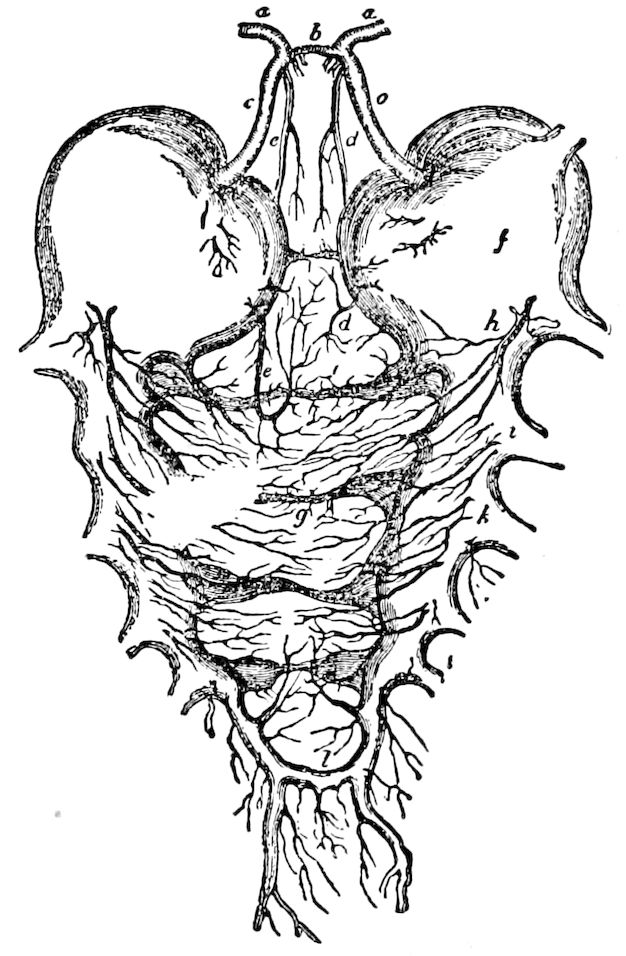
Fig. 427.—The lateral and lower series of sacs of Bombus terrestris, ♂: a, c, longitudinal tracheæ;, connected by b, and dilated at f, and again in the succeeding segments; i, k, funnel-shaped dilatations passing over the dorsal surface of the abdomen and anastomosing (g) with their fellows opposite; at l, communicating directly by a large branch.—After Newport.
There are two chief morphological tracheal systems: 1. The open or normal and primitive (holopneustic) type, and 2. The closed, or secondary and adaptive, i.e. apneustic, type. The open system is characterized by the presence of the stigmata. Through them the air directly enters into the tracheal tubes, whose delicate walls allow the exchange of gases in the blood. This type occurs in all sexually mature individuals, and also in the greater number of larvæ.
The closed or apneustic tracheal system is distinguished either by the want of stigmata, or, if present, they are not open, and do not function, so that the tracheæ cannot communicate with the air. In such cases the direct oxygenation of the blood is effected through the delicate integument, especially over the surface of the body in general, or in certain specialized places where the gill-like expansions of the skin are rich in tracheæ; such outgrowths, generally tubular or leaf-like, are called by Palmén tracheal gills.
This closed form of the tracheal system only occurs in the larval stage of aquatic or parasitic insects, as in the Plectoptera (Ephemeridæ), Perlidæ, Odonata, and Trichoptera, besides single genera of other orders, i.e. among Coleoptera, Gyrinus, Pelobius, Cnemidotus, and the young larva of Elmis; in the aquatic caterpillar of Paraponyx; in certain Diptera (Corethra, Chironomus, etc.), and some of the parasitic Hymenoptera (Microgaster).
Palmén has discovered that in the nymphs of Ephemeridæ, Perlidæ, Odonata, and the larvæ of most Trichoptera the tracheal branch (stigmatal branch) sent from the longitudinal trachea to where the thoracic stigmata would be situated if present, or where their vestiges 460only exist, are aborted, becoming simple solid cords not filled with air (Fig. 436, vf, and 447, f, funiculus or stigmatic cord). In the imago, however, they resume their function, connecting with the open functional stigmata. In Corethra, in its earliest stages, the entire tracheal system is, like the stigmatic branch, a system of solid cords and empty of air. (Palmén.)
Embryology shows that these stigmatal branches are well developed, and are formed at the same time as the stigmata. It was also shown by Dewitz, in a posthumous paper (1890), that in the young larval stage of the Odonata and Ephemeridæ the tracheal system is at first an open one, and in some of the families (Libellulidæ, Agrionidæ, and Ephemeridæ) thoracic stigmata are seen at a very early stage. From numerous experiments Dewitz concludes that in the young stages of Odonata and Ephemeridæ there is an open tracheal system; certainly in very young nymphs the thoracic spiracles allow the air to pass out. Fully grown nymphs of Æschnidæ, Libellulidæ, and Agrionidæ are capable not only of forcing the air out, but also, like the perfect insect, of inhaling it. Moreover, he proved that the gills of Ephemeridæ and Agrionidæ are not indispensable for the maintenance of life, as the insects can live without them, breathing either through the skin or by the rectum, or in both ways. It would seem that while in freshly hatched or very young larvæ of aquatic insects of different orders the skin is so delicate as to allow of dermal respiration, in after life, when the skin becomes thicker and denser, these expansions (gills), provided with a very thin and delicate skin, of a necessity grow out from the walls of the body.
It thus appears that the closure and total or partial abolition of the stigmata are in adaptation to aquatic life, and that such insects have descended from terrestrial air-breathing winged forms. This is an important argument against the view that the wings are modified tracheal gills.
In this connection may be noticed the closure of the 2d and 3d thoracic stigmata in holopneustic insects. We have found on laying open the body of a Sphinx larva that a large number of tracheal branches are seen to arise from the prothoracic and from the first pair of abdominal stigmata. Now between these points there are no spiracles or any external signs of them, there being in Lepidoptera no mesothoracic or metathoracic spiracles. Yet the main lateral trachea between the prothoracic and first abdominal segments deviates from its course and bends down to send off a small shrivelled stigmatal branch or cord to a place where, did a spiracle exist, we should look for it. In the larva of Platysamia cecropia, a similar vestigial stigmata branch is present.
In the larva of Corydalus, also, a trachea as large as the main longitudinal one takes its origin and passes directly under the main trachea. Now both tracheæ send a stigmatal branch opposite to where the mesothoracic stigma should be, if present, i.e. on the hind edge of the segment.
Verson, moreover, has found in the freshly hatched silkworm vestiges of meso- and metathoracic stigmata, each consisting of a circle of high hypodermal cells radially arranged around a common centre. The stigmatal branch is long, but shrivelled; its peritoneum is widened out into several berry-like saccules filled with cell-elements. In profile these rudimentary stigmata appear as a series of high hypodermal cells, which form the basis of a short blind tube.

Lydia M. Hart del.
Plate I.—Examples of metapneustic insects: 1, Bittacomorpha clavipes, larva; 1 a, false foot; 1 b, its pupa; 2, Limnophila luteipennis; 2 a, end of larva; 2 b, its pupa; 3, end of larva of Tipula eluta.—After C. A. Hart.
After the second moult there begins a peculiar transformation of the rudimentary stigmata. The stigmatal branch connected with them sends off at various points thick tufts of capillary tracheæ which press against the base of the blind tube. Gradually lengthening, they form a fold which continues to increase in length. The numerous tufts of tracheal capillaries extend beyond the inner surface of the two layers of which the developing wing consists, the berry-like saccules are drawn into the wing and converted into more or less thick tubes, which finally form the “veins.” It is clear, therefore, says Verson, as Landois claimed, that the wings of Lepidoptera must be regarded as in the fullest sense organs of respiration. (Zool. Anz., 1890, p. 116.)
The number of pairs of stigmata varies, especially in maggots or larval Diptera, in adaptation to their varied modes of life. The larvæ of most flies (Muscidæ) have a pair of peculiarly shaped processes on the prothoracic segment bearing spiracular openings, and two anal spiracles, while in Ctenophora atrata L. only the anal pair are present. In the rat-tailed maggots (Eristalis) the long caudal process ends in two stigmata forming a respiratory tube, which can be thrust out of the water for the reception of air. In the larval mosquito (Fig. 433) and its ally, Mochlonyx, a short thick dorsal tube arises from the penultimate segment of the body, in which the two main tracheæ end, opening outward by a single spiracular aperture. Other dipterous larvæ (Simulium, Tanypus, and Ceratopogon) possess no spiracles, the tracheal system being a closed one.
The larvæ of most water beetles (Dyticidæ, Hydrophilidæ) possess but two spiracles, which, as in maggots, are situated at the end of the body. The aquatic larva of Amphizoa, according to Hubbard, breathes much as in the Dyticidæ, by means of two large valvular spiracles placed close together at the end of the body; “closed or rudimentary stigmata also occur on the mesothorax and on abdominal segments one to seven inclusive.”
Hubbard adds: “The larva of Pelobius is wholly aquatic and breathes by branchiæ, but the obsolete stigmata are indicated precisely as in Amphizoa, with the exception of the last pair, which in Amphizoa are open spiracles, but in Pelobius are suppressed; the terminal eight segments being prolonged in a swimming stylet.”
From a review of the distribution of spiracles, and their atrophy, partial or total, it will be seen that there are intermediate stages between the open (holopneustic) and closed (apneustic) systems. These, following Schiner, Brauer, and Palmén, may be defined thus:
1. Metapneustic type.—The larvæ possess only a single pair of open stigmata situated at the end of the body. (The dipterous Eristalis, Tipula, Culex, Ptychoptera, Bittacomorpha (Plate I.) with certain Tachinidæ, and in Coleoptera, the larvæ of Dyticus, and allies of Hydrophilus and Cyphon.)
4622. Propneustic type.—The pupæ of Corethra, Culex, etc., in which only the most anterior pair of spiracles are open.
Fig. 428.—Visceral tracheal system of the nymph of Æschna maculatissima: o, œsophagus; E, stomach; M, urinary tubes; R, rectum; A, anus; tv, visceral tracheal trunks; td, dorsal trunks.—After Oustalet.
3. Amphipneustic type.—Larvæ with a pair of open spiracles situated at each end of the body, the intermediate spiracles being closed. (Most dipterous larvæ, Musca, after the first moult, Œstridæ, Asilidæ, and Syrphus.)
Fig. 429.—Branchial tuft of nymph of Æschna.
4. Peripneustic type; with prothoracic and abdominal spiracles, the mesothoracic pair atrophied or closed. (The larvæ of Neuroptera, Mecoptera, Trichoptera, Lepidoptera, of most Coleoptera,[72] of most Diptera, and of most of the Hymenoptera.[73])
Fig. 430.—Part of three rows of respiratory folds from cuticular living rectum of Æschna. The shaded parts are abundantly supplied with tracheal tubes. The leaflets appear to be connected with a central trachea, but this is not really the case.—After Miall.
463These differences in the number of functional spiracles are in direct relation with the surroundings of the insects, the physical conditions of existence evidently determining the position of the active functional open spiracles and the closure of those useless to the organism.
i. The rectal tracheal gills, and rectal respiration of larval Odonata and other insects
The remarkable mode of respiration by tracheal gills situated within the intestine of the nymphs of dragon-flies was first described by Swammerdam and afterwards by Réaumur. The most complete and best illustrated modern account is that of Oustalet. In these insects the large rectum is lined with six double longitudinal ridges, in Æschna bearing numerous delicate tubes or papillæ, each of which contains very numerous (by estimate 24,000) tracheal branches (Fig. 431); while in Libellula the gills are lamellate (Fig. 432). The tracheæ arise both from the main dorsal and visceral longitudinal trunks, which give rise to secondary branches passing into the walls of the rectum and sending into the branchial papillæ fine twigs, which, extending to the distal end of the papilla or lamella, recurve and anastomose with the efferent twigs.
Fig. 431.—A small part of one leaflet, highly magnified, showing many fine tracheal branches. The portion shown is marked by a small circle in Fig. 430, lower left-hand corner.—After Miall.
Fig. 432.—Leaves, mh, from a lamellate tracheal gill of Libellula: t, trachea.—This and Fig. 429, after Oustalet.
The anal opening is externally protected by the suranal and lateral triangular chitinous plates, three to five in all. When open, the water passes into the rectum and bathes the rectal gills, where it may be forcibly expelled as if shot out from a syringe, thus propelling the insect forward. In Libellula the anus affords direct access to the intestinal cavity, but in Æschna Oustalet describes “a sort of vestibule separated from the rectum by a circular valvule.” He also states that the inspiration and the repulsion of water is produced at irregular intervals, and rather by the movements of the dorsal and 464sternal arches of the abdomen than by the contractions of the rectum, since the walls of this organ are less muscular than is supposed.
Fig. 433.—Larva of a mosquito (Culex nemorosus) of middle age, seen from above, the tracheal system omitted: at, antennæ; ab, their middle joint; eg, elastic articular membrane; atm, antennal muscle; atn, antennal nerve; zau, compound; eau, simple eye; os, brain; oex, extensor; ofl, flexor of labrum; ha, neck; œ, œsophagus; spd, salivary gland; mau, cœca; ch, chyle stomach; di, contents of intestine; mg, urinary tubes; dd, ileum; ed, rectum; a, anus; s, sipho; z″, its bristles; kb, tracheal gills: k1, k2, k3, closing lobes of the sipho; kn, basal tubercle of tactile hair; g, its ganglion cell; th, tactile hair of the siphon valve.
The nymph of Calopteryx (and probably of all the group Calopteryginæ) possesses rectal gills besides external caudal tracheal gills. There are three double rectal longitudinal folds or ridges, interpenetrated by tracheal twigs. (Dufour, denied by Poletaiew, but confirmed by Hagen.)
Dewitz claims that the caudal gills of the Agrionidæ are not their sole means of respiration, since he cut off the caudal tracheal gills of an Agrionid nymph, which continued to live for a week. Hence he thinks that there may be a rectal respiration, since under the microscope he saw a stream of water pass in and out of the end of the intestine.
Dewitz’ experiments prove that in young Ephemerids there may be besides branchial, both rectal and skin respiration. He saw under the microscope the anus for a while opened and then closed, causing the rectum to move; powdered carmine mixed with water was drawn into and then expelled from the rectum. There was, however, no enlargement and contraction of the abdomen as in the rectal respiration of Æschna. (Zool. Anz. 1890, p. 500.)
Fig. 434.—End of the body of the same larva as in Fig. 431, seen from the side, the branches of the main tracheæ (htr) omitted: kbl, excrementitial pellet in rectum; kb, tracheal gills; b, funnel of the closing apparatus; hz, hollow tooth of the closing apparatus; k1, k2, k3, siphonal lobes; th, tactile hair; as, chitinous plate; str, rudder; l, its thickened edge; sch, its shank; z′, z″, bristles.—This and Fig. 433, after Raschke.
Eaton states that there is a rectal respiration in the nymphs of may-flies, and Palmén observed in young larvæ of Bætis and Cloëon that the rectum took in “by gulps” water colored by carmine and expelled the whole of it at once, in order to fill it again in the same way. “This rectal respiration therefore corresponds to that of Libellulid larvæ.”
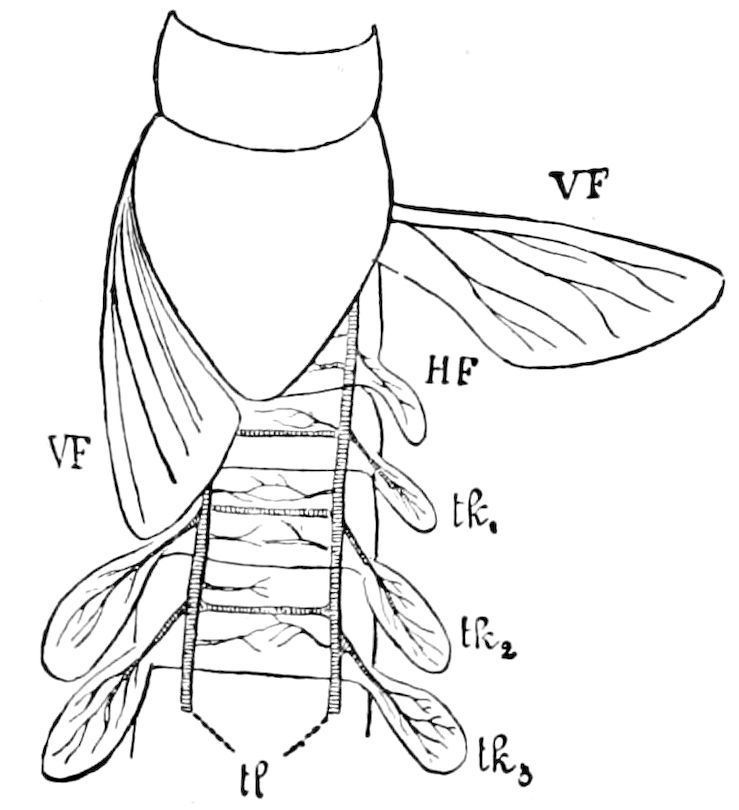
Fig. 435.—Thorax and anterior abdominal segments of the nymph of a may-fly (Cloëon dimidiatum) with tracheal gills (tk1, tk2, tk3) and the rudiments of the fore wings (VF) and hind wing (HF): tl, tracheal longitudinal trunks.—After Graber, from Lang.
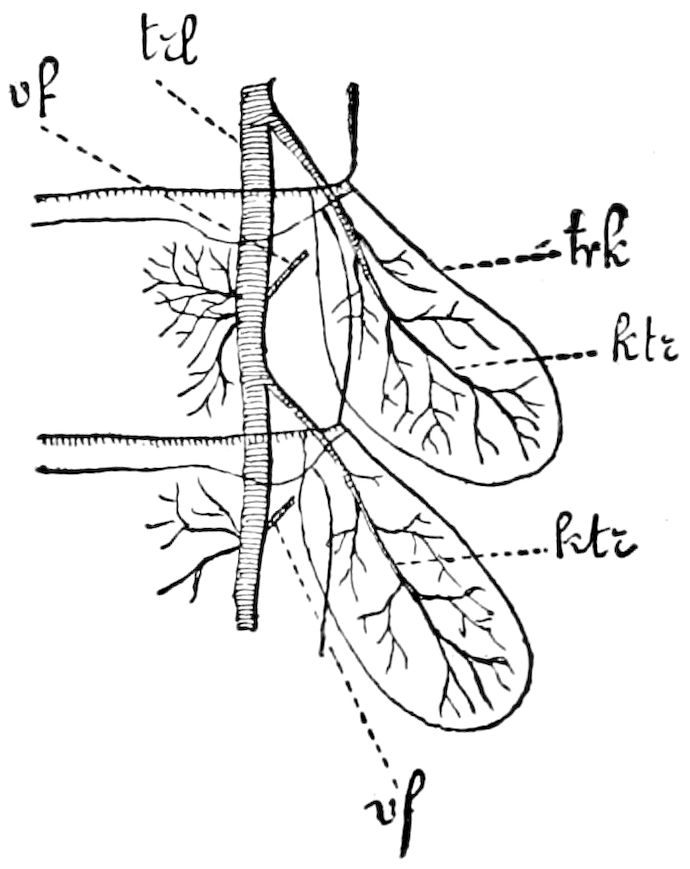
Fig. 436.—Gills on the middle abdominal segments of larva of Bætis binoculatus: trl, longitudinal tracheal trunks; vf, stigmatic cord; ktr, gill-tracheæ; trk, tracheal gills.—After Palmén, from Lang.
Besides breathing by spiracles, by tracheal gills, as well as through the integument, the larva of Culex has been observed by Raschke to have a rectal respiration. At the anterior end of the rectum arises a countless number of fine tracheæ, which pass through the walls and, subdividing, end in numberless very fine twigs in the papilla-like folds situated within the rectum. The supply of tracheal twigs is greatest where the papillæ are largest. (Figs. 433, 434.)
j. Tracheal gills of the larvæ of insects
In many aquatic insects respiration is carried on by tracheal gills. These are delicate, hollow, leaf-like or tubular outgrowths of the integument usually attached to the sides or end of the hind-body, and containing a trachea which usually sends off numerous minute branches, so that the exchange of gases readily takes place in them.
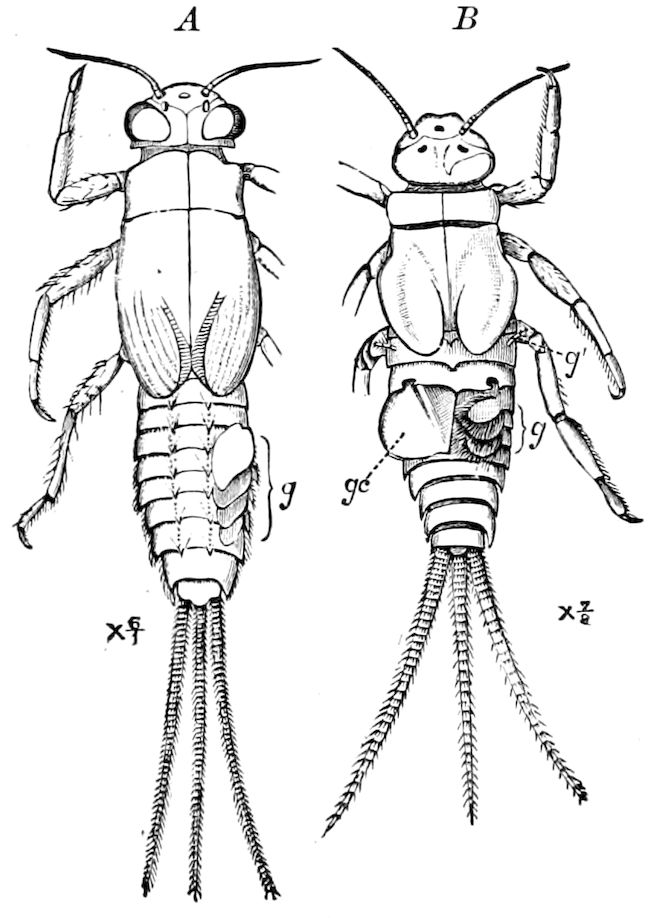
Fig. 437.—A, nymph of Ephemerella ignita, with gills of left side removed; g, gills. B, nymph of Tricorythrus (sp), with gill-cover of right side removed; gc, gill-cover; g, g′, gills.—After Vayssière.
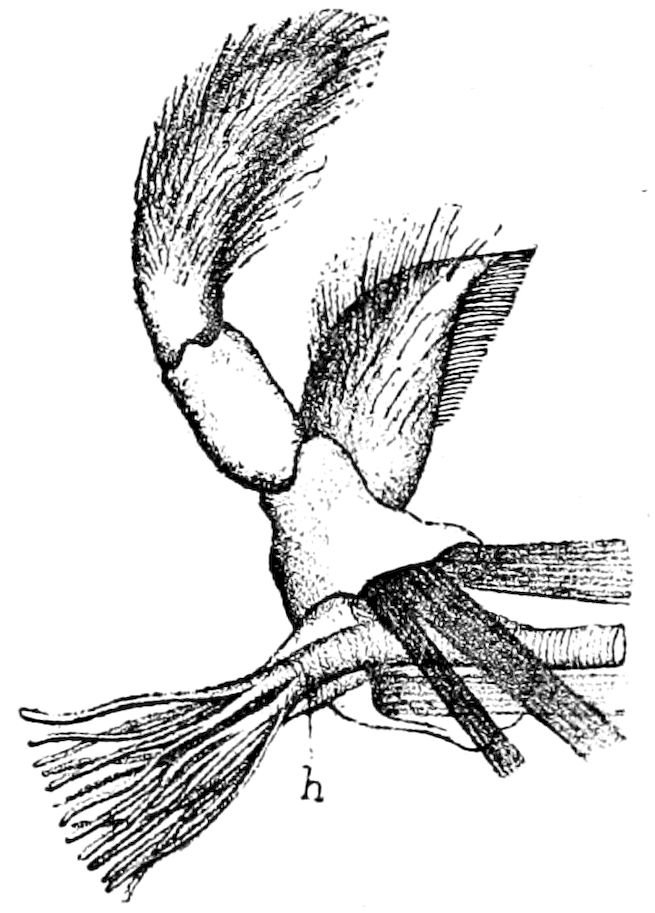
Fig. 438.—Left maxilla of Jolia weselii, with the cephalic tracheal gill (h) inserted at the base on the under side.—After Vayssière.
Palmén has shown that these tracheal gills, as he calls them, are not developed on the same segments as the stigmata, and that the two structures have no genetic connection with each other. It is evident that these gills are secondary, adaptive organs.
In some cases (see p. 475) the tracheæ are wanting, but as such gills are filled with blood, the air contained in the water must pass in through their delicate walls.
In the Plectoptera (Ephemeridæ) the tracheal gills are either foliaceous or filamentous; when foliaceous they form simple or double leaves, with or without branches, or with a fringe of tubules, or under the leaf-like cover-bearing tufts of filaments. They are situated on the (usually) basal seven abdominal segments, at their hinder edge (Figs. 435, 436). In Oligoneuria and Jolia a pair occurs on the under side of the head, attached to the maxillæ, while in 467Jolia there is a pair on the under side of the first thoracic segment at the insertion of each of the legs. In certain genera (Heptagenia, Oligoneuria, and Jolia), they are in the form of a flat cover, under which lies a tuft of respiratory tubes, or (Ephemerella) a small bifid cluster of very delicate leaves (Fig. 437, A). In Cœnis and Tricorythus the tracheal gills of the second pair are modified to form plates covering all the succeeding pairs, those of the first pair being nearly atrophied and well-nigh functionless. (Fig. 437, B.)
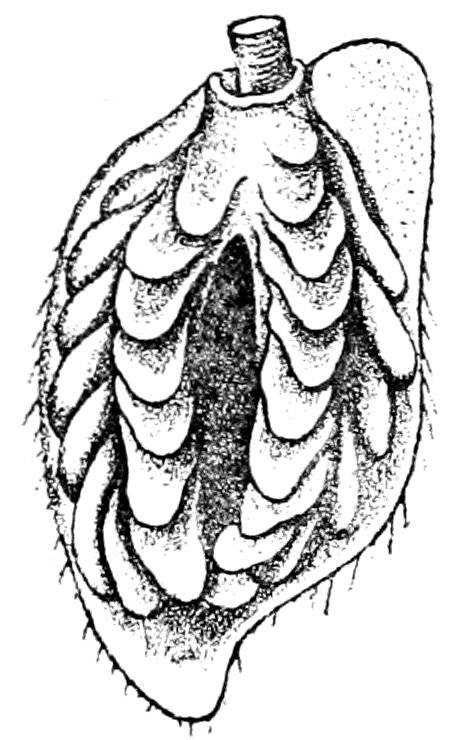
Fig. 439.—Inner side of a gill-cover of the first pair, of Ephemerella, with the tracheal gills.—After Vayssière.
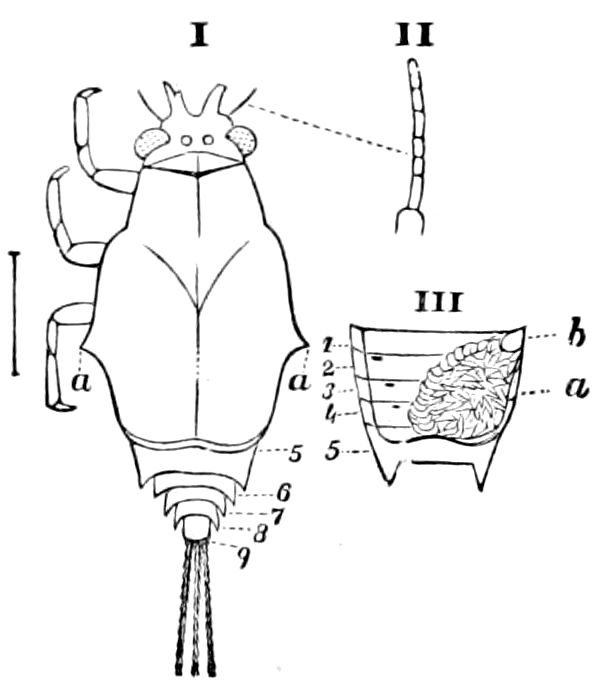
Fig. 440.—Nymph of Bætisca: III, section of abdomen; a, gills; b, flap; 1–9, abdominal segments.—After Walsh.
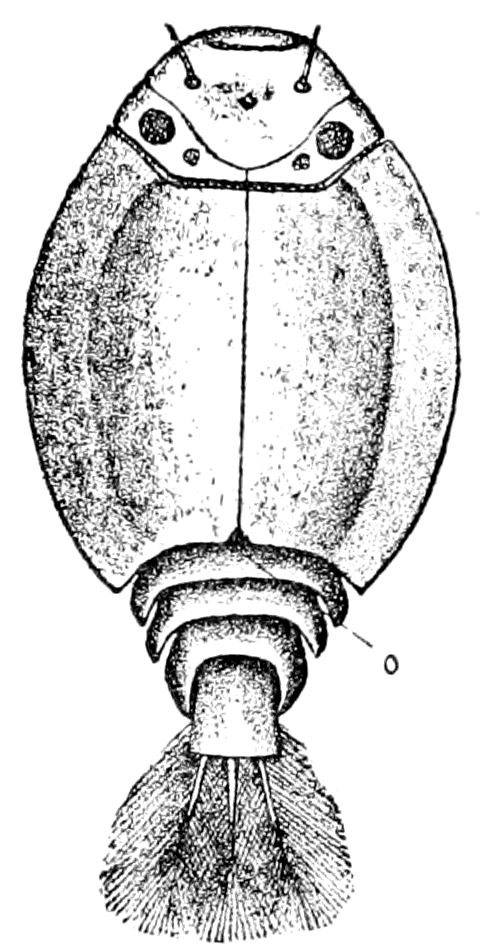
Fig. 441.—Nymph of Prosopistoma punctifrons: o, upper orifice of the respiratory chamber.—After Vayssière.
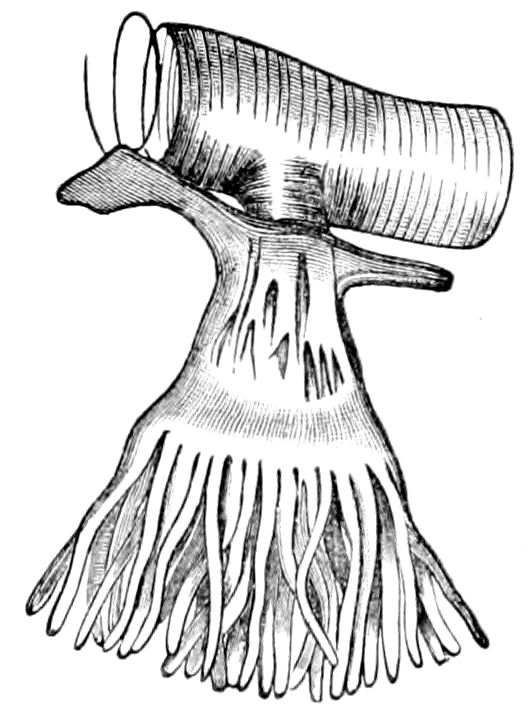
Fig. 442.—Filamentous tracheal gill and part of a trachea of Pteronarcys.—After Newport from Sharp.
Finally, in the highly modified forms Bætisca and Prosopistoma the tracheal gills are entirely concealed and protected by mesothoracic projections so as to form a true respiratory chamber, to which the water has access either by an opening behind, as in Bætisca, or by three openings, two ventral and one dorsal (Fig. 441), as in Prosopistoma.
The slender cylindrical tracheal gills of Heptagenia in the third or fourth nymphal stage are 2–jointed, and the first abdominal pair in Cænis are said by Palmén to be finger-shaped and 2–jointed. In Polymitarcys virgo the gills do not appear until the eighth or tenth day after hatching.
468Dewitz found that young nymphs of Ephemerids will well endure the amputation of their gills, while fully grown ones die. Amputation of the lateral gills hastens ecdysis. After the change of skin, the gills are smaller than before, and at first contain no tracheæ, but in a few weeks they develop as completely as in normal individuals. The caudal gills were also renewed.
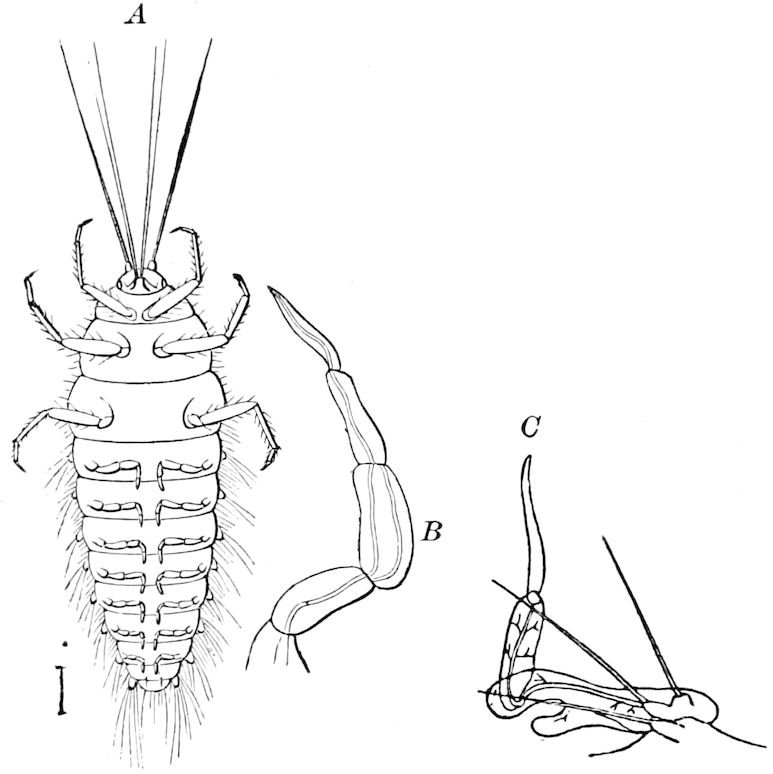
Fig. 443.—A, larva of Sisyra, enlarged. B, one of the hinder gills, with its tracheæ.—After Westwood, from Sharp. C, a gill, showing the branched tracheæ.—After Grube.
In the nymphs of Perlidæ the tracheal gills are usually present, and are either foliaceous (Nemoura) or more commonly filamentous in shape (Fig. 442). They are situated either on the prosternum (Nemoura and Pteronarcys), or on each side of the thorax, or on the sides of the abdomen, or are restricted to a tuft on each side of the anus at the base of the caudal stylets (Pteronarcys and Perla). Unlike the Ephemeridæ the gills persist in certain genera throughout life.
The larvæ of the aquatic Neuroptera, Sisyra, Sialis, and Corydalus possess lateral pointed bristle-like tracheal gills, which in Sisyra are 2–jointed; those of Sialis are, in the living larva, curved upwards and backwards (Fig. 444). Corydalus is also provided with a ventral tuft of delicate filamentous gills, which, however, according to Riley, do not appear until after the first moult.
While the nymphs of Agrionidæ (which have rectal gills) respire chiefly by the large caudal foliaceous gills (Fig. 445), there are, according to Hagen, two genera of the Calopteryginæ (Euphæa, Fig. 445, and Anisopleura) whose nymphs possess seven pairs of external lateral tracheal gills, in shape like those of Sialis, besides three caudal and three rectal tracheal gills.[74]
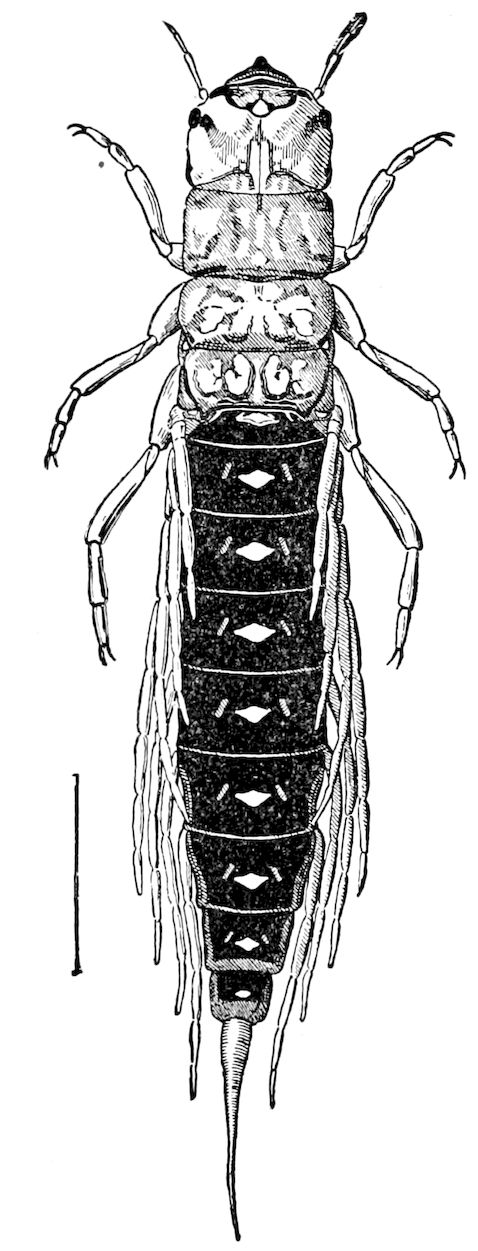
Fig. 444.—Larva of Sialis lutarius.—After Miall.
Fig. 445.—Caudal tracheal gill of nymph of Agrion.
Hagen has also detected in the under side of the 5th abdominal segment of Epitheca and Libellula a pair of sacs of the shape of a Phrygian bonnet, each of which contains a smaller sac lined with epithelium,—as in Æschna they occur in the 5th and 6th, and in Gomphus in the 4th, 5th, and 6th segments. This serial arrangement appears to confirm Hagen’s suggestion that they are survivals of abdominal gills, which in Euphæa are completely evaginated.
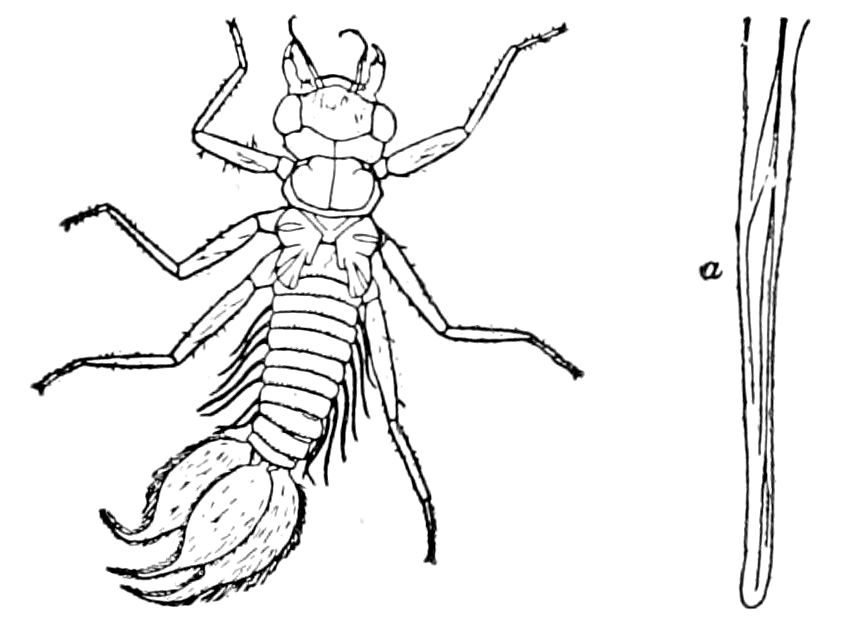
Fig. 446.—Nymph of Euphæa, showing the lateral gills: a, one enlarged.—Folsom del.
In the Trichoptera, all of which, except Enoicyla, are apneustic, and most of which have tracheal gills, the latter are filamentous, and arise either from the dorsal and ventral sides of the abdominal segment, or they grow out from the sides; while in certain genera (Neuronia, Phryganea, etc.) the gills are represented by conical hooks on the sides of the 1st abdominal segment, which are evidently respiratory, as they contain numerous tracheæ. The tracheal gills are either single or more rarely form tufts (Figs. 447, 448).
In Hydropsyche (Fig. 448) the tracheal gills persist throughout life, while in other genera they only last through the pupal stage. When first hatched, the larva of Phryganea lacks gills. The larvæ of most of the Hydropsychidæ, Rhyacophilidæ, and Hydroptilidæ 470have no gills, though they appear well developed in the pupal stage. (Klapálek.)
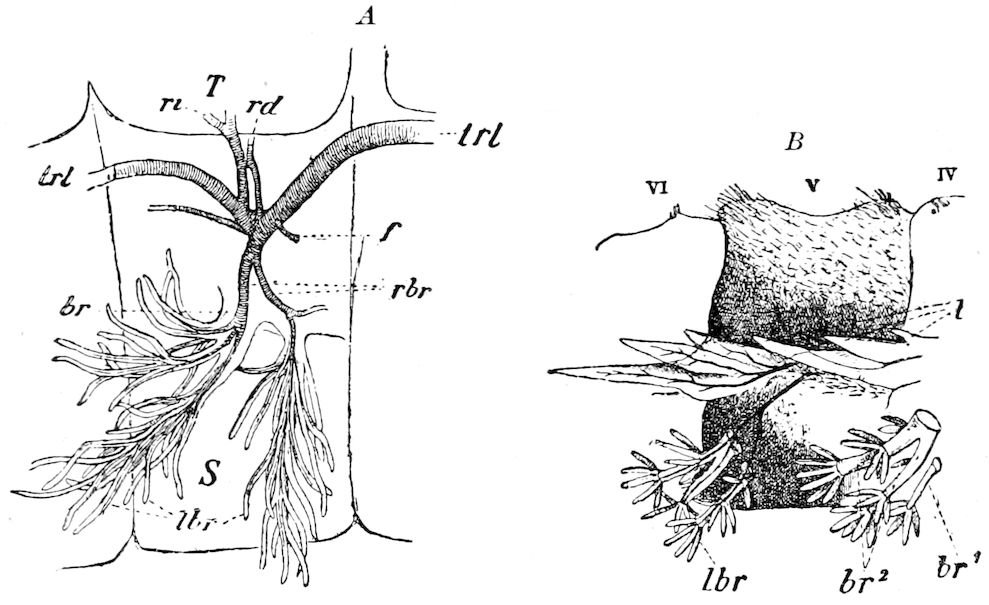
Fig. 447.—A, an abdominal segment of Hydropsyche, with the tracheal gills (lbr): trl, longitudinal tracheal trunk; f, stigmatal branch. B, 5th abdominal segment of pupa of the same; l, the three lateral flaps of the tergite; br1, br2, branchiæ.
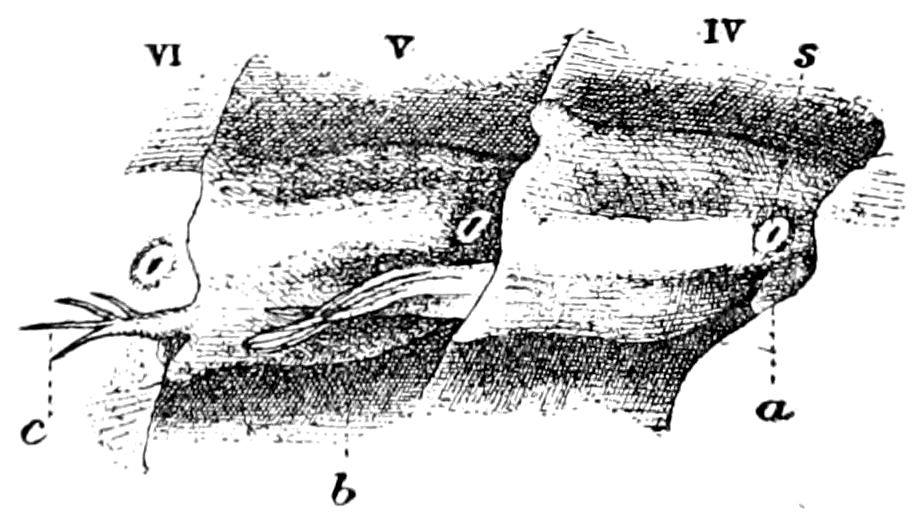
Fig. 448.—Imago, abdominal segments IV to VI, with the gills at a concealed in their natural condition; at b, drawn out with the needle; at c, projecting abnormally and dried.—This and Fig. 447 after Palmén.
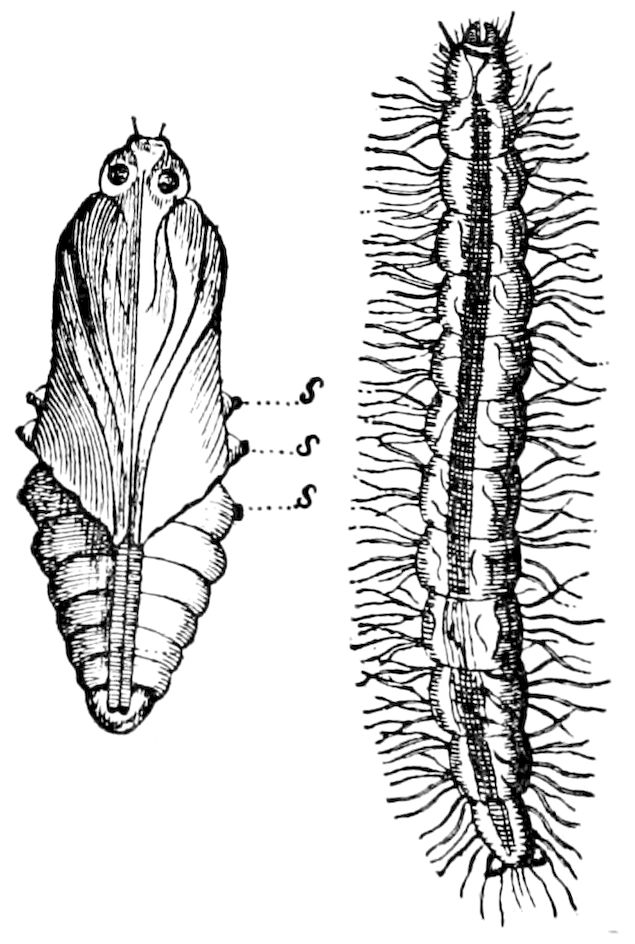
Fig. 449.—Larva and pupa of Paraponyx stratiolata, enlarged; s, spiracle.—After De Geer (compare Hart’s figure of P. obscularis, living in the Illinois River).
The only lepidopterous larva known to be provided with tracheal gills is that of the pyralid genus Paraponyx. Its thread-like gills, arranged in tufts of three or four, arise from a common tubercle situated on the sides of nearly all the segments. Wood-Mason describes the East Indian P. oryzalis as “covered with a perfect forest of soft and delicate white filaments,” arranged in tufts disposed in four longitudinal rows. “The stigmata of the 2d, 3d, and 4th abdominal somites only are clearly discernible.” The caterpillar crawls “free and uncovered” over the submerged leaves 471of the rice plant “in the very midst of the water.” In a Brazilian species of Paraponyx described as Cataclysta pyropalis, by W. Müller, the tufts are reduced to simple unbranched filaments, and the case is more complex than in the European species (Fig. 449).
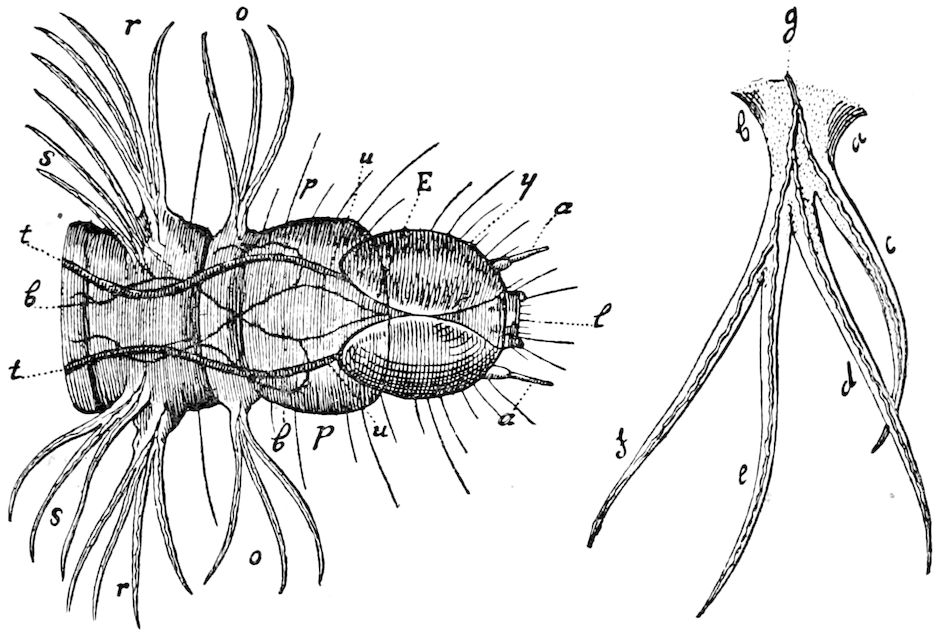
Fig. 450.—Anterior end of larva of P. stratiolata, showing the head and first two thoracic segments, with their gills: A, a tuft of gills, much enlarged.—After De Geer.
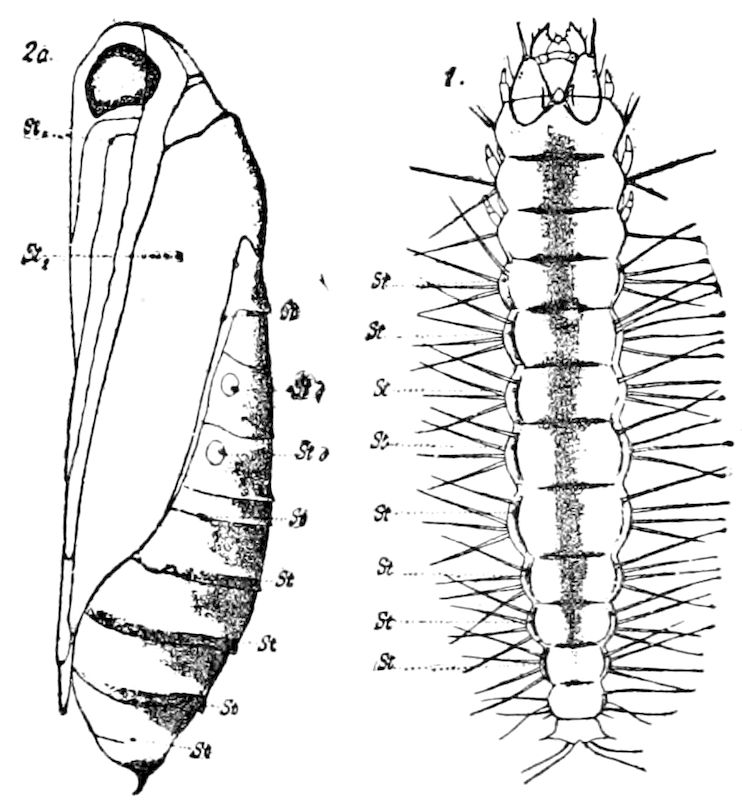
Fig. 451.—Larva (1) and pupa (2a) of Paraponyx pyropalis enlarged: st, stigmata.—After W. Müller.
Of coleopterous larvæ breathing by tracheal gills there are but few. The larva of Gyrinus (Fig. 454) respires by 10 pairs of slender, hairy abdominal gills similar to those of Corydalus, and the stigmata are entirely wanting. Somewhat similar are the tracheal gills of Hydrocharis caraboides. Hydrobius has shorter setose gills, our American species having seven pairs of short setose gills. It has two spiracles at the end of the body, through which the air is taken by thrusting the body out of the water. The larvæ of two other aquatic coleopterous genera, Pelobius and Cnemidotus, also have 472gills; those of the former situated at the base of the coxæ, and brush-like, but containing no tracheæ, though filled with blood, while those of Cnemidotus are very long, bristle-like, jointed, and arising from the dorsal side of the thoracic and abdominal segments. The stigmata are wanting. (Schiödte.)
The larva of the dipterous genus Tanypus respires by two caudal papilliform processes, in each of which a trachea ramifies.
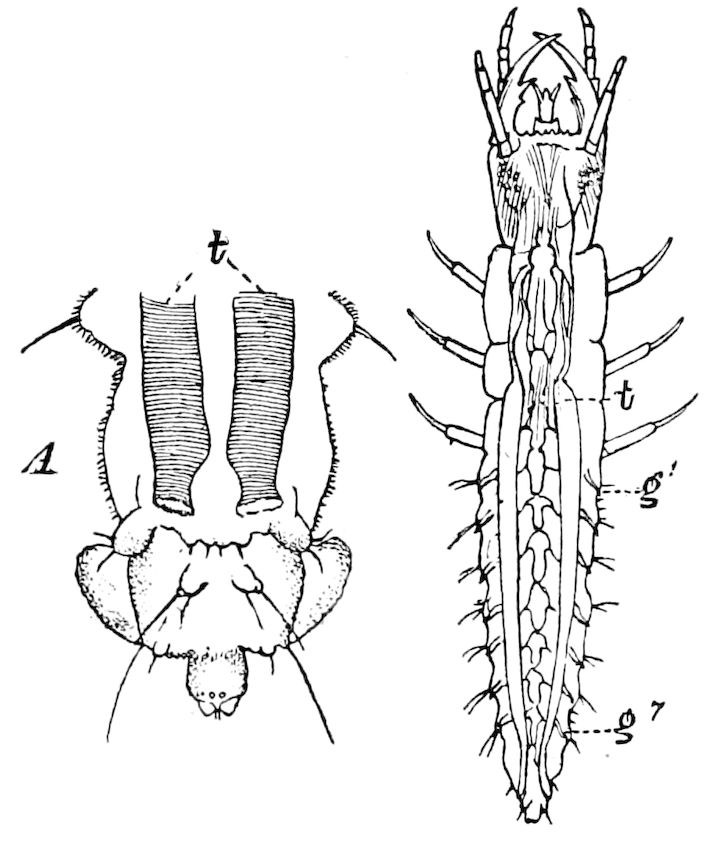
Fig. 452.—Freshly hatched larva of Hydrobius: t, enlarged tracheæ, the heart between them; g1-g7, the seven pairs of gills. A, end of body, enlarged, showing the two terminal stigmata.—Emerton del.
Certain larvæ with both stigmata and tracheal gills are enabled either to live in or out of water or on the surface, as in the case of certain beetles (Cyphonidæ, Elmidæ, Hydrophilidæ, Fig. 452), or the larval mosquito and Psychodes (Fig. 455); also the nymphs of dragon-flies.
The larvæ of the Cyphonidæ (Helodes, Cyphon, Hydrocyphon) possess but a single pair of stigmata, situated in the penultimate abdominal segment, while at the end of the abdomen are delicate tracheal gills. The two main tracheal trunks are much swollen. When on the surface of the water the larva breathes through the stigmata situated near the end of the abdomen; when floating in the water, the larva, like that of Gyrinus, carries along at the end of its body a bubble of air. The gills are only of use, as Rolph thinks, when the insect is compelled to remain a long time under water.
The larva of our native Prionocyphon discoideus (Say) is described by Walsh as “vibrating vigorously up and down a pencil of hairs proceeding from a horizontal slit in the tail”; this pencil is composed “of three pairs of filaments, each beautifully bipectinate. I presume it is used to extract air from the water.” When the larva is at the surface the pencil of hairs touches the surface of the water, and occasionally a bubble of air is discharged from the tail. “The general habit is to crawl on decayed wood beneath the surface, occasionally swimming to the surface, probably for a fresh supply of air.” (Proc. Ent. Soc. Phil., i, p. 117.)
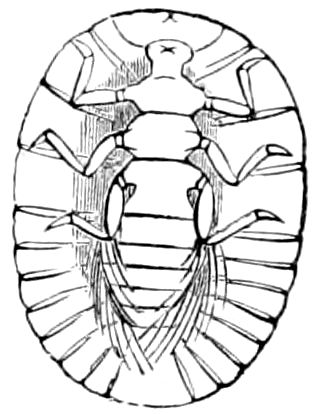
Fig. 453.—Larva of Psephenus, enlarged.
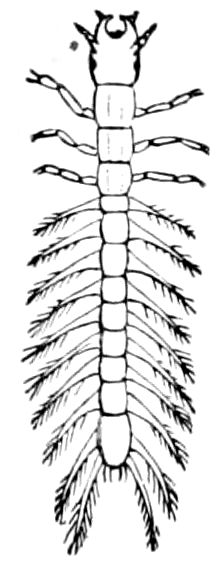
Fig. 454.—Larva of Gyrinus.—After Westwood.
The larvæ of the small water beetles of the family Elmidæ (Elmis, Potamophilus, Macronychus, and Psephenus) have similar habits. That of Elmis has ten dorsally situated pairs of spiracles, and on the end of the body bushy gills which are protruded at pleasure. The young larva is without spiracles, its tracheal system being closed. Macronychus and Potamophilus have similar habits. In the larva of the latter genus, which has nine pairs of spiracles, there are at the end of the body on each side three tufts of thread-like gills which are connected with the two main horizontal tracheæ, while the branches of the abdominal tracheæ are dilated into numerous (64) bladder-like sacs. The larva usually breathes through the caudal gills. When the water is low or dried up, the air is inhaled directly through the spiracles. (Kolbe.)
The larva of Psephenus lecontei, by its broad hemispherical body, is adapted to adhere to the smooth surface of rounded stones, in which situation we have found it. Although it is said by Rolph to have two pairs of spiracles, one pair on the mesothoracic and the other on the 1st abdominal segment, it probably rarely rises to the surface to breathe the air direct.
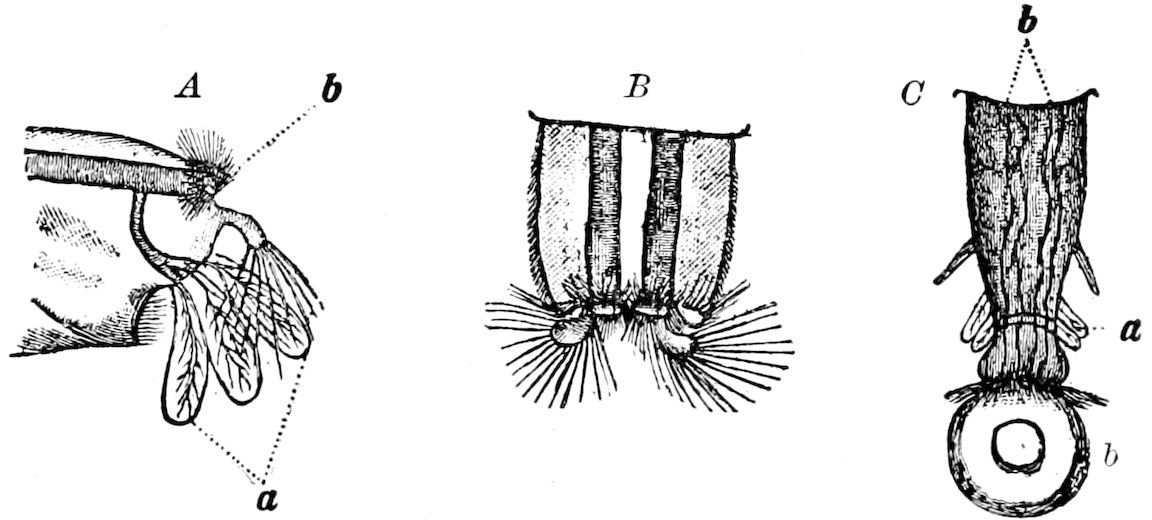
Fig. 455.—End of body of a Psychodes larva: A, end of body of a young, freshly moulted larva, side view: a, the three anal gills; b, the left air-cavity. B, older larva of the same species, with the open air-cavity seen from above. C, end of larva of another species as it goes down into the water with a bubble of air, b, between the crown of hairs of the air-cavity or tube: a, the two pairs of anal gills; b, the two main tracheæ.—After F. Müller.
It possesses five pairs of gills on the under side of the 2d to the 6th abdominal segments. Each gill has finger-shaped processes on its hinder edge, which are “from their constant motion evidently connected with respiration.” Tracheæ may be seen, according to H. J. Clark, entering the gills, and “the circulation of water among 474the branchiæ is kept up by the flapping of the tail-pieces.” The larva of Helichus fastigiatus is said by Leconte to be “very nearly allied, while the remotely allied Stenelmis crenatus has no external branchiæ.”[75]
The larva of the mosquito also has two modes of respiration, breathing either at the surface of the water through the two spiracles situated on the projection (siphon) at the hinder end of the body which is thrust out into the air; or when at the bottom respiring by tracheal gills. The pupa also has a double mode of respiration, either taking in air at the surface by the two thoracic horns with stigmatic openings, or when submerged using its tracheal gills.
Besides its long caudal tracheal air-tubes, the larval Eristalis is said by Chun to thrust out from the anus a number (20) of short tracheal filaments which float about in the water and serve to absorb the air.
An aquatic Brazilian larva of the family Psychodidæ has been found by Fritz Müller to take down under the water a large bubble of air (Fig. 455, C), the main tracheal trunk ending each in an opening at the end of the body (A, B); besides this, while at the bottom it breathes by three digitiform tracheal gills; another species having two pairs (C, a).
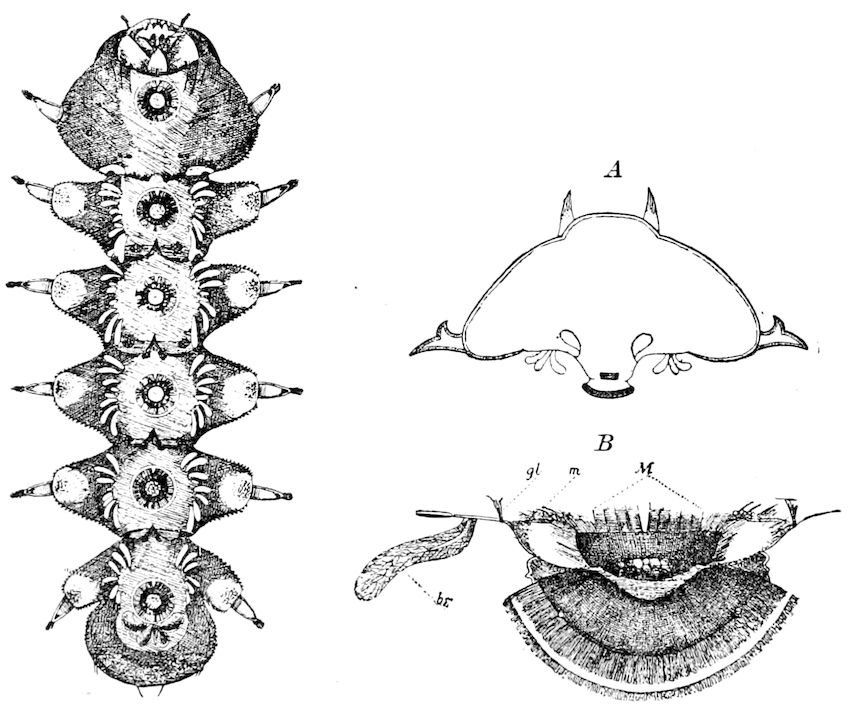
Fig. 456.—Under side of body of larva of Blepharocera. showing the position of the tracheal gills: A, section of the body through a sucker, showing position of the gills. B, section of a sucker: br, gill with numerous tracheæ; gl, outlet of excretory gland; M, m, muscles.—After F. Müller.
The remarkable larvæ of the Blepharoceridæ (represented in the United States by Blepharocera capitata), which live permanently in swift streams, attached by median suckers to stones, are apneustic, 475and breathe solely by leaf-like tracheal gills (Fig. 456, br) attached to the under side of the second to sixth abdominal segments. Those of the European Liponeura are said by Wierzejski to be branched, tree-like. Also immediately in front of the anus and behind the last sucker are four membranous sacs provided with tracheæ, but which are not capable of being withdrawn. These are said by Müller to be the same as what Dewitz states to serve as gills, and by Wierzejski they are homologized with the four anal gills of Chironomus.
The double mode of respiration in the larva of the horse bot-fly has been described by Scheiber. On the hinder end of the body are the stigmatic plates, which contain two lateral gill-plates and the middle stigmatal leaf. Besides this there is a pair of slightly developed prothoracic spiracles. The embryo and also freshly hatched larva of Gastrophilus equi do not possess these gill-plates, but on the end of the body are, according to Joli, two long thread-like gills. The freshly hatched larva of the allied Cephenomyia rufibarbis bears two caudal projections. (Kolbe.) As in shrimps and other Crustacea the gills are kept in constant motion, the water being driven over them by the rapid movements of the telson, so in the larval may-flies, and in the case-worm (Macronema), the gills move more or less rapidly. In case-worms as well as larval Perlidæ, Sialidæ, Paraponyx, and Hydrophilidæ the abdominal region is constantly moved to promote respiration. (Kolbe.)
Blood-gills.—Fritz Müller describes in trichopterous larvæ certain delicate anal tubular processes into which the blood flows, and which do not as a rule contain tracheæ, though occasionally very fine tracheal branches. Müller compares them with the gills of crabs and of shrimps. They are eversible finger-like tubules. They are used when the tracheal gills are temporarily not available. Their number varies even in the same genus. There are six in certain Rhyacophilidæ; five in different Hydropsychidæ; in Macronema there are four, and they are green when filled with the green blood of that insect, the tracheal gills being whitish. In the freshly hatched larva, while the tracheal gills are present, no anal blood-gills are visible. Similar blood-gills also occur in the pupæ of certain caddis-flies. (Pictet.)
Similar anal gills filled with blood occur in the larvæ of the fireflies (Lampyris, etc.), and perhaps, Kolbe thinks, serve for respiration, though other authors believe them to be adhesive organs.
The larva of Pelobius has true blood-gills. (Schiödte. See p. 461.)
The eversible ventral segmental sacs of Scolopendrella, Campodea, and Machilis, as well as the ventral tube (collophore) of Podura, Smynthurus, etc., may, as Oudemans and Haase have suggested, serve a respiratory purpose, though they lack tracheæ, and differ from blood-gills in containing no gases; yet the blood is forced into them, causing their eversion. Oudemans observed 476that Machilis everted its sacs when the vessel in which it was put was filled with warm, damp air. The sacs are only thrust out when the creature is completely at rest.
Structures referable to blood-gills also occur temporarily in the embryo of Orthoptera; Rathke observed them in the mole-cricket; Ayres observed them in Œcanthus niveus, where they form two stalked broad oval appendages on the first abdominal appendages, which he regarded as gills. Patten observed them in Phyllodromia germanica, as pear-shaped structures occurring in the same situation, but regarded them as sense-organs, as did Cholodkowsky. Graber found these structures in the embryo of the May-beetle, which looked like the other embryonic limbs, but survived after the disappearance of the latter, being longer and broader and unjointed. These disappeared shortly before birth. In Hydrophilus they remain, Graber states, after birth. Nussbaum has seen them in Meloë.
Finally, Wheeler has discussed at length these embryonic organs, which he regards as glandular structures, and calls pleuropodia, their primitive function having been that of limbs. He has detected them in the embryo of Periplaneta orientalis, Mantis carolina, Xiphidium ensiferum (Fig. 387); also in the Hemiptera (Cicada septemdecim, Zaitha fluminea), and in Sialis infumata. He discards the view that they were once gills or sense-organs, and concludes that they were glands. But, as we have suggested, their function once that of gills, and still respiratory in Synaptera, has perhaps become in the winged insects glandular and repugnatorial. Instead, then, of being modified abdominal limbs afterwards serving as glands, as Wheeler claims, we are inclined to believe that they functioned as blood-gills.
k. Tracheal gills of adult insects
Tracheal gills are known to be retained by a few insects in the imago stage, the nymphs in all stages breathing by them. The most notable example is the perlid genus Pteronarcys, in which, as Newport states, there are eight sets, comprising 13 pairs of branchial tufts distributed over the under surface of the thoracic and first two abdominal segments.
The first set, consisting of three pairs of tufts, partly encircling the neck like a ruff, arises from the soft membrane connecting the head and prosternum. The thoracic tufts originate between and behind the coxæ, as well as on the front margin of the meso- and metathoracic segments. The number of filaments in each tuft varies from about 20 to 50 or more, the densest tufts being those of the two hinder thoracic segments. Each filament is usually simple, though in a few cases they are branched (Fig. 457, A).
477The adult Pteronarcys is nocturnal, flying only at dewfall or in the night, and Mr. Barnston observed it when on the wing, “constantly dipping on the surface of the water”; by day it hides “in crevices of rocks which are constantly wetted by the spray of falling water, under stones and in other damp places.” It may thus be compared with the Amphibians, Necturus and Proteus, whose gills are retained in adult life. A similar large Chilian Perlid (Diamphipnoa lichenalis Gerst.) differs in completely lacking the thoracic gills, though there are four pairs on the abdomen, i.e. a pair on each of the first four segments. In this form the number of individual filaments in the largest tufts may amount to about 200.
Another Perlid (Dictyopteryx signata) is said by Hagen to have two pairs of gill-tufts on the under side of the head; the first pair situated on the base of the submentum, the second on the membrane connecting the head and prosternum.
Kolbe states that in the imagines of Perla marginata and P. cephalotes on the hinder edge of the thoracic stigmata arise three very small chitinous plates, which, on their under side and on the edges are beset with numerous short white filaments. These completely correspond to the filaments of the tuft-like larval gills. Persistent anal gills also occur in the imagines of Perla.
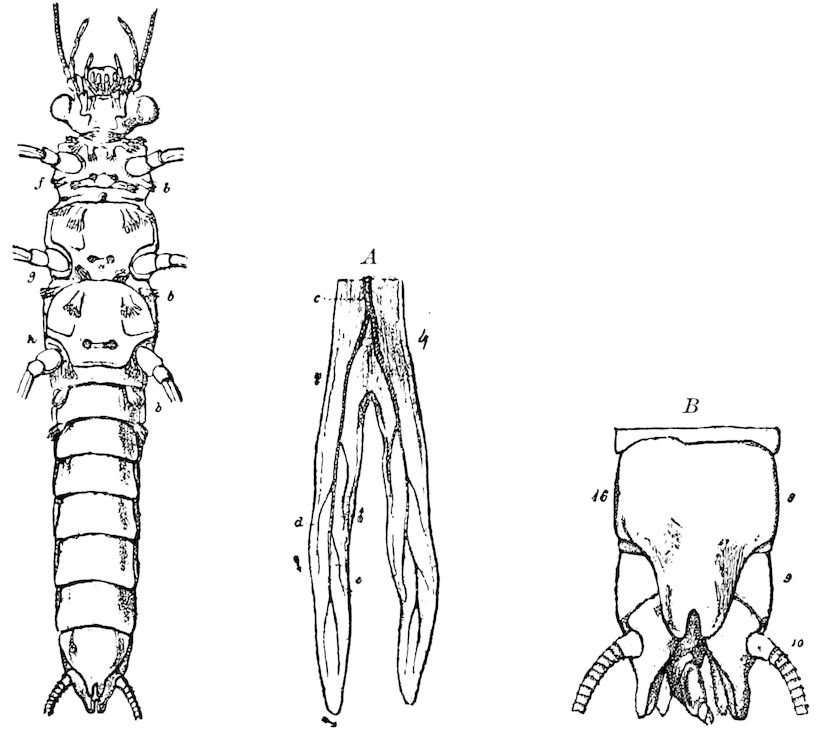
Fig. 457.—Under side of Pteronarcys regalis, showing the situation of the gills (g, b, f) and the sternal orifices: A, a branchial filament showing the direction of the current of blood; c, d, tracheæ. B, end of the abdomen enlarged.—After Newport.
In Nemoura lateralis and cinerea the tracheal gills are differently disposed. On each side of the anterior edge of the prosternum arise delicate tightly twisted filaments, like those of the larva. (Einführung, p. 536.)
Hagen also states that in the dragon-fly, Euphæa, the gills of the nymphs are retained in the imago, and Palmén remarks that in Æschna the rectal gills of the nymph persist in the imago, though not used for respiration.
Palmén gives an instance of a caddis-fly (Hydropsyche, Fig. 448) retaining its gills through the imago stage, but they are unfit for respiration, as they are minute and shrunken.
A walking-stick (Prisopus flabelliformis) found in the mountains of Brazil has the remarkable habit, according to Murray, of spending “the whole of the 478day under water, in a stream or rivulet, fixed firmly to a stone in the rapid part of the stream,” with its head turned up stream; but leaving the water at dark. The under side of the body, including the head, is hollowed so that the creature may adhere, sucker-like, to smooth stones; the claws, claspers, and flaps on the legs aid in retaining its hold, while the outer margin of the legs is dentate and thickly fringed with hair to repel the water.
Another form, closely related to Prisopus, from Borneo (Cotylosoma dipneusticum) is said by Wood-Mason to be even more profoundly modified for an aquatic life, since it has not only spiracles, but also, as he claims, tracheal gills. From each side of the body, in fact along the lower margins of the sides of the metathorax, there stand straight out five equal, small, but conspicuous ciliated oval plates, “which, when the insect is submerged and its stigmata are closed, doubtless serve for respiration.” The author did not note the actual presence of tracheæ in these plates.
LITERATURE ON THE ORGANS AND PHYSIOLOGY OF RESPIRATION
a. On the tracheal system in general
Lyonet, P. Traité anatomique de la chenille qui ronge le bois du saule. (La Haye, 1760; 2d edit., La Haye, 1762, pp. 616, 18 Pls.)
Treviranus, G. R. Beiträge zur Anatomie und Physiologie der Tiere und Pflanzen, 1816.
—— Das organische Leben. Bremen, 1831.
Rengger, J. R. Physiologische Untersuchungen über die tierische Haushaltung der Insekten. Tübingen, 1817. (Germar’s Mag. f. Ent., 1818, iii, pp. 410–413.)
Dufour, L. Recherches anatomiques sur les Carabiques et sur plusieurs autres insectes Coléoptères. Organes de la respiration. (Ann. Sc. nat., viii, 1826, pp. 19–27, 2 Pls.)
—— Études anatomiques et physiologiques sur les insectes Diptères de la famille des Pupipares. Appareil respiratoire. (Ann. Sc. nat. Zool., Sér. 3, iii, 1845, pp. 56–64, 1 Pl.)
—— Description et anatomie d’une larve à branchies externes d’Hydropsche. (Ann. Sc. nat, Zool., Sér 3, 1847, viii, pp. 341–354.)
—— Études anatomiques et physiologiques, et observations sur les larves des Libellules. Appareil respiratoire. (Ann. Sc. nat. Zool., Sér. 3, xvii, 1852, pp. 76–97, 3 Pls.)
—— Recherches anatomiques sur les Hyménoptères de la famille des Urocerates. Appareil respiratoire. (Ann. Sc. nat. Zool., Sér. 4, i, 1854, pp. 203–209, 1 Pl.; see also p. 344.)
Burmeister, H. Handbuch der Entomologie, i, 1832, pp. 169–194, 416–436.
Kirby, W., and W. Spence. Introduction to entomology, 1833, iv, pp. 35–81.
Bowerbank, J. S. Observations on the circulation of blood and the distribution of the tracheæ in the wing of Chrysopa perla. (Ent. Mag., iv, 1837, pp. 179–185.)
Platner, E. A. Mitteilungen über die Respirationsorgane in der Haut bei der Seidenraupe. (Müller’s Archiv f. Physiol., 1844, pp. 38–49.)
Filippi, F. de. Alcuni osservazioni anatomico-fisiologische sugl’ Insetti in generale, ed in particulare sul Bombice del Gelso. (Ann. R. Acad. d’ Agricoltura di Torino, 1850, ii, p. 25, 1 Pl.; Transl. by C. A. Dohrn, Stettin, Ent. Zeit., 1852, xiii, pp. 258–267; xiv, pp. 124–132, 1 Pl.)
479Newport, G. On the formation and the use of the air-sacs and dilated tracheæ in insects. (Trans. Linn. Soc. London, 1851, xx, pp. 419–423.)
Lubbock, J. Distribution of tracheæ in insects. (Trans. Linn. Soc. London, 1860, xxiii, pp. 23–50.)
Landois, L. Anatomie des Phthirius inguinalis Leach. (Zeitschr. f. wissens. Zool., xiv, 1864, pp. 1–26, 5 Taf.)
—— Anatomie des Pediculus vestimenti Nitzsch. (Ibid., xv, 1865, pp. 32–55, 3 Taf.)
—— Anatomie des Hundeflohs (Pulex canis). (Nova Acta Leop.-Carol. Akad. der Naturf., Dresden, 1866, xxxiii, 1867, pp. 67, 7 Taf.)
—— Anatomie der Bettwanze (Cimex lectularius L.). (Ibid., xviii, 1868, pp. 206–224, xix, 1869, pp. 206–233, 4 Taf.)
Meinert, Fr. Campodeæ: en familie af Thysanurernes orden. (Naturhistorisk Tidsskr., 3 Raek., iii, 1864–65, pp. 400–440, 1 Pl.)
Reinhardt, H. Zur Entwicklungsgeschichte des Tracheensystems der Hymenopteren mit besonderer Bezeichung auf dessen morphologische Bedeutung. (Berlin. Ent. Zeitschr., 1865, ix, pp. 187–218, 2 Taf.)
Gerstaecker, A. Bronn’s Klassen und Ordnungen des Tierreichs, v, 1866–1879. Organs of respiration, pp. 119–131.
Pouchet, G. Développement du système trachéen de l’Anophèle (Corethra plumicornis). (Archiv zool. expérimentale, i, 1872, pp. 217–232, 1 Fig.)
Graber, V. Ueber eine Art fibrilloiden Bindegewebes der Insektenhaut und seine lokale Bedeutung als Trachealsuspensorium. (Archiv f. Mikroskop. Anat. x, 1874, pp. 124–144, 1 Taf.)
—— Die Insekten; München, 1877. Organs of respiration, pp. 346–369.
Packard, A. S. On the distribution and primitive number of spiracles in insects. (Amer. Naturalist, viii, 1874, pp. 531–534.)
—— On the nature and origin of the so-called “spiral thread” of tracheæ. (Amer. Naturalist, xx, 1886, pp. 438–442, 2 Figs., p. 558.)
Wolff, O. J. B. Das Riechorgan der Biene nebst einer Beschreibung des Respirationswerkes der Hymenopteren, des Saugrüssels und Geschmacksorganes der Blumenwespen. (Nova Acta d. kais. Leop-Carol. Akad. der Naturf., xxxviii, 1876, pp. 1–251, 8 Taf.)
Palmén, J. A. Zur Morphologie des Tracheensystems. Leipzig, 1877, pp. 140, 2 Taf.
Moseley, H. N. Origin of tracheæ in Arthropoda. (Nature, xvii, 1878, p. 340.)
Poletajew, Olga. Quelques mots sur les organes respiratoires des larves des Odonates. (Horæ Soc. Ent. Ross., xv, 1880, pp. 436–452, 2 Pls.)
Viallanes, H. Sur l’appareil respiratoire de quelques larves de Diptères. (Compt. rend. Acad. Sc., Paris, 1880, pp. 1180–1182.)
MacLeod, J. La structure des trachées et la circulation peritrachéenne. Bruxelles, 1880, pp. 70, 4 Pls.
Hagen, H. A. Beitrag zur Kenntnis des Tracheensystems der Libellenlarven. (Zool. Anzeiger, 1880, pp. 157–162.)
—— Einwürfe gegen Palmens Ansicht von der Entstehung des geschlossenen Tracheensystems. (Ibid., 1881, pp. 404–406.)
Macloskie, G. The structure of the tracheæ of insects. (Amer. Naturalist, 1884, xviii, pp. 567–573, Fig.)
Haase, E. Das Respirationssystem der Chilopoden und Symphylen (Scolopendrellen) vergleichen mit dem der Hexapoden. (Zeitschr. f. Ent. N. F., ix, Breslau, 1884.)
—— Das Respirationssystem der Symphylen und Chilopoden. (Zool. Beiträge, von A. Schneider, i, 1884, pp. 65–95, 3 Taf.; Zool. Anzeiger, 1883, pp. 15–17.)
480Grassi, B. I progenitori degli Insetti e dei Miriapodi. L’Japyx e la Campodea. (Atti d. Accad. Gioenia d. Sc. Nat. Catania, 1885, Sér. 3, xix, pp. 83, 5 Pls.)
—— I progenitori dei Miriapodi e degli Insetti. Anatomia comparata dei Tisanuri. (Reale Accad. d. Lincei di Roma, Anno 284, 1887.)
Meinert, Fr. De eucephale Myggelarver. Sur les larves eucephales des Diptères. (Vidensk. Selsk. Skrifter., 6 Raekke, naturvid. og mathem. Afd. Kjöbenhavn, 1886, iv, pp. 369–493, 4 Pls.)
Cajal, S. R. Coloration par la méthode de Golgi des terminaisons des trachées et des nerfs dans les muscles des ailes des insectes. (Zeitschr. f. wiss. Microscopie, 1890, vii, pp. 332–342, 1 Pl.)
Wistinghausen, C. v. Ueber Tracheenendigungen in den Sericterien der Raupen. (Zeitschr. wissensch. Zool., xlix, 1890, pp. 565–582, 1 Taf.)
Stokes, Alfred C. The structure of insect tracheæ, etc. (Science, 1893, pp. 44–46, 7 Figs.)
Sadones, J. L’appareil digestif et respiratoire larvaire des Odonates. (La Cellule, xi, 1895, pp. 271–325, 3 Pls.)
Holmgren, Emil. Über das respiratorische Epithel der Tracheen bei Raupen. (Festschrift Lilljeborg. Upsala, 1896, pp. 76–79, 2 Taf.) See also p. 437.
Also Gegenbaur’s Comparative Anatomy, Engl. Trans.
b. On the Stigmata
Loewe, C. L. W. De partibus quibus insecta spiritus ducunt. Diss. inaug. Halæ, 1814, pp. 28.
Sprengel, C. Commentarius de partibus quibus Insecta spiritus ducunt. Lipsiæ, 1815, pp. 38, 3 Taf.
Dufour, L. Recherches anatomiques sur l’Hippobosque des chevaux. (Ann. Sc. nat., 1825, vi, pp. 299–322, 1 Pl.)
—— Nouvelles observations sur la situation des stigmates thoraciques dans les larves des Bupresticides. (Ann. Soc. Ent. France, Sér. 2, 1844, ii, p. 203.)
Landois, H. Der Tracheenverschluss bei Tenebrio molitor. (Reichert u. Dubois-Reymond’s Archiv f. Anat., 1866, pp. 391–397, 1 Taf.)
—— und W. Thelen. Der Tracheenverschluss bei den Insekten. (Zeitschr. wissensch. Zool., xvii, 1867, pp. 187–214, 1 Taf.)
—— Der Stigmenverschluss bei den Lepidopteren. (Reichert u. Dubois-Reymond’s Archiv f. Anat., 1886, pp. 41–49, 1 Taf.)
Hagen, H. A. Beitrag zur Kenntnis des Tracheensystems der Libellen-Larven. (Zool. Anzeiger, 1880, pp. 157–162.)
—— Einwürfe gegen Palmén’s Ansicht von der Entstehung des geschlossenen Tracheensystems. (Ibid., 1881, pp. 404–406.)
Krancher, O. Der Bau der Stigmen bei den Insekten. (Zeitschr. wissensch. Zool., xxxv, 1881, pp. 505–574, 2 Taf.; Zool. Anz., 1880, pp. 584–588.)
Meinert, Fr. Spirakelpladen hos Scarabæ-Larverne. (Vid. Meddel. Nat. For. Kjöbenhavn (4), Aarg. iii, 1882, pp. 289–292.)
—— Noget mere om Spiracula cribraria og Os clausum. En Replik. (Ibid. (4), Aarg. v, 1884, pp. 68–91, Fig.)
Schiödte, J. G. Spiracula cribraria—os clausum: lidt om naturvidenskabelig Methode og Kritik. (Nat. Tidsskrift (3), xiii, 1883, pp. 427–473; also Jahresber. Neapel, 1883, p. 105.)
Verson, E. Il meccanismo di chiusura negli stimmati di Bombix mori. (Atti Istit. Veneto. Sc., 1887, p. 9, Pl.)
—— Der Bau der Stigmen von Bombyx mori. (Zool. Anzeiger, 1887, x Jahrg., pp. 561, 562.) See also Zool. Anzeiger, 1890, p. 116.
481Haase, E. Die Stigmen der Scolopendriden. (Zool. Anzeiger, 1887, x Jahrg., pp. 140–142.)
—— Holopneustie bei Käfern. (Biolog. Centralbl., 1887, vii, pp. 50–53.)
Carlet, G. Note sur un nouveau mode de fermeture des trachées, “fermeture operculaire” chez les insectes. (Comp. rend. Acad. Sci. Paris, 1888, cvii, pp. 755–757.)
De Meijere, J. C. H. Über zusammengesetzte Stigmen bei Dipterenlarven [etc.]. (Tijd. Ent., xxxviii, 1895, pp. 65–100, 33 Figs.)
Also the other writings of Palmén, Dufour, Dewitz, Boas, Verson.
c. On tracheal gills and tracheal respiration
Pictet, F. J. Mémoires sur les larves des Némoures. (Annal. Sc. nat., 1832, xxvi, pp. 369–391, 2 Pls.)
—— Recherches pour servir à l’histoire et à l’anatomie des Phryganides. Genève, pp. 235, 20 Pls.
—— Histoire naturelle générale et particulière, des insectes Neuroptères. I, Monographie: Famille des Perlides, Genève, 1841, 1842, pp. 423, 53 Pls.
—— Histoire naturelle générale et particulière des insectes Neuroptères. II, Monographie: Famille des Ephémérines. Genève, 1843–1845, pp. 300, 47 Pls.
Dufour, L. Recherches anatomiques et considerations entomologiques sur les insectes Coléoptères des genres Macronychus et Elmis. (Ann. Sc. nat. Zool., Sér. 2, 1835, iii, pp. 151–174, 1 Pl.)
—— Description et anatomie d’une larve à branchies externes d’Hydropsyche. (Ibid., Sér. 3, 1847, viii, pp. 341–354, Fig.)
—— Recherches anatomiques sur la larve à branchies extérieures du Sialis lutarius. (Ibid., Sér. 3, 1848, ix, pp. 91–99, Fig.)
—— De diverses modes de respiration aquatique chez les insectes. (Compt. rend. Acad. d. Sc. Paris, 1849, xxix, pp. 763–770; Ann. and Mag. Nat. Hist., Sér. 2, 1850, vi, pp. 112–118.)
—— Études sur la larve du Potamophilus. (Ann. Sc. nat., Sér. 4, xvii, 1862, pp. 162–173, 1 Pl., Bericht v. Gerstaecker f. 1862, pp. 16, 17.)
Grube, A. E. Beschreibung einer auffallenden an Süsswasser-schwammen lebenden Larve (Sisyra). (Wiegmanns Archiv f. Naturgesch., 1843, ix, pp. 331–337, Fig.)
Schröder van der Kolk, J. L. G. Mémoire sur l’anatomie et physiologie de Gastrus equi. (Nieuwe Verhandl. d. K. Nederl. Instit. Amsterdam, 1845, ix, pp. 1–155, 13 Pls.; Erichson’s Bericht. f. 1845, p. 109.)
Scheiber, S. H. Vergleichende Anatomie und Physiologie der Œstriden-Larven. Respirationssystem. (Sitzungsber. Akad. Wissensch. Wien. Math.-naturw. Cl., 1862, xlv, pp. 7–39.)
Lubbock, J. On the development of Chloëon dimidiatum. (Trans. Linn. Soc. London, I, 1868, xxiv, pp. 61–78, 2 Pls.; II, 1866, xxv, pp. 477–492.)
Oustalet, E. Note sur la respiration chez les nymphes des Libellules. (Ann. Sc. nat. Zool., Sér. 5, xi, 1869, pp. 370–386, 3 Pls.)
Rolph, W. H. Beitrag zur Kenntnis eininger Insektenlarven. 1 Taf. Inaug. Dissertat. Bonn, 1873.
Chun, C. Ueber den Bau, die Entwicklung und physiologische Bedeutung der Rektaldrüsen bei den Insekten. Frankfurt a. M., 1875.
Haller, G. Die Stechmückenlarve. Kleinere Bruchstücke zur vergleichenden Anatomie der Arthropoden. I. Ueber das Atmungsorgan der Stechmückenlarven. (Archiv f. Naturgesch., xliv, 1878, pp. 91–96, 1 Taf.)
Vayssière, A. Recherches sur l’organisation des larves des Ephémérines. (Ann. d. Sc. nat. Zool., Sér. 6, xiii, 1882, pp. 1–137, 11 Pls.)
482—— Monographie zoologique et anatomique du genre Prosopistoma Latr. (Ibid., Sér. 7, ix, 1890, pp. 19–87, 4 Pls.)
Eaton, A. E. Notes on some species of Cloëon. (Ann. Mag. Nat. Hist, Ser. 3, xviii, 1866, pp. 145–148.)
—— A revisional monograph of recent Ephemeridæ or may-flies. (Trans. Linn. Soc. London, 1883–1887, Ser. 2, iii, 63 Pls.)
Müller, Wilh. Ueber einige im Wasser lebende Schmetterlingsraupen Brasiliens. (Archiv f. Naturgesch., 1884, i Jahrg., pp. 194–212, 1 Taf.)
Vogler. Die Tracheenkiemen der Simulien-Puppen. (Mitteil. Schweiz Ent. Gesellsch., 1887, vii, pp. 277–282.)
Raschke, E. W. Die Larve von Culex nemorosus. Ein Beitrag zur Kenntnis der Insekten-Anatomie und Histiologie. (Archiv für Naturgesch., 1887, liii Jahrg., pp. 133–163, 2 Taf.; Zool. Anz., 1887, x Jahrg., pp. 18, 19.)
Klapálek, Fr. Untersuchungen über die Fauna der Gewässer Böhmens. I. Metamorphose der Trichopteren. (Archiv f. naturwissensch. Landesdurchforschung von Böhmen, Prag, 1888, vi, No. 5, pp. 63; No. 6, 1893, pp. 145, Figs.)
Müller, Fritz. Larven von Mücken und Haarflüglern mit zweierlei abwechselnd thätigen Atemwerkzeugen. (Ent. Nachr., 1888, xiv Jahrg., pp. 273–277; also Zool. Anzeiger, iv, 1881, pp. 499–502.)
Kolbe, H. J. Ueber den Kranzförmigen Laich einer Phryganea. (Sitzungsber. d. Gesellsch. naturforsch. Freunde in Berlin, 1888, pp. 22–26.)
Haase, Erich. Die Abdominalanhänge der Insekten mit Berücksichtigung der Myriopoden. (Morpholog. Jahrbuch, 1889, xv, pp. 331–435, 2 Taf.)
Dewitz, H. Einiger Beobachtungen, betreffend das geschlossene Tracheensystem bei Insectenlarven. (Zool. Anzeiger, xiii, 1890, pp. 500–504, 525–531.)
Miall, L. C. Some difficulties in the life of aquatic insects. (Nature, xliv, London, 1891, pp. 456–462.)
—— Natural History of aquatic insects, 1895, 116 Figs., pp. 1–395.
Weltner, W. (Note on Sisyra.) (Ent. Nachr., p. 145, 1894.)
Also papers by Hagen, Dewitz, Williams, Tömösváry (1884).
d. Literature on rectal respiration
Suckow, F. W. L. Respiration der Insekten, insbesondere über die Darmrespiration der Æschna grandis. (Zeitschrift f. d. organ. Physik, von Heusinger, 1828, ii, pp. 24–29, 4 Taf.)
Dufour, L. Sur la respiration branchiale des larves des grandes Libellules comparée à celle des poissons. (Compt. rend. de l’Acad. Sc. Paris, 1848, xxvi, pp. 301–303.)
—— Études anatomiques et physiologiques et observations sur les larves des Libellules. (Ann. Sc. nat. Zool., Sér. 3, 1852, xvii, pp. 76–97, 3 Pls.)
Gilson, G. and J. Sadones. Larval gills of Odonates. (Journ. Linn. Soc. London, 1897.)
e. Physiology of Respiration
Bonnet, Ch. Recherches sur la respiration des chenilles. (Mém. Math. des Savants Étrangers, Paris, 1768, v, pp. 276–303.)
Treviranus, G. R. Biologie, oder Philosophie der lebenden Natur, für Naturforscher und Aerzte. 6 Bände, Göttingen, 1802–1822. (Atmung, in Bd. iv.)
—— Die Erscheinungen und Gesetze des organischen Lebens. 2 Bände, Bremen, 1831–1833. (Atmung der Insekten in Bd. i.)
483—— Versuche über das Atemholen der niederen Tiere. (Zeitschrift f. d. Physiologie, von F. Tiedemann, G. R. u. L. C. Treviranus, 1832, iv, pp. 1–39.)
Hausmann, J. F. L. De animalium exsanguinum respiratione commentatio. Hannover, 1803, vi, p. 70.
Spallanzani, L. Memoirs on respiration. London, 1804.
Sorg, F. L. A. W. Disquisitiones physiologicæ circa respirationem insectorum et vermium. Rudolstadt, 1805, Part II, p. 146.
Nitzsch, C. L. Commentatio de respiratione animalium. Vitebergæ, 1808, 4º, pp. 56.
—— Ueber das Atmen der Hydrophilen. (Reil’s Archiv f. Physiologie, 1811, x, pp. 440–458.)
Reimarus, J. A. H. Ueber das Atmen, besonders über das Atmen der Vögel und Insekten. (Reil u. Autenrieth, Archiv f. Physiologie, 1812, xi, pp. 229–236.)
Dufour, L. Anatomie de la Ranatre linéaire et de la Nèpe cendrée. (Annal. génér. Scienc. phys., Bruxelles, 1821, vii, pp. 194–213, 1 Pl.)
—— Mémoire pour servir à l’histoire du genre Ocyptera. (Annal. Scienc. natur., 1827, x, pp. 248–261, 1 Pl.)
—— Recherches sur quelques entozoaires et larves parasites des insectes Orthoptères et Hyménoptères. (Ann. Sc. nat. Zool., Sér. 2, 1836, vi, p. 55; Sér. 2, 1837, vii, pp. 5–20.)
—— Note sur le parasitisme. (Compt. rend. Acad. Sc. Paris, 1851, xxxiii, pp. 135–139; Rev. et Mag. de Zool., 1851, pp. 408–412.)
Dutrochet, R. J. H. Du mecanisme de la respiration des Insectes. (Ann. Sc. nat., 1833, xxviii, pp. 31–44; Mém. Acad. Sc. Paris, 1838, xiv, pp. 81–93.)
Newport, G. On the respiration of insects. (Phil. Trans. Roy. Soc., London, 1836, cxxvi, pp. 529–566.)
Coquerel, Ch. Note pour servir à l’histoire de l’Æpus robini. (Ann. Soc. Ent. France, Sér. 2, 1850, viii, pp. 529–532.)
Davy, J. On the effects of certain agents on insects. (Trans. Ent. Soc. London, 1851, pp. 195–212.)
Barlow, W. F. Observations of the respiratory movements of insects. (Phil. Trans. Roy. Soc. London, cxlv, 1855, pp. 139–148.)
Rathke, H. Anatomisch-physiologische Untersuchungen über den Atmungsprozess der Insekten. (Schriften d. k. phys.-ökon. Ges. Königsberg, i Jahrg., 1860, pp. 99–138, 1 Taf.)
Lubbock, J. On two aquatic Hymenoptera, one of which uses its wings in swimming. (Trans. Linn. Soc. London, xxiv, 1863, pp. 135–142, 1 Pl.)
Boyle, R. New pneumatical experiments about respiration. (Phil. Trans., 1870, v, No. 63, pp. 2051–2056.)
Lambrecht. A. Das Atmungsgeschäft der Bienen. (Bienenwirtschaftl. Centralblatt, vii Jahrg., 1871, pp. 20–25.)
—— Luftverbrauch eines Biens und die damit zusammenhängenden Lebensprozesse der Glieder desselben. (Ibid., 1871, pp. 115–120.)
Monnier. Sur la rôle des organes respiratoires chez les larves aquatiques. (Compt. rend. Acad. Sc. Paris, lxxiv, 1872, p. 235.)
Liebe, Otto. Ueber die Respiration der Tracheaten, besonders über den Mechanismus derselben und über die Menge der ausgeatmenten Kohlensäure. (Inaug. Diss. Chemnitz, 1872, pp. 28.)
Plateau, F. Recherches physico-chimiques sur les articulés aquatiques. (Bull. Acad. Roy. Belg., Sér. 2, xxxiv, 1872, pp. 271–321.)
484—— Recherches expérimentales sur les mouvements respiratoires des Insectes. (Mem. Acad. Belg., 1884, xlv, pp. 219, 7 Pls., 56 Figs.)
—— Recherches physico-chimiques sur les articulés aquatiques. Part I. Action de sels en dissolution dans l’eau. Influence de l’eau de mer sur les articulés aquatiques d’eau douce. Influence de l’eau douce sur les Crustacés marines. (Mém. cour. et Mém. des savants étrang. de Belgique, xxxvi, 1871, pp. 68.) Part II. Résistance à l’asphyxie par submersion, action du froid, action de la chaleur, temperature maximum. (Bull. Acad. Roy. de Belgique, Sér. 2, xxxiv, 1872, pp. 271–321.)
—— Les Myriopodes marins et la résistance des Arthropodes à respiration aërienne à la submersion. (Journ. de l’anatomie et de la physiologie, 1890, xxvi, pp. 236–269.)
Bütschli, O. Ein Beitrag zur Kenntnis des Stoffwechsels, insbesondere die Respiration bei den Insekten. (Reichert und Dubois-Reymond’s Archiv f. Anatomie, 1874, pp. 348–361.)
Ritsema Cz., C. Acentropus niveus Oliv., in Zijne levenswijze en verschillende toestanden. (Tijdschr. voor Entom, 1876, xxi, separate, pp. 34, 2 Taf.)
Pott, Rob. Chemical experiments on the respiration of insects. (Psyche, ii, 1878.)
Sharp, D. Observations on the respiratory action of the carnivorous water-beetles. (Journ. Linn. Soc. London, xiii, Zoology, 1878, pp. 161–183.)
Krancher, O. Das Atmen der Biene. (Deutscher Bienenfreund., xvi Jahrg., 1880, pp. 49–51.)
Gissler, C. F. Sub-elytral air-passages in Coleoptera. (Proc. Amer. Assoc. Advanc. Sc. 29 Meet. (1881), 1881, pp. 667–669.)
Langendorff, O. Studien über die Innervation der Atembewegungen. 6. Das Atmungszentrum der Insekten. (Archiv f. Anatomie u. Physiol., Physiol. Abteil., 1883, pp. 80–87.)
Macloskie, G. Pneumatic functions of insects. (Psyche, iii, 1883, pp. 375.)
Chalande, J. Recherches sur le mecanisme de la respiration chez les Myriopodes. (Compt. rend. Acad. Sc. Paris, civ, 1887, pp. 126, 127.)
Comstock, J. H. Note on respiration of aquatic bugs. (Amer. Naturalist, 1887, xxi, pp. 577, 578.)
Fricken, W. v. Ueber Entwicklung, Atmung und Lebensweise der Gattung Hydrophilus. (Tagebl., 60, Versamml. deutscher Naturf. u. Aerzte, 1887, pp. 114, 115.)
Schmidt, E. Ueber Atmung der Larven und Puppen von Donacia crassipes. (Berlin. Ent. Zeitschr., 1887, xxxi Jahrg., pp. 325–334, 1 Taf.)
Dewitz, H. Entnehmen die Larven der Donacien vermittelst Stigmen oder Atemrohren den Luftraumen der Pflanzen die sauerstoffhaltige Luft? (Ibid., 1888, xxxii Jahrg., pp. 5, 6, Fig.)
Müller, G. W. Ueber Agriotypus armatus. (Spengel’s Zoolog. Jahrbücher. Abt. f. Systematik, etc., iv, 1889, pp. 1132–1134.)
—— Noch einmal Agriotypus armatus. (Ibid., v, 1890, pp. 689–691.)
Devaux, H. Vom Ersticken durch Ertrinken bei den Tieren und Pflanzen. (Naturwiss. Rundschau, vi Jahrg., 1891, p. 231; Compt. rend. Soc. de Biol., 1891, Ser. 9, iii, p. 43.)
See also Dewitz, p. 482; Kolbe, p. 482.
THE ORGANS OF REPRODUCTION
Insects are without exception unisexual, the male and female organs existing in different individuals, no insects being normally hermaphroditic. The reproductive organs are situated in the hind-body or abdomen, especially near the end, the genital glands opening externally either in the space between the 7th and 8th, or 8th and 9th, or 9th and 10th abdominal segments, but as a rule between the 8th and 9th segments (Fig. 299).
The primary or essential male organs are the testes, those of the female being the ovaries. As we shall see, the primitive number of seminal ducts and oviducts was two, this number being still retained in Lepisma and the Ephemeridæ. The reproductive organs of both sexes are at first, in their embryonic condition, of the same shape and structure, becoming differentiated in form and function before sexual maturity. These glands and ducts have a paired mesodermal genital rudiment, the ends of the ducts being often connected with corresponding ectodermal invaginations of the cuticle.
The secondary sexual organs mainly comprise the external genital armature of the male, and the egg-laying organs, or ovipositor of the female. Besides these structures there are other more superficial secondary sexual characters, such as differences in the size and ornamentation as well as coloring of the body, or of parts of it.
The primary sexual organs of insects have been conveniently tabulated by Kolbe, thus:—
I. Male reproductive organs.
II. Female reproductive organs.
The ducts of the sexual glands in Peripatus being transformed nephridia or segmental organs, it has been inferred that this is also the case with those of insects, though, as Lang states, there is a considerable difference in the two cases, as the greater part of the ducts in Peripatus arises out of the ectoderm, while in the Myriopoda and insects they come from the mesoderm; but he adds that in the Annelids the greater part of the nephridial duct is of mesodermal origin.
486While in insects there is but a single pair of genital outlets, the serial arrangement of the testicular (Fig. 458) and egg-tubes (Fig. 459) in some Thysanura (Campodea, Japyx, and Lepisma), where the tubes (5 to 7 on each side) open singly one behind the other in segmental succession, indicates that in their ancestors these egg-tubes opened out on different segments situated one behind the other. Each egg-tube independently opens into one of the two oviducts, which extend through the abdomen as straight canals. The two oviducts open externally by a short unpaired terminal portion, which in Machilis is said to be wanting, only the outer aperture of the two oviducts being in this case common to both. In Campodea and in the Collembola the ovaries and testes on each side are simply tubes. It is to be observed that in the young Lepisma Nassonow found that the external openings of the two ejaculatory ducts are paired (Fig. 458 B, ed.).
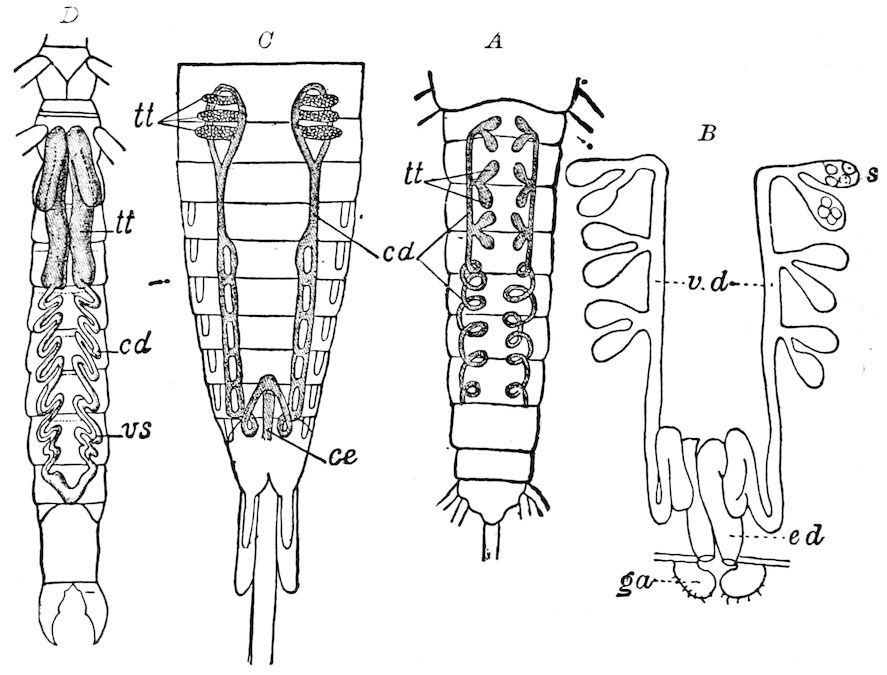
Fig. 458.—Male genital organs of Thysanura: A, Lepisma in which the testes are segmentally arranged.—After Grassi. B, Lepisma saccharina, young ♂: vd, vas deferens; ed, ejaculatory duct; ga, external appendages,—After Nassonow. C, Machilis, the testis lateral and separate, but not corresponding to the segments. D, Japyx, with an undivided testicular tube on each side; tt, testes; cd, vas deferens; vs, seminal vesicle; ce, ejaculatory duct.—After Grassi, from Perrier.
In the Stylopidæ, also, though this may be the result of adaptation to the singular parasitic habits of the females whose bodies are mostly situated in the abdomen of their host, the ends of the oviducts are formed by the invagination 487of the integument of the 2d, 3d, and 4th abdominal segments. In the 2d to 5th segments are situated tubes which open in the cavity of the body with funnel-like ends, so that the ducts have a close resemblance to the segmental organs of worms. (Nassonow.)
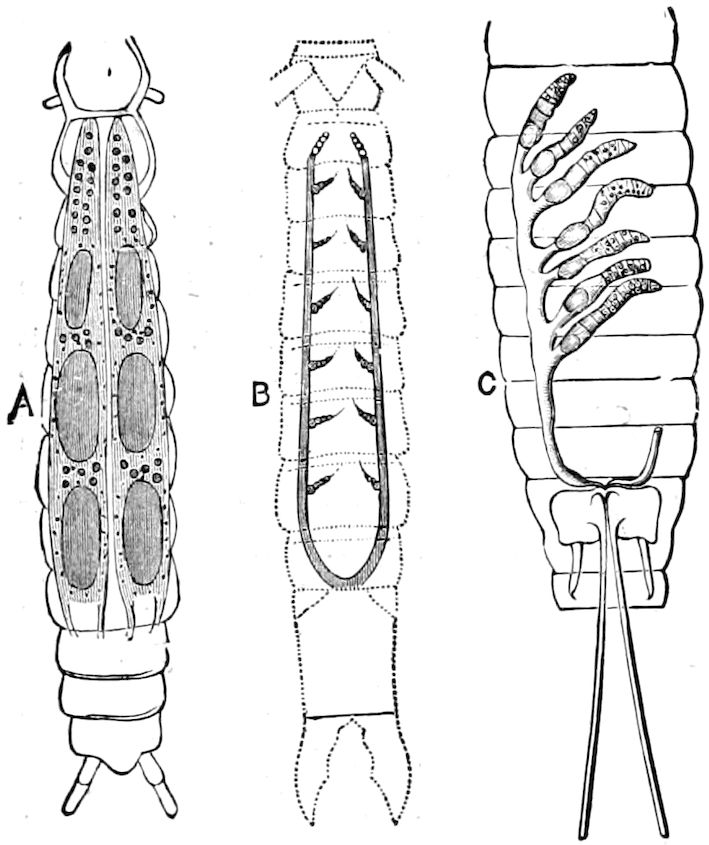
Fig. 459.—Ovaries of Thysanura: A, of Campodea. B, of Japyx.—After Grassi. C, of Machilis.—After Oudemans, from Sharp.
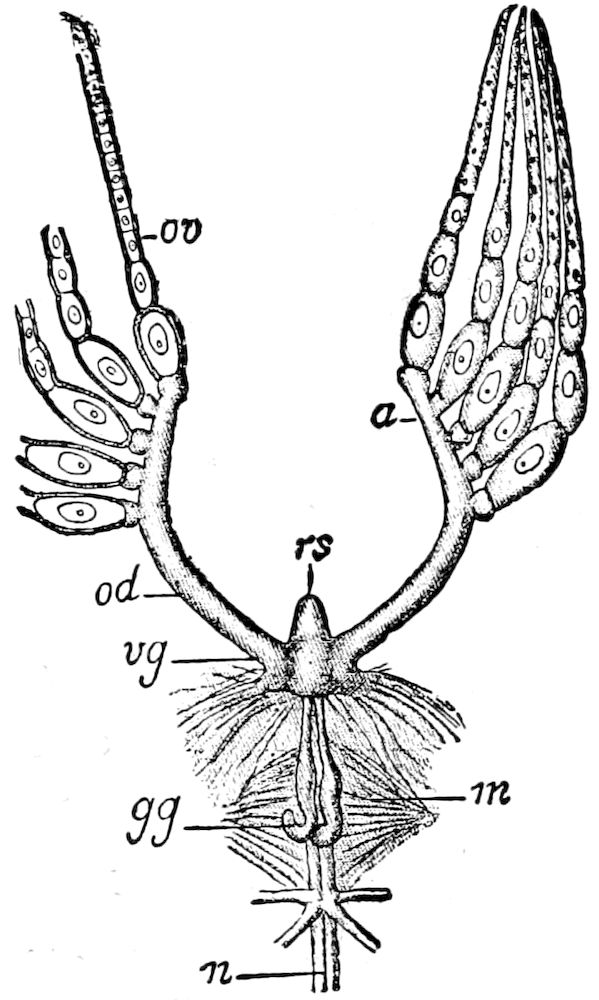
Fig. 460.—Female genital organs of Lepisma saccharina, adult: ov, ovaries; a, part of the oviduct, corresponding to the calyx of winged insects; od, oviduct; vg, vagina; rs, copulatory pouch; gg, accessory glands; m, muscles; n, nervous cord.—After Nassonow, from Perrier.
Among the winged insects the reproductive organs of the cricket (Fig. 466) are perhaps as simple as any. The testes are separate, and the vasa deferentia very long. The seminal vesicles bear numerous large and short utricles (utriculi majores and breviores), the penis being simple and dilated at the end; while in Phyllodromia germanica the testes are functional throughout life, and consist of four lobes each. In the common cockroach (P. orientalis) (Fig. 461) the testes are functional only in the young male; they afterwards shrivel and are functionally replaced by the vesiculæ seminales and their appendages, when the later transformations of the sperm-cells are effected. The accessory glands are numerous and differ both in function and insertion. Two sets of these glands (utriculi majores and breviores) are attached to the vesiculæ seminales and the fore end of the ejaculatory duct, while another appendage, called by Miall and Denny the conglobate gland, opens separately on the exterior upon a double hook, which forms a 488part of the external genital armature. The so-called penis is long, slender, and dilated at the end, but is not perforated.
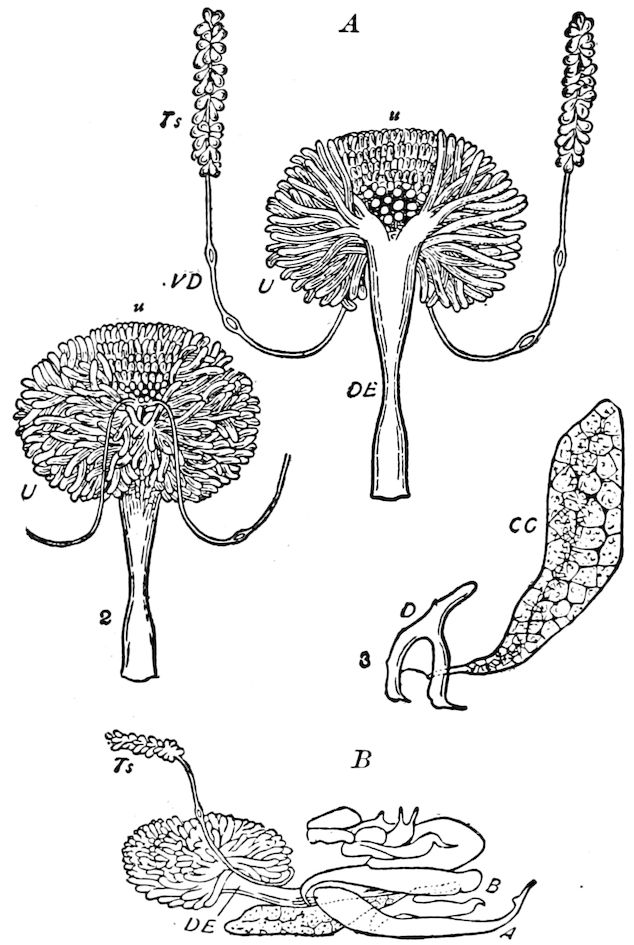
Fig. 461.—A, male organs of the cockroach, ventral view: Ts, testis; VD, vas deferens; DE, ductus ejaculatorius; U, utriculi majores; u, utriculi breviores; 2, dorsal view; 3, CG, conglobate gland and its duct. B, male organs, side view: A, titillator; B, penis; other letters as in A.—After Miall and Denny.
In the locusts (Acrydiidæ) the testes are, unlike those of most other Orthoptera, closely united to each other so as to form a single mass of tubular glands into which penetrate both simple and dilated tracheæ; the entire mass is situated in the 3d, 4th, and 5th abdominal segments, and above the intestine. The anterior end of the testicular mass is rounded and held in place by a broad, thin band, one on each side; two similar bands are situated a little behind the middle of the mass. From the under side, and a little in advance of the middle of the mass, two straight small ducts, as long as the testicular mass, pass obliquely to the sides of the body, at the posterior end of the 7th segment of the abdomen; these are the vasa deferentia. Each vas deferens, with its mate, forms a convoluted mass of tubes, comprising twenty folded bundles (epididymis of Dufour), and two single, long, convoluted tubes, the vesiculæ seminales, which are lobed in the 6th and 7th segments of the abdomen. The two vesiculæ unite over the 5th abdominal ganglion, forming a thick, very short canal (ductus ejaculatorius), which passes into a large spherical muscular mass (præputium), behind which is the large intromittent organ (penis), which forms a short chitinous cylinder, quite complicated in structure, 489being armed with hooks and projections and affording excellent specific characters. It can be seen in place without dissection by drawing back the orbicular convex piece called the velum penis.
In the Hymenoptera the reproductive system is quite simple, as seen in Fig. 462.
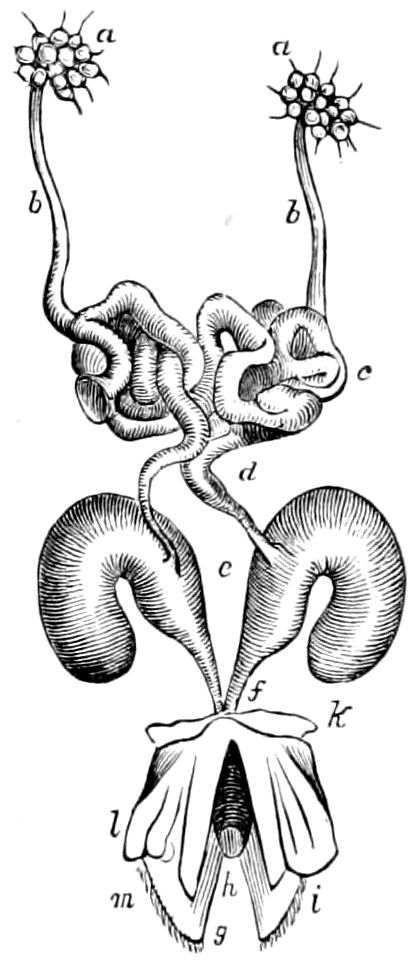
Fig. 462.—Male organs of saw-fly (Athalia centifoliæ): a, a, testes; b, b, epididymis; c, d, vas deferentia; e, vesiculæ seminales; f, ductus ejaculatorius; h, penis (see also p. 180).—After Newport.
The general shape and relations of the female reproductive organs are seen in Fig. 298, of the locust (Acrydiidæ). The ovaries consist of two large bundles of tubes, each bundle tied to the other by slight bands, with air-sacs and tracheæ ramifying among them. These tubes extend along the intestine, passing into the prothorax. The ovarian tubes opening into the oviducts unite to form the vagina, which lies on the floor of the abdomen. (In the cockroach the vagina has a muscular wall and chitinous lining.) Above the opening of the duct, and directly communicating with it, is the copulatory pouch (bursa copulatrix), a capacious pocket lined within with several narrow, longitudinal, chitinous bands. Behind the bursa copulatrix lies, partly resting under the fifth abdominal ganglion, the sebific, cement, or colleterial gland (colleterium; compare Fig. 299, sb), which is flattened, pear-shaped, a little over half as long as a ripe egg of the same insect. From the under side, a little in advance of the middle, arises the sebific duct, which, after making three tight coils next to the ganglion, passes back and empties into the upper side of the bursa copulatrix, dilating slightly before its junction with the latter.
The most primitive type of reproductive organs observed in insects is that of the young Lepisma and the Ephemeridæ, in which the outlets of the oviducts and of the vasa deferentia respectively are double or paired, showing that insects have probably inherited these structures from the segmental organs of their vermian ancestors.
Réaumur had already observed the process of oviposition and seen that the female Ephemera had two openings near the end of the “6th” abdominal segment, from which he saw two masses of eggs pass out at a time (Fig. 463). Eaton afterwards (1871) referred to the oviducts as terminating between the 7th and 8th segments of the abdomen, and after him Joly; but for a detailed monograph on the subject we are indebted to Palmén. He found that the outlets of 490the sexual glands are paired, not only in the larvæ of all stages, but also in the imagines, and in both sexes. In the males the vasa deferentia pass on the ventral side of the 9th segment through two external appendages, both reproductive organs, at whose tips or sides the openings are situated. In the larvæ the female openings are not formed until after the last moult. In the females the two oviducts open on the ventral side of the hind-body between the 7th and 8th segments.
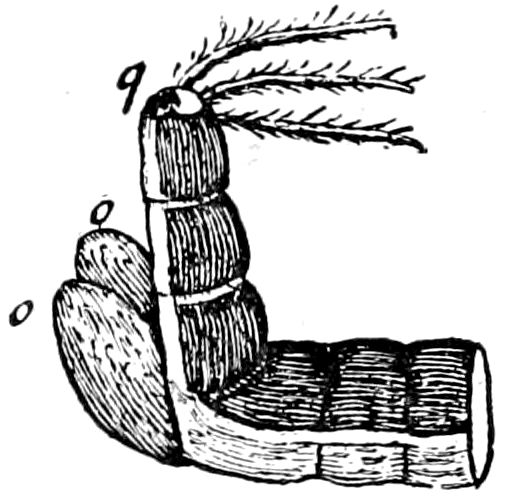
Fig. 463.—Upturned end of body of Ephemera, with two egg-masses (o) issuing at the same time from the double oviducts; q, anus.—After Réaumur.
Palmén suggests that the Ephemerids represent, in respect to the reproductive system among insects, a very primitive type of organization, and he concludes that the inner sexual organs of insects are built up of two different morphological elements; i.e. (a) internal primitive paired structures (testes with vasa deferentia, ovaria with oviducts), and (b) integumental structures, such as the ductus ejaculatorius and vagina.
In the younger larvæ the vasa deferentia form slender cords along which are situated the seminal glands; these cords are inserted in the integument on the hinder edge of the 9th sternite, where afterwards, during the last moult, the copulatory organs grow out. In the older larvæ the sperm collects in the cavities of these cords. Their walls become expanded, and this section then functions as vesiculæ seminales. The ends of the cords remain contracted and act as ductus ejaculatorii. Common unpaired glandular structures are not present. At the last moult the copulatory organs reach their complete development, and the ducts become open externally.
The oviducts in the larva are at first slender, string-like, and bear the egg-follicles. As soon as the eggs pass out of the follicles and collect in the oviducts, the walls of the latter become stretched, and this portion forms two uterus-like structures. The terminal division of the two passages forms their vaginal portions. But since there is no common vagina, there are no unpaired glands and no receptaculum seminis. The two ducts become open after the last shedding of the skin.
Palmén adds that this paired or double nature of the sexual glands and their external ducts in this group of insects occurs in some Myriopoda (Fig. 3, E, F) and a few Arachnida (Fig. 3, C, D, the outlets being in this class unpaired), numerous Crustacea, and most worms; 491and as already stated it is very marked in Limulus, where the paired outlets are in both sexes very simple and wide apart (Fig. 3, A). In the worms the paired genital ducts are modified segmental organs. As we have seen, in the young male Lepisma there are two male genital openings. Hence this double nature of the genital passages in the may-flies seems to be very primitive.
In the Dermaptera, also, the genus Labidura was found by Meinert to have two independent ductus ejaculatorii, opening externally in double external slit-like processes (penes). The two ducts arise from a single seminal vesicle, which is either paired (L. advena), or forms a common passage (L. gigantea). In Forficula (Fig. 464, B) only one ejaculatory duct persists, the other is obliterated, and one of the penes is atrophied, the other assuming a position in the middle line of the body. Thus the single ejaculatory duct and seminal vesicle arise from the primitive vasa deferentia, and not from the integument of the body, as is the case in the following examples.
According to the researches of Dufour, Loew, etc., most species of Orthoptera (Œdipoda), Libellula, Perla, Panorpa, Rhaphidia, Myrmeleon, Sialis, and Trichoptera (Hydropsyche) have double vasa deferentia and seminal vesicles, and two ejaculatory ducts. The male genital passages of Rhaphidia have a double opening, Loew describing “the two seminal vesicles as lying near each other and at last uniting in a common passage, with an external opening, which, however, must be very short, since I could only once clearly observe it.” This opening is a deep invagination of the external integument, at the bottom of which the two ducts open independently of each other. In such insects, Palmén states that the single ejaculatory duct morphologically arises by an invagination of the integument.
In another group, forming, as regards the genital apparatus, a step next above the Ephemeridæ, viz. the Perlidæ, the oviducts open near each other at the bottom of a median single “vagina,” situated between the 7th and 8th abdominal segment; it is covered beneath by a valve-like, enlarged sternite of the preceding segment, and Palmén homologizes it with the ovi-valvula of some Ephemeridæ. He regards this bell-shaped vagina as a cup-like, deep, intersegmental fold, which projects into the body-cavity and there receives the two ducts.
This differentiation in the Perlidæ may be regarded as the type for several groups of insects. But in others occur a complication which in some degree modifies the type. Thus the invagination arises out from one segment alone, but several segments during metamorphosis may become so reduced that the ventral portions of all may be invaginated to form the vagina. Thus in the larva of Corethra, according to Leydig, and also Weismann, the two testes are attached by two cords to the integument; the hinder ones are inserted independently, and share in the development of the outlets.
Graber has observed the same relations in the pupa of Chironomus, the efferent genital tubes in both sexes being separate, so that there are two vaginal passages and two penes present. Palmén comments on these relations in the dipterous insects, remarking that during metamorphosis certain parts of the terminal abdominal segments are reduced, while others are hypertrophied; hence the points 492of insertion of the cords referred to becoming the openings of the vasa are carried within the abdomen; and this part of the integument becomes an unpaired section. In these insects, also, there is an unpaired vesicula seminalis, but its morphological nature (whether formed from the integumental duct or the fused vasa deferentia) can only be settled after special investigation.
In the Lepidoptera, also, it has been shown by Herold, Suckow, Bessels, and recently with full details by Jackson, that the paired larval oviducts are at first solid, but become tubular early in pupal life. A little later, their cavities open into that of the azygos or unpaired oviduct. The paired oviducts open in the female caterpillars on the hind edge of the 7th abdominal segment, afterwards uniting with the unpaired vagina of the 8th segment, which is developed from the hypodermis.
Jackson adds that there are three stages traceable in the evolution of the genital ducts of Lepidoptera: “an ephemeridal stage, which ends towards the close of larval life; an orthopteran stage, indicated during the quiescent period preceding pupation; and a lepidopteran stage, which begins with the commencement of pupal life.”
As a summary of these results it appears that the genital organs of insects consist of two morphologically different elements: 1. the primitive internal paired structures (testes with the vasa deferentia; ovaries with the ovarian tubes), and 2. integumental structures (Fig. 464). In the most primitive winged insects (Ephemeridæ) the latter structures are only represented by the two external sexual openings, the entire reproductive system being paired. The paired parts become in the more highly differentiated forms united into single parts, while, a, a common integumental division, grows in, forming the ductus ejaculatorius, or the vagina; or, b, the inner passages anastomose together, i.e. the openings fuse together; or, c, both of these cases occur at once; or, finally, we have d, where the superfluous paired parts by reduction become single.
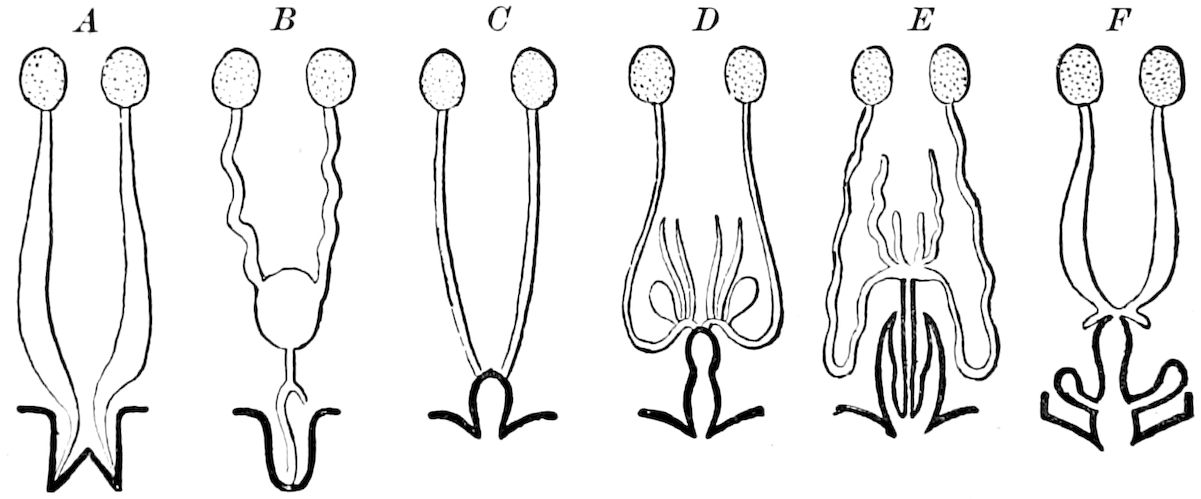
Fig. 464.—Evolution of the unpaired from the paired sexual organs of insects: A-E, male organs. The parts arising by invagination of the integument indicated by thick black lines. A, an Ephemerid. B, Forficula auricularia. C, nymph of Orthoptera in general. D, Œdipoda. E, Cetonia aurata. F, female organs of Æschna.—After Palmén, from Lang.
The male ducts open behind the 9th, the female passages of Ephemerids behind the 7th abdominal segment, those of other 493insects behind the 8th, except in the Stylopidæ (Strepsiptera), in which they open much in front.
Figure 464 graphically shows their relation. In the Odonata (F) the chitinous lining or integumental invagination extends inwards where the two oviducts begin, in the Coleoptera (E) the vagina, bursa copulatrix, and receptaculum seminis being lined by a thick chitinous layer. While in Perla the two seminal ducts pass directly into the copulatory organ, in the Coleoptera they open into the unpaired ductus ejaculatorius at a distance from the copulatory organ.
The morphological results obtained by Palmén, and for the Lepidoptera by Jackson, were apparently confirmed from an embryological point of view by Nusbaum, from observations on the development of the sexual passages in two genera of Pediculidæ, and are as follows:—
1. The prevalent impression that the larval ducts unite with each other and give origin to the whole system of sexual ducts is incorrect; they form only the vasa deferentia or the oviducts.
2. All other parts of the efferent apparatus (uterus, vagina, receptaculum seminis, ductus ejaculatorius, penis, and appended glands) develop from the hypodermis.
3. The connective tissue and the musculature of the efferent apparatus are derived from mesoblast cells present in the body-cavity.
4. The efferent ducts originate as paired rudiments. All unpaired (azygos) parts (uterus, penis, receptaculum seminis, unpaired glands, etc.) are at first paired. The unpaired efferent apparatus of insects must therefore be regarded as morphologically a secondary and more complicated form.[76]
5. The male and female efferent ducts are strictly homologous.
6. The cavities of the oviducts, uterus, vagina in the female, of the vasa deferentia, appended organs, and ductus ejaculatorius of the male arise independently, and come into connection secondarily.
The presence of two genital openings, viz. a bursa copulatrix or copulatory pouch, and of the primitive oviducal orifice behind the 9th segment, is peculiar to Lepidoptera, and the inquiry naturally arises whether they represent the outlets of two pairs of segmental organs. The question has been fully set at rest, however, by Jackson, who shows that the copulatory pouch is a secondary invagination of the ectoderm, being derived from the hypodermis, while the second aperture is a special adaptation. It is, however, the partial homologue of the vaginal orifice in other orders of insects. It opens behind the sternite of the 8th abdominal segment, the typical position of the vaginal aperture as shown by Lacaze-Duthiers. The lateral position of the bursa and its separation 494from the azygos oviduct are probably late features in the phylogenetic history of the Lepidoptera, subsequent even to the closure of the furrow.
“The existence of a second or posterior aperture is probably to be attributed to the advantage gained by a terminal position for the aperture through which the ova are laid. The remarkable way in which this aperture shifts backwards seems to point very distinctly to this explanation, especially as the Lepidoptera are entirely devoid of the outgrowths which form the ovipositor in some orders; e.g. most Orthoptera.”
The original condition of things appears to have been retained in a moth, Nematois metallicus, which, according to Cholodkowsky, possesses but a single external aperture, the bursa opening into the dorsal wall of the unpaired oviduct.
a. The male organs of reproduction
Bearing in mind that the testes with their efferent ducts are, like the ovaries and egg-tubes, primitive structures, there are various secondary or adaptive structures which are either due (1) to modifications of the male efferent ducts, or of the ovarian tubes, or (2) to various accessory organs, mostly glandular, resulting from the invagination of the ectoderm.
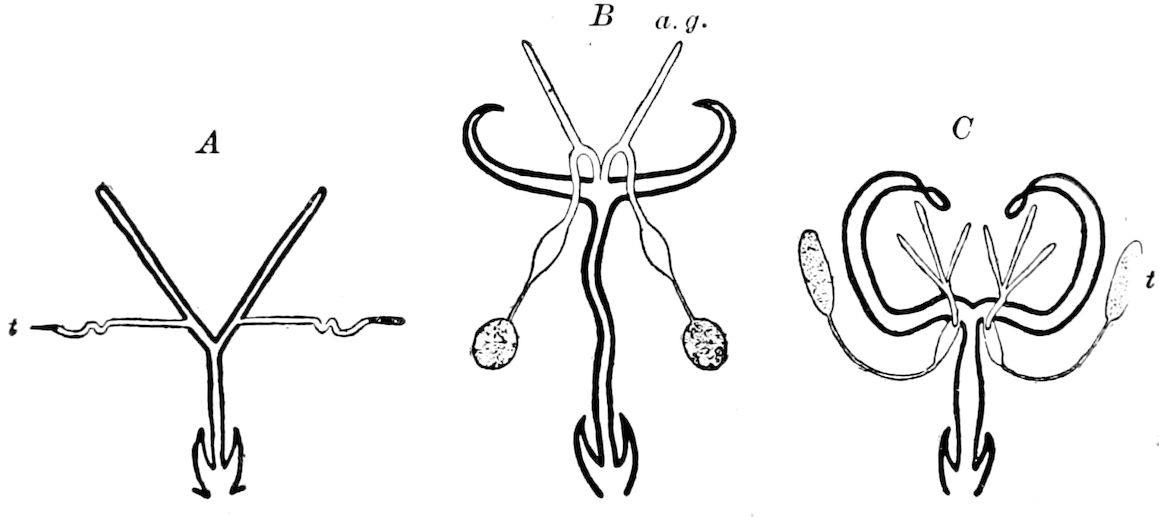
Fig. 465.—A, diagram of male sexual organs of Carabus. B, of Blaps. C, of Hydrophilus. The heavy black lines represent the ectodermal organs; t, testis; a. g., accessory glands.—After Escherich.
The male organs are, then, the following:—
1. Two testes (Figs. 465–469, t, H, ho).
2. The two seminal ducts (vasa deferentia, v, sl, SL), whose lower or outer (distal) division becomes enlarged and acts as a seminal vesicle (vesicula seminalis; Figs. 467–469, bl, SB).
3. The common ejaculatory duct (ductus ejaculatorius), with the penis (Figs. 467–469, ag, uSG).
4. Accessory glands at the base of the vasa deferentia (glandulæ mucosæ, Figs. 465–469, a. g., dr, D), whose secretion mixes with the semen or serves for the formation of the seminal packets (sematophores).
495In his paper on the internal male organs of beetles, Escherich states that those of the Carabidæ illustrate the simplest, most primitive condition (Fig. 465). A simple blind tube on each side produces spermatozoa, stores the elements, and secretes mucus. Each of these tubes opens into a somewhat larger duct, and the two unite in a common ejaculatory canal. The terminal portion in these beetles is lined with chitin, and is therefore ectodermal, and not the result of the union of the mesodermic vasa deferentia. The region corresponding to the testes, vasa deferentia, and seminal vesicles are mesodermic. Blaps (Fig. 465, B) is intermediate between the Carabidæ and Hydrophilus (Fig. 465, C). The accessory glands (a. g.) are developed, and the seminal vesicles are situated in the middle, and not at the lower end of the vasa deferentia, as in Hydrophilus.
The testes.—Each testis is composed of follicles or corresponding parts, which according to the group of insects in which they occur are united in different ways; or each testis consists of a single hank or skein-like blind tube which is enveloped by a membrane, as in the Carabidæ, Dyticidæ, or Lucanidæ.
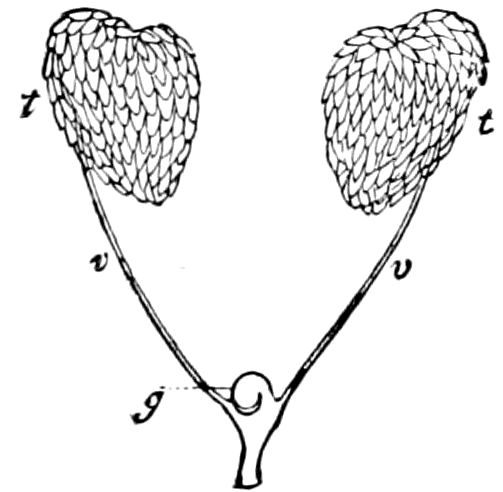
Fig. 466.—t, testis; v, vas deferens; g, seminal vesicle of Acheta campestris.—After Carus, from Gegenbaur.
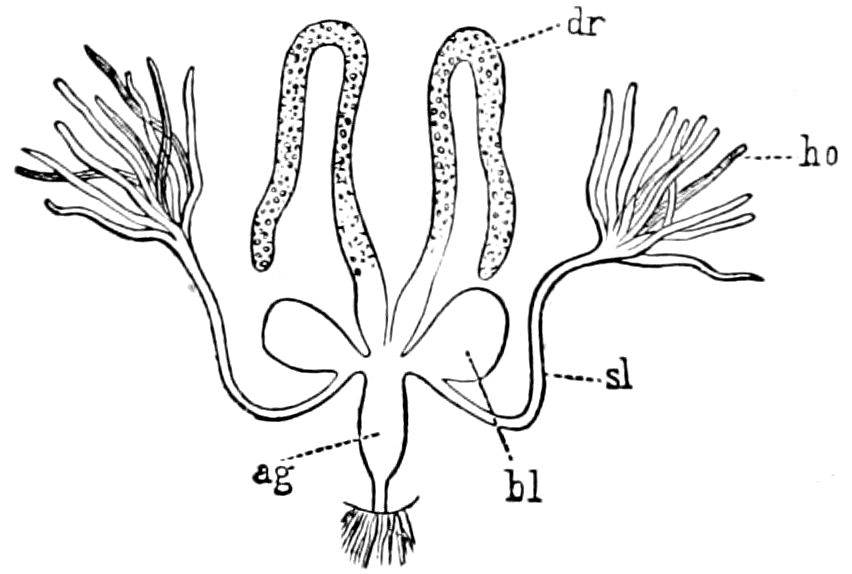
Fig. 467.—Male sexual apparatus of a bark-beetle: sl, vas deferens; ho, testis; bl, seminal vesicle; dr, accessory gland; ag, ductus ejaculatorius.—After Graber.
The number of testicular tubes is small in most Hemiptera, but very great in the Cicadidæ, Orthoptera, Coleoptera, and many Hymenoptera. Although the testes are usually separated from each other, they are closely united in certain Orthoptera (Gryllotalpa, Ephippigera), Coleoptera (Galerucella), in many Lepidoptera, and in a number of Hymenoptera (Scolia, Pompilus, Crabro, and others).
The two testes of most Lepidoptera are so closely grown together or coalesced into a single body that one might regard them as a single testis. But in the different families there occur all grades, from the unpaired testes of most Lepidoptera to Hepialus with separate testes. Cholodkowsky therefore distinguishes four types:—
1. The embryonal or primitive type, with two testes, whose seminal follicles are entirely separate. (Brandt.) These testes are contained, as in all other Lepidoptera, in a well-developed thick chitinous membrane or scrotum, analogous to that of the higher vertebrates, which envelops each separate seminal follicle (Hepialus humuli).
4962. The larval type, with two testes, whose four follicles are enclosed by a common scrotal membrane (Bombyx mori, Gastropacha quercifolia, Ichthyura anachoreta and anastomosis, Saturnia pyri, Aglia tau).
3. The pupal type (since it first occurs in the pupa state), with a single testis, which possesses an external median lace-like covering. (Adela, Lycæna.)
4. The imaginal type, with a single testis enveloped by a lace-like scrotum, within which the follicles are wound around the longitudinal axis of the testis. (Most Lepidoptera.)
In Nematois there are twenty seminal follicles, the number of ovarian tubes being the same. (Cholodkowsky.)
In many insects the testes are not composed of tubes (follicles), but of button-like bodies, each of which has its own duct.
The color of the testes is usually white, but they may be orange (Decticus), yellowish green (Locusta viridissima), or deep yellow (Chrysopa).
The testes of Asilid flies are enveloped by a common dark-red membrane rich in tracheæ, like that in Lepidoptera which clothes the separate testicular follicles. The two testes of Calliphora are enveloped by an orange-yellow capsule, outside of which is a special membrane formed by the fat-body. (Cholodkowsky.)
In the honey-bee the testis has two envelopes, the outer of which is formed by the fat-body, the inner coat of connective tissue. The entire testis corresponds to a portion only of that of Bombyx mori.
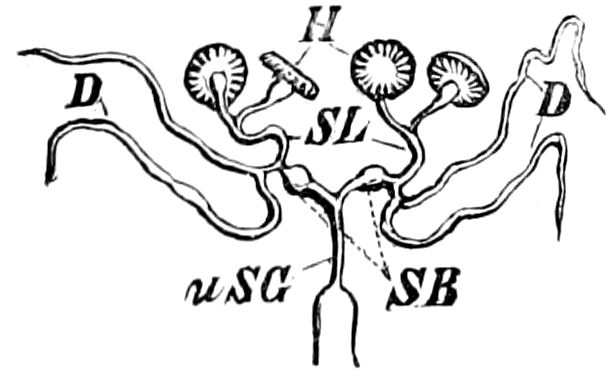
Fig. 468.—Male organs of a weevil, Hylobius abietis: H, testis; SL, vas deferens; D, slime gland; SB, seminal vesicle; uSG, ejaculatory duct.
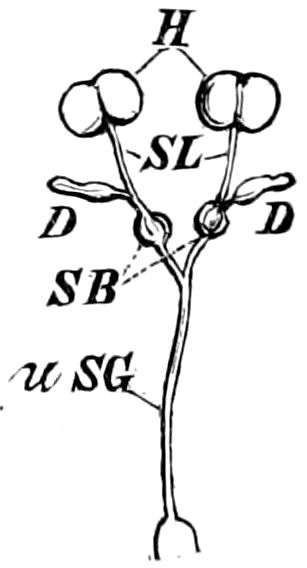
Fig. 469.—Male organs of Tomicus. Lettering same as in Fig. 468.—This and Fig. 468 from Judeich and Nitsche.
The seminal ducts.—The vasa deferentia are fine tubes, which vary much in length; being short in many beetles and locusts, very short in many Diptera (Syrphidæ, etc.), very long in Cicada and many beetles; according to Burmeister, being in Dyticus about five times, in Necrophorus and Blaps eight to ten times, in Cicada 14 times, in Cetonia aurata 30 times, as long as the body. They either resemble a skein of silk, or form a tangled mass.
The distal or lower end of the vasa is in many insects dilated into a sac or seminal vesicle, which serves for the reception and storage of the seminal fluid after it passes through the vasa deferentia. In the honey-bee the vas deferens is given off from the reservoir, forms loops in and outside of the testis, and passes to the seminal vesicle. The canal into which the vesicle narrows does not open into the ductus ejaculatorius, but into the glandulæ mucosæ; its epithelial cells are much vacuolated, and have, therefore, a spongy appearance. (Koschewnikoff.)
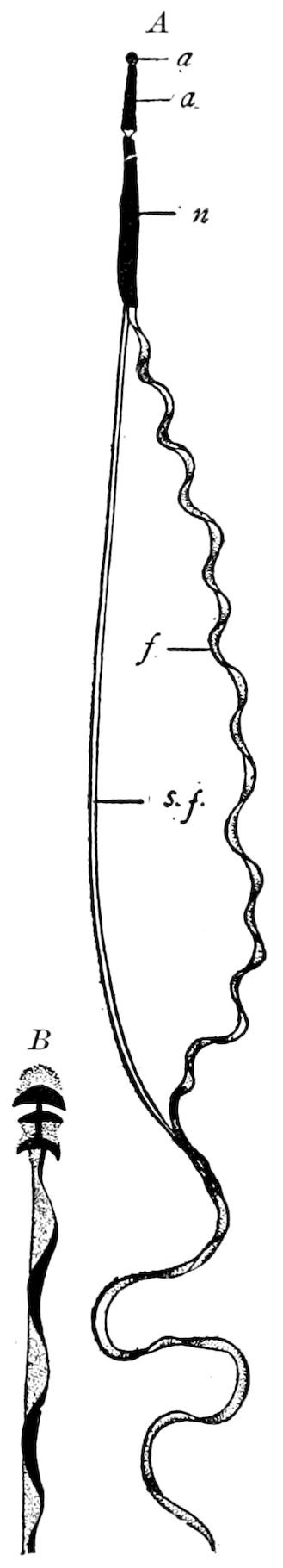
Fig. 470.—A, spermatozoön of a beetle (Copris), partly macerated to show structure of flagellum, which consists of a supporting fibre (s.f) and a fin-like envelope (f); n, nucleus; a, a, apical body divided into two parts. B, anterior part of that of Calathus, with barbed head and finmembrane.—After Ballowitz, from Wilson.
The ejaculatory duct during coition conducts the sperm into the copulatory pouch of the female. In consequence of the stretching of the integumental membrane the end of the duct can be erected and again withdrawn. For this purpose the end of the duct is thickened and is said to be provided with powerful muscles. The evaginable terminal portion is covered by a strong chitinous membrane forming the penis or intromittent organ (Fig: 462, h), which is externally enveloped by a pair of chitinous lobes, which in many beetles are converted into a capsule. The ductus ejaculatorius of the honey-bee is inserted by two chitinous branches into the point of union of the two glandulæ mucosæ; it and the entire copulatory apparatus are devoid of muscles, though it is, however, well developed beneath the mucous glands. (Koschewnikoff.)
The accessory glands of the vasa deferentia are tubes whose secretions either directly mix with the semen, or in many cases form seminal packets (spermatophores). In Coleoptera, Lepidoptera, and Diptera there is usually one pair. In many insects there are several pairs, as in Hydrophilidæ and Elateridæ; they are branched in Hemiptera, and in Orthoptera bushy. The single glandular tubules are very long, and form a skein-like mass. In Orthoptera, in the larger number of accessory glands, two forms may be distinguished, which differ from each other in their contents (Siebold). In the cockroach (Fig. 461) these glands form the “mushroom” shaped gland of Huxley, which was at first regarded as the testis.
The spermatozoa.—These very minute bodies, the sexual homologues of the eggs, abound in the seminal fluid, and are formed in the follicles of the testes from a germinal layer or epithelium, as are the eggs. They are hair- or thread-like, usually consisting of a head, a body or middle-piece, and a long, thread-like tail (flagellum), which vibrates rapidly, causing the spermatozoön to move actively forwards (Fig. 470).
In beetles, according to Ballowitz, there are two main types of spermatozoa, connected, however, by intermediate 498forms. There is a double-tailed type, already described by Bütschli and v. la Valette St. George, and there are others which are single-tailed. Bütschli showed that in the double spermatozoön one tail-filament is straight and stiff, the other being undulating and contractile. Ballowitz describes this type in Calathus (Fig. 470, B), Chrysomela, and Hylobius, etc., and shows that the straight or supporting portion of the tail is elastic, but somewhat stiff, resistant to reagents, and without any fibrillar structure, while the contractile fringe consists of an extremely complicated system of fibrils (Fig. 470). The single-tailed type of spermatozoön, as seen, e.g., in Melolontha and Hydrophilus, has no supporting fibres. The tail is twisted in a spiral, corresponds to the contractile fringe of the double type, and exhibits a complicated fibrillar structure. The fringed type works its way ahead like the screw of a steamer.
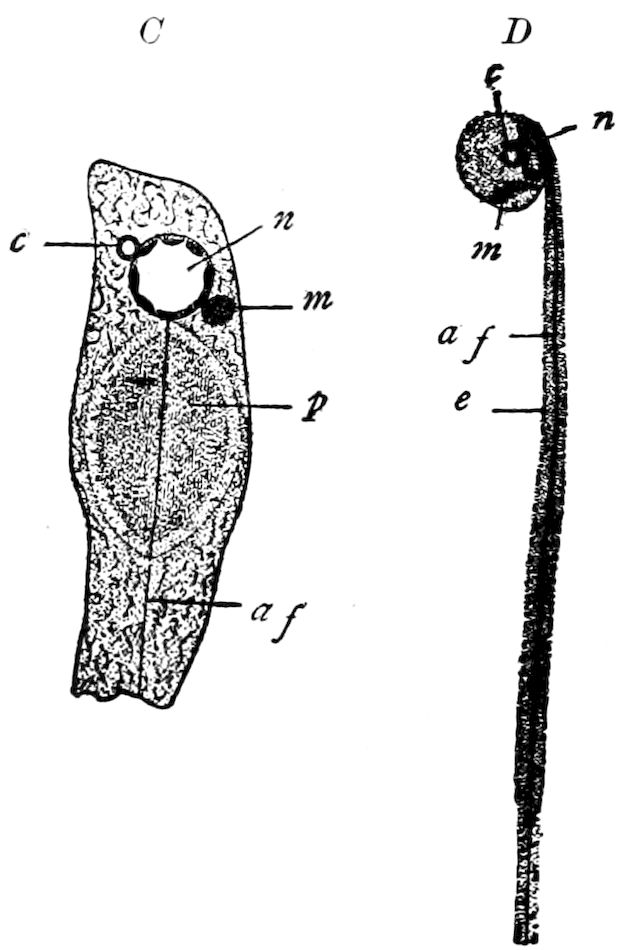
Fig. 471.—C, anterior end of spermatid of a moth (Pygæra). D, young spermatozoön of the same; af, axial filament; c, centrosome; m, middle-piece or mitosoma; n, nucleus; p, paranucleus; e, envelope of the tail.—After Platner, from Wilson.
Each spermatozoön is a modified but complete cell, and the nucleus contains the chromatin, a deeply staining substance of the nuclear network and of the chromosomes and the supposed bearer of heredity.
Formation of the spermatozoön.—It arises from a primordial germcell called spermatogonium. This cell contains a large, pale nucleus and a dark body, the accessory nucleus of Bütschli. The spermatogonia subdivide, but at a certain period pause in their subdivisions, and undergo considerable growth. “Each spermatogonium is thus converted into a spermatocyte, which, by two rapidly succeeding divisions gives rise to four spermatozoa, as follows: The primary spermatocyte first divides to form two daughter-cells, known as spermatocytes of the second order, or sperm mother-cells. Each of these divides again—as a rule without pausing, and without the reconstruction of the daughter-nuclei—to form two spermatids or sperm-cells. Each of the four spermatids is then directly transformed into a single spermatozoön; its nucleus becoming very small and compact, its cytoplasm giving rise to the tail and to certain other structures.... As the spermatid develops into the spermatozoön, it assumes an elongated form, the nucleus lying at one end, while the cytoplasm is drawn out to form the flagellum at the opposite end.” (Wilson’s The Cell, from La Valette St. George.)
499Henking finds that the primordial sperm-cells correspond to the primordial ova, both forms of cells in the insect he studied containing the characteristic number of twenty-four chromosomes.
The spermatogenesis of Laphria, according to Cholodkowsky, is very peculiar, and strongly resembles that described by Verson in Bombyx mori. In the blind end of the testicular tubes lies a colossal cell visible to the naked eye, the spermatogone, from which the entire contents of the testes originate. In Bombyx this spermatogone appears in the larva state. Such colossal spermatogones also occur in Lepidoptera of different families (Hyponomeuta, Vanessa, and in the pupa of Chareas graminis), in Trichoptera, and in Hemiptera (Syromastes); and Cholodkowsky inquires whether they may not be typical of insects. Toyama has observed these colossal cells not only in the testes but also in the ovaries of the silkworm. He regards them as supporting cells.
The spermatozoa are inclined to remain in bundles, and in this state are expelled during copulation. These bundles are either root-like, bushy, string-like, sinuous, or worm-like.
Auerbach has observed the spermatozoa of Dyticus marginalis in their passage through the convoluted seminal vesicles. All those arising from one testicular tube are united in a bundle. Each has a very complex structure, bilateral but unsymmetrical. The right side of the head is concave, the left convex; the whole head is longitudinally curved to right or left; and on the posterior half of the right side there is a projecting ridge bearing a hook-shaped cyanophilous “anchor,” at the free end of which an erythrophilous spherule appears. The most remarkable fact is that the spermatozoa unite in pairs in a perfectly definite way, opposed and crossed in a manner somewhat suggestive of a pair of scissors, with the right sides of the heads in contact. During this conjugation, or “dejugation” as Auerbach calls it, the anchors change their shape, and the little spherules are lost. Hundreds of these double spermatozoa are found together in little balls. The conjugation is a temporary one, but it may permit a molecular exchange of substance, perhaps with the result of mixing the hereditary qualities and limiting variability. (Journ. Roy. Micr. Soc., 1893, p. 622.)
In many insects which lack a true penis, the bundle of spermatozoa are united in the ejaculatory duct, forming packets which are enveloped by the secretion of the accessory glands which stiffens into a hard case. These packets are called spermatophores. They are either introduced into the vagina of the female or simply remain outside. Graber has repeatedly observed that the male crickets, in the absence of the female, let their spermatophores fall to the earth; whether it is afterwards made available is not known, because hitherto no case is reported that females seeking impregnation search, as in the case of the Isopod crustacean, Porcellio, for the spermatophores.
In the Gryllidæ and Locustidæ the spermatophore lies in a cup-like cavity under the penis. This is called the “spermatophore cup” (Chadima, 1871), into which the ejaculatory duct of the testis opens.
500According to the views of Schneider, the spermatophores, with their capsule, usually consist solely of seminal filaments, which stick closely to each other, and only exceptionally have a capsule formed by a glandular secretion. In Locusta, however, and perhaps also in Gryllus, the sperm is enveloped by the secretion of the accessory glands of the seminal ducts; the spermatophores pass, still fluid, out of the sexual opening of the male into that of the female, but become chilled on the outer surface, so that the sperm, without coming in contact with the air, passes into the receptaculum seminis.
The mode of grouping of the spermatozoa of the Locustidæ as they occur in the spermatheca of the female is remarkable. Their heads lie so close to each other that they form a long shaft, while the numerous threads are arranged so as to look like the two vanes of a feather, the entire mass being like a very long heron’s feather. (Siebold.)
In the honey-bee the spermatophore is likewise enveloped by the secretion of the accessory glands, and thereby becomes a sort of seminal cartridge. This is a peculiar oval body which is carried during the marriage-flight into the air within the upper part of the penis, the so-called penis-bulb. (Leuckart.)
b. The female organs of reproduction
The different parts of the female reproductive organs are the following:
1. The two ovaries.
2. The two oviducts.
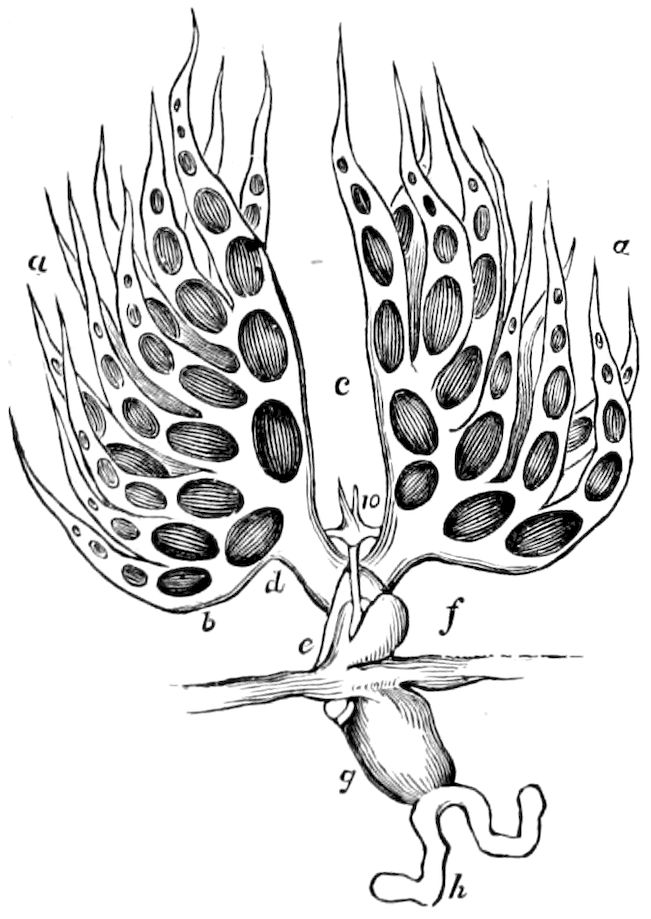
Fig. 472.—Female organs of generation of a saw-fly (Athalia centifoliæ): a, b, c, the 18 ovarial tubes originating from each of the two oviducts (d), and containing the immature eggs; e, common oviduct; f, spermatheca; g, poison-sac; h, poison-glands; 10, last ganglion.—After Newport.
3. The common egg-passage in nearly all insects (its distal or hindermost part forming the uterus or vagina).
4. The receptaculum seminis, or spermatheca.
5. The bursa copulatrix, or copulatory pouch.
6. The accessory glands (cement, sebific, or colleterial glands, or “oil reservoirs,” glandulæ sebaceæ, coleterium).
The ovaries and the ovarian tubes.—As in the testes, so each ovary consists of a variable number of ovarian tubes, by some called ovarioles, united by a thread at the distal end, and at the lower or hinder end opening into the oviduct. Each ovarian or egg tube is divided into three sections: (1) the terminal thread; (2) the terminal chamber, 501and (3) the actual ovarian tube, or chambered main division, this forming the longest part of the egg-tube.
The slender terminal thread serves to attach or suspend each egg-tube near the dorsal vessel (not directly to the heart, as formerly supposed), becoming lost in the fat-body.
Fig. 473.—Ovarian tube of P. orientalis: A, section near the end; tf, base of terminal filament. B, section lower down; ec, egg-cells in egg-chamber.—After Brandt.
The terminal chamber contains undifferentiated cell elements, supposed to be the remains of the ovarian rudiments. From these arise (either in the embryo or larva) first, the follicle epithelium of the ovarian tubes; and, second, the material for the formation of the new eggs, and nutritive cells. “In the terminal chamber these cell-elements remain undifferentiated, excepting when required for the removal of the follicle epithelium, eggs, and nutritive cells in the adult insect.” (Lang.) This portion of the ovariole is called the germarium. In Blatta it is filled with protoplasm in which numerous small nuclei are imbedded. (Wheeler.) The chambered main division of the egg-tube contains the ripening eggs, one in each compartment, the tube appearing like a string of beads.
The egg-tubes are of two types: (1) those without, and (2) those with nutritive cells, the first kind being the simplest, and occurring in the Synaptera (except Campodea) and in Orthoptera. As an example may be cited that of the cockroach (Fig. 473), where in each tube there is a simple continuous row of eggs from the terminal chamber to the oviduct. The tube being constricted between these consecutive eggs, gives it a beaded appearance.
In the cockroach (Periplaneta orientalis) each egg-tube has a beaded appearance. Its wall consists of a transparent elastic membrane, lined by epithelium, with an external peritoneal layer of connective tissue. The terminal filament (tf) is filled with a clear protoplasm, with a few nuclei. In the terminal chamber (tc) are large nucleated cells, with separate nuclei, both entangled in a network of protoplasm. In the third, or egg-chamber (ec), are about twenty ripening eggs, arranged in a single row. “Between and around the eggs the nuclei gradually arrange themselves into one-layered follicles, which are attached, not to the wall of the tubes, but to the eggs, and travel downwards with them. As the eggs descend, the yolk which they contain increases rapidly, and the germinal vesicle and spot (nucleus and nucleolus), which were at first plain, disappear. A vitelline membrane is secreted by the inner surface and a chitinous chorion by the outer surface of the egg-follicle.
502“The lowest egg in an ovarian tube is nearly or altogether of the full size; it is of elongate-oval figure, and slightly curved, the convexity being turned towards the uterus. It is filled with a clear albuminous fluid, which mainly consists of yolk. The chorion now forms a transparent yellowish capsule, which, under the microscope, appears to be divided up into very many polygonal areas, defined by rows of fine dots. These areas probably correspond to as many follicular cells.” (Brandt, from Miall and Denny.)
In the second type, i.e. those egg-tubes with nutritive cells, there are two kinds. In the first the egg-chambers and yolk- or nutritive chambers alternate, each of the latter containing one or more nutritive cells, which serve for the nourishment of the ripening egg contained in the neighboring chamber. “The egg- and yolk-chambers may be distinctly separated externally by constrictions (Hymenoptera and many Coleoptera), or one nutritive and one egg-chamber may lie in each section of the ovarian tube, which is externally visible as a swelling (Lepidoptera, Diptera).”
In the second kind with nutritive cells, the actual tube consists (Fig. 474, C) of ovarian chambers only; the nutritive cells here remain massed together in the large terminal chamber. The single egg in the tube is united with the terminal chamber by connective strands (d. s.), which convey the nutritive material to the eggs. (Lang.)
Egg-cells, nutritive cells, and the cells of the follicle-epithelium (epithelium of the chambers of the ovarian tubes) are, says Lang, according to their origin, similar elements, like the egg and yolk-cells of the flat worms (Platodes); division of labor leads to their later differentiation. Only a few of the numerous egg-germs develop into eggs, the rest serving as envelopes and as food for these few.
Korschelt considers that all the chief elements of the egg-tubes, viz. egg, nutritive, and epithelial cells, arise by a direct transformation of the elements of the terminal chamber, and that the last may be traced to the indifferent elements of the terminal thread, the elements in question originating from the nuclear elements by a breaking down of the syncytium (or masses of protoplasm with nuclei scattered through it) composing it (Fig. 475).
The latest work is that of Wielowiejski (Zoologische Anzeiger, ix, 1886, p. 132), whose observations are based on a study of the ovarian tubes and the growing eggs of the Hemiptera (Pyrrhocoris), the Coleoptera (Telephorus, Saperda, Cetonia and Melolontha, Carabidæ, and Hydradephaga), etc.
Wielowiejski divides the ovaries of insects into three groups:—
1. Comprising such ovaries in the ends of whose egg-tubes (terminal filament) the embryonal cells in the early stages are accumulated, and are transformed into egg-, yolk-, and epithelial cells respectively. (Ovaries of Orthoptera, geodephagous and hydradephagous Coleoptera, Lepidoptera, Diptera, and Hymenoptera).
2. Comprising ovaries whose ends above the egg-cells and egg-germs (Eianlagen) possess throughout life a more or less voluminous solid accumulation of cells (terminal chamber), but which stand in no close relation with the first. (Ovaries of Coleoptera, with the exception of the Geodephaga and Hydradephaga, and Aphidæ in part.)
Fig. 474.—Various types of ovarian tubes, diagrammatic: A, ovarian tube without nutritive cells. B, egg-tube with alternating nutritive and egg-compartments. C, ovarian tubes in which the terminal chamber (ek) is developed into a nutritive chamber, with which the developing eggs remain connected by means of threads (ds); ef, terminal filaments; efa, egg compartments or chambers; fe, follicle epithelium; df, yolk-chambers.—After Lang (C from Claus).
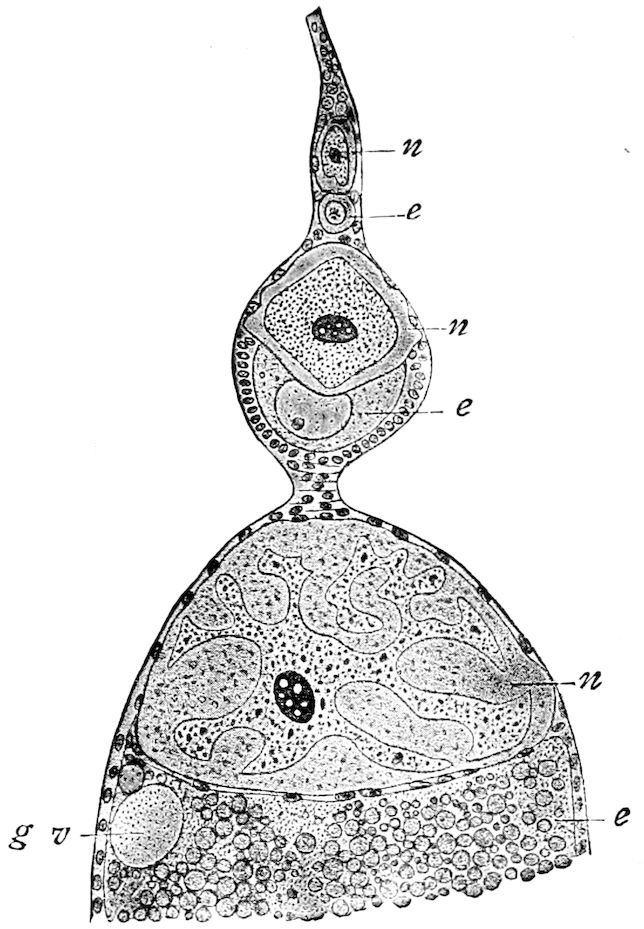
Fig. 475.—Upper portion of the ovary in Forficula, showing eggs and nurse-cells; below, a portion of the nearly ripe egg (e) showing deutoplasm-spheres and germinal vesicle (gv). Above it lies the nurse-cell (n), with its enormous branching nucleus. Two successively younger stages of egg and nurse-cell are shown above.—After Korschelt, from Wilson.
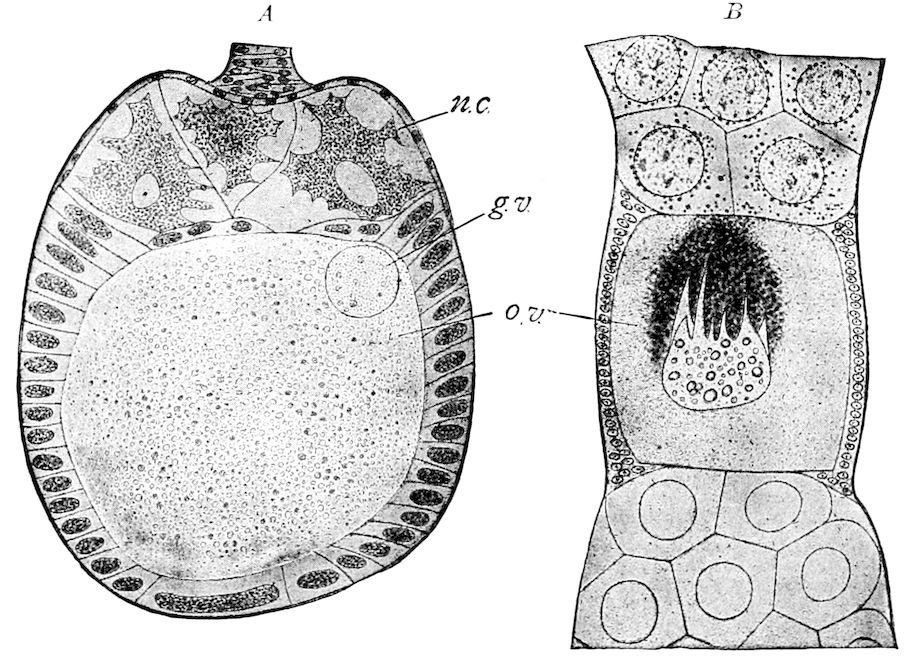
Fig. 476.—A, ovarian egg of a butterfly (Vanessa), surrounded by its follicle; above are the nurse-cells (n. c.), with branching nuclei; g.v, germinal vesicle. B, egg of Dyticus, living; the egg (o.v.) lies between two groups of nutritive cells; the germinal vesicle sends amœboid processes into the dark mass of food-granules.—After Korschelt, from Wilson.
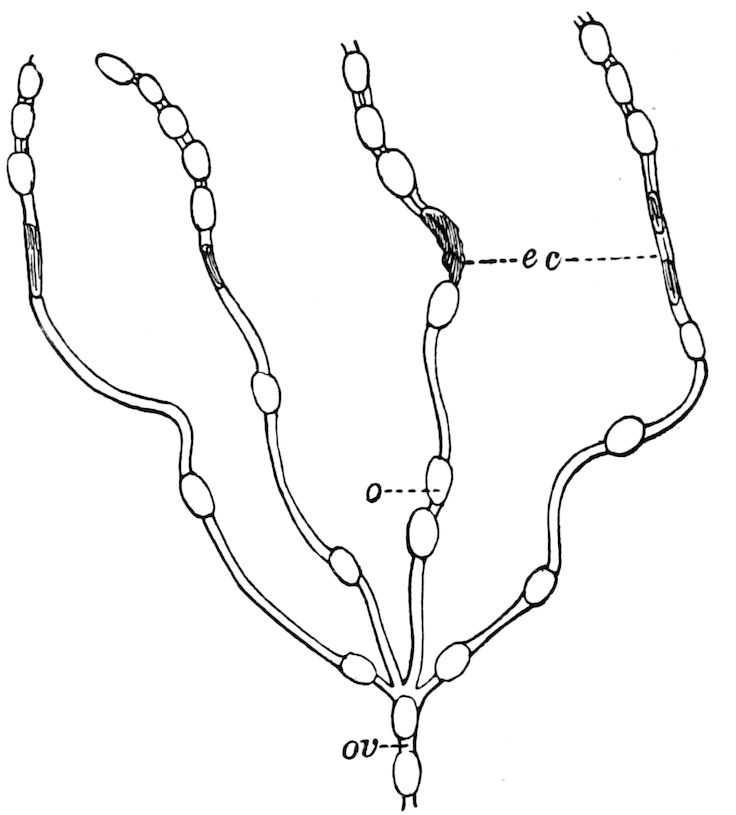
Fig. 477.—A, lower portion of one of the two ovaries of Sphinx ligustri, the four egg-tubes uniting to form the slightly developed calyx (ov). The egg-tubes above contain ripe eggs still surrounded by the follicle; e. c, the empty egg-chamber. Beyond the empty egg-chambers (e. c) are three egg-chambers with ripe eggs and the connecting cord. The whole tube is surrounded by the peritoneal membrane and musculature.—After Korschelt.
3. Comprising ovaries whose ends above the egg-germs contain a well-developed mass of cells functioning as a yolk-forming organ, between whose special elements grow root-like offshoots of nearly ripe egg-cells. (Hemiptera.)
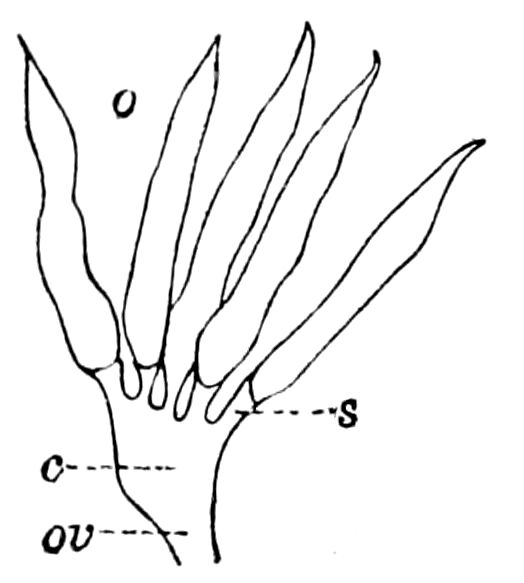
Fig. 478.—Ovary of a beetle, drawn somewhat diagrammatically: o, egg-tube; s, stalk of the same; c, egg-calyx; ov, oviduct.—After Korschelt.
When the egg is ripe the food-chamber disappears because its contents have served for the formation of the egg below it. In Lepidoptera especially, the egg-tubes resemble strings of pearls because most of the numerous eggs ripen simultaneously and are likewise deposited at the same period, which is naturally not the case in those insects whose eggs gradually ripen (Fig. 477). In other cases the egg- or food-compartments are transformed into each other, but only one egg- and one food-compartment can be situated in the same dilatation of the ovarian tube. Finally, there are insects in whose egg-tubes the egg-compartments are arranged in a single row, while the capacious terminal chamber contains a large mass of food-cells.
Egg-cells, nutritive cells, as well as the cells of the follicle epithelium (epithelium of the chambers of the ovarian tubes), originate as similar or homologous elements, division of labor leading to their later differentiation. Only a few of the numerous egg-germs develop into eggs, the rest serving as envelopes and also as food for these few. (Lang.)
In many insects the egg-tubes open into an 504egg-calyx (Fig. 478, c), in which the ripe eggs collect before passing into the oviduct (ov).
As the result of his investigations on the origin of the cellular elements of the ovaries of insects Korschelt concludes:—
1. The different cell-elements of the egg-tubes, eggs, nutritive cells, and epithelium arise from identical undifferentiated elements situated in the contents of the earliest germ of the egg-tubes.
2. The first formation of the cellular elements present, and the differentiation of the individual compartments of the egg-tube, occur during embryonic and larval life.
3. The undifferentiated elements of the terminal chamber correspond to the embryonic condition, while in post-embryonic time, and even during imaginal life, a new formation of the different kinds of cells takes place.
4. The mode of origin of the different kinds of cells from the undifferentiated elements varies greatly in different insects.
5. From their histological nature, and from the mode of origin of their elements, the most complex egg-tubes and those provided with nutritive compartments are phylogenetically derived from those without such nutritive compartments.
6. The nutritive cells in certain cases originate in the same way and at the same time as the germ-cells, and are therefore to be regarded as germ-cells which have abandoned the function of egg-making, and exchanged it for the production of nutritive material.
7. In the egg-tubes with numerous nutritive compartments the nutritive cells can originate at the same place as the egg-cells, and they afterwards still lie intermingled with these in the beginning or upper part of the egg-tubes.
8. While the capability of egg-making of the germ-cells originally situated in the extremity of the terminal chamber gradually becomes transferred to those at the base of the terminal chamber, and the first transform into nutritive cells, egg-tubes with nutritive compartments at the base may be found.
9. The nutritive cells of certain forms arise independently of the germ-cells and therefore could not have previously originated from them.
10. The epithelium has in all forms nearly the same mode of formation; it everywhere shows a close similarity to the undifferentiated elements of the terminal chamber, out of which it directly develops. As to the fact of formation of epithelium through the germ-vesicles (Keimblaschen), nutritive-cell nuclei, or the so-called “oöblasts,” I could not feel certain.
11. Neither the eggs of Hemiptera or of other insects arise through the agency of “oöblasts,” but like the epithelial and nutritive cells arise by a gradual differentiation from the indifferent elements of the ovarian tubes.
12. The different elements of the egg-tubes, also the eggs, have the morphological value of cells.
Origin of incipient eggs in the germ of the testes.—Heymons has detected in the germ of the testes of the male larvæ of Phyllodromia germanica 7 mm. in length, young or incipient eggs, similar to those seen in the ovarian tubes of the female larva of the same size. In another male larva of the same size also occurred short cylindrical tubes each with a terminal thread, which had the appearance of rudimentary egg-tubes. Hence he thinks that every part of the genital germs (Anlagen) in the male, which are not concerned in the formation of testicular follicles, represents the germ of a female genital gland. As is well known, no insects are hermaphroditic, but this case of the practical origin of eggs and egg-tubes in the lowest division of the male efferent passage, which is 505homologous with the egg-producing division of the female ovarian tubes, points back to hermaphroditic ancestors. And Heymons suggests that the frequent occurrence of hermaphroditism in insects probably confirms this view.
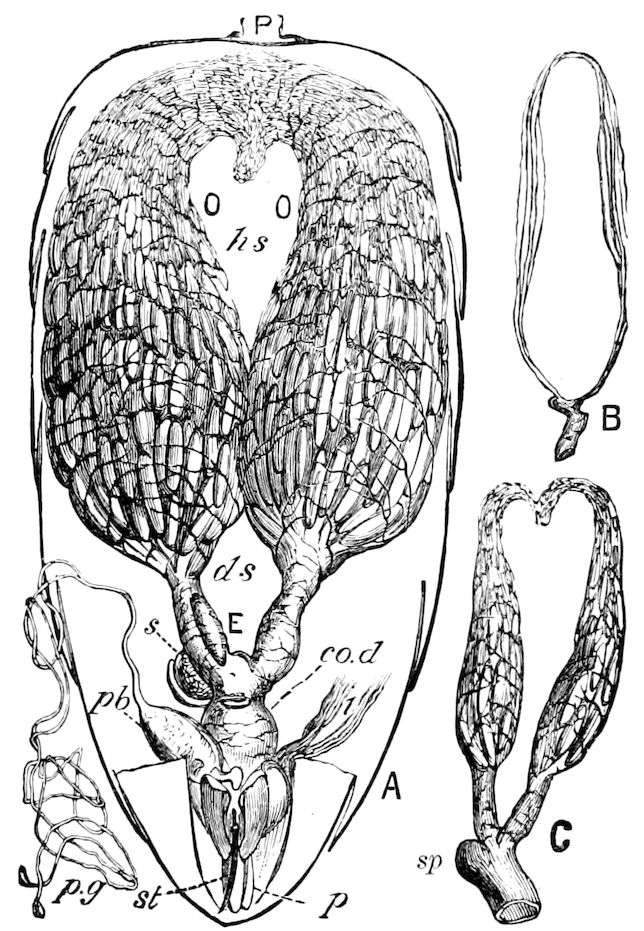
Fig. 479.—Abdomen of queen bee, under side, × 8: P, petiole; o, o, ovaries; hs, position filled by honey-sac; ds, place through which the digestive canal passes; od, oviduct; co.d, common oviduct; E, egg passing oviduct; s, spermatheca; i, intestine: pb, poison-bag; p.g, poison-gland; st, sting; p, palpi. B, vestigial ovaries of ordinary worker; sp, vestigial spermatheca. C, partially developed ovaries of fertile worker; sp, vestigial spermatheca.—After Cheshire.
The bursa copulatrix.—The copulatory pouch in most insects is a special cup-shaped appendage of the vagina adapted for the reception of the male organ during sexual union. Its mode of formation in the cockroach is thus described by Haase:—
“By the retreat of the female sexual aperture, situated in the 8th ventral plate, a considerable space, the genital pouch, is produced; this is formed chiefly by the extended connective membrane between the elongated 7th and 8th ventral plates. This serves for the development of the egg-cocoon, which is retained by the internal appendages of the posterior gonapophyses.”
The fertilization of the female takes place once for all a long time previous to oviposition; the semen in the receptaculum seminis passes out as the eggs slip down the egg-passage, and a spermatozoön gains entrance into the interior of the egg through the micropyle. In Œcanthus, according to Ayers, fecundation probably takes place while the egg is passing into the vagina, “since it is hardly possible that the male element could gain access to the follicles before the chorion is secreted.”
In the Lepidoptera, as has been stated, the copulatory pouch opens separately from the opening of the oviduct (vagina), but a slender canal connects the pouch with the vagina (Fig. 310, bc). The outlet 506(“vagina” of Burgess) of the copulatory pouch opens between the 7th and 8th segments, that of the oviduct (vagina) on the 9th segment being “situated immediately below the anus and hardly separated from it, between the lappets of the 9th segment.” (Burgess.) The opening of the copulatory pouch is, as we have seen, the genuine or primitive sexual opening.

Fig. 480.—Spermatheca of the honey-bee, queen, × 40: a, space filled by a clear fluid; b, mass of spermatozoa; c, duct; d, d, active spermatozoa.— After Cheshire.
Fig. 481.—Female sexual organs of Scolytus: ER, egg-tubes; pEL, paired oviducts; ST, spermatheca; BT, copulatory pouch; KD, cement-glands; Sch, vagina.—After Lindeman, from Judeich and Nitsche.
The spermatheca.—This is a sac or pouch for the reception and storage or preservation of the semen. While in most of the higher insects it opens into the dorsal wall of the vagina (Fig. 472, f), in the cockroach, locusts, and grasshoppers it opens into the bursa; but in other European Orthoptera, as in most insects, it lies upon the dorsal wall of the vagina. (Berlese.) In the cockroach, it is a short tube dilated at the end and wound into a spiral of about one turn. “From the tube a cœcal process is given off, which may correspond with the accessory gland attached to the duct of the spermatheca in many insects (e.g. Coleoptera, Hymenoptera, and some Lepidoptera). The spermatheca is filled during copulation, and is always found to contain spermatozoa in the fertile female. The spermatozoa are no doubt passed into the genital pouch from time to time, and there fertilize the eggs descending from the ovarian tubes.” In Meloë the spermatheca is exceedingly large. (Miall and Denny, pp. 170, 171.)
The colleterial glands.—We have already briefly referred to these glands. Those of the cockroaches form a number of long blind tubes opening into the vagina. They furnish the material for the egg-capsule or oötheca, viz. chitin and large crystals of oxalate of lime.
In Phyllodromia germanica “these glands are glistening white till the time of oviposition approaches, when they assume a yellow tint, and the octahedral crystals are seen imbedded in a viscid substance which fills their lumina. This viscid substance is soluble in potassium hydrate, and is consequently not chitin. When excreted to form the oötheca, it slowly hardens, deepens in color, and becomes insoluble in potassium hydrate. Light has nothing to do with this change, which is possibly produced by the oxygen in the air. It is the same 507change which is undergone by the cuticula of the insect itself immediately after ecdysis.” (Wheeler.)
The vagina or uterus.—This is simply the end of the common oviduct, which, when dilated, is called the vagina, and, in the pupiparous forms, the uterus.
In the cockroach the vagina opens by a median vertical slit situated in the 8th sternite, into the genital pouch or bursa, upon the dorsal wall of which the orifice of the spermatheca is situated. In the sheep-tick the oviduct is enlarged to form the so-called uterus, which furnishes a milk-like secretion for the nourishment of the larva during its intra-uterine life.
In insects in general, the external opening of the vagina is simple, the chitinous structures (valves) at the opening being adapted to receive the male intromittent organ.
When the eggs are to be deposited deep below the surface of the earth, or in wood, or in wood-boring larvæ, or in the body of caterpillars, etc., they are inserted by the ovipositor (see p. 167).
Signs of copulation in insects.—Leydig has collected, partly from his own observations and partly from those of others, a number of cases in which female insects bear traces of having had sexual union, in the form of tags or plates attached to the body, and apparently formed from material secreted by the male. Such probably is the “pouch” on the abdomen of Parnassius apollo, and a somewhat similar structure in Fulgora laternaria, and such is the plate which is found on the hinder part of the abdomen of Dyticus latissimus and D. marginalis. Leydig compares these structures with the white plate in Astacus fluviatilis, and with the little white lid on the spider Argenna, and finds analogues among vertebrates. (Arbeit. Zool. Zoot. Inst. Wurzburg, x, 1891, pp. 37–55, 2 Figs.)
LITERATURE ON THE ORGANS OF REPRODUCTION
a. General
Hunter, J. Observations of bees. (Phil. Trans. Roy. Soc. London, 1792, lxxxii, pp. 128–195.)
Hegetschweiler, J. J. Dissertatio inauguralis zootomica de insectorum genitalibus. Turici, 1820, pp. 28, 1 Pl.
Audouin, V. Recherches anatomiques sur la femelle du Drile jaunatre et sur le male de cette espèce. (Ann. Sc. nat., ii, 1824, pp. 443–462, 1 Pl.)
Müller, Johannes. Ueber die Entwickelung der Eier im Eierstock bei den Gespenstheuschrecken. (Nova Acta Acad. Leop.-Carol., xii, 1825, pp. 555–672, 6 Taf.)
Dufour, L. Recherches anatomiques sur les Carabiques et sur plusieurs autres insectes coléoptères. Organes de la génération (Ann. Sc. nat., vi, 1825, pp. 150–206, 6 Pls.; pp. 427–468, 4 Pls.).
508—— Recherches anatomiques sur l’Hippobosque des cheveaux. (Ibid., 1825, vi, pp. 299–322, 1 Pl.)
—— Recherches anatomiques sur les Labidoures. Appareil de la génération. (Ibid., 1828, xiii, pp. 354–359, 2 Pls.)
—— Recherches anatomiques et considérations entomologiques sur quelques insectes coléoptères, compris dans les familles des Dermestins, des Byrrhiens, des Acanthopodes et des Leptodactyles. Appareil génital. (Ibid., Sér. 2, Zool. i, 1834, pp. 76–82, 2 Pls.)
—— Résumé des recherches anatomiques et physiologiques sur les Hémiptères. (Ibid., Sér. 2, i, pp. 232–239.)
—— Mémoire sur les métamorphoses et l’anatomie de la Pyrochroa coccinea. Appareil génital. (Ibid., Sér. 2, Zool., xiii, pp. 337–339, 1 Pl.)
—— Histoire des métamorphoses et de l’anatomie des Mordelles. (Ibid., Sér. 2, xiv, pp. 235–238, 1 Pl.)
—— Anatomie générale des Diptères. Appareil génital. (Ibid., Sér. 3, Zool., i, 1844, pp. 250–264.)
—— Histoire des métamorphoses et de l’anatomie du Piophila petasionis. Appareil génital. (Ibid., Sér. 3, Zool., i, 1844, pp. 378–386.)
—— Études anatomiques et physiologiques sur les insectes diptères de la famille des Pupipares. Appareil génital. (Ann. Sc. nat., Sér. 3, Zool., iii, 1845, pp. 73–93, 2 Pls.)
—— Recherches sur l’anatomie et l’histoire naturelle de l’Osmylus maculatus. Appareil génital. (Ibid., Sér. 3, Zool., ix, 1848, pp. 349–356, 1 Pl.)
—— Recherches anatomiques sur les Hyménoptères de la famille des Urocerates. Appareil génital. (Ibid., Sér. 4, Zool., i, 1854, pp. 216–234, 1 Pl.)
—— Fragments d’anatomie entomologique. Sur les ovaires du Nemoptera lusitanica. (Ibid., Sér. 4, viii, 1857, pp. 9–10, 1 Pl.)
—— Fragments anatomiques sur quelques Élatérides. (Ibid., Sér. 4, viii, 1857, pp. 365–372, 1 Pl.)
—— Recherches anatomiques et considérations entomologiques sur les Hémiptères du genre Leptopus. Appareil génital. (Ibid., Sér. 10, 1858, pp. 356–362, 1 Pl.)
—— Recherches anatomiques sur l’Ascalaphus meridionalis. Appareil génital. (Ibid., Sér. 4, xiii, 1860, pp. 203–206, 1 Pl.)
—— Sur l’appareil génital male du Coræbus bifasciatus. (Thomson’s Archiv Ent., 1857, i, pp. 378–381.)
Suckow, F. W. L. Geschlechtsorgane der Insekten. (Heusinger’s Zeitschr. f. organ. Physik., 1828, ii, pp. 231–264, 1 Taf.)
Rathke, M. H. Miscellanea anatomico-physiologica. Fasc. 1. De Libellarum partibus genitalibus. Regiomonti, 1832, p. 38, 3 Pls.
Dutrochet, R. J. H. Observations sur les organes de la génération chez les pucerons. (Ann. Sc. nat., 1833, xxx, pp. 204–209.)
Doyère, L. Observations anatomiques sur les organes de la génération chez la Cigale femelle. (Ann. Sc. nat., 1837, vii, pp. 200–206, Fig.)
Siebold, C. Th. E. von. Ueber die weiblichen Geschlechtsorgane der Tachinen. (Wiegmann’s Archiv f. Naturgesch., 1838, iv, pp. 191–201.)
—— Ueber die inneren Geschlechtswerkzeuge der viviparen und oviparen Blattläuse. (Froriep’s Notizen, 1839, xii, pp. 305–308.)
—— Ueber das Receptaculum seminis der Hymenopteren-Weibchen. (Germar’s Zeitschr. f. Ent., 1843, iv, pp. 362–388, 1 Taf.)
Loew, H. Beitrag zur anatomischen Kenntniss der inneren Geschlechtsteile der zweiflügligen Insekten. (Germar’s Zeitschr. f. Ent., 1841, iii, pp. 386–406, 1 Taf.)
509—— Horæ anatomicæ, Abth. I. Entomotomien. Heft i-iii, Posen, 1841.
—— Beiträge zur Kenntniss d. inneren Geschlechtstheile der zweifl. Insecten. (Germar’s Zeitschr. f. Entomologie, iii, 1841, pp. 386–406, 1 Taf.)
Stein, F. Vergleichende Anatomie und Physiologie der Insekten. I, Monographie. Ueber die Geschlechtsorgane und den Bau des Hinterleibes bei den weiblichen Käfern, Berlin, 1847, i, pp. 139, 9 Taf.
Brauer, F. Beitrag zur Kenntniss des inneren Baues und der Verwandlung der Neuropteren. (Verhandl. d. zool. botan., Vereins in Wien, 1855, pp. 1–26, 5 Taf.)
Haliday, A. H. Note on a peculiar form of the ovaries observed in a hymenopterous insect, constituting a new genus and species of the family Diapriadæ. (Nat. Hist. Review, 1857, iv, pp. 166–174, 1 Pl.)
Laboulbène, A. Recherches sur les appareil de la digestion et de la reproduction du Buprestis manca. (Thomson’s Archiv Ent., 1857, i, pp. 204–236, 2 Pls.)
Lubbock, John. On the ova and pseudova of insects. (Phil. Trans. Roy. Soc., London, cxlix, 1860, pp. 341–369.)
Landois, H. Ueber die Verbindung der Hoden mit dem Rückengefäss bei den Insekten. (Zeitschr. f. wissens. Zool., xiii, 1863, pp. 316–318, 1 Taf.)
Leydig, F. Der Eierstock und die Samentasche der Insekten. (Nova Acta Acad. Leop.-Carol., xxxiii, 1867, pp. 88, 5 Taf.)
—— Beiträge zur Kenntniss des thierischen Eies im unbefruchteten Zustande. (Spengel’s Zool. Jahrbücher, 1889. Abth. f. Anat., iii, pp. 287–432, 7 Taf.)
Bessels, E. Studien über die Entwicklung der sexual Drüsen bei den Lepidopteren. (Zeitschr. wissens. Zool., xvii, 1867, pp. 545–563.)
Rajewsky. Ueber die Geschlechtsorgane von Blatta orientalis, etc. (Nachr. d. k. Gesellschaft d. Moskauer Universität, xvi, 1875. Testes of cockroach. In Russian; for abstract, see Hoffmann u. Schwalbe, Jahresbericht, 1875, p. 425.)
Brehm, Siegfr. Comparative structure of the reproductive organs in Blatta germanica and Periplaneta orientalis. (Horæ Ent. Soc. Rossicæ, St. Petersburg, viii, 1880.) (In Russian, male organs only.)
Cholodkowsky, N. A. Ueber die Hoden der Schmetterlinge. (Zool. Anzeiger, iii Jahrg., 1880, pp. 115–117.)
—— Ueber den Bau der Testikel bei Schmetterlingen. (Zool. Anzeiger, 1880, iii, pp. 214–215.)
—— Ueber die Hoden der Lepidopteren. (Zool. Anzeiger, 1884, pp. 564–568.)
—— Ueber den Geschlechtsapparat von Nematois metallicus. (Zeitschr. f. wissens. Zool., xliii, pp. 559–568, 1885.)
Tichomirow, A. Ueber den Bau der Sexualdrüsen und die Entwickelung der Sexualprodukte bei Bombyx mori. (Zool. Anzeiger, iii, 1880, pp. 235–237.)
Nusbaum, F. Zur Entwicklungsgeschichte der Ausführungsgange der Sexualdrüsen bei den Insekten. (Zool. Anzeiger, v, 1882, pp. 637–643.)
——On the developmental history of the efferent passages of the sexual glands in insects. Lemberg, 1884 (in Czech).
Berlese, Ant. Ricerde sugli organi genitali degli ortotteri. (Atti della R. Acad. dei Lincei. Ser 3, xi, 1882.) (Genital organs of European Orthoptera.)
Balbiani, G. Le Phylloxera du chêne et le Phylloxera de la vigne. Paris, 1884, pp. 45, 11 Pls.
—— Contribution à l’étude de la formation des organes sexuel chez les insectes. (Recueil Zool. Suisse, 1885.)
Schneider, Anton. Die Entwicklung der Geschlechtsorgane bei den Insekten. Zool. Beiträge, Breslau, i, 1885.
510Beauregard, H. Recherches sur les insectes vésicants. Suite. (Journal Anat. Phys., Paris, 1887, xxii Année, pp. 528–548, 1 Pl.; xxiii Année, pp. 124–163, 6 Pls.)
—— Les insectes vésicants. Paris, 1890. Chap. v, Appareil de la génération, pp. 103–159, Pls. 10–12.
Nassonow, N. Études morphologiques sur les Lepisma, Campodea et Podura. (Mém. Soc. Imp. Anthropologie et d’Ethn. Moscou, iii, 1887, pp. 85, 2 Pls., 68 Figs.)
—— Xenos rossii; seine Anatomie und Entwicklungsgeschichte. (Bull. de l’Université de Varsovie, 1892, pp. 74, 2 Taf.) (In Russian.)
—— Position des Strepsiptères dans le systeme selon les données du développement post-embryonal et de l’anatomie, p. 11, Warsaw, 1892.
Grassi, B. J. Progenitori dei Miriapodi e degli Insetti. Memoria vii, Anatomia comparata dei Tisanuri. (Atti d. R. Acad. de’ Lincei, Cl. scienc. e fis., Serie 4, iv, 1888, pp. 435–606, 5 Pls.)
Heymons, R. Ueber die hermaphroditsche Anlage der Sexualdrüsen beim Mannchen von Phyllodromia germanica. (Zool. Anzeiger, 1890, pp. 451–457.)
Koschewnikoff, G. Zur Anatomie der männlichen Geschlechtsorgane der Honigbiene. (Zool. Anzeiger, xiv, 1891, pp. 393–396.)
Verhoeff, C. (See p. 186.)
Ingenitzky, J. Zur Kenntniss der Begattungsorgane der Libelluliden. (Zool. Anzeiger, 1893, xvi Jahrg., pp. 405–407, 2 Figs.)
—— On the fauna and organization of dragon-flies of Russian Poland, 1893, 1 Pl. (In Russian.)
Escherich, K. Anatomische Studien über das männliche Genitalsystem der Coleopteren. (Zeitschr. f. wissens. Zool., lvii, pp. 620–641, 1894.)
Kluge, Max H. E. Das männliche Geschlechtsorgan von Vespa germanica. Inaug. Diss. Leipzig, 1895, pp. 1–45, 1 Taf.
Verson, E. La borsa copulatrice nei Lepidotteri (Atti e Mem. Accad. Sc. Lett. ed Arti, Padova, 1896, xii, pp. 369–372, 4 Pls.).
Klapálek, Fr. Über die Geschlechtstheile der Plecopteren, mit besonderer Rücksicht auf die Morphologie der Genitalanhänge. (Sitzungsb. k. Akad. Wissens. Wien. Math.-Naturw. Cl., cv, 1896, pp. 56, 5 Taf.)
Fenard, A. Recherches sur les organes complémentaires internes de l’appareil génital des Orthoptères. (Bull. Sc. France Belg., xxix, 1897, pp. 390–527, 528–533, 5 Pls.)
Consult also the Works of Ayres, Balfour, Burgess, Burmeister, Bütschli, Claus, Dzierzon, Gensch, Henking, Honert, Huxley, Kluge, Kramer, Landois, Leuckart (art. Zeugung), Leydig, Ludwig, Metschnikoff, H. Meyer, Minot, Müller, Pfitzner, Schneider, Seeliger, Scholz, Siebold, Suckow, Swammerdam, Tichomiroff, Wagner, Waldeyer, Weismann, Wheeler, v. Wielowiejski, Will, Witlaczil, and Ziegler.
b. Formation of the egg (oögenesis)
Claus, C. Beobachtungen über die Bildung des Insekteneies. (Zeitschr. wissens. Zool., xiv, 1864, pp. 42–54.)
Brandt, A. Ueber die Eiröhren der Blatta orientalis. (Mém. Acad. Imp. Scienc. de St. Petersbourg, Sér. 7, xxi, 1874, p. 30.)
—— Vergleichende Untersuchungen über die Eiröhren und die Eier der Insekten. (Nachr. d. Gesellsch. Freunde d. naturwiss. Moskau, xxiii, 1876; also xxiv, 1877, pp. 77–79.)
—— Das Ei und seine Bildungsstatte. Ein vergleichenden-morphologischer Versuch mit Zugrundelegung der Insecteneies. Leipzig, 1878.
511Kadyi, H. Beiträge zur Vorgänge beim Eierlegen der Blatta orientalis. Vorläufige Mittheilung. (Zool. Anzeiger, 1879, pp. 632–636.) (Formation of the egg-capsules of cockroach.)
Brass, Arn. Das Ovarium und der Eibildung und der ersten Entwicklungsstadien bei viviparen Aphiden. Halle, 1883. (Zeits. f. Naturwiss. in Halle, Jahrg. 1882.)
—— Zur Kenntniss der männlichen Geschlechtsorgane der Dipteren. (Zool. Anzeiger, 1892, pp. 178–180.)
Will, Ludvig. Zur Bildung der Eies und des Blastoderms bei den viviparen Aphiden. (Arbeiten Zool. Inst. Univ. Würzburg, vi, 1882, pp. 217–258.)
Korschelt, E. Zur Frage nach dem Ursprung der verschiedenen Zellenelemente der Insectenovarien. (Zool. Anzeiger, 1885, pp. 581–586, 599–605.)
Wielowiejski, H. V. Zur Morphologie des Insektenovariums. (Zool. Anzeiger, 1886, ix Jahrgang, pp. 132–139.)
—— Zur Kenntniss der Eibildung bei der Feuerwanze (Pyrrhocoris apterus). (Zool. Anzeiger, 1885, pp. 369–375.)
Blochmann, F. Ueber die Richtungskörper bei Insekteneiern. (Morph. Jahrb., 1887, xii, ix 544.)
Also the writings of Leydig (p. 509).
c. On the spermatozoa
Treviranus, G. R. Ueber die organischen Körper des tierischen Samens und deren Analogie mit dem Pollen der Pflanzen. (Zeitschr. f. d. Physiologie, von F. Tiedemann, G. R. und L. C. Treviranus, 1835, v, pp. 136–153, 2 Taf.)
Siebold, C. Th. E. von. Ueber die Spermatozoen der Crustaceen, Insekten, Gasteropoden und einiger anderer wirbellosen Tiere. (Müller’s Archiv f. Anatomie, 1836, pp. 13–52, 2 Taf.)
—— Fernere Beobachtungen über die Spermatozoen der wirbellosen Tiere. (Ibid., 1836, p. 232; 1837, pp. 381–432, 1 Taf.)
—— Ueber die Spermatozoen der wirbellosen Tiere, iv. (Ibid. 1837, pp. 392–433.)
—— Lange Lebensdauer der Spermatozoen bei Vespa rufa. (Wiegmann’s Archiv f. Naturgesch., 1839, v, pp. 107, 108.)
—— Ueber die Spermatozoiden der Locustinen. (Nova Acta Acad. Leop.-Carol., 1845, xxi, pp. 249–274, 1 Pl.)
Kölliker, A. Beiträge zur Kenntnis der Geschlechtsverhältnisse und der Samenflüssigkeit wirbelloser Tiere, nebst einem Versuch über das Wesen und die Bedeutung der sogenannten Samentiere, Berlin, 1841, pp. 88, 3 Taf.
—— Die Bildung der Samenfaden in Bläschen als allgemeines Bildungsgesetz. (Neue Denkschr. d. allg. Schweiz. Ges., viii, 1847, pp. 28, 3 Taf.)
—— Physiologische Studien über die Samenflüssigkeit. (Zeitschr. f. wissens. Zool., vii, 1856, pp. 201–272, 1 Taf.)
Yersin, A. Observations sur le Gryllus campestris. (Bull. Soc. Vaudoise sc. nat., 1853, iii, pp. 128.)
Lespès, Ch. Mémoire sur les spermatophores des grillons. (Ann. Sc. nat., Sér. 4, iii, 1855, pp. 366–377, 1 Pl.; iv, pp. 244–249, 1 Pl.)
Landois, H. Entwicklung der büschelförmigen Spermatozoiden bei den Lepidopteren. (Schultze’s Archiv f. Anat. u. Physiol., 1866, pp. 50–58, 1 Taf.)
Bütschli, O. Vorlaufige Mitteilungen über Bau und Entwicklung der Samenfaden bei Insekten und Crustaceen. (Zeitschr. f. wissens. Zool., xxi, 1871, pp. 402–415.)
512Bütschli, O. Nähere Mitteilungen über die Entwicklung und den Bau der Samenfaden der Insekten. (Ibid., xxi, 1871, pp. 526–534, 2 Taf.)
La Vallette St. George, A. V. Ueber die Genese der Samenkörper, III. Mitteilung. (Archiv f. Mikroscop. Anat., 1874, x, pp. 495–504, 1 Taf.)
—— Spermatologische Beitrage: II. Mitteilung (Ibid., 1886, xxvii, pp. 1–122 Taf.). IV. Mitteilung (Ibid., 1886, xxviii, pp. 1–13, 4 Taf.) V. Mitteilung (Ibid., 1887, xxx, pp. 426–434, 1 Taf.)
Schneider, A. Das Ei und seine Befruchtung, pp. 88, 10 Taf.; Arthropoden, pp. 57–68 und 79. Taf. 8–10, 1883.
Wielowiejski, H. de. Observations sur la spermatogénèse des Arthropodes. (Archiv Slav. de Biologie, 1886, ii, pp. 28–36.)
Ballowitz, E. Zur Lehre von der Struktur der Spermatozoen. (Anat. Anzeiger, i Jahrg., 1886, pp. 363–376.)
—— Untersuchungen über die Struktur der Spermatozoen, zugleiche in Beitrag zur Lehre von feineren Bau der kontraktilen Elemente. Die Spermatozoen der Insekten. I. Coleopteren. (Zeitschr. f. wissensch. Zool., 1, 1890, pp. 317–407, 4 Taf.)
—— Zu der Mittheilung des Herrn Professor L. Auerbach in Breslau über merkwürdiger Vorgänger am Sperma von Dytiscus marginalis. (Anat. Anzeiger, 1893, viii Jahrg., pp. 505–506.)
—— Die Doppelspermatozoen der Dyticiden. (Zeitschr. f. wissensch. Zool., lxvi, 1895, pp. 458–499, 5 Taf.)
Beauregard, H. Note sur la spermatogénèse chez la cantharide. (Compt.-rend. Soc. Biol., Paris, 1888, iv, pp. 331–333.)
Gilson, G. Étude comparée de la spermatogénèse chez les Arthropodes, in La Cellule, Recueil de Cytologie et d’Histologie gén., i, 1888, 8 Pl.
Verson, E. Zur Spermatogenesis. (Zool. Anzeiger, xii Jahrg., 1889, pp. 100–103, Fig.)
—— La spermatogenesi nel Bombyx mori. (Padova, 1889, 25 pp. und 3 Taf.)
—— Zur spermatogenesis. (Zool. Anzeiger, xii, 1889, pp. 100–103.)
—— Zur spermatogenesis bei der Seidenraupe. (Zeitschr. f. wissens. Zool., lviii, 1894, pp. 303–313, 1 Taf.)
Henking, H. Ueber Reductionsteilung der Chromosomen in den Samenzellen von Insekten. (Internat. Monatsschr. f. Anat. und Phys., 1890, vii, pp. 243–248.)
—— I. Untersuchungen über die erste Entwicklungsorgänge in der Eiern der Insekten. II. Ueber Spermatogenese und deren Beziehung zur Eientwickelung bei Pyrrhocoris apterus. (Zeits. wissens. Zool., xlix, 1890, pp. 503–564; li, 1891, pp. 685–736.) III. Specielles und Allgemeines. (Ibid., 1892, liv, pp. 1–274, 12 Taf.)
Sabatier, A. De la spermatogénèse chez les Locustides. (Comptes rend. Acad. Paris, 1890, cxi, p. 797.)
Cholodkowsky, N. Zur Frage über die Anfangsstadien der Spermatogenese bei den Insecten. (Zool. Anzeiger, 1894, pp. 302–304.) See also Zool. Anzeiger, 1892, p. 179.
Auerbach, Leopold. Ueber merkwürdige Vorgänge am Sperma von Dytiscus marginalis. (Sitz. Ber. Akad., Berlin, 1893, pp. 185–203, 2 Figs.)
—— Zu dem Bemerkungen des Herrn Dr. Ballowitz betreffend das Sperma von Dytiscus marginalis. (Anat. Anzeiger, viii Jahrg., 1893, pp. 627–630.)
Toyama, K. On the spermatogenesis of the silkworm. (Bull, ii, No. 3, Coll. Agric. Imp. Univ. Tokyo, pp. 125–157, 1894, 2 Pls.)
Wilcox, E. V. Spermatogenesis of Caloptenus femur-rubrum and Cicada tibicen. (Bull. Mus. Comp. Zool., xxvii, 1895, pp. 32, 5 Pls.)
513—— Further studies on the spermatogenesis of Caloptenus femur-rubrum. (Ibid., xxix, 1896, pp. 193–202, 3 Pls.)
Wilson, Edmund B. The cell in development and inheritance. (New York, 1896.) Also the writings of Platner, Waldeyer.
d. On the paired genital efferent passages
Loew, H. Abbildungen und Bemerkungen zur Anatomie einiger Neuropterengattungen. (Linnæa Ent., 1848, pp. 345–385, 6 Taf.)
Meinert, F. Anatomia Forficularum, i, Kjöbenhavn, 1863, 1 Pl.
—— Om dobbelte Saedgange hos Insecter. (Naturhist. Tidsskrift, 3 Raekke, v, 1868, pp. 278–294.)
Palmén, J. A. Zur vergleichenden Anatomie der Ausführungsgänge der Sexualorgane bei den Insekten. (Morph. Jahrb., ix, 1883, pp. 169–176.)
—— Ueber paarige Ausführungsgänge der Geschlechtsorgane bei Insekten. Eine morphologische Untersuchung. Helsingfors, 1884, pp. 108 und 5 Taf.
Nusbaum, J. Zur Entwicklungsgeschichte der Ausführungsgänge der Sexualdrüsen bei Insekten. (Kosmos, Lemberg, 1884, ix Jahrg., pp. 256–266, 393–408, 462–474, 2 Taf. In Polish with résumé in German.)
Spichardt, C. Beitrag zur Entwickelung der männlichen Genitalien und ihrer Ausführgänge bei Lepidopteren. (Verhandl. d. naturwiss. Vereins zu Bonn, 1886, xliii Jahrg., pp. 1–34, 1 Taf.)
Jackson, W. H. Studies in the morphology of the Lepidoptera. I. (Zool. Anzeiger, xii Jahrg., 1889, pp. 622–626.) (See p. 389.)
Lowne, B. Th. On the structure and development of the ovaries and their appendages in the blow-fly (Calliphora erythrocephala). (Journ. Linn. Soc. London, 1889, xx, pp. 418–442, 1 Pl.)
See also Meinert (1897), Heymons (1897).
PART II. EMBRYOLOGY OF INSECTS
a. The egg
Fig. 482.—Female Dyticus, laying eggs: A, ovipositor extended. B, egg of Notonecta, attached to stem of rush. C, egg of Dyticus, laid in excavation in rush.—After Régimbart, from Miall.
Insects as a rule arise from eggs which are laid in a great variety of situations, those species which are viviparous being exceedingly few in number compared with the class as a whole. It is noteworthy that Leydig has found in the same Aphis, and even in the same ovary, an egg-tube producing eggs, while a neighboring tube was producing viviparous individuals.[77] The viviparous species are confined to certain May-flies, the Aphidæ, Diptera (Sarcophaga, Tachinidæ, Œstridæ, and Pupipara), and to certain Coleoptera (Stylopidæ and some Staphylinidæ).
The number of eggs laid varies from a very few, as in the Collembola and in the Psocidæ, or 15 or even less in certain fossorial wasps, and from 20 to 35 in some locusts to many thousands in the social insects, the honey-bee laying by estimate over 1,000,000 eggs in the course of her life. Dr. Sharp thinks that from 50 to 100 may perhaps be taken as an average number for one female to produce. The eggs of insects with a complete metamorphosis are said by Brauer to be smaller in proportion to the parent than those laid by ametabolous or heterometabolous insects. In this respect the insects are paralleled by the birds, the highest forms laying smaller eggs than the water birds, ostrich, Apteryx, etc.
Fig. 483.—Eggs (e) of Hydrobius (?) and their capsules, from which the larva, Fig. 452, hatched.—Emerton del.
The egg, or ovum, when laid is not always ripe or perfect, but, as in those of ants, continues to grow after oviposition. Others are laid some time after the embryo has begun to form; and in the flesh-flies the larva hatches before the egg is deposited.
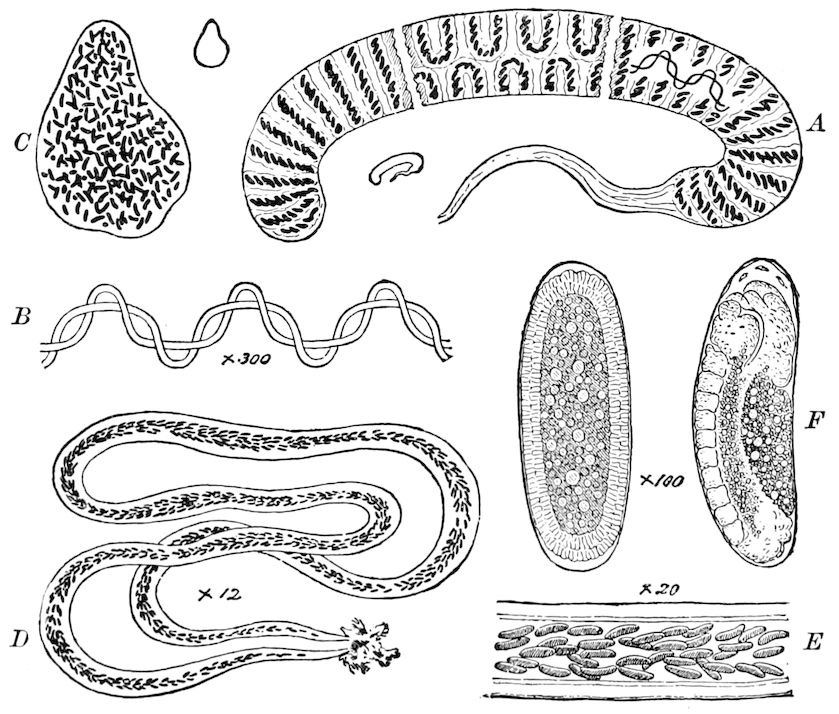
Fig. 484.—Egg-masses of Chironomus: A, string of eggs of C. dorsalis, divided into sections to show both sides. B, twisted fibres which traverse the string of eggs. C, egg-mass of Chironomus (sp). D, egg-mass of a third species. E, part of D, more highly magnified. F, developing eggs, two stages.—After Miall.
Insects as a rule instinctively lay their eggs near or upon objects destined to be the food of the larva; those of caterpillars on leaves, those of many flies on meat or carrion, those of Copris and other dung-beetles in dung, those of aquatic insects in water, while many oviposit in the earth or in plants (Fig. 482), or in the bodies of animals destined to be the hosts of the parasitic larvæ. As the eggs are preyed upon by mites and other animals, the contrivances and modifications of the mode of egg-laying, and the situations in which they are placed, are almost endless. Many insects lay their eggs in a mass, covered with a gummy substance; 517or those laid in the water, as the eggs of dragon-flies, caddis-flies, Chironomus (Fig. 484), etc., are enveloped by a jelly-like mass.
Fig. 485.—Egg-capsule of Periplaneta americana: a, side; b, end view; c, natural size.—After Howard and Marlatt, Bull. 4, Div. Ent. U. S. Dept. Agr.
The oötheca of the cockroach (Fig. 485) is a solid, dense case, which, after being carried about by the mother, can be left without harm in the crevices of the floors of houses. The oötheca of Mantis (Fig. 486) is formed by a large mass of frothy matter, which hardens and is attached to stems of plants.
Fig. 486.—Egg-capsules of Mantis carolina.—After Riley.
On the other hand, the female “walking-stick” (Diapheromera femoratum) drops her eggs, says Riley, loosely upon the ground, from whatever height she may happen to be, and “one hears a constant pattering, not unlike drops of rain, that results from the abundant dropping of these eggs, which, in places, lay so thick among and under the dead leaves that they may be scraped up in great quantities.” (Report for 1879.)
The eggs of the lace-winged flies are supported on pedicels, above the reach of ovivorous mites.
The female Chrysopa usually lays between 40 and 50 eggs. In one case, we observed that 18 egg-stalks were deposited, but there were only nine well-formed eggs in the batch, and nine eggless stalks, some only half the usual height, others with the knob of cement at the end to which the egg is ordinarily fastened. The eggs are evidently stuck on to the end of the pedicel after the latter has been formed, as, in one instance, an egg was glued to the stalk very much out of centre, the insect’s abdomen not having been aimed straight, so to speak, at the mass of cement.
Fig. 487.—Eggs of Chrysopa, with larva and fly.
The eggs of Rhodites are fixed to a long stalk thickened at the end; those of Inquilines and certain Chalcids (Leucospis gigas, Fig. 489, A) are also stalked; and the use of this stalk in the eggs of Cynips (E) is thought by Adler to be respiratory, while, also, he states that the egg-cavity communicates with the egg-stalk, so that a part of the egg-contents can pass into the latter, and this happens at the laying of each egg. The egg of certain ichneumons (Paniscus, Fig. 488) ends in a short stalk, which is inserted 518in the skin of the caterpillar destined to serve as the host of the parasite, the eggs, as stated by De Geer, being retained more firmly in the integument by the stalk so swelling as to form two knobs (Fig. 498, c).
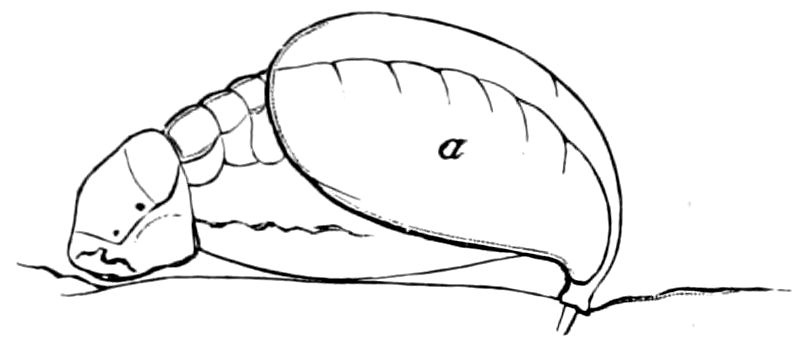
Fig. 488.—Young larva of Paniscus in position of feeding on the skin of a caterpillar: a, the egg-shell.—After Newport, from Sharp.
Certain Homoptera also have stalked eggs, as those of Psylla pyricola (Fig. 489, B), those of Aleyrodes citri (C, a, b), and of an allied form, Aleurodicus cocois (D), and those of Corixa (Fig. 493).
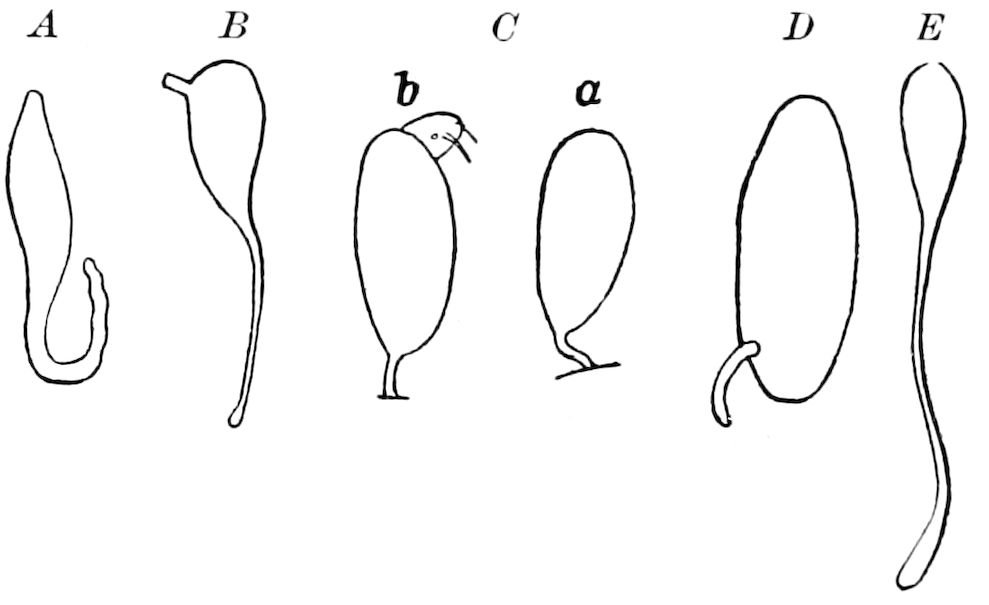
Fig. 489.—Stalked eggs: A, of a Chalcid (after Fabre); B, of Psylla (after Slingerland); C, of Aleyrodes; D, of Aleurodicus (after Riley and Howard); E, of Dryophanta scutellaris (after Adler).
Fig. 490.—Eggs of ox bot-fly, enlarged.—After Riley.
Reference should also be made to the eggs of lice, which are oval and attached to the hairs of their host. Those of the ox bot-fly (Hypoderma lineata) are usually placed four to six together, and fastened to a hair. The lower portion of the egg is admirably adapted for clasping a hair. “It consists of two lobes, forming a bulbous enlargement, which is attached to the egg by a broad, but rather thin, neck, so that, when the latter is viewed sidewise, it appears as a slender pedicel” (Fig. 490, a-d). (Riley in Insect Life, iv, p. 307.) The egg of another fly (Drosophila ampelophila, Fig. 491) bears a pair of long, slender appendages near the anterior end. “The egg is inserted into the soft pulp of the decaying fruit; these appendages leave the ovipositor last, and are spread out upon the surface of the mass. They, in this way, serve to keep the egg in place, and thus insure the emergence of the larva into the open air instead of into the more or less fluid mass in which the egg is situated. The larva issues from the egg just above the base of these appendages.” (Comstock.)
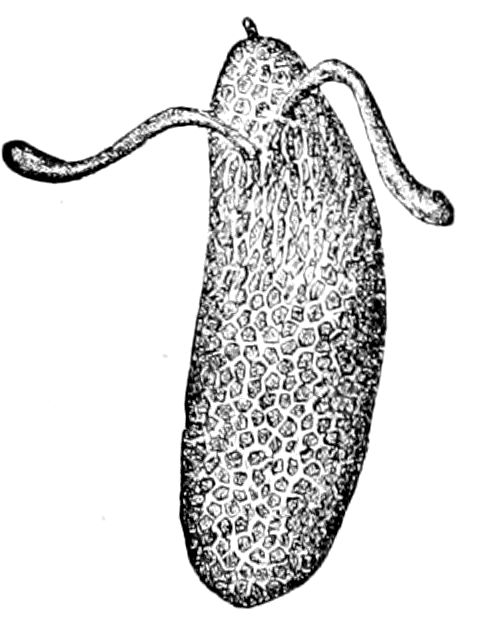
Fig. 491.—Egg of Drosophila.—After Comstock.
Mode of deposition.—The exact process of oviposition has been rarely observed, or at least not observed in detail, 519and further observations are much needed. In the cockroach (Phyllodromia), Wheeler has seen the eggs pass out of the oviduct and become arranged in the oötheca, in a way similar to that in the account published by Kadyi on Periplaneta.
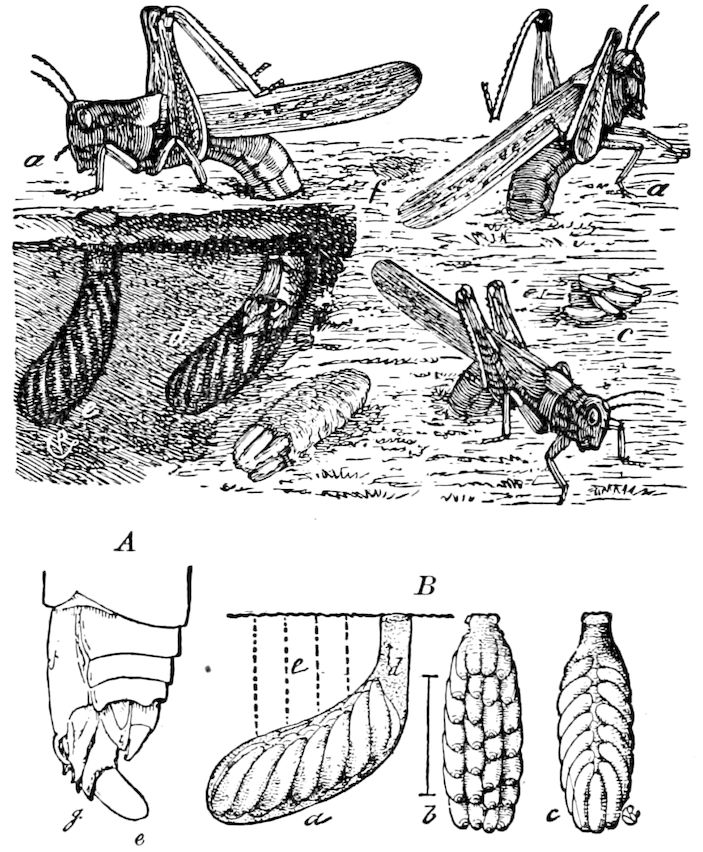
Fig. 492.—Rocky Mountain locust (aa) depositing its eggs (c); d, the earth partially removed, showing (e) an egg-mass already in place, and (d) one being placed; f shows where such a mass has been covered over. A, oviposition; j, position of oviduct; g, egg-guide; e, egg. B, egg-mass of the same; a, from side, b, from beneath, c, from above.—After Riley.
“When about to form the capsule, the female Blatta closes the genital armature, and the two folds of the white membrane which lines the oöthecal cavity close vertically in the middle line. Then some of the contents of the colleterial glands are poured into the chamber, and bathe the inner surface of the posterior wall. The first egg glides down the vagina from the left ovary, describes an arc, still keeping its germarium-pole uppermost, after having pressed the micropylar area against the mouth of the spermatheca, passes to the right side of the back of the chamber, and is placed perpendicularly two-thirds to the right of the longitudinal axis of the insect’s body. The next egg comes from the right ovary, describes an arc to the opposite side of the body, decussating with the path of the first egg, and is placed completely on the left side of the median line. The third egg comes from the left ovary, and is made to lie completely on the right side of the median line; and so the process continues, the ovaries discharging the eggs alternately, and each egg describing an arc to the opposite side of the capsule. The oöthecal chamber soon becomes too small to contain all the constantly accumulating eggs, so the anal armature opens and allows the end of the capsule to project. A raised line, the impression of the edges of the white membrane, runs down the end of the capsule. The last egg deposited comes from the right ovary, and lies two-thirds on the left, and one-third to the right, of the median line. As soon as the egg is laid, a further discharge from the colleterial glands spreads over the vaginal or anterior wall of the cavity, and becomes evenly continuous with the secretion which has before been spread over the back and the sides of the capsule by the white membrane.
“The crista, a cord-like ridge running the full length of the dorsal surface of the capsule, is a thick-walled tube, either half of which is formed by the edge of the side walls of the capsule split into two laminæ. The rhythmical clasping 520of the three pairs of palpi which guard the vaginal opening is registered in an exquisite pattern on the inner face of either half of the crista.”[78]
The mode of oviposition in the locust has been fully described by Riley, who states that the eggs pass down and out of the oviduct, and “guided by a little finger-like style” (Fig. 298), they pass in between the horny valves of the ovipositor, and issue at their tips amid the mucous fluid which forms the egg-capsule (Fig. 492).
Vitality of eggs.—It is well known that the eggs of phyllopod and other fresh-water Crustacea have wonderful vitality, withstanding extreme dryness for several years, at least from two to ten. Such cases are unknown among insects. It has been observed, however, by T. W. Brigham, and also by L. Trouvelot, that the eggs of the walking-stick (Diapheromera femorata) for the most part hatch only after the interval of two years.[79]
The eggs of Bittacus are said by Brauer to lie over unhatched for two years; indeed, the first condition of their hatching is a complete drying of the earth in which the eggs lie, the second is a succeeding thorough wetting of the ground in spring.
Appearance and structure of the ripe egg.—The eggs of insects are on the whole rather large in proportion to the size of the parent, especially so in many minute forms, as the fleas, lice, etc.
Their general shape is spherical or oval, often cylindrical; where the eggs are long and cylindrical a dorsal and ventral side can be distinguished (Fig. 502). They are in the Tortricidæ and Limacodid moths flattened, thin, and scale-like. In the eggs of locusts and grasshoppers, as well as certain Diptera, the ventral side of the embryo corresponds to the convex side, and the concave side of the egg to the dorsal region of the embryo (Figs. 502 and 493).
There is an anterior and posterior end or pole, the anterior end being that which in the body of the parent lies towards her head, or towards the upper or distal end of the ovarian tube. Towards this end lies in the later stages of embryonic life the head-end of the embryo, while the posterior end of the embryo is turned towards the hinder pole of the egg (Figs. 493 and 520).
The egg-shell and yolk-membrane.—The ripe egg is protected by two membranes: 1, an inner or vitelline membrane or oölemma (dh) (Fig. 500, d), produced in the egg by a hardening of the outer layer, and 2, the outer or chorion (c), which is secreted by the cells of the ovarian follicle. The latter is divided into two layers: an inner, the endochorion, and an outer, the exochorion.
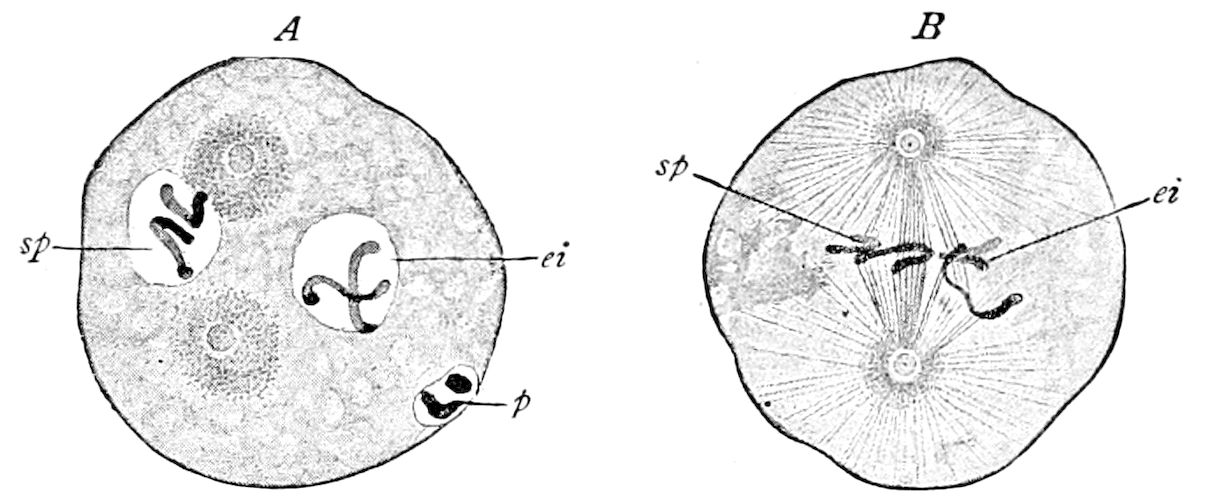
Fig. 503.—Fertilization of the egg of a round-worm (Ascaris megalocephala): A, the ends (centrosomes) of the spindle formed. B, the spindle completed; sp, sperm-nucleus, with its chromosomes; ei, egg-nucleus; p, polar bodies.—After Boveri, from Field’s Hertwig.
Fig. 493.—Eggs of Corixa: A, early stage before formation of the embryo, from one side. B, the same viewed in the plane of symmetry. C, the embryo in its final position; a, anterior, p, posterior, end; l, left, r, right, v, ventral, d, dorsal, aspect. (The letters refer to the final position of the embryo, which is nearly diametrically opposite to that in which it first develops); m, micropyle; p, pedicle.—After Metschnikoff, from Wilson.
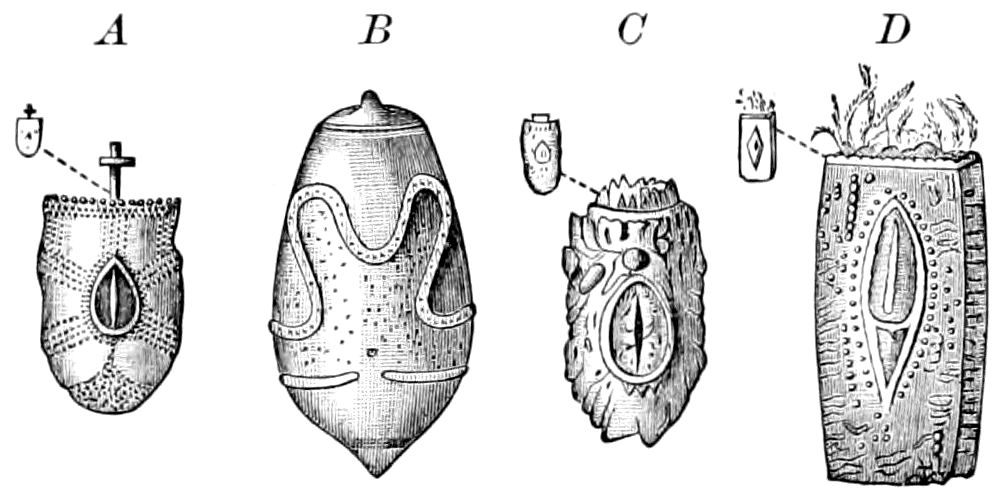
Fig. 494.—Eggs of Phasmidæ: A, Lonchodes duivenbodi. B, Platycrania edulis. C, Haplopus grayi. D, Phyllium siccifolium.—After Kaup, from Sharp.
While the yolk-membrane is usually a completely homogeneous, thin, structureless membrane, the chorion or shell of the egg is usually covered with a network of ridges enclosing polygonal areas, varying in shape according to the species or genus. These external markings are due to the impress of the cellular structure of the epithelium of the ovarian follicle.
In the chorion of the cockroach the surface appears to be finely granular, the finest granules being arranged in large, more or less regularly hexagonal areas, which are bounded by narrow, dark spaces, containing somewhat larger though less dense granules. The surface of the eggs of certain Phasmids are variously sculptured (Fig. 494).
The true structure of the chorion can only be, as Wheeler observes, seen in cross-sections, as shown by Blochmann, and also by Wheeler. The chorion consists of two chitinous laminæ kept in close apposition by means of numerous minute trabeculæ or pillars. It is the ends of these pillars that look like granules. In the spaces between the hexagonal areas, the trabeculæ are more scattered and individually thicker than those of the hexagons.
Fig. 495.—Egg of cotton-worm moth, Aletia: a, top view, showing the micropyle.—After Comstock.
Fig. 496.—Egg of Danais archippus.—After Riley.
These markings are of singular beauty and complexity in the eggs of many Lepidoptera, whose ova are variously ribbed, forming a beautiful fretwork of raised lines (Figs. 495 and 496), while in the Diptera and Hymenoptera the chorion is less solid, and usually smooth under low powers. The exochorion of the egg of the house and meat fly (C. vomitoria) is pitted with elongated hexagonal depressions, which cross the egg transversely. That of the honey-bee is also divided into long hexagonal areas (Fig. 497).
Fig. 497.—Egg with embryo of honey-bee, × 40: ch, chorion; ga, ganglia; s. ga, brain; jm, jaw-muscles forming; c, œsophageal collar; fb, fore intestine; mb, mid-intestine; ab, hind-intestine.—After Cheshire.
Fig. 498.—Micropyle (Mk) of eggs; a, of a fly, Antomyia; b, Drosophila cellaris; c, stalked egg of Paniscus testaceus.—After Leuckart, from Perrier.
When the eggs are deposited in exposed places, and remain in such situations for several days, or weeks, or even through the winter, the shell is either solid and strengthened by the ribs and ridges; or the shell, if of winter eggs, is unornamented, and is dense and solid, to resist extremes in temperature or the attacks of egg-eating birds, mites, etc.
The micropyle.—This is an opening or canal, or, as in most insects, a group of canals situated at the anterior end of the egg for the entrance of the spermatozoa during the process of fertilization of the ovum (Fig. 498). In Acrydians, however, the micropyle is situated at the posterior end of the egg. The micropyle (Fig. 499) is a complicated apparatus within whose circumference the vitelline membrane appears to be firmly attached to the chorion, so that the perforation passes through the chorion as well as the yolk-membrane.
The micropyles of the cockroach are probably as simple and generalized as in any insect. Wheeler states that they are in Phyllodromia scattered over the end of the egg, “over a quadrant of the upper hemisphere, where the beautiful hexagonal pattern of the chorion gives away to an even trabeculation.” The micropyles are wide-mouthed, very oblique, funnel-shaped canals, perforating the chorion, the apertures of the funnels appearing under a low power as 523clear, oval spots, the long axis of which is parallel to the long axis of the egg.
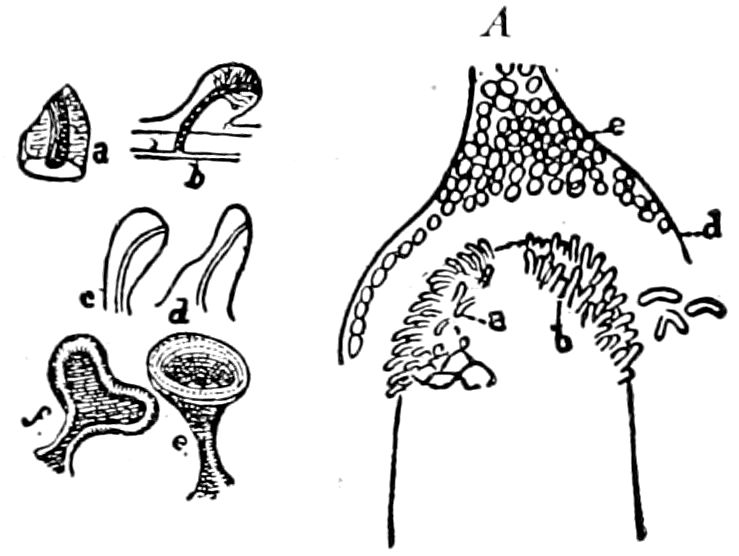
Fig. 499.—a, fragment of a micropylar papilla, showing its lumen; b, optical section of another papilla, in this one the lumen extends to the vitelline membrane, but does not pass beyond it; c, d, e, and f, papillæ of different forms. A, anterior end of an ovarian egg, showing mode of growth of the micropylar papillæ: a, b, two successive stages; c, surface view of modified papillæ from the lower edges of the cap; d, tunica propria of the ovariole; e, remnant of the cell-mass that secreted (?) the micropylar cap.—After Ayers.
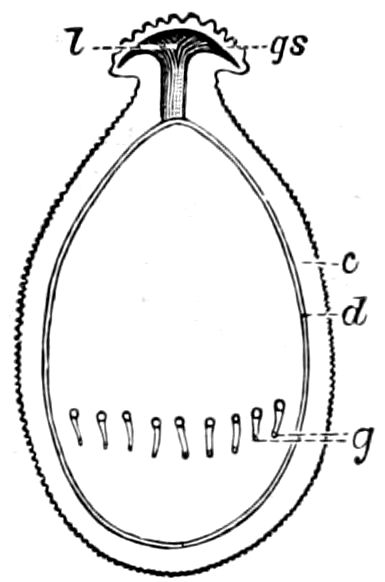
Fig. 500.—Egg of Perla maxima: c, chorion; d, oölemma; gs, glass-like covering of micropyle; l, cavity under same; g, canals penetrating chorion.—After Imhof, from Sharp.
“With a higher power the tube of each funnel is clearly visible as a thin canal which dilates rapidly into the large oval aperture on the outer face of the chorion. The narrow tube is sometimes fully as long as the large orifice. The micropylar perforations are all directed from the germarium to the vaginal pole of the egg. Hence a line, the hypothetical path of the spermatozoön, drawn through one of these oblique micropyles, and continued into the egg, would strike the equatorial plane. The female pronucleus, as we shall see further on, moves in this plane.” (Wheeler, p. 289.)
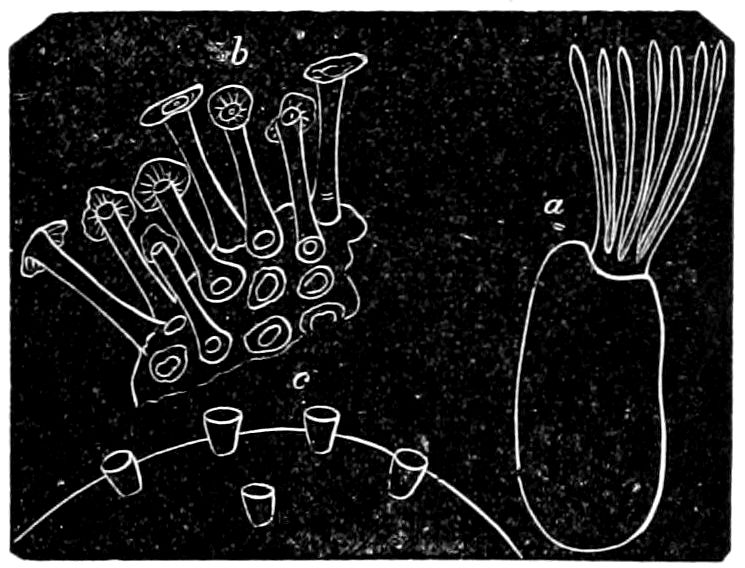
Fig. 501.—Micropyles: a, of Nepa cinerea; b, of Locusta viridissima; c, of a bug (Pyrrhocoris apterus).—From Gerstäcker.
The micropylar region is generally, at least in Orthoptera and Odonata, covered by a gelatinous cap (Figs. 499 and 500, gs), which may form a covering membrane which extends over a large part of the egg, or may envelop the entire outer surface. In some cases micropyles are scattered over the entire surface of the egg, but usually the perforation is situated at the end, and is often guarded by raised processes, either one or several, like bristles, or toadstools, etc., these being especially characteristic of the eggs of certain Hemiptera (Nepa, Fig. 501, a, and Ranatra), or the region is variously sculptured, as in the eggs of butterflies. In the micropylar apparatus of Œcanthus the papillæ have a distinct lumen (Fig. 499), or a channel for the ingress of the male filament.
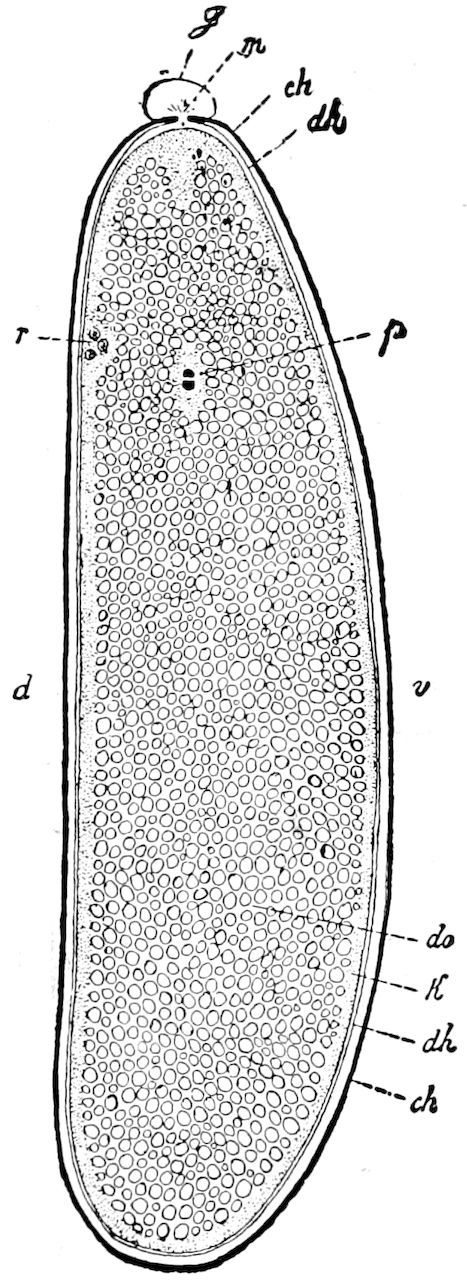
Fig. 502.—Diagrammatic median section through egg of Musca in stage of fertilization (incorporating the figures of Henking and Blochmann): ch, chorion; d, dorsal; v, ventral side of the egg; dh, yolk-membrane; do, nutritive yolk; g, gelatinous cap over the micropyle (m); K, outer layer of plasma (Keimhautblastem); p, male and female pronucleus before copulation; r, directive body (Richtungskörper).—After Korschelt and Heider.
Another use of the micropylar apparatus noticed by Ayers in the egg of the tree-cricket is that it “serves as a thick, roughened plate, against which the insect may push when ovipositing, without injury to the egg, and without danger that the ovipositor slips from its place.” In Chrysopa eggs the micropyle forms a conspicuous button-like knob, resembling the finely milled head of a certain kind of screw.
Internal structure of the egg.—The egg-contents are surrounded by an outer layer of protoplasm or formative yolk, which is separate from the inner parts of the egg (Fig. 502, do), the latter being mostly composed of the nutritive yolk-element. The superficial protoplasmic layer, called by Weismann Keimhautblastem (K) is, in a few cases, afterwards entirely lost, but in most instances forms a very thin layer of clear protoplasm, slight in extent compared with the yolk-mass within.
The eggs of insects are rich in yolk, only certain eggs, such as those of the Aphides and the egg parasites (Proctotrypidæ) being poor in yolk. The eggs of heterometabolous insects have been said by Brauer to contain relatively more yolk than those of the Metabola, particularly the Diptera; though, as Wheeler observes, this rule has some exceptions, the eggs of the 17–year Cicada being very numerous and small.
This he thinks is a greater advantage to the insect than the production of a few large eggs, “when we consider the extremely long period of larval life and the vicissitudes to which the larvæ may be subjected during all this time.” “Similarly, Meloë angusticollis produces a large number of very small eggs, while the eggs of the smaller beetles (Doryphora, e.g.) are much larger. But 525Meloë is a parasitic form, and probably only a few of its many offspring ever succeed in gaining access to the egg of the bee.”
In the eggs of Chrysopa the yolk-granules are remarkably small, so that the primitive band is in strong contrast to the yolk in color and density. When crushed, the yolk does not flow out as a liquid, but in a pasty mass, and we have questioned whether, as in the eggs of Limulus, whose yolk is solid with fine granules, the denseness of the yolk is not connected in the way of cause and effect with their exposed situation.
The central or yolk-mass (Fig. 502, do) consists chiefly of rounded masses of yolk, with fat-globules, between which extends a fine network of protoplasm.
The elements of the yolk are spherical and strongly refractive, by pressure becoming polygonal structureless homogeneous bodies.
The germinal vesicle of the ripe insect-egg lies in the centre of the yolk, where it appears as a large vesicle-like cell-nucleus containing a few chromatin elements.
b. Maturation or ripening of the egg
Before the eggs of animals can be fertilized, they require in some observed cases, and probably in animals in general, to undergo a series of changes, which, as observed in the starfish, etc., consists in the replacement of the germinal vesicle by a very much smaller egg-nucleus, and also at the same time the construction at one pole of the egg of the directive or polar bodies (Fig. 502, r). Towards the end of the ripening process of the insect egg this vesicle, according to Blochmann, passes to the dorsal surface of the egg, and is transformed into the directive spindles (Richtungspindel).
c. Fertilization of the egg
The egg next requires the penetration and admission into the yolk-interior of a spermatozoön.
This process is essentially in insects, as in other animals, the fusion of the sperm-nucleus with the nucleus of the egg. Under normal conditions but a single spermatozoön is required for fertilization. As shown by Hertwig, in the sea-urchin, after the spermatozoön has penetrated into the egg, the head, and the small rounded body, called a centrosome, can still be recognized, but the tail becomes fused with the yolk of the egg. In the protoplasm of the egg (called cytoplasm) the achromatic end of the sperm-nucleus gives rise to 526conspicuous rays, like those observed in ordinary cell-division. Preceded by these rays, the sperm-nucleus or male pronucleus (Fig. 502, p) moves towards the nucleus of the egg, and finally fuses with it, thus forming a new single nucleus. This latter, which is called “the cleavage nucleus,” rapidly forms a nuclear or “cleavage spindle” (Fig. 503). This act gives an impulse to the cleavage of the egg, which is the first step in the formation of the embryo. All these changes have yet to be worked out in detail in insects by microscopic sections of the egg, whose generally hard and opaque egg-shells present great obstacles to such work.
d. Division and formation of the blastoderm[80]
In insects as in most other Arthropoda the segmentation of the yolk is superficial and not total. The ovum is centrolicithal, i.e. the yolk is concentrated at the centre of the egg, and surrounded by a peripheral layer of transparent protoplasm (the Keimhautblastem).
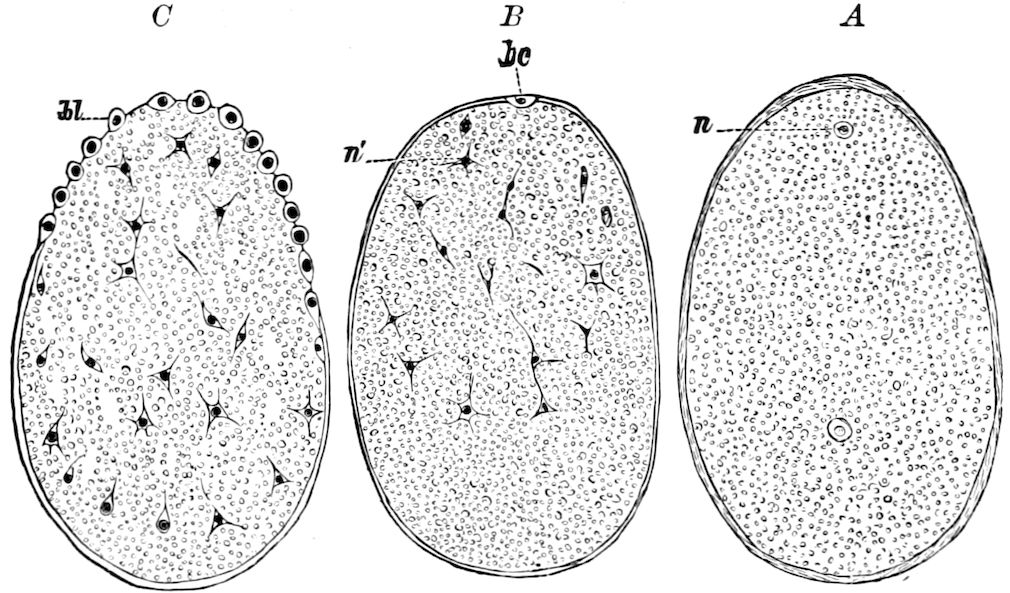
Fig. 504.—Formation of the blastoderm of Pieris cratægi: A, longitudinal section through the egg, with two masses of protoplasm in the yolk. B, a blastoderm-cell at the upper end. C, a later stage, with more blastoderm-cells.—After Bobretsky.
The first step in segmentation is the movement of the first division-nucleus (i.e. that in the fertilized egg arising from the union of the sperm-nucleus with the female pronucleus) towards the interior of the egg in order to multiply itself by the mode of indirect nuclear division (Figs. 504, A, and 507).
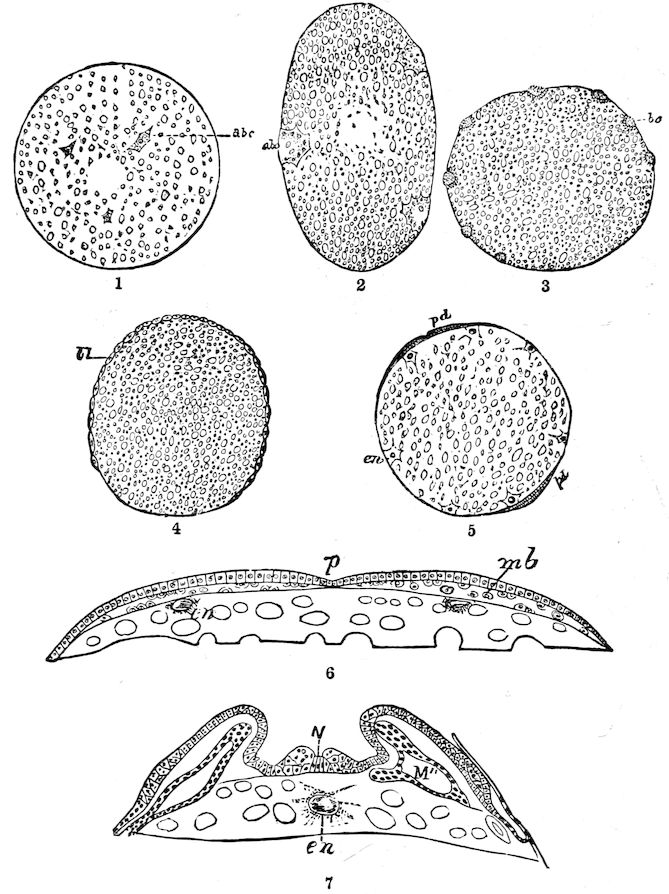
Fig. 505.—Embryology of the mole-cricket: 1, egg in which the amœboid nuclei (abc) are moving toward the surface; 2, egg in which the nuclei (abc) have reached the surface, and show an active nucleus-formation; 3, the blastoderm-cells have no nucleus, and are placed at equal distances apart; 4, the blastoderm-cells now forming a continuous layer; 5, cross-section of the egg with blastodermic disk, also showing the disposition of the endodermal cells; 6, cross-section of the blastodermic disk, with the myoblast cells (mb) already formed; 7, cross-section through the thorax of the embryo, the body-cavity extended into the limbs.
- abc,
- amœboid blastodermic cells.
- bc,
- blastoderm-cells.
- bl,
- blastoderm.
- en,
- endodermal cells.
- M″,
- cavity of the myoblast.
- mb,
- myoblast cells.
- N,
- nerve-furrow.
- P,
- primitive groove.
- pd,
- primitive disk.
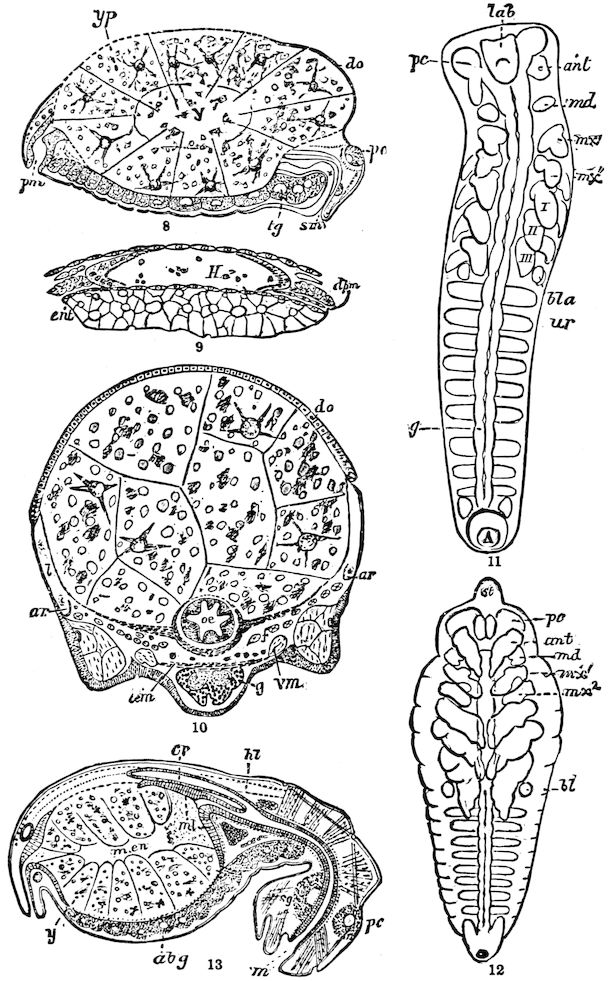
Fig. 505 concluded.—Later stages in the embryology of the mole-cricket: 8, longitudinal section of the embryo; the yolk-pyramids (yp) form a common inner yolk-mass (y). 9, section through the heart; H, cavity of the heart; the two halves of the heart-sinuses having united dorsally, ventrally they are still open and are bounded by the walls of the mesenteron. 10, cross-section of an embryo, showing the blood-lacunæ separated on the back by the dorsal organ (do); the intestinal fasciated layer (Darmfaserblatt) has not completely enclosed the yolk. 11, embryo completely segmented, with the rudiments of the appendages, labrum (lab), and nervous ganglia (pc-ng). 12, a more advanced embryo, showing the stomodæum (st) indicated as a frontal protuberance. 13, section through the recently hatched larva, showing the cells of the mesenteron or chyle-stomach, and the cellular layer on the front surface, also the proventriculus or crop.
- ant,
- antenna.
- ar,
- arterial sinus.
- bl,
- blastoderm.
- bla,
- abdominal vesicles.
- cr,
- proventriculus, or crop.
- dm,
- ventral diaphragm.
- do,
- dorsal organ.
- d pm,
- dorsal diaphragm.
- ent,
- enteric layer.
- f,
- fat-body.
- g,
- ventral ganglion.
- H, ht,
- heart.
- l,
- lacuna.
- m,
- mouth.
- md,
- mandible.
- m.en,
- mesenteron.
- mx′,
- 1st maxilla.
- mx″,
- labium, or 2d maxilla.
- ml,
- leaf-like portion of mesenteron.
- oe,
- œsophagus.
- pc,
- procerebrum.
- pm,
- proctodæum.
- sg,
- subœsophageal ganglion.
- sm,
- stomodæum.
- tg,
- thoracic ganglion.
- vm,
- ventral muscle.
- y,
- yolk.
- yp,
- yolk-pyramids.
- I,
- 1st pair of feet.
- II,
- 2d pair of feet.
- III,
- 3d pair of feet.
The origin of numerous division-nuclei as the offspring of the first has been observed to take place in the eggs of those insects (Aphides, Cecidomyia, and Cynips) which have a slight amount of yolk. Yet in the large, ordinary eggs of insects with an abundance of yolk there is no doubt, say Korschelt and Heider, that these numerous division-nuclei, which soon after the process of oviposition are scattered within the egg between the yolk-spheres, and are enveloped by a star-shaped protoplasmic layer, and which constitute the formative elements of the blastoderm,—there is no doubt but that they have practically arisen through indirect nuclear division from the first division-nucleus.
The process of formation of the blastoderm in ordinary eggs with abundant yolk was first observed by Bobretsky in the eggs of a moth (Porthesia) and Pieris, also by Graber, and more recently by Blochmann in Musca, and by Heider in Hydrophilus.
In the earliest stage observed by Bobretsky there first appear after fertilization a few (the smallest number four) cell-like, minute amœboid masses of protoplasm, each with a distinct nucleus. A few (one at least) of these bodies gradually pass out of the centre of the yolk to the surface of the egg (Fig. 504, A, n), these becoming larger and rounder, and from one or two of these nuclei (B, bc) the blastoderm originates (C, bl). Those nuclei remaining in the yolk increase in number and afterwards become the nuclei of rounded masses of yolk-granules, forming the so-called yolk-spheres which Bobretsky regards as true cells.
To the few blastoderm cells situated on the upper end of the egg are added others which continue to pass from the yolk to the periphery, and then the blastoderm spreads out farther and farther from the upper end of the egg until finally it covers or envelops the whole yolk. This layer of cells is called the blastoderm.
As to the origin of the primitive amœboid cells, Bobretsky is in doubt, but is disposed to think that they are the result of the subdivision of the germinative vesicle or nucleus of the ovarian egg-cell. In this connection may be quoted the observations of Graber, who states that an examination of the ovarian cell at an early period has revealed the presence, in the centre of the yolk, of a number of amœboid cells, which appear to have been formed by the division of the germinal vesicle. These “primary embryonic cells” have a relatively large nucleus and a number of nucleoli. Several may be seen to unite with one another by means of their pseudopodia, and they may also be observed to undergo division. With this account may be compared the results obtained by Korotneff in his work on the embryology of the mole-cricket (Fig. 505).
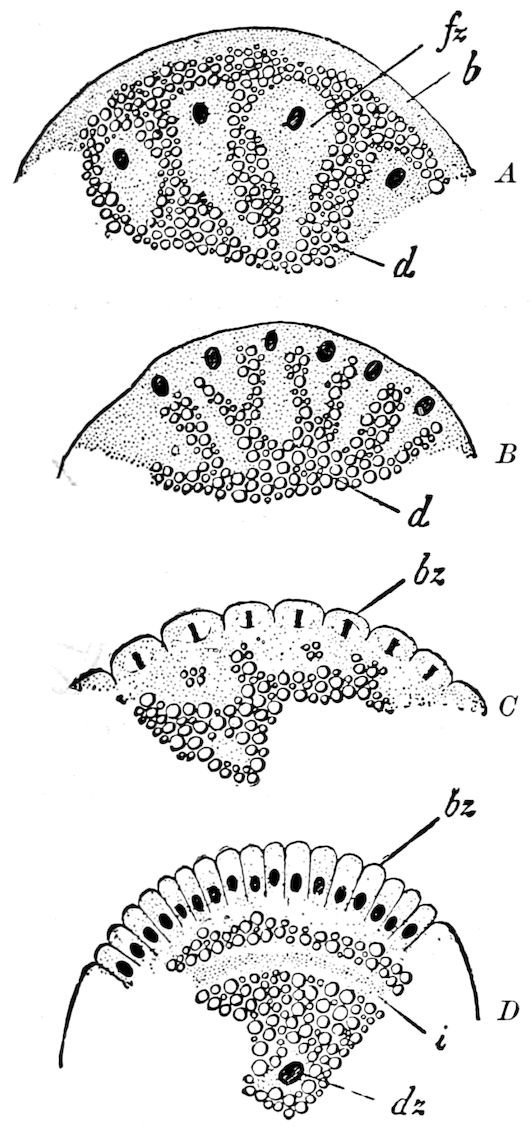
Fig. 506.—Four successive stages in the formation of the blastoderm of Calliphora vomitoria (the figures represent segments of cross-sections through the fly’s egg): A, the nuclei of the division-cells have arranged themselves parallel with the outer surface of the egg. B, the division-cells fused with the “keimhautblastem.” C, the outer surface becomes furrowed by indentations; all the nuclei of the blastoderm-cells in process of division. D, the blastoderm-cells form a high cylinder-epithelium: b, “keimhautblastem”; bz, blastoderm-cells; d, nutritive yolk; dz, yolk-cell; fz, so-called division-cell; i, inner “keimhautblastem.”—After Blochmann, from Korschelt and Heider.
The result of these and of later observations, especially those of Blochmann on Musca, and those of Heider on Hydrophilus, show that the division-nuclei lie near the centre of the egg, along the longitudinal axis (Fig. 507, A). Each of these nuclei is enveloped by a star-shaped mass of protoplasm, and on the whole resembles a wandering amœboid cell. These isolated masses of protoplasm are all connected by a fine network of rays, which unite to form within the yolk a syncytium. Afterwards, in the later stages, these division-cells, as they may be, though somewhat incorrectly, regarded, move nearer the periphery and arrange themselves into a plane parallel with the surface (Figs. 506, A, 507, B). Continuing to divide, they reach the surface and fuse with the peripheral protoplasmic layer (Figs. 506, B, 507, C). Then follows the division into single cell-territories (Figs. 506, B, 507, C), corresponding to the division-nuclei, through the appearance of furrows which pass in from the outer surfaces of the egg into the interior and gradually penetrate the entire “keimhautblastem.” In this way the surface of the egg is covered with an epithelium (blastoderm). In many insects the so-called inner “keimhautblastem” (Fig. 506, D, i) is formed by the separation of a layer of protoplasm which contains larger granules and are accumulated between the blastoderm and the upper surface of the central nutritive yolk-mass. By the addition of this plasmic layer the cells of the blastoderm increase 531in height, and now form a cubical or cylinder epithelium, which continuously envelops the surface of the egg. (Korschelt and Heider.)
e. Formation of the first rudiments of the embryo, and of the embryonic membranes
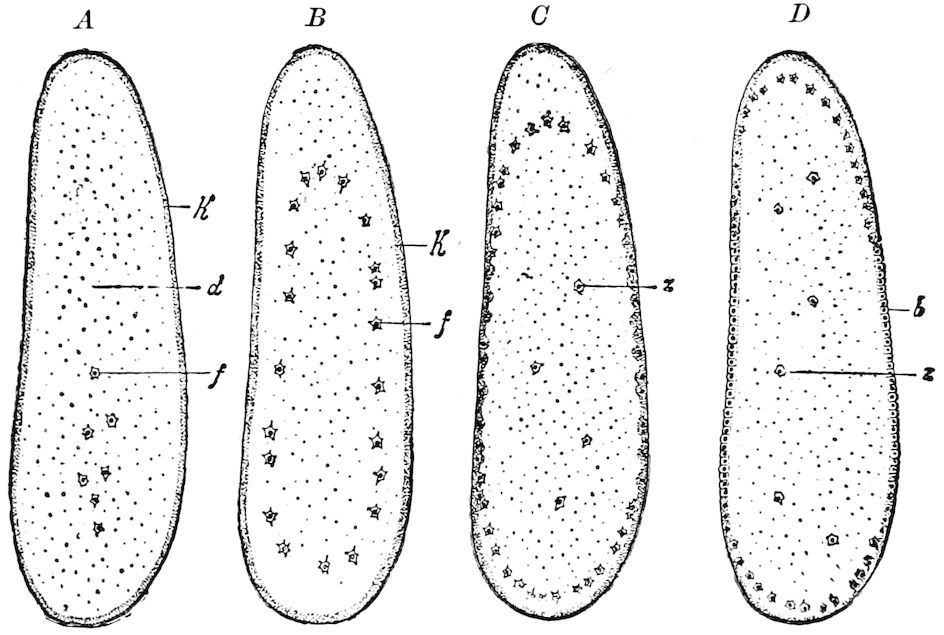
Fig. 507.—Formation of the blastoderm in Hydrophilus: b, completed blastoderm; d, yolk; f, so-called division-cells; k, “keimhautblastem”; z, yolk-cells.—After Heider, from Korschelt and Heider.
The embryo first arises as a whitish streak or band-like thickening on the ventral side of the egg, and is variously called the “primitive streak,” “primitive band,” “germinal band,” or “embryonal streak.” In most cases the primitive band is divided at regular intervals by transverse furrows, indicating the limits of what are to be the body segments.
Cross-sections (Fig. 509) show that the band is composed of several layers, i.e. an outer layer (ectoderm) and an inner layer which comprises the endoderm and mesoderm, and so long as these two layers are not sharply differentiated from one another, this second layer may be called, with Kowalevsky, “the inner lower layer, or ento-mesoderm” (Figs. 508, 509, B, C, u).
It is characteristic of insects, only rarely occurring in other arthropods (e.g. the scorpion), that the primitive streak is not situated on the surface of the egg, but becomes overgrown by a folded structure (Fig. 508, af) rising from its edges, the amnion-fold, so 532that it appears somewhat depressed or sunken in under the upper surface of the yolk. While the amnion-folds are extending from all sides over the primitive band, there becomes formed under it, by the invagination of the outer surface of the egg, a cavity, the amnion-cavity (ah), which, when the amnion-fold has completely overgrown the primitive band and united together (Fig. 509, C), appears completely closed from without.
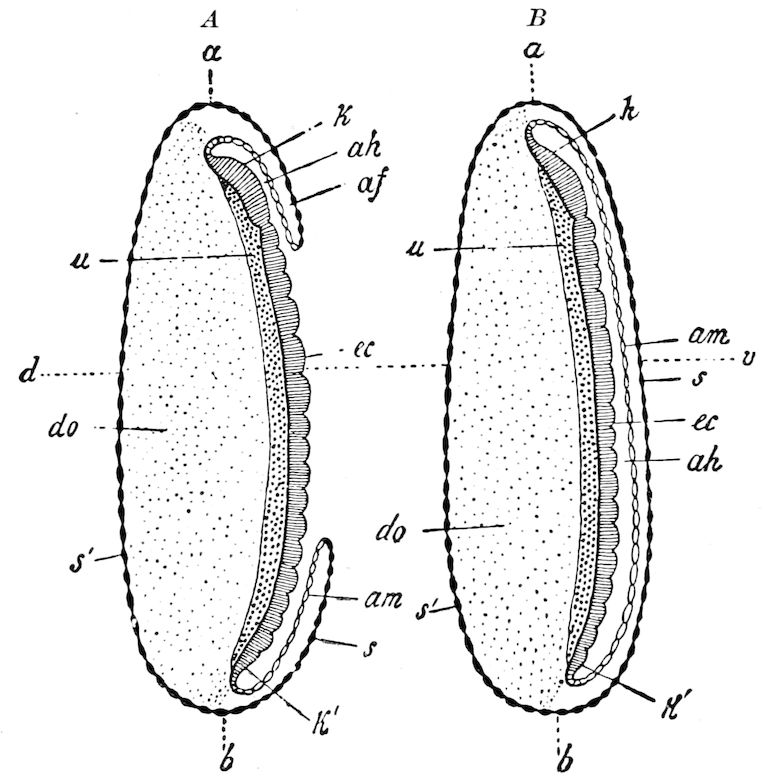
Fig. 508.—Two schematic median sections through an insect-embryo to represent the development of the embryonal membranes. In A the primitive streak is not wholly overgrown by the amnion-fold. In B the amnion-folds have united with each other and completely overgrown the primitive streak: a, fore, b, hind, egg-pole; v, ventral side; d, dorsal side; af, amnion-folds; ah, amnion-cavity; am, amnion; do, yolk; ec, ectoderm; k, head-end, k′, hinder-end, of the primitive streak; s, the part of the serosa arising from the amnion-fold; s′, the part of the serosa arising from the unaltered blastoderm; u, lower layer.—After Korschelt and Heider.
Formation of the embryonic membranes.—The amnion-folds finally completely overgrow the primitive band (Fig. 509, B and C), and form the embryonal membranes. The primitive band is seen after its completion to be overgrown by a double cellular epithelial membrane. The outer of these two membranes, that which arises from the outer leaf or layer of the amnion-fold, is the serosa (Figs. 508, B; 509, C, s; 510). This passes continuously into the unchanged part of the blastoderm, which has no part in the formation of the 533primitive band and germ-layers, and which covers the outer surface of the yolk. Thus the serosa, which is usually held to include this portion also of the blastoderm, forms a closed sac which covers the whole surface of the egg, with one part extending over the surface of the yolk, and the other over the primitive band (Fig. 510).
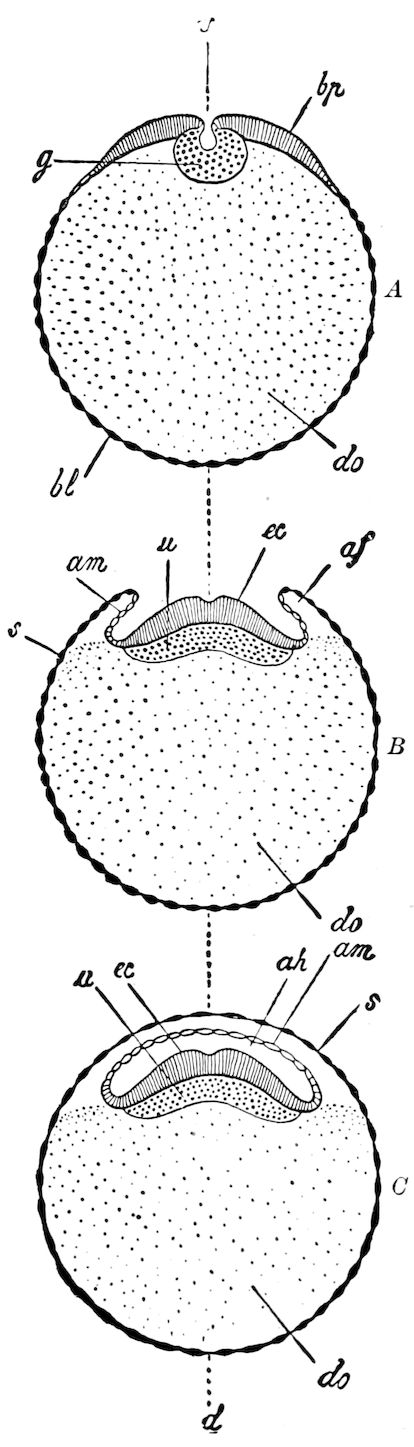
Fig. 509.—Diagrammatic cross-section through three successive stages of the primitive streak, and growing embryonal membranes of insect-embryos. A, formation of the ventral plate and of the gastrula invagination (g). B, upward growth of the amnion-folds (af). C, complete overgrowth of the primitive band through the amnion-folds: v, ventral side; d, dorsal side; af, amnion-folds; ah, amnion-cavity; am, amnion; bl, blastoderm; bp, ventral plate; do, yolk; ec, ectoderm; s, serosa; u, under or inner layer.—After Korschelt and Heider.
The inner of the two layers, called the amnion (Fig. 509, am), is more closely connected with the embryo. The amnion and ectoderm of the primitive band together form a completely closed sac, whose lumen forms the amniotic cavity. Originally connected with the serous membrane, it splits off from the primitive band about the time the appendages begin to bud out, and continues to closely envelop the body and appendages, as seen in Fig. 509. Both of these membranes are, before the time of hatching, either absorbed, or, as in Lepidoptera, retained. The amnion is retained until after hatching in the locust, etc. In certain Coleoptera the serosa is retained, and the amnion is absorbed (Fig. 532), while in Chironomus and the Trichoptera the serosa is absorbed, and the amnion retained, with the egg-shell or chorion. 534Hence we have eight layers in the winged insects[81] during embryonic life:
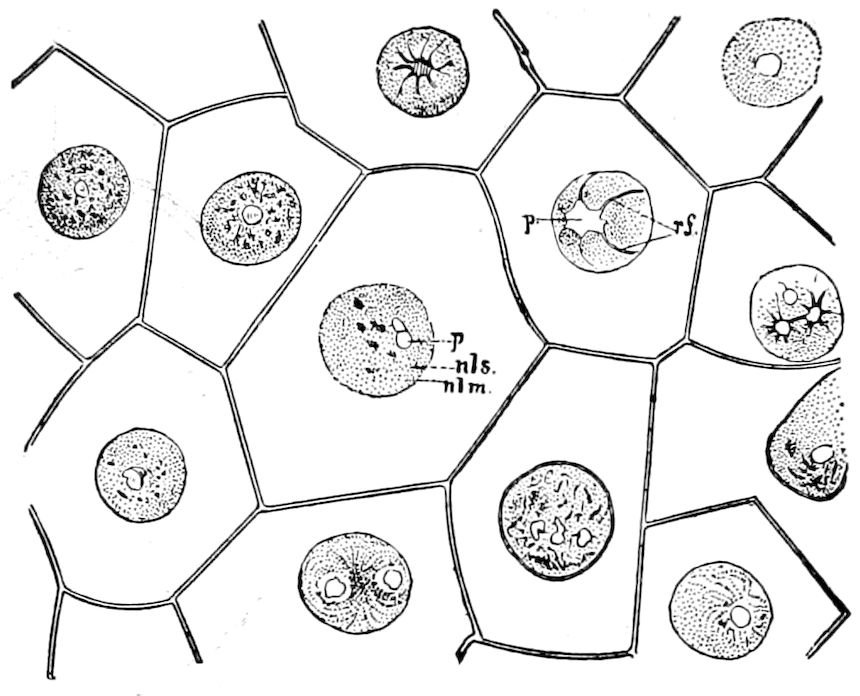
Fig. 510.—Surface view of fresh serosa from an Œcanthus, treated with acetic carmine; the blastoderm completely formed, × 500: p, polar body; rf, radiating fibres; nls, nuclear substance; nlm, nuclear membrane.—After Ayers.
- 1. Exochorion. (Remains of the epithelium of the ovarian follicle.) 2. Chorion. (Egg-shell or cuticle secreted in the ovarian follicle.) 3. Vitelline membrane. (Primary egg-membrane. Yolk-skin or membrane.) 4. Serous or outer germ-membrane. (Serosa.) } 5. Amnion or inner germ-membrane. } Derived from the blastoderm. 6. Ectoderm. } } 7. Mesoderm. } Embryo. } 8. Endoderm. } }
In the embryo of Xiphidium and Orchelimum Wheeler has found and described with much detail a membranous structure which he calls the indusium. “The organ,” he says, “appears to have been retained by the Locustidæ, and completely lost by the embryos of other winged insects.” It arises in Xiphidium, as a simple circular thickening of the blastoderm, between and a little in front of the procephalic lobes (Figs. 511, 512, A-E), and afterwards spreads over nearly the whole surface of the egg, leaving the poles uncovered, as in Fig. 513, where it is divided into two further membranes, the inner and outer indusium, the former lying in contact with the amnion. After this the serosa “is excluded from taking any part in the development of the embryo; both its position and function are now usurped by the inner indusium.”
535Hence in an egg of the Locustidæ Wheeler distinguishes, passing from within outward in a median transverse section of the egg, the following envelopes:
- 1. The chorion. 2. The blastoderm-skin-like cuticle secreted by the serosa. 3. The serosa. 4. The outer indusium. 5. A layer of dark granular secretion (probably some urate). 6. The cuticle secreted by the inner indusium. 7. The inner indusium. 8. The amnion. While envelopes 1–7 invest the whole egg; layer 8, the amnion, covers only the embryo.
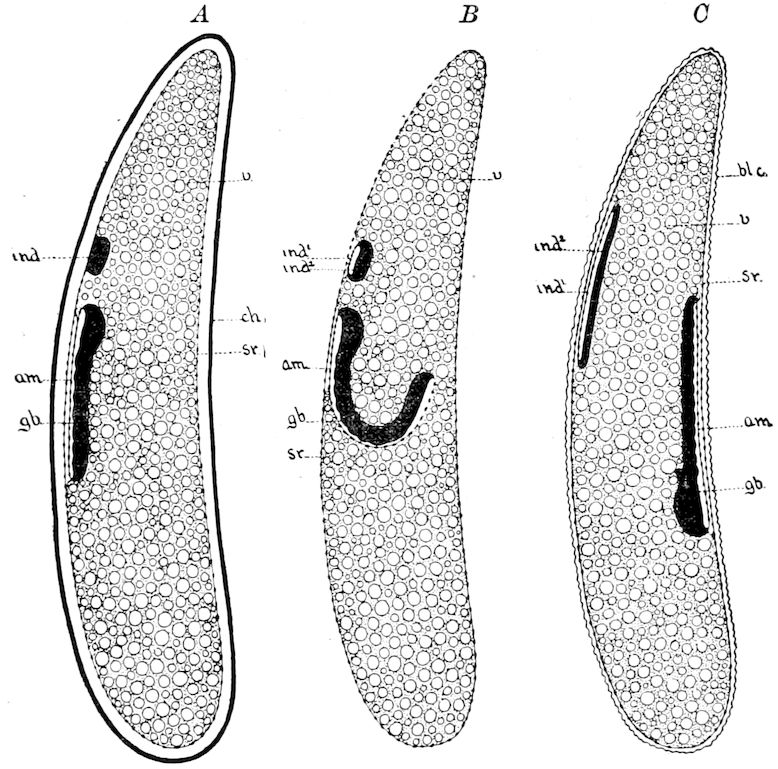
Fig. 511.—Diagrams illustrating the movements and envelopes of the embryo of Xiphidium: A, after the closure of the amnioserosal folds. B, during the embryo’s passage to the dorsal surface. C, just after the straightening of the embryo on the dorsal surface; ind, indusium afterwards forming ind1, the inner, and ind2, the outer indusium; ch, chorion; sr, serosa; am, amnion; gb, germ-band; v, yolk; bl. c, blastoderm membrane.
Wheeler further suggests that the so-called micropyle of the Collembola (Anurida), which has been homologized with the “dorsal organ” of Crustacea, is a possible homologue of the indusium, as also the “primitive cumulus” of spiders, and the “facette” or “cervical cross” of Pentastomids described by Leuckart and also by Stiles.
The gastrula stage.—The primitive band invaginates so as to give the opportunity for the formation of the inner layer. This invagination, which at a certain stage is established along the whole length of the primitive band, forms a median furrow and may be 536regarded as the gastrula-invagination of insects. The lower (inner) layer thus arising afterwards spreads out under the entire primitive band (Fig. 509, B and C), the edges of which become bordered by the growing amnion-fold. (Korschelt and Heider.)
In certain forms the primitive band arises from several separate rudiments which afterwards unite. Thus in Musca and Hydrophilus the anterior and posterior ends develop first, and in Hydrophilus the procephalic lobes originate independently of the rest of the band. In the Aphides, also, according to Will, these lobes arise independently, afterwards uniting with the primitive band.
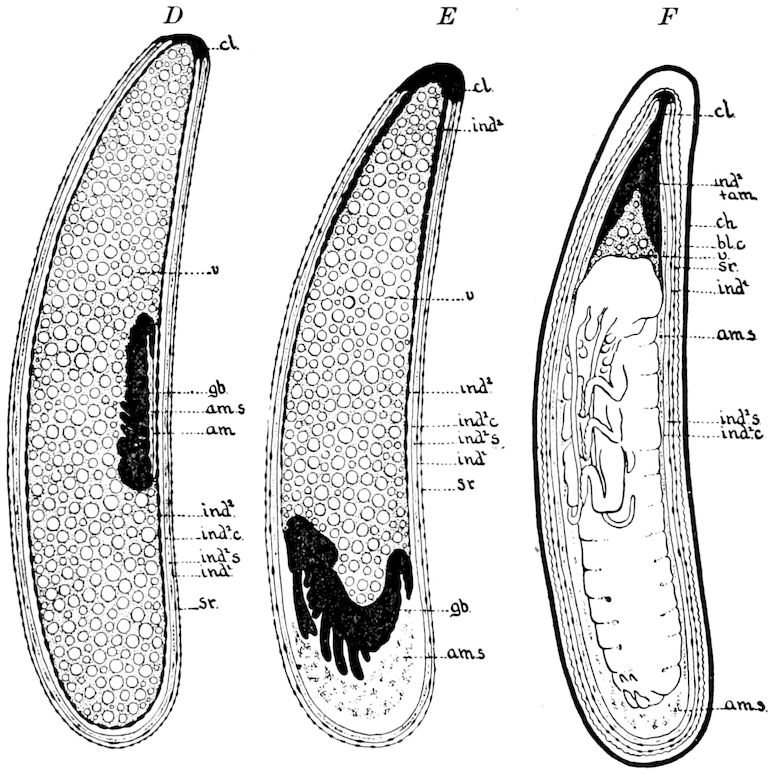
Fig. 512.—Diagrams illustrating the movements and envelopes of the embryo of Xiphidium: D, the stage of the shortened embryo on the the dorsal yolk. E, embryo returning to the ventral surface. F, embryo nearly ready to hatch; ch, chorion; b. lc, blastoderm membrane; sr, serosa; ind1, outer indusium; ind2, inner indusium; ind2 + am, inner indusium and amnion fused; am, amnion; ind1 c, cuticle of the inner indusium; ind2 s, granular secretion of the inner indusium; am. s, amniotic secretion; v, yolk; cl, columella; gb, primitive band.
Division of the embryo or primitive band into body-segments.—Meanwhile the primitive band grows at the expense of the yolk, spreading out more and more over its surface, until in certain cases (Coleoptera, Diptera, Siphonaptera, and Trichoptera) it lies like a broad ribbon over the yolk, so that the two ends nearly meet on the dorsal side. By this time it becomes divided by transversely impressed lines into segments, which correspond to those of the larva and adult. The first of these segments is divided into two broad 537and flaring flaps, which are called the procephalic lobes. It becomes the antennal segment.
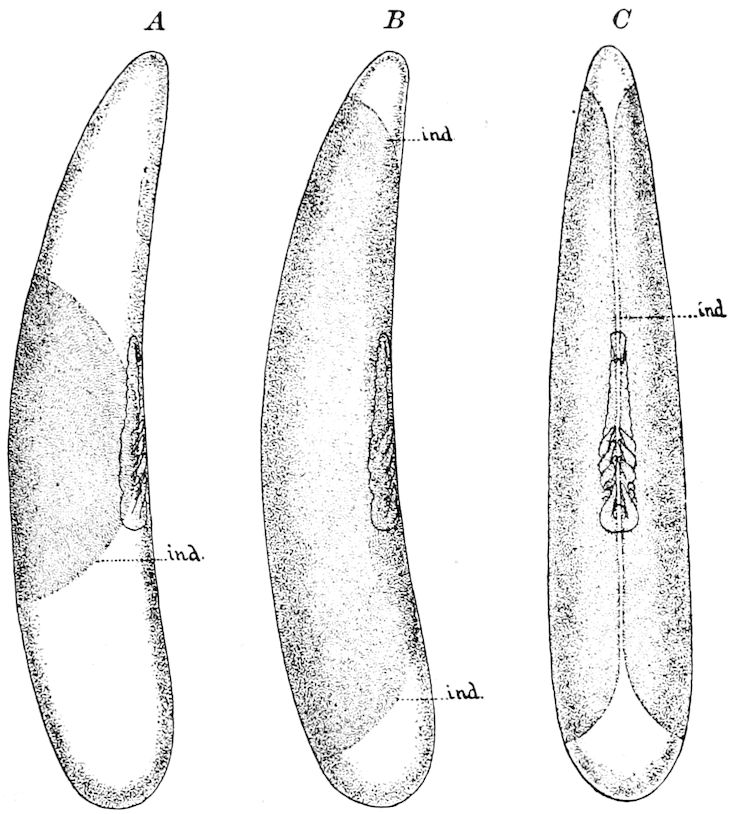
Fig. 513.—Two stages in the spreading of the indusium. A, lateral view of egg just after the arrival of the embryo on the dorsal yolk. B, lateral view of the egg with the indusium nearly reaching the poles. C, same egg seen from the dorsal surface.
The mouth (stomodæum) now develops, and is situated at the anterior,[82] and the rectum (proctodæum,) at the posterior pole, or end of the primitive band.
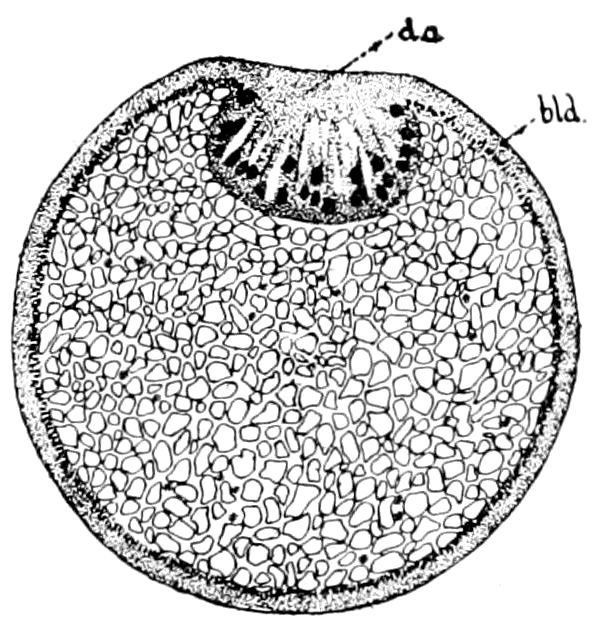
Fig. 514.—Median section of the egg of Anurida maritima: do, “micropyle”; bld, blastoderm.—This and Figs. 511–513, after Wheeler.
In Blatta, Hydrophilus, the Trichoptera, and the Lepidoptera the hindermost part of the primitive band is turned in ventrally (Figs. 534, C).
The preceding account of the relations of the primitive band to the yolk does not apply to all insects, since there are variations which appear to depend on the form of the egg, and on the amount and distribution of the yolk-masses. In certain Coleoptera, the primitive band sinks down and thus becomes immersed into the yolk. In Donacia (Kölliker and Melnikow) and Hydrophilus (Heider), and in the Chrysomelidæ and 538Attelabus, a weevil, as we have observed, the primitive band rests on the outside of the yolk, but in Telephorus fraxini it is immersed. In the Hemiptera it is immersed (Fig. 516), but there is much variation in this respect, the degree of immersion being most marked in the Coccidæ (Aspidiotus), and least so in Corixa. Besides the position of the primitive band, there are in Odonata and Hemiptera differences in the origin of the primitive band itself and of the embryonic membranes.
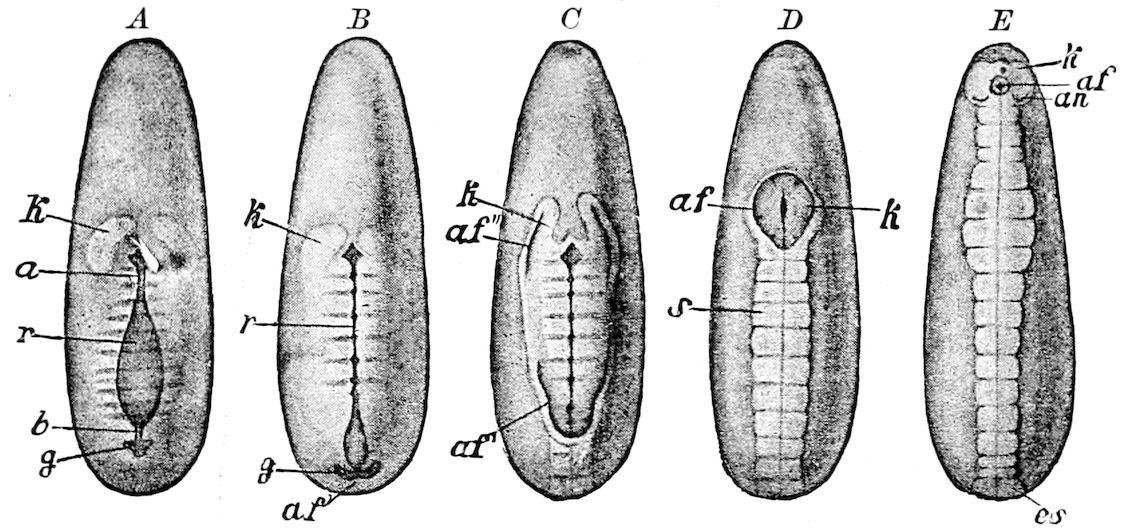
Fig. 515.—Ventral view of five developmental stages of Hydrophilus: a and b, places at which the blastopore contracts; af, edge of the amnion-fold; af′, caudal fold; af″, paired head-fold of the amnion; an, antenna; es, last segment; g, pit-like invagination (first indication of the amniotic cavity); k, head-lobes; r, furrow-like invagination; s, portion of the primitive streak covered by the amnion.—After Heider, from Lang.
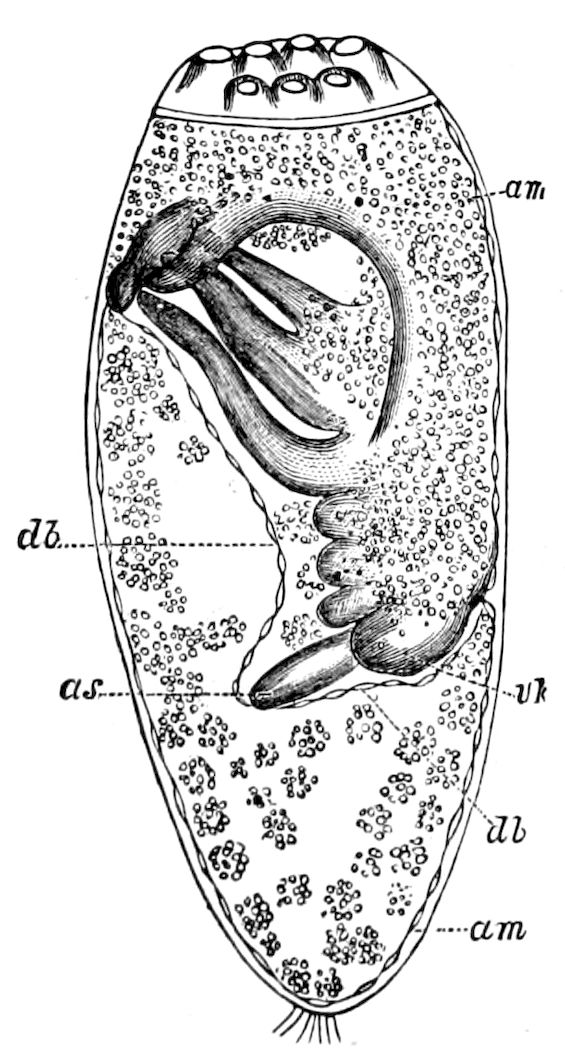
Fig. 516.—Embryo of the louse: am, serosa; db, amnion; as, antenna; vk, clypeus.—After Melnikow.
Korschelt and Heider divide the early embryo of insects into two types:
1. Into those with a superficial primitive band; viz., where there is no passage of yolk-elements into the space between the amnion and serosa. The primitive band has in such cases a relatively superficial position (Figs. 508, 509, 521, 535). Examples are certain Orthoptera (Blatta, Œcanthus, Mantis, Gryllotalpa), also certain Hemiptera (Corixa), certain Coleoptera, and the Trichoptera, Diptera, and Hymenoptera.
2. Into those with an immersed primitive band, with the space between the serosa and amnion filled with yolk (Figs. 517, 518, 534). Examples are the orthopterous Stenobothrus, Odonata, many Hemiptera (the Pediculina and Pyrrhocoris), the Coleoptera already mentioned, and Lepidoptera.
It should be observed, however, that these differences are of little phylogenetic or taxonomic value, since genera of the same order, notably the Coleoptera, differ as to the position of the primitive band, so also two orders so nearly allied as the Trichoptera and Lepidoptera.
Differences between the invaginated and overgrown primitive band.—In respect to the mode of origin of the primitive band and its relative position, 539there are two opposite types, though connected by transitional forms. In the one case the primitive band, i.e. its ventral portion, the “ventral plate” (Fig. 518, b, p) is pushed in or invaginated in the interior of the egg; in the other case it becomes overgrown by the folds of the amnion arising from its edges.
Fig. 517.—Primitive streak of a lepidopter in cross-section: ah, amniotic cavity; am, amnion; c, cœlomic cavity; do, nutritive yolk, divided into single nucleated masses; ec, ectoderm; m, mesoderm; pr, primitive thickenings of the ventral nervous cord; s, serosa.—Combined figure after those of Brobretsky and Hatschek, from Korschelt and Heider.
Fig. 518.—Five diagrammatic median sections representing the growth of a dragon-fly (Calopteryx). A-C, development of the primitive streak (k, k′) by invagination. D, the amnion-fold (af), growing over the head-end of the primitive streak. E, closing of the opening of the amnion-cavity (ah): v, ventral, d, dorsal side; a, fore, b, hind end of egg; bl, blastoderm; bp, ventral plate; do, yolk; k, head-end, k′, caudal end, of the primitive streak; kh, germinal thickening or initial point of invagination; s, serosa.—After Brandt, from Korschelt and Heider.
In insects with an overgrown primitive band, the band at the beginning is generally short and always situated on the ventral side of the egg, with the head-end looking forward, and remains in this position throughout embryonic 540life. There is no revolution of the embryo. The embryonal membranes arise through the formation of folds.
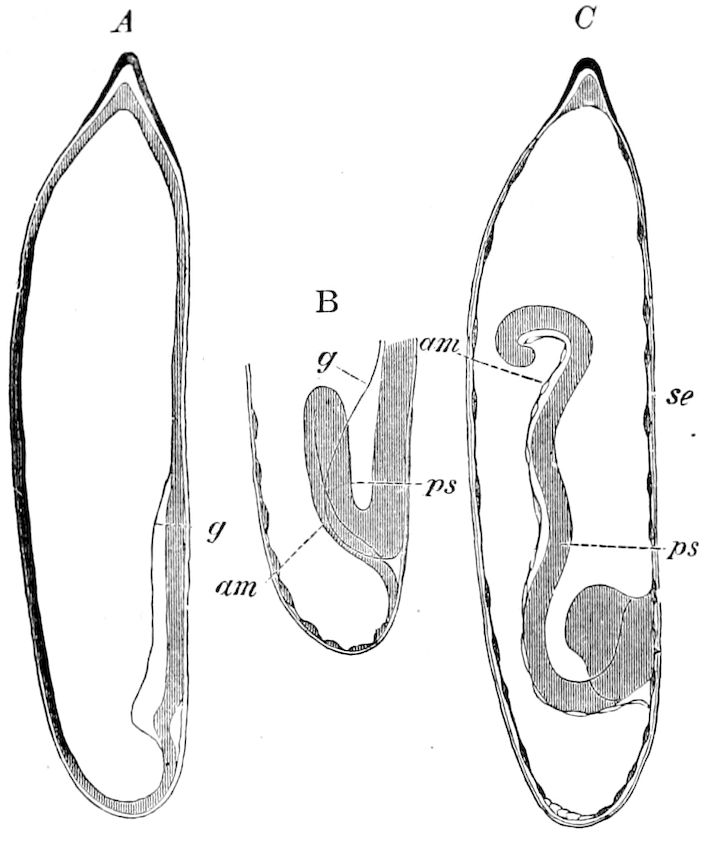
Fig. 519.—Three embryonic stages of Calopteryx: am, amnion; g, edge of the ventral plate; ps, germ of primitive band; se, serosa.—After Brandt, from Balfour.
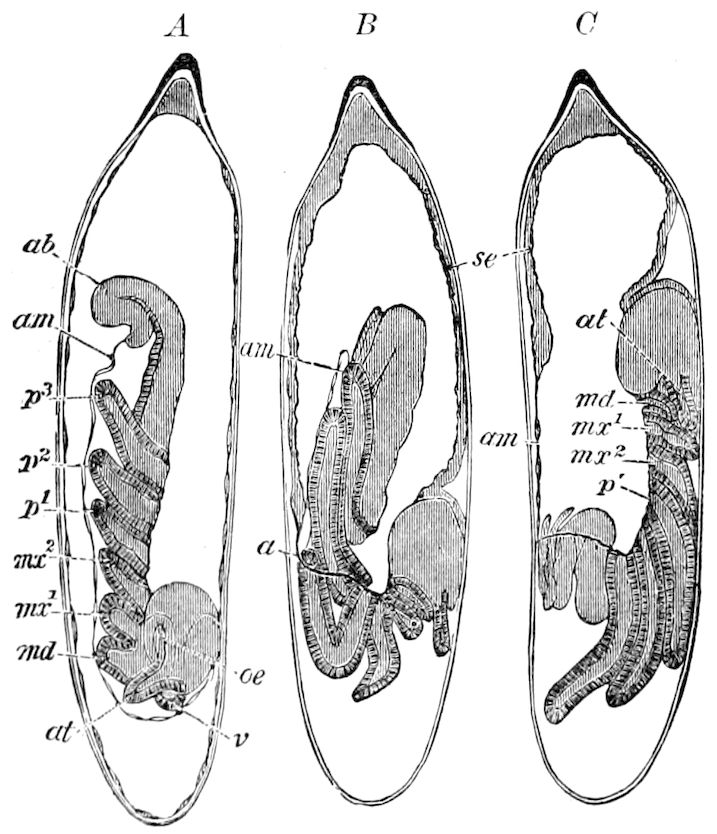
Fig. 520.—Three farther stages of growth of Calopteryx. B and C show the inversion of the embryo: a, opening of the amniotic-cavity, out of which the embryo emerges; ab, abdomen; am, amnion; at, antenna; md, mandible; mx1, mx2, 1st and 2d maxillæ; œ, œsophagus; p1, p2, p3, legs; se, serosa; v, anterior end of the primitive streak.—After Brandt, from Balfour.
In insects with an invaginated primitive band, of which the Odonata afford examples, the first rudiment of the primitive band is in the form of a ventral plate of slight extent passing ventrally in the hinder half of the egg, in whose posterior section a process of invagination (Fig. 518, A, kh), soon occurs. The cavity of this invagination is the first indication of the amnion-cavity (Fig. 518, B, ah), while its wall in its thickened ventral part (K′) is concerned in the formation of the primitive band, and, in its dorsal thin part, in the formation of the amnion (B, C, am).
Revolution of the embryo where the primitive band is invaginated.—At first the head-end of the embryo is directed towards the posterior end of the egg, as in dragon-flies (Fig. 518). Also that surface of the primitive band which afterwards faces the ventral, is at first turned towards the dorsal side of the egg. In order to bring the primitive band into the later relations, there must occur the process of revolution, or turning, of the embryo. The somewhat advanced embryo of the Odonata, after the appearance of the head and thoracic appendages, undergoes a rotating motion around its transverse axis, and at the same time turns out of the amniotic cavity (Fig. 520, B). This process is so managed that near the head-region, the amnion and serosa, there closely situated to each other, are fused together, and at this 541place tear or burst open. Through this rent (a), in the same place in which the original invagination-opening was situated, the amniotic cavity again opens, and through the opening thus formed first the head and then the succeeding segments of the primitive band (Fig. 520, B) pass out, and remain there while the head passes on to the anterior pole of the egg on the ventral side, the embryo thus assuming a position like that of other insects. (Kowalevsky.)
In the parasitic Hemiptera (Pediculina), according to Melnikow, the opening in the membranes near the head remains permanent, and the embryo becomes everted through it, while the yolk, enclosed in the continuous membrane formed by the amnion and serous membrane, forms a yolk-sac on the dorsal surface. The same process occurs in Mallophaga, and also in Œcanthus, as described by Ayers (Fig. 521). Generally as soon as the embryo passes out of the amniotic cavity the latter soon becomes smaller and finally completely disappears.
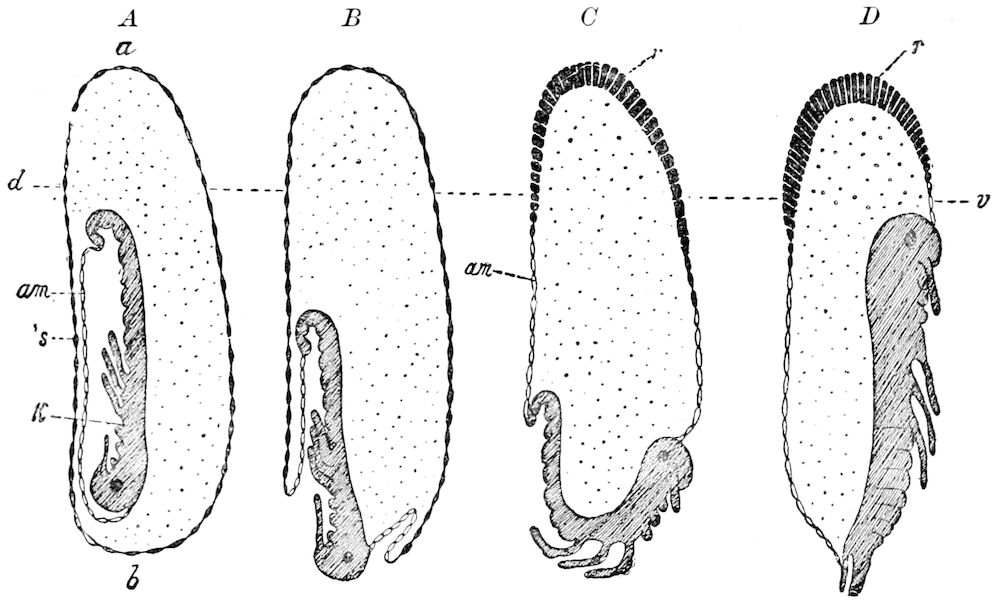
Fig. 521.—Revolution of the embryo of Œcanthus (diagrammatic): a, fore, b, hind end of egg; am, amnion; d, dorsal, v, ventral side of egg; k, primitive streak; r, dorsal plate (originating by the contraction of the serosa (s)).—After Ayers, from Korschelt and Heider.
As the embryo grows, and the sides grow up and the back closes over, the contents of the yolk-sac are soon taken up and absorbed in the intestinal cavity, which communicates with it.
In Phyllodromia, according to Wheeler, the process of revolution is “hurried through by the embryo from the beginning of the 16th to the end of the 17th day.” Several successive stages are represented in Fig. 522. In the 15th day the embryo still occupies the middle of the ventral surface of the egg. Soon the envelopes (amnion and serosa, as) rupture, an irregular slit being formed, and soon the egg and embryo are as seen in Fig. 522, B, the embryo standing out free from its envelopes on the yolk, and the edges of its dorsal growing walls (b) are distinctly marked. The tail now lies at the caudal end of the egg (Fig. 522, C). By the 17th day the walls have closed in the median dorsal line, and the embryo has grown in length to such an extent as to bring its head to the cephalic pole (Fig. 522, E).
Korschelt and Heider consider, since the primitive band of the chilopod myriopods (Geophilus) is curved in at the middle and sinks into the interior of the 542yolk, that in insects the invaginated primitive band is the ancestral or primitive one, the overgrown primitive band being derived from it. The overgrown primitive band by its position may also be better insured against certain mechanical attacks, perhaps also against the danger of drying up.
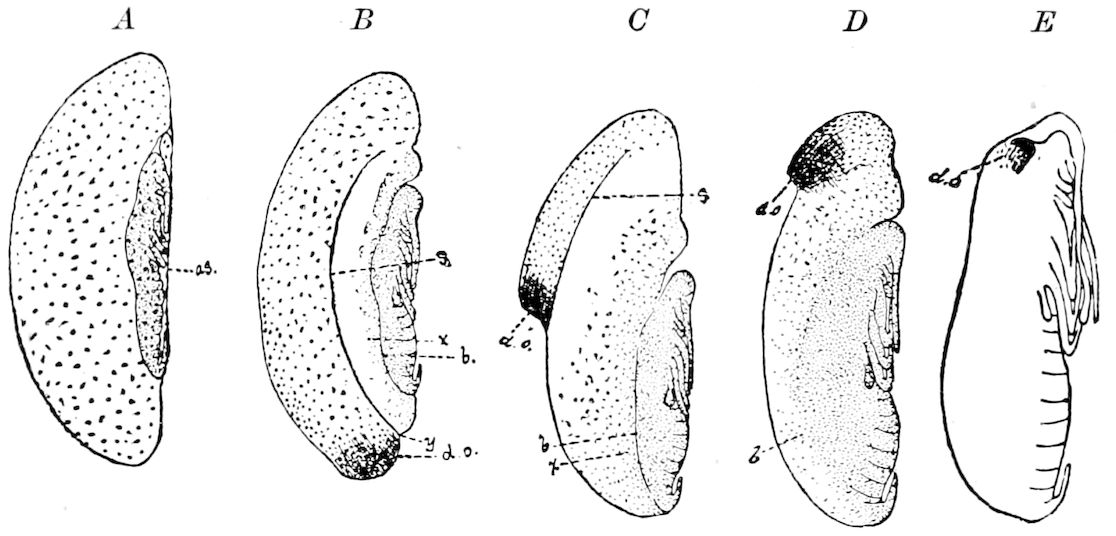
Fig. 522.—Embryo of Phyllodromia, 15 days old; revolution about to begin. The stages in revolution are represented, after the rupture of the amnion and serosa, in A to E, which are from embryos 16, 16½, 16¾, and 17 days old respectively: as, amnion and serosa; s, edge of serosa; b, dorsal growing body-wall; d.o, dorsal organ; x, clear zone covered with scattered amniotic nuclei.—After Wheeler.
f. Formation of the external form of the body
Origin of the body-segments.—As we have seen, the first traces of segments appear very early, the primitive band being divided by superficial transverse furrows into segments. This segmentation into arthromeres (somites or metameres) can be observed in Hydrophilus and Chalicodoma at a time when gastrulation begins (Figs. 515, 536). The segmentation extends not only across the median portion of the primitive band, through whose invagination the inner layer (endomesoderm) results, but also to the lateral portions which become a part of the ectoderm of the primitive band. These transverse furrows correspond to thinner places in the epithelium, which in this stage forms the embryonal rudiment. It thus happens that, in the forms named, after the end of gastrulation not only the ectoderm, but also the endomesoderm, is already segmented.
So early an appearance of segmentation as that observed in Hydrophilus and Chalicodoma we must regard as a falsification of the process of development due to heterochrony. We must consider the conditions observed in other forms as the primitive ones, in which (as, for example, in Lina and in Stenobothrus, according to Graber) the gastrulation and separation of the ectoderm occurs in the still unsegmented primitive band, the division into segments occurring in later stages (Fig. 524). In these forms, then, the segmentation affects the invaginated endomesoderm, as well as the ectoderm. (Korschelt and Heider, p. 789.)
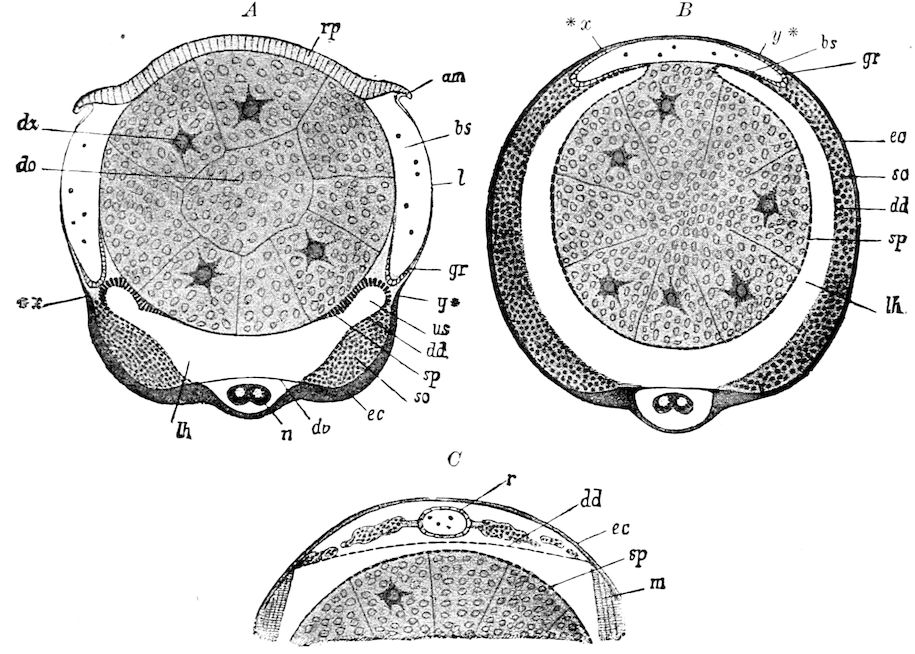
Fig. 523.—Diagrammatic cross-section through three successive stages of Gryllotalpa, showing the formation of the heart. (Compare Fig. 505.) The germs of the glandular intestinal layer (darmdrüsenblatt) are omitted. A, earliest stage; the primitive streak extends from *x to y*. The embryonal membranes are torn and pressed against the back: am, edge of the rent; rp, dorsal plate (serosa); l, lamella (amnion turned up) standing in connection with the ectoderm of the primitive streak. B, second stage; the primitive streak has completely grown around the yolk; the dorsal organ is absorbed. C, third stage, dorsal portion; the formation of the heart is finished: am, vestige of the amnion-fold; bs, blood-sinus; dd, rudiment of the dorsal diaphragm; dv, ventral diaphragm (compare Fig. 505); do, yolk; dz, yolk-cells; ec, ectoderm; gr, vascular groove (rudiment of the heart); l, lamella of the upturned amnion; lh, definite body-cavity; m, transverse muscle; n, nervous cord; r, heart; rp, dorsal plate; sp, splanchnic; so, somatic layer of the mesoderm; us, primitive segmental cavity; *x, y*, lateral terminations of the primitive streak.—After Korotneff, from Korschelt and Heider.
In the completely segmented primitive band may be distinguished two regions of a peculiar appearance (Figs. 515, 527), one at the anterior, and the other at the hinder end. The anterior, the primary head-section, contains the mouth-opening, and is characterized by its lateral expansions, or procephalic lobes. The other section, or posterior section, the so-called anal segment or telson, contains the anus. Between the two sections lies the segmented primary trunk-segment, which in insects consists of 17 segments. Of these the three most anterior are those destined to bear the mandibles and two pairs of maxillæ; the three following are the thoracic, which are succeeded by 10 abdominal segments, besides the 11th or telson (pygidium, or suranal plate).
544It is now generally believed that there are primarily eleven abdominal segments, while Heymons has detected twelve in the embryos of Blattids and Forficula (see p. 162). In the later stages of embryonic development the number of abdominal segments is diminished, the 10th and 11th abdominal segments becoming fused. In Hydrophilus and Lina this is the case, but according to Graber, in the Lepidoptera there is a fusion of the 9th and 10th abdominal segments, the llth remaining free.
According to Wheeler, in Doryphora, and also in Chalicodoma (Carrière), between the primary head-region and the mandibular segment is interpolated a rudimentary and transitory body-segment, the premandibular segment. According to Carrière this segment corresponds to a rudimentary pair of limbs, and also to a ganglion, which participates in the formation of the œsophageal commissure (see p. 51).
Fig. 524.—Three embryonic stages of a leaf-beetle (Lina): A, unsegmented primitive streak; in B and C the segmentation becomes distinct on the lower layer (u). B, with the germs of the gnathal segments (k′-k‴), and in C the three thoracic segments (t’-t‴), with the first two abdominal segments (a′, a″): bl, blastopore; kl, head-lobes; th, extension of the primitive streak into the thoracic region.—After Graber, from Korschelt and Heider.
The procephalic lobes.—The head-lobes, or procephalic lobes, appear at a very early period (Fig. 524, kl), before any traces of the segments of the trunk region. Ayers has shown that in Œcanthus the primitive band, in its earliest condition and before the appearance of the head-lobes, is a simple oval plate or almond-shaped thickening near the posterior end of the egg (Fig. 525, 1, 2). This plate is “soon divided into two tolerably well-marked regions by the enlargement of the head-end,” the first indication of the head-lobes 545(3). A depression next forms in what is to be the middle of the forehead. “It indicates the position of the future labrum, and forms the inner boundaries of the two cephalic ganglia, which are developed on either side of this depression at a much later stage.” Almost simultaneously with the appearance of this depression, two lateral folds are formed in the trunk portion of the band, which are the first indications of the maxillary and thoracic regions, the abdominal portion not yet showing traces of a division into segments (Fig. 525, 5). The thickened outer edges of the head-fold next gradually grow in towards the median line (Fig. 525, 5), and bend forward towards the region of the future mouth. The rounded angle made by the posterior end of the head-fold is the first indication of the antennæ. The embryo is now composed of four well-marked regions: cephalic, maxillary, thoracic, and abdominal. The primitive band then grows much longer, the primitive mouth and anus appear, and the appendages bud out, and eventually the embryo revolves and appears on the ventral side of the egg (Fig. 525, 6).
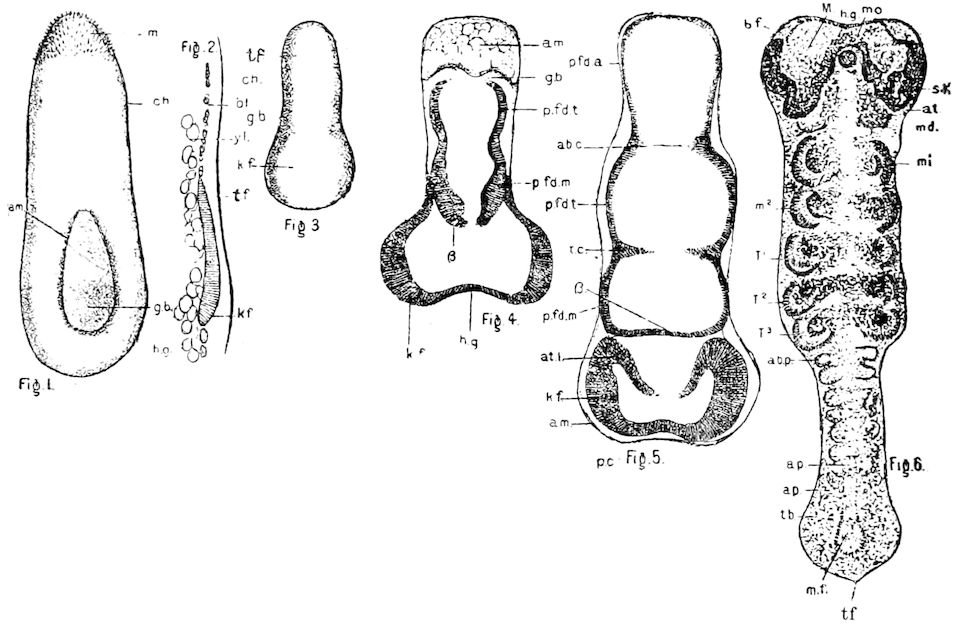
Fig. 525.—Early stages in the embryology of Œcanthus niveus. Fig. 1, the youngest observed primitive band, the serosa not yet formed; 2, longitudinal optic section (diagrammatic) of Fig. 1; 3, the primitive band after the appearance of the head-fold, which is indicated at this time by the more rapid growth and consequent greater breadth of the lower end of embryo, x 25; 4, a young embryo after the appearance of the primitive segment-folds, x 50; 5, a more advanced embryo, with the antennal folds distinctly marked off; the free ends of the primitive folds have united across the embryo posterior to the antennal folds, x 50; 6, ventral view of the embryo with the appendages budding out, x 25 (the embryo in this stage lies dormant through the six colder months of the year): am, amnion; m, micropylar end; ch, chorion; gb, primitive band; bf, brain-fold; yl, yolk; tf, caudal fold; kf, head-fold (procephalic lobe); p.fd.t, primitive thoracic fold; p.fd.m, primitive maxillary fold; p.fd.a, primitive abdominal fold; ab.c, abdominal constriction; t.c, thoracic constriction; at.l, antennal lobe; M, mesoderm; h.g, head groove; mo, mouth; sk invagination of ectoderm to form head-apodeme; md, rudiment of mandible; m1, 1st, m2, 2d maxilla; T1–T5, legs: ab.p, 1st abdominal appendage; ap, other appendages; tb, caudal expansion; mf median furrow; B, primitive unpaired organ (metastomum).—After Ayers.
546These primitive regions of the primitive band, before the segments are formed, are called by Graber macrosomites, and the secondary segments into which they divide (which afterwards become the body-segments), microsomites. The macrosomites are peculiar to insects, and may be an inheritance from a hypothetical ancestral form. With Korschelt and Heider, we should hardly share this view.
Fig. 526.—Older embryo of Œcanthus with the appendages budded out, those of the abdomen distinct: abp, first pair; a.s, anal stylet; pr, proctodæum; am, amnion.—After Ayers.
Our observations on locusts show clearly (1) that the procephalic lobes are the pleural portion of the first cephalic or antennal segment; (2) that the antenna is an appendage or outgrowth of the procephalic lobes; (3) that the eyes are a specialized group of epidermal cells of the upper part of the procephalic lobes, and are not homologues of the antennæ or of the appendages in general; and (4) it seems to follow from a study of the relations and mode of development of the clypeus and labrum, that they arise between the procephalic lobes, and probably represent the tergal part of the antennal segment, forming the roof of the mouth, i.e. closing in from above the pharynx.
In general the formation of the body-segments into the primitive band is in succession from before to the hinder end. This successive appearance has been observed by Graber in genera of different orders (Stenobothrus, Lina, and Hylotoma). For example, in the beetle Lina, after the appearance of the mandibular and two maxillary segments, appear the three thoracic segments, together with the two anterior abdominal segments, the other abdominal segments arising afterwards. In other cases, the formation of segments seems to be simultaneous along the entire length of the primitive band. An exception to the rule has been noticed by Heider in Hydrophilus, as in this beetle the development of the segments of the middle region appears somewhat delayed, while the fore and hind parts of the primitive band are more rapid in development. In Pieris, according to Graber, the thoracic segments are more rapidly developed than the others; soon after, the gnathic segments (mandibles and two pairs of maxillæ) appear, and finally the abdominal segments are formed.
547Fore-intestine (stomodæum) and hind-intestine (proctodæum), Labrum.—The digestive canal of insects consists, as in other animals, of three portions, the fore, mid, and hind gut or intestine. The next change after the completion of the segments of the primitive band is the development of the fore and hind intestine and the appendages. The fore-intestine (stomodæum) arises as an invagination in the area of the primary head-section, and the hind-intestine (proctodæum) in the terminal section (Figs. 300 and 546).
In insects generally the formation of the fore-intestine occurs earlier than that of the hind-intestine. An exception was discovered by Graber and also by Voeltzkow in Muscidæ, where the proctodæum appears earlier.
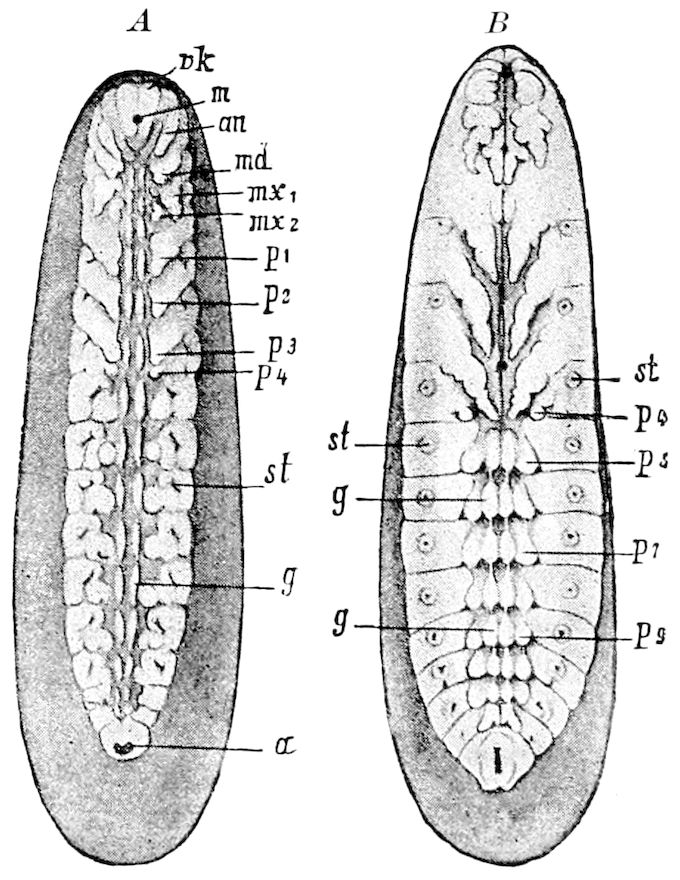
Fig. 527.—Rudiments of the appendages of the embryo of Hydrophilus: an, antenna; md, mandibles; mx1, 1st, mx2, 2d maxilla; vk, clypeal region; m, mouth; p1-p3, legs; p4-p9, rudiments of abdominal appendages, 1–9; st, stigma; a, anus.—After Heider, from Lang.
Usually at the time of origin of the stomodæum a projection arises at the anterior edge of the primary head-region, the so-called forehead (Fig. 527, vk), which is the common rudiment of the clypeus and labrum. In many cases (certain Coleoptera and Lepidoptera) these rudiments first assume the form of paired hooks (see Figs. 83, 102, 104, 105, of Graber’s Keimstreif der Insekten, also Figs. 529 and 546), which afterwards, by fusion in the median line, become single, though notched in the middle; but in the more generalized Blatta and Mantis, as well as in bees, the rudiment is single at the outset.
The view advanced by Patten, and also by Carrière, that the labrum is a first pair of antennæ, is scarcely tenable, and we quite agree with Korschelt and Heider in regarding the clypeo-labral region as homologous with the upper lip of Crustacea, and, we may add, of Merostomes and of Trilobites.
It should be observed that in many insects, in their earlier embryonic state, directly behind the mouth arises, from paired rudiments, what seem provisional lower lip structures (not to be confounded with the 2d maxillæ of insects). This under lip structure was first discovered by Bütschli in the bee (his inner or 2d 548antennæ), and afterwards by Tichomiroff in Lepidoptera. Heider, in his work on Hydrophilus, describes it as the “lateral mouth-lips,” while, more recently, Nusbaum has observed it in Meloë. This under lip structure may be regarded as analogous to the paragnaths of Crustacea, although to attempt to homologize it with these seems useless. (Korschelt and Heider.)
Completion of the head.—Sufficient attention has not been paid to this subject by embryologists. The head is at first, dorsally, mostly composed of the head-lobes, or antennal segment only, and the dorsal or tergal portion of the oral appendages develop at a later period. We have observed in the embryo of dragon-flies (Æschna) that the tergites of the mandibles and first maxillæ are simultaneously fused with the head-lobes, while the much larger tergal region of the 2d maxillæ remains for some time separate from the anterior part of the head, and is continuous with the thoracic segments, and it is only just before hatching that this segment becomes fused with the rest of the head (Fig. 36). In a sense, the 2d maxillary segment when it is free from the head reminds us of the foot-jaw, or 5th segment of chilopod myriopods (see also p. 53).
g. The appendages
As we have seen, nearly or quite simultaneously all the limbs as a rule bud out from each side of the median line of the primitive band. They arise as saccular evaginations or outgrowths of the ectoderm, directed a little backwards. They are at first filled with mesoderm cells, and in the Orthoptera diverticula of the cœlom-sac are taken up into the rudimental limbs, as in Peripatus and Myriopoda. (Graber, Cholodkowsky.) As the antennæ, mouth-parts, legs, and abdominal appendages are all alike at first, their strict homology with one another is thus demonstrated. In insects never more than a single pair of limbs is known to arise from one segment.
The cephalic appendages.—The antennæ evidently arise from the hinder edge of the procephalic lobes (Fig. 527, an). As in Limulus, the first pair of appendages are at first postoral (Fig. 528, at), afterwards moving forward owing to changes in the relative proportions of the parts of the head, and they are in all respects, in their development and position in relation to the segment from which they arise, homologous with the appendages succeeding them.
The occurrence of rudiments of a pair of preantennal appendages in Chalicodoma which is claimed by Carrière, needs confirmation, as other embryologists have not observed them.
549The postoral appendages of the head are the mandibles and the 1st and 2d maxillæ, besides the supposed premandibular segment already referred to on pp. 50–54, which only temporarily exists.
The trophi or oral appendages are all alike at first, but soon differ in shape, acquiring their characteristic form shortly before the embryo leaves the egg. The mandibles of Œcanthus are said by Ayers at the time of revolution of the embryo to be slightly bilobed, and in his Fig. 5, Pl. 19, they are represented as deeply trilobed, but in general they are undivided. The 1st maxillæ are at this time distinctly trilobed. The 2d maxillæ are separate, and distinctly though unequally bilobed, becoming united shortly before birth. In the embryos of dragon-flies they are at an early date very large and long, and directed backwards, and are not fused together until just before hatching, when the extraordinary mask-shaped labium is fully developed.
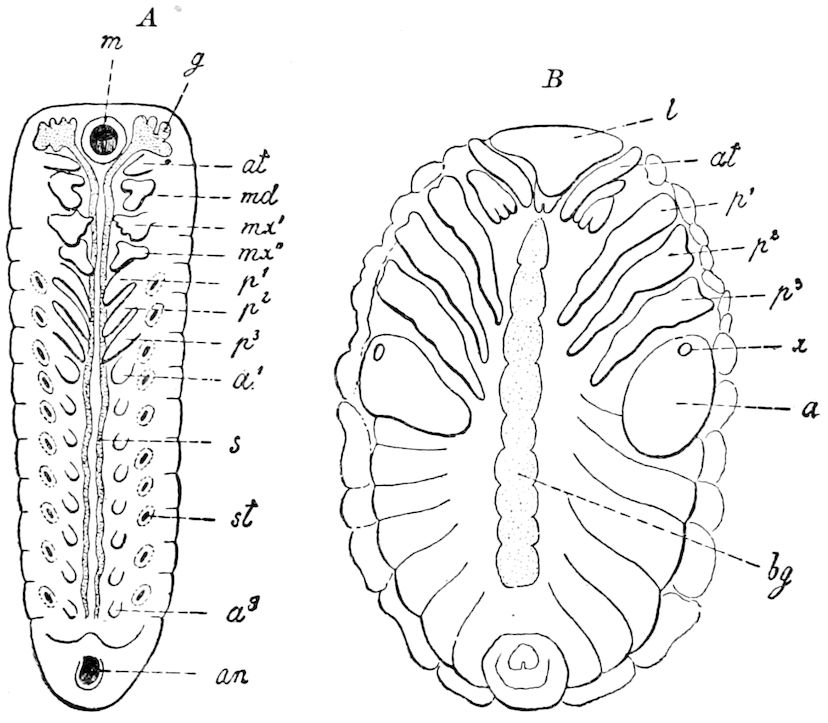
Fig. 528.—Two embryonic stages of the primitive streak of Melolontha. A, younger stage, with rudiments of eight pairs of abdominal appendages (a1–a8). B, older stage, the primitive band now very broad: a, 1st abdominal appendage, in B sac-like; x, place of adhesive disc; g, brain; l, clypeo-labrum; s, lateral cord of the ventral nervous cord; other lettering as in previous figures.—After Graber, from Korschelt and Heider.
The distal parts of the labium, such as the ligula, palpifer, and palpus are elaborated before the mentum and submentum. Many details as to the final changes in the mouth-parts before hatching remain to be worked out.
550The thoracic appendages.—The three pairs of legs arise at the same period and in the same manner in all insects; it is not until the end of embryonic life that they become jointed, and that the claws and onychia are developed. Especial attention has not yet been given to the details of the development of the parts of the last joint of the tarsus.
In many forms the antennæ are the first to appear, the mandibles, maxillæ, and legs appearing at a latter date, though simultaneously. It is thus in Stenobothrus, Hydrophilus, and Melolontha. In Lina, according to Graber, the mandibles precede the antennæ in appearance. In the Libellulidæ, according to Brandt, the legs first appear, then the jaws, and lastly the antennæ. This did not seem to be the case in the embryos of Æschna observed by us, although our observations were more superficial.
On the other hand, in those insects whose larvæ are footless, the rudiments of the legs are retarded and aborted just before hatching (fossorial Hymenoptera and Apidæ), or the rudiments of the legs are not developed at all.
The abdominal appendages.—These appear soon after the thoracic limbs, corresponding in most cases to the latter in shape and position, and their position in the embryo is a matter of the greatest interest. Von Rathke was the first embryologist to detect those of the first abdominal segment, in his examination of the development of Gryllotalpa. Long afterwards Bütschli detected them in the embryo of the honey-bee, observing a pair on each segment. Patten observed them in Trichoptera; Kowalevsky first perceived them in Lepidoptera, Tichomiroff confirming his observations. Graber, Ayers, and Wheeler have observed them in Orthoptera and Coleoptera, and the latter has detected them also in Hemiptera and Neuroptera; and while they do not arise in the embryos of Diptera and of Siphonaptera, they are to be looked for in any or all the lower or more generalized orders.
As the result of these discoveries of polypodous embryos occurring in all but the most specialized order (Diptera), it appears to be a rational deduction that the winged insects have descended from insects in which there were functional legs on each abdominal segment. Such an ancestor was the forerunner of the Thysanura, in which abdominal locomotive appendages still survive, though in a modified, more or less aborted condition. This polypodous ancestral form was apparently allied to Scolopendrella, which has a pair of functional legs on each abdominal segment.
The subject, then, of polypodous embryo insects is one of special interest, and has attracted much attention from Graber, Wheeler, Haase, and others. That these are genuine, though transitory appendages, is shown by the fact that certain pairs persist throughout adult life. The embryology of the Thysanura 551when worked out will throw much light on this subject, but we know that the spring (elater) of Collembola (and possibly the collophore) and the cerci of the winged insects are survivals of these limbs. That the three pairs of appendages forming the ovipositor, or sting, are most probably derived from these appendages is claimed by Wheeler (p. 167), and seems proved by the fact that Ganin and also Bugnion has detected three pairs of imaginal disks in the embryo of parasitic Hymenoptera. Hence the abdominal appendages may ultimately be found to arise in nearly all cases from imaginal disks like those giving origin to the cephalic and thoracic appendages.
As regards the Diptera, Pratt has observed that each of the three thoracic and eight abdominal segments of the embryo brachycerous Diptera (seen especially well in Melophagus) has two pairs of imaginal disks, a dorsal and a ventral pair. He thinks there is no doubt but that the ventral abdominal disks are homologous with the rudimentary appendages which appear in the embryos of all other insects, though not in the brachycerous dipters.
Appendages of the first abdominal segment (pleuropodia).—As early as 1844 Rathke observed in the embryo of the mole-cricket a pair of appendages on the 1st abdominal segment, which he described as mushroom-shaped bodies, and supposed to be embryonic gills. They are called pleuropodia by Wheeler, who, with Patten, Graber, and Nusbaum, ascribes a glandular function to them, while Wheeler suggests that they were odoriferous repugnatorial organs. In Blatta (Phyllodromia) they are of large size, in Melolontha enormous (Fig. 528, B) and filled with blood. Wheeler distinguishes as varieties, beside the mushroom-shaped appendages of Gryllotalpa and Hydrophilus, the reniform (Œcanthus), the broadly pyriform (Blatta), and the elongate pyriform (Mantis carolina). In the European Mantis they are most limb-like, with a digitiform continuation divided by a constriction into two sections. (Graber.) In Meloë they assume the shape of a stalked cup. (Nusbaum.) In the bee and in Lepidoptera the pleuropodia are not present, though the temporary appendages on the succeeding segments appear; Carrière, however, found them on the two first abdominal segments of very young larvæ of the wall-bee (Chalicodoma).
Their cellular structure is peculiar, and they are either formed by evagination or invagination, those of the latter type being subspherical and solid. Those of the former type have a cavity communicating by means of a narrow duct through the peduncle with the body-cavity (Blatta). No tracheæ, nerves, or muscles enter them, though blood-corpuscles have been seen in the cavities. “In some species the pleuropodia produce a secretion from the ends of their enlarged cells. This secretion may be a glairy albuminoid substance (Cicada, Meloë), a granular mass (Stenobothrus), a bundle of threads (Zaitha), or a thick, striated, cuticula-like mass 552(Acilius).” They attain their greatest size during the revolution of the embryo, and they are “mere rudiments of what were probably in remote ages much larger and more complex organs.” (Wheeler.)
Lameere has observed that in Phyllodromia the first pair of abdominal appendages, after becoming of considerable size, undergo an enlargement at their free end, become detached, and fall into the amnion.
Wheeler also calls attention to the homology of these pleuropodia with the 1st abdominal appendages of Campodea, shown by Haase to be originally glandular, but with at present a respiratory function. In the embryos of later, higher orders of insects, these appendages are in size and shape similar to those of the succeeding segments. (See also p. 164.)
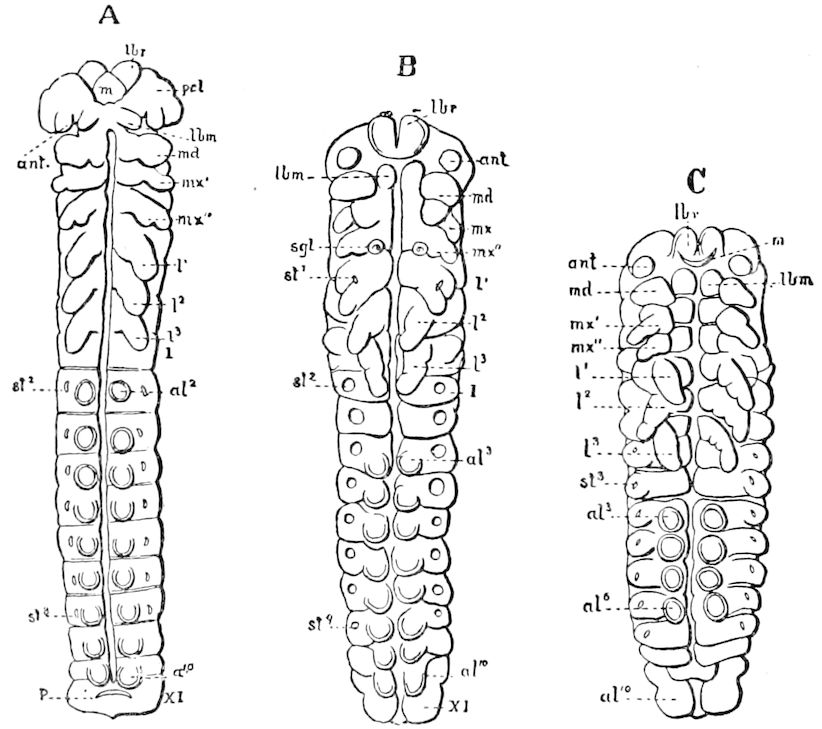
Fig. 529.—Primitive band of Bombyx mori, showing the temporary legs on abdominal segments 2–11: A, early stage, in which the abdominal legs al2–al10 appear. B, later stage, when they are very faint and all except al3–al6 and al10 are about to disappear. C, the persistent abdominal legs al3–al6 and al10; st2, st9, the 2d and 3d pair of stigmata; sgl, silk duct.—After Tichomiroff.
Are the abdominal legs of larval Lepidoptera and phytophagous Hymenoptera true limbs?—The presence of these abdominal legs in the embryos of Sphinx (Kowalevsky), of Bombyx mori (Tichomiroff), and both Bombyx mori and Gastropacha quercifolia (except those of the first segment), as well as in Hylotoma, which has 11 pairs of such appendages, has suggested that the prop or prolegs of caterpillars and saw-fly larvæ are survivals of these outgrowths, and not secondary, adaptive structures. Opinions on this point vary. Balfour, and also, more recently, Cholodkowsky, hold that the prolegs are survivals of the embryonic appendages. Graber cautiously, after a 553lengthy and interesting discussion, says that the question cannot be, in the present state of our knowledge, solved. He, however, seems inclined to believe that the prolegs are not merely secondary structures, and that the rudiments of limbs may remain for a long time in a latent state before their final development. Korschelt and Heider are disposed to regard the abdominal appendages of Lepidoptera and Hymenoptera as true limbs, referring to Balfour’s statement that in the Crustacea there are different examples of the loss and later appearance of limbs, such as the loss of the mandibular palpi of the zoëa of decapods, and the loss in the zoëa of appendages in the Erichthus form of the Squilla larva corresponding to the third pair of maxillipedes and first two pairs of legs of Decapoda, and which are afterwards reproduced; similar cases occurring in the Acarina. In the wasps and bees also, as is well known, the imaginal disks of the thoracic appendages appear, the legs themselves being suppressed in the larva (the imaginal disk probably existing in an indifferent state), to reappear in the pupa and imago. It does not, however, necessarily follow that the numerous pairs of hooked ventral tubercles of certain dipterous larvæ (Ephydra) are true appendages.
It seems to us that it is a strong argument for the view that these prolegs are survivals of primitive limbs, that from similar embryonic paired outgrowths on different segments arise the spring of Podurans, the anal cerci, and three pairs of appendages forming the ovipositor, and the anal legs of the Corydalus larva, as well as those of caddis-worms; at least five abdominal segments throughout the class of insects as a whole bearing appendages in the adult.
On the other hand the view of Haase, that the prolegs of caterpillars are secondary, adaptive characters, is supported by the fact of the rapidity with which two pairs on the 3d and 4th segments nearly disappear in the larvæ of certain Noctuidæ (Catocala, etc.), a reduction evidently due to disuse.
The tracheæ.—The tracheal system arises as ectodermal invaginations on one side of the appendages, appearing soon after the latter. The earliest condition of the tracheal invagination is seen in section at Fig. 539, E, tr; as it deepens, it sends off diverticula or tracheal branches, while the narrow mouth of the invagination forms the stigma. The cup-like cavities situated serially one behind the other, and arising from the single tracheal invaginations, become at the end or bottom of the cup elongated along the length of the body and fused together at their ends; then the two longitudinal stems of the system arise, by a breaking through at the place where the 554original invagination had become fused, thus forming a continuous tube, the lumina opening into each other. (Bütschli.)
The cuticular tracheal intima is differentiated late in embryonic life. The entrance of the air is accomplished in part before the embryo hatches, the air being derived from the tissues and fluids of the body.
The farther development of the tracheal branches is due to the progressive formation of diverticula. The branches thus arising are intercellular formations. On the other hand, the finest twigs are intercellular structures. However, as Schaeffer states, the differences between the two modes of formation are not important.
Wheeler mentions the existence of “two pairs of very indistinct tracheal openings in the 10th and 11th somites” of the abdomen of Doryphora (Fig. 546, t19, t20), and Heider believes that they exist in Hydrophilus.
The tracheal invaginations as a rule begin to appear after the appendages commence to bud out. An exception is met with in the bee (Apis), where the tracheal ingrowths are seen before the rudiments of the legs. Most of the tracheal invaginations appear simultaneously. Only rarely do we see an indication of their successive development from before backwards. Thus in Hydrophilus, Graber observed that the mesothoracic stigmata appeared somewhat earlier than those of the other segments.
h. Nervous system
The rudiments of the nervous and tracheal systems essentially contribute to the building up of the relief of the primitive band of insects. The nervous system is the earliest to appear, being indicated very early, in fact before the appendages begin to grow out. The first traces of the nervous system are two ridges extending along the primitive band, the depression between them being called the primitive furrow. At an early period the segmentation is observed in the primitive ridges, while widened spaces (the rudiments of the ventral ganglia) alternate segmentally with the narrow places which are the incipient longitudinal commissures (Fig. 527, A, g).
The primitive ridges extend anteriorly into the head-lobes; this part must be regarded as the rudiment of the œsophageal commissure. The rudiments of the brain are from their first appearance directly connected with the ventral chain of ganglia.[83]
555Completion of the definite form of the body.—This is accomplished by the growth of the primitive band around the yolk, the band widening, so that its edges behind the head extend up, and finally meet on the back, forming the back or tergum of the embryo, thus enclosing the yolk (Fig. 530, F). The tergal wall of the head is due to the dorsal growth of the head-lobes, and of the clypeo-labral region. In the course of this process the anterior end of the primitive band becomes turned up dorsally, forming a dorsal curve or bend. By this bending up of the primitive band the forehead nearest the mouth forms a transverse ridge, the labrum, while the basal or earlier part of the forehead now is differentiated into the clypeus. This clypeo-labral region likewise forms the roof or palatal region of the mouth. The head-lobes cause by this dorsal growth a rotating motion which carries the rudimental antennæ back over the mouth.
Fig. 530.—Diagram of the formation of the dorsal organ in Hydrophilus. A, cross-section through an egg, whose primitive streak is still covered over by amnion (a) and serosa (s). B, amnion and serosa are grown together in the middle line, then separated and drawn back to form a fold on each side. C, by the contraction of the serosa (s),which becomes converted into the dorsal plate, the folds become drawn up dorsally. D, the contracted serosa becomes partly overgrown by the folds. E, the folds grow together to form the dorsal tube. F, the mid-gut has closed over dorsally and enclosed the dorsal tube (s): a, amnion; d, yolk; ec, ectoderm; h, heart; l, body-cavity; m, rudiment of the mid-gut; n, nervous system; s, serosa (in C and D = dorsal plate, in E and F, dorsal tube); tr, the chief tracheal stem.—After Graber and Kowalevsky, from Lang, and Korschelt and Heider.
556The gnathal or post-antennal segments at first bear but a small part in completing the tergal region of the head, but shortly before hatching the mandibles and their muscles enlarge, giving fulness to the upper and back part of the head.
i. Dorsal closure and involution of the embryonic membranes
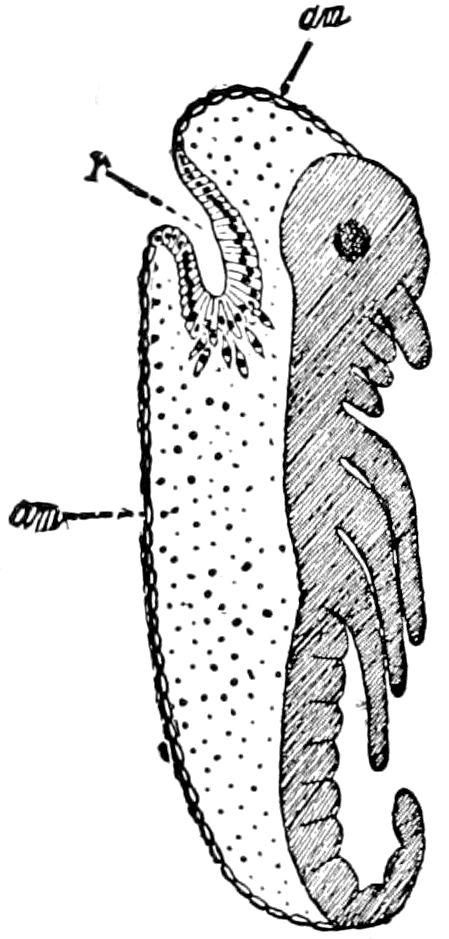
Fig. 531.—Schematic figure of the formation of the dorsal tube by invagination of the dorsal plate (transformed serosa); following after stage Fig. 520, C, and Fig. 521, D; am, amnion (now forming the provisional dorsal closure); r, dorsal tube, whose cells are already breaking away.—After Korschelt and Heider.
In most other Arthropoda (Crustacea, Arachnida, Myriopoda, etc.) development goes on by the formation of a so-called primitive band, but without the appearance of peculiar embryonic membranes. The outer surface of the entire egg becomes, then, in part covered by the band-like embryonic germ, and partly by a portion of the blastoderm which remains unchanged. The dorsal region is formed by the widening and spreading of the primitive band over the greater part of the surface of the egg, while the area of the unchanged section of the blastoderm continually becomes more restricted. It is generally accepted that the latter is concerned in the dorsal closure, because, together with a histological transformation, it becomes involved in the formation of the ectoderm of the primitive band.
A similar form of retrograde structure possibly occurs in the embryos of Poduridæ, in which a dorsal organ has been observed to develop in an early embryonic stage, which bears some relation to the cuticula enveloping the embryo, but whose significance is in general rather obscure.
In most insects the relations are more complicated, since in such cases, the amnion-folds rise on the edges of the primitive band and of the unchanged section of the blastoderm, whose retrograde development is intimately connected with the closure of the back.
A very simple case of dorsal closure, but which certainly is not a primitive one, occurs in Muscidæ and certain other Diptera whose amnion-folds are developed in a rudimentary way. In this case (according to Kowalevsky and Graber), the amnion-folds become smoothed out again. Amnion and serosa become then a simple epithelium, which throughout corresponds to the unmodified type of blastoderm of Crustacea, Arachnida, and Myriopoda, and here seems to share in the formation of the back. More complicated and very manifold relations of dorsal closure and involution of the embryonal membranes occur in other insects, of which Korschelt and Heider distinguish four different types:
5571. Involution under the formation of a continuous dorsal amnion-serosa-sac (Odonata).
2. Involution with exclusively dorsal absorption of the amnion (Doryphora).
3. Involution with exclusively dorsal absorption of serosa and separation of the amnion (Chironomus and Trichoptera).
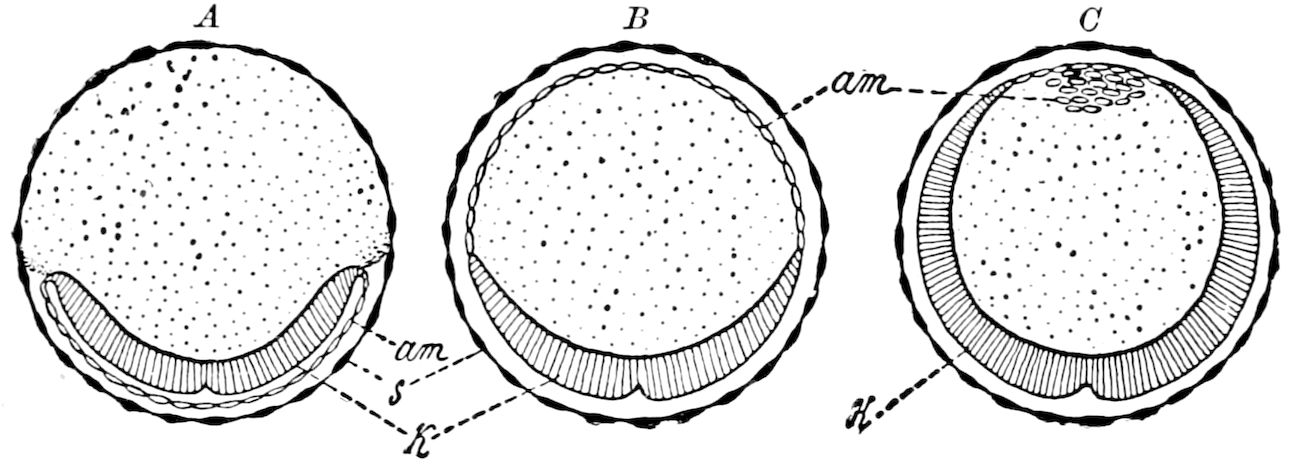
Fig. 532.—Diagram of the formation of the dorsal walls in Doryphora in cross-sections: am, amnion; in B, serving as a provisional dorsal closure, in C, about to break up; k, primitive band; s, serosa.—After Wheeler, from Korschelt and Heider.
4. Involution with separation of both embryonic membranes (Lepidoptera and Hymenoptera, Hylotoma).
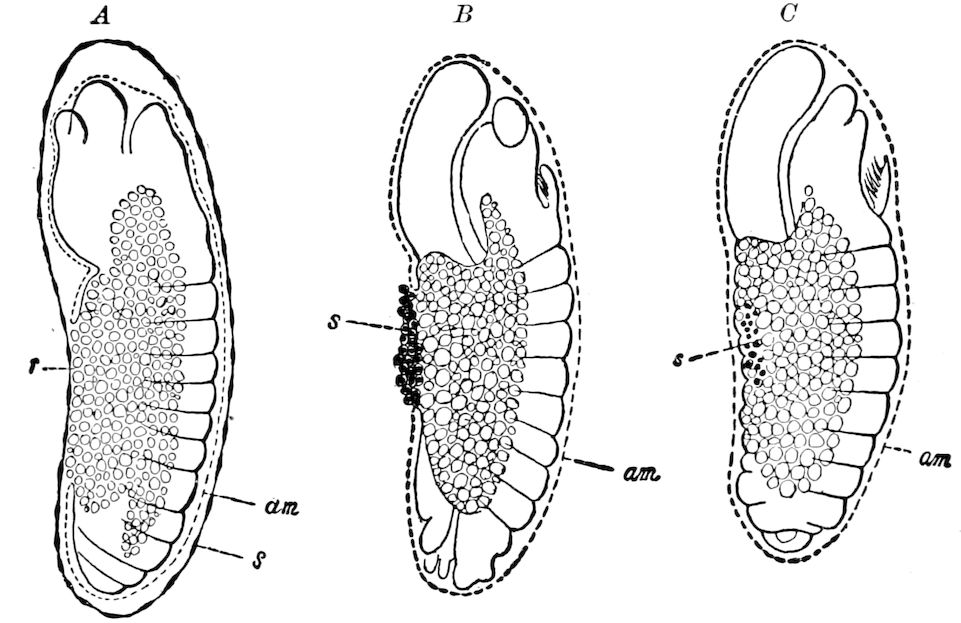
Fig. 533.—Involution of the embryonic membranes of Chironomus: am, amnion; r, dorsal umbilicus; s, serosa, which has withdrawn into the region of the dorsal umbilicus, and in C has passed into the interior of the embryo.—After Graber, from Korschelt and Heider.
The first type occurs in the most primitive order of winged insects. The second type (Coleoptera) appears to be an independently inherited form of dorsal closure. In the first type, the formation of the amnion-serosa-sac is initiated by a rupture of the two fused embryonic membranes. This rupture in the ventral middle line occurs in Odonata only in the region of the head-section. 558In the second type only the amnion, in the third only the serosa are concerned in this rupture, while in the fourth type both membranes remain intact until the slipping out of the larva. (Korschelt and Heider.)
j. Formation of the germ-layers
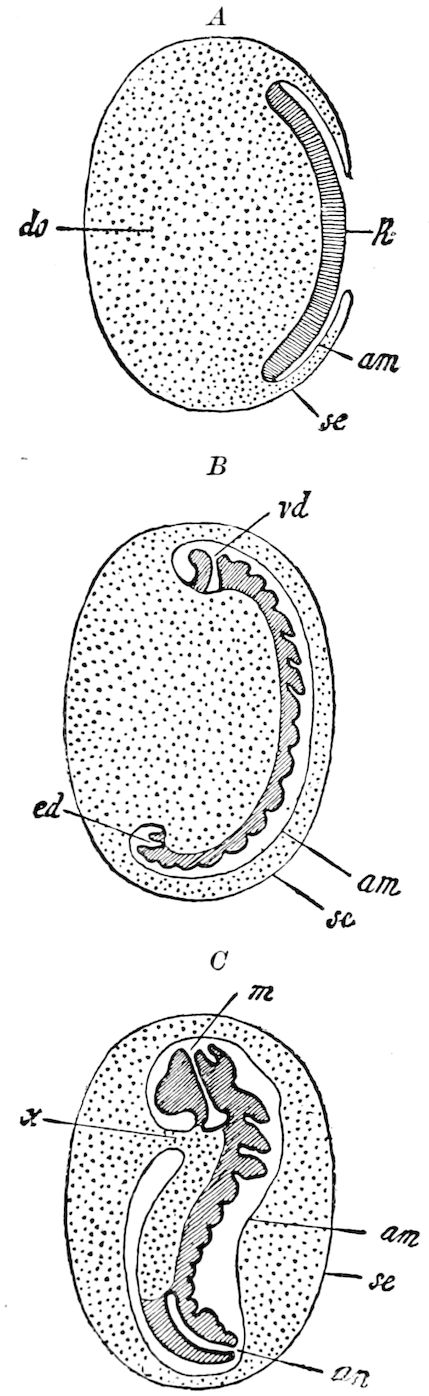
Fig. 534.—Diagram showing the formation of the embryonic membranes in Lepidoptera (A, after Kowalevsky, B and C, after Tichomiroff): k, primitive band; am, amnion: se, serosa; do, yolk; vd, invagination of the fore-gut, ed, of the hind-gut; m, mouth; an, anus; x, dorsal umbilical passage.—From Korschelt and Heider.
The older views on the structure of the layers of the primitive band of insects were thoroughly unsatisfactory. Bütschli first found that in the bee, by a kind of folding process, an inner layer of the primitive band arose. Soon afterwards Kowalevsky, by the employment of section-cutting and thorough researches, laid the foundation of a more exact knowledge of these layers. He found that in Hydrophilus a furrow extended along the whole length of the primitive band (Fig. 515, A, B, r), which, while invaginating or sinking in, gave rise to the inner layer of the primitive band, i.e. the common rudiment of endoderm and mesoderm (Fig. 539, A-C).
Kowalevsky also found similar conditions in the honey-bee (Apis), Lepidoptera, and other forms. The furrow above mentioned must be regarded as a very long gastrula invagination, extending along the entire ventral side of the embryo, and the edges of the furrow as a long-drawnout blastopore. The tube arising in Hydrophilus through the closing of the furrow we may regard as a primitive intestinal canal.
The first rudiment of the gastrula furrow appears in insects as two folds extending along both sides of the median line in the thickened ventral plate (Fig. 536, f), through whose 559formation a more median section of the ventral plate, the so-called middle plate (m), becomes separated from the side plates (s). As the middle plate curves in and becomes overgrown by the folds forming the edges of the blastopore, the gastrula-tube (Fig. 539, A, r) is formed, and furnishes the rudiments of the lower (inner) layer. The ectoderm, then, according to Heider, arises from the lateral plates of the primitive band. The growth of the edges of the blastopore, by which the closure of the gastrula-tube is effected, takes place latest in the region of the most anterior part of the furrow (Fig. 515, B and C), corresponding to that place in the primitive band in which the stomodæum afterwards develops.
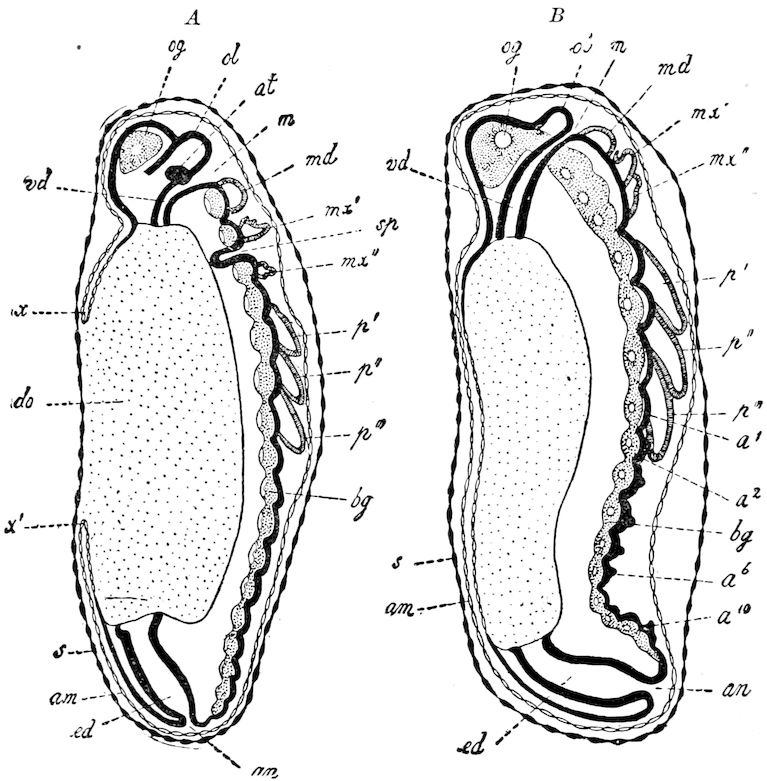
Fig. 535.—Two embryonic stages of a saw-fly (Hylotoma berberidis) in schematic median section: a1–a10, 1st to 10th abdominal segments; bg, ventral nervous cord; og, brain; ol, germ of labrum; sp, salivary gland; ed, hind-gut; x, x′, inner folds of amnion: other letters as before.—After Graber, from Korschelt and Heider.
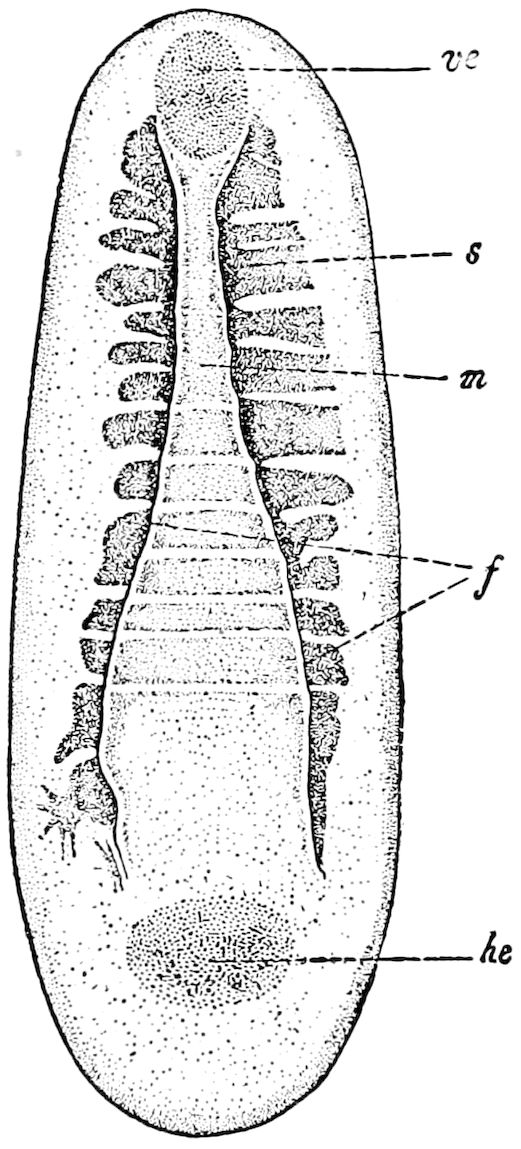
Fig. 536.—Gastrula stage of the wall-bee (Chalicodoma), so-called flask-shaped stage: f, folds which on each side border the middle plate (edge of the blastopore); m, the partly segmented middle plate (here = rudiment of the mesoderm); s, the segmented lateral plate (becoming afterwards the ectoderm of the primitive band); ve, fore, he, hinder entodermal rudiment.—After Carrière, from Korschelt and Heider.
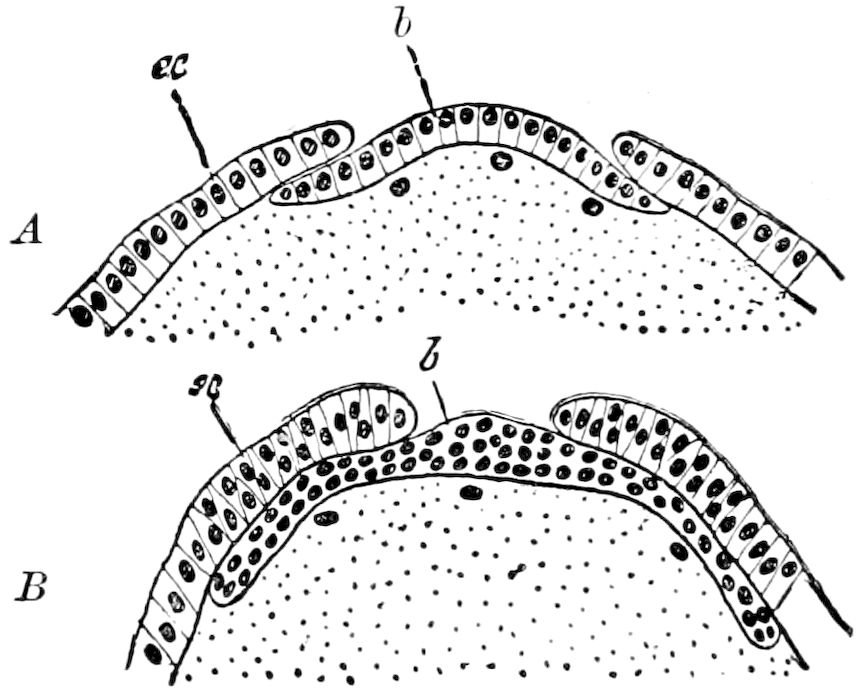
Fig. 537.—Two successive stages in the gastrulation of Apis. Cross-section through the primitive band: b, lower (inner) layer; ec, ectoderm.—After Grassi, from Korschelt and Heider.
During the invagination of the middle plate and its transformation into the gastrula-tube a change takes place in its histological character (Fig. 539, A and B). While it originally consists of a high cylinder epithelium, which after farther changes becomes divided into several layers, since the wedge-shaped single cells push themselves over each other, the cells in later stages become more and more cubical or irregularly polygonal (Fig. 539, B), and are irregularly arranged. At the same time the gastrula-tube is compressed in a dorso-ventral direction. While it in this way spreads out laterally under the side plates (ectoderm), its originally circular primitive lumen passes into the form of a horizontal fissure, which in Hydrophilus long remains as the boundary between the two layers of the inner (or lower) membrane. (Korschelt and Heider.)
There are numerous variations of the process of gastrulation, which are by Korschelt and Heider divided into three types, as follows:—
1. Through invagination and formation of a tube (Fig. 539, A, Hydrophilus, Musca, Pyrrhocoris, etc.).
2. By a lateral overgrowth (Fig. 537, Lepidoptera and Hymenoptera).
3. By an inward growth of cells from a median furrow (Aphides and Trichoptera).
In Doryphora and Lina (Fig. 524) the hinder end of the gastrula furrow is forked.
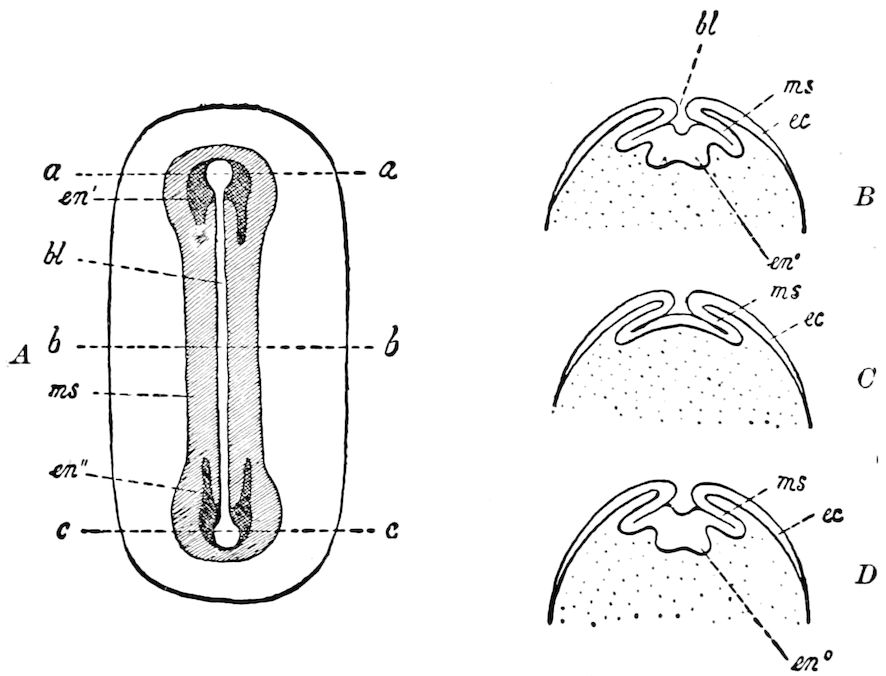
Fig. 538.—Diagrammatic sketch of the formation of the germinal layers in Doryphora: A, view of upper surface. B, cross-section through the fore end of the primitive streak at the line a-a. C, section through the middle of the primitive streak corresponding to the line b-b. D, section through the hinder end of the primitive band corresponding to the line c-c: bl, blastopore; ec, ectoderm; en′, anterior U-shaped; en″, hinder U-shaped germ of the endoderm; ms, mesoderm.—After Wheeler, from Korschelt and Heider.
The cellular layer arising from the gastrula invagination (lower layer) forms the common germ of the endoderm and mesoderm. It has only recently become known how these two germ-layers of insects have become differentiated. Kowalevsky first discovered in Musca that the greatest part of the lower (inner) layer yielded mesoderm exclusively, and that a cell-mass only corresponding to the most anterior and posterior end of the primitive band was used in the formation of the endoderm. We must therefore, in insects, speak of a fore and a hinder endodermal rudiment. In proportion, now, as the ectodermal invaginations, which are destined to form the stomodæum and the proctodæum sink beneath the surface of the embryo, the cell-masses of which the two endodermal rudiments are composed are pushed farther in, and a separation between them and the mesoderm is thus effected. The two endodermal rudiments now form accumulations of cells which lie closely adjacent to the blind ends of the stomodeal and the proctodeal invaginations. They soon widen out into two hour-glass-shaped rudiments, which are directed with their concavities towards each other, but with their convex side towards the nearest pole of the egg. They soon change their form; two lateral stripes grow out from them, and each now assumes the form of a U (Fig. 538, en′). The limbs of the fore 562and hind U-shaped rudiment are directed toward each other, and grow towards each other until they meet, and are fused together. Thus the endodermal rudiments arising out of the fusion of the two U-shaped rudiments form two stripes extending along the primitive band and situated mostly under the primitive segments. At the two ends the endodermal rudiment fuses with the stomodeal and proctodeal invaginations. These lateral endodermal streaks now spread out, and gradually begin to grow over the yolk, on whose outer surface they lie. This overgrowth makes the greater advance on the ventral side, so that the two endodermal streaks first unite in the ventral median line and afterward in the dorsal. The yolk in this way passes completely into the interior of the rudiment of the mid-intestine.
Kowalevsky has already proved that it is the median parts only of the inner layer which at the two ends of the primitive band become separated as endodermal rudiments through the advance of the stomodeal and proctodeal invaginations: the lateral portions become mesoderm.
Kowalevsky has compared the germ-layers of insects with those of Sagitta. This comparison is supported by the later researches of Heider and of Wheeler on Coleoptera. (See Korschelt and Heider, p. 809.)
Relations somewhat different from the common type of formation of germ-layers occur in Hymenoptera. Kowalevsky and also Grassi agree that here also the endoderm originally forms a part of the lower (inner) layer. But the separation of the endoderm from the mesoderm goes on in Apis in such a way that the two ends of the inner layer pass up to the dorsal side of the egg, where the fore and hind rudiments of the endoderm extending along the back of the embryo grow together. When the two horseshoe-shaped rudiments have met each other and become fused, the enclosing of the yolk begins, which accordingly here proceeds from the dorsal towards the ventral side, instead of vice versa. As a result the endodermal cell-layer in Apis (and also Chalicodoma) at first does not lie under the primitive band, but on the dorsal side of the egg under that flat epithelium, which, arising from the amnion-fold, completes the provisional closure of the back.
The yolk-cells and secondary yolk-segmentation are discussed by Korschelt and Heider at this point. The yolk-cells are elements scattered throughout the yolk and which partly remain in the yolk during the formation of the blastoderm (Fig. 507, C and D), but which in part through a later immigration pass out of the blastoderm into the yolk. Graber has proved the fact of the migration of cells from the lower layer into the yolk, and his observations have been confirmed by other authors. Indeed, in certain cases (Melolontha), these later immigrant cells are clearly distinguishable by their histological characters from those originally found in the yolk.
The yolk-cells are regularly scattered throughout the yolk. Their use to the embryo lies in the fact that they absorb the particles of yolk, which they digest and thus reduce to a fluid condition. It usually happens that after the complete formation of the primitive band there results a delimitation of the areas enclosing each yolk-cell, and this occurrence is called secondary yolk-division. 563In special cases (Apis, Musca) this occurrence seems not to take place. The yolk-cells are still, after the complete formation of the mid-intestine, to be recognized in the yolk-remnants filling the interior of the same, and gradually become absorbed.
k. Farther development of the mesoderm. Formation of the body-cavity
We have seen that by means of an invagination extending throughout the entire length of the primitive band a layer of cells is produced which soon spreads out on the inner side of the band and thus forms a second lower (inner) layer (Fig. 539, C). From this inner layer is separated at the anterior and posterior ends of the primitive band, the endoderm, which lies in direct contact with the invaginations of the proctodæum and stomodæum. The remainder, by far the most extensive part of the inner layer, is the mesoderm.
The mesoderm now becomes divided into two lateral streaks (mesodermal streaks), by the withdrawal of its cells from the median line (Fig. 539, D). This withdrawal is not, however, always a complete one. In the free median space thus formed, the yolk often forms the so-called median yolk-ridge. Segmentally arranged cavities soon appear in the lateral region of the mesoderm (the primitive segmental cavities), and the bordering mesoderm-cells arrange themselves in the form of an epithelium, and constitute the wall of the primitive segments or cœlom-sac. (Korschelt and Heider).
The primitive segmental cavities in general arise through a split in the mesoderm. In Phyllodromia, according to Heymons, the primitive segments are very extensive. The mesoderm, at the time of the formation of the rudiments of the appendages, is raised with the ectoderm from the surface of the yolk, and in this way there arise in each segment cavities, which, since they are surrounded by mesodermal elements, become the closed cœlom-sacs (Fig. 540, c, c′, c″).
The cœlom-sacs differ in different groups. They are largest in Orthoptera (Phyllodromia), where they take up almost all the cell material of the mesoderm in their formation, and exhibit certain conditions recalling those of Peripatus. The very large primitive segmental cavities, which in Orthoptera also extend into the rudiments of the appendages (Fig. 540, B, ex), in their later stages are, through the formation of a constriction, divided into a dorsal and a ventral half (Fig. 540, B, c′, c″). The ventral portions of these cavities extending into the extremities soon disappear, while the cells of their walls lose their epithelial nature, and group themselves irregularly into a sort of mesenchym. In this tissue, then, arises, partly through a separation among its cells, partly through the elevation of the same from the upper surface of the yolk, the definite body-cavity. The dorsal portions of the primitive segmental cavities remain unchanged a longer time in order to play a rôle in the formation of the intestinal muscular layer, of the heart, pericardial septum, and sexual organs.
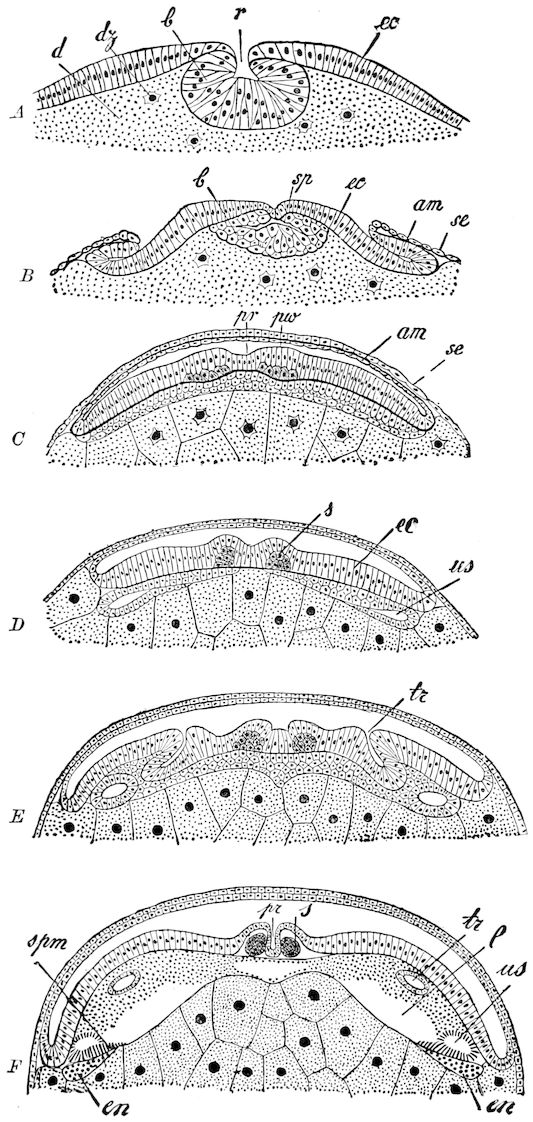
Fig. 539.—Cross-section through the primitive streak of Hydrophilus in six successive stages: A, gastrula-stage (compare Fig. 515, A, corresponding to the point a). B, cross-section through stage, Fig. 515, D, in the most anterior section of the primitive band, where the same is not completely overgrown by the amnion-folds. C, cross-section through the trunk-segment of stage, Fig. 515, E. D, E, F, cross-sections through later stages: am, amnion; b, lower (inner) layer; d, yolk; dz, yolk-cells; ec, ectoderm; en, entoderm; l, definite body-cavity; pr, primitive groove (= neural groove); pw, primitive roll, or strip, of the ventral nerve-cord; r, blastopore; sp, fissure in the mesoderm (remains of the cavity of the primitive intestine); se, serosa; s, lateral cord of the rudiment of the nervous cord; spm, splanchnic layer of the mesoderm; tr, rudiment of a trachea (in E appearing as an invagination of the ectoderm) in F in cross-section; us, primitive segment (= cœlomic sac).—After Heider, from Lang.
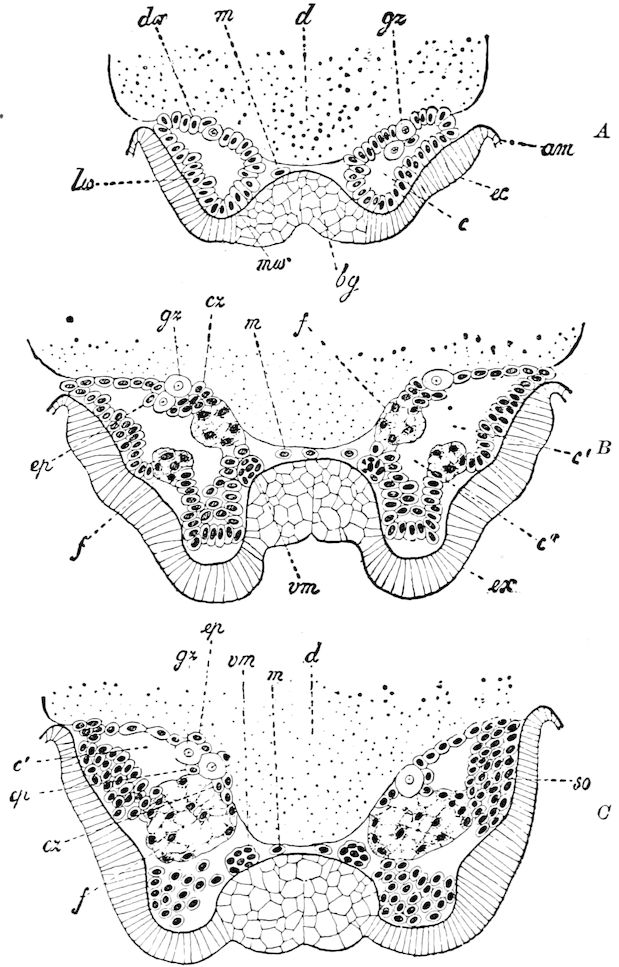
Fig. 540.—Cross-sections through the abdominal part of three successive stages of evolution of Phyllodromia germanica: am, amnion; bg, rudiment of the ventral nervous chord; c, cœlomic cavity; c′, dorsal, and c″, ventral, section of the cœlomic sac; cz, cells of the walls of the primitive segment, which are joined to the genital rudiments; gz, genital cells; dw, dorsal wall of the cœlomic sac; d, yolk; ec, ectoderm; ep, epithelium-cells; ex, rudiment of the abdominal appendages; f, germ of the fat-body; lw, lateral wall of the cœlomic sac; m, mesoderm cells, which take no part in the formation of the cœlomic sac; mw, median wall of the cœlomic sac; so, somatic mesoderm layer; vm, ventral longitudinal muscle.—After Heymons, from Korschelt and Heider.
In the highest groups of insects (Coleoptera, Lepidoptera, and Hymenoptera) the primitive segments are not so extensively developed (Fig. 539, D-F, us). They here form only relatively small sacs situated in the lateral parts of the primitive band which correspond to the dorsal section of the cœlom-sacs of 566Orthoptera. The ventral part is here from the very outset replaced by a mesenchym. As a result in these forms also no cœlomic diverticula occur in the rudiments of the extremities.
The definite body-cavity of insects arises entirely independent of the cœlom cavities, and in fact, as Bütschli showed, through the separation of the primitive band from the yolk (Fig. 539, F, l). It appears bounded on the one hand by the surface of the yolk, on the other side by the irregularly arranged mesenchym cells. Originally we can in cross-sections distinguish three separate cavities of the definite body-cavity (in Hydrophilus according to Heider), a median and two larger paired lateral ones which later fuse with each other and with wide lacunæ (e.g. in the appendages) arising by the separation of the mesenchym cells. We refer the compartments of the definite body-cavity, as in Peripatus, to the primary body-cavity or segmentation-cavity. They are only lacunæ in the area of the mesenchym, and throughout bear the character of a pseudocœl.
In later stages of embryonic development the cœlom-sacs and the definite body-cavity enter into communication with one another (Fig. 523, A, us, lh). (Korschelt and Heider.)
Then the hinder cœlom-sacs unite through the degeneration of the transverse dissepiments which separate them. After this a fissure opens in the median wall of the cœlomic sac, through which its cavity unites with the definite body-cavity. In the subsequent changes which the wall of the cœlom-sacs undergoes, these can be recognised no longer as separate divisions of the whole body-cavity.
l. Formation of organs
The nervous system.—As we have already seen (p. 554), the rudiments of the ventral nervous cord arise, after the gastrula invagination is completed, as two ectodermal thickenings situated on each side of the median line, the so-called primitive rolls or strips (Fig. 528, s), which extend from the centre of the procephalic lobes of the head to the last segment, enclosing between them the single median “primitive groove” (Fig. 539, C, pr, and pw).
Soon after the appearance of the primitive strips, the first traces of segmentation may be detected. The ventral cord is from the first in direct connection and continuous with the brain. From the segmental expansions of the primitive strip arise the ventral nervous ganglia, and from the intersegmental constrictions are developed the paired longitudinal commissures.
Transverse sections of the ectoderm in the region of the primitive strips (Figs. 539, C, and 517) show several layers of cells. Of these cellular layers the deeper ones afterwards, by a kind of delamination, separate from the superficial ones and form the “lateral cords,” i.e. the germs of the longitudinal cords of the ventral ganglionic cord. Meanwhile the primitive groove (pr) deepens and forms an invagination extending between the lateral cords. The cells at the bottom of this invagination form the so-called “median 567cord,” and give rise to the transverse commissures connecting the ganglia.
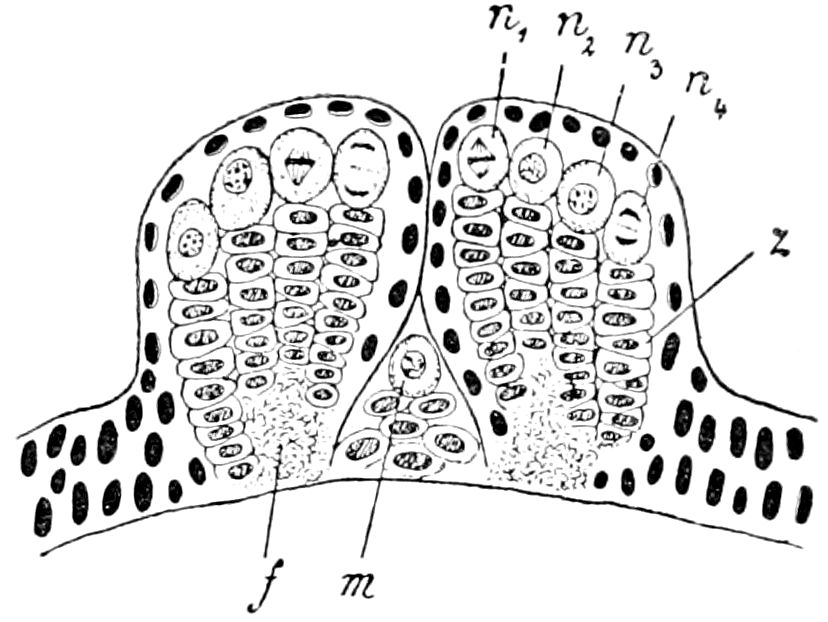
Fig. 541.—Transverse section through the rudiment of the ventral nervous cord of Xiphidium: f, fibrous mass; m, neuroblast cells of the median cord; n1-n4, neuroblasts of the lateral cord; z, pillar of ganglion-cells arising from the neuroblasts.—After Wheeler.
Wheeler has detected in the rudiment of the ventral cord of several Orthoptera, on the upper surface of the lateral cords, four large cells which he calls neuroblasts (Figure 541, n1-n4), from which cells arise by budding and become arranged in vertically arranged layers or pillars (z). Graber has observed them in Stenobothrus and Viallanes in Mantis. These neuroblasts are only present in the inter-ganglionic region, and soon move back to the hinder side of the transverse commissures.
At first there is a pair of ganglia to each of the 16 trunk-segments of the embryo, but afterwards these become more or less fused together; thus those of the three gnathal segments unite to form the subœsophageal ganglion of the adult, and the last abdominal ganglia are fused together and move a little anteriorly (see also pp. 227, 228).
Development of the brain.—The supraœsophageal ganglion is due to the spreading out of the procephalic lobes. The rudiment of the brain is due to a thickening of the ectoderm on the sides of the mouth and of the forehead, this expansion of germinal brain-cells being the direct continuation of the primitive rolls or strips, and which finally becomes differentiated into the protocerebrum, deutocerebrum, and tritocerebrum, as stated on p. 228.
The ganglion opticum, now regarded as a part of the compound eye, arises as an ectodermal thickening on each side of the rudimentary brain. The optic ganglion belongs exclusively to the foremost division of the brain (see also p. 227).
Development of the eyes.—Compound eyes do not appear until the beginning of pupal life, the single eye (ocellus) being the primitive organ of vision. The ocellus of Acilius, according to Patten, arises as a pit or depression of the ectoderm (Fig. 542). The long hypodermal cells which form the walls of this pit or hollow are arranged in a single layer, and bear at their free ends a striated cuticular edge (c), while from their inner or basal end arise the fibres destined to form the common optic nerve.
At a later stage (Fig. 542, B), the eye-pit is closed over, the edges growing over and covering the deeper part of the eye. In this 568way there arises out of the pit-like rudiment a two-layered optic cup. The outer or superficial layer (l) becomes in its central part the crystalline lens, while the peripheral parts form the iris. From the cuticular striated border of these cells arise the chitinous or corneal lens. On its outer edge the superficial layer of the eye passes gradually into the unmodified hypodermis (h).
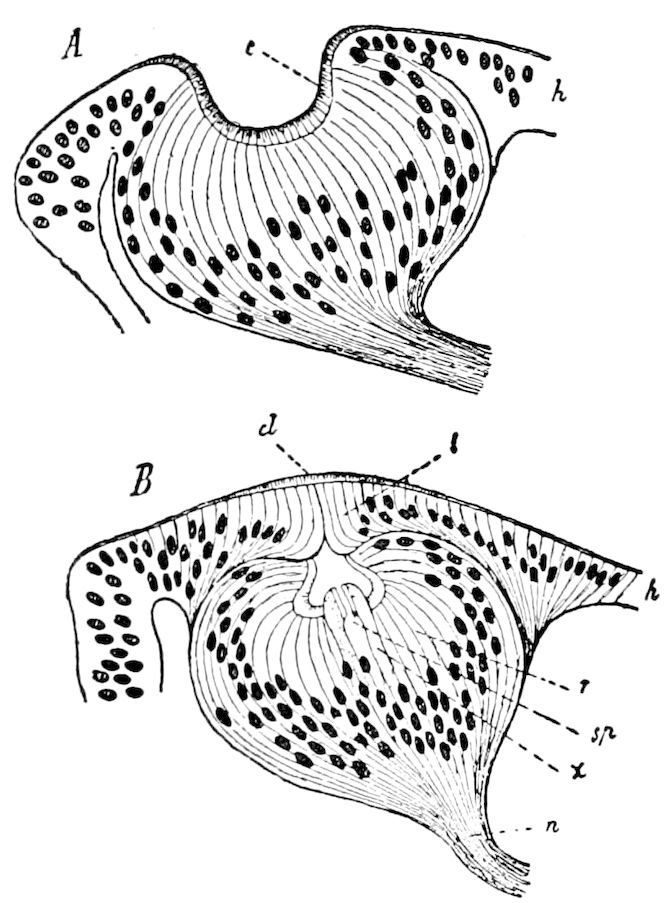
Fig. 542.—Two stages of development of the 5th of the six ocelli of larva of Acilius: c, cuticular striated band; cl, germ destined to form the corneal lens; h, hypodermis; l, crystalline-lens layer; n, optic nerve; r, retinal germ; sp, vertical fissure of the retina; x, the retina-cells bordering this fissure.
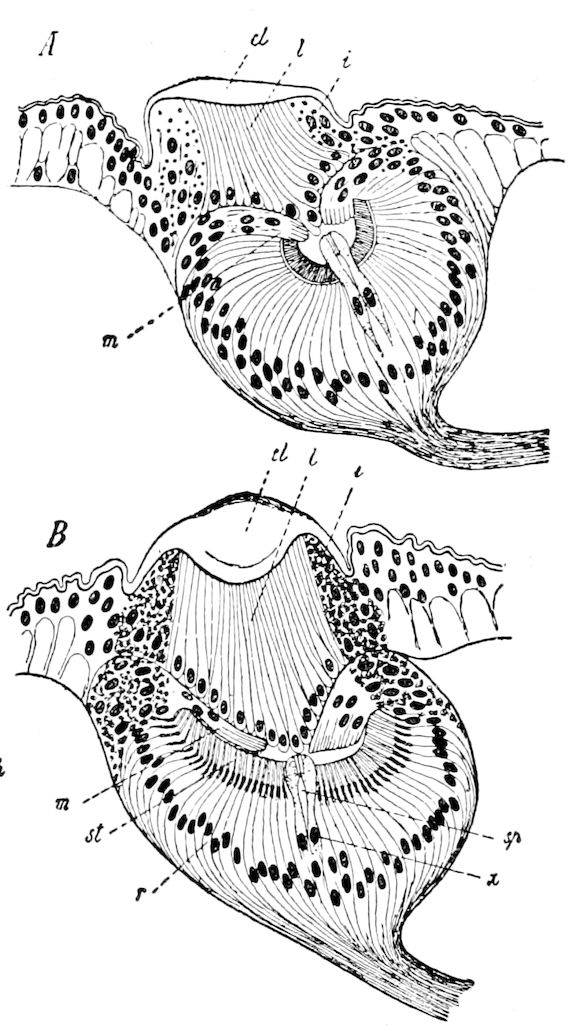
Fig. 543.—Two later stages of development of the same eye as in Fig. 542: i, iris; m, middle inverted layer of the eye; r, retina; sp, vertical fissure of the retina; st, rods; other letters as in Fig. 542.—This and Fig. 542 after Patten, from Korschelt and Heider.
The inner, deeper layer of the eye, which forms the contracted cup-shaped portion, appears to be the rudimentary retina (r). From its cuticular rod-like or fibrous edge arise the visual rods. There soon arise certain peculiarities characteristic of the eye of Acilius, i.e. the fissure (sp) bordered by the horizontally situated rods of the large retina-cells (x).
In the farther developed eye (Fig. 543) there is a flattening of the cup-shaped inner edge, by which the bottom of the eye is levelled and the little rods belonging to it stand up vertically (Fig. 543, B, st). Then the cells belonging to the edge of the retinal cup (m) 569are turned in, forming an inverted layer constituting the germs of a third layer interpolated between the two chief layers of the eye. (Korschelt and Heider, from Patten.) Patten concludes that the structure of the retina in the larval ocelli of insects is much like that of myriopods, and that the whole eye is constructed on the same plan as that of Peripatus and most molluscs.
Intestinal canal and glands.—The intestinal or digestive canal is primitively divided, as already stated on p. 299, into three sections, of which the anterior and posterior are called respectively the stomodæum and proctodæum, and are invaginations of the ectoderm, forming sacs whose blind ends face the future site of the mid-intestine. The fore-intestine (stomodæum) in most cases arises earlier than the proctodæum. Its muscles are derived from the mesoderm. From the stomodæum arises at an early date an unpaired dorsal invagination out of which develops the ganglion frontale and the pharyngeal nerve.
The absorption of the ends of the blind sacs of the fore and hind intestine, and opening up of the passage into the mid-intestine, occur rather early in embryonic life. In the wasps and bees, as well as the larva of the ant-lion, the mid-intestine remains closed at the end, not communicating with the proctodæum, which has an exclusively excretory function (Fig. 497).
The mid-intestine arises from two originally separate rudiments, i.e. the fore and hind endodermal rudiments, which at the outset stand in the most intimate relation with the invagination of the fore and hind intestine. Originating as a simple collection of cells, so closely adjoining these invaginations that Voeltzkow, Patten, and Graber derived them directly through outgrowths of them, they become extended by advancing cell-multiplication until they assume a U-shaped form. The legs of the U-shaped rudiment are in the anterior endodermal mass, directed backwards; those in the posterior mass, on the other hand, are directed anteriorly. These legs grow towards each other until they become fused together, forming two paired endodermal streaks, which pass under the primitive band along its whole length, and are fused with it at the fore and hind ends. In these places they stand in intimate union with the proctodeal and stomodeal invaginations.
The paired endodermal streaks belong to the lateral portions of the primitive band. As a rule, they lie directly under the row of cœlom-sacs (Fig. 539, F). The dorsal wall of the primitive segments stands consequently in intimate contact with the endodermal streaks. On this wall of the primitive segments an active cell-growth takes place, and the cell-material produced in this way, 570which separates from the dorsal wall of the primitive segments, forms the outer or splanchnic layer of the rudiment of the mid-intestine (spm, Figs. 539, F, 544, sp). What remains of the dorsal wall of the cœlom-sacs after this separation joins the genital rudiments and gives rise to the so-called terminal thread-plate (Fig. 544, ef). The endodermal streaks, with the splanchnic layer lying next to them, may now be considered as the rudiments of the mid-intestine (Fig. 530, m, etc.). These are noticeable in the following stages by their considerable lateral growth; they spread out over the upper surface of the yolk, around which they finally entirely grow (Figs. 539, C-F, 544, 545). This growth around the yolk goes on in most cases in such a way as to unite the two mid-intestinal streaks in the region of the ventral median line with each other. Then afterwards their union on the dorsal side takes place (Figs. 539, F, 545). The yolk thus passes completely into the interior of the mid-intestine, and with it the remains of the dorsal tube or dorsal organ, when such an one is present.
Fig. 544.—Cross-section through the abdominal region of a somewhat older primitive band of Phyllodromia germanica: bg, rudiment of the nerve-cord; c, remains of the cœlomic cavity; cz, rudiment of the genital efferent passage; ec, ectoderm; en, endoderm; ef, terminal cord-plate; fk, fat-body tissue; gz, genital cells; h, rudiment of the heart; p, rudiment of the pericardial cavity; ps, rudiment of the pericardial septum; so, somatic mesoderm layer; sp, splanchnic mesoderm layer.
The salivary glands.—These segmentally arranged glands, which open by pairs into the three gnathal segments of the head, arise as ectodermal invaginations originally opening not into the stomodæum, but outwards on the surface of the body; hence Korschelt and Heider 571suggest that they were originally dermal glands, whose mouths became drawn into the buccal cavity.
Fig. 545.—Cross-section through the abdominal region of an embryo of cockroach (P. germanica) after the yolk has been completely enclosed by the primitive band and the closure of the back; s, tracheal stigma; other letters as in Figs. 540, 544.—This and Fig. 544 after Heymons, from Korschelt and Heider.
Fig. 546.—Embryo of Doryphora shortly after the appearance of the appendages, unrolled and isolated: o, stomodæum; lb, labrum; b1–b3, three brain segments; og1–og3, three segments of the optic ganglion; op1–op3, three segments of the optic plate; f1–f5, five pairs of invaginations which form the tentorium, etc.; t7–t20, tracheal invaginations; the two last pairs (t19-t20) either disappear or form the openings of the sexual ducts; at, antennæ; md, mandibles; mx1–mx2, maxillæ: p1–p3, legs; c, commissure connecting the two ganglionic thickenings (g4) of the premandibular segment; gl, ganglia; mst, middlecord thickenings; mpg1–mpg3, rudiments of three pairs of urinary tubes; a, proctodæum.—After Wheeler.
For their serial arrangement, see p. 337. Korschelt and Heider state that they would be inclined to homologize the salivary glands of insects with those glands of myriopods opening into the mouth-cavity, were it not that these glands in myriopods opening into the mouth are in reality transformed nephridia originating from the mesoderm, while the salivary glands of insects are 572clearly ectodermal structures. We must, therefore, they add, leave to later researches the question of the homology of these organs, also of their relations to the similar glands of Peripatus.
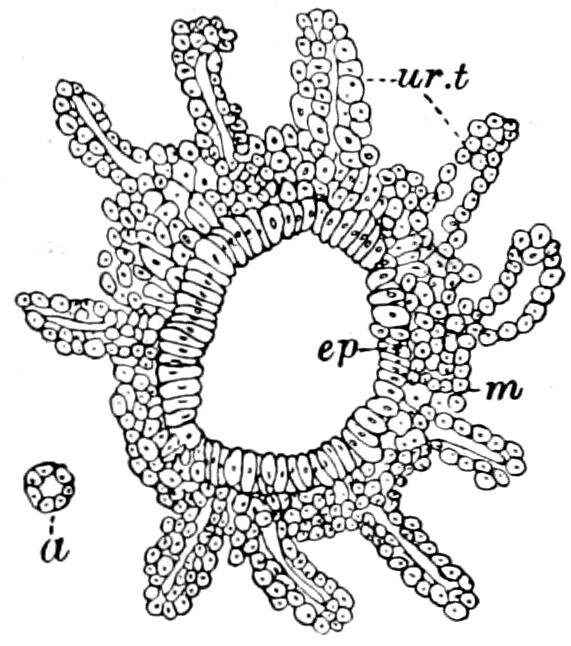
Fig. 547.—Section of proctodæum of embryo locust, showing origin of urinary tubes (ur.t): ep, epithelial or glandular layer; m, cells of outer or muscular layer; a, section of a tube.
The urinary tubes.—These excretory vessels arise as paired evaginations of the hind intestine or proctodæum. They are ectodermal structures arising as lateral diverticula of the intestinal cavity (Fig. 546). Figure 547 represents their mode of origin at the anterior end of the proctodæum of a locust. It will be seen that there are 10 primary tubes. There are 150 such tubes in locusts, or 10 groups of 15 each. The 15 secondary tubes probably arise from the primary ones in the manner described by Hatschek for Lepidoptera (see his Taf. III, Fig. 7).
While the Malpighian tubes usually first arise as diverticula of the proctodæum, in the Hymenoptera (Apis and Chalicodoma) they appear, even before the completion of the proctodæum, as invaginations of the ectoderm which at first open out on the outer surface of the primitive band. They seem, then, in some degree, to be similar to the tracheal rudiments, which perhaps is the reason why they have been homologized with them, a view which we do not share, and in which Carrière does not concur. They afterwards pass, with the growing proctodæum, into the interior of the embryo. (Korschelt and Heider.)
The heart.—The dorsal vessel is first indicated, according to Korotneff, by a long string or row of cells (cardioblasts), which on each side border the mesodermal layer of the primitive band (Figs. 544, h, 548, h). In the advancing growth of the primitive band around the yolk, this rudiment steadily passes up more towards the dorsal side. It is in connection with the wall of the primitive segment (Figs. 544 and 548), and represents the point at which the dorsal wall of the cœlom-sac passes into the lateral wall. According to Korotneff, the cardioblasts arise directly through a migration out from the wall of the primitive segment.
In Gryllotalpa the formation of the dorsal organ, which, as Korotneff states, is in this insect nothing else than a stopper which fills up the dorsal gap of the body-wall of the embryo, is effected by the rupture of the embryonal membranes. The serosa is drawn together to form a thick plate (Fig. 523, A, rp), and the much degenerated amnion-folds (am) which are laterally attached to it have moved from the edges of the primitive streak (*x-*y) far towards the dorsal side (see Fig. 539, C, which represents a similar 573stage). The distance between the rudiment of the amnion-fold and the lateral edge of the primitive band (*x, *y) is occupied by an epithelial lamella (l), in which we recognize the earlier amnion. This lamella does not lie directly on the yolk, but is separated from it by a spacious blood-lacuna (A, bs), in which can be seen numerous blood-corpuscles which have migrated in from the mesoderm of the primitive band. The cardioblasts which have arisen from the wall of the primitive segment (us) are on each side arranged into the form of a furrow (gr), which bounds the blood sinus below.
Fig. 548.—Cross-section through the abdominal part of an older primitive band of P. germanica when beginning to grow around the yolk: vm, ventral longitudinal muscle; other lettering as in Fig. 545.—After Heymons, from Korschelt and Heider.
By the continuous growth of the primitive band around the yolk, after the resulting invagination and degeneration of the dorsal plate, the two blood-lacunæ unite together on the dorsal side into a single one (B, bs). These constitute the first cavity of the heart. The vascular furrows (gr) come in contact with each other and grow together, and the wall of the heart is thus formed. Ayers states that in Œcanthus the heart is formed in the head region only after the yolk-sac has passed entirely within the body. The venous ostia 574arise by two paired invaginations of the lateral walls, forming a split at their bottom.
The rudiment of the heart stands, as we have seen, in intimate union with the primitive segments. Out of the lateral walls of these segments, after giving off the elements of the somatic mesoderm, arises an epithelial plate which becomes the rudiment of the pericardial septum or dorsal diaphragm (Figs. 523, A-C, dd, 544–545, ps). As soon as the two halves of the rudiments of the heart have united with each other in the dorsal middle line, the two halves of the pericardial septum unite with each other and form the wall to the pericardial cavity and shut it off from the rest of the body-cavity. For a long time the pericardial septum remains in union with the wall of the heart. Afterwards, however, it separates from it (Fig. 523, C, dd). (Korschelt and Heider.)
The statements of other authors (Ayers, Grassi, Patten, Tichomeroff, Carrière, Heider, Heymons, etc.) as to the mode of origin of the heart in insects of other orders are all similar to the type described in Gryllotalpa. The difference consists mostly in the fact that the two large blood-lacunæ are wanting or only exist to a slight extent. It results that the rudiment of the cavity of the heart in the earlier stages is of slight extent and often scarcely recognizable.
In Œcanthus (Ayers) and in Gryllotalpa, the hinder section of the heart is the first to develop, the development advancing from behind forward.
The blood-corpuscles.—Blood-cells are said by Korotneff to be, in Gryllotalpa, at an early period present almost everywhere between the yolk and mesoderm; they are derived, as he states, from the cells of the somatic mesoderm layer, which has lost its connection with the other parts of the mesoderm, and fall into the body-cavity. Ayers states that the blood-corpuscles arise from serosa nuclei which have passed into the body-cavity, where they become more vesicular, and ultimately all of the nuclear substance goes to form from one to three spherical bodies, which are surrounded by the common membrane.
“These bodies are blood-corpuscles and are free nucleoli immediately on the rupturing of the vesicle which surrounds them.” (Ayers, Pl. 22, Figs. 1, 3, p. 250.) More recently, Schaeffer has observed in caterpillars certain cell-complexes associated with the fat-body which he has called blood-forming masses.
Musculature, connective tissue, fat-body.—The muscles of various parts of the body, as well as the connective tissue, arise by histological differentiation from the somatic layer of the mesoderm (Fig. 523, so). The fat-body originates from the same source, as shown by the researches of Kowalevsky, Grassi, and of Carrière. In 575Hydrophilus a dorsal band of the fat-body passes over the digestive canal arising by direct transformation of the wall of the cœlom-sacs. But also the other portions of the fat-body, as the fat-body lobes accompanying the tracheal system, are of undoubted mesodermal origin. Heymons’ observations on the cockroach (Phyllodromia) agree with the foregoing view. In this insect at a very early period certain cells in the wall of the cœlom-sacs undergo a change, and may be recognized as the rudiments of what are afterwards fat-body tissues (Fig. 540, B and C, f).
The reproductive organs.—Our knowledge of the mode of development of the genital organs is in a less satisfactory state than that of the other organs. It is now known that the rudiments of the sexual glands belong to the mesoderm, and are developed from the wall of the cœlom-sacs. In the cockroach (Phyllodromia), the most generalized of the winged insects, as Heymons has shown, in the earlier stages of the embryo separate genital cells are already distinguished by their histologically different characters from the other mesodermal cells. The genital cells are larger and show a feebly stained nucleus with a clear nucleolus. These genital cells, which are transformed normal mesodermal cells, lie originally within the mesoderm layer or on the surface of this layer turned towards the yolk, on the edge of the segments. After the complete formation of the cœlom-sacs we find them (Fig. 549, gz) in the dissepiments which separate the successive cœlom-sacs from one another. Here new genital cells are constantly formed through the transformation of mesoderm cells. The development of the genital cells takes place in the 2d to the 7th abdominal segments.
Afterwards the genital cells pass into the interior of the cœlom-sacs, and soon pass to the dorsal wall of the same (Fig. 540, A, gz) and enter between the cells of this wall. The cœlom-sacs (c) show in cross-section in this stage a triangular outline, so that we can distinguish a dorsal, lateral, and median wall. The dorsal wall lies next to the surface of the yolk, and afterwards gives rise by separation or splitting to the splanchnic mesoderm (Fig. 544, sp), while from its remains the terminal thread-plate (ef) originates. The lateral wall, which is turned towards the ectoderm of the primitive band, is intimately concerned in the formation of the somatic layer (Fig. 540, C, so) of the mesoderm. Out of what remains arises the pericardial septum (Fig. 544, ps).
When the genital cells have entered into the dorsal wall of the primitive segments, they are already so numerous that they form a continuous series extending from before backward. The genital 576rudiment consists, then, of a string of cells lying on each side in the dorsal wall of the primitive segments, which extend from the 2d to the 7th abdominal segments. In the formation of these strings or rows of cells not only are the genital cells concerned, but also still undifferentiated mesoderm cells (Fig. 540, B, C), which originate from the dorsal wall of the cœlom-sacs and lie next to the genital cells. Some of these last tend to envelop the genital cells. We designate them the epithelial cells of the genital rudiments (ep), while others form a cellular cord which takes a position medial and ventral to the genital cells.
Fig. 549.—Sagittal (longitudinal) section through the abdominal part of a primitive band of cockroach (Phyllodromia germanica) after the end of the formation of the primitive segments: 1–7, 1st to 7th abdominal segments; from the 8th abdominal segment (8) to the last segment (es) extends the inturned ventral part of the primitive band; am, amnion; c, cœlom-sac; d, yolk; gz, genital cells, lying partly in the dissepiments, partly in the wall or in the cavity of the primitive segments.
From the genital cells in the female arise only the egg-cells (and the nutritive cells in those forms which have such). The follicular epithelium of the egg-tube, on the other hand, also the corresponding cells of the terminal chamber, originate from the epithelial cells. Phyllodromia and Orthoptera in general, to which this description applies, show in this respect tolerably simple relations, since the germinal or terminal compartment of the ovary in them is composed of relatively few cells. In most other insects, and especially those which have a great number of food-cells in the ovary, the germinal chamber (Keimfach) is extraordinarily large.
The ventral cellular cord (cz) develops into the proximal part of the oviduct, which widens out and receives the single egg-tubes.
The cœlom-sacs in the farther course of their development, 577through the retrograde development of the parts extending into the appendages, through the development of the fat-bodies and through the delamination of the somatic and the splanchnic mesoderm layer, become greatly diminished in size. Finally, there remains left of them only a rather small cavity (c), which is bordered on the side by the rudiment of the pericardial septum (ps) and within by the terminal thread-plate (ef). The dorsally situated point where these two lamellæ pass into each other seems to stand in intimate connection with the cells of the rudiment of the heart (h). The cord-like genital rudiment hangs from the terminal thread-plate as from a mesentery (Fig. 549, gz).
Fig. 550.—Longitudinal section through the female genital rudiments of P. germanica. A, with beginning, B, with farther advanced growth of the ovarian tubes: cz, rudiment of the genital efferent passage; ef, terminal threads; ep, nucleus of the epithelial cells; gz, genital cells.—After Heymons, from Korschelt and Heider.
Together with the growth of the primitive band around the yolk, and the formation of the back, the paired rudiments of the heart gradually extend to the neighborhood of the dorsal median line, followed by the genital rudiments which are connected with them by the terminal thread-plates. The genital rudiments advance thus to the dorsal side of the developing mid-intestine (Fig. 545, gz).
The terminal thread-plate (ef) is at first a simple epithelial plate. Soon, however, follows an arrangement of its cells whereby they appear to be arranged in vertical rows, each one of which corresponds to a developing ovarian tube. In this way the terminal thread-plate separates into the separate terminal threads of the ovarian tubes (Fig. 550, ef). In this process of division the uppermost dorsal edge of the terminal thread-plate takes no part. From it afterwards grows a thread which extends anteriorly, which becomes the common terminal thread of all the ovarian tubes, the so-called Müller’s thread. This is originally united with the pericardial 578septum, but seems in later stages to have no longer an intimate connection with it.
The formation of the single ovarian tubes, which in Phyllodromia number about 20, is accomplished by the extension of indentations from the dorsal side towards the ventral side of the ovarian rudiment (Fig. 550). At the same time the epithelial cells (ep), which were originally situated in part between the genital cells, become arranged in the form of an epithelium on the surface of the ovarian tubes, which soon forms on its outer surface a structureless cuticular tunica propria. The outer peritoneal membrane of the ovary becomes formed of the cells of the surrounding tissue of the fat-body.
The genital rudiment originally extends, as already stated, from the 2d to the 7th abdominal segment. In the last, however, the genital cells at first occur only sparingly, and afterwards completely disappear, so that here the genital cord appears composed of epithelial cells only. This part is the rudiment of the oviduct proper, and forms a direct continuation of the above-mentioned cell-cord which is situated ventralward from the genital cells, from which, as we have seen, the proximal cup-shaped section of the oviduct is formed. The hinder section of the oviduct turns down ventrally in order to unite at the boundary between the 7th and 8th abdominal segments with the hypodermis. The rudiment of the oviduct originally forms a solid strand of cells. Afterwards a cavity is formed by the separation of the cells.
In later stages there is a considerable shortening of the genital rudiment, so that it occupies a smaller number of abdominal segments than at first. At the same time the single ovarian tubes pass out of their originally vertical position into one more horizontal.
The paired connections of the rudiments of the oviducts with the hypodermis of the intersegmental furrow between the 7th and 8th abdominal segments reminds us of the conditions in the Ephemeridæ. This is the primitive condition in insects. In the female of Phyllodromia there is developed during larval life, from an ectodermal invagination, an unpaired terminal section of the genital passage, which becomes the genital pouch in which the egg-case (oötheca) is held. This genital pouch is formed, as Haase has already proved, by the withdrawal of the chitinous ventral plate of the 8th and 9th abdominal segment by invagination into the interior of the body.
The development of the efferent passages has been investigated by Nusbaum in the cockroach (Periplaneta) and in the Pediculina. He found that only the vasa deferentia and the oviducts arise from the hinder cord of the germs of the sexual glands, that is, out of 579the mesodermal rudiments, while the other parts of the sexual efferent apparatus (uterus, vagina, receptaculum seminis, ejaculatory duct, penis, and all the accessory glands) develop from the integumental epithelium and are of ectodermal origin. In fact, the unpaired parts (uterus, penis, receptaculum seminis, unpaired glands) have developed from paired rudiments, being outgrowths of the hypodermis. The hinder portions of the rudiments of the sexual glands approach these hypodermal growths and fuse with them. Through a median fusion of the paired hypodermal growths arise the germs of the unpaired organs. These observations are in complete agreement with the results at which Palmén arrived by anatomical investigation (see p. 492).
From the agreement of the position of the sexual openings in Phyllodromia with the conditions observed in the Ephemeridæ, with which the Perlidæ also agree, we conclude that in the entire group of insects an opening between the 7th and 8th abdominal segments is the primitive condition, and that only by a secondary shifting has a more posterior position of the opening (in many forms) been brought about. In this category we must certainly include the Thysanura, in which the sexual opening is single and situated between the 8th and 9th abdominal segments.
Development of the male germinal glands.—These rudiments arise in exactly the same manner as those of the female. Sexual differentiation takes place in the later embryonic stages. We then notice that in the male four masses of genital cells become surrounded by epithelial cells. These masses, which form the germs of the four testicular follicles of Phyllodromia, stand in intimate union with the rudiment of the vas deferens, and in the later stages move in connection with the latter, away from and behind the original genital rudiment. There remains, then, with the terminal thread-plate a remnant of the genital rudiment, which, according to Heymons, forms the female part of the original hermaphroditic genital rudiment, and in special cases may develop even into rudimentary egg-tubes and eggs. The rudimentary organ arising out of this genital rudiment may also be demonstrated in the adult male of Phyllodromia.
In the female the oviduct arises directly out of the originally established efferent passage. In the male, on the contrary, it is not, along its whole length, transformed into the vas deferens, but its distal terminal portion degenerates and is replaced by a newly formed terminal portion of the vas deferens, which then unites with the ectodermal ductus ejaculatorius. (Korschelt and Heider.)
580On reviewing the facts as to the origin of the sexual organs, as in Phyllodromia,[84] as just described, it will be seen that they afford proof that in the derivation of the genital cells from the epithelial cells of the cœlom-sacs, there is a direct agreement with the annelids. In the later development of the paired genital glands, and of an efferent passage standing in direct union with the glands themselves, there is a certain agreement with the conditions in Peripatus. In the first place, the dorsal position of the genital glands is the same in the two groups. On the other hand, the genital glands of Peripatus, according to Sedgwick, are formed by direct fusion of the successive cœlom-sacs (and a similar point of view has been taken by Heathcote for the myriopods), hence it results that in Peripatus the genital cavities arise out of the cœlom-cavities. In the insects, on the other hand, the genital rudiment lies, to be sure, in the wall of the cœlom-sac, but the genital cavity (lumen of the oviducts) in them arises separately from the cœlom-sacs, while the cœlom-cavities finally become a small part of the definite body-cavity. We must consider the conditions in Peripatus and the myriopods as the more primitive, directly pointing to the annelids; on the other hand, those of the insects as derived and secondary.
If we attempt to homologize the sexual efferent passages of insects with those of Peripatus, we are compelled to refer them to a modified pair of nephridia, and the origin of the latter (Peripatus) from the mesoderm agrees with that of insects. In general, however, in the development of the sexual outlets of insects, there are no characters which can be regarded as favorable to such a view. We must here accept the fact that the mode of development is secondary.
Mention should be specially made of the fact we owe to Heymons, that in the genital rudiment of Phyllodromia the genital cells and epithelial cells can be distinguished from each other from the very beginning. This fact does not favor the generally accepted view that the follicle-cells and egg-cells arise through a later differentiation from one and the same kind of cell. From their first origin, indeed, in Phyllodromia, both kinds of cells may be referred to the same source.
The mode of origin of the genital rudiments in Diptera and Aphides deserve special mention. In these groups the sexual germs are present in very early stages of life. This certainly in part is the result of the parthenogenetic and pædogenetic mode of reproduction in the two groups, which leads to an early differentiation of the sexual germs.
In the Diptera the first germs of the genital glands are represented by the polar cells (Fig. 551, pz). In the asexual developing eggs of the oviparous Cecidomyia larva, before the formation of the blastoderm, there separates from the hinder pole (D) a rather large cell rich in granules, which soon divides into two and afterwards four polar cells. After the completion of the blastoderm these polar cells then pass in among the blastoderm cells (G) and into the interior of the embryo, where they are in later stages symmetrically arranged in two groups, and, enveloped by the cells of surrounding tissues, transformed into the genital rudiments. (Metschnikoff.)
In Chironomus (Fig. 552, p), according to Balbiani, two polar cells almost simultaneously separate from the hinder pole of the egg, which, by division, form a group of four and eight cells. Exactly as in the case in Cecidomyia, these cells are taken within the embryo, where they lie divided into two groups on each side of the proctodæum. In all the young, freshly hatched larvæ; these 581two spindle-shaped groups, whose cells soon increase in number, may be seen situated dorsally on the side of the heart, enveloped by a clear cellular membrane which ends before and behind in a ligament-like terminal thread. The anterior terminal thread is the rudiment of the so-called Müller’s thread. The thread at the posterior end is the rudiment of the paired efferent passage of the genital glands. Through a division of the cells lying in the interior of the rudiments of the ovaries, there results the formation of a rosette-shaped group of cells which corresponds to the contents of an ovarian tube. With this view of Balbiani the later observations of Ritter agree.
As in the Diptera, so in the Aphides, the first germs of the genital organs are differentiated very early in life. In the early stage in which through an invagination from the hinder pole of the egg the first rudiment of the amnion-cavity is formed, a group of cells becomes separated from the wall of this invagination before the formation of the lower layer, which at this time lies as an unpaired roundish mass within the embryo. This group of cells, according to Balbiani and Witlaczil, has arisen by division of a single cell. Afterwards it becomes horseshoe-shaped and divides into a number of roundish masses of cells, which are arranged in similar numbers on each side of the median plane of the body, and form the rudiments of the terminal fan (Endfächer). They are covered by an epithelial envelope which passes anteriorly into the terminal threads, posteriorly into the efferent passage. The origin of this epithelial case is unknown. The efferent passages of the separate ovarian tubes are united into a common oviduct, and this fuses with an unpaired ectodermal invagination lying under the hind intestine from which the accessory sexual organs are formed. (Korschelt and Heider from Metschnikoff, Witlaczil, Will.)
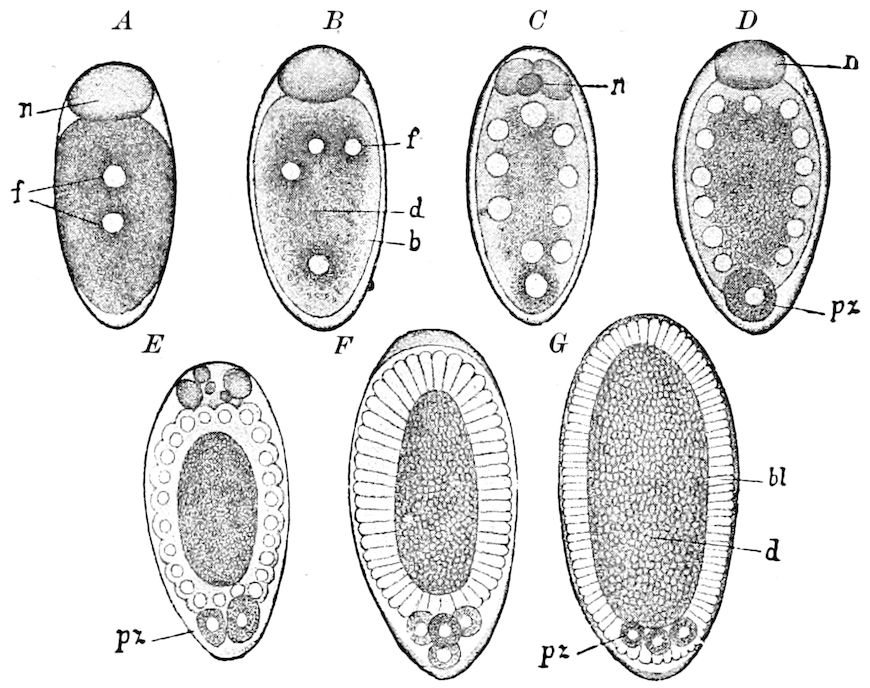
Fig. 551.—First developmental stages of the parthenogenetic eggs of the larva of Cecidomyia: b, peripheral protoplasmic layer (Keimhautblastem); bl, blastoderm; d, central yolk; f, division-nuclei; n, nutritive cell (“corpus luteum”) about to break up; pz, polar cells.—After Metschnikoff, from Korschelt and Heider.
In the Hymenoptera Ganin has observed in the embryo of Platygaster the rudiments of the sexual glands in the form of two rounded masses situated near 582the posterior intestine and apparently derived from the same blastems or buds as the latter.
Uljanin studied these organs in the larva of the honey-bee. They are two reniform bodies in the middle of which will soon appear the ovarian tubes. They also give birth to the internal parts of the excretory ducts, while the external part of the genital tube, as also the accessory glands which are connected with it, are derived by an invagination of the hypodermis at the surface of the penultimate segment.
Dohrn observed in the larva of ants the rudiments of the ovaries in the form of two pyriform masses, each with eight prolongations which he regarded as young ovarian tubes.
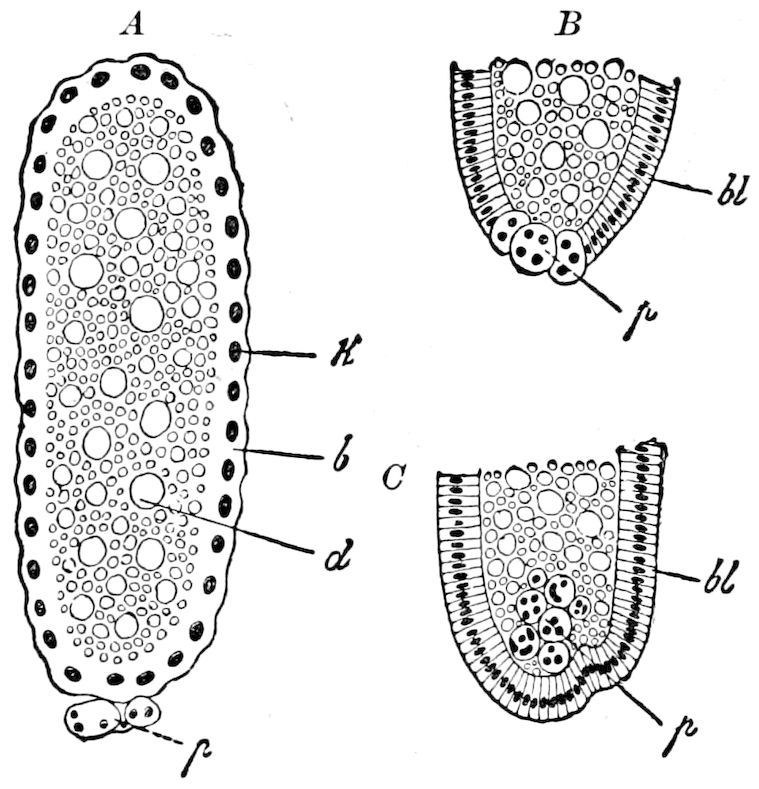
Fig. 552.—Three longitudinal sections through the embryo of Chironomus. In A, the blastoderm (bl) is beginning to form, the polar cells (p) outside of it; in B, the polar cells have pressed in between the blastoderm cells; in C, they lie in the interior of the embryo: b, protoplasmic layer (Keimhautblast); d, yolk; k, nucleus of the forming blastoderm.—After Ritter, from Korschelt and Heider.
In Encyrtus Bugnion observed the rudiments of the sexual glands in the middle of the larval period; they were rounded and with no apparent connection with the neighboring organs. Afterwards these rudiments elongated, approached nearer to the ventral surface, and placed themselves in relation with some small cell-groups which appeared under the rectum, and seemed destined to form the efferent canal (vas deferens) and accessory glands of the genital organs. He thought the sex could be recognized in the second half of larval life, the male gland being distinguished by its rounded shape and smaller size; the ovary by its oval form and larger size. In larvæ ready to be transformed the testis formed a cellular mass enveloped by a cuticle, and at its hinder end prolonged into an epithelial cord, which is undoubtedly the vas deferens. The ovary had a similar envelope, and from its cellular mass arose epithelial cords which were destined to become the ovarian tubes.
m. Length of embryonic life
The duration of embryonic life varies greatly in different insects. The embryo of the blow-fly is fully developed in less than 24 hours, that of the house-fly in 24 hours. In the locusts and tree-cricket the embryos begin to develop at the end of the summer, continuing to grow until the cool weather of autumn, when growth is arrested, the later stages being finished in the latter part of the spring. It is so, likewise, with the embryos of many moths and other insects.
n. The process of hatching
This has been observed only in a few cases, and careful observations as to the exact manner in which the embryo breaks the egg-shell and frees itself from the amnion are much needed. Also the rapid changes of form from that of the embryo within the egg-shell, and that which it immediately assumes after breaking forth from the shell and membranes, have yet to be observed; for these will undoubtedly be found to have special phylogenetic significance. Indeed, the phylogenetic importance of the latest embryonic changes in insects just entering on the nymph or the larval stages is very great, though little attention has as yet been bestowed upon the matter.
As regards the changes at the time of hatching, Wheeler tells us that the cockroach (Phyllodromia), shortly after leaving its narrow place in the egg-capsule, undergoes a peculiar change in shape. Before hatching, and when confined in the egg-shell, the body is about one-third as wide as thick; but soon after breaking out of the chorion its body is much flattened, its dorso-ventral diameter being only about a third as great as its greatest breadth. This shows that the flattened shape of the body of cockroaches, which adapts them for their life under bark and stones, is a very late inheritance, and that these insects have descended from those with more cylindrical bodies. The end of the body, also, which in the egg is bent underneath the abdomen, is, after hatching, bent dorsally, as indicated by the anal stylets, which now point directly upwards and outwards. The spines and claws are developed shortly before hatching. In the Locustidæ (Xiphidium, etc.) Wheeler has observed that the pleuropodia, or 1st pair of abdominal temporary embryonic appendages, are shed during hatching. All the other embryonic appendages have also disappeared, except those which persist and have rapidly become modified to form the cercopods, or the ovipositor.
In locusts, as we have observed[85] in the case of Melanoplus spretus, the egg-shell bursts open at the head end, when the nymph, immediately after extricating itself from the egg, casts off a thin pellicle (the amnion), as we have also noticed in the case of the larvæ of the flea, currant saw-fly, and other insects. Before the amnion is cast off, the young nymph is almost motionless, but by slight movements of the body draws itself, in about five minutes, out of the amnion. The exact process of extraction is as follows: While it lies motionless, 584it puffs out the thin, loose skin connecting the back of the head with the front edge of the prothorax. The distention of this part probably ruptures the skin, which slips over the head, the body meanwhile curved over until the skin is drawn back from the head; when the latter is thrown back, it withdraws its antennæ and legs, and the skin is in a second of time pushed back to near the end of the abdomen; finally, it draws its hind tarsi out of the skin, and in a moment or two more the young locust frees itself, kicks away the cast skin, which resembles a little white crumpled pellet, and which has also been compared to a diminutive mushroom, and walks actively off,—sometimes, however, with the cast skin adhering to the end of the abdomen. Before the shedding of the amnion the body and legs are soft and flabby; immediately after, it walks firmly on its legs. All the eggs hatched—at least one or more hundreds—at about the same time, i.e. before 11 A.M.
Fig. 553.—Locust just before the amnion is cast, enlarged.—Emerton del.
The nymph of Stagmomantis carolina also sheds an amnion-skin, like that of the locust; but the embryo before casting it off is much elongated, and probably, like the European Mantis religiosa, the curious elongated embryos have the same singular habit of suspending themselves by threads, as shown in Fig. 554.
The account by Pagenstecher of the first ecdysis of the European Mantis was so extraordinary that we asked Professor Cockerell to collect the eggs of our Stagmomantis in New Mexico and send them to us. This he has kindly done, writing that he can “hardly recognize a true moult, since all that is cast off is the egg-membrane. In short, Pagenstecher’s account must be not a little fanciful, unless our insect differs very much in its development from Mantis religiosa. The main change is that after leaving the egg the thorax enormously elongates, producing a bulging out, and thrusting the head forward.” Our observations on the alcoholic specimens fully corroborate Cockerell’s conclusions. Pagenstecher’s figure of the embryo appears to be inaccurate. Sharp states that the hatching nymphs remain suspended for some days until the “first change of skin is effected.” This so-called “skin” is evidently the amnion.
The 17–year Cicada, after hatching, is enveloped by the amnion, from which it soon extricates itself, and then drops deliberately to the ground, “its specific gravity being so insignificant that it falls through the air as gently and as softly as does a feather.” (Riley.)
Other insects, as caterpillars, have room enough to turn around within their shell and to eat their way through the walls of the chorion.
585The meat-fly, as we have observed, hatches in the following manner. The embryo moves to and fro, the body twisting until the exochorion is ruptured; the egg-shell splits longitudinally, and in one or two seconds the larva pushes its way out through the anterior end, and in a second or two more extricates itself from the shell. The latter scarcely changes its form, and the larva slips out, leaving the amnion within.
Fig. 554.—Egg-case of Mantis with young escaping: A, the case with young in their position of suspension. B, cerci magnified, showing the suspensory threads.—After Brongniart, from Sharp.
In the case of a fossorial wasp, Specius speciosus, which carries Cicadæ into its burrow, laying an elongated egg on the body under the median thigh of its victim, the larva on hatching, Riley states, “does not emerge from the skin of the egg, but merely protrudes its head and begins at once to draw nourishment from between the sternal sutures of the Cicada.”
The hatching spines.—Animals belonging to quite distinct classes are provided late in embryonic life with hard knobs or spines, which are temporary structures for the purpose of breaking or cutting open the egg-shell, when it is too thick and solid to be ruptured by the movements of the embryo. The embryos of certain lizards, turtles, the blind worm and some snakes, of the crocodile, and even birds, as well as the duckbill and Echidna, are provided with them, always occurring, so far as we are aware, on the end of the upper jaw. In the Arthropoda similar structures have thus far only been met with in myriopods and insects, though an analogous structure on the cephalothorax of the embryo of phalangids has been observed by Balbiani. Metschnikoff describes and figures a low conical spine serving this purpose situated on the embryonal cuticle over the head of the advanced embryo of Strongylosoma, and one on the 3d pair of mouth-parts of Geophilus.
In the winged insects, the embryo of Forficula is said by Heymons to bear a single spine between the eyes, which serves as an egg-tooth. The embryo of the Hemerobiidæ, according to Hagen, “opens the egg with an egg-burster like a saw.” (Proc. Bost. Soc. Nat. Hist., xv, p. 247.) Riley states that the egg-burster, or ruptor ovi, as he calls it, of Corydalus cornutus, has “the form of the common immature mushroom,” and he adds that it is a part of the amnion, being “easily perceived on the end of the vacated shell.” 586Wheeler has observed three pairs of broad-based chitinous “hatching spines” used by Doryphora in rupturing its embryonic envelopes, and which are secreted by pyramidal thickenings of the hypodermis (Figs. 555, 556).
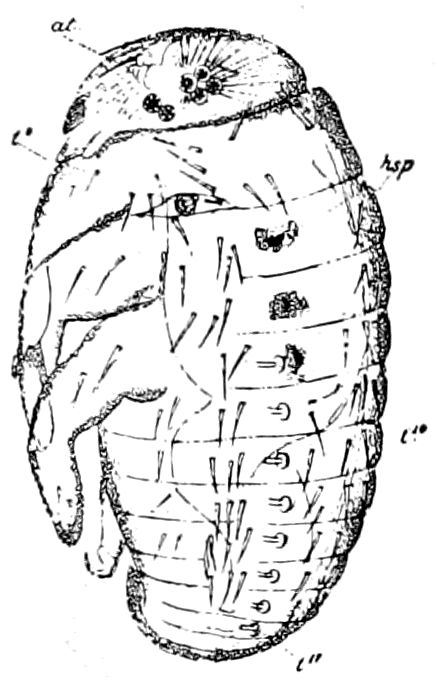
Fig. 555.—The three pairs of hatching spines (hsp) on the late embryo of Doryphora.—After Wheeler.
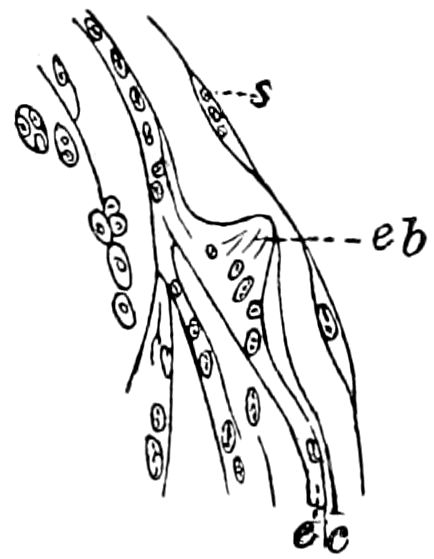
Fig. 556.—Rudiment of the hatching spine: eb, being a thickening of the ectoderm (ec) in embryo Doryphora after formation of the heart; s, serosa.—After Wheeler.
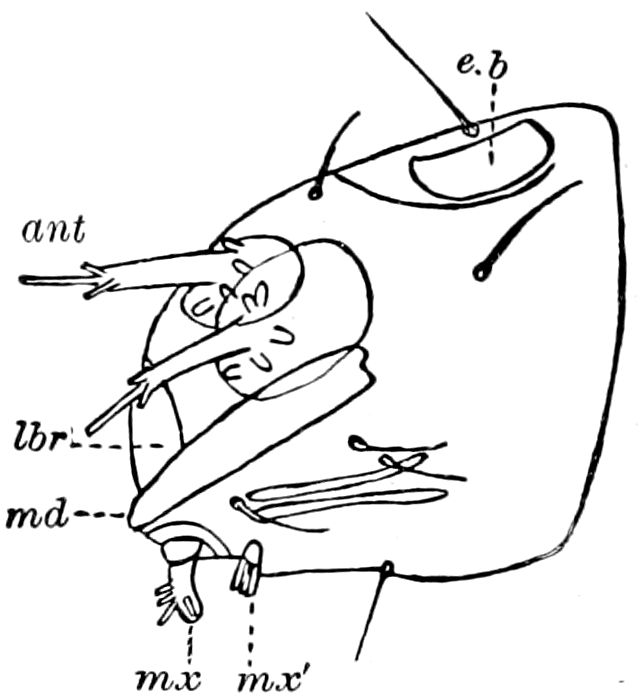
Fig. 557.—Head of freshly hatched larva of Pulex canis: eb, hatching spine; ant, antennæ; md, mandible; mx, maxilla; mx′, 2d maxilla; lbr, labrum.
The hatching spine of Pulex canis (Fig. 557) is a thin vertical plate, like the edge of a knife, situated in the median line of the head very near the posterior end, and is somewhat cultriform, the upper edge slightly hollow, and turned up a little at the anterior end. Though we did not see it working, it is situated at just the point on the head where it would come in contact with the egg-shell, and it was evident that the larva, by moving its head back and forth, would produce a slight split in the chorion and cause it to burst asunder. Later on in larval life it disappears, probably at the first moult.
LITERATURE ON EMBRYOLOGY
Koelliker, Albert. Observationes de prima insectorum genesi, etc. Turici, 1842, pp. 29, 3 Pls.
Rathke, H. Zur Entwickelungsgeschichte der Maulwurfsgrille (Gryllotalpa vulgaris). (Müller’s Archiv, 1844, ii, p. 27, Figs. 1–5.)
Zaddach, G. Untersuchungen über die Entwicklung und den Bau der Gliederthiere. I. Die Entwicklung des Phryganideneies. Berlin, 1854, pp. 138, 5 Taf.
—— Ueber die Entwicklung der Insekten. (Schrift, d. k. phys.-oekon. Gesell. Königsberg, viii Jahrg., 1867, Sitzb., p. 16.)
Leuckart, Rudolph. Die Fortpflanzung und Entwicklung der Pupiparen. Nach Beobachtungen an Melophagus ovinus. (Abhandl. Naturf. Gesell. Halle, iv, pp. 1–82, 3 Taf.) Halle, 1858.
Huxley, T. H. On the organic reproduction and morphology of Aphis. Pt. I, 1858; Pt. II, 1858. (Trans. Linn. Soc., xxii, pp. 193–219, 221–236, 5 Pls.)
587Weismann, A. Die Entwicklung der Dipteren im Ei, nach Beobachtungen an Chironomus sp., Musca vomitoria und Pulex canis. (Zeitschr. f. wiss. Zool., xiii, 1863, pp. i-xvi, 1–263, 14 Taf.)
—— Zur Embryologie der Insecten. (Arch. f. Anat. u. Physiol., 1864.)
—— Beiträge zur Kenntnis der ersten Entwicklungsvorgänge im Insectenei. In: Beiträge zur Anatomie und Embryologie, etc. (Festschrift für J. Henle. Bonn, 1882.)
Kupffer, C. Ueber das Faltenblatt an den Embryonen der Gattung Chironomus. (Arch. Micr. Anat., ii, 1866.)
Metschnikoff, E. Embryologische Studien an Insecten. (Zeitschr. f. wiss. Zool., xvi, 1866, pp. 389–500, 10 Taf.)
Kupffer, Carl. De embryogenesi apud Chironomos Observationes, etc. Kiliæ, 1867, pp. 16, 1 Pl.
Packard, A. S. On the development of a dragon-fly (Diplax) [Æschna?]. (Proc. Bost. Soc. Nat. Hist., xi, 1868, pp. 366–372, 8 Figs.)
—— Embryology of Isotoma, a genus of Poduridæ. (Proc. Bost. Soc. Nat. Hist., xiv, 1870, pp. 13–15, 4 Figs.)
—— Embryological Studies on Diplax [Æschna?], Perithemis, and the thysanurous genus Isotoma. (Mem. Peabody Academy of Science, Salem, i, 1871, pp. 1–21, 3 Pls.)
—— Embryological studies on hexapodous insects. (Ibid., 1872, pp. 1–17, 3 Pls.)
—— The embryological development of the locust. (Chap. X, Third Report U. S. Ent. Commission, Washington, 1883, pp. 263–282, 7 Pls.)
Brandt, A. Beiträge zur Entwicklungsgeschichte der Libelluliden und Hemipteren. (Mém. Acad. St. Pétersbourg (7), xiii, 1869, pp. 1–33, 3 Taf.)
—— Ueber das Ei und seine Bildungsstätte. (Leipzig, 1878, pp. 196, 4 Taf.)
—— Commentare zur Keimbläschentheorie des Eies. I. Die Blastodermelemente und Dotterballen der Insecten. (Arch. f. Micr. Anat., 1880, xvii.)
Melnikow, N. Beiträge zur Embryonalentwicklung der Insecten. (Arch. f. Naturg., xxxv, 1869, pp. 137–189, 4 Taf.)
Bütschli, O. Zur Entwicklungsgeschichte der Biene. (Zeitschr. f. wiss. Zool., xx, 1870, pp. 519–564, 4 Taf.)
—— Bemerkungen über die Entwicklungsgeschichte von Musca. (Morph. Jahrb., xiv, 1888, pp. 170–174, 3 Figs.)
Kowalevsky, A. Embryologische Studien an Würmern und Arthropoden. (Mém. Acad. St. Pétersbourg (7), xvi, 1871, pp. 1–70, 12 Taf.)
—— Zur embryonalen Entwicklung der Musciden. (Biol. Centralbl., vi, 1886, pp. 49–54.)
Müller, Fritz. Beiträge zur Kenntniss der Termiten. (Jena. Zeitschr. Wissens., ix, 1875, pp. 241–263, 4 Taf.)
Oulganine, W. N. (also spelled Uljanin). Sur le développement des Podurelles. (Arch. Zool. Expér., iv, 1875, pp. xxxix-xl, und v, 1876, pp. xvii-xix.)
—— Beobachtungen über die Entwicklung der Poduren. (Russian.) (Nachr. k. Gesellsch. Freunde Naturw., Anthrop. und Ethnogr., xvi, 1875.)
Dohrn, A. Notizen zur Kenntniss der Insectenentwicklung. (Zeitschr. f. wiss. Zool., xxvi, 1876, pp. 112–138.)
Hatschek, B. Beiträge zur Entwicklungsgeschichte der Lepidopteren. (Jena. Zeitschr. f. Naturw., xi, 1877, pp. 38, 3 Taf., 2 Figs.)
Bobretzky, N. Ueber die Bildung des Blastoderms und der Keimblätter bei Insecten. (Zeitschr. f. wiss. Zool., xxxi, 1878, pp. 195–215, 1 Taf.)
Graber, Vitus. Vorläufige Ergebnisse einer grösseren Arbeit über vergl. Embryologie der Insecten. (Arch. f. Micr. Anat., xv, 1878, pp. 630–640, 1 Fig.)
588—— Ueber die Polypodie bei Insectenembryonen. (Morph. Jahrb., xiii, 1888, pp. 586–615, 2 Taf.)
—— Ueber die primäre Segmentirung des Keimstreifs der Insecten. (Morph. Jahrb., xiv, 1888, pp. 345–368, 2 Taf., 4 Figs.)
—— Vergleichende Studien über die Keimhüllen und die Rückenbildung der Insecten. (Denkschr. Acad. Wiss. Wien., lv, 1888.)
—— Vergleichende Studien über die Embryologie der Insecten und insbes. der Musciden. (Denkschr. Acad. Wiss. Wien., lvi, 1889.)
—— Ueber den Bau und die phylogenetische Bedeutung der embryonalen Bauchanhänge der Insekten. (Biol. Centralbl., ix, 1889, pp. 355–363.)
—— Vergleichende Studien am Keimstreif der Insecten. (Denkschr. Acad. Wiss. Wien., lvii, 1890.)
—— Üeber die embryonale Anlage des Blut- und Fettgewebes der Insecten. (Biol. Centralbl., xi, 1891, pp. 212–224.)
—— Zur Embryologie der Insekten. (Zool. Anzeiger, xiv, 1891, pp. 286–291.)
—— Über die morphologische Bedeutung der ventralen Abdominalanhänge der Insekten-Embryonen. (Morph. Jahrb., xvii, 1892, pp. 467–482.)
Barrois, J. Développement des Podurelles. (Assoc. Franc. p. l’Avance. des Sc., 7e Sess., 1879.)
Kadyi, H. Beitrag zur Kenntnis der Vorgänge beim Eierlegen der Blatta orientalis. (Zool. Anzeiger, 1879, ii, pp. 632–636.)
Tichomiroff, A. Ueber die Entwicklungsgeschichte des Seidenwurms. (Zool. Anzeiger, ii Jahrg., 1879, pp. 64–67.)
—— Zur Entwicklungsgeschichte des Seidenspinners (Bombyx mori) im Ei. (Arb. Laborat. Zool. Mus. Moskau, i, 1882, pp. vii, v, 1–80, 3 Taf., 48 Figs.) (Russian.)
—— Ueber die Entwicklung der Calandra granaria. (Biol. Centralbl., x, 1890, p. 424.)
Balfour, Francis M. A treatise on comparative embryology. i, ii. London, 1880. 2d edit., London, 1885.
Hertwig, O. und R. Die Coelomtheorie. Versuch einer Erklärung des mittleren Keimblattes. Jena, 1881. (Jena. Zeitschr., xv, pp. 150, 3 Taf.)
Selvatico, D. S. Sullo sviluppo embrionale dei Bombicini. (Boll. Bachicoltura, viii, 1881.)
Lemoine, V. Recherches sur le développement des Podurelles. (Assoc. Franç. pour l’Avanc. d. Sc. Congrès de la Rochelle, 1882.)
Balbiani, E. G. Sur la signification des cellules polaires des Insectes. (Compt. rend. Ac. Sc. Paris, xcv, 1882.)
—— Contribution à l’étude de la formation des organes sexuels chez les Insectes. (Recueil Zool. Suisse, ii, 1885.)
Korotneff, A. Entwicklung des Herzens bei Gryllotalpa. (Zool. Anzeiger, vi Jahrg., 1883, pp. 687–690, 2 Figs.)
—— Die Embryologie der Gryllotalpa. (Zeitschr. f. wiss. Zool., xli, 1885, pp. 570–604, 3 Taf.)
Nusbaum, J. Vorl. Mittheilung über die Chorda der Arthropoden. (Zool. Anzeiger, vi Jahrg., 1883, pp. 291–295, 3 Figs.)
—— Die Entwicklung der Keimblätter bei Meloë proscarabæus. (Biol. Centralbl., viii, 1888, pp. 449–452, 2 Figs.)
—— Zur Frage der Segmentirung des Keimstreifs und der Bauchanhänge der Insectenembryonen. (Biol. Centralbl., ix, 1889, pp. 516–522, 1 Fig.)
—— Zur Frage der Rückenbildung bei den Insectenembryonen. (Biol. Centralbl., x, 1890, pp. 110–114.)
589—— Ueber die Entwicklungsgeschichte der Ausführungsgänge der Sexualdrüsen bei den Insecten. (In Polish, with German résumé, pp. 39–42.) (“Kosmos” Lemberg, 1884, ix Jahrg.)
—— Zur Embryologie des Meloë proscarabæus, Marscham. (In Polish, with Latin explanation of plates.) (“Kosmos” Lemberg, 1891.)
Schneider, A. Ueber die Entwicklung der Geschlechtsorgane der Insecten. (Zool. Beiträge, herausg. v. A. Schneider, i, 1883.)
Will, L. Zur Bildung des Eies und des Blastoderms bei den viviparen Aphiden. (Arb. Zool. Zoot. Inst. Würzburg, vi, 1883, pp. 217–258, 1 Taf.)
—— Entwicklungsgeschichte der viviparen Aphiden. (Spengel’s Zool. Jahrbücher. Abth. f. Anat. und Ont., iii, 1888, pp. 201–286, 5 Taf.)
Patten, W. The development of Phryganids, with a preliminary note on the development of Blatta germanica. (Quart. Journ. Micr. Sc., xxiv, 1884, pp. 54, 3 Pls.)
—— Studies on the eyes of Arthropods. I. Development of the eyes of Vespa, with observations on the ocelli of some insects. (Journ. of Morphol., Boston, i, pp. 193–226, 1 Pl.)
—— Studies on the eyes of Arthropods. II. Eyes of Acilius. (Journ. of Morphol., Boston, ii, 1888, pp. 97–190, 7 Pl., 4 Figs.)
—— Eyes of molluscs and Arthropods. (Mitth. Zool. Station Neapel., vi, 1888, pp. 542–756, 5 Pls.)
Grassi, B. Interno allo sviluppo delle api nell’ uovo. (Atti Acad. Gioenia. Scienc. Nat. Catania (3), xviii, 1884.)
—— Breve nota intorno allo sviluppo degli Japyx. Catania, 1884, also in I progenitori degli Insetti e dei Miriopodi. 1. L’ Japyx e la Campodea. (Atti Acad. Gioenia Sc. Nat. Catania (3), xix, 1885.)
Ayers, H. On the development of Œcanthus niveus and its parasite Teleas. (Mem. Boston Soc. Nat. Hist., iii, 1884, pp. 225–281, 8 Pls.)
Witlaczil, Em. Entwicklungsgeschichte der Aphiden. (Zeitschr. f. wiss. Zool., xl, 1884, pp. 559–696, 9 Taf.)
Hallez, P. Orientation de l’embryon et formation du cocon chez la Periplaneta orientalis. (Compt. rend. Ac. Sc. Paris, ci, 1885, pp. 444–446.)
—— Sur la loi de l’orientation de l’embryon chez les Insectes. (Compt. rend. Ac. Sc. Paris, ciii, 1886, pp. 606–608.)
Heider, K. Ueber die Anlage der Keimblätter von Hydrophilus piceus. (Abh. k. Acad. Wiss. Berlin, 1885.)
—— Die Embryonalentwicklung von Hydrophilus piceus L. (I. Theil. Jena, 1889, pp. 1–98, 13 Taf., 9 Figs.)
Carrière, J. Kurze Mittheilungen aus fortgesetzten Untersuchungen über die Sehorgane. 7, Die Entwicklung und die verschiedenen Arten der Ocellen. (Zool. Anzeiger, ix Jahrg, 1886, pp. 141–147, 479–481, 496–500.)
—— Die Entwicklung der Mauerbiene (Chalicodoma muraria Fabr.) im Ei. (Arch. f. Micr. Anat., xxxv, 1890, pp. 141–165, 1 Taf.)
—— Die Drüsen am ersten Hinterleibsringe der Insectenembryonen. (Biol. Centralbl., xi, 1891, pp. 110–127, 3 Figs.)
Miall, L. C., and Denny, A. The structure and life-history of the cockroach (Periplaneta orientalis). London, 1886. (The section on embryology by J. Nusbaum.)
Ryder, J. The development of Anurida maritima Guerin. (Amer. Naturalist, xx, 1886, pp. 299–302, 1 Pl)
Stuhlmann, F. Die Reifung des Arthropodeneies nach Beobachtungen an Insecten, Spinnen, Myriopoden und Peripatus. (Ber. Freib. Naturf.-Gesellsch., i, 1886.)
590Blochmann, F. Ueber die Richtungskörper bei Insecteneiern. (Morph. Jahrbuch, xii, 1887, pp. 544–574, 2 Taf.)
Bruce, Adam Todd. Observations on the embryology of insects and arachnids. A memorial volume. Baltimore, 1887, 4º, pp. 31, 6 Pls.
Weismann, A., und Ischikawa, Ch. Ueber die Bildung der Richtungskörper bei thierischen Eiern. (Ber. Naturf. Ges. Freiburg, iii, 1887.)
Cholodkowsky, N. Ueber die Bildung des Entoderms bei Blatta germanica. (Zool. Anzeiger, xi Jahrg., 1888, pp. 163–166, 2 Figs.)
—— Studien zur Entwicklungsgeschichte der Insecten. (Zeitschr. f. wiss. Zool., xlviii, 1889, pp. 89–100, 1 Taf.)
—— Zur Embryologie von Blatta germanica. (Zool. Anzeiger, xiii Jahrg., 1890, pp. 137–138.)
—— Zur Embryologie der Hausschabe (Blatta germanica). (Biol. Centralbl., x, 1890, p. 425.)
—— Ueber die Entwicklung des centralen Nervensystems bei Blatta germanica. (Zool. Anzeiger, xiv Jahrg., 1891, pp. 115–116.)
—— Zur Embryologie der Insecten. (Ibid., 1891, pp. 465–466.)
—— Die Embryonalentwicklung von Phyllodromia (Blatta germanica). (Mém. Acad. St. Pétersbourg (7), xxxviii, 1891.)
Henking, H. Die ersten Entwicklungsvorgänge im Fliegenei und freie Kernbildung. (Zeitschr. f. wiss. Zool., xlvi, 1888, pp. 289–336, 4 Taf., 3 Figs.)
—— Ueber die Bildung von Richtungskörpern in den Eiern der Insekten und deren Schicksal. (Nachr. Ges. Wiss. Göttingen, 1888.)
Platner, G. Die erste Entwicklung befruchteter und parthenogenetischer Eier von Liparis dispar. (Biol. Centralbl., viii, 1888, pp. 521–524.)
Schmidt, F. Die Bildung des Blastoderms und des Keimstreifs der Musciden. (Sitz. Naturf. Ges., Dorpat, viii, 1889.)
Viallanes, H. Sur quelques points de l’histoire du développement embryonnaire de la Mante religieuse. (Rec. Biol. du Nord de la France, ii, 1889–1890.)
Voeltzkow, A. Entwicklung im Ei von Musca vomitoria. (Arb. Zool. Zoot. Inst. Würzburg, ix, 1889, pp. 1–48, 4 Taf.)
—— Melolontha vulgaris, ein Beitrag zur Entwicklung im Ei bei Insecten. (Arb. Zool. Zoot. Inst. Würzburg, ix, 1889, pp. 49–64, 1 Taf.)
Wheeler, W. M. Ueber drüsenartige Gebilde im ersten Abdominalsegment der Hemipterenembryonen. (Zool. Anzieger, xii, 1889, pp. 500–504, 2 Figs.)
—— The embryology of Blatta germanica and Doryphora decemlineata. (Journ. of Morph., Boston, iii, 1889, pp. 291–374, 7 Pls., 16 Figs.)
—— On the appendages of the first abdominal segment of the embryo cockroach (Blatta germanica). (Proceed. Wis. Acad. Science, Arts, and Letters, viii, 1890, pp. 87–140, 3 Pls.)
—— Ueber ein eigenthümliches Organ im Locustidenembryo (Xiphidium ensiferum). (Zool. Anzieger, xiii, 1890, pp. 475–480.)
—— Neuroblasts in the Arthropod embryo. (Journ. of Morphol., iv, 1891, pp. 337–343, 1 Fig.)
—— A contribution to insect embryology. In. Diss. (Journ. Morph., Boston, viii, 1893, pp. 1–160, 6 Pls., 7 Figs.)
Heymons, R. Ueber die hermaphroditische Anlage der Sexualdrüsen beim Männchen von Phyllodromia (Blatta) germanica. (Zool. Anzeiger, xiii Jahrg., 1890, pp. 451–457, 3 Figs.)
—— Die Entstehung der Geschlechtsdrüsen von Phyllodromia (Blatta) germanica L. In. Diss. Berlin, 1891.
—— Die Embryonalentwickelung von Dermapteren und Orthopteren unter besonderer Berücksichtigung der Keimblätterbildung. Monographisch bearbeiteit, 12 Lith. Taf. und 33 Figs. Jena, 1895, 4º, pp. 136.
591—— Ueber die Fortpflanzung und Entwickelungsgeschichte der Ephemera vulgata L. (Sitzungsb. Gesell. Naturf. Freunde, Berlin, 1896, pp. 81–96.)
—— Entwicklungsgeschichtliche Untersuchungen an Lepisma saccharina L. (Zeitschr. wiss. Zool., lxii, 1897, pp. 588–631, 2 Taf.)
Ritter, R. Die Entwicklung der Geschlechtsorgane und des Darmes bei Chironomus. (Zeitschr. f. wiss. Zool., 1, 1890, pp. 408–427, 1 Taf.)
Tichomirowa, O. S. Zur Embryologie von Chrysopa. (Biol. Centralbl., x, 1890, p. 423.)
Pedaschenko, D. Sur la formation de la bandelette germinative chez Notonecta glauca. (In Russian.) (Revue Sc. Natural. St. Pétersbourg, i, 1891.)
Korschelt, E., and Heider, K. Lehrbuch der vergleichenden Entwicklungsgeschichte der wirbellosen Thiere. (Spec. Th. Heft ii, Jena, 1891, many Figs.)
With the writings of Lang (Comp. Anatomy).
PART III.—THE METAMORPHOSES OF INSECTS
We have seen that the embryo rapidly passes through extraordinary changes of form, and now, after hatching, especially in the insects with a complete metamorphosis, the animal continues to undergo striking changes in form, in adaptation to different modes of life.
The life of a winged insect, such as a butterfly, fly, or bee, may be divided into four stages: the embryo, or egg state, the larva, pupa, and imago,—the term metamorphosis being applied to the changes after birth, or post-embryonic stages of life. The transformations of the more specialized orders of insects involve wonderful changes of form, which are only paralleled in other types of animals by the metamorphoses of the echinoderms, of certain worms, and of the Crustacea, as well as by those of the frog. An insect, such as a butterfly or bee, during its post-embryonic life lives, so to speak, three different lives, having distinct bodily structures and existing under quite dissimilar surroundings and habits; so that a caterpillar is practically a different animal from the pupa, and the latter from the imago, with different organs, the appendages and other structures being so modified as to be, so far as regards their functions, radically different. These changes of functions or of habits have also been plainly enough the exciting cause of the divergency in structure of what fundamentally is one and the same organ, the change having been brought about by adaptation of the same organs to quite different uses.
The changes are not only observable in the body and its appendages, but also in the internal organs, and consequently are both structural and physiological. The term larva, as applied to the first stage of animals, is a very variable and indefinite one, that of insects in general being a much more highly organized animal than the larva of a worm, starfish, or crustacean.
a. The nymph as distinguished from the larval stage
As there is no marked difference between the different stages of the young in the insects with an incomplete metamorphosis (Heterometabola), 594the chief difference being the possession of the rudiments of wings and the absence of a resting stage, the terms larva and pupa are in reality scarcely applicable to them, and we much prefer the term nymph, first proposed by Lamarck for the active “pupa” of Orthoptera, Hemiptera, the Odonata and Ephemeridæ, and adopted in part by many. Indeed, in the more generalized and older orders, the larval and pupal stages are not differentiated, though the term larval, in its general sense, will probably always be used; just as we speak of the larval stages of worms, echinoderms, or Crustacea.
Eaton in his elaborate work on the Ephemeridæ employs the term nymph to designate all the aquatic or early stages in the development of the young after hatching, and he urges that the old-fashioned usage of larva and pupa seem scarcely worth retention. “Nymphs are young which live an active life, quitting the egg at a tolerably advanced stage of morphological development and having the mouth-parts formed after the same main type of construction as those of the adult insect.” The word nymph is used in the same sense by McLachlan, and by Cabot. Calvert also applies the term nymph “to the stage of odonate existence between the egg and the transformation into the imago.” On the other hand, Brauer applies the term nymph to the pupa of holometabolous insects. For larval Hyatt proposes the term nepionic.
b. Stages or stadia of metamorphosis
The intervals or periods between the moults or ecdyses of caterpillars and other eruciform larvæ are called stages or stadia; thus, as most caterpillars moult four times, we have five stages or stadia, or stage (stadium) I to V. As observed by Sharp, there is, unfortunately, no term in general use to express the form of the insect at the various stadia; “entomologists say, ‘the form assumed at the first moult,’ and so on.” Hence he adopts a term suggested by Fischer,[86] and calls the insect as it appears after leaving the egg the first instar, and what it is after the first moult the second instar, and so on; hence the pupa, or chrysalis, which assumed that condition after moulting five times would be the sixth instar, and the butterfly itself would be the seventh instar.
c. Ametabolous and metabolous stages
In the Synaptera development is direct, the young differing neither in form, structure, or habits from the adult. Hence they are said to be ametabolous. Since there is an absence of even a tendency to a partial metamorphosis, it is evident that the insects have not inherited a tendency to undergo a transformation, but that it is 595an adaptation induced in the hexapod type after the first winged insects appeared, and which became more marked in the more specialized insects and at a period comparatively late in geological history, i.e. perhaps at or soon after the beginning of the Carboniferous period.[87]
The transformations of the pterygote insects vary greatly in degree, and it is difficult to draw the line between the grades. Those in which the adults differ from the freshly hatched young only or mainly in having wings are generally said to have an incomplete or gradual metamorphosis. There is no inactive, resting, or pupal stage, and the wings are acquired only after successive moults. Insects with an incomplete metamorphosis are the Orthoptera, Dermaptera, Platyptera (Mallophaga, Plecoptera, Corrodentia, Embidæ), Ephemeridæ, Odonata, Thysanoptera, and Hemiptera, with the exception of the male Coccidæ, in which there is a resting or sub-nymph stage. As regards the number of moults in the Synaptera, Grassi states that in Campodea there is a single fragmentary ecdysis, while Sommers tells us that Macrotoma plumbea sheds its skin throughout life, even after attaining its full size.
As an example of the partial metamorphosis of the hemimetabolous insects we may select that of the locust, in which there are five moults and six stages (instars), as seen in Fig. 558, five of which are nymphal. In the first two stages there are no rudiments of wings, these appearing after the second moult. Besides the acquisition of wings there are slight differences after each moult, both in structure and color, besides size, so that we may always recognize the comparative age and the particular stage of growth of any individual.[88]
We have watched the development of Melanoplus spretus from the egg to the imago, and examined thousands of specimens which show the six stages. On the other hand, European authors differ as to whether there are three, four, or five moults in the migratory locust.[89] It is not improbable that, as is the case with many other insects, the number of moults may vary according to the temperature and food, variation in these agencies causing either retardation or rapidity in development.
Those with a complete metamorphosis are said to be metabolous or holometabolous. (Lang.)
596Leach[90] in 1815 gave the name of Ametabola to insects without, and Metabola to insects with a metamorphosis.
Fig. 558.—Partial metamorphosis of Melanoplus femur-rubrum, showing the five nymph stages, and the gradual growth of the wings, which are first visible externally in 3, 3b, 3c.—Emerton del.
597Latreille (1831) called insects with an incomplete metamorphosis homotenous (which means similar to the end of life), and those with a complete metamorphosis, polymorphous. For the different degrees of metamorphosis of insects he employed two terms: for the incomplete degree, metamorphosis dimidia, and for the total or pupal, metamorphosis perfecta.
Westwood in his Introduction to the Modern Classification of Insects (1839), taking into account the relation of the larva with the imago, divided insects into two divisions: the Heteromorpha, or those in which there is no resemblance between the parent and its offspring, and Homomorpha, in which the larva resembles the imago, except in the absence of wings.
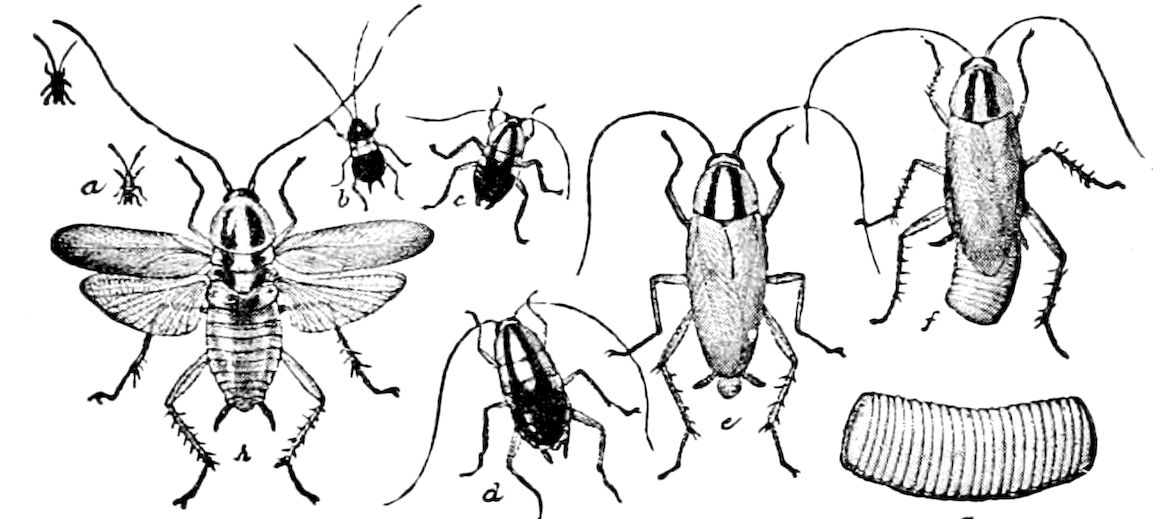
Fig. 559.—Manometabolous metamorphosis of the cockroach (Phyllodromia germanica) with its four nymphal stadia a-d; e, h, adult; f, female with egg-case; g, egg-case.—From Riley.
From the point of view of the degree of metamorphosis, insects have been divided into Heterometabola and Metabola.
I. Heterometabola.—This group may be divided as follows:
1. Manometabola,[91] embracing those forms with a slight or gradual metamorphosis, but which are active in all the stages, without any resting stage. The orders passing through this degree of metamorphosis are the following: Orthoptera, Dermaptera, Platyptera, Thysanoptera, and Hemiptera (Coccidæ excepted).
In all these groups, the only external differences of importance between the freshly hatched nymph and the adult is the presence of wings. The chief difference internally is the complete development of the sexual glands.
It should be observed, however, that in the last nymph stage of the Thysanoptera the articulations of the limbs are enveloped by a membrane and the wings enclosed in short fixed sheaths; the antennæ are turned back on the head, and the insect, though it moves about, is much more sluggish than in the other state. (Haliday.) Hence here we have a close approach to the following degree.
2. Heremetabola,[92] including those forms with a gradual though 598slight or incomplete metamorphosis, but with a quiescent or resting stage at the close of the nymph life. Lang has emphasized this stage, calling attention to the fact that the fore legs of the nymph of the 17–year Cicada, which lives underground on the roots of trees, are thick and adapted for digging. The transition from the nymph to the winged adult is signalized by the decided change in form of the fore legs, as well as by the acquisition of the wings. “The last larval stage is, then, what is called quiescent, i.e. the organization of the imago develops within the chrysalis at the expense of the accumulated reserve material.” (Lang.) There seems to be a resting stage, when the insect does not perhaps suck the sap from the roots, and awaits in its chamber its approaching change to the imago; but we should scarcely apply the term pupa to this stage, though the antennæ of the freshly hatched larva are larger and longer than in the fully grown nymph and are distinctly 8–jointed.
3. Hemimetabola.—In this division, so named by Brauer, the changes are more marked, though there is no truly inactive pupa-like stage. The orders are Perlaria (Plecoptera), Odonata, and Plectoptera (Ephemeridæ). The freshly hatched nymphs of these three groups are much alike in shape, that of Perlidæ, and indeed most of the Platyptera, being more generalized, unless we except that of Chloëon; all closely recall Campodea, and are therefore in the Campodea-stage. These nymphs are indeed more generalized than the freshly hatched nymph of Blattidæ, or any other of the orders mentioned except the Platyptera, to which perlids belong. They all have feet, and the body is more or less flattened. (Fig. 560.)
II. Holometabola.—In this division we have for the first time a true larva, and a pupa stage as distinguished from the imago. Moreover, the insect at each stage is distinguished by radical differences in form, surroundings, and in the nature of the food, while the pupa is inactive, usually immovable, and incapable of taking any food, and is often protected by a cocoon spun by the larva. The holometabolous orders are the Neuroptera, Coleoptera, Mecoptera, Trichoptera, Lepidoptera, Siphonaptera, Diptera, and Hymenoptera.
As we have among worms, echinoderms, and Crustacea certain exceptional species in a metamorphic group whose metamorphosis is suppressed, their development being direct, so there is in pterygote insects, though in a very much less degree, cases of direct development. In the wingless cockroaches such as Pseudoglomeris, etc., of the tribe of Periphæriides, in some of which, however, the males are winged, and in the Hemiptera, occur wingless forms such as the lice and bed-bug. The Mallophaga are all wingless, while certain Dermaptera (Chelidura, Anisolabis) are also apterous. The absence of wings in such cases is due to disuse from parasitism, or to a life under stones or in cracks and 599fissures, where the insects are driven to avoid their enemies, and hence do not need wings. The growth of wings and consequently the development of a metamorphosis is suppressed, so that, as Lang says, “in contrast to the original ametabola of the Apterygota, we have here an acquired ametabola.”
It is rare that, after the rudiments of wings have once appeared in the very young, they should disappear in the late nymph stage; this is, however, said by Walsh to be the case with the Ephemerid Bætisca (Fig. 440). This is a case of retardation in an acquired ametabolesis.
THE LARVA
The term larva is peculiarly applicable to the young of the holometabolous orders. The name (Latin, larva, a mask) was first given to the caterpillar because it was thought by the ancients to mask the form of the perfect insect. Swammerdam supposed that the larva contained within itself “the germ of the future butterfly, enclosed in what will be the case of the pupa, which is itself included in three or more skins, one over the other, that will successively cover the larva.” What led to his conception of the nature of these changes was probably his observations on the semitransparent larva of the gnat, in which the body and limbs of the pupa can be partially seen; for Weismann has shown that the great Dutch observer’s belief that the pupal and imaginal skins were in reality already concealed under that of the larva is partially founded in fact. Swammerdam states: “I can point out in the larva all the limbs of the future nymph, or Culex, concealed beneath the skin,” and he also observed beneath the skin of the larvæ of bees, just before pupating, the antennæ, mouth-parts, wings, and limbs of the adult. But, as we shall see farther on, the discovery by Weismann in the larva of the germs of the imago has completely changed our notions as to the nature of metamorphosis, and revolutionized our knowledge of the fundamental processes concerned in the change from larva to pupa, and from pupa to imago.
Not only are the larvæ of each order of insects characteristic in form, so that the grub or larva of beetles is readily distinguished from those of other orders, or the maggot of flies from the apodous larva of wasps and bees, but within the limits of the larger orders there is great diversity of larval forms, showing that they are the result of adaptation to their surroundings. This is especially the case with the larvæ of the Coleoptera, Lepidoptera, Diptera, and Hymenoptera.
In general, the larvæ of insects may be divided into two types,—the 600Campodea-form, or campodeoid, sometimes called thysanuriform, and the eruciform.
a. The Campodea-form type of larva
This is the most primitive and generalized type of larva (Fig. 560). A Campodeoid larva is one nearest in general shape to Campodea, the form which we have seen to be the nearest allied to the probable ancestor of the insects, and it also resembles the nymphs of the heterometabolous insects, before the appearance of their rudimentary wings.
Brauer, in 1869,[93] first suggested that the larvæ of a great number of insects may be traced back to Campodea and Iapyx. The Campodea-form larva is active, with a more or less flattened body, well developed mandibulate mouth-parts, and usually long legs. The nearest approach to the form of Campodea is the freshly hatched nymph of cockroaches (Blattidæ), Forficula, Perlidæ, Termitidæ, Psocidæ, Embidæ, Ephemeridæ, Odonata, especially the more generalized Agrionidæ, the nymphs of Hemiptera, the larvæ of certain Neuroptera, the active pedate larvæ of the more generalized Coleoptera, such as those of Carabidæ, Cicindelidæ, Dyticidæ, etc., and the first larva (instar) of Stylopidæ and Meloidæ (Fig. 560, d).
While the Campodea-shape is retained throughout nymphal life, of the orders above mentioned the Neuroptera and Coleoptera alone have a true resting pupal stage.
It should also be observed that great changes in the form of the nymph occur within the limits of the Orthoptera; the nymph of all the families except that of the Blattidæ, evidently the most generalized and primitive, being more or less specialized, while the nymphs of the other orders all vary in degree of specialization and modification. The process of adaptation once begun went on very rapidly, as it has in many other orders of insects, as well as in animals of other phyla.

Fig. 560.—Examples of campodeoid nymphs and larvæ: a, Campodea; b, Podura (Degeeria); c, Lepisma; d, triungulin larva of Meloë; e, Perla; f, Forficula; g, Chloëon; h, May-fly (Palingenia); i, Æschna; j, Atropos; k, Myrmeleon; l, Sialis; m, Corydalus; n, Cicada.
b. The eruciform type of larva
Brauer also sagaciously pointed out that “a larger part of the most highly developed insects assume another larva form, which appears not only as a later acquisition, through adaptation to certain definite conditions, but also arises as such before our eyes. The larvæ of Lepidoptera, of saw-flies, and Panorpidæ show the form most distinctly, and I call this the caterpillar form (Raupenform). That this is not the primitive form, but one later acquired, we see illustrated in certain beetles. The larvæ of Meloë and of Sitaris, in their fully grown conditions, possess the caterpillar form, but the new-born larvæ of these genera show the Campodea-form. The last form is lost as soon as the larva begins its parasitic mode of life.... The larger part of the beetles, the Neuroptera (in part), the bees and flies (the last with the most degraded maggot form), possess larvæ of this second form.” In 1871 we adopted these views, giving the name eruciform to this type of larvæ, and afterwards Lubbock adopted Brauer’s views. Brauer considered that the eruciform larva was the result of living a stationary semi-parasitic life on plants, in carrion, or burrowing in the trunks and branches or leaves and buds of trees, where they do not have to move about in search of their food. The change from the Campodea-form to the eruciform larva is a process of degeneration and often of atrophy of the limbs, and, in the footless forms of dipterous and hymenopterous insects, of the gnathites, accompanied by a tendency of the body to become more or less cylindrical.
The first steps in the origination of the eruciform larva were apparently taken in the order Neuroptera, as restricted by Brauer and by myself, where, though the larvæ are campodeoid, there is a true resting pupal stage. The most generalized larval form is perhaps that of the Sialidæ (Fig. 560, l), in which the body tends to be slightly cylindrical, though the legs are long, and the gnathites well developed for seizing and biting their living prey. The terrestrial larvæ of the Hemerobiidæ, though modifications of the sialid larval form, are considerably specialized in adaptation to their active carnivorous habits. But the life-history of Mantispa, where there are two larval stages, gives us plainly enough the key to the mode in which the complete metamorphosis was brought about. The larva, born a true Campodea-like form, with large, long, 4–jointed legs, has a structure which would enable it to move about freely after its prey, beginning at once to live a sedentary life in the egg-sac of a spider; before the first moult it loses the use of its legs, while the antennæ are partly aborted. The result is that, owing to this change of habits and surroundings from those of its active ancestors, it changes its form, and the fully grown larva becomes cylindrical, with small slender legs, and, owing to the partial disuse of its jaws, acquires a small, round head.

Examples of coleopterous larvæ, showing the passage from the campodeoid to the eruciform type of larvæ.
Fig. 561.—Coleopterous larvæ showing passage from campodeoid to eruciform larvæ: a, b,
Harpalus; c, Dyticus; d, Staphylinus; e, Silpha; f, Melanactes; g, Ludius; h, Elater; i, Donacia;
j, Chrysobothris; k, Orthosoma; l, Coccinella; m, Byrrhus; n, Trox; o, p, Lachnosterna; q,
Labidomera; r, Ptinus; s, Anobium; t, Balaninus (entirely apodous).
604Its antennæ, mouth-parts, and legs not only retarded in growth, but retrograding and becoming vestigial, the body meanwhile becoming fat and cylindrical, an apparent acceleration of growth goes on within, with probably an enlargement of the intestine and fat-body, and thus the pupal form is perfected while the larva is full-fed and quiescent. It is not improbable that in the primitive neuropteron, as the result of a mode of life like that of Mantispa, the quiescent life of the later stages graduated into a quiescent, inactive pupal life, allowing the changes going on in the internal organs to result in a complete metamorphosis, which was transmitted to the later Neuroptera, thus making the complete metamorphosis a fixed, normal condition. It thus appears that a change of habits and of food, and more especially the fact that the nymph became so surrounded with an abundance of food close at hand that it did not have to run actively about and seize it in a haphazard manner, were the factors bringing about a change from the Campodea-form nymph to the eruciform larva, thus inducing a hypermetamorphosis.
The larvæ of the Mecoptera (Panorpidæ, Fig. 562, b) are still more caterpillar-like, and besides their cylindrical body, rounded head, small short gnathites, small thoracic legs, they have what appear to be 2–jointed legs to each of the nine abdominal segments, and the close resemblance to caterpillars is farther carried out by the presence of a pair of prothoracic spiracles, none existing on the other two thoracic segments.

Fig. 562.—Examples of eruciform larvæ: a, Phryganea; b, Panorpa; c, Sesia; d, d, caterpillars; e, Selandria; f, Tipula; g, Simulium; h, Chionea; i, Musca; j, Tachina; k, Braula; l, flea; m, Tremex; n, coarctate larva of Meloë; o, bee (Andrena).
606In the Meloidæ (Fig. 560, d) and Stylopidæ the first larval stage is Campodea-form; the changes will be described in the subsequent section on Hypermetamorphosis, and while these cases of change from a campodeoid to an inactive eruciform larva are very salient, if we compare the graduated series of larval forms throughout the order of Coleoptera, as represented by the illustrations in Fig. 561, we shall see that in nearly, if not each, case the form of the boring or mining, or bark or bud or seed-inhabiting grub is the result of a change of habit and commissariat from active predaceous larvæ, like those of the Carabidæ and other adephagous families, together with those of the Staphylinidæ, with their flat body, big mandibles, and well-developed maxillæ, to the cylindrical bodies of such larvæ as those of Dermestes and Anthrenus, which live a more sedentary life, to the root-feeding wire-worm or elaterid larvæ, and scarabæid grubs, onward to the phytophagous Chrysomelidæ, with the mining and boring buprestids and cerambycids,—in all these forms we see a gradual atrophy of the legs, which is fully carried out in the vermiform or maggot-like larva of the weevils. These changes throughout the members of the entire order are epitomized in the life-history of the Meloidæ, in which there are three typical forms of larva: the Campodea-form (triungulin stage), eruciform (second or carabidoid stage), and vermiform (coarctate) larva.
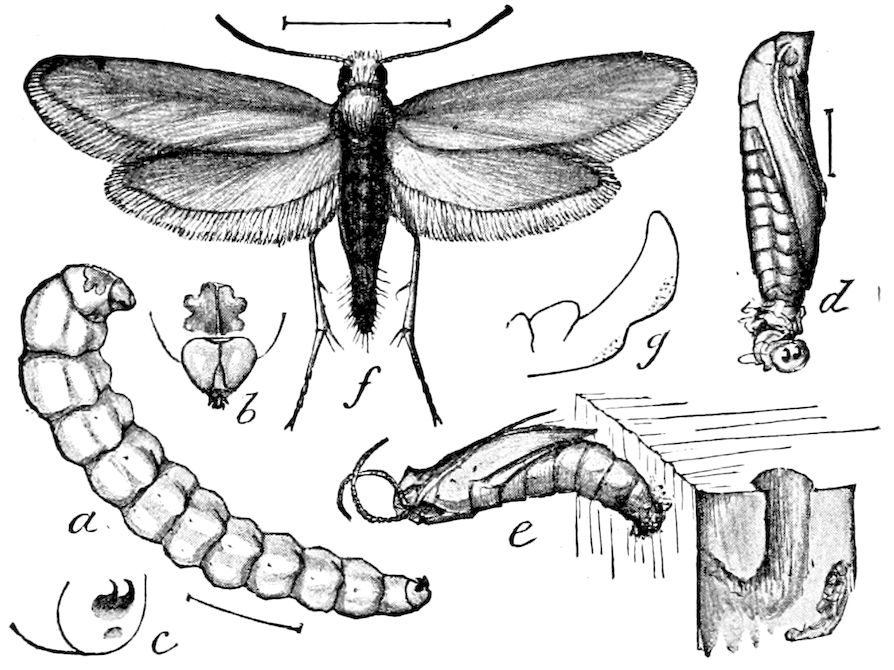
Fig. 563.—Prodoxus cinereus: a, apodous larva; b, head and prothoracic segment; c, anal hooks; d, pupa; e, cast pupal shell protruding from stalk of Yucca; f, female; g, side view of ♂ clasper.—After Riley, from Insect Life.
Fig. 564.—Larva of Limacodes scapha, nat. size.
In the Lepidoptera the eruciform, pedate type is adhered to throughout the order, with the rare exception of the nearly apodous mining larva of Prodoxus (Fig. 563, a), Phyllocnistis, and Nepticula, which have no thoracic legs, and the limacodid larvæ, whose abdominal legs are totally aborted, while the thoracic ones are much reduced (Fig. 564).
In the Hymenoptera the phytophagous forms are eruciform, while by the agency of the same factors as already mentioned, i.e. a sedentary or parasitic life and abundance of food within constant reach, the larvæ lose their legs and become vermiform.
607In the Diptera, which are the most highly specialized of insects, the maggot or vermiform shape, and absence of any legs, prevails throughout the order, though the eucephalous larvæ show their origin from a primitive eruciform type of larva. The highly specialized larvæ of the Culicidæ and Simuliidae are undoubtedly related to the earliest and most generalized types, while the maggots of the parasitic flies (Tachinidæ) and other muscids are later degradational forms, and the result of adaptation induced, as in the previous cases, by a sedentary or parasitic mode of life, living as they do immersed in an abundance of rich nitrogenous food, with the result that the mouth-parts have become atrophied by disuse, while the limbs have become entirely aborted, though the thoracic imaginal discs develop normally in the embryonic or pre-larval stages.
It appears, therefore, highly probable that the metamorphoses of insects are the result of the action of change of conditions, just as the polymorphism of Termites is with little doubt the result of differences of food and other conditions. These matters will be farther discussed under the head of Causes of Metamorphosis.
LITERATURE ON ANCESTRY OF INSECTS, ETC.
Müller, Fritz. Für Darwin, 1869, pp. 144, 67 Figs.
Brauer, Friedrich. Betrachtung ueber die Verwandlung der Insekten im Sinne der Descendenz-Theorie. (Verhandlung d. k.k. zool. bot. Gesell. Wien., 1869, 1 Taf., pp. 1–20.)
Packard, A. S. Amer. Naturalist, iii, p. 45, March, 1869.
—— Proc. Boston Soc. Nat. Hist., xiv, 1870, p. 61.
—— Amer. Nat., iv, Feb. 1871, p. 756; v, 1871, pp. 52, 567.
—— Embryological Studies. (Memoirs Peabody Acad. Sc. Salem, 1871–72.)
—— Our common insects, 1873, Chapter on Ancestry of Insects, pp. 175–178.
—— Third Report U. S. Ent. Commission, 1883, pp. 295–304.
Lubbock, John. On the origin of insects. (Journ. Linn. Soc., London, xl, 1873.)
—— Origin and metamorphoses of insects. (Nature, 1873 [in book form, 1874], pp. 108, 66 Figs.)
Mayer, Paul. Ueber Ontogenie and Phylogenie der Insekten. (Jena. Zeitschr. Wissens., x, 1876, pp. 125–221, 4 Taf.)
Hyatt, A., and Arms, J. M. Insecta. (Bost. Soc. Nat. Hist. Guides for science-teaching, viii.) Boston, 1890, pp. 300, 13 Pls., 223 Figs.
c. Growth and increase in size of the larva
The rapidity of growth and enormous increase in size in early
life is especially noticeable in caterpillars and other phytophagous
larvæ. The latest observations are those of Trouvelot on Telea
polyphemus. When this silkworm hatches, it weighs 1
20 of a grain.
When
| 10 days old it weighs | ½ a grain, or | 10 times the original weight. |
| 20 days old it weighs | 3 grains | 60 times the original weight. |
| 30 days old it weighs | 31 grains | 620 times the original weight. |
| 40 days old it weighs | 90 grains | 1800 times the original weight. |
| 56 days old it weighs | 207 grains | 4140 times the original weight. |
“When,” he says “a worm is 30 days old, it will have consumed about 90 grains of food; but when 56 days old, it is fully grown and has consumed not less than 120 oak leaves, weighing ¾ of a pound; besides this it has drank not less than ½ an ounce of water. So the food taken by a single silkworm in 56 days equals in weight 86,000 times the primitive weight of the worm. Of this about ¼ of a pound becomes excrementitious matter, 207 grains are assimilated, and over 5 ounces have evaporated.”[94]
Dandolo stated that the Asiatic silkworm (Bombyx mori) weighs on hatching
not over 1
100 of a grain, but when fully grown about 95 grains. During this
period, therefore, it has increased 9500 times its original weight, and has eaten
60,000 times its weight of food. Newport thought this estimate of the amount
of food was a little too great. But comparing it with Trouvelot’s estimate for
the American silkworm, which weighs when hatched five times as much, it
would not appear to be so. Newport found that the larva of Sphinx ligustri at
the moment of leaving the egg weighs about 1
80 of a grain, and when fully fed
125 grains, so that in the course of 32 days it increases about 9976 times its
original weight. This proportion of increase is exceeded by the larva of Cossus
ligniperda, which, boring in the trunks of trees, remains about three years in
the larva state, and increases, according to Lyonet, to the amount of 72,000
times its first weight.
Newport adds that those larvæ in which the proportion of increase is the
greatest, are usually those which remain longest in the pupa state, as in the
silkworm. “Thus Redi observed in the maggots of the common flesh-flies a
rate of increase amounting to about 200 times the original weight in 24 hours,
but the proportion of increase in these larvæ does not at all approach that of
the Sphinx and Cossus.” From his observations on the larva of one of the
wild bees (Anthophora retusa) Newport believes that this is also the case with
the Hymenoptera. The weight of the egg of this insect is about 1
150 of a grain,
and the average of a full-grown larva 68
10 grains, so that its increase is about
1020 times its original weight; “which compared with that of the Sphinx of
medium size, is but as 1 to 9¾, and to a Sphinx of maximum size only as 1 to
a little more than 11.”
609The growth is most rapid after the last moult. “Thus a larva of Sphinx ligustri, which at its last change weighed only about 19 to 20 grains, at the expiration of eight days, when it was fully grown, weighed nearly 120 grains.” (Newport.)
d. The process of moulting (ecdysis)
Insects periodically shed the exoskeleton, together with the chitinous lining of their internal organs of ectodermal origin, which thus sloughed off are called the exuvia. The process in the locust has been described by Riley.[95] It occupies from half to three-quarters of an hour (Fig. 565). This process has naturally, from the ease with which it can be observed, been most frequently examined in the Lepidoptera, though careful and detailed observations of the inner and outer changes are still greatly needed, especially in other orders. In the caterpillar of most moths, especially one of the more generalized bombycine moths, on slipping out of its egg-shell the head is of enormous size as compared with the body, but the latter soon fills out after the creature has eaten a few hours; the head, of course, does not during this time increase in size, and the larvæ of different instars may be exactly distinguished, as Dyar has shown, by the measurements of the head.
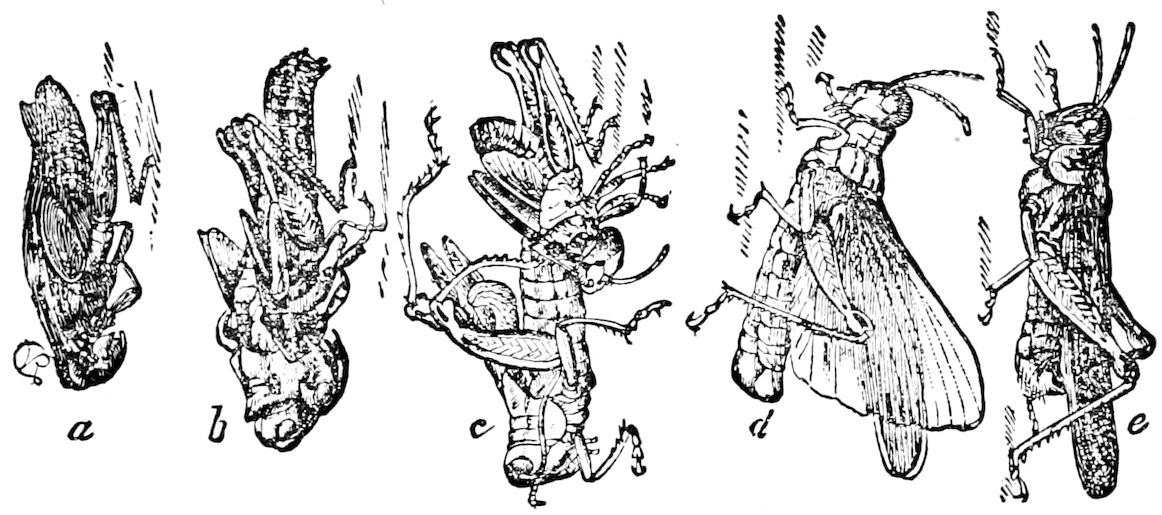
Fig. 565.—Process of moulting from nymph to imago in the locust (M. spretus): a, nymph with skin just split on the back; b, the imago drawing itself out, at c, nearly free; d, the imago, with wings expanded; e, the same with all parts perfect.—After Riley.
Before the caterpillar moults, it stops feeding, and the head is now small compared with the body; the head of the second instar is now large, situated partly under the much-swollen prothoracic segment, and pushes the head of the first instar forward.
Newport has well described the mode of shedding the skin in 610Sphinx ligustri, and his detailed description will apply to most lepidopterous larvæ.
The whole body is wrinkled and contracted in length, and there are occasionally powerful contractions and twitchings of its entire body; the skin becomes dry and shrivelled, and is gradually separated from the new and very delicate one of the next instar beneath. After several powerful efforts of the larva the old skin cracks along the middle of the dorsal surface of the mesothoracic segment, and by repeated efforts the fissure is extended into the 1st and 3d segment, while the covering of the head divides along the vertex and on each side of the clypeus. “The larva then gradually presses itself through the opening, withdrawing first its head and thoracic legs, and subsequently the remainder of its body, slipping off the skin from behind like the finger of a glove. This process, after the skin has once been ruptured, seldom lasts more than a few minutes. When first changed the larva is exceedingly delicate, and its head, which does not increase in size until it again changes its skin, is very large in proportion to the rest of the body.” (Art. Insecta, etc.)
Trouvelot’s account is more detailed and an advance on that of Newport’s view. He explicitly states, and we know that he was a very close observer, that the old skin is detached by “a fluid which circulates between it and the worm.” His account is as follows: The polyphemus worm, like all other silkworms, changes its skin five times during its larval life. The moulting takes place at regular periods, which comes around about every 10 days for the first four moultings, while about 20 days elapse between the fourth and fifth moulting. The worm ceases to eat for a day before moulting, and spins some silk on the vein of the under surface of a leaf; it then secures the hooks of its hind legs in the texture it has thus spun, and there remains motionless; soon after, through the transparency of the skin of the neck, can be seen a second head larger than the first, belonging to the larva within. The moulting generally takes place after four o’clock in the afternoon; a little before this time the worm holds its body erect, grasping the leaf with the two pairs of hind legs only; the skin is wrinkled and detached from the body by a fluid which circulates between it and the worm; two longitudinal bands are seen on each side, produced by a portion of the lining of the spiracles, which at this moment have been partly detached; meanwhile the contractions of the worm are very energetic, and by them the skin is pulled off and pushed towards the posterior part; the skin thus becomes so extended that it soon tears just under the neck, and then from the head. When this is accomplished the most difficult operation is over, and now the process of moulting goes on very rapidly. By repeated contractions the skin is folded towards the tail, like a glove when taken off, and the lining of the spiracles comes out in long white filaments. When about one-half of the body appears, the shell still remains like a cap, enclosing the jaws; then the worm, as if reminded of this loose skull-cap, removes it by rubbing it on a leaf; this done, the worm finally crawls out of its skin, which is attached to the fastening made for the purpose. Once out of its old skin, the worm makes a careful review of the operation, with its head feeling the aperture of every spiracle, as well as the tail, probably for the purpose of removing any broken fragment of skin which might have remained in these delicate organs. Not only is the outer skin cast off, but also the lining of the air-tubes and intestines, together with all the chewing organs and other appendages of the head. After the moulting, the size of the larva is considerably increased, the head is large compared with the body, but 8 or 10 days later it will look small, as the body will have increased very much in size. This is a certain indication that the 611worm is about to moult. Every 10 days the same operation is repeated. From the fourth moulting to the time of beginning the cocoon the period is about 16 days. (Amer. Naturalist, i, pp. 37, 38.)
Little has been recorded as to the exact mode of casting the larval skin in Coleoptera. Slingerland states that Euphoria inda when pupating sheds the larval skin off the anal end in the same way as in caterpillars, while in Pelidnota punctata the larval skin splits down the whole length of the back, retains the larval shape, and forms a covering for the pupa which remains inside. (Can. Entomologist, xxix, p. 52.) The old larval skin in the Coccinellidæ and certain Chrysomelidæ is retained crumpled up at the end of the body, while in Dermestes, Anthrenus, etc., it cloaks the pupa.
Not only is the integument, with its hairs, setæ, and other armatures, as well as the cornea or facets of the eyes, shed, but also all the lining or intima of those internal organs which have been originally derived by an ingrowth or invagination of the ectoderm are likewise cast off, with the probable exception, of course, of the mid-intestine, which is endodermal in its origin. Even so early an observer as Swammerdam noticed that the internal lining of the alimentary canal comes away with the skin. He states that the larva of Oryctes nasicornis sheds both the lining of the colon, and of the smaller as well as the larger branches of the tracheæ.
Careful observations are still needed on the internal changes at ecdysis of most insects. Newport seems to have observed more closely than any one else, notwithstanding the great number who have reared caterpillars but have not carefully observed these points, the extent of the process internally. He informs us: “The lining of the mouth and pharynx, with that of the mandibles, is detached with the covering of the head, and that of the large intestines with the skin of the posterior part of the body, and besides these also the lining of the tracheal tubes. The lining of the stomach itself, or the portion of the alimentary canal which extends from the termination of the œsophagus to the insertion of the so-called biliary vessels, is also detached, and becomes completely disintegrated, and appears to constitute part of the meconium voided by the insect on assuming its imago state.” (Art. Insecta, p. 876.) Newport states on another occasion that he had “noticed the remarkable circumstance [now explained by the fact that the mid-intestine is of endodermal origin] that the mucous lining of the true ventriculus was not cast off with the rest, but was discharged with the fecula.”[96] Burmeister also observed that the smaller tracheæ as well as the internal tunic of the colon of Libellulæ are shed.
In the apodous larvæ of Hymenoptera which live in cells, as we have observed in those of Bombus, during the process of moulting, the delicate skin breaks away in shreds, probably owing to the tension due to the unequal growth of the different parts of the body. “Thus after the skin beneath has fully formed, shreds of the former skin remain about the mouth-parts, the spiracles, and anus. Upon pulling upon these, the lining of the alimentary tube and tracheæ can be drawn out, sometimes, in the former case, to the length of several lines.”[97] We then added, “As all these internal systems of vessels are destined to change their form in the pupa, it may be laid down as a rule in the moulting of insects and Crustacea, that the lining of the internal organs, which is simply a continuation of the outer tegument, or arthroderm, is, in the process of moulting, sloughed off with that outer integument.” We have satisfied ourself that in the larvæ of the Lepidoptera (e.g. Datana) the tracheæ at the time of ecdysis 612undergo a complete histolysis, and arise de novo from hypodermal cells, the so-called spiral threads originating from elongated peritracheal nuclei. (See p. 449, Fig. 412.) This is undoubtedly also the case with the salivary ducts, which are strengthened and rendered elastic by tænidia like those of tracheæ. As the urinary tubes are diverticula of the proctodæum, itself an ectodermal invagination, they may also, though not lined with a chitinous intima, be renewed. With little doubt the intima of the ducts of poison, spinning, and most, if not all the other glands, though certainly the dermal glands, is exuviated. We have found that the lobster in moulting sheds, besides the skin with the most delicate setæ, the lining of the proventriculus, and the apodemes of the head and thorax, hence it is most probable that the tentorium of the head of insects as well as the apodemes and phragmas of the thorax are exuviated.
The formation of the inner skin, or that of any succeeding stage (instar), is due to the secretion of the structureless chitinous layer by the cells of the hypodermis, during the process of histogenesis. These cells at this time are very active, and the formation of the new layer of chitin arrests the supply of nourishment to the old skin, so that it dries, hardens, and with the aid of the fluid thrown out at this time separates from the new chitinous layer secreted by the hypodermis.
Mention of this fluid, which Newport was the first to observe, and which he says causes the separation of the old from the underlying fresh integument of the caterpillar, recalls a passage in Hatchett-Jackson’s Studies in the morphology of the Lepidoptera, which we quote on a succeeding page, where he calls attention to the formation of such a liquid, which in the reptiles facilitates the process of moulting, adding, “Whether such is the case with the moult of the caterpillar, I do not know.” Is it not also possible that the growth of the setæ or tubercles on the cuticle of the caterpillar may likewise serve to loosen and detach the overlying skin about to be cast off? After writing the foregoing, we find that Miall and Denny have suggested that the setæ of the cockroach probably serve the same purpose as the casting-hairs of the crayfish and reptiles.
It is well known that in the crayfish and in lizards the skin is first loosened by the growth of temporary hairs or setæ, which locally grow inward from the old cuticle and push the skin away when it is shuffled off by the movements of the body, jaws, and limbs, as well as the body in general.[98]
Such spines arise in the pupa of many insects, for Verhoeff finds that the spines and teeth of pupal fossorial and other Hymenoptera, as well as Coleoptera, function as moulting-processes for loosening and pushing off the last larval skin, rather than for locomotion. He also claims that the spines of the pupa of the dipterous Anthrax are both for locomotion and for boring, especially the spines on the head and tail. He therefore divides these pupal spines into helcodermatous (boring or tearing) and locomotor spines.
613Gonin has fully confirmed Newport’s discovery of the exuvial fluid. He states that during pupation the outside of the pupa, especially the parts of the head and thorax “is coated with a viscous liquid secreted by special glands.” The parts only harden subsequent to pupation after exposure to the air (p. 41). His observations were made under the direction of Professor Bugnion, who kindly writes us:—
“M. Gonin has proved the formation of a liquid which passes under the cuticle at the time of the last moult and facilitates exuviation. We think that this liquid is secreted by large cells (unicellular glands) which we see especially on the surface of segments 1–3. These cells form part of the hypodermis, and their pores open under the cuticle.”
In a subsequent letter enclosing a sketch kindly made for me by M. Gonin (Fig. 566), Professor Bugnion writes me Aug. 24, 1897, regarding the functions of the large hypodermal cells (l. hy), as follows: “It seems to me, in fact, after having again examined the sections, that the function of these cells is not sufficiently elucidated. Indeed these cells occur only in the section passing through the 1st segment, between the head and 1st thoracic segment. It would seem, if these cells supply the liquid which lubricates the surface at the time of ecdysis, that they should be spread over the entire surface of the body. Moreover, these cells have no distinct orifice, and although there is seen at times to issue streams of a substance (coagulated by the reagents), they cannot be compared with true unicellular glands like those of the epidermis of fishes, amphibians, etc.
“On the other hand, if it is the blood which oozes out on the surface (according to your hypothesis), it would seem that the loss of blood would cause the death of the larva. I believe then it is due to the secretion of the hypodermis which spreads over the whole surface when the cells are still soft (not yet hardened from contact with the air). At all events, there is a liquid spread over the surface; it is this liquid which glues the wings and the legs to the body at the moment the caterpillar issues from the rent in its skin. If at this instant we plunge the pupa in the water the liquid is dissolved, and the feet, wings, etc., are not glued to the body.”
Dr. T. A. Chapman also writes us: “There is no question about the existence of a fluid between the two skins at moulting. In hairy larvae the hairs are always wet at first, or if the skin be renewed rather more quickly than the larva does it naturally, the wetness of both surfaces is obvious. I do not know the nature of the fluid, but it is related to that which hardens into the dense pupal case, and also hardens in a less degree the skin of the larva. I suppose it must contain some chitin in a soluble form. If a newly cast larva skin be taken, there is no difficulty in extending the shrivelled mass to its full length and dimensions, but if a short time elapses, this chitin hardens, and the skin cannot be extended after soaking in water, alcohol, ammonia, or any other solvent I have tried.”
It has been stated that there is a subimaginal pellicle in Lepidoptera, but as Dr. Chapman writes me, “what has been observed has been some of the inner pupal dissepiments, such as the pupal cases of the under wings,” etc. They may be observed in the head of the tineid pupæ, and other small moths. We have thought that the 614delicate, purplish, powdery layer left in the cast shells of the pupæ of saturnians, Catocalæ, and other moths, might possibly be such a pellicle, but this view has been dispelled by the following statement of Professor Bugnion in a letter answering an inquiry whether he had noticed such a pellicle.
“A liquid which is secreted in a few minutes at the time of the last moult, forms in drying a yellowish layer spotted with black (in Pieris brassicæ). This layer extends around the entire pupa, and serves both to protect it and to glue together the wings, legs, etc., in their new position. The dried liquid on the surface of the pupa, and by means of which the appendages are glued to the surface, very likely corresponds to the pellicle of which you speak.” The newly exposed integument is at first pale and colorless, but soon assumes the hues peculiar to the species, and the insect, at first exhausted, after a short rest becomes active.
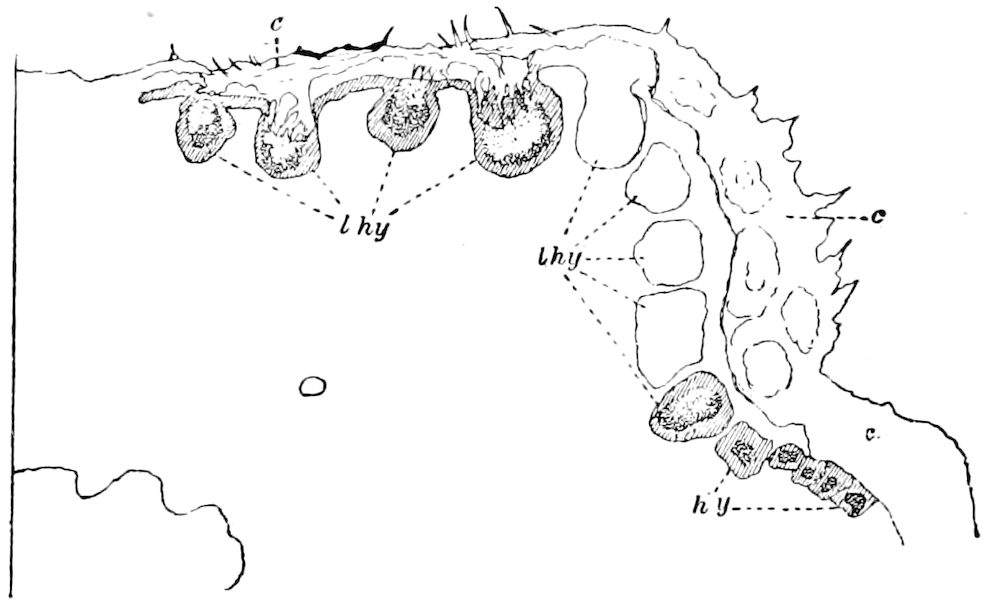
Fig. 566.—Transverse section through the prothoracic segment (ventral face) of larva of Pieris brassicæ, about 12 hours before pupation: c, cuticula; l. hy, large glandular (?) hypodermal cells; gradually passing into normal hypodermal cells (hy).—Gonin del.
E. Howgate has noticed under the microscope peculiar internal movements in a small immature transparent geometrid while moulting. “Each separate segment,” he says, “commencing at the head, elongated within the outer skin, whilst the next ones remained in their former state. Each segment in its turn behaved in this curious manner until the last was reached, when the motion was reversed and proceeded toward the head, when it was again reversed.... The whole proceeding appeared as if the larva was gliding within itself, segment after segment, the outer skin remaining as if held by the other segments, whilst the particular one in motion freed itself within. After remaining motionless for a short interval, the skin near the head swelled and burst, open at the back.... Presently out comes the head of the new caterpillar, pushing forward the old one.... After a short struggle the new true legs appear, pushing off and treading under foot the old ones. Then by violent wriggling movements the abdominal legs were extricated. Then all is clear, and the larva, which is quite exhausted, coils itself up and literally pants for breath.” (The Naturalist, November, 1885, No. 124, p. 366, quoted in Psyche, iv, p. 327, 1887.)
Since the worms and most other ametabolous invertebrates are not known to moult their integument, the body steadily increasing in size without frequent 615changes of skin, it seems that growth may go on and still be accompanied by considerable changes in shape of the body without change of skin. Frequent ecdyses appear, then, to be the result of the great and sudden changes of the body, necessitated by the adaptation of the animal to new or unusual conditions of life. In young Daphnia, a cladocerous crustacean, as many as eight moults were observed in a period of 17 days, and spiders frequently moult even after reaching their full size. The swollen bodies of the gravid female of Gastrophysa, Meloë, or of Termites, and of the honey ant show that the skin can stretch to a great extent, but in the metamorphoses of Crustacea and of insects, whose young are more or less worm-like or generalized in form, with fewer segments and appendages, or with appendages adapted for quite different uses from those of mature life, the necessity for a change of skin is seen to be necessary for mechanical reasons. Hence Crustacea and insects moult most frequently early in life, when the changes of form are most thoroughgoing and radical, while simple growth and increase in size are most rapid at the end of larval life, as seen both in shrimps and crabs, and in insects.
The hibernating caterpillars of certain butterflies are known to moult once oftener than those of the summer brood. Mr. W. H. Edwards has discussed the subject with much detail. “There seems,” he says, “to be a necessity with the hibernators of getting rid of the rigid skin in which the larva has passed the winter; that is, if the hibernation has taken place during the middle stages, as it does in Apatura and Limenitis. In these cases very little food is taken between the moult which precedes hibernation and the one which follows it, and the larva while in lethargy is actually smaller than before the next previous moult. The skin shrinks, and has to be cast off before the awakened larva can grow. Those species (observed) whose larva moults five times in the winter brood require but four moults during the summer.” He adds that while the larva is in lethargy, it is actually smaller than before the next previous moult. Dr. Dyar writes: “I think there is no doubt about the number of stages of arctian larvæ. They seem to have a great capacity of spinning out their life-history by interpolated stages (as regards width of head). I think it is because so many of them hibernate, and only a single brood extends throughout the season.” (Psyche iii, p. 161.)
On the other hand, it is difficult to understand why the caterpillars of arctians moult so frequently, nearly twice as often as in most other caterpillars, though the changes of form and armature are so slight.
Dr. Chapman also writes me: “Arctians resemble bears (Arctos), polar and others, in having long hairs to protect them during winter, and are, in fact, typically hibernators. Many of them have to half-hibernate, having warmth enough to keep them awake, but not enough food for growth, but their tissues, at least the chitinous ones of the cutis, and also probably, and perhaps especially, of the alimentary canal, become old and effete, and require the rejuvenescence acquired by a moult. Other smooth-skinned hibernators have similar capabilities.”
Chapman has shown in his paper on Acronycta that these caterpillars of this genus illustrate how larvæ may lose a moult, and they do so to acquire a sudden change of plumage.
The number of moults in insects of different orders.—It will be seen from the data here presented that the number of moults is as a rule greatest in holometabolic insects with the longest lives, and that an excessive number of ecdyses may at times be due to some physical cause, such as lack of food combined with low temperature.
616In Campodea there is a single fragmentary moult (Grassi), while the Collembola (Macrotoma plumbea) shed their skin throughout life. (Sommer.)
In the winged insects, especially Lepidoptera, the number of moults is dependent on climate. Insects of wide distribution growing faster in warmer climates consequently shed their skins oftener; for example, the same species may moult once oftener in the southern than in the northern States, as in the case of Callosamia promethea, which in West Virginia is double-brooded. Hibernating larvæ moult once oftener than those of the summer brood. (W. H. Edwards.) Weniger by rearing the larvæ of Antheræa mylitta and Eacles imperialis, and which, when reared under normal conditions, actually have six stages, found that when reared in a warm moist atmosphere of about 25° C. they have but five stages, i.e. moult but four times. In the hot and moist climate of Ceylon, A. mylitta has but five stages. (Psyche, v, p. 28.)
Among Orthoptera Acrydians moult five times; Diapheromera femorata but twice (Riley); a katydid (Microcentrum retinervis) moults four times (Comstock). Mantis religiosa, according to Pagenstecher, moults seven times, having eight stages, including that before the amnion is cast, but the first “moult” being an exuviation of the amnion, the number of stages is seven. Cockroaches (Periplaneta americana) are said by Marlatt to “pass through a variable number of moults, there being sometimes as many as seven.”
In the Homoptera there are, in general, from two to four moults; thus in Typhlocyba there are five stages, and in Aphis at least three, and in Psylla four during the nymphal state. Psocus has four. Riley states that the nymph of the female coccid, Icerya purchasi, sheds its skin three times, and that of the male twice. Notwithstanding its slow growth, Riley says, the 17–year Cicada moults oftener than once a year, and the number of larval stages probably amounts to 25 or 30 in all. The bed-bug sheds its skin five times; and with the last moult appear the minute wing-pads characteristic of the adult. In Conorhinus sanguisuga there are “at least two larval stages and pupal stages.” (Marlatt.)
In the dragon-flies moulting occurs, Calvert thinks, many times, since the rudiments of wings are said by Poletaiew to only appear in odonate nymphs after the third or fourth moult.
In the May-fly, Chloëon, the number of ecdyses is 20. The neuropterous Ascalaphus (Helecomitus) insimulans of Ceylon moults three times before pupating. Among the Mecoptera Felt has shown that Panorpa rufescens moults seven times.
617In Coleoptera the normal or usual number is not definitely known; Meloë moults five times, but this is a hypermetamorphic insect; Tribolium confusum has been carried by Mr. Chittenden through seven moults. Phytonomus punctatus, the clover-leaf weevil, moults three times, according to Riley, who has observed that Dermestes vulpinus passes through seven larval stages.
In the breeding jars, with plenty of food and a constant temperature of from 68° to 78° F., the larvæ cast their 1st skin in from four to nine days, the great majority moulting at seven days. Under the same conditions the 2d skin was cast at from four to seven days, the majority moulting at six days; the 3d skin at from three to six days, the majority moulting at five days; and the 4th skin at from three to six days, the majority moulting at five days; the 5th skin at from five to seven days, and the 6th skin at six days. There are thus seven larval stages. (Report for 1885, p. 260.)
Riley has ascertained that by rearing isolated larvæ of Tenebrio molitor, one after being kept nearly a year had moulted 11 times, when it died. A second larva, hatched June 5, had moulted 12 times by June 10 of the following year, (1877), when it also died. Of T. obscurus three larvæ were reared to the imago state. One moulted 11 times by Aug. 30 of the same year, pupated Jan. 20, 1877, and finally became a beetle Feb. 7, 1877. The other two both moulted 12 times, and reached the imago stage Feb. 18 and March 9, respectively. “All were, as nearly as possible, under like conditions of food and surroundings, and in all cases the moult that gave the pupa is not considered among the larval moults.”
Two larvæ of the museum pest (Trogoderma tarsale) were kept by Riley in a tight tin box with an old silkworm cocoon. “They were half-grown when placed in the box. On Nov. 8, 1880, there were in the box 28 larva skins, all very much of a size, the larva having apparently grown but little. The skins were removed and the box closed again as tightly as possible. Recently, or after a lapse of two years, the box was again opened and we found one of the larvæ dead and shrivelled up; but the other was living and apparently not changed in appearance. There were 15 larva skins in the box. He could not tell when the one larva died, but it is certain that within a little more than three and a half years, two larvæ shed not less than 43 skins, and that one larva did not, during that time, appreciably increase in size. We know of no observations which indicate the normal or average length of life, or number of moults in either Tenebrio or Trogoderma, but it is safe to assume from what is known, in these respects, of allied species, that in both the instances here referred to, but particularly in the case of Trogoderma, development was retarded by insufficient nutrition, and that the frequent moulting and slow growth resulted therefrom, and were correlated.”[99] Further observations such as these are greatly needed.
Of the Siphonaptera the common cat and dog flea (Pulex serraticeps) moults three times before pupating. (Howard.)
In Lepidoptera the usual or average number of moults is four, but the number varies considerably, the greatest number yet known occurring in Phyrrarctia isabella, which, Dr. Dyar informs me, moults 10 times.
From Dyar’s observations it appears that there are usually five 618larval stages, but six and seven stages are not infrequent, while there are seven in Seirarctia echo, eight in Ecpantheria scribonia, Scepis, and Apatelodes, and nine and ten in arctians, while the European Nola centonalis moults nine times, other species of this genus shedding their skins six times. (Buckler.) (Psyche, v, pp. 420–422.) Callosamia promethea appears, as a rule, to moult but three times. Orgyia antiqua was found by Hellins to moult from three to five times. Riley found that in O. leucostigma the males moult four times, the female four, but sometimes five times, while Dyar states that in O. gulosa the male larvæ moult three or four times, the female always four times; in O. antiqua, however, there are six stages, and in the female seven. Lithocolletis, Chambers thinks, as a rule, moults eight times, and Comstock thinks that L. hamadryadella casts its skin seven or eight times.
In the blow-fly (Calliphora) Leuckart and Weismann have inferred at least two moults, while Weismann suspected that there are as many as four. In Musca domestica we have observed that the larva moults three times; in Œstridæ there are three larval stadia. (Brauer.) In Corethra there are four larval moults, and Miall thinks there are probably as many in Chironomus. Passing to the phytophagous Hymenoptera, there are three moults or four larval stages in Nematus erichsonii, but Dyar informs us that less than four stages in saw-fly larvæ is very rare, that he has only one record of less than five, and that that is doubtful; “five for nematid, six and seven for others, is certainly the rule. The highest I have is the indication of 11 stages for Harpiphorus varianus, but this again is an inference only, and attended with doubt.” (Can. Ent., xxvii, p. 208.) In Bombus we have observed five different sizes of larvæ, and hence suppose the least number of ecdyses is five, while we are disposed to believe that this insect, as well as wasps and bees, in general shed their skins as many as eight times during their entire existence.
The honey-bee, Cheshire thinks, since he has found the old and ruptured pellicles, probably moults six times before it spins its cocoon, or passes into the semipupa condition. (Bees and Bee-keeping, p. 20.)
As to the cause of the great number of moults in the arctians and in the beetles experimented with by Riley, it would seem that cold and the lack of food during hibernation were the agents in arctians, and starvation or the lack of food in the case of the beetles, such cause preventing growth, though the hypodermis-cells retained their activity.
619Reproduction of lost limbs.—Here might be discussed the subject of the renovation or renewal of maimed or lost limbs, or the reparation of other injuries. As is well known, the cœlenterates, echinoderms, and worms under certain circumstances multiply by self-division, or if artificially mutilated, the parts are gradually restored by cell-proliferation or histogenesis. It is so with the antennæ and legs of crustaceans as well as the digits and tail of salamanders. The experiments first made by Le Pelletier[100] on spiders, and later by Heineken,[101] and others after him, on different spiders, as well as on Orthoptera and Hemiptera (Blatta, Reduvius, etc.), have proved that antennæ and legs and other external parts which have been injured or shortened, or entirely cut off in young individuals, are replaced at the next, or after successive moults, though generally in diminished size. This does not usually occur in adult life, and the process of reparation of lost parts is apparently due to the active growth of the cells of the parts affected during the process of moulting, when the histolysis of the maimed or diseased parts is succeeded by the rapid development of new cells, not only of the hypodermis, but also of the more specialized tissues within. And this tends to prove that such histolysis and making over of the muscles and other structures within occur especially in all metamorphic insects, and also in ametabolous forms, though the process has been most thoroughly examined in the Diptera, where these changes are more marked.
Gonin has found that the thoracic legs of the caterpillar correspond only to the tarsi of the imago (Fig. 608). It results, he says, from this fact that in accordance with the observations of Réaumur (which were wrongly interpreted by Newport and Künckel D’Herculais) that the amputation of the legs of the larva does not involve the entire leg, but only the extremity of the leg of the imago.
Formation of the cocoon.—While the larvæ of many insects, as those of the butterflies, suspend themselves before transforming, and spin no cocoon, or dig into the earth for protection and to secure an immunity from too great changes of temperature, a large proportion of the larvæ of metabolous insects which lead an inactive pupal life, line their earthen cells with silk, or spin a more or less elaborate case of silk, called the cocoon. We have seen that the inactive pupa of the male scale-insects is covered by the scale itself, or even in one case the insect forms a true cocoon of fibres of wax. The aquatic 620larvæ of the Neuroptera and Coleoptera creep out of the water, and by the movements of their bodies make a rude earthen cell in the bank, while that of Donacia spins a dense, leathery cocoon (Fig. 567) in the earth. The larvæ of the Embiidæ are protected by a cocoon, which they renew at each moult. Coniopteryx spins an orbicular cocoon, the Hemerobiidæ a spherical, dense, whitish one. The Trichoptera transform within their larval cases, which thus serve as cocoons, as do certain case-bearing Lepidoptera, notably the Psychidæ.
Fig. 567.—Cocoon (natural size) of Donacia proxima.
Fig. 568.—Cocoon and larva of Lucanus dama.
The pupa of certain leaf-eating beetles (Chrysomelidæ), as well as the Coccinellidæ, Dermestidæ, Hister, etc., are usually protected by the cast larval skin, which is retained, forming a rude shelter. While many beetles spin an oval cocoon (Gyrinus, Silphidæ), the wood-boring species make one of chips glued together, and that of Lucanus, which feeds on decayed wood, is lined with silk (Fig. 568). Anobium constructs a silken cocoon, interweaving the fine particles of its thin castings; the larvæ of weevils also usually spin silken cocoons.
Fig. 569.—Larva (a), puparium (b), and imago (c) of Sarcophaga, enlarged.

Fig. 570.—a, Erax bastardi; b, pupa.—After Riley.
The larval skin of the coarctate Diptera is retained as a protection for the soft-bodied pupa within, the old larval skin separating from 621the integument of the semipupa. To this cocoon-like covering of the coarctate pupa we have restricted the term puparium, originally used by Kirby and Spence to designate the pupa. The puparium is usually cylindrical or barrel-shaped, rounded at each end.
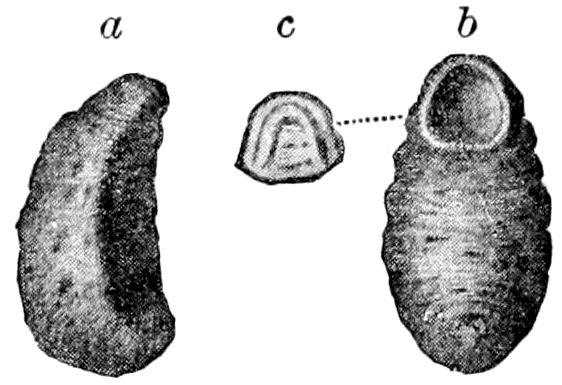
Fig. 571.—Puparium of Hypoderma bovis: a, side; b, ventral view, showing exit hole of adult; c, cap which splits off for exit of fly.—After Clark, from Osborn, Bull. 5, Div. Ent. U. S. Dept. Agr.
In the Diptera cyclorhapha, or common house and flesh flies, etc., the puparium remains in vital connection, by means of four tracheæ, with the enclosed pupa, which escapes from the case through a curved seam or lid at the anterior end and not by a slit in the back, as do the orthoraphous families, represented by the horse-fly (Tabanidæ, Asilidæ, Fig. 570), etc., where in some cases the obtected pupa remains within the loose envelope formed by the old larval skin, which Brauer calls a false puparium. The dry, hard puparium is burst open at the cephalic end when the fly emerges, by means of the frontal vesicle, which is distended with fluid (Fig. 571).
The exact mode of spinning the cocoon by caterpillars has been carefully observed by L. Trouvelot in the case of the polyphemus silkworm.
“When fully grown, the worm, which has been devouring the leaves so voraciously, becomes restless and crawls about the branches in search of a suitable place to build up its cocoon; before this it is motionless for some time, holding on to the twig with its front legs, while the two hind pair are detached; in this position it remains for some time, evacuating the contents of the alimentary canal until finally a gelatinous, transparent, very caustic fluid, looking like albumen, or the white of an egg, is ejected; this is a preparation for the long catalepsy that the worm is about to fall into. It now feels with its head in all directions, to discover any leaves to which to attach the fibres that are to give form to the cocoon. If it finds the place suitable, it begins to wind a layer of silk around a twig, then a fibre is attached to a leaf near by, and by many times doubling this fibre and making it shorter every time, the leaf is made to approach the twig at the distance necessary to build the cocoon; two or three leaves are disposed like this one, and then fibres are spread between them in all directions, and soon the ovoid form of the cocoon distinctly appears. This seems to be the most difficult feat for the worm to accomplish, as after this the work is simply mechanical, the cocoon being made of regular layers of silk united by a gummy substance. The silk is distributed in zigzag lines of about one-eighth of an inch long. When the cocoon is made, the worm will have moved his head to and fro, in order to distribute the silk, about 254,000 times.
“After about half a day’s work, the cocoon is so far completed that the worm can hardly be distinguished through the fine texture of the wall; then a gummy resinous substance, sometimes of a light-brown color, is spread all over the inside of the cocoon. The larva continues to work for four or five days, hardly taking 622a few minutes of rest, and finally another coating is spun in the interior, when the cocoon is all finished and completely air tight. The fibre diminishes in thickness as the completion of the cocoon advances, so that the last internal coating is not half so thick and so strong as the outside ones.” (Amer. Naturalist, i, p. 86.)
The mode of spinning the cocoon of an ichneumon (Microgaster) parasitic on Philampelus has been well described by John P. Marshall, as follows:—
Fig. 572.—Microgaster larvæ; spinning their cocoons: a, enlarged view of 5.—After Marshall.
The first appearance of the parasite is represented in Fig. 572, 1. A warty excrescence appears on the back of the caterpillar, which slowly emerges until it is seen to be a larva enclosed in a delicate transparent membrane, as represented in 2. This it soon succeeds in bursting, and, rising to its full length, balances itself a moment as in 3, then, bending double, it ejects from its mouth a glairy liquid, which instantly changes to silk, and fastens the posterior end to the skin of the caterpillar, as shown in 4, side view. It now begins to spin its cocoon by attaching a silken thread to the silky mass by which it had previously fastened itself to the caterpillar, and forming a series of loops of uniform size, first from right to left, and then back again from left to right, as represented in the front view, 5, and better in the enlarged view, 5a, the arrow heads showing the direction in which the head of the larva moved while forming the loops. The ends of the series, numbered 1, 2, 3, 4, are fastened to the edges of the ventral side of the body, which thus serves as a measure of the width of the cocoon, and also acts as a support for the frail fabric in the first stages of spinning. After the larva has fastened the fabric as far up on its ventral surface as it can, conveniently, it then begins to spin free, as shown in the side view, 6, where it is represented as just completing the first half of its cocoon, which 623resembles in form a slipper. This accomplished, the larva ceases to spin for the time being, bends its head, as in 7, towards its ventral surface, and pushes the half cocoon free from its body. The form of the silken fabric enables it to stand unsupported, while the larva, sliding its head down to the base, holds on firmly until it swings its posterior end into the toe of the slipper.
Figure 572, 8, shows it in the act of changing end for end, and in 9 the larva is seen erect, beginning at the base to complete the other half of its cocoon; 10 shows the larva contracting its body as it spins upward for about half the length of the cocoon, when it again changes end for end, as shown in 11, where it is beginning at the upper part to unite the two sides, finally enclosing itself as represented in 12.
It may now be seen, under the microscope, through the meshes of its cocoon actively engaged in lining the interior with layers of very fine silk ejected from its mouth in great abundance. One half of the cocoon is first lined by a forward and back movement of its head, and then reversing its position, it lines the other half in a similar manner.
In one case the larva was disengaged from the skin of the caterpillar, after beginning its cocoon. It, however, began again, and spun a portion while lying on the table. This was removed, when it began a third time, and completed its cocoon.
In about 10 days the insect made its appearance through a hole in the upper end, as represented in 13. The top was eaten off in a perfect circle and hung by a few threads, so as to resemble a lid as it was thrown back.
One caterpillar observed had between 300 and 400 cocoons on its back and sides, and another was dissected after more than 30 larvæ had escaped, and 130 were discovered in the soft integuments of the back.
The figures from 1 to 13 are magnified five diameters, but in order to observe the spinning of the cocoon a power of 50 is required. (Amer. Naturalist, xii, pp. 559, 560.)
Certain differences observed by W. A. Buckhout in a Microgaster parasitic on the different species of Macrosila, are referred to in the same volume, p. 752.
Fig. 573.—Body of larva of Lithocolletis. swollen and filled with cocoons of Copidosoma, enlarged.
While those chalcidid larvæ which feed internally on their host, as a rule, transform into naked, more or less coarctate pupæ, Howard states that the larvæ of Copidosoma, Bothriothorax, Homalotylus, and perhaps others, which are much crowded within their host, cause a marked inflation of the body of the latter (Figs. 573, 574). The nature of this cocoon-like cell, and how it is produced, is unknown. “Its structure shows it not to be silk, nor yet the last larval skin of the parasite, and whether it is an adventitious tissue of the host-larva or a secretion of the parasite, or is explicable upon other grounds, I cannot say.”
The silken cocoon of an aphidiid ichneumon has been found by Miss Murtfeldt, and also by Dr. Riley, under a rose aphid in which it had lived, and referred by Howard to the genus Praon (Fig. 575).
Sanitary conditions observed by the honey-bee larva, and admission of air within the cocoon.—Cheshire has observed that after the larva of the honey-bee has spun its cocoon or silken lining of its cell, it 624observes the following means of preserving cleanliness. The food given to the larva, especially during the latter part of the growing period, contains much pollen, the cases of the grains of which consist of cellulose, which is indigestible.
Fig. 574.—Coccinellid larva infested by Homalotylus obscurus, enlarged.
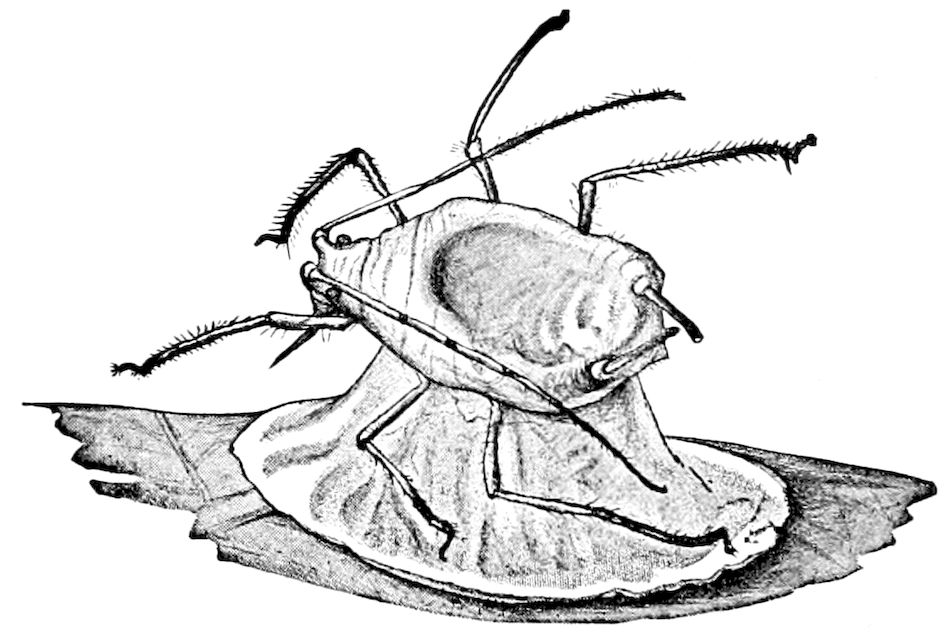
Fig. 575.—Cocoon of Praon under the body of a dead Aphis, enlarged.—This and Figs. 573 and 574 after Howard, from Insect Life.
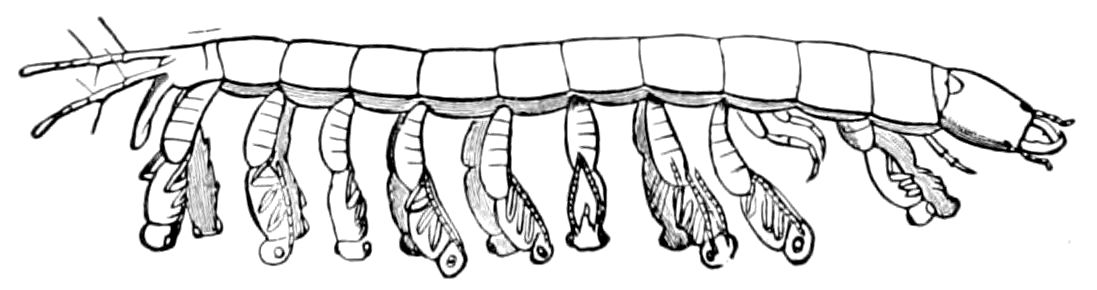
Fig. 576.—Pupation of Proctotrupes in the body of a larva of a beetle, representing a case mentioned by Dr. Sharp, where the parasites have pupated on the outside of the host, a pair of each attached to nearly each segment of the body of their host.—After Sharp.
“These cases, with other refuse matters, collect in quantity within the bowel, which becomes distended, since it has no opening. The imprisoned larva, having little more than enough room for turning, must be freed of these objectionable residua.... In a word, the larva turns its head upon its stomach, and pushes the former towards the base of the cell until its position is reversed, the tail being outwards; and, thus placed, it laps up all residue of food, especially from 625its old clothes previously referred to, until they are dried, and practically occupy no space. It now throws up its stomach and bowel, with all their contents, and without detaching them from its outer skin, which is moulted as before, but in this instance to be pressed against the cell, so as to form for it an interior lining. The dejectamenta of the bowel in this way lie between the cast skin and cell-wall (as seen at e, Fig. 577), and so the larva remains absolutely unsoiled. It now turns its head and resumes its old position, joining its cocoon to the edges of its last cast skin, so that its habitation is relined, it is cleansed, and air can still pass to it through the imperceptible openings left by the bees in the sealing. This point is of radical importance, since breathing is carried on pretty rapidly during the latter part of its subsequent transformations, the absorbed oxygen permitting then of a production of heat, and causing also considerable diminution in weight.”
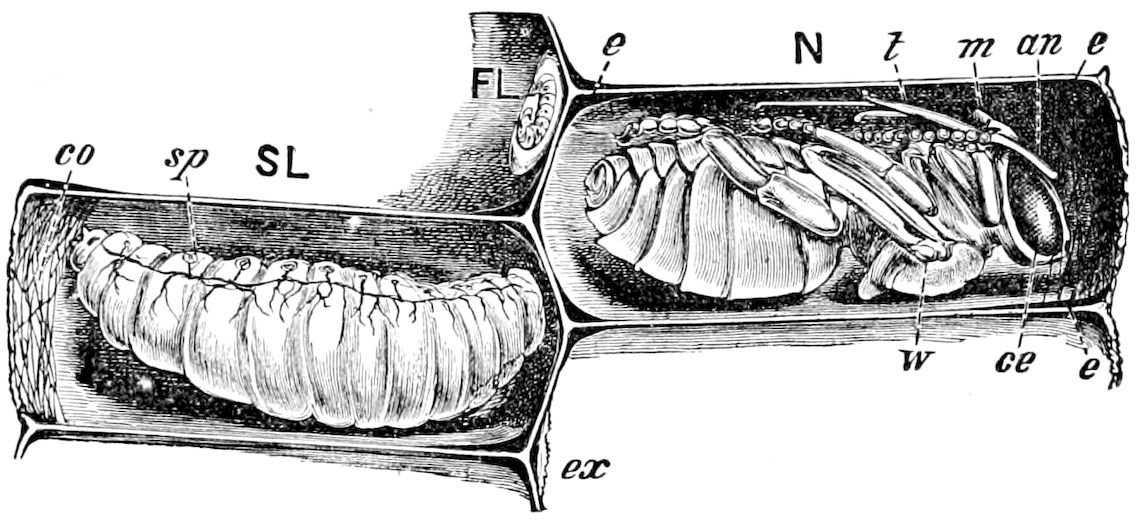
Fig. 577.—Larva and pupa of honey-bee in their cell: SL, spinning-larva; N, pupa; FL, young feeding larva; co, cocoon; sp, spiracles; t, tongue; m, mandible; an, antenna; w, wing; ce, compound eye; e, excrement; ex, exuvium.—After Cheshire.
As to the passage of air into the bee’s cocoon, Cheshire states that before the cocoon can be built, a cover, technically called sealing, is put over the larva by its nurses. These covers are made of pollen and wax, and are pervious to the air. They are more convex and regular in form than those sealing in the honey.[102]
THE PUPA STATE
The word pupa is from the Latin meaning baby. Linnæus gave it this name from its resemblance to a baby which has been swathed or bound up, as is still the custom in Southern Europe. The term pupa should be restricted to the resting inactive stage of the holometabolous insects.
Lamarck’s term chrysalis was applied to the complete or obtected pupa of Lepidoptera and of certain Diptera, and mumia, a mummy, to the pupæ of Coleoptera, Trichoptera, and most Hymenoptera. Latreille (1830) also restricted 626the term pupa to the “oviform nymph,” or puparium, of Diptera. Brauer applies the term nymph to the pupa of metabolous insects.
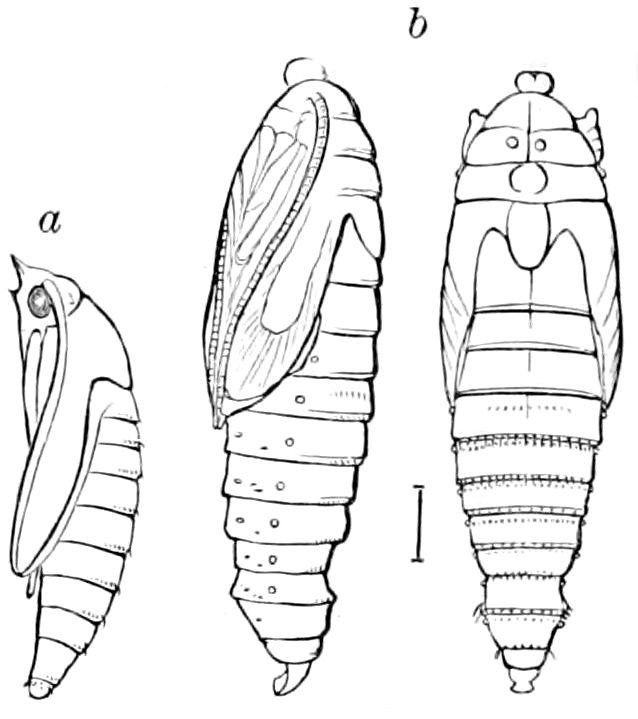
Fig. 578.—Pupa obtecta: a, of Sesia, with its cocoon-cutter on the head; b, of Tortrix vacciniivorana.
The typical pupa is that of a moth or butterfly, popularly called a chrysalis. A lepidopterous pupa in which the appendages are more or less folded close to the body and soldered to the integument, was called by Linnæus a pupa obtecta; and when the limbs are free, as in Neuroptera, Mecoptera, Trichoptera, and the lepidopterous genus Micropteryx it is called a pupa libera (Fig. 579). When the pupa is enclosed in the old larval skin, which forms a pupal covering (puparium), the pupa was said by Linnæus to be coarctate. The pupa of certain Diptera, as that of the orthoraphous families, is nearly as much obtected as that of the tineoid families of moths, especially as regards the appendages of the head; the legs being more as in pupæ liberæ (Fig. 580).
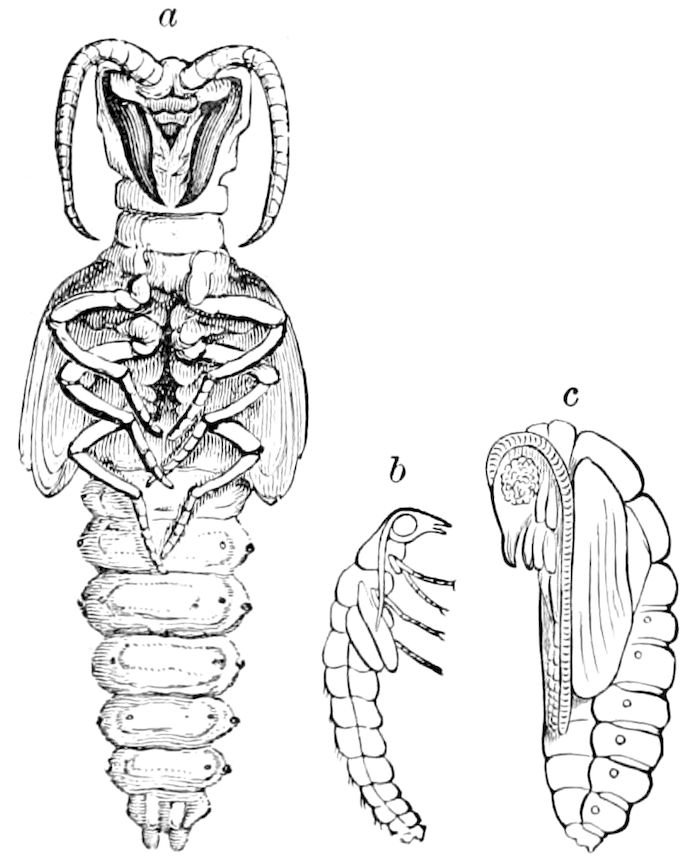
Fig. 579.—Pupa libera of neuropterous insects a, Corydalus cornutus; b, Sialis; c, Hemerobius.
The male Coccid anticipates the metabolous insects in passing through a quiescent state, when, as Westwood states, it is “covered by the skin of the larva, or by an additional pellicle.” The body appears to be broad and flat, the antennæ and fore legs resting under the head, while the two hinder pairs of legs are appressed to the under side of the body. There is but a slight approach to the pupa libera of a metabolous insect.
Riley states that the male larva of Icerya purchasi forms a cocoon waxy in character, but lighter, more flossy, and less adhesive than that of the female egg-cocoon. It melts and disappears when heated, proving its entirely waxy nature. When the mass has reached the proper length, the larva casts its skin, which remains in the hind end of the cocoon, and pushes itself forward into the middle of the cocoon. The pupa (Fig. 581) is of the same general form and size as the larva. All the limbs are free and slightly movable, so that they vary in position, though ordinarily the antennæ are pressed close to the side, 627as are the wing-pads; the front pair of legs are extended forward. “If disturbed, they twist and bend their bodies quite vigorously.” The pupa state lasts two or three weeks. A similar pupa is that of Icerya rosæ. (Riley and Howard.)
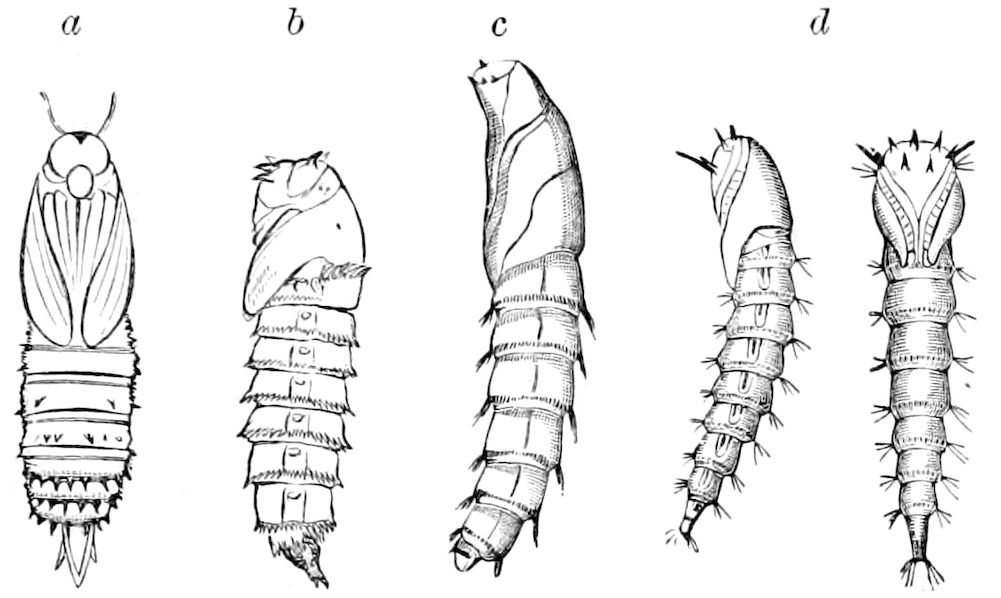
Fig. 580.—Pupa obtecta of Diptera: a, Ptychoptera; b, Tabanus atratus; c, Proctacanthus philadelphicus; d, Midas clavatus.
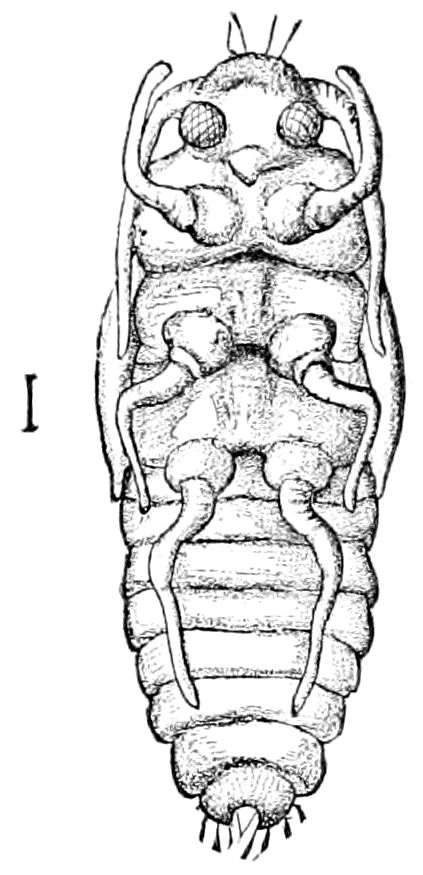
Fig. 581.—Pupa libera of Icerya purchasi, ventral view.—After Riley, Insect Life.
The metamorphosis of Aspidiotus perniciosus is of interest. The male nymph differs much after the first moult from the female, having large purple eyes, while the female nymph loses its eyes entirely. It passes into what Riley terms the pro-pupa (Fig. 582, b), in which the wing-pads are present, while the limbs are short and thick. The next stage is the “true pupa” (Fig. 582, c, d), in which the antennæ and legs are much longer than before. There is no waxy cocoon, but only a case or scale composed of the shed larval skin, i.e. “with the first moult the shed larval skin is retained beneath the scale, as in the case of the female; with the later moultings the shed skins are pushed out from beneath the scale,” and when they transform into the imago they “back out from the rear end of their scale.”
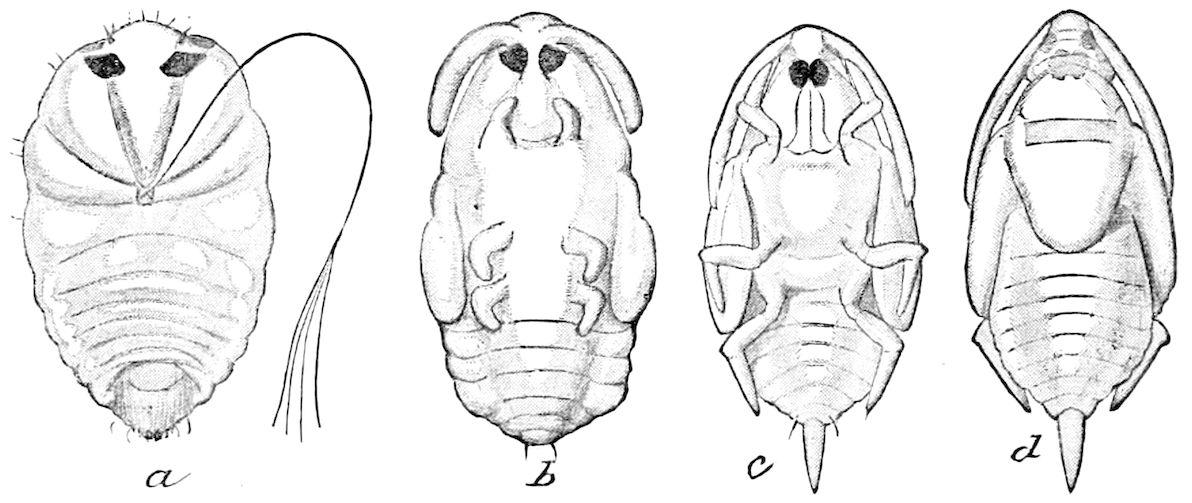
Fig. 582.—Aspidiotus perniciosus, development of male insect: a, ventral view of larva after first moult; b, the same, after second moult (pro-pupa stage); c and d, true pupa, ventral and dorsal views. All greatly enlarged.—After Riley.
628The pupæ of Coleoptera and of Hymenoptera, though there is, apparently, no near relationship between these two orders, are much alike in shape, and, as Chapman pertinently suggests, those of both orders are helpless from their quiescence, and hence have resorted for protection to some cocoon or cell.
But it is quite otherwise with the pupæ of Lepidoptera and Diptera, which vary so much in adaptation to their surroundings, and hence afford important taxonomical and phylogenetic characters. This, as regards the Lepidoptera, was almost wholly overlooked until Chapman called attention to the subject, and showed that the pupæ had characters of their own, of the greatest service in working out the classification, and hence the phylogeny, of the different lepidopterous groups. We have, following the lead of Chapman, found the most striking confirmation of his views, and applied our present knowledge of pupal structures to dividing the haustellate Lepidoptera into two groups,—Paleolepidoptera and Neolepidoptera.
The pupæ of the Neuroptera, Coleoptera, and Hymenoptera differ structurally from the imago, in the parts of the head and thorax being less differentiated. Thus in the head the limits or sutures between the epicranium and clypeus, and the occiput and gula, are obscurely marked, while the tergal and pleural sclerites of the imago are not well differentiated until the changes occurring just before the final ecdysis.
It is easy, however, to homologize the appendages of the pupæ with those of the imago of all the holometabolous orders except in the case of the obtected pupa of the Lepidoptera (and probably of the obtected dipterous pupæ), where the cephalic appendages are soldered together.
That the appendages of the lepidopterous pupa are, as generally supposed, merely cases for those of the imago has been shown by Poulton to be quite erroneous. He says: “If we examine a section of a pupal antenna or leg (in Lepidoptera), we shall find that there is no trace of the corresponding imaginal organ until shortly before the emergence of the imago. In the numerous species with a long pupal period, the formation of imaginal appendages within those of the pupa is deferred until very late, and then takes place rapidly in the lapse of a few weeks. This also strengthens the conclusion that such pupal appendages are not mere cases for the parts of the imago, inasmuch as these latter are only contained within them for a very small proportion of the whole pupal period.” On the other hand, Miall and Hammond claim that there is a strong superficial contrast as to the formation of the imaginal organs, between Lepidoptera 629and tipularian Diptera, the appendages, wings, and compound eyes being substantially those of the imago. “With the exception of the prothoracic respiratory appendages and the tail-fin, there is little in the pupa of Chironomus which does not relate to the next stage.”
The exact homology of the “glazed eye” of the lepidopterous pupæ and of the parts under the head, situated over the maxillæ, is difficult to decide upon, and these points need farther examination. In the dipterous pupa it is interesting to observe that the halteres are large and broad, which plainly indicates that they are modified hind wings. The number and arrangement of the spiracles is different in pupæ from those of the larva and imago.
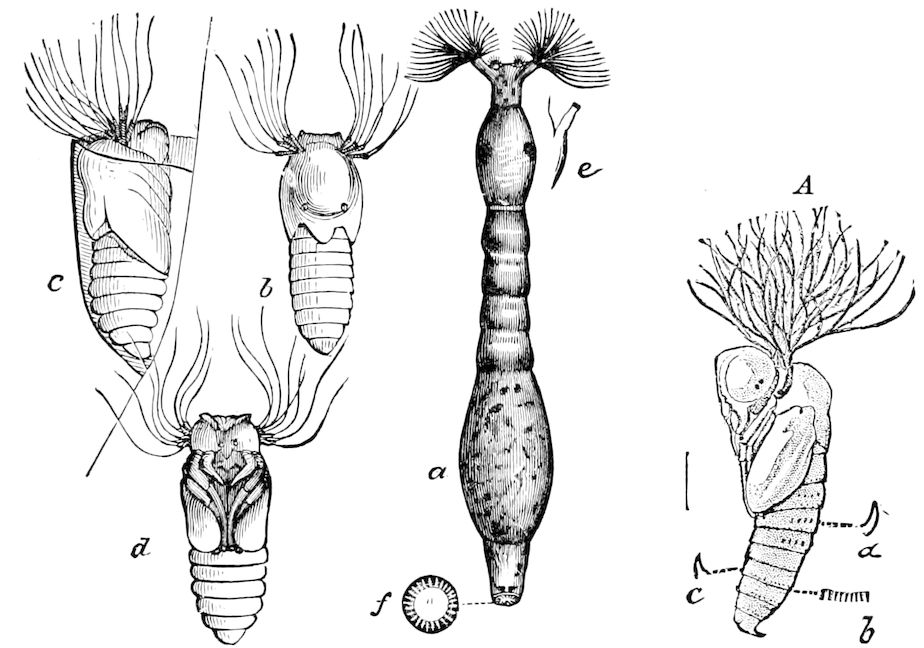
Fig. 583.—Simulium piscicidium: a, larva; b, c, d, pupa; e, thoracic leg; f, row of bristles at end of body. A, S. pecuarum, pupa; a, b, c, adminicula.—After Riley.
There are also secondary adaptive structures peculiar to the pupa, which are present and only of use in this stage. These are the thoracic, spiracular, or breathing appendages of the aquatic Diptera (Fig. 583), the various spines situated on the head or thorax, or on the sides, or more often at the end of the abdomen, besides also the little spines arranged in more or less circular rows around the abdominal segments, the cocoon-breaker, and the cremaster of many pupæ.
In the pupa of certain Diptera, there is a terminal cremaster-like spine, as in that of Tipula eluta (Fig. 584), Tabanus lineola (Fig. 585), besides adminicula or locomotive spines like those of lepidopterous pupæ (Fig. 580, a, b, c).
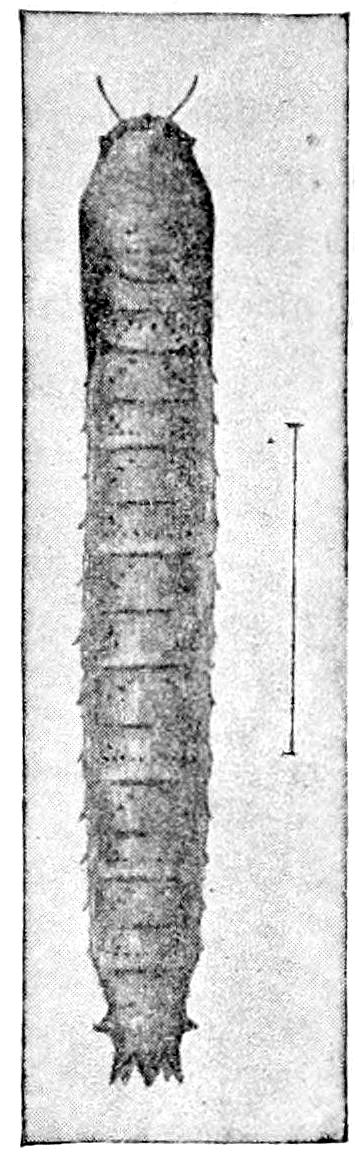
Fig. 584.—Pupa of Tipula eluta.

Fig. 585.—Pupa of Tabanus lineola.—This and Fig. 584 after Hart.
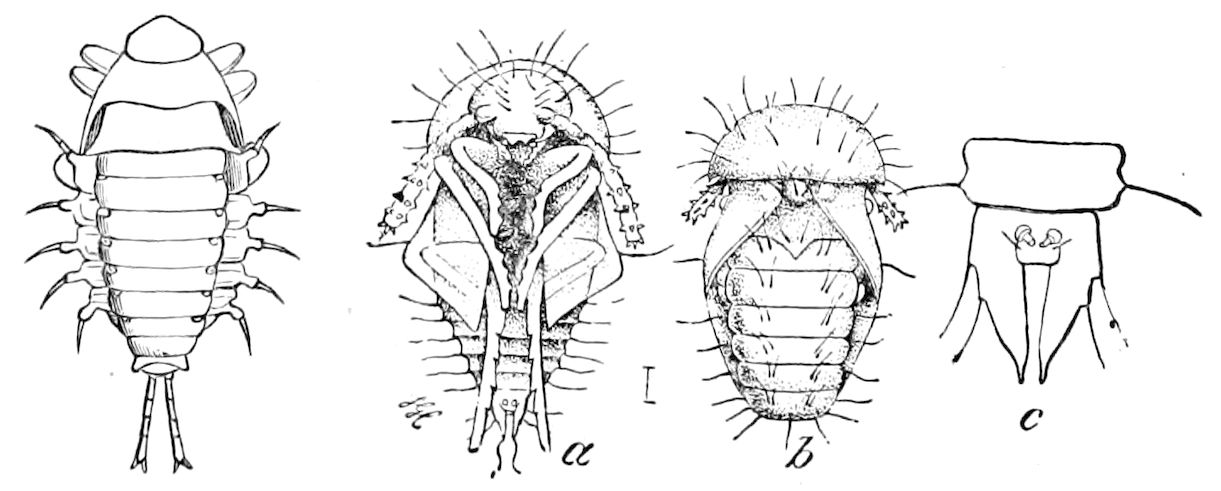
Fig. 586.—Pupa of Galerita lecontei, and of Adelops hirtus (a, b, c).—After Hubbard.
The pupæ of Coleoptera are variously spined or hairy (Fig. 586). Those of Hydrophilus and of Hydrobius are provided with stout spines on the prothorax and abdomen which support the body in its cells, so that, as Lyonet first showed, though surrounded on all sides by moist earth, it is kept from contact with it by the pupal spines; other pupæ of beetles, such as that of the plum weevil, which is also subterranean, possess similar spines. The abdomen of many coleopterous pupæ, such as those of Carabidæ, end in two spines, to aid them in escaping from their cells in wood or in the earth; others have stiff bristles, and others spines along each side of the abdomen (Fig. 586). All these structures are the result of a certain amount of activity in what we call quiescent pupæ, but most of these are for use at the end of pupal life, at the critical moment when by their aid the insect escapes from its cocoon or subterranean cell, or if parasitic, bores out of its host.
If we are to account for the causes of their origin, we are obliged to infer that they are temporary deciduous structures due to the need of support while the body is subjected to unusual strains and stresses in working its way out of its prison in the earth, or its cell within the stems and trunks of plants and similar situations. They are pupal inheritances or heirlooms, and well illustrate the inheritance of characters acquired during a certain definite, 631usually brief, period of life, and transmitted by the action of synchronous heredity.
The pupæ of certain insects are quite active, thus that of Raphidia, unlike that of Sialis, before its final ecdysis regains its activity and is able to run about. (Sharp, p. 448.)
a. The pupa considered in reference to its adaptation to its surroundings and its relation to phylogeny
The form of the pupa is a very variable one, as even in Lepidoptera it is not entirely easy to draw the line between a pupa libera and a pupa obtecta (Fig. 578); and though the period is one of inactivity, yet when they are not in cocoons or in the earth in subterranean cells, their form is more or less variable and adapted to changes in their surroundings. Even in the obtected pupa of butterflies, there is, as every one knows, considerable variability of shape and of armature, which seems to be in direct adaptability to the nature of their environment. Scudder has well shown that in certain chrysalids, such as those of the Nymphalidæ, which are variously tuberculated, and hang suspended by the tail, and often hibernate, these projections serve to protect the body. All chrysalids with projections or ridges on different parts of the body, being otherwise unprotected, move freely when struck by gusts of wind, hence “the greater the danger to the chrysalis from surrounding objects, the greater its protection by horny tubercles and roughened callous ridges.” The greater the protection possessed in other ways, as by firm swathing or a safe retreat, the smoother the surface of the body and the more regular and rounded its contours. The tendency to protection by tubercles is especially noticeable in certain South American chrysalids of nymphalid butterflies. This response to the stimuli of blows or shocks is also accompanied by a sensitiveness to the stimulus of too strong light.
Previously Scudder[103] had made the important suggestion that the smooth crescent-shaped belt of the “glazed eye” or “eyepiece” of chrysalids is, as an external covering of the eye, midway between that of the caterpillar and the perfect insect, and he asks: “May it not be a relic of the past, the external organ of what once was? 632And are we to look upon this as our hint that the archaic butterfly in its transformations passed through an active pupal stage, like the lowest insect of to-day, when its limbs were unsheathed, its appetite unabated?” etc. Scudder also shows that “the expanded base of the sheath covering the tongue affords protection also to the palpi which lie beneath and beside the tongue.”
All this tends to show the importance of studying the structure of the pupa, in order to ascertain how the pupal structures have been brought about, with the final object of discovering whether the pupæ of the holometabolic insects are not descended from active nymphs, and if so, the probable course of the line of descent.
b. Mode of escape of the pupa from its cocoon
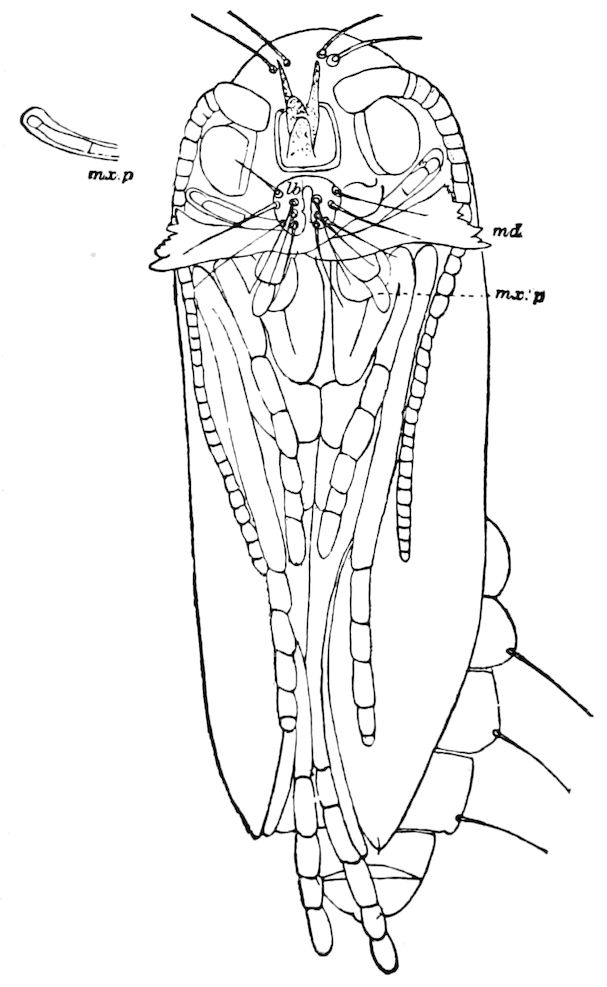
Fig. 587.—Pupa of Micropteryx purpuriella, front view: md, mandibles; mx. p, maxillary palpus, end drawn separately; mx.′ p, labial palpi; lb, labrum.
“In all protected pupæ,” as Chapman says, “the problem has to be faced, how is the imago to free itself from the cocoon or other envelope protecting the pupa.” In the Coleoptera and Hymenoptera the imago becomes perfected within the cocoon or cell, as the case may be, and as Chapman states, “not only throws off the pupal skin within the cocoon, but remains there till its appendages have become fully expanded and completely hardened, and then the mandibles are used to force an outlet of escape,” and he calls attention to the fact that “in many cases, even in some entire families, they are of no use whatever to the imago except in this one particular,” and he cites the Cynipidæ as perhaps the most striking instance of this circumstance.
In those Neuroptera which spin a silken cocoon, e.g. the Hemerobiidæ, the Trichoptera, 633and in Micropteryx (Fig. 588), the jaws used by the pupa for cutting its way out of the cocoon are even larger in proportion than in the pupa of caddis-flies (Fig. 588), being of extraordinary size.
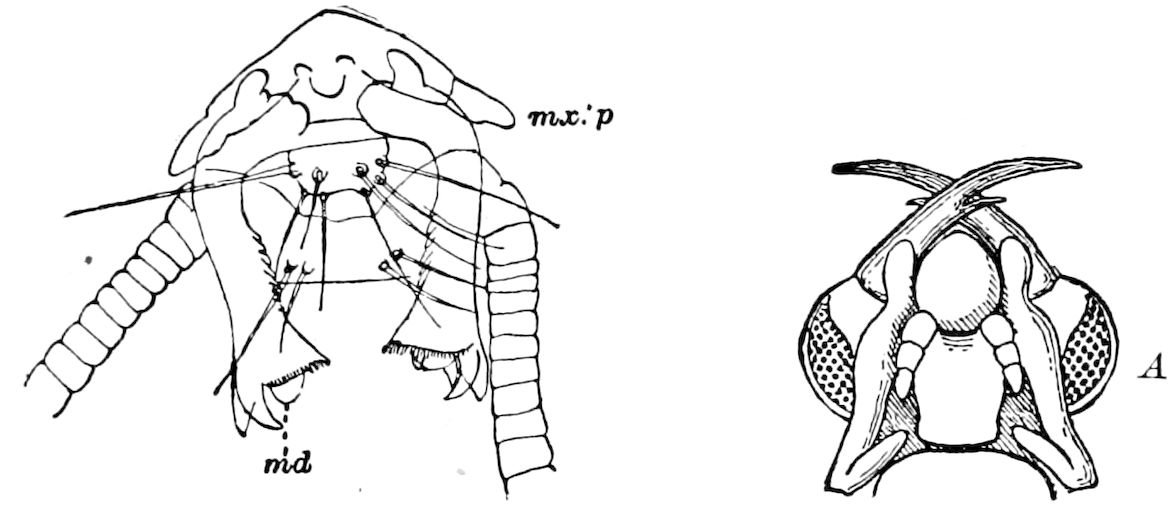
Fig. 588.—Mandibles (md) of Micropteryx purpuriella, enlarged.—Author del. A, pupal head of a hydropsychid caddis-fly, showing the large mandibles.—After Reaumur, from Miall.
In Myrmeleon the pupa pushes its way half out of the cocoon, and then remains, while the imago ruptures the skin and escapes (Fig. 589, a).
Thus in the Neuroptera and Trichoptera we have already established the more fundamental methods of escape from the cocoon, which we see carried out in various ways in the more generalized or primitive Lepidoptera.
The most primitive method in the Lepidoptera of escaping from the cocoon seems to be that of Micropteryx.
Fig. 589.—Larva of Myrmeleon with (a) its cocoon and cast pupa-skin.
“In this genus,” says Chapman, “though it is nominally the pupa that escapes from the cocoon, it is in reality still the imago, the imago clothed in the effete pupal skin. To rupture the cocoon it uses not its own jaws, but those of the pupal skin, energizing them, however, in some totally different way from ordinary direct muscular action, their movements being the result of the vermicular movements of the pupa, acting probably by fluid pressure on the articular structure of the jaws, by some arrangement not altogether different perhaps from the frontal sac of the higher Diptera. In the Micropteryges the jaws of the pupa not only rupture the cocoon, but appear to be the most active agents in dragging the pupa through the opening in the cocoon and through any superincumbent earth, being merely assisted by the vermicular action of the abdominal 634segments, and we find in accordance with this circumstance that the pupal envelope is still very thin and delicate, and has little or no hardening or roughness by which to obtain a leverage against the walls of the channel of escape.” (Trans. Ent. Soc. London, 1896, pp. 570, 571.)
Fig. 590.—Pupa of Talæporia: a, cocoon-cutter; with vestiges of four pairs of abdominal legs, and the cremaster.
Some sort of a beak or hard process, more or less developed, according to Chapman, adapted for breaking open the cocoon exists in nearly all the Lepidoptera with incomplete pupæ (pupæ incompletæ), except the limacodid and nepticulid section. “In all these instances the pupa emerges from the cocoon precisely as in the Micropteryges, that is, the moth it really is that emerges, but does so encased in the pupal skin. To achieve this object, it seems to have been found most efficient to have three, four, or five abdominal segments capable of movement, but to have the terminal sections (segments) soldered together.”
This cocoon-breaker, as we may call it, is especially developed in Lithocolletis hamadryadella. As described by Comstock, it forms a toothed crest on the forehead which enables it to pierce or saw through the cocoon.
“Each pupa first sawed through the cocoon near its juncture with the leaf and worked its way through the gap, by means of the minute backward-directed spines upon its back, until it reached the upper cuticle of the leaf. Through this cuticle it sawed in the same way that it did through the cocoon. The hole was in each case just large enough to permit the chrysalis to work its way out, holding it firmly when partly emerged. When half-way out it stopped, and presently the skin split across the back of the neck and down in front along the antennal sheaths, and allowed the moth to emerge.”[104]
We have observed and figured the cocoon-breaker in Bucculatrix, Talæporia (Fig. 590, a), Thyridopteryx, and Œceticus, and rough knobs or slight projection answering the purpose in Hepialidæ, Megalopyge, Zeuzera, and in Datana.[105] See also the spine on the head of Sesia tipuliformis (Fig. 578).
The imago of the attacine moths cuts or saws through its cocoon by means of a pair of large, stout, black spines (sectores coconis), one on each side of the thorax at the base of the fore wings (Fig. 591), and provided with five or six teeth on the cutting edge (C, D).
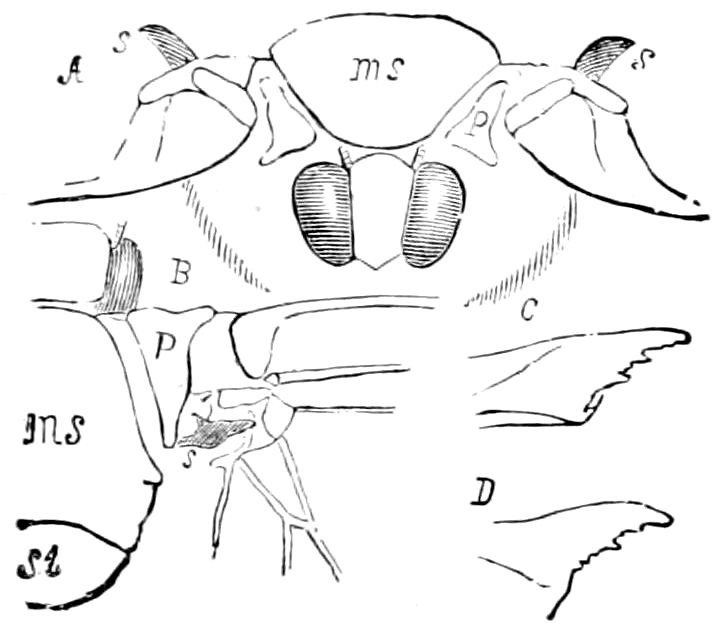
Fig. 591.—Cocoon-cutter of the Luna moth: front view of the moth with the shoulders elevated and the rudimentary wings hanging down: s, cocoon-cutter; p, patagium. B, represents another specimen with fully developed wings: ms, scutum; st, scutellum of the mesothoracic segment; s, cocoon-cutter, which is evidently a modification of one of the pieces at the base of the fore wings; it is surrounded by membrane, allowing free movement. C and D, different views of the spine, magnified, showing the five or six irregular teeth on the cutting edge.
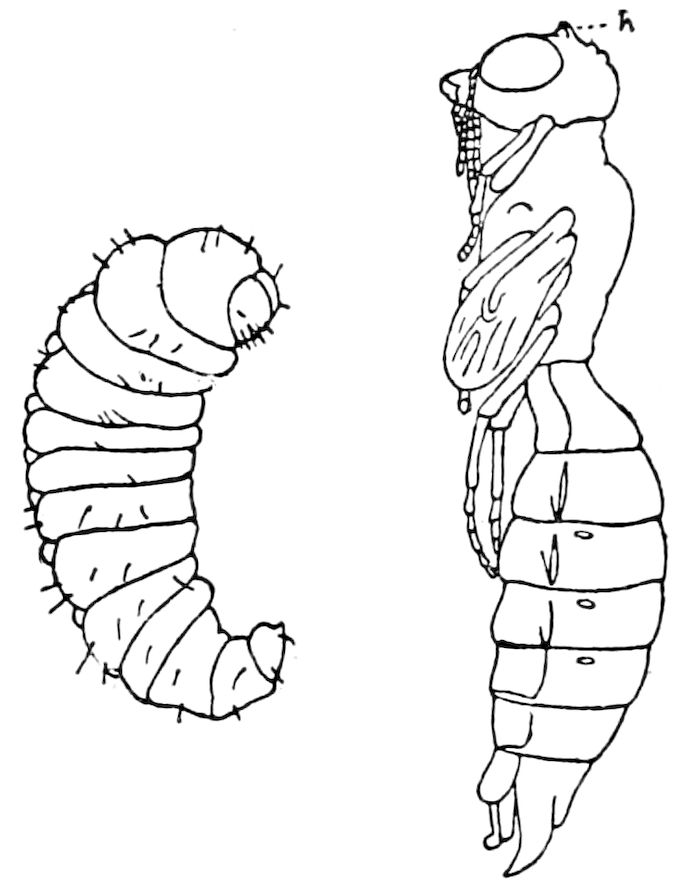
Fig. 592.—Larva and pupa of a wood-wasp (Rhopalum), enlarged: h, temporary locomotive tubercles on head of pupa.—Trouvelot del.
Our attention[106] was drawn to this subject by a rustling, cutting, and tearing noise issuing from a cocoon of Actias luna. On examination a sharp black point was seen moving to and fro, and then another, until both points had cut a rough irregular slit, through which the shoulder of the moth could be seen vigorously moving from side to side. The hole or slit was made in one or two minutes, and the moth worked its way at once out of the slit. The cocoon was perfectly dry. The cocoon-cutter occurs in all the American genera, in Samia cynthia, and is large and well marked in the European Saturnia pavonia-minor and Endromis versicolora. In Bombyx mori the spines are not well marked, and they are quite different from those in the Attaci. There are three sharp points, being acute angles of the pieces at the base of the wing, and it must be these spines which at times perform the cutting through of the threads of the cocoon described by Réaumur, and which he thought was done by the facets of the eyes. It is well known that in order to guard against the moths cutting the threads, silkraisers expose the cocoon to heat sufficient to destroy the enclosed pupa. In Platysamia the cocoon-cutters, though well developed, do not appear to be used at all, and the pupa, like that of the silkworm and other moths protected by a cocoon, moistens the silk threads by a fluid issuing from the mouth, which also moistens the hairs of the head and thorax, together with the antennæ. It remains to be seen whether these structures are only occasionally used, and whether the emission of the fluid is not the usual and normal means of egress of the moth from its cocoon. Dr. Chapman remarks that throughout the obtected moths “there are many devices for breaking through the cocoon: specially constructed weak places in the cocoon, softening fluid, applied by the moth, assisted by special appliances of diverse sorts, such as in Hybocampa[107] and Attacus,” etc.
As to the fluid mentioned above, Trouvelot states that it is secreted during the last few days of the pupa state, and is a dissolvent for the gum so firmly 636uniting the fibres of the cocoon. “This liquid is composed in great part of bombycic acid.” (Amer. Naturalist, i, p. 33.)
The pupa of the dipterous genus Sciara (S. ocellaris O. S.) resembles a tineid pupa, and before transforming emerges for about two-thirds of its length from the cocoon; the pupa-skin remaining firmly attached in this position.[108]
Certain hymenopterous pupæ are provided with temporary deciduous conical processes. Thus we have observed in the pupa of Rhopalum pedicellatum two very prominent acute tubercles between the eyes (h, Fig. 592). As the cocoon is very slight, these may be of use either in extracting itself from the silken threads or in pushing its way along before emerging from the tunnel in the stem of plants. (See also p. 611.)
c. The cremaster
Although this structure is in general confined to lepidopterous pupæ, and is not always present even in them, since it is purely adaptive in its nature, yet on account of its singular mode of development from the larval organs, and the accompanying changes in the pupal abdomen, it should be mentioned in this connection. The cremaster is the stout, triangular, flattened, terminal spine of the abdomen, which aids the pupa in working its way out of the earth when the pupa is subterranean, or in the pupa of silk-spinning caterpillars its armature of secondary hooks and curved setæ enables it to retain its hold on the threads of the interior of its cocoon after the pupa has partially emerged from the cocoon, restraining it, as Chapman well says, “at precisely that degree of emergence from the cocoon that is most desirable.” He also informs us that while in the “pupæ incompletæ the cremaster is attached to an extensible cable, which always allows some emergence of the pupa, in the pupæ obtectæ there is no doubt but that in such cases as the Ichthyuræ, Acronyctæ, and many others, it retains the pupal case in the same position within the cocoon that the living pupa occupied; this is also very usually the case in the Geometræ and in the higher tineids (my pyraloids).”
In many of the more generalized moths there is no cremaster (Micropteryx, Gracilaria, Prodoxus, Tantura, Talæporia, Psychidæ, Hepialidæ, Zeuzera, Nola, Harrisina), though in Tischeria and Talæporia (Fig. 590, but not in Solenobia) and Psychidæ, two stout terminal spines perform the office of a cremaster, or there are simply curved setæ on the rounded, unarmed end of the abdomen, as in Solenobia.
In the obtected Lepidoptera, for example in such a group as the Notodontidæ, where the cremaster is present, though variable in shape, it may from disuse, owing to the dense cocoon, be without the spines and hooks in Cerura, or the cremaster itself is entirely wanting in Gluphisia, and only partially developed 637in Notodonta. In the butterflies whose pupæ are suspended (Suspensi), the cremaster is especially well developed. Reference might here be made to the temporary pupal structures in certain generalized moths, which take the place of a cremaster, such as the transverse terminal row of spines in Tinea, the two stout spines in Tischeria, and the dense rough integument and thickened callosities of the pupal head and end of abdomen of Phassus, which bores in trees with very hard wood; also the numerous stout spines at the end and sides of the abdomen in Ægerians. These various projections and spines, besides acting as anchors and grappling hooks, in some cases serve to resist strains and blows, and have undoubtedly, like the armature in the larvæ and imagines of other insects, arisen in response to intermittent or occasional pressure, stresses, and impacts.
Mode of formation of the cremaster and suspension of the chrysalis in butterflies.—We are indebted to Riley[109] for an explanation of the way the cremaster has originated, his observations having been made on species of over a dozen genera of butterflies (Suspensi).
He shows that the cremaster is the homologue of the suranal plate of the larva.[110] The preliminary acts of the larva have been observed by various authors since the days of Vallisneri, i.e. the larva hanging by the end of the abdomen, turning up the anterior part of the body in a more or less complete curve, and the skin finally splitting from the head to the front edge of the metathoracic segment, and being worked back in a shrivelled mass toward the point of attachment. The critical feat, adds Riley, which has most puzzled naturalists, is the independent attachment of the chrysalis and the withdrawal from and riddance of the larval skin which such attachment implies. Réaumur explained this in 1734 by the clutching of the larval skin between sutures of the terminal segments of the chrysalis, and this is the case, though the sutures act in a somewhat different way.
Before pupation the larva spins a mass or heap of silk, the shape of which is like an inverted settee or a ship’s knee, and “one of the most interesting acts of the larva, preliminary to suspension, is the bending and working of the anal parts in order to fasten the back of the (suranal) plate to the inside of the back of the settee, while the crotchets of the legs are entangled in the more flattened position or seat.”
In shedding the larval skin, the following parts are also shed, and have some part to play in the act of suspension: i.e. 1st, the tracheal ligaments (Fig. 593, tl), or the shed tracheæ from the last or 9th pair of spiracles; 2d, the rectal ligament (Fig. 593, rl), or shed intestinal canal; 3d, the Osborne or retaining 638membrane (membrana retinens, Fig. 593, mr), which is the stretched part of the membrane around the rectum and in the anal legs, and which is intimately associated with the rectal ligament.
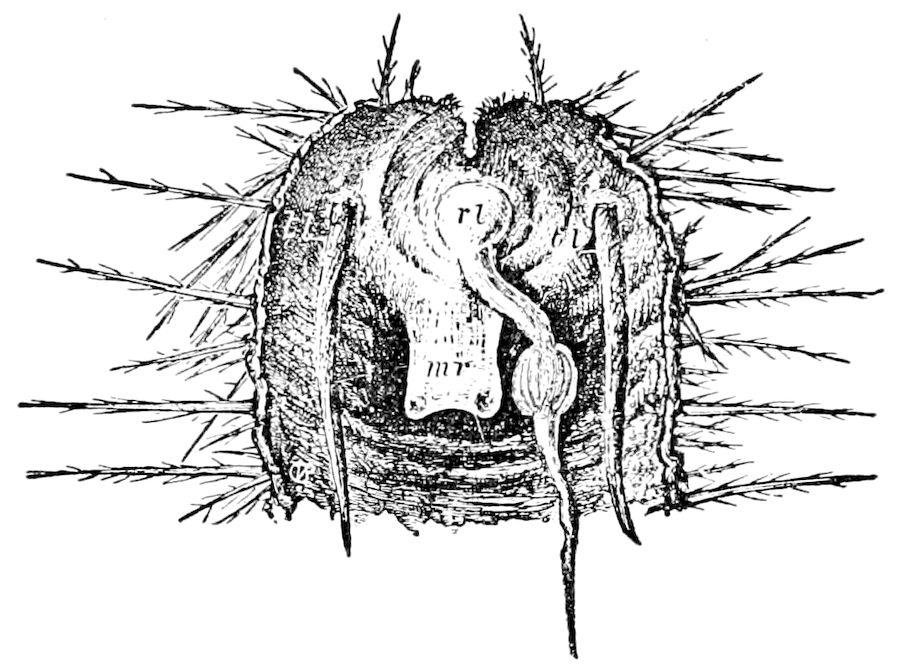
Fig. 593.—Shrunken larval skin of Vanessa antiopa, cut open from the back and showing (mr) the retaining membrane, (rl) the rectal ligament, and (tl) the tracheal ligaments.
The structures in the chrysalis are, first, the cremaster, with its dorsal (Fig. 594, dcr) and ventral (vcr) ridges, and the cremastral hook-pad (chp), said by Riley to be “thickly studded with minute but stout hooks, which are sometimes compound or furnished with barbs, very much as are some of our fishing-hooks, and which are most admirably adapted to the purpose for which they are intended.”
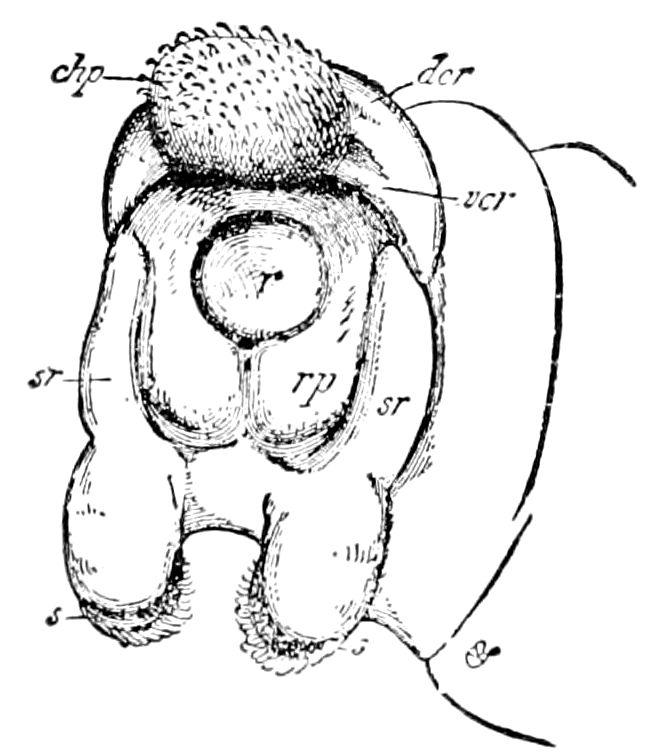
Fig. 594.—Ideal representation of the anal subjoint of Vanessa antiopa, from behind, with the spines removed, and all parts forced apart by pressure so as to show the homologies of the parts in the chrysalis which are concerned in pupation: homologies indicated by corresponding letters in Fig. 595, except that r (the rectum) corresponds with pr in Fig. 595.
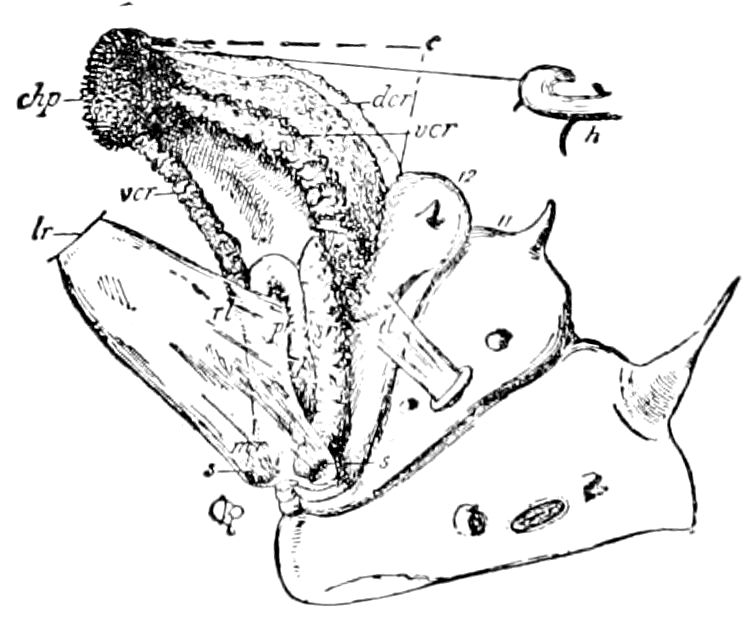
Fig. 595.—Anal parts of chrysalis of Vanessa antiopa, just prior to final extraction from shrunken larval skin: c, cremaster; chp, cremastral hook-pad; h, one of the hooks, more enlarged; vcr, ventral cremastral ridge; dcr, dorsal cremastral ridge; lr, larval rectum; pr, pupal rectum; rp, rectal plate; sr, sustentor ridges; mr, membrana retinens; rl, rectal ligament; tl, tracheal ligament; the 11th or last spiracle-bearing joint and the 12th joint being numbered.
Secondly, there are the other structures, viz., the sustainers (sustentors), two projections which Riley states “homologize with the soles (plantæ) of the anal prolegs, which take on various forms (3), but are always directed forward so as easily to catch hold of the retaining membrane.” These sustentors are, 639however, as Jackson[111] has shown, and as we are satisfied, the vestiges of the anal legs.
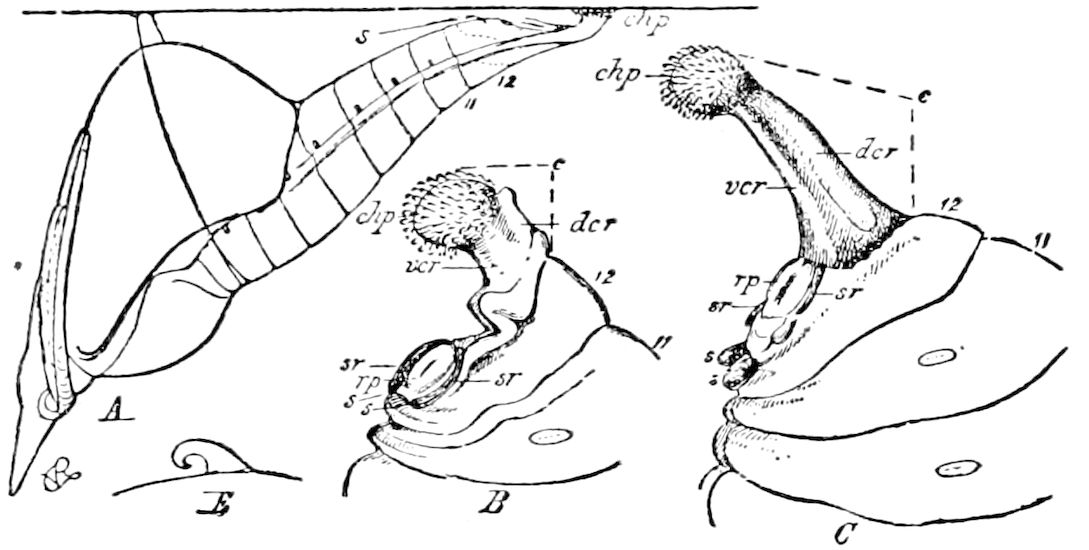
Fig. 596.—A, chrysalis of Terias. B, posterior end of chrysalis of Paphia. C, posterior end of chrysalis of Danais. E, one of the sustainers of Terias, greatly enlarged to show its hooked nature. All the parts of subjoint lettered to correspond with Fig. 595.
Thirdly, the sustentor ridges, which, as Riley states, may be more or less obsolete in some forms, in Paphia (Fig. 596, B) and Limenitis form “quite a deep notch, which doubtless assists in catching hold of the larval skin in the efforts to attach the cremaster.”
Fig. 597.—Pupation of butterflies: a, attachment of larva of Danais archippus; p, attachment of larva of Paphia glycerium; b, ideal larva soon after suspension; d, ideal larva a few hours later, the needle (n) separating the forming membrane from the sustainers; l, ideal larva just before splitting of larval skin, with retaining membrane loosened from the sustainers and showing its connection both with the larval and pupal rectum. In all the figures the joints of the body are numbered; the forming chrysalis is shaded in transverse lines; the intervening space between it and larval skin is dotted: h, is the hillock of silk; hl, hooks of hind legs; ap, anal plate; lr, larval rectum; pr, pupal rectum; mr, retaining membrane; c, cremaster; s, sustainers.—This and Figs. 593–596 after Riley.
“It is principally,” adds Riley, “by the leverage obtained by the hooking of the sustainers in the retaining membrane, which acts as a swimming fulcrum, 640that the chrysalis is prevented from falling after the cremaster is withdrawn from the larval skin. It is also principally by this same means that it is enabled to reach the silk with the cremastral hook-pads.”
“Dissected immediately after suspension, the last abdominal segment of the larva is found to be bathed, especially between the legs and around the rectum, in an abundance of translucent, membranous material.”
“An hour or more after suspension the end of the forming chrysalis begins to separate from the larval skin, except at the tip of the cremaster (Fig. 597, b). Gradually the skin of the legs and of the whole subjoint (10th segment) stretches, and with the stretching, the cremaster elongates, the rectal piece recedes more and more from the larval rectum, and the sustentor ridges diverge more and more from the cremaster, carrying with them, on the sustainers, a part of the soft membrane.” The rectal ligament will sustain at least 10 or 12 times the weight of the chrysalis. That of Apatura seems to rely almost entirely on the rectal ligament, assisted by the partial holding of the delicate larval skin.
FORMATION OF THE PUPA AND IMAGO IN THE HOLOMETABOLOUS INSECTS (THE DIPTERA EXCEPTED)
We have seen that in the incomplete metamorphosis, although there may be as many as five, and possibly seven moults, and in Chloëon as many as 20, and in Cicada septemdecim perhaps 25 or 30, there is but a slight change of form from one stage to another, and no period of inactivity. And this gradual outer transformation is so far as yet known paralleled by that of the internal organs, the slight successive changes of which do not differ from those observed in the growth of ametabolous insects. With the growth of the internal organs there probably goes on a series of gradual regenerative processes, and Korschelt and Heider state that we may venture to assume that each changed cell or group of cells which have become exhausted by the exercise of the functions of life are reabsorbed and become restored through the vital powers of the tissues, so that as the result there goes on a constant, gradual regeneration of the organs.
While the Hemiptera have only an incomplete metamorphosis, the males of the Coccidæ are, as shown by O. Schmidt, remarkable for passing through a complete or holometabolous development, with four stages, three of which are pupal and inactive. Hence, as Schmidt observes, there is here a hypermetamorphosis, like that of the Meloidæ, Stylopidæ, etc.
Shortly before the end of the larval stage of the male appear the imaginal buds of the eyes, legs, and wings. In the 2d or 1st pupal 641stage there is an atrophy of the antennæ and legs. On the other hand, at this stage the female completes its metamorphosis.
The rudiments of the wings arise on the edge of the dorsal and ventral side of the 2d thoracic segment, and this, we would remark, is significant as showing a mode of origin of the wings intermediate between that of the manometamorphic and holometamorphic insects. (See pp. 137–142.) While Schmidt could not ascertain the exact structure of the imaginal buds, he says “in general the process of formation of the extremities is exactly as Weismann has described in Corethra.” The two later pupal stages are “as in other metabolic insects.” (See p. 690, Fig. 637.)
Thus far the internal changes in the metamorphosis of the Coleoptera have not been thoroughly studied. They are less complete than in the other holometabolous insects, the differences between the larva and imago being much less marked than in the more specialized orders, and so far as known all the larval organs pass, though not without some great changes, directly into the imaginal ones, the only apparent exception being the mid-intestine, which, as stated by Kowalevsky, undergoes a complete transformation during metamorphosis. The following account, then, refers almost wholly to the Lepidoptera, Hymenoptera, and Diptera.
a. The Lepidoptera
The first observations on the complete metamorphosis of insects which were in any way exact were those of Malpighi, in 1667, and of Swammerdam, in 1733. While the observations of Swammerdam, as far as they extended, were correct, his conclusions were extraordinary. They were, however, accepted by Réaumur and by Bonnet, and generally held until the time of Herold in 1815, and lingered on for some years after. The rather famous theory of incasement (“emboîtement”) propounded by Swammerdam was that the form of the larva, pupa, and imago preëxisted in the egg, and even in the ovary; and that the insects in these stages were distinct animals, contained one inside the other, like a nest of boxes, or a series of envelopes one within the other, or, to use his own words: “Animal in animali, seu papilio intra erucam reconditus.”
This theory Swammerdam extended to the whole animal kingdom. It was based on the fact that by throwing the caterpillar, when about to pupate, in boiling water, and then stripping off the skin, the immature form of the butterfly with its appendages was disclosed. Malpighi had previously observed the same fact in the 642silkworm, perceiving that before pupation the antennæ are concealed in the head of the larva, where they occupy the place previously taken by the mandibular muscles; also that the legs of the moth grew in those of the larva, and that the wings developed from the sides of the worm.
Even Réaumur (1734) remarked: “Les parties du papillon cachées sous le fourreau de chenille sont d’autant plus faciles à trouver que la transformation est plus proche. Elles y sont neanmoins de tout temps.” He also believed in the simultaneous existence of two distinct beings in the insect. “Il serait très curieux de connaître toutes les communications intimes qui sont entre la chenille et le papillon.... La chenille hache, broye, digere les aliments qu’elle distribué au papillon; comme les mères préparent ceux qui sont portés aux fœtus. Notre chenille en un mot est destineé à nourrir et à defendre le papillon qu’elle renferme.” (T. i, 8e Mémoire, p. 363.)
Lyonet (1760), even, did not expose the error of this view that the larva enveloped the pupa and imago, and, as Gonin says, it was undoubtedly because he did not use for his dissections of the caterpillar of Cossus any specimens about to pupate. Yet he detected the wing-germs and those of the legs, stating that he presumed the bodies he saw to be the rudiments of the legs of the moth (p. 450).
Herold, in his work on the development of the butterfly (1815), was the first to object to this erroneous theory, showing that the wings did not become visible until the very end of larval life; that as the larval organs disappear, they are transformed or are replaced by entirely new organs, which is not reconcilable with a simple putting off of the outer envelope. The whole secret of metamorphosis, in Herold’s opinion, consisted in this fact, that the butterfly in the larva state increases and accumulates a supply of fat until it has reached the volume of the perfect state; then it begins the chrysalis period, during which the organs are developed and take their definite form.[112] (Abstract mostly from Gonin.) Still the old ideas prevailed, 643and even Lacordaire, in his Introduction à l’Entomologie published in 1834, held on to Swammerdam’s theory, declaring that “a caterpillar is not a simple animal, but compound,” and he actually goes so far as to say that “a caterpillar, at first scarcely as large as a bit of thread, contains its own teguments threefold and even eightfold in number, besides the case of a chrysalis, and a complete butterfly, all lying one inside the other.” This view, however, we find is not original with Lacordaire, but was borrowed from Kirby and Spence without acknowledgment. These authors, in their Introduction to Entomology (1828), combated Herold’s views and stoutly maintained the old opinions of Swammerdam. They based their opinions on the fact, then known, that certain parts of the imago occur in the caterpillar. On the other hand, Herold denied that the successive skins of the pupa and imago existed as germs, holding that they are formed successively from the “rete mucosum,” which we suppose to be the hypodermis of later authors. In a slight degree the Swammerdam-Kirby and Spence doctrine was correct, as the imago does arise from germs, i.e. the imaginal disks of Weismann, while this was not discovered by Herold, though they do at the outset arise from the hypodermis, his rete mucosum. Thus there was a grain of truth in the Swammerdam-Kirby and Spence doctrine, and also a mixture of truth and error in the opinions of Herold.
The real nature of the internal changes wrought during the process of metamorphosis was first revealed by Weismann in 1864. His discovery of the germs of the imago (imaginal buds) of the Diptera, and his theory of histolysis, or of the complete destruction of the larval organs by a gradual process, was the result of the application of modern methods of embryology and histology, although his observations were first made on the extremely modified type of the Muscidæ or flies, and, at first, he did not extend his view to include all the holometabolous insects. Now, thanks to his successors in this field, Ganin, Dewitz, Kowalevsky, Van Rees, Bugnion, Gonin, 644and others, we see that metamorphosis is, after all, only an extension of embryonic life, the moults and great changes being similar to those undergone by the embryo, and that metamorphosis and alternation of generations are but terms in a single series. Moreover, the metamorphoses of insects are of the same general nature as those of certain worms, of the echinoderms, and the frog, the different stages of larva, pupa, and imago being adaptational and secondary.
While the changes in form from the larva to the pupa are apparently sudden, the internal histogenetic steps which lead to them are gradual. In the Lepidoptera a few days (usually from one to three) before assuming the pupa stage, the caterpillar becomes restless and ceases to take food. Its excrements are now hard, dry, and, according to Gonin, are “stained carmine red by the secretions of the urinary tubes.” Under the microscope we find that they are almost exclusively composed of fragments of the intestinal epithelium. These red dejections were noticed by Réaumur, and afterwards by Herold, and they are sure indications of the approach of the transformations. It now wanders about, and, if it is a spinner, spins its cocoon, and then lies quietly at rest while the changes are going on within its body. Meanwhile, it lives on the stores of fat in the fat-body, and this supply enables it to survive the pupal period.
The amount of fat is sometimes very great. Newport removed from the larva of Cossus ligniperda 42 grains of fat, being more than one-fourth of the whole weight of the insect, he adds that the supply is soon nearly exhausted during the rapid development of the reproductive organs, “since, when these have become perfected, the quantity that remains is very inconsiderable.”
Although the larval skin of a lepidopterous insect is suddenly cast off, the pupa quickly emerging front it, yet there are several intermediate stages, all graduating into each other. If a caterpillar of a Clisiocampa, which, as we have observed, is much shortened and thickened a day or two before changing to a pupa, is hardened in alcohol and the larval skin is stripped off, the semipupa (pro-nymph, pro-pupa of different authors) is found to be in different stages of development, and the changes of the mouth-parts are interesting, though not yet sufficiently studied.
Newport attributes the great enlargement and changes in the shape of the thoracic segments of the larva of Vanessa urticæ at this time, to the contraction or shortening of the muscles of the interior of those segments, “which are repeatedly slowly extended and shortened, as if the insect were in the act of laborious respiration.” This, he adds, generally takes place at short intervals during 645the two hours immediately preceding the change to the pupa, and increases in frequency as that period approaches. He thus describes the mode of moulting the larval skin: “When the period has arrived, the skin bursts along the dorsal part of the 3d segment, or mesothorax, and is extended along the 2d and 4th, while the coverings of the head separate into three pieces. The insect then exerts itself to the utmost to extend the fissure along the segment of the abdomen, and, in the meantime, pressing its body through the opening, gradually withdraws its antennæ and legs, while the skin, by successive contortions of the abdomen, is slipped backwards, and forced towards the extremity of the body, just as a person would slip off his glove or his stocking. The efforts of the insect to get entirely rid of it are then very great; it twirls itself in every direction in order to burst the skin, and, when it has exerted itself in this manner for some time, twirls itself swiftly, first in one direction, then in the opposite, until at last the skin is broken through and falls to the ground, or is forced to some distance from it. The new pupa then hangs for a few seconds at rest, but its change is not yet complete. The legs and antennæ, which when withdrawn from the old skin were disposed along the under surface of the body, are yet separate, and do not adhere together as they do a short time afterwards. The wings are also separate and very small. In a few seconds the pupa makes several slow, but powerful, respiratory efforts; during which the abdominal segments become more contracted along their under surface, and the wings are much enlarged and extended along the lateral inferior surface of the body, while a very transparent fluid, which facilitated the slipping off of the skin, is now diffused among the limbs, and when the pupa becomes quiet dries, and unites the whole into one compact covering.”
The changes in the head and mouth-parts.—The changes of form from the active mandibulate caterpillar to the quiescent pupa, and then to the adult butterfly, are, as we have seen, in direct adaptation to their changed habits and surroundings, and they differ greatly in details in insects of different orders. In many Lepidoptera and certain Diptera the pupa and imago are without the mandibles of the larva, and, instead, the 1st maxillæ in the former order, and the 2d maxillæ in the latter, are highly developed and specialized. The changes in the shape of the head, with the antennæ, the latter rudimentary in the larvæ of the two orders named, are noteworthy, and will be referred to under those orders. The same may be said of the thorax with the legs and wings, and the abdomen with the ovipositor. Every part of the body undergoes a profound change, though 646in the Coleoptera, Trichoptera, and the more generalized and primitive Diptera, each segment and appendage of the larva are directly transformed into the corresponding parts of the pupa, and subsequently of the imago. We shall see, however, beyond, that this general statement does not apply to the Hymenoptera, in which there is a process of cephalization or transfer of parts headward, peculiar to that order.
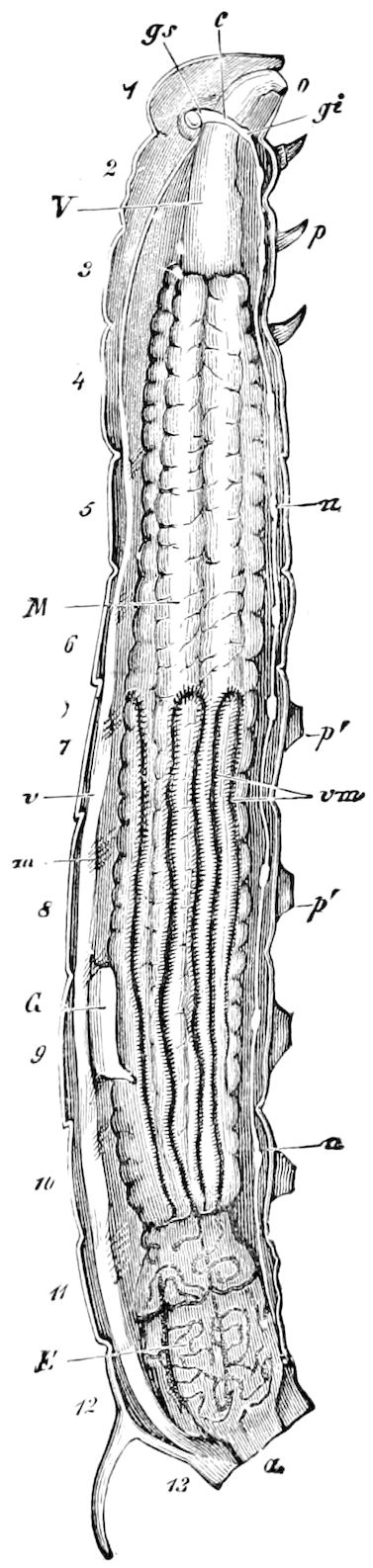
Fig. 598.—Internal organs of Sphinx ligustri: 1, head; 2–4, thoracic, 5–13, abdominal segments; V, fore-, M, mid-, E, hind-intestine; gs, brain; gi, infraœsophageal ganglion; n, ventral ganglion; vm, urinary tubes; c, heart; G, testis; o, œsophagus; a, anus; m, alary muscles of the heart.
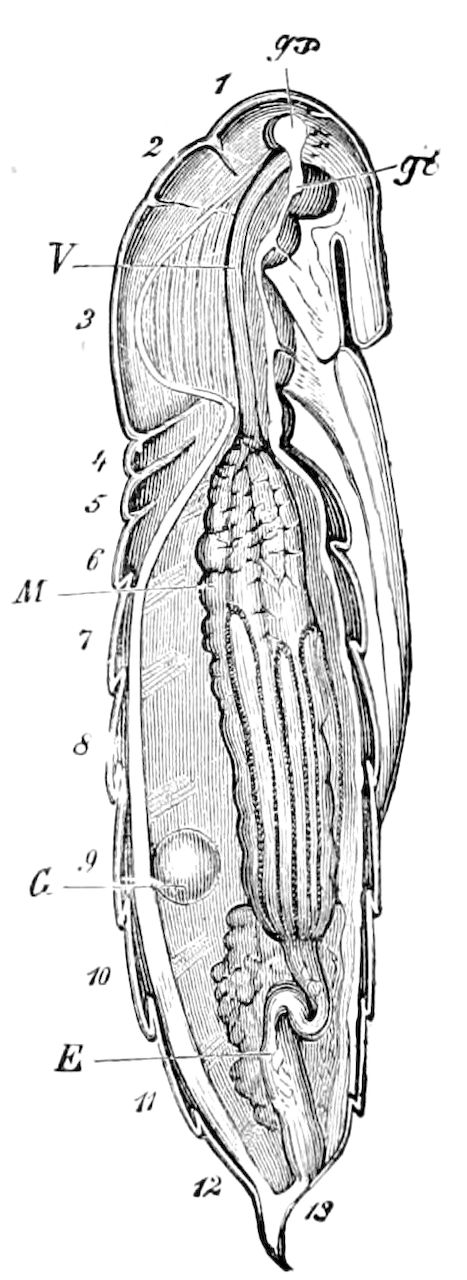
Fig. 599.—Pupa of the same.
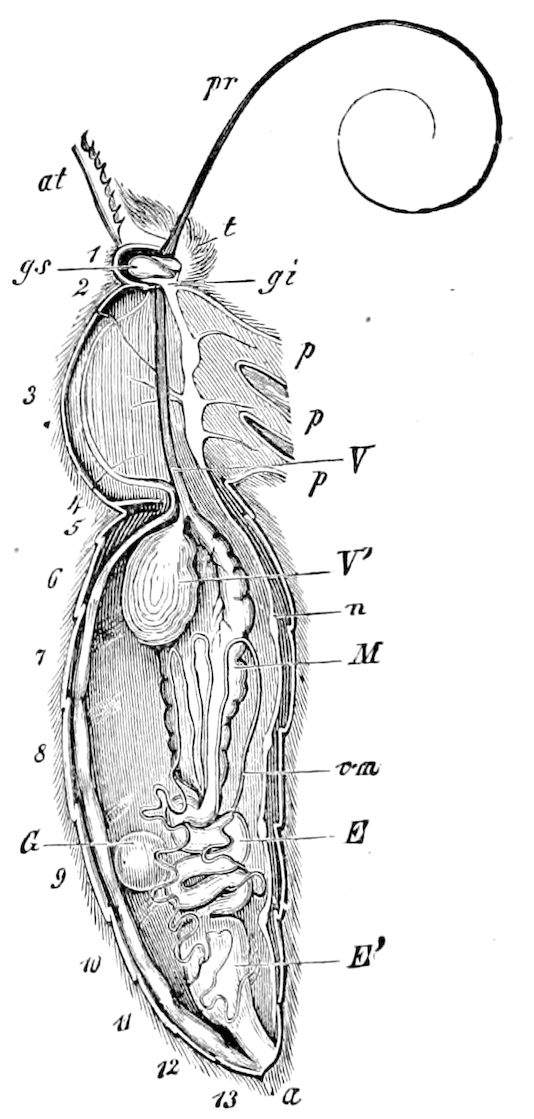
Fig. 600.—Imago of the same.—This and Figs. 598 and 599 after Newport, from Gegenbaur.
Fig. 601.—Nervous system of the larva of Sphinx ligustri.
Fig. 602.—Nervous system of the pupa of Sphinx ligustri, soon after pupation.—This and Fig. 601, after Newport.
The change in the internal organs.—These were especially, as regards the nervous system, first carefully examined and illustrated by that great English entomotomist, Newport, and those of the reproductive organs by Herold as early as 1815. A glance at the figures (598–604), reproduced from Newport’s article Insecta, will show the changes wrought especially in the digestive and nervous systems of Sphinx and Vanessa, his account of the alterations in the muscles having already been quoted. As the pupal form is much nearer to that of the imago than of the larva, so the digestive canal is seen to be nearly as much differentiated in the pupa as in the imago, though the reservoir (“sucking-stomach”) of the imago is not indicated in the pupa. These changes 648are such as occur in an insect which is enormously voracious as a larva, and which often, passing through a period of complete inactivity, taking no food at all, finally becomes an insect which needs to suck in only a minimum quantity of water or nectar, and which practically abstains from all food. The head and genital glands also, as well as the urinary vessels, are nearly the same. On the other hand, the salivary glands have undergone, in the imago, a thoroughgoing reduction.
The changes undergone by the nervous system of Sphinx ligustri and Vanessa urticæ have been described by Newport with fulness of detail. An abstract of his observations on Vanessa urticæ, which undergoes its changes in June in 14 days, and in August in eight days, we will now give, in part verbatim, the subject being rendered much clearer by his figures, which are reproduced.
During the last larval stage, certain changes have already taken place in different parts of the cord, which shows that they had been a long time in progress. Besides the lateral approximation of the cords, the first change consists in a union of the 11th and 12th ganglia, the latter one being carried forwards; these two ganglia being entirely separate before the 3d moult.
Two hours after the larva of Vanessa urticæ has suspended itself in order to pupate, the brain is not yet enlarged, but the subœsophageal ganglion is nearly twice its original size and the ganglia behind are nearer together. “A little while before the old larval skin is thrown off there is great excitement throughout the body of the insect.” About half an hour (Fig. 603, 2) before this occurs the alary nerves and the cerebral, 2d, 3d, 4th, and 5th ganglia are slightly enlarged, and the 1st subœsophageal ganglion very considerably. Immediately after the insect has entered the pupa state (Fig. 603, 3), all the ganglia are brought closer together. One hour after (Fig. 603, 4) pupation the cerebral ganglia are found to be more closely united, the 4th and 5th ganglia are nearer, and the distance between the remaining ganglia is also reduced.
Seven hours after pupation there is a greater enlargement of the cerebral ganglia, optic nerves, and ganglia and cords of the future thoracic segments.
At 12 hours (Fig. 603, 5) the 5th pair of ganglia has almost completely coalesced with the cord and the 4th; at 18 hours (Fig. 603, 6) the whole of the ganglia, cords, and nerves have become more enlarged, especially those of the wings, while the 4th and 5th ganglia of the cords have now so completely united as to appear like an irregular elongated mass. At 24 hours (Fig. 604, 7) the 4th and 5th ganglia are completely united, the 5th being larger than the 4th. At 36 hours (Fig. 604, 8) the optic nerves have attained a size almost equal to that of the brain. The 1st subœsophageal ganglion now forms, with the cerebral ones, a complete ring around the œsophagus, the crura having almost disappeared. The 6th ganglion has now disappeared, but the nerves arising from it remain. At 48 hours (Fig. 604, 9) the cord is straight instead of being sinuous, and the 7th ganglion has disappeared, while the thoracic ganglia are greatly enlarged. At the end of 58 hours the 2d and 3d thoracic ganglia have united, and the double ganglion thus formed is only separated from the large thoracic mass composed of the 4th, 5th, and part of the 6th ganglia, by the short but greatly enlarged cords which pass on each side of the central attachment of the muscles. “The optic and antennal nerves have nearly attained their full development, and those numerous and most intricate plexus of nerves in the three thoracic segments of the larva form only a few trunks, which can hardly be recognized as the same structures. The arrangement of the whole nervous system is now nearly as it exists in the perfect insect. The whole of these important changes are thus seen to take place within the first three days after the insect has undergone its metamorphosis; and they precede those of the alimentary canal, generative system, and other organs, which are still very far from being completed, and indeed, as compared with the nervous system, have made but little progress.” (Art. Insecta, pp. 962–965.)
Fig. 603.—Changes in the nervous system of Vanessa urticæ, during and after pupation.—After Newport.
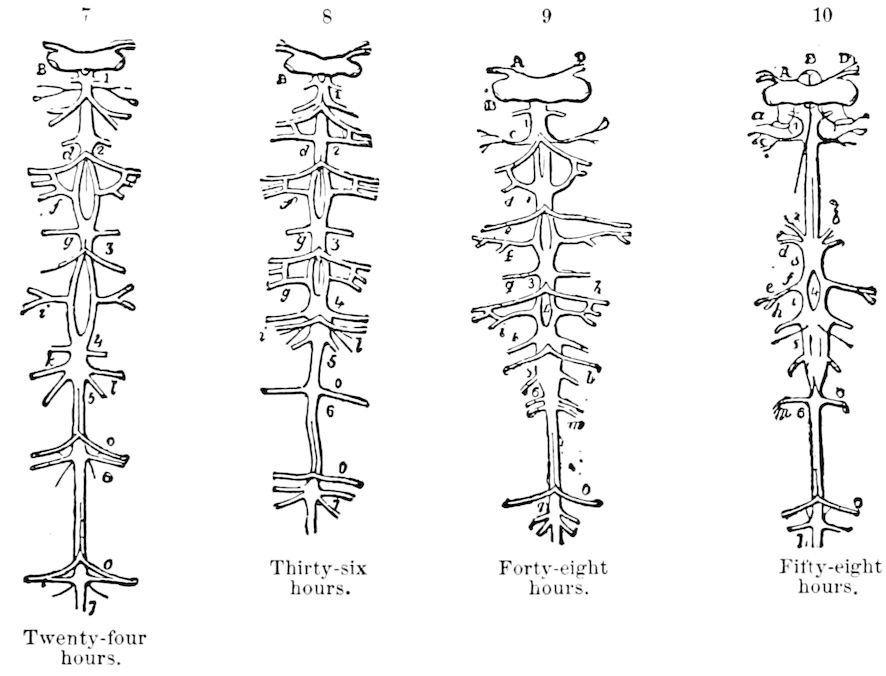
Fig. 604.—Changes in the nervous system of Vanessa urticæ, from 24 to 58 hours after pupation.—After Newport.
650The initial steps and many of the subsequent internal changes escaped the notice of Newport and others of his time, and it was not until the epoch-making work of Weismann on the ultimate processes of transformation of Corethra and of Musca, that we had an adequate knowledge of the subject.
Weismann (1864) was the first to show for the Muscidæ and Corethra that the appendages, wings, and other parts of the imago originate in separate, minute, cellular masses called imaginal disks, buds, or folds (histoblasts of Künckel). These imaginal buds, which arise from the hypodermis, being masses of indifferent cells, are usually present in the very young larva, and even in the later embryonic stages. It has been shown that such imaginal buds exist for each part of the body, not only for the appendages and wings (p. 126), but for the different sections of the digestive canal. During the semipupal stage these buds enlarge, grow, and at the same time there is a corresponding destruction of the larval organs. The process of destruction is due to the activity of the blood corpuscles or leucocytes (phagocytes), the larval organs thus broken up forming a creamy mass, the buds from which the new organs are to arise resisting the attacks of the virulent leucocytes, which attach themselves to the weakened tissue and engulf the pigments (see p. 422). The two processes of destruction of the larval organs (histolysis) and the building up of the imaginal organs (histogenesis) go hand in hand, so that the connection of the organs in question in most cases remains entirely continuous; while the last steps in the destruction of the larval organs only take place after the organs of the imago have assumed their definite shape and size. Other observers have corroborated and confirmed his statements and observations, Gonin extending them to the Lepidoptera and Bugnion to the Hymenoptera.
It is a pity that the observations, such as were set on foot by Weismann, were not first made on the Trichoptera and Lepidoptera, which are much more primitive and unmodified forms than the 651Diptera, but mistakes of this nature have frequently happened in the history of science.
Fig. 605.—Full grown larva of Pieris brassicæ opened along the dorsal line: d, digestive canal; s, silk-gland; g, brain; st I, prothoracic stigma; st IV, 1st abdominal stigma; a, a′, germs (buds) of fore and hind wings; p, bud of thoracic segment;—those of the 3d pair are concealed under the silk-glands; I-III, thoracic rings.—After Gonin.
The latest and most detailed researches are those of J. Gonin on the metamorphoses of Pieris brassicæ, made under the direction of Professor E. Bugnion. They fill an important gap in our knowledge, and show that the Lepidoptera transform in nearly the same manner as described by Weismann in Corethra. We give the following condensed account of Gonin’s observations.
On opening a caterpillar entering on the semipupa state (Fig. 605), 652the relative position of the germs (imaginal buds or folds) of the wings and of the legs are seen.
Fig. 606.—Section through thorax of a tineid larva on sycamore, passing through the 1st pair of wings (w): ht, heart; i, œsophagus; s, salivary gland; ut, urinary tube; nc, nervous cord; m, recti muscles; a part of the fat-body overlies the heart. A, right wing-germ enlarged.
Fig. 607.—Section of the same specimen as in Fig. 606, but cut through the 2d pair of wings (w): i, mid-intestine; h, heart; fb, fat-body; l, leg; n, nervous cord.
These imaginal buds in a more advanced stage are seen in our sections of a tineid larva (Figs. 606, 607).
The number of 12 imaginal buds found by Weismann in the thorax of Muscidæ does not occur in Lepidoptera, since, as in the Hymenoptera (Bugnion), the dorsal buds of the prothoracic segment are wanting. Gonin finds in Pieris that the ventral buds of the 653three thoracic segments are each represented by several distinct folds attached to the femoro-tibial bud and to the tarsal joints.
The imaginal buds serve in some cases for the formation of new organs (wings, legs of insects with apodous larvæ); in others for the growth and the transformation of organs already existing (legs, antennæ, 1st and 2d maxillæ of Lepidoptera).
As to the peripodal sac or hypodermic envelope which contains the imaginal bud, a portion persists and is regenerated, while the other part becomes useless and is detached under the form of débris, as shown by Weismann, Viallanes, and Van Rees in the Muscidæ. On this point Gonin disagrees with Dewitz, who stated that the walls of the wing-sacs are not destroyed, but are only gradually withdrawn at the time of pupation, in order to allow the orifice to distend and let the wing pass out to the exterior.
The portion of the sac which persists (basal portion, peripheral pad of Bugnion, or annular zone of Künckel) serves at first to attach the appendage, while forming, to the hypodermis of the larva, then afterwards to more or less completely regenerate the adjoining portion of the integument. In this way the hypodermis of the thorax is partially, that of the head is almost entirely, replaced by the imaginal epithelium which proliferates at the base of the appendages,[113] while that of the abdominal segments persists, at least in a modified way, and only undergoes (at the end of the pupal period) transformations as regards the appearance of the scales and pigment.
The wings.—The imaginal buds of the wings do not participate in the larval moults. Gonin has observed, contrary to Dewitz, that their surface only forms a cuticle towards the end of the last larval stage.
The network of fine tracheæ of the wing-bud is drawn out at the time of pupation with the internal cuticle of the large tracheæ. The permanent tracheæ of the wing have already appeared at the time of the 3d moult under the form of large rectilinear trunks, the position of which corresponds afterwards to that of the veins, but they are not filled with air until the time of pupation. There are from eight to ten of these tracheæ in each wing (Fig. 159), and they give rise in the pupa to a new system of fine tracheæ (tracheoles) which replaces that of the larva. (For further details the reader is referred to pp. 126–137.)
Development of the feet and the cephalic appendages.[114]—In the apodous larvæ of Diptera and Hymenoptera the rudiments of the legs are, like those of the wings, developed within hypodermal sacs; at times they remain there up to the end of larval life, but sometimes 654they appear early at the surface. This origin of the legs, thanks to Weismann, Künckel, and Van Rees, is well known in the Diptera; in the Hymenoptera it has been proved to be the case with ants by Dewitz, and in Encyrtus by Bugnion. As for the Lepidoptera our knowledge that the legs of the imago arise from the six thoracic legs of the caterpillar, up to the date of Gonin’s paper has not been in advance of that of Malpighi and Swammerdam.
Réaumur, moreover, was supposed to have furnished the proof, having from his experiments concluded that “if the legs of the pupa appear longer and larger than those of the caterpillar wherein they were contained, it is because they were folded and squeezed.” (8e Mém., p. 365.)
This explanation of Réaumur’s has been generally accepted. Graber (Die Insekten, p. 506) accepted it, after examining microscopic sections of the legs, and Künckel averred that “Réaumur, having, in certain caterpillars, completely cut off one of the thoracic legs, had concluded that the butterfly which came from it lacked the corresponding member.” (Rech. sur l’org. et dév. des volucelles, p. 160.)
Newport, it is true, denied this disappearance of the legs, but did not wish to put himself in opposition to received ideas, and supposed that the member cut off was partly reformed in the imago.
Künckel believes that he has found a better solution in his theory of histoblasts or imaginal buds; in his opinion, “Réaumur and Newport are both right,” but “when Réaumur cut off a caterpillar’s leg, he at the same time removed the histoblast, the rudiment of the leg of the butterfly. When Newport repeated this experiment, he simply mutilated the histoblast without completely destroying it: in the first case, the adult insect was born with one leg less; in the second case, it appeared with an atrophied foot.”
“So ingenious an explanation,” says Gonin, “is not necessary.” To prove that the experiments of the two savants are not contradictory, it would have been sufficient to cite, as Künckel did not do, the exact words of Réaumur, for he having cut from a caterpillar “more than half of three of the thoracic legs on the same side,” says he found that the chrysalis had “the three limbs on one side shorter than the corresponding ones on the other side.” The same operation repeated on a somewhat younger caterpillar again showed in the chrysalis three maimed limbs, “so that they could not be said to be entirely absent. These results are like those of Newport; the interpretation only was faulty, as we shall attempt to prove.”
The real relations of the adult legs to the larval legs are thus shown by Gonin.
“If we carefully strip off the skin of a caterpillar near the time of pupation (Fig. 608), we see that the extremity only of the legs of the imago is drawn out of the larval legs; the other parts are pressed against each side of the thorax: near the ventral line a small pad represents the coxa and the trochanter; the femur and the tibia are distinctly recognizable, but soldered to each other and only separated by a slight furrow; they form by their union a very acute knee or bend. The femur is movable on the pad-like coxa, the tibia continues 655without precise limits with the extremity concealed in the larval legs. The three divisions of the latter do not appear to have any relation with the live joints of the perfect state. Under the microscope the rudiment appears very strongly plaited at the level of the tarsus, much less so in the other regions. A large trachea penetrates into the femur with some capillaries; reaching the knee it bends into the tibia at a sharp curve, but does not become truly sinuous in approaching the extremity. It is then the tarsus especially which is susceptible of elongation; it may, on being withdrawn, give rise to the illusion that the whole organ is disengaged from the larval leg.
“This disposition is, we believe, not known. It gives the key to the experiments of Réaumur and of Newport.
“Even when we cut off the limb of the caterpillar at its base, we only remove the tarsus of the imago; the femur and the tibia remain intact. From an evident homology Réaumur has erroneously concluded that there is an identity. His opinion, classical up to this day, that the limb of the butterfly is entirely contained in the leg of the caterpillar, has been found to be inexact and should be abandoned.”
Embryonic cells and the phagocytes.—Up to the last larval stage the legs do not offer, says Gonin, any vestige of an imaginal germ, but they contain a great number of embryonic cells (Fig. 145, ec). They are almost always collected around a nerve or trachea; sometimes they are independent, and sometimes retained in the peritoneal sheath, seeming to arise by proliferation from this sheath. Some thus contribute to the lengthening of the tracheal branches or nerves, and the others, becoming detached, form leucocytes or phagocytes. They are very numerous in the legs, at the beginning of the 4th stage, but are disseminated some days later throughout the whole cavity of the body. At the time of histolysis they attack the larval tissues and increase in volume at their expense; in return they serve for the nutrition of the imaginal parts and exercise no destructive action on them. Van Rees agrees with Kowalevsky in comparing these attacks of the embryonic cells, sometimes victorious and sometimes impotent, to the war which the leucocytes wage against both the attenuated and the virulent bacteria.
Formation of the femur and of the tibia, transformation of the tarsus.—Capillary tracheæ appear in the leg at the same time as in the wing. They arise from the end of a tracheal trunk near the base of the limb on the dorsal and convex side. After the 3d moult the hypodermis thickens near this place; in a few days a pad is formed 656there and then a large imaginal bud with a circular invagination. These buds were noticed by Lyonet, who supposed them to be “les principes des jambes de la phalène.” Nerves and a tracheal branch penetrate into the femoro-tibial bud and form a small bay or constriction which marks the point of junction of the femur with the tibia, and the body-cavity remains in direct communication with the end of the limb.
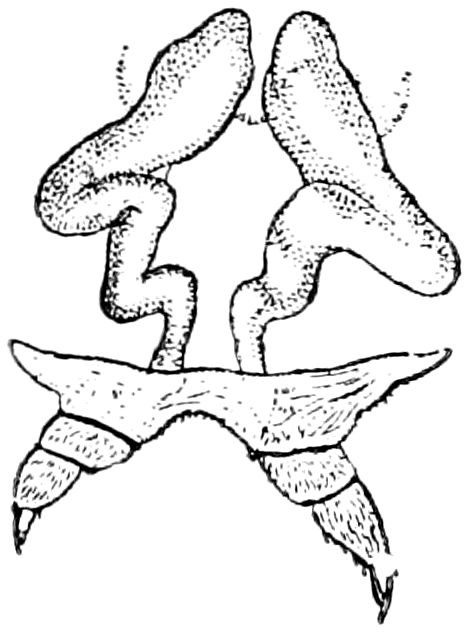
Fig. 608.—Feet of the Pieris butterfly withdrawing from those of the larva.
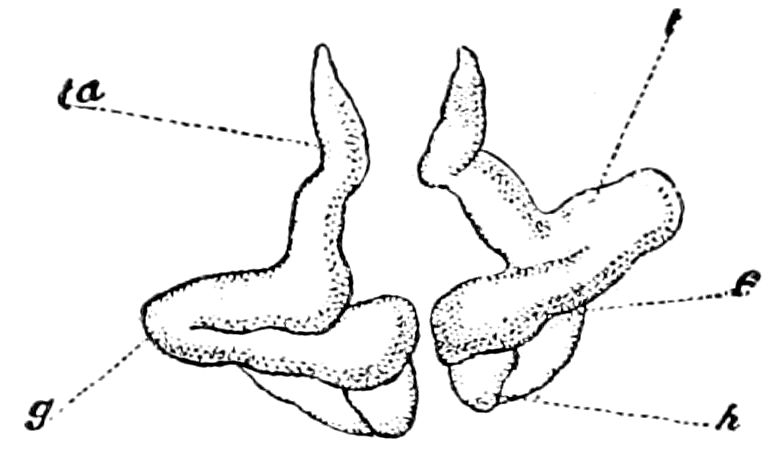
Fig. 609.—Imaginal feet of Pieris uncovered with great care to preserve the position which they had in the larva: ta, tarsus; t, tibia; g, knee; f, femur; h, coxa.
The tarsus undergoes a series of changes; the surface is folded in a very complicated way; at the level of each articulation, but only in the internal and concave region of the leg, is developed a deep fold; on one side there is a hypodermic thickening, on the other a simple leaf of the envelope, which afterwards joins at its base with the parietal hypodermis, and then two leaves are destroyed with the large cells of the setæ. The internal part and end of the tarsus are then reconstituted with the elimination of the débris, while the external and convex region undergoes direct regeneration.
The coxa and trochanter are derived from the base of the larval leg, and only the 1st pair are well separated from the base of the thorax. One or two days before pupation the femoro-tibial bud, after having, until now, preserved its antero-posterior direction, is placed transversely as regards the larva, then becoming directed obliquely forward. This rotatory movement of the coxa may be attributed to the great extension of the fore wings, which push before them the two first pairs of legs. The last pair in their turn are simply covered by the hind wings and are but slightly displaced. This new position of the legs is that of the imago: the knee of the 1st pair is situated in front of the tarsus; that of the 2d a little outward; that of the 3d pair is directed backward. (Gonin.)
The antennæ.—These appendages also have the same relation with those of the caterpillar as in the case of the legs, the larval appendages 657being only the point of departure of the imaginal growth. Weismann has observed in Corethra how at the approach of each moult an invagination like the finger of a glove allows the antenna to elongate from its base. The process, says Gonin, is identical in the caterpillar of Pieris. At the last moult the invagination is so pronounced that it is not effaced with the renewal of the chitinous integument. Several days later it again begins to grow larger. As the imaginal bud gradually sinks into the cavity of the head, it presses back the hypodermic wall and thus forms an envelope around it. Its base, widely opened, gives admission to the nerves, besides capillaries and sometimes a large trachea.
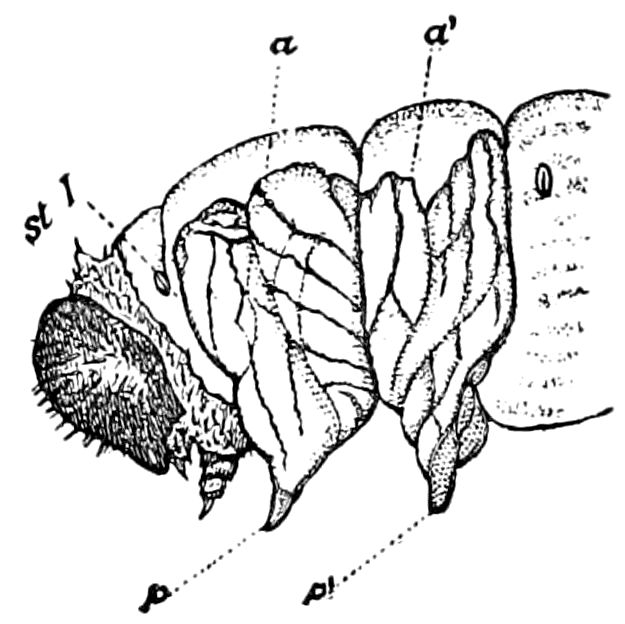
Fig. 610.—Larva in same stage as Fig. 613; side view of head and thorax: a, a′, wings, with the folds on the surface, and the sinuous track of the tracheal bundles; st I, prothoracic stigma; p, p′, ends of the legs.
As soon as it reaches the posterior region of the head, the antenna in lengthening becomes folded and describes the great curves which led Réaumur to compare it to a ram’s horn (Fig. 613). The leaf of the envelope thickens in the interior and all around the base of the organ. Its ultimate rôle is closely like that of the two other hypodermic formations. It is at the outset this layer of cells which in the larva supports the ocelli. This layer, hidden on each side under the parietal region, thickens and regenerates, forming a circular pad which becomes more prominent and finally assumes the form of the compound eye of the imago.
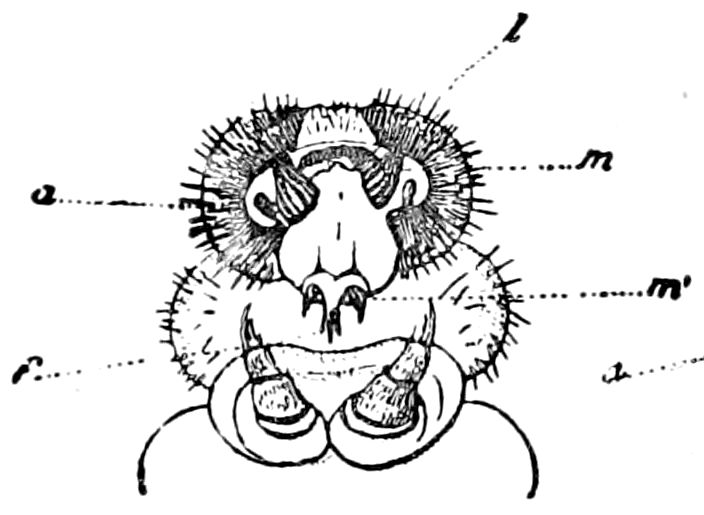
Fig. 611.—Head of the larva just before pupation: between the two mandibles (m) is seen the relief of the tongue or maxillæ (m′); f, spinneret; l, labrum; a, antenna.
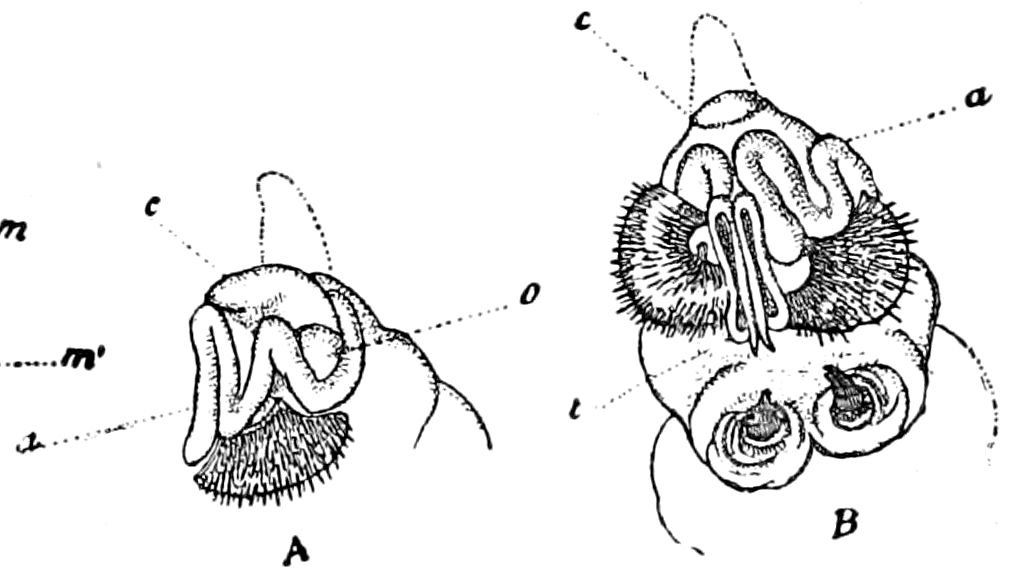
Fig. 612.—Same stage as in Fig. 611, but after the removal of the larval skin, and including the lateral scale: A, side, B, front, view; c, “cimier” (the dotted line shows the position it takes in the pupa); a, antenna; o, eye; t, tongue.—This and Figs. 608–611, after Gonin.
Finally, this layer gives rise to a conical prolongation (Fig. 612, c), which after exuviation appears as a tuft of long hairs, and is called 658by Gonin the crest (cimier, Fig. 612), which is characteristic of the pupæ of Pieridæ. It is only differentiated towards the end of the 4th larval stage in a median depression of the vertex. It is an imaginal bud in the most general sense of the word.
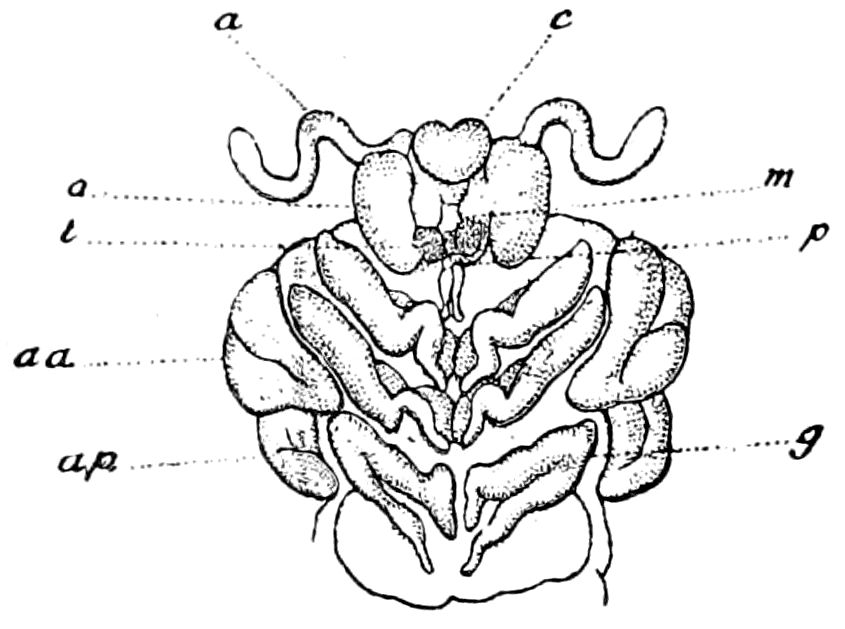
Fig. 613.—Larva of Pieris brassicæ stripped of its skin some minutes before pupation: the antennæ (a) have been displaced, and the tongue cut off, to show the palpi (p); c, cimier: o, eye; m, vestige of a mandible; t, insertion of the tongue (see Fig. 612); aa, fore, ap, hind, wings; g, knee of a foot of the 3d pair.
On each side the base of the antenna comes in contact with the germ of the crest. The envelopes approach each other, and their thickened part constitutes with the ocular disks a new cephalic wall. The head of the butterfly thus marked off is triangular; all the larval parts remaining out of this area then disappear. The muscles and the nerves are resorbed by histolysis, then the external part of the imaginal envelopes and the old parietal hypodermis, reduced very thin and degenerated, is detached in shreds. The antenna becomes external throughout its whole extent. The transformation is in this case, then, almost as complete as in the thorax of Diptera or Hymenoptera. It is necessitated by the change of form and of volume of the head. The region of the ocelli persists unchanged almost alone from the larva to the imago also. The limit is not well marked between the portion which is the replacement or direct renovation of the epithelium.
Maxilla and labial palpi.—The development of the tongue (1st maxillæ) is so like that of the antennæ that it scarcely needs description. Beginning at the last moult, the hypodermic contents of the maxillæ is withdrawn in the cephalic cavity under the form of a hollow bud whose base is turned inward. The invagination remains less distinct than in the antennæ; it does not even reach to the anterior part of the œsophagus. The two symmetrical halves of the tongue approach each other and are thrice folded. When the caterpillar stops feeding, they each curve in in the form of an S, remaining lodged under the floor of the mouth (Fig. 613, t).
Underneath are to be seen two other buds, which by an identical process become the labial palpi (Figs. 614, 615, p).
At the anterior part of the head, where the organs are very close together, the envelopes form several folds without any further use 659(Fig. 615, r). The two leaves then fuse together and decay as at the surface of the tarsus.
Finally, in the mandibles and the labrum, there is only a cellular thickening without any invagination.

Fig. 614.—Section through the anterior region of the head of Pieris larva, four days after the 3d moult: o, œsophagus; m, m, 1st maxillæ containing the two imaginal buds of the tongue; p, p, labial palpi; Tr, trachea.
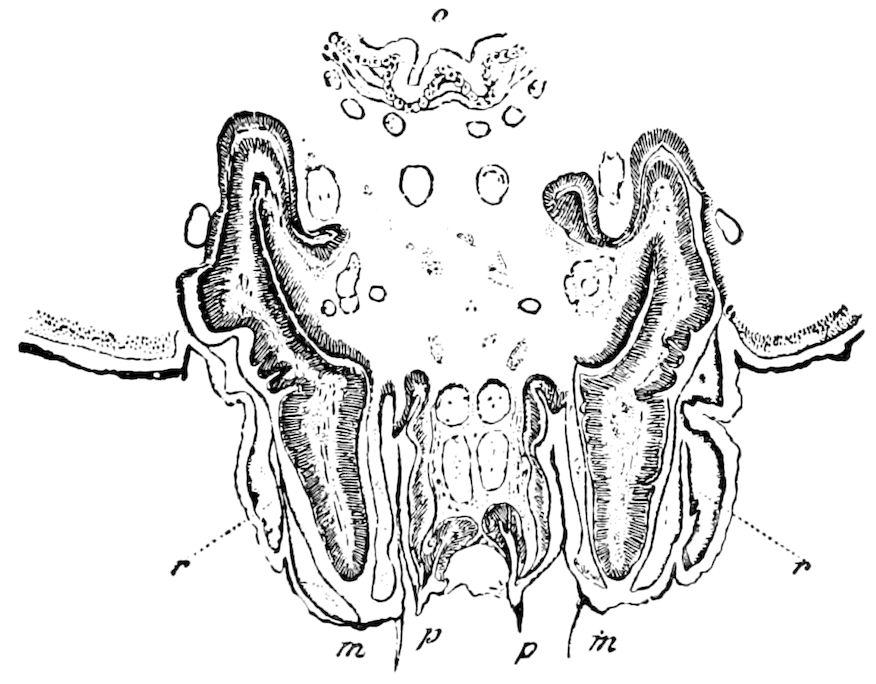
Fig. 615.—Section through the same place as in Fig. 614, 10 days after the 3d moult, the imaginal appendages having grown in size: r, r, caducous folds of the old hypodermis and of the envelopes. Other letters as in Fig. 614.—This and Figs. 613–614, after Gonin.
Process of pupation.—Notwithstanding the great number of persons who have reared Lepidoptera, close and patient observations as to the exact details are still needed. Gonin, who has made the closest observations on Pieris, pertinently asks why the antennæ, which are appendages of the head, are visible in the abdominal region, and why the tongue (maxillæ) is extended between the legs as far as the 3d abdominal segment. To answer these questions he made a series of experiments. Selecting some caterpillars which 660were about to pupate, he produced an artificial metamorphosis by removing the cuticula in small bits. Exposing the appendages in this way, they preserved the position which they are seen to take during growth. Each wing appeared within the limits of the segment from which it grew out (Fig. 610), not extending beyond, as it does in the normal pupa, so that Réaumur was wrong in saying that “the wings are here gathered on each side into a kind of band, which is large enough to lie in the cavity which is between the 1st and 2d segment.” (8e Mém., p. 359.)
All these parts are coated with a viscous fluid secreted by special glands, which hardens after pupation upon exposure to the air. So long as the parts are soft, they can easily be displaced. Gonin drew one of the antennæ like a collar around the head, and one half of the tongue upon the outer side of the wing.
“When pupation is normal, the integument splits open on the back of the thorax, and the pupa draws itself from before backwards. Owing to the feeble adherence which the chitinous secretion gives it, it draws along with it the underlying organs. The legs, antennæ, the two halves of the tongue (maxillæ) retained by their end, each in a small chitinous case, can only disengage themselves from it when in elongating they have acquired a sufficient tension. The curves straighten out and the folds unbend. The chitinous mask of the head in withdrawing from the larval skin follows the ventral line; the tongue and labial palpi free themselves from its median part; the antennæ disengage themselves from the two lateral scales. Between these different appendages a space is left on the surface of the head for the eyes, and on the thorax for the legs. These are not completely extended on account of the lack of freedom of the femoro-tibial articulation; the femur preserves its direction from behind forwards, and the knee in the two first pairs remains at the same height. The wings overlie them and cover the under side of the two basal abdominal segments; their surfaces in becoming united increase much in size.”
As the chitinous frame of each spiracle gradually detaches itself, we see a tuft of tracheæ passing out of the orifice. It is at this moment that the provisional tracheal system is cast off, and it is easy to see that the process is facilitated by the simultaneous elongation of all the appendages. The permanent tracheæ can follow this elongation because they are sinuous, and need only to straighten their curves. It is, however, not the same with the tracheoles, as we have seen in the case of the wings (p. 133), and their extension or stretching is thus explained by a very simple mechanism.
661“The position which the organs assume in the chrysalis is not due to chance, everything is determined in advance, and the microscope shows us that the structure of the hypodermis is specially modified in all the parts which remain external. It is a fact well known to those who rear Lepidoptera that if this normal arrangement is disturbed there are few chances that the perfect insect will survive. A leg lifted up, or an antenna displaced, leaves a surface illy protected against external influences. Almost always this accident causes a drying of the chrysalis.
“Several interesting experiments may be cited as bearing on this subject. If during transformation the chitinous mask of the head is separated from the integument beneath, it is arrested half-way in its development, and the antennæ and tongue are not fully extended. When the case or skin of the caterpillar is drawn, not from before backward, but in the opposite direction, all the appendages of the thorax are placed perpendicularly to the body. Dewitz and Künckel d’Herculais, in stating that the skin of the caterpillar splits open along its whole length, show that they were ignorant of the mechanism; for it is precisely because the chitinous larval skin splits open only in front that it preserves sufficient adherence to the organs beneath to draw them after it in the direction of the abdomen.
“To only read modern authors, one would suppose that the mechanism of pupation had remained hitherto unknown. In reality, it did not escape the notice of Swammerdam or of Réaumur, both of whom have described it with care. The first attached too much importance to the flow of blood, the action of which would be rather to push the organs out than to extend them over the surface of the thorax; the second insists on the movements of the insect. This factor, very admissible in caterpillars, ‘whose under side is situated on a horizontal plane’ (iii, 9e Mémoire, p. 395), cannot be invoked for those which suspend themselves by the tail, as in the genus Vanessa.” (Gonin.)
b. The Hymenoptera
In the Hymenoptera, Ratzeburg was the first to figure and describe the numerous intermediate stages between the larva and pupa, his subjects being the ants, Cynips, and Cryptus, which pass through five stadia before assuming the final pupal shape.
In the bees, as we have observed in the larvæ of Bombus (Proc. Bost. Soc. Nat. Hist., 1866), after hardening a series in alcohol of young in different stages of development, it will be found difficult to draw the line between the different stages since they shade insensibly into each other, those represented in Fig. 616 being selected stages. The head of the incipient semipupa distends the prothoracic segment of the larva whose head is pushed forward and the thoracic segments are much elongated, while the appendages and wings are well developed, and have assumed the shape of those of the pupa. Development both in the head and thorax begins in the most important central parts, and proceeds outwards to the periphery. During this period the “median segment,” or 1st abdominal, has begun to pass forward and to form a part of the thorax.
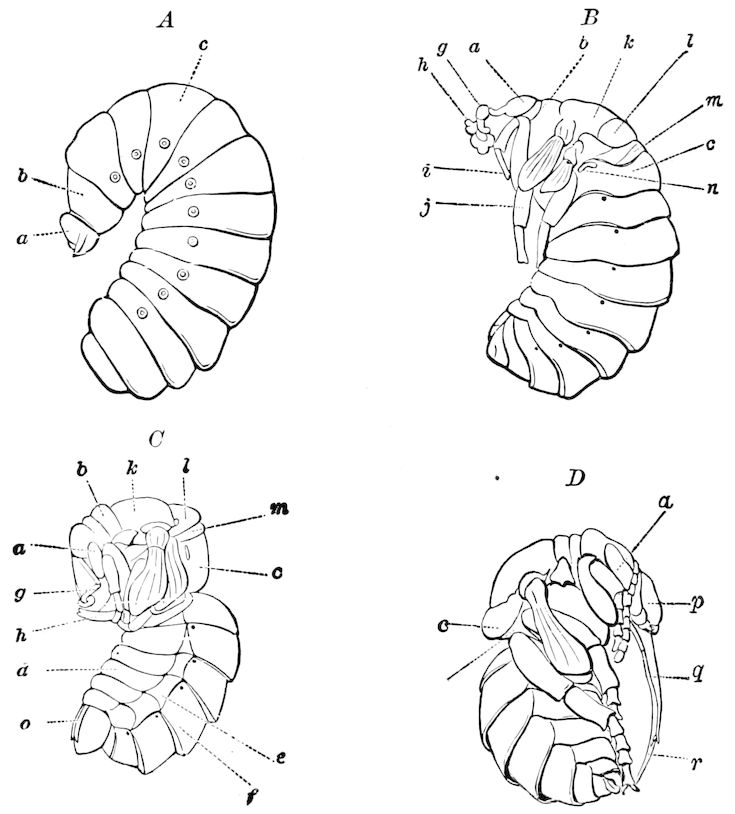
Fig. 616.—Transformation of the bumblebee, Bombus, showing the transfer of the 1st abdominal larval segment (c) to the thorax, forming the propodeum of the pupa (D) and imago: n, spiracle of the propodeum. A, larva; a, head; b, 1st thoracic, c, 1st abdominal, segment. B, semipupa; g, antenna; h, maxillæ; i, 1st, j, 2d leg; k, mesoscutum. l, mesoscutellum; m, metathorax; d, urite (sternite of abdomen); e, pleurite; f, tergite; o, ovipositor; r, lingua; q, maxilla.
In what may be termed the 3d stage (Fig. 616, C), though the distinction is a very arbitrary one, the change is accompanied by a moulting of the skin, and a great advance has been made towards assuming the pupal form. The abdomen is very distinctly separated from the thorax, the propodeum being closely united with the thorax, and the head and thorax taken together are nearly as large as the abdomen, the latter now being shorter and perceptibly changed in form, more like that of the completed pupa, while there are other most important changes in the elaboration of the parts of the thorax, particularly the tergites, and of the head and its appendages. Meanwhile the ovipositor has been completed and nearly withdrawn within the end of the abdomen.
The next to study the transformations of the Hymenoptera was Ganin, who discovered the early remarkable pre-eruciform larvæ, as 663we may call them, of certain egg-parasites (Proctotrypidæ). He discovered the imaginal buds of the wings in the third larva of Polynema (Fig. 185), but his observations, and those of Ayers, need not detain us here, as they have little to do with the subject of the normal metamorphosis of the Hymenoptera, and will be discussed under the subject of Hypermetamorphosis.
To Bugnion we owe the first detailed account of the internal changes in the Hymenoptera, his observations being made on a chalcid parasite, Encyrtus fuscicollis, a parasite of Hyponomeuta. The apodous larva (Fig. 618) moults but once, the next ecdysis being at the time of pupation. It passes through a semipupal stage.
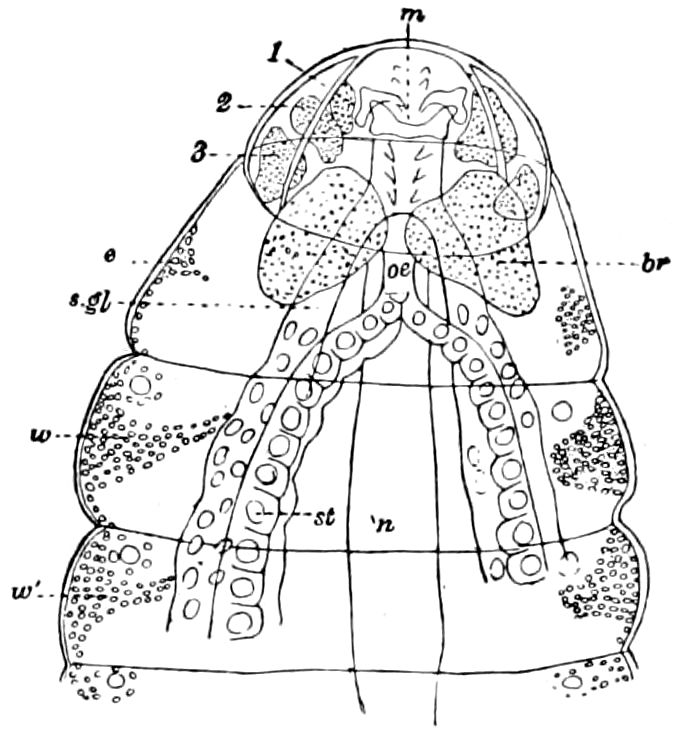
Fig. 617.—Encyrtus larva: 1, 2, 3, ganglia in front of the brain; m, mouth; s. gl, silk-gland; br, brain; n, nervous cord; w, bud of fore, w′, bud of hind, wing.
Bugnion observed in the larva of Encyrtus three pairs of lower thoracic or pedal imaginal buds, two pairs of upper or alary buds, a pair of ocular or oculo-cephalic buds destined to build up all the posterior part of the head, a pair of antennal buds, and three pairs of buds of the genital armature (ovipositor). He also detected the rudiments of the buccal appendages under the form of six small buds (Fig. 619), which do not invaginate, and are not surrounded by a semicircular pad. Also in the abdomen, behind each pair of stigmata, there is a group of hypodermic cells (Fig. 617), which, without doubt, correspond to the wing-buds, but are not differentiated into a central bud and its pad, and does not merit the name of imaginal bud. In fact, except the eye-buds, which are unlike the others, he only observed the imaginal buds of the legs, wings, and ovipositor. The antennal buds are, like those of the buccal appendages, without an annular zone.
The pedal buds were detected in the middle of larval life. They each form a central bud surrounded by a circular thickening. They gradually elongate and become tongue-like and somewhat bent; soon a linear opening or slit appears, forming the mouth of a cavity which communicates with that of the body, allowing the passage into them of the tracheæ, muscles, and nerves, and afterwards of the blood. Finally, the buds grow longer and slenderer, are bent several times, and show traces of the articulations; and soon under the old larval skin, now beginning to rise in anticipation of the moulting, 664we see the coxa, femur, tibia, and tarsus of the perfect insect, the tarsal joints not yet being indicated.
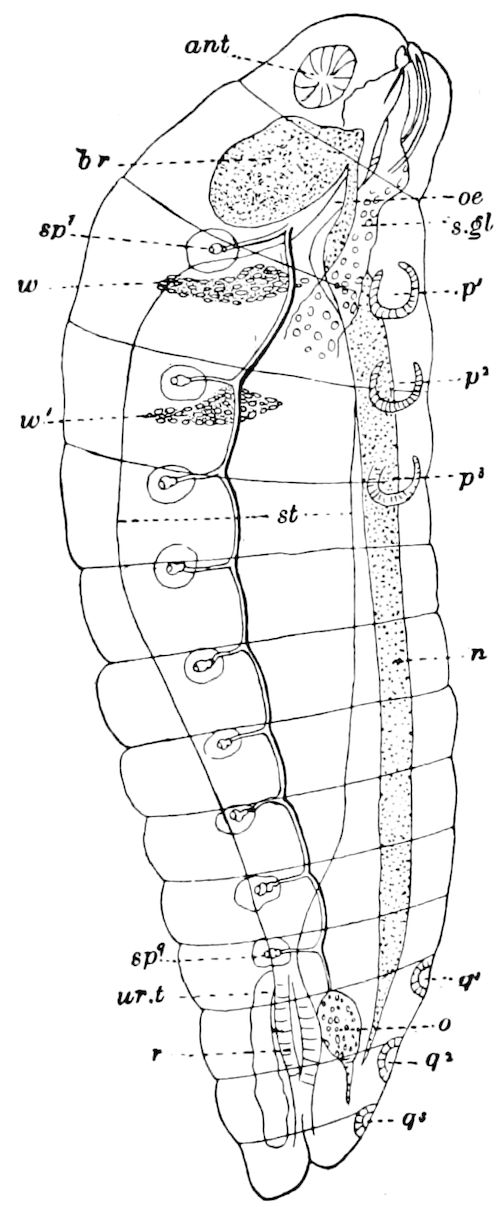
Fig. 618.—Older Encyrtus larva, lateral view, showing the buds of the antennæ (ant), legs, and wings (w, w′): oe, œsophagus; q1, q2, q3, buds of the genital armature; o, rudiment of the sexual gland (ovary or testis); ur.t, urinary tube; st, stomach; i, intestine (rectum); n, ventral nervous cord; r, rectum; sp1-sp9, spiracles.
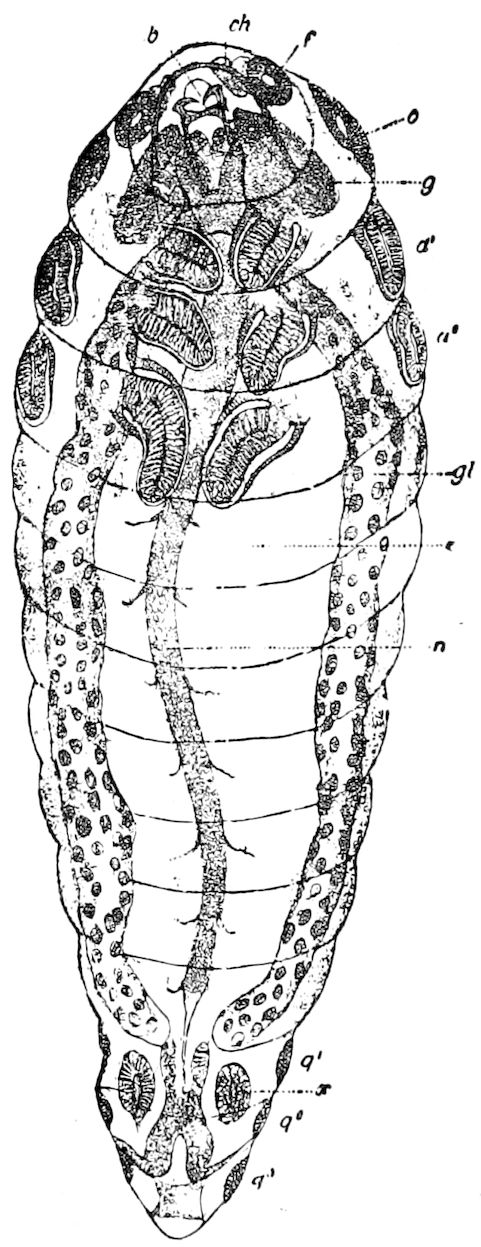
Fig. 619.—A still older larva, ready to transform. The imaginal buds of the antennæ (f), eyes, wings (a1, a2), and legs have become elongated: ch, chitinous arch; b, mouth; o, eye-bud; g, brain; e, stomach; x, rudiment of the sexual glands (either the ovary or testis).—This and Figs. 617 and 618, after Bugnion.
The wing-buds (a1, a2) appear at the same time as those of the legs, as racket-shaped masses of small cells situated directly behind the 1st and 2d pair of stigmata, in contact with the tissue ensheathed by the corresponding tracheal vesicle (Fig. 618). Afterwards they have exactly the form of those of the Lepidoptera (Fig. 619).
The proliferation of the hypodermis is not limited to the thorax, but takes place at corresponding points in the first seven abdominal 665segments. These abdominal agglomerations of cells do not give rise to true buds, but serve simply to reconstitute the hypodermis of the abdominal segments at the time of metamorphosis.
Ocular or oculo-cephalic buds.—The eye of insects, as is well known, is a modification of a portion of the integument, the visual cells being directly derived from the hypodermis, the cornea being a cuticular product of this last, like chitinous formations in general.
The ocular buds appear towards the end of larval life as a simple mass of hypodermic cells, and form a compact layer on the dorsolateral face of the prothoracic segment, and clothe the cephalic ganglion or brain like a skull-cap. The central portion only is destined to form the eye, while the peripheral pad, continuing to thicken, gives rise to a voluminous and rounded mass, which meets on the median line that of the opposite side, and forms the integument of all the posterior part of the head.
Bugnion also observed on the median line a group of small hypodermic cells which he regarded as the rudiment of the anterior ocellus, but he did not detect those of the posterior ocelli.
The antennal buds.—These appear at an early date under the cuticle of the head, as two distinct rounded cellular masses, with a central cavity, but no annular zone (Fig. 619, f). Each one grows longer in a transverse sense, and its summit, extended from the outer side, curves downward. It now forms a hollow tube folded at the end, and terminated by a disk whose centre is perforated (Fig. 619, f). Afterwards, when the larva is ready to transform, it grows longer, becomes folded on itself in its cavity, and, passing beyond on each side the limits of the larval head, encroaches on the prothoracic segment.
The buds of the buccal appendages.—Towards the end of the larval period, the buds of the mouth-parts appear as small digitiform projections, situated on each side and below the mouth. Formed of small epithelial cells pressed against each other, they are all directed anteriorly, and possess no furrow or pad.
The 2d maxillæ (labium) is formed of two separate parts. The imaginal buds of the lower lip appear on each side of the median line, with a fissure indicating the differentiation of the palpus. On each side are to be seen the 1st maxillary buds, bearing each a rudimentary palpus, and, farther in front, the buds of the mandibles.
The buds of the ovipositor.—The six stylets of the ovipositor arise from six small imaginal buds which become visible in the second half of the larval period, on each side of the median line, on the lower face of the three last segments (Fig. 620, q1, q2, q3). 666The bud is differentiated into a central discoidal bud, a furrow, and a marginal, rather thick swelling or pad. Afterwards, these buds elongate and form small papilliform projections directed backwards (Fig. 621); but only during the pupal period do they, as already observed in Bombus, approach each other and assume their definite shape as an ovipositor.
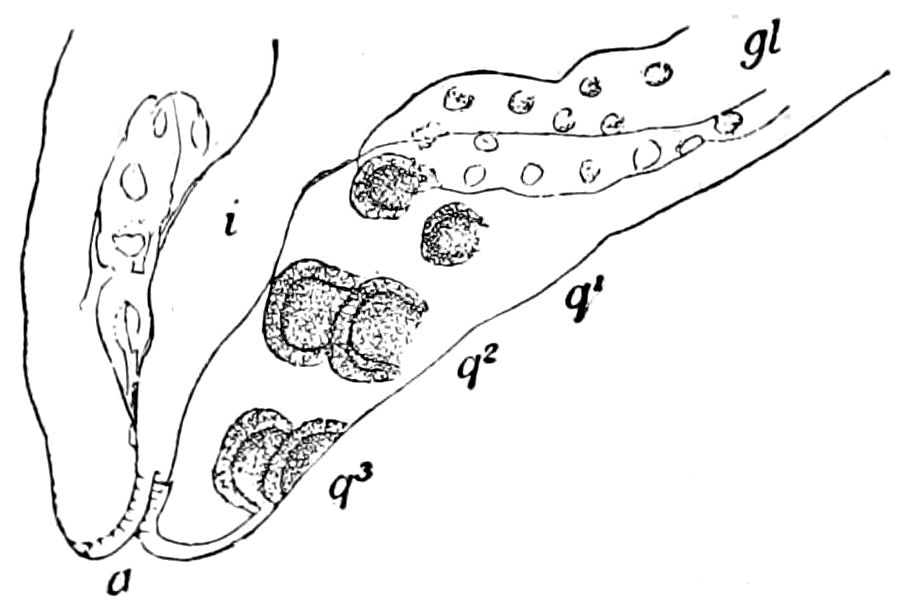
Fig. 620.—End of larva of Encyrtus of 2d stage, showing the three pairs of imaginal buds of the ovipositor q1, q2, q3.
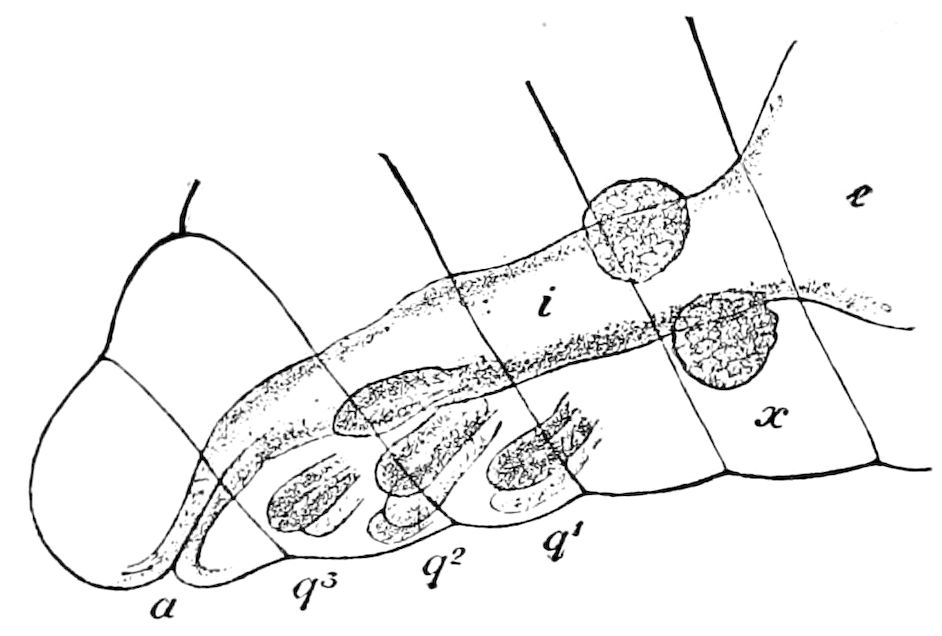
Fig. 621.—The same in an older larva ready to transform: i, intestine; x, genital gland; a, anus.—After Bugnion.
Finally, Bugnion states that while metamorphosis in the Hymenoptera is less highly modified than in the Muscidæ, it is more marked than in the Coleoptera and Lepidoptera. In these orders the pupa moves the abdomen, but in Hymenoptera it is absolutely immovable throughout pupal life, as long as the integument is soft.
DEVELOPMENT OF THE IMAGO IN THE DIPTERA
The flies, particularly the Muscidæ and their allies (Brachycera), are the most highly modified of insects, their larvæ having undergone the greatest amount of reduction and loss of limbs, this atrophy involving even most of the head. The following account has been prepared in part from the works of Weismann, Ganin, Miall, and Pratt, but mostly from the excellent general summarized account given by Korschelt and Heider.
In the holometabolic orders of insects, with their resting pupal stage, during which no food is taken, the entire activity of life 667seems to be turned to deep-seated and complicated internal developmental processes. These inner changes involve an almost complete destruction of many organs of the larva, and their renewal from certain germs (the imaginal buds) already present in the larva, as will be seen in the highly modified Muscidæ. Only a few larval organs become directly transferred into the body of the pupa and imago. Such are the rudiments of the genital system. The heart also, and the central portion of the nervous system, suffer only slight and unimportant, almost trivial, internal changes. On the other hand, most of the other organs of the larva become completely destroyed: the hypodermis, most of the muscles, the entire digestive canal with the salivary glands; while their cells, under the influence of the blood corpuscles (leucocytes), which here act as phagocytes, fall into pieces, which are taken up by them and become digested. Simultaneously with this destructive, histolytic process, the new formation of the organs by the imaginal buds, already indicated in the embryo, is accomplished in such a way that the continuity of the organs in most cases remains unimpaired. This process of transformation can only be understood by considering that of the embryonal germs of the organs, (1) only a part is destined for the use of the larva in growth, and for the performance of certain functions which exhaust themselves during larval life, so that it is no more capable of farther transformation, and finally becomes destroyed; while (2) a second part of the embryonal germs or rudiments persists first in an undeveloped condition, as imaginal buds, in order to undertake during the pupa stage the regeneration of the organs.
Though Swammerdam knew that the rudiments of the wings were already present under the skin of the larvæ, we are indebted, for our present knowledge, to the thorough and profound investigations of Weismann on the metamorphosis of the Diptera, and also to the researches of Ganin and others who have worked on the pupæ of Muscidæ, in which the development is most complicated and modified. In the more generalized and primitive Diptera, such as Corethra, the processes of formation of the pupa and imago are much simpler than in the muscids and Pupipara. These processes are still simpler in the Lepidoptera and Hymenoptera, and for this reason we have given a summary of what has been done on these organs by Newport, Dewitz, and especially by Bugnion.
Our knowledge of this subject is still very imperfect, only the more salient points having been worked out, and, as Korschelt and Heider state, there is still lacking certain proof as to how far the relations of the internal changes known to exist in the Muscidæ also apply to other orders of insects, though it must be considered that in the pupa of Lepidoptera, Hymenoptera, perhaps also the Coleoptera, and we would add in the Neuroptera as well as the male Coccidæ, very similar metamorphic processes take place.
a. Development of the outer body-form
The form of the imago is completely marked out in the pupa, so that the transition from the pupa to the imago is comparatively slight and only depends on the modification and development of the parts already present.
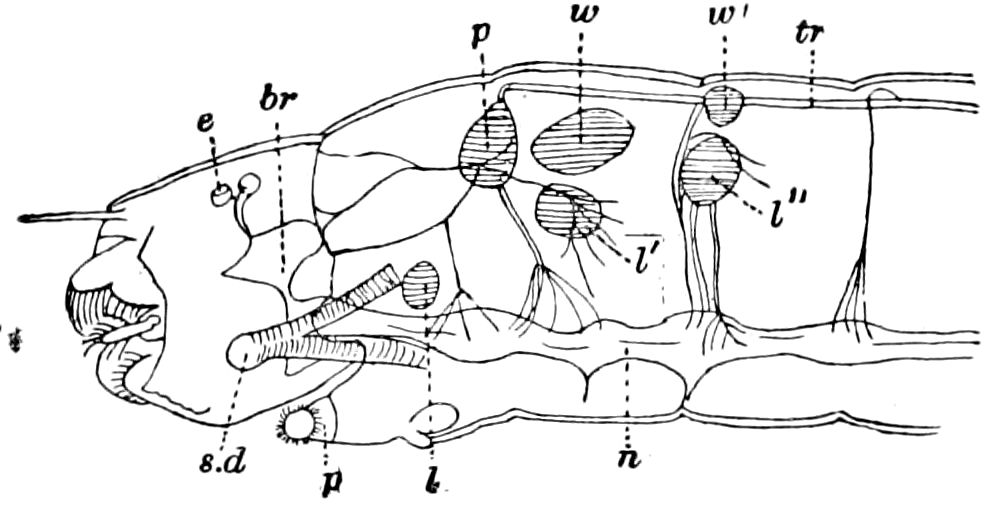
Fig. 622.—Anterior part of young larva of Simulium sericea, showing the thoracic imaginal buds: p, prothoracic bud (only one not embryonic); w, w′, fore and hind wing-buds; l, l′, l″, leg-buds; n, nervous system; br, brain; e, eye; sd, salivary duct; p, prothoracic foot.—After Weismann.
In most cases the modification in question consists of the changes occurring during the passage from the larval form to the imago, the reformation of parts already present being most marked, while the new rudiments only participate in a limited way in the process. Thus, for example, the head of the caterpillar together with the antennæ and mouth-parts, also the thoracic limbs, pass directly and unchanged from the larva into the pupa. The compound eyes and the wings are, however, new formations, the latter arising from imaginal buds. The same is the case with many other Heterometabola, where the passage of the larva into the pupa in general is due to a transformation of parts already present. The changes in the brain, the fusion of certain ganglia of the ventral nervous cord, the changes in the abdomen, involving the reduction in the number of segments and the remodelling of the end of the body, and the formation of the ovipositor or sting, and in the higher Hymenoptera the transfer of the 1st abdominal segment to the thorax, and the origin of the genital armature,—all these should here be taken into account.
It should be observed that in every case where the larvæ are footless, as in Diptera, all the Hymenoptera except the phytophagous ones and certain coleopterous larvæ, the limbs of the imago stage are, in the earliest stages, indicated as new structures in the form of imaginal buds.
Formation of the imago in Corethra.—Corethra may serve as an example of such a relatively simple metamorphosis. Its larva belongs to the group of eucephalous dipterous larvæ. The head of the perfect insect is already indicated in the larva, and its parts, with certain modifications, pass directly into the pupa. The compound 669eyes, and this is a rare exception among the Holometabola, are present in the larva. On the other hand, the thoracic legs, the wings, and halteres are developed out of new rudiments which are present in the last larval stage, before pupation. Each thoracic segment has four of them, two ventral and two dorsal (Fig. 622); the ventral buds becoming the legs. Of the dorsal pairs, that of the mesothorax develops into wings, that of the metathorax into halteres, while from the corresponding rudiments of the prothorax in Corethra arise the stigma-bearing dorsal or respiratory processes of the pupa, and in Simulium a tuft of tracheal gills (Fig. 623, ra; see also Fig. 582).
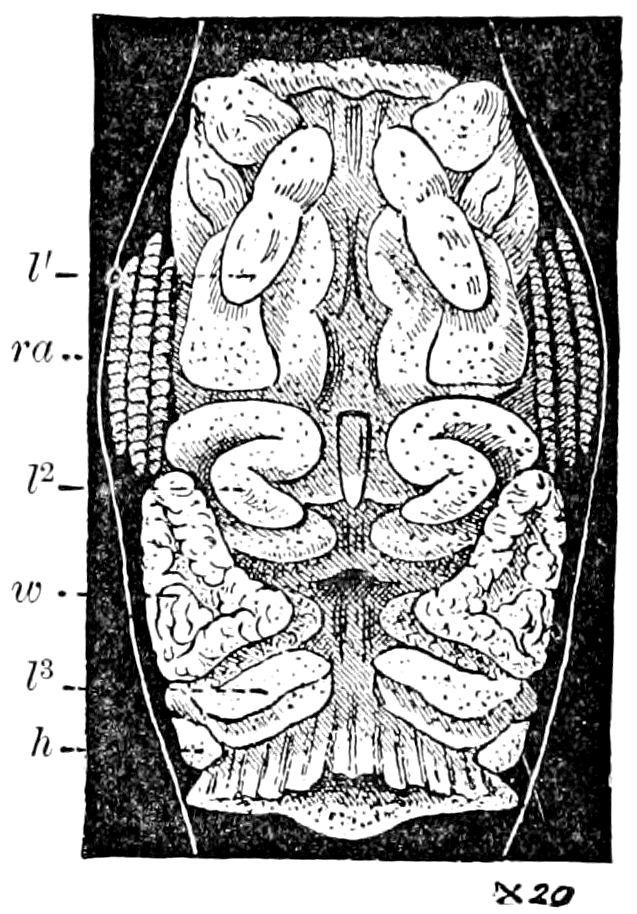
Fig. 623.—Late larva of Simulium, showing the rudiments of the pupal structures within the larval skin: l1, l2, l3, fore, middle, and hind legs of the fly; ra, respiratory appendages of pupa; w, wing of fly; h, halter of fly.—After Miall.
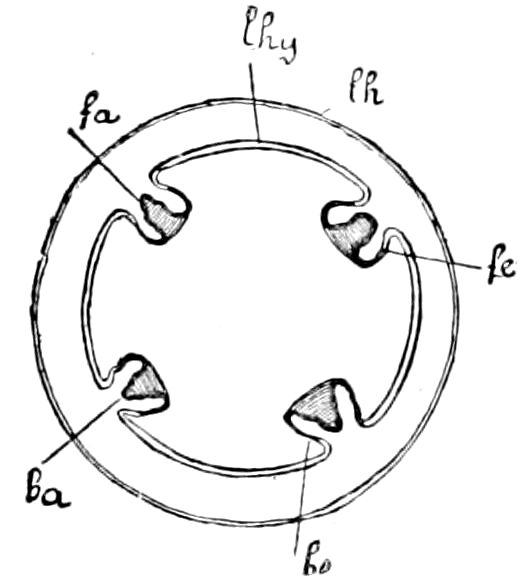
Fig. 624.—Imaginal buds in larva of Corethra (diagrammatic cross-section of thorax): invaginations (fe and be) of the larval hypodermis (lhy) in whose bases the rudiments of wings (fa) and legs (ba) arise; lh, chitinous integument of the larva.—After Lang.
These imaginal buds may be regarded as evaginations of the outer surface of the body. The only difference is that the buds of the appendages as a whole seem sunken below the level of the surface of the body, being situated at the bottom of an evagination, as in the buds of the head and trunk in the Pilidium larva of nemertean worms, and in the rudiments of the lower surface of the body of Echinus present in the pluteus larva.
The lumen of the invagination in which the appendages of Corethra (and other Holometabola) are situated is called by Van Rees the peripodal cavity, and the external sheath bordering it, which is naturally continuous with the hypodermis of the body, the peripodal membrane (Fig. 636, p).
We must adopt the view that the rudiments of the appendages (imaginal buds) are from the first divided into ectodermal and mesodermal portions, which are derived from the corresponding germ-layers of the larva. The ectoderm of the rudiments of the appendages is continuous with the peripodal membrane, and through it with the hypodermis. Weismann was inclined to derive the organs (tracheæ, muscles, etc.) developing within the germs of the appendages from a 670hypertrophy of the neurilemma of a nerve passing down from within into the imaginal bud, and held that nerves and tracheal branches soon after passed into the inner surface of the imaginal bud. (Korschelt and Heider.)
When the imaginal buds of the appendages enlarge, then the peripodal membranes become correspondingly distended, and the limbs within assume a more or less crumpled position, and in Corethra are spirally twisted, while the rudiments of the wings are folded. The completion of the rudimentary limbs is accomplished simply by their passing out of the invagination in which they originated. The limbs thus gradually become free, the peripodal membrane is seen to reach the level of the rest of the hypodermis and become a part of it, and the base of the extremity is no longer situated in a cavity.
The internal organs of Corethra undergo but to a slight degree the destruction (histolysis) which is so thoroughgoing in the Muscidæ. Kowalevsky states that in the mid-intestine of Corethra a histolysis of the larval and reconstruction of the imaginal epithelium goes on in the same way as has been described in Musca. Most of the larval organs pass without histolytic changes directly over into those of the pupal and imaginal stages; the muscles in general are also unchanged, but those of the appendages and wings are made over anew. The last arise, according to Weismann, in the last larval stage from strings of cells which are already present in the embryo.
When we consider how insignificant the internal transformations are during the metamorphosis of the Tipulidæ, of which Corethra serves as an example, we can scarcely doubt that we here have before us conditions which illustrate the passage between an incomplete and a complete metamorphosis. Thus, among other things, should be mentioned the short duration of the pupa stage and the activity of the pupa, as also the early appearance of the germs of the compound eyes, a character which Corethra has in common with the Hemimetabola. (Korschelt and Heider.)
Formation of the imago in Culex.—In respect to the formation of the imaginal head, Culex is still more primitive than Corethra. Miall and Hammond find from Hurst’s partly unpublished descriptions and preparations that there are no deep invaginations for the compound eyes or antennæ of the imago.
“The compound eye forms beneath the larval eye-spots, and is at first relatively simple and composed of few facets. The number increases by the gradual formation of partial and marginal invaginations, each of which forms a new element. The imaginal antenna grows to a much greater length than that of 671the larval antenna, and its base is accordingly telescoped into the head, while the shaft becomes irregularly folded.[115] Culex, though more modified than Chironomus in many respects, e.g. in the mouth-parts, is relatively primitive with respect to the formation of the imaginal head, and shows a mode of development of the eye and antenna which we may suppose to have characterized a remote and comparatively unspecialized progenitor of Chironomus.”
Formation of the imago in Chironomus.—The development of the head of the imago of Chironomus dorsalis has been discussed by Miall and Hammond. The invaginations which give rise to the head of the fly could not be discovered even in a rudimentary state until after the last larval moult.
“Weismann has given reasons for supposing that invaginated imaginal rudiments could not come into existence before the last larval moult in an insect whose life-history resembles that of Corethra or Chironomus. If the epidermis were invaginated in any stage before the ante-pupal one, the new cuticle, moulded closely upon the epidermis, would become invaginated also, and would appear at the next moult with projecting appendages like those of a pupa or imago. This is actually the way in which the wings are developed in some larval insects with incomplete metamorphosis. In Muscidæ the invaginations for the head of the imago have been traced back to the embryo within the egg,[116] but the almost total subsequent separation of the disks from the epidermis renders their development independent of the growth of the larval cuticle and of the moults that probably take place therein.”
The pupal and imaginal cuticles do not follow at all closely the larval skin, but, says Miall, become at particular places folded far into the interior. “The folds which give rise to the head of the fly are two in number and paired. They begin at the larval antenna on each side of the head, and gradually extend further and further backwards. The object of the folds is to provide an extended surface which can be moulded, without pressure from surrounding objects, into the form of the future head. On one part of each fold the facets of the large compound eyes are developed; another part gives rise to the future antenna, a large and elaborate organ, which springs from the bottom of the fold, and whose tip just enters the very short antenna of the larva. The folds for the head ultimately become so large that the larval head cannot contain them, and they extend far into the prothorax. Here a difficulty occurs. If the generating cuticle of the prothorax were also to be folded inwards, the future prothorax would take a corresponding shape. But the prothorax of the fly has a form dictated to it by the limbs which it bears and by the muscles to which it gives attachment. These call for a great reduction in its length, and a peculiar shape, which it is 672not here necessary to describe. It will be enough to realize that the epidermis of the future prothorax cannot be sacrificed to the folds which are to give rise to the head of the fly. All interference between the two developing structures is obviated by the provision of a transverse fold, which pushes into the prothorax from the neck, and forms a sort of internal pocket. The floor of the pocket forms two longitudinal folds, which prolong the folds originating in the larval head. The roof of the pocket shrinks up and forms the connection between the head and thorax of the fly. Ultimately the head-part is drawn out, leaving the prothoracic structures unaffected.”[117]
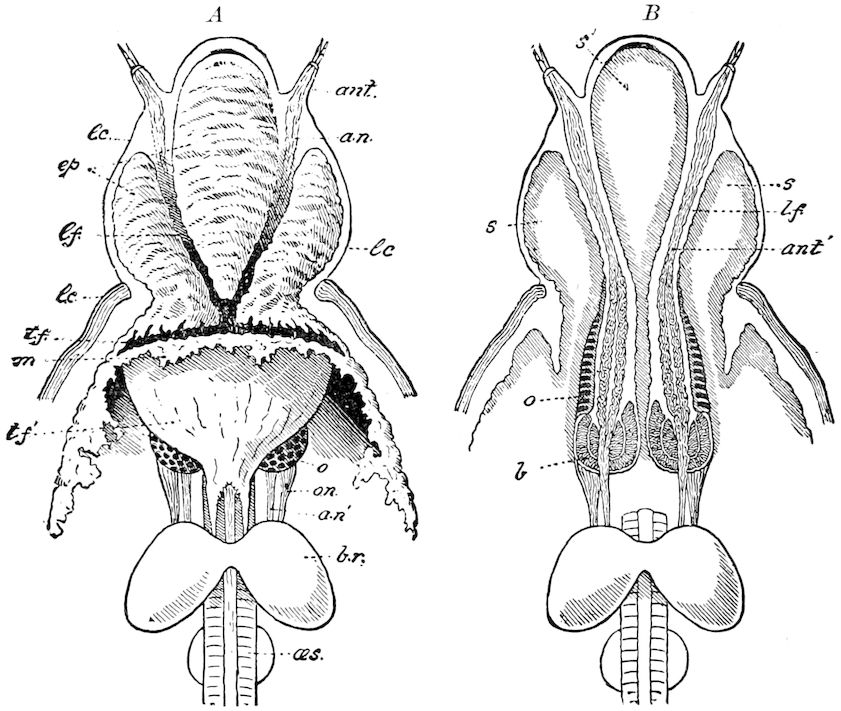
Fig. 625.—Process of formation of the parts of the head of the fly in the larva of Chironomus (male): A, the new epidermis thrown into complicated folds which have been cut away in places to show the parts within. B, the same parts in horizontal section; lc, larval cuticle; tf, transverse fold; tf′, upper wall of the same; m, cut edge of new epidermis; ant, larval antenna; an, nerve to the same; ant′, antenna of fly; lf, longitudinal fold; o, eye of fly; on, optic nerve; an′, root of antennary nerve; br, brain; œs, œsophagus; b, bulb of antenna of fly; s, s, s′, blood-spaces.—After Miall.
The development of the head of the fly of Chironomus appears, as Miall and Hammond state, to be intermediate between the groups Adiscota and Discota of Weismann; i.e. “between the types in 673which the parts of the head of the fly are developed in close relation to those of the larva, and the types in which deep invaginations lead apparently to the formation of similar new parts far within the body, the seeming independence of the new parts being intensified by thoroughgoing histolysis,” and they suggest that possibly types may be discovered intermediate between Chironomus and Muscidæ.
Fig. 626.—A, B, C, D, diagrams of transverse sections showing the development of the wings, legs, and the imaginal hypodermis of muscid flies from the imaginal buds of the larva during metamorphosis: lh, chitinous integument of larva from which the underlying hypodermis (lhy) has withdrawn; iid, imaginal buds of wings, iiv, of legs; is, the cords connecting them with the hypodermis; fl, wing-germs; b, leg-germs; ihy, imaginal hypodermis spreading out in D from the imaginal buds. The imaginal rudiments of the hypodermis are indicated by thick, black outlines, the larval hypodermis by two thin, parallel lines.—After Lang.
We are now prepared to consider the extremely complicated changes, in the Muscidæ, leaving out of consideration the origin of the wings from imaginal buds, which has already been discussed on pp. 126–137.
Formation of the imago in Muscidæ.[118]—In the flesh, and undoubtedly the house, and allied flies the germs or imaginal buds of the legs and wings arise in the same way as in Corethra. But in the Muscidæ, the buds are situated far within the interior of the body, the peripodal cavities appear closed, and the peripodal membrane stands in connection with the hypodermis merely by means of a delicate thread-like stalk. This connecting cord, which was first detected by Dewitz, and whose interpretation was entirely right, shows in its interior, as Van Rees proved, a narrow cavity.
Though the earliest stages in the development of imaginal buds in the embryo of the Muscidæ are still unknown, yet we shall not go far astray if we refer them, like the imaginal buds of Corethra, to hypodermal invaginations. We must, then, regard the stalk-like 674connection just mentioned as the long drawn-out neck of this invagination.
In general, the development of the appendages (Figs. 626, 627) goes on as described in Corethra. The rudiments of the legs enlarge and show at an early date the first traces of the later joints. They are so packed in the peripodal cavities that the single joints of the extremities appear as if pushed in “like the joints of a travelling cup.” (Van Rees.) The evagination of the completely formed buds of the limbs, which occurs on the first day after the beginning of pupation, goes on in such a way that the stalk of the imaginal bud (Figs. 626, B; 627, B) shortens, while its cavity widens so that the limbs finally, as in Corethra, pass out through the widely opened mouth of the peripodal invagination, which at the same time gradually completely disappears. The peripodal membrane is converted into a thickened part of the hypodermis in the region adjoining the base of the leg, and from this thickened hypodermal portion, the formation of the hypodermis of the entire imaginal thorax goes on, as the larval hypodermis is gradually destroyed.
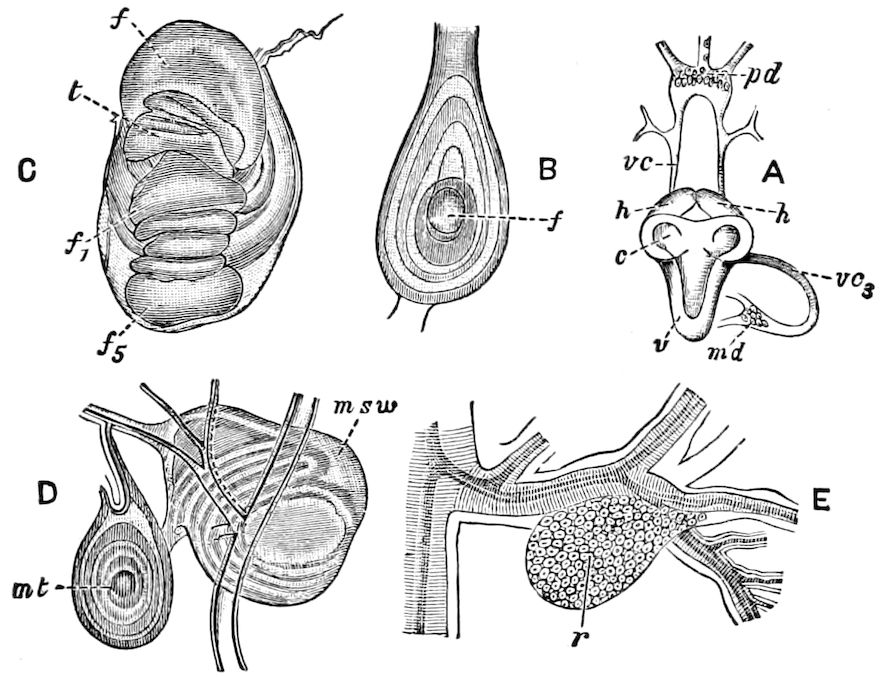
Fig. 627.—Imaginal buds of muscid flies in process of development: A, brain (c) and ventral ganglion (v) of a larva, 7 mm. long, of C. vomitoria; h, head-rudiment; rc, portion of ventral cord; pd, prothoracic rudiment; vc3, third nerve; md, mesothoracic rudiment. B, mesothoracic rudiment more advanced, in a pupa, just formed, of Sarcophaga carnaria, showing the base of the sternum and folds of the forming leg, the central part (f) representing the foot. C, the rudimentary leg of the same more advanced; f, femur; t, tibia; f1, f5, tarsal joints. D, two buds from a larva, 20 mm. long, of Sarcophaga, attached to tracheæ; msw, mesonotal and wing-germ; mt, metathoracic rudiment. E, r, mesothoracic germ of a 7 mm. long larva attached to a tracheal twig.—After Weismann and Graber, from Sharp.
We must here settle the question as to the first origin of the mesodermal portions of the rudiments of the appendages. We can already distinguish in the imaginal buds of the fully grown muscid larva a clear separation between an ectodermal and an inner mesodermal part. Ganin derived the mesodermal 675part through a sort of differentiation and separation of the innermost layer of the ectodermal part, and Van Rees has, in general, confirmed this view. Kowalevsky, on the other hand, is inclined to the view that the mesodermal part of the imaginal bud is derived from the embryonal cells of the mesoderm. He finds scattered throughout the mesoderm, under the hypodermis of the larva, so-called wandering cells (Fig. 632, A, w), which are different in appearance from the leucocytes and from the elements from which the formation of the mesodermal parts of the imaginal rudiments proceed. Kowalevsky is inclined to believe that there are in each segment rudiments of the imaginal mesoderm, but which are so delicate and indifferent that we cannot find them in the first stages of their origin. From these mesodermal imaginal rudiments the above-mentioned wandering cells of the mesoderm are derived, which afterwards come into connection with the ectodermal portion of the imaginal buds.
Still more complicated and difficult to understand is the development of the head-section of muscids. We must remember that in muscid larvæ the head-section exists in its most rudimentary form, being the result of extreme modification and degeneration. The small size of the head is also due to the fact that it is more or less retracted within the thoracic region. Then, as shown by the researches of Weismann, in the last embryonal stages, the forehead, mandibles, and the whole region of the head around the mouth invaginate and form a sunken cavity (Fig. 628, p), in which the chitinous supports of the hooks characteristic of muscid larvæ are soon developed. This sunken part of the head, at whose inner end is the œsophagus, is called by the not entirely appropriate name of “pharynx,” and it must at present be remembered that the hollow space thus named is not a part of the digestive canal. It is an invaginated section of the head, and the formation of the head of the imago mainly depends on the evagination of this region.
The first rudiments of the most important parts of the head (eyes, antennæ, and forehead), occur in the youngest larvæ as paired masses of cells which lie in the thorax next to the two halves of the brain (for this reason called by Weismann “brain-appendages”), which are from their first origin connected with the pharynx, and may be regarded as the imaginal buds of the head. These appear very soon in later stages in the shape of elongated sacs widening at the hinder end (Fig. 628, A and B, h), which from their origin are to be regarded as evaginations of the pharynx. Very soon epithelial thickenings appear in the wall of this sac-shaped brain-appendage, in which the rudiments of the parts of the future head may be recognized.
Disk-shaped thickenings in the hinder widened part of the brain-appendage form the rudiments of the compound eyes, which therefore may be called the eye-buds. On the basal surface of the 676eye-buds is situated a nervous expansion which is connected by a nerve with the supraœsophageal ganglion. This nerve becomes the optic nerve of the perfect animal, while the optic ganglion is clearly separated from the brain.
In the anterior, more cylindrical or tube-like part of the brain-appendage we find the “frontal buds” (ss), on which the antennal rudiments (at) soon bud out, in exactly the same way as the rudiments of the limbs arise from the imaginal buds.
Fig. 628.—Diagrammatic representation of the position of the imaginal buds in the larva (A) and pupa (B) of Musca (the wing rudiments omitted): as, eye-buds; at, antennal germs; b1-b3, germs of the legs; bg, central ganglionic cord: g, brain; h, so-called frontal appendage (Hirnanhang): m, peripodal membrane; o, opening of the frontal appendage into the pharynx; oe, œsophagus; p, so-called “pharynx”; r, rudiment of the proboscis; ss, frontal bud; st, stalk-like connection of the peripodal membrane with the hypodermis; I-III, 1st, 2d, and 3d thoracic segments.—Adapted from Van Rees, by Korschelt and Heider.
Originally (Fig. 628, A) the brain-appendages lie tolerably far behind in the thorax of the larva, so that they connect the hindermost part of the wall of the pharynx with the foremost section of the brain, which they surround in the form of a mushroom. Afterwards, however, subsequent to pupation, they move, together with the central nervous system, farther forward (B), whereby they (if we have correctly understood the descriptions of Weismann and Van Rees) laterally surround the pharynx with their anterior end, which is somewhat ventrally bent. At the same time, there becomes established a gradually widening communication (B, o) between the brain-appendage and the pharynx, which soon extends in the form of a lateral pharyngeal fissure along the entire length of the brain-appendage. As a result, the cavity of the brain-appendage 677and the pharynx so completely unite that the two soon form a single sac, the head-sac or vesicle (Fig. 6–9, k). The walls of this head-vesicle are the later head-wall, the most important parts of which can now be recognized (the antennæ, eyes, rudiments of the beak). It is now necessary that the head-vesicle (Fig. 629, +, +) be, by the eversion of the pharynx, turned outward in order that the head of the pupa may be completed. By this eversion of invaginated parts, the former mouth-opening of the pharynx becomes a neck-section (Fig. 629, +, +) by which head and thorax are now united. (Korschelt and Heider.)
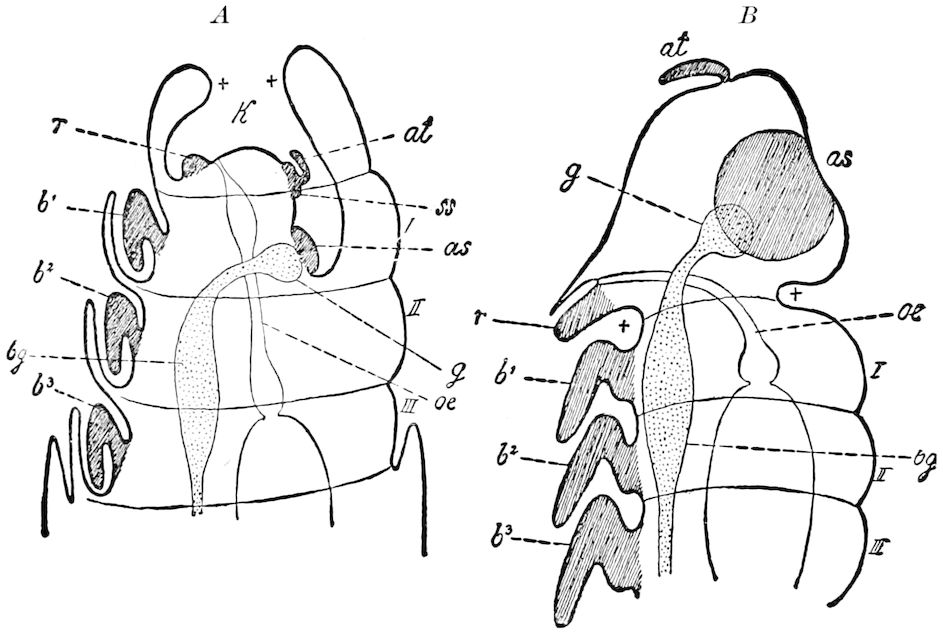
Fig. 629.—Diagram of the changes to pupa of Musca before imago appears; the wing-germs not drawn: as, eye-buds; at, antennal germs; b1-b3, leg-germs; bg, ventral nerve-cord; g, brain; k, head-vesicle (originating from the union of the pharynx with the hypophysis, Hirnanhängen); oe, œsophagus; r, germ of the proboscis; ss, germ of the forehead; I, II, III, 1st, 2d, and 3d thoracic segments.—Based on Kowalevsky and Van Rees, with changes, after Korschelt and Heider.
The cause of the eversion of the head-vesicle, which Weismann directly observed, appears to be due to an increase of the inner pressure through a contraction of the hinder parts of the body. The anterior end of the œsophagus now becomes turned down ventrally corresponding to the conformation of the head of the imago.
It has been shown that the so-called pharynx is only an invaginated part of the outer surface of the larval head. The brain-appendage Korschelt and Heider consider to be the diverticulum of this invagination, in which the single parts of the body lie in an invaginated state. They may throughout be compared to the rudiments of the thoracic limbs. All these imaginal buds have been traced back to the invaginated parts of the outer surface of the body, i.e. the ectoderm.
678It should be borne in mind that the process of development of the head of the highly-modified Muscidæ is much more complex than in the more primitive Diptera.
In their essay on the development of the head of the imago of Chironomus, Miall and Hammond arrange the dipterous types thus far examined, in the order of complexity of the invaginations which give rise to the head of the imago, in the following order:—
- 1. Culex. Relatively simple. Invaginations of the imaginal buds, shallow. 2. Corethra, Simulium. } Intermediate. 3. Chironomus, Ceratopogon. } 4. Muscidæ. Relatively complex. Invaginations deep, and apparently, but not really, unconnected with the epidermis.
b. Development of the internal organs of the imago
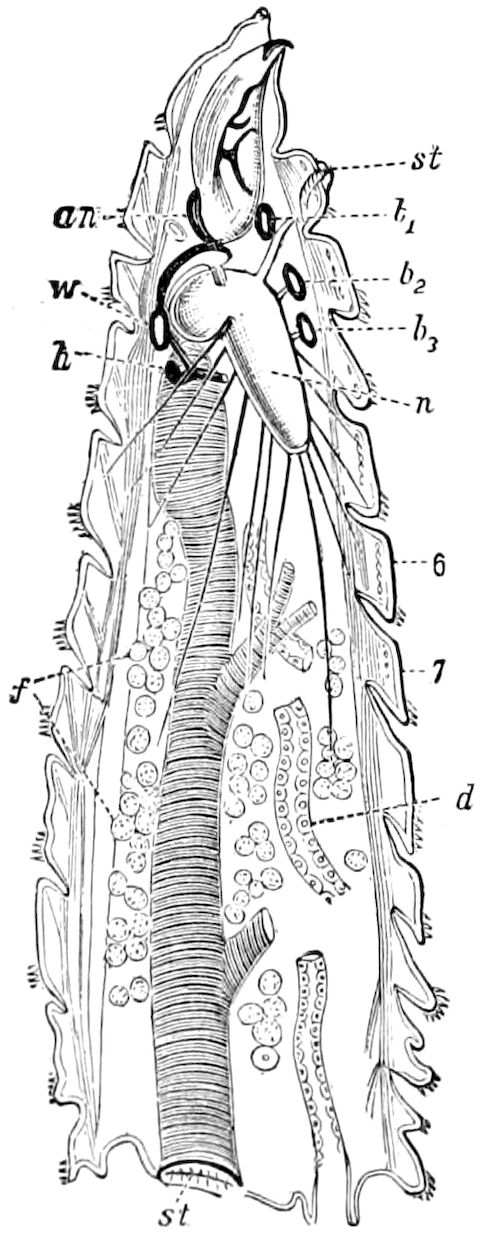
Fig. 630.—Median longitudinal section through larva of blow-fly during the process of histolysis: an, antenna; between an and w, rudiments of eye; w wings; h, halteres; b1-b3, legs; f, fat-body; d, middle of intestine; n, ganglia; st, stigma; 6, 7, 6th and 7th body-segments.—After Graber, from Sharp.
It has already been observed that most of the organs of muscid larvæ (and this applies to most Diptera, Lepidoptera, Coleoptera, and Hymenoptera) are destroyed through the action of leucocytes, and that their reformation is accomplished by definite groups of embryonal cells, the imaginal buds or folds. Destruction and rebuilding occur during the pupa stage in such a way that in many cases while this process is going on the continuity of the organs does not seem to be disturbed. These transformations especially concern the hypodermis, the digestive canal, the muscles, the fat-body, and the salivary glands.
The transformation of the tracheal system is only partial, being in part a simple process of regeneration through cell-division. Slighter changes affect the heart, the central nervous system, and the reproductive system (Fig. 630).
Fig. 631.—Diagram of the formation of the imaginal hypodermis on the abdomen of Muscidæ: hi, imaginal buds of the hypodermis; lh, larval hypodermis.—After Lang.
Fig. 632.—Section through the abdominal bud of the hypodermis of Musca: A, of the larva; B and C, of the pupa; h, larval hypodermis; h′ separated portion of the same attacked by phagocytes; i, imaginal bud; k, phagocytes with what are called cell-wrecks or fragments (so-called granulated cells); k′, phagocytes enclosing hypodermal nuclei; m, mesoderm-germ of the imaginal bud; w, wandering cells.—After Kowalevsky, from Korschelt and Heider.
The hypodermis.—The hypodermis of the imago arises through an extension of the ectodermal part of the imaginal buds. We have already mentioned this for the thorax. As the appendages of the thorax in the pupa gradually attain perfection, the hypodermis layer spreads from the place of their insertion, the layer consisting of numerous small cells whose origin we must refer to the peripodal membrane. This layer continues to spread over the surface of the pupal thorax, while at the same time the area of the larval hypodermis, consisting of large cells, is seen to diminish. Hence the thin edge of the newly-formed hypodermis (Fig. 631, hi) slowly grows into the space between the superficial cuticula and the larval hypodermis (Fig. 632, h), so that at this place the old hypodermis undergoing destruction eventually lies on the inner side of the newly-formed epithelial layer (B). We therefore see from this that, during the replacement of the old hypodermis by the new, the continuity of the superficial epithelium is never interrupted. Since the edges of the two kinds of hypodermis overlap, the surface of the body is nowhere bare of epithelium. The dissolution of the larval 680hypodermis is accomplished under the influence of the leucocytes (Fig. 632, k), which attack the larval hypodermis-cells and absorb their contents piece by piece, and so fill themselves with bits of the hypodermis-cells and their nuclei; since these fragments have the shape of roundish granules, they were called by Weismann granule-balls. These granule-balls, which fill the body-cavity of the later pupal stage, are nothing else than the leucocytes (blood corpuscles) which have absorbed the fragments of tissue of the larval body.
It should here be said that the destruction of the larval tissues is not to be attributed to the previous death of the cells, but is the result of the action of the leucocytes on tissues which, though weakened in their vital power, are still living. While the completely healthy, active tissues, i.e. those of the imaginal buds, withstand the attacks of the leucocytes, the less healthy larval tissues are by the attacks of the leucocytes divided into fragments and eaten and digested by them. This process is most marked in the histolysis of the larval muscles. The destruction of most of the larval organs depends, therefore, on the capacity of the amœboid blood-corpuscles for taking food and on intracellular digestion, as was first shown by Metschnikoff, who has given to these leucocytes the name of “phagocytes.”
This process of histolysis goes on in the same way in the head and abdomen as in the thorax. In the abdomen, as Ganin first proved, there are in each of the eight segments of which it consists in the larva four small cellular islets or imaginal buds (Figs. 631, hi, 632, i), from which originate the new hypodermis.
Van Rees has lately found in the abdominal segments another pair of smaller imaginal buds. The four imaginal buds occurring in the last segment are situated close to each other, encircling the anal opening (Fig. 633, ims), and take part in the formation of the hind-intestine, the rectal pouches and rectal papillæ. To this segment also belong the two pairs of imaginal genital buds (rudiments of the external sexual organs) which were first found by Künckel d’Herculais in Volucella.
The newly formed hypodermis spreads rapidly over the outer surface of the body, so that hypodermal areas corresponding to the separate imaginal buds soon unite. Simultaneously with this completion of the definite epithelial layer the larval hypodermis becomes completely destroyed by the phagocytes.
The muscles.—A similar process of destruction by phagocytes affects the greater number of the larval muscles, except the three pairs of thoracic muscles employed in respiration, and which pass 681intact from the larva to the imago. Indeed, the dissolution of the muscles is the first process which occurs in the metamorphosis. The destruction of the larval muscles is accomplished in such a way that, a great number of leucocytes which have collected on the surface of the muscular fibres, press through the sarcolemma and enter within the muscular tissue, filling the spaces formed between them, By this means the muscles break up into a number of rounded particles which are taken into the interior of the leucocytes. Thus a collection of granule-balls arise from the muscles, which finally separate from each other and become scattered throughout the body-cavity of the pupa. In the same way as the muscular substance, the muscle-nuclei are taken up and digested by the phagocytes.
The imaginal muscles develop from the definitive mesoderm which has originated from the mesoderm of the imaginal buds (Fig. 632, C, m).
The digestive canal.—As in the hypodermis and muscles, the histolysis of the larval digestive tract and its new formation from separate imaginal buds go on simultaneously, so that the continuity of the process is not interrupted.
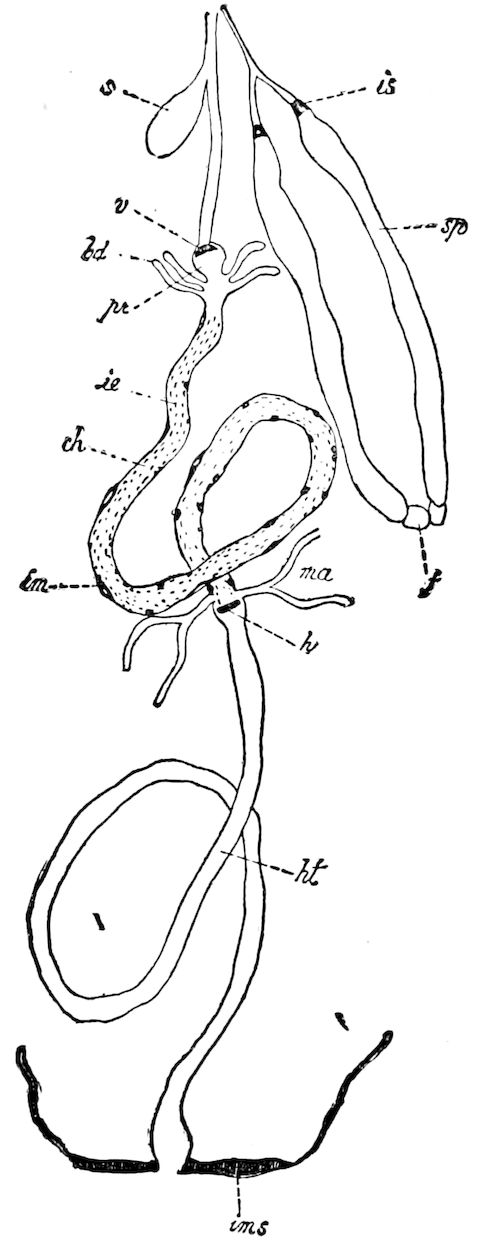
Fig. 633.—Digestive tract of a Musca larva with the imaginal germs: bd, cœca; s, food-reservoir; is, imaginal ring of the salivary gland (sp); f, fat-cells at the end of the salivary gland; pr, proventriculus; r, its ring; ie, imaginal cells of the mid-intestinal epithelium; ch, chyle-stomach; ma, urinary tubes; im, imaginal cells of the mid-intestinal, muscular layer; ims, binder, abdominal, imaginal buds; h, hind intestinal, imaginal bud; ht, hind-intestine.—After Kowalevsky, from Korschelt and Heider.
The imaginal buds of the much-shortened pupal digestive canal occur in the mid-intestine (stomach) in the form of numerous scattered groups of cells (Fig. 633, ie), and in the fore- and hind-intestine in the form 682of rings (v and h) of imaginal tissue. The imaginal ring of the fore-intestine (v) lies in the region of the proventriculus (pr, compare Fig. 635, im), while that of the hind-intestine is situated directly behind the base of the urinary tubes. The regeneration of these two parts of the digestive canal is not entirely accomplished by these two rings, but the imaginal rudiments of the neighboring parts of the outer surface of the body also have a share in it. Thus it appears that the foremost part of the œsophagus is built up from the imaginal buds in the region of the mouth, while the imaginal buds surrounding the anus in the 8th abdominal segment (Fig. 633. ims) produce by invagination the rectal pouches, together with the rectal papillæ.
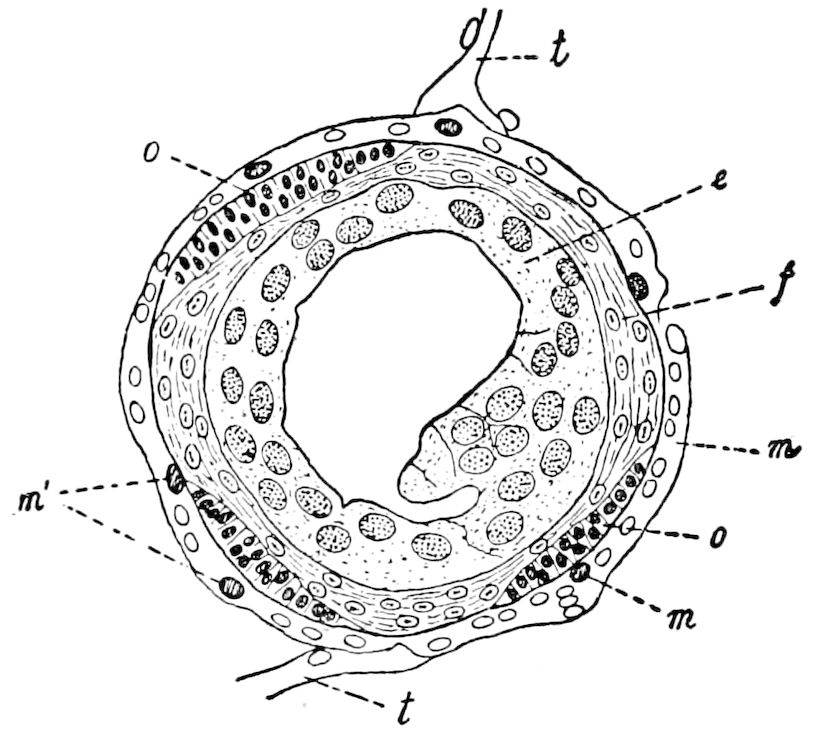
Fig. 634.—Cross-section through the mid-intestine of pupa of Musca: e, rejected and degenerate epithelium of the larval stomach; f, cellular layer newly formed around the same; m, muscular layer; m′, imaginal cell of m; o, imaginal bud of the mid-intestinal epithelium; t, tracheal stem.—After Kowalevsky, from Korschelt and Heider.
Fig. 635.—Longitudinal section through the proventriculus of a muscid larva: im, fore-intestinal, imaginal ring; oe, œsophagus; pr, proventriculus.—After Kowalevsky, from Korschelt and Heider.
The formation of the mid-intestine (stomach) takes place in such a way that the island-like imaginal buds spread out by cell-multiplication over the outer or basal surface of the larval mid-intestinal epithelium (Fig. 634, o), until they finally unite, so as to form the wall of the imaginal mid-intestine (stomach). At the same time the entire larval epithelium (e) is cast in the interior and forms the so-called yellow body, which becomes surrounded by a layer of small cells and a jelly-like mass, and remains until its destruction in the pupal stomach. The larval muscular layer (m) remains intact as long as the imaginal mid-intestine is not fully developed, when 683it is attacked and destroyed by phagocytes. The final muscular layer arises from single cells lying on the outer surface of the imaginal buds (Figs. 633, im, 634, m′), which should be regarded as special imaginal cells of the mid-intestinal muscular layer.
The transformation of the fore-intestine is introduced by a degeneration of the proventriculus and sucking stomach. The proventriculus (Fig. 635, pr), which had been formed from a circular fold of the fore-intestine, disappears by the smoothing out of this folded structure. The sucking stomach also similarly degenerates by withdrawing gradually into the œsophagus, so that instead of the original diverticulum there remains only an enlargement of the œsophageal cavity. At the same time this part of the canal is attacked and destroyed by phagocytes, while the destroyed portions become replaced by the gradually extending imaginal parts of the wall. The imaginal ring of the fore-intestine (Fig. 635, im), which, according to Kowalevsky, is concerned in the formation of a great part of the definitive œsophagus, becomes closed at its hinder end so that the communication with the mid-intestine appears to be interrupted.
The hind-intestine of the imago is rebuilt in exactly the same manner. Here also the imaginal ring widens and forms a tube, which while it grows around the openings into the urinary tubes, closes itself against the mid-intestine, while behind it remains in connection with the larval hind-intestine. In a similar way the larval hind-intestine is attacked by the growth from behind of an imaginal ring, which proceeds from imaginal buds near the anus, until finally, when the entire larval hind-intestine is reduced to granule-balls, the two imaginal sections of the tube are brought into contact with each other. (Kowalevsky in Korschelt and Heider.)
The larval salivary glands (Fig. 633, sp) are completely destroyed by phagocytes. Then succeeds the new formation of these glands from imaginal buds, which, according to Kowalevsky, form rings situated at their anterior ends.
The nature of the transformation undergone by the urinary tubes is not yet well ascertained. According to Van Rees, there is in this case perhaps a regeneration of the larval cells by division, but on the other side there may be a histolysis of these elements.
The above-described method of transformation of the digestive canal seems, according to Korschelt and Heider, to be very common among the holometabolic insects. It has not only been observed in the Diptera, but also in the Lepidoptera (Kowalevsky, Frenzel), Coleoptera (Ganin), and Hymenoptera (Ganin). The stripping off of the epithelium of the mid-intestine was found by Kowalevsky to occur also in Corethra, Culex, and Chironomus.
The tracheal system.—As we have seen (p. 448), the tracheal system of caterpillars just before pupation undergoes disintegration, 684accompanied by a reformation of the peritoneal membrane and tænidia. The larval ectotrachea undergoes histolysis, that of the imago being meanwhile formed; the larval tænidia also break up, dissolve, and are replaced by new tænidia which arise from the nuclei of the peritoneal membrane. That the tracheal system in the Muscidæ during metamorphosis undergoes a transformation is shown, as Korschelt and Heider claim, by the entirely different shape of the system in the maggot, the pupa, and the fly. The air is admitted to the tracheal system of the maggot, not by lateral openings, but through the two stigmata at the end of the body. On the other hand, the pupa breathes by prothoracic spiracles, while the fly has six pairs of lateral stigmata of the normal structure. There may be in the larva and pupa vestigial closed stigmata, as there are in the thorax of caterpillars, with tracheal branches leading to where were once functional stigmata. These stigmatal branches, as well as some other portions of the tracheal system already observed by Weismann, seem, according to Van Rees, to function as imaginal buds for the regeneration of the tracheal matrix, while frequently also a regeneration of this epithelium, by a simple repeated division of cells, may be recognized. The disintegration of the tracheal system is accomplished by means of phagocytes in the manner already described.
The nervous system.—The central nervous system passes directly from the larval into the imaginal stage, since it must continue to exercise most of its functions throughout metamorphosis, though it undergoes important changes of form and position. At the same time, certain histological transformations occur which may be regarded as a histolysis. Such is the destruction and rebuilding in the interior of the organs, which, however, preserve their continuity. Every case of destruction of tissues in the pupa has come to be regarded as a histolysis.
The problem of the transformation of the peripheral nervous system is not yet well understood. Although during the destruction of the larval muscles the motor nerves also in part degenerate, in the case of the nerves distributed to the appendages the conditions are different, as these may be recognized in the larva in the form of the nerve-threads which place the imaginal buds in connection with the central nervous system. These threads, according to Van Rees, pass from the larva into the pupa and imago, so that with the farther development of the rudiments of the extremities, only the distal part of the nerves belonging to them are to be regarded as new formations. (Korschelt and Heider.)
685The fat-body.—The larval fat-body is also destroyed through the activity of the leucocytes in the same way as the other tissues. The reformation of the fat-body seems to begin in the mesoderm of the imaginal buds. Possibly, also, the masses or collections of embryonic cells which are regarded by Schaeffer as “blood-forming cells,” may serve to regenerate the fat-body. At all events, they have been derived from the mesodermal tissues. Though Wielowiejsky saw the fat-body of Corethra arising from a cell-layer situated under the hypodermis, yet it is not necessary to regard this observation as favorable to the view of Schaeffer that in Musca the larval fat-body is derived in part from the hypodermis, and in part from the tracheal matrix, thus from the ectodermal tissues. (Korschelt and Heider.)
Definitive fate of the leucocytes.—We have seen that the formation of the organs of the imago originates in the imaginal buds, in all cases where these do not pass directly from the larva into the pupa. The leucocytes, whose numbers in the pupa are greatly increased, take no direct part in the formation of the tissues. Their importance seems to lie in this, that they destroy those larval organs doomed to destruction, the parts of which they take in and digest, and possibly, by their powers of locomotion, convey particles of food to the developing organs.
What, on the other hand, is the fate of the leucocytes after the developmental processes in the pupa have ceased? There can be no doubt that a part of the so-called granule-cells are again transformed into normal blood-corpuscles. Another, and, as it seems, more considerable, share suffer degeneration. Finally, the leucocytes themselves serve as nourishment for the newly formed tissues. Of interest in this direction is the observation of Van Rees, that numerous leucocytes finally pass into the newly-formed hypodermis and then degenerate in crevices between the hypodermis-cells. (Korschelt and Heider.)
It has been suggested by Van Rees that the phagocytes attack all the larval organs indiscriminately, but that the active metabolism of the imaginal buds preserves them from these attacks. He also thinks that Kowalevsky is probably right in supposing that the buds render themselves immune by some poisonous secretion.
Pratt, however, thinks that the supposition of a protecting or poisonous secretion is scarcely necessary to account for the phenomenon, and suggests that the larval tissues are a prey to the phagocytes, because at the end of larval life they become functionless and inactive, so as to become an easy prey to phagocytes or disintegrating influences of any sort. On the other hand, the imaginal buds “in which there is an exceedingly active metabolism, and all the larval 686organs which remain functional during the metamorphosis are immune from the attacks of the phagocytes. The heart in the muscids continues to beat, as Künckel d’Herculais has observed, during the entire period of the metamorphosis, with the exception of a day or two in the latter half of it. The nervous system must continue functional during the entire time. The three pairs of thoracic muscles which pass intact from the larva to the imago are probably employed in respiration during the metamorphosis. The reproductive glands are, like the imaginal disks, rapidly growing organs.” He adds that among the other holometabolic insects many or most of the larval organs remain functional during metamorphosis, hence there is but little histolysis. “But the larval intestine would always necessarily become unfunctional, and, as we have seen, Kowalevsky is of the opinion that the larval mid-gut in all holometabolic insects contains imaginal disks, and undergoes degeneration during the metamorphosis.”
The post-embryonic changes and imaginal buds in the Pupipara (Melophagus).—The sheep-tick (Melophagus) is still more modified than the Muscidæ; the larva is apodous and acephalous, but, as Pratt observes, much less highly specialized than those of muscids, and in respect to the position of the thoracic buds it closely resembles Corethra. They lie just beneath the hypodermis in two very regular rows, and not in the centre of the body, as in Musca (Figs. 628, 636, C). While, however, in Corethra all the thoracic buds are of larval origin, arising after the last larval moult, in Melophagus, on the other hand, each of these buds, except the dorsal prothoracic, arises in the embryo, as is also the case in Musca.
In the cephalic buds the conditions are similar to those in Musca, but still more complicated. Instead of a single pair of head-buds, there are two pairs, one dorsal and one ventral. “The dorsal pair corresponds to the muscidian head-disks in every respect; they are destined to form the dorsal and lateral portions of the imaginal head, together with the compound eyes. The ventral head-disks have no counterpart in Musca. The fate of these disks or buds is to form the ventral portion of the head, the paired projections forming the rudiments of the proboscis.
“The formation of the head-vesicle proceeds in a way similar to that in Musca. The ventral disk fuses early at its lateral edges with the dorsal pair. The communications between both ventral and dorsal disks and the pharynx rapidly widen (in the old larva they have already become very large), and soon the disks and pharynx form together a single vesicle, which is the head-vesicle.” The imaginal buds of the abdomen Pratt finds to be exactly as in the corresponding ones of Musca.
In the embryo of Melophagus the cephalic and thoracic imaginal buds first appear as local thickenings, followed by the invagination of the ectoderm; the cephalic buds first appear very early in the 687ontogeny of the insect (Fig. 636, C), just as the germs of the digestive canal, nervous system, and tracheæ are appearing. The single median thickening (v) is destined to form the ventral cephalic bud, while the pair of thickenings behind (d) become the dorsal buds, those homologous with the cephalic buds of Musca.
Fig. 636.—Imaginal buds in Musca,—A, in Corethra,—B, in Melophagus,—C, in embryo of Melophagus: dorsal view of head; b, bud; p, peripodal membrane; c, cord; hy, hypodermis; cl, cuticula; st, stomodæum; v, ventral cephalic bud, behind are the two dorsal cephalic buds (d).—After Pratt.
The thoracic buds, which arise as hypodermic thickenings, do not appear until late in embryonic life, until the time of the involution of the head.
Pratt did not observe in the embryo the buds of the internal organs and of the abdominal hypodermis, and thinks it probable that they appear first in the larva.
c. General summary
We have seen that in Coleoptera, Lepidoptera, Diptera, and Hymenoptera, and with little doubt in all the holometabolous insects, the parts of the imago originate in single formative cellular masses (imaginal buds) already present in the larva, and often even in the later embryonic stages. There are such imaginal buds for each part of the body,—for the appendages of the head, for the legs and wings, for the ovipositor, and probably for the cercopods, for the hypodermis of the abdomen, and for the different sections of the digestive 688canal. We have seen, as Korschelt and Heider state, that the formation of the mesodermal organs of the imago (muscles, connective tissue, fat-body) begins in the mesodermal part of the imaginal buds, whose first origin is still obscure. Simultaneously with the formation of the imaginal organs, there goes on under the influence of the leucocytes the destruction of the larval organs. Both processes (destruction and regeneration) therefore go on hand in hand, so that the continuity of the organs in question in most cases remains perfect, inasmuch as the complete destruction only ensues after the formation of the final organs. The only exceptions are most of the muscles of the larva, which are destroyed at a very early period.
Moreover, it is evident that the sharp division into larval, pupal, and imaginal stages only applies to the external surface of the body, since they follow one another after successive moults. The processes of the internal development, on the other hand, form an entirely continuous series of transformations between which is no sharp line of demarcation. Yet as a whole the form of the larva, pupa, and imago are kept distinct in adaptation to their separate environments and habits.
Finally, as Pratt very truly remarks, the epigenetic period in insects, when new organs are forming, does not end with the birth of the larva from the egg, but extends through the larval, and even through the pupal period. “The principal significance of the pupal period and the metamorphosis is that it is the time when the larval characters which were adapted for use during a period of free life in the midst of the development, and which would be valueless to the imago, are corrected or abandoned.”
HYPERMETAMORPHISM
When an insect passes through more than the three normal stages of metamorphosis, i.e. the larval, pupal, and imaginal, it is said to undergo a hypermetamorphosis. The best-known examples are the supernumerary stages of Meloë, Stylops, etc.
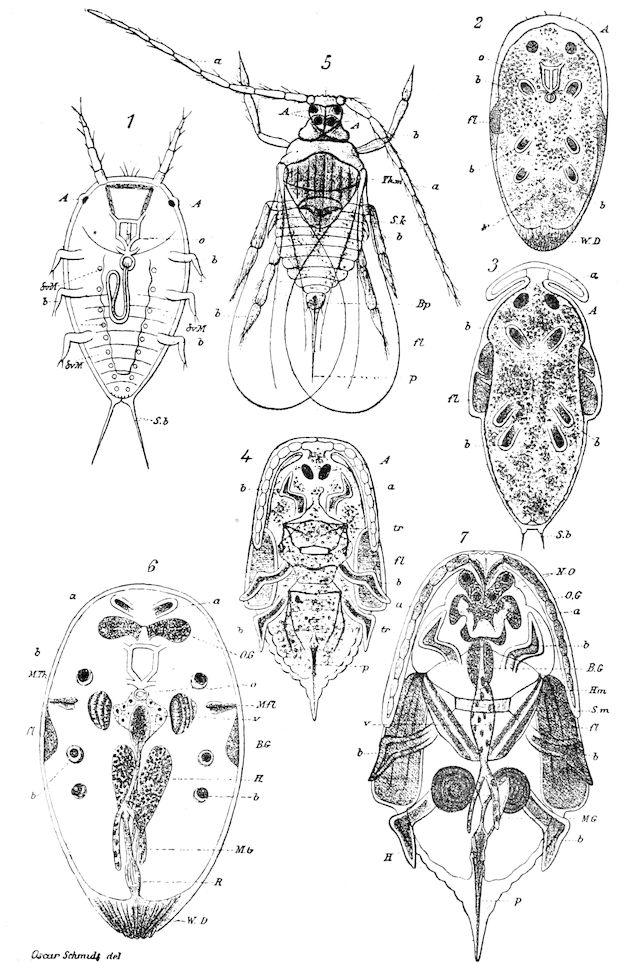
Fig. 637.—Hypermetamorphosis of male of Aspidiotus nerii: 1, freshly hatched larva; 2, larva shortly before pupating; b, rudiments of the legs; fl, of the wings; 3, pupa before moulting; 4, the same after moulting; 6, larva farther advanced than in 2; a, antennal rudiments; b, rudiments of legs; v, stomach; OG, brain; M. Fl, rudiments of the elevator and depressor muscles of the wing; M. Th, rudiments of the dorsal muscles; H, rudiments of the testes; 7, pupa shortly before entering upon the imago state (5); A, eyes; a, antenna; o, mouth; WD, wax-glands; BG, ventral nervous cord; Sb, caudal setæ; tr, tracheæ; p, genital armature.—After Schmidt.
690As has already been observed, Schmidt has shown that in the male of the Coccidæ, there is a true hypermetamorphosis, as shown by Fig. 637. In Aspidiotus nerii there are five stages, there being two larval (1, 2) and two pupal stages (3, 4, 7). Stage 3 (Fig. 637, 2) may be compared with the pro-pupa stage of Riley (Fig. 581).
Fig. 638.—Mantispa interrupta, and side view of the same without wings: natural size.—Emerton del. a, freshly-hatched campodeoid larva of Mantispa styriaca, enlarged; b, the same, but older, before the first moult; enlarged.—Brauer.
We have already, on page 602, described the hypermetamorphosis of the neuropterous insect Mantispa (Fig. 638).
Fig. 639.—Triungulin (a) of a Californian Meloë: b, the three triungulin claws; c, antenna; d, maxillary palpus; e, labial palpus; f, mandible; g, an abdominal joint; h, imago, ♀; i, antenna of ♂.—After Riley.
In Meloë the freshly hatched larva, or “triungulin” (Fig. 639, a), is an active Campodea-like larva, which runs about and climbs up flowers, from which it creeps upon the bodies of bees, such as Anthophora and Andrena, who carry it to their cells, wherein their eggs are situated. The triungulin feeds upon and destroys the eggs of its hostess. Meanwhile its inactive life in the bee’s cell reacts upon the organism; after moulting, the-second larval form (Fig. 640, b) is attained, and now the body is thick, cylindrical, soft, and fleshy, and it resembles a lamellicorn larva, with three pairs of rather long thoracic legs. This is Riley’s carabidoid stage. This second larva feeds upon the honey stored up for the young or larval bees. After 691another moult, there is another entire change in the body; it is motionless, the head is mask-like without movable appendages, and the feet are represented by six tubercles. This is called the semipupa or pseudo-pupal stage. This form moults, and changes to a third larval form (c), when apparently, as the result of its rich, concentrated food, it is overgrown, thick-bodied, without legs, and resembles a larval bee.
Fig. 640.—Oil-beetle: a, first larva; b, second larva; c, third larva; d, pupa.
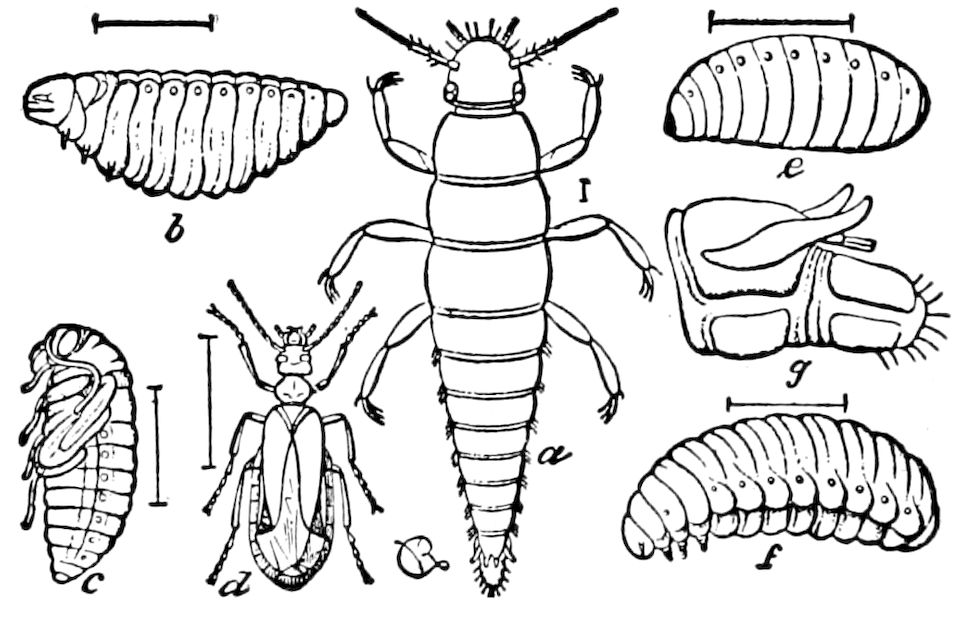
Fig. 641.—History of Sitaris: a, triungulin or 1st larva; g, anal spinnerets and claspers of same; b, 2d larva; e, pseudo-pupa; f, 3d larva; c, true pupa; d, imago, ♀.—After V. Mayet, from Riley.
After thus passing through three larval stages, each remarkably different in structure and in the manner of taking food, it transforms into a pupa of the ordinary coleopterous shape (d).
The history of Sitaris, as worked out by Fabre and more recently by Valery-Mayet, is a similar story of two strikingly different adaptational larval forms succeeding the triungulin or primitive larval stage. The first larva (Fig. 641, a) is in general like that of Meloë, the second (b) is thick, oval, fleshy, soft-bodied, and with minute legs, evidently of no use, the larva feeding on the honey stored by its host. The pseudo-pupal stage is still more maggot-like than in the corresponding stage of 692Meloë, and the third larva (f) is thick-bodied, with short thoracic legs.
In the complicated life-history of another cantharid, Epicauta vittata, as worked out by Riley (Fig. 642), we have the same acquisition of new habits and forms after the first larval stage, which evidently were at the outset the result of an adaptation to a change of food and surroundings. The female Epicauta lays its eggs in the same warm, sunny situation as that chosen by locusts (Caloptenus) for depositing their eggs. On hatching, the active minute carnivorous triungulin, ever on the search for eggs, on happening upon a locust egg gnaws into it, and then sucks the contents. A second egg is attacked and its contents exhausted, when, owing to its comparatively inactive habits and rich nourishing food after a period of inactivity and rest, the skin splits along its back, and at about the eighth day from beginning to take food the second larva appears, with much smaller and shorter legs, a much smaller head, and with reduced mouth-parts. This is the carabidoid stage of Riley. After feeding for about a week in the egg a second moult occurs, and the change of form is slight, though the mouth-parts and legs are still more rudimentary, and the body assumes “the clumsy aspect of the typical lamellicorn larva.” This Riley denominates the scarabæidoid stage of the second larva.
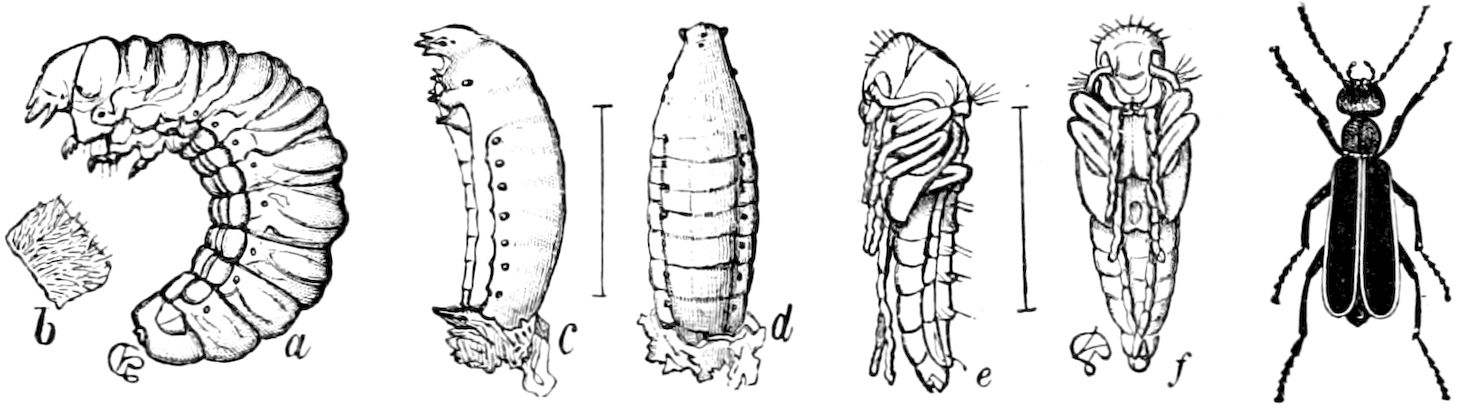
Fig. 642.—Epicauta cinerea: a, end of 2d larval stage; b, portion of dorsal skin; c, d, coarctate larva; e, f, pupa.—After Riley.
After six or seven days there is another transformation, the skin being cast, and the insect passes into another stage, “the ultimate stage of the second larva.” The larva, immersed in its rich nutritious food, grows rapidly, and after about a week leaves the now addled and decaying locust eggs, and burrows into the clear sand, where it lies on its side in a smooth cell or cavity, and where it undergoes an incomplete ecdysis, the skin not being completely shed, and assumes the semipupa stage, or coarctate larval stage of Riley.
In the spring the partly loose skin is rent on the top of the head 693and thorax, and then crawls out of it the “third larva,” which only differs from the ultimate stage of the second larva “in the somewhat reduced size and greater whiteness.” The insect in this stage is said to be rather active, and burrows about in the ground, but food is not essential, and in a few days it transforms into the true pupa state.
These habits and the corresponding hypermetamorphosis are probably common to all the Meloidæ, though the life-history of the other species has yet to be traced.
In the genus Hornia described by Riley, the wings of the imago are more reduced than in any other of the family, both sexes having the elytra as rudimentary as in the European female glow-worm (Lampyris noctiluca). These, with the simple tarsal claws and the enlarged heavy abdomen, as Riley remarks, “show it to be a degradational form.”
Its host is Anthophora, and the beetle itself lives permanently in the sealed cells of the bee, and Riley thinks it is subterranean, seldom if ever leaving the bee gallery. The triungulin is unknown, but the ultimate stage of the second larva, as well as the coarctate larva, is like those of the family in general, the final transformations taking place within the two unrent skins, in this respect the insect (Fig. 643) approaching Sitaris.
It appears, then, that as the result of its semi-parasitic mode of life the Campodea-form or triungulin larva of these insects, which has free-biting mouth-parts like the larvæ of Carabidæ and other carnivorous beetles, instead of continuing to lead an active life and feeding on other insects; living or dead, and then like other beetles directly transforming into the normal pupa, moults as many as five times, there being six distinct stages before the true pupa stage is entered upon. So that there are in all eight stages including the imaginal or last stage.
One cannot avoid drawing the very obvious conclusion that the five extra stages constituting this hypermetamorphosis, as it is so well styled, are structural episodes, so to speak, due to the peculiar parasitic mode of life, and were evidently in adaptation to the remarkable changes of environment, so unlike those to which the members of other families of Coleoptera, the Stylopidæ excepted, have been subjected. The fat overgrown body and the atrophied limbs and mouth-parts are with little doubt due to the abundant supply of rich food, the protoplasm of the egg of its host, in which the insect during the feeding time of its life is immersed. Since it is well known that parthenogenesis is due to over, or at least to abundant nutrition, or to a generous diet and favoring temperature, there is little reason to doubt that the greatly altered and abnormally fat or bloated body of the insect in these supernumerary stages is the result of a continuous supply of rich pabulum, which the insect can imbibe with little or no effort.

Fig. 643.—1, Egg-pod of Caloptenus differentialis with the mouth torn open, exposing the newly hatched larva of Epicauta vittata (1 a) eating into an egg and the passage which it made through the mucous covering; natural size. 2, dorsal view of the 1st larva, or triungulin, of E. vittata; 2 a, one side of the head of same from beneath, greatly enlarged so as to show the mouth-parts; 2 b, terminal joint of maxillary palpus, showing imbrications and flattened inner surface armed with stout points; 2 c, leg, showing more plainly the tarsal spines; 2 e, labrum; 2 d, one of the abdominal joints from above, showing stout points, stigmata, and arrangement of spinous hairs. 3, eggs of E. vittata, the natural size indicated at side. 4, dorsal view of the carabidoid stage of the 2d larva of E. vittata: 4 a, its antenna; 4 b, its right maxilla; 4 c, its leg; 4 d, side view of same, showing its natural position within the locust-egg mass. 5, lateral view of the ultimate or full-grown stage of the 2d larva of E. vittata; 5 a, portion of the dorsal skin, showing short setaceous hairs. 6, third head, or that from the scarabæidoid stage of the 2d larva of E. vittata from beneath, showing the reduction of mouth-parts as compared with the first head (2 a); 6 a, antenna of same; 6 b, maxilla of same; 6 c, mandible of same. 7, fourth head, or that of the full-grown larva of E. vittata, from above; 7 a, leg of same; 7 b, the breastplate or prosternal corneous piece. 8, lateral view of the pseudo-pupa or coarctate larva of E. vittata, with the partially shed skin adhering behind: 8 a, dorsal view of same; 8 b, its head, from the front; 8 c, same from side; 8 d, tuberculous leg; 8 e, raised spiracle; 8 f, anal part of same, 9. lateral view of the true pupa of Epicauta cinerea Forst: 9 a, ventral view of same. 10, Epicauta vittata (lemniscata or trivittate var.). 11, Epicauta cinerea Forst. (= marginata Fabr.). 12, antenna of the triungulin of Epicauta pennsylvanica: 12 a, maxilla of same; 12 b, labial palpus of same. 13. ♂ Hornia minutipennis, dorsal view; 13 a, lateral view of same; 13 b, simple claw of same; 13 c, coarctate larva; 13 d, leg of ultimate stage of 2d larva.—After Riley.
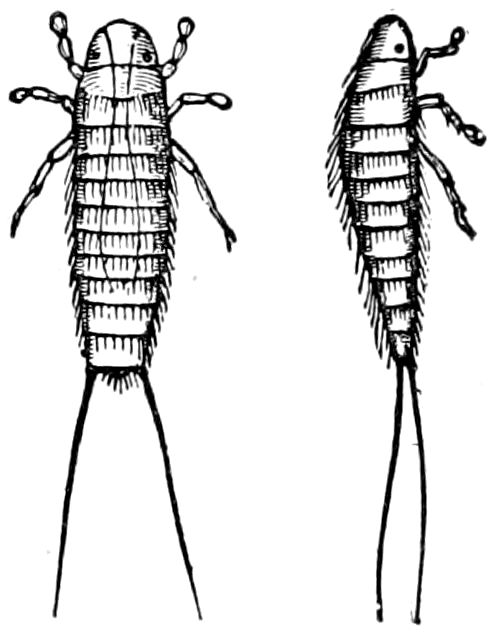
Fig. 644.—Triungulin stage of Stylops childreni.
The life-history of the Stylopidæ is after the same general fashion, though we do not as yet know many of the most important details. The females are viviparous, the young hatching within the body of the parent, as we once found as many as 300 of the very minute triungulin larvæ issuing in every direction from the body of what we have regarded as the female of Stylops childreni in a stylopized Andrena caught in the last of April. The larvæ differ notably from those of the Meloidæ in the feet being bulbous and without claws, yet it is in general Campodea-like and in essential features a triungulin (Fig. 644). The intestine ends in a blind sac, as in the larvæ of bees, and this would indicate that its food is honey. The complete life-history of no Stylopid is completely known. It is probable that, hatched in June from eggs fertilized in April, the larvæ crawl upon the bodies of bees and wasps; finally, after a series of larval stages as yet unknown,[119] penetrating within the abdomen of its host before the latter hibernates, and living there through the winter. 696The females, owing to their parasitic life, retain the larval form, while the free males are winged, not leading in the adult stage a parasitic life, though passing their larval and pupal stages in the body of their host, and are so unlike ordinary beetles as to be referred by good authorities to a distinct order (Strepsiptera).
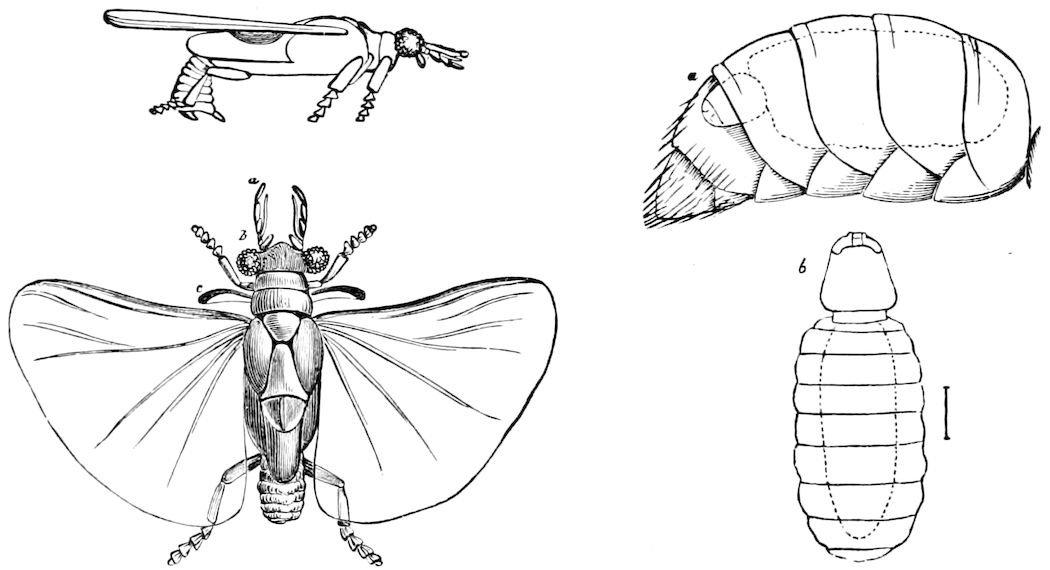
Fig. 645.—Stylops childreni, ♂: a, abdomen of Andrena with ♀ Stylops (b).
The triungulin stage of these insects corresponds in general to the form of the larval Staphylinidæ and allied families, such as the Tenebrionidæ, which are active in their habits, running about and obtaining their food in a haphazard way, often necessarily suffering long fasts. In the external-feeding, less active coleopterous larvæ, like the phytophagous species, which have an uninterrupted supply of nutritious food, we see that the body is thick and fleshy. So also in the larvæ of the Scarabæidæ, Ptinidæ, and the wood-boring groups. In internal feeders, like the larval weevils and Scolytidæ, which live nearly motionless in seeds, fruits, and the sap-wood of plants and trees, with a constant supply of nourishing, often rich food, the eruciform body is soft, thick, and more or less oval-cylindrical. So it is with the larvæ of Hymenoptera, especially in the parasitic forms, and in the ants, wasps, and bees, which are nearly if not quite motionless, at least not walking about after their food.
Now the change from the active triungulin stage to the series of secondary, nearly legless, sedentary, inactive stages is plainly enough due to the change of station and to the change of food. From being an independent, active, roving triungulin, the young insect becomes a lodger or boarder, fed at the expense of its host, and the lack of bodily exertion, coupled with the presence of more liquid food than 697is actually needed for its bare existence, at once induces rotundity of body and a loss of power in the limbs, followed by their partial or total atrophy.
That this process of degeneration may even occur in one and the
same stage of larval existence is very well illustrated by what we
know of the life-history of the wasp-parasite of Europe, Rhipiphorus
paradoxus. Thanks to the very careful and patient observations of
Dr. T. A. Chapman, we have a nearly complete life-history of this
beetle, the representative of a family in many respects connecting
the Meloidæ and Stylopidæ.[120] Where Rhipiphorus lays her eggs is
unknown. Dr. Chapman, however, found a solitary specimen of the
young larva in the triungulin stage. He describes it as “a little
black hexapod, about 1
50 inch (.5 mm.) in length, and 1
120 inch in
breadth, broadest about the fourth segment, and tapering to a point
at the tail; a triangular head with a pair of three-jointed antennæ
nearly as long as the width of the head, with legs very like those of
Meloë larvæ; the tibiæ ending in two or three claws, which are supported
and even obscured by a large transparent pulvillus or sucker
of about twice their length; this was marked by faint striæ radiating
from the extremity of the tibiæ, giving it much the aspect of a
lobe of a fly’s proboscis. Each abdominal segment had a very short
lateral spine pointing backwards; the last segment terminated by a
large double sucker similar to those of the legs; and the little animal
frequently stood up on this, and pawed the air with its feet, as if in
search of some fresh object to lay hold of.”
This almost microscopic larva finds a wasp grub and bores into its
body, probably entering at a point near the back of the first or second
segment behind the head. Dr. Chapman succeeded in finding the
larva of the beetle within that of the wasp, before the latter had
spun up. “Assuming that the wasp larva lives six days in its last
skin before spinning up, I should guess that the youngest of these
had still two or three days’ feeding to do. The Rhipiphorus larvæ
were but a little way beneath the skin of the back, about the fourth
and fifth segments [counting the head as the first], and indifferently
on either side. The smallest of these was 1
16 inch in length, and,
except its smaller size, was precisely like the larger ones I am about
to refer to, having the same head, legs, plates, etc. These were of
the same size as those of the larger larvæ, the difference in size
of the latter being due to the expansion of the intermediate colorless
integument.”
698After the wasp grub has spun the silken covering of its cell the
larva of Rhipiphorus may still be detected in some of them, being
rendered visible by its black legs and dark dorsal and ventral plates.
“On extracting this larva, it bears a general resemblance in size and
outline to the youngest larva of Rhipiphorus that I had found feeding
externally on the wasp grub, but with the very notable exception of
the already mentioned black marks. These are, in fact, a corneous
head, six-jointed legs, and a dorsal and ventral series of plates. I
immediately recognized the head and legs as identical with those of
the little black mite already described, but presenting a ludicrous
appearance in being widely separated from each other by the white
skin of the larva. I have no doubt that the dorsal and ventral series
of black marks are the corresponding plates of the mite-like larva
floated away from each other by the expansion of the intervening
membrane. By measurement also they agree exactly in size, although
the larva extracted from the wasp grub is ten times the length and
six times the width of the little Meloë-like larva. In length it is
⅙ inch (4.5 mm.), and 1
28 inch in breadth.”
The remarkable changes thus described in the larva of this beetle after it has begun its parasitic life within the body of its host are especially noteworthy because the great increase in size and difference in shape, as well as in habits, all take place before the insect has moulted. The rapid development in size, and consequent distension of the body and the separation of the sclerites of the segments behind the head, are paralleled, as Chapman says, by the greatly swollen abdominal region of the body in Sarcopsylla penetrans and in the female of the Termitidæ. In those insects this distension is due to the enlargement of the ovaries and of the eggs contained within them, but in the Rhipiphorus it is due to the comparative inactivity of the larva, and to its being gorged with an unending supply of rich food, the blood and fat of its host. It follows, then, that if a sedentary life and over, or at least abundant, nutrition will have this effect within the short period covered by the single first larval stage of the Rhipiphorus, it is reasonable to infer that the hypermetamorphosis is also due to the same factors.
Chapman then goes on to say that finally, within six hours of the time of spinning up of the wasp grub, the Rhipiphorus larva at the end of Stage 1., which is “usually in motion, and for its situation might be called tolerably active, is seen to lay hold of the interior of the skin with its anterior legs, and keeps biting and scratching with its strong and sharp jaws until it is able to thrust through its head, when, in less than a quarter of an hour, it completely emerges by a 699vermiform movement; and at the same time it casts a skin, together with the black head, legs, plates, etc.”
The larva, now in its second stage, passes forward and seizes hold of the upper or lateral aspect of the prothoracic segment of the wasp grub. On emerging it becomes shorter and thicker, “and very soon loses length by that curving forward of its head which is so marked in the full-grown larva, and which does not exist before its emergence.” The larva is now found “lying like a collar immediately under the head of the wasp grub, and is attached to it by the head, though not very firmly.” At this stage the feeding of the young Rhipiphorus is rather sucking than eating.
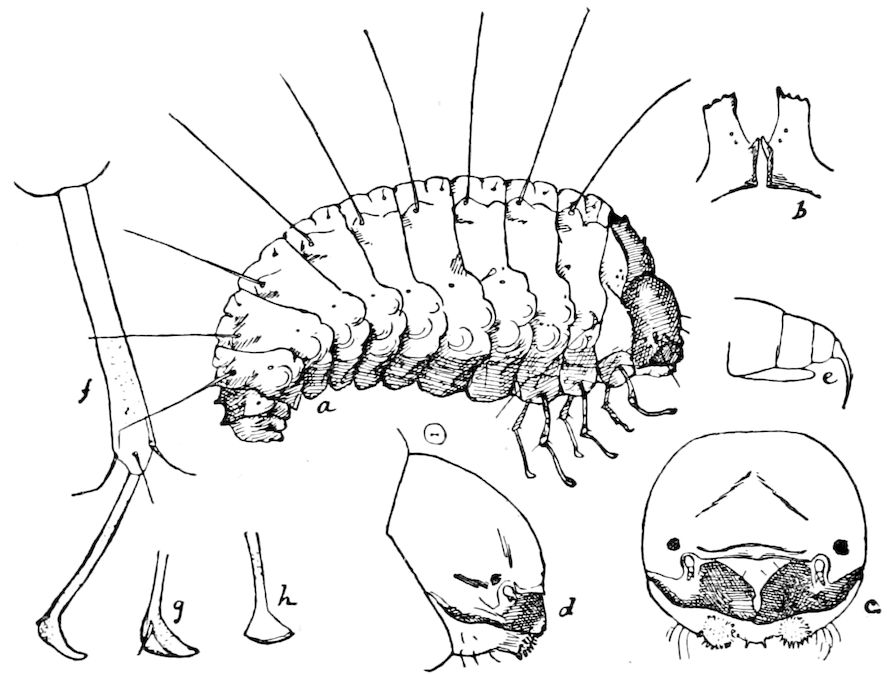
Fig. 646.—First larva (a) of Bruchus fabæ, greatly enlarged; b, thoracic processes; c, head, from front; d, from side; e, antenna; f, thoracic leg; g, rear view of tarsus; h, same, front view.—After Riley.
When about 6 mm. in length it moults a second time, and the full-grown larva closely though superficially resembles a Crabro or Pemphredon larva, the small head being bent over forwards. By the time it is ready to pupate it has wholly eaten the wasp larva, and the temperature of the cell being high, a larva 5 mm. long grows large enough in two days to fill the top of the cell of its host, and the larva is ready to pupate about a week after hatching, so that its development is very rapid. The beetles themselves do not live in the cells. Chapman thinks they hibernate, and that the eggs are laid in the spring or summer.
We thus have in this insect three larval stages, the triungulin, and 700two later stages, the great differences between the first and the last two being apparently due to their parasitic mode of life, the larva spending its second stage within its host, involving an existence in a cell with a high temperature, an uninterrupted supply of rich, stimulating food, and a comparatively sedentary mode of life compared with that of the triungulin at the beginning of its existence. It is quite obvious that the hypermetamorphosis is primarily due to a great change in its surroundings, i.e. the parasitic mode of life of the beetle, habits of very rare occurrence in the Coleoptera, numerous in species as they are.
Fig. 647.—First larval stage of Bruchus pisi: a, egg in pea-pod; b, cross-section of opening of mine; c, young larva and opening on inside of pod by which it has entered, enlarged; d, d, d, eggs, natural size; e, 1st larval stage; f, a leg of same; g, prothoracic spinous processes.—After Riley.
In this connection attention may be drawn to a supernumerary larval stage observed by Riley in the pea- and bean-weevils (Figs. 646 and 647). The larva on hatching has long slender legs, though differing from those of an ordinary coleopterous larva in having but three joints (j, g, h). This stage is very short, and the legs temporary, as, after entering the bean or pea, it casts its skin, losing its legs, and assuming the vermiform shape of the second larval stage. In this case the change from a pedate to an apodous larva is plainly enough due to the change from an external feeder, like a chrysomelid larva, to a larva leading a boring, internal, almost quiescent life.
701Certain ichneumons also appear to have two distinct larval stages, as Ratzeburg inferred that in Anomalon there are four larval stages (Fig. 648).
Fig. 648.—History of Anomalon circumflexum: A, 1st instar or stage. B, 2d instar. C, larva in the 3d or encysted stage removed from its cyst. D, mature larva. E, pupa.—After Ratzeburg, from Sharp.
In another ichneumon, Klapálek detected what he calls the “sub-nymph.” The insect pupates within the case of a caddis-fly, Silo (Fig. 649).
In the Proctotrypidæ there is also a hypermetamorphosis, though the remarkable precocious stages they pass through are rather embryonic than larval.
In a species of Platygaster which is parasitic in the larva of Cecidomia, the first larva (Cyclops stage) is of a remarkable shape, not like an insect, but rudely resembling a parasitic Copepod crustacean. In this condition it clings to the inside of its host by means of its hook-like jaws, moving about, as Ganin says, like a Cestodes embryo with its well-known six hooks. In this stage it has no nervous, vascular, or respiratory system, and the digestive canal is a blind one (Fig. 651).
Fig. 649.—Metamorphosis of Agriotypus: A, larva. B, “sub-nymph.” C, case of the Silo, with the string of attachment formed by Agriotypus. D, section of the case: v1, operculum of case; v2, cocoon; ag, pupa of Agriotypus; e, exuvia of same; w2, wall of cocoon; s, remains of Silo; w1, closure of case.—After Klapálek, from Sharp.
After moulting, the insect entirely changes its form; it is thick oval-cylindrical, 702nearly motionless, with no appendages, but with a digestive canal and a nervous and vascular system (Fig. 652).
After a second moult the third and last larval stage is attained, and the insect is of the ordinary appearance of ichneumon larvæ.
Not less striking is the life-history of Polynema, which lays its eggs in those of a small dragon-fly (Agrion virgo). The first larval stage is most remarkable. It hatches as a microscopic immovable being, entirely unlike any insect, with scarcely a trace of organization, being merely a flask-shaped sac of cells. After remaining in this state five or six days it moults.
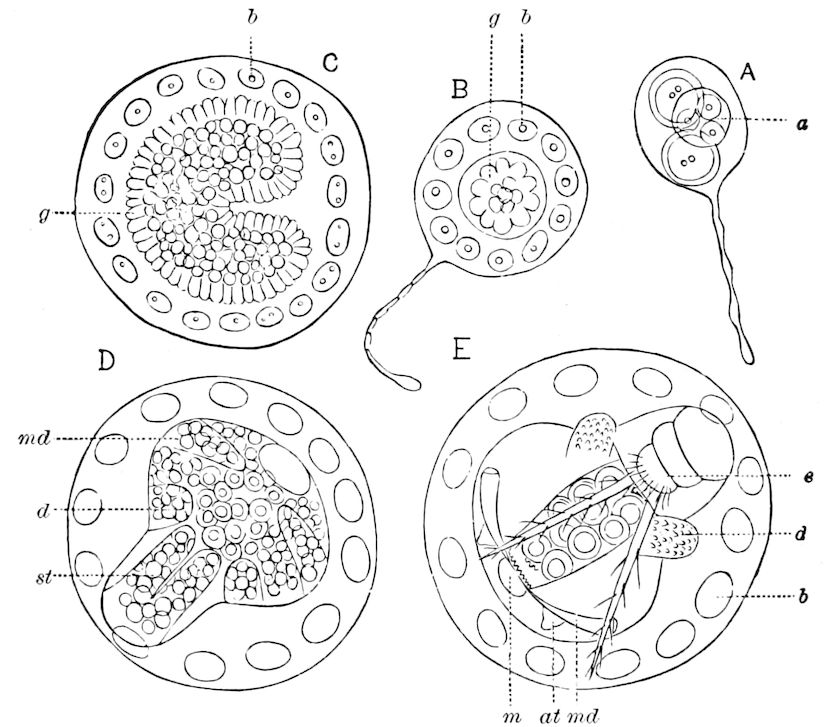
Fig. 650.—Development of Platygaster: A, stalked egg: a central cell giving origin to the embryo. B, g, germ; b, blastoderm cells. C, the same, farther advanced. D, cyclops-like embryo: md, rudiments of mandibles; d, rudimentary pad-like organs, seen more developed in E; st, bilobed tail.
The second stage, or Histriobdella-like form, as Ganin names it, is more like that leech-like worm than an insect.
The third larval form is very bizarre, though more as in insects, having rudimentary antennæ, mouth-parts, legs, and ovipositor. In this condition it lives from six to seven days before pupating (Fig. 653).
The strange history of another egg-parasite (Ophioneurus) agrees in some respects with that of the foregoing forms. It is when hatched of an oval shape, with scarcely any organs, and differs from the genera already mentioned in remaining within its egg-membrane, and not assuming their strange shapes. From the cylindrical sac-like 703non-segmented larva resembling the second larva of Platygaster it passes directly into the pupa state.
A fourth form, Teleas (Fig. 654, A-D), is an egg-parasite of Gerris, and in America one species oviposits in the eggs of Œcanthus.
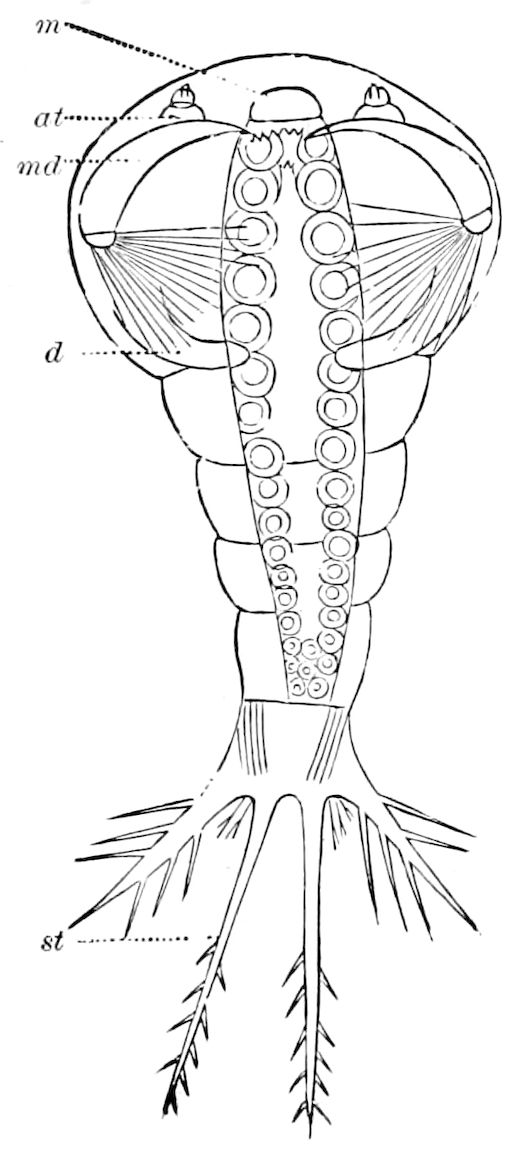
Fig. 651.—First larva of Platygaster: m, mouth; at, rudimentary antenna; md, mandibles; d, tongue-like appendages.
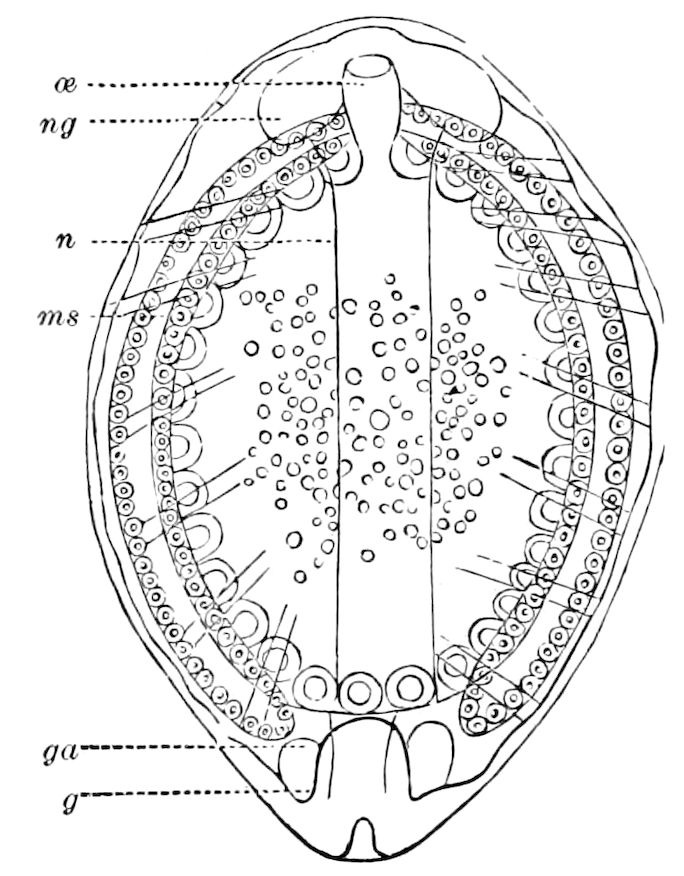
Fig. 652.—Second larva of Platygaster: œ, œsophagus; ng, brain; n, nervous cord; ga and g, genital organs; ms, muscular band.
The spindle-shaped larva in its first stage roughly resembles a trochosphere of a worm rather than the larva of an insect so high in the scale as a Hymenopter. It is active, but after moulting the second larva is oval, still without segments. Dr. Ayers gives a profusion of details and figures of the first and second stages of our Teleas, the second strongly resembling the Cyclops stage of Ganin. He describes three stages, and though he did not complete the life-history of the insect, he thinks it changes to an ovoid flattened form which succeeds the Cyclops stage in other Pteromalidæ, and that there are at least four ecdyses.
It is difficult to account for these strange larval forms, unless we suppose that the embryos, by their rich, abundant food, have undergone a premature development, the growth of the body-walls being greatly accelerated, the insects so to speak having been, under the stimulus of over-nutrition and their unusual environment, and perhaps also the high temperature of the egg, hurried into vermian 704existence on a plane scarcely higher than that of an active ciliated gastrula.
Further observations, difficult though they will be, are needed to enable us to account for the singular prematurity of the embryo of these parasites. That these stages are reversional and a direct inheritance from the vermian ancestors of these insects is not probable, but the forms are evidently the result of adaptation in response to a series of stimuli whose nature is in part appreciable but mostly unknown.
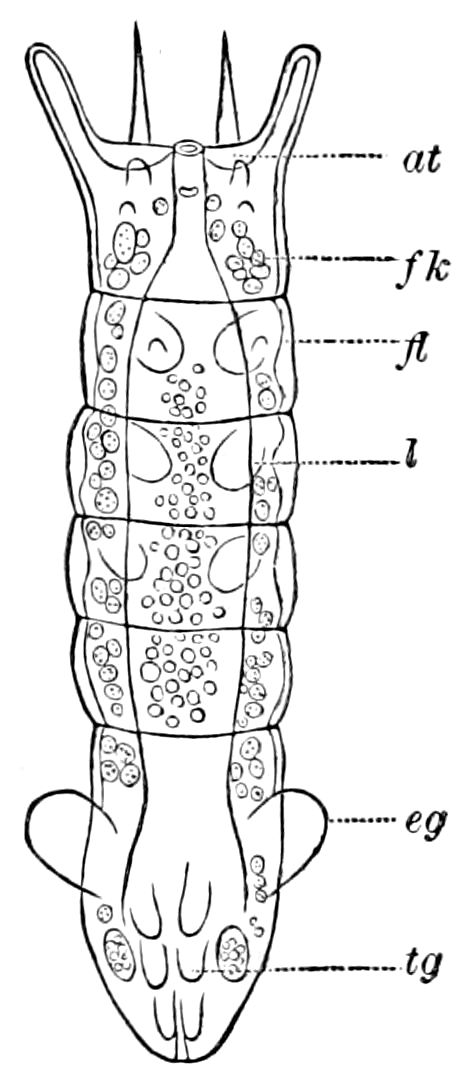
Fig. 653.—Third larva of Polynema: at, antenna; fl, imaginal bud of wing; l, rudimentary legs; tg, buds of one of the three pairs of styles of the ovipositor; fk, fat-body; eg, ear-like process.
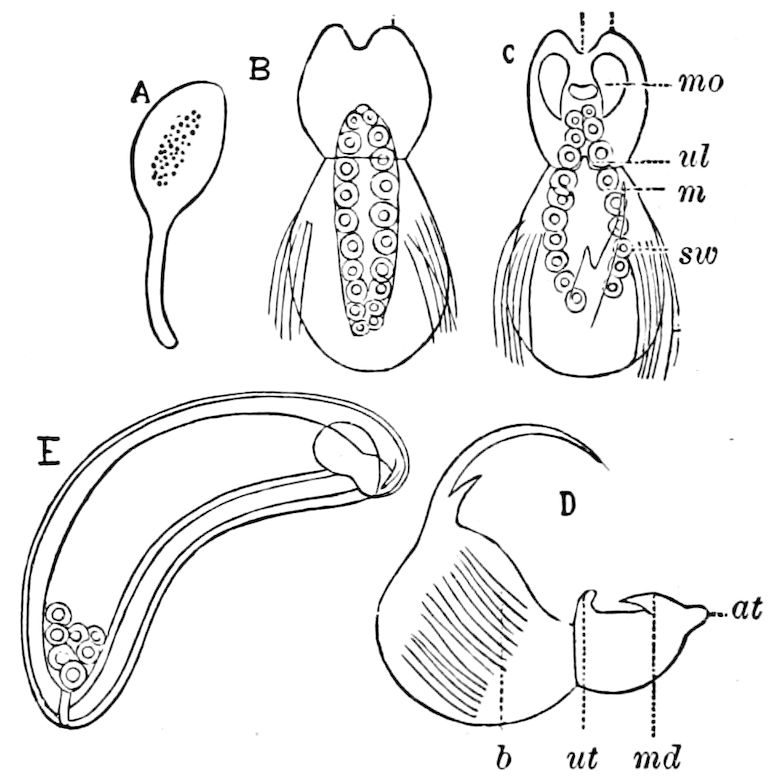
Fig. 654.—A-D, development of Teleas; A, stalked egg; B, C, D, the 1st larval stage: at, antenna; md, hook-like mandibles; mo, mouth; b, bristles; m, intestine; sw, the tail; ul, under lip or labium. E, larva of another parasite, Ophioneurus.—This and Figs. 650–653 after Ganin.
It may be noted, however, that the appearance of a primitive band in the second larval stage suggests the origin of these forms, as well as that of insects in general, from a Peripatus-like, and again from an earlier leech-like Annelid ancestor. Hence the first larval or Cyclops stage is due to a precocious development caused by the unusual environment, and is simply adaptational, and not of phylogenetic significance.
SUMMARY OF THE FACTS AND SUGGESTIONS AS TO THE CAUSES OF METAMORPHISM
An explanation of the causes of metamorphosis is one of the most difficult undertakings in biology, and the phenomenon has been considered as one of the chief difficulties in the way of the acceptance of the theory of descent.
A review, however, of the facts of hypermetamorphism, particularly the life-history of Mantispa, throws much light on the subject, since it is very probable that the supernumerary stages and marked changes of form characterizing them are due to changes of environment, of habits, and of food, causes which have exerted such a profound influence on organic beings throughout all time. Besides these, as the result of changes in the environment and nature of the food, we have the results brought about by the use or disuse of structures brought into existence by the action of stimuli from without, the class of insects abounding in examples of temporary structures which perform a certain function, and then disappear.
Again, if the origin of a hypermetamorphosis can thus be explained, it follows that normal metamorphosis is most probably due to changes of habitat, of seasons, of food, and to accelerated growth resulting from the approach of sexual maturity.
The following facts and conclusions appear to be well established:—
1. The apterous insects (Synaptera) are ametabolous, only the winged insects undergoing a metamorphosis.
2. The complete metamorphosis was not inherited from the primitive ancestor of all insects, but acquired at a later period (F. Müller). The eruciform type is a secondary, adaptive form, derived from the earlier, campodeoid type of larva.
3. The earliest, most primitive pterygote insects passed through only a slight metamorphosis. In other words, as soon as the wings were evolved and insects became adapted to live or take refuge in a new medium, the air, at the approach of the period of adult life, with the ripening or perfection of the reproductive organs, a metamorphosis began to take place, and the number of species greatly multiplied. On the other hand, the Arachnida and Myriopoda, in which as a rule there is no metamorphosis, being confined to a creeping life, with no change of medium, remained poor in number of species.
4. At first the nymphs mainly differed from the adults in lacking 706wings, though having the same habits; in holometabolous insects, the larva became adapted to entirely different habits and environments, so that in Hymenoptera, and especially Diptera, the larva became remarkably unlike the imago.
5. Until the Mesozoic age, or late in the Carboniferous period, there were, so far as we now know, only ametabolous and heterometabolous insects, and these orders (Orthoptera, Dermaptera, Hemiptera, Plectoptera, Odonata, and Neuroptera) were not numerically rich in genera and species, while since early Mesozoic times geological extinction has reduced their numbers.
6. During the Mesozoic age, and since then, the number of species, genera, families, and orders has greatly increased, and insects have become more and more holometabolous. The orders of Coleoptera, Lepidoptera, Hymenoptera, and Diptera are many fold greater in number of species and variety of form than the heterometabolous orders.
The rapid increase in the number and variety of types of insects evidently is correlated with the profound geological changes which took place at the end of the Paleozoic age, involving the appearance of larger continental masses, or a greater land area, thus opening new regions for settlement. Also the origin of flowering plants at about this time undoubtedly had much to do with the genesis of new adaptive structures, such as the changes in the mouth-parts and wings.
7. The process of metamorphosis, at least in the subtropical, temperate, and polar regions, is largely dependent on the change from summer to winter, and, in the tropics, from the rainy to the dry season.
As regards the organization of larval (nepionic) as compared with imaginal forms, the nymphs and larvæ of insects are, with the exception of many Diptera, nearly as perfectly developed as the adult. In this respect the immature insect differs fundamentally from the larvæ of certain worms (for example, the pilidium of Nemerteans) and from the pluteus and brachiolaria stages of echinoderms, which possess only digestive and water-vascular organs.
Insect nymphs and larvæ also differ from the nauplius and zoëa of Crustacea in having at birth all the most important systems of organs (digestive, circulatory, respiratory, nervous, muscular, with sometimes a nearly perfected reproductive system) of the imago, also the same number of cephalic, thoracic, and abdominal segments and appendages. Metamorphism in insects involves (except in the Diptera) rather modifications in the form and functions of organs and appendages already present than the formation of new ones. In 707larval Crustacea, the thoracic and abdominal appendages do not arise until some time after hatching from the egg.
8. While cases of the suppression or abbreviation of larval characters and direct development are not uncommon in echinoderms and crustaceans, in insects this phenomenon occurs only so far as yet known in the Diptera. In these insects the polypody in the embryo is outgrown, or lost, the embryos and larvæ not having even the temporary rudiments of abdominal appendages. The campodeoid characters also are entirely suppressed, dropped, or lost in the more specialized holometabolous orders, Lepidoptera, Hymenoptera, and Diptera, though retained in the more primitive and generalized Coleoptera. (This proves that the Coleoptera are lower or more primitive and generalized than the other orders mentioned.) This abbreviation or loss of organs is, as Hyatt and Arms claim, due to the prepotency of acquired characters in phylogeny, and are also the result of homochronous heredity.
“The Insecta of the more specialized orders, x.-xvi., afford, next to some parasites, the most notable examples of this mode of evolution. Their larval or nepionic, and pupal or neanic, stages are prolonged at the expense of the ephebic, winged stage, and the reasons for this prolongation are found in the great number of new features introduced into these stages of development in these orders as contrasted with those of the more primitive, and, in large part, more ancient orders, i.-ix. The law of tachygenesis has been at work here, as in the former cases alluded to above, and it is shown in the encroachments of the adaptive characteristics of the caterpillar, grub, and maggot upon the inherited characteristics of the Thysanuran stage, which loses its ancestral characteristics, until in most cases they are either obsolete or recognizable with difficulty.” (Hyatt and Arms, Natural Science, 1896, p. 400.)
9. In the holometabolous insects there is a resting, quiescent stage during the pupal period, when the insect takes no food. In this respect the more specialized insects differ from other metamorphic animals. The larva has an abundant supply of fat lasting through pupal life, while in the quiescent pupa, respiration and circulation is much lessened, the animal being as a rule motionless. This resting stage is also necessary for the histolysis and formation of the adult body from the imaginal buds present in the larva.
10. The hypermetamorphosis of Mantispa, Meloë, Stylops, etc., indicate very plainly that the eruciform type of larva is derived from the campodeoid, since one and the same insect passes through these stages before reaching sexual maturity.
11. As observed by Miall, the larva of insects differs from that of other invertebrate animals in being larger than the adult.
12. The metamorphoses of insects are in some important respects 708paralleled by those of the Amphibia. The case of pædogenesis of Chironomus affords a parallel with that of the Siredon, or larva of Amblystoma. Also the organs and appendages of the insects, such as caterpillars, are present, just as the skeleton and other organs of the tadpole are the homologues of those of the adult, although these parts undergo a profound modification, and new structures are added. (See the discussion of this point by Miall, and by Hyatt and Arms.)
Theoretical conclusions; Causes of metamorphosis.—It results from a review of the known facts, together with reasonable inductions from such facts, that so far from opposing the theory of descent, the facts of metamorphosis, and particularly of hypermetamorphosis, seem to afford solid foundation for the theory. While natural selection was not the initiative cause, it plays a part as one of several factors; but the fundamental causes are the same as those which have controlled the origin of species and of the larger groups of animals in general. Owing to the struggle for existence, due to overcrowding, the early insects were forced to take to the air, acquiring wings to enable them to avoid the attacks of creeping and running insects. In the end the insects became, owing to this acquisition of wings, and afterwards to the establishment of a complicated metamorphosis, numerically the most successful type of life in existence, the number of species, extinct and living, mounting into the millions.
All aquatic insects are evidently the descendants of terrestrial forms, and the numberless contrivances and temporary larval organs, particularly of dipterous larvæ, are evidently adaptations to the needs of the insect during its aquatic life, and which are cast aside when the creature passes to a different medium. The sudden or tachygenic appearance of temporary structures, such as hatching spines, various setæ, spines, respiratory organs, so characteristic of dipterous larvæ, and of the protective colors and markings of caterpillars, and which are discarded at pupation, or imagination, are evidently due to the action of stimuli from without, to the primary neolamarckian factors, the characters proper to each larval stadium, and to the pupal and imaginal stadia,—characters probably acquired during the lifetime of the individual,—becoming finally fixed by homochronous heredity.
LITERATURE ON POSTEMBRYONIC DEVELOPMENT AND METAMORPHOSES
Herold, Moritz Johann David. Entwicklungsgeschichte der Schmetterlinge anatomisch und physiologisch bearbeitet. (Cassel, u. Marburg, 1815. 33 Taf., 4º, pp. 1–118, i-xxxiv.)
Ratzeburg, F. T. C. Ueber Entwickelung des fusslosen Hymenopteren-larven, mit besonderer Rücksigt auf die Gattung Formica. (Nova Acta Natur. Curios., xvi, 1832, pp. 145–176.)
Agassiz, Louis. The classification of insects from embryological data. (Smithsonian Contr., ii, Washington, 1851, pp. 28, 1 Pl.)
Ganin, M. Beiträge zur Erkenntniss der Entwicklungsgeschichte bei den Insecten. (Zeitschr. f. wiss. Zool., xix, 1869, pp. 381–451, 4 Taf.)
—— Ueber die Embryonalhülle der Hymenopteren- und Lepidopteren-embryonen. (Mém. Acad. St. Petersbourg (7), xiv, 1869, pp. 18, 1 Pl.)
—— Materialien zur Kenntniss der post-embryonalen Entwicklungsgeschichte der Insecten. (Russian.) Warschau, 1876. (Abdruck aus den Arbeiten der V. Versammlung russischer Naturf. und Aerzte in Warschau, 1876. Abstract by Hoyer in Jahresber. der Anat. und Phys. von Hoffmann und Schwalbe, v, 1876, and in Zeitschr. f. wiss. Zool., xxviii, 1877, pp. 386–389.)
Weismann, A. Die nachembryonale Entwicklung der Musciden nach Beobachtungen an Musca vomitoria und Sarcophaga carnaria. (Zeitschr. f. wiss. Zool., xiv, 1864, pp. 101–263, Taf. 8–14.)
—— Die Metamorphose von Corethra plumicornis. (Zeitschr. f. wiss. Zool., xvi, 1866, pp. 1–83, 5 Taf.)
Packard, A. S. Observations on the development and position of the Hymenoptera, with notes on the morphology of insects. (Proceedings Boston Society of Natural History, 1866, pp. 279–295.)
Künckel d’Herculais, J. Recherches sur l’organisation et le développement des Volucelles. Paris, 1875, Pt. I, pp. 208, 12 Pis.; II, 1881. Atlas of 15 Pls.
Dewitz, H. Beiträge zur Kenntniss der post-embryonalen Gliedmaassenbildung bei den Insecten. (Zeitschr. f. wiss. Zool., xxx, Suppl., 1878, pp. 78–105, 1 Taf.; Nachtrag, Ibid., pp. 25–28.)
—— Ueber die Flügelbildung bei Phryganiden und Lepidopteren. (Berl. Ent. Zeitschr., xxv, 1881, pp. 53–66, 2 Taf.)
Lowne, B. Th. Anatomy, physiology, morphology, and development of the blow-fly. London, Part I, 1880; Part II, 1891.
Viallanes, H. Recherches sur l’histologie des Insectes et sur les phénomènes histologiques qui accompagnent le développement post-embryonnaire de ces animaux. (Ann. Sc. Nat. (6), xiv, 1882.)
Metschnikoff, E. Untersuchungen über intracelluläre Verdauung bei wirbellosen Thieren. (Arb. a. d. zoolog. Inst. zu Wien., v, 1883.)
—— Untersuchungen über die mesodermalen Phagocyten einiger Wirbelthiere. (Biol. Centralbl., iii, 1883.)
Wielowiejsky, H. v. Ueber den Fettkörper von Corethra plumicornis und seine Entwicklung. (Zool. Anzeiger, vi Jahrg., 1883, pp. 318–322.)
Pancritius, P. Beiträge zur Kenntnis der Flügelentwicklung bei den Insecten. In.-Diss. Königsberg, 1884.
Rees, J. van. Over intra-cellulaire spijsverteering en over de beteekenis der witte bloedlichampjes. (Maandblad voor Natuurwetenschappen, xi Jaarg., 1884, pp. 28.)
—— Over de post-embryonale ontwikkeling von Musca vomitoria. (Maandblad voor Natuurwetenschappen, Juli, 1885.)
710—— Beiträge zur Kenntniss der inneren Metamorphose von Musca vomitoria. (Zool. Jahrb. Abth. f. Anat. u. Ontog., iii, 1888, pp. 1–134, 2 Taf., 14 Figs.)
Frenzel, J. Einiges über den Mitteldarm der Insecten, sowie über Epithel-regeneration. (Arch. Micr. Anat., xxvi, 1885.)
Kowalevsky, A. Beiträge zur nachembryonalen Entwicklung der Musciden. (Zool. Anzeiger, viii, 1885, pp. 98, 123, 153.)
—— Beiträge zur Kenntniss der nachembryonalen Entwicklung der Musciden. (I. Theil., Zeitschr. f. wiss. Zool., xlv, 1887, pp. 542–594, 5 Taf.)
Schneider, Ant. Ueber die Anlage der Geschlechtsorgane und die Metamorphose des Herzens bei den Insecten. (Zool. Beiträge, 1885, i, pp. 140–143, 1 Taf.)
Rehberg, A. Ueber die Entwickelung des Insectenflügels (an Blatta germanica). (Marienwerder, 1886, pp. 12, 1 Taf.)
Schäeffer, C. Beiträge zur Histologie der Insecten. (Spengel’s Zool. Jahrb., iii, Abth. f. Anat., 1889, pp. 611–652, 2 Taf.)
Hurst, H. The post-embryonic development of a gnat (Culex). Manchester, 1890, pp. 26, 1 Pl.
Verson, E. Der Schmetterlingsflügel und die sog. Imaginalscheiben desselben. (Zool. Anzeiger, xiii, 1890, pp. 116, 117.)
Bugnion, Edouard. Recherches sur le développement post-embryonnaire, l’anatomie, et les mœurs de l’Encyrtus fuscicollis. (Recueil zool. Suisse, v, 1891, pp. 435–534, 6 Pls.)
Petersen, Wilhelm. Die Entwicklung des Schmetterlings nach dem Verlassen der Puppenhülle. (Deutsch. Ent. Zeitschr., 1891, 2 lepid. Hft., pp. 199–214, 5 Figs.)
Kulagin, Nicolas. Notice pour servir à l’histoire de développement des hyménoptères parasites. (Congrès international de Zoologie, 2e Session, à Moscou, 1892, pp. 253–277. Also in Zool. Anzeiger, xv, 1892, pp. 85–87.)
—— On the development of Platygaster. (Journ. of Friends of Nat. Sc. Moscow. Zool., 1890.) (In Russian.)
—— Beiträge zur Kenntniss der Entwicklungsgeschichte von Platygaster. (Zeitschr. f. wiss. Zool., lxiii, 1897, pp. 195–235, 2 Taf.)
Miall, L. C., and Hammond, A. R. The development of the head of Chironomus. (Trans. Linn. Soc. London, 2d Ser., v, 1892, pp. 265–279, 4 Pls.)
Pratt, Henry S. Beiträge zur Kenntniss der Pupiparen. In.-Diss. Berlin, 1893, pp. 53 (Archiv f. Naturgesch., 1893), 1 Taf.
—— Imaginal discs in insects. (Psyche, viii, 1897, pp. 15–30, 11 Figs.)
Gonin, J. Recherches sur la métamorphose des lépidoptères. De la formation des appendices imaginaux dans la chenille du Pieris brassicæ. (Bull. Soc. Vaud. sc. nat., xxx, 1894, pp. 1–52, 5 Pls.)
Heymons, R. Ueber Flügelbildung bei der Larve von Tenebrio molitor. (Sitz. Ber. Gesell. Natf. Freunde. Berlin, Jahrg. 1896, pp. 142–144, 1 Fig.)
Also the writings of Malpighi, Swammerdam, De Geer, Lyonet, Bonnet, Newport, Brauer, Chapman, Fabre, Valery-Mayet, Riley, Chobaut, Nassonow, Miall (Nature, 1895, pp. 152–158), Hyatt and Arms (Natural Science, 1896, pp. 395–403).
INDEX

- Abantiades, 57.
- Abbreviation of larval characters, 707.
- Abdomen, 162.
- Abdominal appendages, in the embryo, 164;
- Acetabulum, 94.
- Acid, formic, 358;
- uric, 352.
- Acinose salivary glands, 334.
- Acoustic nerve, 290.
- Acronycta, 615;
- hastulifera, 194.
- Acrydium, 421.
- Actias luna, its cocoon-cutters, 634.
- Adelops, 630.
- Adhesive hairs, 111, 113;
- Adiscota, 672.
- Adminicula, 629.
- Adranes cæcus, 57.
- Adult insects, tracheal gills of, 476.
- Æroscepsis, 265.
- Æschna, 53;
- rectal respiration in nymph of, 463.
- Agriotypus, hypermetamorphosis of, 701.
- Aileron, 124.
- Air-sacs, 456;
- use of, 457.
- Aletia xylina, tongue of, 66.
- Aleurodicus, 518.
- Aleyrodes, 518.
- Alitrunk, 90.
- Alluring glands, 391.
- Alula, 123, 125.
- Ametabola, acquired, 599.
- Ametabolia, 596.
- Amnion, 533;
- Amphizoa, 461.
- Anabolia furcata, buccal organs of, 74.
- Anabrus, 49, 73;
- cuticula of, 187.
- Anal glands, 319, 326, 372;
- Androconia, 197, 199.
- Anisomorpha, 371.
- Anisopleura, lateral gills of, 468.
- Anobium, 293, 620.
- Anomalon, hypermetamorphosis of, 701.
- Anophthalmus, brain of, 241;
- Anoplus, 101.
- Ant, cement glands of, 360;
- Antefurca, 92.
- Antennæ, 57;
- Antennal auditory hairs, 292;
- Antheræa, 616.
- Anthrax, 612.
- Anurida, 51;
- maritima, 537.
- Anurophorus, 424.
- Anus, 319;
- Aphides, changes of color in, 205;
- Aphis, 616;
- reduction of tarsal joints of, 103.
- Aphrophora permutata, wings of, 141.
- Apis, premandibular segment in embryo of, 52;
- germ-layers of, 558.
- Apneustic type of tracheal system, 459.
- Apodemes, 92.
- Apodous larvæ, 103.
- Appendages, abdominal jointed appendages, 468;
- Aquatic insects, 459;
- Arachnida, 6.
- Arctia, 391.
- 712Arctian larvæ, 615.
- Argida, 391.
- Armature, 187, 192.
- Arolium, 97, 100, 113.
- Arthromeres, 30.
- Arthropoda, classes of, 3.
- Articerus, 57.
- Ascalaphus, 616.
- Ash, on eversible glands, 377.
- Asilus, mouth-parts of, 79.
- Aspidiotus, 538, 627;
- Ateuchus sacer, 101.
- Attacus, mode of escape from its cocoon, 635.
- Attacine moths, 634.
- Attelabus, 538.
- Auditory organs, 287.
- Audouin, on the median segment, 163;
- on peritreme, 90.
- Autolyca, 371.
- Auzoux, on the salivary glands of silkworm, 332.
- Ayers, on embryonic abdominal appendages, 550;
- Bætisca, 467.
- Balancers, 124.
- Balbiani, on the polar cells of Chironomus, 580.
- Ballowitz, on spermatozoa, 497.
- Band, germinal, 531;
- Bapata, 392.
- Basilar membrane of eye, 253.
- Bee, honey, air-sacs of, 458;
- breathing of, 456;
- cement glands of, 360;
- egg of, 521;
- flight of, 151;
- head, 80;
- moulting of, 611;
- mouth-parts of, 79;
- number of moults of, 618;
- premandibular segment in embryo of, 52;
- salivary glands of, 334;
- sanitary conditions observed by its larva, 623;
- seminal packet of, 500;
- spermatheca, 506;
- tongue of, 80, 81;
- tracheæ of, 458;
- wax glands of, 364.
- Bee’s foot, action of, in climbing, 114;
- sting, 172.
- Bees, twisted hairs of, 189.
- Beetles, anal glands of, 372;
- Benasus griseus, tongue of, 73.
- Bladder, urinary, 35.
- Blanc, on salivary glands of silkworm, 331, 332;
- Blaps, 373;
- Blastoderm, 526, 529.
- Blatta, 43, 69;
- Blattidæ, fœtid glands of, 370.
- Blepharocera, 474.
- Blochman, on embryology of Musca, 530.
- Blood, 407;
- Blow-fly, duration of embryonic life of, 582;
- egg of, 521.
- Boas, on spiracles of Melolontha larva, 439.
- Bobretsky, on embryology of Pieris, 529.
- Body, cavity, formation of, 563, 566;
- Boll, on repellent glands, 371.
- Bombus, 219, 618;
- post-embryonic changes in, 661.
- Bombyx mori, 339, 366, 405, 496, 499, 608;
- embryonic abdominal legs of, 552.
- Bordas, on poison glands, 358;
- on salivary glands of Hymenoptera, 337.
- Bot-fly, of horse, 475;
- of ox, 518.
- Bothriothorax, 623.
- Brain, 222, 226;
- Brauer, on Campodea-form larvæ, 600–602;
- on metamorphosis, 598.
- Breathing, mechanism of, 451;
- rectal, 463.
- Brin, 342.
- Bristles, 188.
- Bruchus, hypermetamorphosis of, 700.
- Buccal appendages, 59.
- Bucculatrix, 634.
- 713Buckton, on change of color in aphides, 205.
- Buds, antennal, 665;
- Bugnion, on composition of head of Hymenoptera, 55;
- Burgess, on colors, 203;
- Burmeister, on organs of smell, 265.
- Bursa copulatrix, 505.
- Busgen, on honey dew, 365.
- Bütschli, on an under-lip structure in bee, 547;
- Butterfly, atrophy of tarsi of, 102;
- Caddis-worm, blood-gills of, 475;
- Cæca of mid-intestine, 300, 325, 347, 348;
- secretion of, 348.
- Calcar, 97.
- Calcaria, 97.
- Calculi in intestine, 325.
- Calliphora vomitoria, 618;
- eggs of, 521.
- Callosamia promethea, 192;
- number of moults of, 616.
- Callosune, 202.
- Caloptenus, 43.
- Calopteryx, 54, 464.
- Caltrops, 189.
- Calypta, 124.
- Calyx of brain, 233.
- Campodea, embryology of, 22, 52;
- Campodea-form larva, 600.
- Campodeoid characters, loss of, in holometabolous insects, 707;
- larvæ, 600.
- Capillary tracheæ, 655.
- Carabidoid stage, 692.
- Carabus, walking, 107;
- tracks of, 109.
- Cardiac valvule, 312.
- Cardioblasts, 572.
- Cardo, 63.
- Carlet, on the poison apparatus of bees, 357;
- Carus, on the circulation, 397, 409.
- Case-worms, blood gills of, 475;
- Caterpillar, actions before pupation, 644;
- Catocala, 392.
- Cauliculus, 233.
- Cavity, peripodal, 669.
- Cecidomyia, 113;
- urinary tubes of, 351.
- Cells, absorbent, 328;
- Cement glands, 360.
- Centrosome, 525.
- Ceratopogon, 678.
- Cerci, 164, 178.
- Cercopoda, 164, 178.
- Cerura, 375.
- Ceuthophilus, 393.
- Chabrier, on use of elytra, 159.
- Chalicodoma, 542.
- Chambers, egg, 502;
- yolk, 502.
- Chapman, on cremaster, 636;
- Chermes, 361.
- Cheshire, on admission of air into bee’s cocoon, 623;
- Chiasma, 231.
- Chironomus, 36, 491;
- Chitin, 29.
- Chlænius, brain of, 241.
- Cholodkowsky, on homologies of propleg or abdominal leg of caterpillars, 552;
- Chordotonal organs, 289.
- Chorion, 520, 534.
- Chromatin, 498.
- 714Chrysalis, 625;
- mode of suspension of, 637.
- Chrysopa, 517, 525.
- Chun, on the tænidia, 445.
- Cicada, shrilling organ of, 295.
- Cicada septemdecim, 616;
- hatching of, 584.
- Cimbex, 374.
- Circulation, of blood, 409;
- Citheronia, 392.
- Claspers, 176, 179.
- Claus, on eversible glands, 374.
- Clavola, 57.
- Climbing, mode of, 116.
- Closure, dorsal, of embryo, 556.
- Clypeus, 546, 547.
- Coarctate Diptera, 620.
- Coccidæ, male, 626;
- metamorphosis of, 641.
- Coccinella, moulting of, 611.
- Coccinellidæ, 375.
- Cockerell, on hatching of mantis, 584.
- Cockroach, 455, 456, 487;
- brain of, 229, 242;
- cement glands of, 360;
- chorion of egg of, 521;
- circulation of blood in wings of, 410;
- colleterial glands, 506;
- deposition of eggs of, 519;
- digestion of, 325;
- egg-tubes of, 501;
- fœtid glands of, 370;
- micropyle of eggs of, 523;
- mode of hatching, 583;
- oötheca of, 517;
- wingless, 598.
- Cocoon, admission of air in, 623;
- Cœcal appendages of stomach, 300, 325, 347.
- Cœcum of colon, 318, 325.
- Cœlom-sac, 563, 566, 576.
- Coleoptera, embryology of, 537;
- gustatory organs of, 284;
- internal changes during metamorphosis of, 641;
- larval types of, 604, 606;
- number of moults of, 617;
- olfactory organs of, 275;
- phosphorescent, 421;
- pupa of, 630;
- salivary glands of, 334;
- seminal ducts of, 496;
- sounds produced by, 293;
- spermatozoa of, 497, 499;
- tongue of, 73.
- Colleterial glands, 506.
- Colon, 317;
- cæcum of, 318.
- Color sense, 260.
- Colors, 201;
- Comb, tarsal, 97.
- Commissure of œsophageal ring, 237.
- Conditions of existence, 463.
- Cone, crystalline, 250, 251.
- Conglobate gland, 487.
- Coniopteryx, 620.
- Conjunctivus, 61.
- Conorhinus, 616.
- Cope, on causes of segmentation of body of arthropods, 33.
- Copidosoma, 623.
- Copris carolina, 61.
- Copulation, signs of, 507.
- Copulatory pouch, 505.
- Cord, stigmatic, 460;
- supraspinal, 240.
- Corethra, 433, 460, 618;
- Corixa, eggs of, 538.
- Cornea, 250.
- Corneal lens, 250.
- Corydalus, 46, 48, 59, 70, 460, 468.
- Corydalus cornutus, hatching spine of, 585.
- Coste, on pigments, 206.
- Cotylosoma, 478.
- Coxa, 95;
- origin of imaginal from larval, 656.
- Coxal glands, 369;
- Cremaster, 636;
- Cremastral hook-spine, 638.
- Cricket, 487;
- anal glands of, 372.
- Crop, 303, 324.
- Crustacea, 4.
- Cucujo, 426.
- Cuénot, on blood, 374, 408;
- Cuilleron, 124.
- Culex, 461, 465, 474, 599;
- Cup, spermatophore, 499.
- Cuticula, 187, 203;
- new layer of, formation of, 612.
- Cyclops stage of Proctotrypid parasites, 701.
- 715Cyphon, 472.
- Cytoplasm, 525.
- Dahl, on constancy of number of six legs, 100;
- Danais, 197, 381;
- Dandolo, on amount of food eaten by the silkworm, 608.
- Datana, 634.
- Datana ministra, moulting of, 611;
- Death-watch, 293.
- Deltochilum gibbosum, 101.
- De Moor, on tracks of insects, 106, 109.
- Dermal glands, 365.
- Dermaptera, wingless, 598.
- Deutocerebrum, 231, 237.
- Development, direct, 598.
- Dewitz’s discovery of imaginal bud-stalks, 673;
- Diapheromera, 371, 616;
- Diaphragm, pericardial, 412.
- Didonis, 381, 391.
- Digestion, 324.
- Digestive canal of imago in the fly, appendages of, 297, 302, 331;
- Dimmock, on hypopharynx, 71;
- Diplopoda, 12.
- Diptera, coarctate, 620;
- cyclorhapha, 621;
- development of imago of, 666;
- food reservoir of, 305;
- germs of genital glands, 580;
- hypopharynx of, 78;
- larval types of, 607;
- mouth-parts of, 78;
- olfactory organs of, 273;
- origin of legs of imago in, 654;
- orthoraphous, 621, 626;
- post-embryonic changes in, 666;
- salivary glands of, 333.
- Dipterous embryo, suppression of polypody in, 707.
- Direct development, 598.
- Discota, 672.
- Division nuclei, 530.
- Donacia, 620.
- Dorsal organ, 535.
- Doryphora, 50;
- Dragon-fly, 53;
- Draught power, 218.
- Drosophila, 518.
- Dryocampa, 187.
- Dubois on the cucujo, 424, 426.
- Ducts, ejaculatory, 496;
- seminal, 496.
- Dufour, gland of, 358.
- Dujardinia, 34.
- Dyar on the number of moults, 615.
- Dyticus, 461;
- Eacles, 616.
- Ears, 288.
- Eaton, on nymph stage, 594;
- of rectal respiration in nymphs of ephemerids, 465.
- Ecdysis, 609, 611.
- Ectoderm, 534;
- formation of, 558.
- Ectotrachea, 432, 448, 684.
- Egg, 515;
- burster, 585;
- capsule, 517, 520;
- cells, 504;
- chambers, 502;
- fertilization of, 525;
- germs, 502;
- guide, 183;
- internal structure of, 524;
- markings of, 521;
- maturation of, 525;
- mode of deposition of, 518;
- number laid, 515;
- ovarian, 501;
- ripe, 520;
- sacs, 361, 517, 520;
- shell, 520;
- smaller in holometabolous insects, 515;
- tubes, 501;
- vitality of, 520.
- Ejaculatory ducts, double openings of, 486;
- origin of, 578.
- Elater of Collembola, 551.
- Electricity, influence of, on action of heart, 412.
- Eleodes, 372.
- Elliott on color sense, 261.
- Elmis, 462, 473.
- Elytra, 124;
- glands in, 125.
- Embidæ, 620.
- Embryo, 531;
- revolution of, 540.
- Embryology of insects, 515.
- Embryonic life, length of, 582.
- Embryonic membranes, involution of, 556.
- 716Embryonic, post-, changes, 650.
- Emery, on homologies of the tracheæ, 443;
- Empodium, 97, 111, 116.
- Empretia stimulea, 192.
- Eucyrtus, 55, 171;
- post-embryonic changes in, 663.
- Endochorion, 520.
- Endoderm, 534, 561.
- Endomesoderm, 542.
- Endosternite, 94.
- Endotrachea, 432, 448.
- Entomoline, 29.
- Entothorax, 92.
- Environment, 463.
- Ephemera, circulation in, 409;
- Ephemerella, 466.
- Ephemeridæ, 459, 460;
- Ephydra, 36, 553.
- Epicauta, life-history of, 692.
- Epilabrum, 13.
- Epimerum, 89.
- Epiopticon, 231, 253.
- Epipharynx, 43, 54.
- Epipleurum, 124.
- Episternum, 88.
- Erichson on sense of smell, 266.
- Eriocephala calthella, mandibles of, 62;
- maxillæ of, 68.
- Eristalis, 189, 461.
- Eruciform type of larva, 602, 605, 705.
- Escherich, on male genital organs of beetles, 495.
- Euphæa, lateral gills of, 468, 477.
- Euphoria inda, moulting of, 611.
- Eupolus, 113.
- Eupsalis minuta, 103.
- Eversible sacs, 475.
- Excretion, defined, 327;
- process of, 328.
- Excretory system, 348.
- Exner, on vision, 258.
- Exochorion, 520, 534.
- Exuvia, 609.
- Exuvium, 609.
- Eye, 249;
- Fabre, on life-history of Sitaris, 69.
- Facet, 250.
- Facets, number of, 249.
- Facetted eye, origin of, 255.
- Faivre, on function of brain, 244.
- Fat, amount of, in caterpillar, 644.
- Fat-body, 419;
- Feet, post-embryonic development of, 653.
- Female reproductive organs, 485, 500;
- origin of, 575.
- Femur, 96;
- formation of, in imago of Lepidoptera, 655.
- Fernald, on rectal glands of Passalus, 318.
- Fertilization of the egg, 525.
- Filator, 340.
- Filippi’s glands, 345.
- Firefly, 426.
- Fischer, on color of butterflies, 200.
- Flagellum, 57.
- Flea, 438;
- Flies, syrphid, 189.
- Flight, 148, 219;
- theory of, 150.
- Fluid, exuvial, 612;
- Fly, blow, hatching of, 585;
- Fold, amnion, 531.
- Folds, gastro-ileal, 317;
- giving rise to head of fly, 671.
- Folsom, on lateral gills of Euphæa, 468.
- Food-reservoir, 305.
- Foot, of beetle, 111;
- of fly, 111.
- Footprints of beetles, 106.
- Fore-stomach, 306.
- Forel, on gustatory organs, 281;
- Forficula, 454, 491;
- Formic acid, 358.
- Frenulum, 122.
- Fulgora, 424.
- Funiculus, 460.
- Funnel, 313.
- 717Galea, 63, 64.
- Galeruca, 113.
- Ganglia, function of, 244;
- Ganglion frontale, 569.
- Ganglion opticum, origin of, 567.
- Ganin, on abdominal imaginal buds, 170;
- on hypermetamorphosis of ichneumon parasites, 701.
- Gastro-ileal folds, 317.
- Gastropacha, 552;
- flattened hairs of, 194.
- Gastrophilus equi, 475.
- Gastrula, 558;
- stage, 535.
- Gegenbauer, on homology of wings with gills, 142.
- Gehuchten, on histology of muscles, 217;
- Gena, 46.
- Genital armature, male, 176;
- Germarium, 501.
- Germ-layers, formation of, 558.
- Giard on urinary tubes, 351.
- Gills, blood, 475;
- Gilson, on anal glands, 373;
- on spinning-glands, 340.
- Gissler on anal glands, 372.
- Gizzard, 311, 324.
- Glands, acid, 358;
- adhesive, 360;
- alkaline, 358;
- alluring, 391;
- of androconia, 199;
- anal, 319, 372;
- accessory of vasa deferentia, 497;
- cement, 360;
- colleterial, 506;
- conglobate, 487;
- corrosive, 374;
- coxal, 369, 383;
- dermal, 365;
- defensive, 368;
- eversible, 368, 382;
- Filippi’s, 346;
- fœtid, 369;
- mucous, 497;
- mushroom, 497;
- odoriferous, 381;
- repugnatorial, 368;
- salivary, 331, 570;
- setiparous, 444;
- sexual, origin of male, 579;
- unicellular, 366;
- wax, 361.
- Glandulæ mucosæ, 497.
- Gnathal segments of embryo, 556.
- Gonapophyses, 167, 168.
- Gonin, on moulting fluid, 613, 614;
- Gorgeret, 170.
- Gottsche on vision, 257.
- Graber, on abdominal legs of caterpillars, 552;
- on blood, 408;
- on blood-gills of embryo, 476;
- on climbing, 116;
- on development of wings, 138;
- on flight, 153;
- on folding of wings, 155;
- on foot-tracks of beetles, 109;
- on heart, 398, 400, 402;
- on mechanics of segmented body and limbs of insects, 31, 38;
- on mechanics of walking, 103;
- on organs of hearing, 290;
- on organs of smell, 267;
- on premandibular segment, 52;
- on respiration, 454;
- on successive appearance of embryonic segments, 546;
- on swimming, 116.
- Grassi, on premandibular segment in Apis, 52;
- on Scolopendrella, 20.
- Grège, 340.
- Grès, 340.
- Griffiths, on pigments, 207.
- Grobben on heart, 399.
- Gromphas, 101.
- Gross, on color sense, 261.
- Gryllotalpa, 572;
- maxilla of, 64.
- Guide, egg, 183.
- Gula, 46, 68.
- Gulo-mental region, 46.
- Gummy layer of silk, 340.
- Gyrinus, 471, 620.
- Haase, on coxal sacs, 14;
- Hadenœcus, 44, 392.
- Hagen, on colors, 201;
- Hair-fields, 197.
- Hair-forming cells, 188.
- Hair-scales, 197;
- tactile, 193.
- Hairs, 188;
- Halteres, 124, 629.
- Hammond, see Miall.
- Hampson, on scent-glands, 391.
- Haplopus, 521.
- Harpes, 180.
- Harpiphorus, 618.
- Harpyia, 375.
- 718Hatching, process of, 583;
- spine, 585.
- Hauser, on organs of smell, 267, 269, 279.
- Head, 42;
- Hearing, organs of, 287.
- Heart, 397;
- Heathcote, on double segments of Diplopods, 14.
- Heider, on embryology of Hydrophilus, 530.
- Heinemann, on the firefly, 426.
- Helcodermatous spines, 612.
- Helecomitus, 616.
- Helichus, 474.
- Heliconidæ, 379.
- Helm, on spinning-glands, 339.
- Hemelytra, 124.
- Hemerobiidæ, hatching spine of, 585.
- Hemimetabola, 598.
- Hemiptera, cardo of, 69;
- Hepialus, 392, 495.
- Heptagenia, 467;
- lingua of, 73.
- Heredity, bearer of, 498;
- homochronous, 708.
- Heremetabola, 597.
- Herold, on the metamorphosis of the butterfly, 642;
- on wing-germs, 128.
- Heterochrony, 542.
- Heterometabola, 597.
- Heymons, on homologies of the labrum, 43;
- Hicks, on auditory organs, 293;
- on olfactory organs, 266.
- Histogenesis, 650.
- Histolysis, 643, 650, 678, 680, 685, 688.
- Hoffbauer, on the structure of elytra, 125.
- Holmgren, on tracheal end-cells, 437.
- Holometabola, 598.
- Holometabolous insects, 595.
- Holopneustic type of tracheal system, 459.
- Holoptic head, 98.
- Homalotylus, 623.
- Homoptera, number of moults of, 616.
- Homotenous insects, 597.
- Honey-dew, 364;
- Hopkins, on pigments, 207.
- Hornia, hypermetamorphosis of, 693.
- Horns, 187.
- Horn, on loss of tarsi, 101;
- on Platypsylla, 62.
- House-fly, thorax of, 88.
- Hum of bee, 295.
- Hunter, John, on the air-sacs, 456.
- Hurst, on the formation of the imago in Culex, 670.
- Hybocampa, 635.
- Hydrobius, 471.
- Hydrophilus, 374, 432;
- Hydropsyche, blood-gills of, 475;
- gills of, at all stages, 469.
- Hydroüs, 49, 50.
- Hylotoma, 52.
- Hymenoptera, composition of the head in, 55;
- Hyperchiria io, 187, 378;
- poisonous spines of, 192.
- Hypermetamorphosis, described, 688;
- causes of, 693.
- Hypodactyle, 73.
- Hypoderma, 518.
- Hypodermis, 188, 612;
- origin of an imago, 678.
- Hypopharynx, 13, 54, 68, 70.
- Hypoptère, 89.
- Icerya, 616, 626.
- Ichneumon, 622;
- Ileum, 317.
- Imaginal buds, 653;
- disks, 653.
- Imago, formation of, in Chironomus, 671;
- 719Incasement theory, 641.
- Indusium, 534.
- Infra-anal lobe, 183.
- Infraœsophageal ganglion, 227.
- Ingluvies, 303.
- Insecta, diagnostic characters of, 26.
- Insects, ancestry of, 17;
- Intestine, fore, embryonic development of, 547;
- Invaginations of the imaginal buds, 678.
- Involution of the embryonic membranes, 556.
- Isotoma, 534.
- Jackson, on the structure of the cremaster and pupa, 639.
- Janet on muscular fibres, 216.
- Japyx, 486.
- Jolia, 466.
- Jugum, 123.
- Julus, larva of, 14.
- Katydid, 616.
- Kellogg, on Androconia, 199;
- Kennel, on origin of tracheæ of Peripatus, 443.
- Kenyon, on double segments of Diplopods, 14;
- on mushroom bodies, 234.
- Kettelhoit, on specific characters of, 195.
- Kingsley, on classification of Myriopoda, 12.
- Kirbach, on salivary duct, 336.
- Kirby and Spence, views of, on metamorphosis, 642.
- Klapálek, on gills of case-worms, 467;
- on sub-nymph of Agriotypus, 701.
- Klemensiewiez, on eversible glands, 377.
- Kolbe, on atrophy of tarsi, 101;
- Korschelt, on the egg-tubes, 502;
- on egg-genesis, 504.
- Korschelt and Heider, on the embryology of insects, 531, 535, 538, 554, 559, 570, 579;
- Koulaguine, on dorsal opening of urinary tubes, 355.
- Kowalevsky, on embryonic abdominal appendages, 550;
- on embryonic membranes, 562;
- on origin of blood corpuscles, 574;
- experiments on feeding maggots with lacmus, 326;
- on fat-body, 420;
- on embryo origin of fat-body, 574;
- on the mesoderm of the rudiments of the appendages, 675;
- on openings of heart, 400;
- on pericardial cells, 420;
- on phagocytes, 421, 422, 655;
- phagocytosis, 686.
- Kraepelin, on homologies of the ovipositor, 168;
- Krancher, on the stigmata, 438.
- Krauss, on eversible glands, 371.
- Krawkow, on chitin, 29.
- Krukenberg, on colors, 202, 205, 206.
- Künckel d’Herculais, on beating of heart throughout the post-embryonic changes, 686;
- on the origin of the imaginal from the larval legs, 654.
- Kupffer, on fine tracheæ, 435.
- Labella, 13, 446.
- Labial palpi, imaginal buds of, 658.
- Labidura, 491.
- Labium, 68, 549.
- Labrum, 42, 79;
- Lacaze-Duthiers on the ovipositor, 167, 169.
- Lace-winged fly, 517.
- Lachnosterna, nervous system of, 225.
- Lachnus, 372.
- Lacinia, 63;
- Lady-birds, 375;
- bug, 375.
- Lagoa crispata, 191, 378.
- Lamarckian factors, 708.
- Lamina supra-analis, 181.
- Lampyris, 425, 451.
- Landois, on pigment, 207;
- 720Lang, on metamorphosis of seventeen-year Cicada, 698;
- Langley, on the light of the firefly, 426.
- Larva, defined, 599;
- Larvæ, apodous, 103, 653.
- Larval insects, tracheal gills of, 466;
- stage, 593.
- Latreille, on the median segment, 163;
- Latzel, on coxal sacs, 14.
- Layer, superficial protoplasmic, of egg, 524, 526;
- germ, formation of, 558.
- Leach, on ametabola, etc., 596.
- Leaping power, 219.
- Legs, abdominal, of lepidopterous larvæ, and larval saw-flies, are they true legs?, 552;
- Lehrman, on organs of smell, 265.
- Leidy, on fœtid glands, 373.
- Lens, crystalline, 250, 251.
- Lendenfeld on flight, 149, 151, 159.
- Léon, on labial palpi of Hemiptera, 68;
- tongue of Hemiptera, 73.
- Lepidoptera, embryology of, 537;
- Lepisma, 52;
- double sexual openings in, 486.
- Leptiform larvæ, 600.
- Leptis, thorax of, 91.
- Leucarctia, 391.
- Leucine, 352.
- Leucocytes, 407, 421, 650, 678, 680, 685;
- size of, 407.
- Leucopis, 113, 517.
- Leydig, on colors, 202, 204;
- Libellula, 463.
- Life, embryonic, length of, 582.
- Light of the firefly, 426;
- its use, 428.
- Ligula, 68.
- Limacodes scapha, 606.
- Limbs, homologies of, 39;
- Limnephilus pudicus, 46;
- maxilla of, 65.
- Limulus, 5.
- Lina, 374, 545, 546.
- Lingua, 68, 70.
- Lip, under, 68.
- Lipochrome, 206.
- Liponeura, 475.
- Lithocolletis, 618;
- its cocoon-cutter, 634.
- Litognatha nubilifasciata, 102.
- Lobe, axillary, 124;
- infra-anal, 183.
- Lobes, œsophageal, 237;
- Lobulus, 124.
- Locomotion, 103;
- on smooth surfaces, 111.
- Locust, air-sacs of, 424, 456;
- brain of, 231;
- cæcal appendages of, 347;
- digestive canal of, 298;
- ear of, 288;
- head-segments of, 546;
- mode of breathing, 451;
- hatching, 583;
- moulting of, 609;
- nervous system of, 223;
- number of stages of, 595;
- number of moults of, 616;
- olfactory organs of, 272;
- oviposition of, 520;
- rectal glands of, 318;
- reproductive organs of, 488, 489.
- Locusta viridissima, rectal glands of, 318.
- Locy, on pulsatile organs in legs of Nepidæ, 405.
- Lonchodes, 521.
- Loop of wing, 122.
- Lophyrus, 59.
- Lora, 68.
- Lubbock, on color sense, 261;
- Lucanus, 59, 620;
- Lucas, on segmental arrangement of salivary glands, 337.
- Luciola, 424, 427, 451.
- Luna moth, its mode of escape from its cocoon, 635.
- Lutz, on blood, 275.
- Lycæna, 381.
- Lymph, 206.
- Lyonet, on the imaginal buds, 656;
- on wing-germs, 128.
- 721Machilis, 164, 223, 369, 476;
- hypopharynx, 72.
- MacLeod, on the tænidia, 445.
- Macloskie, on the tænidia, 445.
- Macrotoma, 616.
- Macrurocampa, 375.
- Maggots, 607;
- Malachius, 374.
- Male reproductive organs, 485, 494;
- origin of, 579.
- Malpighi, on germs of wings, 128;
- Malpighian tubes, 316, 348.
- Mandibles, 59;
- Manometabola, 597.
- Mantidæ, coxal glands of, 372.
- Mantis, oötheca of, 517.
- Mantis religiosa, embryo of, 584;
- Mantispa, 46, 48, 68, 95, 97;
- Marchal on the function of the fat-body, 420.
- Marey, on motion of insects, 111.
- Marey’s views on flight, 148, 151.
- Marshall, on the way Microgaster spins its cocoon, 622.
- Mason, Wood, on jointed structure of mandibles, 60;
- Mastopoda pteridis, 103.
- Maxillæ, first, 62;
- Mayer, A. G., on the development of the wings of Pieris and Danais, 136;
- Mayer, Carl, on scales, 197.
- May-fly, lingua of nymph of, 73;
- Mechanics of walking, 103.
- Mechanism of motion, 32;
- Meconium, 611.
- Mecoptera, maxillæ of, 65;
- number of moults of, 616.
- Median segment, 90, 163.
- Medifurca, 92.
- Megalopyge, 191, 378.
- Meinert, on buccal organs of myriopods, 13;
- Melanoplus, 43, 72, 86, 456;
- Meldola, on yellow pigment, 206.
- Melipona, 359.
- Meloë, 110, 374;
- Melolontha, 213, 438, 455, 456, 458, 498, 549, 550, 551, 562.
- Melophagus, 507;
- post-embryonic changes in, 686.
- Membrane, peritrophic, 313;
- Membranes, embryonic, 531;
- Menge on Scolopendrella, 18.
- Mentum, 54, 68.
- Merostomata, 5.
- Mesoderm, 534, 561, 563;
- cells, 576.
- Mesothorax, 86.
- Metabolia, 596.
- Metabolous insects, 595.
- Metameric structure, 33.
- Metamorphoses of insects, 593;
- Metamorphosis, causes of, 607, 705, 708;
- significance of, 688.
- Metapneustic type of tracheal system, 461.
- Metathorax, 87.
- Metschnikoff, on embryology of myriopods, 13, 16;
- on the germs of the genital glands, 580.
- Miall and Denny, on the blood, 407;
- Miall and Hammond, on the differences between the pupa of Lepidoptera and Diptera, 629;
- on the formation of the imago of Chironomus, 671.
- 722Microcentrum, 616.
- Microgaster, mode of spinning its cocoon, 622.
- Micropteryx, 621, 626;
- Micropyle, 522;
- use of, 524.
- Mid-intestine, 314;
- origin of, 569.
- Minchin, on eversible glands, 370.
- Minot, on cæca of stomach, 347;
- Molar, 61.
- Mole-cricket, 102, 527, 543, 572, 574;
- Mosaic theory of vision, 257.
- Moseley, on circulation of blood, 410;
- on composition of chitin, 30.
- Mosquito, 461, 464, 465;
- Moulting, process of, 609;
- hairs and spines, 612.
- Moults, number of, 615.
- Mouth, 302;
- Müller, F., on blood-gills, 475;
- Müller, J., on alluring glands, 391;
- Müller, W., on gills of Paraponyx, 470.
- Müller’s, J., thread, 577.
- Mumia, 625.
- Musca, 88, 111;
- Muscidæ, appendages of imago, development of, 674;
- Muscles, 31;
- Muscular fibres, 214;
- Musculature, mode of origin of, 574.
- Mushroom bodies, 233.
- Mutilations, inheritance of, 102.
- Myriopoda, 11.
- Myrmeleon, maxilla of, 64;
- palpifer of, 69.
- Mystacides, scent scales of, 199.
- Nagel, on saliva of larval Dyticus, 324.
- Nassonow, on double sexual openings, 486.
- Necrophorus, tracks of, 109.
- Nematois, 496.
- Nematus, 53, 54, 618.
- Nemognatha, epipharynx of, 285;
- Nemoptera, larva of, 42.
- Nemoura, 468.
- Neolamarckian factors, 708.
- Neolepidoptera, 628.
- Nepa, 523.
- Nephridia, 348.
- Nepionic stage or form, 706.
- Nepticula, 606.
- Nerve-centre, 222.
- Nerve-centres, functions of, 243;
- antennal, 650.
- Nerves, motor, 222;
- Nervous system, 222;
- Neuroblasts, 567.
- Neuromeres, 227, 231.
- Neuroptera, 632;
- lingua of, 69.
- Newman, on the median segment, 163.
- Newport, on changes in nervous system of Sphinx during metamorphosis, 648;
- on circulation, 409;
- on heart, 399;
- on larval Julus, 13;
- on the median segment, 163;
- on muscular power, 94;
- on muscles of Sphinx, 213;
- on number of segments of head, 50;
- on occiput, 48;
- on the process of moulting in Sphinx, 610, 611;
- Scolopendrella, 18;
- on sense of smell, 265;
- on tentorium, 49;
- on tracheal gills of Pteronarcys, 476.
- Nola, 618.
- Nucleus, division, 530;
- sperm, 525.
- Nusbaum, on origin of efferent sexual passages, 578.
- 723Nymphalid pupæ, 631.
- Nymph, 706;
- Occipital foramen, 46.
- Occiput, 48, 53.
- Ocellus, 249;
- development of, 567.
- Ockler, on feet of insects, 115.
- Odonata, 53;
- Odors, 368.
- Œcanthus, 476;
- Œceticus, 634.
- Œnocytes, 423.
- Œsophageal valve, 311;
- valvule, 312.
- Œsophagus, 303.
- Œstridæ, 618.
- Oken, on homology of maxillæ with legs, 39.
- Olfactory organs, 264.
- Oligonephria, 354.
- Oligoneuria, 466.
- Ommateum, 250.
- Onychium, 97.
- Oölemma, 520.
- Oötheca, 517.
- Operculum, 181.
- Optic nerves, 650.
- Optic tract, 253.
- Opticon, 253.
- Oral appendages, 549.
- Orchelimum, 534.
- Organ, dorsal, 535, 556.
- Organs, sensory, 249;
- of smell, 264.
- Orgyia, 377, 618;
- poisonous hairs of, 192.
- Orgyia antiqua, number of moults of, 618.
- Orthoptera, fœtid glands of, 369;
- Orya, 424.
- Osmeterium, 377.
- Osten Sacken, on holoptic heads of Diptera, and on running flies, 98.
- Ostia, 397, 400.
- Otiocerus, 58.
- Oudemans, on relation of myriopods to insects, 17.
- Oustalet, on rectal respiration, 463.
- Ovarian tubes, 501;
- formation of, 578.
- Ovaries, 500, 502;
- Oviduct, 500, 503;
- Oviducts, segmental arrangement of, 486.
- Oviposition, 518, 520.
- Ovipositor, 167;
- Ovum, 515, 521, 524.
- Packard, A. A., on muscular power of insects, 219;
- use of air-sacs, 457.
- Pad, peripodal, 653.
- Pædogenesis, 708.
- Paleacrita vernata, maxilla of, 66.
- Paleolepidoptera, 628.
- Palmén, on double sexual openings, 490, 492;
- Palmula, 97.
- Palpifer, 63, 68.
- Palpus, first maxillary, 63, 64, 67;
- Palpi, labial, imaginal buds of, 658.
- Pancritius, on wing-germs, 130, 143.
- Paniscus, 517.
- Panorpa, abdomen of, 162;
- Panorpidæ, 602.
- Papilio, 377.
- Paraglossa, 54, 68.
- Parapodial glands, 444.
- Paraptera, 89.
- Parnassius, 381.
- Paronychium, 97.
- Paraponyx, 470.
- Passalus, 61;
- rectal glands of, 318.
- Pasteur, on the spectrum of the light of the firefly, 426.
- Patagia, 89.
- Patten, on embryonic abdominal appendages, 550;
- Patula, 391.
- Pauropus, 18.
- Paussus, 57.
- Pawlowa, on blood-vessels in the head, 405.
- Pedicel, 57.
- Pediculina, embryology of, 541.
- Pelidnota, moulting of, 611.
- Pellicle, subimaginal, 613.
- Pelobius, 461, 475.
- 724Penis, 180;
- velum of, 181.
- Pericardial cells, 405;
- Periopticon, 231, 253.
- Peripatus, 9, 580;
- Peripodal cavity, 669;
- Periplaneta, 72, 370;
- Peritoneal membrane, 444.
- Peritracheal circulation, 397;
- membrane, 444.
- Peritreme, 90.
- Peritrophic membrane, 313.
- Perlidæ, 69, 468, 491, 493.
- Perris, on organs of smell, 266.
- Phagocytes, 421, 650, 655, 680, 683, 685.
- Phagocytosis, 421.
- Phanæus, 61;
- reduced tarsi of, 101.
- Phanæus pegasus, 188.
- Phaneroptera, 44.
- Pharynx, 302.
- Phasmidæ, eggs of, 521.
- Phosphorescence, 424;
- physiology of, 426.
- Photogenic organ of beetles, 424.
- Phragma, 93.
- Phryganea, 455.
- Phyllium, 521.
- Phyllocnistis, 606.
- Phyllodromia, 506;
- Phytonomus, 617.
- Phytophagous larvæ, 606.
- Pictet, on blood gills, 475.
- Pieris, 39, 614;
- Pigment, 183, 203;
- Pits, olfactory, 271.
- Planta, 638.
- Plantula, 97.
- Plate, extensor, of foot, 116;
- pressure, 116.
- Plateau, on digestion, 324;
- Platycrania, 521.
- Platygaster, hypermetamorphosis of, 701;
- sexual cells of, 581.
- Platypsyllus, 62.
- Platysamia, 460.
- Platyzosteria, 371.
- Plectoptera, 459, 466.
- Pleuropodia, 476, 551, 583.
- Pleurum, 87.
- Plumules, 198.
- Pocock, on classification of myriopoda, 12, 21.
- Poduridæ, 574.
- Poisonous spines, 191, 199.
- Poisons, effect of, on pulsations, 412;
- Poison apparatus, 357;
- Polar cells, 580.
- Polymitarcys, 467.
- Polymorphous insects, 597.
- Polynema, hypermetamorphosis of, 702.
- Polynephria, 354.
- Polyphemus silkworm, 621.
- Polypodous ancestor of insects, 22;
- embryos, 550.
- Polypody, 550;
- suppression of in dipterous embryos, 707.
- Pore-canals, 188.
- Porthesia, 529.
- Postfurca, 92.
- Post-gula, 54, 68.
- Post-retinal fibres, 231, 232.
- Postscutellum, 87.
- Pouch, copulatory, 505.
- Poujade, on flight, 159.
- Poulton, on the differences between the limbs of the pupa and imago, 628.
- Præscutum, 87.
- Pratt, on absence of polypodous embryo in Muscidæ, 55;
- Preantennal appendages, 548.
- Premandibular segment, 51, 52, 549.
- Press of spinning apparatus, 341.
- Primitive band, 531;
- streak, 531.
- Prionocyphon, 472.
- Prisopus, 69, 477.
- Proboscis, 446.
- 725Procephalic lobes, 544.
- Proctodæum, 537, 547, 569.
- Prodoxus, 606.
- Progoneate myriopods, 21.
- Pronotum, 87.
- Pronucleus, 526.
- Propodeum, 163.
- Propupa, 627.
- Prosopistoma, 467.
- Prostheca, 61.
- Prothorax, 86.
- Protocerebrum, 231, 232;
- its representative in annelids, 227.
- Proventricular valvule, 313.
- Proventriculus, 306;
- Prussic acid, 374.
- Psephenus, 462, 473.
- Pseudoglomeris, 598.
- Pseudonychium, 97.
- Pseudo-tracheæ, 446.
- Psocus, 616.
- Psychodes, 474.
- Psylla, 361, 518;
- Pteronarcys, 468, 476.
- Pterygodes, 89.
- Pterygota, 27, 595.
- Pulex, number of moults of, 617.
- Pulex canis, hatching spine of, 586;
- hypopharynx of, 77.
- Pulling power, 218.
- Pulse, 411.
- Pulvillus, 97, 114, 116.
- Pump, pharyngeal, 302;
- Pupa, coarctate, 621, 626;
- Pupa, semi, 691.
- Pupal, pseudo-, stage, 691;
- sustainers, 638.
- Puparium, 621.
- Pupation, mechanism of, 661;
- process of, 659.
- Pupipara, post-embryonic changes in, 686.
- Pushing power, 218.
- Pygidium, 163.
- Pyloric valvule, 315.
- Pyrophorus, 424.
- Pyrrarctia, 617.
- Pyrrhocoris, 372.
- Quiescent pupal life, 598.
- Ranatra, 523.
- Raphidia, 631.
- Raschke, on the rectal respiration of Culex, 465.
- Rath, Vom, on larval Julus, 13.
- Ratzeburg, on composition of head in Hymenoptera, 55.
- Réaumur, on the cremaster, 637;
- Rectal glands, 318;
- tracheal gills, 463.
- Rectum, 318;
- of embryo, 577.
- Reinhard, on head of Hymenoptera, 55;
- on the median segment, 164.
- Reproduction, organs of, 485;
- origin of, 575.
- Repugnatorial glands, distribution of, 382.
- Respiration, 430, 451;
- rectal, 463.
- Respiratory system, 430;
- mechanism of, 451.
- Resting stage, 707.
- Retina, origin of, 568.
- Retinula, cells of, 250, 253.
- Rhabdites, 167, 517.
- Rhabdom, 250.
- Rhabdopoda, 176.
- Rhipiphorus paradoxus, 697.
- Rhopalum, 636;
- pupal spines of, 636.
- Rib, Semper’s, 121.
- Ribs of wings, 146.
- Ridges, primitive, of nervous system, 554.
- Riley, on the cremaster, 637;
- Rods of eye, 253.
- Rods, visual, of eye, origin of, 568.
- Rombouts, on locomotion of insects on smooth surfaces, 114.
- Ruptor ovi, 585.
- Ryder, on loss of tarsi, 101;
- on Scolopendrella, 19.
- 726Sacs, air, 456;
- Saliva, 324;
- poisonous, 359.
- Salivary duct of Stomoxys, 446;
- Savigny on epipharynx, 43;
- Saw-fly, 374.
- Scale-hairs, 198.
- Scales, 187, 193, 202;
- Scape, 57.
- Scent-glands, 39.
- Scent-scales, 198.
- Scepsis, 618.
- Schæffer, on blood, etc., in the pupal wings, 146;
- Schatz, on colors of butterflies, 202.
- Schiemenz, on salivary glands of bees, 334.
- Schindler, on urinary tubes, 351.
- Schiödte on blood-gills, 475.
- Schmidt, on the metamorphosis of male Coccidæ, 640;
- Schneider, on the funnel of the proventriculus, 313;
- on spermatophores, 500.
- Sciara, 348, 636.
- Sclerites, cervical, 46.
- Scolopendrella, 18;
- Scudder on the glazed eye of pupal butterflies, 631.
- Scutellum, 87.
- Scutum, 87.
- Secretion, definition of, 327;
- Sectores coconis, 634.
- Segment, antennal, 227;
- Segmental arrangement of genital glands, 486.
- Segments, number of, in head, 50, 68, 227, 229;
- Seirarctia, 618.
- Selandria larva, mouth-parts of, 68.
- Seminal ducts, 496.
- Semipupa, 691.
- Semper, ground-membrane of, 136;
- on origin of hair-scales, 195.
- Sense organs, special, in flies, 293.
- Sensory organs, 249.
- Serosa, 532.
- Setæ, 188.
- Sexual differences, secondary, 59, 99, 101, 114;
- Sharp, on causes of segmentation of Crustacea, 33;
- Sheep-tick, 507;
- post-embryonic changes of, 686.
- Sialidæ, 602.
- Sialis, 462, 468.
- Siebold, organ of, 290;
- on spermatophores, 500.
- Silk, 340;
- composition of, 346.
- Silk-fibre, composition of, 346.
- Silk-gland, 339;
- Silkworm, 331, 339, 366;
- Simmermacher, on feet of insects, 113.
- Simulium, 668, 678;
- Sinclair on double segments of Diplopods, 14.
- Sisyra, abdominal appendages of, 164.
- Sitaris, 691.
- Slug-worm, 188.
- Smell, experiments on, 269;
- Smith, on lack of fore-tarsi in a moth, 102;
- Sole, extensor, 116.
- Somites, 30.
- Sorby, on change of color in Aphides, 205.
- Sounds, 293.
- Specius, 585.
- Spengel, on color sense, 260.
- Spermatheca, 506.
- Spermatid, 498.
- Spermatocyte, 498.
- 727Spermatogonium, 498.
- Spermatophore cap, 499.
- Spermatophores, 497, 499.
- Spermatozoa, 497, 525;
- formation of, 498.
- Sperm-nucleus, 525.
- Sphinx, 456, 552;
- moulting of, 610.
- Sphinx ligustri, changes during metamorphosis, 646.
- Spilosoma, 391.
- Spindle, directive, 525.
- Spines, 187, 189;
- Spinneret, 342;
- of caterpillars, 75.
- Spinning apparatus, 340;
- Spinules, 189, 197.
- Spiracles, 437;
- types of, 438.
- Spiral thread, 444;
- Spraying apparatus, 370.
- Spring of Collembola, 551.
- Spuler, on pigments, 207, 208;
- Squama, 123.
- Squamæ, 124.
- Squamula, 124.
- Squamule, 89.
- Stadia of metamorphosis, 594.
- Stage, carabidoid, 692;
- Stages, ametabolous, 594.
- Stagmomantis carolina, embryo of, 584;
- hatching of, 584.
- Staphylinus, 61, 454.
- Stenobothrus sibiricus, swollen foretarsus in male, 113.
- Stenosternus, 101.
- Sternum, 87, 89.
- Stigmata, 437;
- Sting, bee’s, 172.
- Stipes, 63.
- Stokes, on the tænidia, 445;
- hairs in, 451.
- Stomach, chyle, 314, 325;
- Stomach-mouth, 309.
- Stomodæum, 537, 547, 569.
- Stomoxys, 446.
- Strauss-Dürckheim, on the heart, 397;
- on muscles of cockchafer, 213.
- Streak, embryonal, 531;
- primitive, 531.
- Streblopus, 101.
- Striæ of scales, 194, 202.
- Styles, abdominal, 176;
- of ovipositor, 167.
- Stylopidæ, 486;
- hypermetamorphosis of, 695.
- Stylops childreni, triungulin larva of, 695.
- Subgalea, 73.
- Submentum, 54, 63, 68, 69.
- Substance, fibrillar nerve, 238.
- Sucking stomach, 302, 305.
- Supra-anal plate, 181.
- Supra-œsophageal ganglion, 231.
- Supra-spinal vessel, 403.
- Suranal plate, 181.
- Surroundings, physical, 463.
- Sustainers of the pupa, 638.
- Swammerdam, on discovery of air-sacs, 456;
- Swimming, act of, 116.
- Symphyla, 18;
- characters of, 22.
- Synaptera, 27, 534, 594, 705.
- Syromastes, 372.
- Tabanus, 629;
- mouth-parts of, 79.
- Tænidia, 444;
- Talæporia, 634.
- Talocera, 57.
- Tanypus, 472.
- Tarsus, 96;
- Taste, organs of, 281.
- Tegeticula yuccasella, 65, 66.
- Tegmina, 124.
- Tegula, 89, 124, 125.
- Telea polyphemus, amount of food eaten by, 608;
- Teleas, hypermetamorphosis of, 703.
- Telephorus, 111, 538;
- tenent hairs of, 99.
- Tenebrio, 617.
- Tenent hairs, 99.
- Tentacle of maxilla, 65.
- Tentorium, 49.
- 728Tergum, 87.
- Termen, 122.
- Termes, abdomen, 162;
- Termes flavipes, 64.
- Termopsis, 48, 64.
- Testes, 487, 495;
- incipient eggs in the germ of the testis, 504.
- Theory of incasement, 641.
- Thomas, on origin of scent-scales, 199.
- Thorax, 86, 95;
- gills on, 468.
- Thread-plate, 575.
- Thread, spiral, 444;
- Thyridium, 124.
- Thyridopteryx, 634.
- Thysanoptera, nymph of, 597.
- Thysanura, 72;
- Tibia, 96;
- formation of, in imago of Lepidoptera, 655.
- Tineid larva, wing-buds of, 652;
- wing-germs of, 129.
- Tipula, 629;
- Tissue, connective, 574.
- Tongue, 70.
- Torulus, 57.
- Tracheæ, 431, 442;
- Tracheal gills, of adult insects, 476;
- rectal, 463.
- Tracheal system, amphipneustic type, 462;
- Tracheoles, 126, 653.
- Tract, optic, 253.
- Trajectory made by wings, 150.
- Tribolium, 617.
- Trichodes, tracks of, 110.
- Trichogen, 188, 199, 366.
- Trichoptera, 469;
- Trilobita, 5.
- Tritocerebrum, 231, 237.
- Triunguline larva, 100, 693, 695.
- Trochanter, 496;
- divided, 97.
- Trochantine, 95.
- Trogoderma, 617.
- Trophi, 54, 59, 549.
- Trouvelot, on the moulting fluid, 610;
- Truxalis, 422.
- Tubercles, 187, 192.
- Tubes, egg, 501;
- Tympanum, 289.
- Typhlocyba, 616.
- Uljanin, on the sexual cells of the honey bee, 582.
- Ungues, 96.
- Uranidin, 206.
- Urates, 352.
- Urea, 352.
- Urech, on pigments, 206, 208.
- Uric acid, 352.
- Urinary bladder, 351;
- Urine, 206.
- Urite, 163.
- Uromeres, 163.
- Uro-patagia, 183.
- Urosome, 163.
- Urosternites, 163.
- Uterus, 507.
- Utriculi, 487.
- Uzel, on the embryology of Campodea, 51, 53;
- Vagina, 507.
- Valery-Mayet, on life-history of Sitaris, 691.
- Vallisneri, on the cremaster, 637.
- Valvule, cardiac, 312;
- Van Bemmelen, on colors, 208.
- Vanessa antiopa, 638.
- Vanessa io, 381.
- Vanessa urticæ, before pupation, 644;
- changes in nervous system during its metamorphosis, 646.
- Van Rees, on post-embryonic changes of Muscidæ, 673, 674, 676, 680, 683, 684.
- Vas deferens, 496;
- origin of, 579.
- Vasa deferentia, 496;
- origin of, 579.
- 729Vayssière, on lingua of Ephemera, 73.
- Veins of wings, 121, 144.
- Velum penis, 181.
- Venomous glands, 358.
- Vent, 319.
- Ventriculus, 314.
- Vermipsylla, tongue of, 77.
- Verson, on serially arranged dermal glands, 366;
- vestigial stigmata, 460.
- Vesicles, air, 456;
- Vespa crabro, 217;
- olfactory organs of, 276.
- Vessel, dorsal, 397;
- Vestigial tracheæ, 460.
- Viallanes, on brain, 231;
- head segments, 51.
- Vision, mode of, by compound eyes, 256;
- by simple eyes, 255.
- Vitelline membrane, 520.
- Viviparous insects, 515.
- Volucella, thorax of, 91.
- Vosseler, on fœtid glands, 369.
- Wagner, on the circulation, 419.
- Walking, mechanics of, 103, 106.
- Walter, on epipharynx of moths, 44;
- on hypopharynx of moths, 75.
- Wasps, taste-organs of, 277, 286.
- Wax, of butterfly, 364;
- Wax-glands, 361, 364.
- Weevil, embryology of, 538;
- bean and pea, hypermetamorphosis of, 700.
- Weismann’s discovery of imaginal buds, 599, 643, 650;
- West, Tuffen, on feet of fly, etc., 100;
- on walking, 99.
- Westwood, on head of Hymenoptera, 55.
- Wheeler, on embryonic abdominal appendages, 550;
- Wielowiejski, on blood tissue, 408;
- Williston, on anal glands, 372.
- Will, on organs of taste, 282.
- Wing-buds, or discs, 127, 129;
- Wingless insects, 598.
- Wings, 120;
- buds of, 664;
- cells, 121;
- circulation of blood in, 410;
- development of tracheæ of, 144;
- development of veins of, 144;
- embryonic development of, 126;
- folding of, 154, 156;
- imaginal buds of, 653;
- mechanism of, 153, 156;
- origin of, 138;
- primitive origin of, 137;
- as respiratory organs, 461;
- rudimentary, ground-membrane of, 136;
- spreading of, 155;
- theory of, 144;
- tracheæ in, 122;
- veins of, 121.
- Wistinghausen, von, on tracheal endings, 436, 447.
- Witlaczil, on honey dew, 364.
- Wood-Mason, on gills of Paraponyx, 470;
- Xiphidium, embryo of, 534;
- Yersin, on results of section of commissures, 245.
- Yolk, amount of, 524, 529;
- Zaitha, 431;
- pleuropodia of, 551.
- Zone, annular, 653.
1. Zool. Anzeiger, xvi, 1893, pp. 271–5.
2. On the morphology of the Myriopoda, Proc. Amer. Phil. Soc. 1883, pp. 197–209.
3. Morphology and classification of the Pauropoda; also American Naturalist, 1897, p. 410.
4. The term which we proposed for this hypothetical ancestor of insects, “Leptus-like” or “Leptiform,” was an unfortunate one, since the name Leptus was originally given to the six-legged larva of a mite (Trombidium), the origin of the mites and other Arachnida being entirely different from that of the myriopods and insects.
5. Proc. Bost. Soc. Nat. Hist., xvi, 1873, p. 3.
6. American Naturalist, May, 1880, pp. 375, 376.
7. Zoologische Anzeiger, Bd. xx, 1897, pp. 125 and 129. He also states that Campodea resembles the myriopods, especially Geophilus, in the primitive band at first lying on the surface of the yolk, and in the absence of an amniotic cavity; also before hatching the abdomen is pressed against the thorax, as in myriopods.
8. “Scolopendrella has very remarkable antennæ; they may be compared each to a series of glass cups strung upon a delicate hyaline and extensible rod of uniform thickness throughout; so that, like the body of the creature, they shrink enormously when the animal is irritated or thrown into alcohol, and they then possess scarcely two-thirds the length they have in the fully extended condition, their cup-like joints being drawn close together, one within the other. Peripatus, Japyx, many (if not all) Homoptera, and the S. Asiatic relatives of our common Glomeris have all more or less extensible antennæ.” (Wood-Mason, Trans. Ent. Soc., London, 1879, p. 155.)
9. Lassaigne gave it the name of entomoline.
10. Miall and Denny ex Krukenberg; Kolbe gives the formula as C9H15NO6 or C18H15NO12. As the result of his recent researches, Krawkow (Zeits. Biol., xxix, 1892, p. 177) states that the chemical composition of chitin may prove to be somewhat variable.
11. On allowing portions of a locust, a piece of the integument of Limulus, a scorpion, and a myriopod to soak for a month in white potash, neither were dissolved or affected by the reagent.
12. We may add, while correcting the proofs of this book, that the important summary, by Uzel, of his work on the embryology of Campodea appears in the Zoologischer Anzeiger for July 5, 1897. He observes that the premandibular segment in the embryo is very distinct, and that the two projections arising from it persist in the adult. “Campodea is now the first example where these appendages are present in the sexually mature insect and function as constituents of the completed mouth parts. I propose for these hitherto overlooked structures the name of intercalary lobes.” They each form a slightly developed chitinous lobe covering a gap between the base of the labium and the fused external lobe and palpus of the first maxillæ (which are inclined near the labium) in place of the mandibles which have sunken inward. Uzel also homologizes these appendages with two similar projections (Höcker) observed in the embryo of Geophilus by Zograf to be situated in front of the mandibles. Heymons has also detected this segment in the embryo of Lepisma.
13. While these pages are still in type, we may add, in confirmation of this view, that Uzel states, from his researches on the embryology of Campodea, that the maxillary tergites of the embryo only slightly share in building up the tergal region (occiput) of the head, but that they form the genæ of the maxillary segments. (Zool. Anzeiger, July 5, 1897, p. 235.)
14. Miall and Denny in their work on the cockroach, in describing the labium, remark: “The upper edge is applied to the occipital frame, but is neither continuous with that structure nor articulated thereto. By stripping off the labium upwards it may be seen that it is really continuous with the chitinous integument of the neck” (p. 95). This is, we think, a mistaken view, as proved by the embryology of the Odonata and of Nematus. Our statements on this subject were first published in part in 1871, and more fully in the third Report, U. S. Ent. Commission, 1883, pp. 284, 285. We also stated that all the gular region of the head probably represents the base of the primitive second maxillæ.
15. After we had arrived at this conclusion, and written the above lines, we received the Zoologischer Anzeiger for March 29, 1897, in which Dr. N. Léon publishes the same view, stating that each side of the submentum is the homologue of the cardo, and each side of the mentum corresponds to the stipes of a single maxilla (p. 74).
16. Miall and Denny were the first to homologize the paraglossæ with the galea and lacinia, showing the complete resemblance of the second maxillæ to the first pair, remarking that “the homology of the labium with the first pair of maxillæ is in no other insects so distinct as in the Orthoptera.” We have also independently arrived at a similar conclusion, but believe that the mentum corresponds to the first maxillary cardo, and the palpifer to the first maxillary stipes, the sclerite of each maxilla being fused to form the base of the labium, i.e. the unpaired mentum and submentum.
17. Uzel states that what is regarded as the ligula of Campodea is formed from the sternite of the first maxillary segment; while the two parts regarded as paraglossæ grow out from the sternite of the mandibular segment, and these three structures together he regards as the hypopharynx. (Zool. Anzeiger, July 5, 1897, p. 234.)
18. See, also, Breithaupt, Ueber die Anatomie und die Functionen der Bienenzunge, 1886. It confirms and extends Cheshire’s work.
19. Cholodkowsky, Zool. Anz., ix, p. 615; x, p. 102.
20. Zool. Anz., ix, p. 711.
21. Ent. Amer., v, p. 110, Pl. II, Fig. 7.
22. In his account of his studies on the locomotion of insects, De Moor states that he obtained the track of each of the feet in different colors by coating them with different pigments; the insect, as it moved, left its track on a strip of paper. (Archives de Biologie, Liège, 1890.)
23. Carlet and also De Moor (1890) confirm Graber’s statement that in beetles the first and last appendages on the same side are in contact with the ground, while the middle one is raised. On the other side of the body the middle appendage is on the ground and the first and last one raised.
24. Trans. Amer. Ent. Soc. xx, p. 168.
25. Proc. Ent. Soc. London. Feb. 19, 1896. Heymons also shows that the germs of the elytra of the larva of Tenebrio molitor in the prepupal stage are like those of other insects. (Sitzungs-Ber. Gesell. natur f. Freunde zu Berlin, 1896, pp. 142–144.)
26. Zur Entwickelungsgeschichte und Reproductionsfähigkeit der Orthopteren. Von Vitus Graber. Sitzungsberichte d. math.-naturw. Classe der Akad. d. Wissensch., Wien. Bd. lv, Abth. i, 1867; also Die Insekten.
27. On the transformations of the common house fly, by A. S. Packard, Jr. Proceedings Boston Society of Natural History, vol. xvi, 1874. See Pl. 3, Figs. 12a, 12b.
28. See our Guide to the Study of Insects, p. 66, Figs. 65, 66.
29. Our Common Insects, 1873, p. 171.
30. Compare the observations of Palmén, Gerstäcker, Vayssière, and others.
31. Beiträge zur Kenntniss der Termiten. Jenaische Zeitschrift für Naturwissenchaft, Bd. ix, Heft 2, p. 253, 1875. Compare, however, Palmén’s Zur Morphologie des Tracheensystems, Helsingfors, 1877, wherein he opposes Müller’s view and adopts Gegenbaur’s. See p. 8, footnote.
32. Pancritius, who also adopted Müller’s views, lays much stress on the fact that in larvæ of some orders the tracheæ do not enter the rudimentary wings until the end of larval life, and hence the wings have not originated from tracheal gills, but were originally “perhaps only protective covers for the body.”
33. Reproduced from the author’s remarks in Third Report U. S. Ent. Commission, pp. 268–271, 1883.
34. Von Lendenfeld, however, points out the fact that Straus-Durckheim proved that the wings of beetles are moved by a complicated system of numerous muscles. “In the Lepidoptera I have never found less than six muscles to each wing, as also in the Hymenoptera and Diptera.” “The motions of the wings of Libellulidæ are the combined working of numerous muscles and cords, and of a great number of chitinous pieces connected by joints.”
35. Heymons, however, denies that the so-called cerci in Odonata are such, and claims that they are the homologues of the “caudal processes” (superior terminal appendages of Calvert), because they arise from the tenth abdominal segment.
36. Amer. Nat., iv, December, 1870.
37. Handbuch der Zoologie, p. 17, 1863, Fig. 162.
38. In my account of the anatomy of Melanoplus spretus, 1st Report U. S. Entomological Commission, p. 259, I have called these the infra-anal flaps or uro-patagia.
39. It has been suggested to us by A. A. Packard that the power possessed by insects of transporting loads much heavier than themselves is easily accounted for, when we consider that the muscles of the legs of an insect the size of a house-fly (¼ inch long), and supporting a load 399 times its own weight, would be subjected to the same stress (per square inch of cross-section) as they would be in a fly 100 inches long of precisely similar shape, that carried only its own weight; from the mechanical law that, while the weight of similar bodies varies as the cube of the corresponding dimensions, the area of cross-section of any part (such as a section of the muscles of the leg) varies only as the square of the corresponding dimensions. In short, the muscles of a fly carrying this great proportional weight undergo no greater tension than would be exerted by a colossal insect in walking.
40. This has been shown to be the case by Michels, who states that each commissure is formed of three parallel bundles of elementary nerve-fibres, which pass continuously from one end of the ventral or nervous cord to the other. “The commissures take their origin neither out of a central punctsubstanz (or marksubstanz), nor from the peripheral ganglion-cells of the several ganglia, but are mere continuations of the longitudinal fibres which decrease posteriorly in thickness, and extend anteriorly through the commissures, forming the œsophageal ring, to the brain.”
41. The following extract from Newton’s paper shows, however, that the infra or subœsophageal ganglion, according to Faivre, has the power of coördinating the movements of the body; still, it seems to us that the brain is primarily concerned in the exercise of this power, as the nerves from the subœsophageal ganglion supply only the mouth-parts. “The physiological experiments of Faivre in 1857 (Ann. des Sci. Nat. tom. viii, p. 245), upon the brain of Dyticus in relation to locomotion, are of very considerable interest, showing, as they appear to do, that the power of coördinating the movements of the body is lodged in the infraœsophageal ganglion. And such being the case, both the upper and lower pairs of ganglia ought to be regarded as forming parts of the insect’s brain.”—Quart. Jour. Micr. Sc., 1879, p. 342.
42. The arthropod protocerebrum probably represents the annelid brain (supraœsophageal ganglion). The antennal segment (deutocerebrum), with the premandibular (intercalary) segment (tritocerebrum) originally postoral, have, as Lankester suggests, in the Arthropoda moved forward to join the primitive brain. See Wheeler, Journ. Morphology, Boston, viii, p. 112.
43. Viallanes’ assertion that the instincts of the horse-flies and dragon-flies are “lower” than those of the locusts, may, it seems to us, well be questioned.
44. A. S. Packard, Experiments on the vitality of insects, Psyche, ii, 17, 1877.
45. Waterhouse, Trans. Ent. Soc., London, 1889, p. xxiv.
46. J. Müller, Physiology of the Senses. Trans. by Baly, copied from Lubbock, p. 176.
47. Hauser here uses the word taster, but this means palpus or feeler. It is probably a lapsus pennæ for teeth (Kegeln).
48. In 1870 I observed these sense-pits in the antennæ and also in the cercopoda of the cockroach (Periplaneta americana). I counted about 90 pits on each cercus. They are much larger and much more numerous than similar pits in the antennæ of the same insect. I compared them to similar pits in the antennæ of the carrion-beetles, and argued that they were organs rather of the smelling than hearing. (Amer. Nat., iv., Dec. 1870.) Organs of smell in the flies (Chrysopila) and in the palpi, both labial and maxillary, of Perla were described in the same journal (Fig. 270). Compare Vom Rath’s account of the organs in the cercopods of Acheta (Fig. 271); also the singular organ discovered by him on the end of the palpus of butterflies, in which a number of hair-like rods (sh) are seated on branches of a common nerve (n, Fig. 272).
49. Forel, however (Recueil Zoologique Suisse, 1887), denies that these tympanic organs are necessarily ears, and thinks that all insects are deaf, with no special organs of hearing, but that sounds are heard by their tactile organs, just as deaf-mutes perceive at a distance the rumbling of a carriage. But he appears to overlook the fact that many Crustacea, and all shrimps and crabs, as well as many molluscs, have organs of hearing. The German anatomist Will believes that insects hear only the stridulation of their own species. Lubbock thinks that bees and ants are not deaf, but hear sounds so shrill as to be beyond our hearing.
50. Weismann, Die nachembryonale Entwicklung der Musciden. Zeitschr. für wissen. Zoologie, xiv, p. 196, 1864.
51. Plateau (1877) states that the digestive fluid of insects, as well as of Arachnids, Crustaceans, and Myriopods, has no analogy with the gastric juice of vertebrates; it rather resembles the pancreatic sugar of the higher animals. The acidity quite often observed is only very accessory in character, and not the sign of a physiological property. “Farther, I have found it in insects; Hoppe-Seyler has demonstrated in the Crustacea, and I have proved in the spiders, that the ferment causing the digestion of albuminoids is evidently quite different from the gastric pepsine of vertebrates; the addition of very feeble quantities of chlorhydric acid, far from promoting its action, retards or completely arrests it.” (Bull. Acad. roy. Belgique, 1877, p. 27.)
52. The word grès we translate as the layer of gum. Not sure of the English equivalent for grès, I applied to Dr. L. O. Howard, U. S. Entomologist, who kindly answers as follows: “I have consulted Mr. Philip Walker, a silk expert, who writes me the following paragraph: ‘Grès, as I understand it, is the gum of the silk fibre, hence the French name for raw silk, grèye, which is in distinction to the silk that has been boiled out in soap after twisting, or throwing, as it is called. As I understand it, the silk fibre is composed of the grès and fibroin. The former is soluble in alkali, like soap water, and the latter is not.’” While Blanc considers the grès as the product of a special secretion of the wall of the reservoir, Gilson regards its production as simultaneous with that of the silk or of the fibroin (l.c. 1893, p. 74).
53. On cytological differences in homologous organs. Report 63d meeting of British Assoc. Adv. Sc. for 1893. 1894. p. 913.
54. See also Giard, Bull. Soc. Ent. France, p. viii, 1894.
55. “The contents of the Malpighian tubules may be examined by crushing the part in a drop of dilute acetic acid, or in dilute sulphuric acid (10 per cent). In the first case a cover-slip is placed on the fluid, and the crystals, which consist of oblique rhombohedrons or derived forms, are usually at once apparent. If sulphuric acid is used, the fluid must be allowed to evaporate. In this case they are much more elongated, and usually clustered. The murexide reaction does not give satisfactory indications with the tubules of the cockroach.” (Miall and Denny, The cockroach, p. 129, footnote.)
56. “There is a curious analogy between the excretory organs of these insects and the mesonephros of some vertebrates, where a second, third, etc., generation of tubules is added to the primitive metameric series. When the embryonic number of Malpighian vessels persists in insects, the demand for greater excreting surface is supplied by a lengthening of the individual vessels.”
57. For the mode of adhesion of Cynips eggs, see Adler in Deutsche Ent. Zeits. 1877, p. 320.
58. Mercaptan is a mercury, belonging to a class of compounds analogous to alcohol, having an offensive garlic odor. Methyl mercaptan is a highly offensive and volatile liquid.
59. Embryonic or temporary glands, the “pleuropodia” of Wheeler, viz. the modified first pair of abdominal legs, occur in Œcanthus, Gryllotalpa, Xiphidium, Stenobothrus, Mantis (occasionally a pair on the second abdominal segment, Graber); Blatta, Periplaneta, Cicada, Zaitha, Hydrophilus, Acilius, Melolontha, Meloë, Sialis, Neophylax. (See Wheeler, Appendages of the First Abdominal Segment, etc., 1890.)
60. These midges owe their phosphorescence to bacteria in their bodies during disease.
61. Untersuchungen zur Anatomie und Histologie der Tiere, 1884, p. 72.
62. Zelle und Gewebe, 1885, p. 43. (See also our p. 217.)
63. Studien über die Lampyriden, Zeits. für wiss. Zool., xxxvii, 1882. Both Wielowiejski and M. Wistinghausen have completely disproved the view of Schultze, that the tracheæ end in star-like cells, where respiration takes place, as the “star-like cells” are simply net-like expansions of the peritoneal membrane of the tracheæ.
64. The following summary compiled from Krancher, is translated, with some minor changes, from Kolbe’s work.
65. Miall and Denny state that in the cockroach the abdominal spiracles are permanently open, owing to the absence of a valve, but communication with the tracheal trunk may be cut off at pleasure by an internal occluding apparatus.
66. Zur Entwicklungsgeschichte der Biene, Zeitschr. wissens. Zoologie, xx, p. 519, 1870.
67. Die Entwicklung der Dipteren im Ei, Zeitschr. wissens. Zoologie, xiii, 1863.
68. Amer. Naturalist, May, 1886, p. 438.
69. Zeitschr. wissens. Zoologie, xl, 1884, Taf. xix, Fig. 8, T.
70. Science, 1893, pp. 44–46.
71. Art. Thorax, Todd’s Cycl. of Anat. and Phys.
72. The mesothoracic stigmata are open in Carabus, Potamophilus, Elmis, Macronychus, Buprestis, Elater, Lampyris, Lycus, Triphyllus, Eucinetus, Dascillus, Psephenus, Ergates, Micralymna, and probably many others. The metathoracic stigmata are open in Lycus and Elmis.
73. In the Hymenoptera the two pairs on the meso- and metathoracic segments are open in the Aculeata, also in the Siricidæ, among which sometimes that on the third segment is closed. In Pimpla and Microgaster (fully grown larvæ) only the mesothoracic stigmata are open.
Palmén adds that most dipterous larvæ are amphipneustic; Cecidomyia, the Mycetophilidæ, Bibionidæ, and Stratiomys are typically peripneustic. (p. 92.)
Moreover, a single insect, as Sialis, may be apneustic as a larva, peripneustic as a pupa, and holopneustic in the imago stage.
74. Mr. J. W. Folsom, who has made the accompanying sketch of the nymph of Euphæa splendens in the Cambridge Museum, finds only seven pairs of gills, there being no traces of them on segments 1, 9, and 10. A stout trachea, he writes us, enters the base of each gill, and subdivides into several long branches, which course along the periphery. Hagen in his original account said there were eight pairs on segments 1–8 respectively.
75. Harris, Correspondence, p. 226, Pl. III., Fig. 7.
76. Nusbaum’s view has been questioned by Heymons, who, from his studies on the embryology of the cockroach (Periplaneta and Phyllodromia), Forficula, and Gryllus, concludes that the ectodermal ends of the sexual outlets owe their origin to an unpaired median hypodermal invagination, and that it is quite doubtful whether the ectodermal portions of the sexual passages of insects were ever paired (p. 104). On the other hand he appears, even throwing out the case of Ephemera, to have overlooked Nassonow’s discovery of paired outlets in the young of Lepisma.
77. Acta Acad. German., xxxiii, 1867, No. 2, p. 81. Quoted by Dr. Sharp, Insecta, p. 142.
78. Journ. Morph., iii, Boston, pp. 299, 300.
79. Proc. Boston Soc. Nat. Hist., xi, pp. 88, 89.
80. In the following general account of the embryology of insects, I have closely followed the admirable arrangement and description of Korschelt and Heider, in their Lehrbuch der vergleichenden Entwicklungsgeschichte der wirbellosen Thiere, pp. 764–846, often translating their text literally, though not omitting to state the results of other writers.
81. Korschelt and Heider state that no cellular embryonal membranes are present in Synaptera, Uljanin finding none in the Podurids. In the embryo of Isotoma walkerii we, however, observed a membrane which we compared to the larval skin of many Crustacea, and both Sommer and Lemoine have detected in eggs of the same group a cuticular larval skin which is provided with spines for rupturing the chorion. The amnion is also wanting in Proctotrupids (Ayers), and is rudimental in Muscidæ (Kowalevsky, Graber), in viviparous Cecidomyidæ, according to Metschnikoff, who also states that in certain ants of Madeira the envelopes are represented only by a small mass of cells in the dorsal region.
82. In Diptera the stomodæum may be dorsal, Dr. Pratt tells us.
83. Will (Aphis) and also Cholodkowsky’s statement (Blatta), as well as Balfour and Schimkewitch’s statements that the brain is at first disconnected from the ventral cord, are apparently erroneous.
84. The description perhaps applies not only to the cockroaches, but, as seen from the similar but fragmentary notices of Heider and of Wheeler on the Coleoptera, may be common to insects in general.
85. Report on the Rocky Mountain locust, etc. Ninth Annual Report U. S. Geol. and Geogr. Survey of the Territories for 1875, pp. 633, 634.
86. Orthoptera Europæa, 1853, p. 37.
87. In his Für Darwin (1863), Fritz Müller gives his reasons for the opinion that the so-called “complete metamorphosis” of insects was not inherited from the primitive ancestor of all insects, but acquired at a later period.
88. For further details see the 1st Report of the U. S. Entomological Commission, 1878, pp. 279–281.
89. See Köppen ueber die Heuschrecken in Südrussland, 1862, pp. 22, 23.
90. In Samouelle’s The Entomologist’s Useful Compendium, 1819. See Westwood’s Class. Insects, i, p. 2; Leach’s Ametabolia comprised the Thysanura (Synaptera) and the lice.
91. From the Greek μανός, scanty; μεταβολή, change.
92. Greek, ἤρεμα, quiet; μεταβολή, change.
93. At the same date (March, 1869) we independently suggested that the insects had originated from some form like the hexapodous young of Pauropus and Podura. In November, 1870, we suggested that the Thysanura and the hexapodous Leptus may have descended from some Peripatus-like worm. Afterwards (1871) we proposed for the ancestral form the term leptiform, which was later abandoned for Brauer’s term Campodea-form.
94. Amer. Naturalist, i, p. 85, 1867.
95. First Rep. U. S. Ent. Commission, p. 281–283.
96. Trans. Ent. Soc. London, iii, p. xv. See also Ashton, R. J., Trans. Ent. Soc. London, iii, 1841–43, pp. 157–159.
97. Proc. Bost. Soc. Nat. Hist., x, 1866, p. 283.
98. See Max Braun’s article entitled Ueber die histologischen Vorgange bei der Hautung von Astacus fluviatilis, with a full bibliography, in Semper’s Arbeiten aus dem Zool. zoot. Institut in Würzburg, ii, pp. 121–166. Also Semper’s Animal Life, p. 20. Trouvelot also discovered the moulting fluid. (Amer. Nat., i, p. 37.)
99. American Naturalist, xvii, May, 1883, pp. 547, 548.
100. Le Pelletier. A. M. L., Bulletin de la Société Philomathique, Paris, April, 1813.
101. Heineken, Carl. Observations on the reproduction of the members in spiders and insects. (Zool. Journ., 1829, vi, pp. 422–432.)
102. Bees and Bee-keeping, pp. 21, 22.
103. Butterflies, their structure, changes, and life-histories. New York, 1881, pp. 37–42. Butterflies of the Eastern United States and Canada, 1888, 1889. Also, Frail children of the air, 1895, pp. 232, 233 a. Dr. Chapman, however, finds that this piece in micropupæ has no connection whatever with the head or eye, but belongs rather with the prothoracic segment. (Trans. Ent. Soc. London, 1893, p. 102.) We have been able to confirm his statements, but still this piece is peculiar to the pupal state.
104. Rep. Ent. U. S. Dept. Agr., 1879, pp. 228, 229, Pl. IV, Fig. 4.
105. Monograph of bombycine moths, Pt. I, 1897. Figs. 24, 28, 29, 33, 34, 40, 77.
106. Amer. Naturalist, xii, pp. 379–383.
107. Hybocampa milhauseni, Dr. Chapman tells me, has a pupal spine (imperfectly present in Cerura) with which it cuts out a lid of the cocoon.
108. Riley’s Report for 1892, p. 203.
109. Philosophy of the pupation of butterflies, and particularly of Nymphalidæ, by Charles V. Riley. (Proc. Amer. Assoc. Adv. Science, xxviii, Saratoga Meeting, August, 1880, pp. 455–463.)
110. The homology of the suranal plate of the larva with the cremaster of the pupa, established by Riley in 1880, is also affirmed by Jackson (1888) and by Poulton, and for some years we have been satisfied that this is the correct view; Professor Hatchett-Jackson discovered it, he states, in 1876.
111. In his remarkable studies on the morphology of the Lepidoptera, Professor W. Hatchett-Jackson states his belief that Riley’s homology of the sustentors with the soles or plantæ of the anal prolegs, and the sustentor ridges with their limbs, is wrong, and that the eminences on either side the anal furrow, or the “anal prominences,” as they are termed by Riley, represent the prolegs, and that the sustentor ridges and sustentors are probably peculiar developments of the body of the 10th somite, found only in some Lepidoptera. From our examination of pupa of different families of moths, we are satisfied that Jackson’s view is the correct one. We have not found the sustentors and their ridges in the pupæ of the more generalized moths, but the vestiges of the anal legs are almost invariably present, their absence in the pupa of Nola and Harrisina being noteworthy.
112. We copy from Kirby and Spence their abstract of Herold’s conclusions: “The successive skins of the caterpillar, the pupa-case, the future butterfly, and its parts or organs, except those of sex, which he discovered in the newly excluded larva, do not preëxist as germs, but are formed successively from the rete mucosum, which itself is formed anew upon every change of skin, from what he denominates the blood, or the chyle after it has passed through the pores of the intestinal canal into the general cavity of the body, where, being oxygenated by the air-vessels, it performs the nutritive functions of blood. He attributes these formations to a vis formatrix (bildende Kraft).
“The caul or epiploon (fett-masse), the corps graisseux of Réaumur, etc., which he supposes to be formed from the superfluous blood, he allows, with most physiologists, to be stored up in the larva, that in the pupa state it may serve for the development of the imago. But he differs from them in asserting that in this state it is destined to two distinct purposes: first, for the production of the muscles of the butterfly, which he affirms are generated from it in the shape of slender bundles of fibres; and, secondly, for the development and nutrition of the organs formed in the larva, to effect which, he says, it is dissolved again into the mass of blood, and being oxygenated by the air-vessels, becomes fit for nutrition, whence the epiploon appears to be a kind of concrete chyle.” (Entwickelungsgeschichte der Schmetterlinge, pp. 12–27.) It seems that Herold was right in deriving the pupa and imago from the hypodermis (his rete mucosum), but wrong in denying that the germs did not preëxist in the young caterpillar, and wrong in supposing that the latter originated from the blood, also in supposing that the muscles owe their origin to the fat-body. Swammerdam, and also Kirby and Spence, were correct in supposing that the imago arose from “germs” in the larva, though wrong in adopting the “emboîtement” theory.
113. In the regions where the imaginal buds are not present (dorsal aspect of the prothorax, and abdomen), the epithelium (hypodermis) may proliferate independently of these buds.
114. We shall translate portions and, when the text allows, make an abstract of parts of Gonin’s clear and excellent account, often using his own words.
115. C. Herbert Hurst, The Pupal Stages of Culex.
116. Lowne on the Blow-fly, new edit., pp. 2, 41, Fig. 7.
117. Miall, Natural History of Aquatic Insects, pp. 136–138. Also Trans. Linn. Soc. London, V, Sept., 1892.
118. This account is translated from Korschelt and Heider, with some omissions and slight changes.
119. Westwood in his excellent account of this group remarks: “Hence, as well as from the account given by Jurine, it is evident that the pupa of the Stylops is enclosed in a distinct skin, and is also in that state enveloped by the skin of the larva, contrary to the suggestion of Mr. Kelly.” (Class. Insects, II. 297.) This is all we know about the supernumerary larval stages.
120. Some facts towards a life history of Rhipiphorus paradoxus. Annals and Magazine of Natural History for October, 1870.
- P. 316, changed “abdominal cells” to “absorbent cells”.
- Silently corrected typographical errors and variations in spelling.
- Archaic, non-standard, and uncertain spellings retained as printed.